Open Access Article
Open Access ArticleH-bond network-facilitated, diversity-oriented, green synthesis of valuable organic compounds via atom-economic, regio- and stereo-selective reactions involving ynamides and anilines
Appanapalli N. V.
Satyanarayana
and
Tanmay
Chatterjee
 *
*
Department of Chemistry, Birla Institute of Technology and Science, Pilani (BITS Pilani), Hyderabad Campus, Jawahar Nagar, Kapra Mandal, Medchal District, Telangana 500078, India. E-mail: tanmay@hyderabad.bits-pilani.ac.in
First published on 13th January 2026
Abstract
Diversity-oriented synthesis (DOS) enables synthetic organic chemists to fabricate structurally diverse classes of molecules, which is highly desirable in the context of medicinal chemistry, thus leading to drug discovery. Herein, we report the H-bonding network-mediated DOS of five structurally diverse classes of molecules, such as acetimidamides, ketene N,N-acetals, α-arylenamides, some heterocycles, such as quinolines, and 3,4-dihydroquinolin-4-ols, starting from readily available ynamides and anilines in the presence of HFIP as the only reagent/solvent. 2-Amidoindoles were also synthesized via a one-pot, two-step process starting from ynamides and primary anilines using CuCl2. The significant benefits of this protocol compared with the previously established synthetic methods are the following: (a) a cost-effective, straightforward, and unified approach for diversity-oriented, green synthesis of various classes of valuable molecules from ynamides and all types of aromatic amines (1°, 2°, and 3°); (b) a metal-free, environmentally friendly, and scalable protocol; (c) mild reaction conditions; (d) 100% atom-economic transformations; (e) HFIP being the sole reagent necessary for the transformations; (f) the dual functionality of HFIP as both a Brønsted acid and a solvent; (g) the regeneration of HFIP post-reaction and its straightforward and effective recovery from the reaction mixture through distillation; (h) the reusability and efficient recyclability of HFIP without compromising the subsequent reaction's results; and (i) the ability to obtain pure products under various conditions without the need for column chromatography (green synthesis).
Sustainability spotlightWe developed a diversity-oriented, versatile synthetic method to access five different classes of valuable organic molecules from readily available ynamides and all types of aromatic amines (1°, 2°, and 3°) with excellent yield and regio- and stereo-selectivity using only HFIP, which was efficiently recovered after the reaction, reused and recycled (five runs) without any compromise on the reaction outcome. Most of the reactions are 100% atom-economic, and pure products were obtained without the need for work-up and column chromatography, resulting in minimal waste generation. Thus, the generality, practicality, cost-effectiveness, sustainability, and circular economy of this protocol were found to be significantly better than those of all existing protocols. This protocol is highly environmentally benign and greener. |
Introduction
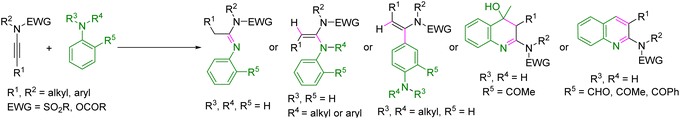
Diversity-oriented synthesis (DOS) mainly aims to produce skeletally diverse small molecules in an efficient manner.1 It is a very important and unique tool in medicinal chemistry and drug discovery, which require the synthesis of libraries of structurally diverse small molecules.2 Over the past few decades, ynamides, a unique type of unsymmetrical nitrogen-containing internal alkynes, have garnered significant interest from synthetic organic chemists as valuable building blocks for the synthesis of various important classes of molecules.3 The polarization of the nitrogen lone pair renders the β-carbon of the ynamides nucleophilic, while α-carbon exhibits electrophilic character. This distinction creates a unique environment that is conducive to regioselective difunctionalization reactions, facilitating the synthesis of tetrasubstituted alkenes. Consequently, several synthetic strategies have been developed for the difunctionalization of ynamides and the synthesis of functionalized enamides.4,5 The synthesis of enamides has garnered considerable interest among synthetic organic chemists due to their prevalence in depsipeptides, pharmaceuticals, and notable natural products.6 Furthermore, enamides serve as versatile reactants in various contexts, including transition metal catalysis, photochemistry, and asymmetric catalysis. To date, some synthetic strategies have been developed for the hydroamination/hydroarylation and cycloaddition reactions of ynamides with anilines. Several metals such as Au,7 Ag,8 Zn,9 and Yb10 have been used to catalyze the hydroamination of ynamides with primary aromatic amines for the synthesis of acetimidamides (Scheme 1A). A metal-free, acid-mediated hydroamination of ynamide with primary and secondary aromatic amines was reported by Junbiao Chang et al. using TfOH (0.5 equiv.) for the synthesis of acetimidamides and α-aminoenamides, respectively (Scheme 1A).11 Moreover, Guo-Qiang Lin et al. developed a Ni-catalysed hydroamination of ynamides with secondary aromatic amines for the synthesis of α-aminoenamides using Ni(OTf)2 as the metal-catalyst under refluxing conditions (Scheme 1A).12 However, when tertiary aromatic amines such as 1-phenylpyrrolidine and N,N-dialkylanilines were reacted with ynamides in the presence of AgNTf2 (10 mol%), hydroarylation of ynamides occurred instead of hydroamination (Scheme 1A).13 Dubovtsev et al. disclosed an Au-catalyzed annulation of 2-aminobenzaldehydes and 2-aminoketones with ynamides using Ph3PAuCl/AgNTf2 and molecular sieves for the synthesis of valuable N-heteroaromatics, i.e., 2-aminoquinolines (Scheme 1B).14 Moreover, a few synthetic strategies have been developed for the direct or one-pot, two-step synthesis of 2-aminoindoles from ynamides and anilines or 2-iodoanilines under Au-catalysis or Pd-catalysis. Youn et al. reported a one-pot two-step synthesis of 2-aminoindoles from ynamides and anilines using catalytic PPh3AuCl and AgOTf in the first step, furnishing the corresponding acetimidamides, which then underwent a Cu-mediated intramolecular oxidative annulation in the second step to produce 2-aminoindoles (Scheme 1B).15 The Ruhland and Skrydstrup group disclosed the Pd-catalyzed direct synthesis of 2-aminoindoles from 2-iodoaniline and terminal ynamides via Sonogashira coupling followed by a base-mediated intramolecular cyclization reaction using Pd(OAc)2 (5 mol%), PPh3 (20 mol%) and NaOAc (4 equiv.).16Despite the notable advancements, the developed strategies suffered from several serious limitations, including the requirements of highly expensive and hazardous transition metal complexes and ligands, or strong acids (TfOH), and harsh reaction conditions, poor stereoselectivity in some cases, and the generation of metal-based and/or organic wastes. Moreover, no synthetic strategy has been developed to date for the diversity-oriented, versatile synthesis of valuable products involving ynamides and all types of aromatic amines (1°, 2°, and 3°) under metal-free and sustainable conditions, which is highly desirable in the context of green and sustainable chemistry. HFIP is an excellent alternative to the noble metals mentioned above and also eliminates the need for harsh reaction conditions.17 Herein, we report a H-bond network-mediated hydroamination or hydroarylation of ynamides with (hetero)aromatic amines (1°, 2°, and 3°) and also cascade hydroamination followed by intramolecular cyclization strategies for the DOS of various useful classes of molecules such as acetimidamides, α-aminoenamides, α-arylenamides, and various bioactive heteroaromatics, such as 2-quinolines and hydroxyquinolines, and indoles, just by using HFIP at room temperature (Scheme 1C). This strategy offers several advantages and overcomes the limitations of the previously developed synthetic methods.
Results and discussion
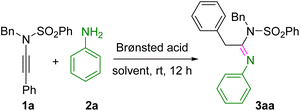
We commenced our investigation by reacting aniline (2a) with N-benzyl-N-(phenylethynyl)benzenesulfonamide (1a) in the presence of HFIP (0.6 M) at ambient temperature, and gratifyingly, the desired product, (E)-N-benzyl-N′,2-diphenyl-N-(phenylsulfonyl)acetimidamide 3aa, was obtained in an excellent yield of 95% (entry 1, Table 1). However, when HFIP was replaced with trifluoroethanol as the solvent, the reaction did not proceed, and the starting material (1a) was completely recovered (entry 2, Table 1). When the reaction was conducted in the presence of a Brønsted acid, i.e., trifluoroacetic acid (1 equiv.), in toluene as the solvent, 3aa was formed in 61% yield, suggesting that HFIP played a crucial role as the Brønsted acid (entry 3, Table 1). Other Brønsted acids, such as acetic acid (1 equiv.) and triflic acid (1 equiv.), also in toluene, provided 3aa, but in lower yields, i.e., 22% and 13%, respectively (entries 4 and 5, Table 1). Notably, when HFIP (2 equiv.) was used as only a Brønsted acid but not as a solvent, the reaction was unsuccessful in toluene (entry 6, Table 1), resulting in the full recovery of the starting material (1a). When HFIP (2 equiv.) was employed only as a reagent in the presence of trifluoroethanol (TFE) as the solvent, the reaction did not proceed well, and the starting material was fully recovered, which revealed the indispensable role of HFIP as a solvent for this transformation. Thus, the addition of 2a (0.1 mmol) and 1a (0.1 mmol) in HFIP (0.6 M) at room temperature was found to be the optimized reaction conditions where HFIP played the dual roles of a Brønsted acid and the solvent.| Entry | Bronsted acid | Solvent (conc.) | Yieldb (%) |
|---|---|---|---|
| a Reactions are conducted on a 0.1 mmol scale. b The NMR yield is reported. c Starting materials are recovered. | |||
| 1 | HFIP | HFIP 0.6 M | 95% |
| 2 | TFE | TFE (0.6 M) | NDc |
| 3 | 1 equiv. TFA | Toluene (0.6 M) | 61% |
| 4 | 1 equiv. CH3COOH | Toluene (0.6 M) | 22% |
| 5 | 1 equiv. TfOH | Toluene (0.6 M) | 13% |
| 6 | 2 equiv. HFIP | Toluene (0.6 M) | NDc |
| 7 | 2 equiv. HFIP | TFE (0.6 M) | NDc |
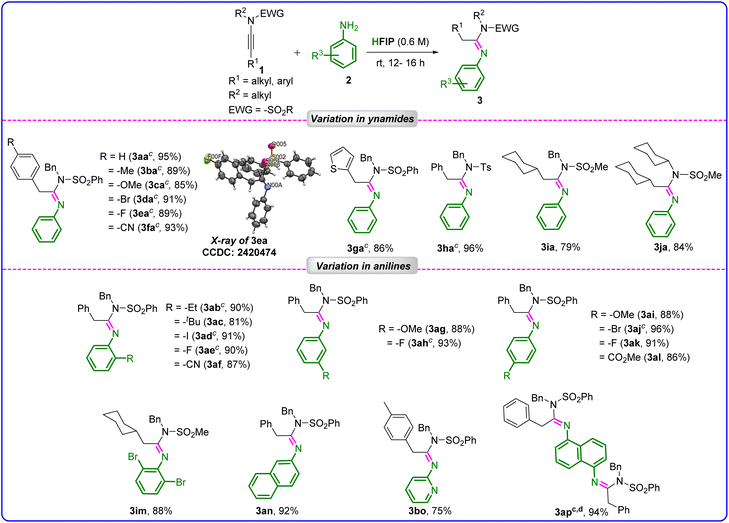
With the optimized conditions in hand, we explored the scope of the reaction (Table 2). The reaction of N-benzyl-N-(phenylethynyl)benzenesulfonamide (1a) with aniline (2a) on a 0.3 mmol scale produced 3aa without any loss in its yield (95%).
We then explored the scope of various ynamides for the hydroamination reaction with aniline. N-Benzyl-N-(arylethynyl)benzenesulfonamides bearing various substituents, such as 4-Me, 4-OMe, 4-Br, 4-F, and 4-CN, in the aryl ring were initially examined, and all reactions proceeded well, irrespective of the electronic nature of the substituents, producing the desired acetimidamides 3ba–3fa in good to excellent yields (85–93%, average yield ∼90%) and also in pure form without the requirement of any workup and column chromatography. The structure and the stereochemistry of 3ea were confirmed by X-ray crystal structure determination (Fig. S1 and Table S1, SI). It is noteworthy that a heteroaromatic (thiophene)-substituted ynamide participated in the reaction, delivering the corresponding product 3ga in 86% yield. Different EWG-substituted (–Ts, and –SO2Me) ynamides (1h, 1i) participated in the reaction, furnishing the desired products 3ha and 3ia in excellent yields of 96% and 79%, respectively. Aliphatic ynamide (1j) reacted with commercially available aniline (2a) to furnish 3aj in 84% yield. Notably, both electron-donating (–Et, –tBu, and –OMe) and electron-withdrawing (–I, –Br, –F, –CN, and –CO2Me), group-substituted anilines 2 smoothly participated in the reaction with 1a, and a series of desired N-arylimines (3ab–3al) was formed in excellent yield, ranging from 81% to 96%. Neither the electronic effect of the substituents nor their positional variations (ortho, meta, and para) impacted the outcome of the reaction. Sterically crowded, 2,6-dibromoaniline also participated in the hydroamination reaction with cyclohexyl-substituted ynamide (1i) without any difficulties and furnished the desired product, 3im, in 88% yield. Naphthalen-2-amine (2n) reacted smoothly with 1a to afford the desired product 3an in 92% yield. Furthermore, the extension of this hydroamination reaction to heteroaryl amines, such as pyridin-2-amine 2o (a poor nucleophile) with 1b, was successful, yielding the desired product 3bo with a 75% yield.
Interestingly, when naphthalene-1,5-diamine (2p) was reacted with 1a (2 equiv.), the bis-functionalization of 2p occurred with 1a, achieving the desired novel product 3ap in a very high yield of 94%. Significantly, most of the reactions were highly efficient and clean, leading to the formation of pure products in yields ranging from very high to excellent, which were then obtained in pure form through a simple washing of the crude product with a small amount (2 × 2 mL) of cold ethanol.
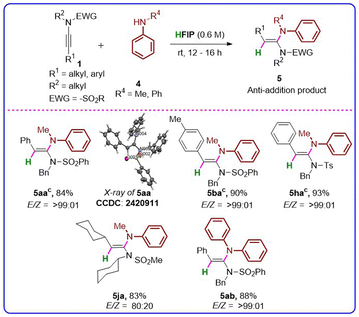
Subsequently, we examined the scope and limitations of the hydroamination of secondary amines with ynamides (Table 3). Interestingly, we observed that unlike primary amines, secondary amines underwent a highly regio-and stereoselective anti-hydroamination with ynamides under the same optimized conditions. Initially, N-methylaniline 4a was reacted with 1a under optimized conditions, resulting in the formation of (E)-N-benzyl-N-(1-(methyl(phenyl)amino)-2-phenylvinyl)benzenesulfonamide (5aa) with a yield of 84% and an excellent E/Z ratio of >99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 01 (exclusively E-stereoselectivity). The structure of 5aa was confirmed by X-ray crystal structure determination (Fig. S2 and Table S1, SI). Various substituted ynamides (both aryl and alkyl ynamides), i.e., 1b, 1h, and 1j, smoothly participated in the reaction with N-methyl aniline (4a), resulting in the desired products (5ba, 5ha, 5ja) with good to excellent yields ranging from 83% to 93% and good to excellent E-stereoselectivity (E/Z = 80
01 (exclusively E-stereoselectivity). The structure of 5aa was confirmed by X-ray crystal structure determination (Fig. S2 and Table S1, SI). Various substituted ynamides (both aryl and alkyl ynamides), i.e., 1b, 1h, and 1j, smoothly participated in the reaction with N-methyl aniline (4a), resulting in the desired products (5ba, 5ha, 5ja) with good to excellent yields ranging from 83% to 93% and good to excellent E-stereoselectivity (E/Z = 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 to >99
20 to >99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Diphenylamine (4b) also successfully participated in the reaction with 1a, producing the desired product (5ab) in 88% yield, and exclusive E-stereoselectivity (E/Z > 99
1). Diphenylamine (4b) also successfully participated in the reaction with 1a, producing the desired product (5ab) in 88% yield, and exclusive E-stereoselectivity (E/Z > 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
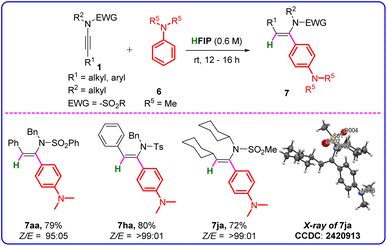
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 01). Notably, most of the reactions of ynamides with secondary amines were clean, and pure products were obtained by the simple cold ethanol washing of the crude product. In our efforts to broaden the substrate scope of the hydroamination of ynamides, we employed tertiary amines to react with ynamides under the optimized conditions, and the results are presented in Table 4. N,N-Dimethylaniline (6a) underwent syn-selective hydroarylation at its para position with ynamide 1a, leading to the formation of the target compound, (Z)-N-benzyl-N-(1-(4-(dimethylamino)phenyl)-2-phenylvinyl)benzenesulfonamide (7aa), with a 79% yield and excellent Z-stereoselectivity (Z/E = 95
01). Notably, most of the reactions of ynamides with secondary amines were clean, and pure products were obtained by the simple cold ethanol washing of the crude product. In our efforts to broaden the substrate scope of the hydroamination of ynamides, we employed tertiary amines to react with ynamides under the optimized conditions, and the results are presented in Table 4. N,N-Dimethylaniline (6a) underwent syn-selective hydroarylation at its para position with ynamide 1a, leading to the formation of the target compound, (Z)-N-benzyl-N-(1-(4-(dimethylamino)phenyl)-2-phenylvinyl)benzenesulfonamide (7aa), with a 79% yield and excellent Z-stereoselectivity (Z/E = 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 05). Both aromatic (1h) and aliphatic ynamides (1j) smoothly underwent the hydroarylation reaction with 6a to afford the desired products 7ha and 7ja in good yields (72–80%) and excellent stereoselectivity (Z/E > 99
05). Both aromatic (1h) and aliphatic ynamides (1j) smoothly underwent the hydroarylation reaction with 6a to afford the desired products 7ha and 7ja in good yields (72–80%) and excellent stereoselectivity (Z/E > 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
1).
The structure of 7aj was confirmed by X-ray crystal structure determination (Fig. S3 and Table S1, SI).
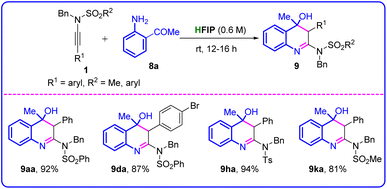
Under the optimized conditions, 2-aminocarbonyls 8 underwent cycloaddition reaction rather than a hydroamination reaction. We then examined the scope of ynamides 1 for the cycloaddition reactions (Table 5). Ynesulfonamide 1a smoothly underwent the cycloaddition reaction with 1-(2-aminophenyl)ethan-1-one (8a) to furnish the desired product N-benzyl-N-(4-hydroxy-4-methyl-3-phenyl-3,4-dihydroquinolin-2-yl)benzenesulfonamide 9aa in an excellent yield, 93%. The mild electron-withdrawing (–Br) group-substituted aryl-ynesulfonamides (1d) also reacted with 8a without any difficulties to furnish the desired product 9da in 87% yield. Upon substituting electron-withdrawing groups (EWG) such as –Ts and –Ms in ynesulfonamides, the reaction proceeded efficiently, yielding the desired products (9ha–9ka) with excellent yields ranging from 81% to 94%.
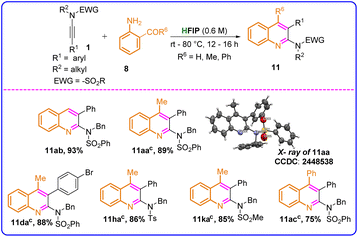
The synthesis of highly functionalized heterocycles in a single step is always challenging and has drawn significant interest from synthetic organic chemists. Ynamide chemistry is an excellent platform to construct different kinds of heterocycles in a single step.18 Among all heterocycles, 2-aminoquinolines represent a significant class of heterocycles, known for their valuable medicinal properties.19 The synthesis of 2-aminoquinolines in a single step through a sustainable approach is highly desirable. When 2-aminobenzaldehyde was reacted with ynamide 1 under the optimized conditions, we unexpectedly obtained an aromatized cyclic product, specifically, a substituted 2-aminoquinoline (Table 6). Phenyl-substituted ynesulfonamide, i.e., N-benzyl-N-((4-bromophenyl)ethynyl)benzenesulfonamide 1d, reacted with 1-(2-aminophenyl)ethan-1-one 8a to furnish N-benzyl-N-(4-methyl-3-phenylquinolin-2-yl)benzenesulfonamide 11da in 88% yield. Under optimized conditions, the reaction of 1-(2-aminophenyl)ethan-1-one (8a) with ynamide (1a) initially yielded a non-aromatized product. Upon heating the reaction mixture at 80 °C for 5 hours, the final desired product, N-benzyl-N-(4-methyl-3-phenylquinolin-2-yl)benzenesulfonamide (11aa), was obtained in an 89% yield. The structure of 11aa was confirmed by X-ray crystal structure determination (Fig. S4 and Table S1, SI). Upon substituting electron-withdrawing groups (EWG) such as –Ts and –Ms in ynesulfonamides, the reaction proceeded efficiently, affording the desired products (11ha–11ka) with excellent yields ranging from 85% to 86%. (2-Aminophenyl)(phenyl)methanone (8c) also underwent the reaction with ynamide (1a) and produced the desired product, N-benzyl-N-(3,4-diphenylquinolin-2-yl)benzenesulfonamide (11ac), in good yield (75%).
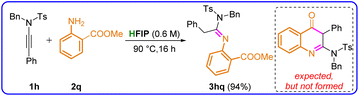
We then explored the feasibility of the cycloaddition reaction of ynamides 1 with ortho-aminobenzoic acid esters for the potential synthesis of 2-amidoquinolin-4(3H)-one. When ynesulfonamide 1h was treated with methyl 2-aminobenzoate (2q), the reaction did not proceed as anticipated, and we observed the formation of the hydroamination product only, 3hq, in 94% yield without the formation of the expected cycloaddition product, suggesting that HFIP could not effectively activate the ester carbonyl group, which is required for cyclization (Scheme 2).
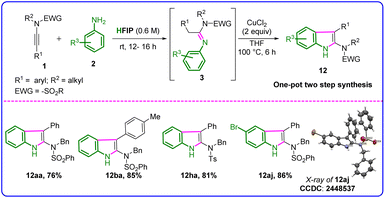
To demonstrate the potential application of our green synthetic protocol, we developed a one-pot, direct synthetic strategy to access highly substituted 2-amidoindoles, a highly potent and biologically active class of molecules, starting from easily accessible ynamides and anilines (Table 7). In the presence of HFIP, 1a and aniline furnished the desired product 3aa in almost quantitative yield. After the evaporation of HFIP, CuCl2 and THF were added to the same pot, and the reaction mixture was heated at 100 °C, which furnished the desired highly substituted 2-amidoindole, i.e., N-benzyl-N-(3-phenyl-1H-indol-2-yl)benzenesulfonamide 12aa in 76% yield. The one-pot direct synthesis of indole from ynamides and aniline was found to be efficient, and the electronic effect of ynamides and substituted anilines did not affect the outcome, resulting in various 2-amidoindoles 12aa–12aj in good to excellent yields (76–86%). The structure of 12aj was confirmed by X-ray crystal structure determination (Fig. S5 and Table S1, SI).
To demonstrate the practicality of our developed protocol, we conducted the model reaction between 1a and 2a on a gram scale (2 mmol scale), successfully obtaining the desired final product without sacrificing yield (Scheme 3A). Upon completion of the reaction, the solvent was recovered via distillation at 50 °C under 400 mbar pressure. The recovery process was efficient, yielding 2.8 mL of HFIP (82% recovery). To demonstrate the reusability of the recovered HFIP, we conducted the reaction between 1a and 2f with the recovered HFIP, and the desired product, (E)-N-benzyl-N′-(2-cyanophenyl)-2-phenyl-N-(phenylsulfonyl)acetimidamide (3af), was obtained with reproducible yields of 87% (Scheme 3B). We also assessed the recyclability of HFIP by conducting a reaction between 1a and 2a on a 3 mmol scale using 5 mL of HFIP (Scheme 3C). After the completion of the reaction, we could recover 4.25 mL of HFIP (85% recovery) and reused it for the second cycle reaction (2.5 mmol scale reaction), which afforded 3aa in 96% yield. After completing the second cycle, we recovered 3.44 mL of HFIP (81% recovery) and reused it for the third cycle reaction (2.06 mmol scale reaction), which afforded 3aa in 96% yield. After completing the third cycle, we recovered 2.82 mL of HFIP (82% recovery) and reused it for the fourth cycle reaction (1.69 mmol scale reaction), which afforded 3aa in 95% yield. After completing the fourth cycle, we recovered 2.42 mL of HFIP (86% recovery) and reused it for the fifth cycle reaction (1.45 mmol scale), which afforded 3aa in 93% yield. After completing the fifth cycle, we recovered 2 mL of HFIP (85% recovery).
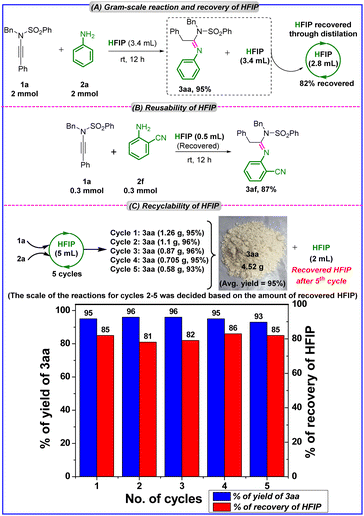 | ||
| Scheme 3 (A) Scale-up batch reaction and recovery of HFIP. (B) Reusability of HFIP. (C) Recyclability of HFIP. | ||
Notably, the recyclability of HFIP was found to be effective and efficient, as 4.52 g of 3aa was synthesized in a total of five cycles with a reproducible yield of 3aa in each cycle (average yield = 95%). These outcomes demonstrate the high sustainability, circular economy, practicality and cost-effectiveness of our protocol.
Based on previous literature reports on ynamide chemistry,4b,d the plausible mechanisms for this HFIP-mediated hydroamination and cycloaddition reaction are shown in Scheme 4. The H-bond network in HFIP, by increasing its acidity (pKa = 9), enables the activation of an ynamide via regioselective protonation at its β-carbon, generating the reactive keteniminium ion intermediate A. For the HFIP-mediated hydroamination of ynamides with primary aromatic amines (path A), the in situ-generated A was subsequently trapped by the nitrogen atom of aromatic amines to afford B. Further deprotonation of B yielded C, which ultimately tautomerized to furnish the final product 3. Similarly, for the hydroamination of ynamides with secondary aromatic amines, the keteniminium ion A was trapped by the nitrogen atom of the secondary aromatic amine 4 from the less hindered side of A to give D (path B). Subsequent deprotonation of species D produced Z-5, which is thermodynamically disfavored in most cases, and under the reaction conditions, it was easily converted to the thermodynamically stable product E-5via the intermediate F and G. Likewise, in the HFIP-mediated hydroarylation of ynamides with tertiary aromatic amines (path c), the resulting keteniminium ion A was trapped by the regioselective para-attack of the aromatic amines to yield intermediate H, followed by deprotonation to afford Z-7. Analogously, during the HFIP-mediated cycloaddition of ynamides with 2-amino aryl carbonyls (path d), the keteniminium ion is trapped by the amino nitrogen in 2-aminoaryl carbonyl 8 to produce I, which is then deprotonated to give J. At the same time, the carbonyl group is activated by HFIP, increasing the electrophilicity of carbon. Due to tautomerism, the electron density on the β-carbon increases, which favours intramolecular cyclization, yielding the cyclized product 9, which is subsequently dehydrated to give 10.
To have a better understanding of the overall sustainability of our developed protocol with respect to the previously reported ones, we enlisted a detailed comparison of various parameters in Table 8, such as the requirement of the starting material being in excess, catalyst, and/or reagents, solvent, reaction temperature, scope of amines, synthesis of products, yield, E-factor, feasibility of the recovery of catalyst or reagent, cost of the catalyst or reagent (cost-effectiveness), and circular economy. This analysis revealed that our protocol is simpler, and more general, cost-effective, practical, sustainable, and environmentally benign, with a broader substrate scope, and has a significantly higher circular economy compared to previously reported protocols. Moreover, the stereoselectivity of this protocol was found to be superior to that of a few previously reported ones.
| Research work | Starting material (required in excess) | Catalyst or reagent | Solvent | Temp. (°C) | Scope of amines | Synthesis of products | Yield (avg. Yield) | E-factor (g waste/g product)a,b | Catalyst or reagent recovery | Circular economy | Cost of the catalyst or reagent (1 g) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| a For the E-factor calculation, the reaction which furnished the highest yield of product was considered. b solvent was considered for the E-factor calculation. | |||||||||||
| Ref. 7 (Skrydstrup) | — | (PPh3)AuNTf2 | DCM | rt | Only 1°-anilines | Acetimidamides | 89–98% (94%) | 5.69 | Difficult | Low | $ 284.2 |
| Ref. 8 (Cai) | Aniline (1.1 eq.) | Ph2P-MCM-41-AuCl, AgNTf2 | DCM | rt | Only 1°-anilines | Acetimidamides | 87–97% (92%) | 3.56 | Effective | High | $ 340.7 |
| Ref. 9 (Wei) | Ynamide (1.5 eq.) | Zn(OTf)2 | Toluene | 80 | Only 1°-anilines | Acetimidamides | 50–93% (68%) | 25.1 | Difficult | Low | $ 9.3 |
| Ref. 10 (Wu) | Aniline (2 eq.) | Yb(OTf)3 | DCE | 85 | Only 1°-anilines | Acetimidamides | 39–89% (75%) | 21.3 | Difficult | Low | $ 27.1 |
| Ref. 11 (Chang) | Aniline (1.2 eq.) | TfOH | DCE | rt | Only 1°- and 2°-anilines | Acetimidamides and 2ketene N,N-acetals | 65–99% (88%) | 6.56 | Difficult | Low | $ 19.4 |
| Ref. 12 (Lin) | Ynamide (1.5 eq.) | Ni(OTf)2 | Toluene | 80 | Only 2°-amines | Ketene N,N-acetals | 54–92% (72%) | 10.7 | Difficult | Low | $ 24.8 |
| Ref. 13 (Lin) | Ynamide (2.0 eq.) | AgNTf2 | CHCl3 | 60–70 | Only 3°-anilines | α-Aryl enamides | 52–90% (72%) | 19.9 | Difficult | Low | $ 147.7 |
| Ref. 14 (Dubovtsev) | — | PPh3AuCl/AgNTf2 (extra 10 mol% MsOH required for ketones) | DCE | 60 | Only ortho carbonyl anilines | Quinolines | 54–98% (77%) | 13.1 | Difficult | Low | PPh3AuCl ($ 316) AgNTf2 ($ 148) |
| Ref. 18a (Youn) | Ynamide (1.2 eq.) | ZnBr2 | Toluene | 140 | Only ortho carbonyl anilines and phenols | Quinolines and coumarins | 84–95% (89%) | 24.1 | Difficult | Low | $ 9.5 |
| This work | — | HFIP | HFIP | rt | 1°, 2°, 3°-Anilines, 1°-hetero aromatic amines, ortho amino benzaldehydes, and ketones | Acetimidamides, ketene N,N-acetals, α-aryl enamides, quinolines,and 4-hydroxy-3,4-dihydroquinolines | 72%-96% (89%) | 0.98–1.27 | Easy and effective | High | $ 0.5 |
Conclusions
The atom-economic, highly regio- and stereo-selective hydroamination of ynamides with primary, secondary, and tertiary aromatic and heteroaromatic amines has been successfully achieved under mild and sustainable reaction conditions. This sustainable protocol yields a wide variety of valuable products, including acetimidamides, ketene N,N-acetals, and α-arylenamides, as well as various heterocycles such as quinolines and 3,4-dihydroquinolin-4-ols, starting from readily available ynamides involving all kinds of aromatic amines (1°, 2°, and 3°). This protocol is workup-free, and most of the pure final products were obtained through a simple organic washing using a small amount of ethanol. The scalability of the protocol is straightforward up to a gram-scale level without compromising reaction yields. The only species used in the reaction, except for the starting materials, was HFIP, which played a crucial dual role as both the activator of the ynamide and the stabiliser of the in situ-generated keteniminium ion, and was regenerated after the reaction. HFIP was recovered effectively through simple distillation and reused for another reaction without losing its properties. HFIP was also recycled efficiently for five consecutive reactions between 1a and 2a, and thus, 4.52 g of 3aa was produced with a reproducible yield in each cycle (average yield = 95%), synthesised in five consecutive batches of reactions involving recycled HFIP. These results indicate that the protocol is highly sustainable in the context of green chemistry. We also described the synthesis of 2-aminoindoles from commercially available aromatic amines. We believe this Diversity-Oriented Synthesis (DOS) will be useful for crafting more powerful small molecules in a more sustainable and cost-effective manner, thereby paving the way for advancements in pharmaceutical development and materials chemistry.Author contributions
A. N. V. Satyanarayana carried out all the experiments, including optimization of the reaction conditions, synthesis of products, their synthetic diversifications, and mechanistic studies. T. Chatterjee conceived and supervised the work and wrote and edited the manuscript. All authors have given their approval to the manuscript's final version.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2420474, 2420911, 2420913, 2448538 and 2448537 contain the supplementary crystallographic data for this paper.20a–eThe data underlying this study are available in the published article, and its experimental and spectroscopic details are included as a part of the online supplementary information (SI). Supplementary information: experimental procedures, X-ray data, analytical data of the synthesized molecules, and 1H, 13C, NOE spectra of the synthesized compounds. See DOI: https://doi.org/10.1039/d5su00899a.
Acknowledgements
T. Chatterjee gratefully acknowledges the financial support from the Science and Engineering Research Board (SERB), Govt. of India [File no. CRG/2023/003045]. The NMR facility at the BITS-Pilani, Hyderabad campus, and the HRMS facility, funded by DST-FIST (Grant number: SR/FST/CS-I/2020/158), at BITS-Pilani, Hyderabad campus, are acknowledged. A. N. V. Satyanarayana thanks CSIR [File No. 09/1026(17969)/2024-EMR-I] for the senior research fellowship (SRF Direct).Notes and references
- (a) R. J. Spandl, M. Díaz-Gavilán, K. M. G. O'Connell, G. L. Thomas and D. R. Spring, Diversity-Oriented Synthesis, Chem. Rec., 2008, 8, 129–142 CrossRef CAS PubMed; (b) R. J. Spandl, A. Bender and D. R. Spring, Diversity-oriented synthesis, a spectrum of approaches and results, Org. Biomol. Chem., 2008, 6, 1149–1158 RSC; (c) C. J. O'Connor, H. S. G. Beckmann and D. R. Spring, Diversity-oriented synthesis: producing chemical tools for dissecting biology, Chem. Soc. Rev., 2012, 41, 4444–4456 RSC.
- (a) M. M. Hann, A. R. Leach and G. Harper, Molecular Complexity and Its Impact on the Probability of Finding Leads for Drug Discovery, J. Chem. Inf. Comput. Sci., 2001, 41, 856–864 CrossRef CAS PubMed; (b) A. Schuffenhauer, N. Brown, P. Selzer, P. Ertl and E. Jacoby, Relationships between Molecular Complexity, Biological Activity, and Structural Diversity, J. Chem. Inf. Model., 2006, 46, 525–535 CrossRef CAS PubMed.
- (a) K. A. DeKorver, H. Li, A. G. Lohse, R. Hayashi, Z. Lu, Y. Zhang and R. P. Hsung, Ynamides: A Modern Functional Group for the New Millennium, Chem. Rev., 2010, 110, 5064–5106 CrossRef CAS PubMed; (b) G. Evano, A. Coste and K. Jouvin, Ynamides: Versatile Tools in Organic Synthesis, Angew. Chem., Int. Ed., 2010, 49, 2840–2859 CrossRef CAS PubMed; (c) X.-N. Wang, H.-S. Yeom, L.-C. Fang, S. He, Z.-X. Ma, B. L. Kedrowski and R. P. Hsung, Ynamides in Ring Forming Transformations, Acc. Chem. Res., 2014, 47, 560–578 CrossRef CAS PubMed; (d) C. C. Lynch, A. Sripada and C. Wolf, Asymmetric synthesis with ynamides: unique reaction control, chemical diversity and applications, Chem. Soc. Rev., 2020, 49, 8543–8583 RSC.
- (a) A. Mishra, S. S. Pati and J. P. Das, Synthesis of Enamides through Hydro-Functionalization of Ynamides, Eur. J. Org Chem., 2024, 27, e202400637 CrossRef CAS; (b) A. N. V. Satyanarayana and T. Chatterjee, Solvent-Mediated C- or N-Center Selective Syn-Hydroheteroarylation of Ynamides with N-Heteroaromatics: Access to a Solvatofluorochromic, Mechanofluorochromic, and Thermochromic Material, J. Org. Chem., 2025, 90, 14892–14906 CrossRef CAS PubMed; (c) A. N. V. Satyanarayana and T. Chatterjee, HFIP-Mediated, Highly Chemo-, Regio-, and Stereoselective Hydrofunctionalizations of Ynamides: Access to Stereodefined Alkenes Bearing Drugs and Natural Products, J. Org. Chem., 2024, 89, 12439–12451 CrossRef CAS PubMed; (d) A. N. V. Satyanarayana, P. Pattanayak and T. Chatterjee, J. Org. Chem., 2024, 89, 11455–11466 CrossRef CAS PubMed; (e) A. N. V. Satyanarayana, P. Pattanayak and T. Chatterjee, HFIP-mediated, regio- and stereoselective hydrosulfenylation of ynamides: a versatile strategy for accessing ketene N,S-acetals, Org. Biomol. Chem., 2025, 23, 2235–2243 RSC; (f) S. Dutta, R. K. Mallick and A. K. Sahoo, Regioselective Difunctionalization and Annulation of Ynamide, Angew. Chem., Int. Ed., 2023, 62, e202300816 CrossRef CAS PubMed.
- (a) A. Maity and A. K. Sahoo, Copper-Catalyzed Regio- and Stereoselective Hydroarylation of Ynamide, J. Org. Chem., 2024, 89, 852–863 CrossRef CAS PubMed; (b) M. Sethi, S. Verma, V. Gandon and A. K. Sahoo, ACS Catal., 2025, 15, 15606–15616 CrossRef CAS.
- (a) T. Courant, G. Dagousset and G. Masson, Enamide Derivatives: Versatile Building Blocks for Total Synthesis, Synthesis, 2015, 47, 1799–1856 CrossRef CAS; (b) T. Kuranaga, Y. Sesoko and M. Inoue, Cu-mediated enamide formation in the total synthesis of complex peptide natural products, Nat. Prod. Rep., 2014, 31, 514–532 RSC; (c) L. Yet, Chemistry and Biology of Salicylihalamide A and Related Compounds, Chem. Rev., 2003, 103, 4283–4306 CrossRef CAS PubMed; (d) K. Okamoto, M. Sakagami, F. Feng, H. Togame, H. Takemoto, S. Ichikawa and A. Matsuda, Total Synthesis of Pacidamycin D by Cu(I)-Catalyzed Oxy Enamide Formation, Org. Lett., 2011, 13, 5240–5243 CrossRef CAS PubMed; (e) M. V. J. Villa, S. M. Targett, J. C. Barnes, W. G. Whittingham and R. Marquez, An Efficient Approach to the Stereocontrolled Synthesis of Enamides, Org. Lett., 2007, 9, 1631–1633 CrossRef CAS PubMed; (f) X. Wang and J. A. Porco, Total Synthesis of the Salicylate Enamide Macrolide Oximidine, J. Am. Chem. Soc., 2003, 125, 6040–6041 CrossRef CAS PubMed.
- S. Kramer, K. Dooleweerdt, A. T. Lindhardt, M. Rottlander and T. Skrydstrup, Highly Regioselective Au(I)-Catalyzed Hydroamination of Ynamides and Propiolic Acid Derivatives with Anilines, Org. Lett., 2009, 11, 4208–4211 CrossRef CAS PubMed.
- D. Liu, Q. Nie, R. Zhang and M. Cai, Regiospecific Hydroamination of Unsymmetrical Electron-Rich and Electron-Poor Alkynes with Anilines Catalyzed by Gold(I) Immobilized in MCM-41, Adv. Synth. Catal., 2018, 360, 3940–3948 CrossRef CAS.
- Z. Chen, X.-D. Nie, J.-T. Sun, A.-M. Yang and B.-G. Wei, Zn(OTf)2-catalyzed hydroamination of ynamides with aromatic amines, Org. Biomol. Chem., 2021, 19, 2492–2501 RSC.
- X. Zeng, Q. Gu, W. Dai, Y. Xie, X. Liu and G. Wu, Yb (OTf) 3-mediated regioselective hydroamination of ynamides with anilines and p-toluenesulfonamide, Synthesis, 2021, 53, 2889–2896 CrossRef CAS.
- Y. Wang, Z. Zhao, S. Lv, L. Ding, Xi. N. Wang and J. Chang, Nonmetal-catalyzed hydroamination of ynamides with amines, Org. Chem. Front., 2021, 8, 6244–6251 RSC.
- X. D. Nie, X. L. Han, J. T. Sun, C. M. Si, B. G. Wei and G. Q. Lin, Nickel-catalyzed regioselective hydroamination of ynamides with secondary amines, J. Org. Chem., 2021, 86, 3433–3443 Search PubMed.
- X. D. Nie, Z. Y. Mao, J. M. Guo, C. M. Si, B. G. Wei and G. Q. Lin, AgNTf2-Catalyzed Regioselective C–H Alkenylation of N,N-Dialkylanilines with Ynamides, J. Org. Chem., 2022, 87, 2380–2392 Search PubMed.
- N. V. Shcherbakov, P. F. Kotikova, E. I. Chikunova, D. V. Dar’in, V. Y. Kukushkin and A. Y. Dubovtsev, Gold-Catalyzed Annulation of Ynamides with Aminocarbonyls as a Route to 2-Aminoquinolines Diversely Substituted at the 4th-Position, Adv. Synth. Catal., 2023, 365, 2428–2434 Search PubMed.
- Y. H. Kim, H. J. Yoo and S. W. Youn, Facile one-pot synthesis of 2-aminoindoles from simple anilines and ynamides, Chem. Commun., 2020, 56, 13963–13966 RSC.
- K. Dooleweerdt, T. Ruhland and T. Skrydstrup, Application of Ynamides in the Synthesis of 2-Amidoindoles, Org. Lett., 2009, 11, 221–224 CrossRef CAS PubMed.
- (a) P. Maciej, M. Joseph and L. David, Difunctionalization Processes Enabled by Hexafluoroisopropanol, ACS Org. Inorg. Au., 2024, 4, 287–300 CrossRef PubMed; (b) F. M. Hashim, M. A. Ahlam, G. C. Jackson, C. C. Thomas, R. K. K. Kimberly, M. N. Verrill, A. Jeffrey and A. HFIP, Organic Synthesis, Chem. Rev., 2022, 122, 12544–12747 CrossRef PubMed; (c) L. David, The rise and future of hexafluoroisopropanol as a solvent, Trends Chem., 2025, 7, 205–207 CrossRef; (d) H. Song, H. Zhou, Y. Shen, H. Wang, H. Song, X. Cai and C. Xu, HFIP as Protonation Reagent and Solvent for Regioselective Alkylation of Indoles with All-Carbon Centers, J. Org. Chem., 2022, 87, 1086–1097 Search PubMed; (e) S. Ghosh, Z.-W. Qu, S. Pradhan, A. Ghosh, S. Grimme and I. Chatterjee, HFIP-Assisted Single C-F Bond Activation of TrifluoromethylKetones using Visible-Light Photoredox Catalysis, Angew. Chem., Int. Ed., 2022, 61, e202115272 Search PubMed; (f) S. Pradhan, S. Das, G. Kumar and I. Chatterjee, Transition-Metal-Free Regioselective Intermolecular Hydroamination of Conjugated 1,3-Dienes with Heterocyclic Amines, Org. Lett., 2022, 24, 2452–2456 CrossRef CAS PubMed; (g) S. Pradhan, V. Sharma and I. Chatterjee, Nitrosoarene-Catalyzed HFIP-Assisted Transformation of Arylmethyl Halides to Aromatic Carbonyls under Aerobic Conditions, Org. Lett., 2021, 23, 6148–6152 CrossRef CAS PubMed.
- (a) H. J. Yoo and S. W. Youn, Zn(II)-catalyzed one-pot synthesis of coumarins from ynamides and salicylaldehydes, Org. Lett., 2019, 21, 3422–3426 CrossRef CAS PubMed; (b) S. Choudhary, S. Gayyur, A. Manhas, R. Kant and N. Ghosh, Copper-Catalyzed and 1,3-Sulfonyl Migration Enabled Installation of Azaindoles in the Periphery of Aryl Rings: Synthesis of Sulfonylated Pyrrolo[2,3-b]quinolines and Investigation of Antimalarial Potency, J. Org. Chem., 2025, 90, 6169–6174 CrossRef CAS PubMed.
- (a) R. S. Keri and S. A. Patil, Quinoline: a promising antitubercular target, Biomed. Pharmacother., 2014, 68, 1161–1175 CrossRef CAS PubMed; (b) O. Afzal, S. Kumar, M. R. Haider, M. R. Ali, R. Kumar, M. Jaggi and S. Bawa, A review on anticancer potential of bioactive heterocycle quinoline, Eur. J. Med. Chem., 2015, 97, 871–910 CrossRef CAS PubMed; (c) A. A. Alhaider, M. A. Abdelkader and E. J. Lien, Design, synthesis and pharmacological activities of 2-substituted 4-phenylquinolines as potential antidepressant drugs, J. Med. Chem., 1985, 28, 1394–1398 CrossRef CAS PubMed; (d) S. F. Campbell, J. D. Hardstone and M. J. Palmer, 2, 4-Diamino-6, 7-dimethoxyquinoline derivatives as. alpha. 1-adrenoceptor antagonists and antihypertensive agents, J. Med. Chem., 1988, 31, 1031–1035 CrossRef CAS PubMed.
- (a) CCDC 2420474: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2m7ptd; (b) CCDC 2420911: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2m84xz; (c) CCDC 2420913: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2m84z1; (d) CCDC 2448538: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n5x3w; (e) CCDC 2448537: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n5x2v.
| This journal is © The Royal Society of Chemistry 2026 |