Stability challenges in non-aqueous Li–O2 batteries and their protective strategies: a comprehensive review on electrode and electrolyte engineering
Sekar
Sandhiya
and
Perumal
Elumalai
 *
*
Electrochemical Energy Storage Lab, Department of Green Energy Technology, Madanjeet School of Green Energy Technologies, Pondicherry University, Puducherry-605014, India. E-mail: drperumalelumalai@pondiuni.ac.in; drperumalelumalai@gmail.com
First published on 10th December 2025
Abstract
Non-aqueous Li–O2 batteries are at the forefront of next-generation energy storage research due to their exceptionally high theoretical energy density, which could surpass that of conventional Li-ion batteries. However, despite their potential, practical implementation is hindered by several critical challenges including poor cycle life and high overpotentials due to the instability of the lithium metal anode, air cathode, and electrolyte. The ineffectiveness of electrocatalysts in non-aqueous Li–O2 systems is critically examined. Although catalysts such as noble metals and transition metal oxides have been explored to enhance the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER), their performance in non-aqueous environments remains suboptimal due to side reactions and catalyst degradation. To provide a detailed understanding of the ORR and OER kinetics, the rotating ring-disk electrode (RRDE) technique is discussed as a valuable tool for evaluating the catalytic activity, identifying reaction intermediates, and probing the role of superoxide species in battery degradation. More detailed discussions of the challenges/strategies with regard to the anode, cathode, electrolyte, redox mediator (RM), and oxygen selective membrane and the recent advances are presented. This review focuses on threefold strategies for protecting the lithium metal anode, air cathode, and electrolyte, which are highly susceptible to attack by reactive oxygen species (ROS) and their intermediates (O2−, LiO2, 1O2, and O22−), lithium metal corrosion, parasitic reactions, electrolyte degradation/volatilization, electrode passivation, and sluggish kinetics. Special emphasis is placed on advanced protective approaches including the design of artificial solid–electrolyte interfaces (SEIs), inducing solution-mediated growth of lithium peroxide (Li2O2), and electrolyte stabilization by a threefold strategy using electrolyte additives, which shows promise in extending the battery cyclability. This review aims to provide a comprehensive understanding of the key issues limiting the performance of non-aqueous Li–O2 batteries and explores potential pathways to overcome these challenges through advanced electrolyte additive engineering and electrochemical techniques.
1. Introduction
The transition to efficient and sustainable energy storage systems is pivotal in meeting the escalating demands of the electric vehicle (EV) market, the growing wearable electronics and smart device sectors. While Li-ion batteries (LIBs) remain the dominant choice of energy source for smaller consumer electronics, their limited specific energy (less than 300 Wh kg−1) renders them insufficient for large-scale applications such as grid storage and the EV traction. This limitation has driven the quest for developing alternative energy storage solutions with high specific energy.1,2 Among the proposed alternatives, Li–Air (Li–O2) batteries have garnered significant attention due to their exceptionally high theoretical specific energy, which can reach as high as 3500 Wh kg−1, even when the mass of air is accounted. Li–O2 batteries refer to battery systems operating in an oxygen environment, while “Li–Air batteries” use ambient air that generally contains N2, H2O, and CO2, which introduce parasitic reactions and performance decay. A typical Li–O2 battery comprises a lithium metal anode, ambient oxygen as the cathode, and a lithium salt dissolved in an organic solvent as the electrolyte. The utilization of the oxygen from the air as the cathode material makes Li–O2 batteries lightweight, cost-effective, and sustainable. Despite their promising attributes, the practical implementation of Li–O2 batteries is hindered by several challenges including electrolyte volatility, high overpotentials, poor cycle life, poor rate capability, and the sluggish oxygen reduction reaction/oxygen evolution reaction (ORR/OER) kinetics due to the insulating nature of the discharge product and unstable cell environment. The inherently slow kinetics of these oxygen reactions significantly limits the efficiency and rechargeability of Li–O2 batteries, underscoring the critical need for effective bifunctional electrocatalysts to enhance these surface reactions. Electrocatalysts in Li–O2 batteries facilitate oxygen adsorption, subsequent reduction, and the desorption of discharge products. The electronic structure and surface properties of the catalyst profoundly influence these catalytic activities. Further, several strategies devoted to improving catalyst performance include altering the chemical composition, defect engineering, and microstructural tuning. However, these modifications are often complex and fail to address the instability of the electrolyte toward reactive superoxide species formed as intermediates during discharge.3–6 Superoxide-induced decomposition of the electrolyte can neglect the catalytic benefits and further compromise the battery performance. Hence, a better strategy needs to be developed to give significant protection to the aprotic Li–O2 battery.7 This review provides a comprehensive analysis of the ORR and OER kinetics pertaining to Li–O2 batteries, with a focus on leveraging advanced diagnostic techniques such as the rotating ring-disk electrode (RRDE) method. The RRDE technique is a valuable tool for evaluating catalytic activity, identifying reaction intermediates, and elucidating the role of O2− in battery degradation. The challenges, strategies, and recent advances in the anode, cathode, electrolyte, redox mediator (RM), and oxygen-selective membrane of the Li–O2 battery are discussed more deeply. In this review, particular emphasis is placed on the development of a threefold protective strategy using electrolyte additives to overcome the limitations of non-aqueous Li–O2 batteries. This approach aims to address the key challenges in all the components, including electrolyte stabilization, air cathode and lithium metal anode protection, paving the way for long-cycle, rechargeable Li–O2 batteries suitable for practical applications.2. Metal–air batteries
Metal–air batteries typically consist of four primary components: a reactive metal anode, an air cathode, an electrolyte, and a separator. During discharge, the metal anode undergoes oxidation, releasing electrons that flow to the cathode through an external circuit. Simultaneously, oxygen diffuses into the cathode, accepts electrons, and is reduced to oxygen-containing species. These reduced oxygen species interact with metal ions transported through the electrolyte to form metal oxides as discharge products, termed ORR. During charging, the reverse processes occur, resulting in metal plating at the anode and oxygen evolution at the cathode, referred to as the OER. Historically, aqueous metal–air batteries such as Fe–Air, Al–Air, and Mg–Air have been studied since the 1960s, with Zn–Air batteries being commercialized as early as 1935.8 Despite their promise, aqueous systems face challenges like metal corrosion and hydrogen evolution.9–11 To mitigate these issues, surface passivation via oxide or hydroxide formation has been employed.9,12 In non-aqueous systems, such as Li–Air and Na–Air batteries, aprotic electrolytes enable the formation of metal superoxide or peroxides, with Li–Air batteries being particularly notable for their high theoretical energy density of 3500 Wh kg−1 and cell voltage of 2.96 V, as shown in Fig. 1(a).132.1 Lithium–air battery components
The components of Li–Air batteries can be categorized based on the type of electrolyte used, including aqueous, non-aqueous, solid-state, and hybrid systems.14,15 The metal anode serves as the negative electrode, where oxidation occurs, releasing electrons. Lithium, with its low reduction potential and high theoretical specific capacity, is a preferred anode material despite challenges such as dendrite formation, which can be mitigated using polymer electrolytes.16,17 The cathode plays a vital role in facilitating the ORR and OER, relying on electrocatalysts with high durability, porosity, and catalytic activity.18 However, ambient air introduces contaminants such as CO2 and H2O, which can react with Li+ ions, forming insulating byproducts such as lithium carbonate (Li2CO3), thereby reducing the efficiency.19 The electrolyte is crucial for ionic conductivity and battery stability. Aqueous electrolytes face issues like parasitic reactions and hydrogen evolution, while aprotic electrolytes including organic solvents and ionic liquids offer broader electrochemical windows but suffer from instability and side reactions with oxygen species.20–222.2 Non-aqueous Li–O2 batteries
Non-aqueous metal–air batteries include Li–Air, Na–Air, and K–Air systems. The schematic of the metal–air battery and reaction mechanism in aqueous and non-aqueous electrolytes is shown in Fig. 1(b). Among these, the Li–Air battery stands out due to its exceptionally high theoretical energy density, as shown in bar graph in Fig. 1(a), superior rechargeability compared to aqueous systems, and high potential for real-world applications like military and civilian uses. Consequently, extensive research efforts are directed toward developing rechargeable non-aqueous Li–Air batteries due to the stable cell environment and simple chemistry.23 In 2006, Bruce demonstrated the favourable electrochemical performance of Li–Air batteries, catalysing global interest in Li–Air battery technology.24Fig. 2 shows the timeline of different metal–air battery chemistries developed, lithium metal protection strategies, and the evolution of RMs over the years.25–50 The foundation for Li–Air batteries was laid in 1996 when Abraham and Jiang proposed a rechargeable Li–Air battery comprising a lithium metal anode, an organic polymer electrolyte membrane for Li+ conduction, and a carbon composite cathode.38 To date, laboratory-scale Li–Air batteries predominantly utilize pure oxygen instead of ambient air to avoid undesirable reactions with components such as H2O and CO2, which can disrupt electrochemical processes. Batteries using pure oxygen are referred to as Li–O2 batteries. The first prototype of Li–O2 was reported by Semkow and Sammells.51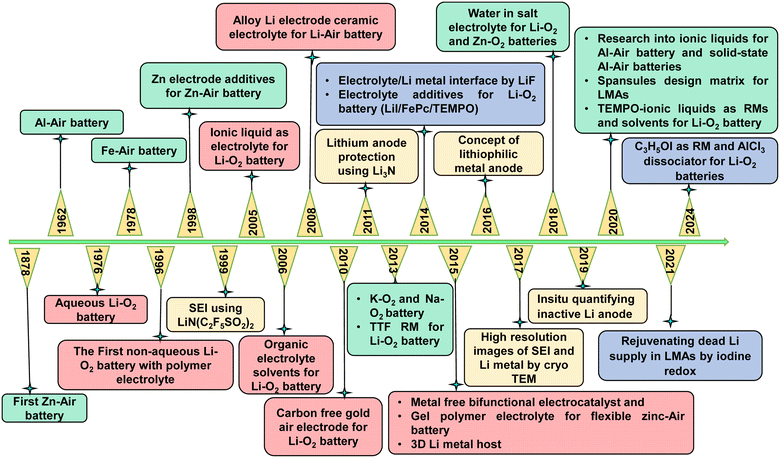 | ||
| Fig. 2 Timeline of the development of different metal–air batteries, lithium metal protection strategies, and RMs proposed over the years. | ||
During discharge, lithium undergoes oxidation at the anode, releasing Li+ ions, while simultaneously, oxygen at the cathode undergoes reduction. Initially, O2 is reduced to form superoxide (O2− and/or LiO2), an unstable intermediate within the electrochemical system. This superoxide rapidly undergoes further reduction, leading to the formation of peroxide species, lithium peroxide (Li2O2), as the final discharge product, accumulating on the porous cathode surface. The 2e− reduction from molecular oxygen to Li2O2 provides better reversibility than the alternative 4e− reduction from O2 to lithium oxide. Conversely, during charging, Li2O2 undergoes oxidation, decomposing into lithium ions and releasing molecular oxygen. Table 1 shows the different discharge products of the non-aqueous Li–O2 battery and their properties. The fundamental electrochemical reaction at the anode involves lithium oxidation:52
| Li → Li+ + e− (E° = −3.05 V) | (1) |
| Characteristic | Lithium peroxide (Li2O2) | Lithium oxide (Li2O) | Lithium superoxide (LiO2) |
|---|---|---|---|
| Formation role | Predominant discharge product | Not typically formed under ambient conditions | Generally, forms an intermediate, subject to disproportionation |
| Electron transfer pathway | 2e− pathway | 4e− pathway | 1e− pathway |
| Gibbs free energy | Low (ΔG = −571 kJ mol−1) | High (ΔG = −561 kJ mol−1) | — |
| O–O bond cleavage | Not required | Required | Not required |
| Discharge reaction | O2 + 2e− + 2Li+ → Li2O2 (E° = 2.96 V) | O2 + 4e− + 4Li+ → 2Li2O (E° = 2.91 V) | Li + O2 + e− → LiO2 |
| Theoretical capacity | 1168 mA h g−1 | 1794 mA h g−1 | 688 mA h g−1 |
| Specific energy | ∼3400 Wh kg−1 | ∼5220 Wh kg−1 | — |
| Performance | High charge overpotential | Low charge overpotential and long cycle life | Theoretically proven to have a long cycle life |
At the cathode, the desired reaction in a non-aqueous system involves the adsorption and deposition of reduced oxygen species.
| O2 + e− → O2− (E° = −0.09 V) | (2) |
Overall reaction is as follows:
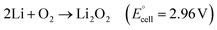 | (3) |
2.3 Discharge mechanisms in Li–O2 batteries
The desired discharge product in the non-aqueous Li–O2 battery is Li2O2, which follows two pathways as shown below, and the different discharge products formed in a non-aqueous Li–O2 battery are shown in Table 1.53| Li(sol)+ + O2(g) + e− → LiO2(sol) | (4) |
LiO2(sol) ↔ O2(sol)− + Li(sol)+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) | (5) |
The LiO2 formed in solution is stabilized by solvent molecules and further undergoes a disproportionation reaction as follows:
| 2LiO2(sol) → Li2O2(sol) + O2/1O2(g) | (6) |
Li2O2 then precipitates out of the solution, forming a crystalline structure.
| O2(g) + e− + Li(sol)+ → LiO2(ads) | (7) |
The surface-adsorbed LiO2 undergoes further reduction to form Li2O2:
| LiO2(ads) + e− + Li(sol)+ → Li2O2(ads) | (8) |
This mechanism typically results in a thin film of Li2O2 on the cathode, which may cause passivation, limiting the capacity and efficiency.
2.4 Charging mechanisms in Li–O2 batteries
Two primary mechanisms for solid Li2O2 oxidation during charging have been proposed:Two-electron process:
| Li2O2 → 2Li+ + O2 + 2e− | (9) |
One-electron process:
| Li2O2 → LiO2 + Li+ + e− | (10) |
| LiO2 → Li+ + O2 + e− | (11) |
The two-step one-electron process is less common due to the poor solubility of Li2O2 in non-aqueous electrolytes. Additionally, the insulating nature of Li2O2 contributes to high charge overpotentials. In the surface-mediated pathway, Li2O2 forms directly on the cathode surface through the reaction of Li+ ions with adsorbed oxygen.54 This results in a thin, uniform Li2O2 layer on the cathode surface due to the high adsorption capability of LiO2, which minimizes the volume of the discharge product and helps maintain cell integrity. The reaction is localized, reducing the complexity of intermediate species transport. It can benefit from conductive cathode surfaces or catalysts that support efficient electron transfer/ion diffusion during charging. The electronic configuration of catalytic metal sites significantly affects their ability to adsorb oxygen species, thereby influencing the morphological evolution of Li2O2 from toroidal to film-like structures. As described by the d-band centre theory, the position of the d-band plays a pivotal role in adsorption behaviour. A higher d-band centre promotes stronger orbital hybridization between the metal's d-orbitals and the oxygen's 2p orbitals, enhancing oxygen binding and boosting the efficiency of the oxygen reduction process. The continuous growth of the Li2O2 grains on the surface can block active sites, leading to a limited capacity. Dense or poorly conductive Li2O2 films (thickness less than 10 nm) may inhibit further reactions and increase the overpotentials, while in the solution-mediated pathway, Li2O2 forms in the electrolyte solution as dissolved Li+ ions and oxygen interact, and the product precipitates onto the cathode or forms as suspended particles. The formation of Li2O2 in the solution minimizes direct deposition on the cathode surface, preserving active sites and improving the cycle life. Li2O2 precipitates often have a toroidal or porous structure (thickness less than 2 µm), which facilitates oxygen and lithium-ion transport, enhancing capacity but with increased overpotential and singlet oxygen issues. The solution-mediated reactions are more compatible with RMs, which further reduce overpotentials and improve the reaction kinetics. This mechanism requires stable electrolytes that can dissolve intermediate species without degradation. Studies show that low-donor-number (DN) solvents promote surface-mediated growth, while high-DN solvents favor solution-mediated pathways.55,56Fig. 3 illustrates the ORR mechanism proceeding in the high and low DN non-aqueous solvents for the non-aqueous Li–O2 battery. To boost the discharge capacity of the Li–O2 battery, researchers have explored high-DN solvents and functional additives (e.g., H2O2 and NO3−) to enhance LiO2 dissolution.57–59 However, superoxide species are highly nucleophilic, accelerating solvent degradation and side reactions.60,61 The disproportionation of LiO2 is a significant source of singlet oxygen, a key driver of parasitic side reactions. High-DN solvents also suffer from lower electrochemical stability due to increased vulnerability to nucleophilic attacks, leading to more byproducts.62–65
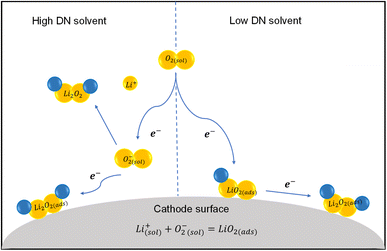 | ||
| Fig. 3 Schematic of the ORR mechanisms occurring in high- and low-DN non-aqueous solvents in Li–O2 batteries. | ||
A promising alternative involves using 2,5-di-tert-butyl-1,4-benzoquinone (DBBQ) as an RM in low-DN solvents, stabilizing LiO2 in the solution, and reducing solvent degradation.66
While ORR mechanisms are well understood, the OER pathway remains debated. A few studies suggest that Li2O2 decomposes directly into O2, while others propose multi-step mechanisms.67–71In situ surface-enhanced Raman spectroscopy (SERS) studies in CH3CN and DMSO electrolytes indicate that LiO2 forms during the ORR but may or may not reappear in the OER, depending on the system.72,73 However, Taylor et al.,74 using the same technique, reported a different perspective in DMSO-based electrolytes. Further investigations using RRDE and X-ray absorption near-edge structure (XANES) reveal that high-DN solvents promote a solution-mediated Li2O2 decomposition pathway via soluble LiO2, whereas low-DN solvents favor a solid surface.75 Although soluble LiO2 improves the charge kinetics, it compromises the solvent stability. Therefore, a deeper understanding of the OER pathway is critical for selecting stable electrolyte systems.
3. Bifunctional electrocatalysts
3.1 Optimizing air-breathing electrode architecture for Li–O2 batteries
The cathode catalyst/air-breathing electrode architecture is critical for enhancing Li–O2 battery performance. Abundant active sites, high catalytic activity, electronic conductivity, nominal adsorption and desorption capabilities, ensuring structural stability and high porosity are essential features of advanced air electrodes.76 Modifications to this structure aim to maximize active sites for the ORR and OER, enhancing the battery performance. The key design of air-breathing electrode architecture must be considered, which enables oxygen diffusion and discharge product storage. The optimal pore size prevents blockage by Li2O2 gains while ensuring sufficient reaction sites, promoting ion transfer, and maximizing the reaction area by enhancing liquid-electrolyte contact with the porous electrode, which accelerates the ORR and OER kinetics, improved through doping to modulate oxygen chemisorption and electron transfer, thus increase the density of low-coordination sites, enhancing the charge transfer. High surface area and tailored pore sizes are critical, as excessively small pores become occluded by the discharge products which reduce the specific capacity. Increasing the electrode surface area enhances the electronic conductivity by donating free electrons.77,78The development of efficient air electrodes hinges on optimizing catalyst properties such as morphology, wettability, and oxygen transport. Organic electrolytes in non-aqueous Li–O2 batteries introduce challenges due to insoluble discharge products (e.g., Li2O2 and Li2CO3) that block the reaction sites. Effective design strategies include enhancing electrolyte compatibility and electrode stability, reducing overpotentials to improve the charge/discharge efficiency, and tailoring the catalyst structure to enable reversible Li2O2 formation and decomposition.84 Future research should prioritize scalable, low-cost materials with high bifunctional catalytic activity to improve energy efficiency, cycle life, and capacity retention in Li–O2 batteries. Additionally, the integration of advanced in situ characterization techniques is essential to gain deeper insights into the complex reaction mechanisms and improve the overall battery performance. A comprehensive and consistent design of the air electrode will be crucial for achieving the promise of the Li–Air batteries, especially for applications in the EVs.
3.2 Oxygen reduction reaction/oxygen evolution reaction
A comprehensive understanding of the ORR and OER mechanisms is critical for the development of efficient bifunctional electrocatalysts. The ORR pathways can proceed via either a two-electron or a four-electron transfer route, depending on the catalytic material and the adsorption method preferred. The ORR involves four key steps: (i) electron transfer from the anode to adsorbed O2, (ii) surface adsorption and diffusion of O2, (iii) elimination of OH− ions into the solution, and (iv) weakening and splitting of O![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bonds.85
O bonds.85
In acidic electrolytes, oxygen reduction follows two primary pathways:
Direct four-electron reduction:
| O2 + 4H+ + 4e− → 2H2O (E° = 1.229 V vs. SHE) | (12) |
Two-electron reduction:
| O2 + 2H+ + 2e− → H2O2 | (13) |
| H2O2 + 2H+ + 2e− → 2H2O | (14) |
| 2H2O2 → O2 + 2H2O | (15) |
The 4e− pathway reduces O2 directly to H2O and can proceed through dissociative or associative mechanisms. The dissociative mechanism is favoured under strong O2 binding or low surface oxygen coverage, while the associative mechanism is preferred under weak O2 adsorption or high surface oxygen coverage. The presence of peroxide intermediates in the two-electron pathway can lead to undesirable degradation of the cell, making it less favourable for practical applications.86
In alkaline electrolytes, ORR mechanisms are more complex due to the formation of intermediates such as O, OH−, O2−, and HO2−. These pathways are categorized as follows:
Four-electron pathway:
| O2 + 2H2O + 4e− → 4OH− (E° = 0.401 V) | (16) |
Two-electron pathway:
| O2 + 2H2O + 2e− → HO2− + OH− (E° = −0.065 V vs. SHE) | (17) |
| HO2− + 2H2O + 2e− → 3OH− (E° = −0.067 V vs. SHE) | (18) |
Each pathway offers distinct advantages and is suitable to specific applications. For instance, the 2e− pathway is desirable for hydrogen peroxide (H2O2) production, while the 4e− pathway is preferred in fuel cells for achieving high current efficiencies.87,88Fig. 4(a) illustrates the proposed ORR mechanisms in acidic and alkaline media, highlighting the associative and dissociative pathways. The intricate interplay of these mechanisms underscores the need for precise catalyst design to optimize the performance for a specific application.
 | ||
| Fig. 4 (a) Schematic of the ORR mechanism and (b) OER mechanism in the acidic medium occurring on the metal oxide cathode catalyst. | ||
The mechanisms for the OER under both acidic and alkaline conditions, involving intermediates such as M–OH and M–O, have been proposed as follows:89
| 2H2O → 2H2 + O2 | (19) |
Acidic conditions:
Cathode reaction
| 4H+ + 4e− → 2H2O (E° = 0 V) | (20) |
Anode reaction
| 2H2O → O2 + 4H+ + 4e− (E° = 1.23 V) | (21) |
Alkaline conditions:
Cathode reaction
| 4H2O + 4e− → 2H2 + 4OH− (E° = −0.83 V) | (22) |
Anode reaction
| 4OH− → O2 + 2H2O + 4e− (E° = −0.40 V) | (23) |
Under acidic conditions, the OER proceeds through either (i) a direct combination of two O* species to generate O2 (blue pathway) or (ii) the formation of the OOH* intermediate (yellow pathway). Under alkaline conditions, represented by the black pathway, the reaction involves the generation of O2 and H2O via the reverse mechanism. Despite the differences in pathways, the OER catalytic performance is commonly influenced by bonding interactions with intermediates such as M–OH, M–O, and M–OOH. Fig. 4(b) illustrates the OER mechanisms on metal-oxide electrocatalysts in alkaline (black pathway) and acidic (blue pathway) media.90,91
The OER is inherently a multi-electron reaction, involving a sequence of steps that include electron transfer and chemical reactions such as association or dissociation. The specific OER mechanism on a given electrocatalyst can often be deduced from the measured Tafel slope, which correlates with the electron transfer coefficient (α). Deeper the understanding of the electrode kinetics for both the ORR and the OER, RRDE measurements were adopted by several groups to investigate key kinetic parameters, which are discussed in the subsequent sections.
3.3 Key parameters for bifunctional ORR/OER activities
The kinetics of ORR and OER was effectively investigated using the RRDE technique. RRDE setup comprises a disk electrode (typically glassy carbon) and a concentric ring electrode (commonly platinum) separated by an insulating barrier, as illustrated in Fig. 5(a and b). The electrode configuration can be customized in terms of materials, dimensions, and designs based on research objectives. For ORR and OER studies, glassy carbon is preferred as the disk substrate due to its negligible intrinsic catalytic activity for the ORR and its compatibility with any catalyst layer fabrication.92 Platinum, however, is often employed as the ring electrode material because of its stability and ability to detect intermediates such as peroxide species. The radii of the disk (r1), inner ring (r2), and outer ring (r3) are critical parameters in the RRDE geometry, influencing the electrochemical analysis. During operation, the rotating motion of RRDE generates centrifugal forces that drive the electrolyte radially outward across the electrode surface while normal flow replenishes the solution at the surface. Increased rotation speeds enhance convection, accelerating the diffusion of the reactants to the electrode surface. The electrochemical reaction at the disk electrode is represented as follows:| O + ne− ↔ R | (24) |
 | ||
| Fig. 5 (a) Schematic of RDE and RRDE. (b and c) Schematic of chemical reactions occurring on the surface of RRDE and RDE. (d) Typical three-electrode setup used for catalytic activity measurements. | ||
When an electrocatalyst is applied to the cathode for the ORR, intermediates or byproducts like H2O2 may form. RRDE enables detection of these species through collection experiments on the ring electrode. The schematic of the chemical reaction on the surface of RRDE is shown in Fig. 5(c).93 A bi-potentiostat is employed to independently control the potentials of the ring and disk electrodes (Er and Ed) relative to the counter and reference electrodes. The bi-potentiostat connections shown schematically in Fig. 5(d).94 By regulating the potentials, cyclic voltammetry and linear sweep voltammetry measurements can be conducted to probe the ORR and OER activity. This experimental approach offers a detailed understanding of electrocatalytic behaviour, making it an indispensable tool for advancing bifunctional catalyst development.
4. Electrochemical techniques/parameters for the ORR/OER kinetics
To assess the catalytic activity of bifunctional electrocatalysts, the RRDE method is among the most effective techniques. Below, several electrochemical methods and parameters essential for evaluating the kinetics of ORR and OER are discussed.95,964.1 Cyclic voltammetry (CV)
Cyclic voltammetry is a fundamental technique for assessing the catalyst activity towards the ORR. Initially, the catalyst-coated electrode is immersed in an oxygen-saturated electrolyte to record cyclic voltammograms. A prominent reduction wave indicates catalytic activity, with the onset potential reflecting the catalyst's redox behaviour. Conversely, in an argon-saturated environment, the absence of this wave highlights the catalyst's inactivity towards the ORR. The experiments are conducted generally in a three-electrode configuration comprising a working electrode, a reference electrode, and a counter electrode. A slow potential scan rate (<10 mV s−1) is typically used to ensure steady-state behaviour, with the ORR potential range spanning 0–1 V versus the reversible hydrogen electrode (RHE) in 0.1 M KOH. To maintain oxygen concentration during the measurements, continuous oxygen bubbling in the electrolyte is employed. Fig. 6(a) depicts a schematic of current–potential curves that can be obtained from RRDE during CV analysis.4.2 Linear sweep voltammetry (LSV)
Linear sweep voltammetry is a widely employed technique to investigate the ORR kinetics. The technique involves recording current–potential curves at various electrode rotation rates in an oxygen-saturated 0.1 M KOH electrolyte, maintaining a potential range of 0–1 V versus RHE and a low scan rate (e.g., 10 mV s−1). The LSV curves typically exhibit three distinct regions, each corresponding to specific electrocatalytic processes. The high-potential region represents surface reaction-controlled kinetics, with the onset potential (Eonset) and half-wave potential (E1/2) serving as quantitative indicators of catalytic activity. A more positive onset potential signifies superior catalytic performance. The mixed kinetic-diffusion and diffusion-controlled regions are also distinguishable, as shown in Fig. 6(b).4.3 Tafel analysis
The Tafel slope (b) provides insights into the reaction pathway and rate-determining step (RDS) and can be derived using the Tafel equation: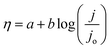 | (25) |
4.4 Koutecky–Levich analysis
The rotating disk electrode (RDE) technique is frequently used to study the electrocatalytic ORR performance. Parameters such as onset potential (Eonset), half-wave potential (E1/2), and overpotential (ηj) at a specific current density, diffusion-limiting current density (jL), and kinetic-limiting current density (jK) can be extracted from polarization curves. The Koutecky–Levich (K–L) equation correlates the electrochemical and hydrodynamic properties of the electrocatalyst: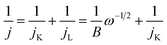 | (26) |
| B = 0.62nFC*D2/3ν−1/6 | (27) |
4.5 Hydrogen peroxide quantification
The H2O2 quantification is conducted using RRDE, equipped with a coaxial platinum ring. The disk generates intermediate species that diffuse to the ring, enabling their detection. Using the following equations, the electron transfer number (n) and percentage of H2O2 are calculated: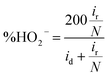 | (28) |
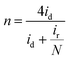 | (29) |
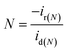 | (30) |
5. Fundamental parameters of a lithium–air battery
For developing an efficient air-breathing electrocatalyst, the aforementioned parameters are vital, which can give information such as the e− transfer number, reaction rate and the mechanism. Besides, the following key parameters are to be considered to evaluate the performance, specific capacity, cell voltage, energy density, power density, cycle life, and coulombic efficiency of a Li–Air battery.5.1 Specific capacity
Specific capacity refers to the total charge stored per unit mass of the active material coated on the electrode. It is a fundamental measure of the battery's capacity and can be calculated for any electrochemical reaction using the equation: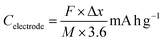 | (31) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1), Δx is the number of electrons involved in the redox reaction, and M is the molar mass of the active material (g mol−1). For a full cell, the specific capacity can be expressed as follows:
485 C mol−1), Δx is the number of electrons involved in the redox reaction, and M is the molar mass of the active material (g mol−1). For a full cell, the specific capacity can be expressed as follows: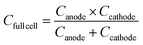 | (32) |
5.2 Cell voltage
The cell voltage (or potential) is the difference in potential between the anode and the cathode, determined by the thermodynamic favourability of the electrode reactions. The Gibbs free energy (ΔG) of the reaction provides the driving force for these reactions and is used to calculate the equilibrium potential: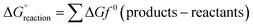 | (33) |
The equilibrium cell voltage is given as follows:
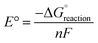 | (34) |
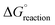 is the Gibbs free energy change under standard conditions, n is the number of electrons transferred, and F is the Faraday constant.
is the Gibbs free energy change under standard conditions, n is the number of electrons transferred, and F is the Faraday constant.
5.3 Energy density and power density
Energy density and power density are critical for understanding the storage and delivery capabilities of a battery. Energy density or specific energy represents the amount of energy stored per unit mass (or volume) of the active material. It is calculated as shown below:| Gravimetric energy (E) = C × V (Wh kg−1) | (35) |
A battery with high energy density can store significant energy in a compact form, whereas high specific energy means a battery can hold high energies in a given small mass.
Power density or specific power indicates the rate at which the energy can be delivered. It is determined by dividing energy by the discharge time as shown below:
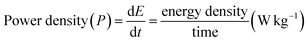 | (36) |
Batteries with high power density (W L−1) can deliver large amounts of energy quickly, whereas batteries with specific power can discharge quickly with small gravimetric mass.
5.4 Coulombic efficiency, cycle life, and shelf life
Coulombic efficiency (CE) is the ratio of discharge capacity to charge capacity, expressed as a percentage: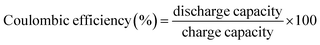 | (37) |
An ideal battery will have a CE of 100%, indicating fully reversible redox reactions.
Cycle life refers to the number of complete charge–discharge cycles a battery can undergo before its capacity significantly degrades. Each cycle causes a loss of active material, leading to a gradual decline in energy retention.
Shelf life is the duration over which a battery retains at least 80% of its capacity during storage. Shelf life depends on the stability of electrode materials, storage temperature, and inactivity period.
6. Challenges, advances, and strategies in non-aqueous Li–O2 batteries
6.1 Non-aqueous electrolytes
Electrolyte instability continues to be one of the most significant challenges in the advancement of Li–O2 batteries. It is the conductive solution of Li salt dissolved in a non-aqueous solvent. The aggressive electrochemical environment, particularly the formation and interaction of the reactive oxygen species (ROS) such as O2−, LiO2, 1O2, and Li2O2 severely impacts the structural and chemical stability of the electrolyte components.97,98 An ideal electrolyte for a Li–O2 battery must combine low viscosity, high ionic conductivity, enhanced Li+ salt and oxygen solubility, wide electrochemical and thermal stability windows, non-flammability, low volatility, and strong chemical compatibility with lithium metal, cathode materials, separators, and current collectors. It should also demonstrate efficient wetting of porous electrodes, resistance to moisture-induced degradation, and the ability to support long-term cycling without decomposition.99–101 The electrolyte solvents for the Li–O2 battery include organic carbonates, ethers, sulfones, esters, nitriles, amides, phosphates, and ionic liquids. Initial research on the Li–O2 battery electrolytes utilized alkyl carbonate solvents, such as ethylene carbonate and propylene carbonate (PC), derived from technology due to their low volatility and broad electrochemical stability.102–104 However, these solvents failed under Li–O2 battery operating conditions, rapidly degrading into Li2CO3 and alkyl lithium carbonates in the presence of the ROS instead of facilitating the desired Li2O2 formation.105–109 This led to the adoption of ether-based solvents, including 1,3-dioxolane (DOL) and dimethoxyethane (DME), which offered improved stability and rate performance.110–112 Nevertheless, ethers also exhibited vulnerability to decomposition via hydrogen abstraction, auto-oxidation, and nucleophilic attack by superoxide species. Advanced in situ techniques such as differential electrochemical mass spectrometry (DEMS), Raman spectroscopy, and X-ray diffraction (XRD) confirmed that the degradation pathways resulted in undesirable byproducts like lithium formate and CO2.105 Zhang et al.113 introduced a fluoro-ether (HFE) solvent that converts these nucleophilic reactions into beneficial pathways, and its decomposition products (LiF and cross-linked polymers) form a robust, flexible cathode electrolyte interface that suppresses polarization and enhances capacity retention. Tetraethylene glycol dimethyl ether (TEGDME) is a stable solvent for Li–O2 batteries due to its low volatility and chemical stability in oxygen-rich environments, but forms unstable SEI, lithium dendrites, and irreversible stripping plating when used with LiTFSI due to strong Li+–TEGDME interactions.114 Introducing trifluoroacetate (TFA−) anions weakens this interaction, favoring anion-rich solvation.115,116To mitigate these degradation mechanisms, researchers have explored the use of stabilizing additives, high-concentration salts, and structural solvent modifications. One of the key degradation contributors, singlet oxygen (1O2), can be quenched by facilitating its conversion to triplet oxygen (3O2) using molecules like 1,4-diazabicyclo[2.2.2]octane (DABCO), N,N-di-(2,2,6,6-tetramethyl-1-oxyl-4-piperidinyl)-perylene-3,4,9,10-tetracarboxylic diimide (PDI-TEMPO), 9,10-dimethylanthracene (DMA), and dimethyl phenazine (DMPZ), which reduce the oxidative side reactions.117–119 However, their poor stability and limited quenching efficiency hinder practical use. Triphenylamine (TPA), with a reported quenching efficiency of 99.3%, shows promise.120 For long-term viability, effective quenchers must combine high reactivity toward 1O2 with strong chemical and electrochemical stability, without adding complexity to the system. Similarly, RMs such as DBBQ were shown to stabilize LiO2 intermediates and facilitate low-overpotential decomposition of the Li2O2, thereby improving reversibility and limiting parasitic reactions.121 The inclusion of DBDMB in TEGDME-based electrolytes has enabled the formation of toroidal Li2O2 structures via a solution-based mechanism, improving the discharge capacity and cycling stability. Moreover, DBDMB's redox potential of 4.2 V vs. Li+/Li allows it to assist in decomposing Li2CO3 and LiOH, which are often irreversible under normal operating voltages.122,123 DMSO-based electrolytes have garnered significant attention due to their high stability against LiO2, enhanced oxygen diffusion coefficient, Li+ conductivity, and high DN, which enhances Li2O2 solubility and promotes longer cycle life.124,125 However, prolonged exposure to ROS caused DMSO to degrade into lithium methyl sulfonates and carbonate species, causing the capacity to fade.126–129 As a result, localized high-concentration electrolytes were developed to retain the benefits of DMSO, possess higher activation energy barriers for C–H bond scission from the CH3 group, while reducing free solvent reactivity. Compared to the conventional solvents such as TEGDME and PC, DMSO-based electrolytes delivered higher discharge voltages (up to 2.8 V vs. Li+/Li) and significantly improved capacity even when paired with standard carbon cathodes.130 Liu et al.131 investigated high-concentration electrolytes for both DMSO and DME in LiTFSI salts; the results indicated improved stabilization of the Li–O2 battery performances; however, the high-concentration electrolytes also lead to high lithium metal anode corrosion and poor conductivity due to high viscosity. In parallel, tetramethylene sulfone (TMS) was introduced, offering advantages such as low volatility, high thermal and electrochemical stability (up to 5.6 V vs. Li+/Li), and good lithium salt solubility. Batteries employing TMS demonstrated remarkable discharge capacities, highlighting its potential as a safe and efficient electrolyte solvent.132 Due to the poor conductivity and insolubility of Li2O2, many electrolyte design strategies have primarily focused on modifying the morphology of the discharge product to boost capacity, often overlooking the impact on lithium metal stability. Guided by Pearson's hard and soft acid–base (HSAB) theory, solvents with high DN have been commonly utilized to enhance the solubility of reactive intermediates like LiO2, which can facilitate the formation of larger Li2O2 structures and improve the discharge performance.133,134 According to Johnson et al.,135 high DN solvents containing stronger solvated Li+ or Li+-containing species with equilibrium shifted to the right (1-methylimidazole, DN = 47) to promote the solubility of LiO2, and in low DN solvents, the solvation is weaker with the equilibrium shifted to the left (CH3CN, DN = 14) to promote surface adsorption of LiO2. Popular choices include DMSO and DME, both of which support higher capacities by promoting intermediate dissolution. However, this increased solubility also intensifies the reactivity of the superoxide species, which are strong nucleophiles. As a result, such solvents become more vulnerable to nucleophilic degradation or proton abstraction, leading to the generation of detrimental side products that accelerate lithium anode corrosion. Following the introduction of the LiCF3SO3 in TEGDME (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 ratio) by Sun et al.,136 this combination gained traction in the field. Nevertheless, issues such as high viscosity, poor ionic transport, and reactivity with superoxide radicals limited their broader applicability. While N,N-dimethylacetamide (DMA) exhibits better resistance to aggressive intermediates like O2− and O22−, it suffers from instability when in contact with lithium metal.137 Ionic liquids (ILs) provide another promising class of electrolytes with high oxidative stability, non-volatility, and wide electrochemical windows.138 It is a molten salt that exists in the liquid phase at a temperature below 100 °C. Ionic liquid is a mixture of organic cations and organic/inorganic anions. The cations are usually pyrrolidinium-, sulfonium-, ammonium-, imidazolium-, pyrazolium, phosphonium, and oxazolium-based salts. Pyridinium- and pyrrolidinium-based ILs have demonstrated resistance to superoxide attack, while imidazolium-based ILs remain susceptible.139–142 Mixing ILs with solvents like DMSO enhances the conductivity and overall performance.143
4 ratio) by Sun et al.,136 this combination gained traction in the field. Nevertheless, issues such as high viscosity, poor ionic transport, and reactivity with superoxide radicals limited their broader applicability. While N,N-dimethylacetamide (DMA) exhibits better resistance to aggressive intermediates like O2− and O22−, it suffers from instability when in contact with lithium metal.137 Ionic liquids (ILs) provide another promising class of electrolytes with high oxidative stability, non-volatility, and wide electrochemical windows.138 It is a molten salt that exists in the liquid phase at a temperature below 100 °C. Ionic liquid is a mixture of organic cations and organic/inorganic anions. The cations are usually pyrrolidinium-, sulfonium-, ammonium-, imidazolium-, pyrazolium, phosphonium, and oxazolium-based salts. Pyridinium- and pyrrolidinium-based ILs have demonstrated resistance to superoxide attack, while imidazolium-based ILs remain susceptible.139–142 Mixing ILs with solvents like DMSO enhances the conductivity and overall performance.143
Amide-based solvents such as dimethylformamide (DMF) and hexamethylphosphoramide (HMPA) exhibit good oxidative stability but pose challenges due to reactivity with lithium metal.144,145 The nitrile-based solvents, particularly acetonitrile (MeCN), have shown promise due to their chemical resistance against the ROS.146 Bruce et al.147,148 demonstrated a two-electron oxygen reduction mechanism in MeCN, while McCloskey et al. highlighted its high rechargeability. Nonetheless, issues such as self-condensation, hydrolysis under moist conditions, and MeCN's reactivity with lithium metal limit its utility. Johnson et al.149 reported hydroperoxide-mediated degradation of MeCN, resulting in acetamide formation, revealing its vulnerability in Li–O2 systems.
To address lithium dendrite formation and improve safety, solid-state electrolytes (SSEs) currently emerge as attractive alternatives. The SSEs are broadly classified into solid-inorganic electrolytes (SIEs) and solid-polymer electrolytes (SPEs).150–152 Inorganic options such as LISICON, NASICON, garnet-type (e.g., LLZO), and perovskite oxides provide high ionic conductivity and wide electrochemical stability windows. Among them, sulfide-based SIEs are generally unsuitable for the Li–Air battery due to the evolution of toxic H2S gas. However, oxide-based electrolytes, LATP and LAGP, are prominent candidates, although LATP reacts unfavourably with lithium, reducing Ti4+ and increasing interfacial resistance.153,154 This issue was addressed by applying buffer layers such as LiPON, BNRA, or Li3N as reported.122,155–158 The LAGP shows better compatibility with lithium metal and supports longer cycle life. The solid-polymer electrolytes and their derivatives offer improved safety, mechanical flexibility, and cost-effectiveness.159,160 Gel polymer electrolytes (GPEs) consist of liquid electrolytes immobilized within polymer matrices such as poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP) or cellulose acetate, which enhance the ionic conductivity. Composite polymer electrolytes (CPEs), incorporating inorganic fillers such as SiO2 or Li10GeP2S12 further improve the electrochemical and mechanical properties. Despite their potential, the SPEs and CPEs still suffer from low ionic conductivity at ambient temperatures and partial permeability to oxygen species, requiring additional improvements for long-term Li–O2 operation.161,162
The role of lithium salts is equally critical. Typical non-aqueous lithium salts are LiPF6, LiClO4, LiBF4, LiCl, Li[B(C2O4)2], Li[N(SO2CF3)2], LiTFSI, LiTf, LiNO3, and LiBOB. Salts such as LiPF6, LiClO4, LiTFSI, LiTf, LiNO3, and LiBOB influence solid electrolyte interface (SEI) formation, ion transport, and electrolyte stability. LiTf and LiTFSI are desirable owing to their high conductivity and stability, but they tend to decompose into LiF.163 However, LiClO4 exhibits better resistance to ROS but presents safety risks.164 LiNO3 stands out for stabilizing Li2O2, lowering charge overpotentials, and reducing carbon electrode degradation.165 Zhang et al.137 optimized the electrolyte by adjusting the lithium salt composition and concentration in DMA. A formulation containing 2 M LiTFSI and 1 M LiNO3 exhibited superior stability with lithium metal. This medium-concentration electrolyte not only reduced cell polarization but also sustained extended cycling over 180 cycles. Post-cycling XRD analysis of the anode showed the absence of LiOH signatures, indicating a stabilized interface facilitated by the optimized electrolyte. Further surface analysis confirmed the presence of LiF and LiNxOy within the SEI, which contributed to the anode's enhanced durability. Given lithium's high inherent reactivity with the most conventional electrolytes in Li–O2 cells, engineering compatible electrolytes presents another route toward anode stabilization. The key approaches include tailoring lithium salts, introducing highly stable separators or membranes, increasing salt concentrations to reduce the solvent activity, adding RM, and deploying gel or SSEs. The type and amount of lithium salts used in the electrolyte play a critical role in determining the stability of the lithium anode. For instance, LiTNFSI has demonstrated superior oxidative resilience and facilitates the development of fluorine-rich interfaces, which are mechanically robust and chemically stable.166 Unlike traditional LiTFSI, LiTNFSI also minimizes unwanted reactions triggered by oxygen species, thereby improving the efficiency of oxygen reutilization during cycling. High-concentration electrolytes have shown efficacy in reducing free solvent molecules, thereby minimizing unwanted lithium solvent reactions. Electrolyte strategies to enhance lithium stability extend to structural electrolyte engineering. For instance, hybrid solid electrolyte designs combining a rigid ceramic core (e.g., LAGP or LLZO) with a flexible polymeric shell (e.g., PVDF-HFP) have demonstrated stable lithium-ion flux and improved interfacial contact. These designs not only suppress dendrite growth but also facilitate better integration of the lithium metal with the SSEs. Advanced interface engineering methods such as nano polishing of the LAGP surfaces and in situ electrolyte gelation reduce interfacial resistance and enhance SEI uniformity. Cathode–electrolyte interfaces (CEIs) remain another critical bottleneck in solid-state Li–Air batteries. Since Li2O2 is poorly conductive and forms insulating discharge films, achieving sufficient triple-phase boundary contact between air, cathode, and SSE is essential.167 Moreover, the inclusion of CO2 and H2O from ambient air introduces irreversible side products such as Li2CO3 and LiOH·H2O. Innovative separator designs including Nafion membranes, graphene–PEO composites, and MOF-based films have been proposed to mitigate oxygen crossover and dendrite penetration. Gel-based and quasi-solid electrolytes bridge the gap between liquid and solid systems. Hybrid gel electrolytes using ionic liquids and LAGP powders have exhibited excellent electrochemical performance.168 Despite significant advances, no universal electrolyte system currently meets all the requirements for long-term, safe, and efficient Li–O2 operation. Each class of electrolyte whether liquid, solid, or gel offers distinct benefits and faces unique limitations. Achieving a balance between oxygen solubility, lithium compatibility, ROS tolerance, and SEI stability will be essential. The electrolyte plays a vital role in facilitating Li+ movement between the two electrodes. Careful selection of the solvents, salts, and additives can promote the formation of a robust SEI on the lithium anode surface, enabling stable Li plating/stripping while minimizing dendrite formation. An ideal electrolyte should not only protect the lithium anode but also exhibit strong resistance to the ROS.
6.2 Lithium anodes
Lithium metal stands as a cornerstone in the pursuit of next-generation, high-energy-density battery technologies, particularly Li–Air batteries, owing to its exceptional theoretical specific capacity of 3860 mA h g−1, low atomic weight of 6.94 g mol−1, and the lowest electrochemical potential of −3.05 V vs. SHE.169 These intrinsic advantages make lithium metal a highly appealing choice as an anode material. However, the realization of practical Li–O2 battery systems using Li metal is significantly hindered by its extreme chemical reactivity due to its high Fermi energy level. Upon exposure to the ambient conditions or the electrolyte environments, lithium readily undergoes a series of degradation processes, including surface corrosion, dendritic growth, electrolyte decomposition, and volume changes during cycling. These issues collectively undermine the safety, electrochemical performance, and life span of Li–Air batteries, thereby necessitating a comprehensive understanding of lithium degradation mechanisms and strategies to mitigate them. The interaction of lithium with atmospheric gases has been an area of research interest dating back to the late 19th century.170 Early studies postulated that lithium remains largely unreactive toward dry gases such as nitrogen (N2), O2, and carbon dioxide (CO2) at room temperature, citing that chemical reactivity with these gases only becomes prominent above certain threshold temperatures (approximately 160 °C for N2, 250 °C for O2, and 300 °C for CO2). However, these observations were primarily based on macroscopic reactions and lacked the resolution to detect subtle surface changes. With the advent of advanced characterization tools such as X-ray photoelectron spectroscopy (XPS), ultraviolet photoelectron spectroscopy (UPS), and density functional theory (DFT) modelling, these classical assumptions have been revisited. It is now well established that lithium can undergo spontaneous surface oxidation even in dry O2 environments at room temperature, leading to the rapid formation of Li2O layers.171–173 These oxide layers, although offering temporary passivation, are often porous and non-uniform, rendering them insufficient as long-term protective barriers. In addition to the oxygen reaction at the cathode catalyst, the Li-metal anode itself can undergo ORR when exposed to the O2 atmosphere, leading to parasitic formation of Li2O and LiOH, surface corrosion, and dendritic growth. Wu et al.174 introduced a dithiobiuret (DTB) additive into an ether-based electrolyte and tailored the Li+ solvation sheath to form an anion-derived, F-rich and O-deficient SEI that effectively resists oxygen penetration and reactive species attack. This regulated SEI architecture not only suppresses dendrite formation but also enables uniform Li deposition and enhanced cycling stability under O2, underscoring the vital role of anode electrolyte interfacial design in mitigating ORR-induced degradation in Li–O2 batteries. While dry nitrogen has traditionally been regarded as an inert background gas in electrochemical systems, emerging studies have shown that nitrogen can participate in electrochemical reactions under specific conditions. In lithium–nitrogen battery systems, where O2 is replaced with N2 as the cathodic gas, lithium reacts with nitrogen to form lithium nitride (Li3N), a compound that exhibits electrochemical reversibility during charge–discharge cycles. The formation of Li3N is associated with a discharge voltage plateau around 1 V, indicating that nitrogen is not entirely inert in the presence of highly reducing lithium metal. This finding has profound implications for the design and understanding of Li–air batteries, where N2 constitutes approximately 78% of the ambient air. Although Li3N can function as a passivating interface, it becomes unstable in the presence of moisture, reacting to form ammonia (NH3), a volatile and potentially harmful byproduct.175–177 This underscores the complexity of lithium's interaction with nitrogen in realistic environments. Carbon dioxide represents another critical atmospheric component that influences the surface chemistry of lithium. Several studies have demonstrated that CO2 not only reacts with lithium directly but also affects the fundamental discharge chemistry of the Li–Air battery. In the presence of CO2, the predominant discharge product shifts from Li2O2 to lithium carbonate (Li2CO3).178 This shift is undesirable for two primary reasons. First, Li2CO3 has much lower electronic conductivity compared to Li2O2, impeding electron transport during discharge and charge processes. Second, Li2CO3 is thermodynamically stable and difficult to decompose electrochemically, which results in higher overpotentials during the charging phase and reduced round-trip energy efficiency. Interestingly, in the environments with extremely low CO2 concentrations (e.g., <100 ppm), Li2O2 remains the dominant product, indicating that the influence of CO2 is highly concentration dependent.179In elevated CO2 atmospheres, Li2CO3 forms via multi-step reactions that involve intermediates such as lithium oxalate (Li2C2O4). Advanced techniques like in situ ambient pressure XPS (APXPS) has confirmed the formation of stratified SEI structures with Li2CO3 layers situated atop Li2O.180 Although Li2CO3 is generally considered detrimental due to its insulating nature, under dry conditions it can function as a stable and protective surface layer that mitigates further lithium degradation. Building on this concept, a hybrid Li–O2/CO2 battery was designed using a palladium-loaded carbon nanotube (Pd/CNT) cathode. Unlike conventional Li–O2 cells that suffer from rapid lithium corrosion and cell failure within 20 cycles, this system achieved over 500 stable cycles.181 Post-cycle analysis revealed that a nanosheet-like Li2CO3 film had formed on the lithium surface, resulting from a reaction between LiOH and CO2. This film acted as an effective passivation barrier against both moisture and chemically aggressive oxygen species. The influence of CO2 on ROS was further elucidated using RRDE measurements. In a pure O2 environment, both disk and ring currents are typically observed due to the migration of O2− intermediates. However, in the O2/CO2 mixture, only disk currents were detected, implying that O2− was rapidly scavenged by CO2 to form stable CO4− intermediates. This reaction suppressed the disproportionation of O2− into singlet oxygen (1O2), a reactive species known to cause oxidative degradation of electrolytes and electrode materials. The combined effect of physical passivation and chemical scavenging allowed the Li–O2/CO2 battery to reach an extended cycling stability of 715 cycles at a current density of 500 mA g−1 and a specific capacity of 500 mA h g−1.181 Our group has studied the influence of CO2 on the performance of Li–O2 batteries, demonstrating that even limited CO2 exposure can impact Li–O2 battery longevity, emphasizing the need for CO2-tolerant catalysts capable of promoting not only the decomposition of Li2O2 but also partial or complete oxidation of Li2CO3.182 The influence of CO2 on reaction pathways, cell stability, and catalyst selection must be considered. Therefore, minimizing CO2 ingress or developing CO2-tolerant electrolytes and catalysts remains crucial for realistic Li–Air operation.183 While the effects of O2, N2, and CO2 are relatively well studied, the role of moisture cannot be overstated. Moisture is universally recognized as the most harmful atmospheric contaminant in Li-based batteries. Even trace levels of water can induce profound changes in interfacial chemistry. Water reacts exothermally with lithium to form LiOH and hydrogen gas, which not only leads to lithium consumption but also increases the risk of thermal runaway, internal pressure buildup, and explosion. The reaction sequence often begins with the formation of Li2O, followed by hydrolysis to LiOH, and ultimately to LiOH·H2O under humid conditions.184 These hydrated hydroxides are hygroscopic, poorly conductive, and mechanically unstable, leading to uneven lithium deposition and dendrite nucleation. Moreover, water influences discharge chemistry by altering the morphology of Li2O2. Under controlled humidity conditions, water has been shown to promote the formation of larger, plate-like Li2O2 particles, which can improve the discharge capacity. However, this morphological benefit is short-lived, as continuous exposure to water leads to the formation of unstable SEI layers and rapid capacity decay. Water also facilitates the degradation of other SEI components such as Li2CO3 and Li3N, further compromising interfacial stability. Consequently, stringent moisture control is imperative for lithium handling and cell assembly. In laboratory settings, lithium is typically stored under inert atmospheres (e.g., argon or nitrogen) with water levels maintained below 0.01 ppm. For commercial applications, lithium components are vacuum-sealed or encapsulated in paraffin wax. Further, the cell assembly is conducted in a glovebox maintained at dew points below −50 °C to prevent atmospheric moisture ingress.185 In parallel, lithium metal reacts readily with the electrolyte components, producing a spontaneously formed SEI. The composition and structure of this SEI are critical in dictating the electrochemical performance of the anode. In the Li–Air battery, the SEI is predominantly composed of LiOH, Li2O, and Li2CO3, with LiOH being the primary component due to reactions with trace water and oxygenated species. The porous and non-uniform nature of the LiOH layers renders them ineffective as long-term barriers, leading to continuous SEI evolution, dendrite formation, and electrolyte depletion. Additional organic components such as ROLi and ROCO2Li are occasionally observed but are less prevalent due to the oxidative nature of the Li–O2 environment.186 Oxygen crossover from the cathode to the anode further complicates SEI chemistry and contributes to parasitic reactions.187 Another serious degradation pathway involves RM shuttling. In the Li–Air battery utilizing soluble RMs to lower overpotentials, the oxidized form of the mediator can diffuse across the electrolyte and react with the lithium metal surface.188–191 This redox shuttling leads to the continuous consumption of the RM and exacerbates lithium corrosion, severely affecting the coulombic efficiency and life span of the cell. These interactions underscore the importance of designing protective strategies such as introducing electrolyte additives, concentrated electrolytes, hybrid ionic liquid electrolytes, high modulus separators, coating protective layers, and solid/gel-type electrolytes, which not only stabilize lithium surfaces but also mitigate mediator crossover.
Given these challenges, extensive efforts have been devoted to the development of robust protective layers (PLs) that can preserve lithium integrity under ambient conditions.188,192 These PLs must meet several criteria: they must be chemically and electrochemically stable, provide mechanical integrity to accommodate volume changes during cycling, exhibit high lithium-ion conductivity, and resist the permeation of moisture and reactive gases. The permeability of the gases through a PL is determined by the product of their solubility coefficient (Sa) and diffusion coefficient (Da).193 Water vapor, due to its small kinetic diameter and high polarity, exhibits both high solubility and diffusivity, making it the most challenging species to exclude. Consequently, hydrophobic, densely packed materials with low free volume are preferred for constructing the PLs with superior environmental resistance.194 Artificial SEIs or engineered interfaces can be synthesized via various strategies. Solid-state methods involve mechanical patterning or the use of pre-structured membranes to improve uniformity and reduce localized current densities. For example, Li surfaces modified with 3D copper mesh frameworks or microstructured patterns showed improved dendrite resistance. Polymer-based membranes such as polydimethylsiloxane (PDMS) provide flexibility and conformal coverage, allowing them to accommodate volume changes during cycling.195–197 Liquid-phase approaches include solution-casting of polymers or composites, as well as in situ chemical reactions using highly reactive solvents/electrolyte additives such as fluoroethylene carbonate (FEC).198–201 These techniques yield conformal, ionically conductive layers that offer both mechanical stability and interfacial passivation. Gas-phase engineering methods such as chemical vapor deposition (CVD), physical vapor deposition (PVD), and direct gas treatments with fluorinated or sulfur-containing species produce ultrathin, defect-free films.202,203 Plasma treatments are also employed for their ability to generate uniform coatings through surface activation under mild conditions.204,205 Beyond coatings, alloying lithium with other elements such as silicon, tin, or aluminium (forming LixMy alloys) improves the structural integrity and reduces the reactivity with air and electrolytes.206 Composite designs incorporating lithium into hydrophobic matrices, such as graphene-wrapped foils or vertically aligned graphene architectures (e.g., rAGA-Li), provide physical shielding against environmental attack. These designs maintain structural integrity even after extended air exposure.207 Dual-metal systems such as Li–Na alloys, where both elements contribute to redox reactions, have shown improved reversibility and reduced dendrite formation. In these systems, discharge products such as Li2O2 form simultaneously, demonstrating the electrochemical activity of both metals.208 In conclusion, lithium degradation in the Li–O2 battery is driven by the complex combination of chemical, electrochemical, and mechanical processes. Protective strategies such as engineered SEIs, composite hosts, and alloying have shown promise but require further optimization to withstand the unique challenges of the Li–O2 environment. Future progress will depend on an integrated understanding of interfacial chemistry, gas–solid interactions, and materials engineering to achieve long-term cycling stability and safety for practical Li–Air battery systems.
6.3 Air electrodes
Air electrochemistry encompasses the electrochemical processes occurring at the interface between the cathode and the electrolyte. The reactivity and nature of these interactions are strongly influenced by both the properties of the cathode material and the composition of the air, including gases such as N2, CO2, and H2O. These factors collectively shape the complexity of the air-based electrochemical systems, directly impacting the feasibility of practical Li–Air batteries. Understanding these mechanisms at a fundamental level is crucial for advancing toward realistic battery applications. For example, varied Li2O2 morphologies have been observed across different cathode materials, demonstrating the essential role of cathode design in modulating air electrochemical reactions. When oxygen is substituted with ambient air, components like N2 and CO2 actively participate in the electrochemical pathways during charge and discharge reactions, which alter the fundamental reaction routes. Therefore, it is critical to explore how these pathways evolve and how species such as N2 and CO2 interact with intermediates like Li2O2, O2−, and LiO2. During the discharge process, Li2O2 accumulates on the cathode surface. This discharge product, being electronically insulating, blocks active sites and hinders the transport of electrons and lithium ions, ultimately increasing polarization, reducing capacity, and minimizing the cycle life. The growth morphology of Li2O2 also significantly influences cell performance. Toroidal or sheet-like structures are preferred over dense layers, as the latter impedes charge transfer more effectively.209–212 The sluggish ORR and OER kinetics are caused by the poor catalytic reaction. Innovative cathode materials and architectures have been developed to mitigate passivation and enhance performance: materials such as carbon nanotubes (CNTs) and graphene provide high conductivity, surface area, and porosity, facilitating the uniform deposition of Li2O2 grains. However, these systems face challenges such as elevated overpotentials and limited cycling stability under high current conditions. Recent advancements indicate that introducing heteroatom dopants or structural defects into carbon frameworks can effectively enhance the ORR/OER kinetics. Additionally, nanostructured carbon-transition metal hybrids exhibit improved resilience against oxidative degradation. Strategies like increasing oxygen vacancy density and tailoring low-energy crystal facets in transition metal oxides have also been shown to significantly accelerate electrochemical reactions in Li–O2 batteries. Enhanced 3D structure with porous or tubular designs enhances oxygen diffusion and provides space for discharge products, reducing polarization. Using electrolyte additives lowers the overpotential by promoting solution-mediated Li2O2 formation and decomposition.213 Precise engineering of electrocatalyst morphology and composition at the sub-nanometre scale plays a pivotal role in enhancing oxygen electrocatalysis for Li–O2 batteries. One-dimensional nanostructures, such as nanowires, offer increased atomic exposure and high aspect ratios, which enhance active site density and facilitate directional charge transport. Concurrently, high-entropy oxides (HEOs), composed of multiple metallic elements, provide advantages such as entropy stabilization, lattice distortion, and synergistic interactions resulting in improved catalytic performance, tunable activity, and structural robustness. The integration of these features in sub-nanometer HEO nanowires has demonstrated promising outcomes; for instance, BiCuFeCeWPtOx-PMA spinel nanowires delivered a high discharge capacity of 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 206 mA h g−1 with stable cycling over 213 cycles as reported by Ge et al.214 Heterostructure phase engineering, supported by theoretical simulations, has revealed that internal electric fields at dissimilar phase interfaces can drive charge redistribution, establishing distinct nucleophilic and electrophilic domains that accelerate both the ORR and the OER. Ternary composite catalysts integrating oxides, sulfides, and phosphides such as NiCo2O4/NiCoP/NiCo2S4 have demonstrated outstanding performance, achieving discharge capacities up to 25
206 mA h g−1 with stable cycling over 213 cycles as reported by Ge et al.214 Heterostructure phase engineering, supported by theoretical simulations, has revealed that internal electric fields at dissimilar phase interfaces can drive charge redistribution, establishing distinct nucleophilic and electrophilic domains that accelerate both the ORR and the OER. Ternary composite catalysts integrating oxides, sulfides, and phosphides such as NiCo2O4/NiCoP/NiCo2S4 have demonstrated outstanding performance, achieving discharge capacities up to 25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 162 mA h g−1 and facilitating vertical Li2O2 growth and efficient decomposition.215 Recent developments emphasize the construction of core–shell architectures, porous frameworks, and materials with engineered internal fields to improve charge transport and catalytic uniformity. Additionally, electronic modulation through defect creation, heterojunction formation, and coupling phases with contrasting electronic properties has proven effective in stabilizing intermediates and reducing energy barriers. These advancements collectively contribute to improved capacity, cycling durability, and reaction reversibility. Two-dimensional (2D) materials, including graphene, MOFs, transition metal chalcogenides, and MXenes, exhibit excellent surface tunability and mechanical flexibility. Despite their promise, issues such as self-restacking and limited conductivity often restrict their independent performance. Mixed-dimensional integration (e.g., 0D/2D or 1D/2D hybrids) has been shown to enhance ion diffusion and active site accessibility. In particular, 2D/2D face-to-face heterostructures unconstrained by lattice matching enable maximized interfacial interactions and tunable electronic properties. For example, Ti0.87O2, a lepidocrocite-type oxide with abundant Ti vacancies, offered strong Li+ affinity but suffered from low conductivity. When coupled with conductive MXene layers via van der Waals and hydrogen bonding, the resulting 2D/2D Ti0.87O2/MXene heterostructure demonstrates an electronic compensation mechanism that enhanced catalytic activity and lowered ORR/OER overpotentials. This hybrid system delivered a high capacity of 13
162 mA h g−1 and facilitating vertical Li2O2 growth and efficient decomposition.215 Recent developments emphasize the construction of core–shell architectures, porous frameworks, and materials with engineered internal fields to improve charge transport and catalytic uniformity. Additionally, electronic modulation through defect creation, heterojunction formation, and coupling phases with contrasting electronic properties has proven effective in stabilizing intermediates and reducing energy barriers. These advancements collectively contribute to improved capacity, cycling durability, and reaction reversibility. Two-dimensional (2D) materials, including graphene, MOFs, transition metal chalcogenides, and MXenes, exhibit excellent surface tunability and mechanical flexibility. Despite their promise, issues such as self-restacking and limited conductivity often restrict their independent performance. Mixed-dimensional integration (e.g., 0D/2D or 1D/2D hybrids) has been shown to enhance ion diffusion and active site accessibility. In particular, 2D/2D face-to-face heterostructures unconstrained by lattice matching enable maximized interfacial interactions and tunable electronic properties. For example, Ti0.87O2, a lepidocrocite-type oxide with abundant Ti vacancies, offered strong Li+ affinity but suffered from low conductivity. When coupled with conductive MXene layers via van der Waals and hydrogen bonding, the resulting 2D/2D Ti0.87O2/MXene heterostructure demonstrates an electronic compensation mechanism that enhanced catalytic activity and lowered ORR/OER overpotentials. This hybrid system delivered a high capacity of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 596 mA h g−1 and maintained stable operation for over 425 cycles at 200 mA g−1.216 Quasi-homogeneous catalysts have also shown potential in overcoming the limitations of immobile solid-state catalysts. Ru-loaded amino-phenanthroline-derived carbonized polymer dots (RuApCPDs), dispersed within the electrolyte, offer both catalytic function and mobility. These dynamic catalysts co-deposit with Li2O2, stabilize intermediates, and reduce charge voltage from 4.46 to 3.75 V, while modifying the Li+ solvation structure and promoting stable SEI formation. Multifunctionality enables long cycling stability up to 168 cycles, outperforming traditional SSC-based systems.217
596 mA h g−1 and maintained stable operation for over 425 cycles at 200 mA g−1.216 Quasi-homogeneous catalysts have also shown potential in overcoming the limitations of immobile solid-state catalysts. Ru-loaded amino-phenanthroline-derived carbonized polymer dots (RuApCPDs), dispersed within the electrolyte, offer both catalytic function and mobility. These dynamic catalysts co-deposit with Li2O2, stabilize intermediates, and reduce charge voltage from 4.46 to 3.75 V, while modifying the Li+ solvation structure and promoting stable SEI formation. Multifunctionality enables long cycling stability up to 168 cycles, outperforming traditional SSC-based systems.217
Single-atom catalysts (SACs) represent a frontier in catalyst design due to their maximum atomic efficiency and precise coordination environment. Anchored onto conductive supports, the SACs such as Pt, Ru, and Pd have demonstrated excellent activity and durability towards the ORR and OER. For instance, Pt atoms anchored on porous g-C3N4 nanosheets (Pt-CNHS) and Ru single atoms on nitrogen-doped carbon (Ru SAs-NC) systems offer improved electron transport and minimal LiO2 binding energy, resulting in low overpotentials. The Pd SACs with N4 coordination structure achieved ultra-low charge voltages (0.24 V), attributed to d-band center shifts and optimal charge redistribution.218–220 Single-atom alloy catalysts, like Pt1Pd nanoplates, further enhance activity through strong Pt–Pd interactions that promote localized electron transfer and suppress overpotential. Structural and spectroscopic analyses confirmed atomic dispersion and coordination, while electrochemical testing demonstrated cycling stability beyond 150 cycles.221 Despite substantial progress, deeper mechanistic insights into SAC function remain limited. In situ techniques capable of capturing intermediate species such as O2−, LiO2, and Li2O2 are crucial for elucidating reaction pathways and guiding future catalyst design. Advanced strategies including coupling high-entropy alloys (HEAs) with SACs have enabled the design of dual-active-site platforms capable of regulating both surface-adsorbed and solution-mediated LiO2 intermediates. Notably, Pt SACs embedded within HEA@Pt nanoparticles exhibited a high reversible capacity of 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 116 mA h g−1 with minimal polarization and prolonged lifespan.222 To further enhance catalytic efficacy, approaches such as d-band center tuning, heteroatom doping, and hybridization with multivalent compounds or high-index facets have been employed to optimize the electronic structure and reaction kinetics. Magnetically active catalysts add another layer of complexity by leveraging spin polarization effects to enhance electron transfer. Ferromagnetic Co-based catalysts with tailored d-orbital configurations, such as Co-r-RCSs, exhibited strong Co 3d–O 2p coupling and significant spin-flip effects, enabling controlled LiO2 nucleation and solution-mediated Li2O2 growth. This approach resulted in an impressive discharge capacity of 18
116 mA h g−1 with minimal polarization and prolonged lifespan.222 To further enhance catalytic efficacy, approaches such as d-band center tuning, heteroatom doping, and hybridization with multivalent compounds or high-index facets have been employed to optimize the electronic structure and reaction kinetics. Magnetically active catalysts add another layer of complexity by leveraging spin polarization effects to enhance electron transfer. Ferromagnetic Co-based catalysts with tailored d-orbital configurations, such as Co-r-RCSs, exhibited strong Co 3d–O 2p coupling and significant spin-flip effects, enabling controlled LiO2 nucleation and solution-mediated Li2O2 growth. This approach resulted in an impressive discharge capacity of 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 429.6 mA h g−1 and prolonged cycling over 240 cycles with low overpotentials.223 Photo electrocatalysis has emerged as a novel strategy to further enhance the round-trip efficiency. Photocathode materials such as BiVO4, g-C3N4, and TiO2 utilize light-induced charge carriers to facilitate Li2O2 decomposition, offering a sustainable approach to reduce charge voltages and accelerate the reaction kinetics.224 In recent years, our research group has systematically developed a range of bifunctional electrocatalysts aimed at addressing the kinetic limitations of oxygen reactions in Li–O2 and Li–CO2 batteries.182,225–228 Complementary work on NiCo-LDH nanosheets dispersed on rGO also yielded notable electrocatalytic behavior, achieving high onset potentials and limiting current density for the ORR and very low Tafel slope for the OER, as shown in Fig. 7(a and d).229 Additionally, a selenium-doped porous carbon synthesized from waste-derived feedstock showed competent ORR activity and enabled stable Li–O2 cell performance, aligning with sustainable material goals.230 Perovskite-based LaMnO3 was successfully prepared as a catalyst to accelerate the ORR and facilitate the 4e− pathway with less than 1% or no HO2− formation, as evidenced from the K–L plot and H2O2 quantification plot, as shown in Fig. 7(b and c).231 In a separate study, a Cu@CuFe2O4 catalyst displayed tolerance to CO2-rich atmospheres and facilitated the decomposition of both Li2O2 and Li2CO3, supporting its function in Li–CO2 cells, and exhibited excellent rate capability for the Li–O2 battery, as shown in Fig. 7(e).182 A hierarchical Ni/NiFe2O4@C composite featuring a cauliflower-like architecture was engineered, which exhibited favorable oxygen reduction characteristics through a 4e− pathway and sustained low H2O2 yield. When integrated into a Li–O2 battery, the system delivered a discharge capacity of 3820 mA h g−1.232 Advancing this work, a Ni/NiO-anchored nitrogen-doped carbon material was synthesized, demonstrating an onset potential of 0.96 V and an electron transfer number close to four, as verified by the RRDE measurements. This catalyst offered impressive reversibility and long-term stability, enabling a Li–Air cell to deliver 3330 mA h g−1 at 100 mA g−1 current density and to retain capacity over 150 cycles, as shown in Fig. 7(f).233 Collectively, these efforts underscore a strategic balance between catalytic performance insights via RRDE and the validation of practical energy storage through full-cell demonstration. The instability of the electrolyte in the presence of the superoxide intermediates generated during discharge renders certain catalysts ineffective.
429.6 mA h g−1 and prolonged cycling over 240 cycles with low overpotentials.223 Photo electrocatalysis has emerged as a novel strategy to further enhance the round-trip efficiency. Photocathode materials such as BiVO4, g-C3N4, and TiO2 utilize light-induced charge carriers to facilitate Li2O2 decomposition, offering a sustainable approach to reduce charge voltages and accelerate the reaction kinetics.224 In recent years, our research group has systematically developed a range of bifunctional electrocatalysts aimed at addressing the kinetic limitations of oxygen reactions in Li–O2 and Li–CO2 batteries.182,225–228 Complementary work on NiCo-LDH nanosheets dispersed on rGO also yielded notable electrocatalytic behavior, achieving high onset potentials and limiting current density for the ORR and very low Tafel slope for the OER, as shown in Fig. 7(a and d).229 Additionally, a selenium-doped porous carbon synthesized from waste-derived feedstock showed competent ORR activity and enabled stable Li–O2 cell performance, aligning with sustainable material goals.230 Perovskite-based LaMnO3 was successfully prepared as a catalyst to accelerate the ORR and facilitate the 4e− pathway with less than 1% or no HO2− formation, as evidenced from the K–L plot and H2O2 quantification plot, as shown in Fig. 7(b and c).231 In a separate study, a Cu@CuFe2O4 catalyst displayed tolerance to CO2-rich atmospheres and facilitated the decomposition of both Li2O2 and Li2CO3, supporting its function in Li–CO2 cells, and exhibited excellent rate capability for the Li–O2 battery, as shown in Fig. 7(e).182 A hierarchical Ni/NiFe2O4@C composite featuring a cauliflower-like architecture was engineered, which exhibited favorable oxygen reduction characteristics through a 4e− pathway and sustained low H2O2 yield. When integrated into a Li–O2 battery, the system delivered a discharge capacity of 3820 mA h g−1.232 Advancing this work, a Ni/NiO-anchored nitrogen-doped carbon material was synthesized, demonstrating an onset potential of 0.96 V and an electron transfer number close to four, as verified by the RRDE measurements. This catalyst offered impressive reversibility and long-term stability, enabling a Li–Air cell to deliver 3330 mA h g−1 at 100 mA g−1 current density and to retain capacity over 150 cycles, as shown in Fig. 7(f).233 Collectively, these efforts underscore a strategic balance between catalytic performance insights via RRDE and the validation of practical energy storage through full-cell demonstration. The instability of the electrolyte in the presence of the superoxide intermediates generated during discharge renders certain catalysts ineffective.
 | ||
| Fig. 7 (a and d) ORR LSV comparison curve at a rotation speed of 1600 rpm in RRDE, and the OER Tafel plots for various electrocatalysts. Reproduced from ref. 229 with permission from Elsevier, copyright [2025]. (b) K–L plots. (c) Calculated number of electrons transferred, and the peroxide formed for the ORR. Reproduced from ref. 231 with permission from Wiley-VCH, copyright [2022]. (e) GCD curves at different current densities of the Li–O2 battery with the Cu@CuFe2O4 air-cathode. Reproduced from ref. 182 with permission from ECS, copyright [2024]. (f) Cycle-life data and coulombic efficiency recorded for the Li–Air battery split cell using NNONC as the air-cathode and an Li disc as the anode. Reproduced from ref. 233 with permission from ECS, copyright [2023]. | ||
While catalysts are intended to enhance the oxygen reduction and evolution reactions, their catalytic activity can inadvertently accelerate the decomposition of the electrolyte as well, exacerbating side reactions and leading to the formation of undesired byproducts. This dual role undermines the long-term stability and efficiency of the battery, highlighting the need for catalysts that are not only highly active but also chemically compatible with the electrolyte to prevent such detrimental interactions. Additionally, it involves exploring electrolyte additives that can stabilize reactive intermediates and lower the charge overpotential.
6.4 Oxygen-selective membranes
Unlike conventional LIBs, the Li–Air batteries operate as semi-open systems, introducing a unique set of challenges, particularly for organic electrolytes and lithium metal anode. To overcome the limitations posed by the ambient air exposure, early strategies enclosed LABs in sealed chambers supplied with pure oxygen. Although this ensured a dry and uncontaminated environment, it significantly reduced energy density, increased system cost and complexity, and compromised the lightweight, flexible design ethos of the LABs. Therefore, there is a critical need for more practical and efficient solutions that preserve the compactness, portability, and full performance potential of the LAB systems.234,235 Thus, beyond direct stabilization strategies such as surface protection of the lithium metal and development of chemically stable electrolytes, structural innovations in cell design are gaining attention. One such approach involves the integration of oxygen-selective membranes/oxygen permeable membranes (OSMs/OPMs) on the cathode exterior. These membranes facilitate the selective passage of O2 while restricting other atmospheric gases, thereby minimizing side reactions that degrade both the cathode and lithium anode by exposure to the moisture or CO2. However, the design of membranes that can exclusively allow O2 transport is nontrivial. The difficulty stems from the fact that gases like H2O (kinetic diameter ∼0.289 nm) and CO2 (∼0.330 nm) are smaller than O2 (∼0.346 nm), making size-based separation highly challenging via Knudsen diffusion. To operate effectively in ambient air, OPMs must exhibit high oxygen permeability, excellent selectivity, water and electrolyte resistance, and long-term mechanical stability. High oxygen flux ensures adequate supply for cathodic reactions, enhancing discharge capacity and rate performance. Meanwhile, water resistance is essential to prevent anode corrosion and electrolyte degradation, as even trace moisture can destabilize the SEI layer. Selectivity toward O2 over similar-sized molecules like H2O and CO2 is achieved through tailored membrane structures using dense polymers or modified porous frameworks. Electrolyte-proof membranes must resist chemical degradation while maintaining oxygen transport, and robust mechanical properties are required to ensure membrane integrity under stress. Integrated design strategies such as using hydrophobic coatings, composite layers, and thermal stabilizers are essential to balance these demands. Overall, OPMs enable ambient–air operation and represent a critical component for the practical realization of next-generation LABs.236–240 The oxygen-selective membranes (OSMs) are generally classified as outer or inner types based on their placement within the battery. Outer-type OSMs are located outside the air cathode, enabling oxygen intake while preventing contaminant ingress. In contrast, inner-type OSMs are positioned between the separator and the air electrode, requiring mechanically stable support to endure chemical exposure and operational stress within the battery. Outer-type OPMs, often composed of hydrophobic polymers such as PDMS, fluorinated polymers (e.g., PTFE, PVDF-HFP, PFPE), and Melinex films, offer robust barrier properties, chemical stability, and oxygen permeability. Advances in material engineering, such as blending with perfluorinated compounds or coating with hydrophobic layers, have significantly improved water resistance and oxygen selectivity, although trade-offs between permeability and selectivity remain a challenge. MOF-based and mixed matrix membranes (MMMs) have also gained attention for their tunable porosity and functionalizability, providing pathways for enhanced oxygen flux and impurity rejection. For inner-type OPMs, substrates like Ni foam, PTFE, electrospun fibers, and carbon frameworks support membrane integrity, improve mechanical strength, and facilitate ion and oxygen transport. These designs help prevent lithium corrosion, reduce electrolyte volatility, and suppress parasitic reactions.As a result, only a few studies have successfully reported functional OSMs for Li–Air batteries. For example, in 2010, Zhang et al.241,242 introduced the use of Melinex membranes for encapsulating pouch-type LABs, marking a key advancement in applying OPMs under ambient conditions. That same year, the development of porous PTFE enabled the stabilization of oxygen-selective liquid membranes. These breakthroughs paved the way for the sequential emergence of diverse OPM designs, significantly advancing the practical realization of LABs operable in Earth's atmosphere. Ruan et al.243 introduced a perfluorocarbon silicone oil-based OSM applied to the exterior of the cathode casing, which significantly improved the battery's electrochemical stability under ambient air conditions. Similarly, Amici et al.244 engineered a composite membrane using PVDF-HFP supported by silicone oil. The synergy between the hydrophobic polymer matrix and oxygen-selective silicone oil allowed Li–O2 to deliver comparable performance in ambient air and pure oxygen atmospheres. Building on these concepts, Huang et al.245 developed a perfluoropolyether (PFPE)-based OSM that exhibited excellent oxygen permeability and water resistance. The membrane effectively reduced organic electrolyte evaporation, stabilized Li2O2 and the lithium anode, and enabled stable cycling for up to 144 cycles at ∼30% relative humidity. Xu et al.246 have recently developed a flexible, superhydrophobic metal–organic framework (MOF) nanosheet membrane with a porous honeycomb-like structure on a polymer substrate. The coating method produced defect-free films capable of withstanding mechanical deformations such as flipping, twisting, and swaying without nanosheet detachment, owing to the strong anchoring of MOFs within the substrate. Despite the inherent rigidity of MOFs and (mixed matrix membranes) MMMs due to their high crystallinity, the resulting membrane exhibited excellent flexibility and a high-water contact angle of 158.3°, significantly surpassing that of conventional PDMS/PVDF membranes. To further enhance the membrane properties, Shao et al.247 embedded silica nanoparticles into a polydimethylsiloxane (PDMS) matrix, creating a cross-linked and flexible OSM. This membrane exhibited high oxygen permeability, strong hydrophobicity, and excellent thermal stability. As a result, both coin-type and pouch-type Li–Air battery showed robust long-term cycling performance, even under severe environmental conditions, with lifetimes reaching up to 700 hours. Despite these promising advances, the overall effectiveness of Li protection strategies remains limited. For instance, although lithium alloy anodes have been explored, their high susceptibility to CO2 and H2O corrosion restricts their practical use. Inspired by natural vortex dynamics, Kim et al.248 employed electrospinning to create carbon nanofiber (CNF) gas diffusion layers (GDLs) combined with high-density carbon paper (HDCP) patterned via microscale mold imprinting. This structural design enhanced internal vortex flow, boosting the oxygen velocity and improving the active site exposure for oxygen reduction. The fully patterned cathode (P-CNF/CP) achieved a higher reduction potential of 2.697 V, outperforming less-structured variants. Additionally, imprinting pressure influenced performance; 10 MPa yielded optimal surface roughness and density, though excessive compaction reduced oxygen permeability, highlighting the need for balanced structural tuning. Similarly, while all-solid-state electrolytes are considered as a barrier against air-induced degradation, their high interfacial resistance and low ionic conductivity hinder their applicability in Li–Air batteries. Gel polymer electrolytes, which offer flexibility, moisture resistance, and electrochemical stability, have extended battery life but often suffer from large polarization due to limited ionic mobility. These strategies, though partially effective, struggle to provide comprehensive protection for the lithium anode, especially in ambient environments. OSMs offer an additional layer of defense by limiting the ingress of moisture and corrosive gases. However, their performance is still hindered by insufficient selectivity for oxygen over other small molecules. Therefore, further optimization of OSM design remains critical. Realizing stable and high-performance Li–Air battery will likely depend on the coordinated application of multiple strategies including lithium surface protection, advanced electrolytes, and oxygen-selective interfaces. Toward this goal, separator engineering has also emerged as a promising avenue. This impacted the development of tissue-based bifunctional separator (TBF) capable of acting both as a physical barrier and as a protective interface for the lithium anode. This dense, stable membrane offers strong resistance against moisture, molecular oxygen, and superoxide radicals (O2−), effectively shielding the anode. As a result, the Li–O2 cells incorporating the TBF separator achieved over 300 stable cycles at 200 mA g−1 with a fixed capacity of 500 mA h g−1.249 However, balancing high oxygen flux with efficient blocking of H2O and CO2 remains a critical bottleneck. While hydrophobicity is essential for moisture rejection, it does not directly translate to superior gas selectivity, especially when dealing with similarly sized molecules like O2 and H2O. Moreover, current membranes often fail to block CO2, which forms electrochemically inert Li2CO3, leading to cathode passivation. Innovations in the membrane architecture, surface functionalization, and multi-layered composites are being explored to address these issues. Additionally, compatibility with battery components, resistance to electrolyte corrosion, and mechanical resilience under operational stresses are vital for practical applications. Although challenges persist, ongoing research studies on OPM materials, structures, and fabrication methods continue to drive progress toward commercially viable LAB systems capable of stable long-term operation in real-world environments. Continued research into synergistic strategies, particularly those integrating functional OSMs, stable separators, and electrolyte innovations, is essential to unlock the full potential of Li–O2 batteries for practical applications.
6.5 Redox mediators
A major bottleneck originates from the formation and decomposition of Li2O2, the primary discharge product in the Li–O2 battery, which is highly insulating and hampers efficient charge transfer across the electrode–electrolyte interface. To address these challenges, RMs have been extensively investigated as either soluble or immobilized charge carriers.250 These mediators facilitate both the ORR during discharge and the OER during charging, thereby reducing energy losses, improving reaction kinetics, and enhancing battery performances.251 Organic, inorganic, and organometallic RMs are typically introduced into the electrolyte to enhance the battery's redox processes.252 Meanwhile, ionic RMs facilitate electron transfer, improving the decomposition efficiency of poorly conductive Li2O2 or aiding its controlled deposition in solution.6.6 Role of ORR RMs
During discharge, oxygen undergoes a stepwise reduction to form Li2O2:| O2 + 2e− + 2Li+ → Li2O2 | (38) |
However, the direct electrochemical reduction of oxygen on the cathode surface is kinetically sluggish and often leads to non-uniform Li2O2 deposition, which blocks active sites and increases cell polarization.
The RM is electrochemically reduced at the cathode:
| RMox + e− → RMred |
The reduced mediator chemically reduces dissolved O2:
| RMred + O2 + 2Li+ → Li2O2 + RMox |
This solution-mediated pathway shifts the ORR from a heterogeneous to a homogeneous mechanism, facilitating uniform Li2O2 deposition with higher crystallinity and reducing electrode passivation. Notably, discharge RMs such as DBBQ form soluble LiDBBQO2 intermediates, promoting solution-phase growth and enhancing discharge capacity.253
6.7 Role of OER RMs
Charging requires the oxidation of Li2O2 back into lithium ions and oxygen:| Li2O2 → O2 + 2e− + 2Li+ | (39) |
This reaction suffers from high overpotentials due to the insulating nature of Li2O2, leading to severe electrolyte decomposition and cathode degradation.
The RM is electrochemically oxidized at the cathode:
| RMred → RMox + e− |
The oxidized mediator then chemically decomposes Li2O2:
| Li2O2 + RMox → O2 + RMred + 2Li+ |
This redox shuttle significantly lowers the charging voltage and prevents direct contact between Li2O2 and the carbon cathode, thereby improving reversibility and suppressing parasitic reactions. Common OER mediators include lithium iodide (LiI), lithium bromide (LiBr), tetrathiafulvalene (TTF), 2,2,6,6-tetramethylpiperidin-1-oxyl (TEMPO), and tris[4-(diethylamino)phenyl]amine (TDPA).254–259
The performance of the RMs in the Li–O2 batteries is intricately influenced by their interactions with other electrolyte components. Strategic pairing of lithium salts, solvents, and RMs plays a crucial role in modulating the ORR/OER kinetics and overall battery efficiency. Lithium salts with high DNs (e.g., LiNO3 and LiTFSI) enhance LiO2 solubility, thus promoting solution-phase Li2O2 formation. This improved solubility facilitates homogeneous ORR pathways, lowers overpotentials, and augments energy efficiency. Solvents with high DNs, such as dimethyl sulfoxide (DMSO), enable effective solvation of O2− and LiO2 intermediates. While such solvents support enhanced ORR activity and improved discharge homogeneity, excessive DNs may trigger solvent degradation and side reactions.260,261 Thus, an optimized balance must be achieved. Experimental observations and computational studies (e.g., density functional theory) underscore that interactions among RMs, lithium salts, and solvents are critical in modulating the ORR kinetics and mediating Li2O2 formation. Tailoring these interactions is essential for utilizing RM for achieving stable and high-efficiency Li–O2 cells. The RMs function by decoupling the electrochemical oxidation of discharge products (e.g., Li2O2) from the cathode surface, thereby reducing the kinetic barriers associated with direct electron transfer. During charging, RMs are oxidized at the cathode and subsequently act as soluble oxidizing agents that chemically decompose Li2O2 in the bulk electrolyte. This indirect oxidation mechanism significantly lowers the required charge potential, often from values >4.2 V to ∼3.7 V, as demonstrated with mediators like TEMPO and LiI.262,263 Beyond voltage reduction, the RMs prevent direct Li2O2 carbon interaction, thereby mitigating side reactions such as carbon corrosion and Li2CO3 formation. Moreover, the RMs modulate the discharge product morphology by promoting solution-mediated Li2O2 crystallization rather than surface passivation. This dual functionality in reducing Li2O2 surface coverage and facilitating its chemical decomposition effectively fixes the charging potential near the redox potential of the RM and enhances the reversibility of the electrochemical processes. Transition metal-based RMs such as iron phthalocyanine (FePc) and cobalt porphyrins further improve the redox kinetics and demonstrate higher catalytic robustness under cycling conditions.264 To mitigate the shuttle effect, polymer-bound mediators such as poly(2,2,6,6-tetramethyl-1-piperidinyloxy-4-yl methacrylate) (PTMA) have been developed. These immobilized mediators enable charge transfer through electron hopping mechanisms along polymer chains, enhancing RM retention and long-term stability.265 ROS, particularly singlet oxygen, play a critical role in side reactions that degrade carbon cathodes and electrolytes. RMs such as DABCO, 4-oxo-TEMP, and 9,10-dimethylanthracene (DMA) have been utilized to quench singlet oxygen, improving the overall electrochemical stability of the Li–O2 battery. Lithium carbonate (Li2CO3), formed through electrolyte decomposition and carbon oxidation, presents a major hurdle due to its high decomposition potential (>4 V vs. Li/Li+). The RMs capable of oxidizing Li2CO3, such as 2,5-di-tert-butyl-1,4-dimethoxybenzene (DBDMB), have shown promise in enhancing long-term battery performance. These mediators accelerate the decomposition of not only Li2CO3 but also Li2O2 and lithium hydroxide (LiOH), extending the cycle life.266 Recent advances demonstrate that halide-based RMs (e.g., Cl−/Cl3−) can shift discharge chemistry from Li2O2 to LiOH formation. Given LiOH's superior ionic conductivity, this transformation significantly reduces charge overpotential and improves cycling stability. Xu et al.267 employed a dual-mediator configuration using DBBQ for the discharging and TEMPO for the charging of the battery. This strategy effectively minimized direct Li2O2 carbon interactions, suppressing Li2CO3 formation and enhancing battery durability. Despite their pivotal role in improving the Li–O2 battery performances, the application of soluble RMs presents significant challenges, particularly related to stability, selectivity, and interfacial compatibility. One of the most critical issues associated with the freely mobile RMs is the shuttle effect, wherein the oxidized form of the mediator (RM+) diffuses from the cathode to the lithium metal anode. This migration can lead to uncontrolled redox reactions at the anode interface, resulting in the degradation of the SEI, loss of active lithium and mediator species, and the formation of parasitic byproducts. Such side reactions compromise coulombic efficiency and accelerate capacity fading over repeated cycles. For instance, oxidized mediators such as TEMPO+ or triiodide (I3−) can undergo spontaneous chemical reduction at the lithium metal surface, leading to mediator decomposition and irreversible lithium consumption. In addition to this shuttle-induced reactivity, many RMs suffer from limited chemical stability, particularly under oxidative potentials or in the presence of ROS like 1O2. Halogen-based mediators such as LiI and LiBr, while effective in reducing overpotential, can also exhibit corrosive behavior, posing compatibility issues with separators, current collectors, and electrolyte components.
7. Threefold strategy to overcome Li–O2 battery drawbacks using electrolyte additives
Electrolyte additives have emerged as a transformative solution to the intrinsic challenges of Li–O2 batteries. By targeting the anode, cathode, and electrolyte, these additives significantly enhance the system stability, mitigate degradation pathways, and optimize the electrochemical performance. This section examines the precise roles and mechanisms of electrolyte additives in addressing key limitations. Fig. 8 illustrates the schematics of a threefold protection strategy using an electrolyte additive. Electrolyte additives address the challenges through the additives that enable the in situ formation of a chemically stable SEI that prevents direct contact between the lithium metal and the electrolyte, mitigating lithium corrosion.Additives suppress side reactions initiated by reactive oxygen intermediates, improving anode lifespan and cycling efficiency. At the cathode, electrolyte additives are instrumental in modulating oxygen electrochemistry, enhancing reaction kinetics, and controlling discharge product formation. Their functions include facilitating smooth ORR and OER processes, reducing the overpotentials and improving the energy efficiency. Enhanced oxygen solubility and adsorption in the electrolyte improve reaction uniformity at the cathode surface. Additives regulate the growth of discharge products (Li2O2), favouring nanosheet-like or toroidal structures that are more electrochemically accessible and easier to decompose during charging. Additives enhance the effectiveness of cathode catalysts, creating a more active and stable reaction environment. Additives mitigate decomposition and volatilization issues through the following mechanisms. Additives reduce the impact of ROS, preventing the breakdown of the electrolyte and extending its lifespan.
• By forming a stabilizing matrix, additives buffer against electrolyte volatilization caused by oxygen attack.
• Additives scavenge reactive oxygen intermediates, reducing parasitic reactions that degrade the electrolyte.
• Additives facilitate the dissolution of oxygen and promote the formation of solution-phase discharge products (Li2O2), enhancing reversibility and cycling performance.
7.1 Integration of electrolyte additive–electrode interactions
The combined effects of electrolyte additives on the anode, cathode, and electrolyte create a synergistic improvement in battery performance. Some key integrated impacts are as follows: (i) suppression of dendrite growth and parasitic reactions reduces the risk of short circuits and thermal runaway, (ii) lower overpotentials for ORR and OER processes minimize energy losses, (iii) stabilized interfaces and reduced degradation pathways extend the operational lifespan of the battery, and (iv) the ability to regulate discharge product morphology and dissolution ensures consistent and reliable cycling. This threefold strategy highlights the critical role of electrolyte additives in advancing Li–O2 battery technology. By addressing core challenges with targeted mechanisms, these additives pave the way for the practical deployment of Li–O2 batteries in high-energy applications.7.2 Functions of electrolyte additives in Li–O2 batteries
Electrolyte additives play a pivotal role in overcoming the inherent limitations of Li–O2 batteries by modifying their chemical, electrochemical, and physical properties. These additives are typically organic compounds or inorganic salts that are integrated into various components of the battery system, including the electrolyte, cathode, and lithium metal surface, to enhance the performance, stability, and longevity. While some additives are redox-active (e.g., RMs), others primarily function through SEI formation (react once during initial cycling to form a passivation layer), solubility control, catalysis, or ionic enhancement without undergoing redox reactions. Their primary functions include facilitating Li-ion transport, stabilizing the SEI, regulating the deposition, dissolution, and decomposition of Li2O2, promoting the solution-mediated growth of toroidal Li2O2, and improving the kinetics of the ORR and OER. The classification of these additives is based on their primary role in addressing specific challenges. Overpotential-reducing additives focus on lowering the high charge and discharge potentials by acting as RMs, thereby enhancing the energy efficiency. The Li2O2 dissolution-promoting additives facilitate the decomposition of the discharge product, preventing cathode passivation and improving rechargeability. Discharge capacity-enhancing additives stabilize the electrode structure, regulate Li2O2 growth morphology, and promote efficient ORR kinetics, leading to higher capacity retention. These additives include functional SEI stabilizers, catalytic and electrochemical performance enhancers, mechanical and structural reinforcements, and electron transport facilitators. The continuous refinement of these categories has led to consistent improvements in battery performance by addressing the shortcomings of the earlier designs. The SEI layer is a crucial component of Li–O2 batteries, acting as a protective barrier that minimizes undesired reactions between lithium metal and the electrolyte while ensuring efficient Li-ion transport. Functional additives such as fluorides, boron compounds, nitrides, and phosphorus-based materials contribute to the formation of a chemically and mechanically stable SEI. During cycling, these additives decompose to form passivation layers rich in fluorine or boron, which enhance the interfacial stability and suppress dendrite formation. Additionally, these materials prevent the diffusion of ROS toward the lithium metal anode, thereby reducing side reactions that lead to capacity fading. A well-regulated SEI not only extends battery lifespan but also improves the cycling efficiency by maintaining a uniform lithium deposition process. Efficient ORR and OER kinetics are fundamental to the operation of Li–O2 batteries, as high overpotentials during these reactions lead to energy losses and performance degradation. Catalytic and electrochemical performance-enhancing additives such as iodides and sulfur-based compounds act as RMs that facilitate charge transfer, thereby accelerating the decomposition of Li2O2 during charging. These additives also aid in the dissolution of Li2O2, ensuring that its accumulation does not lead to pore blockage on the cathode. Additionally, salt additives enhance ionic conductivity, while organic additives improve the solubility and mobility of lithium ions, collectively optimizing the redox reactions and reducing parasitic electrolyte degradation. The mechanical and structural integrity of the battery system is another critical factor in determining its long-term performance. Inorganic nano-fillers such as alumina (Al2O3), silica (SiO2), and titanium dioxide (TiO2) are introduced into the electrolyte or electrode matrix to reinforce mechanical stability and prevent dendrite penetration. These materials also provide thermal resistance, reducing degradation at elevated temperatures. Polymeric additives like polyethylene oxide (PEO) and poly(vinylidene fluoride) (PVDF) enhance electrolyte flexibility and durability, ensuring better compatibility with lithium metal anodes. These reinforcements help maintain battery performance under high-current cycling conditions, thereby improving overall resilience. Lithium deposition irregularities, as well as the insulating nature of Li2O2, pose significant challenges in terms of electronic conductivity and battery efficiency. To address this, conductivity-enhancing additives such as carbonaceous materials including graphene, CNTs, and activated carbon are incorporated into the battery system. These materials create highly conductive networks that facilitate electron transfer and oxygen diffusion, leading to improved ORR and OER kinetics. For Li–O2 batteries to achieve high power density and extended cycle life, it is crucial to minimize interfacial resistance and enhance ionic conductivity. This becomes particularly important under high current densities, where poor ion transport and polarization effects can lead to rapid performance degradation. The continuous development of functional electrolyte additives has significantly improved Li–O2 battery efficiency by addressing key challenges at the molecular and interfacial levels. Fig. 9 shows the schematics of electrolyte additives and their functions for a non-aqueous Li–O2 battery. Through the strategic integration of SEI stabilizers, RMs, structural reinforcements, ionic/electronic conductors, solubility modulation, and catalytic enhancement, these additives collectively optimize the battery performance, paving the way for the practical realization of long-cycle high-energy-density Li–O2 batteries. More than sixty electrolyte additives have been investigated for non-aqueous Li–O2 batteries, each influencing multiple interfacial reactions. Since the function of a specific additive often varies with electrolyte composition and electrode environment, the discussion here is organized chronologically and by additive class, which reflects their historical development and compositional diversity.Within each subsection, the dominant functional roles including SEI stabilization, RM regulation, Li2O2/O2 solubility modulation, and anode/cathode protection are emphasized to highlight mechanistic trends and their influence on overall cell reversibility.
However, NO2 gas tends to evaporate in open-cell structures, preventing the sustained operation of the NO2−/NO2 redox cycle. Interestingly, Uddin et al.270 suggest that LiNO2 can spontaneously react with O2, regenerating LiNO3. This conversion from NO2− to NO3− occurs at a significantly faster rate than NO2 vaporization, enabling its reuse in subsequent cycles. Moreover, solid-state NMR evidence shows that LiNO3 directly stabilizes the interface through its adsorption onto the electrode. He first proposed a regenerative LiNO3 mechanism in Li–O2 batteries, demonstrating that LiNO3 forms soluble nitrate anions that react with lithium metal to form lithium nitrite (LiNO2), which further regenerates LiNO3 upon exposure to oxygen. This self-sustaining cycle prevents the continuous depletion of LiNO3, ensuring long-term electrolyte stability while stabilizing the lithium anode by suppressing side reactions and dendrite formation via forming a passivating Li2O layer on the surface. Their findings highlighted that the introduction of LiNO3 significantly improved the electrochemical performance of Li–O2 batteries by mitigating electrolyte degradation and enabling extended cycling performance without requiring continuous additive replenishment. They proposed a well-defined mechanism for this regeneration, asserting that it does not interfere with Li2O2 oxidation or overall cell efficiency. Further research has highlighted that nitrite ions generated during the electro-oxidation of nitrate can catalyze Li2O2 oxidation via nitric oxide-mediated pathways, effectively reducing the charge potential. Despite these advantages, LiNO3 remains effective only when in direct contact with lithium metal. This requirement poses a major limitation, as lithium metal is often isolated from the electrolyte to mitigate dendrite formation. Nearly half a decade later, Rosy et al.271 expanded on these findings by investigating the bifunctional role of the LiNO3 as both an anode stabilizer and a cathode passivation agent. Their study revealed that the LiNO3 contributes to the formation of an SEI on the lithium anode, which prevents unwanted side reactions and suppresses dendrite growth. Additionally, they demonstrated that LiNO3 plays a crucial role in passivating the cathode surface, significantly reducing carbon degradation, which is a major cause of capacity fading in the Li–O2 batteries. By employing in operando online electrochemical mass spectrometry (OEMS), they were able to deconvolute the surface stabilization and catalytic functions of LiNO3, showing that it effectively enhances both the ORR and OER kinetics, thereby lowering charge overpotential. However, their findings indicated that higher LiNO3 concentrations were necessary to achieve complete cathode passivation and maximize the battery performance. LiNO3 has long been recognized as a crucial electrolyte additive in Li–S batteries due to its ability to stabilize the lithium metal anode. This stabilization occurs through the formation of surface species such as Li2O, Li3N, and LiNxOyvia chemical and electrochemical interactions with lithium metal. Among these, insoluble LiNxOy plays a vital role in passivating the Li surface, preventing direct electron transfer from the metal to the electrolyte, thereby minimizing undesirable side reactions between lithium and polysulfides. In the Li–O2 batteries, several studies have also demonstrated that incorporating LiNO3 into the electrolyte facilitates the formation of a stable, protective SEI on the Li anode. However, it has been observed that NO2− species, a key component of this SEI, can dissolve into the electrolyte and undergo reactions with oxygen, leading to the regeneration of NO3− through a complex reaction pathway. This continuous regeneration process gradually disrupts the SEI, exposing fresh lithium to the electrolyte and resulting in persistent interactions between lithium metal and the oxygen-rich LiNO3-containing electrolyte. Additionally, intermediate nitrogen species such as NO2, N2O4, NO, and NO+, formed during NO2− to NO3− conversion, may trigger undesirable side reactions, compromising the cycling stability of conventional LiNO3-based Li–O2 batteries. The presence of oxygen significantly intensifies NO2− dissolution, further deteriorating the SEI structure. To mitigate this issue, an effective strategy involves designing an SEI with an oxygen-blocking multifunctional layer that encapsulates soluble NO2− species within its structure. The approach by Lin et al.272 ensures the integrity and stability of the LiNO3-derived SEI, suppressing unwanted side reactions, lithium dendrite growth, and corrosion, thereby enhancing both the reversibility and the cycling stability of the system. Lin et al. have demonstrated that an electrochemical polishing technique can be employed to fabricate a molecularly smooth SEI with a layered architecture on alkali metal surfaces, including lithium. By carefully controlling the electrochemical polishing process and electrolyte composition, the SEI's multi-layered structure can be tailored to exhibit both rigidity and elasticity, effectively inhibiting dendrite formation and significantly extending cycle life. Inspired by these advantages, we propose the use of electrochemical polishing to develop a LiNO3-derived multilayered SEI is proposed. In this design, the soluble NO2− species are confined within the inner layers, while an insoluble outer layer acts as a protective barrier against oxygen. The schematic illustration and the performance of the stripping plating experiment for the pristine and polished anodes are shown in Fig. 10(a and b). This innovative SEI architecture is expected to enhance the long-term stability and performance of Li–O2 batteries. When paired with a Ru/rGO cathode, the cell sustained stable operation for 415 cycles, more than double the cycle life observed with a pristine lithium anode (192 cycles). Notably, even with rGO alone as the cathode, the cell incorporating the polished Li anode maintained stability for 375 cycles, which is over tenfold greater than that of the corresponding cell using untreated lithium (36 cycles), as shown in Fig. 10(c). Together, these studies demonstrate the continuous evolution of LiNO3 as a multifunctional additive in Li–O2 batteries. From its initial discovery as a regenerative stabilizing agent to its role in dual-function cathode and anode stabilization, and finally to its modern integration with electrochemical polishing for enhanced SEI stability, LiNO3 has proven to be a key enabler of improved Li–O2 battery performance. These advancements collectively contribute to the development of highly stable, long-lasting Li–O2 batteries by ensuring better electrode protection, reduced side reactions, suppressed dendrite growth, and enhanced electrochemical efficiency.
 | ||
| Fig. 10 (a) Schematic of Li plating/stripping on Li anodes with distinct SEI layers in an O2 atmosphere. (b) Cycling stability of symmetric Li cells with pristine and polished anodes in O2-saturated DMSO at 1 mA cm−2 (1 mA h cm−2 cutoff capacity). (c) Performance and discharge voltage of Li–O2 cells using both anode types at a current density of 400 mA g−1 (500 mA h g−1 cutoff capacity). Reproduced from ref. 272 with permission from the Royal Society of Chemistry, copyright [2021]. | ||
Despite efforts to clarify its role in oxygen reduction under non-aqueous conditions, new debates have emerged due to the exceptionally low charge voltage and extended cycle life observed in iodide-containing aprotic Li–O2 batteries when water is introduced. In these systems, LiOH becomes the predominant discharge product, further complicating the reaction mechanism. The impact of water remains a contentious topic, requiring further investigation. Based on prevailing research and recent findings, water addition in aprotic Li–O2 cells does not appear to affect the dominance of Li2O2 formation, raising uncertainty about the pathway leading to LiOH formation in iodide-containing environments. Further controversy surrounds the oxidation mechanisms mediated by the iodide/triiodide (I3−) and iodide/iodine (I2) redox couples. Since the redox potential of the (I−/I3−) couple (∼3.0 V vs. Li/Li+) is lower than the decomposition voltage of LiOH (3.42 V in alkaline media and 3.82 V in neutral conditions vs. Li/Li+), the proposed mechanism appears thermodynamically unfavorable. Although factors such as water concentration and the hydration enthalpies of lithium salts could influence the redox behavior of LiOH oxidation, no consensus has been reached, further deepening the debate. Liu et al.286 reported that I3− does not directly react with Li2O2 grains, while LiOH oxidation is facilitated by I2, enabling reversible oxygen evolution. Conversely, Burke et al.,288 provided strong support for the interaction between Li2O2 and I3− species but did not confirm Li2O2 formation or reversible oxygen evolution under iodide-containing nonaqueous conditions. Instead, they attributed LiOH decomposition primarily to I2-driven reactions, leading to the irreversible generation of IO3−. Moreover, discrepancies exist in assigning the (I−/I3−) and (I3−/I2) redox couples to distinct charge plateaus, with conflicting interpretations across various studies. Another significant concern is redox shuttling, where species such as water or soluble peroxides migrate between the cathode and lithium anode, interfering with the charge process and introducing uncertainties into iodide-mediated reactions. To eliminate these ambiguities, three key issues must be addressed: (I) the role of iodide in LiOH formation during aprotic oxygen reduction; (II) the reactivity of oxidized iodide species (I3− and I2) toward Li2O2 and LiOH in working cells; and (III) the influence of water on oxygen reduction in iodide-containing Li–O2 batteries. Resolving these questions is crucial for accurately evaluating iodide's viability as an RM in these complex systems. Qiao et al.289 effectively mitigated redox shuttling by employing a solid electrolyte separator, allowing us to systematically reconcile the key areas of debate. During oxygen reduction, iodide catalysis enhances nucleophilic parasitic reactions involving hydroperoxide intermediates, leading to the accumulation of LiOH and other byproducts such as carboxylates. Unlike previous studies that used commercial Li2O2 or LiOH powders as reaction models, they provide direct evidence for oxidized iodide species' reactivity in actual cell systems. Their findings confirm that I3− can oxidize Li2O2, whereas LiOH remains unreactive toward I2. Notably, the presence of additional water can shift the discharge product from irreversible LiOH back to Li2O2, thereby enhancing reversible oxygen evolution. This unexpected “LiOH-to-Li2O2” transformation is attributed to increased alkalinity, which suppresses iodide-catalyzed hydroperoxide decomposition. Through comprehensive analysis and well-supported conclusions, this study offers a transformative perspective on these long-standing debates, providing deeper insights into the stabilization of solvents and the design of functional additives for both Li–O2 and Li–Air battery systems. They systematically analyzed the complex role of LiI in aprotic Li–O2 batteries. While LiI catalyzed the decomposition of peroxide intermediates, it also led to LiOH accumulation, negatively affecting the long-term battery stability. However, their study revealed that water in the electrolyte could buffer these effects by shifting the reaction pathway toward Li2O2 formation instead of LiOH accumulation, mitigating some of the negative consequences. It brought a revolutionary re-understanding of the role of iodide and water in Li–O2 battery systems. To harness the benefits of H2O2 in lowering charge overpotential and enhancing cycling stability, a direct approach would be to introduce H2O2 into the electrolyte while preventing water contamination. Wu et al.290 explored the use of an organic hydrogen peroxide compound, urea hydrogen peroxide (UH2O2), as an electrolyte additive to reduce the charge potential by promoting the decomposition of lithium hydroxide and, more importantly, suppressing side reactions caused by water. Urea has been shown to form deep eutectic electrolytes with LiTFSI, significantly influencing the structural and transport properties of LiTFSI, leading to enhanced Li+ diffusion and improved ionic conductivity. Fig. 11(a) shows the schematic of the Li–O2 cell configuration employing the UH2O2 electrolyte. By introducing a urea-hydrogen peroxide complex, they eliminated water-induced side reactions while maintaining high catalytic efficiency, achieving an ultra-low charge potential of ∼3.26 V and the rate capability of the Li–O2 coin type in the G4-UH2O2 system, as shown in Fig. 11(b). The toroid formation of the Li2O2 discharge product was observed from the SEM image shown in Fig. 11(c and d). This approach also improved the lithium anode stability and cycling performance by preventing unwanted electrolyte degradation. Zhang et al. introduced the concept of a self-protecting RM (InI3), where an in situ pre-deposited indium layer acts as a shield for the lithium anode against chemical degradation caused by the oxidized RM (I3−). This approach effectively enhances both energy efficiency and cycling stability. However, since indium is an electronic conductor, it readily forms a Li–In alloy with lithium metal. Over extended cycling, a portion of the lithium within the alloy remains reactive with the RM, potentially leading to undesired side reactions. Zhang et al.291 introduced organic iodides as dual-function RMs for Li–O2 batteries. These compounds not only help lower the charge overpotential but also offer a novel approach to shielding the lithium anode from I3− corrosion. During the charging process, organic cations present in the TEGDME electrolyte deposit onto the lithium surface, forming an in situ organic film that functions as an SEI-like protective layer. Unlike previously explored metallic indium coatings, this organic layer acts as both a Li-ion conductor and an electronic insulator. Additionally, the SEI-like layer is composed of organic compounds that provide flexibility and strong adhesion to the lithium surface, potentially preventing the lithium anode from degradation caused by soluble I3− while preserving the RM functionality. A wide range of organic iodides including triethylsulfonium iodide, choline iodide, tetramethylammonium iodide, and trimethylsulfoxonium iodide, can serve this purpose. Among them, triethylsulfonium iodide (C6H15SI and TESI) is identified as particularly effective, exhibiting dual functionality as both an efficient RM and an in situ SEI-forming agent.292 This occurs through reductive ethyl detachment followed by oxidation, leading to the formation of a protective layer containing both organic and inorganic components. This layer effectively mitigates I3−-induced degradation while enabling Li-ion transport. As a result, incorporating TESI as an additive significantly enhances the cycling stability and energy efficiency in Li–O2 batteries. Li et al.293 introduced 1-Boc-3-iodoazetidine (BIA) as a bifunctional additive, addressing the shuttle effect commonly associated with iodide-based RMs. BIA reacted with lithium metal to form lithium iodide (LiI) and an organometallic protective layer, which prevented unwanted interactions between free RMs (I3−) and the lithium anode. This in situ-formed protective layer inhibited lithium dendrite growth, reduced charge overpotential, and maintained RM efficiency. Their results showed that Li–O2 batteries with BIA exhibited an extended cycle life of 260 cycles, with significant improvements in charging stability at high current densities (160 cycles at 2000 mA g−1). LiOH is a promising discharge product due to its higher chemical stability than that of Li2O2 and its ability to undergo a 4e− discharge process, which is not feasible under typical aprotic conditions. However, the involvement of LiI as an RM complicates the reaction mechanisms, raising uncertainties about its practical viability. Additionally, soluble RMs introduce internal shuttling, leading to poor coulombic and energy efficiency.
 | ||
| Fig. 11 (a) Schematic of the Li–O2 cell configuration employing the G4-UH2O2 electrolyte. (b) Rate capability of the Li–O2 coin type in the G4-UH2O2 system. (c and d) SEM images of the cathode after discharge and subsequent recharge. Reproduced from ref. 290 with permission from Springer Nature, copyright [2017]. (e) Illustration of IPA-Li composite synthesis and its proposed reaction pathway. (f) Cycling stability of symmetric cells with IPA-Li and bare Li anodes at a current density of 1 mA cm−2 with a fixed capacity of 1 mA h cm−2. (g) Performance of IPA-Li-based Li–Air cells tested in ambient air at a current density of 500 mA g−1 with a limited capacity of 1000 mA h g−1. Reproduced from ref. 294 with permission from Wiley-VCH, copyright [2022]. | ||
Bi et al.295 presented a straightforward alternative of introducing sodium ions as cation electrolyte additives to regulate the solvation environment and shift the reaction pathway toward LiOH formation. This approach significantly lowers the LiOH charging potential from over 4.0 V to 3.3 V, eliminating the complexities and uncertainties associated with iodide oxidation. Notably, no sodium-based products were detected on the cathode, suggesting that sodium ions primarily alter Li-ion solvation and facilitate initial electrolyte degradation to form LiOH without further decomposition in subsequent cycles. Theoretical studies further predicted the mechanism underlying this reversible LiOH formation and decomposition. Bi et al.296 examined lithium iodide (LiI) as an RM, focusing on its role in enhancing the charge/discharge kinetics and reducing the charge overpotential. LiI facilitates the oxidation of Li2O2via iodine species (I3− and I2), improving the battery efficiency and reversibility. Additionally, LiI can alter discharge chemistry, leading to lithium hydroxide (LiOH) formation, which offers potential for a higher capacity but poses reversibility challenges. The study highlights the influence of water on LiI's catalytic activity and debates its precise role in ORR/OER. While LiI significantly enhances the Li–O2 battery performance, issues like side reactions and electrolyte compatibility require further research. Grey286 and colleagues revealed that incorporating LiI and H2O into a dimethoxyethane-based electrolyte enables the reversible formation and decomposition of LiOH in Li–O2 batteries, leading to high specific capacities and minimal overpotentials (0.2 V). When the H2O concentration exceeds 4000 ppm, LiOH replaces Li2O2 as the primary discharge product in the presence of a Ru catalyst. However, the influence of H2O in Li–O2 batteries remains controversial. While small amounts (<1000 ppm) enhance solution-phase reactions and improve discharge capacity, they may also promote the formation of ROS, such as singlet oxygen (1O2) and hydroxyl radicals, accelerating electrolyte degradation. Freunberger et al.268 associated 1O2 production with parasitic side reactions, which intensify as the water content increases. Additionally, Grey et al.287 observed that H2O enhances LiOH solubility, facilitating its interaction with Ru catalysts and generating surface hydroxyl species that contribute to solvent degradation in DMSO-based systems. Johnson et al.268 further demonstrated that trace H2O aids Li2O2 phase transfer catalysis, forming hydroperoxide species that ultimately lead to CH3CN decomposition. Recent studies have explored 3D matrix structures for lithium storage to suppress dendrite growth by reducing the current density. However, these approaches often accelerate electrolyte decomposition. Other strategies including solid electrolytes, artificial protection films, and molten Li infusion have also been investigated. Among them, constructing mixed interfaces with fast Li-ion transport has shown promise. Alloy-based protective layers such as LiZn and Li3Bi enhance ion transfer, while polymer-modified lithiophilic layers improve Li-metal stability, though their mechanical durability remains a concern. Guo et al.294 introduced an iodine-containing polymer/alloy (IPA) interfacial layer on lithium metal, formed via a reaction between ZnI2 and Li, followed by the polymerization of ethyl α-cyanoacrylate (ECA). The illustration of IPA–Li composite synthesis and its proposed reaction pathway are shown in Fig. 11(e). The polymer phase enhances mechanical flexibility and protects Li from air exposure, while LiZn alloy nanoparticles create high-conductivity ion pathways, suppressing dendrite growth. Additionally, soluble iodine species facilitate the reversible decomposition of discharge products, reducing charge voltage. As a result, IPA-Li anodes exhibit ultra-long cycling life, low charge overpotential, and enhanced stability in both oxygen and ambient air environments. The Li symmetric cell achieved stable 1200 h of stripping plating with 20 mV overpotential as shown in Fig. 11(f). Their study demonstrated that IPA-Li-based Li–Air batteries achieved a longer lifespan (120 cycles vs. 25 cycles for bare Li) and a lower overpotential (<1.6 V vs. >4.0 V), making it a promising strategy for stabilizing Li-metal anodes as shown in Fig. 11(g). Together, these studies establish iodide- and hydrogen peroxide-based additives as key enablers for high-performance Li–O2 batteries.
From early discoveries in solvating additives and LiI redox mediation to H2O2-based charge reduction strategies and the introduction of BIA and IPA-Li for lithium anode stabilization, these advancements have significantly improved the charge efficiency, dendrite suppression, and battery lifespan. However, challenges remain, particularly in controlling unwanted side reactions and electrolyte degradation, highlighting the need for further material innovations.
Several studies have highlighted the positive impact of the PFC additives on the Li–O2 battery performance. A pioneering US patent by Yazami revealed that incorporating PFCs into aqueous electrolytes increased the open-circuit voltage, suggesting their influence on cell reactions.319 Subsequent research by Balaish et al.320 and Zhang et al.301 reported increased discharge capacity with PFC and partially fluorinated compound additives, respectively. Wang et al.299 demonstrated that dispersing perfluorotributylamine in PC improved the current density during oxygen reduction, while Nishikami et al.300 observed a 1.5-fold capacity increase by dissolving perfluorohexyl bromide with lithium perfluorooctane sulfonate in tetraglyme. Similarly, literature reports on electrolyte solvent.321–326 confirmed enhanced discharge capacity and current density with 1-methoxyheptafluoropropane in DME and TEGDME-based electrolytes, using RRDE analysis. Despite these benefits, the limited miscibility of the PFCs in organic solvents remains a major challenge for their application in Li–O2 batteries. One strategy to address this issue involves dispersing PFCs as a liquid medium, but this approach often lacks long-term stability due to the instability of the two-phase liquid/liquid system. An alternative strategy involves using PFCs with lower fluorination levels, which can balance solubility in ether-based solvents with high oxygen dissolution capability. Another critical requirement for PFC additives is their chemical stability in the Li–O2 battery environment. Since superoxide radicals form during oxygen reduction and contribute to solvent degradation, including carbonates and glymes, PFC additives must be resistant to oxidative decomposition. Their previous research, using RRDE and CV, suggested the instability of 1-methoxyheptafluoropropane under these conditions. Wijaya et al.327 explored a gamma-fluorinated ether, 1,1,1,2,2,3,3,4,4-nonafluoro-6-propoxyhexane (TE4), as a promising additive for Li–O2 batteries. Density functional theory (DFT) calculations predict that TE4 exhibits superior stability against superoxide radicals compared to 1-methoxyheptafluoropropane, an alpha-fluorinated ether. Furthermore, TE4 is expected to have high oxygen solubility (47.76 cm3/100 mL). Unlike traditional PFCs, TE4 is highly miscible with TEGDME and lithium salts up to approximately 20 vol%, mitigating dispersion-related instability and eliminating the need for surfactants. In this work, they experimentally measure oxygen uptake in pure TE4 and TE4-tetraglyme mixtures at different concentrations. They demonstrate that the addition of TE4 significantly improves both discharge capacity and rate capability in Li–O2 cells. TE4 overcomes this challenge by being fully miscible with TEGDME, leading to a 4-fold increase in oxygen solubility and a 2-fold improvement in oxygen diffusibility. This enhancement results in a 10-fold increase in discharge capacity at high current densities (400 mA gC−1), attributed to improved oxygen availability at the cathode. The chemical stability of TE4 was confirmed using 1H and 19F NMR, showing no significant degradation, while FT-IR and XPS analysis confirmed the formation of the desired Li2O2 discharge product with minimal side reactions. Compared to other perfluorinated additives, TE4 stands out due to its complete miscibility with battery electrolytes, eliminating phase separation issues commonly associated with PFCs. This research highlights TE4 as a promising electrolyte additive that improves the Li–O2 battery performance by facilitating oxygen transport and enhancing discharge capacity without compromising electrolyte stability. Bruce and colleagues326 pretreated lithium anodes with carbonates, forming a relatively stable SEI layer composed of Li2CO3 and ROCO2Li. Boric acid (BA) was introduced by Huang et al.,328 as an electrolyte additive to form a stable and compact SEI layer on the lithium metal anode. This SEI layer primarily consists of lithium borates, carbonates, fluorides, and organic compounds, providing a mechanically strong and ionically conductive barrier that prevents unwanted side reactions. The protective layer formed by BA is particularly beneficial in suppressing dendrite growth, thereby improving the cycle life of Li–O2 batteries by more than sixfold. The SEI formed with BA is covalently bonded and offers significant mechanical strength, which enhances the stability of the Li-metal anode in an oxygen atmosphere. The film is also resistant to side reactions with oxygen or electrolytes. Fig. 12(a) shows the photographs of Li pellets immersed in DMSO with and without BA. In the fine chemicals industry, BA serves as an effective crosslinking agent, facilitating the formation of O–B–O bonds by linking hydroxyl groups on polymers or inorganic particles. It readily reacts with LiOH or Li2O, generating lithium borates with a continuous covalent O–B–O network. Notably, glassy lithium borate has been reported to exhibit Li-ion conductivity in the order of 10−7 S cm−1. Given that LiOH and Li2O are commonly present on the surface of lithium metal anodes in Li–O2 batteries, BA is expected to interact with these species, forming a protective film capable of facilitating Li-ion transport while safeguarding the anode, achieving stable 800 h of stripping plating, as shown in Fig. 12(b), and enhanced cycling performance of 140 cycles at a fixed capacity of 1000 mA h g−1, as shown in Fig. 12(c). Lai et al.329 introduced 1-methyl-3-benzyl-1H-imidazolium bromide (IMPBr) as an electrolyte additive that enhances SEI formation, functions as an RM, and stabilizes oxygen intermediates during the ORR/OER. The positively charged IMP+ is drawn to the anode surface through electrostatic forces, where it forms a protective SEI layer in situ. This barrier prevents DMSO, Br3−, and Br2 from reacting with lithium metal and inhibits dendrite formation. During discharge, IMP+ strongly interacts with the intermediate O2− species, while Br− remains closely associated with Li+, facilitating the solvation process. In the charging phase, Br− plays a crucial role in breaking down discharge products, effectively lowering the charge overpotential. The incorporation of 80 mM IMPBr into the Li–O2 battery electrolyte led to an enhanced discharge capacity of 3154 mA h g−1 when discharged down to 2.2 V, along with outstanding cycling stability, maintaining 50 cycles at 800 mA g−1 under a constrained capacity of 500 mA h g−1.
 | ||
| Fig. 12 (a) Photographs of Li pellets immersed in DMSO with and without boric acid (BA). (b) Cycling studies of symmetric Li cells with (red) and without (black) BA in an O2 atmosphere. (c) Li–O2 battery cycling study comparison with and without BA at a current density of 300 mA g−1 (1000 mA h g−1 capacity limit). Reproduced from ref. 328 with permission from Wiley-VCH, copyright [2018]. (d) Optical images of electrolytes and Li electrodes after 100 charge–discharge cycles. (e) Voltage profiles for Li plating/stripping in symmetric cells using phosphorene-coated Li in 1 M LiTFSI/DMA at ±0.5 mA cm−2 (0.5 h per cycle). Reproduced from ref. 330 with permission from the American Chemical Society, copyright [2018]. | ||
Inorganic passivation layers are among the most widely used artificial SEIs in LIBs, offering outstanding electrochemical stability. They enable operation under high voltage, elevated current densities, and extreme temperatures while maintaining durability in organic electrolyte systems. The study by Guo et al.331 explored the use of a lithium phosphate (Li3PO4) artificial SEI film to enhance the cycling stability of Li–O2 batteries. The researchers introduced a Li3PO4-protected lithium anode in a dimethyl sulfoxide (DMSO)-based electrolyte containing lithium iodide (LiI) as an RM and LiNO3 as a stabilizing agent. This protection strategy significantly improved the electrochemical stability of the lithium anode, leading to a uniform lithium deposition and suppressing the formation of dendritic lithium structures. The battery with the Li3PO4-protected anode exhibited an extended cycling lifespan of 152 cycles at a fixed capacity of 1000 mA h g−1 at 2 A g−1, outperforming conventional lithium anodes in the same electrolyte system. The synergy between the Li3PO4 protection layer and the optimized DMSO-based electrolyte resulted in enhanced coulombic efficiency, reduced charge overpotential, and improved rate capability. The findings suggest that inorganic SEI coatings such as Li3PO4 provide an effective approach to improving lithium anode stability and could be a viable strategy for advancing practical Li–O2 battery applications. The study by Kim et al.330 on two-dimensional phosphorene-derived protective layers for lithium metal anodes in Li–O2 batteries explores the use of phosphorene-derived lithium phosphide (Li3P) as an effective protective layer, and the optical images of electrolytes with and without phosphorene and Li electrodes after 100 charge–discharge cycles are shown in Fig. 12(d). This approach aims to address key challenges such as lithium dendrite growth and electrolyte decomposition, which hinder the practical application of Li–O2 batteries. The researchers demonstrated that Li3P forms a nanoscale protective layer that thermodynamically suppresses electrolyte decomposition due to its higher redox potential relative to the electrolyte solvents. Additionally, Li3P hinders lithium dendrite growth by making lithium plating thermodynamically unfavorable on its surface. The experimental results showed that the Li3P-coated lithium metal electrode exhibited stable cycling in symmetric cells, maintaining electrochemical stability over 500 cycles, as shown in Fig. 12(e).
When applied in Li–O2 batteries, the phosphorene-protected lithium anode prevented capacity fading over 50 cycles, and effectively suppressed dendrite formation. Theoretical calculations further supported the enhanced stability of the Li3P layer, revealing its high mechanical strength and ionic conductivity, which contributed to improved lithium plating/stripping behavior. These findings highlight the potential of phosphorene-derived Li3P as a promising anode protection strategy for extending the lifespan and efficiency of Li–O2 batteries.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 517 mA h g−1 and extended cycle life to 334 cycles, as shown in Fig. 13(a). However, the presence of excess chloride ions in the electrolyte led to unwanted interactions with superoxide species, resulting in the formation of parasitic byproducts that degrade long-term performance and the stripping plating performance with and without Sn(TFSI)2 + LiCl additive, as shown in Fig. 13(b). The introduction of AgTFSI339 in the electrolyte has shown promising results in creating a lithiophilic SEI on the lithium anode. This method involves using Ag-containing additives to form a protective layer on the lithium metal that promotes uniform lithium deposition and reduces nucleation barriers, which helps mitigate dendrite formation. The AgTFSI additive induces the formation of an SEI rich in LiF, Li3N, Ag2O, and Li–Ag alloys, all of which contribute to enhancing the electrochemical performance of the battery by suppressing the redox shuttling effects. This strategy not only provides a continuous protective layer but also enhances the electrochemical stability of Li–O2 batteries by minimizing the shuttling phenomenon, which typically deteriorates the performance of RMs. Ahmadiparidari et al.340 took this concept further by integrating LiTFSI with solvation modulation, which optimized the electrolyte's stability under high current densities. This combination minimized interfacial resistance and maintained a stable ionic environment during extended cycling.
517 mA h g−1 and extended cycle life to 334 cycles, as shown in Fig. 13(a). However, the presence of excess chloride ions in the electrolyte led to unwanted interactions with superoxide species, resulting in the formation of parasitic byproducts that degrade long-term performance and the stripping plating performance with and without Sn(TFSI)2 + LiCl additive, as shown in Fig. 13(b). The introduction of AgTFSI339 in the electrolyte has shown promising results in creating a lithiophilic SEI on the lithium anode. This method involves using Ag-containing additives to form a protective layer on the lithium metal that promotes uniform lithium deposition and reduces nucleation barriers, which helps mitigate dendrite formation. The AgTFSI additive induces the formation of an SEI rich in LiF, Li3N, Ag2O, and Li–Ag alloys, all of which contribute to enhancing the electrochemical performance of the battery by suppressing the redox shuttling effects. This strategy not only provides a continuous protective layer but also enhances the electrochemical stability of Li–O2 batteries by minimizing the shuttling phenomenon, which typically deteriorates the performance of RMs. Ahmadiparidari et al.340 took this concept further by integrating LiTFSI with solvation modulation, which optimized the electrolyte's stability under high current densities. This combination minimized interfacial resistance and maintained a stable ionic environment during extended cycling.
 | ||
| Fig. 13 (a) Terminal voltage and capacity evolution of Li–O2 cells with and without additive at a current density of 100 mA g−1. (b) Symmetric Li cell cycling with and without additive at 0.1 mA cm−2. Reproduced from ref. 338 with permission from Elsevier, copyright [2024]. (c) Cycling studies of Li–O2 cells using bare and IR-SEI-coated Li anodes at 400 mA g−1 with a fixed capacity of 500 mA h g−1. (Inset) ‘XMU’ LED array (48 lights) powered by a Li–O2 battery. (d) Cycling stability of Li symmetric cells with and without GTU at a current density of 10 mA cm−2 with a fixed capacity of 10 mA h cm−2. Reproduced from ref. 341 with permission from Elsevier, copyright [2022]. | ||
A high Li2O2 yield of 90.79% was achieved, accompanied by a remarkable discharge capacity of 46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 mA h gcarbon−1 at a current density of 1000 mA g−1, representing a 23-fold improvement compared to the cell with the additive-free electrolyte. The researchers utilized a liquid alloy, GaSnIn,344 as an electrolyte additive to create a gradient SEI layer composed of a flexible surface-rich polycarbonate matrix and an inorganic LiF-rich core. The liquid alloy facilitated the initial decomposition of LiPF6, leading to the formation of a stable SEI layer with a highly conductive inner structure for enhanced Li-ion transport and a flexible outer layer to accommodate volume expansion. This approach significantly improved lithium metal deposition, suppressed dendrite formation, and enhanced electrochemical performance, particularly at high current densities (10 mA cm−2). The study demonstrated a substantial reduction in voltage polarization and improved the coulombic efficiency due to the SEI stabilization. In Li‖LFP full cells, the liquid alloy-protected lithium anode achieved a high average coulombic efficiency of 99.06% and maintained a stable cycle life of over 2500 cycles at an areal capacity of 3 mA h cm−2. One of the earliest challenges in Li–O2 batteries was the instability of the lithium metal anode due to dendritic growth and electrolyte decomposition. To tackle this, Lee et al.345 introduced vinylpyrrolidone (VP) as an electrolyte additive to create a smooth lithium deposition interface due to dipolar VP molecules. VP molecules act as surface modifiers, ensuring uniform lithium plating, which suppresses dendrite formation. Moreover, VP was found to enhance the decomposition of Li2O2 because the oxygen in the VP could selectively attract Li+ ions, which have a positive charge in Li2O2, thereby improving the cathodic efficiency and reducing the overpotential. The incorporation of VP in the electrolyte notably enhanced the cycling stability of Li‖Li symmetric cells, maintaining a lower and more stable overpotential over 1200 cycles under high current densities, in contrast to the rapid failure of pristine cells, which lasted fewer than 500 cycles. However, VP suffers from gradual chemical instability in high-voltage cycling conditions, necessitating further refinement in electrolyte formulation. Zhang et al.,346 demonstrated that 18-crown-6 ether can boost lithium-salt dissociation, leading to improved solvation of Li+ ions. The addition of 100 mM 18-crown-6 ether significantly reduced overpotential and facilitated solution-phase Li2O2 formation. The introduction of 100 mM 18-crown-6 ether (100-18C6) into the electrolyte significantly improves the electrochemical stability and promotes a solution-phase Li2O2 formation mechanism in Li–O2 batteries, resulting in a high discharge capacity of 10
000 mA h gcarbon−1 at a current density of 1000 mA g−1, representing a 23-fold improvement compared to the cell with the additive-free electrolyte. The researchers utilized a liquid alloy, GaSnIn,344 as an electrolyte additive to create a gradient SEI layer composed of a flexible surface-rich polycarbonate matrix and an inorganic LiF-rich core. The liquid alloy facilitated the initial decomposition of LiPF6, leading to the formation of a stable SEI layer with a highly conductive inner structure for enhanced Li-ion transport and a flexible outer layer to accommodate volume expansion. This approach significantly improved lithium metal deposition, suppressed dendrite formation, and enhanced electrochemical performance, particularly at high current densities (10 mA cm−2). The study demonstrated a substantial reduction in voltage polarization and improved the coulombic efficiency due to the SEI stabilization. In Li‖LFP full cells, the liquid alloy-protected lithium anode achieved a high average coulombic efficiency of 99.06% and maintained a stable cycle life of over 2500 cycles at an areal capacity of 3 mA h cm−2. One of the earliest challenges in Li–O2 batteries was the instability of the lithium metal anode due to dendritic growth and electrolyte decomposition. To tackle this, Lee et al.345 introduced vinylpyrrolidone (VP) as an electrolyte additive to create a smooth lithium deposition interface due to dipolar VP molecules. VP molecules act as surface modifiers, ensuring uniform lithium plating, which suppresses dendrite formation. Moreover, VP was found to enhance the decomposition of Li2O2 because the oxygen in the VP could selectively attract Li+ ions, which have a positive charge in Li2O2, thereby improving the cathodic efficiency and reducing the overpotential. The incorporation of VP in the electrolyte notably enhanced the cycling stability of Li‖Li symmetric cells, maintaining a lower and more stable overpotential over 1200 cycles under high current densities, in contrast to the rapid failure of pristine cells, which lasted fewer than 500 cycles. However, VP suffers from gradual chemical instability in high-voltage cycling conditions, necessitating further refinement in electrolyte formulation. Zhang et al.,346 demonstrated that 18-crown-6 ether can boost lithium-salt dissociation, leading to improved solvation of Li+ ions. The addition of 100 mM 18-crown-6 ether significantly reduced overpotential and facilitated solution-phase Li2O2 formation. The introduction of 100 mM 18-crown-6 ether (100-18C6) into the electrolyte significantly improves the electrochemical stability and promotes a solution-phase Li2O2 formation mechanism in Li–O2 batteries, resulting in a high discharge capacity of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 828.8 mA h gcarbon−1. However, crown ethers are susceptible to oxidative degradation. A novel ionic liquid electrolyte consisting of succinonitrile and LiTFSI has been developed by Man et al.,347 along with fluoroethylene carbonate as an additive to form an artificial SEI. This electrolyte system addresses the instability of traditional organic electrolytes at high voltages (over 4.5 V) and provides excellent performance in Li–O2 batteries. The low volatility and inherent flame-retardant nature of the electrolyte ensured safe and consistent battery operation under ambient air conditions. When tested in a pure oxygen environment, the Li–O2 cell utilizing this electrolyte demonstrated exceptional longevity, achieving over 1000 cycles at a capacity of 200 mA h g−1 and more than 150 cycles at 500 mA h g−1. Remarkably, even without the use of an oxygen-selective membrane, the battery maintained stable cycling for over 350 and 150 cycles at the respective capacities under open–air exposure. The development of artificial layers using electrolyte additives plays a critical role in improving the stability and performance of Li–O2 batteries. Li et al.348 introduced 2-methoxybenzonitrile (2-MBN), which modified the solvation structure and enhanced Li-ion transport. This reduced polarization and improved the overall cell efficiency. The dual-functionality of 2-MBN in simultaneously stabilizing both the anode and the cathode significantly enhances the cycling performance of Li–O2 batteries, achieving 97 stable cycles at 600 mA g−1 with a fixed capacity of 2000 mA h g−1, substantially outperforming the control cells without 2-MBN, which lasted only 28 cycles.
828.8 mA h gcarbon−1. However, crown ethers are susceptible to oxidative degradation. A novel ionic liquid electrolyte consisting of succinonitrile and LiTFSI has been developed by Man et al.,347 along with fluoroethylene carbonate as an additive to form an artificial SEI. This electrolyte system addresses the instability of traditional organic electrolytes at high voltages (over 4.5 V) and provides excellent performance in Li–O2 batteries. The low volatility and inherent flame-retardant nature of the electrolyte ensured safe and consistent battery operation under ambient air conditions. When tested in a pure oxygen environment, the Li–O2 cell utilizing this electrolyte demonstrated exceptional longevity, achieving over 1000 cycles at a capacity of 200 mA h g−1 and more than 150 cycles at 500 mA h g−1. Remarkably, even without the use of an oxygen-selective membrane, the battery maintained stable cycling for over 350 and 150 cycles at the respective capacities under open–air exposure. The development of artificial layers using electrolyte additives plays a critical role in improving the stability and performance of Li–O2 batteries. Li et al.348 introduced 2-methoxybenzonitrile (2-MBN), which modified the solvation structure and enhanced Li-ion transport. This reduced polarization and improved the overall cell efficiency. The dual-functionality of 2-MBN in simultaneously stabilizing both the anode and the cathode significantly enhances the cycling performance of Li–O2 batteries, achieving 97 stable cycles at 600 mA g−1 with a fixed capacity of 2000 mA h g−1, substantially outperforming the control cells without 2-MBN, which lasted only 28 cycles.
| Additive category | Efficiency | Stability |
|---|---|---|
| Iodide-based additives | High charge–discharge efficiency | Moderate stability-iodine shuttling and mediator loss |
| Nitrate-based additives | Moderate efficiency and improves reversibility | High interfacial stability, but may decompose in polar solvents |
| Organic compound-based additives | High kinetic efficiency | Low stability, chemically unstable toward superoxide and Li metal |
| Fluoride-/boron-/phosphorene-based additives | High electrochemical efficiency | Moderately costly, viscous, and may release corrosive species |
| Ionic-liquid additives | Moderate efficiency due to high viscosity | High chemical stability and thermal stability |
| Hybrid/multifunctional additives | High overall efficiency | Enhanced stability, but system complexity increases |
8. Perspectives
Future improvement of Li–O2 batteries will strongly depend on precise catalyst and electrolyte engineering. For cathode catalysts, controlling the particle size is vital for balancing active-site exposure and structural stability. Reducing the particle size increases the surface area and accessible catalytic sites, facilitating Li2O2 formation and decomposition, whereas overly small particles may induce agglomeration or parasitic side reactions. Constructing conductive architectures, for instance, embedding transition-metal oxides within carbon nanotube or graphene networks, enhances electron transport and structural integrity, thereby improving the efficiency and durability. On the electrolyte side, rational design principles should guide the choice of solvent, salt, and additive. High DN solvents improve Li+ solvation but may suffer from oxidative instability; low DN systems enhance stability but limit ion transport. Optimal formulations, therefore, require a balance between ionic conductivity, redox mediator compatibility, and chemical inertness toward superoxide species. Incorporating functional additives and tailored salt anions can further regulate solvation structure, suppress side reactions, and extend the cycle life. These interdependent design strategies for both catalysts and electrolytes are key to realizing practical, high-efficiency Li–O2 batteries.Electrolyte additives have emerged as powerful tools to overcome the intrinsic challenges of aprotic Li–O2 batteries by enabling targeted stabilization of the lithium metal anode, oxygen cathode, and the electrolyte. Going forward, the rational design of multifunctional electrolyte additives will be central to advancing Li–O2 technology toward practical deployment. The future of the additive research lies in developing single-component or synergistic additive systems that can simultaneously regulate redox processes, form stable interfaces, and suppress parasitic reactions. Fig. 14 shows the comparison plot of various additives with their performance. For instance, bifunctional additives like LiNO3 not only promoted stable SEI formation on lithium but also enhanced Li2O2 oxidation kinetics through reversible redox mediation. Similarly, iodide-based additives in the presence of controlled water content showed promise in shifting discharge pathways while maintaining low charge overpotentials, but their long-term stability and redox shuttling effects must be critically addressed. Another important direction is the design of chemically stable, redox-active organic additives that can modulate both the oxygen reduction and evolution reactions without contributing to undesired side reactions.
Additives with tailored donor/acceptor characteristics could selectively stabilize reactive oxygen intermediates, minimise electrolyte decomposition, and enhance discharge product reversibility. Emerging strategies also include fluorine-containing additives that improve oxidative stability, oxygen solubility, and SEI robustness. However, achieving high compatibility across various electrolyte formulations (e.g., ether-based, ionic liquid, or hybrid systems) remains a key challenge. From the collective studies summarized above, it is evident that few additives exhibit multifunctional protection, acting simultaneously on the anode, cathode, and electrolyte interfaces. For instance, LiNO3 and LiI not only stabilize the Li metal through SEI formation but also influence redox mediator stability, while FEC suppresses parasitic oxidation and improves Li2O2 decomposition. Fig. 8 schematically represents this threefold protection strategy, where a single additive can impart synergistic effects across multiple interfaces. Despite significant progress, most reported additives function selectively at one interface, limiting overall stability. Future research should focus on multifunctional or self-adaptive additives capable of simultaneously regulating SEI formation, stabilizing redox mediators, and suppressing electrolyte decomposition. Integrating molecular design with interfacial engineering approaches, such as artificial SEI coatings, polymeric ion-selective layers, or hybrid solid–liquid interfaces, can provide comprehensive protection to both electrodes and the electrolyte. Advanced in situ spectroscopic and computational methods will be key to uncovering these cross-interface mechanisms and guiding rational additive design. These innovations would pave the way for long-life, high-energy, and safe Li–O2 batteries under realistic operating conditions. The different additives utilized for Li–O2 battery are given in Table 3.
| Additive | Electrolyte composition | Device fixed capacity (mA h g−1)/current density (mA g−1)/cycle no. | Ref. |
|---|---|---|---|
| VP | 1 M LiTFSI/TEGDME/100 ppm VP | 600C mA h g−1/100C mA g−1/50 | 345 |
| LiCl and Sn(TFSI)2 | 1 M LiTFSI/TEGDME/6.7 mM LiCl/13.3 mM Sn(TFSI)2 | 500 mA h g−1/100 mA g−1/334 | 338 |
| GTU | 1 M LiTFSI/DME/DOL/2 wt% LiNO3 | 500 mA h g−1/400 mA g−1/489 | 341 |
| BA | 0.5 M LiTFSI/DMSO/20 mM BA | 1000 mA h g−1/300 mA g−1/146 | 328 |
| IPA-Li | LiTFSI/DME/DOL/2.0 wt% LiNO3/0.1 M ZnI2 in the mixed solution of ethyl α-cyanoacrylate (ECA) monomers | 1000 mA h g−1/500 mA g−1/120 | 294 |
| LiI | 1 M LiCF3SO3/TEGDME/0.1 M LiI | 4 mA h g−1/0.1 mA/100 | 296 |
| Na triflate | 1 M Li triflate/TEGDME/0.1 M Na triflate | 0.5 mA h cm−2/50 µA cm−2/30 | |
| TE4 | 0.1 M LiTFSI:TEGDME/20 vol% TE4 | 100C mA h g−1/100C mA g−1/50 | 327 |
| SiO2–LiI-GPE | 1 M LiTFSI/TEGDME/0.05 M LiI/4% SiO2 | 1000 mA h g−1/100 mA g−1/100 | 334 |
| LiNO3 | 1 M LiTFSI/DME-DOL/2 wt% LiNO3 | 500 mA h g−1/400 mA g−1/375 | 272 |
| TMP | 0.5 M LiTFSI/TEGDME/50 mM TMP | 5712.3 mA h g−1/0.1 mA cm−2 | 342 |
| CNTs | 3 wt% CNTs | 0.1256 mA h cm−2/0.05 mA cm−1/55 | 349 |
| KOTf | 1 M LiOTf/TEGDME/0.1 M KOTf/0.1 M KI | 500 mA h g−1/0.1 mA cm−2/30 | 336 |
| BIA | 1.0 M LiTFSI/TEGDME/0.1 mL/100 mM BIA | 1000 mA h g−1/1000 mA g−1/260 | 293 |
| SN | 1 M/L LiTFSI/SN/10% FEC | 200 mA h g−1/200 mA h g−1/1000 | 347 |
| G4-UH2O2 | 1 M LiTFSI/tetraglyme/5 wt% G4-UH2O2 | 1000KB mA h g−1/100 mA g−1/1000KB | 290 |
| 2-MBN | 1 M LiTFSI/DMSO/2-MBN (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 49 v/v) 49 v/v) |
1000 mA h g−1/300 mA g−1/160 | 348 |
| TESI | 1.0 M LiTFSI/TEGDME/50 mM TESI | 1000C mA h g−1/500 mA g−1/60 | 292 |
| AgTFSI | 1 M of LiTFSI/TEGDME/100 mM of LiNO3/70 mM LiI/200 mM AgTFSI | 500 mA h g−1/100 mA g−1/210 | 339 |
| IMPBr | 1 M LiClO4/DMSO/80 mM IMPBr | 500/800/50 | 329 |
| PEO and Al2O3 | NMP/1 M LiPF6/0.5 wt% PEO/1 wt% Al2O3 | 2.5 mA h/0.1 mA cm−2/35 | 350 |
| Li3PO4 | 1 M LiNO3/DMSO/0.05 M LiI | 1000 mA h g−1/2 A g−1/152 | 331 |
| RhB | 1 M LiTFSI/TEGDME/200 mm RhB | 1000 mA h g−1/500C mA g−1/200 | 343 |
| 18-Crown-6 ether | 1 M LiClO4/DMSO/100 mM 18-crown-6 ether | 1000C mA h g−1/400C mA g−1/128 | 346 |
| CF3(CF2)2I | 1 M LiNO3/DMA/0.2 M CF3(CF2)2I | 0.5 mA cm−2/5 mA h cm−2/100 | 351 |
8.1 Practical viability of additive strategies
Beyond laboratory performance, the large-scale implementation of the electrolyte additives must also consider cost, safety, and environmental sustainability. Many high-performance additives such as fluorinated organics and ionic-liquid derivatives remain expensive and difficult to synthesize on a large scale, while some fluorinated compounds pose potential ecological and safety risks. Future efforts should therefore prioritize the design of economically feasible, low-toxicity, and recyclable additives derived from fluorine-free or bio-based precursors. Simplified synthetic routes, solvent-recovery protocols, and material-life-cycle analyses will be critical for bridging laboratory advances with practical Li–O2 battery manufacturing.9. Conclusion
A comprehensive assessment of the electrochemical kinetics, reaction mechanisms, and challenges/strategies/recent advances for aprotic Li–O2 batteries, with a specific focus on electrolyte additive-based threefold protection, is presented in this review. The key points are as follows:(1) Anode stabilization: additives such as LiNO3, boron compounds, and polymer precursors contribute to the formation of robust SEI layers, mitigating dendritic growth and parasitic corrosion of the lithium metal.
(2) Cathode protection: the incorporation of RMs (e.g., LiI, DBBQ, TEMPO) facilitates the decomposition of Li2O2 at lower overpotentials, while functional additives modulate the discharge morphology and enhance the O2 solubility.
(3) Electrolyte stabilization: high-DN solvents and fluorinated additives improve intermediate solvation and suppress ROS-induced degradation, whereas hybrid systems like ionic liquid gels and ceramic-reinforced polymer matrices enable safe, long-term operation.
By categorising and critically analysing over 60 distinct additives, this review mapped the functional roles they play from redox mediation and SEI formation to solubility modulation and interfacial passivation. It is evident that electrolyte additives serve as central enablers for overcoming the intrinsic limitations of Li–O2 systems. In conclusion, while significant progress was achieved, the pathway toward commercially viable Li–O2 batteries hinges on the integrated design of multifunctional additives. Future research should prioritize the development of unified additive systems that can simultaneously regulate redox kinetics, electrolyte decomposition, and electrode degradation, thus routing Li–O2 technology toward practical realization.
Author contributions
SS: writing – original draft, writing – review & editing. PE: supervision, writing – review & editing and funding acquisition.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
No new data were generated as this work is based entirely on existing literature, which is appropriately cited within the article.Acknowledgements
PE thanks Anusandhan National Research Foundation (ANRF)-Partnerships for Accelerated Innovation and Research (PAIR) (ANRF/PAIR/2025/000021/PAIR-A) and the Central Power Research Institute (CPRI), Bangalore, for funding the research (CPRI/R&D/TC/GDEC/2025). SS thanks the CSIR-SRF for the Senior Research Fellowship (09/0559(23742)/2025-EMR-I).References
- E. Karden, S. Ploumen, B. Fricke, T. Miller and K. Snyder, Energy storage devices for future hybrid electric vehicles, J. Power Sources, 2007, 168, 2–11 CrossRef CAS.
- T. Maiyalagan and P. Elumalai, Rechargeable Lithium-Ion Batteries: Trends and Progress in Electric Vehicles, CRC Press, London, 2020. Search PubMed.
- L. Grande, E. Paillard, J. Hassoun, J. B. Park, Y. J. Lee, Y. K. Sun, S. Passerini and B. Scrosati, The lithium/air battery: Still an emerging system or a practical reality?, Adv. Mater., 2015, 27, 784–800 CrossRef CAS PubMed.
- H. C. Lee, J. O. Park, M. Kim, H. J. Kwon, J. H. Kim, K. H. Choi, K. Kim and D. Im, High-Energy-Density Li-O2 Battery at Cell Scale with Folded Cell Structure, Joule, 2019, 3, 542–556 CrossRef CAS.
- J. Lu, L. Li, J. B. Park, Y. K. Sun, F. Wu and K. Amine, Aprotic and aqueous Li-O2 batteries, Chem. Rev., 2014, 114, 5611–5640 CrossRef CAS.
- V. Sankar Devi, M. Athika, S. Sandhiya, and P. Elumalai, Recent Developments of Transition Metal Oxide Nanoparticles on Oxygen Reduction Reaction, Nano-electrocatalyst for Oxygen Reduction Reaction: Fundamentals to Field Applications, 2024, pp. 34–70 Search PubMed.
- Q. Qiu, J. Long, P. Yao, J. Wang, X. Li, Z. Z. Pan, Y. Zhao and Y. Li, Cathode electrocatalyst in aprotic lithium oxygen (Li-O2) battery: A literature survey, Catal. Today, 2023, 420, 114138 CrossRef CAS.
- S. Sathyanarayana and N. Munichandraiah, A new magnesium-air cell for long-life applications, J. Appl. Electrochem., 1981, 11, 33–39 CrossRef CAS.
- A. Zahoor, Z. K. Ghouri, S. Hashmi, F. Raza, S. Ishtiaque, S. Nadeem, I. Ullah and K. S. Nahm, Electrocatalysts for lithium–air batteries: current status and challenges, ACS Sustain. Chem. Eng., 2019, 7, 14288–14320 CrossRef CAS.
- W. Li, C. Han, K. Zhang, S. Chou and S. Dou, Strategies for boosting carbon electrocatalysts for the oxygen reduction reaction in non-aqueous metal–air battery systems, J. Mater. Chem. A, 2021, 2021(9), 6671–6693 RSC.
- Z. L. Wang, D. Xu, J. J. Xu and X. B. Zhang, Oxygen electrocatalysts in metal–air batteries: from aqueous to nonaqueous electrolytes, Chem. Soc. Rev., 2014, 43, 7746–7786 RSC.
- Q. Liu, Z. Pan, E. Wang, L. An and G. Sun, Aqueous metal-air batteries: Fundamentals and applications, Energy Storage Mater., 2020, 27, 478–505 CrossRef.
- W. Chen, Y. F. Gong and J. H. Liu, Recent advances in electrocatalysts for non-aqueous Li–O2 batteries, Chem. Lett., 2017, 28, 709–718 CAS.
- Y. Chen, J. Xu, P. He, Y. Qiao, S. Guo, H. Yang and H. Zhou, Metal-air batteries: progress and perspective, Sci. Bull., 2022, 67, 2449–2486 CrossRef CAS.
- W. Ling, H. Wang, Z. Chen, Z. Ji, J. Wang, J. Wei, Y. Huang, W. Ling, H. Wang, Z. Chen, Z. Ji, J. Wang, J. Wei and Y. Huang, Intrinsic structure modification of electrode materials for aqueous metal-ion and metal-air batteries, Adv. Funct. Mater., 2021, 31(5), 2006855 CrossRef CAS.
- M. A. Rahman, X. Wang and C. Wen, A review of high energy density lithium–air battery technology, J. Appl. Electrochem., 2014, 44, 5–22 CrossRef CAS.
- J. Zhao, D. Wei, C. Zhang, Q. Shao, V. Murugadoss, Z. Guo, Q. Jiang and X. Yang, An overview of oxygen reduction electrocatalysts for rechargeable zinc-air batteries enabled by carbon and carbon composites, Eng. Sci., 2021, 15, 1–19 CAS.
- P. He, T. Zhang, J. Jiang and H. Zhou, Lithium–air batteries with hybrid electrolytes, J. Phys. Chem. Lett., 2016, 7, 1267–1280 CrossRef CAS.
- C. X. Zhao, J. N. Liu, J. Wang, D. Ren, B. Q. Li and Q. Zhang, Recent advances of noble-metal-free bifunctional oxygen reduction and evolution electrocatalysts, Chem. Soc. Rev., 2021, 50, 7745–7778 RSC.
- T. Zhang and H. Zhou, A reversible long-life lithium–air battery in ambient air, Nat. Commun., 2013, 4, 1817 CrossRef.
- A. Manthiram and L. Li, Hybrid and Aqueous Lithium-Air Batteries, Adv. Energy Mater., 2015, 5(4), 1401302 CrossRef.
- J. Lai, Y. Xing, N. Chen, L. Li, F. Wu and R. Chen, Electrolytes for rechargeable lithium–air batteries, Angew. Chem., Int. Ed., 2020, 59, 2974–2997 CrossRef CAS PubMed.
- C. G. Arges, L. Wang and J. Parrondo, et al., A brief history of non-aqueous metal-air batteries, ECS Trans., 2008, 3, 67–71 CrossRef.
- T. Ogasawara, A. Débart, M. Holzapfel, P. Novák and P. G. Bruce, Rechargeable Li2O2 Electrode for Lithium Batteries, J. Am. Chem. Soc., 2006, 128, 1390–1393 CrossRef CAS.
- F. Wang, O. Borodin, T. Gao, X. Fan, W. Sun, F. Han, A. Faraone, J. A. Dura, K. Xu and C. Wang, Highly reversible zinc metal anode for aqueous batteries, Nat. Mater., 2018, 17, 543–549 CrossRef CAS PubMed.
- Q. Dong, X. Yao, Y. Zhao, M. Qi, X. Zhang, H. Sun, Y. He and D. Wang, Cathodically Stable Li-O2 Battery Operations Using Water-in-Salt Electrolyte, Chem, 2018, 4, 1345–1358 CAS.
- J. Park, M. Park, G. Nam, J. Lee, J. Cho, J. Park, M. Park, G. Nam, J. Lee and J. Cho, All-Solid-State Cable-Type Flexible Zinc–Air Battery, Adv. Mater., 2015, 27, 1396–1401 CrossRef CAS.
- J. Zhang, Z. Zhao, Z. Xia and L. Dai, A metal-free bifunctional electrocatalyst for oxygen reduction and oxygen evolution reactions, Nat. Nanotechnol., 2015, 10, 444–452 CrossRef CAS PubMed.
- B. Li, D. Geng, X. S. Lee, X. Ge, J. Chai, Z. Wang, J. Zhang, Z. Liu, T. S. A. Hor and Y. Zong, Eggplant-derived microporous carbon sheets: towards mass production of efficient bifunctional oxygen electrocatalysts at low cost for rechargeable Zn–air batteries, Chem. Commun., 2015, 51, 8841 RSC.
- Y. Chen, S. A. Freunberger, Z. Peng, O. Fontaine and P. G. Bruce, Charging a Li-O2 battery using a redox mediator, Nat. Chem., 2013, 5, 489–494 CrossRef CAS PubMed.
- X. Ren and Y. Wu, A low-overpotential potassium-oxygen battery based on potassium superoxide, J. Am. Chem. Soc., 2013, 135, 2923–2926 CrossRef CAS PubMed.
- Z. Peng, S. A. Freunberger, Y. Chen and P. G. Bruce, A reversible and higher-rate Li-O2 battery, Science, 2012, 337, 563–566 CrossRef CAS PubMed.
- N. Imanishi, S. Hasegawa, T. Zhang, A. Hirano, Y. Takeda and O. Yamamoto, Lithium anode for lithium-air secondary batteries, J. Power Sources, 2008, 185, 1392–1397 CrossRef CAS.
- J. Read, Ether-Based Electrolytes for the Lithium/Oxygen Organic Electrolyte Battery, J. Electrochem. Soc., 2006, 153, A96 CrossRef CAS.
- T. Ogasawara, A. Débart, M. Holzapfel, P. Novák and P. G. Bruce, Rechargeable Li2O2 electrode for lithium batteries, J. Am. Chem. Soc., 2006, 128, 1390–1393 CrossRef CAS.
- T. Kuboki, T. Okuyama, T. Ohsaki and N. Takami, Lithium-air batteries using hydrophobic room temperature ionic liquid electrolyte, J. Power Sources, 2005, 146, 766–769 CrossRef CAS.
- S. Müller, F. Holzer and O. Haas, Optimized zinc electrode for the rechargeable zinc–air battery, J. Appl. Electrochem., 1998, 28, 895–898 CrossRef.
- K. M. Abraham and Z. Jiang, A Polymer Electrolyte-Based Rechargeable Lithium/Oxygen Battery, J. Electrochem. Soc., 1996, 143, 1–5 CrossRef CAS.
- L. Öjefors and L. Carlsson, An iron—air vehicle battery, J. Power Sources, 1978, 2, 287–296 CrossRef.
- S. Zaromb, The Use and Behavior of Aluminum Anodes in Alkaline Primary Batteries, J. Electrochem. Soc., 1962, 109, 1125 CrossRef CAS.
- H. Yuan, J. Nai, H. Tian, Z. Ju, W. Zhang, Y. Liu, X. Tao and X. W. Lou, An ultrastable lithium metal anode enabled by designed metal fluoride spansules, Sci. Adv., 2020, 6, 3112–3118 CrossRef.
- Y. Li, Y. Li, A. Pei, K. Yan, Y. Sun, C. L. Wu, L. M. Joubert, R. Chin, A. L. Koh, Y. Yu, J. Perrino, B. Butz, S. Chu and Y. Cui, Atomic structure of sensitive battery materials and interfaces revealed by cryo–electron microscopy, Science, 2017, 358, 506–510 CrossRef CAS PubMed.
- C. Yang, Y. Yin, S. Zhang, N. Li and Y. Guo, Accommodating lithium into 3D current collectors with a submicron skeleton towards long-life lithium metal anodes, Nat. Commun., 2015, 6, 8058 CrossRef CAS PubMed.
- K. Yan, Z. Lu, H. Lee, F. Xiong, P. Hsu, Y. Li, J. Zhao, S. Chu and Y. Cui, Selective deposition and stable encapsulation of lithium through heterogeneous seeded growth, Nat. Energy, 2016, 1, 16010 CrossRef CAS.
- C. Fang, J. Li, M. Zhang, Y. Zhang and F. Yang, et al., Quantifying Inactive Lithium in Lithium Metal Batteries, Nature, 2019, 572, 511–515 CrossRef CAS.
- M. Wu, Z. Wen, Y. Liu, X. Wang and L. Huang, Electrochemical behaviors of a Li3N modified Li metal electrode in secondary lithium batteries, J. Power Sources, 2011, 196(19), 8091–8097 CrossRef CAS.
- C. Jin, T. Liu, O. Sheng, M. Li, T. Liu, Y. Yuan and J. Nai, et al., Rejuvenating dead lithium supply in lithium metal anodes by iodine redox, Nat. Energy, 2021, 6, 378–387 CrossRef CAS.
- C. Fang, J. Li, M. Zhang, Y. Zhang and F. Yang, et al., Nature and undefined 2019, Quantifying inactive lithium in lithium metal batteries, Nature, 2019, 572, 511–515 CrossRef CAS PubMed.
- X. Liang, Z. Wen, Y. Liu, M. Wu, J. Jin, H. Zhang and X. Wu, Improved cycling performances of lithium sulfur batteries with LiNO3-modified electrolyte, J. Power Sources, 2011, 196, 9839–9843 CrossRef CAS.
- K. Naoi, M. Mori, Y. Naruoka, W. M. Lamanna and R. Atanasoski, The Surface Film Formed on a Lithium Metal Electrode in a New Imide Electrolyte, Lithium Bis(perfluoroethylsulfonylimide) [LiN (C2F5SO2)2], J. Electrochem. Soc., 1999, 146, 462–469 CrossRef CAS.
- K. Semkow and A. Sammells, A lithium oxygen secondary battery, J. Electrochem. Soc., 1987, 134, 2084 CrossRef CAS.
- J. H. Kang, J. Lee, J. W. Jung, J. Park, T. Jang, H. S. Kim, J. S. Nam, H. Lim, K. R. Yoon, W. H. Ryu, I. D. Kim and H. R. Byon, Lithium–air batteries: air-breathing challenges and perspective, ACS Nano, 2020, 14, 14549–14578 CrossRef CAS PubMed.
- Z. Wen, C. Shen and Y. Lu, Air electrode for the lithium–air batteries: Materials and structure designs, ChemPlusChem, 2015, 80, 270–287 CrossRef CAS.
- L. Li, Z. wen Chang and X. B. Zhang, Recent progress on the development of metal-air batteries, Adv. Sustain. Syst., 2017, 1(10), 2056–2080 Search PubMed.
- C. O. Laoire, S. Mukerjee, K. M. Abraham, E. J. Plichta and M. A. Hendrickson, Influence of nonaqueous solvents on the electrochemistry of oxygen in the rechargeable lithium-air battery, J. Phys. Chem. C, 2010, 114, 9178–9186 CrossRef CAS.
- C. O. Laoire, S. Mukerjee, K. M. Abraham, E. J. Plichta and M. A. Hendrickson, Elucidating the mechanism of oxygen reduction for lithium-air battery applications, J. Phys. Chem. C, 2009, 113, 20127–20134 CrossRef CAS.
- C. M. Burke, V. Pande, A. Khetan, V. Viswanathan and B. D. McCloskey, Enhancing electrochemical intermediate solvation through electrolyte anion selection to increase nonaqueous Li-O2 battery capacity, Proc. Natl. Acad. Sci. U. S. A., 2015, 112, 9293–9298 CrossRef CAS.
- N. B. Aetukuri, B. D. McCloskey, J. M. Garciá, L. E. Krupp, V. Viswanathan and A. C. Luntz, Solvating additives drive solution-mediated electrochemistry and enhance toroid growth in non-aqueous Li–O2 batteries, Nat. Chem., 2014, 7(1), 50–56 CrossRef PubMed.
- D. G. Kwabi, V. S. Bryantsev, T. P. Batcho, D. M. Itkis, C. V. Thompson and Y. Shao-Horn, Experimental and Computational Analysis of the Solvent-Dependent O2/Li+-O2− Redox Couple: Standard Potentials, Coupling Strength, and Implications for Lithium–Oxygen Batteries, Angew. Chem., 2016, 128, 3181–3186 CrossRef.
- B. D. McCloskey, A. Speidel, R. Scheffler, D. C. Miller, V. Viswanathan, J. S. Hummelshøj, J. K. Nørskov and A. C. Luntz, Twin problems of interfacial carbonate formation in nonaqueous Li-O2 batteries, J. Phys. Chem. Lett., 2012, 3, 997–1001 CrossRef CAS PubMed.
- S. Freunberger, The lithium-oxygen battery with ether-based electrolytes, Angew. Chem., 2011, 50(37), 8609–8613 CrossRef CAS PubMed.
- A. Khetan, A. Luntz and V. Viswanathan, Trade-offs in capacity and rechargeability in nonaqueous Li-O2 batteries: Solution-driven growth versus nucleophilic stability, J. Phys. Chem. Lett., 2015, 6, 1254–1259 CrossRef CAS PubMed.
- B. D. McCloskey, D. S. Bethune, R. M. Shelby, T. Mori, R. Scheffler, A. Speidel, M. Sherwood and A. C. Luntz, Limitations in rechargeability of li-o2 batteries and possible origins, J. Phys. Chem. Lett., 2012, 3, 3043–3047 CrossRef CAS PubMed.
- E. Mourad, Y. K. Petit, R. Spezia, A. Samojlov, F. F. Summa, C. Prehal, C. Leypold, N. Mahne, C. Slugovc, O. Fontaine, S. Brutti and S. A. Freunberger, Singlet oxygen from cation driven superoxide disproportionation and consequences for aprotic metal–O2 batteries, Energy Environ. Sci., 2019, 12, 2559–2568 RSC.
- N. Mahne, B. Schafzahl, C. Leypold, M. Leypold, S. Grumm, A. Leitgeb, G. A. Strohmeier, M. Wilkening, O. Fontaine, D. Kramer, C. Slugovc, S. M. Borisov and S. A. Freunberger, Singlet oxygen generation as a major cause for parasitic reactions during cycling of aprotic lithium–oxygen batteries, Nat. Energy, 2017, 2(5), 1–9 Search PubMed.
- X. Gao, Y. Chen, L. Johnson and P. G. Bruce, Promoting solution phase discharge in Li–O2 batteries containing weakly solvating electrolyte solutions, Nat. Mater., 2016, 15(8), 882–888 CrossRef CAS PubMed.
- C. O. Laoire, S. Mukerjee, K. M. Abraham, E. J. Plichta and M. A. Hendrickson, Elucidating the mechanism of oxygen reduction for lithium-air battery applications, J. Phys. Chem. C, 2009, 113, 20127–20134 CrossRef CAS.
- K. M. Abraham, Electrolyte-Directed Reactions of the Oxygen Electrode in Lithium-Air Batteries, J. Electrochem. Soc., 2015, 162, A3021–A3031 CrossRef CAS.
- J. Xie, Q. Dong, I. Madden, X. Yao, Q. Cheng, P. Dornath, W. Fan and D. Wang, Achieving Low Overpotential Li-O2 Battery Operations by Li2O2 Decomposition through One-Electron Processes, Nano Lett., 2015, 15, 8371–8376 CrossRef CAS.
- Y. Wang, N. C. Lai, Y. R. Lu, Y. Zhou, C. L. Dong and Y. C. Lu, A Solvent-Controlled Oxidation Mechanism of Li2O2 in Lithium-Oxygen Batteries, Joule, 2018, 2, 2364–2380 CrossRef CAS.
- S. Kang, Y. Mo, S. P. Ong and G. Ceder, A facile mechanism for recharging Li2O2 in Li-O2 batteries, Chem. Mater., 2013, 25, 3328–3336 CrossRef CAS.
- Z. Peng, S. A. Freunberger, L. J. Hardwick, Y. Chen, V. Giordani, F. Bardé, P. Novák, D. Graham, J. M. Tarascon and P. G. Bruce, Oxygen Reactions in a Non-Aqueous Li+ Electrolyte, Angew. Chem., Int. Ed., 2011, 50, 6351–6355 CrossRef CAS PubMed.
- Z. Peng, S. A. Freunberger, Y. Chen and P. G. Bruce, A reversible and higher-rate Li-O2 battery, Science, 2012, 337, 563–566 CrossRef CAS PubMed.
- F. S. Gittleson, W. H. Ryu and A. D. Taylor, Operando observation of the gold-electrolyte interface in Li-O2 batteries, ACS Appl. Mater. Interfaces, 2014, 6, 19017–19025 CrossRef CAS PubMed.
- Y. Wang, N. C. Lai, Y. R. Lu, Y. Zhou, C. L. Dong and Y. C. Lu, A Solvent-Controlled Oxidation Mechanism of Li2O2 in Lithium-Oxygen Batteries, Joule, 2018, 2, 2364–2380 CrossRef CAS.
- S. Cho, H. Jung, M. Park, L. Lyu and Y. M. Kang, Modulating the Configuration of Air Cathodes toward the Extended Triple-Phase Boundaries of Li-O2 Batteries, ACS Energy Lett., 2024, 9, 2848–2857 CrossRef CAS.
- A. Kundu, S. Mallick, S. Ghora and C. R. Raj, Advanced oxygen electrocatalyst for air-breathing electrode in Zn-air batteries, ACS Appl. Mater. Interfaces, 2021, 13, 40172–40199 CrossRef CAS PubMed.
- Y. Wang and S. C. Cho, Analysis of Air Cathode Perfomance for Lithium-Air Batteries, J. Electrochem. Soc., 2013, 160, A1847–A1855 CrossRef CAS.
- B. Liu, Y. Sun, L. Liu, S. Xu and X. Yan, Advances in Manganese-Based Oxides Cathodic Electrocatalysts for Li–Air Batteries, Adv. Funct. Mater., 2018, 28, 1704973 CrossRef.
- D. Liu, Y. Tong, X. Yan, J. Liang and S. X. Dou, Recent advances in carbon-based bifunctional oxygen catalysts for zinc-air batteries, Batteries Supercaps, 2019, 2, 743–765 CrossRef.
- H. Woo, J. Kang, J. Kim, C. Kim, S. Nam and B. Park, Development of carbon-based cathodes for Li-air batteries: present and future, Electron. Mater. Lett., 2016, 12, 551–567 CrossRef CAS.
- Y. Tian, L. Xu, J. Qiu, X. Liu and S. Zhang, Rational design of sustainable transition metal-based bifunctional electrocatalysts for oxygen reduction and evolution reactions, Sustainable Mater. Technol., 2020, 25, e00204 CrossRef CAS.
- D. Yang, L. Zhang, X. Yan and X. Yao, Recent progress in oxygen electrocatalysts for zinc–air batteries, Small Methods, 2017, 1(12), 1700209 CrossRef.
- F. Shi, X. Zhu and W. Yang, Micro-nanostructural designs of bifunctional electrocatalysts for metal-air batteries, Chin. J. Catal., 2020, 41, 390–403 CrossRef CAS.
- N. T. Suen, S. F. Hung, Q. Quan, N. Zhang, Y. J. Xu and H. M. Chen, Electrocatalysis for the oxygen evolution reaction: recent development and future perspectives, Chem. Soc. Rev., 2017, 46(2), 337–365 RSC.
- Z. Liu, Z. Zhao, B. Peng, X. Duan and Y. Huang, Beyond extended surfaces: understanding the oxygen reduction reaction on nanocatalysts, J. Am. Chem. Soc., 2020, 142, 17812–17827 CrossRef CAS PubMed.
- X. Ge, A. Sumboja, D. Wuu, T. An, B. Li, F. W. T. Goh, T. S. A. Hor, Y. Zong and Z. Liu, Oxygen reduction in alkaline media: from mechanisms to recent advances of catalysts, ACS Catal., 2015, 5, 4643–4667 CrossRef CAS.
- Y. Wang, J. Li and Z. Wei, Transition-metal-oxide-based catalysts for the oxygen reduction reaction, J. Mater. Chem. A, 2018, 6, 8194–8209 RSC.
- H. Jin, C. Guo, X. Liu, J. Liu, A. Vasileff, Y. Jiao, Y. Zheng and S. Z. Qiao, Emerging two-dimensional nanomaterials for electrocatalysis, Chem. Rev., 2018, 118(13), 6337–6408 CrossRef CAS PubMed.
- Z. Shi, X. Wang, J. Ge, C. Liu and W. Xing, Fundamental understanding of the acidic oxygen evolution reaction: mechanism study and state-of-the-art catalysts, Nanoscale, 2020, 12(25), 13249–13275 RSC.
- Z. Yan, H. Liu, Z. Hao, M. Yu, X. Chen and J. Chen, Electrodeposition of (hydro) oxides for an oxygen evolution electrode, Chem. Sci., 2020, 11(39), 10614–10625 RSC.
- Z. Jia, G. Yin, J. Zhang, Rotating Ring-Disk Electrode Method, Rotating Electrode Methods and Oxygen Reduction Electrocatalysts, 2014, pp. 199–229 Search PubMed.
- S. Lee, S. Pyun and S. L.-I. J. of, Chemistry and undefined 2008, Fundamentals of rotating disc and ring–disc electrode techniques and their applications to study of the oxygen reduction mechanism at Pt/C electrode for fuel cells, J. Chem., 2008, 48, 215–228 CAS.
- C. Du, Y. Sun, T. Shen, G. Yin and J. Zhang, Applications of RDE and RRDE Methods in Oxygen Reduction Reaction, Elsevier, 2014, pp. 231–277 Search PubMed.
- Z. F. Huang, J. Wang, Y. Peng, C. Y. Jung, A. Fisher and X. Wang, Design of efficient bifunctional oxygen reduction/evolution electrocatalyst: Recent Advances and Perspectives, Adv. Energy Mater., 2017, 7(23), 1700544 CrossRef.
- P. Agbo, An algorithm for the extraction of Tafel slopes, J. Phys. Chem. C, 2019, 123(50), 30252–30264 CrossRef CAS.
- Y. Chen, S. A. Freunberger, Z. Peng, F. Bardé and P. G. Bruce, Li-O2 battery with a dimethylformamide electrolyte, J. Am. Chem. Soc., 2012, 134, 7952–7957 CrossRef CAS.
- S. A. Freunberger, Y. Chen, Z. Peng, J. M. Griffin, L. J. Hardwick, F. Bardé, P. Novák and P. G. Bruce, Reactions in the rechargeable lithium-O2 battery with alkyl carbonate electrolytes, J. Am. Chem. Soc., 2011, 133, 8040–8047 CrossRef CAS PubMed.
- P. Su, W. Pei, X. Wang, Y. Ma, Q. Jiang, J. Liang, S. Zhou, J. Zhao, J. Liu and G. Q. Lu, Exceptional Electrochemical HER Performance with Enhanced Electron Transfer between Ru Nanoparticles and Single Atoms Dispersed on a Carbon Substrate, Angew. Chem., Int. Ed., 2021, 60, 16044–16050 CrossRef CAS.
- Z. Wu, Y. Tian, H. Chen, L. Wang, S. Qian, T. Wu, S. Zhang and J. Lu, Evolving aprotic Li–air batteries, Chem. Soc. Rev., 2022, 51, 8045–8101 RSC.
- W. J. Kwak, N. Rosy, D. Sharon, C. Xia, H. Kim, L. R. Johnson, P. G. Bruce, L. F. Nazar, Y. K. Sun, A. A. Frimer, M. Noked, S. A. Freunberger and D. Aurbach, Lithium-Oxygen Batteries and Related Systems: Potential, Status, and Future, Chem. Rev., 2020, 120, 6626–6683 CrossRef CAS PubMed.
- C. K. Park, S. B. Park, S. Y. Lee, H. Lee, H. Jang and W. I. Cho, Electrochemical Performances of Lithium-air Cell with Carbon Materials, Bull. Korean Chem. Soc., 2010, 31, 3221–3224 CrossRef CAS.
- G. Q. Zhang, J. P. Zheng, R. Liang, C. Zhang, B. Wang, M. Hendrickson and E. J. Plichta, Lithium–Air Batteries Using SWNT/CNF Buckypapers as Air Electrodes, J. Electrochem. Soc., 2010, 157, A953 CrossRef CAS.
- W. Xu, V. V. Viswanathan, D. Wang, S. A. Towne, J. Xiao, Z. Nie, D. Hu and J. G. Zhang, Investigation on the charging process of Li2O2-based air electrodes in Li–O2 batteries with organic carbonate electrolytes, J. Power Sources, 2011, 196, 3894–3899 CrossRef CAS.
- S. A. Freunberger, Y. Chen, N. E. Drewett, L. J. Hardwick, F. Bardé and P. G. Bruce, The Lithium–Oxygen Battery with Ether-Based Electrolytes, Angew. Chem., Int. Ed., 2011, 50, 8609–8613 CrossRef CAS PubMed.
- W. Xu, K. Xu, V. V. Viswanathan, S. A. Towne, J. S. Hardy, J. Xiao, Z. Nie, D. Hu, D. Wang and J. G. Zhang, Reaction mechanisms for the limited reversibility of Li–O2 chemistry in organic carbonate electrolytes, J. Power Sources, 2011, 196, 9631–9639 CrossRef CAS.
- N. Mozhzhukhina, L. P. Méndez De Leo and E. J. Calvo, Infrared spectroscopy studies on stability of dimethyl sulfoxide for application in a Li-air battery, J. Phys. Chem. C, 2013, 117, 18375–18380 CrossRef CAS.
- J. P. Vivek, N. Berry, G. Papageorgiou, R. J. Nichols and L. J. Hardwick, Mechanistic Insight into the Superoxide Induced Ring Opening in Propylene Carbonate Based Electrolytes using in Situ Surface-Enhanced Infrared Spectroscopy, J. Am. Chem. Soc., 2016, 138, 3745–3751 CrossRef CAS.
- Q. Peng, Y. Qiao, K. Kannari, A. Ge, K. I. Inoue and S. Ye, In Situ Spectroscopic Investigations of Electrochemical Oxygen Reduction and Evolution Reactions in Cyclic Carbonate Electrolyte Solutions, J. Phys. Chem. C, 2020, 124, 15781–15792 CrossRef CAS.
- V. S. Bryantsev, V. Giordani, W. Walker, M. Blanco, S. Zecevic, K. Sasaki, J. Uddin, D. Addison and G. V. Chase, Predicting solvent stability in aprotic electrolyte Li-air batteries: Nucleophilic substitution by the superoxide anion radical (O2•-), J. Phys. Chem. A, 2011, 115, 12399–12409 CrossRef CAS PubMed.
- H. G. Jung, J. Hassoun, J. B. Park, Y. K. Sun and B. Scrosati, An improved high-performance lithium-air battery, Nat. Chem., 2012, 4, 579–585 CrossRef CAS.
- B. D. McCloskey, R. Scheffler, A. Speidel, D. S. Bethune, R. M. Shelby and A. C. Luntz, On the efficacy of electrocatalysis in nonaqueous Li-O2 batteries, J. Am. Chem. Soc., 2011, 133, 18038–18041 CrossRef CAS.
- B. Zhang, H. Zhang, H. Luo, H. Hua, X. Wu, Y. Chen, S. Zhou, J. Yin, K. Zhang, H. G. Liao, Q. Wang, Y. Zou, Y. Qiao and S. G. Sun, Manipulated Fluoro-Ether Derived Nucleophilic Decomposition Products for Mitigating Polarization-Induced Capacity Loss in Li-Rich Layered Cathode, Angew. Chem., Int. Ed., 2024, 63(6), e202316790 CrossRef CAS.
- A. Ge, R. Nagai, C. Xu, K. Kannari, B. Peng, K. I. Inoue, A. Morita and S. Ye, Unraveling the Unstable Nature of Tetraglyme-Based Electrolytes toward Superoxide and the Inhibitory Effect of Lithium Ions by Using In Situ Vibrational Spectroscopies, J. Phys. Chem. C, 2022, 126, 2980–2989 CrossRef CAS.
- X. Liang, Y. Liang, Y. Gao, W. Qiao, D. Yin, P. Huang, C. Wang, L. Wang and Y. Cheng, Electrolyte Engineering with TFA− Anion-Rich Solvation Structure to Construct Highly Stable Zn2+/Na+ Dual-Salt Batteries, Small, 2024, 20(49), 2408162 CrossRef CAS.
- H. Xiao, X. Li and Y. Fu, Advances in Anion Chemistry in the Electrolyte Design for Better Lithium Batteries, Nano-Micro Lett., 2025, 17(1), 1–27 CrossRef.
- R. Younesi, M. Hahlin, F. Björefors, P. Johansson and K. Edström, Li-O2 battery degradation by lithium peroxide (Li2O2): A model study, Chem. Mater., 2013, 25, 77–84 CrossRef CAS.
- N. Mahne, B. Schafzahl, C. Leypold, M. Leypold, S. Grumm, A. Leitgeb, G. A. Strohmeier, M. Wilkening, O. Fontaine, D. Kramer, C. Slugovc, S. M. Borisov and S. A. Freunberger, Singlet oxygen generation as a major cause for parasitic reactions during cycling of aprotic lithium–oxygen batteries, Nat. Energy, 2017, 2(5), 1–9 Search PubMed.
- Y. K. Petit, C. Leypold, N. Mahne, E. Mourad, L. Schafzahl, C. Slugovc, S. M. Borisov and S. A. Freunberger, DABCOnium: An Efficient and High-Voltage Stable Singlet Oxygen Quencher for Metal–O2 Cells, Angew. Chem., Int. Ed., 2019, 58, 6535–6539 CrossRef CAS.
- B. Wen, L. Zhou, Y. Wang and F. Li, Recent advances and perspectives of Li–O2 batteries, Chem. Commun., 2025, 61, 6095–6104 RSC.
- X. Gao, Y. Chen, L. Johnson and P. G. Bruce, Promoting solution phase discharge in Li–O2 batteries containing weakly solvating electrolyte solutions, Nat. Mater., 2016, 15(8), 882–888 CrossRef CAS.
- T. Zhang, N. Imanishi, S. Hasegawa, A. Hirano, J. Xie, Y. Takeda, O. Yamamoto and N. Sammes, Li/Polymer Electrolyte/Water Stable Lithium-Conducting Glass Ceramics Composite for Lithium–Air Secondary Batteries with an Aqueous Electrolyte, J. Electrochem. Soc., 2008, 155, A965 CrossRef CAS.
- S. Ko, Y. Yoo, J. Choi, H. D. Lim, C. B. Park and M. Lee, Discovery of organic catalysts boosting lithium carbonate decomposition toward ambient air operational lithium–air batteries, J. Mater. Chem. A, 2022, 10, 20464–20472 RSC.
- Z. Peng, S. A. Freunberger, Y. Chen and P. G. Bruce, A reversible and higher-rate Li-O2 battery, Science, 2012, 337, 563–566 CrossRef CAS PubMed.
- L. Johnson, C. Li, Z. Liu, Y. Chen, S. A. Freunberger, P. C. Ashok, B. B. Praveen, K. Dholakia, J. M. Tarascon and P. G. Bruce, The role of LiO2 solubility in O2 reduction in aprotic solvents and its consequences for Li-O2 batteries, Nat. Chem., 2014, 6, 1091–1099 CrossRef CAS PubMed.
- N. Mozhzhukhina, F. Marchini, W. R. Torres, A. Y. Tesio, L. P. Mendez De Leo, F. J. Williams and E. J. Calvo, Insights into dimethyl sulfoxide decomposition in Li-O2 battery: Understanding carbon dioxide evolution, Electrochem. Commun., 2017, 80, 16–19 CrossRef CAS.
- Z. Peng, S. A. Freunberger, Y. Chen and P. G. Bruce, A reversible and higher-rate Li-O2 battery, Science, 2012, 337, 563–566 CrossRef CAS PubMed.
- N. Mozhzhukhina, L. P. Méndez De Leo and E. J. Calvo, Infrared spectroscopy studies on stability of dimethyl sulfoxide for application in a Li-air battery, J. Phys. Chem. C, 2013, 117, 18375–18380 CrossRef CAS.
- H. Lee, D. J. Lee, J. N. Lee, J. Song, Y. Lee, M. H. Ryou, J. K. Park and Y. M. Lee, Chemical aspect of oxygen dissolved in a dimethyl sulfoxide-based electrolyte on lithium metal, Electrochim. Acta, 2014, 123, 419–425 CrossRef CAS.
- D. Xu, Z. L. Wang, L. L. Zhang and X. B. Zhang, Novel DMSO-based electrolyte for high performance rechargeable Li–O2 batteries, Chem. Commun., 2012, 48, 6948–6950 RSC.
- B. Liu, W. Xu, P. Yan, S. T. Kim, M. H. Engelhard, X. Sun, D. Mei, J. Cho, C. M. Wang and J. G. Zhang, Stabilization of Li Metal Anode in DMSO-Based Electrolytes via Optimization of Salt–Solvent Coordination for Li–O2 Batteries, Adv. Energy Mater., 2017, 7, 1602605 CrossRef.
- D. Xu, Z. L. Wang, J. J. Xu, L. L. Zhang, L. M. Wang and X. B. Zhang, A stable sulfone based electrolyte for high performance rechargeable Li–O2 batteries, Chem. Commun., 2012, 48, 11674–11676 RSC.
- T. Zhang, K. Liao, P. He and H. Zhou, A self-defense redox mediator for efficient lithium–O2 batteries, Energy Environ. Sci., 2016, 9, 1024–1030 RSC.
- C. O. Laoire, S. Mukerjee, K. M. Abraham, E. J. Plichta and M. A. Hendrickson, Influence of nonaqueous solvents on the electrochemistry of oxygen in the rechargeable lithium-air battery, J. Phys. Chem. C, 2010, 114, 9178–9186 CrossRef CAS.
- L. Johnson, C. Li, Z. Liu and Y. Chen, The role of LiO2 solubility in O2 reduction in aprotic solvents and its consequences for Li–O2 batteries, Nat. Chem., 2014, 6, 1091–1099 CrossRef CAS PubMed.
- H. G. Jung, J. Hassoun, J. B. Park, Y. K. Sun and B. Scrosati, An improved high-performance lithium-air battery, Nat. Chem., 2012, 4, 579–585 CrossRef CAS.
- Y. Yu, G. Huang, J. Y. Du, J. Z. Wang, Y. Wang, Z. J. Wu and X. B. Zhang, A renaissance of N,N-dimethylacetamide-based electrolytes to promote the cycling stability of Li–O2 batteries, Energy Environ. Sci., 2020, 13, 3075–3081 RSC.
- G. A. Elia, J. Hassoun, W. J. Kwak, Y. K. Sun, B. Scrosati, F. Mueller, D. Bresser, S. Passerini, P. Oberhumer, N. Tsiouvaras and J. Reiter, An advanced lithium-air battery exploiting an ionic liquid-based electrolyte, Nano Lett., 2014, 14, 6572–6577 CrossRef CAS.
- M. Hayyan, F. S. Mjalli, M. A. Hashim, I. M. Alnashef, S. M. Al-Zahrani and K. L. Chooi, Long term stability of superoxide ion in piperidinium, pyrrolidinium and phosphonium cations-based ionic liquids and its utilization in the destruction of chlorobenzenes, J. Electroanal. Chem., 2012, 664, 26–32 CrossRef CAS.
- F. Soavi, S. Monaco and M. Mastragostino, Catalyst-free porous carbon cathode and ionic liquid for high efficiency, rechargeable Li/O2 battery, J. Power Sources, 2013, 224, 115–119 CrossRef CAS.
- F. Mizuno, K. Takechi, S. Higashi, T. Shiga, T. Shiotsuki, N. Takazawa, Y. Sakurabayashi, S. Okazaki, I. Nitta, T. Kodama, H. Nakamoto, H. Nishikoori, S. Nakanishi, Y. Kotani and H. Iba, Cathode reaction mechanism of non-aqueous Li–O2 batteries with highly oxygen radical stable electrolyte solvent, J. Power Sources, 2013, 228, 47–56 CrossRef CAS.
- M. Hayyan, F. S. Mjalli, M. A. Hashim and I. M. Alnashef, An investigation of the reaction between 1-butyl-3-methylimidazolium trifluoromethanesulfonate and superoxide ion, J. Mol. Liq., 2013, 181, 44–50 CrossRef CAS.
- A. Khan and C. Zhao, Oxygen Reduction Reactions in Aprotic Ionic Liquids Based Mixed Electrolytes for High Performance of Li-O2 Batteries, ACS Sustain. Chem. Eng., 2016, 4, 506–513 CrossRef CAS.
- Y. Chen, S. A. Freunberger, Z. Peng, F. Bardé and P. G. Bruce, Li-O2 battery with a dimethylformamide electrolyte, J. Am. Chem. Soc., 2012, 134, 7952–7957 CrossRef CAS PubMed.
- B. Zhou, L. Guo, Y. Zhang, J. Wang, L. Ma, W. H. Zhang, Z. Fu and Z. Peng, A High-Performance Li–O2 Battery with a Strongly Solvating Hexamethylphosphoramide Electrolyte and a LiPON-Protected Lithium Anode, Adv. Mater., 2017, 29, 1701568 CrossRef.
- V. S. Bryantsev, V. Giordani, W. Walker, M. Blanco, S. Zecevic, K. Sasaki, J. Uddin, D. Addison and G. V. Chase, Predicting solvent stability in aprotic electrolyte Li-air batteries: Nucleophilic substitution by the superoxide anion radical (O2•-), J. Phys. Chem. A, 2011, 115, 12399–12409 CrossRef CAS PubMed.
- Z. Peng, S. A. Freunberger, L. J. Hardwick, Y. Chen, V. Giordani, F. Bardé, P. Novák, D. Graham, J. M. Tarascon and P. G. Bruce, Oxygen Reactions in a Non-Aqueous Li+ Electrolyte, Angew. Chem., Int. Ed., 2011, 50, 6351–6355 CrossRef CAS PubMed.
- B. D. McCloskey, D. S. Bethune, R. M. Shelby, T. Mori, R. Scheffler, A. Speidel, M. Sherwood and A. C. Luntz, Limitations in rechargeability of li-o 2 batteries and possible origins, J. Phys. Chem. Lett., 2012, 3, 3043–3047 CrossRef CAS PubMed.
- R. C. McNulty, K. D. Jones, C. Holc, J. W. Jordan, P. G. Bruce, D. A. Walsh, G. N. Newton, H. W. Lam and L. R. Johnson, Hydroperoxide-Mediated Degradation of Acetonitrile in the Lithium–Air Battery, Adv. Energy Mater., 2023, 13, 2300579 CrossRef CAS.
- T. Zhang, N. Imanishi, S. Hasegawa, A. Hirano, J. Xie, Y. Takeda, O. Yamamoto and N. Sammes, Li/Polymer Electrolyte/Water Stable Lithium-Conducting Glass Ceramics Composite for Lithium–Air Secondary Batteries with an Aqueous Electrolyte, J. Electrochem. Soc., 2008, 155, A965 CrossRef CAS.
- T. Zhang, N. Imanishi, Y. Shimonishi, A. Hirano, J. Xie, Y. Takeda, O. Yamamoto and N. Sammes, Stability of a Water-Stable Lithium Metal Anode for a Lithium–Air Battery with Acetic Acid–Water Solutions, J. Electrochem. Soc., 2010, 157, A214 CrossRef CAS.
- M. Balaish, E. Peled, D. Golodnitsky and Y. Ein-Eli, Liquid-free lithium-oxygen batteries, Angew. Chem., Int. Ed., 2015, 54, 436–440 CrossRef CAS PubMed.
- J. C. Bachman, S. Muy, A. Grimaud, H. H. Chang, N. Pour, S. F. Lux, O. Paschos, F. Maglia, S. Lupart, P. Lamp, L. Giordano and Y. Shao-Horn, Inorganic Solid-State Electrolytes for Lithium Batteries: Mechanisms and Properties Governing Ion Conduction, Chem. Rev., 2016, 116, 140–162 CrossRef CAS.
- C. S. Yang, K. N. Gao, X. P. Zhang, Z. Sun and T. Zhang, Rechargeable solid-state Li-air batteries: a status report, Rare Met., 2018, 37, 459–472 CrossRef CAS.
- T. Zhang, N. Imanishi, Y. Shimonishi, A. Hirano, J. Xie, Y. Takeda, O. Yamamoto and N. Sammes, Stability of a Water-Stable Lithium Metal Anode for a Lithium–Air Battery with Acetic Acid–Water Solutions, J. Electrochem. Soc., 2010, 157, A214 CrossRef CAS.
- P. Chen, F. Bai, J. wen Deng, B. Liu and T. Zhang, Recent progresses and challenges in aqueous lithium–air batteries relating to the solid electrolyte separator: A mini-review, Front. Chem., 2022, 10, 1035691 CrossRef CAS PubMed.
- L. Zhu, Y. Wang, Y. Wu, W. Feng, Z. Liu, W. Tang, X. Wang and Y. Xia, Boron Nitride-Based Release Agent Coating Stabilizes Li1.3Al0.3Ti1.7(PO4)3/Li Interface with Superior Lean-Lithium Electrochemical Performance and Thermal Stability, Adv. Funct. Mater., 2022, 32, 2201136 CrossRef CAS.
- L. Zhu, Y. Wang, Y. Wu, W. Feng, Z. Liu, W. Tang, X. Wang and Y. Xia, Boron Nitride-Based Release Agent Coating Stabilizes Li1.3Al0.3Ti1.7(PO4)3/Li Interface with Superior Lean-Lithium Electrochemical Performance and Thermal Stability, Adv. Funct. Mater., 2022, 32, 2201136 CrossRef CAS.
- R. Khurana, J. L. Schaefer, L. A. Archer and G. W. Coates, Suppression of lithium dendrite growth using cross-linked polyethylene/poly(ethylene oxide) electrolytes: A new approach for practical lithium-metal polymer batteries, J. Am. Chem. Soc., 2014, 136, 7395–7402 CrossRef CAS PubMed.
- C. S. Yang, K. N. Gao, X. P. Zhang, Z. Sun and T. Zhang, Rechargeable solid-state Li-air batteries: a status report, Rare Met., 2018, 37, 459–472 CrossRef CAS.
- X. Liu, X. Xin, L. Shen, Z. Gu, J. Wu and X. Yao, Poly(methyl methacrylate)-Based Gel Polymer Electrolyte for High-Performance Solid State Li-O2Battery with Enhanced Cycling Stability, ACS Appl. Energy Mater., 2021, 4, 3975–3982 CrossRef CAS.
- A. Kondori, M. Esmaeilirad, A. M. Harzandi, R. Amine, M. T. Saray, L. Yu, T. Liu, J. Wen, N. Shan, H. H. Wang, A. T. Ngo, P. C. Redfern, C. S. Johnson, K. Amine, R. Shahbazian-Yassar, L. A. Curtiss and M. Asadi, A room temperature rechargeable Li2O-based lithium-air battery enabled by a solid electrolyte, Science, 2023, 379, 499–505 CrossRef CAS PubMed.
- E. Nasybulin, W. Xu, M. H. Engelhard, Z. Nie, S. D. Burton, L. Cosimbescu, M. E. Gross and J. G. Zhang, Effects of electrolyte salts on the performance of Li-O2 batteries, J. Phys. Chem. C, 2013, 117, 2635–2645 CrossRef CAS.
- R. Younesi, M. Hahlin, F. Björefors, P. Johansson and K. Edström, Li-O2 battery degradation by lithium peroxide (Li 2O2): A model study, Chem. Mater., 2013, 25, 77–84 CrossRef CAS.
- S. A. Rosy, M. Leskes and M. Noked, Bifunctional Role of LiNO3 in Li-O2 Batteries: Deconvoluting Surface and Catalytic Effects, ACS Appl. Mater. Interfaces, 2018, 10, 29622–29629 CrossRef CAS PubMed.
- B. Tong, J. Huang, Z. Zhou and Z. Peng, The Salt Matters: Enhanced Reversibility of Li–O2 Batteries with a Li[(CF3SO2)(n-C4F9SO2)N]-Based Electrolyte, Adv. Mater., 2018, 30, 1704841 CrossRef PubMed.
- K. He, C. Zu, Y. Wang, B. Han, X. Yin, H. Zhao, Y. Liu and J. Chen, Stability of lithium ion conductor NASICON structure glass ceramic in acid and alkaline aqueous solution, Solid State Ionics, 2014, 254, 78–81 CrossRef CAS.
- E. Song, J. Moon, J. Y. Lee, C. O. Lee, W. S. Chi and J. T. Park, High-voltage solar energy conversion based on ZIF-67-derived binary redox-quasi-solid-state electrolyte, J. Electroanal. Chem., 2021, 893, 115264 CrossRef CAS.
- D. Lin, Y. Liu and Y. Cui, Reviving the lithium metal anode for high-energy batteries, Nat. Nanotechnol., 2017, 12, 194–206 CrossRef CAS PubMed.
- M. M. Markowitz and D. A. Boryta, Lithium Metal-Gas Reactions. Interaction of Lithium Metal with Air and its Component Gases, J. Chem. Eng. Data, 1962, 7, 586–591 CrossRef CAS.
- K. R. Zavadil and N. R. Armstrong, Surface chemistries of lithium: Detailed characterization of the reactions with O2 and H2O using XPS, EELS, AND microgravimetry, Surf. Sci., 1990, 230, 47–60 CrossRef CAS.
- K. Wang, P. N. Ross, F. Kong and F. McLarnon, The Reaction of Clean Li Surfaces with Small Molecules in Ultrahigh Vacuum: I. Dioxygen, J. Electrochem. Soc., 1996, 143, 422 CrossRef CAS.
- Y. Li, Y. Li, Y. Sun, B. Butz, K. Yan, A. L. Koh, J. Zhao, A. Pei and Y. Cui, Revealing nanoscale passivation and corrosion mechanisms of reactive battery materials in gas environments, Nano Lett., 2017, 17, 5171–5178 CrossRef CAS PubMed.
- X. Wu, Z. Li, C. Song, L. Chen, P. Dai, P. Zhang, Y. Qiao, L. Huang and S. G. Sun, Regulating the Architecture of a Solid Electrolyte Interface on a Li-Metal Anode of a Li-O2 Battery by a Dithiobiuret Additive, ACS Mater. Lett., 2022, 4, 682–691 CrossRef CAS.
- A. Etxebarria, S. L. Koch, O. Bondarchuk, S. Passerini, G. Teobaldi and M. Á. Muñoz-Márquez, Work Function Evolution in Li Anode Processing, Adv. Energy Mater., 2020, 10, 2000520 CrossRef CAS.
- J. Christensen, P. Albertus, R. S. Sanchez-Carrera, T. Lohmann, B. Kozinsky, R. Liedtke, J. Ahmed and A. Kojic, A Critical Review of Li/Air Batteries, J. Electrochem. Soc., 2011, 159, R1–R30 CrossRef.
- J. Wu, L. Yuan, Z. Li, X. Xie and Y. Huang, Air-stable means more: designing air-defendable lithium metals for safe and stable batteries, Mater. Horiz., 2020, 7, 2619–2634 RSC.
- M. M. Ottakam Thotiyl, S. A. Freunberger, Z. Peng and P. G. Bruce, The carbon electrode in nonaqueous Li-O2 cells, J. Am. Chem. Soc., 2013, 135, 494–500 CrossRef CAS PubMed.
- T. Wang, X. Pan, J. Chen and Y. Chen, Critical CO2Concentration for Practical Lithium-Air Batteries, J. Phys. Chem. Lett., 2021, 12, 4799–4804 CrossRef CAS.
- A. Etxebarria, A. Etxebarria, D. J. Yun, D. J. Yun, M. Blum, M. Blum, Y. Ye, Y. Ye, Y. Ye, M. Sun, K. J. Lee, K. J. Lee, H. Su, H. Su, M. Á. Muñoz-Márquez, P. N. Ross, E. J. Crumlin and E. J. Crumlin, Revealing in Situ Li Metal Anode Surface Evolution upon Exposure to CO2 Using Ambient Pressure X-Ray Photoelectron Spectroscopy, ACS Appl. Mater. Interfaces, 2020, 12, 26607–26613 CrossRef CAS PubMed.
- K. Chen, G. Huang, J. L. Ma, J. Wang, D. Y. Yang, X. Y. Yang, Y. Yu and X. B. Zhang, The Stabilization Effect of CO2 in Lithium–Oxygen/CO2 Batteries, Angew. Chem., Int. Ed., 2020, 59, 16661–16667 CrossRef CAS PubMed.
- S. Pamangadan C., S. Patra and E. Perumal, CO2-tolerant CuFe2O4 as Bifunctional Electrocatalyst for Transition from Rechargeable Li-O2 to Li-CO2 Batteries, J. Electrochem. Soc., 2024, 171, 090511 CrossRef.
- B. Liu, Y. Sun, L. Liu, J. Chen, B. Yang, S. Xu and X. Yan, Recent advances in understanding Li-CO2 electrochemistry, Energy Environ. Sci., 2019, 12, 887–922 RSC.
- C. C. Addison, B. M. Davies, E. F. Mcfarlane, F. C. Tomkins and T. Faruduy, Reaction of nitrogen with stirred and unstirred liquid lithium, J. Chem. Soc. Inorg. Phys. Theor., 1969, 1822–1827 RSC.
- Y. Zhang, W. Lv, Z. Huang, G. Zhou, Y. Deng, J. Zhang, C. Zhang, B. Hao, Q. Qi, Y. B. He, F. Kang and Q. H. Yang, An air-stable and waterproof lithium metal anode enabled by wax composite packaging, Sci. Bull., 2019, 64, 910–917 CrossRef CAS.
- M. H. Cho, J. Trottier, C. Gagnon, P. Hovington, D. Clément, A. Vijh, C. S. Kim, A. Guerfi, R. Black, L. Nazar and K. Zaghib, The effects of moisture contamination in the Li-O2 battery, J. Power Sources, 2014, 268, 565–574 CrossRef CAS.
- X. Yao, Q. Dong, Q. Cheng and D. Wang, Why do lithium–oxygen batteries fail: parasitic chemical reactions and their synergistic effect, Angew. Chem., Int. Ed., 2016, 55, 11344–11353 CrossRef CAS PubMed.
- H. D. Lim, B. Lee, Y. Zheng, J. Hong, J. Kim, H. Gwon, Y. Ko, M. Lee, K. Cho and K. Kang, Rational design of redox mediators for advanced Li-O2 batteries, Nat. Energy, 2016, 1, 1–9 Search PubMed.
- D. Kundu, R. Black, B. Adams and L. F. Nazar, A highly active low voltage redox mediator for enhanced rechargeability of lithium-oxygen batteries, ACS Cent. Sci., 2015, 1, 510–515 CrossRef CAS PubMed.
- Y. Chen, S. A. Freunberger, Z. Peng, O. Fontaine and P. G. Bruce, Charging a Li–O2 battery using a redox mediator, Nat. Chem., 2013, 5, 489–494 CrossRef CAS PubMed.
- D. Wang, X. Mu, P. He and H. Zhou, Materials for advanced Li-O2 batteries: Explorations, challenges and prospects, Mater. Today, 2019, 26, 87–99 CrossRef CAS.
- J. Park, H. R. Yoon, M. A. Khan, S. Cho and M. M. Sung, Selective Infiltration in Polymer Hybrid Thin Films as a Gas-Encapsulation Layer for Stretchable Electronics, ACS Appl. Mater. Interfaces, 2020, 12, 8817–8825 CrossRef CAS PubMed.
- S. Wang, X. Li, H. Wu, Z. Tian and Q. Xin, Advances in high permeability polymer-based membrane materials for CO2 separations, Energy Environ. Sci., 2016, 9, 1863–1890 RSC.
- S. Gadipelli and Z. X. Guo, Graphene-based materials: Synthesis and gas sorption, storage and separation, Prog. Mater. Sci., 2015, 69, 1–60 CrossRef CAS.
- Y. J. Kim, H. S. Jin, D. H. Lee, J. Choi, W. Jo, H. Noh, J. Lee, H. Chu, H. Kwack, F. Ye, H. Lee, M. H. Ryou and H. T. Kim, Guided Lithium Deposition by Surface Micro-Patterning of Lithium-Metal Electrodes, ChemElectroChem, 2018, 5, 3169–3175 CrossRef CAS.
- G. Zheng, S. W. Lee, Z. Liang, H. W. Lee, K. Yan, H. Yao, H. Wang, W. Li, S. Chu and Y. Cui, Interconnected hollow carbon nanospheres for stable lithium metal anodes, Nat. Nanotechnol., 2014, 9(8), 618–623 CrossRef CAS PubMed.
- N. Li, K. Zhang, K. Xie, W. Wei and Y. Gao, Reduced-graphene-oxide-guided directional growth of planar lithium layers, Adv. Mater., 2020, 32, 1907079 CrossRef CAS PubMed.
- Y. Zhao, G. Li, Y. Gao, D. Wang, Q. Huang and D. Wang, Stable Li Metal Anode by a Hybrid Lithium Polysulfidophosphate/Polymer Cross-Linking Film, ACS Energy Lett., 2019, 4, 1271–1278 CrossRef CAS.
- X. Liang, Q. Pang, I. R. Kochetkov, M. S. Sempere, H. Huang, X. Sun and L. F. Nazar, A facile surface chemistry route to a stabilized lithium metal anode, Nat. Energy, 2017, 2, 1–7 Search PubMed.
- Z. Takehara, Z. Ogumi, Y. Uchimoto, K. Yasuda and H. Yoshida, Modification of lithium/electrolyte interface by plasma polymerization of 1,1-difluoroethene, J. Power Sources, 1993, 44(1–3), 377–383 CrossRef CAS.
- C. Yan, R. Xu, Y. Xiao, J. F. Ding, L. Xu, B. Q. Li and J. Q. Huang, Toward Critical Electrode/Electrolyte Interfaces in Rechargeable Batteries, Adv. Funct. Mater., 2020, 30, 1909887 CrossRef CAS.
- L. Fan, H. Zhuang and L. Gao, Regulating Li deposition at artificial solid electrolyte interphases, J. Mater. Chem. A, 2017, 5, 3483–3492 RSC.
- S. Stalin, P. Chen, G. Li, Y. Deng and Z. Rouse, et al., Ultrathin zwitterionic polymeric interphases for stable lithium metal anodes, Matter, 2021, 4(11), 3753–3773 CrossRef CAS.
- K. Chen, R. Pathak, A. Gurung, E. A. Adhamash, B. Bahrami, Q. He, H. Qiao, A. L. Smirnova, J. J. Wu, Q. Qiao and Y. Zhou, Flower-shaped lithium nitride as a protective layer via facile plasma activation for stable lithium metal anodes, Energy Storage Mater., 2019, 18, 389–396 CrossRef.
- J. Zheng, R. Yang, L. Xie, Q. Jianglan, L. Yang and L. Xingguo, Plasma-assisted approaches in inorganic nanostructure fabrication, Adv. Mater., 2010, 22, 1451–1473 CrossRef CAS PubMed.
- X. Wei, X. Wang, Q. An, C. Han and L. Mai, Operando X-ray Diffraction Characterization for Understanding the Intrinsic Electrochemical Mechanism in Rechargeable Battery Materials, Small Methods, 2017, 1, 1700083 CrossRef.
- L. Dong, L. Nie and W. Liu, Water-Stable Lithium Metal Anodes with Ultrahigh-Rate Capability Enabled by a Hydrophobic Graphene Architecture, Adv. Mater., 2020, 32, 1908494 CrossRef CAS.
- J.-L. Ma, F.-L. Meng, Y. Yu, D.-P. Liu, J.-M. Yan, Y. Zhang, X.-B. Zhang and Q. Jiang, Prevention of dendrite growth and volume expansion to give high-performance aprotic bimetallic Li-Na alloy–O2 batteries, Nat. Chem., 2019, 11, 64–70 CrossRef CAS PubMed.
- J. J. Xu, Z. W. Chang, Y. Wang, D. P. Liu, Y. Zhang and X. B. Zhang, Cathode Surface-Induced, Solvation-Mediated, Micrometer-Sized Li2O2 Cycling for Li–O2 Batteries, Adv. Mater., 2016, 28, 9620–9628 CrossRef CAS PubMed.
- Q. C. Liu, J. J. Xu, Z. W. Chang and X. B. Zhang, Direct electrodeposition of cobalt oxide nanosheets on carbon paper as free-standing cathode for Li–O2 battery, J. Mater. Chem. A, 2014, 2, 6081–6085 RSC.
- Q. Liu, Y. Jiang, J. Xu, D. Xu, Z. Chang, Y. Yin, W. Liu and X. Zhang, Hierarchical Co3O4 porous nanowires as an efficient bifunctional cathode catalyst for long life Li-O2 batteries, Nano Res., 2015, 8, 576–583 CrossRef CAS.
- Z. L. Wang, D. Xu, J. J. Xu, L. L. Zhang and X. B. Zhang, Graphene oxide gel-derived, free-standing, hierarchically porous carbon for high-capacity and high-rate rechargeable Li-O2 batteries, Adv. Funct. Mater., 2012, 22, 3699–3705 Search PubMed.
- X. Y. Yang, J. J. Xu, Z. W. Chang, D. Bao, Y. Bin Yin, T. Liu, J. M. Yan, D. P. Liu, Y. Zhang and X. B. Zhang, Blood-Capillary-Inspired, Free-Standing, Flexible, and Low-Cost Super-Hydrophobic N-CNTs@SS Cathodes for High-Capacity, High-Rate, and Stable Li-Air Batteries, Adv. Energy Mater., 2018, 8, 1702242 Search PubMed.
- H. Ge, W. Shi, J. Liu, Y. Zhang and X. Wang, High Entropy Oxide-Polyoxometalate Sub-1 nm Hetero-Nanowires as Cathode Catalysts in Li-O2 Batteries, J. Am. Chem. Soc., 2025, 147(10), 8367–8376 CrossRef CAS PubMed.
- S. Karunarathne, C. Sandaruwan, Y. Y. Kannangara, D. Demko, F. Orange, A. Mija, A. R. Kami and A. M. Abdelkader, Triphase Heterogeneous Electrocatalysts of Ni and Co for High-Performing Li-O2 Batteries, EcoMat, 2025, 7(3), e70002 CrossRef CAS.
- D. Zhang, G. Zhang, R. Liu, R. Yang, X. Li, X. Zhang, H. Yu, P. Zhang, B. W. Li, H. Hou, Z. Guo and F. Dang, Mutually Activated 2D Ti0.87O2/MXene Monolayers Through Electronic Compensation Effect as Highly Efficient Cathode Catalysts of Li–O2 Batteries, Adv. Funct. Mater., 2024, 35, 2414679 CrossRef.
- H. Zhang, Y. Yu, D. Yang, Y. Fan, H. Chen, N. Zhang, Z. Wang, G. Huang and X. Zhang, Multifunctional Quasi-Homogeneous Catalysts as a New Catalytic Strategy to Boost the Performance of Li-O2 Batteries, Adv. Mater., 2024, 37(7), 2413948 CrossRef PubMed.
- W. Zhao, J. Wang, R. Yin, B. Li, X. Huang, L. Zhao and L. Qian, Single-atom Pt supported on holey ultrathin g-C3N4 nanosheets as efficient catalyst for Li-O2 batteries, J. Colloid Interface Sci., 2020, 564, 28–36 CrossRef CAS PubMed.
- J. Zheng, W. Zhang, R. Wang, J. Wang, Y. Zhai and X. Liu, Single-Atom Pd-N4 Catalysis for Stable Low-Overpotential Lithium-Oxygen Battery, Small, 2023, 19, 2204559 CrossRef CAS PubMed.
- X. Hu, G. Luo, Q. Zhao, D. Wu, T. Yang, J. Wen, R. Wang, C. Xu and N. Hu, Ru single atoms on N-doped carbon by spatial confinement and ionic substitution strategies for high-performance Li−O2 batteries, J. Am. Chem. Soc., 2020, 142, 16776–16786 CrossRef CAS PubMed.
- E. Zhang, A. Dong, K. Yin, C. Ye, Y. Zhou, C. Tan, M. Li, X. Zheng, Y. Wang, X. Gao, H. Li, D. Wang and S. Guo, Electron Localization in Rationally Designed Pt1Pd Single-Atom Alloy Catalyst Enables High-Performance Li-O2 Batteries, J. Am. Chem. Soc., 2024, 146, 2339–2344 CrossRef CAS PubMed.
- L. Li, M. Hua, J. Li, P. Zhang, Y. Nie, P. Wang, X. Lin, Z. Zhang, R. Wang, X. Ge, Y. C. Li and L. Yin, Tuning Dual Catalytic Active Sites of Pt Single Atoms Paired with High-Entropy Alloy Nanoparticles for Advanced Li-O2 Batteries, ACS Nano, 2025, 19, 4402 Search PubMed.
- F. Zhang, Z. Hu, J. Lai, N. Chen and Y. Liu, et al., Spin-polarized d-orbital filling in cobalt catalysts boosts solution-mediated Li-O2 batteries, Natl. Sci. Rev., 2025, 12(6), nwaf145 CrossRef CAS.
- H. Chen, J. Cao, D. Zhao and F. Niu, Photocathode Materials for Li–O2 Batteries: Progresses and Perspectives, Adv. Energy Mater., 2025, 15(21), 2500250 CrossRef CAS.
- V. Sankar Devi and P. Elumalai, Selenium heteroatom-doped mesoporous carbon as an efficient air-breathing electrode for rechargeable lithium-oxygen batteries, New J. Chem., 2022, 47, 2189–2201 RSC.
- E. Duraisamy, A. Prasath, V. Sankar Devi, M. N. M. Ansari and P. Elumalai, Sustainably-derived hierarchical porous carbon from spent honeycomb for high-performance lithium-ion battery and ultracapacitors, Energy Storage, 2020, 2(4), e136 CrossRef CAS.
- M. Athika and P. Elumalai, Porous Carbon Networks Decorated with Cobalt on CoFe2O2 as an Air-Breathing Electrode for High-Capacity Rechargeable Lithium-Air Batteries: Role of Metallic Cobalt Nanoparticles, ChemElectroChem, 2020, 7, 4188–4200 Search PubMed.
- S. Sandhiya and P. Elumalai, Compositionally engineered NiCoLDH@rGO as bifunctional cathode catalyst for rechargeable Li-O2/Li-CO2 battery, Electrochim. Acta, 2024, 487, 144195 Search PubMed.
- S. Sandhiya and P. Elumalai, Enhanced active sites using LDH nanosheets@rGO as bifunctional cathode catalyst for high-capacity rechargeable Li-O2 battery, Surf. Interfaces, 2025, 60, 106068 CrossRef CAS.
- V. Sankar Devi and P. Elumalai, Selenium heteroatom-doped mesoporous carbon as an efficient air-breathing electrode for rechargeable lithium–oxygen batteries, New J. Chem., 2023, 47, 2189–2201 RSC.
- V. S. Devi, M. Athika and P. Elumalai, Vacancy-induced LaMnO3 Perovskite as Bifunctional Air-breathing Electrode for Rechargeable Lithium-Air Battery, ChemistrySelect, 2022, 7, e202202554 CrossRef CAS.
- M. Athika, V. S. Devi and P. Elumalai, Cauliflower-Like Hierarchical Porous Nickel/Nickel Ferrite/Carbon Composite as Superior Bifunctional Catalyst for Lithium-Air Battery, ChemistrySelect, 2020, 5, 3529–3538 Search PubMed.
- M. Athika, P. M. Junais and P. Elumalai, Boosting the Bifunctional Oxygen Electrode Activity of Nitrogen-decorated Carbon Networks via Ni/NiO Addition for Li-Air and Zn-Air Batteries, J. Electrochem. Soc., 2023, 170, 030544 CrossRef CAS.
- Z. Guo, Q. Zhang, C. Wang, Y. Zhang, S. Dong and G. Cui, I-containing Polymer/Alloy Layer-Based Li Anode Mediating High-Performance Lithium–Air Batteries, Adv. Funct. Mater., 2022, 32, 2108993 CrossRef CAS.
- D. Du, Z. Zhu, K. Y. Chan, F. Li and J. Chen, Photoelectrochemistry of oxygen in rechargeable Li–O2 batteries, Chem. Soc. Rev., 2022, 51, 1846–1860 RSC.
- D. Aurbach, B. D. McCloskey, L. F. Nazar and P. G. Bruce, Advances in understanding mechanisms underpinning lithium-air batteries, Nat. Energy, 2016, 1, 1–11 Search PubMed.
- S. Soga, F. Bai, T. Zhang, K. Kakimoto, D. Mori, S. Taminato, Y. Takeda, O. Yamamoto and N. Imanishi, Ambient Air Operation Rechargeable Lithium-Air Battery with Acetic Acid Catholyte, J. Electrochem. Soc., 2020, 167, 090522 CrossRef CAS.
- L. Ma, T. Yu, E. Tzoganakis, K. Amine, T. Wu, Z. Chen and J. Lu, Fundamental Understanding and Material Challenges in Rechargeable Nonaqueous Li–O2 Batteries: Recent Progress and Perspective, Adv. Energy Mater., 2018, 8, 1800348 Search PubMed.
- J. P. O. Júlio, B. A. B. Francisco, B. P. de Sousa, J. F. Leal Silva, C. G. Anchieta, T. C. M. Nepel, C. B. Rodella, R. Maciel Filho and G. Doubek, Effect of O2 flow in discharge products and performance of Li-O2 batteries, Chem. Eng. J. Adv., 2022, 10, 100271 CrossRef.
- P. Zhang, M. Ding, X. Li, C. Li, Z. Li and L. Yin, Challenges and Strategy on Parasitic Reaction for High-Performance Nonaqueous Lithium–Oxygen Batteries, Adv. Energy Mater., 2020, 10, 2001789 CrossRef CAS.
- J. Zhang, W. Xu and W. Liu, Oxygen-selective immobilized liquid membranes for operation of lithium-air batteries in ambient air, J. Power Sources, 2010, 195, 7438–7444 CrossRef CAS.
- J. G. Zhang, D. Wang, W. Xu, J. Xiao and R. E. Williford, Ambient operation of Li/Air batteries, J. Power Sources, 2010, 195, 4332–4337 Search PubMed.
- Y. Ruan, J. Sun, S. Song, L. Yu, B. Chen, W. Li and X. Qin, A perfluorocarbon–silicone oil oxygen–selective membrane for ambient operation of aprotic Li–air batteries, Electrochem. Commun., 2018, 96, 93–97 CrossRef CAS.
- J. Amici, C. Francia, J. Zeng, S. Bodoardo and N. Penazzi, Protective PVDF-HFP-based membranes for air de-hydration at the cathode of the rechargeable Li–air cell, J. Appl. Electrochem., 2016, 46, 617–626 CrossRef CAS.
- M. Xie, Z. Huang, X. Lin, Y. Li, Z. Huang, L. Yuan, Y. Shen and Y. Huang, Oxygen selective membrane based on perfluoropolyether for Li-Air battery with long cycle life, Energy Storage Mater., 2019, 20, 307–314 CrossRef.
- L. H. Xu, S. H. Li, H. Mao, Y. Li, A. S. Zhang, S. Wang, W. M. Liu, J. Lv, T. Wang, W. W. Cai, L. Sang, W. W. Xie, C. Pei, Z. Z. Li, Y. N. Feng and Z. P. Zhao, Highly flexible and superhydrophobic MOF nanosheet membrane for ultrafast alcohol-water separation, Science, 2022, 378, 308–312 CrossRef CAS PubMed.
- X. Zou, K. Liao, D. Wang, Q. Lu, C. Zhou, P. He, R. Ran, W. Zhou, W. Jin and Z. Shao, Water-proof, electrolyte-nonvolatile, and flexible Li-Air batteries via O2-Permeable silica-aerogel-reinforced polydimethylsiloxane external membranes, Energy Storage Mater., 2020, 27, 297–306 CrossRef.
- H. C. Lee, J. O. Park, M. Kim, H. J. Kwon, J. H. Kim, K. H. Choi, K. Kim and D. Im, High-Energy-Density Li-O2 Battery at Cell Scale with Folded Cell Structure, Joule, 2019, 3, 542–556 CrossRef CAS.
- J. J. Xu, Q. C. Liu, Y. Yu, J. Wang, J. M. Yan and X. B. Zhang, In Situ Construction of Stable Tissue-Directed/Reinforced Bifunctional Separator/Protection Film on Lithium Anode for Lithium–Oxygen Batteries, Adv. Mater., 2017, 29, 1606552 CrossRef PubMed.
- X. Zeng, L. Leng, F. Liu, G. Wang, Y. Dong, L. Du, L. Liu and S. Liao, Enhanced Li-O2 battery performance, using graphene-like nori-derived carbon as the cathode and adding LiI in the electrolyte as a promoter, Electrochim. Acta, 2016, 200, 231–238 CrossRef CAS.
- C. K. Lee and Y. J. Park, CsI as Multifunctional Redox Mediator for Enhanced Li-Air Batteries, ACS Appl. Mater. Interfaces, 2016, 8, 8561–8567 CrossRef CAS PubMed.
- G. Ahmed, F. Butt, F. Raza, S. Hashmi, G. Gnana Kumar and M. Christy, The study of different redox mediators for competent Li–air batteries, J. Power Sources, 2022, 200, 231–238 Search PubMed.
- X. Gao, Y. Chen, L. Johnson and P. G. Bruce, Promoting solution phase discharge in Li–O2 batteries containing weakly solvating electrolyte solutions, Nat. Mater., 2016, 15(8), 882–888 CrossRef CAS PubMed.
- G. Leverick, M. Tułodziecki, R. Tatara, F. Bardé and Y. Shao-Horn, Solvent-Dependent Oxidizing Power of LiI Redox Couples for Li-O2 Batteries, Joule, 2019, 3, 1106–1126 CrossRef CAS.
- B. J. Bergner, A. Schürmann, K. Peppler, A. Garsuch and J. Janek, TEMPO: A mobile catalyst for rechargeable Li-O2 batteries, J. Am. Chem. Soc., 2014, 136, 15054–15064 CrossRef CAS PubMed.
- X. Gao, Y. Chen, L. Johnson and P. G. Bruce, Promoting solution phase discharge in Li–O2 batteries containing weakly solvating electrolyte solutions, Nat. Mater., 2016, 15(8), 882–888 CrossRef CAS PubMed.
- Y. Chen, S. A. Freunberger, Z. Peng, O. Fontaine and P. G. Bruce, Charging a Li-O2 battery using a redox mediator, Nat. Chem., 2013, 5, 489–494 CrossRef CAS PubMed.
- D. Kundu, R. Black, B. Adams and L. F. Nazar, A highly active low voltage redox mediator for enhanced rechargeability of lithium-oxygen batteries, ACS Cent. Sci., 2015, 1, 510–515 CrossRef CAS.
- Z. Liang and Y.-C. Lu, Critical role of redox mediator in suppressing charging instabilities of lithium–oxygen batteries, J. Am. Chem. Soc., 2016, 138, 7574–7583 CrossRef CAS PubMed.
- J. P. Vivek, N. Berry, G. Papageorgiou, R. J. Nichols and L. J. Hardwick, Mechanistic Insight into the Superoxide Induced Ring Opening in Propylene Carbonate Based Electrolytes using in Situ Surface-Enhanced Infrared Spectroscopy, J. Am. Chem. Soc., 2016, 138, 3745–3751 CrossRef CAS PubMed.
- Y. S. Hong, C. Z. Zhao, Y. Xiao, R. Xu, J. J. Xu, J. Q. Huang, Q. Zhang, X. Yu and H. Li, Safe Lithium-Metal Anodes for Li−O2 Batteries: From Fundamental Chemistry to Advanced Characterization and Effective Protection, Batteries Supercaps, 2019, 2, 638–658 CrossRef CAS.
- B. J. Bergner, A. Schürmann, K. Peppler, A. Garsuch and J. Janek, TEMPO: A Mobile Catalyst for Rechargeable Li-O2 Batteries, J. Am. Chem. Soc., 2014, 136, 15054–15064 CrossRef CAS PubMed.
- B. J. Bergner, C. Hofmann, A. Schü, D. Schrö, K. Peppler, P. R. Schreiner and J. Rgen Janek, Understanding the fundamentals of redox mediators in Li–O 2 batteries: a case study on nitroxides, Phys. Chem. Chem. Phys., 2015, 17, 31769 RSC.
- D. Sun, Y. Shen, W. Zhang, L. Yu, Z. Yi, W. Yin, D. Wang, Y. Huang, J. Wang, D. Wang and J. B. Goodenough, A solution-phase bifunctional catalyst for lithium–oxygen batteries, J. Am. Chem. Soc., 2014, 136, 8941–8946 CrossRef CAS PubMed.
- Y. Ko, H. Park, K. Lee, S. J. Kim, H. Park, Y. Bae, J. Kim, S. Y. Park, J. E. Kwon and K. Kang, Anchored Mediator Enabling Shuttle-Free Redox Mediation in Lithium-Oxygen Batteries, Angew. Chem., Int. Ed., 2020, 59, 5376–5380 CrossRef CAS PubMed.
- Q. Xiong, G. Huang and X. B. Zhang, High-Capacity and Stable Li-O2 Batteries Enabled by a Trifunctional Soluble Redox Mediator, Angew. Chem., Int. Ed., 2020, 59, 19311–19319 CrossRef CAS PubMed.
- J. J. Xu and X. B. Zhang, Li–air batteries: Decouple to stabilize, Nat. Energy, 2017, 2, 1–2 Search PubMed.
- L. Johnson, C. Li, Z. Liu, Y. Chen, S. A. Freunberger, P. C. Ashok, B. B. Praveen, K. Dholakia, J.-M. Tarascon and P. G. Bruce, The role of LiO2 solubility in O2 reduction in aprotic solvents and its consequences for Li–O2 batteries, Nat. Chem., 2014, 6, 1091–1099 CrossRef CAS PubMed.
- D. Sharon, D. Hirsberg, M. Salama, M. Afri, A. A. Frimer, M. Noked, W. Kwak, Y. K. Sun and D. Aurbach, Mechanistic Role of Li+ Dissociation Level in Aprotic Li-O2 Battery, ACS Appl. Mater. Interfaces, 2016, 8, 5300–5307 CrossRef CAS PubMed.
- J. Uddin, V. S. Bryantsev, V. Giordani, W. Walker, G. V. Chase and D. Addison, Lithium nitrate as regenerable SEI stabilizing agent for rechargeable Li/O2 batteries, J. Phys. Chem. Lett., 2013, 4, 3760–3765 CrossRef CAS.
- S. A. Rosy, M. Leskes and M. Noked, Bifunctional Role of LiNO3 in Li-O2 Batteries: Deconvoluting Surface and Catalytic Effects, ACS Appl. Mater. Interfaces, 2018, 10, 29622–29629 CrossRef CAS PubMed.
- X. D. Lin, Y. Gu, X. R. Shen, W. W. Wang, Y. H. Hong, Q. H. Wu, Z. Y. Zhou, D. Y. Wu, J. K. Chang, M. Sen Zheng, B. W. Mao and Q. F. Dong, An oxygen-blocking oriented multifunctional solid-electrolyte interphase as a protective layer for a lithium metal anode in lithium-oxygen batteries, Energy Environ. Sci., 2021, 14, 1439–1448 RSC.
- Y. Qiao, S. Wu, J. Yi, Y. Sun, S. Guo, S. Yang, P. He and H. Zhou, From O2− to HO2− : Reducing By-Products and Overpotential in Li-O2 Batteries by Water Addition, Angew. Chem., 2017, 129, 5042–5046 CrossRef.
- N. B. Aetukuri, B. D. McCloskey, J. M. Garciá, L. E. Krupp, V. Viswanathan and A. C. Luntz, Solvating additives drive solution-mediated electrochemistry and enhance toroid growth in non-aqueous Li-O2 batteries, Nat. Chem., 2015, 7, 50–56 CrossRef CAS.
- S. Meini, N. Tsiouvaras, K. U. Schwenke, M. Piana, H. Beyer, L. Lange and H. A. Gasteiger, Rechargeability of Li-air cathodes pre-filled with discharge products using an ether-based electrolyte solution: Implications for cycle-life of Li-air cells, Phys. Chem. Chem. Phys., 2013, 15, 11478–11493 RSC.
- Y. Chen, S. A. Freunberger, Z. Peng, O. Fontaine and P. G. Bruce, Charging a Li-O2 battery using a redox mediator, Nat. Chem., 2013, 5, 489–494 CrossRef CAS PubMed.
- B. J. Bergner, A. Schürmann, K. Peppler, A. Garsuch and J. Janek, TEMPO: A mobile catalyst for rechargeable Li-O2 batteries, J. Am. Chem. Soc., 2014, 136, 15054–15064 CrossRef CAS PubMed.
- D. Kundu, R. Black, B. Adams and L. F. Nazar, A highly active low voltage redox mediator for enhanced rechargeability of lithium-oxygen batteries, ACS Cent. Sci., 2015, 1, 510–515 CrossRef CAS PubMed.
- D. Sun, Y. Shen, W. Zhang, L. Yu, Z. Yi, W. Yin, D. Wang, Y. Huang, J. Wang, D. Wang and J. B. Goodenough, A solution-phase bifunctional catalyst for lithium-oxygen batteries, J. Am. Chem. Soc., 2014, 136, 8941–8946 CrossRef CAS PubMed.
- W. J. Kwak, D. Hirshberg, D. Sharon, H. J. Shin, M. Afri, J. B. Park, A. Garsuch, F. F. Chesneau, A. A. Frimer, D. Aurbach and Y. K. Sun, Understanding the behavior of Li-oxygen cells containing LiI, J. Mater. Chem. A, 2015, 3, 8855–8864 RSC.
- J. S. Lee, C. Lee, J. Y. Lee, J. Ryu and W. H. Ryu, Polyoxometalate as a Nature-Inspired Bifunctional Catalyst for Lithium-Oxygen Batteries, ACS Catal., 2018, 8, 7213–7221 CrossRef CAS.
- H. D. Lim, H. Song, J. Kim, H. Gwon, Y. Bae, K. Y. Park, J. Hong, H. Kim, T. Kim, Y. H. Kim, X. Leprõ, R. Ovalle-Robles, R. H. Baughman and K. Kang, Superior rechargeability and efficiency of lithium-oxygen batteries: Hierarchical air electrode architecture combined with a soluble catalyst, Angew. Chem., Int. Ed., 2014, 53, 3926–3931 CrossRef CAS PubMed.
- W. H. Ryu, F. S. Gittleson, J. M. Thomsen, J. Li, M. J. Schwab, G. W. Brudvig and A. D. Taylor, Heme biomolecule as redox mediator and oxygen shuttle for efficient charging of lithium-oxygen batteries, Nat. Commun., 2016, 7, 12925 CrossRef CAS.
- W. J. Kwak, D. Hirshberg, D. Sharon, M. Afri, A. A. Frimer, H. G. Jung, D. Aurbach and Y. K. Sun, Li-O2 cells with LiBr as an electrolyte and a redox mediator, Energy Environ. Sci., 2016, 9, 2334–2345 RSC.
- J. B. Park, S. H. Lee, H. G. Jung, D. Aurbach and Y. K. Sun, Redox Mediators for Li–O2 Batteries: Status and Perspectives, Adv. Mater., 2018, 30, 1704162 CrossRef.
- T. Liu, M. Leskes, W. Yu, A. J. Moore, L. Zhou, P. M. Bayley, G. Kim and C. P. Grey, Cycling Li-O2 batteries via LiOH formation and decomposition, Science, 2015, 350, 530–533 CrossRef CAS.
- T. Liu, G. Kim, J. Carretero-González, E. Castillo-Martínez, P. M. Bayley, Z. Liu and C. P. Grey, Response to comment on ‘cycling Li-O2 batteries via LiOH formation and decomposition, Science, 2016, 352, 667 CrossRef.
- C. M. Burke, R. Black, I. R. Kochetkov, V. Giordani, D. Addison, L. F. Nazar and B. D. McCloskey, Implications of 4 e- Oxygen Reduction via Iodide Redox Mediation in Li-O2 Batteries, ACS Energy Lett., 2016, 1, 747–756 CrossRef CAS.
- Y. Qiao, S. Wu, Y. Sun, S. Guo, J. Yi, P. He and H. Zhou, Unraveling the Complex Role of Iodide Additives in Li-O2 Batteries, ACS Energy Lett., 2017, 2, 1869–1878 CrossRef CAS.
- S. Wu, Y. Qiao, S. Yang, M. Ishida, P. He and H. Zhou, Organic hydrogen peroxide-driven low charge potentials for high-performance lithium-oxygen batteries with carbon cathodes, Nat. Commun., 2017, 8, 15607 CrossRef CAS PubMed.
- T. Zhang, K. Liao, P. He and H. Zhou, A self-defense redox mediator for efficient lithium–O2 batteries, Energy Environ. Sci., 2016, 9, 1024–1030 RSC.
- X. P. Zhang, Y. Y. Sun, Z. Sun, C. S. Yang and T. Zhang, Anode interfacial layer formation via reductive ethyl detaching of organic iodide in lithium–oxygen batteries, ACS Appl. Mater. Interfaces, 2021, 13(14), 16437–16444 CrossRef PubMed.
- Y. N. Li, F. L. Jiang, Z. Sun, O. Yamamoto, N. Imanishi and T. Zhang, Bifunctional 1-Boc-3-Iodoazetidine Enhancing Lithium Anode Stability and Rechargeability of Lithium-Oxygen Batteries, ACS Appl. Mater. Interfaces, 2021, 13, 16437–16444 CrossRef CAS.
- Z. Guo, Q. Zhang, C. Wang, Y. Zhang, S. Dong and G. Cui, I-containing Polymer/Alloy Layer-Based Li Anode Mediating High-Performance Lithium–Air Batteries, Adv. Funct. Mater., 2022, 32(12), 2108993 CrossRef CAS.
- X. Bi, M. Li, C. Liu, Y. Yuan, H. Wang, B. Key, R. Wang, R. Shahbazian-Yassar, L. A. Curtiss, J. Lu and K. Amine, Cation Additive Enabled Rechargeable LiOH-Based Lithium–Oxygen Batteries, Angew. Chem., Int. Ed., 2020, 59, 22978–22982 CrossRef CAS PubMed.
- X. Bi, J. Li, M. Dahbi, J. Alami, K. Amine and J. Lu, Understanding the Role of Lithium Iodide in Lithium–Oxygen Batteries, Adv. Mater., 2022, 34(1), e2106148 CrossRef PubMed.
- M. Balaish, A. Kraytsberg and Y. Ein-Eli, Realization of an artificial three-phase reaction zone in a Li-Air battery, ChemElectroChem, 2014, 1, 90–94 CrossRef.
- O. Wijaya, A. Rinaldi, R. Younesi and R. Yazami, The Origin of Li-O2 Battery Performance Enhancement Using Fluorocarbon Additive, J. Electrochem. Soc., 2016, 163, A2660–A2664 CrossRef CAS.
- Y. Wang, D. Zheng, X. Q. Yang and D. Qu, High rate oxygen reduction in non-aqueous electrolytes with the addition of perfluorinated additives, Energy Environ. Sci., 2011, 4, 3697–3702 RSC.
- Y. Nishikami, T. Konishi, R. Omoda, Y. Aihara, K. Oyaizu and H. Nishide, Oxygen-enriched electrolytes based on perfluorochemicals for high-capacity lithium–oxygen batteries, J. Mater. Chem. A, 2015, 3, 10845–10850 RSC.
- S. S. Zhang and J. Read, Partially fluorinated solvent as a co-solvent for the non-aqueous electrolyte of Li/air battery, J. Power Sources, 2011, 196, 2867–2870 CrossRef CAS.
- S. Zhang, Sources and undefined 2011, Partially fluorinated solvent as a co-solvent for the non-aqueous electrolyte of Li/air battery, J. Power Sources, 2011, 196(5), 2867–2870 CrossRef CAS.
- Y. Wang, F. Bai, A. Wang, Z. Cui, D. Wang, S. Shi and T. Zhang, Perfluorinated organics regulating Li2O2 formation and improving stability for Li–oxygen batteries, Chem. Commun., 2021, 57, 3030 RSC.
- E. Nasybulin, W. Xu, M. H. Engelhard, Z. Nie, S. D. Burton, L. Cosimbescu, M. E. Gross and J. G. Zhang, Effects of electrolyte salts on the performance of Li-O2 batteries, J. Phys. Chem. C, 2013, 117, 2635–2645 CrossRef CAS.
- E. Yoo and H. Zhou, Enhanced Cycle Stability of Rechargeable Li-O2 Batteries by the Synergy Effect of a LiF Protective Layer on the Li and DMTFA Additive, ACS Appl. Mater. Interfaces, 2017, 9, 21307–21313 CrossRef CAS.
- Y. Yu, G. Huang, J. Z. Wang, K. Li, J. L. Ma and X. B. Zhang, In Situ Designing a Gradient Li+ Capture and Quasi-Spontaneous Diffusion Anode Protection Layer toward Long-Life Li−O2 Batteries, Adv. Mater., 2020, 32, 2004157 CrossRef CAS PubMed.
- Q. C. Liu, J. J. Xu, S. Yuan, Z. W. Chang, D. Xu, Y. Bin Yin, L. Li, H. X. Zhong, Y. S. Jiang, J. M. Yan and X. B. Zhang, Artificial Protection Film on Lithium Metal Anode toward Long-Cycle-Life Lithium-Oxygen Batteries, Adv. Mater., 2015, 27, 5241–5247 CrossRef CAS PubMed.
- N. S. Choi, G. Jeong, B. Koo, Y. W. Lee and K. T. Lee, Tris(pentafluorophenyl) borane-containing electrolytes for electrochemical reversibility of lithium peroxide-based electrodes in lithium–oxygen batteries, J. Power Sources, 2013, 225, 95–100 CrossRef CAS.
- S. S. Zhang, K. Xu and J. Read, A non-aqueous electrolyte for the operation of Li/air battery in ambient environment, J. Power Sources, 2011, 196, 3906–3910 CrossRef CAS.
- J. Read, K. Mutolo, M. Ervin, W. Behl, J. Wolfenstine, A. Driedger and D. Foster, Oxygen Transport Properties of Organic Electrolytes and Performance of Lithium/Oxygen Battery, J. Electrochem. Soc., 2003, 150, A1351 CrossRef CAS.
- C. Xia, C. L. Bender, B. Bergner, K. Peppler and J. Janek, An electrolyte partially-wetted cathode improving oxygen diffusion in cathodes of non-aqueous Li–air batteries, Electrochem. Commun., 2013, 26, 93–96 CrossRef CAS.
- J. G. Riess, Highly fluorinated systems for oxygen transport, diagnosis and drug delivery, Colloids Surf., A Physicochem. Eng. Asp., 1994, 84, 33–48 CrossRef CAS.
- J. G. Riess and J. G. Riess, Oxygen carriers (‘blood substitutes’) - Raison d’etre, chemistry, and some physiology, Chem. Rev., 2001, 101, 2797–2919 CrossRef CAS.
- M. P. Krafft and J. G. Riess, Perfluorochemical-Based Oxygen Therapeutics, Contrast Agents, and Beyond, Fluorine and Health: Molecular Imaging, Biomedical Materials and Pharmaceuticals, 2008, pp. 447–486 Search PubMed.
- J. Lu, L. Li, J. B. Park, Y. K. Sun, F. Wu and K. Amine, Aprotic and aqueous Li-O2 batteries, Chem. Rev., 2014, 114, 5611–5640 CrossRef CAS PubMed.
- C. Ó. Laoire, S. Mukerjee, E. J. Plichta, M. A. Hendrickson and K. M. Abraham, Rechargeable Lithium/TEGDME- LiPF6/O2 Battery, J. Electrochem. Soc., 2011, 158, A302 CrossRef CAS.
- H. G. Jung, J. Hassoun, J. B. Park, Y. K. Sun and B. Scrosati, An improved high-performance lithium-air battery, Nat. Chem., 2012, 4, 579–585 CrossRef CAS PubMed.
- R. Battino, T. R. Rettich and T. Tominaga, The Solubility of Oxygen and Ozone in Liquids, J. Phys. Chem. Ref. Data, 1983, 12, 163–178 CrossRef CAS.
- R. Yazami, Smart Battery Charging System, US Pat., US20120043929A1, 2012 Search PubMed.
- M. Balaish, A. Kraytsberg and Y. Ein-Eli, A critical review on lithium–air battery electrolytes, Phys. Chem. Chem. Phys., 2014, 16, 2801–2822 RSC.
- B. D. McCloskey, D. S. Bethune, R. M. Shelby, G. Girishkumar and A. C. Luntz, Solvents critical role in nonaqueous Lithium-Oxygen battery electrochemistry, J. Phys. Chem. Lett., 2011, 2, 1161–1166 CrossRef CAS.
- V. S. Bryantsev, V. Giordani, W. Walker, M. Blanco, S. Zecevic, K. Sasaki, J. Uddin, D. Addison and G. V. Chase, Predicting solvent stability in aprotic electrolyte Li-air batteries: Nucleophilic substitution by the superoxide anion radical (O2•-), J. Phys. Chem. A, 2011, 115, 12399–12409 CrossRef CAS.
- R. Black, S. H. Oh, J. H. Lee, T. Yim, B. Adams and L. F. Nazar, Screening for superoxide reactivity in Li-O 2 batteries: Effect on Li2O2/LiOH crystallization, J. Am. Chem. Soc., 2012, 134, 2902–2905 CrossRef CAS PubMed.
- K. U. Schwenke, S. Meini, X. Wu, H. A. Gasteiger and M. Piana, Stability of superoxide radicals in glyme solvents for non-aqueous Li–O2 battery electrolytes, Phys. Chem. Chem. Phys., 2013, 15, 11830–11839 RSC.
- K. R. Ryan, L. Trahey, B. J. Ingram and A. K. Burrell, Limited stability of ether-based solvents in lithium-oxygen batteries, J. Phys. Chem. C, 2012, 116, 19724–19728 CrossRef CAS.
- S. A. Freunberger, Y. Chen, Z. Peng, J. M. Griffin, L. J. Hardwick, F. Bardé, P. Novák and P. G. Bruce, Reactions in the rechargeable lithium-O2 battery with alkyl carbonate electrolytes, J. Am. Chem. Soc., 2011, 133, 8040–8047 CrossRef CAS PubMed.
- O. Wijaya, P. Hartmann, R. Younesi, I. I. E. Markovits, A. Rinaldi, J. Janek and R. Yazami, A gamma fluorinated ether as an additive for enhanced oxygen activity in Li-O2 batteries, J. Mater. Chem. A, 2015, 3, 19061–19067 RSC.
- Z. Huang, J. Ren, W. Zhang, M. Xie, Y. Li, D. Sun, Y. Shen and Y. Huang, Protecting the Li-Metal Anode in a Li–O2 Battery by using Boric Acid as an SEI-Forming Additive, Adv. Mater., 2018, 30(39), 1803270 CrossRef PubMed.
- J. Lai, N. Chen, F. Zhang, B. Li, Y. Shang, L. Zhao, L. Li, F. Wu and R. Chen, Imidazolium bromide: A tri-functional additive for rechargeable Li-O2 batteries, Energy Storage Mater., 2022, 49, 401–408 CrossRef.
- Y. Kim, D. Koo, S. Ha, S. C. Jung, T. Yim, H. Kim, S. K. Oh, D. M. Kim, A. Choi, Y. Kang, K. H. Ryu, M. Jang, Y. K. Han, S. M. Oh and K. T. Lee, Two-Dimensional Phosphorene-Derived Protective Layers on a Lithium Metal Anode for Lithium-Oxygen Batteries, ACS Nano, 2018, 12, 4419–4430 CrossRef CAS PubMed.
- H. Guo, G. Hou, J. Guo, X. Ren, X. Ma, L. Dai, S. Guo, J. Lou, J. Feng, L. Zhang, P. Si and L. Ci, Enhanced Cycling Performance of Li-O2 Battery by Using a Li3PO4-Protected Lithium Anode in DMSO-Based Electrolyte, ACS Appl. Energy Mater., 2018, 1, 5511–5517 CrossRef CAS.
- A. A. Uludağ, M. Tokur, H. Algul, T. Cetinkaya, M. Uysal and H. Akbulut, Int. J. Hydrogen Energy, 2016, 41, 6954–6964 CrossRef.
- M. Tokur, H. Algul, T. Cetinkaya, M. Uysal and H. Akbulut, Improved Electrochemical Performance of Lithium Oxygen Batteries with N-methyl-2-pyrrolidone Based Composite Polymer Electrolytes, J. Electrochem. Soc., 2016, 163, A1326–A1335 CrossRef CAS.
- Z. Guo, J. Li, Y. Xia, C. Chen, F. Wang, A. G. Tamirat, Y. Wang, Y. Xia, L. Wang and S. Feng, A flexible polymer-based Li-air battery using a reduced graphene oxide/Li composite anode, J. Mater. Chem. A, 2018, 6, 6022–6032 RSC.
- B. Tong, J. Huang, Z. Zhou and Z. Peng, The Salt Matters: Enhanced Reversibility of Li–O2 Batteries with a Li[(CF3SO2)(n-C4F9SO2)N]-Based Electrolyte, Adv. Mater., 2018, 30(1), 1704841 CrossRef.
- I. Landa-Medrano, M. Olivares-Marín, B. Bergner, R. Pinedo, A. Sorrentino, E. Pereiro, I. Ruiz De Larramendi, J. Janek, T. Rojo and D. Tonti, Potassium Salts as Electrolyte Additives in Lithium-Oxygen Batteries, J. Phys. Chem. C, 2017, 121, 3822–3829 CrossRef CAS.
- Q. Zhang, Y. Zhou, W. Dai, X. Cui, Z. Lyu, Z. Hu and W. Chen, Chloride Ion as Redox Mediator in Reducing Charge Overpotential of Aprotic Lithium-Oxygen Batteries, Batteries Supercaps, 2021, 4, 232–239 CrossRef CAS.
- S. Ding, C. Zhang, L. Wu and X. Yuan, Anion-cation coupled electrolyte additive enhancing the performance of lithium-oxygen batteries, Chem. Eng. J., 2024, 500, 156757 CrossRef CAS.
- Y. Zhang, Q. Chen, D. Li, S. Qi, Y. Liu, J. Liu, H. Xie and J. Zhu, AgTFSI Pretreated Li Anode in LiI-Mediated Li-O2 Battery: Enabling Lithiophilic Solid Electrolyte Interphase Generation to Suppress the Redox Shuttling, CCS Chem., 2024, 32(2), 2106984 Search PubMed.
- A. Ahmadiparidari, S. Fuladi, L. Majidi, S. Plunkett, E. Sarnello, H. Gholivand, Z. Hemmat, S. Rastegar, S. N. Misal, N. Jimenez, P. C. Redfern, J. Wen, T. Li, A. T. Ngo, F. Khalili-Araghi, L. A. Curtiss and A. Salehi-Khojin, Enhancing the performance of lithium oxygen batteries through combining redox mediating salts with a lithium protecting salt, J. Power Sources, 2021, 491, 229506 CrossRef CAS.
- P. Xu, X. Lin, Z. Sun, K. Li, W. Dou, Q. Hou, Z. Zhou, J. Yan, M. Zheng, R. Yuan and Q. Dong, A robust interphase via in-situ pre-reconfiguring lithium anode surface for long-term lithium-oxygen batteries, J. Energy Chem., 2022, 72, 186–194 CrossRef CAS.
- M. Song, C. Chen, T. Huang and A. Yu, Tetramethylpyrazine: an electrolyte additive for high capacity and energy efficiency lithium-oxygen batteries, RSC Adv., 2021, 11, 24320–24325 RSC.
- H. Wan, Y. Sun, W. Cai, Q. Shi, Y. Zhu and Y. Qian, A Friendly Soluble Protic Additive Enabling High Discharge Capability and Stabilizing Li Metal Anodes in Li–O2 Batteries, Adv. Funct. Mater., 2022, 32(2), 2106984 CrossRef CAS.
- W. Xu, X. Liao, W. Xu, C. Sun, K. Zhao, Y. Zhao and C. Hu, Gradient SEI layer induced by liquid alloy electrolyte additive for high rate lithium metal battery, Nano Energy, 2021, 88, 106237 CrossRef CAS.
- J. S. Lee, K. Shin, S. Y. Jun, S. Kim and W. H. Ryu, Dual-functional vinylpyrrolidone electrolyte additive as anode surface leveler and cathode catalyst for lithium Metal-Oxygen batteries, Chem. Eng. J., 2023, 458, 141383 CrossRef CAS.
- F. Zhang, J. Lai, Z. Hu, A. Zhou, H. Wang, X. Hu, L. Hou, B. Li, W. Sun, N. Chen, L. Li, F. Wu and R. Chen, Lithium Salt Dissociation Promoted by 18-Crown-6 Ether Additive toward Dilute Electrolytes for High Performance Lithium Oxygen Batteries, Angew. Chem., 2023, 62(16), e202301772 CrossRef CAS PubMed.
- Z. Man, H. Tian, X. Zhu and Z. Lü, High Charging Voltage Stable and Air Atmosphere Stable Li-O2 Batteries with an Electrolyte Based on Succinonitrile and In Situ Artificial SEI Construction, ACS Sustain. Chem. Eng., 2024, 12, 3996–4006 CrossRef CAS.
- Z. Li, Y. Yu, D. Yang, J. Wang, J. Yan, G. Huang, T. Liu and X. Zhang, A bifunctional nitrile additive for high-performance lithiumoxygen batteries, Nano Res., 2024, 17, 6119–6126 CrossRef CAS.
- Y. L. Liang, Y. Yu, Z. W. Li, J. Wang, J. M. Yan, G. Huang and X. B. Zhang, Co-deposition of conductive additives and lithium peroxide during discharge to boost the performance of lithium-oxygen batteries, J. Mater. Chem. A, 2024, 24977–24982 RSC.
- M. Tokur, H. Algul, S. Ozcan, T. Cetinkaya, M. Uysal, M. O. Guler and H. Akbulut, Stability effect of polymer-based additives on EMITFSI-LiTFSI electrolyte in lithium-air battery, Solid State Ionics, 2016, 286, 51–56 CrossRef CAS.
- M. G. Jeong, H. H. Lee, H. J. Shin, Y. Jeong, J. Y. Hwang, W. J. Kwak, G. Oh, W. Kim, K. Ryu, S. Yu, H. D. Lim, M. Lee and H. G. Jung, A fluoroalkyl iodide additive for Li–O2 battery electrolytes enables stable cycle life and high reversibility, J. Mater. Chem. A, 2023, 11, 15246–15255 RSC.
| This journal is © The Royal Society of Chemistry 2026 |







