Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
The effect of thioester linkages on the stability of the ferroelectric nematic phase
Gytis
Stepanafas
 a,
Stevie
Brown
a,
Stevie
Brown
 a,
Ewan
Cruickshank†
a,
Ewan
Cruickshank†
 a,
Grant J.
Strachan
a,
Grant J.
Strachan
 ab,
John M. D.
Storey
ab,
John M. D.
Storey
 a,
Corrie
Imrie‡
a,
Corrie
Imrie‡
 a and
Rebecca
Walker
a and
Rebecca
Walker
 *a
*a
aDepartment of Chemistry, University of Aberdeen, Old Aberdeen, AB24 3UE, UK. E-mail: rebecca.walker@abdn.ac.uk
bFaculty of Chemistry, University of Warsaw, Zwirki i Wigury 101, 02-089 Warsaw, Poland
First published on 2nd February 2026
Abstract
The synthesis and characterisation of thirteen sulfur-based liquid crystals are reported, based on the molecular backbone of RM734 and containing thioester linkages. Ten of these materials are new examples of ferroelectric nematogens. Phase transition temperatures are compared for systematic changes to molecular structure including the position and extent of fluorination, and the nature of lateral and terminal substituents. The new materials are compared with their ester-linked counterparts. In line with expected behaviour of the nematic phase, replacement of an ester with a thioester raised TNI, but NF phase stability upon making this change proved more variable. These findings remain in line with the general view that the formation of the NF phase is not solely governed by the magnitude of the molecular dipole moment, but is able to be tuned by the interplay of steric and electronic effects endowed by different molecular fragments.
Introduction
The recent groundbreaking discovery of proper ferroelectricity in low molar mass liquid crystals1 heralded a new era of both fundamental and experimental research in the field, aiming to understand and exploit the fantastic properties of the ferroelectric nematic phase, NF, for electrooptical applications. A polar variant of the conventional nematic phase, N, the NF phase has huge application potential, exhibiting switchable electric polarisation and non-linear response comparable in magnitude to that of solid crystalline ferroelectrics while retaining the fluidity and self-healing ability vital in electrooptical devices.2–10 The vast majority of compounds exhibiting the phase can be categorised by a limited number of molecular templates. The molecular archetypes for the NF phase are the two ‘flagship’ materials of groups in Europe and Asia, RM73411,12 and DIO,13 respectively and UUQU-4N,14 for which a room-temperature stable NF phase was discovered, Fig. 1. Liquid crystal chemists sought to establish design rules for the NF phase through the creation of large libraries of molecules with structures based on these three templates.15–18 While some fundamental features quickly became apparent: the necessity for a large longitudinal molecular dipole moment to encourage ferroelectric ordering of molecules, and some degree of lateral bulk, robust structure–property relationships have yet to be established. Nonetheless, the surge in new materials has led to the discovery of numerous other new proper ferroelectric phases in the ‘ferroelectric nematic realm’, including polar variants of the chiral nematic, NF*,19–22 SmA and SmC phases (SmAF and SmCF),23–32 the antiferroelectric NX33–37 and SmAAF phases,38,39 and heliconical ferroelectric nematic (NTBF)40–42 and smectic (SmCHP) phases.38,43Sulfur-containing liquid crystal materials are attractive candidates for use in liquid crystal display technologies,44 liquid crystal lenses,45 and liquid crystal lasers,46 owing to their enhanced values of optical birefringence47,48 compared with structurally analogous molecules instead containing oxygen-based functionalities. A wide range of sulfur-based fragments have been used in the design of low molar mass and dimeric liquid crystals, including terminal alkylthio chains,49–56 alkylthio spacers,57–62 thiophene moieties,33,63 thiocyanate terminal groups,37,47,64,65 pentafluorosulfanyl terminal groups,12,66,67 and thioester linking groups.68–70 Such functionalities have recently been incorporated into molecular structures based on the RM734 archetype53,71,72 but examples of sulfur-containing ferroelectric nematogens remain minimal.
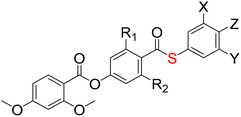
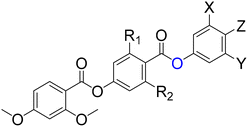
Here we report thirteen thioester-containing materials, eleven of which exhibit the NF phase (Table 1). These are RM734-type molecules, with our parent compound GS36 simply being RM734 with a thioester in place of the ester adjacent to the polar terminus. A selection of structural modifications are made to this backbone, including the position and extent of fluorination, and the nature of lateral and terminal substituents, and the effect of these changes on the phase behaviour is assessed, particularly the stability of the ferroelectric nematic phase. We also compare the thioester-containing compounds to their conventional ester analogues, Table 2, the data for which has either been extracted from our previous literature or re-synthesised for comparison.
Experimental
Synthesis
The synthesis of compounds GS36–48 consisted of two stages: the preparation of a selection of sulfur-containing intermediates, followed by the synthesis of the final products, see Scheme 1. The preparation of intermediates 4a–4f, involved three different routes depending on the availability of the starting materials and the different synthetic steps required. In the first step, 4-fluoro-2-methoxybenzonitrile is treated with base to obtain 1a. In all three routes, the appropriate phenol is reacted with dimethylthiocarbamoyl chloride to produce the O-aryl thiocarbamate intermediates 2a–2f. These are subjected to high temperatures and undergo intramolecular rearrangement, also known as Newman–Kwart rearrangement, to obtain the S-aryl thiocarbamate intermediates 3a–3f, followed by base hydrolysis to yield the required thiophenol-based intermediates, 4a–4f. The intermediates 4d, 4e and 4f were obtained as mixtures in which the fluorine atom was substituted by a methoxy group, resulting in products A and B. Further details of this are provided in the SI.The intermediates 5a–d was prepared by methods reported elsewhere73,74 and the target molecules GS36–48 were formed by a Steglich esterification reaction between these and intermediates 4a–f. A detailed description of the preparation of all the target compounds along with analytical data can be found in the SI.
Characterisation
Mesophase identification from their characteristic optical textures was performed by POM, using an Olympus BH2 polarising light microscope equipped with a Linkam TMS 92 hot stage. Studied samples were sandwiched between two untreated glass coverslips and for characteristic comparison materials were viewed within cells treated for planar alignment, purchased from INSTEC with a thickness between 2.9–3.5 µm and an ITO conducting layer.Thermal properties of the materials were studied using differential scanning calorimetry performed by a Mettler Toledo DSC1 or DSC3 differential scanning calorimeter equipped with TSO 801RO sample robots and calibrated using indium and zinc standards. Heating and cooling rates were maintained at 10 °C min per minute and all samples were measured under a nitrogen atmosphere. Transition temperatures, which are assigned as the peak maxima, and associated enthalpy changes were extracted from the first heating run unless stated otherwise.
Results and discussion
The thirteen thioester-containing compounds will first be discussed in two sets, split according to the nature of their Z terminus: GS36–43 contain a terminal nitro group and GS44–S48 a terminal nitrile group. We will begin by varying the number and substitution pattern of lateral fluorine atoms and methoxy groups within each set of molecules and assessing the resultant mesophase behaviour. Differences in such behaviour upon changing the nature of the terminal group will be considered, and the transitional properties compared to those of corresponding molecules containing an ester rather than thioester linkage.The effect of lateral substitution in nitro-terminated compounds (GS37–43)
The transitional properties of GS36–43 are summarised in Table 3. We note that GS36 has been published elsewhere and transition temperatures obtained here are in excellent agreement with those previously reported.71 Phase assignments were primarily based on the observation of characteristic textures when the samples were viewed through the polarised optical microscope. Specifically, nematic (N) phases were identified in untreated glass slides by schlieren textures containing two- and four-point brush defects that flashed when subjected to mechanical stress (Fig. 2a), and in cells treated for planar alignment by a uniform texture (Fig. 2b). Additional defect lines appeared when the sample was cooled from the N phase to the lower temperature mesophase; the effect was particularly distinct for samples viewed in planar-aligned cells (Fig. 2c) and this so-called ‘banded’ texture has become characteristic of the ferroelectric nematic phase NF. Such a texture was also observed following coalescence of spherical droplets upon direct transition from the isotropic to the NF phase in GS38, 41 and 43 (Fig. 2d). These materials are highly monotropic and required significant supercooling to observe the NF phase, as such many of the temperatures reported were obtained solely from microscope observations. However, where possible scaled entropy changes were obtained from DSC (Table 3). Values of ΔS/R associated with the direct NF–I transitions of GS38 and GS43 are several times larger than those for N–I transitions in other compounds. This is in accord with data reported for other ferroelectric nematogens having direct NF–I transitions and is thought to reflect the additional entropic contribution associated with the ordering of the dipoles in the NF phase.75–78| μ/D | M.P./°C (ΔS/R) | T NFN/°C *TNFI/°C (ΔS/R) | T NI/°C (ΔS/R) | |
|---|---|---|---|---|
| a Values extracted from DSC cooling traces. b Measured using the polarised optical microscope. [dec.] indicates decomposition prior to clearing. | ||||
| GS36 | 12.4 | 174 (9.0) | 107b | 215 (0.13) |
| GS37 | 13.5 | 186 (16.9) | 112b | 171a (0.10) |
| GS38 | 13.5 | 148 (13.1) | *95a (0.51) | — |
| GS39 | 13.1 | 184 (14.5) | 125b | 185a (0.13) |
| GS40 | 12.8 | 180 (13.4) | 132b | 141b |
| GS41 | 13.0 | 165 (13.0) | *108b | — |
| GS42 | 12.8 | 151 (11.3) | 102b | 104b |
| GS43 | 13.0 | 172 (18.8) | *49a (0.93) | — |
| GS44 | 12.3 | 191 (11.6) | — | 240 [dec.] |
| GS45 | 12.3 | 212 (14.2) | — | 205a (0.15) |
| GS46 | 14.0 | 164 (10.7) | 143 (0.23) | 169a (0.11) |
| GS47 | 10.5 | 196 (19.2) | 95b | 123b |
| GS48 | 10.9 | 201 (16.7) | *121a (1.31) | — |
The material containing no additional lateral substituents on the central and right-terminal rings, GS36, will be used as a reference against which the effects of fluorine and methoxy substitution will be assessed in molecules GS37–43 (Fig. 3). The high value of TNI seen for GS36 may attributed, at least in part, to the thioester linkage which is known to enhance nematic behaviour68,69,79 and we will return to this point later.
Introducing an additional lateral methoxy substituent in compounds GS41–43 greatly destabilised the nematic phase and this presumably reflects the large change in shape, a lowering of the molecular anisotropy and thus a reduction in the ability of the molecule to form antiparallel dimers which drive a conventional nematic arrangement of molecules. Adding the methoxy substituent to the middle ring (GS41) led to a loss of the nematic phase entirely, corresponding to a decrease in TNI of at least 100 K compared to the parent compound GS36, and of at least 69 K compared to its fluorine substituted analogue GS37. Moving the lateral methoxy group from the middle ring to the nitro-substituted terminus to give GS42 reduced TNI by 111 K compared to GS36. GS43 has both a fluorine and a methoxy substituent on the terminal ring, and, as for the di-fluoro substituted GS38, the nematic phase is not observed. This is consistent with previous observations.74
The addition of a fluorine atom ortho to the right-terminal nitro group (GS37) enhances TNFN by 5 K. This has been attributed to the fluorine substituent acting to spread electron density more evenly around the terminal ring, reducing the amplitude of the charge density wave and thus stabilising the NF phase. The effect of adding a fluorine substituent to the middle ring is even more pronounced: the TNFN of GS39 is increased by 18 K compared to that of the non-fluorinated GS36. Comparing the two mono-fluorinated compounds, GS37 and GS39, moving the single additional F atom from the right-terminal to the central ring increases TNFN (by 13 K), despite a reduction in the overall molecular dipole moment by 0.4 D§ (Table 3) which further highlights that just the dipolar magnitude alone is not enough to predict the behaviour of these compounds as has been previously observed.15 These observations can be interpreted as an increase in the amplitude of the charge density wave associated with the central ring due to the fluorine substituent, reducing the relative amplitude of the waves at either end of the molecule, and thus stabilising the NF phase. It is important to note, however, that the accompanying change in molecular shape is not accounted for in this model as it assumes the molecules can be described by similar cylindrical rods. This effect is even greater for GS40, with two fluorine substituents on the central ring. GS40 has the highest TNFN of this trio, elevated by 25 K and 7 K from GS36 and GS39, respectively. These differences may be attributed to a combination of (i) a favourable change in molecular shape and (ii) a further increase in the amplitude of the charge density wave on the fluorine-substituted central ring relative to that of the terminus. However, this framework does not fully explain the behaviour of GS38, with two fluorine substituents on the right-terminal ring. For this material, the addition of the second fluorine atom has a detrimental effect on the stability of the NF phase, lowering TNFI by 17 K and 12 K compared to GS37 and GS36, respectively. In terms of the Madhusudana model, the reason for this decrease is not immediately apparent.80 The addition of a second F atom would contribute to a further spread of the electronic charge more evenly around the right-terminus, reducing the amplitude of the charge density wave, and thus an increase in TNFI might have been expected. Presumably, this effect is offset by an unfavourable change in the molecular shape.
The application of this framework is further complicated by the more pronounced changes in molecular shape produced by methoxy substituents. According to the Madhusudana model, the electron donating methoxy group acts to increase electron density in the ring to which it is attached.80 In such terms, the inclusion of a methoxy substituent on the middle ring (GS41) would be expected to promote the NF phase. However, TNFI of GS41 is essentially the same as TNFN of GS36. In addition, TNFI for GS41 is lower by 17 K than TNFN of its fluorine-substituted analogue GS39. This appears to highlight the importance of shape effects from the larger methoxy group. GS42, in which the methoxy substituent is added ortho to the terminal nitro group, has a substantially lower TNI – by over 100 K – than the unsubstituted GS36, attributed to a decrease in molecular anisotropy, as discussed in the previous section. However, TNFN for GS42 is significantly less affected, with a decrease of only 5 K. This suggests that, in this case, the detrimental effect of molecular shape is at least somewhat counteracted by favourable electronic factors which act to stabilise the ferroelectric nematic phase. Namely, in the same manner as ortho fluorine substituents in GS37 and GS38, the methoxy group acts to decrease the amplitude of the charge density wave at the end of the molecule by spreading the charge around the terminal substituents. The net effect is therefore a much smaller decrease in TNFN than in TNI, which is less sensitive to electronic effects.
A fluorine atom is added ortho to the terminal nitro group in GS42 to give the 3,4,5-terminated GS43 and this results in a decrease in TNF− of 53 K. This is consistent with the trend seen with the di-fluoro substituted GS38, for which the second substituent ortho to the terminal nitro group destabilises the NF phase. A final comparison between GS38 (3,4,5-terminated where positions 3 and 5 are F) and GS43 (3,4,5-terminated where position 3 is OMe and 5 is F) sees a reduction of TNFI by 46 K on substituting F with OMe. This is an even more pronounced decrease than that produced by the analogous substitution between GS37 and GS42 (10 K). These observations suggest that, in nitro-terminated molecules, this di-substitution is not conducive to the parallel association of the molecules, i.e. a ferroelectric arrangement, and this is exacerbated by the associated change in shape.
The effect of lateral substitution in nitrile-terminated compounds (GS44–48)
We now turn our attention to five nitrile-terminated compounds for which the additional substituents on this terminal ring are varied while the rest of the molecule remains unchanged. The phase behaviour for these is also summarised in Table 2 and assignments again made based on characteristic optical textures. GS44 is the nitrile-terminated analogue of GS36, and we will use this as a reference against which the effects of structural modifications are assessed (Fig. 4).Somewhat surprisingly, GS47, with a methoxy substituent ortho to the nitrile group, also exhibits a monotropic NF–N transition. The addition of a fluorine atom ortho to the terminal nitrile group in GS47 to give GS48 resulted in a direct NF–I transition. The value of TNFI for GS48 is 26 K higher than TNFN for GS47. In contrast, comparing GS48 with GS46, the change from a fluorine to a methoxy substituent reduced TNFN by at least 22 K.
It seems that while a single substituent ortho to the nitrile terminus is detrimental to both nematic phases, the NF phase is re-stabilised by the introduction of a second substituent ortho to CN, presumably as a result of the double substitution effectively disrupting antiparallel packing of molecules and promoting a ferroelectric arrangement.
The trend in the values of TNFN/TNFI between these two groups of compounds is more variable. Of the three pairs for which a comparison is possible, the nitrile-substituted compound shows significantly higher transition temperatures in two cases (GS38/46 and GS43/48), but this trend is reversed in the third. It is apparent that there is no simple relationship with the dipole moments of these pairs. It appears that two substituents at positions ortho to a nitrile group are required to significantly inhibit the antiparallel associations which promote N phase behaviour, and when this is the case, the resulting shape of these molecules is more conducive to the formation of the NF phase than that of their nitro-terminated analogues. For nitrile-terminated materials with only one ortho-substituent, (e.g.GS47) the formation of antiparallel dimers is still possible, which decreases the stability of the NF phase relative to the N phase, and hence the inversion of TNFN for GS47 and GS42 is seen. The early onset of crystallisation observed for GS45 prevented this comparison from being extended to the equivalent single fluorinated derivative.
Thioester vs. ester
Nine of the new thioester-containing materials GS36–41 and 44–46 can be compared to the corresponding compounds containing an ester link. The structures of the ester-containing molecules are given in Table 2, and their transitional properties in Table S1. These materials have previously been published by us and others, and the transitional properties obtained here are generally in excellent agreement with literature. An exception is compound GO9, which contains F atoms in the two positions ortho to a nitrile terminal group. GO9 was found to exhibit a direct NF–I transition at 150 °C, while previously it was reported to be solely nematogenic.For all pairs, TNI is higher for the thioester-containing compounds, as can be seen in Fig. 6. This is consistent with previously observed trends for liquid crystals comparing thioester and ester linkages. The thioester linkage makes the overall molecular shape longer and more anisotropic than the corresponding ester71 and the larger, more electron-rich, and highly polarisable sulfur atom acts to enhance intermolecular interactions.
Conversely, in all but two cases, TNFN/TNFI is reduced by the presence of the sulfur-based group. This can be rationalised by electronic differences between oxygen and sulfur atoms. Specifically, delocalisation of electrons between the heteroatom (either O or S) and the carbonyl group is less effective for sulfur because the energy gap between sulfur's 3p orbital and the carbonyl π-system is larger compared to that of the equivalent oxygen orbital (2p). However, sulfur's electrons can more effectively interact with the π-system of the terminal ring such that, in contrast to the ester-linked materials, the terminal ring is more electron-rich. Consequently, the amplitude of the charge density wave at this terminus is increased and the NF phase is destabilised. We will return to the exceptional cases below.
Increasing the number of F substituents on the polar terminus has a similar effect on both ester- and thioester-containing materials (O1–3 and S36–38) such that TNI is highest for the non-fluorinated materials (O1/S36) and the singly fluorinated analogues (O2/S37) show the highest temperature NF phase, the reasons for which were discussed earlier. The addition of lateral substituents on the central ring (O4–O6, S39–41) appears to have a greater effect on the NF phase for the thioesters. GS39, with one F substituent, has a lower TNFN compared to GO4. However, the effect of the second fluorine is dependent on the nature of the linkage: ester-linked material GO5 has a lower TNFN than GO4, while TNFN of the thioester-linked GS40 is enhanced compared to GS39; GO5 and GS39 therefore coincidentally have identical values of TNFN. For both the thioester and ester linked compounds, the electronic effects of adding a single fluorine to the middle ring enhances TNFN, as discussed earlier, and appears to overcome any detrimental changes to molecular shape. However, while the addition of a second fluorine also has this favourable electronic effect, the accompanying changes to molecular shape appear to vary depending on the nature of the linking group. Specifically, the unsubstituted thioester GS36 has a more anisotropic shape than the corresponding oxygen material RM734, which has been interpreted as being unfavourable due to the potential for antiparallel interactions which according to the predictions of Madhusudana inhibits the formation of the NF phase.80 This is indeed reflected in their respective NF–N transition temperatures, and this is also true for the singly substituted (GS39 and GO4). For the more favourably shaped oxygen-based materials, the addition of a second fluorine substituent appears to have a detrimental effect on molecular shape, to a large enough extent that it offsets the favourable electronic changes. In contrast, the detrimental effect on the molecular shape endowed by the inclusion of the second fluorine atom appears to be less significant in the thioester compounds, due to their less favourable starting shape, such that the cumulative steric and electronic effects are dominated by the latter and enhance the stability of the NF phase. This is even more pronounced for compounds with a single lateral methoxy group in the middle ring, which acts to drastically reduce TNI in both types of material: GO6vs.GO1, GS41vs.GS36. In the ester compounds TNF− is also significantly reduced, while its thioester counterpart maintains a nearly identical TNF− to the non-substituted material, such that now TNFIGS41 > TNFIGO6.
Molecules with a nitrile terminus (GO7–9vs.GS44–46) share the expected increase in TNI on replacing an ester with a thioester linkage. Only the pair with two F atoms ortho to the polar terminus (GO9, GS46) exhibit an NF phase, for reasons discussed earlier, but the sulfur-containing material is the only one to show both N and NF phases. This is presumably a consequence of the stability endowed to the N phase by the thioester moiety, in conjunction with that provided by the nitrile group. The molecular dipole moment of GS46 is only 0.1 D higher than its ester-containing counterpart, and the latter has the higher TNFN by 6 K. The C–S–C bond in the thioester enhances the overall linearity of the molecules compared to the C–O–C bond of an ester, and consequently causes the magnitude of the overall molecular dipole moment to be higher as well as the value of TNI.71 The value of TNFN, however, falls and this is likely due to the mesogens favouring antiparallel correlations preferable for the conventional nematic phase rather than the parallel correlations favoured by the NF phase thanks to the shape and electronic profile of the molecule.
Conclusions
We have reported thirteen sulfur-based liquid crystals based on the molecular backbone of RM734 and containing thioester linkages, ten of which are new ferroelectric nematogens. Phase transition temperatures are compared for systematic changes to molecular structure including the position and extent of fluorination, and the nature of lateral and terminal substituents. The new materials are compared with their ester-linked counterparts, including one for which phase behaviour has been reassigned from solely nematic to solely ferroelectric nematic. In line with expected behaviour of the nematic phase, replacement of an ester with a thioester raised TNI, but NF phase stability upon making this change proved more variable. These findings remain in line with the general view that the formation of the NF phase is not solely governed by the magnitude of the molecular dipole moment, but able to be tuned by the interplay of steric and electronic effects endowed by different molecular fragments.Author contributions
GS, SB and EC synthesised the reported compounds. GS, SB, EC, RW performed thermal and optical analyses. Interpretation of results was performed by GS, CTI, EC, GJS and RW. CTI and JMDS proposed the project and acquired funding. RW, GJS and EC wrote and edited the original manuscript; all authors provided input to the final manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5sm01206f.Acknowledgements
Thank you to Marijus Juodka for helpful discussions regarding Orca calculations. The authors would like to acknowledge the immense contributions to this project, as well as past, ongoing present, and future work by Professor CT Imrie, who passed away suddenly in January 2025. CTI and JMDS acknowledge the financial support of the Engineering and Physical Sciences Research Council [EP/V048775/1].References
- X. Chen, E. Korblova, D. Dong, X. Wei, R. Shao, L. Radzihovsky, M. A. Glaser, J. E. MacLennan, D. Bedrov, D. M. Walba and N. A. Clark, Proc. Natl. Acad. Sci. U. S. A., 2020, 117, 14021–14031 CrossRef CAS PubMed.
- F. Caimi, G. Nava, R. Barboza, N. A. Clark, E. Korblova, D. M. Walba, T. Bellini and L. Lucchetti, Soft Matter, 2021, 17, 8130–8139 RSC.
- P. Rudquist, Sci. Rep., 2021, 11, 24411 Search PubMed.
- C. L. Folcia, J. Ortega, R. Vidal, T. Sierra and J. Etxebarria, Liq. Cryst., 2022, 49, 899–906 CrossRef CAS.
- H. Nishikawa, K. Sano, S. Kurihara, G. Watanabe, A. Nihonyanagi, B. Dhara and F. Araoka, Commun. Mater., 2022, 3, 89 CrossRef CAS.
- C. Parton-Barr, H. F. Gleeson and R. J. Mandle, Soft Matter, 2024, 20, 672–680 RSC.
- A. Erkoreka and J. Martinez-Perdiguero, Phys. Rev. E, 2024, 110, L022701 CrossRef CAS PubMed.
- J. Szydlowska, P. Majewski, M. Čepič, N. Vaupotič, P. Rybak, C. T. Imrie, R. Walker, E. Cruickshank, J. M. D. Storey, P. Damian and E. Gorecka, Phys. Rev. Lett., 2023, 130, 216802 Search PubMed.
- M. T. Máthé, B. Farkas, L. Péter, Á. Buka, A. Jákli and P. Salamon, Sci. Rep., 2023, 13, 6981 Search PubMed.
- N. Sebastián, R. J. Mandle, A. Petelin, A. Eremin and A. Mertelj, Liq. Cryst., 2021, 48, 2055–2071 Search PubMed.
- R. J. Mandle, S. J. Cowling and J. W. Goodby, Phys. Chem. Chem. Phys., 2017, 19, 11429–11435 RSC.
- R. J. Mandle, S. J. Cowling and J. W. Goodby, Chem. – Eur. J., 2017, 23, 14554–14562 CrossRef CAS PubMed.
- H. Nishikawa, K. Shiroshita, H. Higuchi, Y. Okumura, Y. Haseba, S. I. Yamamoto, K. Sago and H. Kikuchi, Adv. Mater., 2017, 29, 1702354 Search PubMed.
- A. Manabe, M. Bremer and M. Kraska, Liq. Cryst., 2021, 48, 1079–1086 Search PubMed.
- E. Cruickshank, ChemPlusChem, 2024, e202300726 Search PubMed.
- J. Li, Z. Wang, M. Deng, Y. Zhu, X. Zhang, R. Xia, Y. Song, Y. Hisai, S. Aya and M. Huang, Giant, 2022, 11, 100109 Search PubMed.
- R. J. Mandle, Soft Matter, 2022, 18, 5014–5020 Search PubMed.
- Y. Song, S. Aya and M. Huang, Giant, 2024, 19, 100318 Search PubMed.
- D. Pociecha, R. Walker, E. Cruickshank, J. Szydlowska, P. Rybak, A. Makal, J. Matraszek, J. M. Wolska, J. M. D. Storey, C. T. Imrie and E. Gorecka, J. Mol. Liq., 2021, 361, 119532 Search PubMed.
- X. Zhao, J. Zhou, J. Li, J. Kougo, Z. Wan, M. Huang and S. Aya, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2111101118 Search PubMed.
- C. Feng, R. Saha, E. Korblova, D. Walba, S. N. Sprunt, A. Jákli, C. Feng, S. N. Sprunt, A. Jákli, R. Saha, E. Korblova and D. Walba, Adv. Opt. Mater., 2021, 9, 2101230 CrossRef CAS.
- H. Nishikawa and F. Araoka, Adv. Mater., 2021, 33, 2101305 Search PubMed.
- J. Hobbs, C. J. Gibb, D. Pociecha, J. Szydłowska, E. Górecka and R. J. Mandle, Angew. Chem., Int. Ed., 2025, 64, e202416545 CrossRef CAS PubMed.
- Y. Song, M. Deng, Z. Wang, J. Li, H. Lei, Z. Wan, R. Xia, S. Aya and M. Huang, J. Phys. Chem. Lett., 2022, 13, 9983–9990 CrossRef CAS PubMed.
- X. Chen, V. Martinez, P. Nacke, E. Korblova, A. Manabe, M. Klasen-Memmer, G. Freychet, M. Zhernenkov, M. A. Glaser, L. Radzihovsky, J. E. Maclennan, D. M. Walba, M. Bremer, F. Giesselmann and N. A. Clark, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2210062119 CrossRef CAS PubMed.
- H. Nishikawa, M. Kuwayama, A. Nihonyanagi, B. Dhara and F. Araoka, J. Mater. Chem. C, 2023, 11, 12525–12542 Search PubMed.
- H. Kikuchi, H. Matsukizono, K. Iwamatsu, S. Endo, S. Anan and Y. Okumura, Adv. Sci., 2022, 9, 2202048 CrossRef PubMed.
- H. Kikuchi, H. Nishikawa, H. Matsukizono, S. Iino, T. Sugiyama, T. Ishioka and Y. Okumura, Adv. Sci., 2024, 11, 2409827 Search PubMed.
- J. Hobbs, C. J. Gibb and R. J. Mandle, Small Sci., 2024, 4, 2400189 Search PubMed.
- G. J. Strachan, S. J. Ramsay, M. Juodka, D. Pociecha, J. Szydlowska, J. Storey, N. Vaupotič, R. Walker, E. Gorecka, G. J. Strachan, D. Pociecha, J. Szydlowska, E. Gorecka, S. J. Ramsay, M. Juodka, J. M. Storey, R. Walker and N. Vaupotič, Angew. Chem., 2025, e202516302 Search PubMed.
- H. Nishikawa, Y. Okumura, D. Kwaria, A. Nihonyanagi and F. Araoka, Adv. Mater., 2025, 2501946 CrossRef CAS PubMed.
- Y. Song, X. Huang, X. Zhang, M. Deng, S. Aya, M. Huang, Y. Song, X. Huang, X. Zhang, M. Deng, S. Aya and M. Huang, Adv. Sci., 2025, 2414317 Search PubMed.
- E. Cruickshank, P. Rybak, M. M. Majewska, S. Ramsay, C. Wang, C. Zhu, R. Walker, J. M. D. Storey, C. T. Imrie, E. Gorecka and D. Pociecha, ACS Omega, 2023, 8, 36562–36568 CrossRef CAS PubMed.
- P. Nacke, A. Manabe, M. Klasen-Memmer, X. Chen, V. Martinez, G. Freychet, M. Zhernenkov, J. E. Maclennan, N. A. Clark, M. Bremer and F. Giesselmann, Sci. Rep., 2024, 14, 1–13 Search PubMed.
- X. Chen, V. Martinez, E. Korblova, G. Freychet, M. Zhernenkov, M. A. Glaser, C. Wang, C. Zhu, L. Radzihovsky, J. E. Maclennan, D. M. Walba and N. A. Clark, Proc. Natl. Acad. Sci. U. S. A., 2022, 120, e2217150120 CrossRef PubMed.
- M. Mrukiewicz, M. Czerwiński, N. Podoliak, D. Repček, P. Perkowski, R. J. Mandle and D. Węgłowska, J. Mater. Chem. C, 2024, 12, 7214–7224 Search PubMed.
- R. J. Mandle, Liq. Cryst., 2023, 50, 534–542 CrossRef CAS.
- C. J. Gibb, J. Hobbs, D. I. Nikolova, T. Raistrick, S. R. Berrow, A. Mertelj, N. Osterman, N. Sebastián, H. F. Gleeson and R. J. Mandle, Nat. Commun., 2024, 15, 5845 CrossRef CAS PubMed.
- D. Pociecha, J. Szydlowska, N. Vaupotič, K. Kwiatkowska, M. Juodka, J. Spiess, J. M. D. Storey, C. T. Imrie, R. Walker and E. Gorecka, Adv. Sci., 2025, e08405 Search PubMed.
- J. Karcz, J. Herman, N. Rychłowicz, P. Kula, E. Górecka, J. Szydlowska, P. W. Majewski and D. Pociecha, Science, 2024, 384, 1096–1099 CrossRef CAS PubMed.
- H. Nishikawa, D. Okada, D. Kwaria, A. Nihonyanagi, M. Kuwayama, M. Hoshino and F. Araoka, Adv. Sci., 2024, 11, 2405718 Search PubMed.
- E. Gorecka, M. Majewska, L. Fekete, J. Karcz, J. Żukowska, J. Herman, P. Kula and D. Pociecha, Mater. Horiz., 2025, 12, 5352–5356 RSC.
- J. Hobbs, C. J. Gibb and R. J. Mandle, Nat. Commun., 2025, 16, 7510 Search PubMed.
- Z. Raszewski, W. Piecek, L. Jaroszewicz, E. Nowinowski-Kruszelnicki, P. Perkowski, L. Soms, R. Dabrowski, J. Kedzierski, M. Olifierczuk, M. Mrukiewicz, E. Miszczyk, P. Morawiak, R. Mazur and K. Kowiorski, Adv. Mater. Res., 2014, 909, 12–18 Search PubMed.
- N. Bennis, T. Jankowski, O. Strzezysz, A. Pakua, D. C. Zografopoulos, P. Perkowski, J. M. Sánchez-Pena, J. M. López-Higuera and J. F. Algorri, Sci. Rep., 2022, 12, 012107 Search PubMed.
- M. Reuter, N. Vieweg, B. M. Fischer, M. Mikulicz, M. Koch, K. Garbat and R. D
![[a with combining cedilla]](https://www.rsc.org/images/entities/char_0061_0327.gif) browski, APL Mater., 2013, 1, 012107 CrossRef.
browski, APL Mater., 2013, 1, 012107 CrossRef. - M. Hird, A. J. Seed, K. J. Toyne, J. W. Goodby, G. W. Gray and D. G. McDonnell, J. Mater. Chem., 1993, 3, 851–859 RSC.
- Y. Arakawa, Y. Sasaki, N. Haraguchi, S. Itsuno and H. Tsuji, Liq. Cryst., 2018, 45, 821–830 CrossRef CAS.
- E. Cruickshank, G. J. Strachan, J. M. D. Storey and C. T. Imrie, J. Mol. Liq., 2021, 346, 117094 Search PubMed.
- Y. Arakawa, S. Inui and H. Tsuji, Tetrahedron, 2022, 122, 132958 Search PubMed.
- Y. Arakawa, Y. Ishida, T. Shiba, K. Igawa, S. Sasaki and H. Tsuji, CrystEngComm, 2022, 24, 1877–1890 Search PubMed.
- E. Cruickshank, R. Walker, G. J. Strachan, C. H. F. Goode, M. M. Majewska, D. Pociecha, E. Gorecka, J. M. D. Storey and C. T. Imrie, J. Mol. Liq., 2023, 391, 123226 Search PubMed.
- G. Stepanafas, E. Cruickshank, S. Brown, M. M. Majewska, D. Pociecha, E. Gorecka, J. M. D. Storey and C. T. Imrie, Mater. Adv., 2024, 5, 525–538 RSC.
- M. Alaasar, C. Anders, R. Pashameah and A. F. Darweesh, Liq. Cryst., 2023, 50, 2397–2412 CrossRef CAS.
- R. Saha, C. Feng, C. Welch, G. H. Mehl, J. Feng, C. Zhu, J. Gleeson, S. Sprunt and A. Jákli, Phys. Chem. Chem. Phys., 2021, 23, 4055–4063 RSC.
- E. Cruickshank, G. J. Strachan, M. M. Majewska, D. Pociecha, E. Gorecka, J. M. D. Storey and C. T. Imrie, J. Mater. Chem. C, 2025, 13, 20156–20168 RSC.
- E. Cruickshank, M. Salamończyk, D. Pociecha, G. J. Strachan, J. M. D. Storey, C. Wang, J. Feng, C. Zhu, E. Gorecka and C. T. Imrie, Liq. Cryst., 2019, 46, 1595–1609 Search PubMed.
- Y. Arakawa, K. Komatsu and H. Tsuji, New J. Chem., 2019, 43, 6786–6793 RSC.
- Y. Arakawa, Y. Arai, K. Horita, K. Komatsu and H. Tsuji, Crystals, 2022, 12, 1734 CrossRef CAS.
- Y. Arakawa, Y. Ishida, K. Komatsu, Y. Arai and H. Tsuji, Tetrahedron, 2021, 95, 132351 CrossRef CAS.
- Y. Arakawa and Y. Arai, Materials, 2024, 17, 3278 CrossRef CAS PubMed.
- E. Cruickshank, G. J. Strachan, A. Pearson, D. Pociecha, E. Gorecka, J. M. D. D. Storey and C. T. Imrie, Phys. Chem. Chem. Phys., 2025, 27, 6111–6121 Search PubMed.
- A. Seed, Chem. Soc. Rev., 2007, 36, 2046–2069 RSC.
- G. J. Cross, A. J. Seed, K. J. Toyne, J. W. Goodby, M. Hird and M. C. Artal, J. Mater. Chem., 2000, 10, 1555–1563 Search PubMed.
- R. J. Mandle, E. J. Davis, C. C. A. Voll, C. T. Archbold, J. W. Goodby and S. J. Cowling, Liq. Cryst., 2015, 42, 688–703 CAS.
- M. V. Ponomarenko, N. Kalinovich, Y. A. Serguchev, M. Bremer and G. V. Röschenthaler, J. Fluorine Chem., 2012, 135, 68–74 CrossRef CAS.
- J. A. Smith, R. A. DiStasio, N. A. Hannah, R. W. Winter, T. J. R. Weakley, G. L. Gard and S. B. Rananavare, J. Phys. Chem. B, 2004, 108, 19940–19948 CrossRef CAS.
- A. J. Seed, K. J. Toyne, J. W. Goodby and D. G. McDonnell, J. Mater. Chem., 1995, 5, 1–11 RSC.
- Z. Fang and C. Wu, Liq. Cryst., 2020, 47, 1086–1099 CrossRef CAS.
- A. U. Petersen, M. Jevric, R. J. Mandle, M. D. Kilde, F. P. Jørgensen, J. W. Goodby and M. B. Nielsen, Aust. J. Chem., 2018, 71, 422–434 Search PubMed.
- Y. Arakawa, Q. Ning, S. Karthick and S. Aya, J. Mater. Chem. C, 2024, 12, 16206–16217 Search PubMed.
- P. Guragain, A. Ghimire, M. Badu, N. P. Dhakal, P. Nepal, J. T. Gleeson, S. Sprunt, R. J. Twieg and A. Jákli, Mater. Horiz., 2025, 12, 8153–8164 Search PubMed.
- S. Brown, E. Cruickshank, J. M. D. Storey, C. T. Imrie, D. Pociecha, M. Majewska, A. Makal and E. Gorecka, ChemPhysChem, 2021, 22, 2506–2510 Search PubMed.
- E. Cruickshank, N. Tufaha, R. Walker, S. Brown, E. Gorecka, D. Pociecha, J. M. D. Storey and C. T. Imrie, Liq. Cryst., 2024, 51, 401–415 Search PubMed.
- N. Tufaha, E. Cruickshank, D. Pociecha, E. Gorecka, J. M. D. Storey and C. T. Imrie, Chem. – Eur. J., 2023, 29, e202300073 Search PubMed.
- E. Cruickshank, R. Walker, J. M. D. Storey and C. T. Imrie, RSC Adv., 2022, 12, 29482–29490 RSC.
- E. Cruickshank, R. Walker, M. M. Majewska, E. Gorecka, D. Pociecha, J. M. D. Storey and C. T. Imrie, ACS Omega, 2025, 10, 23609–23619 Search PubMed.
- E. Cruickshank, R. Walker, G. J. Strachan, E. Górecka, D. Pociecha, J. M. D. Storey and C. T. Imrie, J. Mater. Chem. C, 2025, 13, 3902–3916 Search PubMed.
- U. Jarek-Mikulska and Z. Galewski, Liq. Cryst., 2009, 36, 187–195 CrossRef CAS.
- N. V. Madhusudana, Phys. Rev. E, 2021, 104, 014704 Search PubMed.
- J. E. Fearon, G. W. Gray, A. D. Ifill and K. J. Toyne, Mol. Cryst. Liq. Cryst., 1985, 124, 89–103 Search PubMed.
- F. Neese, Wiley Interdiscip. Rev.:Comput. Mol. Sci., 2012, 2, 73–78 Search PubMed.
- F. Neese, F. Wennmohs, U. Becker and C. Riplinger, J. Chem. Phys., 2020, 152, Art. No. L224108 Search PubMed.
- F. Neese, Wiley Interdiscip. Rev.:Comput. Mol. Sci., 2022, 12, e1606 Search PubMed.
- F. Neese, Chem. Phys. Lett., 2000, 325, 93–98 Search PubMed.
- F. Neese, J. Comput. Chem., 2022, 1–16 Search PubMed.
- F. Neese, J. Comput. Chem., 2003, 24, 1740–1747 CrossRef CAS PubMed.
Footnotes |
| † Present Address: School of Pharmacy, Applied Sciences and Public Health, Robert Gordon University, Aberdeen, AB10 7GJ, UK. |
| ‡ Deceased January 2025. |
| § The molecular dipole moments were calculated using Orca 6.0.1,82–87 performed using the r2SCAN-D4 functional with the def2-TZVP basis set. |
| This journal is © The Royal Society of Chemistry 2026 |