Catalytic aerobic oxidation of 5-hydroxymethylfurfural to 2,5-furandicarboxylic acid using Co–Mn/N–C catalysts in aqueous media
Received
5th October 2025
, Accepted 25th March 2026
First published on 26th March 2026
Abstract
Aerobic oxidation of 5-hydroxymethylfurfural (HMF) can yield numerous furan-based chemicals, and enhancing the selectivity for specific products poses a significant challenge. In this study, a novel non-noble bimetallic catalyst supported on N-doped carbon (Co–Mn/N–C) was designed and synthesized, and various characterization techniques, including XRD, BET analysis, TEM, XPS and VSM studies, were employed to investigate the catalyst's structure and properties. Catalytic testing studies demonstrated that Co–Mn/N–C exhibits superior catalytic performance in the selective oxidation of HMF to 2,5-furandicarboxylic acid (FDCA) in aqueous media, outperforming both Co/N–C and Mn/N–C catalysts. Nearly 100% HMF conversion and 98.3% selectivity towards FDCA were achieved using the Co–Mn/N–C catalyst at 150 °C with Na2CO3 (0.5 mmol) as a base. Furthermore, the Co–Mn/N–C catalyst exhibited good stability, maintaining its catalytic activity throughout the recycling experiments. This stable performance is attributed to the robust interaction between the metal and nitrogen-carbon materials.
Sustainability spotlight
As humanity's rapid development continues, the once-abundant reserves of fossil resources are steadily dwindling, intensifying concerns over the long-term sustainability of energy and raw material supplies. In this study, the oxidation of 5-hydroxymethylfurfural (HMF) to furan-based chemicals was achieved under an O2 atmosphere in water using Co–Mn/N–C catalysts. This approach offers a promising strategy for developing non-precious-metal catalytic processes under mild conditions. The findings presented here are expected to significantly advance the field of biomass conversion, aligning with the United Nations Sustainable Development Goals 12 (responsible consumption and production).
|
1 Introduction
As human society continues its rapid development, the once-plentiful reserves of fossil resources are gradually diminishing, thereby raising a pressing concern for the sustainable future of energy and raw material supplies.1 The continuing decline in the supply of fossil resources highlights the urgent need to explore alternative resources to meet the growing needs of an evolving human civilization.2 Biomass resources, the most abundant renewable resource on the earth, have garnered significant attention from the perspective of scientific research and industrial applications.3–6 Among the biomass-based platform chemicals, 5-hydroxymethylfurfural (HMF) can be further converted into a series of various fine chemicals with promising application prospects via esterification,7,8 hydrogenation9–12 and oxidation.13–22 As shown in Scheme 1, there are several industrially valuable furan compounds such as 2,5-diformylfuran (DFF), 5-formyl-2-furancarboxylic acid (FFCA), 5-hydroxymethyl-2-furancarboxylic acid (HFCA) and 2,5-furandicarboxylic acid (FDCA), which are oxidized from HMF.15,23–28 As an oxidation product of HMF, FDCA possesses two carboxylic acid groups, making it akin to terephthalic acid (TPA). In addition, FDCA can be utilized in the production of polyethylene furanoate (PEF) polymers, a bio-based polymer that can serve as a sustainable alternative to polyethylene terephthalate (PET).29,30 Besides, FDCA, as a promising furan chemical, can be further used for synthesizing various derivatives such as succinic acid, 2,5-dihydroxymethylfuran, 2,5-dihydroxymethyl tetrahydrofuran, and 2,5-furandicarbaldehyde.31 Considering the promising potential of FDCA, numerous endeavors have been undertaken to devise effective chemocatalytic approaches to efficiently convert HMF into FDCA.32 Due to the variability in oxidation products resulting from variations in the degree of HMF oxidation, achieving high selectivity in the production of FDCA remains a formidable challenge for most researchers.33,34
 |
| | Scheme 1 Reaction pathways for HMF oxidation. | |
In recent years, there have been many studies focusing on the development of non-noble metal catalysts for the catalytic oxidation of HMF to produce FDCA.16,35–37 Transition metals typically exhibit tunable pH adaptability, redox capabilities, crystal defects, and diverse oxygen species. This is attributed to the variable three-dimensional electron orbital configurations of transition metals. These characteristics can effectively improve the selectivity of desired products of the oxidation reaction.38 Transition metals such as manganese (Mn), cobalt (Co), and iron (Fe) were investigated as promising candidates for the catalytic oxidation of HMF. This may be motivated by the desire to harness the unique properties of these metals for targeted oxidation processes.39,40 For example, Han et al. prepared a MnOx-CeO2 bimetal oxide catalyst and applied it in the aerobic oxidation of HMF to prepare FDCA, and the yield of FDCA was as high as 91.0%, and the catalyst could be reused 5 times.41 Lu et al. introduced Fe3O4 into α-MnO2 to form complex metal oxides, and studied the oxidation behavior of these Mn–Fe oxides catalyzing HMF. Among them, Mn8Fe3Ox showed the best catalytic performance for HMF oxidation to synthesize FDCA.42 Under the optimal conditions, HMF can be completely transformed, and the yield of FDCA was 76.9%. Yang et al. synthesized nano-spherical mesoporous silica (NS-MS) with modification by bimetal (Co–Mn) oxides via a modified micelle template method.43 The HMF conversion rate was close to 100%, and the yield of FDCA was up to 72.4%. Despite the significant progress made in the utilization of non-precious metal catalysts for the oxidation of HMF, the quest for achieving high selectivity towards the desired products under conditions that are both mild and environmentally benign remains a challenge that necessitates further research and development.44
Based on our recent work toward oxidation catalysts for HMF conversion,45–47 we herein synthesize a novel rod-like magnetic Co–Mn/N–C bimetal supported on N-doped carbon, using citric acid, melamine, MnSO4 and CoCl2 as precursors. Characterization techniques, including XRD, nitrogen adsorption–desorption studies, XPS, and TEM, were employed to analyze the structure of the catalyst. As expected, the Co–Mn/N–C catalyst was successfully obtained. The catalytic performance of this catalyst in the oxidation process for the production of FDCA from HMF in water was evaluated. In addition, the effect of various reaction parameters on the oxidation of HMF was investigated. A HMF conversion of nearly 100% with an FDCA selectivity of 98.3% was achieved under optimal conditions.
2 Experimental section
2.1 Chemicals
5-Hydroxymethylfurfural (97.0%), 2,5-furandicarboxylic acid (98.0%), CoCl2·6H2O (≥99.0%) and melamine (≥99.0%) were purchased from Shanghai Macklin Biochemical Co., Ltd (Shanghai, China). Citric Acid (≥99.5%), MnSO4·H2O (≥99.0%), NaHCO3 (≥99.5%) and Na2CO3 (≥99.8%) were obtained from Sinopharm Group Co., Ltd (Shanghai, China).
2.2 Synthesis of Co–Mn/N–C
The catalysts were prepared according to the procedure presented in Scheme 2. Citric acid primarily acts as a chelating agent to coordinate with Co2+ and Mn2+ ions, and melamine is employed as a nitrogen-rich precursor for the N-doped carbon support. Firstly, CoCl2·6H2O, MnSO4·H2O, citric acid and melamine were dissolved in deionized water at 100 °C for 3 h. Subsequently, the mixture was washed and filtered, followed by drying in an oven at 100 °C. The resulting sample was calcined at 700 °C for 3 h, yielding the product denoted as Co–Mn/N–C. Under identical operational conditions, Co/N–C and Mn/N–C were synthesized. The Co/N–C catalyst was derived from a precursor mixture comprising CoCl2·6H2O, citric acid, and melamine. Similarly, the Mn/N–C catalyst was prepared from a precursor mixture comprising MnSO4·H2O, citric acid, and melamine.
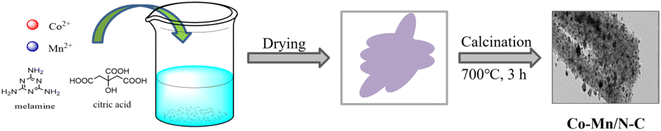 |
| | Scheme 2 Schematic of the synthesis steps for the Co–Mn/N–C catalyst. | |
2.3 Characterizations techniques
The catalyst morphology was characterized by transmission electron microscopy (TEM, JEM 2100F). Thermogravimetry-differential thermal analysis (TG-DTG, Germany) curves were recorded in an N2 flow on a Netzsch Model STA 409 PC instrument using α-Al2O3 as the standard material. Data were recorded at a temperature range from room temperature to 800 °C with a heating rate of 10 °C min−1. X-ray diffraction (XRD) studies were carried out using a Bruker diffractometer with Cu Kα radiation to survey the crystal structure and diffraction angles (2θ) ranging from 10° to 80°. The magnetic properties were probed using a Lake Shore superconducting vibrating sample magnetometer (VSM). XPS was conducted using a Thermo Scientific K-Alpha+ spectrometer, and the XPS Peak 4.1 software was utilized to fit the curves using the Shirley-type background subtraction.
2.4 HMF oxidation reaction
The catalytic oxidation reaction of HMF was carried out according to the following procedure: HMF (0.5 mmol) was dissolved in 10 mL of water, while the catalyst (20 mg) and Na2CO3 (0.5 mmol) were added. At 5 bar O2 pressure, the reaction was carried out at 100 °C for 6 h in an autoclave with a Teflon liner. After the reaction, the reactor was cooled to room temperature. The reaction mixture was further analyzed using an Agilent 1260 HPLC system equipped with a Venusil XBP C18 chromatographic column (4.6 × 250 mm, 5 µm, Phenomenex, USA) and a UV-Vis (280 nm) detector. Quantification was carried out using the external standard method. The mobile phase consisted of an acetic acid solution (0.1 wt%) and acetonitrile with a volume ratio of 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, and the samples were eluted at a flow rate of 0.7 mL min−1 at 30 °C. Typical HPLC results and 1H NMR analysis for HMF oxidation products are shown in Fig. S1 and S2 in the SI. The HMF conversion and the selectivity of the main products were calculated using the following equations:
5, and the samples were eluted at a flow rate of 0.7 mL min−1 at 30 °C. Typical HPLC results and 1H NMR analysis for HMF oxidation products are shown in Fig. S1 and S2 in the SI. The HMF conversion and the selectivity of the main products were calculated using the following equations:
| HMF conversion = (1 − moles of HMF/added moles of HMF) × 100% |
| FDCA selectivity = moles of FDCA/(added moles of HMF − moles of HMF) × 100% |
| HFCA selectivity = moles of HFCA/(added moles of HMF − moles of HMF) × 100% |
| FFCA selectivity = moles of FFCA/(added moles of HMF − moles of HMF) × 100% |
3 Results and discussion
3.1 Catalyst characterization
The X-ray diffraction (XRD) spectra of Co–Mn/N–C, Mn/N–C, and Co/N–C are shown in Fig. 1. Co–Mn/N–C, Co/N–C, and Mn/N–C showed a wide peak centered at a 2θ value of 26°, which was attributed to the (002) reflection plane graphitic carbon (JCPDS 41-1487).48 In Fig. 1, the peaks at about 44.2°, 51.5° and 75.9° correspond to the (111), (200) and (220) lattice planes, respectively, of the cubic Co phase (JCPDS 15-0806).49 In the XRD patterns of Mn/N–C, the peaks at 2θ values of 34.9°, 40.5°, 58.7°, 70.2° and 73.8° were attributed to the characteristic peaks of MnO.50 The addition of a low content of Mn improves the dispersion of the Co–Mn phase. It was noted that Mn species were not observed in the Co–Mn/N–C spectra due to the Mn elements being homogenously dispersed on the Co–Mn/N–C catalyst.
 |
| | Fig. 1 XRD spectra of Co–Mn/N–C, Co/N–C, and Mn/N–C. | |
TEM analysis was employed to further investigate the morphology of the Co–Mn/N–C catalyst, as depicted in Fig. 2. As shown in Fig. 2a, the Co–Mn/N–C catalyst exhibits a rod-like structure. Many Co nanoparticles were randomly embedded in the carbon layer, as observed in Fig. 2b. Fig. 2d represents an enlarged portion of Fig. 2c. The measured lattice spacing of 0.29 nm and 0.14 nm corresponds to the (002) crystal plane of graphite carbon and the (200) crystal plane of cobalt, respectively (Fig. 2d). The elemental distribution of Mn, Co, N, and C on the Co–Mn/N–C catalyst was investigated and visually represented using distinct colors, as depicted in Fig. 3a. As illustrated in Fig. 3b, some agglomeration of metallic Co was observed. This observation is consistent with the presence of diffraction peaks associated solely with metal Co in the XRD spectra. Both Mn and N elements exhibited a uniform distribution on the carbon support. This phenomenon may be attributed to the relatively low content of Mn. Furthermore, the strong coordination between Mn and N may effectively avoid the aggregation of Mn atoms, resulting in a homogeneous dispersion on the Co–Mn/N–C catalyst.51
 |
| | Fig. 2 HRTEM images of Co–Mn/N–C at different resolutions of (a) 200 nm, (b) 50 nm, and (c) 5 nm and (d) the corresponding zoomed-in area. | |
 |
| | Fig. 3 Elemental mappings of Co–Mn/N–C: (a) overall, (b) Co, (c) C, (d) N and (e) Mn. | |
The magnetic property of the Co–Mn/N–C catalyst was measured by a VSM at a magnetic field of ±1.5 T/MH at room temperature, and the results are shown in Fig. 4. From Fig. 4, the magnetic hysteresis curve the coercivity field (Hc) and saturation magnetization (Ms) values were 436.1 G and 15.4 emu per g, respectively. The results indicate that Co–Mn/N–C belongs to hard magnetic materials, suggesting that it can be used as a magnetic-supported catalyst.
 |
| | Fig. 4 VSM curve of Co–Mn/N–C. | |
The N2 adsorption–desorption isotherms of Co/N–C, Mn/N–C, and Co–Mn/N–C were investigated. In Fig. 5a, N2 adsorption isotherms of both Co/N–C and Co–Mn/N–C are type IV according to the IUPAC classification, indicating the existence of mesoporous structures. Moreover, N2 adsorption isotherms of Co/N–C and Co–Mn/N–C did not show any adsorption limitation at high relative pressure, indicating a type H3 hysteresis loop, which may be caused by the slit pores of the catalyst. Table 1 reveals that Mn/N–C exhibits the lowest specific surface area and pore volume (10.0 m2 g−1 and 0.03 cm3 g−1, respectively), whereas Co/N–C possesses significantly higher values (239.1 m2 g−1 and 0.36 cm3 g−1). The primary reason for the very low surface area and pore volume of Mn/N–C is likely due to the inability of manganese to facilitate the formation of a porous carbon framework during pyrolysis unlike cobalt. Notably, Co–Mn/N–C exhibits a larger specific surface area and pore volume, reaching 210.7 m2 g−1 and 0.25 cm3 g−1, respectively. A greater specific surface area and higher pore volume are advantageous for facilitating contact between the catalyst and reactants, thus enhancing the catalytic activity of the catalyst.52
 |
| | Fig. 5 N2 adsorption–desorption isotherm (a) and pore size distribution curves (b) of Co–Mn/N–C, Co/N–C, and Mn/N–C. | |
Table 1 Surface area and pore volume of the Mn/N–C, Co/N–C and Co–Mn/N–C catalysts
| Entry |
Catalyst |
S
BET (m2 g−1) |
V
p (cm3 g−1) |
S
p (nm) |
| 1 |
Mn/N–C |
10.0 |
0.03 |
16.2 |
| 2 |
Co/N–C |
239.1 |
0.36 |
7.7 |
| 3 |
Co–Mn/N–C |
210.7 |
0.25 |
6.7 |
XPS characterization was further performed to investigate the composition and valence states of elements. Elemental signals of C, N, O, Mn and Co are observed in the XPS survey spectrum of Co–Mn/N–C (Fig. 6a), whereas only C, N, O and Co are detected for Co/N–C (Fig. S3), and C, N, O and Mn for Mn/N–C (Fig. S4). The fitted peak at 778.3 eV observed in the high-resolution spectrum of Co 2p3/2 (Fig. 6b) indicates the presence of Co0, while the peak at 779.6 eV corresponds to Co–Nx with a pair of satellite peaks at 782.2 and 803.2 eV.53 It is worth noting that the Co 2p binding energies of Co–Mn/N–C exhibit a slight shift compared with those of Co/N–C, attributable to the electronic-structure modulation induced by Mn introduction (Fig. S5). Fig. 6c shows the high-resolution spectra of Mn 2p in Co–Mn/N–C catalysts, and the Mn 2p3/2 peak at 641.3 eV is attributed to the Mn–Nx moieties.51 Similarly, the binding energy of Mn in Co–Mn/N–C differs from that in Mn/N–C due to the presence of Co (Fig. S6). The O 1s high-resolution spectra were further analyzed to explore the surface oxygen species (Fig. 6d). The peak at ca. 529.6 eV corresponds to lattice oxygen provided by metal–oxygen bonds. The peak at a binding energy of 531.1 eV is attributed to oxygen-related defects and consequent adsorbed oxygen, including free oxygen molecules, which act directly in catalytic reactions on the surfaces/interfaces of different metal elements and carbon. The peak at a binding energy of 532.8 eV indicates the presence of surface oxygen species with low reactivity. In the C 1s spectrum (Fig. 6e), the three peaks at the binding energies of 284.6, 286.1 and 287.4 eV belong to C–C, C–N and C–O bonds, respectively. The presence of the C–N bond in the catalyst implies the possibility of rapid electron transfer during the reaction, thereby enhancing the catalytic activity of the catalyst.54 The high-resolution spectrum of N 1s (Fig. 6f) can be divided into three peaks corresponding to pyridinic N (398.4 eV), pyrrolic N (399.7 eV) and graphitic N (400.7 eV).55 These results prove the successful integration of Mn and N into the carbon matrix.
 |
| | Fig. 6 XPS spectra for Co–Mn/N–C: (a) survey scan; (b) Co 2p; (c) Mn 2p; (d) O 1s; (e) C 1s; and (f) N 1s. | |
3.2 Catalytic evaluation of the Co–Mn/N–C catalysts
As presented in Table 2, all the samples demonstrated good catalytic activity in the conversion of HMF, producing HFCA and FFCA as the main products. The monometallic Mn/N–C catalyst primarily facilitates the oxidation of the alcohol group, leading to high FFCA selectivity (Table 2, Entry 1). In contrast, the Co/N–C catalyst shows a greater tendency to activate molecular oxygen, promoting further oxidation toward FDCA (Table 2, Entry 2). Notably, the Co–Mn/N–C catalyst exhibits superior activity and FDCA selectivity (Table 2, Entry 3), likely due to a synergistic interaction between the Co and Mn sites that enhances oxygen activation and facilitates the sequential oxidation pathway from HMF to FDCA. 78.0% HMF conversion and 9.5% FDCA selectivity were achieved using Na2CO3 as the base, and the carbon balance of the reaction reached 99.3%. More importantly, the addition of a base was also crucial, as evidenced by the low conversion (19.6%) in its absence (Table 2, Entry 4). Different bases, such as NaOH and NaHCO3, were further screened under the same reaction conditions. It was observed that more FFCA and less FDCA were produced under strong alkaline conditions (Table 2, Entry 5). This suggests that strong alkalinity efficiently promotes the oxidation of the alcohol group to form FFCA, but may inhibit the final step of FFCA oxidation to FDCA potentially due to excessive deprotonation of the reaction intermediates or catalyst surface. However, the conversion of HMF decreased significantly when the weaker base, NaHCO3 was added (Table 2, Entry 6). Therefore, Na2CO3 as a suitable base was further investigated for the oxidation of HMF over the Co–Mn/N–C catalyst. The by-product DFF was not detected in the reaction solutions for any of the three catalysts, underscoring that the oxidation of HMF predominantly proceeds via the aldehyde group pathway (HMF → HFCA → FFCA → FDCA). The possible reaction pathway for HMF oxidation is proposed, as shown in Scheme 3. Notably, FFCA accumulates with high selectivity, which indicates that the oxidation step (FFCA → FDCA) is rate-limiting. Based on these findings, Co–Mn/N–C was selected for further investigation.
Table 2 Oxidation of HMF in the presence of various catalysts and bases in watera
| Entry |
Catalyst |
Base |
HMF conv. (%) |
Sel. (%) |
| HFCA |
FFCA |
FDCA |
Others |
|
Reaction conditions: 0.5 mmol HMF, 0.5 mmol base, 20 mg catalyst, 10 mL H2O, 6 h, 100 °C, and 0.5 MPa O2.
|
| 1 |
Mn/N–C |
Na2CO3 |
62.8 |
25.0 |
56.5 |
0 |
18.5 |
| 2 |
Co/N–C |
Na2CO3 |
62.3 |
22.3 |
64.2 |
5.3 |
8.2 |
| 3 |
Co–Mn/N–C |
Na2CO3 |
78.0 |
20.0 |
69.4 |
9.5 |
1.1 |
| 4 |
Co–Mn/N–C |
None |
19.6 |
40.2 |
47.3 |
0 |
12.5 |
| 5 |
Co–Mn/N–C |
NaOH |
63.6 |
37.4 |
55.0 |
4.5 |
3.1 |
| 6 |
Co–Mn/N–C |
NaHCO3 |
43.7 |
18.1 |
75.3 |
3.6 |
3.0 |
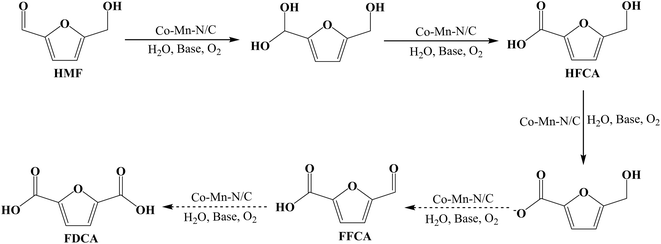 |
| | Scheme 3 Possible pathway for the HMF oxidation catalyzed by the Co–Mn/N–C catalyst. | |
The effect of Na2CO3 amount on HMF oxidation was also studied, and the results are shown in Fig. 7. The HMF conversion increased with the increase in the amount of Na2CO3, up to 0.5 mmol. It was clearly observed that the presence of Na2CO3 can facilitate the oxidation reaction of HMF, which probably increases the pH of the solution, thus favoring the oxidation of the aldehyde groups of HMF.56 However, with a further increase in the amount of Na2CO3, the FDCA selectivity decreased, which could be explained by the generation of by-products such as other furan-based intermediates and/or humins in the reaction mixture.
 |
| | Fig. 7 Effects of the Na2CO3 amount on the HMF oxidation over Co–Mn/N–C in water. Reaction conditions: 0.5 mmol HMF, 100 °C, 20 mg Co–Mn/N–C, 10 mL H2O, 6 h, and 0.5 MPa O2. | |
The effect of Co–Mn/N–C dosage on HMF oxidation was studied. In Fig. 8, as expected, the conversion of HMF exhibits a gradual increase with the increase in the catalyst amount. Upon incrementing the catalyst dosage from 20 mg to 70 mg, a discernible decrease in the selectivity of the intermediate products, 5-hydroxymethyl-2-furancarboxylic acid (HFCA) and 5-formyl-2-furancarboxylic acid (FFCA), was observed. The selectivity for 2,5-furandicarboxylic acid (FDCA) exhibited a consistent and progressive enhancement. At a catalyst dosage of 70 mg, the conversion of HMF and the selectivity towards FDCA reached a maximum of 99.6% and 50.4%, respectively. Based on the above results, a catalyst dosage of 70 mg was opted for subsequent experimental studies.
 |
| | Fig. 8 Effects of the catalyst amount on the HMF oxidation in water. Reaction conditions: 0.5 mmol HMF, 0.5 mmol Na2CO3, 100 °C, 6 h, 10 mL H2O, and 0.5 MPa O2. | |
The effect of temperature on HMF oxidation was investigated between 100 and 150 °C, and the results are displayed in Fig. 9. It was observed that complete conversion of HMF was achieved at 110 °C. In addition, the selectivity for 2,5-furandicarboxylic acid (FDCA) increased considerably with rising temperature, giving 98.3% selectivity for FDCA at 150 °C. Furthermore, during the reaction process, the intermediate products HFCA and FFCA were detected, whereas the by-product DFF was not detected. Meanwhile, the detected intermediate products, FFCA and HFCA, decreased with increasing temperature. This phenomenon can be explained by the further oxidation of both FFCA and HFCA to FDCA at high reaction temperatures.
 |
| | Fig. 9 Effects of temperature on HMF oxidation over Co–Mn/N–C in water. Reaction conditions: 0.5 mmol HMF, 0.5 mmol Na2CO3, 70 mg Co–Mn/N–C, 10 mL H2O, 0.5 MPa O2, and 6 h. | |
The recyclability of the catalyst is very important for HMF oxidation in water. The Co–Mn/N–C catalyst can be easily separated using magnets after each use owing to the magnetic properties of the catalyst. As presented in Fig. 10, the selectivity of FDCA decreased from 97.1% to 66.2% after the first use. The ICP-OES analysis showed that no Co or Mn element was detected in the solution after the reaction, indicating that the Co and Mn elements on the catalyst were stable. The used catalyst was further characterized by XRD to compare with the fresh catalyst, and a new diffraction peak at 36.8°, which belonged to Co3O4, was observed (Fig. S7), which may lead to a decrease in the catalyst activity. Therefore, the catalyst was calcined in a H2 atmosphere before the next use. Notably, the catalytic activity of Co–Mn/N–C could be partly restored, giving relatively acceptable selectivity towards FDCA.
 |
| | Fig. 10 Recyclability experiments of Co–Mn/N–C. Reaction conditions: 0.5 mmol HMF, 0.5 mmol Na2CO3, 70 mg Co–Mn/N–C, 10 mL H2O, 0.5 MPa O2, 6 h, and 150 °C. | |
The catalytic performances of various catalysts for the selective oxidation of HMF to FDCA were further compared using O2 as an oxidant in an aqueous medium. As shown in Fig. 11, manganese dioxides with different crystal structures, such as β-MnO2-HS57 and Mn2O3 nanoflakes,58 show high activity for the aerobic oxidation of HMF. Moreover, bimetals of Co and Mn, such as MnCo2O4,59 Co–Mn-0.25 (ref. 60) and Co-MnOx,61 can be rationally designed and prepared and exhibit high effectiveness for the oxidation of HMF into FDCA. It can be found that the Co–Mn/N–C catalyst exhibits a relatively high HMF conversion and satisfactory FDCA selectivity under mild conditions.
 |
| | Fig. 11 Comparison of various catalysts for the selective oxidation of HMF to FDCA. | |
4 Conclusion
A novel rod-like magnetic catalyst, Co–Mn/N–C, was obtained using citric acid, melamine, manganese sulfate, and cobalt chloride as precursors through a calcination process at 700 °C. Co–Mn/N–C catalyst shows excellent catalytic performance for the oxidation of HMF into FDCA in water. Under optimal reaction conditions, HMF could be completely converted and the selectivity of FDCA could reach as high as 98.3%. Additionally, the magnetic properties of the catalyst facilitated easy separation from the catalytic system. This study provides a significant and challenging effort in the utilization of non-noble metal catalysts supported on N–C materials for the conversion of HMF into FDCA using molecular oxygen as the oxidant.
Author contributions
Shuolin Zhou: investigation, visualization, writing – reviewing and editing, project administration, funding acquisition. Wen Sha: investigation, data curation, formal analysis, visualization. Ying Liu: investigation, visualization. Xianxiang Liu: conceptualization, methodology, project administration, funding acquisition, supervision.
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability
Data supporting this article have been included as part of the supplementary information (SI) with the original data available from the corresponding authors on request. Supplementary information: details of the HPLC results, 1H NMR analysis, XPS and XRD. See DOI: https://doi.org/10.1039/d5su00783f.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 22278121), the Hunan Provincial Natural Science Foundation of China (Grant No. 2026JJ81172 and 2026JJ50394), and the Scientific Research Fund of the Hunan Provincial Education Department (Grant No. 22B1095).
References
- P. A. Østergaard, N. Duic, Y. Noorollahi and S. Kalogirou, Renewable Energy, 2022, 199, 1145–1152 Search PubMed.
- S. Biswas, B. Dutta, A. Mannodi-Kanakkithodi, R. Clarke, W. Song, R. Ramprasad and S. L. Suib, Chem. Commun., 2017, 53, 11751–11754 RSC.
- J. Nie and H. Liu, Pure Appl. Chem., 2011, 84, 765–777 Search PubMed.
- R. Fang, R. Luque and Y. Li, Green Chem., 2016, 18, 3152–3157 RSC.
- R. Mori, RSC Sustain., 2023, 1, 179–212 RSC.
- F. Brienza, D. Cannella, D. Montesdeoca, I. Cybulska and D. P. Debecker, RSC Sustain., 2024, 2, 37–90 RSC.
- A. He, Q. Gu, X. Shen, J. Zheng, L. Hu, X. Wang, Y. Jiang, Z. Wu, J. Xu and J. Song, Green Chem., 2023, 25, 2349–2360 RSC.
- L. G. Tonutti, B. O. Dalla Costa, G. Mendow, G. L. Pestana, N. S. Veizaga and J. M. Grau, Micropor. Mesopor. Mater., 2022, 343, 112145 CrossRef CAS.
- W. Zhao, X. Zhu, Z. Zeng, J. Lei, Z. Huang, Q. Xu, X. Liu and Y. Yang, Mol. Catal., 2022, 524, 112304 CAS.
- Z. Huang, Z. Zeng, X. Zhu, W. Zhao, J. Lei, Q. Xu, Y. Yang and X. Liu, Front. Chem. Sci. Eng., 2023, 17, 415–424 CrossRef CAS.
- W. Zhao, Z. Huang, L. Yang, X. Liu, H. Xie and Z. Liu, Appl. Surf. Sci., 2022, 577, 151869 CrossRef CAS.
- R. Guo, Y. Zeng, L. Lin, D. Hu, C. Lu, S. Conroy, S. Zhang, C. Zeng, H. Luo, Z. Jiang, X. Zhang, X. Tu and K. Yan, Angew. Chem., Int. Ed., 2025, 64, e202418234 CrossRef CAS PubMed.
- C. A. Antonyraj, N. T. T. Huynh, K. W. Lee, Y. J. Kim, S. Shin, J. S. Shin and J. K. Cho, J. Chem. Sci., 2018, 130, 156 CrossRef.
- T. K. T. Le, S. Kongparakul, H. Zhang, J. Zhao, G. Guan, N. Chanlek, T. T. V. Tran and C. Samart, Mol. Catal., 2023, 539, 113017 CAS.
- C. A. Celaya, R. Oukhrib, M. A. El Had, Y. Abdellaoui, H. A. Oualid, H. Bourzi, R. Chahboun, D. Zhao, S. M. Osman, V. S. Parmar and C. Len, Mol. Catal., 2022, 519, 112117 CAS.
- J. Lai, F. Cheng, S. Zhou, S. Wen, D. Guo, W. Zhao, X. Liu and D. Yin, Appl. Surf. Sci., 2021, 565, 150479 CrossRef CAS.
- X. Liu, J. Xiao, H. Ding, W. Zhong, Q. Xu, S. Su and D. Yin, Chem. Eng. J., 2016, 283, 1315–1321 Search PubMed.
- J. Lai, S. Zhou, F. Cheng, D. Guo, X. Liu, Q. Xu and D. Yin, Catal. Lett., 2020, 150, 1301–1308 Search PubMed.
- J. Han, M. Song, Y. Li, Y. Yao, S. Lu and X. Liao, React. Chem. Eng., 2024, 9, 148–159 RSC.
- J. Li, G. Wang, X. Wang, Y. Zhao, Y. Zhao, W. Sui, D. Wang and C. Si, ACS Catal., 2024, 14, 16003–16013 CrossRef CAS.
- K. Zhao, B. Wen, Q. Tang, F. Wang, X. Liu, Q. Xu and D. Yin, Green Chem., 2024, 26, 9957–9992 Search PubMed.
- Z. Zheng, K. Li, L. Lin, Z. Jiang, Y. Wang and K. Yan, Green Energy Environ., 2025, 10, 898–916 Search PubMed.
- H. Zhang, Z. Feng, Y. Zhu, Y. Wu and T. Wu, J. Photochem. Photobio. A: Chem., 2019, 371, 1–9 CrossRef CAS.
- C. Chang, X. Ma and P. Cen, Chin. J. Chem. Eng., 2006, 14, 708–712 Search PubMed.
- M. Ventura, M. Aresta and A. Dibenedetto, ChemSusChem, 2016, 9, 1096–1100 CrossRef CAS PubMed.
- P. Pan, M. Li, Y. Liu, Y. Feng and X. Li, Mol. Catal., 2022, 530, 112622 CAS.
- Z. Zeng, L. Yang, X. Zhu, W. Zhao, X. Liu, Z. Huang, Q. Xu and W. Zhong, React. Chem. Eng., 2023, 8, 455–464 Search PubMed.
- B. S. Solanki and C. V. Rode, J. Saudi Chem. Soc., 2019, 23, 439–451 CrossRef CAS.
- H. Zhou, H. Xu, X. Wang and Y. Liu, Green Chem., 2019, 21, 2923–2927 RSC.
- L. Gao, K. Deng, J. Zheng, B. Liu and Z. Zhang, Chem. Eng. J., 2015, 270, 444–449 CrossRef CAS.
- M. Sajid, X. Zhao and D. Liu, Green Chem., 2018, 20, 5427–5453 RSC.
- G. Totaro, L. Sisti, P. Marchese, M. Colonna, A. Romano, C. Gioia, M. Vannini and A. Celli, ChemSusChem, 2022, 15, e202200501 CrossRef CAS PubMed.
- D. Zhao, T. Su, Y. Wang, R. S. Varma and C. Len, Mol. Catal., 2020, 495, 111133 CAS.
- C. Chen, L. Wang, B. Zhu, Z. Zhou, S. I. El-Hout, J. Yang and J. Zhang, J. Energy Chem., 2021, 54, 528–554 CrossRef CAS.
- E. Hayashi, T. Komanoya, K. Kamata and M. Hara, ChemSusChem, 2017, 10, 654–658 CrossRef CAS PubMed.
- F. Cheng, D. Guo, J. Lai, M. Long, W. Zhao, X. Liu and D. Yin, Front. Chem. Sci. Eng., 2021, 15, 960–968 CrossRef CAS.
- F. Wang, J. Lai, Z. Liu, S. Wen and X. Liu, Biomass Convers. Bior., 2023, 13, 16887–16898 CrossRef CAS.
- T. Gao, Y. Yin, G. Zhu, Q. Cao and W. Fang, Catal. Today, 2020, 355, 252–262 CrossRef CAS.
- E. Hayashi, Y. Yamaguchi, K. Kamata, N. Tsunoda, Y. Kumagai, F. Oba and M. Hara, J. Am. Chem.Soc., 2019, 141, 890–900 CrossRef CAS PubMed.
- S. Chen, Y. Cheng, H. Ban, Y. Zhang, L. Zheng, L. Wang and X. Li, Ind. Eng. Chem. Res., 2020, 59, 17076–17084 CrossRef CAS.
- X. Han, C. Li, X. Liu, Q. Xia and Y. Wang, Green Chem., 2017, 19, 996–1004 RSC.
- H.-z. Lu, J.-f. Bai, F. Yan, X.-y. Zhang, Y. Jin, J.-y. Wang, P. Chen and M.-d. Zhou, J. Fuel Chem. Technol., 2021, 49, 312–321 CrossRef.
- F. Yang, Y. Ding, J. Tang, S. Zhou, B. Wang and Y. Kong, Mol. Catal., 2017, 435, 144–155 Search PubMed.
- C. Chen, L. Wang, B. Zhu, Z. Zhou, S. I. El-Hout, J. Yang and J. Zhang, J. Energy Chem., 2021, 54, 528–554 CrossRef CAS.
- L. Yang, J. Liu, F. Cheng, S. Zhou, Q. Xu, D. Yin and X. Liu, Renewable Energy, 2024, 226, 120409 CrossRef CAS.
- F. Wang, J. Lai, Z. Liu, S. Wen and X. Liu, Biomass Convers. Bior., 2023, 13, 16887–16898 CrossRef CAS.
- Z. Zeng, Q. Tang, B. Wen, L. Luo, X. Liu, Q. Xu, W. Zhong and J. Enviorn, Chem. Eng., 2024, 12, 112190 CAS.
- S. K. Singh, M. J. Akhtar and K. K. Kar, Ind. Eng. Chem. Res., 2020, 59, 9076–9084 Search PubMed.
- Y. Chen, M. Lu, J.-E. Zhou, X. Zhang, Y. Li, X. Lin, A. Zeb and Z. Xu, Electrochim. Acta, 2022, 435, 141355 CrossRef CAS.
- K. Hou, T. Wang, X. Jing and L. Zhang, Mater. Chem. Phys., 2023, 303, 127744 CrossRef CAS.
- H. Zhou, H. Xu and Y. Liu, Appl. Catal. B: Environ., 2019, 244, 965–973 CrossRef CAS.
- Y. Wang, H. Arandiyan, J. Scott, A. Bagheri, H. Dai and R. Amal, J. Mater. Chem. A, 2017, 5, 8825–8846 RSC.
- T. Jing, S. Yang, Y. Feng, T. Li, Y. Zuo and D. Rao, Nano Res., 2023, 16, 6670–6678 CrossRef CAS.
- R. Kumar, Z. Zhu, C. Chen, W. Cai, J. Woon-Chung Wong and J. Zhao, ChemSusChem, 2022, 15, e202201333 CrossRef CAS PubMed.
- J. Yu, Q. Zhou, X. Xue, H. Zhang, X. Li, F. Wang, Q. Chen and H. Zhu, New J. Chem., 2021, 45, 14608–14615 RSC.
- L. Zhang, X. Luo and Y. Li, J. Energy Chem., 2018, 27, 243–249 CrossRef.
- E. Hayashi, Y. Yamaguchi, K. Kamata, N. Tsunoda, Y. Kumagai, F. Oba and M. Hara, J. Am. Chem. Soc., 2019, 141, 890–900 CrossRef CAS PubMed.
- L. Bao, F.-Z. Sun, G.-Y. Zhang and T.-L. Hu, ChemSusChem, 2020, 13, 548–555 CrossRef CAS PubMed.
- S. Zhang, X. Sun, Z. Zheng and L. Zhang, Catal. Commun., 2018, 113, 19–22 CrossRef CAS.
- K. T. V. Rao, J. L. Rogers, S. Souzanchi, L. Dessbesell, M. B. Ray and C. Xu, ChemSusChem, 2018, 11, 3323–3334 CrossRef CAS PubMed.
- K. Yu, Y. Liu, D. Lei, Y. Jiang, Y. Wang, Y. Feng, L.-L. Lou, S. Liu and W. Zhou, Catal. Sci. Techol., 2018, 8, 2299–2303 RSC.
|
| This journal is © The Royal Society of Chemistry 2026 |
Click here to see how this site uses Cookies. View our privacy policy here.  Open Access Article
Open Access Article *a,
Sha
Wen
b,
Ying
Liu
a and
Xianxiang
Liu
*a,
Sha
Wen
b,
Ying
Liu
a and
Xianxiang
Liu
 *b
*b
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, and the samples were eluted at a flow rate of 0.7 mL min−1 at 30 °C. Typical HPLC results and 1H NMR analysis for HMF oxidation products are shown in Fig. S1 and S2 in the SI. The HMF conversion and the selectivity of the main products were calculated using the following equations:
5, and the samples were eluted at a flow rate of 0.7 mL min−1 at 30 °C. Typical HPLC results and 1H NMR analysis for HMF oxidation products are shown in Fig. S1 and S2 in the SI. The HMF conversion and the selectivity of the main products were calculated using the following equations:













