 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Towards sustainable biogas upgrading: MIL-120(Al) as a cost-effective water stable MOF for CO2/CH4 separation
Marta Bordonhos ab,
Rosana Vieira Pinto
ab,
Rosana Vieira Pinto c,
Tânia Fradea,
Bingbing Chenc,
Farid Nouarc,
Georges Mouchaham
c,
Tânia Fradea,
Bingbing Chenc,
Farid Nouarc,
Georges Mouchaham c,
José R. B. Gomes
c,
José R. B. Gomes b,
Christian Serrec and
Moisés Luzia Pinto
b,
Christian Serrec and
Moisés Luzia Pinto *a
*a
aCERENA, Departamento de Engenharia Química, Instituto Superior Técnico, Universidade de Lisboa, Av. Rovisco Pais, No. 1, 1049-001 Lisboa, Portugal. E-mail: moises.pinto@tecnico.ulisboa.pt
bCICECO – Aveiro Institute of Materials, Department of Chemistry, University of Aveiro, Campus Universitário de Santiago, 3810-193 Aveiro, Portugal
cInstitut des Matériaux Poreux de Paris (IMAP), ESPCI Paris, École Normale Supérieure de Paris, CNRS, PSL University, 75005 Paris, France
First published on 9th February 2026
Abstract
The microporous MIL-120(Al) MOF has been tested for CO2/CH4 separation. The material has been synthesised in powder form at the kilogram scale and shaped into spherical beads without significant loss in adsorption capacity (on average, ca. 7% for CO2). MIL-120(Al) is more selective towards CO2 (IAST mean selectivity of 10.6–35 between 0.1–10 bar), showing a good adsorption capacity and a moderate enthalpy of adsorption (−36.7 to −39.1 kJ mol−1 in the low loading regime). Molecular simulation studies have revealed a probable rotation of the µ2-OH groups with increasing CO2 pressure altering the CO2 adsorption capacity at higher pressures. Breakthrough experiments have confirmed the CO2/CH4 selectivity of MIL-120(Al) and have shown that the material can retain its original adsorption separation performance after regeneration, following consecutive cycles (10 cycles) and prolonged exposure to high amounts of water vapour, demonstrating the potential of this MOF for biogas upgrading.
Introduction
Over the last years, the demand for sustainable energy sources has been growing, driving intensified research into biogas upgrading, a critical process to enhance methane (CH4) purity in raw biogas by removing carbon dioxide (CO2). Biogas is a renewable energy source that originates from the anaerobic digestion of organic matter and can serve as a substitute for fossil natural gas. However, raw biogas can typically contain up to 50% (V/V) in CO2, along with other contaminants and trace impurities such as hydrogen sulphide (H2S) and moisture, which lower its energy value and limit its direct use in conventional energy grids or as fuel. To maximise its utility, efficient CO2 separation is essential to obtain biomethane with a high CH4 molar content (>96%), making it comparable to conventional natural gas.1,2Various technologies have been explored for CO2/CH4 separation in biogas upgrading, including absorption, adsorption, and membrane separation.1,2 Among these, adsorption-based cyclic processes using porous materials, like pressure swing adsorption (PSA), have garnered significant attention due to their potential for energy-efficient and cost-effective separations. Metal–organic frameworks (MOFs), crystalline micro- or mesoporous hybrid solids, with their high surface area, tuneable pore structures, and selective gas adsorption properties, have emerged as promising candidates for this application.3,4 However, many MOFs suffer from hydrothermal and/or acid–base instability, complex synthesis routes, and high production costs, limiting their large-scale implementation.5,6 MIL-120(Al),7 an aluminium-based ultramicroporous MOF, emerges as a promising alternative for biogas upgrading due to its high stability toward coordinating and corrosive gases, such as water, NOx or SOx.8 MIL-120(Al) is among the most cost-effective adsorbent materials, with an estimated production cost of approximately 13 $ kg−1 and a scalable synthesis protocol.8 Moreover, it exhibits excellent CO2 adsorption capacity up to 1 bar,8 enabling more energy-efficient separation processes. Herein, we investigate the CO2/CH4 separation performance of MIL-120(Al) in the context of biogas upgrading (Fig. 1) from a combination of single-component adsorption experiments and molecular simulations up to 10 bar, and gas mixture breakthrough experiments at ca. 1 bar. These allowed the assessment of its adsorption capacity, pore structure, behaviour under dry and wet conditions, selectivity and recyclability, while benchmarking its performance against other adsorbent materials. MIL-120(Al) demonstrates a remarkable potential for industrial biogas purification, offering a breakthrough solution to the pressing challenges of economic scalability and operational feasibility in sustainable energy applications.
Results and discussion
Adsorption performance under dry conditions
MIL-120(Al) has been synthesised at the kilogram scale using an environmentally friendly, scalable and high-yield synthesis method, following a previous protocol developed by some of us.8 The characterisation details, provided in Section S1 in the SI, confirm the structural integrity, porosity and thermal stability of the material. Single-component CO2 and CH4 adsorption isotherms measured at 25 °C for the powder form of MIL-120(Al) are represented in Fig. 2A. Additional details are described in the Experimental section and in Section S2.A in the SI. The experimental data and corresponding isotherm model fittings are reported in Tables S2 and S3 in the SI. As expected, MIL-120(Al) shows a higher adsorption capacity for CO2 than for CH4 in the entire pressure range studied, suggesting the potential of this MOF for biogas upgrading. In comparison with other adsorbent materials (additional details in Section S2.D in the SI), the CO2 uptake of MIL-120(Al) at 1 bar, ca. 3.6 mmol g−1, is higher than for the reviewed activated carbons9–12 (AC), carbon molecular sieves13,14 (CMS), pillared clays,15 silica-based materials,16–18 and periodic mesoporous organosilicas19 (PMO). MIL-120(Al) also shows a higher or similar CO2 uptake at 1 bar than the other MOFs reviewed in the SI,13,20–23 with the exception of MIL-160(Al) (as a powder)24 and CuBTC.13 In comparison with zeolites,13,25–30 MIL-120(Al) shows a mixed uptake performance: higher than some, including zeolites beta,25 5A![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28 and Na-ZSM-5,30 but lower than zeolites 13X
28 and Na-ZSM-5,30 but lower than zeolites 13X![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26
26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) )/NaX,13 4A
)/NaX,13 4A![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27 and NaY.29
27 and NaY.29
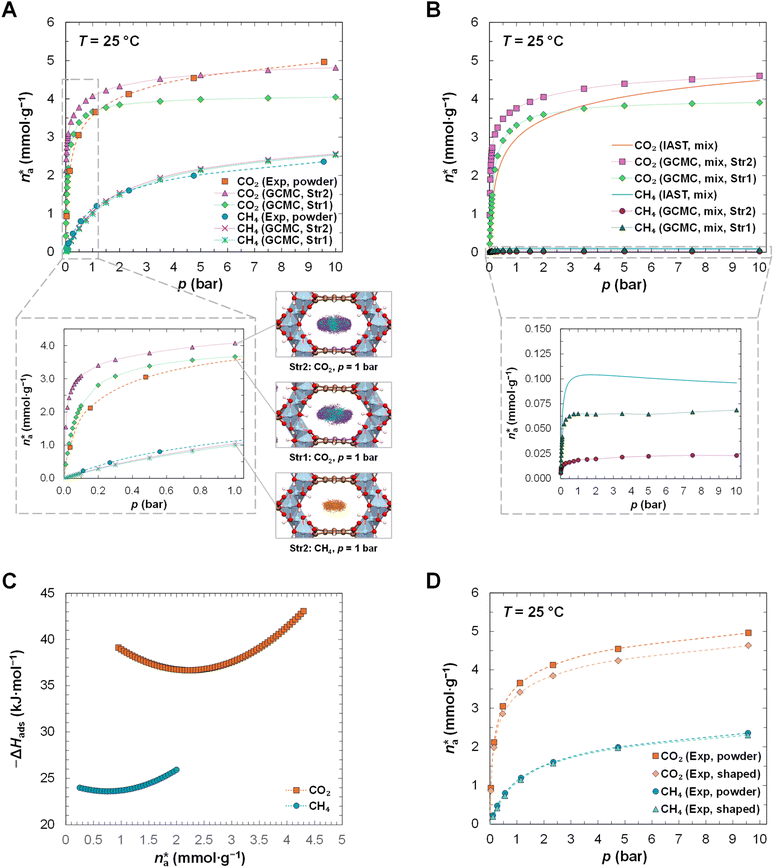 | ||
Fig. 2 (A) Single-component adsorption isotherms of CO2 and CH4 at 25 °C measured for the powder form of MIL-120(Al) and calculated by GCMC for Str2 and Str1, and density plots generated from GCMC simulations for the single-component adsorption at 1 bar of CO2 in Str2 and Str1 and of CH4 in Str2; (B) mixture isotherms calculated by IAST and GCMC (Str2 and Str1) for a binary CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 mixture with % (V/V) = (50 CH4 mixture with % (V/V) = (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50)%; (C) isosteric enthalpy of adsorption (ΔHads) calculated from the Clausius–Clapeyron equation from the single-component experimental data measured at 25, 35 and 45 °C; (D) comparison of the single-component adsorption isotherms of CO2 and CH4 at 25 °C measured for the powder and shaped forms of MIL-120(Al). All isotherms are reported in absolute adsorbed amounts, 50)%; (C) isosteric enthalpy of adsorption (ΔHads) calculated from the Clausius–Clapeyron equation from the single-component experimental data measured at 25, 35 and 45 °C; (D) comparison of the single-component adsorption isotherms of CO2 and CH4 at 25 °C measured for the powder and shaped forms of MIL-120(Al). All isotherms are reported in absolute adsorbed amounts,  . The dashed lines in (A) and (D) represent the non-linear least-squares fit of the isotherm model to the experimental data (cf. Table S3 in the SI). The dotted lines in (A), (B) and (C) serve only as a visual guide of the data trends to the reader. The density maps in (A) were generated from the combined snapshots of GCMC simulations collected every 100 cycles of the production run. Colour code for framework atoms in the density maps: carbon (C, brown); hydrogen (H, white); oxygen (O, red); aluminium (Al, blue). Colour code for CO2 atoms in the density maps: C (green); O (violet). Colour code for the C atoms from CH4 (UA) in the density maps: C (orange). CO2 and CH4 (UA) atoms were drawn without bonds (where applicable) and on a 1 . The dashed lines in (A) and (D) represent the non-linear least-squares fit of the isotherm model to the experimental data (cf. Table S3 in the SI). The dotted lines in (A), (B) and (C) serve only as a visual guide of the data trends to the reader. The density maps in (A) were generated from the combined snapshots of GCMC simulations collected every 100 cycles of the production run. Colour code for framework atoms in the density maps: carbon (C, brown); hydrogen (H, white); oxygen (O, red); aluminium (Al, blue). Colour code for CO2 atoms in the density maps: C (green); O (violet). Colour code for the C atoms from CH4 (UA) in the density maps: C (orange). CO2 and CH4 (UA) atoms were drawn without bonds (where applicable) and on a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 radii scale relative to framework atoms in the density maps. Structure drawing and density maps produced by VMD 1.9.4.36 8 radii scale relative to framework atoms in the density maps. Structure drawing and density maps produced by VMD 1.9.4.36 | ||
Grand-canonical Monte Carlo (GCMC) simulations have been run in RASPA 2.0,31 at 25, 35 and 45 °C and up to 10 bar, and contemplated two different structures for MIL-120(Al), Str2 (Fig. S7 in the SI) and Str1 (Fig. S8 in the SI), in accordance with the strategy and nomenclature employed in a previous work by our colleagues up to 1 bar.8 The models of these two structures differ mainly in the position of eight H atoms of the µ2-OH groups in the unit cell (four different pairs of H atoms) and have been optimised by density functional theory (DFT) in VASP32–35 6.1.2. Additional details can be found in the Experimental section and in Section S2.B in the SI. Regarding CO2 adsorption, Str2 presents higher uptakes than Str1. In MIL-120(Al), CO2 interacts preferentially with the µ2-OH groups of the metal clusters8 on both sides of the pore, as can be seen in the density maps (25 °C, 1 bar) in Fig. 2A, and in greater detail for 0.01, 0.1, 1 and 10 bar in Fig. 3. The CO2 density maps at 25 °C are also represented for the full simulation box in the SI in Fig. S9 to S16 for Str2, and S21 to S28 for Str1. The interaction of CO2 with these µ2-OH groups is likely due to the quadrupole moment of CO2, that leads to strong electrostatic interactions between the O atoms in CO2 and the H atoms in the µ2-OH groups.
 | ||
Fig. 3 Density maps of the single-component adsorption of CO2 at 25 °C and 0.01, 0.1, 1 and 10 bar obtained for Str2 (top) and Str1 (bottom) from GCMC calculations, as viewed from two directions. For the complete set of density maps, the reader is referred to Section S2.B in the SI. Colour code for framework atoms: C (brown); H (white); O (red); Al (blue). Colour code for CO2 atoms: C (green); O (violet). CO2 atoms are drawn without bonds and on a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 radii scale relative to framework atoms. Density maps generated from the combined snapshots of GCMC simulations collected every 100 cycles of the production run. Structure drawing and density maps produced by VMD 1.9.4.36 8 radii scale relative to framework atoms. Density maps generated from the combined snapshots of GCMC simulations collected every 100 cycles of the production run. Structure drawing and density maps produced by VMD 1.9.4.36 | ||
As the orientation of some of these groups in Str2 and Str1 is different, the resulting CO2 isotherms are different, as also verified and discussed by our colleagues in a previous work.8 The predicted Str1 isotherm more closely resembles the experimental isotherm until ca. 1 bar. The small differences observed until 1 bar between this work and the previous study can be attributed to both the experimental pore volume, ca. 0.28 cm3 g−1 in Chen et al.8 (value converted from Fig. S1D for MIL-120-AP powder for p/p° ≈ 0.83) vs. 0.21 cm3 g−1 (for p/p° ≈ 0.85, cf. Table S1 in the SI) in this work that can be due to minor structural differences in the synthesised materials, and the volume of the crystallographic unit cells of the simulated structures, ca. 1030 Å3 for Str1 and ca. 1056 Å3 for Str2 in Chen et al.8 (values reported originally for the primitive cell, i.e., half of the total volume of the unit cell) vs. ca. 1045 Å3 (for the unit cell, cf. Tables S4 and S5 in the SI) for both structures in this work. As pressure increases, the trend of the experimental isotherm starts deviating from Str1 and moving towards Str2, following Str2 more closely from 5 bar onwards, as can be seen more clearly in the semi-logarithmic plot of the isotherms in Fig. 4. This indicates that there is probably a rotation of the µ2-OH groups as pressure increases, and neither Str1 nor Str2 can fully represent CO2 adsorption on MIL-120(Al) on their own. As such, we suspect that the real structure can be seen as a composite of not only Str1 and Str2 but also of other structures with other orientations of the µ2-OH groups, highlighting the hydroxyl rotational flexibility of this framework. Indeed, this flexibility has recently attracted scientific attention and is currently being investigated with machine learning potentials by different research groups.37,38 For the adsorption of CH4 there is no noticeable difference between Str2 and Str1. This is because CH4 does not have a quadrupole moment that can induce strong (electrostatic) interactions with the µ2-OH groups, thus it is less sensitive to their orientation, unlike CO2. Its lower uptake is mostly due to weaker van der Waals interactions with the chemical environment inside the pores, that are the same for both types of structures. In comparison with the experimental CH4 isotherm at 25 °C, there is only a minor increase in CH4 uptake from ca. 2 bar onwards in the simulated isotherms, that is acceptable within the uncertainty of both methods. Looking into the density of gas inside the pores at 25 °C, the increase in CO2 and CH4 density from 0.01, to 0.1, to 1, to 10 bar in Str2 and Str1 can be seen in greater detail from two directions in Fig. 3 (only CO2) and in Fig. S9 to S32 in the SI.
 | ||
Fig. 4 Semi-logarithmic plot of the CO2 and CH4 isotherms (25 °C) represented in Fig. 2A. All isotherms are reported in absolute adsorbed amounts,  . The dashed lines represent the non-linear least-squares fit of the Virial isotherm model to the experimental data (cf. Table S3 in the SI), and the dotted lines serve only as a visual guide of the simulated data trends to the reader. . The dashed lines represent the non-linear least-squares fit of the Virial isotherm model to the experimental data (cf. Table S3 in the SI), and the dotted lines serve only as a visual guide of the simulated data trends to the reader. | ||
Complementary radial distribution functions (RDFs) for the interaction of each framework atom-type of Str2 (Table S7 in the SI) and Str1 (Table S8 in the SI) with the C or O atoms of CO2 at 25 °C have also been computed in RASPA31 and are plotted for the same pressures in Fig. S33 to S46 in the SI. Interestingly, for CO2 adsorption in Str2, where the pore-facing µ2-OH groups are oriented toward the same plane inside the pore channel (cf. Table S4 and Fig. S7 in the SI), it can be seen in the density maps in Fig. 3, top, that CO2 molecules align both perpendicularly and axially to the pore channel length, as observed previously,8 and that as pressure increases more CO2 molecules start adsorbing axially to the pore channel length and slightly closer to the metal chain on the side wall of the pore, resulting in a rectangle-like shape of adsorbed CO2 molecules along the length of the pore channel (cf. Fig. 3, top). In the RDF plots in Fig. S33 to S36 in the SI, it can be seen in general that the intensity of some of the first RDF peaks between CO2 atoms and framework atom-types increases from lower to higher pressures. This can be attributed to the increase in CO2 density inside the pores, that leads to a closer (denser) and more ordered packing of the CO2 molecules (cf. Fig. 3, top). It should be noted that RDF peaks at distances greater than 5–6 Å can also represent the interaction of adsorbate atoms with framework atoms of neighbouring pores, as the maximum pore dimensions, measured from the centre of mass of framework atoms and without taking into account the van der Waals radii, are ca. 7 Å (between the organic linkers at the top and bottom of the unit cell, cf. Fig. S7 in the SI) and ca. 9.4 Å (between opposite oxygens from the metal clusters with atom-type label 8, cf. Fig. S7 and Table S7 in the SI). For Str1 (cf. Fig. 3, bottom), where the rotated pore-facing µ2-OH groups on each side of the pore have different orientations relative to each other (cf. Table S5 and Fig. S8 in the SI), CO2 molecules are adsorbed in alignment with these µ2-OH groups, but with a higher density along and in parallel to the pore channel (cf. Fig. 3, bottom). The RDF plots for this structure, represented in Fig. S37 to S46 in the SI, show, in general, notably higher intensity peaks at 0.01 bar than at higher pressures. This can be due to the highly specific interactions of CO2 at 0.01 bar, that corresponds to a low loading of ca. 0.4 mmol g−1 (cf. Fig. 4), with the framework atoms that results in a well-defined coordination of this low amount of CO2 molecules with the adsorption sites inside the pores. At higher pressures, as more CO2 occupies the available pore space, the molecules are more dispersed, and the intensity of the peaks decreases sharply. This is in contrast with the observations for Str2 (cf. Fig. S33 to S36 in the SI), as in Str2 the CO2 uptake at 0.01 bar is ca. 1.5 mmol g−1, and the CO2 molecules are already more dispersed (cf. density maps at 0.01 bar in Fig. 3). Nevertheless, a slight increase in peak intensity can also be seen from 0.1 to 10 bar in some RDFs for Str1, as seen for Str2. For CH4, the density maps for both structures (cf. Fig. S17 to S20 for Str2 and S29 to S32 for Str2 in the SI) show a similar density of adsorbed CH4 molecules, that is located along and in the middle of the pore channel.
Adsorption isotherms were calculated for an equimolar CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 mixture at 25 °C based on the Ideal Adsorbed Solution Theory (IAST),39,40 from the Virial isotherm model adjusted to the single-component experimental data (cf. Tables S2 and S3 in the SI), and by GCMC for Str2 and Str1 (cf. Table S6 in the SI). The mixture isotherms, represented in Fig. 2B, show a high selectivity for CO2 over CH4, as the mixture uptakes of CO2 are close to the uptakes of the single-component CO2 isotherms, whereas for CH4, the mixture uptakes are much lower (up to two orders of magnitude for Str2) than those of the single-component CH4 isotherms. The results for CO2 are consistent with the observations made for its single-component adsorption: the IAST-predicted mixture isotherm is more similar to Str1 at lower pressures and to Str2 at higher pressures. For CH4, IAST overestimates its mixture uptake in comparison with the GCMC-calculated uptake for both Str2 and Str1.
CH4 mixture at 25 °C based on the Ideal Adsorbed Solution Theory (IAST),39,40 from the Virial isotherm model adjusted to the single-component experimental data (cf. Tables S2 and S3 in the SI), and by GCMC for Str2 and Str1 (cf. Table S6 in the SI). The mixture isotherms, represented in Fig. 2B, show a high selectivity for CO2 over CH4, as the mixture uptakes of CO2 are close to the uptakes of the single-component CO2 isotherms, whereas for CH4, the mixture uptakes are much lower (up to two orders of magnitude for Str2) than those of the single-component CH4 isotherms. The results for CO2 are consistent with the observations made for its single-component adsorption: the IAST-predicted mixture isotherm is more similar to Str1 at lower pressures and to Str2 at higher pressures. For CH4, IAST overestimates its mixture uptake in comparison with the GCMC-calculated uptake for both Str2 and Str1.
The isotherms measured at 35 and 45 °C, represented in Fig. S4 in the SI, show lower gas uptakes with increasing temperature, with lower slopes and an overall similar isotherm shape. The isotherms calculated by GCMC at 35 and 45 °C for Str1 and Str2 show similar trends to those calculated at 25 °C, as can be seen in Fig. S47 in the SI: for CO2, Str1 and Str2 are closer to the experimental isotherms at lower and higher pressures, respectively, and for CH4 both Str1 and Str2 follow the experimental isotherms. The isosteric enthalpy of adsorption, ΔHads, has been calculated from the Clausius–Clapeyron equation41 using the isotherm fittings from the experimental data for the three temperatures within the same loading range (cf. Tables S2 and S3 in the SI). The results are represented in Fig. 2C. CO2 shows a moderate enthalpy of adsorption, between ca. −36.7 and −39.1 kJ mol−1 in the lower loading regime (≤3 mmol g−1), and CH4 shows a lower enthalpy of adsorption, with values between ca. −23.6 and −24.0 kJ mol−1 in the lower loading regime (≤1.2 mmol g−1). The estimated values for the isosteric enthalpy of adsorption of CO2 are in agreement with the ones previously reported (ca. −36.3 kJ mol−1 on average, in the range 1–2.5 mmol g−1),8 and in agreement with the average heat of desorption obtained from the GCMC calculations ran in this work (ca. 37.0–43.4 kJ mol−1 within the same loading range). The increase observed above 2.25 mmol g−1 for CO2 and above 0.75 mmol g−1 for CH4 is due to adsorbate–adsorbate interactions, that become more dominant as loading increases and the available adsorption sites in MIL-120(Al) become gradually occupied (i.e., compression inside the pores). In comparison with other materials (cf. Table S10 in the SI), the CO2 enthalpy of adsorption estimated for MIL-120(Al) is higher than for the reviewed AC,21–24 CMS,13,14 the MCM-41 silica,42 zeolites Silicalite-1,25 NaY29 and Na-ZSM-5,30 and the majority of MOFs13,20,21,23,43 reviewed, and generally in line with the remaining zeolites13,25–28 reviewed.
MIL-120(Al) has been further shaped into beads to make it more suitable for continuous operation in industrial biogas upgrading systems like PSA. Millimetre-sized beads have been obtained via the wet granulation method using 5% (w/w) bentonite as binder (cf. the Experimental section and Fig. S2 in the SI), achieving similar characterisation properties as the powder material (cf. Section S1.B in the SI) and similar to the previous study in which a slightly different shaping method had been used.8 The performance of the shaped form of MIL-120(Al) has also been evaluated. The single-component CO2 and CH4 adsorption isotherms at 25 °C, represented in Fig. 2D, are similar to those measured for the powder form of MIL-120(Al), with only a small decrease in adsorption capacity (on average, ca. 6% for CH4 and ca. 7% for CO2), attributed to the presence of a non-porous binder (5% (w/w)) (cf. Fig. S3C and Table S1 in the SI). This suggests that the shaping procedure did not impact the overall adsorption properties of the MOF. IAST has also been used to predict the mean selectivity and phase diagrams of the separation of CO2 and CH4 at 25 °C, based on a method proposed by Myers39,40 described in previous works.15,44 The IAST mean selectivity, represented in Fig. S5 in the SI, shows a similar increase of the mean selectivity with pressure for both materials (shaped and powder). MIL-120(Al) also shows a higher IAST-predicted mean selectivity of CO2 over CH4 at 5 bar than other materials reviewed (cf. Table S10 in the SI), with the notable exception of two amine-functionalised SBA-15 materials, on which CO2 is strongly chemisorbed.16 The IAST-predicted phase diagrams at 5 bar are shown in Fig. S6 in the SI. For the binary mixture of CO2 and CH4 at a molar fraction of 0.5 in the gas phase (yCH4) – the composition of a typical biogas feed1,2 – the molar fraction of CH4 in the adsorbed phase (xCH4) is ca. 0.02 for both samples (cf. Fig. S6A in the SI). This means that the adsorbed phase is much richer in CO2 than in CH4 for both materials, further highlighting the high selectivity of MIL-120(Al) for this separation. The complete phase diagrams in Fig. S6B in the SI further confirm that both materials present a similar performance in terms of selectivity and adsorption capacity of CO2.
Following these promising results, the adsorption of an equimolar CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 gas mixture has been studied under dynamic conditions, by measuring the breakthrough curves at 25 °C and ca. 1 bar, in a fixed-bed adsorption system illustrated in Fig. S48 in the SI. Additional experimental details are described in the Experimental section, in Section S2.C in the SI and in a previous work.24 To evaluate the recyclability of the material, a series of ten consecutive adsorption–desorption cycles have been performed, with a duration of 1 h per cycle (30 min for each adsorption and desorption branch). The ten sets of curves are represented consecutively in Fig. 5 and overlapped in Fig. S49 in the SI. The breakthrough curves confirm that MIL-120(Al) is more selective towards CO2, as CH4 breaks through the column ahead of CO2. The selectivity calculated from breakthrough run 1, ca. 89, is higher than for a biomass-based AC,10 similar to that for benchmark zeolite 13X
CH4 gas mixture has been studied under dynamic conditions, by measuring the breakthrough curves at 25 °C and ca. 1 bar, in a fixed-bed adsorption system illustrated in Fig. S48 in the SI. Additional experimental details are described in the Experimental section, in Section S2.C in the SI and in a previous work.24 To evaluate the recyclability of the material, a series of ten consecutive adsorption–desorption cycles have been performed, with a duration of 1 h per cycle (30 min for each adsorption and desorption branch). The ten sets of curves are represented consecutively in Fig. 5 and overlapped in Fig. S49 in the SI. The breakthrough curves confirm that MIL-120(Al) is more selective towards CO2, as CH4 breaks through the column ahead of CO2. The selectivity calculated from breakthrough run 1, ca. 89, is higher than for a biomass-based AC,10 similar to that for benchmark zeolite 13X![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 21 and lower than that for NH2-MIL-53(Al)21 (cf. Table S10 in the SI). Additionally, a roll-up of CH4, indicative of the displacement of the weakly adsorbed CH4 by CO2,45 is also clearly visible in all adsorption branches, further emphasising the higher affinity of the material towards CO2. The adsorption uptakes calculated from the experimental curves are listed and compared with those predicted from IAST in Table S9 in the SI. The adsorption uptakes calculated from the experimental curves for run 1 for CO2 are slightly lower but very close to those predicted from IAST, however for CH4 these differences are higher which can be attributed to both experimental error in the quantification of the low amount of CH4,24 and the assumptions of the model. Due to these differences in CH4 uptake, the selectivities obtained from IAST and breakthrough experiments are different. We recall that IAST values are estimated from the mixture adsorbed amounts in the material, using the experimental data of the pure components' adsorption, which may contribute in part to the observed selectivity differences. Furthermore, considering the GCMC gas mixture isotherms, obtained for a theoretical perfect crystal structure, the selectivity at 1 bar varies between ca. 51 for Str1 and ca. 201 for Str2. Nevertheless, the overall agreement is a good indicator of the validity of the methodologies and assumptions used. From run 1 to run 2 there is a loss in CO2 adsorption capacity of ca. 35% (ca. 1 mmol g−1) that is due to the incomplete regeneration of the material in 30 min. After run 2, the adsorption capacity is maintained throughout the remaining cycles, as seen by the overlapped breakthrough curves and calculated uptakes (cf. Fig. S49 and Table S9 in the SI). This shows that the loss in CO2 adsorption capacity is not cumulative and is only a function of the regeneration conditions, reflecting the reversible nature of the host–guest interactions between CO2 and the chemical environment inside the pores of the material, namely the hydrogen bonds between CO2 and the µ2-OH groups in MIL-120(Al). For CH4, the weakly adsorbed component in the mixture, 30 min is sufficient time to purge the material of all adsorbed CH4 and the adsorption capacity is maintained (cf. Table S9 in the SI).
21 and lower than that for NH2-MIL-53(Al)21 (cf. Table S10 in the SI). Additionally, a roll-up of CH4, indicative of the displacement of the weakly adsorbed CH4 by CO2,45 is also clearly visible in all adsorption branches, further emphasising the higher affinity of the material towards CO2. The adsorption uptakes calculated from the experimental curves are listed and compared with those predicted from IAST in Table S9 in the SI. The adsorption uptakes calculated from the experimental curves for run 1 for CO2 are slightly lower but very close to those predicted from IAST, however for CH4 these differences are higher which can be attributed to both experimental error in the quantification of the low amount of CH4,24 and the assumptions of the model. Due to these differences in CH4 uptake, the selectivities obtained from IAST and breakthrough experiments are different. We recall that IAST values are estimated from the mixture adsorbed amounts in the material, using the experimental data of the pure components' adsorption, which may contribute in part to the observed selectivity differences. Furthermore, considering the GCMC gas mixture isotherms, obtained for a theoretical perfect crystal structure, the selectivity at 1 bar varies between ca. 51 for Str1 and ca. 201 for Str2. Nevertheless, the overall agreement is a good indicator of the validity of the methodologies and assumptions used. From run 1 to run 2 there is a loss in CO2 adsorption capacity of ca. 35% (ca. 1 mmol g−1) that is due to the incomplete regeneration of the material in 30 min. After run 2, the adsorption capacity is maintained throughout the remaining cycles, as seen by the overlapped breakthrough curves and calculated uptakes (cf. Fig. S49 and Table S9 in the SI). This shows that the loss in CO2 adsorption capacity is not cumulative and is only a function of the regeneration conditions, reflecting the reversible nature of the host–guest interactions between CO2 and the chemical environment inside the pores of the material, namely the hydrogen bonds between CO2 and the µ2-OH groups in MIL-120(Al). For CH4, the weakly adsorbed component in the mixture, 30 min is sufficient time to purge the material of all adsorbed CH4 and the adsorption capacity is maintained (cf. Table S9 in the SI).
 | ||
| Fig. 5 Consecutive CH4 (dashed lines) and CO2 (full lines) breakthrough curves for experimental runs 1 to 10 measured at 25 °C and ca. 1 bar for the shaped MIL-120(Al). | ||
Adsorption performance under moisture
The impact of water on the adsorption capacity of MIL-120(Al) has also been studied. Water is an important contaminant in biogas streams and during an adsorption process it can compete for the adsorption sites and significantly reduce the adsorption capacity. While most of the water can be removed during the compression stage of the raw biogas feed, some traces are always present, and its complete removal is very costly. Thus, as a first assessment, a small amount of water vapour (ca. 0.16 mmol g−1, corresponding to only traces of water in the gas feed8) was pre-adsorbed onto the powder, and the single-component adsorption isotherms of CO2 and CH4 were measured at 25 °C. The resulting CO2 and CH4 isotherms are compared with the isotherms initially measured under dry conditions in Fig. 6A, and it can be seen that pre-adsorbing ca. 0.16 mmol g−1 does not affect the adsorption capacity of MIL-120(Al) (in powder form), as both sets of isotherms overlap. This is a clear advantage over some classical zeolites (e.g., 13X) that are very hydrophilic due to the binding of water on the surface cations.The performance of the shaped material has been tested under humid conditions with an equimolar CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 gas mixture at ca. 1 bar and 25 °C in the fixed-bed dynamic adsorption system, by pre-loading the material with water vapour in two tests (additional details in the Experimental section and in Section S2.C in the SI). On the first test, a helium (He) flow carrying water vapour at a relative humidity (RH) of 50% was passed through the packed bed for ca. 3 h and 30 min, after which the equimolar CO2
CH4 gas mixture at ca. 1 bar and 25 °C in the fixed-bed dynamic adsorption system, by pre-loading the material with water vapour in two tests (additional details in the Experimental section and in Section S2.C in the SI). On the first test, a helium (He) flow carrying water vapour at a relative humidity (RH) of 50% was passed through the packed bed for ca. 3 h and 30 min, after which the equimolar CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 gas mixture was passed through the packed bed. The resulting breakthrough curves, illustrated in Fig. 6B, overlap with those of run 1 (performed under dry conditions, cf. Fig. 5). With these conditions of %RH and contact time, the material is still not at saturation equilibrium due to the slow water adsorption kinetics, and thus, it does not appear to have any effect on the CO2 and CH4 adsorption capabilities of the material. To further understand the effect of the presence of water vapour inside the pores of MIL-120(Al), an additional experiment was run under more severe humid conditions, by flowing He carrying water vapour at RH = 80% for a prolonged period of exposure of 48 h. The higher humidity content of the stream and the much longer exposure time can better ensure the pores inside the material are saturated with water vapour. The equimolar CO2
CH4 gas mixture was passed through the packed bed. The resulting breakthrough curves, illustrated in Fig. 6B, overlap with those of run 1 (performed under dry conditions, cf. Fig. 5). With these conditions of %RH and contact time, the material is still not at saturation equilibrium due to the slow water adsorption kinetics, and thus, it does not appear to have any effect on the CO2 and CH4 adsorption capabilities of the material. To further understand the effect of the presence of water vapour inside the pores of MIL-120(Al), an additional experiment was run under more severe humid conditions, by flowing He carrying water vapour at RH = 80% for a prolonged period of exposure of 48 h. The higher humidity content of the stream and the much longer exposure time can better ensure the pores inside the material are saturated with water vapour. The equimolar CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 breakthrough run performed after this prolonged exposure to water vapour, represented by the blue curves in Fig. 6B, now clearly shows the effect of the presence of water vapour inside the pores. The curves of both gases are shown to break through ahead of the curves of previous runs (ca. 2 min and 30 s for CH4 and 5 min and 30 s for CO2) and the calculated uptakes are reduced by half for CO2 and ca. ten times for CH4 (cf. Table S9 in the SI). Despite this significant loss in adsorption capacity, the slopes of these and the other breakthrough curves remain the same, which indicates that the presence of even this significantly high amount of water vapour inside the pores does not appear to affect the adsorption kinetics of the gases. Additionally, a final dry breakthrough experiment, run 11, has been performed after this last humid run, and following material activation for 12 h with temperature and He flow (cf. Section S2.C in the SI). The results of run 11 have shown that the material has been completely regenerated, as seen by the overlapped breakthrough curves in pink in Fig. 6B and calculated gas uptakes in Table S9 in the SI, demonstrating that the high amount of pre-adsorbed water has not altered permanently the physical and chemical environment inside the pores of MIL-120(Al). This performance of MIL-120(Al) is in clear contrast with the cases of hydrophilic zeolites (with low Si/Al ratios) that are severely affected by the presence of water.46,47
CH4 breakthrough run performed after this prolonged exposure to water vapour, represented by the blue curves in Fig. 6B, now clearly shows the effect of the presence of water vapour inside the pores. The curves of both gases are shown to break through ahead of the curves of previous runs (ca. 2 min and 30 s for CH4 and 5 min and 30 s for CO2) and the calculated uptakes are reduced by half for CO2 and ca. ten times for CH4 (cf. Table S9 in the SI). Despite this significant loss in adsorption capacity, the slopes of these and the other breakthrough curves remain the same, which indicates that the presence of even this significantly high amount of water vapour inside the pores does not appear to affect the adsorption kinetics of the gases. Additionally, a final dry breakthrough experiment, run 11, has been performed after this last humid run, and following material activation for 12 h with temperature and He flow (cf. Section S2.C in the SI). The results of run 11 have shown that the material has been completely regenerated, as seen by the overlapped breakthrough curves in pink in Fig. 6B and calculated gas uptakes in Table S9 in the SI, demonstrating that the high amount of pre-adsorbed water has not altered permanently the physical and chemical environment inside the pores of MIL-120(Al). This performance of MIL-120(Al) is in clear contrast with the cases of hydrophilic zeolites (with low Si/Al ratios) that are severely affected by the presence of water.46,47
Conclusions
The microporous aluminium tetracarboxylate MOF MIL-120(Al) has been successfully synthesised at the kilogram scale, shaped and tested for the separation of CO2 and CH4, under dry and humid conditions. The single-component adsorption isotherms measured for CO2 and CH4 at 25 °C, on both powdered and shaped forms of MIL-120(Al), have confirmed the great potential of the material for the separation of these gases, both in terms of sorption capacity and high selectivity towards CO2. Molecular simulation studies suggest a rotation of the µ2-OH groups in MIL-120(Al), that shifts the material between two different configurations, Str1 to Str2, as pressure increases. To further understand this phenomenon, future work could include in situ experimental studies, namely neutron scattering, solid state nuclear magnetic resonance (ssNMR) or extended X-ray absorption fine structure (EXAFS) spectroscopies. The isosteric enthalpies of adsorption for CO2 in the powdered MIL-120(Al) indicate a consistent affinity for CO2 molecules. The breakthrough experiments carried out for an equimolar CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 gas mixture at 25 °C and ca. 1 bar on the shaped material have confirmed MIL-120(Al) to be more selective towards CO2, as it breaks through the packed column after CH4, in line with the best sorbents studied so far for this application. Indeed, the selectivity values estimated for an equimolar mixture at ca. 5 bar from IAST (ca. 31) and from the breakthrough curves at ca. 1 bar (ca. 89) place MIL-120(Al) clearly above other reported MOFs (except for NH3-MIL-53, where chemisorption is expected) and in line with the best zeolites, although in MIL-120(Al) the water presence does not permanently impair the adsorption separation as in the case of some zeolites with low Si/Al ratios. The cyclability of the material has also been tested under dry conditions, and a loss in CO2 adsorption capacity related with the short regeneration time has been observed. However, this loss has not been cumulative, as it has only been observed from the first to the second cycle. The adsorption capacity of CO2 and CH4 in the MIL-120(Al) powder remains unaffected by trace exposure to humid conditions. The breakthrough experiments under humid conditions in the shaped material have further revealed that even though a prolonged exposure to water vapour at a high RH (80%) had a significant impact on the adsorption uptake of both gases, the adsorption kinetics appeared unaffected. Furthermore, after complete regeneration, the performance of the material was re-established, showing no permanent damage to its adsorption capabilities after the prolonged exposure to water vapour. In addition to its hydrothermal stability, sustainable, cheap and scalable character, these overall results highlight the great potential of MIL-120(Al) for biogas upgrading. This strongly supports the continuation of further studies to optimise the regeneration conditions and the cyclic separation of biogas, and ascertain the stability of the material in the presence of corrosive gases.
CH4 gas mixture at 25 °C and ca. 1 bar on the shaped material have confirmed MIL-120(Al) to be more selective towards CO2, as it breaks through the packed column after CH4, in line with the best sorbents studied so far for this application. Indeed, the selectivity values estimated for an equimolar mixture at ca. 5 bar from IAST (ca. 31) and from the breakthrough curves at ca. 1 bar (ca. 89) place MIL-120(Al) clearly above other reported MOFs (except for NH3-MIL-53, where chemisorption is expected) and in line with the best zeolites, although in MIL-120(Al) the water presence does not permanently impair the adsorption separation as in the case of some zeolites with low Si/Al ratios. The cyclability of the material has also been tested under dry conditions, and a loss in CO2 adsorption capacity related with the short regeneration time has been observed. However, this loss has not been cumulative, as it has only been observed from the first to the second cycle. The adsorption capacity of CO2 and CH4 in the MIL-120(Al) powder remains unaffected by trace exposure to humid conditions. The breakthrough experiments under humid conditions in the shaped material have further revealed that even though a prolonged exposure to water vapour at a high RH (80%) had a significant impact on the adsorption uptake of both gases, the adsorption kinetics appeared unaffected. Furthermore, after complete regeneration, the performance of the material was re-established, showing no permanent damage to its adsorption capabilities after the prolonged exposure to water vapour. In addition to its hydrothermal stability, sustainable, cheap and scalable character, these overall results highlight the great potential of MIL-120(Al) for biogas upgrading. This strongly supports the continuation of further studies to optimise the regeneration conditions and the cyclic separation of biogas, and ascertain the stability of the material in the presence of corrosive gases.
Experimental
Large scale green synthesis of MIL-120(Al) at ambient pressure
Deionised (DI) water (16 litres) was heated to 80 °C in a 30-litre reactor (Fig. S1 in the SI). Once the temperature was reached, 1,2,4,5-benzene-tetracarboxylate (H4BTeC) (2.13 mol, 542 g) and aluminium acetate, Al(OH)(OAc)2·1.38H2O, (8.57 mol, 1.39 kg) were added. The resulting suspension was heated to 100 °C and stirred at 200 rpm for 24 h. The white crude product was collected by filtration, yielding approximately 1.5 kg of the as-synthesised sample (STY = 60 kg m−3 d−1). It was then washed twice with boiling DI water overnight. The washed sample was dried in air.Shaping of MIL-120(Al) into beads
The MIL-120(Al) powder was shaped into spheres by wet granulation followed by spheronisation: at first, the MOF was mixed with 5% (w/w) of an inorganic binder – bentonite – using a mixer. After that, the powder blend was transferred into the wet granulator bowl and water was added dropwise while mixing until uniform granules were formed. The wet granules were then fed to the spheroniser with the speed set to 250 rpm for 10 min to obtain spherical beads. The resulting spheres, ranging in diameter, dp, from 0.5 to 3 mm, were sieved to achieve a more uniform size distribution. Beads selected for experimental measurements were between 1.4 and 2 mm in diameter (Fig. S2 in the SI). The spheres were calcined at 50 °C under vacuum for 12 h.Characterisation of MIL-120(Al) powder and beads
Powder X-ray diffraction (PXRD) data were collected using a high-throughput Bruker D8 Advance diffractometer working on transmission mode and equipped with a focusing Göbel mirror producing Cu Kα radiation (λ = 1.5418 Å) and a LynxEye detector. Nitrogen sorption data at 77 K was collected on a Micromeritics Tristar instrument. Prior to the measurement, the sample was degassed at 150 °C using a Micromeritics SmartVacPrep degas unit. Thermogravimetric analysis was conducted using a Mettler Toledo TGA/DSC 2, STAR System apparatus with a heating rate of 5 °C min−1 under oxygen flow. Mettler Toledo FiveEasyTM Plus pH mV−1 bench meter. Scanning electron microscopy (SEM) micrographs of the MIL-120(Al) powder were captured using a FEI Magellan 400 scanning electron microscope.Experimental single-component adsorption studies
Single-component adsorption equilibrium isotherms of CO2 (99.998%, Air Liquide) and CH4 (99.995%, Air Liquide) were measured for pressures up to 10 bar on MIL-120(Al) in powder form at 25, 35 and 45 °C and on the shaped beads at 25 °C. The gas adsorption isotherms were measured in a lab-made stainless steel volumetric apparatus used in previous works.17,24,48 The samples were first degassed in situ at 150 °C (heating rate of 5 °C min−1) for 8 h, under vacuum in the range of 10−9 bar. The experimental isotherms were fit using the Virial isotherm model,49–52 and binary gas mixture selectivity and phase diagrams were estimated based on IAST,39 following a method proposed by Myers40 that is described in detail in previous works.15,44 The Clausius–Clapeyron equation was used to determine the isosteric enthalpies of adsorption.41 The experiments performed under humid conditions were run as described in a previous work,17 with activation in situ at 150 °C (heating rate of 5 °C min−1) for 16 h, under vacuum in the range of 10−9 bar, before each isotherm measurement. Additional details in Section S2.A in the SI.Molecular simulation studies
The two crystalline structures considered for MIL-120(Al), Str1 and Str2, were optimised by DFT calculations with VASP,32–35 version 6.1.2. GCMC calculations53 were run in RASPA,31 version 2.0, in both MIL-120(Al) structures to compute single-component (at 25, 35 and 45 °C) and mixture adsorption isotherms (at 25 °C) for pressures up to 10 bar, with partial atomic charges obtained with the REPEAT method.54 All RASPA runs were computed in a 4 × 2 × 5 simulation box, with 2 × 104 cycles for initialisation followed by 1 × 105 cycles for production. Additional details in Section S2.B in the SI.Experimental multi-component adsorption studies
Binary breakthrough experiments of CO2 (99.998%, Air Liquide) and CH4 (99.995%, Air Liquide) mixtures were conducted on the shaped MIL-120(Al) beads (particle size with dp [1.4–2] mm) under dry and wet (with pre-adsorbed water vapour) conditions at 25 °C and ca. 1 bar, in a lab-made fixed-bed dynamic adsorption system, represented in Fig. S48 in the SI. For the breakthrough experiments under humid conditions, the activated material was initially exposed to a flow of He carrying water vapour at the desired relative humidity, after which the material was exposed to a flow of the dry equimolar CO2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CH4 mixture. Additional details in Section S2.C in the SI.
CH4 mixture. Additional details in Section S2.C in the SI.
Author contributions
The manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: characterisation of the materials; detailed description of the experimental and computational adsorption studies; additional experimental and computational results; a comparison with other adsorbent materials. See DOI: https://doi.org/10.1039/d5ta07225e.Acknowledgements
This research was developed in the scope of the Projects FCT-UIDB/04028/2025 and FCT-UIDP/04028/2025 (CERENA), and UID/50011/2025 with DOI: 10.54499/UID/50011/2025 and LA/P/0006/2020 with DOI: 10.54499/LA/P/0006/2020 (CICECO - Aveiro Institute of Materials), financed by Portuguese funds through FCT/MCTES, and when applicable co-financed by ERDF under the PT2020 Partnership Agreement. M. B. acknowledges FCT for the PhD Grant Reference SFRH/BD/147239/2019. B. C. is grateful for support from a CSC grant (grant number 201804910475). G. M. and C. S. acknowledge the European Union's Horizon 2020 research and innovation program under grant agreement No. 831975 (MOF4AIR project) for providing financial support.References
- C. A. Grande, in Biofuel's Engineering Process Technology, InTech, 2011, pp. 65–84 Search PubMed.
- L. Paz, S. Gentil, V. Fierro and A. Celzard, J. Environ. Chem. Eng., 2024, 12, 114870 Search PubMed.
- S. Chaemchuen, N. A. Kabir, K. Zhou and F. Verpoort, Chem. Soc. Rev., 2013, 42, 9304–9332 Search PubMed.
- A. Khan, M. A. Qyyum, H. Saulat, R. Ahmad, X. S. Peng and M. Lee, Appl. Mater. Today, 2021, 22, 100925 Search PubMed.
- R. Sahoo, S. Mondal, D. Mukherjee and M. C. Das, Adv. Funct. Mater., 2022, 32, 2207197 Search PubMed.
- D. Chakraborty, A. Yurdusen, G. Mouchaham, F. Nouar and C. Serre, Adv. Funct. Mater., 2024, 34, 1–23 Search PubMed.
- C. Volkringer, T. Loiseau, M. Haouas, F. Taulelle, D. Popov, M. Burghammer, C. Riekel, C. Zlotea, F. Cuevas, M. Latroche, D. Phanon, C. Knöfelv, P. L. Llewellyn and G. Férey, Chem. Mater., 2009, 21, 5783–5791 CrossRef CAS.
- B. Chen, D. Fan, R. V. Pinto, I. Dovgaliuk, S. Nandi, D. Chakraborty, N. García-Moncada, A. Vimont, C. J. McMonagle, M. Bordonhos, A. Al Mohtar, I. Cornu, P. Florian, N. Heymans, M. Daturi, G. De Weireld, M. Pinto, F. Nouar, G. Maurin, G. Mouchaham and C. Serre, Adv. Sci., 2024, 11, 2401070 Search PubMed.
- C. A. Grande, R. Blom, A. Möller and J. Möllmer, Chem. Eng. Sci., 2013, 89, 10–20 Search PubMed.
- I. Durán, N. Álvarez-Gutiérrez, F. Rubiera and C. Pevida, Chem. Eng. J., 2018, 353, 197–207 Search PubMed.
- N. Álvarez-Gutiérrez, M. V. Gil, F. Rubiera and C. Pevida, Fuel Process. Technol., 2016, 142, 361–369 CrossRef.
- N. Álvarez-Gutiérrez, S. García, M. V. Gil, F. Rubiera and C. Pevida, Energy Fuels, 2016, 30, 5005–5015 CrossRef.
- A. D. Wiersum, J. S. Chang, C. Serre and P. L. Llewellyn, Langmuir, 2013, 29, 3301–3309 CrossRef CAS PubMed.
- X. Song, L. Wang, X. Ma and Y. Zeng, Appl. Surf. Sci., 2017, 396, 870–878 CrossRef CAS.
- J. Pires, V. K. Saini and M. L. Pinto, Environ. Sci. Technol., 2008, 42, 8727–8732 CrossRef CAS PubMed.
- L. Mafra, T. Čendak, S. Schneider, P. V. Wiper, J. Pires, J. R. B. Gomes and M. L. Pinto, Chem. Eng. J., 2018, 336, 612–621 Search PubMed.
- M. Pacheco, M. Bordonhos, M. Sardo, R. Afonso, J. R. B. Gomes, L. Mafra and M. L. Pinto, Chem. Eng. J., 2022, 443, 136271 Search PubMed.
- Y. Belmabkhout and A. Sayari, Chem. Eng. Sci., 2009, 64, 3729–3735 CrossRef CAS.
- M. A. O. Lourenço, C. Siquet, M. Sardo, L. Mafra, J. Pires, M. Jorge, M. L. Pinto, P. Ferreira and J. R. B. Gomes, J. Phys. Chem., 2016, 120, 3863–3875 CrossRef PubMed.
- N. Singh, S. Dalakoti, A. Sharma, R. Chauhan, R. S. Murali, S. Divekar, S. Dasgupta and Aarti, Sep. Purif. Technol., 2024, 341, 012072 CrossRef.
- S. A. Peter, G. V. Baron, J. Gascon, F. Kapteijn and J. F. M. Denayer, Adsorption, 2013, 19, 1235–1244 CrossRef CAS.
- A. D. Wiersum, C. Giovannangeli, D. Vincent, E. Bloch, H. Reinsch, N. Stock, J. S. Lee, J. S. Chang and P. L. Llewellyn, ACS Comb. Sci., 2013, 15, 111–119 Search PubMed.
- H. N. Wamba, S. Dalakoti, N. Singh, S. Divekar, J. Ngoune, A. Arya and S. Dasgupta, Ind. Eng. Chem. Res., 2023, 62, 19773–19783 CrossRef CAS.
- M. Bordonhos, M. I. S. Neves, A. Marandi, F. Nouar, M. Jorge, J. R. B. Gomes, C. Serre and M. L. Pinto, Chem. Eng. J., 2025, 524, 169276 CrossRef CAS.
- J. Yang, J. Li, W. Wang, L. Li and J. Li, Ind. Eng. Chem. Res., 2013, 52, 17856–17864 Search PubMed.
- S. Cavenati, C. A. Grande and A. E. Rodrigues, J. Chem. Eng. Data, 2004, 49, 1095–1101 Search PubMed.
- K. G. Wynnyk, B. Hojjati, P. Pirzadeh and R. A. Marriott, Adsorption, 2017, 23, 149–162 CrossRef CAS.
- M. Mofarahi and F. Gholipour, Microporous Mesoporous Mater., 2014, 200, 1–10 Search PubMed.
- A. Sharma, A. Verma, U. Kumar, N. Singh, S. Dalakoti, R. Chauhan, S. Bhandari, S. Divekar, S. Dasgupta and Aarti, Microporous Mesoporous Mater., 2024, 367, 112984 Search PubMed.
- M. Rahmani, B. Mokhtarani, M. Mafi and N. Rahmanian, Ind. Eng. Chem. Res., 2022, 61, 6600–6610 Search PubMed.
- D. Dubbeldam, S. Calero, D. E. Ellis and R. Q. Snurr, Mol. Simul., 2015, 42, 81–101 Search PubMed.
- G. Kresse and J. Hafner, Phys. Rev. B:Condens. Matter Mater. Phys., 1993, 47, 558–561 Search PubMed.
- G. Kresse and J. Hafner, Phys. Rev. B:Condens. Matter Mater. Phys., 1994, 49, 14251–14269 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Comput. Mater. Sci., 1996, 6, 15–50 CrossRef CAS.
- G. Kresse and J. Furthmüller, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS PubMed.
- W. Humphrey, A. Dalke and K. Schulten, J. Mol. Graphics, 1996, 14, 33–38 CrossRef CAS PubMed.
- X. Jin, Y. Li, K. Gaedecke, X. Zhang and B. Smit, ChemRxiv, 2025, preprint, DOI:10.26434/chemrxiv-2025-dqdkf.
- D. Fan, F. L. Oliveira, M. Wahiduzzaman and G. Maurin, arXiv, 2025, preprint, arXiv:2508.20608, DOI:10.48550/arXiv.2508.20608.
- A. L. Myers and J. M. Prausnitz, AIChE J., 1965, 11, 121–127 Search PubMed.
- A. L. Myers, Adsorption, 2003, 9, 9–16 CrossRef CAS.
- A. Nuhnen and C. Janiak, Dalton Trans., 2020, 49, 10295–10307 Search PubMed.
- Y. Belmabkhout, R. Serna-Guerrero and A. Sayari, Chem. Eng. Sci., 2009, 64, 3721–3728 Search PubMed.
- D. Damasceno Borges, P. Normand, A. Permiakova, R. Babarao, N. Heymans, D. S. Galvao, C. Serre, G. De Weireld and G. Maurin, J. Phys. Chem. C, 2017, 121, 26822–26832 CrossRef CAS.
- M. L. Pinto, J. Pires and J. Rocha, J. Phys. Chem. C, 2008, 112, 14394–14402 Search PubMed.
- N. S. Wilkins, A. Rajendran and S. Farooq, Adsorption, 2021, 27, 397–422 CrossRef CAS.
- J. M. Kolle, M. Fayaz and A. Sayari, Chem. Rev., 2021, 121, 7280–7345 Search PubMed.
- D. G. Boer, J. Langerak and P. P. Pescarmona, ACS Appl. Energy Mater., 2023, 6, 2634–2656 Search PubMed.
- M. Bordonhos, M. Lourenço, J. R. B. Gomes, P. Ferreira and M. L. Pinto, Microporous Mesoporous Mater., 2021, 317, 110975 CrossRef CAS.
- D. M. Ruthven, Principles of Adsorption and Adsorption Processes, John Wiley & Sons, 1984 Search PubMed.
- F. J. Wilkins, Proc. R. Soc. London, Ser. A, 1938, 164, 496–509 CAS.
- R. M. Barrer and R. M. Gibbons, Trans. Faraday Soc., 1963, 59, 2875–2887 RSC.
- A. V. Kiselev, J. Colloid Interface Sci., 1968, 28, 430–442 CrossRef CAS.
- D. Dubbeldam, A. Torres-Knoop and K. S. Walton, Mol. Simul., 2013, 39, 1253–1292 CrossRef CAS.
- C. Campañá, B. Mussard and T. K. Woo, J. Chem. Theory Comput., 2009, 5, 2866–2878 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |


