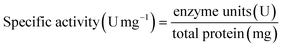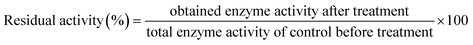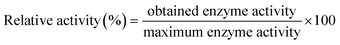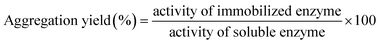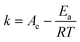Open Access Article
Open Access ArticleEnhanced catalytic turnover and robustness of laccase from Trametes pubescens through carrier-free aggregation
Afsheen Aman *a,
Muhammada,
Faiza Shahid
*a,
Muhammada,
Faiza Shahid b,
Asma Ansari
b,
Asma Ansari a and
Muhammad Sohail
a and
Muhammad Sohail c
c
aThe Karachi Institute of Biotechnology and Genetic Engineering (KIBGE), University of Karachi, Karachi 75270, Pakistan. E-mail: afaman@uok.edu.pk; Tel: +92-3332314423
bDow College of Biotechnology, Dow University of Health Sciences, Karachi 75270, Pakistan
cDepartment of Microbiology, University of Karachi, Karachi 75270, Pakistan
First published on 30th April 2026
Abstract
Green biocatalysis has revolutionized industrial processing by replacing whole-cell systems with robust immobilized biocatalysts, significantly streamlining downstream purification. This study reports the fabrication and biocatalytic characterization of carrier-free Crosslinked Enzyme Aggregates (CLEAs) using laccase produced by the white rot fungus Trametes pubescens through solid state fermentation. The resulting L-CLEAs exhibited superior thermal robustness and catalytic performance compared to the soluble laccase (SL). Kinetic analysis revealed that while the apparent substrate binding affinity (Km) increased 5.4-fold due to mass transfer limitations, this was also largely compensated for by a 4.1-fold enhancement in the catalytic rate constant (kcat). Furthermore, L-CLEAs demonstrated a significant 2.2-fold reduction in activation energy (Ea) and an increased inactivation energy (Ed), indicating a fortified structural conformation. Thermal stability assays confirmed that L-CLEAs nearly doubled the enzyme's half-life (t1/2) at 60 °C, extending it to 406 minutes. Moreover L-CLEAs also retained >80% activity after 20 consecutive cycles of use, demonstrating excellent operational durability and ease of recovery through simple centrifugation. These findings, coupled with exceptional reusability, position L-CLEAs as a highly efficient and stable hybrid system for the industrial degradation of phenolic compounds and other green chemistry applications.
Sustainability spotlightEnzymes are catalytic modalities that help in sustaining life by controlling metabolic systems. Reusing them multiple times, for the same purpose, assists in reducing their production cost and also minimizes carbon emission. Our goal was to establish a stable insoluble version of the enzyme (laccase) that could be recycled in order to cut down the biochemical processing industrial steps. Laccase is capable of oxidizing phenolic compounds; therefore, laccase based crosslinked enzyme aggregates (L-CLEAs) could be used to clean water-based contaminants. The present research work highlights the affordable and clean energy (SDG-7), industry, innovation and infrastructure (SDG-9) and, to some extent, the climate action (SDG-13) sustainable development goals. |
1 Introduction
Enzymes, due to their green nature, have found several industrial applications and subsequently have limited the use of conventional chemical catalysts for the manufacture of different types of metabolites. Microbial enzymes could either be utilized in soluble form or immobilized form depending upon their application. Numerous researchers have highlighted the need to overcome the barriers associated with the use of soluble or free enzymes as they cannot be easily recovered or reused multiple times, and they also have low stability. One of the alternative options developed to overcome the related issues was the use of enzyme immobilization technology where compatible carrier materials (carrier-bound matrices) were applied to restrain enzyme molecules on a solid support. With the passage of time, immobilization of biocatalysts on different types of organic or inorganic matrices has become one of the most explored areas in research. This carrier-bound technology was also found to be one of the most suitable alternative methods that would facilitate industries in overcoming the stability and reusability issues related to soluble enzymes. Immobilized single-enzymes, immobilized multiple-enzymes or immobilized microbial cells have been frequently investigated and used in academia and industry. However, the immobilization technique itself has also exhibited several limitations including loss in enzyme activity and stability because of the complex immobilization process and the type of matrix used. Cost and scalability are also some of the issues related to the use of immobilized enzymes in various sectors. There are several important aspects of immobilization that, if considered carefully, may result in improved implementation of this technique. Selection of a suitable immobilization method (physical adsorption, encapsulation, ionic bonding, covalent binding, entrapment and crosslinking techniques), the type of compatible support system used (inorganic material, biopolymers, synthetic polymers, and hydrogels), an efficient enzyme recovery process and improved kinetic properties of the immobilized enzyme are some of the parameters that could result in scalable applications of immobilized enzymes.To overcome the aforementioned challenges, a new carrier-free crosslinking immobilization technology was introduced where the main emphasis was on the improvement of enzyme's operational stability. Currently, there are many different types of crosslinking strategies on which researchers are extensively working and these include crosslinked enzyme aggregates (CLEAs), crosslinked enzyme crystals (CLECs), magnetic crosslinked enzyme aggregates (MCLEAs), porous crosslinked enzyme aggregates (PCLEAs) and combi- and multi-crosslinked enzyme aggregates (CCLEAs).1–8 While these strategies have been applied across various enzyme classes, the choice of immobilization technique is dictated by the specific primary amino acid sequence and structural motifs rather than the enzyme's functional classification. Among various oxidoreductases, laccase has been immobilized using some of the aforementioned methods using different types of matrices or supports. This is because laccases are reported to have high catalytic properties and are explored frequently in developing green applications for water remediation. It has been immobilized using hydrogels,9 magnetic nanoparticles5 and other solid supports10–12 and has also been studied in the form of CLEAs.13–15
As per the sustainable development goals (SDGs) by the United Nations, it has become imperative to explore and utilize different types of sustainable materials to support a green environment. This trend has also affected the use of sustainable biological materials like enzymes in various processing setups. In the pursuit of sustainable biocatalysis, the field of enzyme immobilization has increasingly prioritized carrier-free strategies. These matrix-less techniques, such as crosslinked enzyme aggregates, continue to be extensively exploited to maximize volumetric productivity and minimize the environmental footprint of industrial processes. Herein, a supportless enzyme immobilization approach is studied to support SDGs in industrial processing. CLEAs of laccase are considered as a versatile approach for the bioremediation of pollutants and are therefore currently being explored extensively;16 however, their kinetic performance has not been discussed in detail elsewhere. Another important aspect of utilizing CLEAs of laccase is their simplicity and robustness as this approach could be employed on crude enzymes without extra purification steps involved. Therefore, the current study reports its production from the white rot fungi Trametes pubescens MB89 through solid state fermentation, its partial purification and its fabrication into CLEAs. The kinetics of laccase based CLEAs are also studied in detail. Here we report, for the first time, the use of laccase from Trametes pubescens to create laccase-based crosslinked enzyme aggregates, which are subsequently fully characterized for kinetic studies.
2 Experimental
2.1 Chemicals and reagents
All chemicals used during the study were of analytical grade and were procured from different sources/vendors. ABTS [2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid)] disodium salt, 98% HPLC; kraft lignin (alkali); copper(II) sulfate pentahydrate (ACS grade) and poly(ethylene glycol)-6000 were purchased from Sigma Aldrich whereas Tween-80 and isopropanol were purchased from Scharlau. Glutaraldehyde (25%) aqueous solution was from Daejung, Korea.2.2 Production of laccase
Laccase was extracellularly produced by the white rot fungi Trametes pubescens MB89 through solid state fermentation for up to consecutive 10.0 days at 30 °C with pretreated sugarcane bagasse as a substrate matrix. Initially, the fungal strain was cultivated in a culture medium (100 ml) containing potato extract (30 ml: prepared from 50 g diced potatoes) and dextrose (0.5%).17 Sugarcane bagasse was collected from the local market in Karachi in July, 2020 and was initially subjected to extensive washing for the removal of dust particles and any other contaminating matter followed by drying at 100 °C. This oven dried sugarcane bagasse was crushed into small particles (100 µm) and the fungal strain, which was earlier cultivated in potato-dextrose liquid medium, was finally mixed with mineral salt medium containing 0.1% kraft lignin and 0.5 mM Cu2+ to stimulate laccase production as inducers for the extracellular production of laccase.2.3 Extraction of the crude enzyme and partial purification of fungal laccase
Crude laccase was extracted using one litre of 100 mM citrate phosphate buffer (pH-4.0) which was supplemented with Tween-80 (0.01%) to maximize recovery. Fungal biomass and the sugarcane bagasse were recovered by filtration through Whatman filter paper no. 1 followed by secondary filtration through a 0.45 µm membrane. The resulting cell free filtrate (crude laccase) was then utilized for enzyme assay and subsequent experiments.Crude laccase was partially purified using ammonium sulphate (70.0%) and was desalted through dialysis with a cutoff value of 2000 Daltons (Servapor®) against citrate phosphate buffer (100 mM, pH-4.0). The protein content in the desalted sample was equivalent to 1200 ± 110 units, with 1.3 ± 0.058 mg protein. This desalted enzyme was stored at −20 °C and was used for the characterization of the soluble enzyme as well as for the development of CLEAs.
2.4 Enzyme assay for soluble laccase and laccase-CLEAs
For the laccase assay, the partially purified soluble enzyme was used with a chromogenic substrate known as 2,2′-azinobis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS). ABTS solution (1.0 mM) was utilized as the working concentration for the enzyme assay, and it was mixed with laccase (10.0 µl) and the reaction mixture was kept at 50 °C for 5.0 minutes. The absorbance was measured at 420 nm. The same procedure was followed using 5.0 mg of the CLEAs for the enzyme activity for the immobilized enzyme at 50 °C but for 10.0 minutes.Total protein content in the samples was analyzed using Lowry's protocol.18 Bovine serum albumin (250 µg ml−1) was used as a standard protein.
2.5 Enzyme unit calculation
Laccase activity was determined spectrophotometrically by monitoring the oxidation of 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS). Enzyme units were calculated as described earlier19 using the following equation:where A is the absorbance measured at a wavelength of 420 nm; V is the total volume of the reaction mixture; t is the time in minutes used for the reaction; ε is the substrate molar extinction coefficient (ε = 36
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 M−1 cm−1) and Ve is the volume (ml) of enzyme used in the reaction.
000 M−1 cm−1) and Ve is the volume (ml) of enzyme used in the reaction.
One unit of laccase activity is defined as “the amount of enzyme that is required to oxidize 1.0 µM of ABTS into its radical ABTS+ form in one minute under standard assay conditions.”
Specific activity was calculated as:
Percent residual enzymatic activity was calculated as:
Percent relative enzymatic activity was calculated as:
2.6 Characterization of soluble laccase
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 60 to ensure that the enzymatic reaction remained within the linear range of the spectrophotometer. This dilution maintained absorbance values between 0.1 and 1.0 at 420 nm under standard assay conditions, accounting for the high catalytic rate of the soluble enzyme. The experimental parameters are detailed below:
60 to ensure that the enzymatic reaction remained within the linear range of the spectrophotometer. This dilution maintained absorbance values between 0.1 and 1.0 at 420 nm under standard assay conditions, accounting for the high catalytic rate of the soluble enzyme. The experimental parameters are detailed below:To determine the optimal reaction time for the conversion of ABTS to its radical cation (ABTS+), the reaction was monitored at 420 nm over 20 minutes at 05-minute intervals. With the reaction time subsequently fixed at 10 minutes, the effect of temperature was also evaluated between 30 °C and 70 °C. Substrate concentration was varied from 0.01 mM to 1.0 mM in 100 mM citrate phosphate buffer (pH 4.0). Finally, the effect of pH on soluble laccase activity was investigated using four buffer systems (all at 100 mM): citrate phosphate (pH-3.0 to pH-8.0), sodium acetate (pH-4.0 to pH-7.0), potassium phosphate (pH-6.0 to pH-9.0) and glycine–NaOH (pH-9.0 to pH-12.0).
2.7 Development of a laccase-based biocatalytic system (laccase-CLEAs)
Insoluble laccase based crosslinked enzyme aggregates (L-CLEAs) were developed by precipitating the soluble enzyme with an appropriate agent, followed by the addition of a crosslinker. Based on the preliminary precipitation experiments described above, ammonium sulfate was selected as the optimal precipitating agent for this process. However, for the development of laccase-CLEAs the concentration of this salt (50%, 60% and 70%; w/v) and the concentration of the crosslinking agent (glutaraldehyde: 50.0 mM to 150.0 mM) were varied to ensure effective development of laccase based insoluble enzyme aggregates. The crosslinking factors including reaction temperature (20 °C), shaking time (72.0 hours) and the agitation speed (150 rpm) were adopted as described earlier.20 After 72.0 hours, the synthesized insoluble enzyme aggregates (laccase-CLEAs) were harvested using centrifugation (2000×g) for 30.0 minutes at 4.0 °C. The supernatant was used to evaluate the remaining total protein content and the enzyme units that failed to aggregate during the crosslinking reaction. Laccase-CLEAs were washed multiple times with citrate phosphate buffer (pH 4.0, 100 mM) to remove unbound crosslinker molecules and the unbound soluble laccase. The total protein content and enzyme units were also calculated for the washes. After consecutive washing steps, laccase-CLEAs were subjected to drying using silica beads as a desiccant in a vacuum chamber at 4.0 °C. The developed laccase-CLEAs were stored at 4.0 °C for further characterization.2.8 Scanning electron microscopy
Surface topographical images of the developed laccase-CLEAs were captured at different magnification levels through a JSM 6389 A Jeol, Japan.2.9 Calculation of aggregation yield (%) and aggregation efficiency (%)
The aggregation yield represents the efficiency of the crosslinking process, quantifying the conversion of soluble laccase into insoluble aggregates. In this study, the yield was determined after 72 hours of incubation under regulated environmental parameters (20 °C and 150 rpm). The following equation was used:Whereas percent aggregation efficiency is the amount of protein concentration that has been successfully aggregated and is calculated as follows:
2.10 Characterization of crosslinked laccase aggregates (laccase-CLEAs)
After synthesizing functionally active laccase-CLEAs, they were characterized using kinetic performance by optimizing various crosslinking parameters and were compared with the soluble laccase.Catalytic reaction time interaction was also optimized for the substrate and the laccase-CLEAs for up to 20.0 minutes with an interval of 5.0 minutes. The optimized temperature for laccase-CLEAs was determined by performing the catalytic reaction in the range of 30 °C to 70 °C with an interval of 10 °C. ABTS concentration was varied in the range of 0.5 mM to 2.75 mM for the calculation of maximum velocity (Vmax), substrate binding affinity (Km), substrate specificity (Vmax/Km), catalytic rate constant (kcat) and catalytic efficiency (kcat/Km). Four different buffers were also tested including citrate phosphate buffer (pH-3.0 to 8.0), sodium acetate buffer (pH-4.0 to 7.0), potassium phosphate buffer (pH-6.0 to 9.0) and glycine NaOH buffer (pH-9.0 to 12.0) for measuring effective catalytic activity.
2.11 Thermal and storage stabilities of laccase-CLEAs
Thermal stability of laccase-CLEAs was determined by placing the CLEAs at 50 °C, 60 °C and 70 °C for up to 180 minutes. Enzymatic activity was measured in terms of percent residual activity. Storage stability of enzyme aggregates was analyzed by performing enzyme assays periodically for up to 120 days with a time interval of 06 days. Laccase CLEAs were also stored at 4 °C as was the soluble laccase.2.12 Reusability of laccase-CLEAs
The reusability of laccase-CLEAs was evaluated by performing successive enzyme assays under optimized conditions. The activity measured in the initial cycle was defined as 100% residual activity. To ensure the removal of any residual substrate or product, the laccase-CLEAs were washed three times with 100 mM citrate phosphate buffer (pH-4.0) after each cycle.2.13 Determination of thermal activation energy of soluble laccase and laccase-CLEAs
The activation energy of soluble laccase and laccase-CLEAs was determined at varying temperatures ranging from 20 °C to 50 °C in the case of soluble laccase and between 30 °C and 50 °C for laccase CLEAs. Activation energy was calculated using the following Arrhenius equation:where k is the rate constant of a reaction; Ae is the Arrhenius exponential factor; Ea is the activation energy; R is the gas constant and T is the temperature in kelvin.
Activation energy was calculated using the following equation, a derived equation from the Arrhenius equation and the graph was plotted between ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k and 1/T from the Arrhenius equation:
k and 1/T from the Arrhenius equation:
| Ea = −slope × R |
2.14 Determination of thermal deactivation of soluble laccase and laccase-CLEAs
Various parameters including deactivation energy (Ed), change in Gibbs energy (ΔG), change in enthalpy (ΔH) and change in entropy (ΔS) of the soluble laccase and laccase-CLEAs were estimated at various temperatures (40 °C to 70 °C) for a specific time interval. Percent residual activity for both the soluble laccase and laccase-CLEAs was also calculated for the estimation of thermal inactivation constant (kd). Natural log(ln) of residual activity was calculated for each temperature. A graph was plotted using natural log of percent residual activities versus time. The following equation was used for the calculation of deactivation constant:where A is the residual activity after thermal treatment; A0 is the initial activity; t is the time of exposure to heat.
The temperature at which 50.0% reduction in enzyme activity was observed was referred to as the half-life of an enzyme molecule. The half-life (t1/2) of both the soluble laccase and laccase-CLEAs was calculated as described earlier21 using the following equation:
Decimal reduction time (D-value) is defined as the time required at a specific temperature to achieve a 90% reduction (1-log cycle) in initial enzymatic activity. The Z-value represents the temperature increase required to result in a ten-fold (1-log) reduction of the D-value and the following equation was used:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) D and temperature in °C:
D and temperature in °C:Finally, deactivation energy was calculated considering the non-linear Arrhenius equation by plotting a graph of ln of the deactivation constant (Kd) against the inverse of absolute temperature in K.
| Ed = −slope × R |
Change in Gibbs energy (ΔG), change in enthalpy (ΔH) and change in entropy (ΔS) were also calculated as described previously22 using the following equations:
| Enthalpy (ΔH) = Ed − RT |
where R is the gas constant; T is the temperature in kelvin; H is Plank's constant; kb is the Boltzmann constant; Ed is the deactivation energy and kd is the deactivation constant.
3 Results and discussion
Generally, in stoichiometric chemical reactions, specific amounts of reactants are used for the production of a specific number of product molecules. In these types of chemical reactions all of the reactant molecules are completely consumed and therefore have an increased economic impact. Most of the time these stoichiometric reactions are very slow and require high energy input and at the same time during the reaction some unwanted byproducts are also produced parallel to the specific synthesis of a product. Many chemical processes are expensive, and the byproducts formed are also a hazard to the environment. With the passage of time, many biocatalysts from different classes of enzymes have surpassed the conventional setups for the synthesis of several chemical metabolites and intermediates. Different types of biocatalysts were introduced afterwards in the processing steps to facilitate quick chemical reactions. Free enzyme molecules used in bioprocesses were environmentally friendly, but they were not recovered after the reaction was completed because of their soluble nature and sometimes caused interference during the product purification step. Keeping this in view, the current research work was undertaken to support sustainable green chemistry processes which might help industries in reducing their production cost and could also support in reutilizing the biocatalyst reactant molecules multiple times as the focus is towards an insoluble enzyme version. Many different types of enzymes have been used in different industrial chemical processes, but these soluble biocatalysts are not recoverable from the reaction mixture after the chemical reaction is completed. Hence, to overcome this drawback various immobilization techniques were introduced previously with different types of matrices or support systems. Again, these insoluble matrices were not feasible on a large scale; thus, researchers introduced a new form of supportless insoluble enzyme immobilization approach known as crosslinked enzyme aggregates. Laccase [E.C. 1.10.3.2.] is one of the most stable and robust biocatalysts with broad industrial applications and in the present study, a detailed kinetic characterization of the soluble and laccase based CLEAs has been explored to support the thermodynamics and stability of the supportless immobilization technique for its potential application in the biodegradation of toxic compounds.3.1 Production and purification of soluble fungal laccase
Laccase [E.C. 1.10.3.2] is ubiquitously produced by a variety of bacteria, plants and fungi whereas, fungal laccase has been extensively studied for its various applications in industrial and environmental setups. Previously, fungal laccase from white rot fungi Trametes versicolor, Fomes fomentarius and Cerrena sp. have been extensively studied.15,20,23 So far, laccase from Trametes pubescens has proved to be more potent in catalytic performance than other laccases from different fungal species. This is because it has been shown to exhibit having significantly higher catalytic efficiency i.e. approximately four to five times higher than that of commercial T. versicolor laccase when acting on certain phenolic substrates with stronger affinity towards certain diphenols like caffeic and gallic acids.24 In this same study the authors found that amperometric measurements indicated that biosensors developed using T. pubescens laccase could be more active than biosensors based on laccase from Trametes versicolor and this was because of the particular specificity of the enzyme. In general, the choice of T. pubescens laccase is often driven by a combination of its efficient kinetic performance and the high-yield, low-cost production potential achievable on waste streams. Therefore, in this study laccase was produced from Trametes pubescens MB 89 through solid state fermentation using sugarcane bagasse. Agricultural biomass is a complex source of nutrients particularly carbon and nitrogen that accelerates the fungal growth rate without any supplementation; therefore, utilization of agricultural leftovers and lignocellulosic material is an ecofriendly and cost-effective approach for the production of useful biomaterials and industrially important enzymes.25,26 In this current research, fungal culture was grown under solid state fermentation using agro-industrial waste that gave a crude laccase activity of 200 U with a specific activity of 434 U mg−1 (Table 1). The fermentation was performed at 30 °C for 10 days. Similarly, other studies have also reported the utilization of natural biowaste materials as effective substrates for the production of soluble laccase.27–29 To maximize the recovery of soluble laccase from biomass, the extracellular enzyme was extracted using citrate phosphate buffer (pH-4.0) supplemented with Tween-80. The extraction was performed at a constant agitation speed of 180 rpm. In this study the provision of buffer was to maintain the physiological conditions and the stability of the enzyme; however, it was reported that sterile distilled water can also be used for the extraction of enzyme.27,30 Afterwards, filtration was carried out using a 0.45 µM nitrocellulose membrane disc (Merck) to ensure the removal of residual biomass.| Precipitating agents and concentrations | Total volume (ml) | Enzyme activity (U) | Total protein (mg) | Specific activity (U mg−1) | Fold purification (%) | Yield (%) |
|---|---|---|---|---|---|---|
| Crude laccase | 1000 | 200 ± 50 | 0.46 ± 0.008 | 434 ± 20 | 1.0 | 100 |
| Ammonium sulphate (70%) | 100 | 1200 ± 110 | 1.3 ± 0.058 | 923 ± 40 | 2.13 | 60 |
| Desalted laccase | 90 | 1434 ± 120 | 1.3 ± 0.05 | 1103 ± 80 | 2.54 | 72 |
The nature of the precipitating agent plays an important role in the precipitation of biocatalysts; some of the precipitating agents may lead to the loss of enzymatic activity. Some of the water-miscible organic solvents can promote enzyme denaturation, as demonstrated by Öztürk et al. in the case of serine alkaline proteases.31 Initially soluble laccase was precipitated using three different types of precipitating agents (ammonium sulphate, PEG-6000 and isopropanol) and it was observed that laccase was effectively precipitated using salt as a precipitating agent. This suggested that polymeric or solvent based precipitating agents may have led to loss in enzymatic activity or denaturation of the soluble enzyme. The results revealed that the crude enzyme precipitated with 70.0% ammonium sulphate gives an enzymatic activity of 1200 U (Table 1) whereas much lower laccase activity was observed in the case of PEG-6000 (25.0%) and isopropanol (50.0%) (data not shown). Afterwards, gradient precipitation for ammonium sulphate was performed with three different concentrations and it was observed that at the highest ammonium sulphate concentration of 70.0% the maximum laccase activity was observed with a specific activity of 1103 U mg−1. Previous studies have also reported that ammonium sulphate may be an effective precipitating agent for the partial purification of laccase.15,32
3.2 Development of crosslinked aggregates of laccase (L-CLEAs)
The conventional process for the synthesis of CLEAs is a simple, fast and economical binary process that involves physical and chemical interactions between the biocatalyst molecules in the presence of a suitable dialdehyde linker molecule.33,34 Physical enzyme aggregates have been previously developed through precipitation of enzyme molecules while chemical crosslinkers among these aggregates result in the synthesis of an insoluble, stable form of enzymes.35 Earlier, the development and utilization of laccase-based enzyme aggregates have been used as an effective and ecofriendly approach for the decolorization, oxidation and biodegradation of dyes and phenolic compounds.23 Therefore, for the current study the extracellularly produced crude soluble laccase with specific activity of 434.0 ± 20.0 U mg−1 was initially precipitated through a physical interaction method by the addition of ammonium sulphate salt (70.0%) and a specific activity of 923.0 ± 40.0 U mg−1 was obtained (Table 1). The precipitated enzyme molecules were desalted (1103.0 ± 80.0 U mg−1) and were chemically crosslinked together by combining glutaraldehyde molecules (50.0 mM) as crosslinkers under specified conditions. The feasibly recovered insoluble form of laccase, in the form of aggregates, were named laccase-CLEAs (L-CLEAs) which exhibited a specific activity of 1639.0 ± 20.0 U mg−1 thus demonstrating successful aggregation of the catalytically active laccase molecules.The complete protocol for production, purification and synthesis of L-CLEAs is depicted in the schematic presentation (Fig. 1).
 | ||
| Fig. 1 Schematic presentation for the fabrication of laccase based crosslinked enzyme aggregates (L-CLEAs). | ||
3.3 Scanning electron micrographs of L-CLEAs
According to the classical nucleation theory, the covalent crosslinking of enzyme molecules in the form of aggregates results from the variation in free energy between the soluble and insoluble state of enzyme molecules. The structure and size of the synthesized aggregates depend on the ratio of nucleation and growth of aggregates. The structural elucidation of CLEAs through SEM helps to categorize the aggregates into one of the two different types which may be Type-I and Type-II, as proposed earlier.36 The physical appearance of L-CLEAs was in the form of a fine powder (Fig. 2A) while surface topography through scanning electron micrographs at different magnifications revealed a uniform porous structure of the aggregates, resembling the characteristics of Type-II form (Fig. 2B–D). One plausible reason for the synthesis of Type II form of L-CLEAs could be the high glycosylation as well as the greater hydrophilic surface of fungal laccase.37 | ||
| Fig. 2 Scanning electron microscopy of laccase based crosslinked enzyme aggregates (L-CLEAs) at different magnification levels (A–D). | ||
3.4 Optimization of crosslinking parameters for the development of L-CLEAs
Further improvement in catalytic activity was carried out by optimizing crosslinking parameters that included the specific concentrations of the precipitating agent and the crosslinker molecule (Fig. 3). After the assessment of different precipitating agents (ammonium sulphate, PEG- 6000 and isopropanol) for soluble laccase, ammonium sulphate was selected due to its efficient precipitating property for the development of laccase-based enzyme aggregates. Ammonium sulphate is reported as the most common inorganic salt that has been used for the synthesis of CLEAs; however, the appropriate concentration of salt should be optimized to obtain a higher yield of functionally active CLEAs.33 Therefore, in the current study, three different concentrations of ammonium sulphate (50.0%, 60.0% and 70.0%) were analyzed, and it was observed that 70.0% concentration of ammonium sulphate resulted in a maximum aggregation efficiency of approximately 53.0% (Fig. 3A). While at concentrations of 50.0% and 60.0%, lower aggregation efficiency was observed as compared to 70.0% concentration. The reason for lower aggregation efficiency is the insufficient amount of precipitation agent which would have been required for the aggregation of laccase molecules. Similar effects were also reported previously by other researchers as well.38,39 Nevertheless, in addition to ammonium sulfate, PEG-4000 has also been reported to attain maximum precipitation of laccase produced from Trametes versicolor IBL-04.32Enzyme aggregates can be fabricated by incorporating a crosslinker at various stages: prior to, during or following the precipitation of the crude enzyme.40 In the current study, to achieve effective binding, glutaraldehyde (GA) was incorporated as a crosslinker molecule after the precipitation of laccase enzyme. Glutaraldehyde is frequently used for the synthesis of CLEAs due to its higher degree of crosslinking capacity, feasible cost and availability in sufficient quantity.34 GA is a dialdehyde and its aldehyde groups undergo Schiff base formation upon nucleophilic attack by amino groups of lysine moieties present in the enzyme or protein molecules. Under acidic conditions, at which the laccase likely precipitates out, the dialdehyde molecules exist in monomeric cyclic hemiacetal and multimeric forms and both of these react with the protein molecules through the amino group of lysine, thus also improving the stability of the enzyme. Therefore, it has been earlier reported that the presence of such amino acids on the surface of the protein molecules and their absence or even low quantity in the catalytic domain facilitates the synthesis of a functionally active form of CLEAs.32,33,41 While studying the structure of laccase through literature it was revealed that histidine and cysteine residues are the most critical amino acids in the catalytic domain of fungal laccase instead of lysine.42–44 In addition, selection of an appropriate ratio of enzyme and crosslinker molecule is also a critical task to develop catalytically active crosslinked aggregates.45 Hence, three different concentrations of glutaraldehyde were selected in the present study (50 mM, 100 mM and 150 mM) for efficient crosslinking of soluble laccase/precipitated laccase and it was noted that 100 mM concentration of glutaraldehyde gave the highest aggregation efficiency of about 63.0% that remained constant to some extent even after increasing the concentration up to 150 mM (Fig. 3B). On the other hand, a low aggregation yield of approximately 33.0% was observed at 50 mM concentration of glutaraldehyde. A plausible reason for the low catalytic activity of aggregates at low crosslinker concentrations could be the intramolecular crosslinking of dialdehyde within the same enzyme, which restricts the intermolecular crosslinking between enzymes that facilitates the synthesis of efficient enzyme crosslinked system. In contrast, excessive amounts of crosslinker result in excessive inter and intra molecular linkages that compact the conformational structure of protein, making it rigid and thus creating mass transfer limitations that ultimately lessen the enzyme activity of CLEAs.46–48 Another possibility could be that, because of the more compact structure of the CLEAs, the substrate binding pocket is prevented from docking of the specific substrate thus resulting in delayed enzyme–substrate reaction.
3.5 Optimization of reaction time, reaction temperature and reaction pH
The impact of catalytic performance on time, reaction pH and reaction temperature was also investigated for both soluble and L-CLEAs and the results are expressed as percent relative activity. The initial activity for each parameter was set as 100%.The experiments conducted to explore the optimum catalytic reaction time of soluble laccase and L-CLEAs depicted that in the soluble state, laccase exhibited maximum catalytic action within 05 minutes of reaction while, after the change in the state from soluble to aggregate, the time extended up to 10 minutes (Fig. 4A). It is possible that the crosslinking of the enzyme units through the crosslinker molecules could have brought the enzyme molecules close together in a small dimension and thus resulted in the limitation of availability of substrate molecules. Another possibility is the mass transport limitation of substrate molecules due to the aggregated form of enzyme that ultimately prolonged the enzyme–substrate reaction time.36,48
To discover the optimal reaction temperature for the effective catalytic performance of L-CLEAs, the enzyme activity was conducted at various temperatures (30 °C to 70 °C) along with soluble laccase (Fig. 4A). The experimental data suggested that the activity of soluble laccase started from 30 °C and reached to its peak point at 50 °C and approximately 50.0% decline in the activity was noticed at 60 °C and 70 °C. This pattern is also observed by the previously mentioned activity of the biocatalyst which mentions that the structural configuration of the enzyme is responsible for any temperature sensitivity; thus, denaturation is noticed at increasing temperatures.49 The comparative analysis revealed that the catalytic activity of L-CLEAs was five times lower at 30 °C, than that of the soluble counterpart, which gradually increased with increasing temperature and attained maximum activity at a similar temperature (50 °C) as that of soluble laccase, followed by a decline thereafter (Fig. 4B). The results are in accordance with former studies as well which reported similar findings of temperature optima for soluble laccase and L-CLEAs.14,15 While in another study, a shift in temperature optima from 40 °C (soluble laccase) to 60 °C (Lac-CLEAs) was also reported with a significant difference in the stability of Lac-CLEAs at high temperature in contrast to the stability of soluble laccase.32,50 Covalent bonding between the enzyme and the crosslinker likely stabilized the active site against conformational changes, thereby minimizing the loss of enzymatic activity at elevated temperatures. Likewise, the majority of the studies on enzyme aggregate synthesis have reported a shift in maximum activity of aggregates at high temperatures compared to the soluble version of the enzyme.51
Selection of a suitable buffer with specific pH has attained critical consideration for efficient catalytic performance of biocatalysts. Therefore, using various buffers and pH at constant ionic strength for assessing the catalytic performance of soluble and L-CLEAs was studied. The result of this study indicates that the catalytic performance of L-CLEAs is highly buffer dependent, with optimal activity (98% relative activity) achieved in citrate–phosphate buffer at pH-4.0 (Fig. 4C). While both soluble laccase and laccase-CLEAs exhibited a clear preference for acidic conditions, the CLEAs demonstrated significantly broader pH stability. Notably, the immobilized aggregates maintained catalytic functionality in neutral and slightly alkaline environments, whereas the soluble enzyme was completely inactive under these conditions. This extended pH tolerance suggests that the covalent crosslinking provides a robust structural framework, shielding the enzyme active site from the denaturing effects of deprotonation or hydroxyl ion interference typically encountered at higher pH levels. Previous studies have shown that immobilizing laccase on various solid supports or developing it into aggregates significantly enhances its catalytic efficiency across a wider pH range.15,52 There is a possibility that higher concentration of hydroxyl ions in soluble laccase may cause hindrance in redox reactions at copper sites which are present in every type of laccase, thus resulting in the reduction of catalytic performance under alkaline conditions.53 However, the formation of covalent bonds between the enzyme and the crosslinker, during aggregation, significantly alters the enzyme's microenvironment. This structural rigidification likely accounts for enhanced performance across a broader pH range compared to the soluble enzyme.54
3.6 Steady-state kinetic and thermodynamics parameters of soluble laccase and laccase aggregates
The enzyme kinetic parameters were estimated using the Hanes–Woolf plot for soluble laccase and laccase aggregates when ABTS was used as a substrate (Table 2). In the case of laccase aggregates, it exhibited a much higher Km value as compared to the Km value of soluble laccase which indicates that the immobilized laccase showed less affinity towards its substrate and therefore, the laccase aggregates also have a higher turnover rate and lower catalytic efficiency. This reduction in substrate affinity is attributed to several factors, including conformational changes in the protein structure post-immobilization, electrostatic interactions between the crosslinker and the substrate molecule, and increased mass transfer resistance limiting substrate diffusion to the enzyme active site.55 In the case of diffusion limitations of the laccase based CLEAs, the dense cross-linking of laccase molecules creates a porous matrix where, substrate mass transfer is hindered. This typically results in a higher Km value because a higher bulk concentration of the substrate is required to saturate the enzymes located within the interior of the developed enzyme aggregate. At the same time in terms of structural considerations, it cannot be entirely ruled out that minor conformational distortion or steric hindrance (active site occlusion) may also be caused by the glutaraldehyde cross-linking. However, the retention of significant activity suggests that the laccase active sites remain largely accessible and functional even after crosslinking. While it was also observed that the catalytic efficiency (kcat/Km) decreased, the trade-off is justified by the drastic improvement in operational stability. In industrial biocatalysis, the ability to recover and reuse the enzyme over multiple cycles and its enhanced resistance to thermal/solvent denaturation is often more economically critical than the initial catalytic rate. The catalytic efficiency (kcat/Km) shows a slight decrease (from 11.86 to 9.09 s−1 mM−1) and this is driven entirely by the five-fold increase in apparent Michaelis–Menten constant (Km,app). Crucially, the catalytic rate constant (kcat) actually increased more than four-fold in the CLEA form (5.355 s−1 compared to 1.281 s−1 for the soluble enzyme). This suggests that once the substrate reaches the active site, the catalytic transformation is significantly faster in the immobilized state. Earlier studies have reported that laccase aggregates from the fungal isolate Oudemansiella canarii have a higher Km for the immobilized enzyme in comparison to the soluble enzyme.56 In another study, crosslinked enzymes prepared by ordered-crosslinking had better reducing activity and a higher NADPH production rate than those prepared by fixed point crosslinking and random crosslinking, obtained by glutaraldehyde mediated crosslinking of pure aldo–keto reductase and alcohol dehydrogenase proteins.35 This suggests that there is a difference in enzyme reaction catalysis when CLEAs are prepared under controlled conditions. Similarly, a separate study demonstrated that the ordered crosslinking yielded lower Km values and superior catalytic efficiency compared to both site-specific disordered and random crosslinking methods. These results suggest that enzymes within ordered-CLEAs maintain a higher substrate affinity, likely due to more favorable conformational orientations.57 Consequently, it can be inferred that substrate transfer is maximized when enzyme molecules are assembled with minimal inter-enzyme distance and when crosslinking sites are optimally oriented towards one another.58 In the present study, a decrease in activation energy (Ea) from 7.423 kJ mol−1 to 3.329 kJ mol−1 for the CLEAs was also observed. A lower Ea indicates that the energetic barrier for the catalytic reaction has been reduced, which strongly suggests that the active site remains fully functional and is perhaps even stabilized in a more transition-state-favorable conformation. Inactivation energy (Ed) was observed to increase from 48.55 to 53.30 kJ mol−1, providing evidence that the CLEAs are more resistant to thermal unfolding. Similarly, the change in Gibbs free energy (kJ−1 mol−1 K−1) was also observed to increase from 95.56 to 100.10 kJ mol−1, thus confirming the superior thermodynamic stability of the CLEA matrix. Thermal stability of soluble laccase and laccase aggregates was also assessed. The activation energy (Ea) for L-CLEAs was lower (3.329 kJ mol−1) than that of soluble laccase (7.432 kJ mol−1) whereas, the inactivation energy (Ed) of L-CLEAs was higher (53.29 kJ mol−1) than that of soluble laccase (48.55 kJ mol−1), thus suggesting that CLEAs are more stable and require less energy for chemical reaction. Overall, the decrease in Ea and the concurrent four-fold increase in kcat t suggest that the crosslinking process does not compromise the intrinsic catalytic mechanism. When coupled with the significant enhancement in half-life at 60 °C (t1/2) and higher inactivation energy Ed, the CLEAs demonstrate a clear net benefit for long-term operational use despite the apparent reduction in substrate affinity. Similarly, when both the enzyme forms were exposed to three different temperatures including 50.0 °C, 60.0 °C and 70.0 °C for the calculation of half-life (t1/2) the results again supported the assumption that L-CLEAs have a more stable half-life, specifically at 60.0 °C then the soluble version. Approximately a 2.0-fold increase was noted in the half-life of the L-CLEAs when exposed to 60.0 °C. The slightly higher values of change in Gibbs free energy (ΔG), change in enthalpy (ΔH) and change in entropy (ΔS) indicate the thermal stability of the enzyme as well. In this study, the data suggested that immobilized laccase has higher Gibbs-energy which could be corelated to the thermal stability of the enzyme. Previous literature has also suggested that the development of enzyme aggregates and the process of immobilization result in the enhancement of the thermal stability and thermal tolerance of immobilized enzymes.59,60 The increased heat tolerance of an immobilized enzyme is due to the structural stability during the process of immobilization and aggregate development.32| Parameters | Units | Laccase produced by Trametes pubescens MB 89 | |
|---|---|---|---|
| Soluble laccase | CLEAs of laccase | ||
| Maximum velocity (Vmax) | U mg−1 | 1794 | 1639 |
| Substrate binding affinity (Km) | mM | 0.108 | 0.589 |
| Substrate specificity (Vmax/Km) | U mg−1 mM−1 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 611.11 611.11 |
2782.68 |
| Catalytic rate constant (kcat) | s−1 | 1.281 | 5.355 |
| Catalytic efficiency (kcat/Km) | s−1 mM−1 | 11.86 | 9.09 |
| Gibbs free energy (ΔG) | kJ−1 mol−1 K−1 | 95.56 | 100.10 |
| Enthalpy of activation (ΔH) | kJ−1 mol−1 K−1 | 45.94 | 50.53 |
| Entropy of activation (ΔS) | kJ−1 mol−1 K−1 | −0.155 | −0.145 |
| Free energy for formation of activation complex (ΔGE–T) | kJ mol−1 | −6867.95 | −6129.56 |
| Activation energy (Ea) | kJ mol−1 | 7.423 | 3.329 |
| Inactivation energy (Ed) | kJ mol−1 | 48.554 | 53.295 |
| Half-life (t1/2) at 50 °C | min−1 | 423 | 494 |
| Half-life (t1/2) at 60 °C | min−1 | 210 | 406 |
| Half-life (t1/2) at 70 °C | min−1 | 127 | 154 |
3.7 Storage stability and recycling efficiency of soluble laccase and laccase aggregates
Long-term storage of enzymes is considered as important parameter for industrial applications. Therefore, storage stability was also determined for both the soluble and L-CLEAs and it was observed that at 4 °C the soluble laccase was able to retain its enzymatic activity only up to 24 days (90.0%) while L-CLEAs exhibited similar stability up to 72 days (Fig. 5A). After this time period a gradual decline was noticed in both cases. Contrary to soluble laccase, the developed laccase aggregates were able to retain the enzymatic activity even after 120 days of experimentation, thus suggesting that L-CLEAs could be used for a longer period of time with effective catalytic performance.Reutilization of an enzyme is also an important parameter in utilizing the enzyme at an industrial scale. Development of immobilized versions of an enzyme is an effective method to enhance the recycling efficiency of the biocatalytic molecule in a commercial setup.21 The current study suggests that the L-CLEAs remained more than 85.0% catalytically active up to 16 cycles out of 20 cycles (Fig. 5B) which is much higher than previously reported data. In the case of matrix dependent immobilization, the recycling efficiency was reported previously to be reduced to 08–12 cycles only.61,62 This loss in catalytic performance could be due to the leakage or deactivation of immobilized enzyme molecules during multiple washing steps that are involved after each cycle of study.63 Thus, development of enzyme aggregates might be an advantage over matrix dependent immobilization in terms of reutilization of enzyme molecules.
4 Conclusions
Laccase is applied in various green chemistry processes including polymer synthesis and bioremediation. Sensitivity to environmental and process conditions impedes its widespread use. This study concludes that cross-linked enzyme aggregates could work as an effective technology to improve operational stability of fungal laccase. Our findings demonstrate that while the aggregate matrix introduces diffusion-related increases in Km, it simultaneously optimizes the enzyme's catalytic and structural resilience. The four-fold increase in the catalytic rate constant (kcat) and the significant reduction in activation energy (Ea) confirm that the laccase active site remains highly accessible and catalytically efficient within the crosslinked framework. Furthermore, the other parameters emphasize the success of this approach; the increase in inactivation energy (Ed) and the doubling of the half-life at 60 °C illustrate a superior protective effect against thermal unfolding. Ultimately, this study proves that for industrial laccase applications, the gain in operational longevity and turnover frequency effectively outweighs the apparent reduction in substrate affinity, providing a robust biocatalytic platform capable of withstanding rigorous process conditions. The immobilized laccase (L-CLEA) retained enzymatic activity for up to 120 days and remained catalytically active for 16 out of 20 cycles. Future work on the application of L-CLEAs in biomass pretreatment, remediation of textile effluent and synthesis of aromatic polymers will widen the scope of enzyme-based green chemistry.Author contributions
Afsheen Aman: conceptualization, funding acquisition, project administration, resources, supervision, validation, roles/writing – original draft, and writing – review & editing. Mohammad: data curation, formal analysis, methodology. Faiza Shahid: validation, visualization, roles/writing – original draft. Asma Ansari: methodology, writing – review & editing. Muhammad Sohail: resources.Conflicts of interest
The authors declare that they have no financial interests/personal relationships which may be considered as potential competing interests.Data availability
The authors do not have other results to share as all data are shown in the present article.Acknowledgements
The authors acknowledge the indigenous support, laboratory facilities and indigenous funding provided by The Karachi Institute of Biotechnology & Genetic Engineering (KIBGE), University of Karachi, Pakistan.References
- E. Hajili, A. Sugawara and H. Uyama, Biomacromolecules, 2024, 25, 3486–3498, DOI:10.1021/acs.biomac.4c00109.
- Q. Hao, P. Guo, H. Wang, M. Su, C. Li, Y. Hu and W. Su, Particuology, 2024, 85, 102–112, DOI:10.1016/j.partic.2023.03.023.
- T. Charoenwongpaiboon, K. Wangpaiboon, R. A. Field, M. Prousoontorn and R. Pichyangkura, Mol. Catal., 2023, 535, 112827, DOI:10.1016/j.mcat.2022.112827.
- G. N. Lucena, C. C. dos Santos, G. C. Pinto, R. D. Piazza, W. N. Guedes, M. J. Júnior, A. V. de Paula and R. F. C. Marques, J. Magn. Magn. Mater., 2020, 516, 167326, DOI:10.1016/j.jmmm.2020.167326.
- M. Primožič, G. Kravanja, Ž. Knez, A. Crnjac and M. Leitgeb, J. Cleaner Prod., 2020, 275, 124121, DOI:10.1016/j.jclepro.2020.124121.
- E. Araya, P. Urrutia, O. Romero, A. Illanes and L. Wilson, Food Chem., 2019, 288, 102–107, DOI:10.1016/j.foodchem.2019.02.024.
- M. Sharifi, A. Y. Karim, N. Q. M. Nanakali, A. Salihi, J. F. M. Aziz, R. H. Hong, A. K. Khan, A. K. Saboury, A. Hasan, O. K. Abou-Zied and M. Falahati, J. Biomol. Struct. Dyn., 2019, 38, 2746–2762, DOI:10.1080/07391102.2019.1643787.
- M. Q. Xu, S. S. Wang, L. N. Li, J. Gao and Y. W. Zhang, Catalysts, 2018, 8, 460, DOI:10.3390/catal8100460.
- X. Su, X. Yang, H. Long, Y. Li, W. Sun, T. Mo, H. Lyu, A. Cavaco-Paulo, H. Wang and J. Su, Int. J. Biol. Macromol., 2024, 134929, DOI:10.1016/j.ijbiomac.2024.134929.
- M. Mohammadi, M. A. As’habi, P. Salehi, M. Yousefi, M. Nazari and J. Brask, Int. J. Biol. Macromol., 2018, 109, 443–447, DOI:10.1016/j.ijbiomac.2017.12.102.
- S. K. Patel, V. C. Kalia, J. H. Choi, J. R. Haw, I. W. Kim and J. K. Lee, J. Microbiol. Biotechnol., 2014, 24, 639–647, DOI:10.4014/jmb.1401.01025.
- J. H. Park, H. Xue, J. S. Jung and K. Ryu, Korean J. Chem. Eng., 2012, 29, 1409–1412, DOI:10.1007/s11814-012-0024-1.
- D. M. Braga, T. Brugnari, C. W. I. Haminiuk and G. M. Maciel, Process Biochem., 2024, 141, 132–143, DOI:10.1016/j.procbio.2024.03.012.
- A. Zerva, C. Pentari and E. Topakas, Molecules, 2020, 25, 2221, DOI:10.3390/molecules25092221.
- M. Vršanská, S. Voběrková, A. M. Jimenez, V. Strmiska and V. Adam, Int. J. Environ. Res. Public Health, 2018, 15, 23, DOI:10.3390/ijerph15010023.
- G. Liu, L. Wang, A. Zhao, L. Guo, L. Zhou, Y. He, L. Ma, Y. Liu, J. Gao and Y. Jiang, Chem. Eng. J., 2024, 479, 147635, DOI:10.1016/j.cej.2023.147635.
- N. Enayatizamir, J. H. Liu, L. Wang, X. Lin and P. Fu, J. Water Process Eng., 2020, 37, 101357, DOI:10.1016/j.jwpe.2020.101357.
- O. H. Lowry, N. J. Rosebrough, A. L. Farr and R. J. Randall, J. Biol. Chem., 1951, 193, 265–275 CrossRef CAS.
- R. A. Abd El Monssef, E. A. Hassan and E. M. Ramadan, Ann. Agric. Sci., 2016, 61, 145–154, DOI:10.1016/j.aoas.2015.11.007.
- J. Yang, X. Xu, X. Yang, X. Ye and J. Lin, J. Taiwan Inst. Chem. Eng., 2016, 65, 1–7, DOI:10.1016/j.jtice.2016.04.025.
- A. Karim, Z. Bibi, H. U. Rehman, A. Aman, S. A. U. Qader and M. H. Rashid, Colloids Surf., B, 2021, 200, 111583, DOI:10.1016/j.colsurfb.2021.111583.
- A. P. Tavares, C. G. Silva, G. Dražić, A. M. Silva, J. M. Loureiro and J. L. Faria, J. Colloid Interface Sci., 2015, 454, 52–60, DOI:10.1016/j.jcis.2015.04.054.
- M. Bilal, T. Rasheed, F. Nabeel, H. M. N. Iqbal and Y. Zhao, J. Environ. Manage., 2019, 234, 253–264, DOI:10.1016/j.jenvman.2019.01.001.
- M. Shukri, T. Cherneva, A. Peshkov, M. Nikolova, I. Iliev and N. Dimcheva, Appl. Food Biotechnol., 2025, 12, 1–11, DOI:10.22037/afb.v12i1.46955.
- M. E. Arias, A. Blánquez, M. Hernández, J. Rodríguez, A. S. Ball, N. T. Jiménez-Morillo, J. A. González-Vila and J. A. González-Pérez, J. Anal. Appl. Pyrolysis, 2016, 122, 202–208, DOI:10.1016/j.jaap.2016.09.023.
- A. Blánquez, A. S. Ball, J. A. González-Pérez, N. T. Jiménez-Morillo, F. González-Vila, M. E. Arias and M. Hernández, PLoS One, 2017, 12, e0187649, DOI:10.1371/journal.pone.0187649.
- I. Stoilova, A. Krastanov and V. Stanchev, Adv. Biosci. Biotechnol., 2010, 1, 2368 CrossRef . http://www.SciRP.org/journal/abb/.
- F. Wang, L. Xu, L. Zhao, Z. Ding, H. Ma and N. Terry, Microorganisms, 2019, 7, 665, DOI:10.3390/microorganisms7120665.
- E. M. Rodrigues, S. G. Karp, L. C. Malucelli, C. V. Helm and T. M. Alvarez, J. Basic Microbiol., 2019, 59, 784–791, DOI:10.1002/jobm.201900084.
- K. N. Niladevi, R. K. Sukumaran and P. Prema, J. Ind. Microbiol. Biotechnol., 2007, 34, 665–674, DOI:10.1007/s10295-007-0239-z.
- D. C. Öztürk, D. Kazan, A. A. Denizci, D. Grimoldi, F. Secundo and A. Erarslan, J. Mol. Catal. B:Enzym., 2010, 64, 184–188, DOI:10.1016/j.molcatb.2009.02.017.
- M. Bilal, S. Noreen, M. Asgher and S. Parveen, Environ. Technol. Innovation, 2021, 101364, DOI:10.1016/j.eti.2021.101364.
- N. Chen, B. Chang, N. Shi, W. Yan, F. Lu and F. Liu, Crit. Rev. Biotechnol., 2023, 43, 369–383, DOI:10.1080/07388551.2022.2038073.
- O. Barbosa, C. Ortiz, Á. Berenguer-Murcia, R. Torres, R. C. Rodrigues and R. Fernandez-Lafuente, RSC Adv., 2014, 4, 1583–1600, 10.1039/C3RA45991H.
- L. Qiao, Z. Luo, R. Wang, X. Pei, S. Wu, H. Chen, T. Xie, R. A. Sheldon and A. Wang, Green Chem., 2023, 25, 7547–7555, 10.1039/D3GC01898A.
- R. Schoevaart, M. W. Wolbers, M. Golubovic, M. Ottens, A. P. Kieboom, F. van Rantwijk, L. A. van der Wielen and R. A. Sheldon, Biotechnol. Bioeng., 2004, 87, 754–762, DOI:10.1002/bit.20184.
- L. Arregui, M. Ayala, X. Gómez-Gil, G. Gutiérrez-Soto, C. E. Hernández-Luna, M. Herrera De Los Santos, L. Levin, A. Rojo-Domínguez, D. Romero-Martínez, M. C. N. Saparrat, M. A. Trujillo-Roldán and N. A. Valdez-Cruz, Microb. Cell Fact., 2019, 18, 1–33, DOI:10.1186/s12934-019-1248-0.
- S. S. Nadar, M. R. Ladole and P. U. Joshi, Int. J. Biol. Macromol., 2016, 84, 69–78, DOI:10.1016/j.ijbiomac.2015.11.082.
- H. W. Yu, H. Chen, X. Wang, Y. Y. Yang and C. B. Ching, J. Mol. Catal. B:Enzym., 2006, 43, 124–127, DOI:10.1016/j.molcatb.2006.07.001.
- F. López-Gallego, L. Betancor, A. Hidalgo, N. Alonso, R. Fernández-Lafuente and J. M. Guisán, Biomacromolecules, 2005, 6, 1839–1842, DOI:10.1021/bm050088e.
- P. Monsan, G. Puzo and H. Mazarguil, Biochimie, 1976, 57, 1281–1292, DOI:10.1016/S0300-9084(76)80540-8.
- A. Sekretaryova, S. M. Jones and E. I. Solomon, J. Am. Chem. Soc., 2019, 141, 11304–11314, DOI:10.1021/jacs.9b05230.
- R. Mehra, J. Muschiol, A. S. Meyer and K. P. Kepp, Sci. Rep., 2018, 8, 1–16, DOI:10.1038/s41598-018-35633-8.
- D. S. Yaver, F. Xu, E. J. Golightly, K. M. Brown, S. H. Brown, M. W. Rey, P. Schneider, T. Halkier, K. Mondorf and H. Dalbøge, Appl. Environ. Microbiol., 1996, 62, 834–841, DOI:10.1128/aem.62.3.834-841.1996.
- O. R. Zaborsky, Immobilization of enzymes by adsorption, in Biomedical Applications of Immobilized Enzymes and Proteins, Springer, Boston, MA, US, 1977, vol. 1, pp. 37–46 Search PubMed.
- R. Wang, J. Zhang, Z. Luo, T. Xie, Q. Xiao, X. Pei and A. Wang, Int. J. Biol. Macromol., 2022, 205, 682–691, DOI:10.1016/j.ijbiomac.2022.02.171.
- H. Yamaguchi, Y. Kiyota and M. Miyazaki, Catalysts, 2018, 8, 174, DOI:10.3390/catal8050174.
- S. Z. Mazlan and S. A. Hanifah, Int. J. Polym. Sci., 2017, 2017, 5657271, DOI:10.1155/2017/5657271.
- H. Bisswanger, Perspect. Sci., 2014, 1, 41–55, DOI:10.1016/j.pisc.2014.02.005.
- T. Hassani, S. Ba and H. Cabana, Bioresour. Technol., 2013, 128, 640–645, DOI:10.1016/j.biortech.2012.10.058.
- B. S. Aytar and U. Bakir, Process Biochem., 2008, 43, 125–131, DOI:10.1016/j.procbio.2007.11.001.
- Y. Gu, P. Xue and K. Shi, J. Porous Mater., 2020, 27, 73–82, DOI:10.1007/s10934-019-00786-y.
- C. Munoz, F. A. T. Guillén and M. J. Martinez, Appl. Environ. Microbiol., 1997, 63, 2166–2174, DOI:10.1128/aem.63.6.2166-2174.1997.
- J. M. Bolivar and B. Nidetzky, Molecules, 2019, 24, 3460, DOI:10.3390/molecules24193460.
- A. M. Girelli, L. Quattrocchi and F. R. Scuto, J. Biotech., 2020, 318, 45–50, DOI:10.1016/j.jbiotec.2020.05.004.
- T. M. Uber, A. J. D. R. Buzzo, G. Scaratti, S. M. Amorim, C. V. Helm, G. M. Maciel, R. A. Perlta, R. F. P. M. Moreira, A. Bracht and R. M. Peralta, Biocatal. Biotransform., 2022, 40, 17–28, DOI:10.1080/10242422.2020.1835873.
- J. U. Bowie, S. Sherkhanov, T. P. Korman, M. A. Valliere, P. H. Opgenorth and H. J. Liu, Trends Biotechnol., 2020, 38, 766–778, DOI:10.1016/j.tibtech.2019.12.024.
- C. C. Roberts and C. E. A. Chang, J. Chem. Theory Comput., 2015, 11, 286–292, DOI:10.1021/ct5007482.
- N. Seow and K. L. Yang, Colloids Surf., B, 2017, 151, 88–94, DOI:10.1016/j.colsurfb.2016.12.006.
- J. Yang, Z. Wang, Y. Lin, T. B. Ng, X. Ye and J. Lin, Sci. Rep., 2017, 7, 1–9, DOI:10.1038/s41598-017-16771-x.
- D. Spinelli, E. Fatarella, A. Di Michele and R. Pogni, Process Biochem., 2013, 48, 218–223, DOI:10.1016/j.procbio.2012.12.005.
- A. P. Tavares, C. G. Silva, G. Dražić, A. M. Silva, J. M. Loureiro and J. L. Faria, J. Colloid Interface Sci., 2015, 454, 52–60, DOI:10.1016/j.jcis.2015.04.054.
- S. Shleev, A. Jarosz-Wilkolazka, A. Khalunina, O. Morozova, A. Yaropolov, T. Ruzgas and L. Gorton, Bioelectrochemistry, 2005, 67, 115–124, DOI:10.1016/j.bioelechem.2005.02.004.
| This journal is © The Royal Society of Chemistry 2026 |