 Open Access Article
Open Access ArticleMicrowave assisted construction of a SnOx/ZnSn(OH)6 heterojunction for photocatalytic CO2 cycloaddition
Soumita
Sarkar
 ,
Soumalya
Banerjee
,
Soumalya
Banerjee
 ,
Sk Afsar
Ali
,
Sk Afsar
Ali
 ,
Sunny
Sarkar
,
Sunny
Sarkar
 and
Astam K.
Patra
and
Astam K.
Patra
 *
*
Department of Chemistry, University of Kalyani, Kalyani 741235, West Bengal, India. E-mail: astamchem18@klyuniv.ac.in
First published on 2nd February 2026
Abstract
The utilization of photochemical reactions to convert CO2 and epoxides into valuable cyclic organic carbonates is a highly sought-after process in various industries. The development of a high-performance photocatalyst for this cycloaddition of CO2 presents a significant challenge, particularly in the context of sustainability initiatives. To effectively harness light energy for CO2 value-added processes, we have engineered a SnOx-decorated perovskite-type ZnSn(OH)6 photocatalyst. Through analysis using UV-Vis and Mott–Schottky plots, we have gained insights into the electronic structure of the materials, including adsorption, band gap, and band position. The incorporation of SnOx into the ZnSn(OH)6 cube has significantly improved charge transfer, as demonstrated by photocurrent and Nyquist measurement. This molecular framework lays the foundation for the high photocatalytic performance and stability exhibited by SnOx/ZnSn(OH)6 in facilitating the cycloaddition of CO2 and various epoxides to produce cyclic carbonates. The SnOx/ZnSn(OH)6 catalyst demonstrates high performance, achieving an impressive yield of ≈95% and reaction activity of 4750.0 µmol g−1 h−1 with high values of green chemistry metrics (AE = 100%, RME = 92.98%, PMI = 5.99 and E factor = 4.99). This performance surpasses that of several other catalysts. Further examination through LSV, EPR and electron–hole scavenger tests has provided a deeper understanding of the reaction mechanism under light irradiation. Additionally, the materials have demonstrated good stability, highlighting their potential in sustainable processes.
Sustainability spotlightMost of the current processes for the transformation of CO2 into value added chemicals have various limitations from an experimental point of view such as (i) requiring extreme temperature or pressure with a lengthy reaction time; (ii) use of hazardous catalysts; (iii) use of non-ecofriendly solvents. In this work, we have carried out the photocatalytic conversion of CO2 and epoxides into valuable cyclic organic carbonates using the SnOx/ZnSn(OH)6 material at ambient temperature and pressure. The catalysts were synthesized using a commercial microwave reactor with a short reaction time of 2 min. The catalysts have a number of beneficial qualities like simpler separation and recyclability. Utilising light is a sustainable, affordable, adaptable, and energy-efficient process. The SnOx/ZnSn(OH)6 catalyst demonstrates exceptional performance, achieving an impressive yield of ≈95%, and a reaction activity of 4750.0 µmol g−1 h−1 for the photocatalytic cycloaddition of CO2 using an eco-friendly solvent (PEG 600), SnOx/ZnSn(OH)6 and TBAB cocatalyst. Our synthesized catalyst also provides high values of green chemistry metrics (AE = 100%, RME = 92.98%, PMI = 5.99 and E factor = 4.99). Furthermore, the catalyst's reusability was assessed, showing that it maintained photocatalytic activity for up to six consecutive cycles, highlighting its potential for reuse. |
1. Introduction
The transformation of carbon dioxide (CO2) into valuable chemical products has attracted significant attention as a promising approach and crucial area of research, offering potential solutions to mitigate climate change and utilize this abundant carbon source.1–3 One of the most promising pathways involves the photochemical reaction of CO2 with epoxides, which shows great potential for the eco-friendly production of cyclic carbonates.4,5 Photochemical CO2 fixation presents a sustainable and energy-efficient substitute for conventional thermal or catalytic techniques, utilizing light as a driving force to activate CO2.6–10 The use of light for driving CO2 conversion not only provides a mild reaction environment but also enables selective transformations that are difficult to achieve under conventional conditions.11–14 The photocatalytic CO2 cycloaddition process is highly sought after due to its 100% CO2 utilization and significant economic benefits.15 One example of its effectiveness is the conversion of styrene oxide to styrene carbonate by simply adding CO2. The resulting cyclic organic carbonates are widely used as aprotic polar solvents,16 polycarbonate precursors,17 and electrolytes for secondary batteries.18 However, the challenge lies in the poor electron-withdrawing capacity and strong steric hindrance of the phenyl group in styrene oxide, making the cycloaddition of CO2 difficult.19 Developing effective catalysts for CO2 fixation to high-value compounds is therefore a worthwhile but challenging endeavor. Various catalysts have been developed for the CO2 cycloaddition process, such as semiconductors,20–23 single-atom catalysts,24,25 organocatalysts,26,27 MOF-based catalysts,28 semiconductor quantum dots,29,30 and nanoclusters31,32 which facilitate the activation of CO2 and epoxide substrates under light irradiation. Studies have explored various strategies to optimize reaction conditions, such as the use of co-catalysts, solvent-free conditions, and solar-driven processes, to improve yields and selectivity.33,34 Unfortunately, many of these catalysts require harsh conditions, leading to laborious post-reaction treatment, environmental issues, and increased energy consumption. As a result, there is a pressing need for the green and sustainable synthesis of CO2 cycloaddition catalysts. Researchers are turning their attention to the photocatalytic CO2 cycloaddition reaction with epoxides due to its sustainable process and environmentally friendly nature.35Semiconductor heterojunctions play a crucial role in the advancement of new photocatalysts.36–38 These heterojunctions are created at the interface between two materials with different semiconductor band gaps, usually composed of distinct semiconductor compounds.36 The distinct properties found at these interfaces, including customized band alignments, effective charge carrier transport, and decreased recombination rates, allow for the development of high-performance materials. Through precise engineering of these interfaces, significant progress has been made in the fields of photovoltaics and photocatalysis.39 The key to achieving optimal performance in semiconductor heterojunctions lies in the careful selection of materials, the precise control of interface quality, and the understanding of charge transfer mechanisms at the junction. With the development of new materials, such as perovskites and two-dimensional materials, and advancements in fabrication techniques, the scope of semiconductor heterojunctions continues to expand.40–42 In particular, metal oxide/hydroxide heterojunction materials have shown promise in a wide range of applications due to their tunable electronic properties and efficient carrier dynamics.43 Among the emerging material systems, the SnOx/ZnSn(OH)6 heterojunction has shown considerable promise because of its unique electronic properties, stability, and efficient charge separation capabilities.44–46 SnOx (tin oxide) is widely recognized for its high conductivity, optical transparency, and suitability as an electron transport material in solar cells and photocatalysts,47,48 while ZnSn(OH)6 offers a distinct advantage due to its mixed-valence structure and tunable band gap.49 The combination of these two materials into a heterojunction structure can significantly enhance the performance of devices such as photocatalysts for water splitting, sensors, and field-effect transistors. Recently, Chen and his colleagues synthesized a small quantity of SnO2-decorated ZnSn(OH)6, which proved to be an extremely efficient photocatalyst for the decomposition of gaseous benzene.44 Their study aimed to uncover the synergistic effect between SnO2 and ZnSn(OH)6.
In this study, we demonstrate the inclusion of SnOx into ZnSn(OH)6, resulting in the high conversion of CO2 gas to organic carbonate. The presence of SnOx on ZnSn(OH)6 plays a crucial role in reducing charge recombination. The SnOx/ZnSn(OH)6 catalyst demonstrates high performance, achieving an impressive yield of ≈95% and a reaction activity of 4750.0 µmol g−1 h−1 with AQY 722.89 × 10−5.
2. Results and discussion
2.1 Morphology, phase and structural analysis
Cuboid-shaped zinc hydroxystannate [ZnSn(OH)6] was successfully synthesized using a microwave-assisted synthetic route without the need for a template. The synthetic process included zinc nitrate hexahydrate, tin(IV) chloride, sodium hydroxide, and deionized water. This innovative method facilitated the efficient production of zinc hydroxystannate cuboids in an aqueous solution, leading to a remarkably short reaction time of just 2 minutes. Furthermore, the SnOx-decorated zinc hydroxystannate [SnOx/ZnSn(OH)6] was synthesized by utilizing zinc hydroxystannate cubes and tin(IV) chloride through a microwave-assisted synthetic route. The specific procedures for these reactions are outlined in Scheme 1 and the Experimental section.
Fig. 1a, b and c, d shows the SEM pictures of ZnSn(OH)6 and SnOx/ZnSn(OH)6 materials respectively. Fig. 1a and b displays a SEM image of cuboid-shaped materials ranging in size from 0.5 to 1.7 µm in length, 0.5 to 1.6 µm in width, and 0.4 to 1.4 µm in height. The surface morphology of materials remains consistent across the entire product. But the SnOx/ZnSn(OH)6 material shows a cuboid shape with small-size nanoparticles on the surfaces. Furthermore, the SnOx particles on the surface of the ZnSn(OH)6 cuboid are examined through high-resolution transmission electron microscopy (TEM) analysis, as depicted in Fig. 2. The powder XRD features obtained from ZnSn(OH)6 and SnOx/ZnSn(OH)6 (Fig. 1e) clearly indicate the crystalline nature of these cuboids. But there were no additional peaks for the SnO2 phase or for other phases in SnOx/ZnSn(OH)6. This indicates that the SnOx nanoparticles are amorphous in nature. Both the materials displayed primary peaks at 19.93, 22.99, 32.70, 36.68, 38.59, 40.32, 46.91, 52.80, and 58.17° corresponding to the (111), (200), (220), (310), (311), (222), (400), (420), and (422) planes, respectively, of the zinc hydroxystannate phase.44 These diffraction peaks are matched to a pure cubic phase with lattice parameter a = b = c = 7.800 Å and the space group of Pn![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) (JCPDS no. 96-901-2079). The distinct diffraction patterns observed in the as-prepared samples clearly indicate that the sample is crystalline in nature and possesses high purity. The material [ZnSn(OH)6] synthesized using microwave radiation was crystalline in nature and composed of ZnO6 octahedra and SnO6 octahedra (JCPDS no. 96-901-2079).
(JCPDS no. 96-901-2079). The distinct diffraction patterns observed in the as-prepared samples clearly indicate that the sample is crystalline in nature and possesses high purity. The material [ZnSn(OH)6] synthesized using microwave radiation was crystalline in nature and composed of ZnO6 octahedra and SnO6 octahedra (JCPDS no. 96-901-2079).
 | ||
| Fig. 2 High-resolution TEM images of (a–c) ZnSn(OH)6 and (d–f) SnOx/ZnSn(OH)6 microcubes at different magnifications. | ||
Additionally, FT-IR spectroscopy measurements were conducted to analyze the bonding environment of the materials.50,51 The FT-IR spectra in Fig. 1f show the molecular vibrations between Zn, Sn, O and H. Fig. 1f shows the FT-IR spectra of the ZnSn(OH)6 material. The bands of absorption are detected at 3210, 1628, 1172, 1104, 777, and 536 cm−1. These bands demonstrate strong correlation with the ZnSn(OH)6 material previously reported.52,53 The infrared spectrum displays a prominent, narrow band at approximately 3210 cm−1, indicating the presence of O–H stretching vibration. The small band at 1628 cm−1 is ascribed to the adsorbed water molecules on the materials. The sharp band at 1172 cm−1 may arise due to the Sn–OH bending vibrations.53 The sharp band at 1108 cm−1 arises due to the out-of-plane deformation vibration of Zn–OH, while the stretching vibrations of Sn–O–Sn are observed at 536 cm−1 and 777 cm−1.52
Fig. 2 and S1 show high-resolution transmission electron microscopy (HR TEM) images of ZnSn(OH)6 and SnOx/ZnSn(OH)6 materials at different magnifications. In Fig. 2a, the high-resolution TEM image shows that ZnSn(OH)6 is a cuboid-shaped particle with a length of 0.7 µm. Fig. 2b and c also show a similar texture at high magnifications for the ZnSn(OH)6 material. Even after the loading of SnOx, the particle's cuboid shape remains intact, with small SnOx nanoparticles deposited on the surfaces (Fig. 2d). The surface SnOx is more clearly visible in the highly magnified images in Fig. 2e and f. Furthermore, the presence of SnOx on the surface of the ZnSn(OH)6 material was established by elemental mapping (Fig. S1).
2.2 Surface analysis
To investigate the surface chemical composition of the ZnSn(OH)6 microcubes, X-ray photoelectron spectroscopy (XPS) analysis was performed. For the ZnSn(OH)6 and SnOx/ZnSn(OH)6 materials, the survey profile, high-resolution XPS spectra of Zn 2p core level, Sn 3d core level and O 1s core level are shown in Fig. S2 and 3. The survey profile of ZnSn(OH)6 and SnOx/ZnSn(OH)6 microcubes across the 0–1200 eV region is depicted in Fig. S2 showing the presence of Zn, Sn and O elements. For ZnSn(OH)6, the high-resolution Zn 2p spectrum (Fig. 3a) displayed two broad peaks at 1021.74 eV for Zn 2p3/2 and 1044.84 eV for Zn 2p1/2.53,54 The spin–orbit splitting of 23.1 eV is indicative of the Zn2+ oxidation state in the ZnSn(OH)6 microcubes.55,56 The high-resolution Sn 3d spectrum (Fig. 3b) displayed peaks at 486.73 eV for Sn 3d5/2 and 495.29 eV for Sn 3d3/2.53,54 The spin–orbit splitting of 8.56 eV is indicative of the Sn4+ oxidation state in the ZnSn(OH)6 microcubes.55 The Zn 2p peak (Fig. 3d) and Sn 3d peak (Fig. 3e) in SnOx/ZnSn(OH)6 demonstrate similar observation to ZnSn(OH)6. The O 1s peak in ZnSn(OH)6 microcubes displays asymmetry and a noticeable shoulder, which can be further divided into three distinct Gaussian–Lorentzian components (Fig. 3c). The main peak of O 1s at 531.45 eV is related to oxygen components (e.g., OH) within ZnSn(OH)6, adsorbed water nearly at 533.14 eV and the other one at around 530.01 eV is assigned to oxygen in metal oxides (e.g., Sn–O–Zn).54 Upon SnOx loading on ZnSn(OH)6, the population of Sn–O–Zn and Sn–O–Sn increases compared to Sn/Zn–OH which was reflected in the O 1s spectra (Fig. 3f) of SnOx/ZnSn(OH)6. The elemental composition of the ZnSn(OH)6 surface was analyzed using XPS peak areas to determine the semi-quantitative atomic percentages due to its surface sensitivity (∼10 nm). The ZnSn(OH)6 microcubes were found to contain 10.40% Zn, 13.08% Sn, and 76.52% O. The SnOx/ZnSn(OH)6 microcubes were found to contain 9.61% Zn, 19.75% Sn, and 70.63% O. To further verify the composition, we conducted elemental mapping and energy-dispersive X-ray spectroscopy (EDX) analysis (surface-sensitive ∼1–3 µm). The elemental mapping and EDX spectrum of Zn, Sn, and O are presented in Fig. 4 and S3, revealing the presence of Zn, Sn, and O throughout the microcubes. The elemental compositions of ZnSn(OH)6 using EDX are 13.37% Zn, 11.45% Sn, and 75.18% O. After loading SnOx onto the ZnSn(OH)6 surface, the composition changed to 10.41% Zn, 8.34% Sn, and 81.25% O. Both XPS and EDX show similar results of the ZnSn(OH)6 composition, which align with theoretical values.Significant discrepancy between the EDS and XPS data for ZnSn(OH)6 (e.g., Sn/Zn ratio) is expected due to their different sampling depths and surface-sensitivities. But both semi-quantitative analyses show similar results (e.g., Sn/Zn ratio) and the calculations in Fig. S3 show that the amount of Sn and O increased in the SnOx/ZnSn(OH)6 material with respect to ZnSn(OH)6. Additionally, we conducted measurements of the surface properties, oxygen vacancies, and defects, utilizing electron spin resonance (ESR) techniques. The ESR spectra provide compelling evidence of a higher concentration of oxygen vacancies (OVs) in SnOx/ZnSn(OH)6 compared to ZnSn(OH)6, with a g-value of 2.04 (Fig. S4).57,58 Under light illumination, SnOx/ZnSn(OH)6 exhibits a significantly higher peak intensity, indicating a greater ease of electron formation upon photoirradiation.58 This observation highlights its superior ability to generate photoinduced electrons, which positively influences its photoactivity.
2.3 Electronic structure and electrochemical study
Fig. 5a reveals that the ZnSn(OH)6 microcubes exhibit strong ultraviolet light absorption at approximately 215 and 250 nm, with a band gap energy of 3.98 eV (as shown in the inset of Fig. 5a).53 On the other hand, the SnOx/ZnSn(OH)6 microcubes demonstrate strong ultraviolet light absorption around 215 and 254 nm, with a slightly lower band gap energy of 3.90 eV (as depicted in the inset of Fig. 5a). A comparison between ZnSn(OH)6 and SnOx/ZnSn(OH)6 reveals that the adsorption spectra of the latter shifts to longer wavelengths due to the deposition of SnOx on the surface ZnSn(OH)6 cubes, resulting in a lower band gap energy. In Fig. S5 and 5b, the electrochemical Mott–Schottky plots of ZnSn(OH)6 and SnOx/ZnSn(OH)6 materials are presented, respectively. Both plots exhibit n-type responses, and the flat band potential of the conduction band (CB) of ZnSn(OH)6 and SnOx/ZnSn(OH)6 was calculated from the intercepts on the horizontal axis. The flat band potential of ZnSn(OH)6 was determined to be −0.577 V (0.0182 V vs. RHE), while for SnOx/ZnSn(OH)6, it was found to be −0.582 V vs. Ag/AgCl (0.0132 V vs. RHE).53 Based on the results presented above, the band energy structures of ZnSn(OH)6 and SnOx/ZnSn(OH)6 are proposed, as depicted in Fig. 5c. The highest conduction band potential of both ZnSn(OH)6 and SnOx/ZnSn(OH)6 materials shows a positive value close to the H+/H2 potential. To investigate the rates of charge transfer and the efficiency of separating photogenerated electrons and holes, transient photocurrent response and impedance spectroscopic (EIS) measurements were conducted. The transient photocurrent density of ZnSn(OH)6 and SnOx decorated ZnSn(OH)6 is shown in Fig. 5d. Typically, the intensity of the photocurrent response is directly linked to the efficiency of separating photogenerated electrons and holes. This finding confirms that SnOx/ZnSn(OH)6 effectively inhibits the recombination of charge pairs better than ZnSn(OH)6, which aligns with the results obtained from the EIS analysis. The results indicate that the presence of SnOx on the surface of ZnSn(OH)6 forms a heterojunction and effectively reduces recombination rates. This phenomenon is visually represented in Fig. 5e and f, where it can be observed that ZnSn(OH)6 and SnOx/ZnSn(OH)6 display a smaller semicircular radius on the Nyquist curve when exposed to light as opposed to darkness. This reduction in radius suggests a decrease in interfacial charge transfer resistance under light conditions, which is highly beneficial for facilitating electron and hole transfer processes. Furthermore, the steady-state photoluminescence (PL) spectrum was utilized to assess the separation efficiency of the photogenerated carriers (see Fig. S6).57,59 Notably, the fluorescence intensity of SnOx/ZnSn(OH)6 was found to be lower than that of ZnSn(OH)6. This observation indicates that the incorporation of SnOx effectively mitigates the recombination of photogenerated carriers, likely due to the presence of oxygen vacancies and defects.57,59 The impedance spectra were meticulously analyzed using the equivalent Randles circuit R1 + Q2/R2, with R1 representing electrolytic resistance, R2 representing charge transfer resistance, and Q2 representing constant phase elements.60 The values of R1, Q2 and R2 under both light and dark conditions are detailed in Table S1. The charge transfer resistance (R2) value of ZnSn(OH)6 under light conditions is 644.3 Ω, which is significantly lower than the value observed in the dark, which was 1155.0 Ω. Additionally, the loading of SnOx on ZnSn(OH)6 resulted in further reduction in charge transfer resistance under both dark and light conditions. The R2 value of SnOx/ZnSn(OH)6 under light conditions is 464.1 Ω, which is significantly lower than the value observed in the dark, which was 746.0 Ω. To evaluate charge separation and interfacial charge transfer resistance under photocatalytic reaction conditions (i.e., under 254 nm light), we conducted photocurrent generation and EIS Nyquist measurements. The findings are summarized in Fig. S7 and Table S1. The results demonstrate that SnOx-coated ZnSn(OH)6 exhibits a higher photocurrent density compared to ZnSn(OH)6. Notably, both ZnSn(OH)6 and SnOx/ZnSn(OH)6 show a reduced semicircular radius on the Nyquist plot when illuminated with 254 nm light, in contrast to measurements taken in darkness. This reduction in radius indicates a decrease in interfacial charge transfer resistance under light conditions, which significantly facilitates the transfer processes of electrons and holes.2.4 Photocatalytic activity
The optical and electrochemical properties observed in SnOx/ZnSn(OH)6 have inspired us to further explore its potential as a heterogeneous photocatalyst for the CO2 cycloaddition reaction. The CO2 cycloaddition reaction with styrene oxide was chosen as a representative model for our initial optimization experiments. Remarkably, this reaction resulted in 93% yield of the desired product, styrene carbonate, when employing SnOx/ZnSn(OH)6 as the photocatalyst, TBAB as the co-catalyst, a 1 atm CO2 balloon as the source of CO2, and PEG 600 as the solvent, all under the influence of 8 watt UV light (254 nm) at room temperature (Table 1, entry 1). The 8 watt UV light (254 nm) was chosen based on the catalyst's absorption edge (Fig. 5a). The optimization conditions and time courses of the reaction are detailed in Table 1. Furthermore, the product of the reaction was thoroughly characterized using 1H and 13C NMR spectroscopy.| Entry | Variation from the standard conditions | Time (h) | Yield (%) | Rate (µmol h−1) | Activity (µmol h−1 g−1) | AQYb × 105 |
|---|---|---|---|---|---|---|
| a Standard conditions: SnOx/ZnSn(OH)6 catalyst (50 mg), styrene oxide (3 mmol), PEG 600 (2 mL), 1 atm CO2 balloon, TBAB (0.3 mmol), 8 watt UV light (254 nm), room temperature, 12 h (PEG – polyethylene glycol). b Apparent quantum yield. c The standard reaction was carried out in white light (50 W). d Simulated solar light (using a 420 nm cut-off filter). AE, RME, PMI and E factor are green chemistry metrics. | ||||||
| 1 | Standard conditionsa | 12 | 93.0 | 232.5 | 4650.0 | 722.89 |
| 2 | No SnOx/ZnSn(OH)6 (no catalyst, TBAB only) | 12 | 19.0 | 47.5 | 950.0 | 147.69 |
| 3 | No light (dark) | 12 | 15.6 | 39.0 | 780.0 | 121.26 |
| 4 | No CO2 (Ar) | 12 | 0.00 | 0.0 | 0.0 | 0.00 |
| 5 | No TBAB | 12 | 0.00 | 0.0 | 0.0 | 0.00 |
| 6 | ZnSn(OH)6 | 12 | 74.0 | 185 | 3700.0 | 575.20 |
| 7 | ZnO | 12 | 53.0 | 132.5 | 2650.0 | 411.97 |
| 8 | SnO2 | 12 | 29.0 | 72.5 | 1450.0 | 225.42 |
| 9 | Acetonitrile | 12 | 53.0 | 132.5 | 2650.0 | 411.97 |
| 10 | DMF | 12 | 18.0 | 45.0 | 900.0 | 139.91 |
| 11 | Ethylene glycol | 12 | 22.0 | 55.0 | 1100.0 | 171.01 |
| 12 | PEG 600 | 12 | 93.0 | 232.5 | 4650.0 | 722.89 |
| 13 | Ethanol | 12 | 39.0 | 97.5 | 1950.0 | 303.15 |
| 14 | H2O + ethanol | 12 | 59.0 | 147.5 | 2950.0 | 458.61 |
| 15 | H2O | 12 | 8.0 | 20.0 | 400.0 | 62.18 |
| 16 | Without solvent | 12 | 73.0 | 182.5 | 3650.0 | 567.43 |
| 17 | White lightc | 12 | 19.0 | 47.5 | 950.0 | — |
| 18 | Simulated solar light (<420 nm)d | 06 | 24.0 | 120.0 | 2400.0 | — |
After 6 h of reaction time, 72% formation of the product was observed through 1H NMR analysis (see the SI, Fig. S8 and S9). This percentage steadily increased as the reaction time progressed, ultimately reaching complete conversion after 12 h of irradiation. The isolated yield was an impressive 93% (see SI, Fig. S8 and S10). In order to comprehend the significance of the SnOx/ZnSn(OH)6 photocatalyst in this cycloaddition process, a control experiment was carried out without the presence of SnOx/ZnSn(OH)6 (Table 1, entry 2). The NMR analysis (see SI, Fig. S11) of the reaction mixture revealed a decreased conversion of the reactant to the product, indicating the crucial role of SnOx/ZnSn(OH)6 in facilitating this cycloaddition reaction. Without light, the catalyst yields 15.6% (see Table 1, entry 3, SI, Fig. S12), implying that it contains both acidic and basic sites. Similarly, control experiments were conducted in the absence of CO2 (Table 1, entry 4), and TBAB (Table 1, entry 5), resulting in no detectable product formation. Additionally, a time-resolved light-on/light-off experiment was carried out (see the SI, Fig. S13), which conclusively demonstrated that the reaction only occurred when the light was on. The rate of the reaction displayed a first-order dependence on the epoxide (Fig. S14). This observation strongly suggests that the reaction follows a photocatalytic pathway.
Various catalysts, such as ZnSn(OH)6 (Table 1, entry 6, Fig. S15), ZnO (Table 1, entry 7, Fig. S16), and SnO2 (Table 1, entry 8, Fig. S17), were tested instead of SnOx/ZnSn(OH)6. However, these alternatives resulted in poor product formation. The activity of the catalyst was compared with several reported catalysts and results are shown in Table S2.61,62 The reaction was also conducted in various solvent systems (Table 1, entry 9–15), revealing that PEG 600 is the most effective solvent for this cycloaddition reaction. Wu et al. demonstrated that PEGs with varying molecular weights significantly influenced the outcomes of the cycloaddition reaction between CO2 and propylene oxide.63 Their findings indicate that the yield of propylene carbonate decreases markedly as the PEG chain length increases from PEG 200 to PEG 1000, likely due to mass-transport limitations. The reaction was also carried out without the use of any solvent, resulting in a yield of 73% (Table 1, entry 16). The reaction was also carried out with white light (50 W) and simulated solar light (using a 420 nm cut-off filter), resulting in a poor conversion of styrene oxide to cyclic carbonate (Table 1, entries 17 and 18). These results show the wide band gap limitation of the catalyst. In addition to optimizing the process, we have also calculated the rate (µmol h−1), activity (µmol h−1 g−1), and apparent quantum yield which are detailed in Table 1. Different quantities of SnOx/ZnSn(OH)6 were evaluated, with the results depicted in Fig. 6a and Table S3 (entry 1–5). Furthermore, the catalyst's reusability was assessed, showing that it maintained photocatalytic activity for up to six consecutive cycles (Fig. 6b), highlighting its potential for reuse. The compound SnOx/ZnSn(OH)6 can be effortlessly isolated from the reaction mixture through centrifugation and can be directly utilized in subsequent runs without the need for any additional treatment or reactivation process. The recycled SnOx/ZnSn(OH)6 maintained its initial structure and crystallinity, as evidenced by the unchanged FT-IR, powder XRD, SEM, Nyquist and XPS analysis results (see the SI, Fig. S18, S19 and Table S4). FT-IR and XRD analyses demonstrate good structural stability. Additionally, XPS analysis conducted before and after the reaction reveals that the oxidation states of the metals remain unchanged. This suggests the good stability of the SnOx/ZnSn(OH)6 photocatalyst.
 | ||
| Fig. 6 (a) Amount of SnOx/ZnSn(OH)6 variation and (b) recyclability of SnOx/ZnSn(OH)6. The reactions were performed under standard conditions as shown in Table 1, entry 1. | ||
After optimizing the conditions, the potential of the SnOx/ZnSn(OH)6 photocatalyst for synthesizing cyclic organic carbonates was investigated by testing various sources of epoxides. The results, as displayed in Table 2, demonstrate that with the addition of a small amount of the SnOx/ZnSn(OH)6 catalyst, a range of epoxy substrates successfully produced the desired products with high isolated yields ranging from 87% to 95% (Table 2, entries 1–6). The reaction exhibited high compatibility with both aromatic and aliphatic epoxides. The results presented here showcase the promising potential of the SnOx/ZnSn(OH)6 photocatalyst for practical applications.
| Entry | Reactant | Product | Time (h) | Yield (%) (SD) | AQY × 105 |
|---|---|---|---|---|---|
| a Reaction conditions: SnOx/ZnSn(OH)6 catalyst (50 mg), epoxide (3 mmol), PEG 600 (2 mL), 1 atm CO2, TBAB (0.3 mmol), 8 watt UV light (254 nm), room temperature, 12 h. SD-standard deviation, reactions were performed three times. | |||||
| 1 |
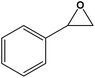
|

|
12 | 94.00 (1) | 730.66 |
| 2 |
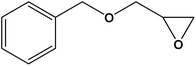
|

|
12 | 94.66 (0.33) | 735.79 |
| 3 |

|
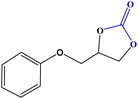
|
12 | 94.50 (0.25) | 734.55 |
| 4 |

|

|
12 | 91.50 (0.375) | 711.23 |
| 5 |

|

|
12 | 89.33 (2.33) | 694.36 |
| 6 |

|

|
12 | 87.00 (1) | 676.25 |
Literature indicates that researchers have delved into the mechanism of the CO2 cycloaddition reaction. Several groups have proposed that epoxides are oxidized by photogenerated holes to form epoxides˙+, while CO2 is reduced by photogenerated electrons to CO2˙−. These species then combine in the presence of a halide co-catalyst to create an organic carbonate.64 However, other researchers have suggested that epoxides are actually reduced to epoxides˙− and that CO2 is neither reduced nor oxidized in this process.61
In order to gain a comprehensive understanding of the CO2 cycloaddition reaction mechanism involved in this photocatalysis process, a series of meticulously controlled experiments were carried out and carefully monitored using 1H NMR analysis. The reactions were executed under the most optimized conditions, in the presence of AgNO3 (electron scavenger), KI (hole scavenger), Na2C2O4 (hole scavenger), and 5,5-dimethyl-1-pyrroline-N-oxide (DMPO) (radical scavenger) (see Table S5 and Fig. 7a for details). The presence of either AgNO3 or Na2C2O4 resulted in a decrease in product formation (see Fig. S20 and S21). This suggests that both reductive electrons and oxidative holes play a role in the photocatalytic process. Additionally, the presence of DMPO led to a significant decrease in the yield of the desired product by 35%, indicating that the reaction follows a radical pathway (see Fig. S22). The anomalous behavior observed when using KI as a hole scavenger will be discussed later.
The results from linear sweep voltammetry (LSV) curves in the potential range −0.4 to −1.2 V vs. RHE indicate that SnOx/ZnSn(OH)6 exhibited a higher current response under CO2 in the presence of light compared to under an argon atmosphere (see Fig. 7b). This suggests that the catalyst has the capability for CO2 reduction and the current density increases due to CO2 to CO2˙− conversion.65,66 Subsequently, the cyclic voltammetry (CV) of SnOx/ZnSn(OH)6 was examined in argon saturated and CO2 saturated 0.5 M Na2SO4 electrolyte at a scan rate of 10 mV s−1 in the dark (see Fig. 7c).67 The results from linear sweep voltammetry (LSV) curves in the potential range 0.6 to 1.6 V vs. RHE indicate that SnOx/ZnSn(OH)6 exhibited a higher current response in the presence of styrene oxide (see Fig. 7g). This suggests that the epoxides are oxidized to form epoxides˙+. Recently, Zhang and co-workers identified the epoxide˙+ radical generation for CO2 cycloaddition reaction by ESR analysis, with TEMPO, DMPO, and MNP as spin trapping agents.68Fig. 7d presents the ESR spectrum of the SnOx/ZnSn(OH)6 microcubes, revealing an anisotropic one-line pattern with a g-value of 2.04. Upon the introduction of CO2 (DMPO–CO2˙−, aN = 14.1 & 14.0, see Fig. 7e) or styrene oxide (DMPO–epoxyalkyl radical, aN = 14.1 & 13.9, see Fig. 7f) into the SnOx/ZnSn(OH)6 and DMPO system under 254 nm light irradiation, the signal attenuation transformed into a sharp, isotropic three-line pattern characteristic of a nitroxide radical (Fig. S23). This observation indicates an increased separation and transfer of photogenerated electrons and holes from SnOx/ZnSn(OH)6 to the reactants (CO2 and styrene oxide) when exposed to light. Furthermore, the role of the co-catalyst was examined (Fig. 7h). TBAB only can perform the reaction with a low yield (Table 1, entry 2). However, on addition of KI as a hole scavenger (i.e., TBAB & KI both are present, Fig. S24) in the reaction, the yield of the product increased. But when Na2C2O4 was used as the hole scavenger in the reaction, the yield of the product decreased. This implies that a halide has a relevant role in the reaction. So, we used KI, KBr, and TBAI instead of TBAB, the reaction showed 99, 37 and 87% yields respectively (see Fig. S25–S27). This implies that iodide shows better performance than bromide in the reaction. We also performed the reaction with KI only (without the catalyst); 50% conversion was observed in the 1H NMR analysis (see Fig. S28). This suggests that KI as the catalyst has the capability for styrene oxide conversion. A similar observation was reported in the literature.69,70
According to our controlled experiments, electrochemical studies, and previous literature, a reaction mechanism is proposed, as shown in Fig. 7i. Styrene oxide was adsorbed onto the Zn/Sn species of the SnOx/ZnSn(OH)6 catalyst through oxygen atoms, while CO2 was adsorbed at Lewis basic sites. When exposed to light, SnOx/ZnSn(OH)6 produced photogenerated electrons and holes. The photogenerated holes then oxidized styrene oxide, creating carbon-centered radicals with a positive charge. Following this, Br− ions in TBAB attacked the less hindered carbon atom in styrene oxide through a nucleophilic attack, resulting in ring-opening and the formation of a bromopropoxy intermediate (I). At the same time, photogenerated electrons reduced CO2 to CO2˙− species, which then combined with the bromopropoxy intermediate (I) to form intermediate (II). Ultimately, the intramolecular cyclization of intermediate (II) led to the creation of styrene carbonate, regenerating the SnOx/ZnSn(OH)6 catalyst and TBAB in the process.
3. Conclusion
We have successfully developed a SnOx-decorated ZnSn(OH)6 catalyst for use in photocatalytic CO2 cycloaddition reactions. The presence of SnOx on the surface of ZnSn(OH)6 makes it an ideal photocatalyst, as evidenced by high charge separation demonstrated through photocurrent density and Nyquist analysis. The SnOx/ZnSn(OH)6 catalyst exhibited outstanding catalytic performance, achieving approximately 93% conversion and a reaction rate of 4650 µmol g−1 h−1 in CO2 cycloaddition reactions with styrene oxide under light irradiation. This performance was approximately 1.3, 1.8 and 2.1 times higher than that of the ZnSn(OH)6, ZnO and SnO2 catalyst respectively. The catalyst also demonstrated high yields (87–95%) when tested with various substrates. Furthermore, the catalyst showed good recyclability, making it a promising option for sustainable catalytic processes.4. Experimental section
4.1 Synthesis of zinc hydroxystannate microcubes [ZnSn(OH)6]
Cuboid shaped zinc hydroxystannate was synthesized by a microwave assisted technique without any template. In a standard synthesis procedure, 1 mmol of Zn(NO3)2·6H2O was dissolved in 20 mL of water and stirred for a few minutes. In a separate beaker, 1 mmol of SnCl4·5H2O was dissolved in 20 mL of water. The second solution was then slowly added to the first solution. The pH of the solution was adjusted to 8 by the dropwise addition of 1 (N) NaOH. The resultant mixture was stirred for 45 minutes at room temperature. After that the solution mixture was transferred into a polypropylene bottle and placed in a microwave reactor. The microwave power (Commercial Panasonic Microwave, model number NN-CT644M, level P100, 900 W) was set to 900 watt for 2 minutes (1 atm, temperature 100 °C). After the reaction, the bottle was allowed to cool to room temperature and the precipitate was collected by centrifugation. The sample was washed several times using water and ethanol.4.2 Synthesis of SnOx decorated ZnSn(OH)6 microcubes [SnOx/ZnSn(OH)6]
100 mg as-synthesized ZnSn(OH)6 powder was dispersed in 35 mL water and sonicated for 10 minutes. Then 10 mg SnCl4·5H2O was dissolved in 5 mL water. The solution was added to the former solution. The pH of the solution was adjusted to 8 with 2 (N) NaOH followed by sonication and stirring. The well-mixed solution was then transferred into a 50 mL polypropylene bottle and placed in a microwave reactor at 900 watt for 2 minutes (1 atm, temperature 100 °C). The final product was collected by centrifugation and washed with ethanol.4.3 Catalytic activity
Herein, SnOx/ZnSn(OH)6 microcube catalysts were prepared and employed in the photocatalytic cycloaddition reaction of CO2 and different epoxides at ambient pressure and room temperature. The catalytic experiments were conducted using a CO2 balloon as the source of CO2. In the catalytic reaction, a round bottom flask was used to combine 3 mmol of styrene oxide, 2 mL of PEG 600 solvent, and 0.3 mmol of TBAB. Subsequently, 50 mg of the SnOx/ZnSn(OH)6 catalyst was introduced into the solution, which was then sealed with a silicone rubber septum. CO2 was purged into the solution using the CO2 balloon connected with a stainless steel needle. The solution was stirred under a light illumination of 254 nm UV light (8 watt, Genei Make) at room temperature. The reaction progress was monitored using TLC. Once the reaction was complete, the catalyst was recovered and the product was obtained using a mixture of ethyl acetate and water. The products were then separated using column chromatography. The optimization of the reaction and different substrate scope results are listed in Tables 1, 2 and S3–S5. The products were analyzed using 1H and 13C NMR spectroscopy, as depicted in the SI file.Author contributions
Soumita Sarkar: data curation, formal analysis, methodology, resources, software, validation, visualization, writing – original draft, writing – review & editing. Soumalya Banerjee: data curation, methodology, software, visualization, writing – review & editing. Sk Afsar Ali: data curation, methodology, software, validation, writing – review & editing. Sunny Sarkar: data curation, methodology, software, validation, writing – review & editing. Astam K. Patra: conceptualization, formal analysis, funding acquisition, investigation, project administration, resources, software, supervision, validation, visualization, writing – original draft, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Data are also available upon request from the authors. Supplementary information: experimental details and different characterization experiments, XPS, EDX, EPR, Mott–Schottky plots, PL spectra, photocurrent responses of the catalyst under 254 nm light, green chemistry metrics, reaction kinetics, light-on/light-off experiment, assessment of quenchers, 1H and 13C NMR spectra, and photocatalyst performance comparison. See DOI: https://doi.org/10.1039/d5su00789e.Acknowledgements
Ms Soumita Sarkar acknowledges the Swami Vivekananda Merit Cum Means PhD Scholarship, Government of West Bengal, India. Mr Soumalya Banerjee and Mr Sk Afsar Ali acknowledge the University Grants Commission (UGC), Government of India for the Junior Research Fellowship. Mr Sunny Sarkar acknowledges University of Kalyani for Fellowship and Personal Research Grant. Dr A. K. Patra acknowledges the Anusandhan National Research Foundation (ANRF), Government of India for funding (File No. EEQ/2021/000008). Dr A. K. Patra also acknowledges the University of Kalyani for providing the basic infrastructure. All authors acknowledge IACS Kolkata and IISER Kolkata for instrument facility. The help received from DST PURSE II grant of the University of Kalyani and DST FIST LEVEL II (SR/FST/CS-II/2019/96) grant of the Department of Chemistry, University of Kalyani is also acknowledged by Dr A. K. Patra.References
- J. Artz, T. E. Müller, K. Thenert, J. Kleinekorte, R. Meys, A. Sternberg, A. Bardow and W. Leitner, Chem. Rev., 2018, 118, 434–504 CrossRef CAS PubMed.
- L. Qiu, B. Li, J. Hu, A. Ganesan, S. Pramanik, J. T. Damron, E. Li, D.-e. Jiang, S. M. Mahurin, I. Popovs, C. A. Steren, J. Fan, Z. Yang and S. Dai, J. Am. Chem. Soc., 2024, 146, 29588–29598 CrossRef CAS PubMed.
- M. Wang, M. Yabushita, K. Okuma, T. Shono, Y. Nakagawa and K. Tomishige, Green Chem., 2025, 27, 7174–7190 Search PubMed.
- A. J. Kamphuis, F. Picchioni and P. P. Pescarmona, Green Chem., 2019, 21, 406–448 Search PubMed.
- Y. Feng and J. Yao, Sep. Purif. Technol., 2025, 356, 130027 CrossRef CAS.
- A. J. David and A. Das, Green Chem., 2025, 27, 851–862 RSC.
- J. Zhao, Z. Li, P. Wang, P. Miao, R. Shi, G. I. N. Waterhouse, L.-Z. Wu and T. Zhang, Matter, 2024, 7, 1696–1709 CrossRef CAS.
- M. Sain, D. Dey, R. Sen and S. Chowdhury, RSC Sustainability, 2025, 3, 2733–2749 RSC.
- G. Fu, M. Jiang, J. Liu, K. Zhang, Y. Hu, Y. Xiong, A. Tao, Z. Tie and Z. Jin, Nano Lett., 2021, 21, 8824–8830 CrossRef CAS PubMed.
- W. Neamsung, N. Kitjanukit, A. Karawek, N. Chongkol, N. Lertthanaphol, P. Chotngamkhum, K. Khumsupa, P. Phadungbut, W. Jonglertjunya, P. Kim-Lohsoontorn and S. Srinives, RSC Sustainability, 2025, 3, 3520–3529 RSC.
- G. Yang, Q. Wang, Y. Kuwahara, K. Mori and H. Yamashita, Chem Bio Eng., 2024, 1, 289–311 CrossRef CAS PubMed.
- Q. Chen, H. Yu, H. Li, K. Liu and M. Liu, Chem. Commun., 2025, 61, 19403–19420 RSC.
- L. Eisele, B. Hulaj, M. Podsednik, F. Laudani, P. Ayala, A. Cherevan, A. Foelske, A. Limbeck, D. Eder and K. Bica-Schröder, RSC Sustainability, 2024, 2, 2524–2531 RSC.
- Y. Xiong, W. Zhao, D. Gu, Z. Tie, W. Zhang and Z. Jin, Nano Lett., 2023, 23, 4876–4884 Search PubMed.
- L. Qiu, N. Mokhtarinori, H. Liu, D.-e. Jiang, Z. Yang and S. Dai, Mater. Today Energy, 2025, 47, 101740 CrossRef CAS.
- B. Schäffner, F. Schäffner, S. P. Verevkin and A. Börner, Chem. Rev., 2010, 110, 4554–4581 CrossRef PubMed.
- W. Yu, E. Maynard, V. Chiaradia, M. C. Arno and A. P. Dove, Chem. Rev., 2021, 121, 10865–10907 CrossRef CAS PubMed.
- P. Markewitz, W. Kuckshinrichs, W. Leitner, J. Linssen, P. Zapp, R. Bongartz, A. Schreiber and T. E. Müller, Energy Environ. Sci., 2012, 5, 7281–7305 Search PubMed.
- C. Martín, G. Fiorani and A. W. Kleij, ACS Catal., 2015, 5, 1353–1370 Search PubMed.
- L. A. Siddig, R. H. Alzard, H. L. Nguyen and A. Alzamly, Inorg. Chem. Commun., 2022, 142, 109672 Search PubMed.
- H. Eliasson, Y.-T. Chiang, T. P. Araújo, X. Li, R. Erni, S. Mitchell and J. Pérez-Ramírez, Adv. Mater., 2025, 37, 2419859 Search PubMed.
- Y. Ge, A. J. Martín, M. Suvarna, T. Zou, F. Krumeich and J. Pérez-Ramírez, Appl. Catal., B, 2025, 378, 125578 Search PubMed.
- J. Zhang, C. Shang, Z. An, Y. Zhu, H. Song, Z. Chai, X. Shu, L. Zheng and J. He, Angew. Chem., Int. Ed., 2023, 62, e202312068 Search PubMed.
- S. P, J. John, T. P. D. Rajan, G. M. Anilkumar, T. Yamaguchi, S. C. Pillai and U. S. Hareesh, J. Mater. Chem. A, 2023, 11, 8599–8646 RSC.
- J. Koppe, A. V. Yakimov, D. Gioffrè, M.-E. Usteri, T. Vosegaard, G. Pintacuda, A. Lesage, A. J. Pell, S. Mitchell, J. Pérez-Ramírez and C. Copéret, Nature, 2025, 642, 613–619 Search PubMed.
- W. d. A. Bezerra, J. L. S. Milani, C. H. d. J. Franco, F. T. Martins, Â. de Fátima, Á. F. A. da Mata and R. P. das Chagas, Mol. Catal., 2022, 530, 112632 CAS.
- A. Chowdhury, S. Bhattacharjee, R. Chatterjee and A. Bhaumik, J. CO2 Util., 2022, 65, 102236 CrossRef CAS.
- M. Pander, M. Janeta and W. Bury, ACS Appl. Mater. Interfaces, 2021, 13, 8344–8352 Search PubMed.
- F. Hernandez, M. Yang, N. Nagelj, A. Y. Lee, H. Noh, K. P. Hur, X. Fu, C. J. Savoie, A. M. Schwartzberg and J. H. Olshansky, Nanoscale, 2024, 16, 5624–5633 RSC.
- S. Roy, B. Banerjee, A. Bhaumik and S. M. Islam, RSC Adv., 2016, 6, 31153–31160 Search PubMed.
- G. Li, X. Sui, X. Cai, W. Hu, X. Liu, M. Chen and Y. Zhu, Angew. Chem., Int. Ed., 2021, 60, 10573–10576 CrossRef CAS PubMed.
- S. Gratious, E. N. Nahan, R. Jin and S. Mandal, Acc. Mater. Res., 2024, 5, 1291–1302 CrossRef CAS.
- R. R. Shaikh, S. Pornpraprom and V. D'Elia, ACS Catal., 2018, 8, 419–450 Search PubMed.
- S. Bhattacharjee, S. Chongdar, A. Modak, P. Bhanja, B. K. Jena and A. Bhaumik, Green Chem., 2022, 24, 8853–8862 Search PubMed.
- B. Limburg, C. Maquilón and A. W. Kleij, in CO2 as a Building Block in Organic Synthesis, 2020, pp. 1–28, DOI:10.1002/9783527821952.ch1.
- A. Balapure, J. Ray Dutta and R. Ganesan, RSC Appl. Interfaces, 2024, 1, 43–69 RSC.
- A. K. Patra, V. Amoli, A. K. Sinha and D. Kim, ChemCatChem, 2020, 12, 2315–2323 Search PubMed.
- A. K. Patra, N. T. Vo and D. Kim, Appl. Catal., A, 2017, 538, 148–156 CrossRef CAS.
- H. Li, Y. Zhou, W. Tu, J. Ye and Z. Zou, Adv. Funct. Mater., 2015, 25, 998–1013 Search PubMed.
- S. Sarkar, S. A. Ali, S. Sarkar, S. Banerjee, D. Kim and A. K. Patra, ACS Appl. Nano Mater., 2025, 8, 16398–16409 CrossRef CAS.
- S. A. Ali, S. Sarkar, S. Sarkar, S. Banerjee and A. K. Patra, Surf. Interfaces, 2025, 72, 107061 CrossRef CAS.
- S. Sarkar, S. Banerjee, S. Sarkar, S. A. Ali and A. K. Patra, J. Mol. Struct., 2025, 1348, 143414 CrossRef CAS.
- R. Gogoi, P. Garg, P. P. Das, A. K. Rajak, N. Nath, S. Roy and K. Raidongia, Nano Energy, 2024, 131, 110187 CrossRef CAS.
- X. Fu, J. Wang, D. Huang, S. Meng, Z. Zhang, L. Li, T. Miao and S. Chen, ACS Catal., 2016, 6, 957–968 Search PubMed.
- J. Hu, T. Ma, W. Shen, J. Wang, Z. Chen, L. Liang, Y. Zhang, J. Chen and Z. Li, J. Alloys Compd., 2022, 920, 165800 Search PubMed.
- L. Chen, K. Li, Y. Yang, T. Xue, H. Wang, B. Lei, J. Sheng, F. Dong and Y. Sun, J. Hazard. Mater., 2023, 444, 130436 CrossRef CAS PubMed.
- G. K. Dalapati, H. Sharma, A. Guchhait, N. Chakrabarty, P. Bamola, Q. Liu, G. Saianand, A. M. Sai Krishna, S. Mukhopadhyay, A. Dey, T. K. S. Wong, S. Zhuk, S. Ghosh, S. Chakrabortty, C. Mahata, S. Biring, A. Kumar, C. S. Ribeiro, S. Ramakrishna, A. K. Chakraborty, S. Krishnamurthy, P. Sonar and M. Sharma, J. Mater. Chem. A, 2021, 9, 16621–16684 RSC.
- P. Bhanja, B. Mohanty, A. K. Patra, S. Ghosh, B. K. Jena and A. Bhaumik, ChemCatChem, 2019, 11, 583–592 CrossRef CAS.
- L. Du, K. Gu, M. Zhu, J. Zhang and M. Zhang, Sens. Actuators, B, 2019, 288, 298–306 Search PubMed.
- S. Sarkar, S. Sarkar and A. K. Patra, ChemNanoMat, 2022, 8, e202200294 CrossRef CAS.
- S. Banerjee, S. Sarkar, S. Sarkar and A. K. Patra, Inorg. Chem. Commun., 2024, 160, 111925 Search PubMed.
- S. Paul, S. Basak and W. Ali, ACS Omega, 2019, 4, 21827–21838 Search PubMed.
- X. Lian, Z. Chen, X. Yu, T. Fan, Y. Dong, H. Zhai, W. Fang and X. Yi, Nanoscale, 2019, 11, 9444–9456 RSC.
- L. Mohapatra, M. Nagaraju, S. Suman and A. K. Kushwaha, J. Alloys Compd., 2022, 928, 167127 CrossRef CAS.
- W. Marynowski, M. Saski, B. Pałuba, M. Pisarek and J. Lewiński, ACS Appl. Electron. Mater., 2022, 4, 2253–2263 CrossRef CAS.
- L. Dai, C. Strelow, T. Kipp, A. Mews, I. Benkenstein, D. Eifler, T. H. Vuong, J. Rabeah, J. McGettrick, R. Lesyuk and C. Klinke, Chem. Mater., 2021, 33, 275–284 Search PubMed.
- B. Chen, Y. Duan, J. Tan, Y. Li, S. Li and D. Zhang, Adv. Funct. Mater., 2025, 35(19), 2425956 CrossRef CAS.
- L. Lu, Y. Cheng, Z. Liang, S. Yan, G. Qiao and Z. Zou, Inorg. Chem., 2023, 62, 2934–2941 Search PubMed.
- Y. Li, B. Chen, P. Ouyang, Y. Duan, W.-J. Ong and F. Dong, Chem. Eng. J., 2023, 451, 138695 CrossRef CAS.
- S. A. Ali, S. Sarkar and A. K. Patra, ACS Appl. Mater. Interfaces, 2024, 16, 38061–38072 CrossRef CAS PubMed.
- Q. Yang, H. Peng, Q. Zhang, X. Qian, X. Chen, X. Tang, S. Dai, J. Zhao, K. Jiang, Q. Yang, J. Sun, L. Zhang, N. Zhang, H. Gao, Z. Lu and L. Chen, Adv. Mater., 2021, 33, 2103186 CrossRef CAS PubMed.
- Y. Wu, X. Song, S. Xu, Y. Chen, O. Oderinde, L. Gao, R. Wei and G. Xiao, Dalton Trans., 2020, 49, 312–321 RSC.
- K. Wu, T. Su, D. Hao, W. Liao, Y. Zhao, W. Ren, C. Deng and H. Lü, Chem. Commun., 2018, 54, 9579–9582 RSC.
- C. Liu, H. Niu, D. Wang, C. Gao, A. Said, Y. Liu, G. Wang, C.-H. Tung and Y. Wang, ACS Catal., 2022, 12, 8202–8213 CrossRef CAS.
- H. Liu, J. Xia, N. Zhang, H. Cheng, W. Bi, X. Zu, W. Chu, H. Wu, C. Wu and Y. Xie, Nat. Catal., 2021, 4, 202–211 CrossRef CAS.
- H. Liu, H. Wang, Q. Song, K. Küster, U. Starke, P. A. van Aken and E. Klemm, Angew. Chem., Int. Ed., 2022, 61, e202117058 CrossRef CAS PubMed.
- W. Zhang, Y. Yan, J. Wang, Z. Yang, T. Li, H. Li, S. Yan, T. Yu, W. Fan and Z. Zou, Dalton Trans., 2023, 52, 7129–7135 RSC.
- Y. Wang, H. Liu, Q. Shi, Z. Miao, H. Duan, Y. Wang, H. Rong and J. Zhang, Angew. Chem., Int. Ed., 2024, 63, e202404911 CrossRef CAS PubMed.
- Q. Li, H. Yang, M. Li, K. Zhao, B. Duan, C. Zhang, H. Wang, C. Qiao, H. Chang and T. Lin, Ind. Eng. Chem. Res., 2020, 59, 8136–8144 Search PubMed.
- W. Natongchai, S. Posada-Pérez, C. Phungpanya, J. A. Luque-Urrutia, M. Solà, V. D'Elia and A. Poater, J. Org. Chem., 2022, 87, 2873–2886 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |







