 Open Access Article
Open Access ArticleLarge scale valorization of steel slag combined with membrane-based direct air capture for carbon mineralization: a techno-economic evaluation
Vitor
Gama†
a,
Kyle
Shank†
b,
Madison
Morgan
a,
Owen
Gerdes
a,
Savannah
Sakhai
a,
Fernando V.
Lima
a,
Shang
Zhai
 *bc and
Oishi
Sanyal
*a
*bc and
Oishi
Sanyal
*a
aDepartment of Chemical and Biomedical Engineering, West Virginia University, Morgantown, WV, USA. E-mail: oishi.sanyal@mail.wvu.edu
bDepartment of Mechanical and Aerospace Engineering, The Ohio State University, Columbus, OH, USA. E-mail: zhai.218@osu.edu
cSchool of Earth Sciences, The Ohio State University, Columbus, OH, USA
First published on 9th April 2026
Abstract
The removal of carbon dioxide (CO2) from the atmosphere and from hard-to-abate industrial processes such as steel production remains a significant challenge for meeting climate goals. Progressive studies on membrane-based direct air capture (m-DAC) have emerged to propose this technology as a promising option for addressing legacy emissions. Previous studies discussed computational techniques that can search for favorable operational regions and suitable membrane material properties that give satisfactory CO2 capture performance and system energy efficiency. The combination of operability studies with membrane modeling and simulation represented a novel pathway to investigate fundamental material/process parameters tied to large-scale metrics. In this work, we explore a two-fold approach aiming to intensify CO2 capture and utilization: (i) capturing CO2 from ambient air (425 ppm) using m-DAC and concentrating it (25–50) times, and (ii) treating steel slags through CO2 mineralization for the synthesis of cementitious materials. Each process was initially investigated separately, and their integration was then analyzed based on the overall technoeconomic viability and CO2 removal efficiency per ton captured. The m-DAC process provides a low-purity (1–2%) CO2 stream suitable for combination with mineralization. Slag-water CO2 mineralization using 1% and 2% CO2 achieves conversion rates of 55.4% and 43.3%, respectively, producing carbonated slag. Additionally, the slag can be reacted with steam to yield 16.99 std. ml H2 per gram, utilizing waste heat from steelmaking. This dual process enables revenue generation from both H2 production and carbonated slag, which can be sold as a supplementary cementitious material.
Sustainability spotlightThis work advances sustainable carbon management by integrating membrane-based direct air capture with the mineralization of CO2 with steelmaking slag, transforming two environmental challenges into a unified climate solution. By permanently storing captured CO2 in stable mineral products while valorizing industrial waste from steel production, the research promotes circular resource use and reduces emissions from hard-to-abate sectors. By coupling carbon removal with industrial waste valorization, this work promotes sustainable industrial innovation, supports resilient built environments, and contributes to long-term strategies for mitigating climate change. The techno-economic evaluation provides a clear pathway for scaling carbon capture and utilization technologies alongside renewable electricity. |
1 Introduction
In March 2025, global atmospheric carbon dioxide levels reached an all-time high of 426.4 parts per million (ppm),1 surpassing the previous year's record of 423.4 ppm.3 This notable annual increase underscores the urgent demand to address the worldwide carbon dioxide crisis. To meet the goal of limiting the annual global temperature increase to 1.5 °C, emissions must be reduced by half by 2030, requiring the removal of tens of billions of metric tons of CO2 within the coming years.4 Thus development of scalable, economical and energy-efficient systems is critical to meet the net-zero target by 2050.5 This target remains unachievable with existing technologies, as the largest operational direct air capture (DAC) facilities remove about 0.01 megatons of CO2 per year.6 At this time, technologies including Carbon Engineering's potassium hydroxide solvent system and Climeworks' solid-sorbent units remain in early commercial phases, with limited scalability.7 Meanwhile, m-DAC systems remain at the laboratory, but offer a fundamentally different pathway for capturing CO2 from highly diluted air streams. Membranes may operate as standalone units or in hybrid configurations to selectively produce low-purity CO2, making them well suited for air-based capture where conventional sorbent and solvent systems face scalability constraints.This study presents a techno-economic analysis (TEA) of a combined CO2 capture and utilization process. The capture process consists of an m-DAC configuration represented by hollow fiber membrane modules, in which the individual membranes operate via a facilitated transport mechanism, as demonstrated in the author's prior work.2 The CO2 mineralization process, on the other hand, uses steelmaking by-products to generate supplementary cementitious materials (SCMs), thereby locking CO2 into a stable carbonate form. Membrane technologies offer several advantages in DAC applications, including modularity and operational flexibility.8 Despite these benefits, current limitations include high estimated removal costs ranging from $3000 to $10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 per ton of CO2 captured for a small-scale two-stage process, which are not competitive with other commercial DAC technologies.9 However, to the best of our knowledge, no DAC processes have yet reached the $100 per ton CO2 target set by DOE.
000 per ton of CO2 captured for a small-scale two-stage process, which are not competitive with other commercial DAC technologies.9 However, to the best of our knowledge, no DAC processes have yet reached the $100 per ton CO2 target set by DOE.
The m-DAC system analyzed in this study was simulated using AVEVA Process Simulation, targeting outlet purities of 1% and 2% CO2 for potential downstream applications including mineral carbonation or algae cultivation.10
To enhance the value proposition of m-DAC, this study examines a specific downstream CO2 utilization strategy, with the integration of m-DAC capture with steel slag carbonation, to produce SCMs. These SCMs not only enable permanent mineralization of CO2, but also improve the mechanical and hydraulic properties of construction-grade concrete.11
The mineralization pathway for producing SCMs is particularly attractive given the cement industry's high process emissions and significant material demand associated with ordinary Portland cement production. The production of ordinary Portland cement is a major source of CO2 emissions, primarily due to the endothermic decomposition of limestone (CaCO3) into lime (CaO) and CO2, followed by the high temperature formation of calcium silicates – the main reactive components in cement.12 Due to the energy and carbon intensity involved in both the calcination reaction and the calcium silicates formation reaction, there have been attempts to decrease cement usage by partially replacing it with SCMs.
Ground granulated blast furnace slag is a widely used SCM, whereas basic oxygen furnace (BOF) steel slag has limited SCM utilization due to low pozzolanic reactivity and the presence of expansive free CaO.13 Carbonation of BOF slag converts free lime to a stable carbonate, mitigating expansion while permanently storing CO2.
BOF slag carbonation has been investigated via gas–solid, direct aqueous, and indirect aqueous pathways.13 Gas–solid routes typically require elevated pressures or temperatures, while indirect aqueous processes rely on additional chemical reagents, increasing cost.14 Direct aqueous carbonation is therefore attractive, as it enables rapid CaO dissolution and CO2 mineralization under mild conditions.
In addition, BOF slag contains reduced iron that can react with steam to produce hydrogen (H2).15 In this study, BOF slag is first used for H2 generation through steam oxidation and then for CO2 mineralization via a modified direct aqueous carbonation process.
In this work, we simulate a membrane-based DAC system that concentrates atmospheric CO2 to 1–2% for slag-based CO2 mineralization. We then experimentally assess the performance of these streams in a modified direct aqueous carbonation process using BOF steel slag following hydrogen generation. Finally, we integrate the capture and utilization pathways into a unified techno-economic analysis, evaluating capital costs under membrane and electricity zero-cost scenarios, and performing breakeven analyses based on product revenues. Together, these steps provide a comprehensive assessment of both the technical performance and economic feasibility of coupling facilitated-transport m-DAC with BOF-slag-based CO2 mineralization for scalable and durable CO2 removal.
2 Background
2.1 Gas transport in membranes
Gas permeation through membranes can occur by different mechanisms depending on the material characteristics and the presence of fixed or mobile carriers. In general terms, the flux of a gas component i is governed by the pressure difference across the selective layer and the intrinsic permeability of the material: | (1) |
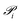 is the permeability of component i, pi,f and pi,p are its partial pressures on the feed and permeate sides, and l is the selective layer thickness.9 When l is not precisely known, the property is commonly reported as permeance (P′), measured in gas permeation units (GPU):
is the permeability of component i, pi,f and pi,p are its partial pressures on the feed and permeate sides, and l is the selective layer thickness.9 When l is not precisely known, the property is commonly reported as permeance (P′), measured in gas permeation units (GPU):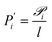 | (2) |
 and solubility
and solubility 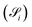 :
: | (3) |
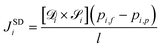 | (4) |
Although widely applied, SD membranes are limited by Robeson's upper bound, which highlights the trade-off between permeability and selectivity for polymer-based systems.16,17
| Jtotal = JSD + JFT | (5) |
 | (6) |
Compared to complex multi-parameter models, this framework represented by eqn (6), by Xu et al.20 balances simplicity with physical accuracy. It enables direct regression of kinetic and thermodynamic parameters from experimental data. This makes it particularly useful for process-level modeling and performance prediction under varying feed conditions.21
It is important to note that facilitated-transport theory indicates that the separation performance is limited by coupled diffusion-reaction behavior, including carrier saturation and the existence of an optimal binding strength; increasing Keq. without bound does not monotonically increase facilitation and can introduce release/saturation limitations.21 The aspirational values considered in this work are used as a bounding upper-limit case representative of a hypothetical high-carrier-density reactive membrane regime.
2.2 Background on cost estimation techniques
| Class | Definition (%) | Accuracy range |
|---|---|---|
| 5 – Order-of-magnitude | 0–2 | −50 to −20% (low); +30 to +100% (high) |
| 4 – Study estimate | 1–15 | −30 to −15% (low); +20 to +50% (high) |
| 3 – Budget/target | 10–40 | −20 to −10% (low); +10 to +30% (high) |
| 2 – Control/engineering | 30–70 | −15 to −5% (low); +5 to +20% (high) |
| 1 – Definitive/final | 65–100 | −3 to −1% (low); +3 to +15% (high) |
2.3 Equipment cost estimation models
When detailed project data are unavailable, equipment cost estimation models provide a practical approach to accelerate profitability evaluation. These models rely on correlations developed from empirical data, which link equipment cost to process characteristics such as throughput, phase of material (liquid, solid, or gas), or construction materials.22,23 | (7) |
The value of n typically ranges from 0.5 to 0.85 depending on the equipment and plant type, with 0.6 being widely used in the chemical industry—hence the name six-tenths rule.22,25 This equation captures the principle of economies of scale, in which larger equipment tends to cost less per unit capacity. While more detailed methods, such as vendor quotes or factorial approaches, may provide greater accuracy, the rule of thumb remains appropriate for preliminary screening studies such as the techno-economic evaluation proposed by this work.
| CBM,i = CDE,i + CIDE,i | (8) |
| CDE,i = CP0 + CM + CL | (9) |
| CIDE,i = CFIT + CO + CE | (10) |
• The direct costs (eqn (9)) include the base equipment cost (CP0), material installation costs (CM), and labor costs (CL).
• The indirect costs (eqn (10)) account for freight, insurance, and taxes (CFIT); overhead (CO) such as vacation, retirement, and unemployment expenses; and engineering and project management services (CE).
3 Methods
3.1 Membrane-based direct air capture (m-DAC) system
The TEA presented here builds on our previous work on facilitated transport membranes (FTMs) for direct air capture.1,2 A single-stage membrane module was simulated to enrich CO2 from ambient concentration (≈425 ppm) up to two different outlet conditions: 1 and 2% CO2. While the base scenario, which considered the regressed intrinsic properties from ref. 20 of Keq. = 2.63 × 10−5 Pa−1 and DCO2 = 3.79 × 10−11 × 10−8 mol m−1 s−1, was able to achieve this target purity. However, the corresponding recovery was too low to justify cost evaluation. An NLP-based optimization framework was then applied to identify the property ranges that satisfied capture requirements of at least 50% recovery and CO2 purity between 1–1.75%.2 The optimal properties identified were Keq. = 4.04 ± 1.23 Pa−1 and DCO2 = 6.57 × 10−8 ± 2.44 × 10−8 mol m−1 s−1. These values are 4–5 orders of magnitude higher than the regressed base-case membrane properties obtained in the study by,26 which employed the model suggested by.20 Therefore, only scenarios with recoveries <50% are considered in the economic analysis. In Section 5 of the SI for this work, the limited recovery simulation is shown to demonstrate its poor performance, emphasizing that the baseline techno-economic scenario presented here is not commercially viable under current conditions. The results indicate that positive economic performance is only achieved under optimistic assumptions regarding the membrane properties assumed (Fig. 1).The facilitated transport mechanism was modeled using eqn (6), where DCO2 and Keq. represent the intrinsic membrane properties of diffusivity and binding constant.20Fig. 2 presents the process flow diagram (PFD) for the two CO2 purities discussed.
3.2 Mineralization process
| 3FeO(s) + H2O(g) → Fe3O4(s) + H2(g); ΔH (600 °C) = −55.72 kJ mol−1 | (11) |
After H2 production, the steel slag is cooled and ground into a fine powder to serve as the feedstock for subsequent hydration and mineralization steps. During hydration, water reacts with the principal calcium-bearing phases in the slag—Ca3SiO5, Ca2SiO4, and CaO—as represented in (eqn (12)–(14)). Hydration of the calcium silicates yields calcium silicate hydrate (3CaO·2SiO2·8H2O, commonly referred to as CSH). The reaction pathways and corresponding heats of hydration for Ca3SiO5 and Ca2SiO4 are taken from the work of Mindess et al.27
2Ca3SiO5(s) + 11H2O(l) → 3CaO·2SiO2·8H2O(s) + 3Ca(OH)2(aq.)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ΔH = −0.52 kJ gCa3SiO5−1 ΔH = −0.52 kJ gCa3SiO5−1 | (12) |
2Ca2SiO4(s) + 9H2O(l) → 3CaO·2SiO2·8H2O(s) + Ca(OH)2(aq.)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ΔH = −0.26 kJ gCa2SiO4−1 ΔH = −0.26 kJ gCa2SiO4−1 | (13) |
CaO(s) + H2O(l) → Ca(OH)2(aq.)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ΔH (45 °C) = −84.2 kJ mol−1 ΔH (45 °C) = −84.2 kJ mol−1 | (14) |
Following hydration, the CSH and other solid components in the steel slag can be separated from the liquid phase. The remaining Ca(OH)2 solution reacts with CO2 to precipitate CaCO3. Because only part of the water consumed in eqn (12)–(14) is regenerated in eqn (15), the overall process results in a net water deficit.
5CO2(g) + 5Ca(OH)2(aq.) → 5CaCO3(s) + 5H2O(l)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ΔH (45 °C) = −468.7 kJ mol−1 ΔH (45 °C) = −468.7 kJ mol−1 | (15) |
The precipitated CaCO3 is then filtered from the liquid. At this stage, fresh steel slag and makeup water can be introduced, allowing the cycle to continue. The reaction enthalpies were calculated with data from Bale et al. (2002),28 Bale et al. (2009)29 and Reed.30
Samples of BOF steel slag were sourced from Cleveland-Cliffs – Cleveland Works. The slag was collected directly from the BOF prior to the slagging-off process and was crushed into a fine powder for experimental testing. X-ray diffraction (XRD) analysis indicated that the slag was mostly composed of Ca3SiO5, Ca2SiO4, CaO, Ca2Fe2O5, and Mg0.8Fe0.2O (Fig. S1).
H2 production performance was evaluated at 600 °C by flowing 100 sccm of 2% H2O vapor in argon over 6 g slag. Under these conditions, the slag produced 16.99 std. ml H2 per gram of slag (Fig. S2).
For mineralization, experiments were conducted using 1% and 2% CO2/Ar mixtures, with water only blank runs performed for comparison. Each cycle used 0.5 g of slag with a 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 water-to-slag mass ratio. The experiments consisted of three cycles, each containing two sequential steps: hydration followed by CO2 mineralization, with solid–liquid separation after each hydration step. During hydration, slag was reacted with water at 45 °C for 30 minutes. After separation, the mineralization step was carried out by flowing 40 sccm of either 1% or 2% CO2 through the solution for 15 minutes, followed by a 20-minute argon purge to quantify the remaining CO2 in the reactor. Fresh slag was added to the solution after the mineralization step. At the end of each hydration and mineralization step, the temperature and pH were measured. Fig. 3 presents the CO2 mineralization results for 1% and 2% CO2 feeds, showing 55.4% and 43.3% conversion respectively.
1 water-to-slag mass ratio. The experiments consisted of three cycles, each containing two sequential steps: hydration followed by CO2 mineralization, with solid–liquid separation after each hydration step. During hydration, slag was reacted with water at 45 °C for 30 minutes. After separation, the mineralization step was carried out by flowing 40 sccm of either 1% or 2% CO2 through the solution for 15 minutes, followed by a 20-minute argon purge to quantify the remaining CO2 in the reactor. Fresh slag was added to the solution after the mineralization step. At the end of each hydration and mineralization step, the temperature and pH were measured. Fig. 3 presents the CO2 mineralization results for 1% and 2% CO2 feeds, showing 55.4% and 43.3% conversion respectively.
The mass and energy balance was initiated by scaling up the slag and water quantities proportionally to match the CO2 flow rate produced by the DAC membrane system. The corresponding mass and energy balance of the mineralization system are summarized in Fig. 4 for 1% and 2% CO2 cases, alongside the overall process schematic. All three reactions in the mineralization process are exothermic. Energy balances were performed for each reactor to evaluate the impact of reaction heat on reactor temperatures. From the net heat generated or removed over the reaction period and the heat capacity of the reactor contents, the resulting temperature change was estimated and found to be negligible (ΔT < 3 °C) for the H2 production and hydration reactors. Similarly, for the CO2 capture reactor, the liquid phase was assumed to remain at 45 °C, while the inlet gas stream at 25 °C removes reaction heat as it exits the system. Between the high-temperature H2 production step and the low-temperature milling step prior to hydration, the slag cools from 600 °C to 45 °C during conveying. Since no internal process steps require additional thermal energy, this sensible heat was not recovered in the current analysis. In future process designs, the slag waste heat could potentially be utilized for low- to medium-grade heat applications, for example by coupling the cooling stage to a steam generator. Secondary unit operations such as milling, filtration, and pumping are assumed to operate isothermally at 45 °C.
3.3 Capital cost estimation
| Equipment | Capacity parameters | Units |
|---|---|---|
| a Vendor reference sizing (e.g., solids capacity × length2.5) used for quoting. | ||
| m-DAC equipment | ||
| Vacuum pump | Power | kW |
| Compressor | Power | kW |
| Heat exchanger | Heat-transfer area | m2 |
| Fan | Volumetric flow rate | m3 s−1 |
| Membrane module | Membrane area | m2 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
||
| Mineralization equipment | ||
| Water splitting reactor | Reactor volume | m3 |
| Mill | Solids throughput | Mg h−1 |
| Hydration reactor | Reactor volume | m3 |
| CO2 capture spray tower | Gas flow rate | L s−1 |
| Fan, centrifugal | Gas flow rate | L s−1 |
| Pump, centrifugal | Liquid flow rate | L s−1 |
| Rotary drum filter—slag | Filter area | m2 |
| Rotary drum filter—CaCO3 | Filter area | m2 |
| Conveyor | Vendor reference indexa | (Mg h−1) m2.5 |
To estimate total fixed capital cost, the methodology developed by Towler, Gavin P., Sinnott, Ray K. (2021) was applied. Multipliers for installation, piping, control, and other items were included, leading to a factor of 6.0 on purchase cost for the m-DAC, since the CO2 capture operation only handles gases, while the factor of 6.05 was used for the mineralization, since the latter involves both solids and gases.22 Additionally, as noted in Section 3.2.2, the 1% permeate system required 4 units of membrane based capture, while 2% requires 5 units. Fig. 5 translate this difference in associated CAPEX for each CO2 concentration considered. With that in mind, a multiplier for the total m-DAC cost was introduced to meet this objective based on the overall yield for each permeate condition, ultimately leading to the highest yield mineralization system to require less m-DAC units.
Using this method, the total fixed capital cost was estimated, considering 2015 as the base year (CEPCI = 556.8), and later updated using the CEPCI index for 2024 (CEPCI = 801.9) to correct the inflation effects.
For labor, a fixed staffing assumption was used for both process configurations. Specifically, five operators per shift were assumed in total, including three operators for the m-DAC section and two operators for the mineralization section, with an annual wage of $63![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 210 per operator.33 This corresponds to an annual labor cost of approximately $316
210 per operator.33 This corresponds to an annual labor cost of approximately $316![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 in both the 1% and 2% CO2 feed cases. Therefore, the larger labor share reported in the 2% case reflects the lower total OPEX of that case, driven primarily by reduced electricity demand, rather than a substantial increase in absolute labor requirements.
000 in both the 1% and 2% CO2 feed cases. Therefore, the larger labor share reported in the 2% case reflects the lower total OPEX of that case, driven primarily by reduced electricity demand, rather than a substantial increase in absolute labor requirements.
 | (16) |
A positive NPV (NPV > 0) indicates that discounted revenues exceed the total investment, making the project economically attractive, whereas NPV < 0 shows that revenues are insufficient to offset costs. The breakeven point occurs at NPV = 0, when discounted revenues exactly match capital and operating expenses and the internal rate of return equals the assumed discount rate. In addition to NPV, the breakeven analysis is used to determine when cumulative revenues offset total investment, using both simple (undiscounted) and discounted payback times. Together, these metrics provide a quantitative basis for evaluating long-term project viability under varying electricity prices, slag prices, and feed CO2 conditions.
 | (17) |
The total annualized cost is then given by:
 | (18) |
 represents the average annual operating expenditure, including electricity, labor, maintenance, and other recurring costs. This approach can also compute any major component replacements or overhauls that may occur during the project lifetime, their future costs are discounted to present value and annualized using the same CRF formulation. Salvage values, if/when applicable, are discounted similarly and subtracted from the total capital cost. Annualizing costs enables consistent comparison among design alternatives that differ in scale or lifetime. This facilitates the calculation of levelized metrics (e.g., cost per ton of CO2 captured or mineralized), allowing direct evaluation of the influence of electricity price, slag price, and number of m-DAC units on the system's overall economic performance.
represents the average annual operating expenditure, including electricity, labor, maintenance, and other recurring costs. This approach can also compute any major component replacements or overhauls that may occur during the project lifetime, their future costs are discounted to present value and annualized using the same CRF formulation. Salvage values, if/when applicable, are discounted similarly and subtracted from the total capital cost. Annualizing costs enables consistent comparison among design alternatives that differ in scale or lifetime. This facilitates the calculation of levelized metrics (e.g., cost per ton of CO2 captured or mineralized), allowing direct evaluation of the influence of electricity price, slag price, and number of m-DAC units on the system's overall economic performance.
4 Results and discussion
4.1 Capital expenses
As the first step in the economic analysis of the capture, the process equipment estimated cost was calculated considering eqn (7), using the appropriate capacity factor required. It is important to note that the m-DAC capture and the mineralization had to be adapted for two different CO2 concentrations—1% and 2%. In order to properly report the associated costs with the process, all of the base purchase costs were assumed to have 2015 as reference year and consequently corrected to 2024. The productivity of a single m-DAC system has to be scaled-up by assuming parallel units to achieve the goal of removing at least 1000 tons of CO2 per year, seeking to qualify for the 45Q tax credit. Prior to cost estimation, the APS flowsheets for the m-DAC system were optimized, focusing on reducing cooling-water demand in the compression train and adjusting heat-exchange duties to maintain equipment within safe operating limits.Both cases show a similar CAPEX breakdown, with the major contribution attributed to the m-DAC units. This could be primarily attributed to the vacuum pumps and compressors used in the m-DAC process. Notably, the setup incorporating additional m-DAC units to offset reduced mineralization efficiency leads to a marginally lower overall CAPEX proportion for m-DAC (60.5% compared to 64.3%). This suggests that, from a capital investment standpoint, scaling up m-DAC may be more cost-effective than increasing the yield or throughput of the mineralization stage. This finding aligns with modularity-driven economies of scale: m-DAC units could often be scaled incrementally with relatively minor cost penalties, while mineralization systems may face nonlinear CAPEX growth due to solid handling, reactor sizing, or thermal integration constraints.
4.2 Operational expenses
The operating expenditure (OPEX) shares for both cases, shown in Fig. 6, are dominated by electricity consumption, which accounts for over 74% of total OPEX in each case. Notably, the process with fewer m-DAC units and higher mineralization performance exhibits a slightly higher electricity share (75.7%) compared to the alternative case with more m-DAC units (74.3%).This increase reflects the fact that the 1% case requires higher permeation across the membrane to meet the same capture target. The resulting higher permeate flow imposes a larger load on the compression train, increasing compressor power requirements and increasing the electricity share of the overall OPEX. However, because the labor expenses were treated as a fixed annual operating cost in both cases, its share of total OPEX increases in the 2% CO2 case as the overall OPEX decreases, mainly due to lower electricity demand. Utility and maintenance costs remain relatively constant, indicating that the economic breakeven point mostly lies on energy consumption and product revenue. Overall, this comparison shows the delicate balance between modularity, and energy efficiency in capturing and mineralizing CO2.
The advantage of coupling capture and utilization is discussed here to envision a scenario where a “carbon economy” drives investment to DAC and decarbonization processes through the 45Q tax credit. The revenue breakdown shown in Fig. 7 includes the U.S. 45Q tax credit, assumed at $100 per ton of CO2 permanently stored via mineralization. This value corresponds to the 45Q incentive level adopted in this study and is treated as a fixed revenue stream directly linked to the amount of CO2 mineralized. In addition, product revenues are calculated using a hydrogen selling price of $2 per kg and a carbonated slag selling price of $26 per ton. These values are held constant in the baseline NPV analysis; only the carbonated slag price is varied in the breakeven analysis (Section 4.4), where it is back-calculated to determine the price required to achieve NPV = 0 over the project lifetime. Additionally, during the carbonation of the steel slag, besides the carbonated slag, hydrogen is generated as a byproduct which fortunately can also be sold, adding value to the combined process. In addition to these direct revenue streams, utilizing BOF steel slag offers an implicit economic advantage by avoiding disposal and handling costs. Steel slag is often sent to landfills due to its limited reactivity, creating long-term management burdens for steel producers. Although these avoided costs were not explicitly quantified in this TEA, they represent a meaningful indirect economic benefit that could further improve the viability of the integrated m-DAC and mineralization process.
4.3 NPV analysis
In the 1% case, the analysis spans a 45-year operational horizon with a 10% discount rate. Although a 10% discount rate is typically associated with shorter economic evaluations, it was held constant here due to the early-stage nature of m-DAC and the lack of established discounting practices for long-term DAC infrastructure.Maintaining a fixed rate provides a consistent baseline for comparison while acknowledging the uncertainty surrounding appropriate risk-adjusted rates for emerging carbon-removal technologies. The key TEA results for this case are:
• Initial CAPEX: −$57.5 million
• Annual revenue: $2.33 million
• Annual OPEX: −$2.14 million
• Annual profit (undiscounted): $188![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000
• Resulting NPV: −$55.6 million
• Payback time: over 300 years (unrealistic within project lifetime)
• Discounted breakeven: not reached within 45 years
The NPV plot, in Fig. 8, confirms the above outcomes, showing a flat, negative trend throughout the plant's life. Despite modest annual profits, the magnitude of the CAPEX investment vastly outweighs the cumulative returns. Importantly, the NPV does not show a trend toward zero at any point, which implies that under current economic and technical assumptions, this configuration is fundamentally nonviable from a financial standpoint. This result suggests that improvements in revenue generation (e.g., product pricing or carbon credit incentives) or cost reduction (especially CAPEX) would be essential to make this design attractive.
 | ||
| Fig. 8 NPV profile for the 1% CO2 feed case, based on discounted cash-flow analysis using CO2 permeate from the m-DAC capture unit as the feed to the mineralization step. | ||
In contrast to the 1% scenario, the 2% configuration has a reduced capital expenditure but fails to achieve any positive yearly profit, resulting in a slightly less negative but still unfavorable NPV.
Key results include:
• Initial CAPEX: −$40.3 million, about 30% lower than the 1% case.
• Annual revenue: $1.50 million, substantially reduced due to lower mineralization performance and possibly lower throughput.
• Annual OPEX: −$1.81 million, moderately lower than in the 1% case.
• Annual profit: negative at −$302![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, meaning the plant operates at a net annual loss.
000, meaning the plant operates at a net annual loss.
• NPV @ 10%: −$43.3 million
• Simple payback: not applicable, due to no profitable years.
• Discounted breakeven: not reached within the 45-year horizon
The cash flow plot further confirms the poor economic outcome: the cumulative discounted cash flow declines continuously, although with a lower slope compared to the 1% case. This reflects lower upfront investment and operating costs, but the inability to cover them due to insufficient revenue generation. Although the 2% case benefits from a lower capital cost, the drop in revenue and the shift from marginal profit to annual losses results in a project that is still economically unattractive. This result highlights a critical insight: lower CAPEX does not guarantee better economic performance if operational revenues decline disproportionately, particularly when fixed OPEX and depreciation continue to apply. For both configurations, the root issue is insufficient annual cash flow to justify the scale of investment. It should be further noted that the m-DAC simulations considered membrane material properties such as DCO2 and Keq. to be 4–5 orders of magnitude times higher than the values regressed from pior literature. Therefore, these values are already aspirational, and further improvements in their magnitudes cannot be expected.
When comparing the discounted cumulative cash-flow trend for the 1% and 2% feed cases, the key difference in their shapes arises from the sign of the annual operating profit. In the 1% configuration, annual revenue ($2.33 million) marginally exceeds annual OPEX ($−2.14 million), yielding a small positive yearly profit of approximately $188![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. Even after discounting, this positive rate contributes to slight incremental gains each year, producing the small upward drift observed in the cumulative discounted cash flow curve, although the magnitude of the annual profit is far too small to offset the large initial CAPEX. In contrast, the 2% case operates at an annual loss, as revenue ($1.50 million) falls below OPEX ($−1.81 million), resulting in a negative yearly profit of −$302
000. Even after discounting, this positive rate contributes to slight incremental gains each year, producing the small upward drift observed in the cumulative discounted cash flow curve, although the magnitude of the annual profit is far too small to offset the large initial CAPEX. In contrast, the 2% case operates at an annual loss, as revenue ($1.50 million) falls below OPEX ($−1.81 million), resulting in a negative yearly profit of −$302![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. Consequently, each operational year adds a discounted deficit rather than a surplus, causing the cumulative cash flow to decline steadily over the 45-year horizon.
000. Consequently, each operational year adds a discounted deficit rather than a surplus, causing the cumulative cash flow to decline steadily over the 45-year horizon.
It is important to note that a direct comparison between the present m-DAC system and conventional DAC technologies is not strictly equivalent. Most large-scale DAC projects are designed to produce high-purity CO2 streams (95–99%), which are suitable for compression and geological storage, with reported capture costs in the range of $600–900 per ton CO2 removed (e.g., McQueen et al.37). In contrast, the present system is intentionally designed to deliver a low-purity (1–2%) CO2 stream for direct downstream mineralization. Since separation cost strongly depends on the target purity and pressure specifications, comparing a 1–2% product to a 95–99% product would not reflect equivalent system objectives.
4.4 Breakeven analysis
Given the high capital and operating expenditures associated with m-DAC and mineralization processes, additional NPV calculations were conducted to assess the economic sensitivity of the system to two critical parameters: membrane capital cost and electricity price. These parameters were selected due to their influence on the capital (membranes) and operating (electricity) sides of the techno-economic analysis, as shown in the CAPEX/OPEX breakdowns in Fig. 5 and 6 and NPV evaluations in Fig. 8 and 9. To isolate the role of electricity cost, the NPV profiles of both configurations were recalculated assuming free electricity, effectively removing the dominant OPEX component (previously ≈75%). | ||
| Fig. 9 NPV profile for the 2% CO2 feed case, based on discounted cash-flow analysis using CO2 permeate from the m-DAC capture unit as the feed to the mineralization step. | ||
In the 1% case, Fig. 10(a), the removal of electricity cost significantly flattens the cash flow trajectory but does not enable breakeven within the 45-year window. The NPV remains negative, though the magnitude of the loss decreases. In the 2% case, Fig. 10(b), the reduced OPEX from free electricity improves performance similarly. The NPV becomes less negative, indicating partial mitigation of financial losses. This scenario demonstrates that electricity price is a critical limiting factor, but not the sole barrier to economic viability. Even in the absence of energy costs, the high CAPEX and low revenue (especially from carbonated slag) continue to hinder positive returns. The next analysis explores the impact of eliminating membrane CAPEX, simulating a best-case scenario where membrane technology becomes low-cost or is subsidized. When combined with the previously analyzed free electricity scenario, this will help identify whether current unprofitability is primarily driven by capital intensity, energy dependence, or insufficient revenue streams. The discounted cumulative cash flow plots for both 1% and 2% scenarios under the free membrane cost assumption provide a sharp contrast with the earlier free electricity cases (Table 3):
| Scenario | 1% CO2 feed case | 2% CO2 feed case | Impact |
|---|---|---|---|
| Free electricity | Moderate recovery but still negative | Slope reduction, smaller NPV loss | High |
| Free membrane | Minor improvement, near-flat trend | Slightly smaller initial loss | Low |
With this framing in place, understanding the important role of electricity in cost and disregarding the influence of the membrane impact, the slag price will be assessed seeking to find a breakeven point by determining the minimum slag selling price that would make either case (or both) financially viable. To identify the market conditions under which the process may become economically viable, a breakeven analysis was conducted by adjusting the selling price of the carbonated slag product while keeping all other parameters constant. This approach directly addresses the revenue deficit observed in the baseline NPV analysis.
The breakeven analyses for both scenarios were designed by setting the project lifetime (45 years) as a parameter and adjusting the carbonated slag selling price to ensure economic viability within that time frame. This approach provides insight into the minimum viable product price necessary to recover investment and achieve a NPV = 0 under a fixed operational horizon and discount rate. For the 1% CO2 use case, a slag price of $99.26 per ton enables breakeven exactly at year 45 (Fig. 11). This value was selected to remain under the $100 per ton threshold, reflecting a potential upper limit for market feasibility in commodity applications. The curve shows a consistent rise in cumulative value with time, driven by positive annual cash flows, but the expressive CAPEX at year 0 underscores the barrier for profitability. This extended payback period, combined with the relatively high product price needed, suggests that the economic viability of this configuration is highly sensitive to slag market conditions.
A similar breakeven analysis was performed for the 2% CO2 use configuration by adjusting the slag selling price such that cumulative discounted cash flows reach zero by year 45. As shown in Fig. 12, a slag price of $116.17 per metric ton is required for the process to achieve breakeven precisely at the end of the 45-year operational period. Compared to the 1% case, the required slag price is higher by approximately $17 per ton. This increase can be attributed to the greater capital and operational costs of the 2% CO2 permeate design, which includes one additional m-DAC unit. The presence of this extra unit increases not only the initial investment but also the recurring OPEX, particularly in terms of electricity and labor, as previously noted in the OPEX breakdown. Notably, in both cases, the breakeven was not an unknown variable in the TEA, but rather a targeted constraint used to back-calculate the necessary revenue (via slag pricing). This allowed us to precisely identify a critical threshold for the carbonated slag and ultimately inform market development efforts and policy actions. Table 4 summarizes the TEA analysis comparing the levelized costs per ton of CO2 that ultimately is mineralized and the cost considering just capture without utilization.
 | ||
| Fig. 12 Discounted cumulative cash flow for the 2% CO2 feed case over 50 years at 10% discount rate, assuming a slag selling price of $116.17 per ton. Breakeven occurs at year 45. | ||
| Cost category | 1% feed | 2% feed |
|---|---|---|
| Overall capture & mineralization | $8203 | $6225 |
| Overall capture only | $3221 | $1868 |
4.5 Emissions impact
The carbon intensity of the capture process was evaluated by coupling the m-DAC and mineralization systems with different electricity supply scenarios, ranging from fully powered by the US grid to fully renewable energy and mixed configurations. While grid-powered operation results in net-positive emissions due to the high carbon footprint of conventional electricity generation, progressively increasing the renewable share leads to a clear reduction in total emissions. At high renewable fractions, both 1% and 2% feed cases achieve net-negative operation, with the 2% feed configuration consistently showing greater CO2 removal owing to its higher process efficiency and improved mineralization performance.With grid power, both cases are net positive emitters because electricity-related emissions dominate the carbon balance.
Under 100% renewables, both systems become net negative; the 2% feed removes slightly more CO2 overall (Table 7).
With a 75% renewable share, both configurations achieve net-negative emissions, with deeper removal for the 2% feed.
5 Conclusions
The techno-economic evaluation indicates that the viability of this process remains strongly dependent on its integration with a downstream utilization pathway. When considered by itself, the capture step alone exhibits prohibitively high costs—reaching $3221.33 and $1868.03 per ton of CO2 for the 1% and 2% CO2 permeates, respectively (Table 4). These costs are well above current benchmarks for DAC technologies, confirming that membranes, though promising for continuous modular operation, require strong economic incentives and/or a revenue-generating utilization process to be competitive.When capture is coupled with carbonated slag production, the cost per ton of CO2 utilized are $8203.05 and $6224.62 for the 1% and 2% CO2 feed conditions, respectively. Despite proposing a new pathway to associate capture and utilization of CO2, the overall process remains capital-intensive, with initial investments dominated by the m-DAC subsystem (approximately 60–65% of total CAPEX; Fig. 5), mostly due to the multiple number of m-DAC units required. Furthermore, electricity contributes over 74% of OPEX (Fig. 6), emphasizing that operational viability hinges on the availability of low-cost, low-carbon power. This relationship was further seen by the free-electricity sensitivity tests, which showed that even complete removal of energy costs could not ensure a positive NPV within a 45-year lifetime. Hence, the energy intensity of the process is found to be the main bottleneck, followed by limited product revenue.
A key implication of this bottleneck is that m-DAC is unlikely to become competitive through bulk air compression alone. Prior m-DAC studies have shown that, because atmospheric CO2 is extremely dilute, the main technical challenge is generating sufficient transmembrane driving force without surpassing prohibitive power demands. In this context, permeate-side vacuum is generally more energy-favorable than feed compression, although it also increases membrane-area requirements and places greater importance on low pressure-drop module design.9,38,39 At the materials level, the literature further indicates that progress will depend not only on increasing intrinsic CO2/N2 selectivity and CO2 permeance, but also on translating these properties into scalable ultra-thin asymmetric or facilitated-transport membranes that retain performance under humid, oxygen-containing DAC conditions while resisting physical aging, swelling, and carrier instability. Thus, the most credible pathway forward is a lower-energy m-DAC system in which membranes operate as modular pre-concentration units. This system would be supported by vacuum- or sweep-assisted operation and low pressure-drop contactors. It would also rely on hybrid integration with downstream mineralization or other utilization steps that can handle intermediate purity CO2 streams, rather than requiring high purity CO2 at the capture stage.
The breakeven analyses also reinforce these findings. For the process to achieve economic neutrality within the project lifetime (45 years), the carbonated slag must be sold at $99.26 per ton for the 1% configuration and $116.17 per ton for the 2% configuration (Fig. 11 and 12). These prices are three to five times higher than the projected market value of carbonated steel slag ($20–$30 per ton) and exceed the selling price of ground granulated blast furnace slags (GGBFS) (≈50 $ per ton). This suggests that the process would only be profitable if the carbonated slag can be further refined as a SCM or as a material that can qualify for carbon credit incentives.
From an environmental standpoint, the system's carbon emissions exhibited a high correlation to the electricity source. When fully powered by the current U.S. grid mix, both configurations act as net emitters, releasing 2023 and 1012 tons of CO2 per year for the 1% and 2% cases, respectively (Table 5). Transitioning to renewable energy drastically shifts the outcome, achieving net-negative emissions of up to −720 and −783 tons CO2 per year under 100% renewable supply (Table 6). Even partial renewable integration (e.g., 75% renewable) yields small but measurable net removal, confirming that decarbonizing the electricity supply is a prerequisite for this technology to contribute effectively to negative emissions targets.
| 1% feed | 2% feed | |
|---|---|---|
| a Renewable sources represent a share of 40% (including nuclear power sources) of the total U.S. grid power generation. | ||
| Total CO2 emitted [ton per year] | 2994.10 | 1958.92 |
| Total CO2 captured [ton per year] | 970.96 | 947.17 |
| Net CO2 emissions [ton per year] | 2023.14 | 1011.75 |
| 1% feed | 2% feed | |
|---|---|---|
| Total CO2 emitted [ton per year] | 250.87 | 164.13 |
| Total CO2 captured [ton per year] | 970.96 | 947.17 |
| Net CO2 emissions [ton per year] | −720.09 | −783.04 |
| 1% feed | 2% feed | |
|---|---|---|
| Total CO2 emitted [ton per year] | 961.76 | 702.57 |
| Total CO2 captured [ton per year] | 970.96 | 947.17 |
| Net CO2 emissions [ton per year] | −34.28 | −65.13 |
Overall, this study shows that although membrane-based DAC paired with BOF steel-slag mineralization can technically deliver continuous carbon capture and sequestration, its economic feasibility hinges on favorable market and policy conditions; particularly scenarios that combine low-carbon electricity, lower membrane and compression costs, and effective valorization of both carbonated slag and hydrogen byproducts. Under these conditions, and with continued progress in renewable-energy integration and material upgrading, the proposed hybrid process could become a scalable, regionally deployable pathway for permanent CO2 removal.
Conflicts of interest
There are no conflicts to declare.Data availability
Data supporting this publication are available from the corresponding author upon reasonable request.Supplementary information (SI): additional experimental, modeling, and techno-economic details supporting the analysis presented in the main text; CO2 mineralization reaction data and procedures, mass and energy balance calculations, supplementary m-DAC simulation discussions, and detailed equipment sizing parameters used for cost estimation to support transparency and reproducibility. See DOI: https://doi.org/10.1039/d5su00924c.
Acknowledgements
V. G., F. V. L. and O. S. acknowledge funding from NSF RII Track-2 FEC (OIA-2119688) and NASA Established Program to Stimulate Competitive Research through Grant #80NSSC22M0027. K. S. and S. Z. acknowledge support from the Sustainability Research Seed Grant at The Ohio State University. The authors thank Cleveland-Cliffs – Cleveland Works for providing steel slag samples. Support from the Center for Electron Microscopy and Analysis (CEMAS) at The Ohio State University is gratefully acknowledged. K. S. is supported by the Distinguished University Fellowship and the ENGIE-Axium Fellowship at The Ohio State University.Notes and references
- V. Gama, B. Dantas, O. Sanyal and F. V. Lima, ACS Eng. Au, 2024, 4, 394–404 CrossRef CAS PubMed.
- V. Gama, D. Roy, F. V. Lima and O. Sanyal, Ind. Eng. Chem. Res., 2025, 64, 8375–8389 CrossRef CAS PubMed.
- Global Monitoring Laboratory, Trends in CO2, NOAA Global Monitoring Laboratory, 2025, https://gml.noaa.gov/ccgg/trends/monthly.html, accessed on: 2025-08-07 Search PubMed.
- U. Nations, Goal 13: Take urgent action to combat climate change and its impacts, 2025, https://www.un.org/sustainabledevelopment/climate-change/.
- U. Nations, Net Zero Coalition, Publisher, United Nations, 2025, https://www.un.org/en/climatechange/net-zero-coalition, accessed 7 August 2025 Search PubMed.
- Direct Air Capture – Energy System, 2025, https://www.iea.org/energy-system/carbon-capture-utilisation-and-storage/direct-air-capture.
- EFI Foundation, Direct Air Capture: The Rapidly Expanding Landscape – From Kiloton to Megaton Scale, 2025, https://efifoundation.org/foundation-reports/annual-report-2024/.
- Y. Jia, K. Wong, C. Z. Liang, J. Wu, T.-S. Chung and S. Zhang, Prog. Mater. Sci., 2024, 146, 101324 CrossRef CAS.
- C. Castel, R. Bounaceur and E. Favre, Front. Chem. Eng., 2021, 3, 668867 CrossRef.
- H. Leflay, J. Pandhal and S. Brown, J. CO2 Util., 2021, 52, 101657 CrossRef CAS.
- Q. Song, M.-Z. Guo, M. Zhang and T.-C. Ling, J. Clean. Prod., 2024, 469, 143214 CrossRef CAS.
- T. Hottle, T. R. Hawkins, C. Chiquelin, B. Lange, B. Young, P. Sun, A. Elgowainy and M. Wang, J. Clean. Prod., 2022, 363, 131834 CrossRef CAS.
- Y. Zhang, L. Yu, K. Cui, H. Wang and T. Fu, Chem. Eng. J., 2023, 455, 140552 CrossRef CAS.
- Y. Wang, J. Liu, X. Hu, J. Chang, T. Zhang and C. Shi, J. Sustainable Cem.-Based Mater., 2023, 12, 471–486 CrossRef CAS.
- P. Li, Y. Chen, X. Li, B. Yan, D. Chen and H. Guo, Int. J. Hydrogen Energy, 2020, 45, 17140–17152 CrossRef CAS.
- L. M. Robeson, J. Membr. Sci., 2008, 320, 390–400 CrossRef CAS.
- R. Sidhikku Kandath Valappil, N. Ghasem and M. Al-Marzouqi, J. Ind. Eng. Chem., 2021, 98, 103–129 CrossRef CAS.
- Y. Chen, L. Zhao, B. Wang, P. Dutta and W. Winston Ho, J. Membr. Sci., 2016, 497, 21–28 CrossRef CAS.
- Z. Tong and W. S. W. Ho, Sep. Sci. Technol., 2017, 52, 156–167 CrossRef CAS.
- H. Xu, S. G. Pate and C. P. O'Brien, Chem. Eng. J., 2023, 460, 141728 CrossRef CAS.
- R. Rea, M. De Angelis and M. Baschetti, Membranes, 2019, 9, 26 CrossRef PubMed.
- G. P. Towler and R. K. Sinnott, Chemical Engineering Design : Principles, Practice and Economics of Plant and Process Design, Butterworth-Heinemann, 2nd edn, 2021 Search PubMed.
- Analysis, Synthesis, and Design of Chemical Processes, ed. R. Turton, Prentice Hall, Boston, 5th edn, 2018 Search PubMed.
- D. R. Woods, Rules of Thumb in Engineering Practice, Wiley, 1st edn, 2007 Search PubMed.
- S. Lemmens, Energies, 2016, 9, 485 CrossRef.
- Y.-Y. Lee, N. P. Wickramasinghe, R. Dikki, D. L. Jan and B. Gurkan, Nanoscale, 2022, 14, 12638–12650 RSC.
- S. Mindess, J. F. Young and D. Darwin, Concrete, Prentice Hall, 2nd edn, 2003 Search PubMed.
- C. Bale, P. Chartrand, S. Degterov, G. Eriksson, K. Hack, R. Ben Mahfoud, J. Melançon, A. Pelton and S. Petersen, Calphad, 2002, 26, 189–228 CrossRef CAS.
- C. Bale, E. Bélisle, P. Chartrand, S. Decterov, G. Eriksson, K. Hack, I.-H. Jung, Y.-B. Kang, J. Melançon, A. Pelton, C. Robelin and S. Petersen, Calphad, 2009, 33, 295–311 CrossRef CAS.
- J. J. Reed, The NBS Tables of Chemical Thermodynamic Properties: Selected Values for Inorganic and C1 and C2 Organic Substances in SI Units, 1989, https://data.nist.gov/od/id/mds2-2124.
- 45Q Tax Credit for Carbon Capture Projects. Carbon Capture Coalition, https://carboncapturecoalition.org/resource/45q-tax-credit-for-carbon-capture-projects/, accessed on: 2026-03-11.
- Hydrogen Production. Energy.gov, https://www.energy.gov/eere/fuelcells/hydrogen-production, acessed on: 2026-03-17.
- Chemical Equipment Operators and Tenders. Bureau of Labor Statistics, https://www.bls.gov/oes/2023/may/oes519011.htm, acessed on: 2026-03-11.
- U.S. Energy Information Administration, Energy Implications of Potential Iron and Steel-Sector Decarbonization Pathways, U.S. Department of Energy, ieo2021 issues in focus, 2022 Search PubMed.
- K. Shank, H. Xu, Y. Xu, A. A. Kermani, J. Qin and S. Zhai, Energy Environ. Sci., 2026, 19, 2194–2212 RSC.
- N. Badger, R. Boylu, V. Ilojianya, M. Erguvan and S. Amini, Energy Adv., 2024, 3, 2311–2327 RSC.
- N. McQueen, K. V. Gomes, C. McCormick, K. Blumanthal, M. Pisciotta and J. Wilcox, Prog. Energy, 2021, 3, 032001 CrossRef CAS.
- S. Fujikawa, R. Selyanchyn and T. Kunitake, Polym. J., 2021, 53, 111–119 CrossRef CAS.
- P. Panja, C. Manankandayalage, M. M. Alam and M. Deo, J. Appl. Polym. Sci., 2023, e54802 Search PubMed.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |









