Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Emerging and persistent challenges of transitioning to solid-state electrolytes for hydrogen production from water splitting
Adrian P.
Dowling
 a,
Ioana M.
Popa
a,
Ioana M.
Popa
 b,
Yagya N.
Regmi
c and
Leila
Negahdar
b,
Yagya N.
Regmi
c and
Leila
Negahdar
 *a
*a
aSchool of Chemistry, University College Dublin, Belfield, Dublin 4, Ireland. E-mail: leila.negahdar@ucd.ie
bPaul Scherrer Institute (PSI), 5232 Villigen, Switzerland
cManchester Fuel Cell Innovation Centre, Department of Natural Sciences, Manchester Metropolitan University, Manchester M1 5GD, UK
First published on 10th March 2026
Abstract
One of the biggest challenges we are faced with in the 21st century is closing the anthropogenic carbon cycle and shifting away from a fossil-fuel dependent society. Hydrogen, one of the alternative fuels, is seeing unprecedented investment and deployment. However, obtaining cost competitive green hydrogen at industrial scale remains a challenge. Optimizing water electrolysis is, in this context, an increasingly important research area. Currently, a major portion of electrolytic green hydrogen is generated by aqueous alkaline electrolytes, but their limitation to relatively low current densities, low operating pressures and the corrosion of metallic components over prolonged use, as well as other fluctuating parameters, raise the price of hydrogen to an uncompetitive level. Water electrolysers using solid-state electrolytes (SSEs) have been gaining more interest in recent years in an attempt to accelerate cost competitive electrolytic green hydrogen production at scale. Despite many advantages, SSE-based electrolysers are still facing limitations such as sub-optimal ionic conductivity, inefficient electrode/electrolyte interface and short operating lifetimes. In this review, we aim to offer an overview of both the current state-of-the-art SSE technologies, as well as the most recent material designs addressing these limitations. We outline advancements made in the field of electrolysis technologies which we believe could serve as inspiration for the further improvement of SSEs in water electrolysis. Lastly, we summarize the remaining open questions and emerging trends, hoping that further research will use the improvements made in various fields to improve the efficiency of SSEs in water electrolysis.
1. Introduction
Access to abundant and affordable clean green energy is one of the biggest challenges of the 21st century in the wake of the accelerated technological progress since the industrial revolution and the unprecedented population growth it enabled over the following decades and centuries.1,2 Particularly, the emission of greenhouse gases, especially CO2, has increased dramatically, bringing the pressing environmental crisis to the forefront of the research community.1,3–7 During the last decade, several research groups have tackled the problem of the anthropogenic carbon footprint, with two general approaches to mitigating the emerging crisis. One solution is to aim to close the anthropogenic carbon cycle, either by carbon-capture and storage techniques, or by activating the CO2 molecule and using it as a carbon-source to synthesize platform chemicals such as methanol or olefines.8 Another solution is to deviate from the carbon-containing fuels that cause the greenhouse gas emissions in the first place by developing sustainable fuels with a low to non-existent carbon footprint.4,9,10One needs to look no further than the first row of the periodic table to find one such promising fuel. Hydrogen, whose combustion yields harmless water, is considered one of the most viable “fuels of the future” and has thus been the study object of many investigations in recent years.9–13 Amongst other challenges that prevent us from currently being a “hydrogen society”, such as its storage and infrastructure limitations, a crucial question is finding a reliable hydrogen source. Currently, hydrogen produced from fossil fuels is the lowest cost option, but does not ensure a sustainable future due to the high carbon footprint associated with it.9,12,14 A readily available, cheap and accessible hydrogen source is water. However, extracting hydrogen through water by means of thermal decomposition, photochemical approaches or photoelectrochemical water splitting remains difficult due to problems related to corrosion, high temperatures or catalyst design, among others.15–18
Presently, electrochemical water splitting remains one of the best alternatives to produce green hydrogen, as it allows for the integration of renewable energy sources such as solar and wind energy to deliver high-purity hydrogen.9,13,19,20 Despite the environmental advantages of this carbon-free solution, only about 4% of the worldwide hydrogen production derives from water electrolysis, due to both the intermittent nature of the renewable energies used to power the production of green hydrogen and the suboptimal efficiency of the electrochemical process (4.5–5.5 kW h per m3 H2).19–21 It is crucial, then, to improve the efficiency of water electrolysis such that it becomes an economically viable alternative for hydrogen production.22
One of the key components in increasing the electrochemical efficiency is designing (electro)chemically stable electrolytes and optimizing the electrode–electrolyte interface.23 The role of an electrolyte is to enable the ion transport, but not the electron transport—this need for a high ionic mobility is one of the main reasons why liquid electrolytes are commonly used in electrochemical processes. The liquid state does not only allow for an easy ion exchange and minimizes the electrolyte's resistance, but it also grants uncomplicated access to parameters such as pH, temperature and ion activity in the solution, which can significantly enhance the underlying electrochemical reactions.24,25 While these electrolytes show good ionic conductivity, especially at higher concentrations, they also tend to corrode the electrode material over a longer period of time.26 Moreover, gas-crossover (the recombination of the freshly created oxygen and hydrogen gases) must be avoided through the separation of the cathodic and anodic space through a membrane, which itself needs to be chemically and mechanically stable under the reaction conditions. In systems generating gaseous products such as water splitting, a common occurrence is also a decrease in efficiency as a result of the occupation of active sites by gas bubbles.27 Fluctuations in the electrolyte pH through evaporation, CO2 dissolution from the atmosphere, as well as local thermal and concentration gradients are additional factors that make liquid electrolytes very complex systems to uniformly control.28
Solid-state electrolytes (SSE) have been studied and deployed in recent decades in many electrochemical applications, such as fuel cells, electrochemical CO2 reduction and water-splitting, in order to circumvent the limitations of liquid electrolytes.29–34 Their thermal and mechanic stability allows SSEs to operate at elevated temperatures (200 °C or higher) in some cases and high voltages without the risk of degradation, which enhances the electrochemical efficiency.35,36 The main setback of SSEs lies in their limited ionic conductivity when compared to the liquid electrolytes. Thus, numerous recent studies have dedicated much effort to overcoming this barrier – developing SSEs which match or even outperform liquid electrolytes at room temperature.35,37–39 Generally regarded as safer and more efficient for large-scale applications, the investigation of SSEs in the water electrolysis process has been a topic of interest for many decades.
Alternatives to the traditional liquid electrolytes have been known for decades, and have been the recent subject of a resurging research interest.40,41 Amongst these, solid oxide and anion exchange membrane water electrolysers (SO-EL and AEM-EL, respectively) are very promising, while proton exchange membrane water electrolysers (PEM-EL) have already been commercialized at 100 s of MW scales.20,41–43 The recent advancements of characterization techniques on the micro-, nano and atomic scale, have awakened a renewed interest in understanding the fundamental working principle of the electrochemical water splitting with SSEs, and with it, a resurgence in the targeted material design for this process. While these technologies require more development before they can reliably replace current electrolyser cells, they promise an efficient integration of renewable energy sources for a sustainable future.20,41,42
This tutorial review aims to provide an introduction to the application of SSEs in electrochemical water splitting, relying on both previous studies on this subject and the known properties of solid electrolytes. In recent years, there has been a rapid increase of publications in this area (Fig. 1) – here, we will mainly focus on the most frequently investigated PEMs, AEMs and SOs, offering a concise and succinct summary of the state-of-the-art before diving into the newest material developments, aimed at overcoming and mitigating the challenges that SSEs face with respect to water electrolysis. While PEMs and AEMs are often hydrated, and thus their classification as solid-state electrolytes is sometimes disputed, the introduction of non-porous polymeric electrolytes represented a significant advancement in electrochemical water splitting.32,44 For this reason, AEMs and PEMs will be considered when referring to SSEs. Persisting barriers, as well as emerging trends and research questions that have yet to be exhaustively addressed are outlined, while less frequently discussed areas are considered in order to deepen the understanding of this promising avenue to green hydrogen.
2. Fundamental principles in electrochemical water splitting
Hydrogen was first obtained from water through an electrochemical route in 1789 by Adriaan Paets van Troostwijk and Johan Rudolph Deiman, but it was not until Alessandro Volta introduced the Volta Pila at the beginning of the 19th century that electrochemistry became an intensely researched field of science.45 In 1888, the first plans for an industrial water electrolysis technique were developed by Dmitry Lachinov, marking the beginning of the industry's interest in this process.46 Presently, electrochemical water splitting has been experiencing resurgent commercial activities and research interest. In this section, we discuss the fundamental principles of water splitting and the most common industrial applications with their advantages and technical limitations.Water electrolysis is an endothermic reaction (eqn (1)) which converts electrical energy into chemical energy while generating oxygen and hydrogen from water. The current flows between two electrodes, known as the anode where the oxidation half reaction takes place and the cathode where the reduction half reactions occur. The two electrodes are adjacent to the electrolyte to facilitate the ionic conductivity, and thus, completing the electrochemical circuit.15,47,48
Depending on the pH of the electrolyte the reaction mechanisms for both the cathodic hydrogen evolution reaction (HER) and anodic oxygen evolution reaction (OER) are different as illustrated below (eqn (2)–(5)) and Fig. 2). Alkaline solutions are generally preferred due to the relatively low price of the electrolyte and lower equipment demands, their high conductivity, and the fact that they are compatible with noble metal-free bifunctional HER-OER catalysts.49–51
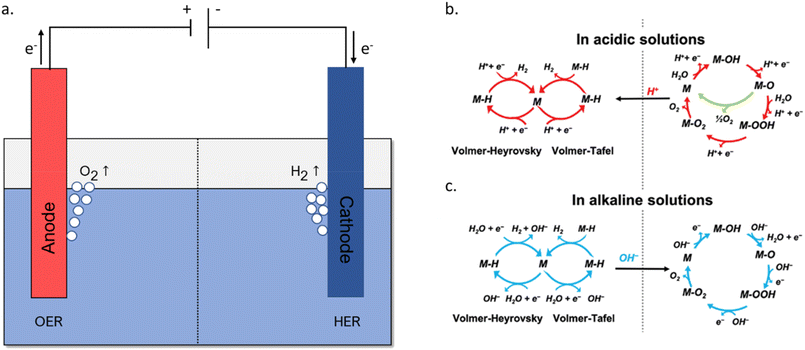 | ||
| Fig. 2 (a) Schematic representation of the conventional water electrolysis process in an alkaline electrolyte, reproduced from ref. 52 with permission from Wiley, copyright 2021; (b and c) general mechanism of the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) in acidic and alkaline media respectively, reproduced from ref. 53 with permission from Wiley Advanced, copyright 2020. | ||
| Total reaction: 2H2O → 2H2 + O2 | (1) |
| Cathode: 2H+ + 2e− → H2 | (2) |
| Anode: 2H2O → 4H+ + O2 + 4e− | (3) |
| Cathode: 2H2O + 2e− → H2 + 2OH− | (4) |
| Anode: 4OH− → 2H2O + O2 + 4e− | (5) |
Indeed, the majority of the investigations on electrochemical water splitting have been focused on the catalyst research, especially on searching and optimizing nonprecious metal catalysts (which lower the cost of electrolysis) capable of efficiently driving both the OER and HER.54–56 Despite the thermodynamic potential needed being 1.23 V, in particular because of the sluggish kinetics of the four-electron transfer OER mechanism, the actual potential to drive water splitting is much higher. This potential is an irreversible loss also known as the overpotential η and can broadly be divided into three terms: the activation overvoltage, related to the electrode kinetics and highly dependent on the catalytic pathway; the ohmic losses, which refer to the losses associated with the resistance of the different cell elements, and the concentration overvoltage, induced by mass transport limitations. The minimization of the activation overpotential is the main drive for the intensive work put into the catalyst development efforts.15,49,52,57 For an exhaustive review on the topic, the authors recommend the recent review by Quan et al.51 More recently, single atom catalysts have also been considered for this purpose.58
The four-electron OER has been intensely studied, and different mechanistic pathways have been proposed for different reaction conditions and electrocatalysts.57 While an in-depth analysis of these mechanisms is beyond the scope of this review, the mini review of Feng et al. presents the most recently proposed OER mechanisms.50 Regardless of the mechanism, however, the formation of the intermediates (O*, HO* and HOO*) on the surface of the electrocatalyst is crucial and rate-determining.57
The HER has been investigated for a plethora of transition metal catalysts, and has been generally described as a two-step process.54,59 First, the hydrogen adsorbs on to the active site followed by either a reaction with a water molecule in an electron transfer step to yield H2 and HO−, or the recombination of two hydrogen species to form the gas molecule.19,49 While the strength of the metal–hydrogen bond is the decisive mechanistic factor, the pH is also an important consideration.49 In alkaline conditions, the HER reaction rate is at least two orders of magnitude lower than the reaction rate in acidic electrolytes, yet alkaline electrolytes are the preferred industrial choice due to their enhanced operational stability.19
Despite the intensive studies on the HER and OER mechanisms, a unanimous agreement on the rate-limiting step and precise pathway has yet to be reached. Beyond different composition and structure of the catalysts steering the reaction down specific pathways, the work of Shih et al. has shown that the applied overpotential can influence the rate-limiting step as well.60 It follows, then, that local changes in pH, impurities, ion fugitivity, potential and viscosity can greatly affect the kinetics and completely change the operating regime of the reaction, and all of this on the laboratory scale.60
3. SSE in water electrolysis: ion transport, challenges and advancements
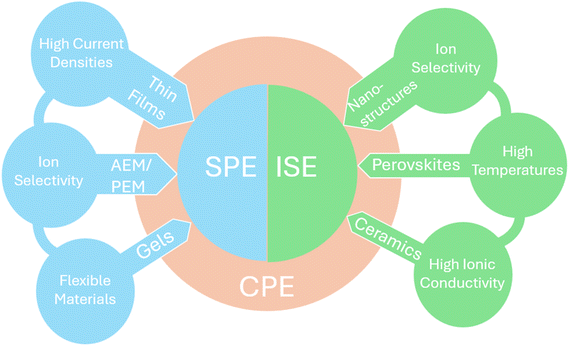
In order to shift away from the many varying factors bound to the use of aqueous electrolytes, solid state electrolytes prove to be a viable alternative. Nevertheless, SSEs for water electrolysis themselves face many other challenges involving ionic conductivity, mass transport, and material degradation.Ionic conductivity is a measure of the electrical conductivity due to the motion of ions in a material.61 Research into maximising the ionic conductivity is a core topic in SSE research for water electrolysis, as generally SSEs have lower ionic conductivities relative to liquid electrolytes. An understanding of how ion conductivity affects water electrolysis, and how the ion conductivity is affected by materials or external parameters, is key for understanding the state-of-the-art performance of SSEs. This section will examine ion transport mechanisms of various types of solid-state electrolytes used for water electrolysis, the material challenges that are presented for different SSE types and the strategies researchers have used to overcome them. In general, SSEs in water electrolysis can be split in three different categories: inorganic solid electrolytes (ISE), solid polymer electrolytes (SPE) and composite polymer electrolytes (CPE) (Fig. 3). While polymer-based electrolytes are usually used in low to intermediate temperature electrolysers, ISEs such as ceramics and layered double hydroxides (LDH) can be used in intermediate to high temperature applications (Fig. 3 and 8b). CPEs typically consist of ISE materials integrated into a polymer structure, to create a hybrid of SPE and ISE (Fig. 3).
3.1 Inorganic solid-state electrolytes (ISE)
ISEs tend to have higher bulk ionic conductivity relative to other SSE types.62 Ceramic structures used for ISEs include garnets, oxides, perovskites, oxyhydroxides and hydroxides.63,64 The ionic conductivity in ceramic materials is greatly influenced by active sites in the materials (Fig. 4a). Of particular interest is their ability for selective single-ion conduction at lower temperatures, which is ideal for efficient water electrolysis.65 Vacancies (Schottky defects) or interstitial sites (Frenkel defects) in the ISE can allow greater ion diffusion due to disruption of the lattice network, which allows for ion hopping to help restore the neutrality of the lattice (Fig. 4a).66,67 Crystal lattices of solid ionic compounds have some ion mobility in the lattice, which is the origin of their conductivity.68 Ion site hopping requires ions to travel from site to site, either as single ions or more commonly in multi-ion hops (Fig. 4c).63 In water electrolysis, neutral particles diffuse through voids in the material, so efficient ISEs need to be highly porous and have good ionic selectivity. ISEs should conduct ions of certain charge such as hydroxide anions, carbonates etc. for alkaline water electrolysis and protons and cations for acidic water electrolysis. | ||
| Fig. 4 (a) Ion transport via defects and sites in the inorganic solid electrolyte. Figure re-used with permission from John Wiley and sons.87 (b) Ion conductivity through grain boundaries, grains are represented by blue spheres and ion paths by arrows. The green arrow path has to overcome a large energy barrier due to impedance for ions to conduct across the grain boundary. (c) Ion transport for single ions and multi-ions in Inorganic Solid Electrolytes. Transport of ions through the ceramic occurs through ion hopping. Ion hopping from site to site involves an energy barrier that the ion must overcome. The energy barrier for multiple-ion hops is lower as result of ion–ion interactions, reproduced from ref. 88 with permission from Nature, copyright 2017. | ||
Single ion diffusion occurs due to particularly low energy barriers for specific ion types in the material with porous ion-conductive channels in the ceramic lowering the energy barrier.67 Control over defects in the material synthesis will greatly affect the ionic conductivity of ceramic electrolytes.69 Thermodynamic processes in the material cause defect formation and site movement. ISEs tend to be highly ordered and crystalline, and although typically the bulk ionic conductivity is high, the conductivity of ISEs across grain boundaries and at the electrode–electrolyte interface tends to be low due to impedance (Fig. 4c).70 Typically, there is a resistance increase at the grain boundary due to crystallographic orientation changes, effectively changing the orientation of ion paths through the material.71,72 Many inorganic electrolytes are prone to cracking, which occurs over time due to volume changes during cycling.73,74 Cracks and void formation from volume changes negatively affect ion conductivity through the electrolyte by increasing the impedance and the area specific resistance of the cell.75,76 Crack formation can also lead to reduced interfacial contact with the electrode, reducing the operational efficiency of an electrochemical cell.73,75
Redox reactions within the SSE also cause further degradation of the material. In particular for water electrolysis, any salts or metals dissolved in the water may deposit onto the solid electrolyte, which over time may negatively affect performance.77–79 This is especially problematic with alkaline, buffer liquid salts (such as KOH or KHCO3), or seawater.78,79 The ionic conductivity of ISEs also depends on bulk properties such as the thickness of the electrolyte (thicker electrolyte can reduce ionic conductivity as there are greater ohmic losses) as well as porosity of the ISE (neutral particles such as water molecules travel through voids in the porous material, whilst ions can travel through the material itself via active site hopping).80,81
Generally the effects of grain boundaries can be somewhat negated through sintering, i.e., compression of the material without melting it.82 Ceramic electrolytes present advantages over liquid or polymer electrolytes in their chemical stability and mechanical strength.62,71 Their hardness and brittleness causes ceramic electrolytes to have low interfacial contact area with the electrode surface, leading to high interfacial resistance at the electrode contact.83 Ionic conductivity in ceramics can be improved through thinning, although this effect may also affect mechanical strength.84 Alternatively ceramics can be inserted as fillers into polymer matrices, creating CPEs, to improve ion conductivity at the electrode electrolyte interface (this is discussed in Section 3.3).85 Temperature is also a significant factor in ionic conductivity, as heating the ISE can provide enough energy to ions in the material to overcome the ion hopping energy barrier.86
SSEs are often subjected to high pressure and temperature environments in solid oxide electrolyzers (SO-ELs). Mechanical stability is one persistent issue during heating and cooling of reactors.89 Heat and pressure changes affect the structure of the ISE through reactions between electrolyte and electrode.89 Proton ceramic electrolysis cells (PCEC) for water splitting usually operate between 500 and 700 °C.90,91 SO-ELs operate at generally higher temperatures between 700 and 1000 °C.92,93 They use ceramic electrolytes that at high temperatures either conduct oxide anions or protons.94 These dense electrolytes used in SO-ELs can be anion conducting or proton conducting, and can be tailored at the nanoscale to improve their conduction.94
Although the ionic conductivity of ISEs is relatively high compared to SPEs, they are typically less ionically conductive than many traditional liquid-based electrolytes. One of the strategies to improve the performance of ceramic electrolytes is through selective metal-doping of the electrolyte itself. Metal doping in ceramics leads to the creation of vacancies and active sites within the material, leading to improvements in ion transport and conductivity.95 Additionally, doping of the electrolyte can improve interface conductivity by altering material properties.
Lu et al. investigated doping a SrTiO3 perovskite electrolyte with Li to improve the surface charge transfer.96 The addition of Li to the perovskite electrolyte created oxygen vacancies which typically improves ion conductivity (Fig. 4a).96 They observed improved proton conduction via X-ray photoelectron spectrometry analysis (XPS) and increased electrochemical performance after surface doping.96 Li doping helps to preserve and maintain defects in the lattice and on the surface which improves the proton transport at the electrode/electrolyte interface.96 Shah et al. also observed enhanced performance due to oxygen vacancies in doped perovskite electrolyte.97 They doped a SrTiO3 electrolyte with Al which affected the energy band structure and allowed operation at relatively low temperatures (for SO-EL) of 520 °C.97 At 520 °C they observed a peak power density of 0.69 W (higher than the undoped SrTiO3 of 0.52 W).97 It is evident from this research that doping is an excellent strategy to overcome many of the drawbacks present in ISEs.
3.2 Solid polymer electrolytes (SPE)
Ion transport through solid polymer electrolytes (SPEs) mostly occurs through segmental motion of the polymer chains and thus results in relatively poor ionic conductivity (ranging from 10−7 to 10−5 S cm−1, compared to ISE conductivities of around 10−2 S cm−1) at room temperature.62,98 The motion of polymer chains in the SPE repeatedly creates new coordination sites which causes ions to move with the chain through vehicle mechanisms, as well as inter and intrachain site hopping (Fig. 5a).99,100 Segmental motion of the polymer chains occurs above the glass transition temperature (below which they will be crystalline, ordered and immobile). This suggests that there should be zero ionic conductivity in SPEs below the glass transition temperature; however, this is not always the case. At a given temperature, SPEs may possess various amorphous and crystalline phases, making the study of ion transport through SPEs quite complex, given that there are several potential ion pathways.99 Some SPEs have relatively high ionic conductivity in largely crystalline phase, although this, too, depends on the polymer composition.100 SPEs typically have higher ionic conductivity at high temperatures, as the polymer matrix is in the amorphous phase. The increased segmental motion of polymer chains enables greater ion conductivity.62 Ion selective conduction within solid polymer electrolytes usually occurs via functional groups.79,99 | ||
| Fig. 5 (a) Ion transport mechanisms for solid polymer electrolytes. Ion transport in solid polymer electrolytes occurs via several mechanisms. Ions can hop via clusters (via ions of opposite charge) along the chain or to other nearby chains in the polymer. Increasing the temperature makes the polymer amorphous, giving increased mobility to polymer chains thus leading to an increase in ion mobility/conductivity, reproduced from ref. 108 with permission from the Royal Society of Chemistry, copyright 2015. (b) Diagram of Grotthuss ion transport mechanism for OH− in water. OH− anions in water travel via bond formation and cleavage with H2O molecules. For optimal ion conductivity in polymer-based electrolytes, the electrolyte needs high water absorptive capabilities with some cation functionalization, reproduced from ref. 109 with permission from the Royal Society of Chemistry, copyright 2019. | ||
Interestingly, partially crystalline SPEs also allow for anionic/cationic selectivity, dependant on the material.100,101 For example, crystalline and amorphous phase SPEs made from polyethylene oxide (PEO) have high selectivity to Li+ cation conduction in lithium-ion batteries, due to the polymer forming channels with functional groups which attract cations and repel anions.100,101 This is useful for the development of next generation water electrolysis proton exchange membranes (PEMs), as the higher selectivity prevents oxygen radicals from degrading the material or recombination of products in the membrane.102 Likewise, solid polymer electrolytes can be designed for use as anion exchange membranes for alkaline water electrolysis. An interesting example was proposed in report by S. Z. Oener et al., who designed a combination of proton exchange membrane (PEM) and anion exchange membrane (AEM) to reduce overpotentials associated with the OER and HER. They made use of the selectivity of each membrane to allow for the use of optimal basic and acidic conditions, respectively.103 Ion selectivity within the material depends on nanochannels formed in the material, where ion conduction selectivity comes from inhomogeneous distribution of ions in the channel interior.103 The overall material retains charge neutrality to maintain system stability. Thus, ion conduction selectivity within the channels of SPEs can deviate from the bulk ion conduction selectivity.103 The controlled formation of SPEs at the nanoscale offers a great degree of control over the electrolyte properties such as ionic conduction selectivity.103 Nagao demonstrated how higher molecular ordering in sulfonated polyimide thin films enhanced the ionic conductivity.104 They drew a strong correlation between higher molecular weight and increased proton conductivity in the polyimide.104 They reduced the degrees of freedom of the polymer chains by a combination of growing the SPE on a substrate and increasing hydration.104 This in turn leads to increased proton conductivity within the electrolyte.
In most polymer-based electrolytes, ion transport can occur through vehicular ion transport or via the Grotthuss mechanism in hydrated SPEs. The Grotthuss mechanism in water is visualised in Fig. 5b. This mechanism is a model used to describe the transfer of ions through water. For AEM water electrolysis, OH− anions are transported via water, where OH− anion transportation occurs through formation and cleavage of covalent bonds with water molecules in the membrane.105–107 The formation of bonds via OH− bonding with protons causes subsequent water molecules to undergo similar operations. The Grotthuss mechanism is a very fast and efficient mode of ion transport for ion transport through porous materials.107 For example, low functionalized polymers with a high degree of swelling will typically favour ion transport via the Grotthuss mechanism.107 Highly functionalized polymers (such as block polymers), on the other hand, will mostly have ion transport occurring via a vehicular mechanism where OH− anions are transported via overlapping hydration shells in a polymer chain.107
To overcome the low ionic conductivity of solid polymer electrolytes, thin film design is often employed to reduce ohmic losses.110 The state-of-the-art thin film PEM electrolyte is the Nafion® ionomer, which is chemically stable and durable over a long time.79,111,112 Nafion® has high water absorptive capabilities and contains sulfonic polar groups which guide ions through porous channels in the polymer.79,112 The use of ionomers such as Nafion® for proton exchange membranes is commonplace due to their high efficiency and ion selectivity, but can present environmental concerns regarding perfluorinated compounds leaking into the environment.111,113,114 Due to these environmental concerns with Nafion®, research into alternative thin, even higher performance PEMs is ongoing.110,113 Thin film manufacturing also presents some challenges with stability over prolonged usage and degradation at high temperatures due to polymer membranes drying out.110
SPEs generally have poor ionic conductivity at room temperatures.85 To improve the performance for SPEs, the operating temperature can be increased to overcome overpotentials in the system and increase the ionic conductivity. High temperature SPE-based PEMs can be used for steam electrolysis, which is less electrochemically energy intensive than water electrolysis.115 Heating and cooling of the SPE can lead to poisoning of the electrode from the electrolyte due to degradation, reducing the effectiveness of the material.116 Poisoning can also occur via salt deposition from water in hydrated SPEs.79 Currently, most liquid filled membranes will dry out and fail at temperatures above 80 °C.110,117 This can be counteracted with increased humidification and pressure.110 Additional degradation issues occur from ion deposition due to salts (such as KOH) dissolved in the liquid portion of the electrolyte.79 These salts can clog the channels of the membrane and damage the structure of the membrane, leading to large ohmic losses and structural failure.79 Polymer membranes in water electrolysers are typically not durable or stable over long periods of continuous use which is problematic for commercialization. However, they can be synthesized from environmentally sustainable materials and avoid the use of corrosive substances which can damage electrolyser systems over time.118,119
3.3 Advantages and properties of composite polymer electrolytes (CPE)
As has been mentioned, some of the limitations of ISEs and SPEs are overcome by combining properties of the two SSE types into CPEs. CPEs typically consist of polymer matrices with inorganic fillers providing them with good mechanical strength and high chemical stability. As a result of their mixed properties, CPEs are imbued with higher ionic conductivity than SPEs and greater interfacial contact than ISEs.85 The hydrophilic nature of inorganic fillers such as MXenes, layered double hydroxides, etc. also helps augment the water absorptive capabilities of polymer membranes.113,120Ion transport through CPEs is multifaceted. Generally, it is understood as a combination of ionic relocation in ISEs and SPEs (Fig. 6), that is, site hopping through the inorganic phase, through segmental motion of the polymer chains in the polymer phase, or a combination of the two.62,85 The ionic conductivity in CPEs is increased due to decrease in crystallinity and glass transition temperature.85 Structural changes also occur in the polymer due to Lewis-acid/base effects, where ion dissociation in the polymer is affected by Lewis acid/base centres in the inorganic material.121 Lewis acid/base groups form trapped surface charges, which affect ion concentration and flux in the surrounding phase.62 The high selectivity for ionic conduction in CPEs makes them ideal for use in water electrolysis. In literature there has been ample reports done into developing CPEs as proton/anion exchange membranes for acidic/alkaline water electrolysis respectively.106,113,122
 | ||
| Fig. 6 Possible ion pathways through composite polymer electrolytes (CPEs). Like solid polymer electrolytes, CPE ionic conductivity increases with temperature as polymer chain segment mobility increases. The ionic conductivity of CPEs is greatly affected by material properties of the inorganic filler, reproduced from ref. 85, with permission from the Royal Society of Chemistry, copyright 2022. | ||
Increasing the water absorptivity helps increase ion conductivity via the previously mentioned Grotthuss mechanism, as reported by Waribam et al.113 Their group investigated sulfonated polyether ether ketone (SPEEK), a hydrocarbon-based polymer which has high mechanical, thermal and oxidative stability and chemical resistance.113 To avoid excessive swelling and improve the proton conductivity they created a CPE by inserting 2D nanomaterial Ti3AlC2 MXenes.113 They found that their composite material had comparable conductivities to that of commercial Nafion® (around 0.010 S cm−1) for a range of inorganic filler concentrations from 2% to 6%.113 Notably they observed that their composite membrane had a H2 faradaic efficiency of 95.2% compared to the 66.7% of the SPEEK membrane.113 A research paper by Choi et al. described the use of thin film composite polymer membranes for use in alkaline water electrolysis.106 They constructed composite polymer membranes using a quaternary ammonium layer on a porous polyethylene support.106 They selected the polyethylene polymer matrix due to its high mechanical and thermochemical stability and thin film design which can minimise mass transport phenomena. Their membrane design had low gas crossover and long-term stability of about 1000 hours of operation.106 Quaternary ammonium cationic groups inserted into the polyethylene structure provided the OH− anion conduction necessary for the AWE.106 They observed improved results from the composite electrolyte when compared to hydrophilized polyethylene.106
4. Industrial water electrolysers
At industrial and stack levels, the hurdles are more complex than those faced in a laboratory-scale single cell (or half-cell) system. For instance, one issue occurs during operation at elevated temperatures and current densities leading to the production of large quantities of gas bubbles.123 Their build-up creates considerable mechanical pressure in the flow reactor (electrolyser cell), leading to quick performance degradation and even structural oscillations of the catalysts.123–125 While fundamental studies on the half-cell level answer important research questions about interface phenomena and catalytic processes, the results obtained under such bespoke conditions may be insufficient in addressing the key challenges in full-cell level setups. The heterogenous potential distribution over large electrodes, the ohmic drops caused by operating at high currents and the concentrations and thermal gradients which develop under industrially relevant conditions are difficult to simulate in a half-cell and even in <25 cm2 single cell, set-ups.60For this reason, it is important to have a more in-depth look at the current industrial setups and understand the demands and challenges of a scaleup of the water electrolysis process. According to Thissen et al., the most basic laboratory-scale system to mimic industrial processes is a full single cell with continuous flow. Its complexity and cost makes this setup out of reach for widespread academic use while the difficulty of accommodating reference electrodes for investigations render deconvolution of underlying electrochemical phenomena impractical.126
There are three widespread low temperature technologies: alkaline water electrolysers (AW-EL), proton exchange membranes electrolysers (PEM-EL) and anion exchange membranes electrolysers (AEM-EL), with solid oxide electrolysers (SO-EL) operating at high temperatures, as summed up in Fig. 7. We will consider the critical raw materials (CRM), techno-economic and life cycle assessment (TEA/LCA), United Nations Sustainable Development Goals (UNSDG) adopted in 2015 to address the environmental challenges by 2030 and lifetime and robustness of the technologies we will discuss in this section.127
 | ||
| Fig. 7 Schematic representations of the industrially relevant water electrolysers: (a) alkaline water electrolyser (AW-EL), (b) proton exchange membranes electrolysers (PEM-EL), (c) anion exchange membranes electrolysers (AEM-EL), reproduced from ref. 124 with permission from Joule. Copyright 2021, and (d) solid oxide electrolysers (SO-EL) re-used with permission from Elsevier, copyright 2008, ref. 128. | ||
AW-Els, despite operating with aqueous electrolytes, remain the most mature of the three low temperature technologies and the dominant one at a commercial level thanks to their relatively low cost and long history of operation and innovation.15,19 To understand how the transition to solid electrolytes fares in comparison to this well-established industrial procedure, we must first take a look at the AW-EL system. The electrolysers showcase remarkable stability and are generally operated at 80–90 °C in 6–8 M KOH electrolyte solution to ensure good ionic conductivity.51,126 The anodic and cathodic spaces are separated by a porous diaphragm which is impermeable to gases but permeable for OH−, typically Zirfon membranes, meant to prevent the recombination of the product gases.129,130
Some of the most significant advancements in AW-ELs are the development of ion exchange inorganic membranes to be used a diaphragm, the design of electrolysers which allow an increase of the working temperature up to 150 °C, and the research conducted into developing electrocatalysts to reduce the applied overpotential.15,49,51,131,132 While the electrolyser cells are largely cost-effective, the current densities and thus the dynamics are often slower.42 Strategies to reduce the energy consumption without greatly changing the electrolyser setup include additives to tune the electrolyte–electrode interactions and using ionic liquids as electrolytes.133,134 Moreover, the handling of the alkaline liquid electrolyte may pose a problem in terms of water contamination, thus not fully aligning with the outlined environmental goals.
4.1 Proton exchange membrane electrolysers
The emergence of SPEs over the past few decades marks a shift towards the integration of SSEs in electrochemical water splitting.43,60,135–139 This generation of electrolysers replaces the liquid electrolyte for a humidified ion-exchange polymer membrane.15,129,140 While AW-ELs often have difficulty in adapting to the fluctuating loads characteristic of renewable energy sources, the ultrathin polymer membrane based PEM-ELs and AEM-ELs are more suitable for integrating solar and wind energy sources.135Fig. 7b illustrates the operating principle of the PEM cell, which involves the oxidation of water at the anode; the protons then travel through the polymeric membrane to the cathode, where they are reduced to form gaseous hydrogen.15,141 The PEM, such as Nafion®, need to be resistant to the chemical degradation that occurs due to the resulting oxygen and hydrogen gases. Proton transport in Nafion® is believed to occur via a combination of ion hopping and vehicular transport (see Section 3), where the high proton mobility is determined by the hydration shell of the acid –SO3H groups. This minimizes the ohmic resistance losses, which helps reduce the applied overpotential.140,142 The proton conductivity can be enhanced through higher molecular ordering and an in-plane oriented structure of the polymer network, as proven by Nagao.104 The water uptake of the membranes can also be fine-tuned through the pre-conditioning temperature.130,142
The PEM-ELs operate at current densities of >2 A cm−2 below 2 V cell potentials. The PEM-EL performance is thus superior to the maximum allowable current density in AW-ELs at relevant potentials, and is not the limiting factor in the industrial application of PEM-EL.15,20,123 Instead, PEM-ELs are still limited by their high investment costs, shorter lifetimes compared to AW-ELs and poor component recyclability, which constitute some of the biggest bottlenecks in terms of TEA and LCA.143 The reduced lifetimes for PEM-ELs are mostly due to membrane-thinning or degradation though chemical attack by radicals, as well as catalyst dissolution/deactivation in acidic environment.43,141,144 The true bottleneck, however, arises from the need to use noble metal Pt and Ir catalysts, which significantly increases the cost and scalability of the water electrolysis using PEM.43 An environmental concern arises from the use of fluoropolymers such as Nafion® as the membrane, which remain the state-of-the-art materials.20,43,145 Finding alternatives to these materials is crucial in conforming with the environmental goals set out by UNSDG, in particular SDG 12, which postulates avoiding the use of scarce or non-recyclable materials.
It thus becomes evident that PEM-ELs are limited not by their performance, but rather by the materials, and that further research is imperative for the PEM-ELs to become more industrially widespread. Developing alternatives to Ir as the OER catalyst has been a generational challenge, due to most transition metal oxides rapidly dissolving under the acidic PEM-EL conditions. Thus, research on the OER electrocatalysts for PEM-EL has also been focused on strategies to minimize the use of Ir.43 Research efforts dedicated to developing cost-effective, fluorine-free PEMs is more promising, but not without its challenges either. Materials such as the acid-doped polybenzimidazole membranes have received attention due to their promising efficiency despite their limited stability.144 In a 2023 review, Kumar and Lim highlight the recent advances in PEM development and stress that these Nafion®-alternatives do not yet match the high current densities and chemical durability of the state-of-the art PEMs.130 Hydrocarbon backbones are more vulnerable to radical attacks and to the strongly oxidizing conditions at the anode interface, which prevents them from becoming reliable replacements for fluorinated ionomer polymers.43
While they are the benchmark, Nafion®-based PEM-ELs exhibit their own weaknesses, too. A limiting factor is the operating temperature limited to below 80 °C, the membrane undergoes mechanical and chemical degradations at high temperatures, which limits the hydration-shell dependent proton transport and thus the overall efficiency. Moreover, the higher temperature regime and decrease in membrane thickness increases the hydrogen permeation, running the risk of exceeding the safety limit of 2% hydrogen in the oxygen product stream.146 Not only is the hydrogen permeation affected by the membrane thickness, but also the film swelling, water absorption and solubility are affected by it.110
In situ characterization methods have been used to understand the proton conductivity, the water-gas distribution in the cell in operation regime and the degradation processes of these materials, which is vital for the rational design of new ionomers for PEM-ELs.147–150 It must be mentioned, however, that a recent review by Corti concludes that the intermediate-temperature operation of PEM-ELs is not necessarily a rewarding affair.144 In fact, the author outlines several other research directions, such as understanding the nanostructure of PEMs and evaluating alternative membranes through the ratio of proton conductivity and hydrogen crossover under state-of-the-art operating conditions instead.144
4.2 Anion exchange membrane electrolysers
AEM-ELs are considered a third-generation technology of electrolysers, combining the advantages of the AW-ELs and PEM-ELs.117,123,151–155 The main attraction of AEM-ELs lies in the possibility of developing noble-metal free systems, integrating renewable energy sources and rendering it economically more competitive in comparison to PEM-EL technologies.117,156Here, the diaphragm used in AW-EL systems is replaced by a SPE anion exchange membrane (AEM). Additional ionic conductivity is provided by a 0.1–1 M KOH supporting electrolyte solution, much lower in concentration than in its AWE counterpart and thus reducing the corrosion risk.20 Significantly, unlike in PEM-ELs, the electrocatalysts are based on relatively abundant, and thus, low-cost transitional metals.117,130,157 Water electrolysis with AEMs follows the same principle as the AW-ELs, whereby the AEM facilitates the OH− transport and prevents the recombination of gases.158 While still at the late developmental stages of research and demonstrations, it is for these reasons that the AEM technology is increasingly seen as a significant contributor towards electrolysis in the future.
Unlike with PEMs, where a benchmark polymeric membrane with good performance in electrochemical water splitting already exists, AEMs with high ionic conductivity, remarkable mechanical processes and stability under strong alkaline conditions have yet to be developed.43,123 AEM membranes undergo nucleophilic attack of the OH− ions on the cationic groups and backbone degradation, which causes them to lose conductivity within 1000–5000 h under realistic conditions, posing a significant durability problem.43,159 AEM-ELs are also particularly sensitive towards ppm-level CO2, which changes the local pH and reduces the conductivity of the membrane.117 All of these are reasons why, despite the low capital expenditures the AEM-EL shares with its AW-EL “parent”, the overall operating expenditures are higher and not industrially attractive. As per the UNSDG, scaling this technology up for commercialization (SDG 9) remains a challenge.
Since AEM-ELs are still being developed and are considered in the pre-commercialisation stage, universally accepted state-of-the-art parameters are yet to be defined.117,160 Commercially available AEMs are aromatic polymers with mostly quaternary ammonium functional groups, often crosslinked or with microphase separation to increase the membrane's durability.158,161 To increase their ionic conductivity, the number of anion exchange functionalities must be increased, leading to a trade-off between mechanical strength and conductivity.158 A possible mitigation arises in the form of 3D design of the membranes, that contain simultaneous in-plane and through-plane transport channels for water and anion diffusion.155 Organic-inorganic CPEs also exhibit good hydroxide conductivity and better mechanical stability under operation conditions on single-cell tests, but have yet to be thoroughly investigated.162
A recent review by Ng et al. offers an in-depth discussion of recent studies investigating different AEMs in water electrolysis and how the choice of catalyst in combination with the membrane is crucial, while a review by Park et al. dives into the synthesis of aryl ether-free anion exchange polymers, which exhibit better stability under alkaline conditions.117,163 Another overview by Henkensmeier et al. analyses a range of commercial membranes and their stability.159 Notably, there is no current benchmark that allows the comparison between different investigation results, an unanimously agreed upon way to evaluate alkaline stability, nor a definitive model for the electrochemical device.156,164
While the design of PEM-ELs benefit from the developments achieved by PEM fuel cells – not only manufacturing procedures such as membrane casting and thickness control, catalyst-coating and stack compression, but also the maturity of an already well-defined supply chain – AEM-ELs do not have a similar predecessor to aid with the manufacturing maturity. Cell architecture itself is still being optimized, as it can help improve the ionic conductivity of the membranes.155 Scale-up attempts of lab-scale AEM-ELs have been investigated: Loh et al. describe zero-gap configurations of AEMs of 10 cm2 and 100 cm2 sizes, respectively, testing several parameters such as electrolyte concentrations, temperature and the choice of membrane over 72 h.160,165 Despite promising results from such studies, the lifespan of AEM-EL technologies remains one order of magnitude under that of PEM electrolysers, with most of the SPE membranes ceasing operation after less than 500 h.152,162,166 AEM-ELs are thus far from being considered a replacement for PEM-ELs, but they have been garnering more and more academic attention thanks to their potential as low-cost, environmentally-friendly systems.
4.3 Solid oxide electrolysers
Apart from the low-temperature water electrolysis systems, high-temperature electrolysis can be performed in solid oxide electrolysers (SO-ELs) using ISEs. SO-ELs were first conceptualized in the 1960s, and much of the information required for their operation was available thanks to the work done on solid oxide fuel cells (SOFCs).167,168 Due to this inheritance, the progress on SO-ELs was significantly sped up and it has recently entered the demonstration and scale-up phase.92 These are operated between 500 and 900 °C and exhibit a much higher efficiency than their low temperature counterparts.15,129,130 While the high temperatures lead to increase in thermal energy demand for the water-splitting reaction, the electrical energy demand is lowered remarkably.41,128 As such, a much higher percentage of the required energy can be supplied as heat, which is advantageous and would represent a big step in reducing green H2 costs as waste heat generated through existing industry can be efficiently utilised.15,20,92,123,130,169 The nuclear energy sector is interested in the SO-EL research, considering the possibility for the high-temperature gas cooled nuclear reactors to act as a heat source.15In SO-ELs, steam is fed to the cathode, where the water is reduced to form hydrogen and oxide anions. These are then transported through the solid electrolyte to reach the anode, where they recombine to form oxygen and release electrons (see eqn (6), (7) and Fig. 7d).
| H2O(g) + 2e− → H2(g) + O2− | (6) |
 | (7) |
A cost-intensive process is the separation of the hydrogen from the steam at the cathode.15 To circumvent the latter problem, ISEs consisting of perovskite-type oxides with protonic conductivity at ca. 700 °C have been investigated. The half-cell reactions are then identical with the ones in a PEM-EL, so that hydrogen is produced at the cathode and no further separation from the steam is required.170
The state-of-the-art electrolyte for SO-EL is the gas-tight yttria-stabilized zirconia (YSZ), in the form of a thin film, with other rare earth oxide alternatives posing a CRM limitation and going against SDG12.15,92,171 Unlike in the AEM-EL, the challenges related to SO-EL do not stem from the immaturity of the field or lack of benchmark electrolyte materials, but rather, from the high-temperature stability and operational durability of the ceramics, which are sensitive to thermal cycling and thus limit their lifetimes. Due to the high-temperature operation, one must also consider the thermal expansion coefficient of the electrode and electrolyte materials, since a mismatch could lead to mass transfer limitations and poor charge exchanges. For example, Elangovan et al. discuss the promising ionic conductivity of lanthanum gallate electrolytes, but the widespread use of these materials is limited by their thermal incompatibility with the Ni-cathode.172 A common solution is the introduction of an intermediate “barrier layer”, especially needed between the anode and the electrolyte, such as yttrium-doped cerium or gadolinium-doped cerium, which ensures good thermal compatibility between the two.169
The problems of the cell degradation due to electrolyte aging and electrode deactivation, however, have yet to be tackled.15 Recent research has also focused on the development of solid oxide cells (SOCs), which can be used reversibly as both electrolysis and fuel cells.173–179 A study by Tietz et al. investigated the degradation of the electrolyte layer, which showed granular porosity and pore formation along grain boundaries after 9000 h of operation, likely due to the buildup of oxygen within the closed pores of the electrolyte.180 These deformations where associated with an increase in ohmic electrolyte resistance and higher overpotentials, which resulted in a lower cell performance.180 A later study by Graves et al. claimed that reversible cycling between the electrolysis and fuel-cell modes can eliminate the formation of microstructural damage, studying a SOC which was in operation for 4000 h to support these claims.178
Unlike other water electrolysis technologies, SO-ELs are not limited by the electrochemistry, but rather by system integration and the material physics, as opposed to the catalysis. They are a promising avenue to green hydrogen, especially when industrial heat integration can be implemented. For this reason, further research is required to optimize the electrode and electrolyte materials (Table 1).
| Criteria | AW-EL | PEM-EL | AEM-EL | SO-EL |
|---|---|---|---|---|
| a Criteria includes critical raw materials (CRM) such as yttria stabilised zirconia (YSZ), the technoeconomic and life-cycle assessment (TEA/LCA), judged in terms of capital expenditure (CAPEX), the United Nations Sustainable Development Goals (UNSDG) addressed by the challenges posed by each type of electrolyser, lifetime & robustness and their materials maturity. Electrolyser technologies include alkaline water electrolysis (AW-EL), proton exchange membrane electrolysis (PEM-EL), anion exchange membrane electrolysis (AEM-EL) and solid oxide electrolysis (SO-EL). | ||||
| CRM | Ni-catalysts low risk | Ir-catalysts high risk | Transitional metal catalysts low risk | YSZ electrolyte high risk |
| TEA/LCA | Low CAPEX | High CAPEX | Low-moderate CAPEX | High CAPEX |
| UNSDG | Water contamination | Scarce materials | Challenging scaleup | Scarce materials |
| Lifetime & robustness | Long lifetime > 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h 000 h |
Long lifetime 40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–60,000 h 000–60,000 h |
Low lifetime 1000–10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h 000 h |
Moderate lifetime 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–40 000–40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h 000 h |
| Materials maturity | High | High | Low | Moderate |
4.4 The status quo of current water electrolysis technologies
For SPEs (PEM and AEM), their high cost, temperature-limited regime and membrane thinning were identified as some of the key challenges, limiting their lifetime. Despite this, the high efficiencies of up to 80% (2.2 V, 0.6 A cm−2) justify the research efforts in trying to develop more durable polymeric membranes.181 For the solid oxide ISEs, their stability at high temperatures, the thermic coefficient mismatch with the electrode material, as well as the separation of the hydrogen product from the steam stream are problematic. However, its industrial integration and high efficiency values of over 90% render it a promising technology for the future.181The following section will take a brief look at some of the potential applications of SSEs in water splitting technologies, and some relevant recent developments in material science regarding solid state electrolytes.
5. Applications, emerging trends and future prospects
5.1 Advanced materials for use as SSEs
Promising solid state electrolytes with desirable and tuneable physical properties come in the form of nanostructured inorganic materials, which can act as both electrolytes and have active catalytic sites for water splitting.182 Layered Double Hydroxides (LDH) are inorganic materials composed of 2D inorganic layers, each layer being composed of divalent and substituting trivalent metal cations octahedrally coordinated to hydroxide groups (see Fig. 8b).183,184 LDH are highly porous with large surface areas, enabling good particle diffusion and absorption, with catalytically active metal sites regularly dispersed through the material.185 The metal-hydroxide layers have a net positive charge due to partial substitution of M2+ with M3+ cations, which is neutralised through the intercalation of anions and water in the interlayer space.184 These play an important role in hydroxide ion conduction, as these are conducted along the interlayers in water electrolysis.182,186 LDH have tuneable properties desirable for electrocatalysis, such as high surface areas, high porosity which both increase the anion mobility, thus increasing the ionic conductivity.184 The anion conductivity of LDHs was seen to increase with the interlayer distance and adsorbed water content according to a 2013 paper by Jung et al.186 Jung et al. synthesized several LDH with (Mg(1−x)Znx)2Al layers (varying the metal ratios) for use in an electrochemical fuel cell. They reported through their experimentation that the binding energies of the metal and oxygen orbitals changed based on changes to the electron density on the elements. The binding energy of elements in the LDH affects the ion conductivity through the material. Changes in the interlayer distance of the LDH were seen to occur, driven by the substitution of Mg2+ by the larger Zn2+ ions. Ion conductivity was reported to increase proportionally to increases in the interlayer distance and water absorbed.186 | ||
| Fig. 8 (a) “Brick and mortar” design of bio-inspired solid-state electrolyte. The image presented here is intended for a Li-ion battery electrolyte, where the ceramic “bricks” are joined to each other with polymer “mortar”, creating a composite electrolyte. This electrolyte design exhibits the exceptional hardness and strain of nacre. Re-used with permission from Wiley-VCH, copyright 2020.201 (b) Layered double hydroxides. Blue, grey, orange polygons represent metal cation layers. Anions and water occupy the interlamellar space. (c) Arrhenius plot of ionic conductivities of different ice salt electrolytes. The ionic conductivities of doped ice are significantly higher than pure ice, reproduced from ref. 205 with permission from Wiley-VCH, copyright 2019. (d) Ion transport via composite polymer electrolyte using modified Zeolite Imidazolate Framework 8 with piperidine functional groups as filler in a polymer matrix. Reproduced from ref. 198 with permission from the American Chemical Society, copyright 2025. | ||
Zeng et al. describes efficient hydroxide anion conduction by an anion exchange membrane, used in AWE, synthesized from Mg–Al layered double hydroxides (Mg–Al-LDH).182 The positively charged layer was compensated by carbonate anions in the interlayer regions. The LDH in question was shown to have a relatively high ionic conductivity of 0.012 S cm−1 at a temperature of about 80 °C. As a comparison, a 2020 review by Brauns et al. examining the state of alkaline water electrolysis put the ionic conductivity of KOH at 95 S cm−1 at a temperature of 50 °C.129 LDH electrolytes tend to have greater stability and chemical resistance than traditional AWE liquid electrolytes.184 Lv et al. found through AWE testing that the NiFe LDH also doubled as an excellent catalyst for OER, reducing the overpotential of electrochemical reactions.185 While attractive for their dual use as electrolytes and electrocatalysts, LDH materials face stability issues over long-term use, such as component leaching or structural changes.187 Integration of LDH materials into a polymer matrix helps to negate these stability issues. Lv et al. integrated their LDH materials as a filler into an anion exchange membrane using a quaternary ammonium poly matrix for AWE.185 LDH electrolytes have potential usage in seawater electrolysis due to their ability to withstand salt corrosion and resistance to the effects of microorganisms in seawater.78,188 The wide choice of non-noble metals for LDH is also promising for sustainable production of SSEs, as is the possibility of using seawater as the hydrogen source.78
Low-dimensional materials such as graphene have potential as solid-state electrolyte materials due to their high conductivity, ease of synthesis, and flexibility. Moreover, they can be incorporated into liquid electrolyte solutions, as discussed by Uddin et al., where they tested the results of a water splitting experiment using three different acidic electrolyte solutions containing nanoparticles: graphene nanoflakes, multi-wall carbon nanotubes and indium tin oxide.189 They reported that increases in the graphene nanoflake concentration led to increases in the hydrogen production rate for various applied voltages. Concentrations of 1 wt% or greater led to agglomeration of nanoparticles, which reduced its ionic and charge carrying capacity, subsequently reducing the hydrogen production rate.189 They observed the highest hydrogen production rates at 0.5 wt% graphene flake concentration. Similar results were observed on hydrogen production rate measurements for the electrolyte solution with the multi-walled carbon nanotubes. In performance comparison to the iridium tin oxide nanoparticle solution, they determined that the high conductivity and surface area of the graphene-based materials enabled high ion transport enhancing hydrogen productivity. Anion exchange membranes suffer more from ohmic losses than proton exchange membranes due to hydroxide mobility being about 57% of proton mobility.190–192 Thus for anion-conductive CPEs a filler with high ion exchange capacity is required.190 Graphite is an ideal filler due to its high conductivity.193 A paper by Chu et al. investigated the use of graphene as an inorganic filler for anion exchange membranes.190 They tested various wt% of graphene oxide filler in the membrane, and observed the highest hydroxide ion conductivity for a 1 wt% filler at 115 mS cm−1 at 90 °C.190 They attributed the improved ionic conductivity of the polymer membrane to the increased π–π interactions between the polymer chain and graphene aromatic chain.190 They also observed good resistance from the graphene oxide doped membrane in strong alkaline solution.190 Graphene is a cheap, relatively easy material to synthesize and is environmentally friendly. Further research on graphene as an inorganic filler is a promising avenue of further research.
Metal Organic Frameworks (MOFs) have potential as SSEs or as a bridging material at the electrode–electrolyte interface.41 MOFs are highly crystalline, ordered materials composed of metal cations covalently bonded with organic linkers.98,122 These porous materials, with their large internal surface areas, form highly functionalised nanochannels which can be used for selective ion conduction.98,122 Integrating MOFs into polymer frameworks to create CPEs can lead to improved cell performances.98,194,195 As they are bulk insulators, conductive materials (such as graphene) are usually inserted into MOFs post-synthesis to make them more electrically conductive.196 It should be noted however that even without post-synthetic treatment the regular dispersion of active sites inside of MOFs makes them ideal for ion conduction.194 In addition, MOFs contain metal nodes, which can act as active catalytic sites for the OER and HER, reducing overpotentials within the cell.197 Li et al. designed highly stable AEMs incorporating Zeolite Imidazolate Framework-8 (ZIF-8, a zinc MOF) into poly terphenyl piperidinium polymer matrix.198 They reported high hydroxide anion conductivity for their CPEs and high stability over long periods of operation.198 They modified the ZIF-8 filler surface with functional groups to enhance interfacial contact with the polymer matrix which also resulted in a highly stable CPE (Fig. 8d).198 Jiao et al. reported high proton conductivity in dye-encapsulating MOFs (dye@MOFs) used for water electrolysis, and observed good stability in the MOF structure even after several test runs for HER.196 The ionic conductivity of the MOF was greatly improved through the addition of the dye.196 These dye@MOFs were applied to the surface of nickel foam to prepare a working electrode and used in concert with a liquid electrolyte. Hong et al. measured the performance of their MOF-CPE in a Li-ion battery and found that addition of the MOF improved the ionic conductivity by a factor of 20 times when compared to the unmodified CPE.199 Hong et al. made use of a MOF with pore sizes greater than the lithium ion diameter which enabled good ion transportation through the MOF-CPE.199 The large surface area of MOFs increases their interactions with polymer chains in CPEs, enabling enhanced ion transport.122
Bio-inspired SSE designs take the knowledge of biological systems and apply their unique and advantageous properties to synthetic electrolyte designs. Although in literature there has been some level of research for bio-inspired SSEs for fuel cells and Zinc Ion Batteries (ZIB), there has been minimal research for bio-inspired designs for water electrolysis. For the purpose of this review, some of the conclusions from research into bio-inspired SSEs for ZIBs and fuel cells still can be put into context for water electrolysis reactions. Nacre, the material composing mollusc shells, has been used as inspiration for advanced solid electrolyte materials with their advantageous “brick and mortar” design (Fig. 8a).200,201 He et al. investigated a nacre-inspired architecture for a proton-conductive electrolyte membrane.202 They found that their electrolyte design had a higher power density than the commercially available Nafion® 212, yet due to the nacre inspired design it had far more tensile strength and greater resistance to fractures.202 A paper by Dou et al. described the design of biomass-based SSE for use in flexible zinc-air batteries.203 They synthesized cellulose nanofibers from bamboo modified with an ion-conducting polymer to create a CPE AEM. The electrolyte conducted OH− anions through nanochannels formed in the material. They reported high water retention and ionic conductivity up to 31.7 g g−1 and 0.175 S cm−1 respectively which is higher than alternative membranes such as Nafion®.203 The higher water retention (which is responsible for increased ion conductivity in hydrated AEMs) was largely driven by the bamboo nanofibers. Many plant-based materials can be viable as fillers in water electrolysis due to their naturally water absorptive properties. Mokete et al. also utilised plant-based fillers in their proton-conducting CPE PEMs.204 Mokete et al. created a polymer matrix using a polyvinyl pyrrolidone (pvp) and polyvinyl alcohol (pva).204 They processed Chinese tallow seeds and pine bark into fillers, creating several CPEs to test in water electrolysers.204 The organic fillers used were chosen for their low-cost and environmentally friendly materials which avoid the use of damaging compounds present in commercial electrolytes.204 They observed good proton conductivity in their electrolytes with high faradaic efficiencies of ∼88% for H2 production.204 Plant-based fillers are not only environmentally sustainable as materials, but are also naturally abundant and do not have to be derived from mining or oil. It is also noted that plant-based SSEs are reportedly reaching performance levels close to that of commercial electrolytes.204
Some research has been done into water ice as a SSE as it is a highly ordered crystal which is also naturally abundant.205 Metal ions can be easily incorporated into a water ice electrolyte via dissolution in the liquid phase and freezing. Water ice is a promising material as an environmentally sustainable electrolyte due to its natural abundance and relative ease of processing. Deng et al. tested an ice SSE doped with 0.5 M H2SO4 and 1 M KOH for H+ and OH− conduction respectively. They observed high conductivities for both protons and hydroxide anions of up to 33.5 mS cm−1 and 27.3 mS cm−1 in the acidic and basic ice respectively at −10 °C.206 At −30 °C they observed ionic conductivities of 10 mS cm−1 and 7.1 mS cm−1 for acidic and basic ice respectively. Through the use of this electrolyser, they were able to conduct electrochemical ice splitting. They observed a decrease in current with decreased doping due to the reduced conductivity of the doped ice. Additionally, they tested various ice SSEs doped with various alkalis and acids, and determined that the ice splitting occurs via ion transport through the ice lattice as opposed to acid radical ions or metal cations.206 A paper by Guo et al. details their investigation into acidic ice doped with various metal ions and measurements of their ionic conductivities (Fig. 8c).205 They created metal-sulfuric acid solutions and froze them to produce metal-acid doped ice blocks which were tested in a circuit. They tested their doped ice electrolytes at temperatures between −5 °C and −20 °C. Similarly to Deng et al. they found that at higher temperatures (closer to 0 °C) the ionic conductivity was increased. Water ice SSEs could have potential applications for water splitting in extreme environments such as the arctic regions or in space exploration.
5.2 Advanced applications of SSEs in water electrolysis
Seawater electrolysis is ideally a sustainable method to harvest sea water to be converted into hydrogen for fuel using green energy.207 Using seawater directly instead of purified water helps to reduce energy costs in water purification and avoid depletion of freshwater sources. In most research revolving around seawater electrolysis, issues arise involving the dissolved ionic salts in water leading to corrosion of the electrodes and degradation of the membrane and electrolyte.208 Moreover, the differences in the physicochemical properties induced by the different geographic locations and seasons make this a difficult water source to standardize.207An electrolyte-specific challenge in seawater electrolysis presents itself in the Chlorine Evolution Reaction (CER) (due to the presence of Cl− in seawater), which has a similar thermodynamic potential to the OER at about 1.36 V (OER is 1.23 V) and as such is a competitive reaction to the OER. The equations for CER are presented in the following, under acidic and alkaline conditions respectively:
Acidic conditions
| 2Cl− → Cl2 + 2e− | (8) |
| Cl− + H2O → HClO + H+ + 2e− | (9) |
Alkaline conditions
| Cl− + 2OH− → ClO− + H2O + 2e− | (10) |
Due to the two electron transfer step, CER at high current densities can be favoured over OER,208 although this depends on the applied potential. In alkaline conditions, the overpotential for CER is greater than for OER at about 0.480 V, thus alkaline seawater electrolysis is preferential to OER and CER can be avoided.188 Another aspect to consider is the small concentration of bromine ions in seawater which undergo similar reactions, as well as microorganisms and bacteria.188,208 LDH-based materials were discussed in 5.1 for their potential as electrolytes based on their catalytic behaviour to the OER, which can improve the seawater electrolyser efficiency.78,188,208 A chloride ion-selective layer over the anode to suppress the CER (and in turn improve the OER and HER) may improve electrolyser performance, and this can also be controlled through the use of ion selective membranes as demonstrated in work by Xie et al.77 Tang et al. investigated direct seawater electrolysis using a gel electrolyte which had high OH− ion conduction.209 Their gel electrolyte was highly effective at self-dampening, a process where the seawater passing through the electrolyte is spontaneously diffused through the membrane as a vapour, avoiding ion crossover and CER (Fig. 9a).209,210 Their gel-based electrolyte was synthesized from polyvinyl alcohol which was frozen as an aqueous solution to form a porous membrane (Fig. 9b). It was immersed in aqueous KOH for the tests.209 They tested their reactor in a lab environment and found it operated continuously for about 400 hours. They also experimented with scaling-up the reactor and testing in a river with promising results.209 Inorganic solid electrolytes such as sodium ion super ionic conductor (NASICON) have also yielded good results as electrolytes for seawater electrolysis (Fig. 9c and d).211 NASICON has been extensively researched as an electrolyte in seawater batteries, however its properties as an electrolyte can also be applied to seawater electrolysis.211,212 Effective SSEs for seawater electrolysis must filter the sweater impurities, either through a self-dampening process or through effective ion conduction.
 | ||
| Fig. 9 Direct seawater electrolysis using solid electrolytes. (a) Tang et al. used a gel electrolyte and PTFE membrane to cause self-dampening for seawater to spontaneously evaporate into the electrolyte. (b) Gel electrolyte and PTFE membrane interface. Re-used under creative commons license, from ref. 209. (c) Sodium Ion Super Ionic Conductor (NASICON) solid state electrolyte structure, showing the ion transport through the material. (d) Seawater electrolysis using a NASICON solid electrolyte can help avoid the chlorine reduction reaction, improving water electrolysis performance. Re-used under creative commons license, from ref. 211. | ||
There has also been a significant amount of research into SSEs for photoelectrochemical (PEC) water splitting, and although not the topic of this review, it is worth briefly exploring some of the state-of-the-art technologies in this area. PEC devices present a potential method for direct conversion of solar energy into chemical energy in the form of hydrogen.213 Semiconductor materials used as photoelectrodes must have bandgaps with energies corresponding to the visible light range, high stability in alkaline or acidic environments, high charge carrier diffusion length, and appropriate band edge positions relative to redox potentials for water splitting.213 Wang et al. report the development of quasi-solid state electrolytes for PEC devices.214 They developed a CoFe dispersed polyacrylamide (PAM) hydrogel protector for the electrodes using the solid-state network structure of PAM hydrogels, with potassium salt used for ion transfer through the electrolyte. With high concentration of ionic solvent, this effectively makes an ionically conductive quasi-solid state electrolyte.214 The increased hydrophilicity of the PAM hydrogel, thanks to the water molecules contained within it, enhances the photocurrent performance of the photoelectrodes by facilitating particle absorption from the electrolyte. Its low viscosity also allows it to penetrate into the photoelectrode pores. The authors compared the ionic conductivities of the liquid and quasi-solid-state electrolyte (2.52 mS cm−1 and 1.96 mS cm−1 respectively), which also resulted in slightly lower current densities at the photoanode using a quasi-solid state electrolyte.214 This was determined to result in reductions in ion diffusion capability due to the fixing of potassium ions to the solid network.214
While we will not dwell on these advancements, worth mentioning are also the efforts to decouple water splitting through redox mediators, such that the OER is coupled with the reduction of the mediator rather than the direct HER.42,215 Lastly, we would like to refer the readers to a review on mixed ionic-electronic conducting (MIEC) membrane reactors, which aim to enhance the hydrogen production rate by extracting the produced oxygen.40
6. Conclusions
Climate change is driven by increased pollution and carbon emissions caused by human activity and the use of fossil fuels. It is evident that alternative green forms of energy must be developed as replacements to fossil fuel sources. Wind and solar energy have been implemented at large-scale around the globe, yet their unpredictable fluctuations make them unreliable alternatives. Hydrogen energy generated from electrochemical water splitting could be utilised as a form of chemical energy storage and green energy source. The majority of green hydrogen produced today is produced via reactors utilising aqueous electrolytes which often results in side-effects such as corrosion leading to electrode degradation and instability in the electrolyte pH, reducing reactor efficiency. Whilst solid state electrolytes (SSE) have been known since the 20th century, their use in water electrolysis has only recently resurged to the forefront of the research community, with a particular focus on the development of novel materials. Inorganic solid electrolytes (ISEs), such as solid oxides and perovskites, have seen use in solid oxide water electrolysis and other high temperature applications due to their high ion conductivity. Solid polymer electrolytes (SPEs) are currently also used as electrolytes in the water splitting process, in particular for anion and proton exchange membrane water electrolysers. Despite their implementation in industrial water electrolysers and advantages over liquid electrolytes, SSEs have many practical limitations to overcome for more mainstream use in water electrolysis. In the case of ISEs, the high operation temperatures lead to mismatches in thermal expansion coefficients, formation of cracks and low contact between the electrolyte and electrode. Polymers are faced with lower ionic conductivity, failure due to drying at temperatures above 80 °C and environmental concerns surrounding the use of fluorinated compounds. Many research groups have dedicated their efforts to overcoming these limitations through targeted material designs, such as doping strategies to close the gap in thermal expansion coefficients and using layered double hydroxide materials to merge the roles of the electrolyte and electrocatalyst. On the polymer front, nano-film alternatives to Nafion® using hydrocarbons with quaternary ammonium ions are being researched, but despite the possibility of using them at higher-temperature operation regimes, their low ionic conductivity is the limiting factor. A solution to these problems comes in the form of composite polymer electrolytes: polymer matrices with inorganic filler, combining the high ionic conductivity of ISEs with the high interfacial contact of SPEs. More novel materials use metal organic frameworks and graphene films as electrolyte modifiers. Nanoscale SSEs and composite materials have opened new avenues in the search for new, promising ways to overcome the limitations of not only the already-existing materials, but also of the traditional aqueous electrolytes.While there are many outstanding advancements in terms of material design, there are just as many unanswered questions about the evolution of these materials under reaction conditions. In particular, replicating the electrocatalytic water splitting under industry-relevant conditions is challenging, yet imperative, since many of the problems encountered under operational conditions are hard to extrapolate from lab-scale experiments alone. Electrochemistry has been developing hand in hand with advanced characterization methods and material science, and research in this area must be continued on both a fundamental and industrial level in order to realize a society fuelled by green hydrogen.
Author contributions
Adrian Dowling and Ioana-Maria Popa are joint first authors and contributed equally to the manuscript.Conflicts of interest
There are no conflicts of interest to declare.Data availability
All data supporting this study are included in the article. Additional derived data that support this work are available from the corresponding author upon reasonable request.Acknowledgements
Dr Leila Negahdar wishes to thank Science Foundation Ireland for the support (grant number 22/FFP–P/11527).References
- X. Zhao, Highl. Sci. Eng. Technol., 2024, 90, 123–128 CrossRef.
- M. Crippa, D. Guizzardi, E. Pisoni, E. Solazzo, A. Guion, M. Muntean, A. Florczyk, M. Schiavina, M. Melchiorri and A. F. Hutfilter, Environ. Res. Lett., 2021, 16, 074033 CrossRef CAS.
- C. Sulaiman and A. S. Abdul-Rahim, Sage Open, 2018, 8, 215824401876591 CrossRef.
- A. Al-Mamoori, A. Krishnamurthy, A. A. Rownaghi and F. Rezaei, Energy Technol., 2017, 5, 834–849 CrossRef.
- Z. Zhao, K. Chong, J. Jiang, K. Wilson, X. Zhang and F. Wang, Renewable Sustainable Energy Rev., 2018, 97, 580–591 CrossRef CAS.
- S. C. Patil, S. T. M. Fahim, J. H. Lee and K. L. Gilliard-AbdulAziz, ACS Energy Lett., 2024, 10, 371–392 CrossRef.
- T. M. Gür, Energy Environ. Sci., 2018, 11, 2696–2767 RSC.
- Y. Zheng, J. Wang, B. Yu, W. Zhang, J. Chen, J. Qiao and J. Zhang, Chem. Soc. Rev., 2017, 46, 1427–1463 RSC.
- J. Gómez and R. Castro, Energies, 2024, 17, 3110 CrossRef.
- P. Kut, K. Pietrucha-Urbanik and M. Zeleňáková, Energies, 2024, 17, 1862 CrossRef CAS.
- C. Alves, G. Castro, R. Coelho and L. Hocevar, Hydrogen Technologies: Recent Advances, New Perspectives, and Applications, in Hydrogen Technologies - Advances Insights, and Applications, ed. Z. Abdallah and N. Aldoumani, IntechOpen, 2024, ch. 1. Search PubMed.
- J. G. Ingersoll, Hydrogen, 2022, 3, 84–111 CrossRef CAS.
- K. Ayers, N. Danilovic, R. Ouimet, M. Carmo, B. Pivovar and M. Bornstein, Annu. Rev. Chem. Biomol. Eng., 2019, 10, 219–239 CrossRef CAS PubMed.
- R. Ali, M. Elhoweity, A. Zahran and E. Elfadaly, Int. J. Environ. Res., 2023, 2, 105–119 Search PubMed.
- A. Ursua, L. M. Gandia and P. Sanchis, Proc. IEEE, 2012, 100, 410–426 CAS.
- Q. Wang, S. Okunaka, H. Tokudome, T. Hisatomi, M. Nakabayashi, N. Shibata, T. Yamada and K. Domen, Joule, 2018, 2, 2667–2680 CrossRef CAS.
- M. Medhat, A. M. Elshaer, M. Soliman, S. Ebrahim and M. Khalil, Integr. Nano, 2024, 1, 1–9 CrossRef.
- T. Hisatomi, J. Kubota and K. Domen, Chem. Soc. Rev., 2014, 43, 7520–7535 RSC.
- Z. Y. Yu, Y. Duan, X. Y. Feng, X. Yu, M. R. Gao and S. H. Yu, Adv. Mater., 2021, 33, 2007100 CrossRef CAS PubMed.
- X. Gao, Y. Chen, Y. Wang, L. Zhao, X. Zhao, J. Du, H. Wu and A. Chen, Nano-Micro Lett., 2024, 16, 237 CrossRef PubMed.
- H. He, Y. Zhang, W. Zhang, Y. Li, X. Zhu, P. Wang and D. Hu, ACS Appl. Mater. Interfaces, 2022, 14, 834–849 CrossRef CAS PubMed.
- Z. Taie, X. Peng, D. Kulkarni, I. V. Zenyuk, A. Z. Weber, C. Hagen and N. Danilovic, ACS Appl. Mater. Interfaces , 2020, 12, 52701–52712 CrossRef CAS PubMed.
- M. Li, C. Wang, Z. Chen, K. Xu and J. Lu, Chem. Rev., 2020, 120, 6783–6819 CrossRef CAS PubMed.
- T. Shinagawa and K. Takanabe, ChemSusChem, 2017, 10, 1318–1336 CrossRef CAS PubMed.
- C. Ding, J. Shi, Z. Wang and C. Li, ACS Catal., 2016, 7, 675–688 CrossRef.
- Y. Lv, Y. Xiao, L. Ma, C. Zhi and S. Chen, Adv. Mater., 2022, 34, 2106409 CrossRef CAS PubMed.
- J. Jin, L. Li, X. Nie, T. Xiao and Z. Liu, Adv. Mater. Interfaces, 2022, 9, 2101478 CrossRef CAS.
- M. Schalenbach, Y. E. Durmus, H. Tempel, H. Kungl and R.-A. Eichel, Research Square Platform LLC, 2022 Search PubMed.
- L. M. Aeshala, R. G. Uppaluri and A. Verma, J. CO2 Util., 2013, 3–4, 49–55 CrossRef CAS.
- M. Faraj, M. Boccia, H. Miller, F. Martini, S. Borsacchi, M. Geppi and A. Pucci, Int. J. Hydrogen Energy, 2012, 37, 14992–15002 CrossRef CAS.
- I. Ganesh, J. Solid State Electrochem., 2023, 29, 149–176 CrossRef.
- Y. Leng, G. Chen, A. J. Mendoza, T. B. Tighe, M. A. Hickner and C. Y. Wang, J. Am. Chem. Soc., 2012, 134, 9054–9057 CrossRef CAS PubMed.
- A. Luthfiah and C. W. Lee, ChemCatChem, 2023, 15(21), e202300702 CrossRef CAS.
- E. J. Park, C. B. Capuano, K. E. Ayers and C. Bae, J. Power Sources, 2018, 375, 367–372 CrossRef CAS.
- F. P. Lohmann-Richters, M. Müller and M. Carmo, J. Electrochem. Soc., 2020, 167, 084512 CrossRef CAS.
- L. Wang, J. Li, G. Lu, W. Li, Q. Tao, C. Shi, H. Jin, G. Chen and S. Wang, Front. Mater., 2020, 7, 111 CrossRef.
- L. Hu, X. Gao, H. Wang, Y. Song, Y. Zhu, Z. Tao, B. Yuan and R. Hu, Small, 2024, 20(31), 2312251 CrossRef CAS PubMed.
- J. H. Park, S. Jung, P. L. Handayani, N. Aluru, T. Kim, S. B. Lee, U. H. Choi and J. Lee, J. Phys. Chem. C, 2022, 126, 16777–16784 CrossRef CAS.
- P. Oh, H. Lee, S. Park, H. Cha, J. Kim and J. Cho, Adv. Energy Mater., 2020, 10, 2000904 CrossRef CAS.
- B. Wang, T. Li, Z. Wang, M. H. D. Othman, S. Liu and R. Xiao, Green Chem., 2023, 25, 6930–6948 RSC.
- S. S. Jolly, A. R. Twinkle, B. S. Arun Sasi and R. Reshma, Next Energy, 2025, 8, 100343 CrossRef CAS.
- P. J. McHugh, A. D. Stergiou and M. D. Symes, Adv. Energy Mater., 2020, 10(44), 2002453 CrossRef CAS.
- P. A. Garcia-Salaberri, L. van Eijk, W. Bangay, K. J. Ferner, M. H. Ha, M. Moore, I. Perea, A. Kusoglu, M. Secanell, P. K. Das, N. Firas, S. Pylypenko, M. Novy, M. Yandrasits, S. C. Saha, A. Bayat, S. Litster and I. V. Zenyuk, ACS Appl. Energy Mater., 2025, 8, 13050–13121 CrossRef CAS PubMed.
- K. E. Ayers, E. B. Anderson, C. B. Capuano, B. D. Carter, L. T. Dalton, G. Hanlon, J. Manco and M. Niedzwiecki, ECS Trans., 2010, 33, 3–15 CrossRef CAS.
- R. De Levie, J. Electroanal. Chem., 1999, 476, 92–93 CrossRef CAS.
- S. Simon Araya, V. Liso, X. Cui, N. Li, J. Zhu, S. L. Sahlin, S. H. Jensen, M. P. Nielsen and S. K. Kær, Energies, 2020, 13, 596 CrossRef.
- S. Crawford, E. Thimsen and P. Biswas, J. Electrochem. Soc., 2009, 156, H346 CrossRef CAS.
- S.-W. Ryu, Y. Zhang, B. Leung, C. Yerino and J. Han, Semicond. Sci. Technol., 2012, 27, 015014 CrossRef.
- Y. Yan, B. Y. Xia, B. Zhao and X. Wang, J. Mater. Chem. A, 2016, 4, 17587–17603 RSC.
- Z. Feng, C. Dai, P. Shi, X. Lei, R. Guo, B. Wang, X. Liu and J. You, Chem. Eng. J., 2024, 485, 149992 CrossRef CAS.
- L. Quan, H. Jiang, G. Mei, Y. Sun and B. You, Chem. Rev., 2024, 124, 3694–3812 CrossRef CAS PubMed.
- M. Yu, E. Budiyanto and H. Tüysüz, Angew. Chem., Int. Ed., 2022, 61(1), e202103824 CrossRef CAS PubMed.
- H. Jin, B. Ruqia, Y. Park, H. J. Kim, H. S. Oh, S. I. Choi and K. Lee, Adv. Energy Mater., 2021, 11, 2003188 CrossRef CAS.
- A. A. Feidenhansl, Y. N. Regmi, C. Wei, D. Xia, J. Kibsgaard and L. A. King, Chem. Rev., 2024, 124, 5617–5667 CrossRef PubMed.
- C. C. L. McCrory, S. Jung, J. C. Peters and T. F. Jaramillo, J. Am. Chem. Soc., 2013, 135, 16977–16987 CrossRef CAS PubMed.
- C. C. L. McCrory, S. Jung, I. M. Ferrer, S. M. Chatman, J. C. Peters and T. F. Jaramillo, J. Am. Chem. Soc., 2015, 137, 4347–4357 CrossRef CAS PubMed.
- A. Raveendran, M. Chandran and R. Dhanusuraman, RSC Adv., 2023, 13, 3843–3876 RSC.
- S. Tosoni, G. Di Liberto, I. Matanovic and G. Pacchioni, J. Power Sources, 2023, 556, 232492 CrossRef CAS.
- A. Lasia, in Handbook of Fuel Cells, 2010 Search PubMed.
- A. J. Shih, M. C. O. Monteiro, F. Dattila, D. Pavesi, M. Philips, A. H. M. Da Silva, R. E. Vos, K. Ojha, S. Park, O. Van Der Heijden, G. Marcandalli, A. Goyal, M. Villalba, X. Chen, G. T. K. K. Gunasooriya, I. McCrum, R. Mom, N. López and M. T. M. Koper, Nat. Rev. Methods Primers, 2022, 2, 84 CrossRef CAS.
- J. Owen, in Comprehensive Polymer Science and Supplements, ed. G. Allen, J. C. Bevington, Pergamon, Amsterdam, 1989, pp. 669–686 Search PubMed.
- S. C. Sand, J. L. M. Rupp and B. Yildiz, Chem. Soc. Rev., 2025, 54, 178–200 RSC.
- T. Norby, Solid State Ionics, 1999, 125, 1–11 CrossRef CAS.
- Y. Wang, X. Fu, Y. He, X. Li and Y. Feng, Appl. Surf. Sci., 2025, 693, 162729 CrossRef CAS.
- L. Devaraj, S. V. Thummalapalli, N. Fonseca, H. Nazir, K. Song and A. M. Kannan, Mater. Today Sustain., 2024, 25, 100614 Search PubMed.
- X. Yu and A. Manthiram, Energy Storage Mater., 2021, 34, 282–300 CrossRef.
- H. Yang and N. Wu, Energy Sci. Eng., 2022, 10, 1643–1671 CrossRef CAS.
- S. Kundu, A. Kraytsberg and Y. Ein-Eli, J. Solid State Electrochem., 2022, 26, 1809–1838 CrossRef CAS.
- S. Shi, Y. Qi, H. Li and L. G. Hector Jr, J. Phys. Chem. C, 2013, 117, 8579–8593 CrossRef CAS.
- Z. Sun, M. Liu, Y. Zhu, R. Xu, Z. Chen, P. Zhang, Z. Lu, P. Wang and C. Wang, Sustainability, 2022, 14, 9090 CrossRef CAS.
- Z. Huang, L. Chen, B. Huang, B. Xu, G. Shao, H. Wang, Y. Li and C.-A. Wang, ACS Appl. Mater. Interfaces, 2020, 12, 56118–56125 CrossRef CAS PubMed.
- G. Baure, H. Zhou, C.-C. Chung, M. A. Stozhkova, J. L. Jones and J. C. Nino, Acta Mater., 2017, 133, 81–89 CrossRef CAS.
- H. Qu, T. Ding, X. Zhang, D. Qiu, P. Chen, D. Zheng, D. Lu and D. Qu, Nano Energy, 2025, 110887 CrossRef CAS.
- J. G. Swallow, W. H. Woodford, Y. Chen, Q. Lu, J. J. Kim, D. Chen, Y. M. Chiang, W. C. Carter, B. Yildiz, H. L. Tuller and K. J. Van Vliet, J. Electroceram., 2014, 32, 3–27 CrossRef CAS.
- R. Raj, MRS Commun., 2021, 11, 644–649 CrossRef CAS.
- J. Tippens, J. C. Miers, A. Afshar, J. A. Lewis, F. J. Q. Cortes, H. Qiao, T. S. Marchese, C. V. Di Leo, C. Saldana and M. T. McDowell, ACS Energy Lett., 2019, 4, 1475–1483 CrossRef CAS.
- H. Xie, Z. Zhao, T. Liu, Y. Wu, C. Lan, W. Jiang, L. Zhu, Y. Wang, D. Yang and Z. Shao, Nature, 2022, 612, 673–678 CrossRef CAS PubMed.
- H. Wang, H. Wang, Z. Wang, J. Qiu, B. Wang, R. Guo, J. You, X. Lei and X. Liu, J. Alloys Compd., 2024, 1002, 175368 CrossRef CAS.
- M. Schalenbach, L. Keller, B. Janotta, A. Bauer, H. Tempel, H. Kungl, M. Bonnet and R.-A. Eichel, J. Electrochem. Soc., 2022, 169, 094510 CrossRef CAS.
- A. Luthfiah and C. W. Lee, ChemCatChem, 2023, 15, e202300702 CrossRef CAS.
- S. Xu, P. Zhang, L. Li, M. W. Moon, C. H. Chung, H. Li, J. Y. Lee and P. J. Yoo, Small, 2025, 21(24), 2502000 CrossRef CAS PubMed.
- H. Yamada, D. Tsunoe, S. Shiraishi and G. Isomichi, J. Phys. Chem. C, 2015, 119, 5412–5419 CrossRef CAS.
- R. Xu, F. Liu, Y. Ye, H. Chen, R. R. Yang, Y. Ma, W. Huang, J. Wan and Y. Cui, Adv. Mater., 2021, 33, 2104009 CrossRef CAS PubMed.
- M. Balaish, J. C. Gonzalez-Rosillo, K. J. Kim, Y. Zhu, Z. D. Hood and J. L. M. Rupp, Nat. Energy, 2021, 6, 227–239 CrossRef CAS.
- J. Fu, Z. Li, X. Zhou and X. Guo, Mater. Adv., 2022, 3, 3809–3819 RSC.
- N. Schichtel, C. Korte, D. Hesse and J. Janek, Phys. Chem. Chem. Phys., 2009, 11, 3043–3048 RSC.
- C. Wang, B. B. Xu, X. Zhang, W. Sun, J. Chen, H. Pan, M. Yan and Y. Jiang, Small, 2022, 18, 2107064 CrossRef CAS PubMed.
- X. He, Y. Zhu and Y. Mo, Nat. Commun., 2017, 8, 15893 CrossRef CAS PubMed.
- A. Pandiyan, A. Uthayakumar, R. Subrayan, S. W. Cha and S. B. Krishna Moorthy, Nanomater. Energy, 2019, 8, 2–22 CrossRef.
- W. Li, B. Guan, T. Yang, Z. Li, W. Shi, H. Tian, L. Ma, T. L. Kalapos and X. Liu, J. Power Sources, 2021, 495, 229764 CrossRef CAS.
- W. Tang, H. Ding, W. Bian, C. Y. Regalado Vera, J. Y. Gomez, Y. Dong, J. Li, W. Wu, W. Fan, M. Zhou, C. Gore, B. M. Blackburn, H. Luo and D. Ding, Small, 2022, 18, 2201953 CrossRef CAS PubMed.
- A. Hauch, R. Küngas, P. Blennow, A. B. Hansen, J. B. Hansen, B. V. Mathiesen and M. B. Mogensen, Science, 2020, 370, eaba6118 CrossRef CAS PubMed.
- Q. Shao, D. Jin, Y. Lu, Y. Yu, L. Luo, X. Sun, C. Guan and J.-Q. Wang, Int. J. Hydrogen Energy, 2024, 57, 709–716 CrossRef CAS.
- Y. Zheng, Z. Chen and J. Zhang, Electrochem. Energy Rev., 2021, 4, 508–517 CrossRef CAS.
- Z. Liu, D. Cheng, Y. Zhu, M. Liang, M. Yang, G. Yang, R. Ran, W. Wang, W. Zhou and Z. Shao, Chem. Eng. J., 2022, 450, 137787 CrossRef CAS.
- Y. Lu, M. Y. Shah, M. Khalid, N. Mushtaq, M. Yousaf and N. Akbar, Ceram. Int., 2024, 50, 49748–49758 CrossRef CAS.
- M. A. K. Y. Shah, Y. Lu, N. Mushtaq, M. Yousaf, S. Rauf, M. I. Asghar, P. D. Lund and B. Zhu, Sustainable Energy Fuels , 2022, 6, 3794–3805 RSC.
- G. Lu, G. Meng, Q. Liu, L. Feng, J. Luo, X. Liu, Y. Luo and P. K. Chu, Adv. Powder Mater., 2024, 3, 100154 CrossRef.
- Z. Li, J. Fu, X. Zhou, S. Gui, L. Wei, H. Yang, H. Li and X. Guo, Advanced Science, 2023, 10, 2201718 CrossRef CAS PubMed.
- Z. Gadjourova, Y. G. Andreev, D. P. Tunstall and P. G. Bruce, Nature, 2001, 412, 520–523 CrossRef CAS PubMed.
- S. Cheng, D. M. Smith and C. Y. Li, Macromolecules, 2014, 47, 3978–3986 CrossRef CAS.
- Z. A. R. Gautama, Y. A. Hutapea, B. Hwang, J. Matsuda, A. Mufundirwa, T. Sugiyama, M. Ariyoshi, S. Fujikawa, S. M. Lyth, A. Hayashi, K. Sasaki and M. Nishihara, J. Membr. Sci., 2022, 658, 120734 CrossRef CAS.
- S. Z. Oener, S. Ardo and S. W. Boettcher, ACS Energy Lett., 2017, 2, 2625–2634 CrossRef CAS.
- Y. Nagao, Langmuir, 2017, 33, 12547–12558 CrossRef CAS PubMed.
- N. Agmon, Chem. Phys. Lett., 1995, 244, 456–462 CrossRef CAS.
- J. Choi, H. Kim, S. Jeon, M. G. Shin, J. Y. Seo, Y. I. Park, H. Park, A. S. Lee, C. Lee, M. Kim, H. S. Cho and J. H. Lee, Small, 2023, 19(37), 2300825 CrossRef CAS PubMed.
- D. Dong, W. Zhang, A. C. T. van Duin and D. Bedrov, J. Phys. Chem. Lett., 2018, 9, 825–829 CrossRef CAS PubMed.
- Z. Xue, D. He and X. Xie, J. Mater. Chem. A, 2015, 3, 19218–19253 RSC.
- M. Hellström, V. Quaranta and J. Behler, Chem. Sci., 2019, 10, 1232–1243 RSC.
- S. Garbe, J. Futter, T. J. Schmidt and L. Gubler, Electrochim. Acta, 2021, 377, 138046 CrossRef CAS.
- M. Feng, R. Qu, Z. Wei, L. Wang, P. Sun and Z. Wang, Sci. Rep., 2015, 5, 9859 CrossRef CAS PubMed.
- H. Ito, T. Maeda, A. Nakano and H. Takenaka, Int. J. Hydrogen Energy, 2011, 36, 10527–10540 CrossRef CAS.
- P. Waribam, K. Jaiyen, C. Samart, M. Ogawa, G. Guan and S. Kongparakul, Catal. Today, 2023, 407, 96–106 CrossRef CAS.
- A. Wang, C. Breakwell, F. Foglia, R. Tan, L. Lovell, X. Wei, T. Wong, N. Meng, H. Li, A. Seel, M. Sarter, K. Smith, A. Alvarez-Fernandez, M. Furedi, S. Guldin, M. M. Britton, N. B. McKeown, K. E. Jelfs and Q. Song, Nature, 2024, 635, 353–358 CrossRef CAS PubMed.
- H. R. Corti, Curr. Opin. Electrochem., 2022, 36, 101109 CrossRef CAS.
- Z. Zhang, Z. Xia, J. Huang, F. Jing, S. Wang and G. Sun, J. Energy Chem., 2022, 69, 569–575 CrossRef CAS.
- W. Ng, W. Wong, N. Rosli and K. Loh, Separations, 2023, 10, 424 CrossRef CAS.
- B. Motealleh, Z. Liu, R. I. Masel, J. P. Sculley, Z. R. Ni and L. Meroueh, Int. J. Hydrogen Energy, 2021, 46, 3379–3386 CrossRef CAS.
- S. Xu, P. Zhang, L. Li, M.-W. Moon, C.-H. Chung, H. Li, J. Y. Lee and P. J. Yoo, Small, 2025, 21, 2502000 CrossRef CAS PubMed.
- Y. Pan, L. Li, D. Liu, F. Wang, M. Xia and F. Wang, J. Environ. Chem. Eng., 2025, 13, 115284 CrossRef CAS.
- W. Wieczorek, J. R. Stevens and Z. Florjańczyk, Solid State Ionics, 1996, 85, 67–72 CrossRef CAS.
- M. U. Javed, S. Muhammad, Z. Wang, J. Li, X. Wu, A. Chukwuka, Y. Zhang, K. Wang and B. Guo, J. Power Sources, 2025, 640, 236720 CrossRef CAS.
- H. Sun, X. Xu, H. Kim, W. Jung, W. Zhou and Z. Shao, Energy Environ. Mater., 2023, 6(5), e12441 CrossRef CAS.
- F.-Y. Chen, Z.-Y. Wu, Z. Adler and H. Wang, Joule, 2021, 5, 1704–1731 CrossRef CAS.
- B. Han, K. A. Stoerzinger, V. Tileli, A. D. Gamalski, E. A. Stach and Y. Shao-Horn, Nat. Mater., 2017, 16, 121–126 CrossRef CAS PubMed.
- N. Thissen, J. Hoffmann, S. Tigges, D. A. M. Vogel, J. J. Thoede, S. Khan, N. Schmitt, S. Heumann, B. J. M. Etzold and A. K. Mechler, ChemElectroChem, 2024, 11(1), e202300432 CrossRef CAS.
- The Sustainable Development Goals Report 2025, U. Nations, 2025 Search PubMed.
- A. Brisse, J. Schefold and M. Zahid, Int. J. Hydrogen Energy, 2008, 33, 5375–5382 CrossRef CAS.
- J. Brauns and T. Turek, Processes, 2020, 8, 248 CrossRef CAS.
- S. S. Kumar and H. Lim, Sustainable Energy Fuels , 2023, 7, 3560–3583 RSC.
- W. A. P. Vermeiren, J. Moreels and R. Leysen, Int. J. Hydrogen Energy, 1998, 23, 321–324 CrossRef.
- K. Zhang and R. Zou, Small, 2021, 17, 2100129 CrossRef CAS PubMed.
- K. Zeng and D. Zhang, Prog. Energy Combust. Sci., 2010, 36, 307–326 CrossRef CAS.
- R. F. de Souza, J. C. Padilha, R. S. Gonçalves and J. Rault-Berthelot, Electrochem. Commun., 2006, 8, 211–216 CrossRef CAS.
- K. Zhang, X. Liang, L. Wang, K. Sun, Y. Wang, Z. Xie, Q. Wu, X. Bai, M. S. Hamdy, H. Chen and X. Zou, Nano Res. Energy, 2022, 1, e9120032 CrossRef.
- F. Barbir, Sol. Energy, 2005, 78, 661–669 CrossRef CAS.
- U. Babic, M. Suermann, F. N. Büchi, L. Gubler and T. J. Schmidt, J. Electrochem. Soc., 2017, 164, F387–F399 CrossRef CAS.
- M. Carmo, D. L. Fritz, J. Mergel and D. Stolten, Int. J. Hydrogen Energy, 2013, 38, 4901–4934 CrossRef CAS.
- S. Grigoriev, V. Porembsky and V. Fateev, Int. J. Hydrogen Energy, 2006, 31, 171–175 CrossRef CAS.
- Z. P. Ifkovits, J. M. Evans, M. C. Meier, K. M. Papadantonakis and N. S. Lewis, Energy Environ. Sci. , 2021, 14, 4740–4759 RSC.
- D. S. Falcão and A. M. F. R. Pinto, J. Cleaner Prod., 2020, 261 CrossRef.
- S. Stucki, G. G. Scherer, S. Schlagowski and E. Fischer, J. Appl. Electrochem., 1998, 28, 1041–1049 CrossRef CAS.
- A. Bayat, P. K. Das, G. Saha and S. C. Saha, Eng, 2025, 6, 72 CrossRef.
- H. R. Corti, Curr. Opin. Electrochem., 2022, 36 CAS.
- M. B. Z. Hegazy, K. Harrath, D. Tetzlaff, M. Smialkowski, D. Siegmund, J. Li, R. Cao and U. P. Apfel, iScience, 2022, 25, 105148 CrossRef CAS PubMed.
- S. Garbe, J. Futter, T. J. Schmidt and L. Gubler, Electrochim. Acta, 2021, 377 CrossRef CAS.
- G. M. Su, I. A. Cordova, M. A. Yandrasits, M. Lindell, J. Feng, C. Wang and A. Kusoglu, J. Am. Chem. Soc., 2019, 141, 13547–13561 CrossRef CAS PubMed.
- C. Rakousky, U. Reimer, K. Wippermann, M. Carmo, W. Lueke and D. Stolten, J. Power Sources, 2016, 326, 120–128 CrossRef CAS.
- O. Panchenko, E. Borgardt, W. Zwaygardt, F. J. Hackemüller, M. Bram, N. Kardjilov, T. Arlt, I. Manke, M. Müller, D. Stolten and W. Lehnert, J. Power Sources, 2018, 390, 108–115 CrossRef CAS.
- M. A. Hoeh, T. Arlt, I. Manke, J. Banhart, D. L. Fritz, W. Maier and W. Lehnert, Electrochem. Commun., 2015, 55, 55–59 CrossRef CAS.
- Y. Yang, P. Li, X. Zheng, W. Sun, S. X. Dou, T. Ma and H. Pan, Chem. Soc. Rev., 2022, 51, 9620–9693 RSC.
- D. Li, A. R. Motz, C. Bae, C. Fujimoto, G. Yang, F.-Y. Zhang, K. E. Ayers and Y. S. Kim, Energy Environ. Sci. , 2021, 14, 3393–3419 RSC.
- N. Du, C. Roy, R. Peach, M. Turnbull, S. Thiele and C. Bock, Chem. Rev., 2022, 122, 11830–11895 CrossRef CAS PubMed.
- S. A. Lee, J. Kim, K. C. Kwon, S. H. Park and H. W. Jang, Carbon Neutralization, 2022, 1, 26–48 CrossRef.
- U. K. Ghorui, G. Sivaguru, U. B. Teja, A. M, S. Ramakrishna, S. Ghosh, G. K. Dalapati and S. Chakrabortty, ACS Appl. Energy Mater., 2024, 7, 7649–7676 CrossRef CAS.
- R. Bernat, J. Milewski, O. Dybinski, A. Martsinchyk and P. Shuhayeu, Energies, 2024, 17, 5030 CrossRef CAS.
- A. R. Motz, D. Li, A. Keane, L. D. Manriquez, E. J. Park, S. Maurya, H. Chung, C. Fujimoto, J. Jeon, M. K. Pagels, C. Bae, K. E. Ayers and Y. S. Kim, J. Mater. Chem. A, 2021, 9, 22670–22683 RSC.
- I. Vincent and D. Bessarabov, Renewable Sustainable Energy Rev., 2018, 81, 1690–1704 CrossRef CAS.
- D. Henkensmeier, M. Najibah, C. Harms, J. Žitka, J. Hnát and K. Bouzek, J. Electrochem. Energy Convers. Storage, 2021, 18, 1–46 Search PubMed.
- M. Chatenet, B. G. Pollet, D. R. Dekel, F. Dionigi, J. Deseure, P. Millet, R. D. Braatz, M. Z. Bazant, M. Eikerling, I. Staffell, P. Balcombe, Y. Shao-Horn and H. Schäfer, Chem. Soc. Rev., 2022, 51, 4583–4762 RSC.
- N. Chen and Y. M. Lee, Prog. Polym. Sci., 2021, 113, 101345 CrossRef CAS.
- C. Liu, Z. Geng, X. Wang, W. Liu, Y. Wang, Q. Xia, W. Li, L. Jin and C. Zhang, J. Energy Chem., 2024, 90, 348–369 CrossRef CAS.
- E. J. Park, P. Jannasch, K. Miyatake, C. Bae, K. Noonan, C. Fujimoto, S. Holdcroft, J. R. Varcoe, D. Henkensmeier, M. D. Guiver and Y. S. Kim, Chem. Soc. Rev., 2024, 53, 5704–5780 RSC.
- M. Muhyuddin, C. Santoro, L. Osmieri, V. C. Ficca, A. Friedman, K. Yassin, G. Pagot, E. Negro, A. Konovalova and G. Lindquist, Chem. Rev., 2025, 125, 6906–6976 CrossRef CAS PubMed.
- A. Loh, X. Li, S. Sluijter, P. Shirvanian, Q. Lai and Y. Liang, Hydrogen, 2023, 4, 257–271 CrossRef CAS.
- C. Li and J.-B. Baek, Nano Energy, 2021, 87, 106162 CrossRef CAS.
- H. B. T. Smolinka, J. Garche and M. Kusnezoff, Electrochemical Power Sources: Fundamentals, Systems, and Applications, 2022 Search PubMed.
- S. D. Ebbesen, S. H. Jensen, A. Hauch and M. B. Mogensen, Chem. Rev., 2014, 114, 10697–10734 CrossRef CAS PubMed.
- A. Nechache and S. Hody, Renewable Sustainable Energy Rev., 2021, 149, 111322 CrossRef CAS.
- T. Sakai, S. Matsushita, H. Matsumoto, S. Okada, S. Hashimoto and T. Ishihara, Int. J. Hydrogen Energy, 2009, 34, 56–63 CrossRef CAS.
- M. A. Laguna-Bercero, J. Power Sources, 2012, 203, 4–16 CrossRef CAS.
- S. Elangovan, J. J. Hartvigsen and L. J. Frost, Int. J. Appl. Ceram. Technol., 2007, 4, 109–118 CrossRef CAS.
- K. Chen, S.-S. Liu, N. Ai, M. Koyama and S. P. Jiang, Phys. Chem. Chem. Phys., 2015, 17, 31308–31315 RSC.
- S. P. Jiang, Asia-Pac. J. Chem. Eng., 2016, 11, 386–391 CrossRef CAS.
- J. T. S. Irvine, D. Neagu, M. C. Verbraeken, C. Chatzichristodoulou, C. Graves and M. B. Mogensen, Nat. Energy, 2016, 1, 15014 CrossRef CAS.
- A. Hauch, S. H. Jensen, S. Ramousse and M. Mogensen, J. Electrochem. Soc., 2006, 153, A1741 CrossRef CAS.
- A. Nechache, M. Cassir and A. Ringuedé, J. Power Sources, 2014, 258, 164–181 CrossRef CAS.
- C. Graves, S. D. Ebbesen, S. H. Jensen, S. B. Simonsen and M. B. Mogensen, Nat. Mater., 2015, 14, 239–244 CrossRef CAS PubMed.
- Y. Wang, W. Li, L. Ma, W. Li and X. Liu, J. Mater. Sci. Technol., 2020, 55, 35–55 CrossRef CAS.
- F. Tietz, D. Sebold, A. Brisse and J. Schefold, J. Power Sources, 2013, 223, 129–135 CrossRef CAS.
- S. Sebbahi, N. Nabil, A. Alaoui-Belghiti, S. Laasri, S. Rachidi and A. Hajjaji, Mater. Today: Proc., 2022, 66, 140–145 CAS.
- L. Zeng and T. S. Zhao, Nano Energy, 2015, 11, 110–118 CrossRef CAS.
- G. Arrabito, A. Bonasera, G. Prestopino, A. Orsini, A. Mattoccia, E. Martinelli, B. Pignataro and P. Medaglia, Crystals, 2019, 9, 361 CrossRef CAS.
- Y. Wang, Z. Ying, Y. Gao and L. Shi, Chem. – Eur. J., 2024, 30(6), e202303025 CrossRef CAS PubMed.
- B. Lv, Y. Yang, C. Yang, Z. Huang, Y. Zhou, W. Song, J. Hao and Z. Shao, Int. J. Energy Res., 2022, 46, 11892–11902 CrossRef CAS.
- H. Jung, H. Ohashi, G. M. Anilkumar, P. Zhang and T. Yamaguchi, J. Mater. Chem. A, 2013, 1, 13348 RSC.
- A. Mavrič and C. Cui, ACS Appl. Energy Mater., 2021, 4, 12032–12055 CrossRef.
- X.-J. Zhai, Q.-X. Lv, J.-Y. Xie, Y.-X. Zhang, Y.-M. Chai and B. Dong, Chem. Eng. J., 2024, 496, 153187 CrossRef CAS.
- M. N. Uddin, V. V. Nageshkar and R. Asmatulu, Energy, Ecol. Environ., 2020, 5, 108–117 CrossRef.
- J. Y. Chu, K. H. Lee, A. R. Kim and D. J. Yoo, Composites, Part B, 2019, 164, 324–332 CrossRef CAS.
- T. C. T. To, A. Le Mong and D. Kim, J. Mater. Chem. A, 2025, 13, 14280–14292 RSC.
- N. Agmon, Chem. Phys. Lett., 2000, 319, 247–252 CrossRef CAS.
- S. Gahlot and V. Kulshrestha, Int. J. Hydrogen Energy, 2020, 45, 17029–17056 CrossRef CAS.
- B. Yang, Y. Shi, D. J. Kang, Z. Chen and H. Pang, Interdiscip. Mater., 2023, 2, 475–510 Search PubMed.
- C. Yuan, J. Li, P. Han, Y. Lai, Z. Zhang and J. Liu, J. Power Sources, 2013, 240, 653–658 CrossRef CAS.
- C. Jiao, M. Hu, T. Hu and J. Zhang, J. Solid State Chem., 2023, 322, 123978 CrossRef CAS.
- W. Li, B. Guo, K. Zhang, X. Chen, H. Zhang, W. Chen, H. Chen, H. Li and X. Feng, J. Colloid Interface Sci., 2024, 668, 181–189 CrossRef CAS PubMed.
- W. Li, J. Li, Y. Zeng, D. Pan, R. Sa, X. Wang, M. Xiao, Z. Wang and J. Zhu, ACS Sustain. Chem. Eng., 2025, 13, 11474–11484 CrossRef CAS.
- Z. Hong, P. Li, Q. Zou, L. Gu, J. Wang, L. Deng, C. Wang, Y. Zhang, M. Li, J. Chen, R. Si and C. Yang, ACS Appl. Energy Mater., 2024, 7, 11967–11976 CrossRef CAS.
- J. Mei, T. Liao, H. Peng and Z. Sun, Small Methods, 2022, 6, 2101076 CrossRef CAS PubMed.
- A. Li, X. Liao, H. Zhang, L. Shi, P. Wang, Q. Cheng, J. Borovilas, Z. Li, W. Huang, Z. Fu, M. Dontigny, K. Zaghib, K. Myers, X. Chuan, X. Chen and Y. Yang, Adv. Mater., 2020, 32, 1905517 CrossRef CAS PubMed.
- G. He, M. Xu, J. Zhao, S. Jiang, S. Wang, Z. Li, X. He, T. Huang, M. Cao and H. Wu, Adv. Mater., 2017, 29, 1605898 CrossRef PubMed.
- H. Dou, M. Xu, Z. Zhang, D. Luo, A. Yu and Z. Chen, Adv. Mater., 2024, 36 CrossRef PubMed.
- R. Mokete, F. Mikšík, R. Selyanchyn, N. Takata, K. Thu and T. Miyazaki, Energy Adv., 2024, 3, 1019–1036 RSC.
- Z. Guo, T. Wang, H. Wei, Y. Long, C. Yang, D. Wang, J. Lang, K. Huang, N. Hussain, C. Song, B. Guan, B. Ge, Q. Zhang and H. Wu, Angew. Chem., Int. Ed., 2019, 58, 12569–12573 CrossRef CAS PubMed.
- B. Deng, G. Yu, W. Zhao, Z. Zhang, X. Li, K. Huang, L. Wang, H. Peng, H. Gao, X. Li and H. Wu, J. Am. Chem. Soc., 2025 Search PubMed.
- S. Xu, P. Zhang, L. Li, M. W. Moon, C. H. Chung, H. Li, J. Y. Lee and P. J. Yoo, Small, 2025, 21, e2502000 CrossRef PubMed.
- H. Fei, R. Liu, T. Liu, M. Ju, J. Lei, Z. Wang, S. Wang, Y. Zhang, W. Chen, Z. Wu, M. Ni and J. Wang, Adv. Mater., 2024, 36(17), 2309211 CrossRef CAS PubMed.
- W. Tang, Z. Zhao, D. Yang, Y. Liu, L. Zhu, Y. Wu, C. Lan, W. Jiang, Y. Wu and T. Liu, Energy Environ. Sci., 2025, 18(14), 7048–7059 RSC.
- Y. Zheng and S.-Z. Qiao, Joule, 2023, 7, 20–22 CrossRef.
- N. Kim, S. Kim, S. Jeong, H. Jin, H. Lee, Y. Kim, W.-G. Lee and J.-S. Park, Front. Batter. Electrochem., 2023, 2, 1301806–1301826 CrossRef.
- Q. Tang, Y. Liu, R. Meng, Z. Pan, Y. He, C. Zhang, G. Ling, W. Chen and Q.-H. Yang, Adv. Energy Mater., 2025, 15, 2502407 CrossRef CAS.
- S. Singh, H. Chen, S. Shahrokhi, L. P. Wang, C.-H. Lin, L. Hu, X. Guan, A. Tricoli, Z. J. Xu and T. Wu, ACS Energy Lett., 2020, 5, 1487–1497 CrossRef CAS.
- H. Wang, R.-T. Gao, N. T. Nguyen, J. Bai, S. Ren, X. Liu, X. Zhang and L. Wang, ACS Nano, 2023, 17, 22071–22081 CrossRef PubMed.
- I. Slobodkin, E. Davydova, M. Sananis, A. Breytus and A. Rothschild, Nat. Mater., 2024, 23, 398–405 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |