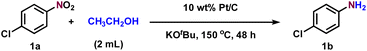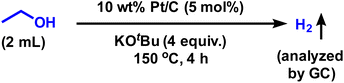Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Ethanol as a H2 source: transfer hydrogenation of sulfur and halogen containing nitroarenes with an anti-poisoning platinum on carbon catalyst
Chitrarekha
Dewangan
a,
Reeshma
Rameshan
b,
Suresh
Perumal
b,
Narayana V.
Kalevaru
 c,
Sebastian
Wohlrab
c,
Sebastian
Wohlrab
 *c,
Rajenahally V.
Jagadeesh
*c,
Rajenahally V.
Jagadeesh
 *c and
Kishore
Natte
*c and
Kishore
Natte
 *a
*a
aDepartment of Chemistry, Indian Institute of Technology Hyderabad, Kandi, Sangareddy, Telangana 502285, India. E-mail: kishore.natte@chy.iith.ac.in
bLaboratory for Energy and Advanced Devices (LEAD), Department of Materials Science and Metallurgical Engineering, Indian Institute of Technology Hyderabad (IITH), Kandi, Sangareddy, 502284, Telangana, India
cLeibniz-Institut für Katalyse e.V., Albert-Einstein-Str. 29A, Rostock 18059, Germany. E-mail: sebastian.wohlrab@catalysis.de; jagadeesh.rajenahally@catalysis.de
First published on 12th January 2026
Abstract
Developing environmentally friendly transfer hydrogenation protocols using sustainable and economically viable hydrogen donors is highly desirable in the organic synthesis toolbox. In this respect, the high hydrogen content (13.1 wt%) and its low toxicity, including its production from renewable substances, make ethanol a promising hydrogen source. Herein, we report an attractive, recyclable, and anti-poisoning platinum on carbon (Pt/C) catalyst for the chemoselective reduction of functionalized nitroarenes comprising reactive functional groups, including multiple sulfur functionalities (–SO2Me, –SO2, –SO3H, –SMe, etc.), halogens (–F, –Cl, –Br, and –I), carbonyl, amide, and acid groups, selectively delivering structurally diverse aniline products in high yields (up to 95%). Therefore, the current approach may open new possibilities for syntheses, especially with respect to chemical building blocks for pharmaceutical synthesis. The use of viable ethanol as a hydrogen source also makes this catalytic process sustainable.
Sustainability spotlightEthanol represents a highly sustainable hydrogen donor in chemical synthesis due to its unique combination of environmental, economic, and practical advantages. As a bio-derived compound, ethanol can be readily produced from renewable biomass sources, minimizing dependence on fossil fuels and reducing greenhouse gas emissions throughout its lifecycle. In this regard, our work exemplifies a meaningful stride toward aniline synthesis from nitroarenes by advancing a transfer hydrogenation protocol that leverages ethanol. Ethanol's bio-derived origins and favorable hydrogen density (13.1 wt%) make it an economically viable and environmentally benign choice. The development of a recyclable, anti-poisoning Pt/C catalyst further reinforces the sustainability of this approach by minimizing waste and extending catalyst lifespan. |
Introduction
Among the numerous renewable raw materials, ethanol (EtOH) is an important C2 raw material that can be regarded as a liquid organic chemical hydrogen carrier, the production of which is useful in chemicals and fuel technologies.1 EtOH is mainly produced by yeast fermentation from starch-rich crops such as corn, wheat, and cassava.2 The annual production of renewable EtOH is constantly increasing, and it reached around 31.1 billion gallons in 2024, making it the largest volume of small renewable feedstock molecules.3 Currently, the majority of bioethanol is used as a transportation fuel.4 Additionally, EtOH is also used in the clinical sector for disinfection and sterilization, including alcoholic beverages.5 Furthermore, EtOH can also be used to produce a few key chemicals, such as ethylene, acetic acid, acetic anhydride, and ethyl acetate.6 However, implementing ethanol as a renewable C2 feedstock to access a variety and large number of commodity chemicals,7 including catalytic hydrogen transfer reactions,8 is less developed and merits greater attention in organic synthesis.Catalytic transfer hydrogenation (CTH) with an organic hydrogen donor has emerged as a synthetically powerful and practical tool and has attracted significant attention from both academia and industry as a complementary technology over the classical direct hydrogenation process.9 Also, the CTH strategy offers a number of advantages, such as (a) easily available and cost-effective hydrogen sources, (b) enhanced safety measures, (c) no demand for a high-pressure experimental set-up, (d) no external H2 supply, and (e) operational simplicity. Alcohols are potential sources of hydrogen surrogates.10 Specifically, alcohols like methanol, ethanol, isopropanol, and polyols (glycerol and sugars) are notable examples of organic hydrogen donors in CTH chemistry.10 After the vast use of isopropanol (IPA) as a hydrogen source in chemical reactions,11 methanol has become an excellent substitute for IPA owing to its production from renewable resources or natural gas.12 Despite the potential use of methanol in CTH reactions, it also has a few drawbacks like the generation of formaldehyde, which can be converted to toxic CO gas that sometimes hampers the activity and selectivity of the reaction or may also poison the catalyst (depending on the type of catalyst and the reaction conditions applied).12a,f In comparison to both IPA and methanol, ethanol is less studied in transfer hydrogenation reactions.8,13 The high hydrogen content (13.1 wt%) and the low toxicity make ethanol a promising hydrogen source.14 Furthermore, the dehydrogenation of ethanol produces exclusively hydrogen, which is consumed during the reaction, and acetaldehyde, which is a relatively environmentally friendly side product.15 Ever since the pioneering work of Grützmacher in 2008 on the transfer hydrogenation of ketones with ethanol as the hydrogen donor, ethanol has emerged as a particularly attractive hydrogen source and has increasingly come into focus in modern synthetic methodology.13b
Aniline and its derivatives are found in many pharmaceuticals, natural products, agrochemicals, polymers, and more (Fig. 1A).16 Furthermore, sulfur and halogen-containing scaffolds are widely present in many drug components owing to their unique physicochemical and pharmacokinetic properties.17 Anilines comprising well-known functional groups such as thiol, thioether, sulfone, sulfonamide, and sulfide attract attention in drug discovery.18 Although there are a plethora of methodologies available for the synthesis of structurally diverse anilines, the preparation of sulfur and halogen-containing anilines is still a formidable challenge. Moreover, catalytic approaches are very limited,18,19 limiting the chemical space in aniline chemistry and its applications in drug discovery. Therefore, a general and efficient protocol to prepare these sulfur and halogen-containing anilines will facilitate understanding and application in pharmaceutical chemistry. On the other hand, it is worth noting that sulfur-resistant (transfer) hydrogenation catalysts are much fewer in number and are typically applied in petrochemical industries, particularly for the hydrodesulfurization (HDS) of naphtha feedstocks.20 In this context, very recently, Mitsudome and co-workers developed a ruthenium phosphide (Ru2P/SiO2) catalyst18a and ruthenium phosphide nanoparticles supported on activated carbon (Ru2P/C)21 for the hydrogenation and reductive amination of challenging sulfur-containing molecules. In another study, Wang and Zhang together reported that triphenylphosphine counteracts S-poisoning during Pd/C catalyzed 4-nitroanisole reduction.19 The Glorius group also reported a heterogeneous Ru–S catalyst for the selective hydrogenation of sulfur-containing heterocycles.17d
 | ||
| Fig. 1 (A) Phenylamine-containing pharmaceuticals. (B) General overview of bioethanol production and its utilization as a hydrogen source in transfer hydrogenation reactions. | ||
Research on transfer hydrogenation is still yearning for a robust protocol for sulfur and halogen-containing anilines with wide substrate compatibility. Based on our previous reports and interest in exploiting hydrogen transfer chemistry,12a,f,g,18b we have focused on ethanol utilization as a hydrogen source for the synthesis of sulfur and halogen-containing anilines in the presence of commercially available Pt/C catalysts. Pt/C-catalyzed reactions have been of great interest over the past due to their particular advantages, such as high catalytic efficiency and easy recyclability.12g,22 A ligand-free synthetic system is always considered clean and environmentally friendly in modern organic synthesis. This manuscript reports the straightforward synthesis of sulfur-, halogen-, and other functional groups containing anilines, through catalytic transfer hydrogenation using ethanol as a hydrogen donor under an anti-poisoning Pt/C catalyst (Fig. 1B).
Results and discussion
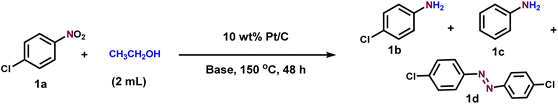
Initially, the transfer hydrogenation of 4-chloronitrobenzene (1a) with ethanol as both a solvent and hydrogen source was investigated by exploring different bases and catalytic systems at 150 °C for 48 h (Table 1). First, various bases were surveyed, and they played a very important role in this reaction (entries 1–7). Bases like KOH, K2CO3, NaOH, NaOMe, TEA (triethylamine), DIPEA (diisopropylamine), and Cs2CO3 did not show much influence on the reaction and gave the desired product 1b in moderate yields and 1c in very low yields. Remarkably, KOtBu was found to be very effective and afforded 1b in 95% isolated yield and 5% of 1c (entry 8). In fact, KOtBu was also successful in related transfer hydrogenation reactions.12f,g Since KOtBu is a powerful and non-nucleophilic base, it can easily abstract a proton from alcohols.12f,g,15b Control experiments revealed that the presence of Pt/C and KOtBu is very crucial in the transformation (entries 9–11). Other commercial catalysts like Pd/C, Ru/C, and Rh/C did not show improved results (entries 12–14). We also conducted the reaction of 1a with ethanol at various temperatures (100–150 °C) and noticed that our targeted product 1b gradually increased with increasing temperature (Fig. 2A). An elevated temperature of 150 °C is required to promote ethanol dehydrogenation. At lower temperatures, ethanol dehydrogenation is slow, limiting metal-hydride formation and hydrogen availability at the catalyst surface. Similarly, the reaction was also conducted at different time intervals (12–48 h), and an increase in 1b yield was observed at 48 h (Fig. 2B). These results clearly suggest that an elevated temperature (150 °C) and higher reaction time (48 h) are necessary to achieve the full conversion and selectivity. Additionally, under optimized reaction conditions, we also conducted the transfer hydrogenation reaction with other alcohols such as isopropanol, butanol, and pentanol. However, only a satisfactory yield of 1b was achieved (see SI Table T1).| Entry | Catalyst | Base (4 equiv.) | Selectivity 1b (%) | Yield 1b (%) | Yield 1c (%) | Yield 1d (%) |
|---|---|---|---|---|---|---|
| a General reaction conditions: 1a (0.5 mmol), ethanol (2 mL), 10 wt% Pt/C (25 mg), base (4 equiv.), 150 °C, 48 h. | ||||||
| 1 | 10 wt% Pt/C | KOH | 64 | 28 | 4 | 12 |
| 2 | 10 wt% Pt/C | K2CO3 | 59 | 10 | 2 | 5 |
| 3 | 10 wt% Pt/C | NaOH | 61 | 19 | 5 | 7 |
| 4 | 10 wt% Pt/C | NaOMe | 82 | 28 | 2 | 4 |
| 5 | 10 wt% Pt/C | TEA | 100 | 5 | 0 | 0 |
| 6 | 10 wt% Pt/C | DIPEA | 53 | 8 | 2 | 5 |
| 7 | 10 wt% Pt/C | Cs2CO3 | 66 | 55 | 12 | 16 |
| 8 | 10 wt% Pt/C | KOtBu | 95 | 95 | 5 | 0 |
| 9 | 10 wt% Pt/C | — | — | n.d. | n.d. | n.d. |
| 10 | — | KOtBu | — | n.d. | n.d. | n.d. |
| 11 | — | — | — | n.d. | n.d. | n.d. |
| 12 | 5 wt% Pd/C | KOtBu | 91 | 78 | 6 | 2 |
| 13 | 5 wt% Ru/C | KOtBu | 75 | 48 | 14 | 2 |
| 14 | 5 wt% Rh/C | KOtBu | 89 | 72 | 9 | 0 |
To determine the electronic states of Pt and C in Pt/C XPS spectra of C 1s and Pt 4f were recorded and are presented in Fig. S1 (see SI Fig. S1). The C correction was done for the XPS spectra with respect to the C 1s spectra at 284.8 eV. From Fig. S1a, the C 1s spectra show peaks at 284.3 eV, 285.6 eV, and 287.1 eV, which correspond to the sp2, sp3 hybridized C, and C–O, respectively. The Pt 4f spectra show a 4f doublet, consisting of the Pt 4f5/2 and Pt 4f7/2 peaks, which were well-separated by ∼3.3 eV. The deconvolution of the Pt 4f shows doublet peaks at 70.9 eV and 74.1 eV corresponding to Pt0 and 72.1 eV and 75.4 eV corresponding to Pt2+ (see Fig. S1b). The quantitative deconvolution of the Pt 4f XPS spectra demonstrates that the surface Pt species in the catalyst consist of approximately 52.2% metallic Pt0 and 47.8% Pt2+. Fig. 2C illustrates the transmission electron micrograph of the powder of the Pt/C catalyst used in this study (product no. GRM4777-1G). From the micrograph (see Fig. 2C), it is clear that fine nano-sized Pt particles are embedded in the matrix of carbon (C), which are highlighted with yellow circles. Fig. 2D depicts the particle size distribution of Pt calculated from Fig. 2C, which shows that the particle size of Pt in the C-matrix varies from 2 nm to 4 nm, and the maximum particles were in the average size of ∼2.34 nm. The XRD pattern of the Pt/C catalyst is shown in Fig. S2 (see SI Fig. S2). The broad diffraction peak centered at ∼24.3° corresponds to the (002) plane of amorphous carbon, indicating a disordered carbon support. The 2θ reflections at ∼39.8°, 46.2°, 67.5°, and 81.3° are indexed to the (111), (200), (220), and (311) planes of face-centered cubic metallic Pt, respectively (JCPDS no. 04-0802). The broad nature of the Pt peaks suggests that nanosized Pt crystallites with high dispersion are present on the carbon support. The optimized conditions were applied next to the chemoselective transfer hydrogenation of a structurally diverse array of nitroaromatics with ethanol, and the results are summarized in Scheme 1. As described in the Introduction, sulfur-resistant hydrogenation catalysts for the reduction of sulfur-containing nitroarenes to their respective anilines are scarcely reported in the literature. In this regard, for the first time, we have applied commercially available Pt/C for the reduction of a variety of sulfur-containing nitroarenes. Interestingly, a wide range of sulfur-based functional groups were well tolerated in this reaction. For example, thiomethyl (1), diphenyl sulfide derivatives (2–4), S-heterocycles (5–6), organic sulfide (7), sulfonamide (8), sulfonic acid and mesylate substituents (9–12), and diphenyl sulfone (13–14) were all amenable to this protocol and afforded S-containing anilines in good to excellent isolated yields. The functional group tolerance of carbon–halogen bonds of nitroarenes offers exciting opportunities for the synthesis of small-molecule drugs, agrochemicals, and some new materials. In line with this response, various halogenated substrates were highly tolerated and allowed for smooth transfer hydrogenation, affording desired halo-anilines in excellent isolated yields (15–28). Remarkably, no reductive dehalogenation of the C–X bond or byproduct took place. The developed catalytic system is able to chemoselectively reduce keto (29), acid (30), amide (31–32), hydroxyl (33), methoxy (34), –CH2OH (35), dioxolane (36), pyrene (37), and methyl substituents (38–41) including simple nitrobenzene (42) were successfully reduced to the corresponding anilines without any byproducts. Fascinatingly, dinitroarenes were also surveyed and can be conveniently applied to give the corresponding dianilines in acceptably good yields (43–44). Furthermore, this transfer hydrogenation protocol could be readily applied to heterocyclic nitroarenes, allowing heterocyclic arylamines to be obtained in very good yield (45–48). Despite varying the reaction conditions, flutamide was found to be non-compatible.
We also applied our Pt/C catalytic system for the gram-scale synthesis of 4-chloroaniline and furnished the corresponding product in 89% isolated yield, thereby enhancing the practical utility of our protocol (Scheme 2).
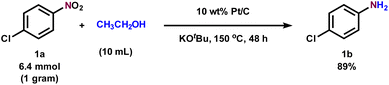 | ||
| Scheme 2 Gram-scale synthesis of 4-chloroaniline (for reaction conditions, see the reaction procedure). | ||
To check the reusability of the Pt/C catalyst, the reaction of 4-chloronitrobenzene (1a) with ethanol was demonstrated under the optimized reaction conditions (Table 2). In the first cycle, which afforded 4-chloroaniline (1b) in 90% yield, the Pt/C was readily recovered by the filtration process. Next, the recovered catalyst was reused in the subsequent four reaction cycles, and a decline in yield was observed. The TEM image of cycle 4 reveals the formation of agglomerates, which could be the more probable reason for the low activity of Pt/C (Fig. 3).
| Cycle | Cycle 0 | Cycle 1 | Cycle 2 | Cycle 3 | Cycle 4 |
|---|---|---|---|---|---|
| a Reaction conditions: 1a (0.5 mmol), ethanol (2 mL), 10 wt% Pt/C (25 mg), KOtBu (4 equiv.), 150 °C, 48 h. | |||||
| Yield (%) | 95 | 90 | 85 | 73 | 60 |
To understand the reaction mechanism of this transfer hydrogenation reaction, ethanol degradation experiments were performed. Ethanol was reacted with Pt/C and KOtBu at 150 °C for 4 h (Scheme 3). GC analysis revealed the formation of hydrogen species (see SI Fig. S3), clearly evidencing the participation of ethanol as a hydrogen donor in nitro reduction reactions.
On the basis of our optimization studies, ethanol degradation experiments, and literature,12a,b,19b we propose a plausible reaction mechanism (Fig. 4). Initially, in the presence of Pt/C and base (KOtBu), EtOH dehydrogenates and produces in situ hydrogen. This hydrogen interacts with the Pt/C and leads to the formation of active Pt–H species on the surface. Then Pt–H species will reduce the nitro group to nitroso (I), and further reduction followed by dehydrogenation gives phenyl hydroxylamine (II). In the next step, nitroso and phenyl hydroxylamine produce azoxy intermediate (III) via the condensation reaction. The azoxy intermediate further undergoes reduction to produce azo compound (IV) and again reduction to form hydrazo (V), and finally, the hydrazo compound is reduced to form aniline.
 | ||
| Fig. 4 Plausible reaction mechanism for the Pt/C catalyzed transfer hydrogenation of nitro compounds with ethanol. | ||
Conclusions
In summary, we could show that ethanol can be effectively used as a hydrogen donor for the transfer hydrogenation of nitroarenes to anilines using a recyclable and anti-poisoning Pt/C catalyst. The catalytic protocol tolerated numerous nitroarenes bearing sensitive functional groups such as sulfur functionalities (–SO2Me, –SO2, –SO3H, –SMe, etc.), halogen (–F, –Cl, –Br, and –I), –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, –CO2H, –CONH2, etc., selectively delivering the structurally diverse aniline products in moderate to high yields that are hard to access by the reported methods, therefore, opening up new possibilities for synthetic and medicinal applications.
O, –CO2H, –CONH2, etc., selectively delivering the structurally diverse aniline products in moderate to high yields that are hard to access by the reported methods, therefore, opening up new possibilities for synthetic and medicinal applications.
Conflicts of interest
There are no conflicts to declare.Data availability
The data underlying this study are available in the published article and its supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5su00864f.Acknowledgements
K. N. acknowledges DST-SERB, New Delhi (CRG/2023/005734), for financial support. C. D. is thankful to UGC-SRF.Notes and references
- B. L. Tran, S. I. Johnson, K. P. Brooks and S. T. Autrey, ACS Sustainable Chem. Eng., 2021, 9, 7130–7138 CrossRef CAS.
- F. da Silva Fernandes, É. S. de Souza, L. M. Carneiro, J. P. Alves Silva, J. V. B. de Souza and J. da Silva Batista, Int. J. Microbiol., 2022, 7878830 CAS.
- https://www.statista.com/statistics/274142/global-ethanol-production-since-2000/#:%7E:text=Globalfuelethanolproduction2016D2024%26text=Worldwidefuelethanolproductionclimbed,fromcropssuchascorn, accessed on March 27, 2025.
- N. N. Deshavath, V. V. Goud and V. D. Veeranki, Bioref., 2024, 14, 3551–3563 CAS.
- (a) M. Hans, Y. Lugani, A. K. Chandel, R. Rai and S. Kumar, Biomass Convers. Bioref., 2023, 13, 7423–7440 CrossRef CAS PubMed; (b) S. Onuki, J. A. Koziel, W. S. Jenks, L. Cai, D. Grewell and J. H. van Leeuwen, J. Inst. Brew., 2016, 122, 588–598 CrossRef CAS.
- (a) H. Xiang, R. Xin, N. Prasongthum, P. Natewong, T. Sooknoi, J. Wang, P. Reubroycharoen and X. Fan, Resour. Chem. Mater., 2022, 1, 47–68 CAS; (b) J. Sun and Y. Wang, ACS Catal., 2014, 4, 1078–1090 CrossRef CAS; (c) L. He, B.-C. Zhou, D.-H. Sun, W.-C. Li, W.-L. Lv, J. Wang, Y.-Q. Liang and A.-H. Lu, ACS Catal., 2023, 13, 11291–11304 CrossRef CAS; (d) M. Zhang and Y. Yu, Ind. Eng. Chem. Res., 2013, 52, 9505–9514 CrossRef CAS; (e) Y. W. Cheng, C. C. Chong, C. K. Cheng, K. H. Ng, T. Witoon and J. C. Juan, J. Clean. Prod., 2020, 249, 119323 CrossRef CAS; (f) A. Mohsenzadeh, A. Zamani and M. J. Taherzadeh, ChemBioEng Rev., 2017, 4, 75–91 CrossRef; (g) N. Xiang, P. Xu, N. Ran and T. Ye, RSC Adv., 2017, 7, 38586–38593 RSC; (h) K. Inui, T. Kurabayashi and S. Sato, J. Catal., 2002, 212, 207–215 CrossRef CAS; (i) A. Rodríguez-Gómez, F. Dorado, P. Sánchez and A. R. de la Osa, J. Energy Chem., 2022, 70, 394–406 CrossRef.
- (a) C. C. Meyer, N. P. Stafford, M. J. Cheng and M. J. Krische, Angew. Chem., Int. Ed., 2021, 60, 10542–10546 CrossRef CAS PubMed; (b) S. Yadav and M. R. Kuram, Eur. J. Org Chem., 2023, 26, e202201344 CrossRef CAS; (c) C. C. Meyer and M. J. Krische, J. Org. Chem., 2023, 88, 4965–4974 CrossRef CAS PubMed; (d) M. Roca Jungfer, J. L. Schwarz, F. Rominger, T. Oeser, R. Paciello, A. S. K. Hashmi and T. Schaub, ChemCatChem, 2024, 16, e202301588 CrossRef CAS.
- (a) Y. Wang, Z. Huang, X. Leng, H. Zhu, G. Liu and Z. Huang, J. Am. Chem. Soc., 2018, 140, 4417–4429 CrossRef CAS PubMed; (b) S. Revathi and T. Ghatak, J. Catal., 2024, 429, 115207 CrossRef CAS; (c) D. Panja, S. Dey, R. Saha, R. Sahu, G. K. Das, P. Bhobe and S. Kundu, Green Chem., 2023, 25, 9374–9387 RSC; (d) P. Weingart, Y. Sun and W. R. Thiel, ChemCatChem, 2020, 12, 6223–6233 CrossRef CAS; (e) R. D. Patil and S. Pratihar, RSC Sustain., 2025, 156, 155439 CAS.
- (a) D. Wang and D. Astruc, Chem. Rev., 2015, 115, 6621–6686 Search PubMed; (b) C. G. Santana and M. J. Krische, ACS Catal., 2021, 11, 5572–5585 CrossRef CAS PubMed; (c) V. Vermaak, H. C. M. Vosloo and A. J. Swarts, Coord. Chem. Rev., 2024, 507, 215716 Search PubMed; (d) D. Sharma, P. Choudhary, P. Mittal, S. Kumar, A. Gouda and V. Krishnan, ACS Catal., 2024, 14, 4211–4248 CrossRef CAS; (e) D. Sharma, P. Choudhary, S. Kumar and V. Krishnan, J. Colloid Interface Sci., 2024, 657, 449–462 Search PubMed; (f) M. N. Shaikh, M. A. Aziz, A. N. Kalanthoden, A. Helal, A. S. Hakeem and M. Bououdina, Catal. Sci. Technol., 2018, 8, 4709–4718 Search PubMed; (g) M. N. Shaikh, A. Helal, A. N. Kalanthoden, B. Najjar, M. A. Aziz and H. D. Mohamed, Catal. Commun., 2019, 119, 134–138 CrossRef CAS; (h) M. N. Shaikh, RSC Adv., 2019, 9, 28199–28207 Search PubMed; (i) S. A. Nafiu, M. A. Aziz and M. N. Shaikh, J. Organomet. Chem., 2020, 922, 121361 CrossRef CAS; (j) M. N. Shaikh, M. M. Abdelnaby, A. S. Hakeem, G. A. Nasser and Z. H. Yamani, ACS Appl. Nano Mater., 2021, 4, 3508–3518 Search PubMed.
- (a) M. Trincado, D. Banerjee and H. Grützmacher, Energy Environ. Sci., 2014, 7, 2464–2503 Search PubMed; (b) A. Bisarya, S. Karim, H. Narjinari, A. Banerjee, V. Arora, S. Dhole, A. Dutta and A. Kumar, Chem. Commun., 2024, 60, 4148–4169 RSC; (c) M. Nielsen, A. Kammer, D. Cozzula, H. Junge, S. Gladiali and M. Beller, Angew. Chem., Int. Ed., 2011, 50, 9593–9597 CrossRef CAS; (d) T. L. Lohr, A. R. Mouat, N. M. Schweitzer, P. C. Stair, M. Delferro and T. J. Marks, Energy Environ. Sci., 2017, 10, 1558–1562 Search PubMed; (e) B. Taleb, R. Jahjah, D. Cornu, M. Bechelany, M. A. Ajami, G. Kataya, A. Hijazi and M. H. El-Dakdouki, Molecules, 2023, 28, 7541 Search PubMed.
- M. Brodt, K. Muller, J. Kerres, I. Katsounaros, K. Mayrhofer, P. Preuster, P. Wasserscheid and S. Thiele, Energy Technol., 2021, 9, 2100164 Search PubMed.
- (a) V. Goyal, T. Bhatt, C. Dewangan, A. Narani, G. Naik, E. Balaraman, K. Natte and R. V. Jagadeesh, J. Org. Chem., 2023, 88, 2245–2259 CrossRef CAS; (b) J. Sklyaruk, V. Zubar, J. C. Borghs and M. Rueping, Org. Lett., 2020, 22, 6067–6071 Search PubMed; (c) N. Garg, H. P. Somasundharam, P. Dahiya and B. Sundararaju, Chem. Commun., 2022, 58, 9930–9933 RSC; (d) G. Huang, L. Wang, H. Luo, S. Shang, B. Chen, S. Gao and Y. An, Catal. Sci. Technol., 2020, 10, 2769–2773 Search PubMed; (e) A. Mandal, K. Ganguli, M. Pradhan, A. Gorai and S. Kundu, ChemSusChem, 2023, 16, e202300683 CrossRef CAS; (f) N. Sarki, V. Goyal, N. K. Tyagi, A. Puttaswamy, A. Narani, A. Ray and K. Natte, ChemCatChem, 2021, 13, 1722–1729 CrossRef CAS; (g) V. Goyal, J. Gahtori, A. Narani, P. Gupta, A. Bordoloi and K. Natte, J. Org. Chem., 2019, 84, 1534–1543 CrossRef PubMed; (h) V. Goyal, N. Sarki, K. Natte and A. Ray, J. Indian Chem. Soc., 2021, 98, 100014 Search PubMed.
- (a) H. Wang, B. Liu, F. Liu, Y. Wang, X. Lan, S. Wang, B. Ali and T. Wang, ACS Sustainable Chem. Eng., 2020, 8, 8195–8205 CrossRef CAS; (b) T. Zweifel, J.-V. Naubron, T. Büttner, T. Ott and H. Grützmacher, Angew. Chem., Int. Ed., 2008, 47, 3245–3249 Search PubMed; (c) N. Castellanos-Blanco, A. Arévalo and J. J. García, Dalton Trans., 2016, 45, 13604–13614 RSC; (d) R. Ghosh, N. C. Jana, S. Panda and B. Bagh, ACS Sustainable Chem. Eng., 2021, 9, 4903–4914 Search PubMed; (e) R. A. Farrar-Tobar, B. Wozniak, A. Savini, S. Hinze, S. Tin and J. G. de Vries, Angew. Chem., Int. Ed., 2019, 58, 1129–1133 CrossRef CAS PubMed; (f) R. Ghosh, R. R. Behera, S. Panda, S. K. Behera, N. C. Jana and B. Bagh, ChemCatChem, 2023, 15, e202201062 Search PubMed.
- (a) G. A. Deluga, J. R. Salge, L. D. Schmidt and X. E. Verykios, Science, 2004, 303, 993–997 Search PubMed; (b) M. Ni, D. Y. C. Leung and M. K. H. Leung, Int. J. Hydrogen Energy, 2007, 32, 3238–3247 Search PubMed.
- (a) Y. Wu, X. Zhu, S. Du, G. Huang, B. Zhou, Y. Lu, Y. Li, S. P. Jiang, L. Tao and S. Wang, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2300625120 Search PubMed; (b) J. P. Barham, G. Coulthard, K. J. Emery, E. Doni, F. Cumine, G. Nocera, M. P. John, E. Leonard, A. Berlouis, T. McGuire, T. Tuttle and J. A. Murphy, J. Am. Chem. Soc., 2016, 138, 7402–7410 Search PubMed.
- (a) D. Formenti, F. Ferretti, F. K. Scharnagl and M. Beller, Chem. Rev., 2019, 119, 2611–2680 CrossRef CAS PubMed; (b) H. K. Kadam and S. G. Tilve, RSC Adv., 2015, 5, 83391–83407 RSC; (c) V. Goyal, N. Sarki, B. Singh, A. Ray, M. Poddar, A. Bordoloi, A. Narani and K. Natte, ACS Appl. Nano Mater., 2020, 3, 11070–11079 CrossRef CAS; (d) R. V. Jagadeesh, A.-E. Surkus, H. Junge, M.-M. Pohl, J. Radnik, J. Rabeah, H. Huan, V. Schünemann, A. Brückner and M. Beller, Science, 2013, 342, 1073–1076 Search PubMed.
- (a) K. A. Scott and J. T. Njardarson, Top. Curr. Chem., 2018, 376, 5 Search PubMed; (b) M. Feng, B. Tang, H. S. Liang and X. Jiang, Curr. Top. Med. Chem., 2016, 16, 1200–1216 Search PubMed; (c) C. Zhao, K. P. Rakesh, L. Ravidar, W.-Y. Fang and H.-L. Qin, Eur. J. Med. Chem., 2019, 162, 679–734 CrossRef CAS PubMed; (d) L. Lückemeier, T. De Vos, L. Schlichter, C. Gutheil, C. G. Daniliuc and F. Glorius, J. Am. Chem. Soc., 2024, 146, 5864–5871 Search PubMed; (e) P. Mäder and L. Kattner, J. Med. Chem., 2020, 63, 14243–14275 Search PubMed.
- (a) H. Ishikawa, N. Nakatani, S. Yamaguchi, T. Mizugaki and T. Mitsudome, ACS Catal., 2023, 13, 5744–5751 Search PubMed; (b) C. Dewangan, S. Kumawat, T. Bhatt and K. Natte, Chem. Commun., 2023, 59, 14709–14712 Search PubMed.
- (a) R. Xiong, W. Ren, Z. Wang and M. Zhang, ChemCatChem, 2021, 13, 548–552 Search PubMed; (b) H. U. Blaser, H. Steiner and M. Studer, ChemCatChem, 2009, 1, 210 CrossRef CAS.
- (a) I. Mochida and K.-H. Choi, J. Jpn. Pet. Inst., 2004, 47, 145–163 Search PubMed; (b) J. A. Anabtawi, D. S. Redwan, A. M. Al-Jarallah and A. M. Aitani, Fuel Sci. Technol. Int., 1991, 9, 1–23 CrossRef CAS; (c) P. Grange and X. Vanhaeren, Catal. Today, 1997, 36, 375–391 Search PubMed.
- H. Ishikawa, S. Yamaguchi, T. Mizugaki and T. Mitsudome, ACS Catal., 2024, 14, 4501–4509 CrossRef CAS.
- (a) L. He, K. Natte, J. Rabeah, C. Taeschler, H. Neumann, A. Brückner and M. Beller, Angew. Chem., Int. Ed., 2015, 54, 43204324 Search PubMed; (b) S. Nishimura, Handbook of Heterogeneous Catalytic Hydrogenation for Organic Synthesis, Wiley-Interscience: New York, 2001, pp. 43–44, 271, 336, 499 Search PubMed; (c) M. N. Shaikh, M. A. Aziz and Z. H. Yamani, Catal. Sci. Technol., 2020, 10, 6544–6551 RSC.
| This journal is © The Royal Society of Chemistry 2026 |