 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Complex molecular dynamics of symmetric model discotic liquid crystals: comparison of hexakis(hepta-alkanoyloxy)triphenylene (HOT6) with hexakis(hexa-alkyloxy)triphenylene (HAT6)
Christina
Krause
a,
Paulina
Szymoniak
a,
Wiebke
Lohstroh
b,
Fanni
Juranyi
c,
Michaela
Zamponi
d,
Bernhard
Frick
 e,
Dominik
Al-Sabbagh
a,
Franziska
Emmerling
e,
Dominik
Al-Sabbagh
a,
Franziska
Emmerling
 af,
Reiner
Zorn
g and
Andreas
Schönhals
af,
Reiner
Zorn
g and
Andreas
Schönhals
 *ah
*ah
aBundesanstalt für Materialforschung und -prüfung (BAM), Department Materials Chemistry, Unter den Eichen 87, 12205 Berlin, Germany. E-mail: Andreas.Schoenhals@bam.de; Fax: +49 30/8104-73384; Tel: +49 30/8104-3384
bHeinz Maier-Leibnitz Zentrum (MLZ), Technische Universität München, Lichtenbergstraße 1, 85748 Garching, Germany
cPSI Center for Neutron and Muon Sciences, 5232 Villigen PSI, Switzerland
dForschungszentrum Jülich GmbH, Jülich Centre for Neutron Science at MLZ, Lichtenbergstr. 1, 85748 Garching, Germany
eInstitut Laue-Langevin, 71 avenue des Martyrs, 38042 Grenoble Cedex 9, France
fInstitut für Chemie, Humboldt Universität zu Berlin, Brook-Taylor-Straße 2, 12489 Berlin, Germany
gForschungszentrum Jülich GmbH, Jülich Centre for Neutron Science (JCNS-1), 52425 Jülich, Germany
hInstitut für Chemie, Technische Universität Berlin, Straße des 17. Juni 135, 10623 Berlin, Germany
First published on 6th February 2026
Abstract
This study investigates the complex molecular dynamics of discotic liquid crystals (DLCs) by comparing two structurally similar compounds: hexakis(hepta-alkanoyloxy)triphenylene (HOT6) and hexakis(hexa-alkyloxy)triphenylene (HAT6) having the same triphenyl core and the same length of the alkyl side chain. The difference of both materials is that the alkyl chain is linked by an oxygen bridge to the triphenylene core for HAT6 and by a ester group for HOT6. Using a combination of broadband dielectric spectroscopy, differential scanning calorimetry, X-ray scattering, and neutron scattering techniques, the research explores the glass transition phenomena and relaxation processes in these materials. HOT6, featuring ester linkages, exhibits distinct dynamic behavior compared to HAT6, including two separate glass transitions indicated by the α1- and α2-relaxation found by dielectric spectroscopy which are assigned to the glassy dynamics of the alkyl side chain in the intercolumnar space and that of the columns, respectively. The study reveals that the ester group in HOT6 leads to increased molecular rigidity and altered packing in the intercolumnar space, as evidenced by X-ray scattering and the vibrational density of states. Neutron scattering confirms localized methyl group rotations and a further relaxation process which relates to the γ-relaxation revealed by dielectric spectroscopy. The findings contribute to a deeper understanding of glassy dynamics in partially ordered systems and highlight the influence of molecular architecture on relaxation behavior in DLCs.
Introduction
The molecular dynamics associated with the glass transition represent a central and unresolved issue in condensed matter physics (see for instance ref. 1–9). Despite extensive research, the fundamental mechanisms underlying the transition of a glass-forming system into a disordered, amorphous state – rather than crystallizing – remain only partially understood. A particularly challenging aspect is the pronounced increase in both the structural relaxation time, τ, and the viscosity as the system is cooled toward the thermal glass transition temperature, Tg.Upon reducing the temperature by approximately a factor of two while approaching Tg, the relaxation time τ increases by more than 14 orders of magnitude. This dramatic change bridges microscopic and macroscopic time scales, highlighting the complexity of the underlying molecular processes. This phenomenon is referred to as α-relaxation (structural relaxation) or the dynamic glass transition.
At temperatures near Tg, the temperature dependence of the relaxation time τ, or equivalently the relaxation rate fp = 2π/τ, can be empirically described by the Vogel–Fulcher–Tammann (VFT) equation,10–12 which reads
 | (1) |
Here, f∞ denotes the relaxation rate in the high-temperature limit, while B is a constant related to the fragility of the system. The concept of fragility in the context of the glass transition provides a valuable framework for categorizing glass-forming systems.13,14 Systems are referred to as fragile when fp(T) exhibits a pronounced deviation from Arrhenius behavior, indicating a super-Arrhenius temperature dependence. In contrast, strong glass formers display a relaxation time that closely follows an Arrhenius-type temperature dependence. The fragility is characterized by the fragility strength parameter D = B/T0. The temperature T0, often termed the Vogel temperature or the ideal glass transition temperature, is derived from empirical fits using the VFT equation to fp(T). It typically lies 30–70 K below Tg as determined by conventional techniques such as differential scanning calorimetry (DSC). It is worth noting that it is known for a longer time that a transition from the VFT to an Arrhenius-like behavior has been observed above a temperature TB ≈ 1.2…1.3Tg (see for instance15,16).
In the cooperativity-based approaches to the glass transition, which have been extensively discussed in the literature (see for instance ref. 17–20), the pronounced increase in relaxation time near the glass transition temperature is attributed to the notion that molecular rearrangements occur in a cooperative manner. Specifically, the movement of a given molecule requires the simultaneous reorganization of neighboring molecules within a complex and rugged energy landscape to reach a local or global energy minimum.
In addition to the primary α-relaxation process, further dynamical processes relevant to the glass transition have been identified. Notably, Goldstein and Johari introduced the concept of a so-called slow β-relaxation, which is considered a precursor to the α-relaxation, particularly in cases where the observed secondary relaxation cannot be attributed to internal molecular degrees of freedom (i.e., a genuine β-relaxation). The temperature dependence of the β-relaxation rates typically follows an Arrhenius behavior, indicating a truly thermally activated process. The Arrhenius equation reads
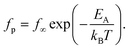 | (2) |
Here EA denotes the activation energy which is found in the range of 20 to 40 kJ mol−1. kB is the Boltzmann constant.
Besides relaxation processes, it is known that glasses show characteristic excess contributions to the low frequency vibrational density g(ω) (VDOS) of states compared with the prediction of the Debye theory which gives g(ω) ∼ ω2 based on the propagation of sound waves. In the reduced representation depicting g(ω)/ω2versus the angular frequency ω a peak is observed in the frequency range of 1.5 to 7.5 ps−1 (1 to 5 meV) which is referred to as the Boson peak.21 Despite the Boson peak manifesting on picosecond timescales, its potential relevance to the glass transition – characterized by relaxation processes occurring on timescales of approximately 100 seconds or longer – remains a topic of active scientific debate.22,23 The molecular origin of the Boson peak remains contentious. Theoretical models are commonly grouped into three categories. Within the soft potential model, the Boson peak is attributed to quasi-localized vibrations of atomic groups in soft potentials arising from structural disorder in amorphous systems which differ principally from sound waves (see for instance ref. 24–26). A second class of models interprets the Boson peak as a broadened and shifted analogue of the Van Hove singularity, assuming an underlying crystalline reference structure, with disorder-induced broadening and frequency shifts.27 Notably, ref. 28 suggests that coupling between optical and acoustic modes may produce a pseudo Van Hove singularity. A third approach, proposed by Schirmacher et al., combines elements of both previous discussed models, attributing the Boson peak to harmonic vibrations in a disordered elastic medium.29 While retaining sound-wave characteristics and incorporating ‘soft spots’ akin to the soft potential model, it does not treat the Boson peak as a modified Van Hove singularity. Recently, a unified theoretical approach considering both Van Hove singularity and the Boson peak has been presented where the propagation and damping of phones is discussed as a mechanism.30,31
As discussed above, the cooperative dynamics underlying the glass transition were initially theorized by Adam and Gibbs, who introduced the concept of cooperatively rearranging regions (CRRs) to describe structural rearrangements within supercooled liquids.17 Wynne et al. proposed that the Boson peak originates from molecular clusters consisting of approximately 20 molecules, which may correspond to cooperatively rearranging regions implicated in the dynamics of the glass transition.32 Further evidence supporting this interpretation has been obtained through molecular dynamics simulations conducted on systems composed of symmetric molecules.33
The most extensively studied systems exhibiting glass transitions include low-molecular-weight liquids, silica-based glasses, and polymers. In addition to these well-known materials classes, glass transition phenomena can also occur in materials characterized by partial structural order and restricted molecular mobility.13,14 Seki et al. were the first to report the occurrence of a glass transition in plastic crystals by calorimetry.34 Additional examples of glass transitions in plastic crystalline systems are discussed in the literature (see for instance35–39). It is noteworthy that discotic liquid crystals also exhibit glass-like dynamic behavior, as discussed in previous studies.40–50 To date, the molecular mechanisms underlying glassy dynamics in systems characterized by restricted mobility and partial order remain unresolved. Studies on such systems, including liquid crystals, may provide valuable insights into the general phenomena associated with the glass transition.
Discotic liquid crystals (DLCs) represent a class of advanced soft matter materials with significant potential for instance for applications in electronic devices (see for instance ref. 51–53 and references cited therein). DLCs are typically composed of molecules having a rigid, disc-shaped aromatic core or a self-assembled disc-like structure with flexible alkyl chains attached to the periphery. At low temperatures, DLCs often exhibit a plastic crystalline phase, which transitions upon heating to a hexagonal columnar liquid crystalline phase. In this phase, the discotic molecules stack into columns that behave as one-dimensional fluids,54 with the columns themselves arranged on a two-dimensional hexagonal lattice. The alkyl side chains fill the intercolumnar space. The unique properties and potential applications of DLCs are primarily associated with this hexagonally ordered liquid crystalline phase. In addition to the hexagonal columnar phase, nematic phases etc. can also be observed in certain DLC systems. Upon further temperature increase beyond the stability range of the columnar phase, DLCs undergo an isotropization transition, resulting in a disordered, isotropic liquid state.
Recently the molecular mobility of a triphenyl-based discotic liquid crystal, hexakis(hexa-alkyloxy)triphenylene (HAT6) was investigated by a combination of broadband dielectric spectroscopy, advanced dynamical calorimetry and quasielastic neutron scattering.44 Here a comparable triphenyl-based DCL with an equal length of the alkyl side chains, hexexakis(hepta-alkanoyloxy)triphenylene (HOT6) was studied. Interest in the hexexakis(n-alkanoyloxy)triphenylenes comes from the additional fact that they are photoconductive and doped with few mol of an oxidant they become semiconductive.55 The obtained results for HOT6 will be compared in detail with that of HAT6.
Materials und methods
Materials
Hexakis(hepta-alkanoyloxy)triphenylene (HOT6) was obtained from Synthon Chemicals (Bitterfeld, Germany). Its chemical structure is given in Fig. 1a in comparison with that of Hexakis(hexa-alkyloxy)triphenylene (HAT6) (see Fig. 1b). Both materials possess alkyl side chains of identical length, defined by the number of carbon atoms in the chain. The difference in the chemical structure of both materials is that the alkyl side chains are linked via an ester group to the triphenylene core for HOT6 while for HAT6 the alkyl side is connected to the core by an oxygen bridge. The ester group in HOT6 will dynamically decouple the alkyl side chain more from the triphenylene core than the oxygen bridge in HAT6. Moreover, as the ester linkage in HOT6 is bulkier than the oxygen group in HAT6, the HOT6 molecule is expected to be larger than the HAT6 one. It is worth noting that due to the presence of an ester group, HOT6 exhibits increased susceptibility to hydrolytic degradation under humid conditions compared with HAT6. HOT6 was used without further purification. The purity given by the producer is 94%. Fig. 1c presents a schematic of the self-assembly of the disc-like molecules to the columnar hexagonal liquid crystalline phase. | ||
| Fig. 1 Chemical structure of HOT6 (a) and HAT6 (b). (c) Schematics of the self-assembly of the discotic liquid crystal. | ||
Methods
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 K s−1 can be achieved. Nitrogen was employed as the purge gas at a constant flow rate of 40 mL min−1. The base temperature control was ensured using a Huber TC100 intercooler. Calibration of the Flash DSC 1 was performed in accordance with the specification of the manufacturer. This means that each calorimetric chip was calibrated by the producer. During the measurement the calibration data were automatically downloaded from the database of the producer.56 The calibration was checked by measurements on standards leading to a maximal error in temperature of ±5 K in the whole considered temperature range. Thermal lag effects are minimized by considering low samples masses in the nanogram range and a thin film sample geometry. The typical error due to thermal lag under these conditions is smaller than the overall uncertainty in temperature. During the FSC experiment the heating rate was varied. To ensure the same history of the sample before each heating run the sample was cooled from the isotropic state to 193 K with a cooling rate of 1000 K s−1.
000 K s−1 can be achieved. Nitrogen was employed as the purge gas at a constant flow rate of 40 mL min−1. The base temperature control was ensured using a Huber TC100 intercooler. Calibration of the Flash DSC 1 was performed in accordance with the specification of the manufacturer. This means that each calorimetric chip was calibrated by the producer. During the measurement the calibration data were automatically downloaded from the database of the producer.56 The calibration was checked by measurements on standards leading to a maximal error in temperature of ±5 K in the whole considered temperature range. Thermal lag effects are minimized by considering low samples masses in the nanogram range and a thin film sample geometry. The typical error due to thermal lag under these conditions is smaller than the overall uncertainty in temperature. During the FSC experiment the heating rate was varied. To ensure the same history of the sample before each heating run the sample was cooled from the isotropic state to 193 K with a cooling rate of 1000 K s−1.
Scattered X-ray intensities were recorded 820 mm downstream from the sample position using a two-dimensional X-ray detector (MarMosaic, CCD, 3072 × 3072 pixels), featuring a point spread function with an approximate width of 100 µm. A comprehensive description of the beamline setup can be found in ref. 57. The acquired scattering patterns were processed and transformed into intensity profiles as a function of the scattering vector q, where q = (4π/λx)sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ with θ being half the scattering angle 2θ, and λx the wavelength of the X-rays. Data reduction and analysis were performed using the software package FIT2D.58
θ with θ being half the scattering angle 2θ, and λx the wavelength of the X-rays. Data reduction and analysis were performed using the software package FIT2D.58
For the X-ray scattering experiments, the samples were loaded into borosilicate glass capillaries (WJM-Glas Glastechnik & Konstruktion, Germany) with an outer diameter of 0.3 mm. To ensure consistency with the DSC measurements, the samples were heated at a controlled rate of 10 K min−1 during the experiments.
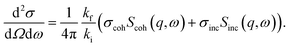 | (3) |
For the neutron scattering investigations different methods are combined:
An effective mean squared displacement, 〈u2〉eff, is derived from the measured elastic scattering intensity, Iel(q), by fitting the function65
 | (4) |
Neutron backscattering spectra were taken at the high-resolution spectrometer SPHERES. SPHERES is a third-generation cold neutron backscattering spectrometer equipped with focusing optics and a rotating phase-space transform chopper. It is operated at the Heinz Maier-Leibnitz Zentrum by the Jülich Centre Neutron Science (JCNS), Forschungszentrum Jülich GmbH.68,69 Measurements were conducted in the standard configuration, utilizing unpolished Si(111) monochromators and analyzers. The incident neutron wavelength was 6.271 Å, corresponding to an energy resolution of 0.65 µeV (full width at half maximum, FWHM) for the high-angle detectors. In this setup, the accessible momentum transfer range spans from 0.1 Å−1 to 1.8 Å−1, but only data at and above 0.6 Å−1 were used for analysis because of the worse resolution and statistics for the small-angle detectors. The Doppler drive was set to achieve a maximal energy transfer of ±31 µeV. Like for TOFTOF the resolution of SPHERES R(q,ΔE) was obtained by measuring the sample at 4 K. Data obtained from the SPHERES spectrometer were processed using the SQW software,70 which performs backscattering normalization, vanadium standard normalization, and corrections for self-attenuation. Additionally, the software applies an attenuation correction to the background prior to its subtraction. Fig. S3 presents the incoherent dynamic structure factor of HOT6 as measured with the SPHERES backscattering spectrometer at various temperatures. A temperature-dependent quasielastic broadening of the spectra relative to the instrumental resolution is observed, indicating an enhanced molecular mobility.
The analysis of the TOF and BS data by INX and SQW yield an effective dynamic structure factor, Sinc(q,ΔE), weighted by the respective scattering cross sections. Due to the significant difference in energy resolution between the TOFTOF and SPHERES spectrometers (see Fig. S2 and S3), the incoherent dynamic structure factors, Sinc(q,ΔE) were Fourier transformed and subsequently normalized by the Fourier transformation of the respective instrumental resolution functions. This procedure yields absolute values of the incoherent intermediate scattering function, Sinc(q,t), enabling a consistent time-domain analysis of data acquired from both instruments. The Sinc(q,t) datasets obtained from TOF and BS neutron spectroscopy were corrected for multiple scattering using a time-domain-based approach.71 Due to the inability to precisely determine the scattering geometry, the multiple scattering contribution was treated as a fitting parameter and adjusted to satisfy the physical constraint Sinc(q → 0,t) = 1. The optimized multiple scattering fractions were determined to be 31% (HAT6)/33% (HOT6) for SPHERES and 21% (HAT6)/22% (HOT6) for TOFTOF, relative to the single-scattering intensity.
 | ||
| Fig. 2 (a) Heat flow versus temperature for HOT6. Blue – cooling, red – heating. Second cooling and heating. Rate 10 K min−1. (b) Comparison of the heat flow for HOT6 (red) and HAT6 (blue). Data for HAT6 were taken from ref. 44. Second cooling, cooling rate 10 K min−1. The heat flow for HOT6 was shifted along the y-scale for clarity. (c) Heat flow versus temperature for HOT6 at lower temperatures during cooling. (d) Derivative of the heat flow given in part c with respect to temperature. The arrows indicate the glass transition temperatures. | ||
Results and discussion
Differential scanning calorimetry and X-ray scattering
Fig. 2a gives the heat flow versus temperature for HOT6 for cooling and heating. At low temperature HOT6 has a plastic crystalline phase. At ca. 288 K (heating) HOT6 shows a crystal/crystal transition. This is different for HAT6 where such a crystal/crystal transition is not observed (see Fig. 2b). Most probably the observed crystal/crystal transition of HOT6 is due to the presence of the ester group which decouples the alkyl side chain dynamically more from the triphenyl core than the oxygen linkage in HAT6. At ca. 382 K HOT6 undergoes the transition from the plastic crystalline to the liquid crystalline Colh state followed by the phase transition to the “isotropic” state at ca. 391 K. The assignment of the phases corresponds to that given in the literature.72 The phase transition temperatures of HOT6 in cooling and heating are given in comparison with data from the literature in Table 1. As known for other DCL systems there is a hysteresis between heating and cooling. Moreover, the values for the phase transitions temperatures measured here agree with the data given by the producer73 and approximately with results presented in the literature.72 This agreement supports the assignment of the phases. Like for HAT6 it is assumed for HOT6 that in the crystalline phases the tripenylene cores are arranged in columns where the alkyl side chains fill intercolumnar space.| T Cry1,Cry2 [K] | T Cry2,Colh [K] | T Colh,Iso [K] | |
|---|---|---|---|
| Cooling | 277 | 367 | 383 |
| Heating | 288 | 382 | 390 |
| Producer | — | 379 | 381 |
| Literature | — | 342 | 366 |
Fig. 2b compares the heat flow for the second cooling for HOT6 and HAT6. This comparison shows that the temperature range of the Colh mesophase for HOT6 is much smaller than that of HAT6. Probably, the more flexible ester group of HOT6 compared with the oxygen bridge in HAT6 is the reason for the smaller mesophase range. Besides, the narrower temperature range of the mesophase Fig. 2b shows further that the phase transition enthalpies of all phase transition of HOT6 are much smaller than that of HAT6. Again, the lower phase transition enthalpies are due to the presence of the ester groups which might lead to less stable phases at higher temperatures.
Besides the peaks in the heat flow of HOT6 associated with the phase transition, a step-like change in the heat flow is observed in the temperature range between 150 K and 225 K (see Fig. 2a). This step-like change may indicate a glass transition of HOT6. A more detailed inspection of the low-temperature heat flow reveals a double-step structure (see Fig. 2c), which may suggest the presence of two distinct glass transitions. This becomes clearer considering the derivative of the heat flow with respect to temperature where a step-like change in the heat flow results in a peak. Although the derivative data shows some scattering, Fig. 2d reveals two peaks which are well separated in temperature. From the maximum positions of the peaks the thermal glass transition temperatures are estimated. The glass transition observed at higher temperatures is denoted as glass transition 1 and that at lower temperatures as glass transition 2. It is worth noting that for HAT6 also two glass transitions are detected by calorimetric methods.48 It is worth noting that the numeration of both glass transitions chosen here for HOT6 is analogues to that given for HAT648 for compatibility. The phenomenon will be discussed in more detail below.
Fig. 3 depicts the X-ray pattern for HOT6 in the different phases. In the Cry2 phase an X-ray pattern of a crystalline material with several sharp reflection peaks is observed (see Fig. 3a). Besides the sharp Bragg peaks in the q value range from ca. 10 nm−1 to 20 nm−1 an amorphous halo is observed which corresponds to average carbon–carbon correlations of the alkyl side chains.74 In the columnar phase (see Fig. 3b), besides the amorphous halo the X-ray pattern, one relatively sharp reflection peak is visible. It corresponds to the average column–column distance d given by75
 | (5) |
From eqn (5)d = 2.1 nm is calculated for HOT6. Due to the presence of the bulkier ester group in HOT6 the intercolumnar distance is larger than for HAT6 (d = 1.8 nm) which features the smaller oxygen bridge.83 Theoretically for a columnar phase higher order reflections at q values  should be observed. These higher order reflections are not observed for HOT6. That might point to a less perfect columnar order in HOT6 Cry2 phase compared with that of HAT6 as it is already concluded from the phase transition enthalpies. However, an additional reflection is observed at a smaller q value than
should be observed. These higher order reflections are not observed for HOT6. That might point to a less perfect columnar order in HOT6 Cry2 phase compared with that of HAT6 as it is already concluded from the phase transition enthalpies. However, an additional reflection is observed at a smaller q value than  at ca. 4.6 nm.
at ca. 4.6 nm.
A more detailed analysis of the X-ray diffraction pattern in the vicinity of the principal reflection peak for both the crystalline Cry2 and columnar phases reveals a distinct peak splitting into two subpeaks (see insets of Fig. 3a and b). This feature is absent in the corresponding patterns of HAT6. Given that the subpeaks occur at different q values for the crystalline and columnar phases, the possibility of an experimental artifact can be reasonably excluded. The splitting of the peaks is discussed considering that the ester group can adopt different conformations. The specific conformations depend on several factors like the rotation around the single bond between the carbonyl carbon and the oxygen group/atom of the alkyl moiety, steric interactions with neighboring groups or molecules, intramolecular interactions, such as hydrogen bonding or π-interactions or crystal packing or supramolecular organization, which is particularly relevant in columnar phases. For simple esters, like present in HOT6, conformations are typically classified as syn or anti, depending on the relative orientation of the carbonyl group and the alkyl chain. These conformational differences can influence the effective length of the side chain, thereby affecting molecular packing and lattice parameters. These effects can lead to the observed peak splitting. However, to be visible as separate peak in the X-ray pattern there must be some clusters of a preferred conformation. The change of packing and lattice parameter are probably also the reason that higher order reflexes  are not observed. It might be further concluded that the different packing and lattice parameters results also in a different structure of the alkyl side chain in the intercolumnar space of HOT6 compared with HAT6.
are not observed. It might be further concluded that the different packing and lattice parameters results also in a different structure of the alkyl side chain in the intercolumnar space of HOT6 compared with HAT6.
In the isotropic state one broad Bragg peak is found (see Fig. 3c) which indicated a correlation of the triphenyl molecules also in this phase. The width of this reflection is much broader than that in the Colh. This increased broadness of the peak which evidence a higher disorder in the “isotropic” state compared to Colh phase. Also, the amorphous halo is found in the isotropic phase as expected.
Fig. 3d compares the X-ray pattern of HOT6 and HAT6 at T = 303 K. At this temperature HOT6 is the in the plastic crystal Cry2 state. Compared with HAT6 the X-ray scattering pattern that of HOT6 is less detailed. This points to a less ordered plastic crystalline structure in the Cry2 state compared with the plastic crystal state of HAT6. Unfortunately, no X-ray measurements could be carried out at temperatures where HOT6 is in the Cry1 phase which could be more comparable or even more ordered than the plastic crystal phase of HAT6.
 | ||
| Fig. 4 Dielectric loss versus frequency and temperature in a 3D representation: (a) HOT6; (b) HAT6. The figure for HAT6 was adopted from ref. 44. | ||
The γ-relaxation of HOT6 is analyzed by standard methods fitting the model function of Havriliak/Negami (see SI, Section fitting of dielectric data by the HN-function) to the dielectric loss. For details see ref. 76. From the fits, the frequency of maximal loss fp is obtained which is called relaxation rate. Fig. 5 depicts the temperature dependence of the of the logarithm of the relaxation rates of the γ-relaxation versus inverse temperature in the relaxation map or Arrhenius diagram. Fig. 5a gives the data for HOT6 and Fig. 5b for HAT6. fp(T) for the γ-relaxation is linear when plotted versus 1/T and can be therefore described by the Arrhenius equation. For HOT6 a value of the activation energy of 29.2 kJ mol−1 is obtained while for HAT6 a value of 20.6 kJ mol−1 is reported in ref. 44. The γ-relaxation of HAT6 and of other discotic liquid crystals is assigned to localized fluctuations of methylene units of the alkyl chains filling the intercolumnar space involving also polar groups.42–50 Therefore, also the γ-relaxation of HOT6 is assigned to localized fluctuations of CH2 units of the alkyl chains in the intercolumnar space. In ref. 48 the activation energy of the γ-relaxation for the series homologous of HATn (n = 5, 6, 8, 10, 12) with that of polyethylene. The value of the activation energy of the γ-relaxation estimated for HOT6 is closer to the activation energy of ca. 40 kJ mol−1 of the γ-relaxation of polyethylene (PE)77 one must conclude that the molecular fluctuations responsible for the γ-relaxation of HOT6 are more restricted than that of HAT6. At the first glance that seems to disagree with the conclusion drawn from the X-ray measurements that the Cry2 phase is less ordered that the plastic crystalline state of HAT6. However, the γ-relaxation of HOT6 takes place at lower temperature in the Cry1 phase of HOT6. Although no X-ray data for the Cry1 phase are available it is concluded that the Cry1 state of HOT6 is more ordered than that of HAT6.
 | ||
| Fig. 5 (a) Relaxation map for HOT6 in Arrhenius coordinates: asterisks – methyl group rotation. The dashed dotted–dotted line is a common fit of the Arrhenius equation to the data of HOT6 and HAT6. Squares – dielectric γ-relaxation. The solid lines are fits of the Arrhenius equation to the corresponding data. Pentagons – second process measured by neutron scattering. The dashed dotted line is a fit of the Arrhenius equation to the relaxation rates of the second process measured by neutron scattering. The red dashed-dotted line is a fit of the Arrhenius equation to the data of HOT6. Circles – dielectric α2-relaxation. Solid triangles – dielectric α1-relaxation diamonds – heating rate (HR) versus inverse glass transition temperature estimated from the FSC measurements. Open triangles – HR versus inverse Tg taken from the DSC measurements. The dotted line is a linear fit to heating rates of FSC and Tg,1 measured by DSC. Phase transitions temperatures are indicated by black dotted lines. (b) Relaxation map for HAT6 in Arrhenius coordinates. All data were taken from ref. 48: asterisks – methyl group rotation. The dashed dotted–dotted line is a common fit of the Arrhenius equation to the data of HOT6 and HAT6. Squares – dielectric γ-relaxation. The solid lines are fits of the Arrhenius equation to the corresponding data. Pentagons – second process measured by neutron scattering. The solid blue line is a fit of the Arhenius equation to the data of the γ-relaxation. Triangles – thermal data for the α1-relaxation. The dashed line is a fit of the VFT equation to the thermal data. Phase transitions temperatures are indicated by black dotted lines. | ||
At low temperature the α2-relaxation of HOT6 can be also analyzed by fitting the HN model function directly to the data. However, at higher temperatures the α2-relaxation overlaps with the γ-relaxation which prevents a reliable analysis of the α2-relaxation (see Fig. 4a). Therefore, that process is further analyzed by a so-called derivative approach.78 It has been demonstrated that the Debye function exhibits the following behavior:
 | (6) |
Owing to the squared dependence in the derivative of the dielectric function,  , the width of the conduction-free loss peak is narrower than that of the primary dielectric loss function ε″ leading to a separation of overlapping relaxation processes. Details and examples for this analysis are given in the SI (Section Analysis of dielectric spectra by the derivative approach). Fig. 5 compares the temperature dependence of the relaxation rates of the α2-relaxation of HOT6 (Fig. 5a) with that of HAT6 (Fig. 5b). The α2-relaxation of HOT6 is observed in a comparable temperature/frequency window than that of HAT6. In ref. 44 evidence was presented that the α2-relaxation is related to a glassy mobility of the columns, which can be considered as glassy dynamics of a one-dimensional liquid. As the core of HOT6 is analogous to that of HAT6 and because the α2-relaxation for HOT6 is observed in a comparable temperature/frequency, the α2-relaxation of HOT6 is also assigned to glassy mobility of the columns. Like for HAT6 the temperature dependence of the relaxation rates of the α2-relaxation is quite complex and can be described neither by the Arrhenius nor by the VFT equation. Firstly, fp(T) increases relatively strongly with increasing temperature up to ca. 210 K following approximately the Arrhenius law. Secondly, at higher temperatures fp(T) increases further Arrhenius-like but with a lower apparent activation energy (see Fig. 5). For HAT6 the change in fp(T) correlates with the thermal glass transition temperature Tg,1 (see Fig. 5b). Although Tg,1 of HOT6 is found close to the change of fp(T), no exact correlation is found for HOT6. Nevertheless, it is argued that the change in the temperature dependence of the α2-relaxation is related to the glass transition 2 which is assigned to the glass transition of the alkyl side chains in the intercolumnar space.
, the width of the conduction-free loss peak is narrower than that of the primary dielectric loss function ε″ leading to a separation of overlapping relaxation processes. Details and examples for this analysis are given in the SI (Section Analysis of dielectric spectra by the derivative approach). Fig. 5 compares the temperature dependence of the relaxation rates of the α2-relaxation of HOT6 (Fig. 5a) with that of HAT6 (Fig. 5b). The α2-relaxation of HOT6 is observed in a comparable temperature/frequency window than that of HAT6. In ref. 44 evidence was presented that the α2-relaxation is related to a glassy mobility of the columns, which can be considered as glassy dynamics of a one-dimensional liquid. As the core of HOT6 is analogous to that of HAT6 and because the α2-relaxation for HOT6 is observed in a comparable temperature/frequency, the α2-relaxation of HOT6 is also assigned to glassy mobility of the columns. Like for HAT6 the temperature dependence of the relaxation rates of the α2-relaxation is quite complex and can be described neither by the Arrhenius nor by the VFT equation. Firstly, fp(T) increases relatively strongly with increasing temperature up to ca. 210 K following approximately the Arrhenius law. Secondly, at higher temperatures fp(T) increases further Arrhenius-like but with a lower apparent activation energy (see Fig. 5). For HAT6 the change in fp(T) correlates with the thermal glass transition temperature Tg,1 (see Fig. 5b). Although Tg,1 of HOT6 is found close to the change of fp(T), no exact correlation is found for HOT6. Nevertheless, it is argued that the change in the temperature dependence of the α2-relaxation is related to the glass transition 2 which is assigned to the glass transition of the alkyl side chains in the intercolumnar space.
From the DSC measurements a thermal relaxation rate can be estimated by79
 | (7) |
The heating or cooling rate is denoted as Ṫ and ΔTg is the width of the glass transition. ΔTg is estimated as temperature difference between onset and endset of the glass transition step a is a constant of the order of one. The thermal relaxation rate estimated for the glass transition 2 is added to the relaxation map (Fig. 5a). It correlates approximately with the lower temperature branch of the α2-relaxation which supports the assignment of the α2-relaxation to the glass-like mobility within the columns.
In addition to the well-established α2-relaxation, the dielectric spectra of HOT6 reveal an additional an α1-process, which is not detectable in the dielectric response of HAT6 (see Fig. 4) but has been identified through advanced calorimetric techniques. The α1-relaxation in HOT6 was further analyzed using the derivative approach described previously (see SI). The corresponding relaxation rates were incorporated into the relaxation map (Fig. 5a). The dielectric α1-process occurs in a comparable temperature/frequency window as the α1-relaxation of HAT6 measured by a combination of calorimetric methods. In ref. 44 the α1-relaxation was attributed to the glassy dynamics of the alkyl chains in the intercolumnar space. A similar interpretation is made here for the molecular origin of the α1-relaxation of HOT6, supported by the correlation between dielectric and thermal relaxation rates derived from the glass transition 1 measured via DSC.
To investigate the glassy dynamics of HOT6 further fast scanning calorimetric measurements employing the Flash DSC1 have been carried out. An example for these measurements is given in the SI (Section Fast scanning calorimetry). From these measurements a thermal glass transition temperature is estimated by a derivative technique as described for the DSC measurements. As the data show considerable scatter, no thermal relaxation rates for FSC measurements according to eqn (7) can be extracted as the width ΔTg cannot be estimated unambiguously. Therefore, the heating rates (HR) are plotted versus the inverse of the estimated glass transition temperature in Fig. 5a (right y-axis). The absolute values and the temperature dependence of the FSC data agrees with that of the dielectric α1-relaxation of HOT6. Moreover, the heating rate and corresponding Tg estimated for the glass transition 1 measured by DSC were added to the heating rate plot. Both data sets agree with each other and can be described by a common fit of the Arrhenius equation. From these agreements it is concluded that the dielectric α1-relaxation of HOT6 is really a glass transition.
While the temperature dependence of the relaxation rates of the α1-process in HAT6 follows the VFT behavior, that of the α1-relaxation of HOT6 seems to follow the Arrhenius equation. Within the framework of the fragility concept to the glass transition, this suggests that the α1-relaxation in HOT6 exhibits stronger (i.e., less fragile) glassy dynamics compared to that of HAT6. Consistent with conclusions drawn regarding the γ-relaxation, the Cry1 phase of HOT6 is structurally more ordered than the plastic crystalline phase of HAT6. This increased order likely imposes stronger constraints on the mobility of the alkyl side chains, resulting in more localized fluctuations of the alkyl chains relative to those of HAT6.
It is worth mentioning that glass transition 2 could not be investigated by FSC due to the limitations of Flash DSC1.
Neutron scattering
 | (8a) |
The exponential term e−2W(q) is the Debye–Waller factor and δ represents the delta function. ![[m with combining macron]](https://www.rsc.org/images/entities/i_char_006d_0304.gif) gives the average atomic mass. Owing to the high incoherent scattering cross-section and the low atomic mass of hydrogen, its dynamical contributions are disproportionately amplified in the neutron scattering signal. Nevertheless, if the vibrational modes associated with hydrogen atoms are uniformly distributed across the relevant frequency range, the resulting spectral signal may be considered a valid representation of the VDOS from a thermodynamic perspective.
gives the average atomic mass. Owing to the high incoherent scattering cross-section and the low atomic mass of hydrogen, its dynamical contributions are disproportionately amplified in the neutron scattering signal. Nevertheless, if the vibrational modes associated with hydrogen atoms are uniformly distributed across the relevant frequency range, the resulting spectral signal may be considered a valid representation of the VDOS from a thermodynamic perspective.
The experimentally observed scattering intensity, Sobs(q,ω), corresponds to the convolution of the intrinsic scattering response of the sample with the instrumental resolution function of the spectrometer. Given that the Boson peak typically appears as a broad spectral feature, this convolution can be effectively approximated by a discrete summation. Under this approximation, eqn (8a) simplifies to:81
 | (8b) |
R(q,ω) represents the resolution of the spectrometer. Eqn (8b) describes a linear relationship between g(ω) and R(q,ω). By acquiring scattering data at two distinct temperatures (in this case, 4 K and 80 K), it is possible to estimate both g(ω) and R(ω) from eqn (8b). The detailed procedure for the calculation of the VDOS is given in the SI. This calculation also includes a correction for multiple scattering events. At this point it must be mentioned that in earlier publication78 this correction has not been made, leading to apparently higher values of g(ω). But due to the linear nature of the correction, this does not affect the shape and peak position of the result.
Fig. 6 compares g(ω)/ω2versus frequency for HOT6 and HAT6. As known from ref. 82g(ω)/ω2versus ω shows a broad Boson peak for HAT6 with a maximum frequency ωBP of ca. 2.77 ps−1. This is a bit different for HOT6. Besides a main peak at a frequency of approximately 3.1 ps−1, g(ω)/ω2versus frequency shows a broad shoulder in the frequency range of 4 ps−1 to 8 ps−1 which is not present for HAT6.
Firstly, the shift in the main maximum frequency position of HOT6 in comparison to that of HAT6 will be discussed. Within the framework of elastic disorder models addressing the Boson peak, the characteristic frequency of the BP is commonly associated with the elastic moduli of the material.83 In materials with a reduced stiffness (softer materials) on a local scale, the Boson peak is observed to shift toward lower frequencies compared with stiffer (harder) counterparts. To validate this correlation, experimental studies have been conducted in which the mechanical stiffness of the material was systematically modulated – either through continuous structural modifications82 or by incorporating an additional component into the host matrix.84 Recently, this approach has been extended to investigate the correlation between the frequency position of the Boson peak and the Brunauer–Emmett–Teller (BET) surface area in intrinsic microporous polymers.85–87 The higher value of ωBP for HOT6 in comparison with that of HAT6 would imply that HOT6 is stiffer on a molecular scale in comparison to HAT6. This conclusion agrees with the results obtained by dielectric spectroscopy that the alkyl chains in the intercolumnar space are more constrained for HOT6 than that of HAT6 leading to a higher local elastic modulus.
Secondly, the shoulder in g(ω)/ω2versus ω observed in the frequency range of ca. 4 ps−1 to 8 ps−1 for HOT6 will be addressed (see Fig. 6). From the dielectric experiments, it was concluded that the structure of the alkyl side chains in the intercolumnar space is changed for HOT6 in comparison to that of HAT6. Most probably the changed packing and structure of the alkyl chains for HOT6 leads to additional vibrations giving rise to the shoulder in the VDOS of HOT6.
Fig. 7b compares the temperature dependence of 〈u2〉eff of HOT6 and HAT6 in the lower temperature range. In the temperature where the vibrations are revealed in the elastic scans the effective mean squared displacement for both materials are approximately similar as the vibrations are localized. As the methyl group rotation is also a localized relaxation process, the mean squared displacement is also approximately similar for this relaxation process. For temperatures above than characteristic for the methyl group rotation, 〈u2〉eff(T) is essential higher for HOT6 compared with that of HAT6. This behavior indicates that the molecular mobility in HOT6 is higher than for HAT6. Moreover, for HOT6 the temperature dependence of 〈u2〉eff seems to be more complex than that of HAT6.
 | (9) |
 | ||
| Fig. 8 (a) Incoherent intermediate scattering function SInc(q,t) of HOT6 versus time at T = 150 K at different values of the q vector: black squares q = 1.78 Å−1; blue circles q = 1.42 Å−1, red asterisk –q = 1.04 Å−1 and green triangle –q = 0.69 Å−1. Lines are fits of eqn (9) to the data. (b) EISF versus q for HOT6 (solid red circles) and HAT6 (open blue squares). The dashed line is the prediction of eqn (10a). The solid line is calculated by eqn (10b) with Cfix = 0.785. | ||
The q dependence of the EISFM can be most effectively described by a model based on jump rotation within a threefold rotational potential. This potential is given by V(ϕ) ∼ (1 − cos(3ϕ))/2 which reflects the presence of three energetically equivalent minima corresponding to the rotational symmetry of the methyl group around its axis. This model captures the essential physics of the rotational dynamics and leads to an analytical expression for the EISFM90,91
 | (10a) |
The spatial arrangement of the hydrogen nuclei in a methyl group defines a circular trajectory with a radius of r = 1.027 Å. The theoretical prediction of eqn (10a) is given in Fig. 8b. During the deduction of eqn (10a) it was assumed that all hydrogen nuclei in a system undergo the methyl group rotation. Fig. 1a shows that is not true as HOT6 contains 84 hydrogen nuclei into total of which only 18 are in the methyl groups. Therefore eqn (10a) must be corrected by
| EISFM,corr(q) = (1 − Cfix)EISFM(q) + Cfix | (10b) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) group)/nH,total = 0.785. Fig. 8b reveals that this approach describes the data well. Fig. 8b shows further that the q dependence of the EISF for HOT6 is identical to that of HAT6 within error bars. This should be expected as the number of hydrogen nuclei in the methyl groups is identical for both materials, and the methyl group rotation is a localized process and thus independent of the linkage of the alkyl side chain to the triphenylene core.
group)/nH,total = 0.785. Fig. 8b reveals that this approach describes the data well. Fig. 8b shows further that the q dependence of the EISF for HOT6 is identical to that of HAT6 within error bars. This should be expected as the number of hydrogen nuclei in the methyl groups is identical for both materials, and the methyl group rotation is a localized process and thus independent of the linkage of the alkyl side chain to the triphenylene core.
From the relaxation time τM obtained by the fit of eqn (9) to the data a mean relaxation time can be calculated by
 | (11) |
In eqn (11)Γ(x) denotes the Gamma function. The relaxation times for the methyl group rotation are independent of the q value which is expected for a localized relaxation process. Therefore, an arithmetically average relaxation time is calculated for each temperature. The relaxation rates for the methyl group of HOT6 are included in Fig. 5a and compared with those of HAT6. Both sets of data for the methyl group rotation can be described by a common Arrhenius equation with an average activation energy of approximately 11.3 kJ mol−1. This is expected as the methyl group rotation is a localized relaxation process and should be independent from the linker of the alkyl side chain to the triphenyl core.
Fig. 9a compares the time dependence of Sinc(q,t) for HOT6 and HAT6 for a q value of 1.79 Å−1 at T = 250 K. As already concluded from the elastic scans, for HOT6 the intermediate scattering function decays much stronger for HOT6 than for HAT6. As most of the scattering is due to the hydrogen nuclei located in the intercolumnar space, that indicates that the structure of the intercolumnar state is different for HOT6 compared to HAT6. Moreover, for temperatures higher than 150 K, Sinc(q,t) exhibits a two-step decay. This indicates two underlying relaxation processes where the process at shorter times is methyl group rotation. To analyze the intermediate (time dependent) scattering function for these temperatures, two stretched exponentials are fitted to Sinc(q,t) for temperatures higher than T = 150 K. The whole fit function reads
 | (12) |
 | ||
| Fig. 9 (a) Comparison of the incoherent intermediated scattering function SInc(q,t) of HOT6 (red circles) and HAT6 (blue squares) for a q vector of 1.78 Å−1 at T = 250 K. Lines are guides to the eyes. (b) SInc(q,t) of HOT6 versus time at T = 250 K for different values of the q vector: black squares q = 1.78 Å−1; blue circles q = 1.42 Å−1, red asterisk –q = 1.04 Å−1 and green triangle –q = 0.69 Å−1. Lines are fits of eqn (9) to the data. | ||
EISFγ, τγ and βγ are the corresponding elastic incoherent structure factor, the relaxation time and the shape parameter for the relaxation process taking place observed at longer times. Examples for the fit of eqn (12) to the data are given in Fig. 9b. To reduce the number of free fit parameters it was assumed that the elastic incoherent structure factor is independent of temperature. This assumption is supported by the result that the q dependence follows eqn (10b) with a value of Cfix estimated from the chemical structure of HOT6. Therefore, the values of the EISFM obtained at T = 150 K were fixed for the fits of eqn (12) to the data measured at higher temperatures. Moreover, for βγ the value obtained from the dielectric experiment was taken and fixed during the fits. The average relaxation for the process at longer times is calculated by eqn (11). As for the methyl group rotation, the relaxation times for the process observed at longer times are independent of the q vector within error bars which points to a localized relaxation process. The relaxation times were arithmetically averaged with respect to q. The corresponding relaxation rates are included in Fig. 5a.
For HAT6 the relaxation rates of the low frequency process observed with neutron scattering correspond in both its absolute values and its activation energy with that of the γ-relaxation found by dielectric spectroscopy (see Fig. 5b). This is a bit different for HOT6. Here the values of the relaxation rates observed by neutron scattering are by ca. 1 decade higher than that estimated by dielectric spectroscopy. However, the deduced activation energy of 29 kJ mol−1 for the relaxation process observed by neutron scattering is similar to that found for the γ-relaxation by dielectric spectroscopy (29.2 kJ mol−1) (see Fig. 5a). Therefore, it is concluded that the relaxation process observed by neutron scattering is at least related to the dielectric γ-relaxation. It might be further argued that dielectric and neutron scattering sense different aspects of the same process.
Conclusion
This study presents a comprehensive comparison of the molecular dynamics of two structurally related discotic liquid crystals, HOT6 and HAT6, using a multi-technique approach including dielectric spectroscopy, calorimetry, X-ray scattering, and neutron scattering. The substitution of an oxygen bridge in HAT6 with an ester linkage in HOT6 significantly alters the dynamic behavior and structural organization of the material in the intercolumnar space. Also, the dielectric loss of HOT6 is enhanced compared to that of HAT6 which is due to the presence of the polar ester group in HOT6. HOT6 exhibits two distinct glass transitions, indicated by dielectric α1- and α2-relaxations. The α1-relaxation is assigned to the glassy dynamics of the alkyl side chains in the intercolumnar space while the α2-relaxation is attributed to the glassy dynamics of the columns. This process can be considered as a glass transition of a one-dimensional liquid. Besides the α-relaxations a γ-relaxation is observed by dielectric spectroscopy. Compared to HAT6 the activation energy of the γ-relaxation is higher for HOT6 which points to a more constrained molecular mobility in the intercolumnar space. This conclusion is supported by a modified low frequency vibrational density of states measured by inelastic neutron scattering.X-ray diffraction reveals a peak splitting and increased intercolumnar distances in HOT6, indicating altered packing due to conformational variability of the ester groups. Neutron scattering confirms the localized methyl group rotations for HOT6 which are identical to that of HAT6. An additional relaxation process is found by quasielastic neutron scattering. This process shows a close resemblance to the γ-relaxation revealed by dielectric spectroscopy.
Overall, the findings underscore the sensitivity of glassy dynamics to subtle changes in molecular architecture and highlight the importance of combining complementary experimental techniques to unravel the intricate behavior of partially ordered soft matter systems. These insights contribute to the broader understanding of glass transition phenomena in discotic liquid crystals and may inform the design of advanced functional materials for electronic applications.
Author contributions
CK: dielectric spectroscopy and analysis of the dielectric measurement, differential scanning calorimetry, neutron scattering; PS: fast scanning calorimetry, writing; WL and FJ local contacts inelastic and quasielastic neutron time-of-flight scattering; MZ and BF local contacts for neutron backscattering; DAS and FE: X-ray scattering, analysis of the raw X-ray data; RZ: neutron scattering, analysis of raw neutron scattering; AS: data analysis, conception of the project, supervising, writing.Conflicts of interest
There are no conflicting interests.Data availability
Supplementary information (SI): examples for raw data of the neutron scattering experiments, section fitting of dielectric data by the HN-function, analysis of dielectric spectra by the derivative approach, estimation of the low frequency density of states, comparison of different analysis methods for elastic scans of HAT6. See DOI: https://doi.org/10.1039/d5sm01247c.Data will be available from the corresponding author on a reasonable request.
Acknowledgements
The Institut Laue-Langevin (Grenoble/France) and the Heinz Maier-Leibnitz Zentrum (Garching/Germany) are thanked for enabling the neutron scattering experiments. The German Science Foundation is acknowledged for financial support (DFG project numbers 158088440, 282247454 and 430146019).References
- P. W. Anderson, Through the Glass Lightly, Science, 1995, 267, 1615–1616 Search PubMed.
- C. A. Angel, Formation of Glasses from Liquids and Biopolymers, Science, 1995, 267, 1924–1935 CrossRef PubMed.
- P. G. Debenedetti and F. H. Stillinger, Supercooled Liquids and the Glass Transition, Nature, 2000, 410, 259–267 Search PubMed.
- J. C. Dyre, Colloquium: The Glass Transition and Elastic Models of Glass-Forming Liquids, Rev. Mod. Phys., 2006, 78, 953–972 Search PubMed.
- S. Kivelson and G. Tarjus, In Search of a Theory of Supercooled Liquids, Nat. Mater., 2008, 7, 831–833 CrossRef CAS PubMed.
- J.-H. Hung, T. Patra, V. Meenakshisundaram, J. H. Mangalara and D. S. Simmons, Universal localization transition accompanying glass formation: insights from efficient molecular dynamics simulations of diverse supercooled liquids, Soft Matter, 2019, 15, 9529–9540 Search PubMed.
- V. Lubchenko, Theory of the Structural Glass Transition: A Pedagogical Review, Adv. Phys., 2015, 64, 283–443 CrossRef CAS.
- Q. Zheng, Y. Zhang, M. Montazerian, O. Gulbiten, J. C. Mauro, E. D. Zanotto and Y. Z. Mauro, Understanding Glass through Differential Scanning Calorimetry, Chem. Rev., 2019, 119, 7848–7939 CrossRef CAS PubMed.
- M. Ediger and P. Harrowell, Perspective: Supercooled Liquids and Glasses, J. Chem. Phys., 2012, 137, 080901–080915 CrossRef CAS PubMed.
- H. Vogel, Das Temperaturabhängigkeitsgesetz der Viskosität von Flüssigkeiten, Phys. Z., 1921, 22, 645–646 CAS.
- G. S. Fulcher, Analysis of Recent Measurements of the Viscosity of Glasses, J. Am. Ceram. Soc., 1925, 8, 339–355 CrossRef CAS.
- G. Tammann and W. Hesse, Die Abhängigkeit der Viscosität von der Temperatur bei unterkühlten Flüssigkeiten, Z. Anorg. Allg. Chem., 1926, 156, 245–257 CrossRef CAS.
- C. A. Angell, Relaxation in Liquids, Polymers and Plastic Crystals—Strong/Fragile Patterns and Problems, J. Non-Cryst. Solids, 1991, 131–133, 13–31 CrossRef CAS.
- C. A. Angell, Entropy and Fragility in Supercooling Liquids, J. Res. Natl. Inst. Stand. Technol., 1997, 102, 171–185 CrossRef CAS PubMed.
- A. Schönhals, F. Kremer, A. Hofmann, E. W. Fischer and E. Schlosser, Anomalies in the scaling of the dielectric α-relaxation, Phys. Rev. Lett., 1993, 70, 3459 Search PubMed.
- A. Schönhals, Universal evidence for a crossover behavior of the dynamic glass transition, Europhys. Lett., 2001, 56, 815 Search PubMed.
- G. Adam and J. H. Gibbs, On the Temperature Dependence of Cooperative Relaxation Properties in Glass-Forming Liquids, J. Chem. Phys., 1965, 43, 139–146 CrossRef CAS.
- E. J. Donth, Relaxation and thermodynamics in polymers: glass transition, Akademie Verlag, Berlin, 1992 Search PubMed.
- L. Berthier, G. Biroli, J.-P. Bouchaud, L. Cipelletti, D. El Masri, F. Ladieu and M. Pierno, Direct Experimental Evidence of a Growing Length Scale Accompanying the Glass Transition, Science, 2005, 310, 1797–1800 CrossRef CAS PubMed.
- T. Bauer, P. Lunkenheimer and A. Loidl, Cooperativity and the Freezing of Molecular Motion at the Glass Transition, Phys. Rev. Lett., 2013, 111, 225702-1–225702-5 Search PubMed.
- R. Zallen, The physics of amorphous solids, Wiley, New York, 1983 Search PubMed.
- M. Beiner, S. Kahle, S. Abens, E. Hempel, S. Höring, M. Meissner and E. Donth, Low-temperature heat capacity, glass-transition cooperativity, and glass-structure vault breakdown in a series of poly(n-alkyl methacrylate)s, Macromolecules, 2001, 34, 5927–5935 Search PubMed.
- X. Monnier, J. Colmenero, M. Wolf and D. Cangialosi, Reaching the ideal glass in polymer spheres: Thermodynamics and vibrational density of states, Phys. Rev. Lett., 2021, 126, 118004 CrossRef CAS PubMed.
- V. K. Malinovsky, V. N. Novikov and A. P. Sokolov, Investigation of structural correlations in disordered materials by Raman scattering measurements, J. Non-Cryst. Solids, 1987, 90, 485–488 CrossRef.
- U. Buchenau, Y. M. Galperin, V. L. Gurevich, A. D. Parshin, M. A. Ramos and H. R. Schober, Interaction of soft modes and sound waves in glasses, Phys. Rev. B: Condens. Matter Mater. Phys., 1992, 46, 2798–2808 CrossRef PubMed.
- B. B. Laird and H. R. Schober, Localized low-frequency vibrational modes in a simple model glass, Phys. Rev. Lett., 1991, 66, 636–639 Search PubMed.
- S. N. Taraskin, Y. L. Loh, G. Natarajan and S. R. Elliott, Origin of the boson peak in systems with lattice disorder, Phys. Rev. Lett., 2001, 86, 1255–1258 Search PubMed.
- A. I. Krichikov, A. Jezowski, D. Szewczyk, O. A. Korolyuk, O. O. Romantsova, L. M. Buravtseva, C. Cazoria and J. L. Tamarit, Role of optical phonons and anharmonicity in the appearance of the heat capacity Boson peak-like anomaly in fully ordered molecular crystals, J. Phys. Chem. Lett., 2022, 13, 5061–5067 CrossRef PubMed.
- W. Schirmacher, G. Diezemann and C. Ganter, Harmonic vibrational excitations in disordered solids and the “boson peak”, Phys. Rev. Lett., 1998, 81, 136–139 CrossRef CAS.
- G. Ding, E. Ma, F. Jiang, S. Cai, N. Xu, B. Cui, L. Dai and M. Jiang, Unified theory of phones in solids with phase diagram of non-Debye anomalies, Nat. Phys., 2025, 21, 1911–1919 Search PubMed.
- A. Zaccone, A unified picture of phonon anomalies in crystals and lasses, Sci. China:Phys., Mech. Astron., 2026, 69, 226131 Search PubMed.
- M. Gonzalez-Jimenez, T. Barnard, B. A. Russell, N. V. Tukachev, U. Javonik, L.-A. Hayes, A. J. Farrell, S. Guinane, H. M. Senn, A. J. Smith, M. Wilding, G. Mali, M. Nakano, Y. Miyazaki, P. McMillan, G. C. Sosso and K. Wynne, Understanding the emergence of the boson peak in molecular glasses, Nat. Commun., 2023, 14, 215 Search PubMed.
- E. Lerner and E. Bouchbinder, Boson-peak vibrational modes in glasses feature hybridized phononic and quasilocalized excitations, J. Chem. Phys., 2023, 158, 194503 CrossRef CAS PubMed.
- H. Suga and S. Seki, Thermodynamic Studies on Glassy States of Pure Simple Compounds, J. Non-Cryst. Solids, 1974, 16, 171–194 CrossRef CAS.
- L. M. Wang and R. Richert, Dynamics of glass-forming liquids. IX. Structural versus dielectric relaxation in monohydroxy alcohols, J. Chem. Phys., 2004, 121, 11170–11176 CrossRef CAS PubMed.
- D. L. Leslie-Pelecky and N. O. Birge, Universal scaling of the relaxation near a model glass transition, Phys. Rev. Lett., 1994, 72, 1232–1236 CrossRef CAS PubMed.
- B. Kuchta, M. Descamps and F. Affouard, Molecular dynamics study of orientational disorder in cyanoadamantane plastic crystal, J. Chem. Phys., 1998, 109, 6753–6763 CrossRef CAS.
- S. Benkhof, T. Blochowicz, A. Kudlik, C. Tschirwitz and E. Rössler, The dielectric response of glass forming propylene carbonate: Slow β-process and excess wing, Ferroelectrics, 2000, 236, 193–207 CrossRef CAS.
- R. Brand, P. Lunkenheimer and A. Loidl, Relaxation dynamics in plastic crystals, J. Chem. Phys., 2002, 116, 10386–10401 CrossRef CAS.
- B. Glüsen, A. Kettner, J. Kopitzke and J. H. Wendorff, On the character of the glass transition in columnar discotics, J. Non-Cryst. Solids, 1998, 241, 113–120 CrossRef.
- H. Groothues, F. Kremer, P. G. Schouten and J. M. Warman, Charge transport and molecular dynamics in columnar stacks of liquid crystalline phthalocyanine derivatives, Adv. Mater., 1995, 7, 283–286 Search PubMed.
- C. Krause, H. Yin, C. Cerclier, D. Morineau, A. Wurm, C. Schick, F. Emmerling and A. Schönhals, Molecular dynamics of a discotic liquid crystal investigated by a combination of dielectric relaxation and specific heat spectroscopy, Soft Matter, 2012, 8, 11115–11122 RSC.
- A. Yildirim, A. Bühlmeyer, S. Hayashi, J. C. Haenle, K. Sentker, C. Krause, P. Huber, S. Laschat and A. Schönhals, Multiple Glass Dynamics in Dipole Functionalized Triphenylene Based Discotic Crystals Revealed by Broadband Dielectric Spectroscopy and Advanced Calorimetry – Assessment of the Molecular Origin, Phys. Chem. Chem. Phys., 2019, 21, 18265–18277 Search PubMed.
- A. Yildirim, C. Krause, R. Zorn, W. Lohstroh, G. J. Schneider, M. Zamponi, O. Holderer, B. Frick and A. Schönhals, Complex molecular dynamics of a symmetric model discotic liquid crystal revealed by broadband dielectric, thermal and neutron spectroscopy, Soft Matter, 2020, 16, 2005–2016 RSC.
- A. Yildirim, M. Kolmangadi, A. Bühlmeyer, P. Huber, S. Laschat and A. Schönhals, Ele ctrical conductivity and multiple glassy dynamics of crown-ether based columnar liquid crystals, J. Phys. Chem. B, 2020, 124, 8728–8739 CrossRef CAS PubMed.
- A. Yildirim, P. Szymoniak, K. Sentker, M. Butschies, A. Bühlmeyer, P. Huber, S. Laschat and A. Schönhals, Dynamics and Ionic Conductivity of Ionic Liquid Crystals Forming a Hexagonal Columnar Mesophase, Phys. Chem. Chem. Phys., 2018, 20, 5626–5635 RSC.
- M. A. Kolmangadi, A. Yildirim, K. Sentker, M. Butschies, A. Bühlmeyer, P. Huber, S. Laschat and A. Schönhals, Molecular dynamics and electrical conductivity of guanidium based ionic liquid crystals: Influence of cation headgroup configuration, J. Mol. Liq., 2021, 330, 115666 CrossRef CAS.
- A. Yildirim, C. Krause, P. Huber and A. Schönhals, Multiple glassy dynamics of a homologous series of triphenylene-based columnar liquid crystals – A study by broadband dielectric spectroscopy and advanced calorimetry, J. Mol. Liq., 2022, 358, 119212 CrossRef CAS.
- M. A. Kolmangadi, G. J. Smales, L. ZhuoQing, A. Yildirim, E. Wuckert, S. Eutionnat, F. Demel, P. Huber, S. Laschat and A. Schönhals, Side Chain Length-Dependent Dynamics and Conductivity in Self-Assembled Ion Channels, J. Phys. Chem. C, 2022, 126, 10995–11006 Search PubMed.
- M. A. Kolmangadi, A. R. Raab, P. Szymoniak, Z. Li, P. Huber, S. Laschat and A. Schönhals, Molecular mobility and electrical conductivity of amino acid-based (DOPA) ionic liquid crystals in the bulk state and nanoconfinement, Phys. Chem. Chem. Phys., 2025, 27, 18162 Search PubMed.
- Handbook of Liquid Crystals, ed. D. Demus, J. Goodby, G. W. Gray, H. W. Spiess and V. Vill, Wiley-VCH, Weinheim, 1998 Search PubMed.
- S. Laschat, A. Baro, N. Steinke, F. Giesselmann, C. Hägele, G. Scalia, R. Judele, E. Kapatsina, S. Sauer, A. Schreivogel and M. Tosoni, Discotic Liquid Crystals: From Tailor-Made Synthesis to Plastic Electronics, Angew. Chem., Int. Ed., 2007, 46, 4832–4887 Search PubMed.
- S. Sergeyev, W. Pisula and Y.-H. Geerts, Discotic liquid crystals: a new generation of organic semiconductors., Chem. Soc. Rev., 2007, 36, 1902–1929 Search PubMed.
- T. Wöhrle, I. Wurzbach, J. Kirres, A. Kostidou, N. Kapernaum, J. Litterscheidt, J. C. Haenle, P. Staffeld, A. Baro, F. Giesselmann and S. Laschat, Discotic liquid crystals, Chem. Rev., 2016, 116, 1139–1241 CrossRef PubMed.
- N. Boden, R. J. Bushby, J. Clements, M. V. Jesudason, P. F. Knowles and G. Williams, One-dimensional electronic conductivity in discotic liquid crystals, Chem. Phys. Lett., 1988, 152, 94–99 Search PubMed.
- V. Mathot, M. Pyda, T. Pijpers, G. Vanden Poel, E. van De Kerkhof, S. van Herwaarden, F. van Herwaarden and A. Leenaers, The Flash DSC 1, a power compensation twin-type, chip-based fast scanning calorimeter (FSC): First findings on polymers, Thermochim. Acta, 2011, 522, 36–45 CrossRef CAS.
- O. Paris, C. Li, S. Siegel, G. Weseloh, F. Emmerling, H. Riesemeier, A. Erko and P. Fratzl, J. Appl. Crystallogr., 2007, 40, S466 Search PubMed.
- A. P. Hammersley, FIT2D: a multi-propose data reduction, analysis and visualization program, J. Appl. Crystallogr., 2026, 49(Part 2), 646–652 Search PubMed.
- F. Kremer and A. Schönhals, Broadband dielectric measurement techniques, in Broadband Dielectric Spectroscopy, ed. F. Kremer and A. Schönhals, Springer, 2003, pp. 36–57 Search PubMed.
- M. Bée, Quasielastic neutron scattering, Principles and applications in solid state chemistry, biology and materials science, Adam Hilger, 1988 Search PubMed.
- Heinz Maier-Leibnitz Zentrum, TOFTOF: Cold neutron time-of-flight spectrometer, J. Large-Scale Res. Facil., 2015, 1, A15 Search PubMed.
- F. Rieutord, INX-Program for time-of-flight data reduction, ILL internal publication 90RI17T, 1990 Search PubMed.
- B. Frick, The neutron backscattering spectrometer IN16 at the ILL: High energy resolution with high intensity and excellent signal-to-noise ratio, Neutron News, 2002, 13, 15–22 Search PubMed.
- A. Schönhals, B. Frick, C. Krause and R. Zorn, Elastic Sans on a Homologous Series of Discotic Liquid Crystals, Institut Laue-Langevin (ILL), 2013 DOI:10.5291/ILL-DATA.6-05-930.
- P. Szymoniak, M. A. Kolmangadi, M. Böhning, B. Frick, M. Appel, R. A. Mole, N. R. Souza, R. Zorn and A. Schönhals, Confined Segmental Diffusion in Nanophase Separated Janus-Polynorbornenes as Investigated by Quasielastic Neutron Scattering, Macromolecules, 2024, 57, 8562–8575 CrossRef CAS.
- K. S. Singwi and A. Sjölander, Diffusive motion in water and cold neutron scattering, Phys. Rev., 1960, 119, 863 Search PubMed.
- R. Zorn, Neutron scattering studies of dynamics in confined geometries, Nucl. Instrum. Methods Phys. Res., Sect. A, 2009, 603, 439–445 Search PubMed.
- Heinz Maier-Leibnitz Zentrum, SPHERES: Backscattering spectrometer, J. Large-Scale Res. Facil., 2015, 1, A30 CrossRef.
- J. Wuttke, A. Budwig, M. Drochner, H. Kämmerling, F.-J. Kayser, H. Kleines, V. Ossovyi, L. C. Pardo, M. Prager, D. Richter, G. J. Schneider, H. Schneider and S. Staringer, SPHERES, Jülich's high-flux neutron backscattering spectrometer at FRM II, Rev. Sci. Instrum., 2012, 83, 075109 CrossRef PubMed.
- O. G. Randl, SQW – A comprehensive user manual, ILL internal publication 96RA07T, 1996 Search PubMed.
- R. Zorn, B. Frick and L. Fetters, Quasielastic neutron scattering study of the methyl group dynamics in polyisoprene, J. Chem. Phys., 2002, 116, 845–853 CrossRef CAS.
- A. N. Cammidge and R. J. Bushby, Chapter VII: Synthesis and Structural Features, in Handbook of Liquid Crystals, ed., D. Demus, J. Goodby, G. W. Gray, H.-J. Spiess and V. Vill, 1st edn, 1998, vol. 2B, pp. 693–748 Search PubMed.
- https://shop.synthon-chemicals.com/en/LIQUID-CRYSTALS/DISCOTICS/TRIPHENYLENES/2-3-6-7-10-11-Hexakis-heptanoyloxy-triphenylene.html .
- G. W. Stewart, Theory of X-Ray Diffraction in Liquids, Phys. Rev., 1928, 32, 558 CrossRef CAS.
- S. Laschat, A. Baro, N. Steinke, F. Giesselmann, C. Hägele, G. Scalia, R. Judele, E. Kapatsina, S. Sauer, A. Schreivogel and T. Tosoni, Discotic Liquid Crystals: From Tailor-Made Synthesis to Plastic Electronics, Angew. Chem., 2007, 119, 4916–4950 CrossRef.
- A. Schönhals and F. Kremer, Analysis of dielectric spectra, in Broadband Dielectric Spectroscopy, ed. F. Kremer and A. Schönhals, Springer, Berlin, 2003, pp. 59–98 Search PubMed.
- O. van den Berg, W. G. F. Sengers, W. F. Jager, S. J. Picken and M. Wübbenhorst, Dielectric and Fluorescent Probes to Investigate Glass Transition, Melt, and Crystallization in Polyolefins, Macromolecules, 2004, 37, 2460–2470 Search PubMed.
- M. Wübbenhorst and J. van Turnhout, Analysis of Complex Dielectric Spectra. I. One-Dimensional Derivative Techniques and Three-Dimensional Modelling, J. Non-Cryst. Solids, 2002, 305, 40–49 Search PubMed.
- E. Donth, The Size of Cooperatively Rearranging Regions at the Glass Transition, J. Non-Cryst. Solids, 1982, 53, 325–330 CrossRef CAS.
- S. W. Lovesey, Theory of Neutron Scattering from Condensed Matter, Oxford University, New York, 1987, vol. 1, p. 121 Search PubMed.
- R. Zorn, L. Hartmann, B. Frick, D. Richter and F. Kremer, J. Non-Cryst. Solids, 2002, 307, 547–554 CrossRef.
- C. Krause, R. Zorn, F. Emmerling, J. Falkenhagen, B. Frick, P. Huber and A. Schönhals, Vibrational density of states of triphenylene based discotic liquid crystals: dependence on the length of the alkyl chain, Phys. Chem. Chem. Phys., 2014, 16, 7324–7333 RSC.
- W. Schirmacher, B. Schmid, C. Tomaras, G. Viliani, G. Baldi, G. Ruocco and T. Scopigno, Phys. Status Solidi C, 2008, 5, 862–866 CrossRef CAS.
- M. T. Cicerone and C. L. Soles, Fast Dynamics and Stabilization of Proteins: Binary Glasses of Trehalose and Glycerol, Biophys. J., 2004, 86, 3836–3845 CrossRef CAS PubMed.
- R. Zorn, H. Yin, W. Lohstroh, W. Harrison, P. M. Budd, B. R. Pauw, M. Böhning and A. Schönhals, Anomalies in the low frequency vibrational density of states for a polymer with intrinsic microporosity – the Boson peak of PIM-1, Phys. Chem. Chem. Phys., 2018, 20, 1355–1363 RSC.
- R. Zorn, P. Szymoniak, M. Kolmangadi, M. Wolf, D. Alentiev, M. Bermeshev, M. Böhning and A. Schönhals, Low frequency vibrational density of state of highly permeable super glassy polynorbornenes – The Boson peak, Phys. Chem. Chem. Phys., 2020, 22, 18381–18387 RSC.
- R. Zorn, P. Szymoniak, M. A. Kolmangadi, R. Malpass-Evans, N. B. McKeown, M. Tyagi, M. Böhning and A. Schönhals, Low frequency vibrations and diffusion in disordered polymers bearing an intrinsic microporosity as revealed by neutron scattering, Crystals, 2021, 11, 1482 CrossRef CAS.
- M. F. T. Telling, Elastic and inelastic fixed window scans, A practical guide to quasi-elastic neutron scattering, RSC eTextbook Collection, 2020, ch. 7, 105–119 Search PubMed.
- A. Schönhals, R. Zorn and B. Frick, Inelastic Neutron Spectroscopy as a Tool to Investigate Nanoconfined Polymer Systems, Polymer, 2016, 105, 393–406 CrossRef.
- J. Colmenero, A. Moreno and A. Alegria, Neutron scattering investigations on methyl group dynamics in polymers, Prog. Polym. Sci., 2005, 30, 1147–1184 CrossRef CAS.
- M. Prager and A. Heidemann, Rotational Tunneling and Neutron Spectroscopy: A Compilation, Chem. Rev., 1997, 97, 2933–2966 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |



