Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Life cycle assessment of seaweed-based biorefineries: environmental impacts, hotspots, and pathways for a circular bioeconomy
SantanKumar
Chaurasiya
 a,
Nathan
Preuss
a and
Fengqi
You
a,
Nathan
Preuss
a and
Fengqi
You
 *abcde
*abcde
aCollege of Engineering, Cornell University, Ithaca, New York 14853, USA. E-mail: fengqi.you@cornell.edu
bRobert Frederick Smith School of Chemical and Biomolecular Engineering, Cornell University, Ithaca, New York 14853, USA
cCornell University AI for Science Institute, Cornell University, Ithaca, New York 14853, USA
dCornell AI for Sustainability Initiative (CAISI), Cornell University, Ithaca, New York 14853, USA
eCornell Institute for Digital Agriculture (CIDA), Cornell University, Ithaca, New York 14853, USA
First published on 22nd January 2026
Abstract
Seaweed-based biorefineries are increasingly recognized as promising contributors to the circular bioeconomy, offering renewable pathways for fuels, materials, fertilizers, cosmetics, and bioactive compounds. This review synthesizes approximately sixty life cycle assessments (LCA) studies covering diverse product categories, including biofuels, bioplastics, fertilizers and biostimulants, cosmetics, protein and feed, construction materials, food additives, and biochemicals. Across these applications, drying and energy-intensive extraction/crosslinking processes consistently emerge as dominant hotspots, often accounting for 50–70% of total GWP. Consequently, analysis shows that substituting greener solvents (reducing GWP by 20–40%) and implementing renewably powered drying (reducing GWP by ∼15–25%) are the most critical levers for impact reduction. Fertilizers and biostimulants show potential to reduce greenhouse gas emissions through substitution of synthetic inputs, while bioplastics and biofuels highlight trade-offs between energy use and co-product valorization. Cosmetics, food additives, and construction uses remain underexplored but demonstrate niche opportunities for high-value and low-carbon products. Cross-cutting analysis reveals methodological gaps, including inconsistent functional units, narrow impact category coverage, and limited integration of techno-economic analysis. Cultivation-focused LCAs further underscore the influence of farming practices, seasonality, and feedstock quality on downstream performance. Key challenges include high moisture content, ash and salt constraints in thermochemical conversion, insecure feedstock supply, and fragmented system modeling. Addressing these requires harmonized LCA methodologies, improved pretreatment strategies, and integration of techno-economic analysis to bridge environmental and economic performance. Compared to previous reviews, this study advances the field by synthesizing product-specific LCAs alongside cultivation studies, highlighting underrepresented products such as biostimulants and construction materials, and framing hotspots at process, system, and policy levels. Seaweed biorefineries present significant opportunities for climate mitigation, resource efficiency, and sustainable industry development, provided that future assessments expand in scale, scope, and methodological rigor.
Sustainability spotlightThis comprehensive review synthesizes the life cycle environmental impacts of seaweed-based bioproducts, which are poised to become a cornerstone of the circular bioeconomy. By identifying persistent hotspots (e.g., thermal drying, solvent-based extraction) and proven mitigation levers (e.g., renewable energy integration, green solvents), this work provides an actionable roadmap for researchers and industry. The findings directly support the advancement of several UN Sustainable Development Goals, including SDG 9 (Industry, Innovation, and Infrastructure), SDG 12 (Responsible Consumption and Production), and SDG 13 (Climate Action), by guiding the development of viable, low-carbon, and resource-efficient alternatives to fossil-based products. |
1. Introduction
Modern society faces escalating challenges from climate change, resource depletion, food insecurity, and the growing demand for sustainable products.1,2 The global population is projected to reach 9.7 billion by 2050, which places immense pressure on food, energy, and material supply chains.3,4 To maintain global economic growth while minimizing environmental degradation, the utilization of renewable feedstocks and clean energy alternatives must be accelerated. This transition is essential for reducing dependency on fossil fuels, conserving natural resources, and enabling a shift toward a more sustainable bio-based economy.5 An increasing number of countries are recognizing bioeconomy as a strategic pathway to sustainable development. For instance, the European Union has adopted a Bioeconomy Strategy,6 while the U.S. has issued an Executive Order on Advancing Biotechnology and Biomanufacturing.7 Similarly, countries like China, India, and Brazil are investing in bio-based innovation ecosystems and biomass valorization programs.8 These efforts aim to meet several of the 17 Sustainable Development Goals (SDGs) set by the United Nations (UN), particularly those related to climate action (SDG 13), responsible consumption and production (SDG 12), and affordable and clean energy (SDG 7).9,10 These efforts also follow some of the 12 principles of Green Chemistry, such as using renewable feedstocks (Principle 7), being energy efficient (Principle 6), designing for degradation (Principle 10), and using safer solvents and auxiliaries (Principle 5).11Biorefineries, which convert biomass into marketable products such as biofuels, bioplastics, food ingredients, pharmaceuticals, and fertilizers, are central to this transition. They enable the eco-innovative and sustainable transformation of biological resources through integrated processes, many of which align with SDG targets for clean energy (SDG 7), economic growth (SDG 8,9,12), and climate mitigation (SDG 13).10 For example, ethanol derived from agricultural residues or biodiesel from algae directly substitutes fossil-based fuels and reduces carbon emissions. Importantly, because biomass is of biogenic origin, carbon dioxide (CO2) released during its conversion is considered part of the natural carbon cycle. Under current life cycle assessment (LCA) guidelines and IPCC recommendations, these biogenic emissions are accounted for separately and typically not modeled as contributing to a net increase in atmospheric CO2 levels.12 Biorefineries are categorized into three generations based on the type of feedstock used and the technological advancements involved. In order to develop sustainable and profitable bio-based industries, these generations must be recognized.13 First-generation biorefineries rely on food crops such as corn, sugarcane, and wheat,14 while these systems are commercially established and have well-developed supply chains, they pose concerns related to food-versus-fuel conflicts, land use change, and biodiversity loss. Second-generation biorefineries utilize non-food lignocellulosic biomass, including crop residues, forestry waste, and dedicated energy crops.15 These offer improved environmental benefits by reducing competition with food systems and utilizing agricultural waste; however, they require more complex and energy-intensive pretreatment processes. Third-generation biorefineries are based on algal and microbial feedstocks, including microalgae and seaweed, which are fast-growing, high-yielding, and capable of growing in saline or wastewater environments.16 Unlike earlier generations, these systems do not require arable land or freshwater, can perform nutrient bioextraction to mitigate coastal eutrophication, and provide circular residues for co-product valorization, significantly reducing the environmental footprint of biorefineries. Later generations are preferred not only because they reduce pressure on land and freshwater resources, but also because they align more closely with circular bioeconomy principles and global climate goals. The transition to third-generation biorefineries therefore represents a strategic evolution toward low-impact, high-efficiency, and carbon-neutral production systems, as depicted in Fig. 1.13–16
 | ||
| Fig. 1 Classification of seaweed-based biorefinery systems based on biorefinery generations, illustrating technological evolution from traditional use (first generation) to advanced cascading extraction and integration with bioenergy and co-products (third generation).17 | ||
Seaweed-based biorefineries, often classified as third-generation systems, offer several unique advantages over first-generation (e.g., corn, sugarcane), second-generation (e.g., lignocellulosic biomass), and other third-generation options such as microalgae. Unlike 1 generation and 2nd feedstocks, seaweed cultivation requires no arable land, freshwater, or synthetic fertilizer inputs, significantly reducing competition with food systems and freshwater stress (SDG 2 & 6).8 In comparison with microalgae, seaweed cultivation is often lower in energy demand due to its passive nutrient uptake from seawater and ease of harvesting without expensive photobioreactors or closed systems. From an environmental perspective, several LCA studies suggest that seaweed cultivation offers lower Global Warming Potential (GWP) and eutrophication impacts per kg of biomass compared to first-generation (e.g., corn, sugarcane), second-generation (e.g., lignocellulosic biomass), and other third-generation options such as microalgae, particularly when integrated into near-shore or offshore farming systems with minimal land, fertilizer, and freshwater inputs. However, challenges persist. Seaweed exhibits high moisture content (80–90%), necessitating energy-intensive drying or dewatering unless co-located with processing units.18 To provide a more intuitive comparison of GWP-related drivers across biorefinery generations, a qualitative “traffic-light” matrix is presented in Fig. 2, summarizing key conditions such as land use, fertilizer demand, drying intensity, offshore logistics, and renewable heat integration.
Additionally, the geographic constraints, favoring temperate or tropical coastal zones, may limit year-round cultivation and scalability in inland or landlocked regions. Cost-wise, seaweed cultivation remains labor- and infrastructure-intensive, especially where mechanization or offshore logistics are not well-developed. While microalgae may yield higher-value compounds (e.g., lipids, pigments), their cultivation costs and energy footprints are generally higher. In contrast, seaweed biorefineries can enable cascaded extraction (e.g., protein, biostimulants, and polysaccharides) that promotes circularity and whole-biomass utilization. Thus, seaweed-based biorefineries strike a balance between ecological sustainability and process feasibility, positioning them as a promising feedstock for blue bioeconomy development in suitable coastal geographies. Seaweed species, in particular, are fast-growing, with cultivation cycles typically ranging from 3 to 8 months and achieving annual productivities of 1–5 tonnes of dry weight per hectare.19 This high-yield, rapid rotation potential makes them attractive feedstocks capable of growing in saline or wastewater environments. When evaluating their role in biogenic carbon sequestration, this short rotation period must be accounted for using dynamic assessment methods (e.g., dynamic LCA, GWPbio) that consider the temporal balance between carbon uptake and release, and the ultimate fate of the carbon in end-use products or sequestration pathways.20,21
Macroalgae, commonly referred to as seaweeds, possess significant potential to completely or partially replace terrestrial biomass and generate sustainable bioenergy and biomaterials.22 This is because the cultivation of seaweed does not require either land or freshwater.23 Furthermore, seaweed has the potential to provide a vast range of sustainable products that include food, cosmetics, biodiesel, biofuel, biogas, bioplastics, pharmaceutical, fertilizers, and other chemicals.1,24 Moreover, seaweed cultivation directly decarbonizes multiple sectors by absorbing CO2 and provides co-benefits such as protecting marine biodiversity, reducing eutrophication, and conserving land and freshwater resources.25,26
Previous reviews on seaweed biorefineries have primarily focused on technological pathways, conversion processes, or economic feasibility, often treating environmental performance only briefly or in isolation.24,27,28 In contrast, this review provides the systematic synthesis of life cycle assessment (LCA) studies across eight major product categories, biofuels, bioplastics, fertilizers and biostimulants, cosmetics, protein and feed, construction materials, food additives, and biochemicals. Unlike earlier reviews, it explicitly integrates cultivation-focused LCAs to highlight the role of farming practices, seasonality, and feedstock quality in shaping downstream impacts. This paper also advances the literature by identifying cross-cutting environmental hotspots (e.g., drying, transport, extraction, crosslinking, infrastructure) and linking them with methodological challenges such as inconsistent functional units, limited Techno-Economic Analysis (TEA)-LCA integration, and underrepresentation of impact categories. Moreover, the review emphasizes underexplored product streams, such as biostimulants, cosmetics, and construction applications, thereby expanding the scope beyond conventional biofuels and bioplastics. Together, these novelties position the study to not only consolidate fragmented evidence but also to outline research priorities and opportunities for scaling seaweed biorefineries within the circular bioeconomy.
2. Materials and methods
2.1. Literature search strategy and selection of LCA studies
This study conducted a systematic literature review of LCA studies related to seaweed biorefineries. Two major academic databases were used: Scopus (https://www.scopus.com, accessed on 1 May 2025) and Web of Science (https://www.webofscience.com, accessed on or before 1 May 2025). The following search strings were employed:• On Scopus: “LCA” OR “life cycle analysis” AND “environmental impact” AND “seaweed” (searched within title, abstract, and keywords).
• On Web of Science: “life cycle assessment” OR “LCA” AND “seaweed” AND “environmental impact” (topic search across all indexed fields).
After compiling records from both sources, duplicates were removed. The remaining entries underwent a two-stage screening process. First, titles and abstracts were reviewed for relevance. Then, full-text articles were assessed against a set of inclusion and exclusion criteria.
• Studies that clearly apply and report LCA methodology with quantifiable environmental impacts.
• Focus on seaweed-derived bioproducts (e.g., fuels, plastics, fertilizers, protein, cosmetics).
• Reviews, commentaries, or conceptual works not including formal LCA methodology.
A total of 361 records were retrieved initially (Scopus = 205; Web of Science = 156). After screening and eligibility assessment, 60 studies were included in the final review.
2.2. Data extraction and thematic analysis of LCA studies
The studies were systematically reviewed to extract relevant methodological and contextual information. The data extraction focused on the following parameters:• Study origin (country).
• Seaweed species and product type (e.g., brown, red, green; bioplastic, biofuel, fertilizer, etc.)
• Functional unit (FU).
• System boundary (e.g., cradle-to-gate, cradle-to-grave, cradle-to-cradle).
• Impact assessment method and impact categories.
• Identified environmental hotspots.
The extracted information is synthesized in Table 3, which provides an overview of key characteristics and findings across all reviewed LCA studies on seaweed-based biorefineries.
3. Overview of seaweed cultivation, harvesting, and biomass conversion technologies
This section reviews key aspects of macroalgal resources that are foundational to seaweed-based biorefineries. It begins with an overview of major seaweed types classified based on their pigmentation and biochemical profiles: red, brown, and green. This is followed by a summary of global cultivation trends and harvesting techniques. These elements are essential, as species-specific composition and farming practices strongly influence both environmental impacts and techno-economic performance in biorefinery pathways. The section concludes with an outline of key value-added products (e.g., bioplastics, biofuels, biostimulants) derived from seaweed, highlighting their relevance to circular bioeconomy and sustainable development. Understanding this foundation is critical for linking upstream biomass characteristics with downstream processing, environmental assessment, and product development.3.1. Overview of seaweed and type
Seaweed cultivation is rapidly emerging as a key component of sustainable aquaculture, offering ecological benefits such as biodiversity enhancement and habitat provision while supporting expanding production across Asia. Asian countries such as China, Indonesia, the Philippines, Japan, and South Korea lead global seaweed production due to favorable coastal environments and well-established farming infrastructure. From 2000 to 2020, global seaweed production increased significantly from 10.6 million tons to 35 million tons, driven by demand for hydrocolloids and sustainable materials. The most dominant cultivated species include Undaria, Pyropia, Gracilaria, Eucheumatoids, and Saccharina.87 The cultivation of Phaeophyceae (brown algae) and Rhodophyta (red algae) has been increased by 3.1 million tons to 16.4 million tons, and 1 million tons to 18.3 million tons respectively. However, a significant decline has been observed in Chlorophyta seaweed (green algae).88 Over 95% of global macroalgae production is dominated by a few key seaweed groups, including Kappaphycus and Eucheuma (33.5%), Gracilaria (10.5%), Porphyra/Pyropia (8.6%), Undaria (7.4%), and Laminaria saccharina (35.4%), refer Fig. 3. In contrast, Europe contributed only 287![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 033 tonnes of seaweed in 2019, representing just 0.8% of global production.89 A World Bank analysis of 10 emerging seaweed markets estimates that the global commercial potential of the seaweed industry could reach USD 11.8 billion by 2030, driven by demand in bio-based materials, food additives, pharmaceuticals, and climate-focused applications.90 Despite this forecast, much of the sector's economic value remains untapped due to limited processing capacity, policy support, and market access, especially in developing nations. Moreover, another World Bank report in 2016 projects that increasing seaweed cultivation by 14% annually could result in production of 500 million dry tons by 2050, potentially contributing to a 10% increase in global food supply. This expansion is also linked to improved income generation for coastal communities and enhanced quality of life, although these two latter benefits are more qualitative and context-dependent rather than direct 10% increases.91 Macroalgal biomass contains various polysaccharides, primarily made up of glucose units known as glucans. The dominant glucans differ by algal type: in green algae, cellulose and starch are prevalent; in red algae, cellulose and floridean starch are common; while brown algae mainly feature cellulose and laminarin.92,93
033 tonnes of seaweed in 2019, representing just 0.8% of global production.89 A World Bank analysis of 10 emerging seaweed markets estimates that the global commercial potential of the seaweed industry could reach USD 11.8 billion by 2030, driven by demand in bio-based materials, food additives, pharmaceuticals, and climate-focused applications.90 Despite this forecast, much of the sector's economic value remains untapped due to limited processing capacity, policy support, and market access, especially in developing nations. Moreover, another World Bank report in 2016 projects that increasing seaweed cultivation by 14% annually could result in production of 500 million dry tons by 2050, potentially contributing to a 10% increase in global food supply. This expansion is also linked to improved income generation for coastal communities and enhanced quality of life, although these two latter benefits are more qualitative and context-dependent rather than direct 10% increases.91 Macroalgal biomass contains various polysaccharides, primarily made up of glucose units known as glucans. The dominant glucans differ by algal type: in green algae, cellulose and starch are prevalent; in red algae, cellulose and floridean starch are common; while brown algae mainly feature cellulose and laminarin.92,93
 | ||
| Fig. 3 Global seaweed production by major groups, showing Laminaria/Saccharina leading at 35.33%, carrageenan seaweeds at 33.46%, Agar seaweeds at 10.48%, Porphyra/Pyropia at 8.59%, other seaweeds at 7.38%, and Undaria at 4.76%; key production regions noted for each group (e.g., China, South Korea, Japan, Canada).94 | ||
Seaweeds are broadly classified into three main groups based on the pigments they use for photosynthesis: green (Chlorophyta), red (Rhodophyta), and brown (Phaeophyta).95 These marine macroalgae typically consist of 80–90% water and, on a dry weight (DW) basis, contain approximately 50% carbohydrates, 1–3% lipids, and 7–38% minerals.96 Their protein content is highly variable across species, ranging from 10% to as high as 47% DW, and is rich in essential amino acids.
Around 1500 recognized species of green macroalgae occur in shallow coastal environments such as bays, tidal pools, and estuaries. Prominent genera include Ulva, Codium, and Halimeda, which contain chlorophylls A and B as well as carotenoids, typical of the broader Chlorophyta group.97 A characteristic feature of this division is the presence of the complex sulfated heteropolysaccharide ulvan in their cell walls.98,99 In addition, Chlorophyta are rich in carbohydrates, with cellulose and hemicellulose representing 53–70% of their dry weight.100 Ulvan displays a variety of biochemical and functional properties that make it attractive for pharmaceutical and agricultural uses. However, its weak gelling capacity currently limits large-scale commercial exploitation. Interestingly, ulvan solutions are pseudoplastic; they exhibit low viscosity that decreases further when shear rate increases.101
Nearly 4000 identified species of red macroalgae occur in both cold, deeper waters and in warmer, shallower seas, making them one of the most widely distributed groups of marine algae.102 Most species typically inhabit submerged or subtidal environments, although only a limited number can endure desiccation or direct exposure (p). A considerable portion of these algae contain a high percentage of carbohydrates in their dry weight (53–76%), primarily in the form of polysaccharides such as agar, cellulose, and carrageenan. Under favorable conditions, members of Rhodophyta can reach lengths of about four feet.103 Asexual propagation takes place through non-motile spores, whereas sexual reproduction involves the union of spermatia with carpogonia (female gametangia), resulting in heterogamous offspring. These gametes are well differentiated into non-motile male and female cells. Some species allow calcium carbonate deposition on their fronds, contributing to the growth of algal reefs in marine ecosystems. Economically, red algae hold great value: for example, Gelidium is a major source of agar, while Irish moss (Chondrus crispus) is widely utilized for carrageenan production.104,105
Around 1500 different species of brown macroalgae exist across genera such as Fucus, Laminaria, Himanthalia, Undaria, Alaria, and Ascophyllum, all of which belong to the Phaeophyta division.106 They are generally distributed in cooler waters of shallow marine zones. Brown macroalgae exhibits a diverse metabolic structure, with carbohydrate content ranging between 34% and 76% of their dry mass.102 The distinctive brown coloration arises from a dark pigment that masks chlorophyll. Nowadays, seaweeds are utilized in various sectors including the production of industrial phycocolloids, animal and human nutrition, and agriculture, particularly as biofertilizers. Despite their impressive nutritional and functional potential, the full dietary value of seaweeds remains underutilized, largely due to the complexity of their structural polysaccharides.107
3.2. Seaweed cultivation and harvesting
In the past, seaweed was mostly collected from wild or natural habitats, which often led to overharvesting and strain on marine ecosystems. To make seaweed use more sustainable, researchers and farmers began developing new farming techniques to domesticate native species. The success of large-scale seaweed farming has depended on several factors, like how well a species can regenerate, its physical structure, and how it responds to environmental conditions such as temperature, light, nutrients, and water movement.108 The cultivation method used usually depends on the specific type of seaweed. Some genera like Eucheuma, Kappaphycus, Chondrus, and Gracilaria can be grown through simple vegetative propagation. Others, such as Laminaria, Undaria, Porphyra, and Enteromorpha, rely on spores and need a more complex, two- or multi-step farming process because they don't survive well when propagated vegetatively. Whether a species grows clonally or as individual units also affects how it's farmed.29,109 Clonal genera like Gracilaria and Kappaphycus, which can grow from fragments, are often directly planted into farming systems. There are many ways to farm seaweed, ranging from controlled environments like tanks and ponds to open-water systems using longlines or rafts. For species like kelp, hatcheries or nurseries are often needed for early growth stages, which makes the process more industrial. While many cultivation techniques have been explored, the most successful ones tend to be those that are cost-effective and easier to manage.110 Different farming methods, such as fixed off-bottom, raft, or net systems, are used around the world (Table 1). Each method has its pros and cons; for instance, approaches like the off-bottom method, single raft, and hanging longline are particularly effective in shallow water, but may not perform as well in deeper or more turbulent conditions.87| Cultivation method | Advantages | Disadvantages |
|---|---|---|
| Floating raft | Promotes favorable environmental conditions; low capital cost; minimal infrastructure; suitable for nutrient-rich waters; enables passive nutrient uptake | Sensitive to weather changes; higher ecological risks due to drifting; uneven biomass growth patterns |
| Tube net | Lower risk from weather; uniform growth; suitable for deep water with minimal infrastructure | Requires more seedlings; limited reusability; higher material input and setup time |
| Off-bottom monoline | Easy to manage during low tide; low-cost setup | High seedling loss; sensitive to wind, tides, and storms; hard to control epiphytes; difficult to remove in bad weather |
| PVC pipe raft | Lightweight and easy to handle; low labor demand; reusable materials | Not biodegradable; higher fossil-based plastic footprint; less durable in high wave energy regions |
| Cage systems | Variety of cage designs possible; good for epiphyte control; can withstand harsh weather | High implementation and maintenance costs; risk of biofouling |
| Multiple raft longline | Suitable for large-scale operations; cost-effective per unit biomass; lower energy input per kg yield | Potential ecological impact (e.g., shading, growth of unwanted organisms); risk of loss in extreme weather |
| Spider web | High productivity in constrained areas; supports polyculture systems; increases space-use efficiency | Susceptible to epiphyte growth; weather-sensitive |
Seaweed harvesting methods fall into two main categories: manual and mechanical (Table 2). In manual harvesting workers hand-pick or cut seaweed fronds; it is energy-efficient and allows for selective removal of mature biomass, which helps maintain high quality for downstream processing. It also minimizes damage to the holdfast and surrounding ecosystem, supporting regrowth in the next cycle. However, manual harvesting is labor-intensive, time-consuming, and generally limited to small-scale operations or high-value products.111
| Criteria | Mechanical harvesting | Manual harvesting |
|---|---|---|
| Technologies/techniques | Mechanical cutters, automated harvesters with quality sensors | Hand-picking |
| Scalability | High; ideal for large-scale bioplastics feedstock | Low; best for niche or high-value bioplastics |
| Costs | High initial investment, low labor costs; suited for bulk production | Low initial cost, high ongoing labor; suitable for high-value bioplastics |
| Impact on quality and yield | Medium: potential damage could affect biopolymer yield and extraction quality | High; careful selection preserves quality |
| Energy consumption | High; necessary for large-scale operations | Low; minimal energy usage, labor intensive |
| Extraction efficiency | Medium: risk of contamination or damage during harvesting could lower efficiency | High; selective harvesting ensures high-quality inputs for extraction |
| Suitability for bioplastics production | Best for mass production where quantity is prioritized | Suitable for high-quality, high-value bioplastics |
Mechanical harvesting uses specialized cutters, conveyor systems, or sensor-guided boats to collect large volumes quickly. This approach is well-suited for industrial-scale farms targeting bulk commodities like bioplastics feedstock.112 Mechanical cutters can strip entire lines of seaweed in a single pass, dramatically reducing labor costs and turnaround time. On the downside, mechanical methods often cause more breakage of seaweed tissue and greater disturbance to the seabed and non-target organisms. The higher energy input and risk of mixed-species or immature fronds entering the process stream can also impact overall product quality and extraction efficiency.30 Balancing these trade-offs is key when choosing the right harvesting strategy for a given farm size, target product, and environmental setting.
3.3. Seaweed biomass conversion technologies
Utilizing seaweed biomass to produce a wide array of bio-based products is a key strategy in advancing sustainable marine biorefineries. This section explores the various thermochemical, biochemical, and physicochemical processes used to transform seaweed into biofuels, bioplastics, biostimulants, and high-value co-products. Key technologies include anaerobic digestion (AD), fermentation, hydrothermal liquefaction (HTL), and enzymatic extraction, each offering distinct pathways for valorizing different fractions of seaweed biomass, refer Fig. 4. The ongoing research and technological advancements in seaweed processing and product development are creating new opportunities for green technology and infrastructure investment. | ||
| Fig. 4 Schematic representation of the macroalgal biorefinery pathways,.4 The diagram illustrates the sequential stages from cultivation and harvesting to post-harvest processing, fractionation, and conversion routes for macroalgae. Both whole and fractionated biomass streams can undergo biochemical or thermochemical processes to yield a variety of value-added bio-products, such as bio-ethanol, bio-diesel, bio-oil, bio-char, and bio-syngas, as well as high-value compounds including pigments, proteins, lipids, and polysaccharides. Residues generated from extraction or conversion steps can further serve as feedstocks for energy recovery or the production of biochemicals and bio-materials, ensuring comprehensive biomass utilization within a circular bioeconomy framework, adapted from ref. 4 with permission from Elsevier (Kostas et al., Renewable and Sustainable Energy Reviews, 2021), copyright 2026. | ||
Bioethanol derived from algal biomass represents an environmentally sustainable and renewable route for biofuel generation.116 The fermentation process can be conducted through two main approaches: Separate Hydrolysis and Fermentation (SHF) and Simultaneous Saccharification and Fermentation (SSF).117 In the SHF method, hydrolysis and fermentation occur as two distinct, consecutive steps.118 In both cases, the hydrolysable sugars in seaweed (e.g., laminaran and alginate) enter the SP, where they are converted into simple sugars that serve as substrates for microbial fermentation. These sugars are then fermented into bioethanol, while some intermediates can also feed into the VFA pathway. Non-glucans such as agar, carrageenan, and alginate require additional hydrolysis, and efficient conversion of glucans and non-glucans into fermentable sugars is essential to achieve higher ethanol yields.117–119 In the SP, enzymatic hydrolysis is used to break down seaweed polysaccharides like cellulose and laminaran into fermentable sugars, without the need for acid pretreatment due to the absence of lignin in Laminaria japonica. The hydrolysate is then fermented using engineered Escherichia coli to yield ethanol at 0.281 kg kg−1 of dry Laminaria japonica biomass, followed by multi-step distillation and membrane separation to achieve 99.5 wt% ethanol purity.120,121 The SP offers relatively high ethanol yields but often requires genetically engineered strains and strict control of fermentation conditions, which may increase operational complexity.
The VFA pathway leverages partial AD to convert biomass into VFAs (yield: 0.35 kg per kg dry seaweed), with methanogenesis inhibited to maximize acid production. VFAs are recovered using methyl tert-butyl ether (MTBE) extraction, distilled, and hydrogenated catalytically (290 °C, 60 atm) to produce a mixture of alcohols (ethanol, propanol, butanol).122,123 The VFA pathway is advantageous for producing a broad spectrum of alcohol and intermediates; however, it depends heavily on solvent extraction and energy-intensive hydrogenation. Lastly, the MP converts volatile solids into biogas through complete AD. For example, AD of Laminaria digitata can yield 0.3 m3 CH4 per kg VS as biogas, which can be combusted to generate 770 kWh electricity per t dry seaweed, with 82% of the electricity sold externally.124 The MP provides a direct energy route with moderate yields but stands out for its ability to integrate with combined heat and power systems, offering energy self-sufficiency.125,126 Each technology also recovers digestate solids that could be sold as fertilizer, emphasizing circularity in seaweed biorefineries. Selecting the appropriate pathway depends on the desired product profile, energy requirements, and compatibility with downstream valorization strategies.
Pre-treatment of seaweed biomass is a critical yet often overlooked stage in large-scale biorefinery operations. Industrial-scale utilization requires the removal of impurities such as sand, silt, epiphytes, inorganic salts (e.g., NaCl, CaCO3), and heavy metals accumulated during cultivation or harvesting.137 Technologies employed for this purpose include freshwater or seawater washing, flotation separation, mechanical dewatering, and low-speed sedimentation. Some systems use air bubbling or vibratory sieving to dislodge fine particulates. However, these approaches are often energy- or water-intensive and may generate wastewater with high salt and nutrient loads, posing environmental burdens unless properly managed.
Recent studies have highlighted pre-treatment as a grand bottleneck in macroalgal valorization due to variability in feedstock composition and the difficulty in standardizing protocols across regions and species.138,139 For example, brown seaweeds such as Saccharina latissima may require extra washing cycles due to their mucilage content, while red seaweeds like Gracilaria may require alkaline conditioning to remove surface-bound phosphorus or sulfate residues.140,141 The integration of modular, low-energy pre-treatment units into farm-side infrastructure is a priority for scalable biorefinery deployment.
4. LCA framework and methodology
The LCA framework of seaweed-based biorefineries follows a structured approach to assess environmental impacts across the life cycle of a product or process. LCA is standardized by the International Organization for Standardization (ISO)142,143 and further supported by the International Reference Life Cycle Data System (ILCD).144 According to these standards, an LCA typically consists of four core stages: goal and scope definition, life cycle inventory analysis (LCI), impact assessment (LCIA), and interpretation.In practice, LCAs of biorefineries often face methodological challenges, including limited process data, inconsistencies in system boundary definitions, allocation rules, or functional units, and recurring issues such as defining goal, scope, and functional units; allocating feedstock and co-products; land use assumptions; and modeling biogenic carbon and emission timing.145 Additional concerns include the selection of impact categories, simplification of feedstock and process models, regional variations, and uncertainties in foreground and background datasets. These challenges can be mapped across different stages of the LCA framework, as illustrated in Fig. 5. This schematic serves as a visual guide to understand where methodological uncertainties emerge, from inconsistent goal definitions to underrepresentation of key environmental impacts and lack of sensitivity analysis. Each stage is critical in shaping the outcomes and reliability of sustainability assessments.
In the goal definition stage, the study must clarify its intended application (e.g., comparing seaweed-based vs. fossil-derived bioplastics), system boundary (e.g., cradle-to-gate or cradle-to-grave), and stakeholder audience.146,147 The choice between attributional LCA and consequential LCA further determines whether the analysis aims to describe the current system or explore broader system-level consequences.148 Recent studies have expanded traditional LCA by integrating social LCA and prospective LCA frameworks.149 These approaches assess labor conditions, social equity, and anticipate future impacts, which are increasingly relevant for evaluating seaweed-based biorefineries as part of a sustainable blue economy. Within the inventory phase, process data are collected across major life cycle stages, such as seaweed cultivation, harvesting, biorefinery processing, usage, and end-of-life management. Depending on the study's objective, this data may include energy inputs, chemical use, emissions, and co-product flows.150
The impact assessment stage applies characterization models to quantify how emissions and resource flows contribute to environmental impact categories, such as climate change, eutrophication, acidification, human toxicity, resource depletion, and water use. Common LCIA methods include ReCiPe, CML, TRACI, and ILCD,151–153 each offering different midpoint and endpoint indicators based on geographic focus and methodological assumptions. For example, ReCiPe enables detailed tracking of flows to both human health and ecosystem-level damages.154
The interpretation phase of an LCA typically involves identifying environmental hotspots, assessing uncertainties, and conducting sensitivity analyses.155 However, as observed in the reviewed literature, these steps are often either omitted or inconsistently reported,156 thereby reducing comparability across studies and weakening the robustness of conclusions. The selection and implementation of methodological elements, such as system boundaries, allocation strategies, and impact assessment methods, must be tailored to the study's goals, data availability, regional specificity, and intended audience.157 A rigorously designed LCA ensures consistent interpretation of trade-offs and environmental burdens, empowering stakeholders to make evidence-based decisions in sustainable product development, policy-making, and investment planning for seaweed-based biorefineries.158
4.1. Summary of key LCA studies
In recent years, a growing number of LCA studies have focused on evaluating the environmental performance of seaweed-based biorefineries across various product categories such as biofuels, bioplastics, fertilizers and biostimulants, cosmetics, construction material, protein and feed, food/additive and biochemicals/single cell oil. These studies provide valuable insights into process-level environmental burdens, highlight critical hotspots, and support the identification of opportunities for improvement throughout the life cycle. This section synthesizes key findings from 60 LCA studies, organized by product type, to identify common challenges, methodological trends, and potential strategies for sustainability enhancement within seaweed biorefineries.The most dominant hotspots in seaweed-to-fuel LCA studies are associated with (1) high-temperature processing (HTL, fermentation, etc.), (2) feedstock moisture content, and (3) energy source for heating. Moisture reduction prior to HTL can lower GWP by ∼15–25%, depending on the method. Integrated energy recovery systems (e.g., using HTL aqueous phase or digestate) are critical for improving net energy balance. A major unresolved contradiction lies in co-product allocation; studies diverge on whether to allocate emissions based on energy content, mass, or economic value, resulting in large swings in impact results. Sensitivity analyses show energy mix (renewables vs. fossil) is the single biggest driver of impact variation.
The environmental performance of seaweed-derived bioplastics hinges primarily on three factors: (1) energy use during drying and casting, (2) solvent and crosslinker choices, and (3) film yield per unit of dry biomass. Several studies reported that thermal drying alone can account for up to 50–70% of total GWP and CED. Replacing fossil-based crosslinkers (e.g., glutaraldehyde) with safer alternatives (e.g., genipin) and using renewable electricity for casting processes are strong levers for impact reduction. However, inconsistencies remain in how biodegradability and end-of-life are modeled, some studies assume full compostability, others landfill. Functional unit definition (e.g., 1 kg vs. 1 m2 film) and assumptions about solvent recovery efficiency strongly influence toxicity and energy-related results.
What matters for greenness: for seaweed-based fertilizers and biostimulants, environmental impacts are mostly driven by (1) solvent extraction methods, (2) processing energy (e.g., evaporation), and (3) transportation from farm to application site. Studies using water or ethanol as solvents report 20–40% lower GWP and toxicity compared to acid or alkali-based extraction routes. However, allocation methods (especially when fertilizer is a co-product from biorefineries) vary widely, affecting comparability. Another unresolved issue is the end-use modeling, whether benefits such as reduced synthetic fertilizer use or soil health improvements are credited in LCA. Transport distance and bulk density significantly influence outcomes, making moisture content and concentration strategies critical sensitivity levers.
Key levers for improving greenness in seaweed-based feed LCAs include (1) drying and pelletizing energy use, (2) farmed vs. wild biomass sourcing, and (3) functional substitution assumptions (i.e., replacing what feed?). Farmed seaweed generally shows lower impacts than wild-collected biomass due to more efficient logistics and less variability, though this depends on cultivation method (rope vs. tank). End-use assumptions matter greatly, whether seaweed replaces soy, fishmeal, or acts as a methane-reducing additive (e.g., Asparagopsis) can change outcomes dramatically. Few studies model land use change (LUC) savings or enteric methane reductions robustly, and this is an area needing clearer assumptions and harmonized methods.
Seaweed's application for construction materials (e.g., insulation, panels) is still emerging, with limited but promising LCA data. The top levers for greenness include (1) binder choice (bio-based vs. synthetic), (2) drying and processing energy, and (3) fire retardant or additive selection. Studies using lime or natural clay binders report lower impacts than epoxy- or resin-based systems. However, durability trade-offs and fire resistance remain technical gaps, often leading to conservative assumptions in LCA. The greatest sensitivity comes from functional unit choice (e.g., mass vs. thermal insulation delivered over time), which strongly affects results when comparing to mineral wool or fiberglass.
For blue carbon LCAs, the main sensitivities are (1) carbon sequestration assumptions (e.g., burial vs. degradation), (2) life span of stored carbon, and (3) system boundaries (farm-gate vs. full fate modeling). Studies estimating long-term sequestration from seaweed sinking to the ocean floor show wide variation in permanence assumptions (years to centuries), heavily influencing climate credits. The most critical unresolved issue is methodological standardization, some LCAs use net GHG balances with sequestration credits, while others exclude them entirely due to uncertainty. Sensitivity analyses consistently show that carbon accounting method and system boundary definitions are the largest levers affecting net climate benefit results.
4.2. Cross-cutting findings
The summary of LCA studies (Table 3) includes the applications, functional unit (FU), system boundary (i.e., cradle-to-gate, gate-to-gate, gate-to-grave, cradle-to-grave, and cradle-to-cradle), impact categories, and major environmental hotspots. Reviewed LCA studies on seaweed cultivation and product valorization reveal several key trends. First, most studies adopt a cradle-to-gate boundary for early-stage or intermediate product assessments, whereas cradle-to-grave boundaries are more commonly applied to biofuel and energy-related pathways.38,39,43 This distinction is important because it influences comparability across studies and determines whether downstream burdens, such as end-of-life management, are captured. Second, climate change (CC) and fossil depletion (FD) are the most frequently reported categories, though other midpoints such as eutrophication (EP), human toxicity (HT), and water depletion (WD) are inconsistently covered.40,46,49 These biases limit understanding of broader ecological trade-offs, suggesting the need for wider indicator coverage. Third, environmental hotspots are generally concentrated in upstream cultivation (e.g., diesel use, seeded ropes, hatchery inputs) and downstream processing (e.g., drying, extraction energy, solvent use), while fewer hotspots are reported in intermediate stages such as storage or packaging.31,32 Recognizing this distribution is critical, as it helps practitioners focus modeling efforts on stages with the greatest leverage for impact reduction. Additionally, several studies highlight the influence of methodological choices, including allocation rules, system boundaries, and functional unit definition, on results.41,42 For LCA practitioners, this underscores the need to clearly justify assumptions and perform sensitivity analyses to ensure robust conclusions. Finally, very few studies integrate social, economic, or prospective aspects, indicating an opportunity for more holistic sustainability assessments of seaweed-based biorefineries that move beyond environmental indicators alone. For example, a key study by Sadhukhan et al. (2019) evaluated an integrated biorefinery for the simultaneous extraction of protein, sugar, salt, nutrient, and mineral platforms. This work stands out by combining LCA and TEA with a Social Life Cycle Assessment (S-LCA), demonstrating that integrated marine systems can be economically superior to terrestrial lignocellulosic biorefineries due to lower production costs and higher-value co-products. Their findings highlight that protein, sugar-based chemicals, and inorganic products offer climate change impact savings of approximately 12, 3, and 1 kg CO2 eq. per kg of product, respectively. Furthermore, their S-LCA identified Indonesia, China, and the Philippines, which represent 93% of global seaweed production, as the regions with the highest potential for ‘avoided social impacts’ when seaweed-derived products displace animal-based proteins and minerals.39| Study | Study origin | Seaweed species | Functional unit | System boundary | Allocation method | Product | Impact categories | Hotspot | Green lever |
|---|---|---|---|---|---|---|---|---|---|
| 31 | Denmark | Brown | 1 kg bioplastic film | Cradle to grave | Mass-based allocation | Bioplastic film production | ReCiPe method, CC, OD, IR, POF, PM, AC, EU, EC, HT, RUMM, RUF, WU | Crosslinking chemicals (e.g., glutaraldehyde or glycerol) | Switch to genipin or enzymatic crosslinkers; lower-temperature crosslinking |
| 32 | Denmark | Brown | 1 kg bioplastic film | Cradle to grave | Mass-based allocation | Bio-based plastic | ReCiPe method, CC | Crosslinking and drying process | Use solar or heat-pump drying; improve film yield per kg of seaweed |
| 33 | Malaysia | Brown | 1 kg nanocomposite | Cradle to gate | No allocation | Nanocomposite | CC, AC, EC, OD, RUMM, RUF, HT, POF | Alginate extraction and beads transport | Optimize transport logistics; improve yield during extraction with greener solvents |
| 34 | USA | Brown | 1 kg extracted sodium alginate | Cradle to factory gate | No allocation | Sodium alginate extraction | CML baseline v2.05 method, RUF, AC, EU, CC, OD, HT, EC, POF | Diesel fuel for harvesting and electricity for extraction | Electrify logistics; improve alginate extraction efficiency |
| 18 | Norway | Brown | 1 kg bioplastic film | Cradle-to-grave | Mass-based allocation | Bioplastic (alginate-based transparent film) | ReCiPe 2016 midpoint (E), 18 impact categories | Glycerol (crosslinking) dominates CC; hatchery electricity; seaweed cultivation inputs; drying energy | Replace glycerol with greener crosslinker (e.g., citric acid); renewable electricity; improve drying efficiency |
| 19 | Norway | Brown | 1 kg alginate | Cradle-to-grave | Mass-based allocation | Bioplastic films, alginate-cellulose microfibers, PLA composites | ReCiPe 2016 midpoint (E) | Glycerol (film fabrication), NaHCO3 alkaline treatment, electricity in extraction, drying | Replace crosslinker (GC #3, #4); use green reagents; improve drying heat recovery |
| 35 | Norway | Brown | 1 kg bioplastic film | Cradle-to-grave | Mass-based allocation | Bioplastic film (alginate-based, bio-based & biodegradable) | ReCiPe 2016 midpoint (E), GWP, LU, FRS, WU, HCT, HNCT | Film fabrication (esp. Glycerol: 2.1 kg CO2-eq per kg film); co-product recirculation needs H2O2 & ethanol; EoL scenario (composting vs. incineration) | Replace glycerol with low-impact crosslinkers (e.g., citric acid); recover H2O2; prioritize composting in EoL scenarios |
| 36 | Caribbean (trinidad & tobago, Mayaro/Chamomile coast) | Brown | 1 kg Ca(Alg)2 bioplastic | Cradle-to-gate | Mass-based allocation | Calcium alginate composite bioplastic (Ca(Alg)2) | ReCiPe 2016 midpoint (H) | Chemicals: Na2CO3 (10–18%), H2SO4 (12–52%), NaOCl (15–22%); energy heating (61% GHG) | Use mild acid/base alternatives; recover energy via heat integration; avoid NaOCl by switching to H2O2 or filtration |
| 37 | UK | Brown | 1 kg packaging material | Cradle-to-gate | System expansion, mass allocation or economic allocation | Protein, fucoidan, laminarin, alginate–cellulose polymeric packaging material | ReCiPe 2016 midpoint (H), 18 impact categories | Filtration wastewater (up to 60% of GWP); HCl in acid extraction (45–97% in several categories); drying (esp. SD3); heating demand | Water reuse in filtration; replace HCl with organic acids; dry via low-temperature solar/heat pump |
| 38 | UK | Brown | 1 MJ of energy | Cradle to factory gate | Energy content-based allocation (based on biogas vs. bioethanol energy yields) | Bioethanol and biogas | CC, AC, EU, OD, POF, HT | Diesel fuel during transport and rope materials used in cultivation | Shift to low-impact mooring systems; electrify boats or switch to biodiesel |
| 39 | Denmark | Brown | 1 ton of dry seaweed biomass (Laminaria digitata) | Cradle to factory gate | No allocation | Biofuels | CC, AC, EU | Glycerol in film compounding and incineration of residues | Use greener plasticizers; valorize residues via composting or biochar |
| 40 | France | Green | Production and combustion of 1 MJ of usable energy in a passenger car engine | Cradle to grave | Energy content-based allocation | Bioethanol | CC, OD, HT, PM, IR, POF, EU, AC, LU, WU, RUMM, RUF | Enzyme production and electricity use in infrastructure | Improve enzyme efficiency; source electricity from renewables |
| 41 | Italy | Brown | Generation of 1.02 kWh of electricity and 10.92 MJ of heat, together with 1.86 kg of compost as a co-product from the biorefinery process | Cradle to grave | Mass based allocation | Biomethane production | HT, CC, EC, AC, EU, LU, RUMM | Collection and storage of poultry manure | Poultry manure improve pre-treatment and sanitation of co-substrates; integrate anaerobic digestion with renewable-powered thermal drying for digestate management |
| 42 | Ireland | Brown | 1 MJ of compressed biomethane | Cradle to gate | System expansion to account for digestate valorization as fertilizer substitute | Biomethane | AC, CC, EU | Digestate handling and field application, a dominant hotspot, causing >80% of eutrophication and acidification impacts | Improve nutrient recovery; optimize digestate reuse in agriculture |
| 43 | USA | Brown | 1-ton dry seaweed | Cradle to grave | Energy content-based allocation | Seaweed-derived bioethanol | The CML-IA method, RUMM, RUF, CC, OD, HT, EC, POF, AC, EU | Drying of seaweed biomass | Use low-moisture feedstock; integrate waste heat recovery for drying |
| 44 | Israel | Brown | 1 ton dried seaweed's product | Cradle to cradle | Not clearly stated, likely system expansion or no allocation since entire system energy is assessed cradle-to-cradle | Assessment of environmental sustainability in solar-based seaweed biorefinery configurations | ReCiPe method, EU, EC, RUMM, AC, WU, CC, OD, HT, POF, PM, IR, RUF | Total energy use in solar-biorefinery system | Increase solar capture efficiency; phase out fossil backup heating |
| 45 | South Korea | Brown | 1 kL of bioethanol | Cradle to gate | Not explicitly mentioned, assumed mass-based or energy-based allocation between bioethanol and residues | Biofuel production | ReCiPe method, CC | Bioethanol production | Optimize fermentation yields; recover process heat; integrate HTL where viable |
| 46 | France | Brown | 1 kg fresh weight seaweed | Cradle to farm gate | No allocation; biogenic carbon considered neutral and co-products excluded | Biofuel production | ReCiPe method, CC, RUMM, RUF, OD, HT, EC, POF, AC, EU | Hatchery infrastructure, and energy consumption | Electrify nursery operations; reduce infrastructure material use |
| 47 | Mexico (Caribbean coast, Yucatán) | Brown | Flow rate of 40 kg dry per h | Gate-to-gate (oxidative pretreatment process only) | Not applicable, as gate-to-gate study only addresses pretreatment with no downstream products | Pretreated biomass (oxidative delignification for biofuel/bioproduct valorization) | GREENSCOPE (30 environmental indicators), 5 areas: hazardous materials, human health, ecosystem (air, water), process sustainability (solids) | Energy demand (dryer ∼50 kWh); hydrogen peroxide use; residual solid stream | Replace H2O2 with greener oxidants (GC Principle #3); recover energy from solids (biochar/combustion) |
| 48 | Denmark | Brown | 1 ha offshore cultivation | Cradle to factory gate | System expansion used to credit protein and nutrient recovery functions | Energy, feed, and protein generation | ReCiPe method, CC, RUF, EU, HT | Substitution of chemical fertilizer and iron bars | Renewable nutrient recovery; switch to low-impact cultivation materials |
| 49 | Sweden | Red and Brown | 1 MJ product | Cradle to factory gate | No allocation, system boundary ends at pyrolysis product output with no co-product handling | Biofuel from pyrolysis of beach-cast seaweed, syngas | RUF, CC | High ash content in raw biomass reducing efficiency | Explore HTL over pyrolysis; recover energy; remove heavy metals before process |
| - Energy-intensive drying and pyrolysis steps | |||||||||
| - Heavy metal (e.g., cadmium) risk in residues/biochar | |||||||||
| 50 | Russia | Green | 1 Mg (1000 kg) of fuel produced | Gate-to-gate | No allocation mentioned | Biofuels (liquid bio-oil from pyrolysis & HTL) | ReCiPe 2016 midpoint (H), CC (GWP100), fossil depletion (FD), human toxicity (HTP), ozone depletion (ODP), terrestrial acidification (TA) | Pyrolysis: drying energy (natural gas); HTL: electricity & high-pressure heating; wastewater treatment burdens | Shift to renewable heat (biogas, solar); integrate HTL heat exchangers; valorize aqueous residue |
| 51 | Indonesia | Red | 1 kg ethanol | Gate-to-gate | Not clearly stated, but likely mass-based or economic allocation between ethanol and solid waste | Bioethanol (3rd-gen, from extraction solid waste) | CC, ADP, ADP fossil, ODP, HT, FET, MET, TET, POCP, AP, EP | Enzyme production (20%), fermentation (75%), electricity (5%) | Optimize enzyme and fermentation yield per unit product; switch to biobased enzyme sources |
| 52 | North Atlantic (case: Laminaria digitata, Maritimes, Canada) | Brown | 1 MJ energy (biofuel) and 1 MJ electricity | Cradle-to-gate | System expansion used to account for co-products (fertilizers, feed, protein) | Bioenergy carriers (biogas, bioethanol, combined) + waste valorization (fertilizers, feed, protein) | IMPACT World+ (midpoint & endpoint, weighted results in EUR2003) | CH4 leaks in anaerobic digestion; SHF distillation energy; combined burden | CH4 capture systems, heat recovery in SHF, multi-product valorization to share burdens |
| 53 | India | Red | 1 kL (1 m3) of Gracilaria seaweed extract | Cradle-to gate | No allocation | Environmental cost from Gracillariid seaweed extract, biostimulants | LU, CC, WU, EC, EU, HT, IR, RUMM, PM, OD, POF, AC | Plastic use for packaging and energy-intensive extrusion | Use biodegradable packaging; optimize process heat with renewable energy |
| 54 | India | Red | 1 kL (1 m3) of Kappaphycus alvarezii sap | Cradle to gate | No allocation | Biostimulant production | LU, CC, RUF, EC, WU, HT, IR, EU, PM, POF AC | Packaging plastic, extrusion, electricity demand | Use biodegradable packaging, solar-assisted extrusion, and cleaner energy grid |
| 55 | Denmark | Brown | 1 ton dry weight | Cradle to cradle | No allocation | Seaweed for circular nutrient management, fertilizer | ReCiPe method, CML, EU | Plastic ropes and nutrient bio-extraction | Replace ropes with biodegradable alternatives; recover excess nutrients |
| 56 | India | Red | 1 ton rice | Cradle to rice production | Substitution approach; avoided burden method using CO2-equivalent of conventional rice inputs | Seaweed as biofertilizer | ReCiPe method, CC, EC, WU, EU, RUF, HT, IR, PM, OD, POF, AC | CO2-equivalent emissions from conventional rice inputs | Broad adoption of seaweed fertilizer; reduce methane by improved field drainage |
| 57 | India | Red | 1 ton cane production | Cradle-to-cane production | Substitution/avoided burden; CO2-equivalent from synthetic nitrogen avoided | Biostimulants for sugarcane production | ReCiPe method, CC, EU, LU, WU | CO2-equivalent from cane inputs | Promote foliar biostimulant application; replace synthetic N |
| 58 | India | Red | 1 ton of sugar cane | Cradle to cane production | Substitution/avoided burden; avoided impact from conventional sugarcane system | Use as fertilizer | ReCiPe method, LU, CC, RUF, EC, HT, IR, EU, RUMM, PM, OD, POF, AC, WU | High GHG from conventional sugarcane systems | Support full substitution with seaweed-derived nutrients |
| 59 | Spain | Red | 1 batch process (varies by extraction method, e.g., 175–338 kg extract per batch) | Cradle-to-gate | Mass-based allocation (likely, based on batch extract yield distribution) | Biostimulant extracts (for wheat and other crops) | ReCiPe 2016 midpoint (H), 18 impact categories | Electricity and steam use (scenarios 1 & 2); CO2 compression (scenario 3) | Use heat recovery in extraction; reduce CO2 compression needs; green solvent alternatives |
| 60 | Denmark | Brown | 1 square meter of timber wall with 376.5 mm seaweed insulation, evaluated over a 50-year lifetime | Cradle to grave | No allocation | LCA of biobased construction materials | ReCiPe method, EU, CC, LU, RUF, EC | Steel infrastructure in wall panel insulation | Replace steel with bamboo or recycled wood; optimize insulation-to-weight ratio |
| 61 | Brazil | Brown | 1 m3 lightweight ceramic aggregate | Cradle-to-gate (clay/sargassum acquisition, prep, sintering; excludes distribution, use, disposal) | No allocation | Construction material (lightweight ceramic clay aggregates with sargassum particles) | ReCiPe 2016 midpoint, 18 impact categories | Electricity for sintering (esp. Conventional ovens), clay extraction, transport of sargassum, land use for drying | Use solar sintering or heat recovery; optimize sargassum transport and drying via local sourcing |
| 62 | Sweden | Brown | 1 kg protein ingredient | Cradle to gate | Mass-based allocation | Protein extraction from herring side streams using antioxidant-enriched co-feedstocks | CC, RUF, LU, RUM | Centrifugation and grinding (high energy use) | Use low-energy dehydration or passive separation techniques; valorize grinding heat |
| 63 | Norway | Brown | 1 ton seaweed protein concentrate | Cradle to gate | System expansion/substitution (compared with conventional fish feed systems) | Fish feed | CC | Drying process | Use solar or low-energy drying (e.g., heat pump); harvest at lower moisture levels |
| 64 | Norway | Brown | 1 kg protein | Cradle to gate | Economic allocation among extracted protein fractions | LCA of protein production | CML 2016, CC, RUF, OD, AC, EU, POF, RUMM | Energy use across processing stages | Switch to renewable energy sources and improve process efficiencies |
| 65 | Ireland | Brown | 1 ton seaweed | Cradle to factory gate | Mixed method (mass-based and economic allocation across full seaweed biorefinery value chain) | Seaweed value chain | ReCiPe method, CC, OD, IR, POF, PM, HT, AC, EC, EU, LU, WU, RUF, RUMM | Fuel consumption, drying, and inoculum tank | Optimize inoculum re-use; low-impact transportation; solar drying |
| 64 | Norway | Brown | 1 kg crude protein | Cradle-to-gate | System expansion (compared SPP from seaweed vs. soy protein concentrate) | Seaweed protein product (SPP) vs. soy protein concentrate (SPC) | CML 2016 midpoint | Drying energy (>60% GWP with fossil gas); protein extraction rate; farm infrastructure lifetime | Improve drying efficiency; recover latent heat; enhance protein yield per biomass; extend infrastructure life via modular design |
| 66 | Spain | Red | 1 kg cosmetic product | Cradle to factory gate | No allocation | Seaweed for cosmetics | ReCiPe method, CC, RUF, RUMM, WU, EU | Drying process | Use waste heat from other processes; explore low-temperature dehydration |
| 67 | UK | Brown | 1 ton single cell oil | Cradle to factory gate | No allocation | Single cell oil production | Recipe method, CC, EU, HT, EC, AC, WU | Fermentation and acid pretreatment, and enzymatic hydrolysis | Reduce enzyme input via immobilization or reuse; integrate renewable heat |
| 68 | Netherland | Brown | 1 kg salt replacement, processed and packed at supermarket | Cradle to grave | Mass allocation | Use as food ingredients, 100% salt replacement | CC, EU, RUF, WU, LU | Seaweed cultivation (transportation) | Use electric transport options; optimize harvest location vs. demand centers |
| 69 | Indonesia | Red | 1 kg carrageenan flour | Cradle to factory gate | No allocation | Seaweed in carrageenan flour products | CML-IA baseline version 3.06., CC | Carrageenan flour production phase (the use of coal as boiler fuel, potassium hydroxide, and solid waste) | Switch to biomass/solar for steam; reduce chemical inputs via green chemistry pretreatment |
| 70 | Spain, Estonia, Latvia | Red | Annual catch (464.5 t seaweed) and 1 batch of extraction | Cradle-to-gate (harvesting, transport, extraction, purification up to factory gate) | Economic allocation between pigment (R-phycoerythrin) and residual biomass or alternative extraction outcomes | Pigment (R-phycoerythrin) | ReCiPe 2016 midpoint (H) & endpoint, CC, AC (TA), FE, ME, TET, FET, MET, HCT, HNCT, FRS, SOD | Electricity & steam demand in extraction; freeze-drying; sodium acetate buffer; marine diesel for trawler | Switch to solar-assisted extraction/drying; use low-toxicity buffer alternatives; optimize trawler logistics |
| 69 | Indonesia | Red | 1 kg carrageenan flour | Cradle to factory gate | No allocation | Seaweed in carrageenan flour products | CML-IA baseline version 3.06., CC | carrageenan flour production phase (the use of coal as boiler fuel, potassium hydroxide, and solid waste) | Switch to biomass/solar for steam; reduce chemical inputs via green chemistry pretreatment |
| 71 | China | Red | 1 kg agar product | Cradle-to-gate | No allocation | Hydrocolloid (agar) | ReCiPe 2016 midpoint (H) + EP&L monetization | Energy & chemicals in agar extraction; activated carbon use | Use green solvents (GC #5); improve efficiency of activated carbon use; valorize side streams |
| 72 | Germany | Red | 1 kg fat- and protein-corrected milk (FPCM) | Cradle-to-farm gate | Mass allocation between milk and co-products (e.g., manure, based on nutrient content) | Dairy milk with mitigation measures (seaweed feed, cow toilet, slurry acidification | CML-IA baseline, CC, EP, AP, AD | Enteric CH4 emissions, manure NH3 emissions, feed-related impacts | Methane-reducing feed additives (e.g., seaweed); improved manure handling; precision feeding strategies |
| 73 | Sweden | Red | 1 kg dry seaweed | Cradle to factory gate | No allocation | Environmental impact of seaweed production | CC, AC, EC, EU, HT, RUMM, WU, RUF, OD, POF, IR | Salt use in inoculum tank stage | Substitute or reduce salt input; integrate salt recycling systems |
| 74 | Denmark | Brown | 1 ha sea surface | Cradle to grave | System expansion (bio-extraction and fuel substitution included) | Offshore seaweed production for biorefinery | ReCiPe method, CC, RUF, EU, HT | Iron bars for cultivation, N & P bio-extraction, substituted gasoline | Use recycled/reused steel; optimize bio-extraction rate; replace gasoline with electricity |
| 75 | Denmark | Brown | 1 ton dry seaweed (annual production and processing) | Cradle-to-seaweed drying | No allocation | Dry seaweed biomass | ReCiPe method, CC | Drying, fermentation, liquid fertilizer spreading | Use solar/waste-heat drying; reduce nutrient leaching in fertilizer spreading |
| 76 | Sweden | Brown | 1 ton of fresh kelp | Cradle to grave | No allocation | Comparing methods to preserve seaweed | CML 2 baseline 2000, CC, RUMM, AC, EU, OD, HT, EC, POF, RUF | Equipment production (hatchery, cultivation), the preservation and storage of harvested seaweed, which requires energy for refrigeration and transportation | Reuse/recycle infrastructure; improve preservation with low-temp or solar refrigeration |
| 77 | Sweden | Brown | 1 kg fresh seaweed | Cradle to farm gate | No allocation | Nutrient uptake potential | CML, CC, EU, RUF | Transport, steel infrastructure, and of polyethylene or polypropylene components | Optimize site location to reduce transport; replace plastics with biodegradable options |
| 78 | Belgium | Brown | 1 kg fresh weight seaweed | Cradle to farm gate | No allocation | Seaweed production | ReCiPe method, CC, EU | Cultivation infrastructure and energy use | Use renewable energy for pumping/lighting; switch to bio-based ropes and anchors |
| 79 | Netherland | Brown | 1 ton protein content | Cradle-to-seaweed drying | Mass-based allocation | Sustainable cultivation system for seaweed production | CML 2001 and cumulative energy demand, RUMM, AC, EU, CC, OD, HT, EC, RUF | Drying, and infrastructure (production of the chromium steel chains and polypropylene rope, and infrastructure, and drying for toxicity related impacts | Avoid toxic materials (e.g., chromium steel); use waste heat for drying |
| 80 | Denmark | Brown | 1 ha cultivation area | Cradle-to-gate | No allocation | Seaweed as bio-extractor | ReCiPe method, CC, EU | Process heating and product drying | Use low-energy heating and drying (solar, waste heat recovery) |
| 81 | Denmark | Red and Brown | 1 kg dry matter feedstock processed | Gate to gate | No allocation | Techno-economic and environmental assessment of biorefinery | CC, WU, RUMM | Energy use for heating and drying | Electrify biorefinery with renewables; optimize moisture content before drying |
| 82 | China (Dalian, Liaoning) | Brown | 1 ton wet seaweed | Cradle-to-shore | No allocation | Seaweed production | ReCiPe 2016 midpoint (H), 18 impact categories | Diesel combustion for marine transport/harvest (∼50% CC); polyethylene lines (∼28–33%); buoys (∼9–12%) | Electrify harvesting; shift to biodegradable/biocomposite ropes and buoys |
| 83 | Europe (France, Ireland, Denmark, Sweden) | Brown | 1-ton fresh seaweed | Cradle-to-farm gate | No allocation | Seaweed production | ReCiPe 2016 midpoint, CC (kg CO2-eq), FE (kg P eq), ME (kg N eq) | Plastics (ropes, buoys), metals (anchors, chains), electricity in hatchery; boat fuel (diesel vs. tkm modelled) | Replace fossil-derived plastics; integrate renewable electricity; streamline anchor systems |
| 84 | Ireland | Brown | 1 kg fresh seaweed | Cradle-to-gate | No allocation | Seaweed production | IPCC GWP100, CEENE/CExD (exergy), GWP (kg CO2-eq) | Wild harvesting: boat fuel (mechanical), cultivation: capital equipment, boat fuel, hatchery energy | Electrify boats; switch to passive intake hatcheries; minimize capital intensity |
| 85 | Baltic sea region (Estonia, Iceland, literature-based Denmark) | Red | 1 ton fresh seaweed | Cradle-to-gate | No allocation | Seaweed production | Environmental footprint (EF) 3.0, CC, PM, AP, EF (freshwater/marine), ECF, LU, WU, RF, RM&M | Wild harvest (WH): diesel in boats. On-shore cultivation (ONC): electricity for artificial lighting. Off-shore cultivation (OFC): ropes, buoys, boat fuel | Renewable electricity; bio-based cultivation infrastructure; hybrid/methanol boat systems |
| 86 | Global | Brown, green, red | 1 kg dry macroalgae | Cradle-to-grave | Mass allocation | Integrated (protein, FDCA, Lactic/Succinic acid, salts, nutrients) | CC (GWP), FD, HT, WD (Aquatic ecotoxicity), social impacts | Energy intensive upstream process | External fossil energy for drying/pretreatment |
5. Broader impacts, future direction and opportunities
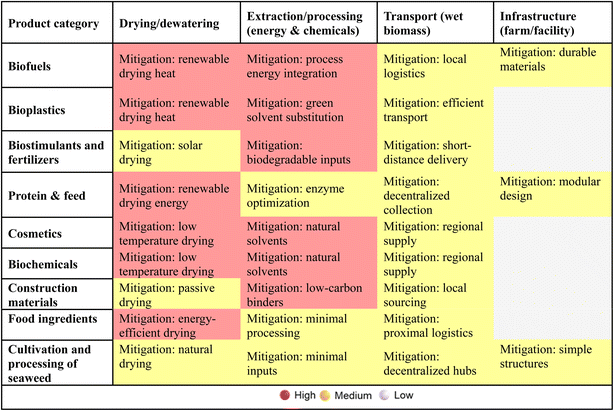
In order to support the transition toward more sustainable and circular seaweed-based biorefineries, this review synthesizes common environmental hotspots and highlights effective mitigation strategies. A critical cross-comparison across product categories reveals that drying and extraction phases are the most frequent contributors to environmental burdens. Transport and infrastructure also play a role, especially in large-scale or offshore systems. The following tables summarize these findings and provide a forward-looking roadmap to guide innovation and policy.Table 4 presents a heatmap of environmental hotspot intensity across nine major product categories, along with the most effective “green levers” observed across studies. Table 5 outlines an actionable roadmap, organized by time horizon, linking each intervention to relevant Green Chemistry Principles. Together, these tools offer a strategic framework for researchers, technologists, and policymakers aiming to align seaweed valorization with climate, circularity, and sustainability goals.
| Time horizon | Priority area | Recommended action/strategy | Linked principle(s) | Sustainability focus |
|---|---|---|---|---|
| a P1-waste prevention, P2-atom economy, P3-less hazardous synthesis, P4-designing safer chemicals P5-safer solvents & auxiliaries, P6-energy efficiency, P7-use of renewable feedstocks, P8-reduce derivatives, P9-use of catalysis, P10-design for degradation, P11-real-time analysis, P12-safer systems design. | ||||
| Near-term (0–2 years) | Drying | Switch to renewable heat sources (biomass, solar, industrial waste heat); apply heat integration where possible | P6 (energy efficiency), P7 (renewables) | GHG reduction, process energy efficiency |
| Crosslinking & extraction | Adopt greener solvents/crosslinkers (e.g., citric acid, ionic liquids); minimize glycerol and toxic agents | P3 (less Hazardous synthesis), P5 (safer solvents & auxiliaries) | Reduced toxicity and eutrophication impacts | |
| Decentralized processing | Promote on-site dewatering or preprocessing at seaweed farms to minimize transport of water-heavy biomass | P1 (Prevent waste), P2 (Atom economy) | Water-energy nexus, reduced logistics impact | |
| End-of-life planning | Ensure design for compostability, especially for bioplastics; assess realistic EoL fate (e.g., incineration vs. biodegradation) | P10 (design for degradation) | Reduced landfill burden, enhanced circularity | |
| Mid-term (2–5 years) | Modeling standards | Develop standardized FU templates and harmonized system boundaries for biorefineries across LCA studies | P11 (real-time analysis), P12 (inherently safer design) | Inter-study comparability, database improvement |
| Indicator set | Move beyond GWP; include acidification, eutrophication, land use, and toxicity indicators as standard | P4 (designing safer chemicals), P12 (safer systems) | More holistic environmental accounting | |
| Biogenic carbon handling | Integrate time-explicit biogenic carbon modeling with clear assumptions (e.g., temporary sequestration, re-release) | P6 (energy efficiency), P12 (safer systems) | Accuracy in carbon-neutrality claims | |
| Long-term (5–10 years) | LCA-TEA integration | Simultaneously evaluate economic and environmental trade-offs (e.g., low GWP vs. high cost of green solvents) | P9 (Catalysis), P12 (system design) | Decision support for commercialization |
| Residue cascades | Incorporate multi-output valorization (e.g., protein + biochar + fertilizer) using cascading LCA approaches | P1 (waste Prevention), P2 (Max Atom economy) | Maximizing resource recovery | |
| Regional siting & logistics | Use spatial LCA models to assess optimal farm and facility locations based on resource availability and impacts | P7 (renewables), P6 (energy efficiency) | Reduced transport footprint, improved siting efficiency | |
5.1. Sustainability design levers: insights from green chemistry and SDG alignment
The process hotspots and mitigation levers identified across the reviewed LCA studies align with several design strategies rooted in Green Chemistry and the UN (SDGs). For example, replacing toxic organic solvents with ethanol or water-based alternatives (aligned with Principle 5: safer solvents) showed up to 70% reduction in toxicity-related impacts.32,77 Energy-intensive drying and extraction stages, identified as dominant hotspots, can be addressed by shifting to renewable-powered, thermal systems and decentralized pre-processing units, consistent with Principle 6: energy efficiency and SDG 7 (affordable and clean energy).43Moreover, the use of biodegradable materials and co-product valorization directly supports Principle 10: design for degradation and SDG 12 (responsible consumption & production). In several bioplastic and biostimulant case studies, implementing crosslinkers derived from bio-based inputs reduced global warming potential by 30–60%. These interventions demonstrate the critical role of early design decisions in improving sustainability performance, particularly when supported by consistent LCA data and regional energy profiles.35
By embedding these principles into upstream decisions, such as solvent choice, energy source, and end-of-life planning, seaweed-based biorefineries can significantly lower their environmental footprint while delivering on multiple SDG targets including SDG 2 (Zero Hunger), SDG 13 (Climate Action), and SDG 14 (Life Below Water).
5.2. Climate impact: integrating policy relevance and LCA perspectives
Seaweed cultivation and valorization have gained increasing attention as nature-based solutions for mitigating climate change. This is primarily due to their dual capacity for carbon sequestration during growth and displacement of fossil-derived products in various applications, refer Fig. 6. | ||
| Fig. 6 Schematic of integrated seaweed farming and valorization pathways. Offshore seaweed cultivation can contribute to carbon sequestration through detritus burial in seafloor sediments and, in some cases, export to the deep sea. Cultivation can also support nearby blue-carbon habitats such as seagrass beds. During processing, seaweed biomass yields multiple products, including hydrocolloids (e.g., alginate), proteins, lipids, bio-crude, and aqueous fractions. Valorized uses range from biofuels (ethanol, methane) to biostimulants that improve soil health, and feed additives that can reduce cattle methane emissions. These products can substitute fossil-based alternatives, lowering emissions across sectors. Adopted from ref. 161 (Jones et al., BioScience, 2022), licensed under CC BY 4.0. | ||
Although seaweed is widely promoted in policy and research narratives as a sustainable resource, it is essential to rigorously assess and quantify these benefits through LCA. Several studies reviewed in this paper (e.g., ref. 42, 43, 31, and 58) report notable reductions in climate change impact when seaweed-based fuels, bioplastics, and biofertilizers are compared to fossil-based or conventional alternatives. These benefits are often more pronounced when: (1) renewable energy is used in cultivation or drying stages, (2) residues are valorized as co-products (e.g., biofertilizers, feed).
Substitution scenarios modeled in various studies demonstrate up to 60–80% of the GHG reduction compared to base scenario,31 but these results vary significantly depending on assumptions related to: system boundaries (cradle-to-gate vs. cradle-to-grave), allocation approaches (economic, mass-based, substitution), biogenic carbon accounting, and energy source used during processing. This variability is important because it shows that reported climate benefits of seaweed biorefineries are highly sensitive to methodological choices, making transparency and harmonization in LCA critical for producing results that can reliably inform policy and industrial decision-making.
Although many studies show that seaweed biorefineries can help reduce greenhouse gas emissions, there are still important gaps in how these benefits are measured in LCA. For example, most studies do not clearly show how carbon captured by seaweed is stored in the long term, such as in soils, sediments, or durable bioproducts. Also, methods like modeling how carbon emissions change over time or vary by location, commonly used in land-based biomass LCA, are rarely applied to marine systems. Because of this, results from different studies can be hard to compare, and the true climate benefits of seaweed might be underestimated.
To improve future LCAs, researchers should consider tracking carbon overtime (e.g., how long it stays stored) and modeling the products replaced by seaweed (e.g., fossil-based plastics or synthetic fertilizers) under what real-world conditions. Adding these elements would make the climate impact assessments of seaweed more reliable and would also provide evidence for policymakers considering blue carbon strategies. However, it is important to note that product substitution credits are not commonly recognized in carbon credit markets due to challenges in verifying counterfactual baselines; instead, such benefits are typically addressed through LCA studies or national greenhouse gas. Fig. 7 illustrates the multifaceted ecosystem services of seaweed cultivation and their alignment with various SDGs, highlighting the environmental, social, and economic co-benefits that warrant future inclusion in LCA frameworks.
5.3. Industrial/technology advancement
The development of seaweed biorefinery technologies is driving innovation at the forefront of the global bioeconomy. These technologies enable the conversion of seaweed into a diverse portfolio of bio-based products, including food ingredients, pharmaceuticals, cosmetics, biofuels, bioplastics, and biostimulants.162 Evidence from recent LCA studies reviewed in Section 4 shows that industrial and technological advances will be most impactful in addressing recurring hotspots such as energy-intensive drying, chemical crosslinking, and electricity-dependent processing stages. For example, bioplastic LCAs highlight crosslinking as a dominant burden,32 while biofuel pathways consistently identify drying as the main contributor to climate impacts.43 Similarly, biostimulant production studies reveal that electricity use during extraction remains a critical challenge.53,54 Addressing these bottlenecks through innovations in processing methods and integrated valorization strategies could significantly improve energy efficiency and product yield while reducing waste and emissions.163 This growing industrial relevance is drawing attention from policymakers, investors, and entrepreneurs. The combined economic and environmental potential of seaweed-based technologies positions them as key enablers of the UN Sustainable Development Goals, particularly SDG 9 (Industry, Innovation, and Infrastructure) and SDG 12 (Responsible Consumption and Production).164,165 Continued investment in seaweed innovation ecosystems is expected to accelerate the transition toward a more circular, inclusive, and low-carbon industrial landscape.5.4. Circular bioeconomy
A circular economy aims to maximize the value extracted from natural resources while minimizing waste and environmental impact.166 In the context of seaweed, this means leveraging its renewable nature to create value-added products while ensuring that waste materials are minimized, reused, or recycled. Fig. 8 illustrates how seaweed biorefineries connect three functions: a bio-based economy (primary products such as polysaccharides, proteins, lipids, biofuels, and bioplastics), a circular economy (reuse, recycling, and cascading of residues), and a green economy (ecosystem services such as carbon uptake and nutrient removal). Our review shows these benefits are pathway-specific: cultivation often provides measurable ecosystem services, yet many LCAs report that energy and chemical-intensive processing steps (e.g., drying, extraction, and crosslinking) and long-distance transport can significantly reduce or erase net gains (see Sections 4.2–4.4 and Table 3). In contrast, integrated biorefinery approaches that valorize multiple fractions and route residues to anaerobic digestion, soil amendments, or feed generally show better environmental and economic outcomes because of co-product credits and internal energy recovery.87 To realize the promise shown in Fig. 8, research and policy should prioritize integrated processing, reductions in processing energy and chemical use, and regionally optimized supply chains. This will reduce waste and help seaweed systems truly benefit the environment.5.5. Challenges
Building on the synthesis of LCA studies presented in Section 4.1 and Table 3, several systemic and technical challenges still hinder the commercial deployment of seaweed biorefineries. These are discussed as follows.Future research must prioritize integrated LCA-TEA studies that utilize industrial-scale data to provide decision support for investors and policymakers. Evaluation of “sleeping giant” chemicals like FDCA should be a priority given their high market value compared to bioethanol.
Future studies should investigate decentralized preprocessing hubs at the farm site to perform partial dewatering. Implementing solar-assisted drying or heat integration can reduce GWP by up to 25%.
Future work will help to developing cascading extraction protocols to remove salts and minerals before energy conversion can turn these impurities into secondary revenue streams.
Future work can include large-scale aquaculture expansion, and the development of low-energy preservation methods (e.g., ensiling) are required to ensure a stable, year-round feedstock supply.
A key methodological challenge observed across reviewed studies involves the treatment of biogenic carbon within cradle-to-gate boundaries. While seaweed cultivation systems sequester atmospheric CO2via photosynthesis, most studies either assume carbon neutrality at the gate or omit downstream emissions, potentially overestimating climate benefits if the biomass is rapidly combusted or decomposed. This is particularly problematic when biogenic CO2 uptake is accounted for as a negative flow (−1), but end-of-life emissions (+1) occur faster than biomass regeneration, creating a temporal mismatch.20 Future LCA studies of seaweed-based biorefineries should clearly state carbon accounting conventions, adopt consistent system boundaries, and where possible, implement time-differentiated modeling approaches such as dynamic LCA or GWP to better capture the climate relevance of short-lived biogenic carbon flows. Also, future frameworks should adopt “Triple Bottom Line” sustainability assessments, integrating LCA, TEA, and Social LCA, to quantify holistic benefits like “avoided social impacts” from displacing fossil-derived products.
Overall, this review highlights that seaweed biorefineries hold strong potential to contribute to the circular bioeconomy by providing low-carbon fuels, materials, and agricultural inputs. While LCA studies consistently identify hotspots such as drying, extraction, and transport, they also demonstrate opportunities through co-product valorization, nutrient recycling, and substitution of fossil-based products. Future research should prioritize industrial-scale validation, integration of techno-economic and social dimensions, and broader coverage of impact categories to establish seaweed biorefineries as robust pathways for sustainable development.
5.6. Guidelines for greener seaweed biorefineries
Across the reviewed literature, certain product pathways (e.g., biostimulants, fertilizers) and process configurations (e.g., solar-assisted drying, mechanical dewatering) consistently demonstrate more favorable environmental profiles. Biostimulants and fertilizers derived from minimally processed seaweed biomass tend to yield the lowest environmental burdens, primarily due to the avoidance of energy-intensive drying, solvent use, or chemical extraction. In contrast, biofuels and bioplastics typically exhibit higher impacts due to extensive downstream processing, though their net climate benefits may improve when paired with renewable energy sources and green solvents.From a technological standpoint, modular and decentralized biorefineries co-located with cultivation sites can significantly reduce transportation energy and water–energy trade-offs. Key enabling technologies include mechanical dewatering, low-temperature drying, and non-toxic extraction solvents. Among biorefinery integration strategies, multi-product cascading systems that extract both high-value (e.g., protein, pigments) and low-value bulk products (e.g., alginate, biochar) from the same biomass stream offer promising routes toward resource efficiency and climate mitigation. For example, combinations such as biostimulants and fertilizer, alginate and biofuel, protein, pigments, and bioplastics deserve further LCA-informed exploration.
In order to advance the field, future studies should prioritize multi-product biorefineries under harmonized system boundaries and functional units, ideally supported by scenario analysis. Ultimately, the most environmentally beneficial designs will strike a balance between ecological sustainability, economic feasibility, and circularity, tailored to coastal geographies where seaweed thrives.
Conflicts of interest
The authors declare no conflicts of interest.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The authors acknowledge the support from the Environmental Defense Fund and funding from the Cornell Atkinson Center for Sustainability.References
- J. DeAngelo, B. T. Saenz, I. B. Arzeno-Soltero, C. A. Frieder, M. C. Long, J. Hamman, K. A. Davis and S. J. Davis, Nat. Plants, 2023, 9, 45–57 CrossRef PubMed.
- T. Rose Daphnee, O. Helen and N. Hugue, Sci. Total Environ., 2024, 920, 171047 CrossRef PubMed.
- S. V. Mohan, G. N. Nikhil, P. Chiranjeevi, C. N. Reddy, M. Rohit, A. N. Kumar and O. Sarkar, Bioresour. Technol., 2016, 215, 2–12 CrossRef PubMed.
- T. K. Emily, M. M. A. Jessica, A. R. Héctor, D.-J. Gabriela and J. L. Gary, Renewable Sustainable Energy Rev., 2021, 151, 111553 CrossRef.
- A. Sanz-Hernández, E. Esteban and P. Garrido, J. Cleaner Prod., 2019, 224, 107–119 CrossRef.
- R. Nicolas, G. Jacopo, A. Rita, A. Marios, B. Elisabetta, I. B. José, B. Bettina, B. William, B. Maria Teresa, B. Claudia, C. Andrea, F. Gianluca, F. Marco, G. Patricia, L. Alessandra, L. Maria, M. Luisa, M. B. Robert, P. Claudia, P. George, R. Tévécia, S. Serenella, S. Javier and M. Sarah, New Biotechnol., 2020, 59, 10–19 CrossRef PubMed.
- B. T. Office, National Biotechnology and Biomanufacturing Initiative Will Advance the Department of Energy Bioenergy Technologies Office's Mission, https://www.energy.gov/eere/bioenergy/articles/national-biotechnology-and-biomanufacturing-initiative-will-advance, accessed 19th Sept, 2025.
- R. Raghu, S. Aswathy, V. K. Naveen, M. Suresh and N. Prema, iScience, 2025, 28, 112157 CrossRef PubMed.
- UN General Assembly, Transforming our world : the 2030 Agenda for Sustainable Development, A/RES/70/1, 2015, https://www.refworld.org/legal/resolution/unga/2015/en/111816, [accessed 23 January 2026] Search PubMed.
- T. Heimann, Earth's Future, 2019, 7, 43–57 CrossRef.
- P. T. Anastas, J. C. Warner, P. T. Anastas and J. C. Warner, in Green Chemistry: Theory and Practice, Oxford University Press, 2000, DOI:10.1093/oso/9780198506980.003.0004.
- A. Tursi, Biofuel Res. J., 2019, 6, 962–979 CrossRef CAS.
- N. Khandelwal, S. Kumari, S. Poduval, S. K. Behera, A. Kumar and V. V. Gedam, Sci. Rep., 2025, 15, 13135 CrossRef CAS PubMed.
- J. Castilla-Archilla, V. O'Flaherty and P. N. L. Lens, in Biorefinery: Integrated Sustainable Processes for Biomass Conversion to Biomaterials, Biofuels, and Fertilizers, ed. J.-R. Bastidas-Oyanedel and J. E. Schmidt, Springer International Publishing, Cham, 2019, pp. 3–35, DOI:10.1007/978-3-030-10961-5_1.
- C. D. Pinales-Márquez, R. M. Rodríguez-Jasso, R. G. Araújo, A. Loredo-Trevino, D. Nabarlatz, B. Gullón and H. A. Ruiz, Ind. Crops Prod., 2021, 162, 113274 CrossRef.
- W. Zhong, H. Li and Y. Wang, BioDesign Res., 2023, 5, 0021 CrossRef CAS PubMed.
- K. Choon Gek, D. Yaleeni Kanna, L. Man Kee and L. Keat Teong, Bioresour. Technol., 2019, 292, 121964 CrossRef PubMed.
- M. Ayala, Ø. Arlov, K. Nøkling-Eide, M. Sœther, C. Dore, J. Vidal, Q. Zhou, S. Wang, L. Michalak, A. Kyvik, B. Jolain, L. Aubel, S. Strand Jacobsen and M. Pizzol, J. Cleaner Prod., 2024, 444, 141248 CrossRef CAS.
- M. Ayala, N. Goosen, L. Michalak, M. Thomsen and M. Pizzol, Sustain. Prod. Consum., 2024, 52, 416–426 Search PubMed.
- S. Füchsl, J. Huber, M. Fröhling and H. Röder, Int. J. Life Cycle Assess., 2025, 30, 2300–2313 Search PubMed.
- F. Rod, A. Simona, B. Jennifer, B. Poppy, H. B. Alejandro, C. Max, C. Jamie, A. D. Kristen, G. John Barry, G. Rebecca, L. G. Rebecca, K. Kristin, M. Monica, P. Nichole, R. Loretta, T. John and Y. Charles, Mar. Pol., 2023, 155, 105747 CrossRef.
- A. J. Kyung, L. Seong-Rin, K. Yoori and P. Jong Moon, Bioresour. Technol., 2013, 135, 182–190 CrossRef PubMed.
- C. A. Pfister, Ecology, 1995, 76, 2341–2341 CrossRef.
- W. Muhammad Ahmed, H. Fatemeh, M. Lisbeth and K. Marie Trydeman, Sustain. Prod. Consum., 2024, 48, 123–142 CrossRef.
- C. D. Bullen, J. Driscoll, J. Burt, T. Stephens, M. Hessing-Lewis and E. J. Gregr, Sci. Rep., 2024, 14, 15021 CrossRef CAS PubMed.
- W. R. R. Finnley, W. B. Philip, F.-D. Karen, W. Kenta, O. Alejandra, K.-J. Dorte, L. Catherine, F. A. S. Calvyn, T. B. Lennart, M. D. Carlos, S. Oscar, B. John, T. Patrick and I. M. Peter, Sci. Total Environ., 2023, 885, 163699 CrossRef PubMed.
- S. Michele, H. Xiaoru, B. Simone, B. Anne-Belinda and T. Marianne, J. Cleaner Prod., 2016, 137, 1158–1169 CrossRef.
- D. Manikandan and P. Anuchit, Algal Res., 2025, 90, 104136 CrossRef.
- V. V. Durga, V. Veeraprakasam, S. Sivaraj, M. Meivelu, S. Ramachandran, K. Prabhu and I. Kapilkumar Nivrutti, Total Environ. Res. Themes, 2022, 3–4, 100016 Search PubMed.
- T. Virginia Martin, F. Sara, K. R. Uttam, O. Uche and a. Lorna Anguilano, J. Appl. Phycol., 2025, 6, 52–73 CrossRef.
- M. Ayala, M. Thomsen and M. Pizzol, Algal Res., 2023, 76, 103313 CrossRef.
- M. Ayala, M. Pizzol and M. Thomsen, Life cycle assessment of brown-seaweed-based plastic, in SETAC Copenhagen–SETAC Europe 32nd Annual Meeting: Towards a Reduced Pollution Society, 2022 Search PubMed.
- C. S. C. Chiew, W. Gourich, P. Pasbakhsh, P. E. Poh, B. T. Tey, C. P. Song and E.-S. Chan, J. Water Proc. Eng., 2022, 45, 102531 CrossRef.
- P. V. Rey, PhD Thesis, Universidade de Santiago de Compostela, 2015.
- M. Ayala, M. Thomsen and M. Pizzol, Algal Res., 2023, 71, 103036 CrossRef.
- A. Mohammed, K. Ward, K.-Y. Lee and V. Dupont, Green Chem., 2023, 25, 5501–5516 RSC.
- L. Amponsah, C. Chuck and S. Parsons, Int. J. Life Cycle Assess., 2023, 29, 174–191 CrossRef.
- D. Aitken, C. Bulboa, A. Godoy-Faundez, J. L. Turrion-Gomez and B. Antizar-Ladislao, J. Cleaner Prod., 2014, 75, 45–56 CrossRef CAS.
- M. Alvarado-Morales, A. Boldrin, D. B. Karakashev, S. L. Holdt, I. Angelidaki and T. Astrup, Bioresour. Technol., 2013, 129, 92–99 Search PubMed.
- D. Brockmann, C. Pradinaud, J. Champenois, M. Benoit and A. Hélias, Biofuel Bioprod. Biorefining, 2015, 9, 696–708 CrossRef CAS.
- A. Cappelli, G. Emanuele, R. Francesco, S. Silvano, B. Dagnija, P. Massimiliano and G. Elisa, Energy Procedia, 2015, 72, 3–10 CrossRef.
- M. M. Czyrnek-Delêtre, S. Rocca, A. Agostini, J. Giuntoli and J. D. Murphy, Appl. Energy, 2017, 196, 34–50 Search PubMed.
- P. Fasahati, R. Dickson, C. M. Saffron, H. C. Woo and J. J. Liu, Renewable Sustainable Energy Rev., 2022, 157, 112011 Search PubMed.
- A. Golberg, M. Polikovsky, M. Epstein, P. M. Slegers, D. Drabik and A. Kribus, Energy Convers. Manage., 2021, 246, 114679 CrossRef CAS.
- K. A. Jung, S. R. Lim, Y. Kim and J. M. Park, Environ. Prog. Sustainable Energy, 2017, 36, 200–207 CrossRef CAS.
- J. Langlois, J. F. Sassi, G. Jard, J. P. Steyer, J. P. Delgenes and A. Hélias, Biofuels, Bioprod. Biorefin., 2012, 6, 387–404 CrossRef CAS.
- E. T. Quintanar-Orozco, K. J. Azcorra-May, E. Olguin-Maciel, L. Alzate-Gaviria and R. Tapia-Tussell, Biofuels, Bioprod. Biorefin., 2025, 19, 1088–1099 CrossRef CAS.
- M. Seghetta, D. Romeo, M. D'este, M. Alvarado-Morales, I. Angelidaki, S. Bastianoni and M. Thomsen, J. Cleaner Prod., 2017, 150, 1–15 CrossRef CAS.
- Y. Wen, S. Wang, Z. Shi, Y. Jin, J.-B. Thomas, E. S. Azzi, D. Franzén, F. Gröndahl, A. Martin and C. Tang, Water Res., 2022, 222, 118875 CrossRef CAS PubMed.
- Y. Kulikova, G. Ilinykh, N. Sliusar, O. Babich and M. Bassyouni, Energy Convers. Manage.:X, 2024, 23, 100647 CAS.
- A. M. H. Putri, M. Safaat, H. Prasetia, F. Zulpikar, J. Renyaan and R. Noor, Int. J. Technol., 2023, 14, 78–89 CrossRef.
- M. Kiehbadroudinezhad, H. Hosseinzadeh-Bandbafha, J. Pan, W. Peng, Y. Wang, M. Aghbashlo and M. Tabatabaei, Energy, 2023, 278, 127871 CrossRef CAS.
- K. G. V. Anand, K. Eswaran and A. Ghosh, J. Cleaner Prod., 2018, 170, 1621–1627 CrossRef.
- A. Ghosh, K. G. V. Anand and A. Seth, Algal Res., 2015, 12, 513–521 CrossRef.
- M. Seghetta, D. Tørring, A. Bruhn and M. Thomsen, Sci. Total Environ., 2016, 563, 513–529 CrossRef PubMed.
- L. Sharma, M. Banerjee, G. C. Malik, V. A. K. Gopalakrishnan, S. T. Zodape and A. Ghosh, J. Cleaner Prod., 2017, 149, 968–975 CrossRef CAS.
- I. Singh, S. Solomon, V. A. K. Gopalakrishnan and A. Ghosh, Sugar Tech, 2023, 25, 440–452 CrossRef CAS.
- I. Singh, K. V. Anand, S. Solomon, S. K. Shukla, R. Rai, S. T. Zodape and A. Ghosh, J. Cleaner Prod., 2018, 204, 992–1003 CrossRef.
- A. Arias, G. Feijoo and M. T. Moreira, Sustain. Prod. Consum., 2024, 48, 169–180 CrossRef.
- R.G.D Campos, Master’s Thesis, Aalborg University, 2022, pp. 1–54.
- G. Pitolli Lyra, A. J. F. P. Duran, I. M. da Silva Parente, C. Bueno, F. G. Tonin and J. A. Rossignolo, J. Mater. Civ. Eng., 2025, 37, 04025236 Search PubMed.
- C. R. Coelho, G. Peters, J. Zhang, B. Hong, M. Abdollahi and I. Undeland, Future Foods, 2022, 6, 100194 CrossRef CAS.
- J. Halfdanarson, M. Koesling, N. P. Kvadsheim, J. Emblemsvåg and C. Rebours, APMS 2019, IFIP Advances in Information and Communication Technology, Springer, Cham, 2019, vol. 567, pp. 127–134 Search PubMed.
- M. Koesling, N. P. Kvadsheim, J. Halfdanarson, J. Emblemsvåg and C. Rebours, J. Cleaner Prod., 2021, 307, 127301 Search PubMed.
- A. E. Nilsson, K. Bergman, L. P. G. Barrio, E. M. Cabral and B. K. Tiwari, Algal Res., 2022, 65, 102725 CrossRef.
- F. Pagels, A. Arias, A. Guerreiro, A. C. Guedes and M. T. Moreira, Phycology, 2022, 2, 374–383 Search PubMed.
- S. Parsons, M. J. Allen, F. Abeln, M. McManus and C. J. Chuck, J. Cleaner Prod., 2019, 232, 1272–1281 CrossRef CAS.
- P. M. Slegers, R. J. K. Helmes, M. Draisma, R. Broekema, M. Vlottes and S. W. K. van den Burg, J. Cleaner Prod., 2021, 319, 128689 Search PubMed.
- S. Zuhria, Earth and Environmental Science, 2022, 1063, 012013 Search PubMed.
- A. Arias, E. Entrena-Barbero, T. Ilmjarv, R. Paoli, F. Romagnoli, G. Feijoo and M. T. Moreira, Nat. Biotechnol., 2025, 86, 73–86 CAS.
- R. Zhang, Q. Wang, H. Shen, Y. Yang, P. Liu and Y. Dong, Algal Res., 2024, 78, 103384 Search PubMed.
- R. Meite, L. Bayer, M. Martin, B. Amon and S. Uthes, Heliyon, 2024, 10, e29389 CrossRef CAS PubMed.
- J. Nilsson and M. Martin, Sustain. Prod. Consum., 2022, 30, 413–423 CrossRef.
- M. Seghetta, X. Hou, S. Bastianoni, A.-B. Bjerre and M. Thomsen, J. Cleaner Prod., 2016, 137, 1158–1169 Search PubMed.
- M. Seghetta, M. Marchi, M. Thomsen, A.-B. Bjerre and S. Bastianoni, Algal Res., 2016, 18, 144–155 CrossRef.
- J.-B. Thomas, M. Sodré Ribeiro, J. Potting, G. Cervin, G. M. Nylund, J. Olsson, E. Albers, I. Undeland, H. Pavia and F. Gröndahl, ICES J. Mar. Sci., 2021, 78, 451–467 Search PubMed.
- J. B. E. Thomas, R. Sinha, Å. Strand, T. Söderqvist, J. Stadmark, F. Franzén, I. Ingmansson, F. Gröndahl and L. Hasselström, J. Ind. Ecol., 2022, 26, 2136–2153 CrossRef CAS.
- S. E. Taelman, J. Champenois, M. D. Edwards, S. De Meester and J. Dewulf, Algal Res., 2015, 11, 173–183 CrossRef.
- R. van Oirschot, J.-B. E. Thomas, F. Gröndahl, K. P. Fortuin, W. Brandenburg and J. Potting, Algal Res., 2017, 27, 43–54 CrossRef.
- X. Zhang, T. Boderskov, A. Bruhn and M. Thomsen, Algal Res., 2022, 64, 102686 CrossRef.
- X. Zhang and M. Thomsen, Algal Res., 2021, 60, 102499 CrossRef.
- H. Wu, X. Liu, L. Tang, H. Zhao, F. Wei, C. Liu and G. Song, Environ. Impact Assess. Rev., 2025, 111, 107735 CrossRef.
- J.-B. E. Thomas, E. Ahlgren, S. Hornborg and F. Ziegler, J. Cleaner Prod., 2024, 450, 141987 CrossRef.
- C. Vance, P. Pollard, J. Maguire, J. Sweeney and F. Murphy, Algal Res., 2023, 75, 103294 CrossRef.
- R. Paoli, B. Bjarnason, T. Ilmjärv and F. Romagnoli, Environ. Clim. Technol., 2023, 27, 606–626 CAS.
- J. Sadhukhan, S. Gadkari, E. Martinez-Hernandez, K. S. Ng, M. Shemfe, E. Torres-Garcia and J. Lynch, Green Chem., 2019, 21, 2635–2655 RSC.
- K. Nida, K. Sudhakar and R. Mamat, Heliyon, 2024, 10, e28208 CrossRef PubMed.
- S. Fahmida, W. Md Abdul, N. Md, M. Md, I. Mohammad Zafar, S. Abrar, M. Abdullah-Al, K. Md Sadequr Rahman, W. LiLian and A. Md, Aquacult. Fish., 2023, 8, 463–480 Search PubMed.
- FAO, The State of World Fisheries and Aquaculture 2020, Sustainability in action, Rome, 2020, DOI:10.4060/ca9229en.
- N. Khan, K. Sudhakar and R. Mamat, Heliyon, 2024, 10, e28208 CrossRef PubMed.
- World Bank Group, Seaweed Aquaculture for Food Security, Income Generation and Environmental Health in Tropical Developing Countries, World Bank, 2016, http://hdl.handle.net/10986/24919.
- C. S. Lobban and P. J. Harrison, Seaweed Ecology and Physiology, Cambridge University Press, 1994 Search PubMed.
- M. Yanagisawa, S. Kawai and K. Murata, Bioengineered, 2013, 4, 224–235 Search PubMed.
- L. Zhang, W. Liao, Y. Huang, Y. Wen, Y. Chu and C. Zhao, Food Prod., Process. Nutr., 2022, 4, 23 CrossRef.
- Seaweeds: Biodiversity, Environmental Chemistry and Ecological Impacts, ed. M. Miranda, M. López-Alonso, M. García-Vaquero, and P. Newton, 2017 Search PubMed.
- N. G.-C. Maria, C. P. Ana, L. Irene, R. José and F. Q. Maria, J. Nutr., 2007, 137, 2691–2695 CrossRef PubMed.
- C. Nag Dasgupta, Algae as a Source of Phycocyanin and Other Industrially Important Pigments, 2015 Search PubMed.
- W. Niklas, N. Filip, M.-B. Eric, S. Karin, E. Ulrica, W. Gunnar and A. Eva, Carbohydr. Polym., 2020, 233, 115852 CrossRef PubMed.
- C. Filote, S. C. Santos, V. I. Popa, C. M. Botelho and I. Volf, Environ. Chem. Lett., 2021, 19, 969–1000 CrossRef CAS.
- Z. Enio, D. Eya, P. Bhavish, B. Tobias, P. Horst, P. Adrian and L. Christian, Algal Res., 2021, 56, 102288 CrossRef.
- L.-A. Tziveleka, E. Ioannou and V. Roussis, Carbohydr. Polym., 2019, 218, 355–370 CrossRef CAS PubMed.
- R. V. Hemavathy, Y. P. Ragini, S. Shruthi, S. Ranjani, S. Subhashini and P. Thamarai, Ind. Crops Prod., 2025, 224, 120282 CrossRef CAS.
- P. G. Del Río, J. S. Gomes-Dias, C. M. Rocha, A. Romaní, G. Garrote and L. Domingues, Bioresour. Technol., 2020, 299, 122613 CrossRef PubMed.
- H. Chen, D. Zhou, G. Luo, S. Zhang and J.-M. Chen, Renewable Sustainable Energy Rev., 2015, 47, 427–437 CrossRef CAS.
- F. Offei, M. Mensah, A. Thygesen and F. Kemausuor, Fermentation, 2018, 4, 99 CrossRef CAS.
- B. L. Tagliapietra and M. T. P. S. Clerici, Food Res. Int., 2023, 167, 112655 CrossRef CAS PubMed.
- K. Sinah, A. Romero, B. Christin, B. Leon, A. Garabed, L. Andreas and K. Martin, Biomass Bioenergy, 2024, 183, 107105 CrossRef.
- K. T. Brijesh and J. T. Declan, in Seaweed Sustainability, ed. K. T. Brijesh and J. T. Declan, Academic Press, San Diego, 2015, pp. 1–6, DOI:10.1016/B978-0-12-418697-2.00001-5.
- D. Sahoo and C. Yarish, Mariculture of Seaweeds, 2005 Search PubMed.
- L. Hayashi, A. Q. Hurtado, F. E. Msuya, G. Bleicher-Lhonneur and A. T. Critchley, in Seaweeds and Their Role in Globally Changing Environments, ed. J. Seckbach, R. Einav and A. Israel, SpringerNetherlands, Dordrecht, 2010, pp. 251–283, DOI:10.1007/978-90-481-8569-6_15.
- M. Michéal Mac, C. Lynn, M. Liam, A. Rita and T. C. a. Alan, Eur. J. Phycol., 2017, 52, 371–390 Search PubMed.
- L. Amponsah, C. Chuck and S. Parsons, Sustainability Sci. Technol., 2024, 1, 012001 CrossRef CAS.
- A. Vergara-Fernández, G. Vargas, N. Alarcón and A. Velasco, Biomass Bioenergy, 2008, 32, 338–344 CrossRef.
- O. Obata, J. C. Akunna and G. Walker, Biomass Bioenergy, 2015, 80, 140–146 Search PubMed.
- A. Dave, Y. Huang, S. Rezvani, D. McIlveen-Wright, M. Novaes and N. Hewitt, Bioresour. Technol., 2013, 135, 120–127 Search PubMed.
- R. P. John, G. Anisha, K. M. Nampoothiri and A. Pandey, Bioresour. Technol., 2011, 102, 186–193 Search PubMed.
- Z. Kádár, Z. Szengyel and K. Réczey, Ind. Crops Prod., 2004, 20, 103–110 Search PubMed.
- K. Öhgren, O. Bengtsson, M. F. Gorwa-Grauslund, M. Galbe, B. Hahn-Hägerdal and G. Zacchi, J. Biotechnol., 2006, 126, 488–498 Search PubMed.
- S. Kumar, R. Gupta, G. Kumar, D. Sahoo and R. C. Kuhad, Bioresour. Technol., 2013, 135, 150–156 CrossRef CAS PubMed.
- P. Fasahati, H. C. Woo and J. J. Liu, Appl. Energy, 2015, 139, 175–187 CrossRef CAS.
- A. J. Wargacki, E. Leonard, M. N. Win, D. D. Regitsky, C. N. S. Santos, P. B. Kim, S. R. Cooper, R. M. Raisner, A. Herman and A. B. Sivitz, Science, 2012, 335, 308–313 CrossRef CAS PubMed.
- T. N. Pham, W. J. Nam, Y. J. Jeon and H. H. Yoon, Bioresour. Technol., 2012, 124, 500–503 CrossRef CAS PubMed.
- P. Fasahati, C. M. Saffron, H. C. Woo and J. J. Liu, Energy Convers. Manage., 2017, 135, 297–307 CrossRef CAS.
- D. Humbird, R. Davis, L. Tao, C. Kinchin, D. Hsu, A. Aden, P. Schoen, J. Lukas, B. Olthof and M. Worley, Process Design and Economics for Biochemical Conversion of Lignocellulosic Biomass to Ethanol: Dilute-Acid Pretreatment and Enzymatic Hydrolysis of Corn Stover, National Renewable Energy Lab.(NREL), Golden, CO (United States), 2011 Search PubMed.
- A. Border, A. Tolessa, J. Görgens and N. Goosen, Waste Biomass Valorization, 2025, 16, 3689–3703 CrossRef CAS.
- M. Farghali, I. M. A. Mohamed, A. I. Osman and D. W. Rooney, Environ. Chem. Lett., 2023, 21, 97–152 CrossRef CAS PubMed.
- M. Schumacher, J. Yanık, A. Sınağ and A. Kruse, J. Supercrit. Fluids, 2011, 58, 131–135 CrossRef CAS.
- M. Turhan, T. Aysu, M. Harbi Çalımlı and M. M. Küçük, Energy Sources, Part A, 2017, 39, 90–96 CrossRef CAS.
- F. Safari, O. Norouzi and A. Tavasoli, Bioresour. Technol., 2016, 222, 232–241 CrossRef CAS PubMed.
- K. Anastasakis and A. Ross, Fuel, 2015, 139, 546–553 CrossRef CAS.
- W. Yang, X. Li, S. Liu and L. Feng, Energy Convers. Manage., 2014, 87, 938–945 CrossRef CAS.
- Q.-V. Bach, M. V. Sillero, K.-Q. Tran and J. Skjermo, Algal Res., 2014, 6, 271–276 CrossRef.
- T. Priscila, S. Janaína Pires, C. Fungyi and Y. A. C. Déborah, Algal Res., 2019, 37, 288–306 CrossRef.
- X. Fengwei, G. Chengcheng and A. Luc, Mater. Sci. Eng., R, 2024, 159, 100799 CrossRef.
- F. Joël, Trends Food Sci. Technol., 1999, 10, 25–28 CrossRef.
- P. Ashkan, K. Alireza and D.-L. Carolin, Technol. Forecast. Soc. Change, 2025, 216, 124157 CrossRef.
- M. P. Sudhakar, M. Ravel, K. Arunkumar and K. Perumal, Biomass Bioenergy, 2016, 90, 148–154 CrossRef CAS.
- J. W. van Hal, W. J. J. Huijgen and A. M. López-Contreras, Trends Biotechnol., 2014, 32, 231–233 CrossRef CAS PubMed.
- E. T. Kostas, D. A. White and D. J. Cook, BioEnergy Res., 2020, 13, 271–285 CrossRef CAS PubMed.
- L. Richen, D. Chen, D. Lingkan, B. Archishman and D. M. Jerry, Energy Convers. Manage., 2019, 196, 1385–1394 CrossRef.
- S. Dhakal, A. O. Jüterbock, X. Lei and P. Khanal, Anim. Nutr., 2024, 19, 153–165 CrossRef CAS PubMed.
- I. O. f. Standardization, Environmental Management: Life Cycle Assessment; Principles and Framework, ISO, 2006 Search PubMed.
- I. O. f. Standardization, Environmental Management: Life Cycle Assessment: Requirements and Guidelines. Amendment 2, International Organization for Standardization, 2020 Search PubMed.
- J. R. Centre, I. f. Environment and Sustainability, International Reference Life Cycle Data System (ILCD) Handbook – General guide for life cycle assessment – Detailed guidance, Publications Office of the European Union, 2010 Search PubMed.
- S. Ahlgren, A. Björklund, A. Ekman, H. Karlsson, J. Berlin, P. Börjesson, T. Ekvall, G. Finnveden, M. Janssen and I. Strid, Biofuels, Bioprod. Biorefin., 2015, 9, 606–619 CrossRef CAS.
- A. Bjørn, A. Laurent, M. Owsianiak and S. I. Olsen, in Life Cycle Assessment: Theory and Practice, Springer, 2017, pp. 67–74 Search PubMed.
- A. Bjørn, M. Owsianiak, A. Laurent, S. I. Olsen, A. Corona and M. Z. Hauschild, in Life Cycle Assessment: Theory and Practice, Springer, 2017, pp. 75–116 Search PubMed.
- J. Giuntoli, C. Bulgheroni, L. Marelli, S. Sala, R. Pant, M. Lusser and M. Avraamides, Brief on the use of Life Cycle Assessment (LCA) to evaluate environmental impacts of the bioeconomy, 2019 Search PubMed.
- O. Mateo, M.-I. Miguel, H. Almudena and S. Bernhard, J. Cleaner Prod., 2023, 383, 135331 CrossRef.
- R. Julio, J. Albet, C. Vialle, C. Vaca-Garcia and C. Sablayrolles, Biofuels, Bioprod. Biorefin., 2017, 11, 373–395 CrossRef CAS.
- E. Crenna, M. Secchi, L. Benini and S. Sala, Int. J. Life Cycle Assess., 2019, 24, 1851–1877 CrossRef CAS.
- J. Bare, Clean Technol. Environ. Policy, 2011, 13, 687–696 CrossRef CAS.
- J. R. Centre, I. f. Environment and Sustainability, International Reference Life Cycle Data System (ILCD) Handbook – General Guide for Life Cycle Assessment: Provisions and Action Steps, Publications Office, 2011 Search PubMed.
- M. A. J. Huijbregts, Z. J. N. Steinmann, P. M. F. Elshout, G. Stam, F. Verones, M. Vieira, M. Zijp, A. Hollander and R. van Zelm, Int. J. Life Cycle Assess., 2017, 22, 138–147 CrossRef.
- I. Standard, Environmental management-Life cycle assessment-Requirements and guidelines, ISO, London, 2006 Search PubMed.
- A. Laurent, S. I. Olsen and M. Z. Hauschild, Environ. Sci. Technol., 2012, 46, 4100–4108 CrossRef CAS PubMed.
- K. Simonen, Life Cycle Assessment, Routledge, London, 1st edn, 2014, DOI:10.4324/9781315778730.
- S. Hellweg and L. Milà i Canals, Science, 2014, 344, 1109–1113 CrossRef CAS PubMed.
- N. Collins, M. K. Mediboyina, M. Cerca, C. Vance and F. Murphy, Bioresour. Technol., 2022, 346, 126637 CrossRef CAS PubMed.
- J. Li, K. Bergman, J. E. Thomas, Y. Gao and F. Grondahl, Sci. Total Environ., 2023, 903, 166861 CrossRef CAS PubMed.
- A. R. Jones, H. K. Alleway, D. McAfee, P. Reis-Santos, S. J. Theuerkauf and R. C. Jones, BioScience, 2022, 72, 123–143 CrossRef PubMed.
- S. W. Mrunal, S. Sowjanya, N. Pinku Chandra, C. Arnab, A. Rajshree, M. Bishwambhar, M. Awdhesh Kumar and M. Yugal Kishore, Process Saf. Environ. Prot., 2024, 183, 708–725 CrossRef.
- P. B. Joseph, F. R. Laura and D. G. Leonardo, Algal Res., 2023, 75, 103248 CrossRef.
- A. G. Olabi, S. Nabila, S. Enas Taha, R. Cristina, A. Ruth Chinyere, R. Callum and A. Mohammad Ali, Sci. Total Environ., 2023, 854, 158689 CrossRef CAS PubMed.
- V. Vincenzo, C. Antonello, M. Francesca and C. Mauro, Sustain. Prod. Consum., 2024, 47, 87–104 CrossRef.
- A. K. Priya, A. Alagumalai, D. Balaji and H. Song, RSC Sustainability, 2023, 1, 746–762 Search PubMed.
- S. Bleakley and M. Hayes, Foods, 2017, 6, 33 CrossRef PubMed.
- S. B. Ravi, Chem. Eng. J., 2023, 454, 140177 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |