 Open Access Article
Open Access ArticleNanocomposite electrodes with CuFe layered double hydroxides and hydrochar for alkaline HER and HOR: a multifunctional platform for hydrogen electrocatalysis
Michelle
Saliba
ab,
Philippe
Knauth
 *a,
Emily
Bloch
a,
Luca
Pasquini
a,
Emanuela
Sgreccia
b,
Riccardo
Narducci
*a,
Emily
Bloch
a,
Luca
Pasquini
a,
Emanuela
Sgreccia
b,
Riccardo
Narducci
 b,
Alessandra
Varone
b and
Maria Luisa
Di Vona
b,
Alessandra
Varone
b and
Maria Luisa
Di Vona
 *b
*b
aAix Marseille Univ, CNRS, MADIREL (UMR 7246) and International Laboratory: Ionomer Materials for Energy, Campus St Jérôme, 13013 Marseille, France. E-mail: philippe.knauth@univ-amu.fr
bUniversity of Rome Tor Vergata, Dep. Industrial Engineering and International Laboratory: Ionomer Materials for Energy, 00133 Roma, Italy. E-mail: divona@uniroma2.it
First published on 1st April 2026
Abstract
The hydrogen evolution reaction (HER) and hydrogen oxidation reaction (HOR) in alkaline media remain limited by slow kinetics and the reliance on noble metal catalysts. Copper-iron layered double hydroxides (CuFe-LDHs), composed of non-rare and low-toxicity elements, offer a sustainable alternative, though their performance is restricted by low conductivity and limited accessibility of active sites. In this work, CuFe-LDHs were synthesized hydrothermally and modified through three complementary strategies: ammonium fluoride (NH4F) as a structure-directing agent poly(2,6-dimethyl-1,4-phenylene oxide) functionalized with trimethylammonium (PPO-LC) as a hydroxide-conducting ionomer, and a nitrogen-doped hydrochar derived from pine needles as an electron-conducting additive. Structural tuning with 1.5% NH4F produced highly crystalline LDH-1.5 with increased exposure of catalytic centers, while PPO-LC enhanced OH− transport and electrode integrity. Hydrochar contributed additional conductive domains, facilitating charge transfer and improving electrode architecture. The materials were characterized using BET surface area analysis, scanning electron microscopy with EDS element mapping, X-ray diffraction, and Fourier-transform infrared spectroscopy. All catalytic electrodes demonstrated a high electrochemically active surface area. The LDH-1.5/PPO-LC electrode delivered the best HER activity with an overpotential of −332 mV at −10 mA cm−2. For the HOR, hydrochar-containing electrodes displayed reduced onset potential values: the LDH-1.5/PPO-LC/HC composite reached the lowest onset potential of 38 mV, though the current densities remained limited. Tafel analysis indicated that the HER is governed by the Volmer step associated with water dissociation, whereas the HOR follows a Heyrovsky–Volmer or Tafel–Volmer pathway depending on the electrode composition. These results highlight the effectiveness of combining structural, ionic, and electronic modifications to enhance the performance of CuFe-LDH-based bifunctional electrodes in alkaline environments, while indicating that further optimization is required to overcome intrinsic kinetic limitations.
Introduction
Hydrogen is considered as a key energy vector in the transition toward sustainable and low-carbon energy systems. Its high gravimetric energy density, storability over long timescales, and ability to be used across diverse applications, ranging from transportation and industrial decarbonization to large-scale energy storage, make it an attractive carrier for a low-carbon economy.1–3 Realizing this potential requires efficient and scalable routes for both hydrogen generation and utilization. Electrochemical technologies, such as water electrolysis and fuel cells, are among the most promising solutions; they rely on the hydrogen evolution reaction (HER) and hydrogen oxidation reaction (HOR), respectively. Alkaline systems offer several advantages over acidic environments, such as lower corrosion rates, reduced system costs, and compatibility with catalysts derived from earth-abundant elements. However, the kinetics of the HER and HOR under alkaline conditions are slow, with exchange current densities that can be up to two orders of magnitude lower than those achieved in acidic electrolytes.4–6 This kinetic barrier raises energy losses and reduces the efficiency of hydrogen conversion. Consequently, the design of electrocatalysts that are both efficient and composed of abundant, non-noble elements is a central challenge for the advancement of hydrogen-based technologies.Among the wide range of nanomaterials explored for clean energy conversion, layered double hydroxides (LDHs) have emerged as promising electrocatalysts for alkaline water splitting, owing to their tunable composition, hydroxide conductivity, high surface area, and favorable redox properties.7–13 Their general formula, [(M2+)1−x(M3+)x(OH)2]x+(Am−1)x/m·nH2O, describes a lamellar structure consisting of positively charged hydroxide layers with divalent (M2+) and trivalent (M3+) metal cations, whose charge is balanced by exchangeable interlayer anions. This brucite-derived architecture enables precise modulation of catalytic properties via targeted cation substitution. NiFe-LDH has long served as a benchmark system, due to the synergistic interaction between Ni2+ and Fe3+, which enhances adsorption of reaction intermediates and improves the catalytic performance.14 However, recent studies show that replacing Ni with Cu in LDH structures can further enhance catalytic activity. Furthermore, copper is generally considered “greener” than nickel due to its lower toxicity, wider abundance and lower cost, better recyclability, and moderate environmental impact when used in catalytic amounts. In LDH, Cu2+ contributes to electronic conductivity via its partially filled 3d orbitals, accelerating the Volmer step of the HER.15 Simultaneously, Fe3+ facilitates electron delocalization, ensuring efficient charge transfer throughout the catalytic cycle.15–17 Additionally, the presence of iron oxides, particularly Fe2O3, has been reported to enhance the hydrogen evolution kinetics by promoting water dissociation and optimizing hydrogen adsorption. This enhancement is attributed to strong electronic coupling with adjacent metal centers, which modulates the hydrogen binding free energy and lowers energy losses.18,19 Although CuFe-LDHs have mainly been explored for the HER, their redox flexibility, intrinsic hydroxide conductivity, stability in alkaline media, and structural adaptability also make them strong candidates for the HOR, pointing to their potential as bifunctional electrocatalysts.
Despite these advantages, CuFe-based LDHs suffer from inherent drawbacks such as low electronic conductivity, limited crystallinity, and restricted access to active sites. Strategies to overcome these issues have included coupling with conductive scaffolds (e.g. CuFe-LDH on Ni foam or NiFe-LDH on a CuxO support) and introducing ternary compositions (e.g. CoCuFe-LDH on graphene) to enhance charge transport and exploit redox synergies.20–22 More recently, attention has turned toward in situ structural engineering during synthesis using ammonium fluoride (NH4F).23 Acting as a mild etchant and coordinating agent, NH4F promotes the formation of ultrathin, porous, and defect-rich nanosheets, thereby increasing the surface area, enhancing the electrolyte accessibility, and exposing more catalytically active edge sites.23–25 Such morphological control has been shown to substantially improve electrochemical performance, as demonstrated for NiCo-LDHs that reach high specific capacitances while retaining stability over thousands of cycles.26
In this work, we investigated CuFe-LDH synthesized without a structure-directing agent and with two different concentrations of NH4F added during the synthesis (1.5% and 4.5%). In parallel with structural tuning, ionic transport across the catalyst layer remains a critical factor for overall efficiency. Incorporating hydroxide-ion conducting ionomers such as poly(2,6-dimethyl-1,4-phenylene oxide) functionalized with trimethylammonium (PPO-LC) provides a means to improve OH− conductivity. The long, flexible side chains of PPO-LC promote nanophase separation and hydrophilic network connectivity while also reinforcing mechanical integrity and adhesion, making it an effective additive for electrocatalytic systems.27,28 Furthermore, growing attention has been directed toward the integration of biomass-derived carbon materials, particularly biochar, to further improve the electronic conductivity and electrocatalytic performance. Hydrochar (HC), produced from pine needles via hydrothermal carbonization in the presence of urea, offers a nitrogen-doped porous carbon matrix with moderate conductivity and abundant functional groups (–OH, –COOH, and –NH2). These features improve the dispersion of active phases, facilitate charge transport, and enhance electrolyte penetration.29 HC has already shown promise in reactions such as oxygen and carbon dioxide reduction, where hydrochar-based electrodes displayed favorable onset potentials and supported efficient multi-electron transfer processes.30 Incorporating HC is a promising approach to improve electron mobility, stabilize the dispersion, and optimize the interfacial architecture within the catalytic layer, but its use in CuFe-LDH systems for the HER and HOR has not been explored.
The novelty of this work lies in the preparation of CuFe-LDH-based electrodes via a hydrothermal route, systematically combining NH4F for structural optimization, PPO-LC for hydroxide conduction, and pine needle-derived hydrochar (HC) for electronic conduction and porous support. This design enables the evaluation of not only the individual effects of each additive but also their potential synergistic contributions to governing HER and HOR activity under alkaline conditions. Through this approach, the study aims to identify key structure–property relationships that can guide the development of efficient, low-cost, and sustainable electrocatalysts for hydrogen technologies.
Materials and methods
Materials
Semi-dried pine needles were collected from the vicinity of the University of Perugia, Italy. Urea (99%, carbamide, CO(NH2)2, Merck KGaA, Darmstadt, Germany) was used as the nitrogen precursor. All other reagents were used as received from Sigma-Aldrich, including copper(II) nitrate trihydrate (Cu(NO3)2·3H2O, 99%), iron(III) nitrate nonahydrate (Fe(NO3)3·9H2O, > 98%), n-butyllithium (BuLi, 2.5 M in hexane), trimethylamine (TMA, 31–35 wt% in ethanol, 4.2 M), and poly(2,6-dimethyl-1,4-phenylene oxide) (PPO, MW = 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1). Carbon paper (AvCarb EP55) was purchased from the Fuel Cell Store. Potassium hydroxide (KOH, 85%) used for electrochemical measurements was supplied by LabCAM.
000 g mol−1). Carbon paper (AvCarb EP55) was purchased from the Fuel Cell Store. Potassium hydroxide (KOH, 85%) used for electrochemical measurements was supplied by LabCAM.
Synthesis of CuFe-LDH
CuFe-LDH was synthesized by a hydrothermal method using Cu(NO3)23H2O, Fe(NO3)3·9H2O, and urea as precursors. Aqueous solutions of Cu2+ (0.25 M) and Fe3+ (0.125 M) were mixed at a Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe molar ratio of 2
Fe molar ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and stirred for 20 min. Different amounts of NH4F (0, 1.5 mol%, or 4.5 mol% with respect to Cu2+) were then introduced, followed by the addition of urea (molar ratio urea: Fe = 10
1 and stirred for 20 min. Different amounts of NH4F (0, 1.5 mol%, or 4.5 mol% with respect to Cu2+) were then introduced, followed by the addition of urea (molar ratio urea: Fe = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). After stirring for 1 h, the suspension was transferred to a Teflon-lined autoclave and heated at 90 °C for 24 h. The precipitate was collected by centrifugation, washed with deionized water, and dried at 60 °C for 3 days.
1). After stirring for 1 h, the suspension was transferred to a Teflon-lined autoclave and heated at 90 °C for 24 h. The precipitate was collected by centrifugation, washed with deionized water, and dried at 60 °C for 3 days.
Synthesis of hydrochar
Hydrochar was prepared as reported in ref. 31. In brief, 2 g of untreated pine needle waste was hydrothermally treated in 20 mL of deionized water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 solid-to-liquid ratio) at 230 °C for 1 h. The product was filtered, washed with hot water until colorless, and dried under vacuum at 105 °C. The dried hydrochar was then mixed with melamine (1
10 solid-to-liquid ratio) at 230 °C for 1 h. The product was filtered, washed with hot water until colorless, and dried under vacuum at 105 °C. The dried hydrochar was then mixed with melamine (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 wt ratio) and pyrolyzed at 800 °C for 1 h under an inert atmosphere to obtain N-doped hydrochar, which was stored in an airtight container until use. Elemental composition (wt%): C 75.2, N 10.5, H 1.0, O 4.1.
1 wt ratio) and pyrolyzed at 800 °C for 1 h under an inert atmosphere to obtain N-doped hydrochar, which was stored in an airtight container until use. Elemental composition (wt%): C 75.2, N 10.5, H 1.0, O 4.1.
Synthesis of CuFe-LDH supported on hydrochar (CuFe-LDH/HC)
CuFe-LDH/HC was synthesized under the same conditions in the presence of hydrochar. Hydrochar (15 wt% relative to the total salt mass) was dispersed in deionized water by ultrasonication for 20 min prior to metal salt addition. Cu2+ and Fe3+ were introduced at a Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe molar ratio of 2
Fe molar ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (0.25 M and 0.125 M, respectively), followed by NH4F (1.5 mol% relative to Cu2+) and, after 10 min, urea (urea:Fe = 10
1 (0.25 M and 0.125 M, respectively), followed by NH4F (1.5 mol% relative to Cu2+) and, after 10 min, urea (urea:Fe = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The mixture was stirred for 1 h, hydrothermally treated at 90 °C for 24 h, centrifuged, washed, and dried at 60 °C for 3 days.
1). The mixture was stirred for 1 h, hydrothermally treated at 90 °C for 24 h, centrifuged, washed, and dried at 60 °C for 3 days.
Synthesis of PPO-LC
The synthesis of PPO-LC was performed as described in ref. 32. Briefly, PPO (41.7 meq) was dissolved in THF (500 mL) and reacted with BuLi (62.6 mmol, 2.5 M in hexane) at 55 °C for 3 h. The reaction mixture was then cooled to −70 °C, followed by the addition of 1,4-dibromobutane (125 mmol). The mixture was stirred overnight at room temperature. The product was precipitated and washed with ethanol. Quaternization was carried out via nucleophilic substitution in N-methylpyrrolidone (NMP) using excess TMA. After heating at 80 °C for 72 h, the polymer was precipitated in water, filtered, washed, and vacuum-dried at 60 °C. The obtained ionomer, PPO-LC, exhibited a degree of amination of 0.38 and an IEC of 0.99 meq g−1.Catalyst ink and electrode preparation
Catalyst inks were prepared by dispersing 40 mg of hydrothermally obtained powder (LDH, LDH-NH4F, or LDH-NH4F-HC), with or without 10 mg PPO-LC, in dimethyl sulfoxide (DMSO). The suspensions were stirred at room temperature and sonicated overnight. A 5 µL aliquot of ink was drop-cast onto acid-treated carbon paper (pretreated according to ref. 33) and dried under vacuum at 40 °C for 4 h. The final catalyst loading was (0.23 ± 0.03) mg per electrode. The electrodes were fixed to glassy carbon disks with 6 mm diameter for electrochemical testing. The electrode compositions are reported in Table 1.| Electrode code | NH4F | Ionomer | Hydrochar (HC) |
|---|---|---|---|
| LDH/PPO-LC | — | PPO-LC | — |
| LDH-1.5/PPO-LC | 1.5 | PPO-LC | — |
| LDH-4.5/PPO-LC | 4.5 | PPO-LC | — |
| LDH/HC | — | — | 230_1800_1 |
| LDH-1.5/HC | 1.5 | — | 230_1800_1 |
| LDH-1.5/PPO-LC/HC | 1.5 | PPO-LC | 230_1800_1 |
Characterization
The electrode morphology was investigated by scanning electron microscopy (ZEISS Gemini SEM 500), coupled with an EDAX Octane Silicon Drift Detector for elemental mapping (EDS).XRD patterns were collected using Cu-Kα radiation (λ = 0.1508 nm), in step-scanning mode with a 2θ step of 0.05° and counting time per step of 5 s. Phase identification was performed by comparing the experimental pattern with the JCPDS-ICDD database.
Fourier Transform Infrared (FTIR) spectra were obtained using a PerkinElmer Spectrum 2 IR spectrometer equipped with an ATR ZnSe crystal.
The N2 adsorption–desorption isotherms were measured at 77 K on a BelSorp MAX from BelJapan. The specific surface area was determined by the Brunauer, Emmett and Teller (BET) method and the pore volume using the t-plot.34 Prior to adsorption, the samples were outgassed at 250 °C overnight under vacuum using the BelPrep apparatus.
Electrochemical measurements
Electrochemical measurements were performed in a conventional H-cell, using a three-electrode configuration. The setup consisted of a rotating disk electrode (RDE, 0.28 cm2, OrigaTrod, OrigaLys) as the working electrode, platinum foil (4 cm2) as the counter electrode, and an Ag/AgCl reference electrode positioned close to the working electrode. All experiments were carried out in 1 M KOH at room temperature for both the HER and the HOR. A BioLogic VMP3 potentiostat was employed to perform cyclic voltammetry (CV), linear sweep voltammetry (LSV), chronopotentiometry (CP), and electrochemical impedance spectroscopy (EIS). CV curves were recorded at scan rates ranging from 20 to 100 mV s−1, while EIS spectra were collected in the frequency range of 1 MHz to 1 Hz with a perturbation amplitude of 20 mV. LSV measurements were conducted at a scan rate of 5 mV s−1 with electrode rotation at 1000 rpm to remove hydrogen bubbles during the HER and to enhance mass transport during the HOR. All measured potentials were converted to the reversible hydrogen electrode (RHE) scale using the equation: ERHE/V = EAg/AgCl + 0.059 pH + 0.197 and ohmic drop correction was applied.Results and discussion
CuFe-LDH was synthesized hydrothermally using Cu(NO3)2, Fe(NO3)3, and urea. In the absence of NH4F, the slow release of OH− from urea hydrolysis promoted LDH precipitation with carbonate anions from solution or atmospheric CO2 occupying the interlayer. However, the resulting material displayed relatively broad and low intensity diffraction peaks, suggesting poor crystallinity and possible coexistence of amorphous hydroxide phases. When NH4F was introduced as an additional mineralizer, fluoride ions could interact with Cu2+ and Fe3+ during nucleation, modifying the growth mechanism. The mechanism of F− regulation is illustrated in ref. 35. At a molar ratio of NH4F: Cu = 0.015 (LDH-1.5), the XRD pattern (Fig. 1a) exhibited sharper and more intense reflections, indicating improved crystallinity. This enhancement can be attributed to the ability of F− to stabilize metal hydroxide layers and regulate crystal growth, leading to larger and more ordered LDH domains. In contrast, at higher NH4F content (NH4F![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu = 0.045, LDH-4.5), less intense diffraction peaks and reduced phase purity were observed. The XRD pattern of LDH-1.5 confirms the formation of the LDH phase. The characteristic LDH reflections corresponding to the (003), (006), (012), and (015) planes are observed at 11.9°, 24.2°, 32.3°, and 37.1°, respectively. Additional diffraction peaks are detected and attributed to Fe2O3 (ICDD PDF 40-1139) and FeOOH (ICDD PDF 08-0098), indicating the presence of minor secondary phases.
Cu = 0.045, LDH-4.5), less intense diffraction peaks and reduced phase purity were observed. The XRD pattern of LDH-1.5 confirms the formation of the LDH phase. The characteristic LDH reflections corresponding to the (003), (006), (012), and (015) planes are observed at 11.9°, 24.2°, 32.3°, and 37.1°, respectively. Additional diffraction peaks are detected and attributed to Fe2O3 (ICDD PDF 40-1139) and FeOOH (ICDD PDF 08-0098), indicating the presence of minor secondary phases.
As shown in Fig. 1a, the experimental diffraction pattern is compared with the standard pattern of LDH (ICDD PDF card No. 41-1428) and the positions of the main reflections are in good agreement with the reference one, confirming the formation of the LDH structure. A slight shift of the experimental diffraction peaks is observed, which may indicate variations according to Bragg's law; the lattice parameters of the LDH structure were calculated and the values a = 0.33 nm and c = 2.22 nm are consistent with those reported for Fe–Cu LDH systems.24
To further investigate the chemical structure and functional groups of the composite material, FTIR analysis was performed. The FTIR spectrum of CuFe-LDH grown on hydrochar (CuFe-LDH/HC) is compared with that of pristine hydrochar in Fig. 1b. A broad absorption band centred around 3400 cm−1 is especially prominent in the CuFe-LDH/HC sample, and is attributed to O–H stretching vibrations from both interlayer water and hydroxide ions within the LDH layers. The corresponding H–O–H bending vibration of interlayer water appears at 1670 cm−1, where it overlaps with the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching bands from carboxyl or carbonyl groups in the hydrochar matrix.36 A sharp peak near 3770 cm−1 is observed and can be assigned to isolated O–H stretching vibrations, which may arise from non-hydrogen-bonded terminal OH groups on LDH crystallites, or from coexisting phases such as Fe2O3. In the lower wavenumber region, bands at 535 cm−1 and 678 cm−1 correspond to Cu–O and Fe–O vibrational modes, respectively.36,37 A feature at 942 cm−1 may be attributed to M–O–H bending vibrations, consistent with the layered hydroxide structure.24 Notably, the CuFe-LDH/HC spectrum exhibits a strong absorption band at about 1500 cm−1 and a weaker one near 860 cm−1, both characteristic of the carbonate anion (CO32−). This confirms that carbonate is the predominant interlayer anion, likely incorporated from atmospheric CO2 during synthesis, which employed urea hydrolysis under controlled pH conditions. Additionally, a distinct band at 1265 cm−1 is observed, which may result from interactions between the LDH layers and the aromatic moieties of the hydrochar surface.
O stretching bands from carboxyl or carbonyl groups in the hydrochar matrix.36 A sharp peak near 3770 cm−1 is observed and can be assigned to isolated O–H stretching vibrations, which may arise from non-hydrogen-bonded terminal OH groups on LDH crystallites, or from coexisting phases such as Fe2O3. In the lower wavenumber region, bands at 535 cm−1 and 678 cm−1 correspond to Cu–O and Fe–O vibrational modes, respectively.36,37 A feature at 942 cm−1 may be attributed to M–O–H bending vibrations, consistent with the layered hydroxide structure.24 Notably, the CuFe-LDH/HC spectrum exhibits a strong absorption band at about 1500 cm−1 and a weaker one near 860 cm−1, both characteristic of the carbonate anion (CO32−). This confirms that carbonate is the predominant interlayer anion, likely incorporated from atmospheric CO2 during synthesis, which employed urea hydrolysis under controlled pH conditions. Additionally, a distinct band at 1265 cm−1 is observed, which may result from interactions between the LDH layers and the aromatic moieties of the hydrochar surface.
To complement the structural characterization, the textural properties of the carbon support were investigated. The adsorption/desorption isotherm of hydrochar (Fig. 1c) shows clearly the existence of micropores (type I isotherm). The BET surface area is 353 m2 g−1 and the pore volume is 0.14 cm3 g−1. A high temperature heat treatment of hydrochars is known to increase significantly their surface area.38
Representative SEM images (Fig. 2) of (a) LDH-1.5/PPO-LC and (b) LDH-1.5/PPO-LC/HC on carbon paper show fibers uniformly coated with thin LDH/PPO-LC films. In sample (a), the coating appears smoother with smaller porous domains and only a few larger openings. With hydrochar (b), the structure becomes more heterogeneous, showing both small and larger interconnected pores. The addition of HC therefore increases surface heterogeneity and pore variety while preserving the fiber scaffold. The spatial distribution of elements in the LDH-1.5/PPO-LC electrode was further examined by EDS mapping on a high-resolution SEM image (Fig. 2c). The elemental maps of Fe and Cu revealed a uniform distribution of both metals across the surface, while a zoomed-in view of the brightest regions (Fig. 2d) highlighted LDH domains, confirming the successful formation of the CuFe-LDH phase (Fig. 2e and f). The higher Fe intensity is attributed to the presence of iron oxides, as confirmed by XRD. Such homogeneous dispersion of active components within the three-dimensional electrode framework is expected to increase the density of accessible active sites, thereby facilitating the adsorption–desorption processes during the HER and HOR.39 The corresponding EDS results for LDH-1.5/PPO-LC/HC show similar elemental distributions.
The electrochemically active surface area (ECSA) is a fundamental parameter that reflects the density of catalytically accessible sites. To obtain the ECSA, the double-layer capacitance (Cdl) must first be determined, and in this work, it was evaluated using two complementary electrochemical methods. The first approach was electrochemical impedance spectroscopy (EIS). Nyquist plots (Fig. 3a) were fitted using the equivalent circuit R1−(R2‖CPE2)−CPE3, where R1 represents the electrolyte–electrode resistance, R2 the charge-transfer resistance, CPE2 accounts for interfacial processes, and CPE3 corresponds to the electrode capacitance. The intercept on the real axis yielded similar R1values for all electrodes (average 6.8 Ω; Table 2), indicating comparable ohmic resistance. The impedance of a constant phase element is given by the following relation:
| Z = [Q(iω)n]−1 | (1) |
| Electrode | C dl (mF) | ESCA (cm2) | R 1 (Ω) | R 2 (Ω) | Q 2 (mF sn−1) | n 2 | Q 3 (mF sn−1) | n 3 | Overpotential (mV)/HER @ −10 mA cm−2 | E onset (mV)/HOR |
|---|---|---|---|---|---|---|---|---|---|---|
| LDH/PPO-LC | 0.9 | 30 | 6 | 1.4 | — | — | 1.1 | 0.7 | −592 | — |
| LDH-1.5/PPO-LC | 2.4 | 80 | 7.7 | 1.7 | 19 | 0.45 | 2.9 | 0.91 | −332 | 80 |
| LDH-4.5/PPO-LC | 1.7 | 57 | 7.7 | 1.4 | 16.5 | 0.58 | 1.8 | 0.94 | −513 | — |
| LDH/HC | 1.9 | 63 | 6.6 | 1.1 | 56 | 0.46 | 2.4 | 0.97 | −407 | 70 |
| LDH-1.5/HC | 2.8 | 93 | 6 | 1.2 | 3.6 | 0.53 | 3.3 | 0.93 | −403 | 60 |
| LDH-1.5/PPO-LC/HC | 3.2 | 107 | 6.9 | 1.7 | 0.3 | 0.71 | 3.4 | 0.93 | −381 | 38 |
A second, independent determination of Cdl was obtained from cyclic voltammetry (CV) performed in the non-faradaic region, where the voltammograms exhibit the characteristic rectangular shape of double-layer charging (Fig. 3b). The capacitive current (Δi/2) was plotted as a function of scan rate (Fig. 3c), and the slope of the linear fit yielded Cdl according to the following relation:
 | (2) |
The Cdl values obtained from CV showed excellent agreement with those extracted from EIS (Cdl ≈ CPE3), confirming the reliability of the capacitance determination.
The ECSA was calculated using the following equation:
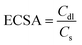 | (3) |
A clear trend was observed in the calculated ECSA values among the different electrodes. The LDH/PPO-LC electrode showed the lowest ECSA of 30 cm2, consistent with a compact structure due to polymer blocking of active sites, as previously reported.43–45 Incorporation of highly crystalline LDH-1.5 increased the ECSA to 80 cm2, demonstrating the role of fluoride as a structure-directing agent that etches layers, generates edge sites, and achieves a favorable balance between defect creation and structural stability. Similar behavior has been reported for NiFe- and CoFe-LDHs, where fluoride facilitated the formation of ultrathin nanosheets with enhanced defect density and edge exposure, thereby boosting ECSA and catalytic activity.23,46 However, increasing the NH4F concentration to 4.5% during synthesis resulted in a lower ECSA of 57 cm2, reflecting the detrimental effect of excessive fluoride, which induces lattice disorder.47 Hydrochar provided an additional enhancement: when incorporated alone (LDH/Hydrochar), it increased the ESCA to 63 cm2. Owing to its sp2-carbon network and intrinsic porous structure, hydrochar forms a conductive framework that improves electron transport and electrolyte accessibility.48–50 The LDH-1.5/PPO-LC/HC composite achieved the highest ESCA (107 cm2), reflecting the synergistic interplay between NH4F-induced defect generation and hydrochar. This combination maximized the density of accessible sites and improved charge-storage capacity, consistent with reports showing that LDHs modified by anion exchange, fluoride-assisted synthesis, or integration with biomass-derived carbons exhibit enhanced capacitances and faster kinetics compared to their unmodified counterparts26,51,52.
The catalytic activity toward the hydrogen evolution reaction (HER) was subsequently evaluated by linear sweep voltammetry. The HER polarization curves (Fig. 3d) confirmed the trends inferred from the capacitance analysis. The LDH/PPO-LC electrode exhibited the lowest catalytic activity, requiring −592 mV to reach −10 mA cm−2. In contrast, LDH-1.5/PPO-LC achieved a significantly lower overpotential of −332 mV, while LDH-4.5/PPO-LC gave an intermediate value (−513 mV), in line with the structural disorder discussed in the ECSA section. These results highlight the importance of optimizing the NH4F concentration and demonstrate that HER performance is governed by surface structuring and ordering of LDH: when optimized, NH4F expands the accessible area and facilitates charge/mass transport, whereas when excessive or poorly controlled, it induces lattice disorder and reduces activity.
Electrodes containing hydrochar displayed intermediate activity, with LDH/HC and LDH-1.5/HC requiring overpotentials of −407 and −403 mV, respectively. This suggests that hydrochar plays a non-negligible role by providing electronic conductivity and facilitating electron transfer. However, in systems with HC but without PPO-LC, ionic conduction occurs mainly through OH− transport within the LDH layers, which is less efficient than ionomer-assisted conduction. Consequently, the best performance was obtained when highly crystalline LDH was combined with the ionomer binder: LDH-1.5/PPO-LC reached the lowest overpotential (−332 mV), followed closely by LDH-1.5/PPO-LC/HC (−381 mV).
Some LDH-based catalysts reported in the literature exhibit lower overpotentials, as summarized in Table 3. However, these performances were obtained on highly conductive and catalytically active supports such as Ni foam or by introducing less common or even toxic metals such as Co. In contrast, our electrodes were prepared on carbon paper via a simple hydrothermal process, relying on the intrinsic catalytic properties of CuFe-LDH without additional metals. Overall, these results demonstrate that a sustainable strategy, combining optimized crystallinity with ion and electron conducting pathways, can deliver competitive HER activity without relying on scarce or environmentally harmful elements. This approach provides a viable direction for designing eco-friendly electrocatalysts that balance performance and sustainability; however, further optimization is required to fully close the gap with state-of-the-art catalysts.
To further evaluate the intrinsic catalytic efficiency of the electrodes, the hydrogen generation rate (rH2) and the mass activity (MA) were calculated. The hydrogen production rate was determined according to the following equation:
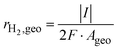 | (4) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485 C mol−1), and Ageois the geometric electrode area (0.28 cm2). The factor of 2 accounts for the two electrons required for the formation of one molecule of H2. A faradaic efficiency of 100% was assumed for the calculation.
485 C mol−1), and Ageois the geometric electrode area (0.28 cm2). The factor of 2 accounts for the two electrons required for the formation of one molecule of H2. A faradaic efficiency of 100% was assumed for the calculation.
The mass activity was calculated as follows:
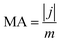 | (5) |
To gain further insight into the reaction kinetics governing the HER process, the Tafel slopes were extracted from the LSV curves (Fig. 3e). The electrodes with low crystallinity LDH exhibit higher Tafel slopes, whereas the incorporation of LDH-1.5 and HC decreases the slope through improved charge transfer. Despite these improvements, all slopes remain above 120 mV dec−1, highlighting the intrinsic kinetic limitations of LDH catalysts in alkaline HER. In alkaline electrolytes, the HER typically proceeds through an initial Volmer step involving water dissociation, generally associated with a Tafel slope of ∼120 mV dec−1:
| H2O + e− + * → H* + OH− | (6) |
This step can be followed by either the Tafel recombination step (characterized by a Tafel slope of ∼30 mV dec−1):
| H* + H* → H2 | (7) |
| H2O + e− + H* → H2 + OH− | (8) |
In the present work, the Tafel slopes (Fig. 3e) indicate that the rate-determining step (RDS) of the HER is the Volmer reaction, which involves water dissociation. This behavior is attributed to the intrinsic kinetic barrier associated with H–OH bond cleavage and is consistent with previous reports on HER kinetics in alkaline media8,57,58.
Finally, chronopotentiometric tests (Fig. 3f) demonstrated good stability for all four active electrodes, including the hydrochar-containing variant. A decrease in overpotential after 12 h at −10 mA cm−2 was observed for electrodes containing LDH-1.5, consistent with a better availability of active sites, whereas the overpotential increased for the less crystalline LDH samples, indicating their lower stability in alkaline medium.
Linear sweep voltammograms for the HOR are presented in Fig. 5a. The onset potential (Eonset, Table 2) decreased from 80 mV for LDH-1.5/PPO-LC to 70 mV with LDH/HC, 60 mV with LDH-1.5/HC, and 38 mV for the LDH-1.5/PPO-LC/HC electrode. In contrast, electrodes containing poorly crystalline LDH and no HC (LDH/PPO-LC and LDH-4.5/PPO-LC) showed negligible HOR activity. Importantly, hydrochar-containing electrodes exhibited the lowest onset potentials, suggesting the direct involvement of HC in the catalytic process. The very low value of 38 mV obtained with the optimized formulation reflects the synergistic effects of NH4F improving crystallinity, HC providing supplementary catalytic sites and an electronically conductive framework, and the PPO-LC ionomer enhancing hydroxide transport. These combined factors increase the electrocatalytic activity at the electrode–electrolyte interface, enabling the HOR to initiate more easily.
 | ||
| Fig. 5 HOR activity of electrodes with different compositions in 1 M KOH: (a) linear sweep voltammograms (5 mV s−1, 1000 rpm) recorded in H2-saturated electrolyte and (b) corresponding Tafel slopes. | ||
However, although hydrochar lowers the onset potential, the corresponding current densities are smaller than those of the electrodes without it, suggesting that its presence partially blocks some hydrogen diffusion pathways. In all cases, the current density did not exceed 1 mA cm−2, with the highest value (∼0.6 mA cm−2) achieved by LDH-1.5/PPO-LC. The main limitation is related to mass-transport effects in alkaline media, where hydrogen solubility and diffusion are low, and even the most active catalysts typically reach only a few mA cm−2 under rotating-disk conditions.59–63 This underscores the need for further electrode engineering to improve hydrogen delivery and utilization in order to translate the improved onset potentials into higher practical HOR currents in alkaline fuel cells.
The Tafel slope analysis for the HOR (Fig. 5b) further supports the LSV results. The four best-performing electrodes, particularly those incorporating hydrochar (HC), exhibit faster kinetics for the HOR than for the HER. LDH-1.5/PPO-LC and LDH/HC show Tafel slopes of approximately 50 mV dec−1, values typically associated with a Heyrovsky–Volmer regime in which the RDS involves the reaction between adsorbed hydrogen species and OH−. When the highly crystalline LDH-1.5 phase is combined with HC, the slope decreases markedly to 26 mV dec−1 for LDH-1.5/PPO-LC/HC, a value among the lowest reported for non-noble metal HOR catalysts.64 According to established microkinetic analyses of the reversible hydrogen reaction,56,57 slopes close to ∼30 mV dec−1 are generally attributed to a Tafel–Volmer pathway, indicating that the dissociation of adsorbed hydrogen gas becomes the rate-determining step.
If we compare the HER and HOR results, the role of hydrochar differs between these two reactions. Although the incorporation of HC improves the HOR onset potential and kinetics and increases the capacitive response, this effect does not translate into improved HER activity. This difference arises from the distinct rate-determining steps of the two reactions. The HOR mainly requires efficient hydrogen oxidation and rapid electron transfer from the catalytic sites to the electrode, which is facilitated by the conductive carbon framework of hydrochar. In contrast, the HER in alkaline media is limited by the water dissociation step, which requires highly active catalytic sites capable of breaking the O–H bond. Since hydrochar does not significantly promote this step and may partially cover the active LDH surface, its contribution to the HER remains limited. Consequently, hydrochar mainly improves electrode conductivity and structure, favoring HOR kinetics, while HER activity remains largely governed by the intrinsic LDH catalytic sites.
Conclusions
Layered Double Hydroxides (LDHs) are versatile materials for a wide range of applications, and CuFe-LDH, based on abundant and non-toxic elements, represents an appealing alternative to LDHs containing less benign and more rare metals, such as Co or Ni. In this study, we reported the preparation of six different CuFe-LDH-based electrodes and demonstrated that their electrocatalytic response is strongly governed by composition and structural features. The presence of an optimal concentration of 1.5% NH4F as a structure-directing agent during hydrothermal synthesis enhanced crystallinity and increased the electrochemically active surface area (ECSA), while hydrochar boosted the ECSA through conductive networks and hierarchical porosity. The incorporation of a long-side-chain ionomer (PPO-LC) further improved the electrocatalytic performance.The most crystalline ionomer-containing electrode (LDH-1.5/PPO-LC) delivered a HER current of −10 mA cm−2 at an overpotential of −332 mV and retained excellent stability, whereas its hydrochar analogue achieved comparable activity. In contrast, less crystalline LDH samples showed consistently lower performance.
For the HOR, LDH-1.5/PPO-LC reached the highest current density (0.6 mA cm−2), while all hydrochar-containing electrodes exhibited lower onset potentials, with the most significant reduction to 38 mV observed for LDH-1.5/PPO-LC/HC, highlighting the role of hydrochar in improving electrocatalytic activity for hydrogen oxidation.
These results are consistent with the kinetic insights derived from the Tafel analysis. For the HER, the large slopes (>120 mV dec−1) indicate that the reaction is limited by the Volmer step involving water dissociation. In contrast, the Tafel slopes for the HOR are consistent with a Heyrovsky–Volmer pathway for LDH-1.5/PPO-LC and LDH/HC, while the lower slope (∼26 mV dec−1) observed for LDH-1.5/PPO-LC/HC suggests a shift toward a Tafel–Volmer mechanism, reflecting faster hydrogen adsorption and improved HOR kinetics.
Overall, further optimization is required to overcome the intrinsic kinetic limitations of the HER, especially the water dissociation step, and the mass-transport barriers for the HOR, thereby enabling the development of more efficient bifunctional CuFe-LDH-based electrocatalysts for alkaline hydrogen technologies via a sustainable, scalable, and cost-effective route.
Conflicts of interest
There are no conflicts to declare.Data availability
The supporting data have been provided as figures and tables in the main manuscript and as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6se00103c.Acknowledgements
M. S., P. K. and L. P. gratefully acknowledge support from the French government under the France 2030 investment plan, as part of the Initiative d’Excellence d’Aix-Marseille Université – A*MIDEX project “ION-MEET”, AMX-22-IN1-41.References
- J. Gómez and R. Castro, Green Hydrogen Energy Systems: A Review on Their Contribution to a Renewable Energy System, Energies, 2024, 17(13), 3110 CrossRef.
- M. G. Rasul, M. A. Hazrat, M. A. Sattar, M. I. Jahirul and M. J. Shearer, The Future of Hydrogen: Challenges on Production, Storage and Applications, Energy Convers. Manage., 2022, 272, 116326 CrossRef CAS.
- T. Terlouw, L. Rosa, C. Bauer and R. McKenna, Future Hydrogen Economies Imply Environmental Trade-Offs and a Supply-Demand Mismatch, Nat. Commun., 2024, 15(1), 7043 CrossRef CAS PubMed.
- X. Mu, S. Liu, L. Chen and S. Mu, Alkaline Hydrogen Oxidation Reaction Catalysts: Insight into Catalytic Mechanisms, Classification, Activity Regulation and Challenges, Small Struct., 2023, 4(4), 2200281, DOI:10.1002/sstr.202200281.
- M. D. Woodroof, J. A. Wittkopf, S. Gu and Y. S. Yan, Exchange Current Density of the Hydrogen Oxidation Reaction on Pt/C in Polymer Solid Base Electrolyte, Electrochem. Commun., 2015, 61, 57–60 CrossRef CAS.
- J. Zheng, W. Sheng, Z. Zhuang, B. Xu and Y. Yan, Universal Dependence of Hydrogen Oxidation and Evolution Reaction Activity of Platinum-Group Metals on pH and Hydrogen Binding Energy, Sci. Adv., 2016, 2(3), e1501602, DOI:10.1126/sciadv.1501602.
- S. Nagappan, S. Yang, A. Adhikari, R. Patel and S. Kundu, A Review on Consequences of Flexible Layered Double Hydroxide-Based Electrodes: Fabrication and Water Splitting Application, Sustain. Energy Fuels, 2023, 7(16), 3741–3775 RSC.
- A. M. Tucker-Quiñónez, B. F. Rivadeneira-Mendoza, M. L. Gorozabel-Mendoza, I. B. Pérez-Almeida, A. J. García-Guerrero, A. A. Dueñas-Rivadeneira, K. K. Yadav, L. A. Zambrano-Intriago and J. M. Rodríguez-Díaz, Challenges and Potential of Layered Double Hydroxides as Electrocatalytic Materials for Hydrogen Production from Water: A Review of Recent Advances and Applications, Energy Nexus, 2025, 100399 CrossRef.
- J. Chen, Y. Wu, H. Zheng, M. Chen, H. Sun, T. Zhou, G. Na, D. Li, Q. Lu and B. Zi, Facile Room-Temperature Synthesis of Pt/NiCo LDH for Enhanced Hydrogen Evolution Reaction, Int. J. Hydrogen Energy, 2024, 72, 41–48 CrossRef CAS.
- Y. Zhang, Z. Zhang, X. Zhang, J. Li and R. Guo, Research Progress on NiCo-LDH Electrocatalysts for Oxygen Evolution Reaction, Int. J. Hydrogen Energy, 2025, 102, 304–320 CrossRef CAS.
- M. Chettri, N. Diyali and B. Biswas, Tracking in situ transformation of metal–organic frameworks into layered double hydroxides during synthesis and alkaline water oxidation through operando mechanistic studies, Dalton Trans., 2026, 55(4), 1548–1583 RSC.
- M. Tayebi, et al., Production of H2 and glucaric acid using electrocatalyst glucose oxidation by the Ta NiFe LDH electrode, ACS Appl. Mater. Interfaces, 2024, 16(20), 26107–26120 CrossRef CAS PubMed.
- Y. Shajirati, et al., Facile synthesis of interlaced flower-like layered double hydroxides grown on porous CoMoP as a highly efficient electrocatalyst for hydrogen evolution reaction, Energy, 2023, 278, 127840 CrossRef CAS.
- R. Xu, X. Wang, Z. Yang, Y. Chang, X. Chen, J. Wang and H. Li, Electrodeposition Fabrication of La-Doped NiFe Layered Double Hydroxide to Improve Conductivity for Efficient Overall Water Splitting, ACS Appl. Energy Mater., 2024, 7(9), 3866–3875, DOI:10.1021/acsaem.4c00246.
- M. Bhavanari, K.-R. Lee, C.-J. Tseng, I.-H. Tang and H.-H. Chen, CuFe Electrocatalyst for Hydrogen Evolution Reaction in Alkaline Electrolysis, Int. J. Hydrogen Energy, 2021, 46(72), 35886–35895 CrossRef CAS.
- X. Li, S. Xu, J. Li, S. Zhang, B. Zhang, R. Zhao, D. Zhao and F. Wu, NiFe-LDH as a Bifunctional Electrocatalyst for Efficient Water and Seawater Electrolysis: Enhanced Oxygen Evolution and Hydrogen Evolution Reactions, Nanoscale Adv., 2025, 7, 5546–5560 RSC.
- Y. Zhuang, S. Meng, Y. Wu, J. Li, X. Yang, C. Yuan, T. Peng and D. Guo, Enhanced Overall Water Splitting Performance in Alkaline Solutions Using Hollow Nanocage Nickel-Cobalt-Iron Layered Double Hydroxide Derived from Zeolitic Imidazolate Framework-67: The Synergy of Fast Etching and Metallic Ions, J. Colloid Interface Sci., 2025, 138645 CrossRef CAS PubMed.
- B. H. Suryanto, Y. Wang, R. K. Hocking, W. Adamson and C. Zhao, Overall Electrochemical Splitting of Water at the Heterogeneous Interface of Nickel and Iron Oxide, Nat. Commun., 2019, 10(1), 5599 CrossRef CAS PubMed.
- M. S. S. Danish, Exploring Metal Oxides for the Hydrogen Evolution Reaction (HER) in the Field of Nanotechnology, RSC Sustain., 2023, 1(9), 2180–2196 RSC.
- M. Bhavanari, K.-R. Lee, C.-J. Tseng, B.-J. Su, J.-M. Chen, J.-K. Chang, A. J. Bhattacharyya and C.-Y. Su, New Insights into Interface Charge-Transfer Mechanism of Copper-Iron Layered Double Hydroxide Cathodic Electrocatalyst in Alkaline Electrolysis, J. Environ. Chem. Eng., 2022, 10(2), 107287 CrossRef CAS.
- D. Alves, R. A. Moral, D. Jayakumari, E. Dempsey and C. B. Breslin, Factorial Optimization of CoCuFe-LDH/Graphene Ternary Composites as Electrocatalysts for Water Splitting, ACS Appl. Mater. Interfaces, 2024, 16(38), 50846–50858, DOI:10.1021/acsami.4c10870.
- F. Arshad, A. Munir, A. Tahir, S. Z. Hussain, A. Jilani, A. Hussain, N. Ullah, F. Sher and I. Hussain, Microwave-Assisted Growth of Spherical Core-Shell NiFe LDH@ CuxO Nanostructures for Electrocatalytic Water Oxidation Reaction, Int. J. Hydrogen Energy, 2023, 48(12), 4719–4727 CrossRef CAS.
- X. Feng, X. Hao, X. Zhao, Y. Guo, S. Ma, B. Xu, H. Liu and B. Gao, Tailoring Nanostructured NiFe-LDH Catalysts via Ammonium Fluoride-Mediated Hydrothermal Synthesis: Enhanced Electrocatalytic Performance in Oxygen Evolution Reactions, CrystEngComm, 2025, 27(10), 1483–1490 RSC.
- P. Gholami, L. Dinpazhoh, A. Khataee, A. Hassani and A. Bhatnagar, Facile Hydrothermal Synthesis of Novel Fe-Cu Layered Double Hydroxide/Biochar Nanocomposite with Enhanced Sonocatalytic Activity for Degradation of Cefazolin Sodium, J. Hazard Mater., 2020, 381, 120742 CrossRef CAS PubMed.
- W. Y. Lim and G. W. Ho, Nickel-Cobalt Layered Double Hydroxides for Photocatalytic Degradation under Visible Light Irradiation, Procedia Eng., 2017, 215, 163–170 CrossRef CAS.
- C. Xing, F. Musharavati, H. Li, E. Zalezhad, O. K. S. Hui, S. Bae and B.-Y. Cho, Synthesis, Characterization, and Properties of Nickel–Cobalt Layered Double Hydroxide Nanostructures, RSC Adv., 2017, 7(62), 38945–38950, 10.1039/C7RA06670H.
- A. R. Nallayagari, E. Sgreccia, L. Pasquini, M. Sette, P. Knauth and M. L. Di Vona, Impact of Anion Exchange Ionomers on the Electrocatalytic Performance for the Oxygen Reduction Reaction of B-N Co-Doped Carbon Quantum Dots on Activated Carbon, ACS Appl. Mater. Interfaces, 2022, 14(41), 46537–46547, DOI:10.1021/acsami.2c11802.
- A. R. Nallayagari, E. Sgreccia, L. Pasquini, F. Vacandio, S. Kaciulis, M. L. Di Vona and P. Knauth, Catalytic Electrodes for the Oxygen Reduction Reaction Based on Co-Doped (BN, Si-N, SN) Carbon Quantum Dots and Anion Exchange Ionomer, Electrochim. Acta, 2022, 427, 140861 CrossRef CAS.
- X. Meng, X. Liu, D. Zeng, Y. Huang, H. Wang, Z. Li and C. Yu, Recent Advances in Biomass-Derived Hydrochar for Photocatalytic and Electrocatalytic Applications, Chem. Eng. Sci., 2025, 121435 CrossRef CAS.
- A. Marrocchi, E. Cerza, S. Chandrasekaran, E. Sgreccia, S. Kaciulis, L. Vaccaro, S. Syahputra, F. Vacandio, P. Knauth and M. L. Di Vona, Hydrochar from Pine Needles as a Green Alternative for Catalytic Electrodes in Energy Applications, Molecules, 2024, 29(14), 3286 CrossRef CAS PubMed.
- S. Chandrasekaran, R. Narducci, E. Sgreccia, A. Haider, L. Pasquini, A. Marocchi, E. Cerza, M. L. Di Vona and P. Knauth, Sustainable Electrodes Based on Biomass-Derived Catalysts and Ionomers for the Oxygen Reduction Reaction, J. Mater. Sci.: Mater. Energy, 2025, 1, 7 Search PubMed.
- R.-A. Becerra-Arciniegas, R. Narducci, G. Ercolani, E. Sgreccia, L. Pasquini, M. L. Di Vona and P. Knauth, Model Long Side-Chain PPO-Based Anion Exchange Ionomers: Properties and Alkaline Stability, J. Phys. Chem. C, 2020, 124(2), 1309–1316, DOI:10.1021/acs.jpcc.9b10480.
- K. Kordek, L. Jiang, K. Fan, Z. Zhu, L. Xu, M. Al-Mamun, Y. Dou, S. Chen, P. Liu, H. Yin, P. Rutkowski and H. Zhao, Two-Step Activated Carbon Cloth with Oxygen-Rich Functional Groups as a High-Performance Additive-Free Air Electrode for Flexible Zinc–Air Batteries, Adv. Energy Mater., 2019, 9(4), 1802936, DOI:10.1002/aenm.201802936.
- J. Rouquerol, F. Rouquerol, P. Llewellyn, G. Maurinand K. Sing, in. Adsorption by Powders and Porous Solids: Principles, Methodology and Applications, Academic press, 2013 Search PubMed.
- S. Sun, Y. He, T. Chen, C. Sun and C. Wu, Morphology Regulated Synthesis of NiFe-Layered Double Hydroxide Nanostructures on Nickel Foam toward Efficient Oxygen Evolution Reaction, J. Alloys Compd., 2023, 963, 171304 CrossRef CAS.
- M. Shabanian, M. Hajibeygi, A. Raeisi, FTIR Characterization of Layered Double Hydroxides and Modified Layered Double Hydroxides. In Layered Double Hydroxide Polymer Nanocomposites, Elsevier, 2020, pp 77–101 Search PubMed.
- K. Nejati, S. Davary and M. Saati, Study of 2, 4-Dichlorophenoxyacetic Acid (2, 4-D) Removal by Cu-Fe-Layered Double Hydroxide from Aqueous Solution, Appl. Surf. Sci., 2013, 280, 67–73 CrossRef CAS.
- R. Khoshbouy, F. Takahashi and K. Yoshikawa, Preparation of High Surface Area Sludge-Based Activated Hydrochar via Hydrothermal Carbonization and Application in the Removal of Basic Dye, Environ. Res., 2019, 175, 457–467 CrossRef CAS PubMed.
- S. Anantharaj, K. Karthick, M. Venkatesh, T. V. Simha, A. S. Salunke, L. Ma, H. Liang and S. Kundu, Enhancing Electrocatalytic Total Water Splitting at Few Layer Pt-NiFe Layered Double Hydroxide Interfaces, Nano Energy, 2017, 39, 30–43 CrossRef CAS.
- S. M. Gateman, O. Gharbi, H. G. De Melo, K. Ngo, M. Turmine and V. Vivier, On the Use of a Constant Phase Element (CPE) in Electrochemistry, Curr. Opin. Electrochem., 2022, 36, 101133 CrossRef CAS.
- Z. Xie, W. Qu, E. A. Fisher, J. Fahlman, K. Asazawa, T. Hayashi, H. Shirataki and H. Murase, Capacitance Determination for the Evaluation of Electrochemically Active Surface Area in a Catalyst Layer of NiFe-Layered Double Hydroxides for Anion Exchange Membrane Water Electrolyser, Materials, 2024, 17(3), 556 CrossRef CAS PubMed.
- F. Dionigi, Z. Zeng, I. Sinev, T. M. S. Deshpande, M. B. Lopez, S. Kunze, I. Zegkinoglou, H. Sarodnik, D. Fan and A. Bergmann, et al., In-situ structure and catalytic mechanism of NiFe and CoFe layered double hydroxides during oxygen evolution, Nat. Commun., 2020, 11, 2522 CrossRef CAS PubMed.
- G.-F. Li, D. Yang and P.-Y. Abel Chuang, Defining Nafion Ionomer Roles for Enhancing Alkaline Oxygen Evolution Electrocatalysis, ACS Catal., 2018, 8(12), 11688–11698, DOI:10.1021/acscatal.8b02217.
- P. Mardle, B. Chen and S. Holdcroft, Opportunities of Ionomer Development for Anion-Exchange Membrane Water Electrolysis: Focus Review, ACS Energy Lett., 2023, 8(8), 3330–3342, DOI:10.1021/acsenergylett.3c01040.
- M. K. Cho, H.-Y. Park, H. J. Lee, H.-J. Kim, A. Lim, D. Henkensmeier, S. J. Yoo, J. Y. Kim, S. Y. Lee and H. S. Park, Alkaline Anion Exchange Membrane Water Electrolysis: Effects of Electrolyte Feed Method and Electrode Binder Content, J. Power Sources, 2018, 382, 22–29 CrossRef CAS.
- S. Wang, F. Yuan, G. Yang, S. Luo, M. Chen, T. Fan and J. Ma, In Situ Construction of CoFe-LDH by Regulating the Co/Fe Molar Ratio for Promoting Oxygen Evolution Reaction, Mol. Catal., 2022, 525, 112339 CrossRef CAS.
- Y. Yang, Y. Wu, D. Guo and L. Liu, Effect of Fluorine Content in Layered Double Hydroxide Catalyzing Oxygen Evolution, Appl. Surf. Sci., 2024, 644, 158810 CrossRef CAS.
- C. G. Díaz-Maroto, F. Verdugo, J. Fermoso, P. Pizarro, D. P. Serrano, I. Moreno and J. Fermoso, Hydrochars Derived from Real Organic Wastes as Carbonaceous Precursors of Activated Carbons for the Removal of NO from Contaminated Gas Streams, Sci. Total Environ., 2024, 945, 173897 CrossRef PubMed.
- K.-W. Jung, S. Y. Lee, J.-W. Choi, M.-J. Hwang and W. G. Shim, Synthesis of Mg–Al Layered Double Hydroxides-Functionalized Hydrochar Composite via an in Situ One-Pot Hydrothermal Method for Arsenate and Phosphate Removal: Structural Characterization and Adsorption Performance, Chem. Eng. J., 2021, 420, 129775 CrossRef CAS.
- S. Yadav, A. Ahmad, C. Gulati, M. M. Ghangrekar and B. K. Dubey, Zn-Al@ LDH Infused Hydrochar as Cathode Catalyst for Upgrading Tetracycline Degradation and Hospital Wastewater Treatment: A Synergy of Fenton-like and Bio-Electrochemical Systems, J. Environ. Chem. Eng., 2024, 12(5), 113874 CrossRef CAS.
- W. Zou, W. Guo, X. Liu, Y. Luo, Q. Ye, X. Xu and F. Wang, Anion Exchange of Ni–Co Layered Double Hydroxide (LDH) Nanoarrays for a High-Capacitance Supercapacitor Electrode: A Comparison of Alkali Anion Exchange and Sulfuration, Chem. Eur. J., 2018, 24(72), 19309–19316, DOI:10.1002/chem.201804218.
- Y. Wang, Y. Liu, Z. Chen, M. Zhang, B. Liu, Z. Xu and K. Yan, In Situ Growth of Hydrophilic Nickel-Cobalt Layered Double Hydroxides Nanosheets on Biomass Waste-Derived Porous Carbon for High-Performance Hybrid Supercapacitors, Green Chem. Eng., 2022, 3(1), 55–63 CrossRef CAS.
- L. Zhang, C. Han, Z. Ye, D. Cui, J. Yang, H. Xu, H. Song, Z. Guo, X. Wu and Y. Zhao, Mo-Doped CoFe-Layered Double Hydroxides as an Efficient Bifunctional Electrocatalyst for Overall Water Splitting: Effects of Different Molybdenum Additions on Catalytic Performance, Int. J. Hydrogen Energy, 2024, 93, 355–363 CrossRef CAS.
- A. Chaudhari, M. R. Kandel, A. Ali, D. R. Paudel, B. Dahal and S. Subedi, Ni-Doped Co-Layered Double Hydroxides as High-Performance Electrocatalyst for the Hydrogen Evolution Reaction, J. Nepal Chem. Soc., 2025, 45(1), 66–75 CrossRef.
- H. S. Jadhav, A. Roy, B. Z. Desalegan and J. G. Seo, An Advanced and Highly Efficient Ce Assisted NiFe-LDH Electrocatalyst for Overall Water Splitting, Sustain. Energy Fuels, 2020, 4(1), 312–323 RSC.
- T. Shinagawa, A. T. Garcia-Esparza and K. Takanabe, Insight on Tafel Slopes from a Microkinetic Analysis of Aqueous Electrocatalysis for Energy Conversion, Sci. Rep., 2015, 5(1), 13801 CrossRef PubMed.
- H. Prats and K. Chan, The Determination of the HOR/HER Reaction Mechanism from Experimental Kinetic Data, Phys. Chem. Chem. Phys., 2021, 23(48), 27150–27158 RSC.
- S. Anantharaj, S. Noda, V. R. Jothi, S. Yi, M. Driess and P. W. Menezes, Strategies and Perspectives to Catch the Missing Pieces in Energy-Efficient Hydrogen Evolution Reaction in Alkaline Media, Angew. Chem., Int. Ed., 2021, 60(35), 18981–19006, DOI:10.1002/anie.202015738.
- R. B. Patil, M. Kaur, S. D. House, L. Kavalsky, K. Hu, S. Zhong, D. Krishnamurthy, V. Viswanathan, J. Yang and Y. Yan, Reversible Alkaline Hydrogen Evolution and Oxidation Reactions Using Ni–Mo Catalysts Supported on Carbon, Energy Adv., 2023, 2(9), 1500–1511 RSC.
- P. J. Rheinländer, J. Herranz, J. Durst and H. A. Gasteiger, Kinetics of the Hydrogen Oxidation/Evolution Reaction on Polycrystalline Platinum in Alkaline Electrolyte Reaction Order with Respect to Hydrogen Pressure, J. Electrochem. Soc., 2014, 161(14), F1448 CrossRef.
- W. Sheng, Z. Zhuang, M. Gao, J. Zheng, J. G. Chen and Y. Yan, Correlating Hydrogen Oxidation and Evolution Activity on Platinum at Different pH with Measured Hydrogen Binding Energy, Nat. Commun., 2015, 6(1), 5848 CrossRef CAS PubMed.
- R.-B. Lin and S.-M. Shih, Effects of Mass Transfer on Kinetics of Hydrogen Oxidation Reaction at Nafion/Pt-Black Thin-Film Electrodes, J. Taiwan Inst. Chem. Eng., 2013, 44(3), 393–401 CrossRef CAS.
- F. B. Spingler, A. Phillips, T. Schuler, M. C. Tucker and A. Z. Weber, Investigating Fuel-Cell Transport Limitations Using Hydrogen Limiting Current, Int. J. Hydrogen Energy, 2017, 42(19), 13960–13969 CrossRef CAS.
- X. Zhao, X. Li, L. An, K. Iputera, J. Zhu, P. Gao, R.-S. Liu, Z. Peng, J. Yang and D. Wang, Nitrogen-Inserted Nickel Nanosheets with Controlled Orbital Hybridization and Strain Fields for Boosted Hydrogen Oxidation in Alkaline Electrolytes, Energy Environ. Sci., 2022, 15(3), 1234–1242 RSC.
| This journal is © The Royal Society of Chemistry 2026 |




