Water exotic states are responsible for the hydrovoltaic effect and controllable evaporation over carbon fibers – spectroscopic and thermodynamic studies
Marek
Wiśniewski
 *,
Julia
Moszczyńska
*,
Julia
Moszczyńska
 ,
Agnieszka
Marczyńska
,
Marita
Samburska
and
Dominika
Olszowy
,
Agnieszka
Marczyńska
,
Marita
Samburska
and
Dominika
Olszowy
Department of Materials Chemistry Adsorption and Catalysis, Faculty of Chemistry, Nicolaus Copernicus University in Toruń, Gagarina 7, 87-100 Torun, Poland. E-mail: marekw@umk.pl
First published on 28th October 2025
Abstract
Carbon materials dominate among materials applied in hydrovoltaic energy-generating and photothermal conversion devices as Janus evaporators. This work investigated the hydrovoltaic effect and photothermal conversion on gradually oxidized carbon fibers (CFs). We used the controllable and unique properties of the tested materials, such as conductivity and mechanical and surface properties, to explain the mechanism of water interaction with the CF surface. With known changes in the specific surface area of CFs, we could directly correlate changes in polarity, enthalpy, or acidity of surface energy components with exotic structures of water clusters. We have proven experimentally that these structures are responsible for the observed phenomena. The increase in surface polarity is the direct cause of the increase in the strength of the electrostatic field at the surface, which for the most oxidized fibers is 4.22 GV m−1. Such a high field intensity value immobilizes water clusters. Reducing the surface electrostatic field intensity to 1.3 GV m−1 causes a significant reduction of the Stern layer, and its complete disappearance occurs for the sample with 0.35 GV m−1. The higher the electrostatic field, the higher the hydrovoltaic effect observed. Water adsorption enthalpy, especially at high relative humidity, is also correlated with the observed phenomena. Stronger interaction of water with the CF surface is not beneficial for a strong stream of water vapor in the evaporator (4.67 kg m−2 h−1). The strongest vapor stream (equal to 7.10 kg m−2 h−1) was observed for the sample with 1.3 GV m−1, for which water adsorption enthalpy values decrease to a minimum at high relative pressure.
Introduction
It has been reported in the literature that when a carbonaceous material is placed in water, its negative surface polarizes adjacent water molecules and attracts counterions such as hydronium ions, and forms an electrical double layer at the solid–liquid interface.1 This happens even when the material has previously been degassed at high temperature due to oxygen chemisorption.2,3 Water and counterions then flow up the highly porous layer and evaporate from the surface. This process induces a jet current and is the source of different surface phenomena, among others a constant potential difference due to evaporation.1,4According to Kumar et al.,1 this mechanism requires an efficient water flow. The authors claim that the hydrovoltaic layer can quickly become saturated if water–adsorbent interactions are too strong. Thus, the evaporation rate is insufficient. They also believe that saturation of the surface significantly impedes the capillary flow, which is a necessary condition for the continuous generation of potential and streaming current.
Solar-powered interfacial evaporation is an innovative technology for obtaining clean water from the sea and wastewater, using solar energy directly.5–8 When the process is based on two-dimensional (2D) evaporators, at the air–water interface,9 it is characterized by relatively low evaporation efficiency, and the efficiency in converting solar energy into steam remains limited by a theoretical maximum threshold.10,11
A breakthrough was the development of an additional solid phase in evaporator structures,7,12–15 for which spatial architecture allows increasing the evaporation surface, obtaining additional energy from the environment, and intensifying the process using external convection. Thanks to these mechanisms, efficiencies exceeding theoretical limits were achieved, which opens the way to practical applications. Nevertheless, further development of advanced solid structures remains crucial for increasing the evaporation rate and expanding the range of possibilities for purifying various water sources.
Since there are many literature studies describing the above-mentioned phenomena, the authors avoid explaining the mechanism of the process. In this work, we want to look into the phenomenon to optimize the surface properties of the carbon material, proving at the same time that delicate modifications of the carbon fibers bring the most expected benefits.
The attempt to understand the mechanism of water interaction with the surface of the carbon material also allows for combining those seemingly distant experiments.
To the best of our knowledge, there are no literature reports experimentally analyzing the structure of the aqueous adsorption phase. We aimed in this work to look closely for diffusive and Stern layers from FTIR and thermodynamic perspectives, and correlate the outcomes with ab initio results.
Experimental
Materials and methods
Carbon fibers (CFs) in the form of 12 mm braid sleeves were purchased from Carbon Center S.C. (Poland) and functionalized according to the scheme presented in Table 1.| Sample label | Atmosphere | Temperature (°C) | Time (h) | Surface area (m2 g−1) |
|---|---|---|---|---|
| CF-1 | Vacuum | 600 | 6 | 4 |
| CF-2 | Air | 300 | 1 | 5 |
| CF-3 | Air | 350 | 1 | 8 |
| CF-4 | O2 | 350 | 12 | 12 |
| CF-5 | O2 | 375 | 2 | 16 |
| CF-6 | O2 | 375 | 24 | 25 |
The full characterization studies of the carbon materials, including low-temperature N2 adsorption, FTIR, Raman, XRD, SEM, TEM, and calorimetric measurements, were performed according to recently published procedures.16 The samples were identified by X-ray powder diffraction (XRD) using an X'Pert Pro θ–2θ diffractometer (PANalytical) with Cu Kα radiation. X'Pert Plus and Data Viewer software were used for analyzing the X-ray diffraction (XRD) data.
The carbon samples were also characterized by scanning electron microscopy (SEM) using a Quanta 3D FEG (EHT = 30 kV) instrument. The obtained nanoparticles were analyzed by high-resolution transmission electron microscopy (HRTEM). The images were taken using a F20X-TWIN (FEI-Tecnai) transmission electron microscope operated at 200 kV. The Raman spectra were measured using a Senterra micro-Raman spectrometer (Bruker Optiks, Billerica, MA, USA). The spectral parameters were set as follows: laser power, 2 mW at 532 nm; resolution, 4 cm−1; CCD temperature,−64 °C; laser spot, 2 µm; total integration time, 100 s (50 × 2 s); an objective 20× was used.
FTIR spectroscopic data were acquired by using a Hyperion 3000 FTIR microscope connected to the Vertex V70 (Bruker Optics) apparatus in the frequency range of 600–7000 cm−1. The spectral changes of the CF surfaces were collected after soaking the samples in deionized water and compared to the spectrum of the initial dry sample. Typically, 32 scans were collected using an instrument resolution of 4 cm−1 every 60 s.
Water adsorption isotherms were determined at a temperature of 25 °C using a gravimetric adsorption apparatus equipped with Baratron pressure transducers (MKS Instruments, Germany). Each sample was thermally desorbed before the measurement under high vacuum until a constant mass was obtained (after ca. 2 days).3,16
Immersion enthalpy and surface energy determinations were performed using the Tian–Calvet isothermal calorimeter constructed in our laboratory.17N-Heptane, deionized water, and formamide were used as standards. Each measurement was repeated at least three times.
The major assumption of the van Oss–Good–Chaudhury (VGC) model used in this study is the independence of the dispersive and acid–base interactions. Eqn (1) describes the enthalpy of immersion to the surface energy components of both the solid surface and the wetting liquid.
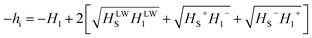 | (1) |
Moreover, the separation of the above components allows calculation of the sum i.e. total surface enthalpy (HTS).
| HTS = HLWS + HpolS | (2) |
Here the polar term is defined as:
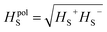 | (3) |
The energy components of the different probe liquids used in this study were from ref. 18 and 19.
The enthalpies of immersion of all samples in heptane, 1-chlorobutane, and 1-nitropropane were measured for electrostatic field strength determination (according to our earlier reports17). Each measurement was repeated at least three times, and the error was not larger than 0.5 mJ m−2.
As was previously described, the enthalpy of immersion ΔHi consists of the dispersion (hid), the polarization (hia), and the electrostatic forces (hiµ), which represent the interaction between the permanent dipole of the liquid and the static electric field on the surface of the solid.20,21
By assuming that for a given adsorbent the elements: (hid) and (hia), are constant, the changes in measured ΔHi correspond directly to the (hiµ). On the other hand, (hiµ) is usually defined as hiµ = −zµF, where F represents the strength of the surface electrostatic field of the solid, µ is the dipole moment of the liquid and z is the number of adsorbed molecules per unit surface area. Thus, for the enthalpy of immersion obtained by using a series of adsorbates differing in the dipole moments and having almost the same cross-sectional areas (for the mentioned above molecules ca. 20 Å2), it is possible, using the mentioned relation to obtain the intensity of the static electric field of the tested surface. For the studied molecules z comes roughly to 5 × 1018 molecules per m2.22 The dipole moment (µ) of n-heptane, 1-chlorobutane, and 1-nitropropane is equal to 0.00, 2.04 and 3.57 Debye, respectively.
Low temperature N2 adsorption isotherms were measured using an ASAP2010 volumetric adsorption analyzer from Micromeritics (Norcross, GA) at liquid nitrogen temperature (77 K) in the relative pressure range from about 10−6 up to 0.99. Before the measurements, the samples were outgassed for at least 12 h at 413 K (Fig. S1).
The hydrovoltaic measurements were performed by partially immersing a CF-X braid in Milli-Q water and then connecting the working and counter electrodes of the device to a multimeter (Fig. S2). Platinum was employed to rule out the contribution of any redox processes between the electrodes.
For the solar evaporation experiment, the fabricated evaporators were placed under a 50 W, 800 nm LED lamp (0.8 W cm−2). The evaporator was placed on an electronic balance (0.1 mg) to record the time-dependent mass change. A thermocouple was used to measure the surface temperature of the evaporator.
For the ab initio study, all of the calculations, at B3LYP levels of theory using 6-311G(d,p) basis sets, were performed using geometries optimized for the couple of compounds, as shown in Fig. 6 and S7. These calculations were performed using the Gaussian'09 program. For the calculations mimicking the OH-bond dissociation, the relaxed scan of a modredundant optimization was performed. Option “field” in the range of X = −000 to X = −200 was used to simulate the electrostatic field strength up to 10.28 V nm−1.
Results and discussion
Carbon fibers (CFs) were gently functionalized under high-temperature gas phase (air or O2) treatment to introduce oxygen functional groups (–C–OH, –CO, –COOH, and C–O) without drastically changing the morphology and electronic structure (Fig. 1). The effect of the process was confirmed through multiple techniques, including SEM (Fig. 1A and B), EDX (Fig. S2 and 3), Raman spectroscopy (Fig. 1C), XRD (Fig. 1D), and immersion calorimetry (Fig. 1E and F).Comparison of the images from microscopic analysis of the initial materials and after maximum oxidation, i.e., 24 hours at 375 °C in O2, proves the assumed hypothesis of intact fiber morphology (Fig. 1A and B). The material's surface is smooth in both cases, with no visible roughness.
The absence of morphological changes is also reflected in the absence of structural changes, as confirmed by XRD studies (Fig. 1C). The XRD pattern of CF-6 shows the same crystal planes as the initial CF-0 sample with no new peak formation, confirming its amorphous structure with typical wide and low-intense peaks at 25.6 and 44.1° attributed to the (002) and (100) planes, respectively.23
The first-order Raman spectra of carbonaceous materials in the range from about 1000 to 1800 cm−1 (Fig. 1D) are, despite their apparent simplicity, very complex. The dominant signals are located near 1350 and 1600 cm−1 and are known as the D1 and G bands, respectively. The G-band is associated with the carbon atoms of highly graphitized structures with sp2 hybridization and results from the presence of a doubly degenerate E2g peak centered in the Brillouin zone, while the D1 band is associated with the defective structure and results from a complex interaction with the electron band structure near the K-point.
Additional signals are usually detectable in this range of 1000–1800 cm−1, including a D2 peak near 1540 cm−1, which is formed by a similar mechanism to that of the D-band, and D3 and D4 bands near 1480 and 1150 cm−1, respectively. A comparison of Raman scattering spectra (Fig. 1D) of CF-1 and CF-6 materials confirms the statement that during the oxidation process, the relative changes in the degree of graphitization or disorder of carbon structures were minimal (G and D1 peaks remain practically unchanged, and the IG/ID is 0.33 for both materials). Nevertheless, the intensities of the D2 and D4 peaks increase while the intensity of D3 decreases. Thus, Raman spectra of materials after the process of gentle oxidation can be correlated with microstructural defects.
The EDX analysis of the tested samples reveals an increase in the atomic percentage of oxygen from 0.2 for the initial to 3.02% for the sample after 24 h of O2 treatment at 375 °C (Fig. S3 and S4). This increase is due to the formation of new carbon–oxygen bonds on the fiber surface. Based on the elemental analysis, the increase in oxygen amount is from 0.08 to 0.97% m m−1. Such results evidenced that oxygen is built only on the surface of CFs.
Surface oxidation of the CF surface is unfortunately hard to detect by IR spectroscopy, due to the high absorption of the carbon matrix in the infrared range. Thus, to prove the assumption, we need to look closer at the surface via analysis of adsorbent–adsorbate interactions. A simple and elegant solution in such cases is measuring the enthalpies of immersion in different probe liquids to characterize the surface energy components using the van Oss–Good–Chaudhury (VGC) approximation. In this model it is possible to separate the Lifshitz-Van der Waals component of the surface energy, HLWS, from the polar ones i.e. acidic, HS+, and the basic, HS−. Such a procedure leads to calculation of the total surface energy, HTS, as a sum of HLWS and HpolS.24 In order to achieve the enthalpies of immersion in different probe liquids (viz. water, n-hexane, and formamide), (hi) values were measured, and the results are displayed in Fig. 1E.
The characterization of the surface energy shows that values of both components increase during the oxidation process. The polar term increases from the value of 5 mJ m−2 for the starting material to 91 mJ m−2 after 24 h O2 treatment at 375 °C (Fig. 1E). Concurrently, the value of the non-polar surface energy increases from 14 to 65 mJ m−2. Such changes are easily explained by an increase in the number of surface functional groups and are in agreement with the literature data, where the increase in both energies was observed upon carbon fiber functionalization.25
The separation of the acidic and basic components of the surface free energy results from the ionic nature of the functional groups formed on the surface. The results summarized in Fig. 1F show a sharp increase in the acidic nature of oxidized CFs. The calculated HS+/HS− ratio is ca. 23 times higher for the CF-6 sample than for the initial material (CF-1). The increase in the acidic character causes the increase in surface polarity (defined as the HpolS/HTS ratio) and in the surface electrostatic field strength.
The enthalpy of immersion for solids in a variety of organic liquids as probe molecules has been reported22,26–29 and those studies focused mainly on inorganic heteropolar materials such as TiO2,22 zeolites,28 ZnO,29etc. For carbon materials, Graphon carbon black was used as a reference, non-porous, hydrophobic, homopolar solid.22 Also, Morita and Takami30 reported results for B-doped graphitized carbon fibers immersed in n-heptane and nitropropane. The authors concluded that the strength of the surface electrostatic field increased with the rise in concentration of oxygen on the carbon surface due to the enhancement of surface polarity.
The authors estimated the value of 4.83 GV m−1 for the material annealed for 1 hour in air at 750 °C, for which they estimated an O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C ratio of 0.05. A slightly lower electrostatic field strength value was obtained for the CF-6 sample (4.22 GV m−1) with a measured O
C ratio of 0.05. A slightly lower electrostatic field strength value was obtained for the CF-6 sample (4.22 GV m−1) with a measured O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) C ratio of 0.033. This clearly proves that the values of electrostatic field strength presented in this report perfectly agree with the results shown by Morita and Takami.30
C ratio of 0.033. This clearly proves that the values of electrostatic field strength presented in this report perfectly agree with the results shown by Morita and Takami.30
When a pair of materials, such as carbon fibers and Pt, is placed in water, their surfaces polarize adjacent water molecules, giving the possibility for the appearance of exotic structures by forming an electrical double layer at the solid–liquid interface. As a result, a measurable potential difference is created, strongly dependent on the surface chemistry, temperature, and resistance of the system (Fig. 2A and B). The temperature and conductivity of the external load have a negative effect on the values of the obtained potentials, while the oxidation state of the carbon fiber surface has a positive effect. The values of over 370 mV at a temperature of 22 °C at a load of 10 MΩ for CF-6 (material obtained after 24 h oxidation in O2) are achievable.
When the external load is reduced, the value of the flowing current increases, reaching the value of 8.2 µA for CF-6 at a load of 11 kΩ (Fig. 1C). Such high current values allow for achieving significant power values, which for the most oxidized sample reaches a maximum of close to 1 µW (Fig. 2D).
Charged water clusters are transported up through the material by capillary forces, where the water evaporates from the surface. This process generates the streaming current and the observed potential, the values of which are constant due to evaporation. The measured potential is strongly related to surface electrostatic field strength. A linear correlation is observed for each resistance used (Fig. S5), and not surprisingly, the relationships are parallel, thus proving the same mechanism of dependence on surface functional groups.
It has been proven in the literature that while an effective water flow is essential for the hydrovoltaic material, saturation must be taken care of. Kumar et al.1 proved that a hydrovoltaic surface can become saturated if the flow of water is too easy and the rate of evaporation is insufficient. The saturation of the device significantly hinders the capillary flow, an essential condition for the continuous generation of the potential and streaming current. To counteract this, the authors added a hydrophobic material without going into the essence of the phenomenon.
A seemingly different experiment is related to solar evaporators; carbon materials are also widely used in such systems (Fig. 3). By studying the evaporation rate of water from a series of modified carbon fibers, it can be concluded that the functionalization of the surface, i.e., its polarity, is also a decisive factor influencing the capillary flow rate.
 | ||
| Fig. 3 Water mass change using CF-braids as evaporators. The system under irradiation with an 800 nm laser (0.8 W cm−2) corresponding to the solar-to-vapor generation under an equivalent of 0.5 sun. | ||
The slowest evaporation of water was observed for the CF-1 sample (desorbed at 600 °C), which is understandable due to its hydrophobicity. However, the water did not evaporate the fastest from the most oxidized sample, CF-6, with the highest hydrophilicity. The best results, slightly better than for a typical Janus composite, were observed for the slightly oxidized CF-3 material, for which the surface had a potential of 1.3 GV m−1. We conclude here that the force of the water–surface interaction competes with the capillary flow. The weak water–surface interaction characteristic of hydrophobic surfaces (CF-1) does not induce any capillary flow. However, too strong an interaction inhibits the flow of evaporating water. This effect is perfectly visible when we correlate the evaporation rate with surface electrostatic field strength (Fig. S6). The rate reaches a maximum for a value of about 1.3 GV m−1. Decreasing this value causes a steep drop in the rate. On the other hand, an increase in the surface energy causes the effect to stabilize at a good level of about 4 kg m−2 h−1.
In order to explain why better results than the typical Janus-type system are observed for materials with lower hydrophilicity, two additional experiments were conducted: (i) the in situ phenomenon was investigated, recording spectral changes via FTIR, and (ii) the enthalpy of water adsorption on the studied materials was investigated calorimetrically.
Fig. 4 and S7 show spectral changes collected in the form of a map from the surface of the studied carbon fibers. It is worth explaining that the background was always a spectrum of the dry surface of the fiber, which means that any changes are caused by the presence of an H2O layer on the surface of the tested material. For clarity, on the right side, there are individual spectra from places marked with appropriate arrow colors.
The results presented here prove that the overall mechanism is based on different surface structures formed during different CF exposures to the water layer. The formation of an aqueous layer on the CF-1 material (Fig. 4A) is associated with the appearance of three characteristic bands: stretching, bending, and wagging of H2O molecules, at 3600, 1630, and ca. 800 cm−1, respectively. The additional signal visible as an shoulder on the lower wavenumber side of ν(OH) is attributed to the hydrogen bonding of water molecules. A change in the intensity ratio of the 3050/3600 cm−1 bands (Fig. S8) allows for the assessment of the size and durability of the layer closest to the surface.
The evaporation process causes a decrease in the intensity of the above-mentioned bands and the appearance of additional IR bands attributed to the water surface structures at 1400, 2600, and over 4000 cm−1. Their metastability causes fast disappearance with evaporation.
Different effects are observed during H2O desorption from oxidized materials (Fig. 4B and C). Stronger hydrophilicity of the surface causes the formation of stronger long-range hydrogen bonds. Fig. S6 for the CF-6 material proves that the ratio of 3050/3600 is close to unity after just 5 minutes of evaporation. This means that the adsorbate–adsorbent interaction in this case is very strong.
An intermediate situation occurs for the CF-3 material, where the value of the ratio of the 3050/3600 bands remains at about 0.5 up to 40 minutes of the process. Existing, but not very strong, hydrogen bonds can cause a weaker capillary flow, but the interactions with the adsorbent surface are not strong enough to inhibit the evaporation of the adsorbate. Interestingly, the FTIR spectra of the surface of this material (bands 1000–1600 cm−1 and 2000–2800 cm−1) prove the presence of Eigen–Zundel surface structures. Similar spectral changes are not described in the literature.
The main assumption here is to create a stable Stern layer at the surface of the carbon material. Strong, numerous hydrogen bonds stabilize the water clusters, which are mobile in the diffusion layer. The evaporation of water, causing continuous thinning of the diffusion layer, causes the shortening of hydrogen bonds, visible as shifting signals towards higher wavenumbers. The increasingly strong oxidation of the surface layer of the material causes the enlargement of the Stern layer, stabilizing exotic water structures.
The results of the calorimetric tests confirm different values of the interaction energies of water with the surfaces of the tested materials (Fig. 5). As expected, the highest water adsorption was observed for the most oxidized material, CF-6, and the lowest for CF-1, a material heated at 600 °C in an inert gas atmosphere.
The C-1 material shows the greatest hydrophobic properties; the course of the water adsorption isotherm is almost horizontal up to the value of 0.6 p/po, then it increases gradually to the value of ca. 40 µmol m−2 at maximum relative pressure (Fig. 5A).
In the case of the CF-6 sample, the strong adsorbate–adsorbent interaction raises the isotherm already from low p/po values to a value of ca. 73 µmol m−2. The material labeled CF-3 was characterized by intermediate values of water adsorption. The shape of the second type of isotherm proves the presence of both strong specific and weaker dispersion interactions at higher relative pressures.
The analysis of thermodynamic parameters confirms the above conclusions by supplementing them with further data. H2O adsorption on all tested materials is spontaneous, as evidenced by negative Gibbs energy values (Fig. 5C). For all materials, it was also observed that for the initial values of relative adsorption (A/Amax), the absolute value of adsorption enthalpy increases, reaching the maximum value at A/Amax of 01–0.2 (Fig. 5B). Such changes are typical for a well-cleaned surface of the carbon material and are associated with the dissociative adsorption of adsorbate molecules.2,3 The increase in adsorbate pressure (and adsorption) is accompanied by a gradual decrease in the absolute ΔH value, reaching a value close to 0 for the CF-1 sample and −2.85 kJ mol−1 for the CF-3 material. Only the most oxidized material (CF-6), with A/Amax about 0.4, is characterized by an increase in the absolute heat of adsorption (−15.54 kJ mol−1). Note that even for this sample, the measured value of ΔH is far from the condensation enthalpy (−40.7 kJ mol−1). Nevertheless, the strong correlation of integrated enthalpies of adsorption with surface electrostatic field strength (Fig. S9) was observed.
The interaction of the adsorbate with the surface is proportional to the surface energy of the material being tested, which translates into adsorption and the exothermic effect of the process. However, the evaporation rate does not correlate so easily. Equally interesting is the analysis of changes in adsorbate entropy, which, for low A/Amax, is positive, proving the correctness of the assumption about dissociative adsorption of water. For higher values of A/Amax, the entropy change should take negative values, indicating increasing order in the adsorption phase. This is only the case with the CF-6 sample. For other materials, ΔS is close to 0 for the CF-3 or positive for the CF-1 sample (Fig. 5D).
These results directly explain the reasons for the different rates of water evaporation from the tested materials. The strong adsorbate–adsorbent (CF-6) interactions result in high adsorption enthalpy values and a decrease in degrees of freedom, which Kumar et al.1 determined as saturation. Conversely, the hydrophobicity of the surface of the tested material, with a close to zero ΔH value and a positive TΔS, should mean very easy evaporation of water from the fiber surface. Such an effect proves the combination of three parameters: the surface wettability (manifested in the form of the capillary effect), the adsorption enthalpy (as a derivative of the surface energy), and the entropy factor, which, for lower energy surfaces, disables the formation of stable adsorptive layers.
Small absolute values of adsorption enthalpy for the CF-3 material, close to zero ΔS, with surface electrostatic potential ca. 1.3 GV m−1, give the best results. It is worth mentioning that such an effect has not yet been described in the literature.
In order to show that the results obtained for CF materials are not unique, comparative studies were carried out using model hydrophilic surfaces, i.e., graphene oxide and detonated nanodiamond (Fig. S10 and 11). On both considered surfaces, structures with a different geometry than typical water molecules are formed and disappear during the drying process. The presence of bands at 2820 and 2580 cm−1 for both materials clearly confirms the hypothesis.
In addition, spectral changes in the range of 1800–1200 cm−1 are also observed for both tested materials. Here, too, we find confirmation of the hypothesis that surface functional groups affect the geometry of water molecules in the adsorption layer.
The faithful simulation of water cluster compositions is very complicated. Therefore, in an attempt to reproduce the dynamics of water molecules during adsorption and desorption, we used a simplification defining the effect of an electrostatic field on two water molecules during dissociation.
The results presented in Fig. 6 and S12 show the energy gain that the system of two H2O molecules receives near the polarized surface. The energy of the system of two H2O molecules decreases gradually by about 72 kJ mol−1 in a field gradually raised up to 150 a.u. (7.71 V nm−1). The excess energy can then be used for stretching and/or possible dissociation of the OH bond. The fluctuations of the OH bond are accompanied by changes in the dipole moment of the 2-molecular system, visible as wandering IR bands. Their presence in the spectral regions of 900–1300 and 2000–3000 cm−1 has been repeatedly observed in the literature31,32 and interpreted as the formation of Eigen and/or Zundel-type structures. The simulated IR spectra prove that (i) the formation of E–Z structures is energetically possible, (ii) characteristic IR bands around 1200 and 3000 cm−1 can be expected in the IR spectra near polar functional groups, (iii) increasing the field intensity affects the intensity of the bands and not their position in the spectrum, and finally (iv) complete dissociation of the OH bond is energetically much easier near a polar functional group.
The results presented here are universal and can be directly transferred to other adsorption systems: metallic, oxide, or zeolite.
Conclusions
The main, proven assumption of the work was the formation of a stable Stern layer at the surface of oxidized carbonaceous materials. Caused by the oxidation process, an increase in the surface polarity enables the fluctuation of OH bonds. This increase manifests in the appearance of additional IR bands. Strong, numerous hydrogen bonds stabilize water clusters – Eigen–Zundel structures, which are unstable in the diffusion layer. By disintegrating and combining into new structures, they cause the formation of wrinkles in the time-resolution IR spectra.The water evaporation process, causing the continuous thinning of the diffusion layer, causes the shortening of hydrogen bonds, visible as shifting signals towards higher wavenumbers. It is worth mentioning that such an effect has not yet been described in the literature.
Finally, we explained how the functionalization of the fiber surface through changes in thermodynamic functions affects the capillary flow and thus the rate of water evaporation. We have shown that it is possible to obtain a material hydrophilic enough to have an effective capillary flow, while at the same time, low values of adsorption enthalpy, for which entropy changes are small enough to make water desorption highly effective.
In our opinion, the materials presented in this work can be directly applied on a larger scale both in real hydrovoltaic equipment and in seawater desalination systems.
Author contributions
Conceptualization, MW; methodology, MW, JM, AM, MS, and DO; validation, MW and JM; investigation, MW, JM, AM, MS, and DO; data curation, JM, MW, AM, MS, and DO; original draft preparation, MW; review & editing, MW, JM, AM, MS, and DO; visualization, MW, AM, and MS; supervision, MW.Conflicts of interest
There are no conflicts to declare.Data availability
All the datasets obtained during the current study are available from the corresponding authors on reasonable request.Supplementary information: among others, the low-temperature N2 adsorption isotherms of the tested samples; the EDX results for CF-1 and CF-6 samples; the operando results registered during H2O evaporation from the carbon fiber, graphene oxide, and nanodiamond surfaces. See DOI: https://doi.org/10.1039/d5ta06447c.
Acknowledgements
MW & JM gratefully acknowledge the financial support from the NSC Opus 23 project: UMO-2023/49/B/ST11/01341.Notes and references
- R. Kumar, G. Kay, G. B. G. Liu and K. Stamplecoskie, ACS Appl. Mater. Interfaces, 2023, 15, 7511–7517 CrossRef CAS PubMed.
- M. Wiśniewski, J. Moszczyńska and X. Liu, Langmuir, 2024, 40, 193–200 CrossRef PubMed.
- M. Wiśniewski, Int. J. Mol. Sci., 2022, 23, 14292 CrossRef.
- X. Yangyi, L. Chenxing, Z. Xiaojiao, T. Ningyao and Y. Miaomiao, ACS ES&T Water, 2021, 1, 2423–2430 Search PubMed.
- L. Zhang, B. Tang, J. Wu, R. Li and P. Wang, Adv. Mater., 2015, 27, 4889–4894 CrossRef CAS PubMed.
- F. Zhao, Y. Guo and X. Zhou, Nat. Rev. Mater., 2020, 5, 388–401 CrossRef.
- X. Li, J. Li, J. Lu, N. Xu, C. Chen, X. Min, B. Zhu, H. Li, L. Zhou, S. Zhu, T. Zhang and J. Zhu, Joule, 2018, 2, 1331–1338 CrossRef CAS.
- L. Cui, C. Ma, P. Wang, H. Che, H. Xu and Y. Ao, Appl. Catal., B, 2023, 337, 122988 CrossRef CAS.
- P. Tao, G. Ni and C. Song, Nat. Energy, 2018, 3, 1031–1041 CrossRef.
- F. Zhao, X. Zhou, Y. Shi, X. Qian, M. Alexander, X. Zhao, S. Mendez, R. Yang, L. Qu and G. Yu, Nat. Nanotechnol., 2018, 13, 489–495 CrossRef CAS PubMed.
- H. Ghasemi, G. Ni, A. M. Marconnet, J. Loomis, S. Yerci, N. Miljkovic and G. Chen, Nat. Commun., 2014, 5, 4449 CrossRef CAS.
- Y. Guo, C. M. Dundas, X. Zhou, K. P. Johnston and G. Yu, Adv. Mater., 2021, 33, 2102994 CrossRef CAS PubMed.
- X. Wu, Z. Wu, Y. Wang, T. Gao, Q. Li and H. Xu, Adv. Sci., 2021, 8, 2002501 CrossRef CAS.
- J. Zhou, Y. Gu, P. Liu, P. Wang, L. Miao, J. Liu, A. Wei, X. Mu, J. Li and J. Zhu, Adv. Funct. Mater., 2019, 29, 1903255 CrossRef CAS.
- W. Zhang, C. Wang, L. Wang, F. Wang, P. Tan, J. Ma, J. Jin, Z. Geng, H. Xie and L.-D. Zhao, Energy Environ. Sci., 2025, 18, 7916–7927 RSC.
- M. Wiśniewski, J. Moszczyńska, A. Marczyńska, A. Bielicka, Y. Yao and X. Liu, Small, 2025, 2412754 CrossRef.
- M. Wiśniewski, G. Rychlicki and A. Arcimowicz, Chem. Phys. Lett., 2010, 485, 331–334 CrossRef.
- J.-M. Douillard, F. Salles, M. Henry, H. Malandrini and F. Clauss, J. Colloid Interface Sci., 2007, 305, 352–360 CrossRef CAS.
- G. Gąsior, M. Grodzicka, T. Jędrzejewski, M. Wiśniewski and A. Radtke, J. Funct. Biomater., 2023, 14, 1–18 Search PubMed.
- J. J. Chessick and A. C. Zettlemoyer, Adv. Catal., 1959, 6, 263 Search PubMed.
- S. Hagiwara, K. Tsutsumi and H. Takahashi, Carbon, 1971, 9, 693 CrossRef CAS.
- F. H. Healey, J. J. Chessick, A. C. Zettlemoyer and G. J. Young, J. Phys. Chem., 1954, 58, 887 CrossRef CAS.
- T. Liu, X. Li, C. Xu and H. Zhang, ACS Appl. Mater. Interfaces, 2017, 9, 4626–4633 CrossRef CAS PubMed.
- J. Taguta, B. McFadzean and C. O'Connor, Miner. Eng., 2019, 143, 105954 CrossRef CAS.
- P. Han, L. Yang, S. Zhang and Z. Gu, Nanomaterials, 2022, 12, 2778 CrossRef CAS.
- T. Takei and M. Chikazawa, J. Colloid Interface Sci., 1998, 208, 570 CrossRef CAS PubMed.
- J. Silvestre-Albero, C. Gomez de Salazar, A. Sepulveda-Escribano and F. Rodriguez-Reinoso, Colloids Surf., A, 2001, 151, 187–188 Search PubMed.
- K. Tsutsumi and H. A. Takahashi, J. Phys. Chem., 1970, 74, 2710 CrossRef CAS.
- T. Morimoto and Y. Suda, Langmuir, 1985, 1(239), 8–12 Search PubMed.
- T. Morita and N. Takami, Electrochim. Acta, 2004, 49, 2591 CrossRef CAS.
- W. Kulig and N. Agmon, J. Phys. Chem. B, 2014, 118, 278–286 CrossRef CAS PubMed.
- S. Das, S. Imoto, S. Sun, Y. Nagata, E. H. G. Backus and M. Bonn, J. Am. Chem. Soc., 2020, 142, 945–952 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |





