 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
A synthetic toolbox for sustainable methods in porphyrin chemistry
Karolina Urbańska *a,
Liam Cribbina,
Brendan Twamleyb,
Fiach O Ceallaigha and
Mathias O. Senge
*a,
Liam Cribbina,
Brendan Twamleyb,
Fiach O Ceallaigha and
Mathias O. Senge a
a
aSchool of Chemistry, Trinity Biomedical Sciences Institute, Trinity College Dublin, The University of Dublin, 152–160 Pearse Street, D02 R590 Dublin, Ireland. E-mail: urbanskk@tcd.ie
bSchool of Chemistry, Trinity College Dublin, The University of Dublin, Dublin 2, Ireland
First published on 7th May 2026
Abstract
Greener synthesis for red dyes. As the days of chlorinated organic solvents are numbered, we propose a sustainable synthetic protocol for porphyrin synthesis to facilitate the transition into a more viable, safer and greener lab. Here we present a facile flow-assisted method for the sustainable synthesis of 40 meso-substituted planar porphyrins of the A4-, A2-, and trans-A2B2-type, including 10 examples of dodecasubstituted nonplanar porphyrins. We set forth a sustainable approach towards porphyrin synthesis, saving up to 90% of chlorinated solvents and drastically limiting waste generation by merging a flow synthesis process via a continuous stirred tank reactor (CSTR) with a traditional batch oxidation method.
Sustainability spotlight“A synthetic toolbox for sustainable methods in porphyrin chemistry” falls into goal 12 of the UN Sustainable Development Goals (Sustainable consumption and production patterns). One of the targets of chapter 20 of Agenda 21 is preventing or minimizing the generation of hazardous waste. In porphyrin chemistry, the bulk of hazardous waste generated during porphyrin synthesis consists of chlorinated solvents. We aim to limit the generation of hazardous waste in porphyrin synthesis by reducing the volume of solvent required for the synthesis. This is enabled by continuous flow; merging a flow synthesis process via a continuous stirred tank reactor (CSTR) with a traditional batch oxidation step, we reduced the volume of chlorinated solvent required for the synthesis by up to 90%. |
Introduction
Minimising solvent use in chemical reactions is one of the twelve principles of green chemistry1 and the ten objectives of green and sustainable chemistry, proposed by the United Nations.2 Specifically, seeking alternatives to chlorinated solvents was identified as one of the key green chemistry research areas by the ACS Green Chemistry Institute in 2005.3 While a suitable drop-in replacement for dichloromethane (DCM) and its homologues has not yet been identified,4 limiting their use is strongly encouraged. This is exemplified by the restriction for specific industrial applications of DCM in the European Union since 2010 (ref. 5) and most uses of DCM introduced in the United States by the Environmental Protection Agency in 2024.6Macrocycles constitute a class of functional molecules applied in sensing, catalysis, medicine, cosmetics, agriculture and nanotechnology.7 Their synthesis requires high dilution to promote cyclisation over formation of linear products.8 This is called the high dilution principle, and its implications can be circumvented by experiment design,9 pseudodilution10 and catalysis.11
Porphyrins are aromatic tetrapyrrolic macrocycles. meso-Substituted porphyrins, such as 5,10,15,20-tetraphenylporphyrin (H2TPP, Fig. 1) are artificial dyes, counterparts to naturally occurring porphyrinoids, like heme B, chlorophyll a, or vitamin B12 (Fig. 1).12 Due to the ease of their synthesis and versatile chemical and physical properties, meso-substituted porphyrins are highly sought after as feasible targets for research areas including catalysis,13 quantum technologies,14 molecular probes15 and supramolecular assemblies,16 applications in dye-sensitised solar cells,17 remediation,18 imaging, photodynamic therapy,19 and other areas of biomedicine.20
 | ||
| Fig. 1 Structures of chlorophyll a, heme B, 5,10,15,20-tetraphenylporphyrin (H2TPP) and vitamin B12. | ||
Perhaps the most thoroughly studied and certainly most frequently applied method for porphyrin synthesis is a two-step protocol involving a condensation of aldehydes with pyrroles under acidic conditions, followed by an oxidation of the tetrapyrrolic macrocycle.21 The first general method toward porphyrin synthesis was initially reported by Rothemund22 and further developed by Adler,23 and called for harsh conditions, such as high temperatures and acidic or basic solvents (Table 1).
| Protocol | Rothemund | Adler–Longo | Lindsey |
|---|---|---|---|
| Steps | 1 | 1 | 2 |
| Concentration | High 3.6 M | High 0.1–0.3 M | Low 0.001–0.1 M |
| Temperature | 220 °C | 140 °C | 20 °C |
| Solvent | Pyridine | Propionic acid | Chlorinated |
| Catalyst | None | cat. = solvent | BF3·Et2O, TFA |
| Conditions | sealed vessel | Aerobic | Inert |
| Oxidant | None | Nitrobenzene | Quinone |
| Yields | <10% | <30% | 35–60% |
Porphyrins synthesised via either Rothemund or Adler protocols were contaminated with the corresponding chlorins (<10%); this could be evaded by treatment with a strong quinone oxidant, such as 2,3-dichloro-5,6-dicyanobenzoquinone (DDQ). In Lindsey's method, oxidation is an inherent step following the mild, room temperature cyclisation carried out in acidified chlorinated solvents, at low concentrations.24 Currently, the Lindsey protocol is the mildest and most effective method for the synthesis of meso-substituted porphyrins, both A4 5,10,15,20-tetra-25,26 (Fig. 2) and 2,3,5,7,8,10,12,13,15,17,18,20-dodecasubstituted.27,28 The higher yields and wider functional group tolerance are related to the mild reaction conditions.
The Lindsey protocol is currently the standard method for porphyrin synthesis in academia and industry.29 One major drawback of this method is the requirement for large volumes of chlorinated solvents for the reaction, which calls for 1–100 mM reactant concentrations to promote macrocyclisation over formation of linear side products.30,31 This constitutes a sustainability issue and improvements to the current methods or development of new protocols for porphyrin synthesis are needed to overcome this problem.
Protocols employing microwave irradiation and mechanochemistry, as well as the use of ionic liquids as solvents, have been developed over the past two decades to increase the sustainability of meso-substituted porphyrin synthesis.32 Recently, two contributions describing the use of continuous flow in porphyrin synthesis were reported. These involve the Gonsalves protocol,33,34 and a process for Lindsey synthesis of meso-substituted porphyrins.35 Both articles constitute landmarks in porphyrin synthesis in continuous flow; however, there is still room for improvement in the sustainability-related aspects of porphyrin synthesis. Reduction of solvent volume required for the synthesis, application of green solvents and finally, generalisation of solventless methods are green directions in porphyrin synthesis. In this contribution we build up on the preliminary studies reported for application of continuous flow in porphyrin synthesis to achieve improved sustainability (E-factor) and propose a universal method for porphyrin synthesis. We present a semi-continuous protocol for porphyrin synthesis under Lindsey conditions, incorporating a continuous stirred tank reactor (CSTR) at the condensation step. In this approach, we show that a significant (10×) increase in reactant concentration is not only beneficial for the reaction yield, but it also allows reduction in the volume of chlorinated solvents required for macrocyclisation by up to 90%.
Experimental
Materials and methods
Dipyrromethanes 5c and 5d (Scheme 4) were purchased from Frontier Scientific. Column chromatography was carried out using Supelco Silica Gel (pore size 60 Å, 230–400 mesh; Merck). CSTR-assisted synthesis was carried out using Asynt fReactor with five 2 mL modules (total reactor volume 10 mL). Detailed description of the experimental setup, including photographs, is provided in SI (page S6). Full compound characterisation data and 1H NMR, 13C NMR, 19F NMR, HRMS, IR, UV/vis spectra are provided in SI.Synthetic procedures
Dipyrromethanes 5a, 5b, 5e, 5f (Scheme 4) and 3,4-diethylpyrrole 9 (Scheme 5) were prepared according to literature procedures.36–38 | ||
| Scheme 2 Scope and limitations of CSTR-assisted synthesis of A4 porphyrins. Yields in grey were reported in literature for batch syntheses.44 aLiterature yield for Alder–Longo method. | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) were taken up in syringes and injected into fReactor at equal flow rate of 1 mL min−1. After injection was completed, an additional 10 mL of DCM was pumped through the reactor to wash fully wash out the crude porphyrinogen 10a–af (Scheme 6) into a round bottom flask equipped with a stir bar and shielded from light. DDQ (750 mg, 3.30 mmol) was added to the crude mixture as powder and the mixture was stirred at RT for 1 h, then neutralised with TEA (150 µL, 1.1 mmol). The crude porphyrins 11a–s (Scheme 6) were isolated as green solids by precipitation with methanol. Recrystallisation from KOH/EtOH 0.4% w/v afforded ‘free base’ dodecasubstituted porphyrins as brown/purple crystals. Porphyrin 11d (Scheme 6) was isolated as a dication salt, in the form of a green solid. The yields were determined based on two experiments.
1, v/v) were taken up in syringes and injected into fReactor at equal flow rate of 1 mL min−1. After injection was completed, an additional 10 mL of DCM was pumped through the reactor to wash fully wash out the crude porphyrinogen 10a–af (Scheme 6) into a round bottom flask equipped with a stir bar and shielded from light. DDQ (750 mg, 3.30 mmol) was added to the crude mixture as powder and the mixture was stirred at RT for 1 h, then neutralised with TEA (150 µL, 1.1 mmol). The crude porphyrins 11a–s (Scheme 6) were isolated as green solids by precipitation with methanol. Recrystallisation from KOH/EtOH 0.4% w/v afforded ‘free base’ dodecasubstituted porphyrins as brown/purple crystals. Porphyrin 11d (Scheme 6) was isolated as a dication salt, in the form of a green solid. The yields were determined based on two experiments.Results and discussion
A4 porphyrins
With the goal to limit the volume of chlorinated solvents required for porphyrin synthesis, we envisioned a semi-continuous process, aided by a CSTR reactor (continuous stirred tank reactor). In the first step, an in-flow condensation of aldehydes 2a–af (Fig. S1) with pyrrole 1 was performed. Due to the poor solubility of DDQ in chlorinated solvents, in-line oxidation would be counterproductive to the goal of limiting the need for chlorinated solvents in porphyrin synthesis.39 DDQ was selected as the oxidant for this method (rather than its weaker tetrachlorinated derivative p-chloranil) in order to eliminate the need for heat supply during the oxidation step40 and to comply with principle 6 of green chemistry (“design for energy efficiency”).1 Hence, both the oxidation and neutralisation steps were conducted in-batch.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio. The optimisation of in-flow condensation conditions involved the flow rate and the concentration of the reactants. The concentration range investigated started at 0.02 M, (a typical concentration for in-batch condensation)24 and was increased up to c = 1.00 M (50 times more concentrated). For each concentration, we examined four different flow rates to assess the influence of residence time on the yield. Isolated yields listed in Table 2 were determined for the pure isolated product of condensation, oxidation and neutralisation (i.e., ‘free base’ porphyrin 4a).
1 molar ratio. The optimisation of in-flow condensation conditions involved the flow rate and the concentration of the reactants. The concentration range investigated started at 0.02 M, (a typical concentration for in-batch condensation)24 and was increased up to c = 1.00 M (50 times more concentrated). For each concentration, we examined four different flow rates to assess the influence of residence time on the yield. Isolated yields listed in Table 2 were determined for the pure isolated product of condensation, oxidation and neutralisation (i.e., ‘free base’ porphyrin 4a).
| Entry | Conc. [M] | Flow rate [mL min−1] | Yield [%] |
|---|---|---|---|
| 1 | 0.02 | 0.5 | 15 |
| 2 | 0.02 | 1.0 | 12 |
| 3 | 0.02 | 1.5 | 10 |
| 4 | 0.02 | 2.0 | 9 |
| 5 | 0.05 | 0.5 | 22 |
| 6 | 0.05 | 1.0 | 16 |
| 7 | 0.05 | 1.5 | 15 |
| 8 | 0.05 | 2.0 | 11 |
| 9 | 0.10 | 0.5 | 27 |
| 10 | 0.10 | 1.0 | 24 |
| 11 | 0.10 | 1.5 | 23 |
| 12 | 0.10 | 2.0 | 20 |
| 13 | 0.15 | 0.5 | 30 |
| 14 | 0.15 | 1.0 | 30 |
| 15 | 0.15 | 1.5 | 29 |
| 16 | 0.15 | 2.0 | 29 |
| 17 | 0.20 | 0.5 | 28 |
| 18 | 0.20 | 1.0 | 28 |
| 19 | 0.20 | 1.5 | 27 |
| 20 | 0.20 | 2.0 | 25 |
| 21 | 0.30 | 0.5 | 27 |
| 22 | 0.30 | 1.0 | 26 |
| 23 | 0.30 | 1.5 | 24 |
| 24 | 0.30 | 2.0 | 24 |
| 25 | 0.50 | 0.5 | 22 |
| 26 | 0.50 | 1.0 | 20 |
| 27 | 0.50 | 1.5 | 20 |
| 28 | 0.50 | 2.0 | 19 |
| 29 | 1.00 | 0.5 | 12 |
| 30 | 1.00 | 1.0 | 12 |
| 31 | 1.00 | 1.5 | 13 |
| 32 | 1.00 | 2.0 | 13 |
Overall, the yields obtained in this optimisation varied from 9 to 30% (Table 2, entry 4 and entries 13 and 14, respectively). Interestingly, increasing the concentration of the reactants from standard batch conditions (c = 0.02 M) up to 10 times (c = 0.15, 0.20 M) returned best results; the highest yield recorded for c = 0.02 M was 15% (entry 1), and the best yield overall was 30% (c = 0.15 M, entries 13, 14). No further improvement was observed increasing the reactant concentration to c = 1.00 M – unsurprisingly, a gradual drop in isolated yields to 28% (0.20 M, entry 17), 27% (0.30 M, entry 21), 22% (0.50 M, entry 25) and 13% (1.00 M, entries 31, 32) was observed. The optimal concentration of reactants for the in-flow condensation reaction was 0.15 M.
The impact of flow rate and residence time on the yield of 5,10,15,20-tetraphenylporphyrin 4a was well-pronounced at low concentrations (0.02–0.10 M, entries 1–12, Table 2). Here, decreasing the flow rate from 2.0 mL min−1 to 0.5 mL min−1 contributed to 6, 12 and 7% yield increase at 0.02, 0.05 and 0.10 M, respectively. The maximum yields obtained for this range of concentrations were 15, 22 and 27%, increasing with the reactant concentration and residence time (entries 4, 8, 12). The influence of residence time on the isolated yield of H2TPP (4a) was negligible in more concentrated reaction mixtures (0.15–1.00 M), with 3–8% differences in isolated yields. At the most effective reagent concentration of 0.15 M, both 0.5 mL min−1 and 1.0 mL min−1 returned the same highest yield of 30%. Thus, we concluded that the optimal flow rate for in-flow condensation of aldehydes and pyrroles under acid catalysis is 1.0 mL min−1, translating to 15 min residence time.
Having established the optimal concentration and flow rate of the reactants for condensation, we examined other solvents commonly used in porphyrin synthesis – acetonitrile (MeCN), as a non-chlorinated alternative, and CHCl3 (Table 3). The reactions were carried out at c = 0.3 M concentrations at 1.0 mL min−1 flow rate. No product was detected in the reaction in MeCN (entry 1), and in CHCl3 the yield was 10% – over 2.5 times lower than in DCM (entries 2 and 3, respectively). While DCM is more volatile than CHCl3, the latter is classified as significantly more hazardous for health according to the CLP Regulation.41,42 Thus, we selected DCM as the more promising choice of solvent for this synthetic method.
| Entry | Conc. [M] | Flow rate [mL min−1] | Solvent | Yield [%] |
|---|---|---|---|---|
| 1 | 0.30 | 1.0 | MeCN | 0 |
| 2 | 0.30 | 1.0 | CHCl3 | 10 |
| 3 | 0.30 | 1.0 | DCM | 26 |
Next, we carried out experiments with other acids used as catalysts in the Lindsey method of porphyrin synthesis, i.e., boron trifluoride diethyl etherate (BF3·Et2O) and p-toluenesulfonic acid (TSA). Both acids promoted the condensation, however neither performed as well as TFA: BF3·Et2O yielded 17% of H2TPP (entry 1, Table 4) and TSA 25% (entry 2). Additionally, we examined the influence of the amount of acid catalyst used on the yield, on the example of TFA. Reducing the amount of acid to 0.5 eq. resulted in a yield decrease to 8% (entry 4), while using 2.0 eq. of TFA only slightly increased the yield from 30 to 34% (entry 5). This result is in line with the 31% yield of 4a reported by Parveen et al.35 Given the modest improvement in yield triggered by the 100% increase in TFA quantity (entry 5), we concluded that TFA in 1.0 eq. is the optimal catalyst for condensation of aldehydes with pyrrole in a CSTR reactor. Taken together, the optimised conditions used for the determination of scope of flow-assisted synthesis of porphyrins under Lindsey conditions at high concentrations were a c = 0.15 M concentration of reagents in DCM with 1.0 eq. of TFA, at 1.0 mL min−1 flow rate. These optimised conditions require 7.5 times less DCM for porphyrin synthesis (c = 0.15 M). Additionally, a further decrease in solvent requirement is possible with a negligible drop in yield (from 30 to 28%), to 10 times less DCM than in batch conditions (using c = 0.20 M).
| Entry | Conc. [M] | Flow rate [mL min−1] | Acid | Acid eq. | Yield [%] |
|---|---|---|---|---|---|
| 1 | 0.15 | 1.0 | BF3·Et2O | 0.1 | 17 |
| 2 | 0.15 | 1.0 | TSA | 1.0 | 25 |
| 3 | 0.15 | 1.0 | TFA | 1.0 | 30 |
| 4 | 0.15 | 1.0 | TFA | 0.5 | 8 |
| 5 | 0.15 | 1.0 | TFA | 2.0 | 34 |
The best yields were obtained for condensations involving activated benzaldehydes, 4-tert-butylbenzaldehyde 2d (4d, 43%), 4-tolualdehyde 2b (4b, 33%) and 3,5-di-tert-butylbenzaldehyde 2q (4q, 33%). 4a was obtained in 31% yield from 2a. Condensations with isomeric anisaldehyde 2e and 2i yielded 5,10,15,20-tetrakis(4-methoxyphenyl)porphyrin (4e, 26%) and 5,10,15,20-tetrakis(3-methoxyphenyl)porphyrin (4i, 24%). The presence of a bulky substituent in the ortho position of a benzaldehyde negatively impacts the yield,43 as evidenced by 5,10,15,20-tetrakis(2-methoxyphenyl)porphyrin 4l and 5,10,15,20-tetrakis(2,6-dimethoxyphenyl)porphyrin 4n, which were formed in low yields of 8 and 4%, from aldehydes 2l and 2n, respectively. While p-bromobenzaldehyde 2c gave 20% yield of the corresponding porphyrin 4c, meta and ortho isomers both yielded 7% yield of porphyrins 4h and 4k, respectively. Interestingly, 4f and 4g were not isolated despite moderate yields reported in literature for batch synthesis of these porphyrins under Adler–Longo and Lindsey conditions, respectively.44j,k ortho-Substituted 2,6-difluorobenzaldehyde 2m and 2,4,6-trifluorobenzaldehyde 2t produced porphyrins (4m, 13%) and (4t, 11%) in yields lower than reported in literature for batch synthesis. The low yields of condensation observed for benzaldehydes bearing bulky substituents at the ortho-positions in comparison to H2TPP result directly from the steric hindrance caused by bromine and fluorine atoms and methoxy groups, compared to a hydrogen atom.43 Porphyrins 4k and 4l were obtained as mixtures of atropisomers.45
Polycyclic aromatic aldehydes, naphthaldehyde 2y, 4-methoxynaphthaldehyde 2z, 9-phenantrenecarbaldehyde 2ab, 9-antracenecarbaldehyde 2aa and 1-pyrenecarbaldehyde 2ac were subjected to condensations with pyrrole to yield π-rich porphyrins which have previously found applications in two-photon absorption46 and nanomaterials,47 with moderate results. 5,10,15,20-Tetrakis(1-naphthyl)porphyrin 4y and 5,10,15,20-tetrakis(4-methoxy-1-naphthyl)porphyrin 4z were isolated in 27 and 20% yields, while phenanthrene and pyrene carbaldehydes 2ab and 2ac produced porphyrins 4ab and 4ac in 16% and 8% yields, respectively. Not surprisingly, the sterically hindered 5,10,15,20-tetrakis(9-anthryl)porphyrin 4aa was not isolated out of the reaction mixture. Previous efforts to synthesise this porphyrin resulted in only 0.2% yield for Rothemund conditions,48 0.4% for modified Rothemund conditions (in pyridine and glacial acetic acid)49 and 2.6% and 4% for the carbinol route.50,51 Among heterocyclic derivatives, tetrakis(2-furyl)porphyrin 4w was obtained in 14% yield, while 5,10,15,20-tetrakis(1-methyl-2-imidazolyl)porphyrin 4x was not isolated. To further demonstrate the wide applicability of this method, three aliphatic aldehydes were included in the scope. These typically produce lower yields than their aromatic counterparts.52 Valeraldehyde 2ad and iso-butylaldehyde 2ae formed porphyrins 4ad and 4ae in 8% yields, but pivaldehyde 2af did not produce the 5,10,15,20-tetrakis(tert-butyl)porphyrin 4af. Porphyrins 4d, 4w, 4z and 4ae (Scheme 2, framed) were isolated in yields surpassing those reported in literature for batch methods.53–55 Yields determined for 4b, 4i, 4q, 4ab and 4ac were comparable with those reported in literature for conventional methods of porphyrin synthesis according to the Lindsey protocol.56–59 The detailed comparison of the yields obtained with this method and those reported previously in literature is presented in Scheme 2.
The limitations of this method correspond with those observed in traditional porphyrin synthesis. Bulky substituents installed near the formyl group of the aldehyde can hamper or preclude condensation.43 This was observed for mesitylaldehyde 2u, 2,6-dimethylbenzaldehyde 2o, 2,6-dichlorobenzaldehyde 2p and 2,4,6-trimethoxybenzaldehyde 2v as corresponding porphyrins 4u, 4o, 4p and 4v were not isolated.
In summary, we prepared 21 5,10,15,20-tetrasubstituted porphyrins in 4–43% yields. The yield range in this library is in line with that reported by Parveen et al., albeit exact comparison of numerical values cannot be made due to negligible overlap in scopes.35
A2 and trans-A2B2 porphyrins
As [2 + 2] condensations are typically carried out at higher dilutions,64 we anticipated an improvement in the isolated yield of H2DPP upon decreasing the reagent concentration. Indeed, at concentrations c = 0.10, c = 0.07 and c = 0.05 M the recorded yields gradually increase to 29, 31 and 35%, respectively (Table 5, entries 2, 3 and 4). Further dilution to c = 0.03 and c = 0.01 M resulted in yield drop to 19 and 17%, respectively (entries 5 and 6). Similarly to the case of A4 porphyrins described above, deviation from 1.0 eq. of TFA brought no improvement to the yield, as 0.5 eq. of acid produced 26% of H2DPP (entry 7), and 2.0 eq. of TFA yielded only 9% of the porphyrin (entry 8). Taken together, under the best yielding conditions for [2 + 2] condensation of meso-unsubstituted dipyrromethane 5a and benzaldehyde 2a, catalyzed by TFA, at c = 0.05 M concentration in dichloromethane, at 1.0 mL min−1 flow rate, H2DPP 7a was isolated in 35% yield. These conditions were used for scope determination for [2 + 2] condensations producing A2 and trans-A2B2 porphyrins. While the isolated yield of H2DPP is lower than that obtained under traditional batch conditions, our optimised CSTR-assisted method uses only 10% of the DCM volume, comprising a significant reduction of the environmental cost of porphyrin synthesis.
| Entry | Conc. [M] | Flow rate [mL min−1] | TFA eq. | Yield [%] |
|---|---|---|---|---|
| 1 | 0.15 | 1.0 | 1.0 | 27 |
| 2 | 0.10 | 1.0 | 1.0 | 29 |
| 3 | 0.07 | 1.0 | 1.0 | 31 |
| 4 | 0.05 | 1.0 | 1.0 | 35 |
| 5 | 0.03 | 1.0 | 1.0 | 19 |
| 6 | 0.01 | 1.0 | 1.0 | 17 |
| 7 | 0.05 | 1.0 | 0.5 | 26 |
| 8 | 0.05 | 1.0 | 2.0 | 9 |
 | ||
| Scheme 4 Scope and limitations of CSTR-assisted synthesis of A2 and trans-A2B2 porphyrins. Yields in grey were reported in literature for batch syntheses.65 aLiterature yield for Alder–Longo method. bYield not reported in literature. | ||
Similarly to Lindsey condensations, 3,5-di-tert-butylbenzaldehyde 2q produced porphyrin 7q in the highest yield, 42%, close to the yield reported for batch synthesis (48%).64 This was the only A2 porphyrin obtained with higher yield than H2DPP 7a, which was formed in 35%. p-Tolualdehyde 2b and p-tert-butylbenzaldehyde 2d yielded 7b and 7d in 20 and 18%, respectively, while for p-nitrobenzaldehyde 2f, a significant drop in yield was observed (3%). iso-Butyraldehyde 2ae was selected as an aliphatic example and yielded 16% of porphyrin 7ae. 5,15-Bis(2,6-difluorophenyl)porphyrin 7m was formed in 26% yield, but condensations with more hindered ortho-aldehydes, mesitylaldehyde 2u and 2,6-dimethoxybenzaldehyde 2n, did not produce the anticipated porphyrins 7u and 7n. In contrast to the A4 porphyrin 4w, the heterocyclic 5,15-bis(2-furyl)porphyrin 7w was not formed. Formation of A2 porphyrins substituted with polycyclic aromatic hydrocarbons was also impeded, as neither the naphthalene porphyrin 7y, nor the anthracene derivative 7aa were isolated.
In the second part of the scope, benzaldehyde 2a was subjected to [2 + 2] MacDonald condensations with a small library of meso-substituted dipyrromethanes, i.e. 5-phenyldipyrromethane 5b, 5-(4-bromophenyl)dipyrromethane 5c, 5-(4-nitrophenyl)dipyrromethane 5d, 5-(4-carboxymethylphenyl)dipyrromethane 5e and 5-(4-acetamidophenyl)dipyrromethane 5f. However, under the optimised conditions (Table 5), the crude mixtures contained unreacted dipyrromethanes. Thus, flow rate was adapted to 0.5 mL min−1 to allow longer residence time, and to drive the reactions to completion. Gratifyingly, this modification helped achieve full consumption of starting materials. Utilising aryl substituted dipyrromethanes 5b–f the tetrarylporphyrins 4a, 8c, 8f and 8g were formed in 28, 20, 11 and 5% yields, respectively. These values are lower than those reported previously for batch synthesis of the respective porphyrins 4a, 8c, 8f and 8g.59,66 8ag was not formed under flow conditions. No scrambling was observed in these experiments.59
Dodecasubstituted porphyrins
Recognising the unique properties arising from the shape of dodecasubstituted porphyrins, and their potential for applications in sensing,67 organocatalysis68 and photocatalysis69 we focused on the third library of interest – 5,10,15,20-tetraaryl-2,3,7,8,12,13,17,18-octaethylporphyrins (H2OETAP). To the best of our knowledge, this is the first flow-assisted method for the synthesis of dodecasubstituted porphyrins.| Entry | Acid | MeOH [%v/v] | Conc. [M] | Yield [%] |
|---|---|---|---|---|
| a 0.1 eq. of BF3·Et2O was used.b 0.5 eq. of BF3·Et2O was used. | ||||
| 1a | BF3·Et2O | 0 | 0.010 | 0 |
| 2a | BF3·Et2O | 0 | 0.100 | 0 |
| 3b | BF3·Et2O | 0 | 0.010 | 0 |
| 4 | TFA | 0 | 0.010 | 0 |
| 5 | TFA | 0 | 0.050 | 0 |
| 6 | TFA | 0 | 0.100 | 0 |
| 7 | TSA | 0 | 0.010 | 0 |
| 8 | TSA | 0 | 0.050 | 6 |
| 9 | TSA | 0 | 0.100 | 16 |
| 10 | TSA | 0.1 | 0.050 | 10 |
| 11 | TSA | 1.0 | 0.050 | 20 |
| 12 | TSA | 1.0 | 0.100 | 30 |
| 13 | TSA | 1.0 | 0.200 | 21 |
Using 1.0 eq. of TSA, we observed formation of porphyrin 11a with 6% isolated yield at c = 0.05 M concentration (Table 6, entry 8) and 16% at c = 0.1 M (Table 6, entry 9); at c = 0.01 M (typical batch concentration) porphyrin 11a was not isolated (entry 9). The low solubility of TSA in DCM was a significant limitation in these experiments. Hence, we examined the influence of methanol as a solubilising additive. A significant increase in isolated yield, from 6 to 10%, was observed when 0.1% (v/v) of MeOH was used (entries 8 and 10). The yield was increased 2-fold when 1% of MeOH was used (20%, entry 11). With these satisfactory results, we assessed the effect of the concentration of reactants on the yield, keeping in mind our aim to reduce the volume of chlorinated solvents required for porphyrin synthesis. Pleasingly, at c = 0.1 M porphyrin 11a was isolated in 30% yield (entry 12), but at c = 0.2 M the yield dropped to 21% (entry 13). Hence, the optimised conditions for the synthesis of dodecasubstituted porphyrins involve TSA as acid catalyst, c = 0.1 M in DCM/MeOH, 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v and 1.0 mL min−1 flow rate. As p-toluenesulfonic acid has not been used in the synthesis of dodceca substituted porphyrins to date, we carried out batch experiments using TSA for formation of 2,3,7,8,12,13,17,18-octaethyl-5,10,15,20-tetraphenylporphyrin (11a, H2OETPP); however no macrocycle was isolated at low (c = 0.01 M) or high (c = 0.1 M) concentrations.
1, v/v and 1.0 mL min−1 flow rate. As p-toluenesulfonic acid has not been used in the synthesis of dodceca substituted porphyrins to date, we carried out batch experiments using TSA for formation of 2,3,7,8,12,13,17,18-octaethyl-5,10,15,20-tetraphenylporphyrin (11a, H2OETPP); however no macrocycle was isolated at low (c = 0.01 M) or high (c = 0.1 M) concentrations.
Following the TSA-catalyzed condensation, we synthesised a library of ten dodecasubstituted porphyrins from a range of aldehydes and 3,4-diethylpyrrole 9 (Scheme 6). The highest yield in this library was observed for porphyrin 11e (45%), 2% higher than previously reported in literature76a and 19% higher than the corresponding planar porphyrin 4e (26%, Scheme 2) under flow conditions. Electronically similar H2OETPP 11a and octaethyltetra-p-tolylporphyrin 11b were isolated in 30 and 29% yields, respectively, with the latter having no comparable value in literature.76b Both meta-disubstituted 3,5-dimethoxybenzaldehyde 2r and 3,5-dibromobenzaldehyde 2s produced the corresponding porphyrins 11r and 11s in good yields (29 and 35%, respectively). However, the condensation yield determined for the reaction with 3,5-di-tert-butylbenzaldehyde 2q was only 6% for 11q, a yield 10% lower than reported for batch synthesis.76c Similarly, p-tert-butylbenzaldehyde 2d only produced 4% of the dication salt of porphyrin 11d. 11d was the only porphyrin in this series isolated as a dication, possibly due to an increased basicity of the porphyrin core due to activating electronic effects. p- And m-bromobenzaldehydes 2c and 2h yielded 20% and 19% of 11c and 11h, respectively the nonplanar meta-derivative was once again obtained in a higher yield than the planar 4h (7%, Scheme 2); the para-derivative with an isolated with a yield 13% lower than reported for batch conditions.76d On the other hand, o-bromobenzaldehyde 2k was the poorest starting material in this library, with only a 2% isolated yield for 11k. This is likely due to the increased steric hindrance near the reacting atoms.39 Alkyl groups substituting pyrrole Cβ positions may augment this effect significantly; 11k was the only porphyrin isolated out of five ortho-substituted aldehydes investigated. Alkyl aldehydes 2ad and 2ae, furfural 2w and p-nitrobenzaldehyde 2f, and ortho-hindered benzaldehydes 2m, 2n, 2p failed to produce the corresponding porphyrins 11f, 11m, 11n, 11p, 11w, 11ad and 11ae (Scheme 6), contrary to the results reported for batch synthesis.72
 | ||
| Scheme 6 Scope and limitations of CSTR-assisted synthesis of dodecasubstituted porphyrins. Yields in grey were reported in literature for batch syntheses.76 aYield not reported in literature. bCompound isolated as a dication salt. cFor porphyrins 11a and 11k, single crystal X-ray structures were obtained, full refinement details and analysis are provided in the SI (CCDC: 2521006 and 2521007). | ||
E-Factors
To validate our methods as sustainable, we calculated E-factor (environmental impact factor)77 for the protocols presented in this manuscript and compare them with corresponding values determined for batch protocols. E-Factor is one of the key descriptors used to assess the sustainability of a chemical reaction, considering the effectiveness in product formation over waste produced in the process. We selected E-factor as a measure of success in achieving our goal, which was to develop a sustainable and versatile flow-assisted protocol for porphyrin synthesis, reducing the volume of chlorinated solvent required for cyclisation.In eqn (1) below, waste includes unreacted starting materials, catalysts, solvents, side products formed over the course of the reaction and materials and chemicals used in the work-up. Detailed analysis of waste contributions is disclosed in SI (Table S1).
 | (1) |
Table 7 presents a comparison of E-factors calculated for protocols developed in this project and the corresponding values obtained for literature batch protocols. All values are presented for 4a (H2TPP), 7a (H2DPP) and 11a (H2OETPP) syntheses as examples. The range of E-factor values for fine chemicals in industry is 5–50.77a In porphyrin chemistry, the E-factor values for traditional synthesis of 4a (H2TPP) range between 158 for Alder–Longo method and 2252 for Lindsey method.78 Porphyrin synthesis is an environmentally taxing process, which is in part due to high dilutions required for macrocyclisation, combined with the side reactions predominant in acidic conditions.79 We circumvented some of the problems associated with the synthesis of meso-substituted porphyrins. By incorporating flow chemistry at the condensation step, we successfully accessed higher global concentration of reactants, which directly translates to lower demand for chlorinated solvents, and decreasing the volume of waste produced in the process. This effect is quantified by E-factor. The comparison of E-factors for H2TPP 4a, H2DPP 7a and H2OETPP 11a synthesis in flow and in batch shows significant improvement for all three methods presented in this contribution. The E values were reduced by 48% for 4a (from 2252 to 1180), 38% for 7a (from 2282 to 1416) and 35% for 11a (from 2015 to 1314). The reaction yield, type of acid catalyst, oxidant and possible additives have negligible influence on the E-factor (Table S1); the major contribution comes from the mass of solvents used for the synthesis and purification. Hence, the 90% decrease in solvent volume required for porphyrin synthesis in our method directly translates to significant improvements in sustainability parameters.
| Porphyrin | 4a | 7a | 11a | |||
|---|---|---|---|---|---|---|
| Method | Flow | Batch | Flow | Batch | Flow | Batch |
| Scale [mmol] | 2.25 | 0.75 | 1.10 | |||
| Waste [g] | 130 | 495 | 85 | 251 | 92 | 262 |
| Product [g] | 0.11 | 0.22 | 0.06 | 0.11 | 0.07 | 0.13 |
| E-Factor77 | 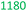 |
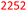 |
 |
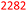 |
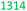 |
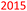 |
Crystallography
Many examples of X-ray crystal structures of dodecasubstituted nonplanar porphyrins are found in the literature.74,76,80 However, many H2OETAP derivatives are often reported in their dicationic form with just one other example of a ‘free base’ OETAP being previously reported (11a, crystallised from ethanolic KOH).81 Here, the X-ray crystal structures of the ‘free base’ dodecasubstituted porphyrins 11a (from deuterated benzene) and 11k are reported, with 11k adopting the α3β atropisomeric conformation (Fig. 3).82Analysis of the view of the molecular structures in the crystal of 11a and 11k reveals the expected saddled shaped (B2u) nonplanar distortion. For porphyrin 11k intramolecular hydrogen bonding interactions (2.29–2.34 Å) between both opposing enamine hydrogen atoms (N-21 and N-23) and the adjacent imine nitrogen atoms (N-22) are observed. A preferential hydrogen bonding of N-24 with the hydrogen atom of the CHCl3 solvate is noted (Fig. 3). This solvent binding event is not observed in the case of 11a where enamine hydrogen atoms (N-21 and N-23) hydrogen bond (2.29–2.31 Å) to both adjacent imine nitrogen's (N-22 and N-24).
In contrast to the X-ray crystal structure of 11k, porphyrin 11a by when analyzed by NSD analysis has 3.4° greater tilt in the pyrroles from the mean porphyrin plane. Additionally, the total Δoop distortion of the ‘free base’ analogue (defined as the average deviation of the 24-atom macrocycle with respect to the mean porphyrin plane, Δz = 0)83 is 3.48 Å in porphyrin 11a compared to 3.13 Å in 11k.
Comparing the crystal structure of ‘free base’ 11k with the analogous Ni(II) metalated cis-α2β2 atropisomeric derivative of 11k from literature76e is difficult as conformationally these two porphyrins are quite different, nonetheless, the pyrrole tilt angle for the ‘free base’ 11k is 26° almost 3° less than the Ni(II) derivative. The total Δoop distortion of the ‘free base’ analogue is 0.64 Å less compared to the Ni(II) counterpart with a total Δoop distortion of 3.77 Å. Nevertheless, it is very difficult to make a direct comparison between the two structures due to disparate atropisomeric conformations as well differing crystallisation76d and crystal packing effects.84
Conclusions
In conclusion, we designed and optimised flow-based synthetic methods for meso-substituted porphyrin synthesis and demonstrated their applicability to three libraries of porphyrins: A4, trans-A2B2 (including A2) and dodecasubstituted A4 porphyrins. These sustainable methods were optimised to operate at significantly higher reactant concentrations than traditional batch methods, leading to a dramatic decrease in the volume of chlorinated solvents required for porphyrin synthesis (up to 90% solvent saving), and a significant improvement in E-factors over batch conditions (up to 48% reduction of E values). The synthesis of 40 porphyrins is disclosed, alongside their structural details. We hope that this contribution proves useful to porphyrin chemists across the globe and prompts the use of sustainable protocols in porphyrin and other chemical laboratories.Author contributions
Karolina Urbańska – conceptualisation, funding acquisition, methodology, investigation, project administration, writing – original draft; Liam Cribbin – conceptualisation, funding acquisition, investigation, writing – original draft; Brendan Twamley – investigation, formal analysis; Fiach O Ceallaigh – investigation; Mathias O. Senge – resources, supervision, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article is included as part of the supplementary information (SI) (compound characterisation, NMR, HRMS, UV/vis and IR spectra and refinement details). References cited in the SI (ref. 85–118) are listed in the article reference list. Supplementary information: compound characterisation data, spectra, CIF files, E-factor calculation. See DOI: https://doi.org/10.1039/d6su00176a.CCDC 2521006 (11a) and 2521007 (11k) contain the supplementary crystallographic data for this paper.119a,b
Acknowledgements
This project has received funding from Royal Society of Chemistry under Sustainable Laboratories Grant (award L23-8636817163) and from Taighde Éireann – Research Ireland (award 21/FFP-A/9469, PORPHYSHAPE).Notes and references
- P. T. Anastas and J. C. Warner, Green Chemistry: Theory and Practice, Oxford University Press, New York, 1998 Search PubMed.
- A. Halpaap, Green and Sustainable Chemistry: Framework Manual, The United Nations, 2020 Search PubMed.
- D. J. C. Constable, P. J. Dunn, J. D. Hayler, G. R. Humphrey, J. J. L. Leazer, R. J. Linderman, K. Lorenz, J. Manley, B. A. Pearlman, A. Wells, A. Zak and T. Y. Zhang, Green Chem., 2007, 9, 411–420 Search PubMed.
- A. Jordan, P. Stoy and H. F. Sneddon, Chem. Rev., 2021, 121, 1582–1622 CrossRef CAS PubMed.
- Regulation (EC) No 1907/2006 of the European Parliament and of the Council of 18 December 2006 concerning the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH), establishing a European Chemicals Agency, amending Directive 1999/45/EC and repealing Council Regulation (EEC) No 793/93 and Commission Regulation (EC) No 1488/94 as well as Council Directive 76/769/EEC and Commission Directives 91/155/EEC, 93/67/EEC, 93/105/EC and 2000/21/EC.
- United States Environmental Protection Agency, Fed. Regist., 2024, 89, 39254 Search PubMed.
- F. Davis and S. Higson, Macrocycles: Construction, Chemistry, and Nanotechnology Applications, John Wiley & Sons, 2011 Search PubMed.
- A. K. Yudin, Chem. Sci., 2015, 6, 30–49 RSC.
- A. C. Bédard, S. Régnier and S. K. Collins, Green Chem., 2013, 15, 1962–1966 RSC.
- Q. Fan, T. Wang, J. Dai, J. Kuttner, G. Hilt, J. M. Gottfried and J. Zhu, ACS Nano, 2017, 11, 5070–5079 CrossRef CAS PubMed.
- F. Garnes-Portolés and A. Leyva-Pérez, ACS Catal., 2023, 13, 9415–9426 CrossRef.
- P. J. Brothers and M. O. Senge, Fundamentals of Porphyrin Chemistry, Wiley, 2022 Search PubMed.
- L. Feng, K. Y. Wang, E. Joseph and H. C. Zhou, Trends Chem., 2020, 2(6), 555–568 CrossRef CAS.
- H. Lee, K.-I. Hong and W.-D. Jang, Coord. Chem. Rev., 2018, 354, 46–73 CrossRef CAS.
- (a) F. Santanni and A. Privitera, Adv. Opt. Mater., 2024, 12(18), 2303036 CrossRef CAS; (b) D. Ranieri, A. Privetera, F. Santanni, K. Urbanska, G. J. Strachan, B. Twamley, E. Salvadori, Y. K. Liao, M. Chiesa, M. O. Senge, F. Totti, L. Sorace and R. Sessoli, Angew. Chem., Int. Ed., 2023, 62(48), e202312936 CrossRef CAS PubMed.
- H. Lee, H. Park, D. Y. Ryu and W. D. Jang, Chem. Soc. Rev., 2023, 52, 1947–1974 RSC.
- W. M. Campbell, A. K. Burrell, D. L. Officer and K. W. Jolley, Coord. Chem. Rev., 2004, 248, 1363–1379 CrossRef CAS.
- R. Das, P. Kumar Verma and C. M. Nagaraja, Coord. Chem. Rev., 2024, 514, 215944 CrossRef CAS.
- D. Xu, Q. Duan, H. Yu and W. Dong, J. Mater. Chem. B, 2023, 11, 5976–5989 RSC.
- J. Chen, Y. Zhu and S. Kaskel, Angew. Chem., Int. Ed., 2021, 60(10), 5010–5035 CrossRef CAS PubMed.
- D. Dolphin, J. Heterocycl. Chem., 1970, 7, 275–283 CrossRef CAS.
- P. Rothemund and A. R. Menotti, J. Am. Chem. Soc., 1941, 63, 267–270 CrossRef CAS.
- A. D. Adler, F. R. Longo and W. Shergalis, J. Am. Chem. Soc., 1964, 86, 3145–3149 CrossRef CAS.
- J. S. Lindsey, H. C. Hsu and I. C. Schreiman, Tetrahedron Lett., 1986, 27, 4969–4970 CrossRef CAS.
- K. M. Smith, New J. Chem., 2016, 40, 5644–5649 RSC.
- M. O. Senge, N. N. Sergeeva and K. J. Hale, Chem. Soc. Rev., 2021, 50, 4730–4789 RSC.
- T. Ishizuka, N. Grover, C. J. Kingsbury, H. Kotani, M. O. Senge and T. Kojima, Chem. Soc. Rev., 2022, 51, 7560–7630 RSC.
- M. O. Senge, Chem. Commun., 2006, 243–256 RSC.
- B. Habermeyer and R. Guilard, Photochem. Photobiol. Sci., 2018, 17, 1675–1690 CrossRef CAS PubMed.
- P. Ruggli, Adv. Cycloaddit., 1912, 392, 92–100 CAS.
- K. Ziegler, H. Eberle and H. Ohlinger, Adv. Cycloaddit., 1933, 504, 94–130 CAS.
- S. M. A. Pinto, C. A. Henriques, V. A. Tomé, C. S. Vinagreiro, M. J. F. Calvete, J. M. Dąbrowski, M. Piñeiro, L. G. Arnaut, M. M. Pereira and J. Porphyrins, Phthalocyanines, 2016, 20, 45–60 CrossRef CAS.
- A. M. d'A Rocha Gonsalves, J. M. T. B. Varejão and M. M. Pereira, J. Heterocycl. Chem., 1991, 28, 635–640 CrossRef.
- P. B. Momo, B. S. Bellete, T. J. Brocksom, R. O. M. A. de Souza and K. T. de Oliveira, RSC Adv., 2015, 5, 84350–84355 RSC.
- F. Parveen, H. J. Morris, H. West and A. G. Slater, J. Flow Chem., 2024, 14, 23–31 CrossRef CAS.
- T. E. O. Screen, K. B. Lawton, G. S. Wilson, N. Dolney, R. Ispasoiu, T. Goodson III, S. J. Martin, D. D. C. Bradley and H. L. Anderson, J. Mater. Chem., 2001, 11, 312–320 RSC.
- O. Yadav, A. Varshney and A. Kumar, Inorg. Chem. Commun., 2017, 86, 168–171 CrossRef CAS.
- S. D. Roth, T. Shkindel and D. A. Lightner, Tetrahedron, 2007, 63, 11030–11039 CrossRef CAS PubMed.
- In a recent paper by Parveen et al. (ref. 35), the authors designed a fully continuous process with the steps of condensation, oxidation and neutralisation arranged in three consecutive coil reactors.
- J. S. Lindsey, I. C. Schreiman, H. C. Hsu, P. C. Kearney and A. M. Marguerettaz, J. Org. Chem., 1987, 52, 827–836 CrossRef CAS.
- Regulation (EC) No 1272/2008 of the European Parliament and of the Council of 16 December 2008 on classification, labelling and packaging of substances and mixtures, amending and repealing Directives 67/548/EEC and 1999/45/EC, and amending Regulation (EC) No 1907/2006.
- (a) Merck Life Sciences Ltd, Chloroform, Safety Data Sheet, Version 8.9, Revision date 25.12.2025, https://www.sigmaaldrich.com/IE/en/sds/SIAL/288306 Search PubMed; (b) Merck Life Sciences Ltd, Dichloromethane, Safety Data Sheet, Version 8.12, Revision date 23.12.2025, https://www.sigmaaldrich.com/IE/en/sds/SIAL/270997 Search PubMed.
- (a) J. B. Kim, J. J. Leonard and F. R. Longo, J. Am. Chem. Soc., 1972, 94, 3986–3992 CrossRef CAS PubMed; (b) J. S. Lindsey and R. W. Wagner, J. Org. Chem., 1989, 54, 828–836 CrossRef CAS.
- (a) G. J. Capitosti, C. D. Guerrero, D. E. Binkley Jr, C. S. Rajesh and D. A. Modarelli, J. Org. Chem., 2003, 68, 247–261 CrossRef CAS PubMed; (b) I. Yslas, M. G. Alvarez, C. Marty, G. Mori, E. N. Durantini and V. Rivarola, Toxicology, 2000, 149, 69–74 CrossRef CAS PubMed; (c) C. A. Hunter, M. C. Misuraca and S. M. Turega, J. Am. Chem. Soc., 2011, 133, 582–594 Search PubMed; (d) D. J. Walaszek, K. Maximova, K. Rybicka-Jasińska, A. Lipke and D. Gryko, J. Porphyrins Phthalocyanines, 2014, 18, 493–505 CrossRef CAS; (e) Y. Ishimizu, Z. Ma, M. Hada and H. Fujii, Inorg. Chem., 2021, 60, 17687–17698 CrossRef CAS PubMed; (f) M. Blanco, M. Lunardon, M. Bortoli, D. Mosconi, L. Girardi, L. Orian, S. Agnoli and G. Granozzi, J. Mater. Chem. A, 2020, 8, 11019–11030 RSC; (g) D. Oulmi, P. Maillard, J.-L. Guerquin-Kern, C. Huel and M. Momenteau, J. Org. Chem., 1995, 60, 1554–1564 CrossRef CAS; (h) A. C. Kaelin and G. D. Zanelli, J. Labelled Compd. Radiopharm., 1990, 28, 343–354 CrossRef CAS; (i) L. Huang, Y. Chen, G.-Y. Gao and X. P. Zhang, J. Org. Chem., 2003, 68, 8179–8184 CrossRef CAS PubMed; (j) D. Yadav, A. Kumar, J. Y. Kim, N.-J. Park and J.-O. Baeg, J. Mater. Chem. A, 2021, 9, 9573–9580 RSC; (k) R. Soman, D. Raghav, S. Sujatha, K. Rathinasamy and C. Arunkumar, RSC Adv., 2015, 5, 61103–61117 RSC.
- J. W. Dirks, G. Underwood, J. C. Matheson and D. Gust, J. Org. Chem., 1979, 44, 2551–2555 CrossRef CAS.
- N. Sheng, B. Gu, B. Ren, J. Zhang, Y. Wang, J. Wang and J. Sha, Dyes Pigm., 2017, 142, 116–120 CrossRef CAS.
- I. Radivojevic, I. Likhtina, X. Shi, S. Singh and C. M. Drain, Chem. Commun., 2010, 46, 1643–1645 RSC.
- J. M. Cense and R.-M. Le Quan, Tetrahedron Lett., 1979, 3725–3728 CrossRef CAS.
- A. Treibs and N. Häberle, Liebigs Ann. Chem., 1968, 718, 183–207 CrossRef CAS PubMed.
- H. Volz and G. Herb, Z. Naturforsch., B: Anorg. Chem., Org. Chem., 1984, 39, 1393–1398 CrossRef.
- M. Baljozović, J. Pijeat, S. Campidelli and K.-H. Ernst, J. Am. Chem. Soc., 2024, 146, 34600–34608 CrossRef PubMed.
- M. O. Senge, I. Bischoff, N. Y. Nelson and K. M. Smith, J. Porphyr. Phthalocyanines, 1999, 3, 99–116 CrossRef CAS.
- M. M. Martin, C. Oleszak, F. Hampel and N. Jux, Eur. J. Org Chem., 2020, 2020, 6758–6762 CrossRef CAS.
- I. Gupta and M. Ravikanth, Tetrahedron, 2003, 59, 6131–6139 CrossRef CAS.
- T. Ema, M. O. Senge, N. Y. Nelson, H. Ogoshi and K. M. Smith, Angew. Chem., Int. Ed., 1994, 33, 1879–1881 CrossRef.
- V. V. Borovkov, M. Anikin, K. Wasa and Y. Sakata, Photochem. Photobiol., 1996, 63, 477–482 CrossRef CAS.
- S. Banfi, E. Caruso, S. Caprioli, L. Mazzagatti, G. Canti, R. Ravizza, M. Gariboldi and E. Monti, Bioorg. Med. Chem., 2004, 12, 4853–4860 CrossRef CAS PubMed.
- G. D. Carlo, A. O. Biroli, M. Pizzotti, F. Tessore, V. Trifiletti, R. Ruffo, A. Abbotto, A. Amat, F. De Angelis and P. R. Mussini, Chem.–Eur. J., 2013, 19, 10723–10740 CrossRef PubMed.
- G. R. Geier III, B. J. Littler and J. S. Lindsey, J. Chem. Soc., Perkin Trans. 2, 2001,(5), 701–711 RSC.
- K. S. Suslick, C. T. Chen, G. R. Meredith and L. T. Cheng, J. Am. Chem. Soc., 1992, 114, 6928–6930 CrossRef CAS.
- M. J. A. Reis, A. M. V. M. Pereira, N. M. M. Moura and M. G. P. M. S. Neves, ACS Omega, 2024, 9, 31196–31219 CrossRef CAS PubMed.
- D. Pan, P. Liang, X. Zhong, D. Wang, H. Cao, W. Wang, W. He, Z. Yang and X. Dong, ACS Appl. Bio Mater., 2019, 2, 999–1005 CrossRef CAS PubMed.
- G. P. Arsenault, E. Bullock and S. F. MacDonald, J. Am. Chem. Soc., 1960, 82, 4384–4389 CrossRef CAS.
- J. Jökel, F. Schwer, M. von Delius and U. P. Apfel, Chem. Commun., 2020, 56, 14179–14182 RSC.
- (a) X. Jiang, T. Zhang and F. Gou, Mol. Catal., 2024, 555, 113865 CAS; (b) S. Arrechea, A. Molina-Ontoria, A. Aljarilla, P. de la Cruz, F. Langa and L. Echegoyen, Dyes Pigm., 2015, 121, 109–117 CrossRef CAS; (c) C. Schissler, E. K. Schneider, S. Lebedkin, P. Weis, G. Niedner-Schatteburg, M. M. Kappes and S. Bräse, Chem.–Eur. J., 2021, 27, 15202–15208 CrossRef PubMed; (d) M. C. Kuo, H. F. Chen, J. J. Shyue, D. M. Bassani and K. T. Wong, Chem. Commun., 2012, 48, 8051–8053 RSC; (e) M. Handayani, S. Gohda, D. Tanaka and T. Ogawa, Chem.–Eur. J., 2014, 20, 7655–7664 CrossRef CAS PubMed; (f) S. M. S. Ló, D. R. B. Ducatti, M. E. R. Duarte, S. M. W. Barreira, M. D. Noseda and A. G. Gonçalves, Tetrahedron Lett., 2011, 52, 1441–1443 CrossRef; (g) R. Elliott, A. A. Ryan, A. Aggarwal, N. Zhu, F. W. Steuber, M. O. Senge and W. Schmitt, Molecules, 2021, 26, 2955 CrossRef CAS PubMed; (h) Y. Xie, F. Zhang, P. Liu, F. Hao and H. a. Luo, Can. J. Chem., 2014, 92, 49–53 Search PubMed; (i) S. M. Kuzmin, S. A. Chulovskaya and V. I. Parfenyuk, J. Electroanal. Chem., 2023, 943, 117594 CrossRef CAS; (j) J. Deyerling, B. Berionni Berna, D. Biloborodov, F. Haag, S. Tömekce, M. G. Cuxart, C. Li, W. Auwärter and D. Bonifazi, Angew. Chem., Int. Ed., 2025, 64, e202412978 CrossRef CAS PubMed.
- Y. B. She, H. Y. Fu, Z. L. Zhang, X. F. Song and Z. C. Sun, Heterocycles, 2014, 89, 503–514 CrossRef.
- K. Norvaiša, K. J. Flanagan, D. Gibbons and M. O. Senge, Angew. Chem., Int. Ed., 2019, 58, 16553–16557 CrossRef PubMed.
- M. Roucan, M. Kielmann, S. J. Connon, S. S. R. Bernhard and M. O. Senge, Chem. Commun., 2018, 54, 26–29 RSC.
- T. Ishizuka, S. Ohkawa, H. Ochiai, M. Hashimoto, K. Ohkubo, H. Kotani, M. Sadakane, S. Fukuzumi and T. Kojima, Green Chem., 2018, 20, 1975–1980 RSC.
- Only 434 Records Found for “Nonplanar Porphyrin” in a Scopus Search, Compared to 82,358 Records for the Keyword “Porphyrin”, Making up <1% of the Field, (accessed January 2026) Search PubMed.
- B. Evans, K. M. Smith and J. H. Fuhrhop, Tetrahedron Lett., 1977, 18, 443–446 CrossRef.
- (a) A. Neuberger and J. J. Scott, Proc. R. Soc. London, Ser. A, 1952, 213, 307–326 CrossRef CAS; (b) O. S. Finikova, A. V. Cheprakov, P. J. Carroll, S. Dalosto and S. A. Vinogradov, Inorg. Chem., 2002, 41, 6944–6946 CrossRef CAS PubMed.
- M. Kielmann and M. O. Senge, Angew. Chem., Int. Ed., 2019, 58, 418–441 CrossRef CAS PubMed.
- M. O. Senge, T. P. Forsyth, L. T. Nguyen and K. M. Smith, Angew. Chem., Int. Ed., 1995, 33, 2485–2487 CrossRef.
- M. O. Senge and W. W. Kalisch, Z. Naturforsch., 1999, 54b, 943–959 CrossRef.
- (a) Yu. B. Ivanova, N. Zh. Mamardashvili, A. V. Glazunov and A. S. Semeikin, Russ. J. Gen. Chem., 2014, 84, 1404–1410 CrossRef CAS; (b) E. I. Sagun and E. I. Zenkevich, Opt. Spectrosc., 2013, 115, 727–738 CrossRef CAS; (c) K. Norvaiša, K. Yeow, B. Twamley, M. Roucan and M. O. Senge, Eur. J. Org Chem., 2021, 2021, 1871–1882 CrossRef PubMed; (d) K. J. Flanagan, B. Twamley and M. O. Senge, Inorg. Chem., 2019, 58, 15769–15787 CrossRef CAS; (e) L. Cribbin, B. Twamley, N. Buga, J. E. O'Brien, R. Bühler, R. A. Fischer and M. O. Senge, Beilstein J. Org. Chem., 2025, 20, 2784–2798 CrossRef PubMed; (f) K. M. Barkigia, M. D. Berber, J. Fajer, C. J. Medforth, M. W. Renner and K. M. Smith, J. Am. Chem. Soc., 1990, 112, 8851–8857 CrossRef CAS; (g) K. M. Kadish, M. Lin, E. V. Caemelbecke, G. De Stefano, C. J. Medforth, D. J. Nurco, N. Y. Nelson, B. Krattinger, C. M. Muzzi, L. Jaquinod, Y. Xu, D. C. Shyr, K. M. Smith and J. A. Shelnutt, Inorg. Chem., 2002, 41, 6673–6687 CrossRef CAS PubMed; (h) A. Hoshino, Y. Ohgo and M. Nakamura, Tetrahedron, 2005, 46, 4961–4964 Search PubMed.
- (a) R. A. Sheldon, Chem. Ind., 1992, 903–906 CAS; (b) R. A. Sheldon, J. Chem. Technol. Biotechnol., 1997, 68, 381–388 CrossRef CAS.
- C. A. Henriques, S. M. A. Pinto, G. L. B. Aquino, M. Pineiro, M. J. F. Calvete and M. M. Pereira, ChemSusChem, 2014, 7(10), 2821–2824 CrossRef CAS PubMed.
- H. Hölzel, M. Muth, D. Lungerich and N. Jux, Chem.:Methods, 2021, 1, 142–147 Search PubMed.
- C. J. Kingsbury, M. Kielmann, B. Twamley and M. O. Senge, Molecules, 2022, 27, 7060 CrossRef CAS PubMed.
- A. Regev, T. Galili, C. J. Medforth, K. M. Smith, K. M. Barkigia, J. Fajer and H. Levanon, J. Phys. Chem., 1994, 98, 2520–2526 CrossRef CAS.
- S. Maguire, G. Strachan, K. Norvaiša, C. Donohue, L. Gomes-da-Silva and M. O. Senge, Chem.–Eur. J., 2024, 30, e202401559 CrossRef CAS PubMed.
- W. Jentzen, X.-Z. Song and J. A. Shelnutt, J. Phys. Chem. B, 1997, 101, 1684–1699 CrossRef CAS.
- W. R. Scheidt and Y. J. Lee, in Structure and Bonding, ed. J. W. Buchler, Springer-Verlag, Berlin, 1987, vol. 64, p. 1 Search PubMed.
- R. A. W. Johnstone, M. L. P. G. Nunes, M. M. Pereira, A. M. d'A, R. Gonsalves and A. C. Serra, Heterocycles, 1996, 43, 1423–1437 CrossRef CAS.
- F. Liu, L. Duan, Y. Wang, Q. Zhang and J. Wang, Synth. Commun., 2009, 39, 3990–3998 CrossRef CAS.
- O. V. Malkova, V. G. Andrianov, B. D. Berezin, A. S. Semeikin and I. I. Zhilina, Zh. Org. Khim., 1988, 24, 1546–1550 CAS.
- G. Mele, R. D. Sole, G. Vasapollo, E. Garcia-Lopez, L. Palmisano and M. Schiavello, J. Catal., 2003, 217, 334–342 CrossRef CAS.
- Y. Naruta, F. Tani, N. Ishihara and K. Maruyama, J. Am. Chem. Soc., 1991, 113, 6865–6872 CrossRef CAS.
- M. Yaseen, M. Ali, M. N. Ullah, M. A. Munawar and I. Khokbar, J. Heterocycl. Chem., 2009, 46, 251–255 CrossRef CAS.
- H. Lv, B. Yang, J. Jing, Y. Yu, J. Zhang and J. L. Zhang, Dalton Trans., 2012, 41, 3116–3118 RSC.
- I. Gupta and M. Ravikanth, Tetrahedron Lett., 2002, 43, 9453–9455 CrossRef CAS.
- C. H. da Silveira, E. N. Garoforo, O. A. Chaves, P. F. B. Goncalves, L. Streit and B. A. Iglesias, Inorg. Chim. Acta, 2018, 482, 542–553 CrossRef.
- P. Kus, G. Knerr and L. Czuchjakowski, J. Heterocycl. Chem., 1990, 27(4), 1161–1166 CrossRef CAS.
- T. Zoltan, F. Vargas, C. Rivas, V. Lopez, J. Perez and A. Biasutto, Sci. Pharm., 2010, 78, 767–790 CrossRef CAS PubMed.
- N. Sheng, P. H. Zhu, C. Q. Ma and J. Z. Jinag, Dyes Pigm., 2009, 81, 91–96 CrossRef CAS.
- R. Plamont, Y. Kikkawa, M. Takasahi, M. Kanesato, M. Giorgi, A. C. K. Shun, C. Roussel and T. S. Balaban, Chem.–Eur. J., 2013, 19, 11293–11300 CrossRef CAS PubMed.
- A. Auger, A. J. Muller and J. C. Swarts, Dalton Trans., 2007, 33, 3623–3633 RSC.
- J. S. Manka and D. S. Lawrence, Tetrahedron Lett., 1989, 30, 6989–6992 Search PubMed.
- C. Ryppa, M. O. Senge, S. S. Hatscher, E. Kleinpeter, P. Wacker, U. Schilde and A. Wiehe, Chem.–Eur. J., 2005, 11, 3427–3442 CrossRef CAS PubMed.
- D. Bonifazi, G. Accorsi, N. Armaroli, F. Song, A. Palkar, L. Echegoyen, M. Scholl, P. Seiler, B. Jaun and F. Diedrich, Helv. Chim. Acta, 2005, 88(7), 1839–1884 CrossRef CAS.
- R. Shediac, M. H. B. Gray, H. T. Uyeda, R. C. Johnson, J. T. Hupp, P. J. Angiollo and M. J. Therien, J. Am. Chem. Soc., 2000, 122, 7017–7033 CrossRef CAS.
- X. Z. Song, W. Jentzen, S. L. Jia, L. Jaquinod, D. J. Nurco, C. J. Medforth, K. M. Smith and J. A. Shelnutt, J. Am. Chem. Soc., 1996, 118, 12975–12988 CrossRef CAS.
- B. Temelli and C. Unaleroglu, Tetrahedron, 2009, 65, 2043–2050 CrossRef CAS.
- W. Anannarukan, S. Tantayanon, D. Zhang, E. A. Alemán, D. A. Modarelli and F. W. Harris, Polymer, 2006, 47, 4936–4945 CrossRef CAS.
- M. B. Bakar, M. Oelgemöller and M. O. Senge, Tetrahedron, 2009, 65, 7064–7078 CrossRef CAS.
- N. Rana, S. Kumari and M. Sankar, Chem.–Asian J., 2025, 20, e202400719 CrossRef CAS PubMed.
- C. R. Jen, R. J. Cheng, C. C. Chen, G. H. Lee, S. M. Peng and P. P. Y. Chen, Inorg. Chem., 2014, 53, 8848–8850 CrossRef PubMed.
- H. Hope, in Progress in Inorganic Chemistry, ed. K. D. Karlin, John Wiley & Sons, 1994, vol. 41, pp. 1–19 Search PubMed.
- Bruker, SAINT, v8.41B, Bruker AXS SE, Karlsruhe, Germany, 2004 Search PubMed.
- L. Krause, R. Herbst-Irmer, G. M. Sheldrick and D. Stalke, J. Appl. Crystallogr., 2015, 48, 3–10 CrossRef CAS PubMed.
- G. M. Sheldrick, Acta Crystallogr., 2015, A71, 3–8 CrossRef PubMed.
- G. M. Sheldrick, Acta Crystallogr., 2015, C71, 3–8 CrossRef PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. J. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- C. J. Kingsbury and M. O. Senge, Coord. Chem. Rev., 2021, 431, 213760 CrossRef CAS.
- C. J. Kingsbury and M. O. Senge, Angew. Chem., Int. Ed., 2024, 63, e202403754 CrossRef CAS PubMed.
- C. R. Groom, I. J. Bruno, M. P. Lightfoot and S. C. Ward, Acta Crystallogr., 2016, B72, 171–179 Search PubMed.
- D. Kratzert, FinalCif., https://dkratzert.de/finalcif.html.
- (a) CCDC 2521006: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qm9sg; (b) CCDC 2521007: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qm9th.
| This journal is © The Royal Society of Chemistry 2026 |





