 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Formation of mixed-phase NixB/Co3O4/Co(OH)2 and its application as a pre-catalyst for the oxygen evolution reaction
Gillian
Collins
a,
Ramaraj
Sukanya
 a,
Daniele
Alves
a,
Thamaraiselvi
Kanagaraj
b,
Raj
Karthik
a,
Daniele
Alves
a,
Thamaraiselvi
Kanagaraj
b,
Raj
Karthik
 a,
Tara N.
Barwa
a,
Jae-Jin
Shim
a,
Tara N.
Barwa
a,
Jae-Jin
Shim
 b and
Carmel B.
Breslin
b and
Carmel B.
Breslin
 *ac
*ac
aDepartment of Chemistry, Maynooth University, Maynooth, Co. Kildare, Ireland. E-mail: Carmel.Breslin@mu.ie
bSchool of Chemical Engineering, Yeungnam University, 280 Daehak-ro, Gyeongsan, Gyeongbuk 38541, Republic of Korea
cKathleen Lonsdale Institute, Maynooth University, Maynooth, Co. Kildare, Ireland
First published on 5th February 2026
Abstract
Herein, a mixed-phase nickel boride/cobalt oxide/cobalt hydroxide, NixB/Co3O4/Co(OH)2, composite was formed to give a scalable and non-precious pre-catalyst for the oxygen evolution reaction (OER) in alkaline medium. The material was synthesised via a simple chemical reduction method, where amorphous NixB particles were deposited onto cobalt oxide rods undergoing partial transformation into Co(OH)2 sheets. This approach suppresses the agglomeration of NixB particles, increasing surface accessibility. The optimised composite (NixB/Co3O4-100 mg/Co(OH)2) achieved a low overpotential of 370 mV at 100 mA cm−2. It retained 99.2% of its activity after more than 90-h of continuous operation at 100 mA cm−2 for the OER in 1.0 M KOH, indicating excellent stability. Electrochemical impedance analysis revealed a 20-fold increase in the electroactive surface area compared to the bare substrate. XPS analysis following stability testing under OER conditions confirmed a full surface reconstruction, with the loss of boron from the surface and formation of catalytically active NiOOH and CoOOH species, indicating that NixB, Co3O4 and Co(OH)2 act as precursors to the true active phase. This work highlights a practical strategy for designing robust, non-precious OER catalysts through the phase reconstruction of boride/oxide hybrids.
1. Introduction
Hydrogen production via water electrolysis is a promising option for sustainable energy, but its efficiency is limited by the oxygen evolution reaction (OER) half-reaction, which is a complex, four-electron process that demands a substantial energy input.1 As the rate-limiting step, OER directly influences the energy requirement and cost of hydrogen production, necessitating the development of high-performing, non-precious OER catalysts critical to lowering the overall energy requirements for water-splitting applications.2 Conventional OER catalysts, such as iridium and ruthenium oxides, deliver high performance but are costly and limited in supply, necessitating the exploration of new materials with comparable catalytic efficacy.3,4Transition metal (TM)-based compounds, particularly pre-catalysts that undergo in situ surface reconstruction to form active (oxy)hydroxide layers under OER conditions, have emerged as viable electrocatalysts due to their enhanced active-site density and stability.5–8 This surface transformation process is associated with improved catalytic performance. Indeed, numerous studies report that the in situ oxidation of chalcogenides results in significantly higher surface areas than their TM-oxide equivalents, through surface transformation, enhancing the OER activity.5,8–10
Among these, transition metal borides (TMBs) have gained attention as a promising class of materials for catalytic applications due to their structural versatility, electronic characteristics, and high specific capacity.11,12 The performance of NixB is rooted in its electron-rich nickel centres, created by partial electron transfer from boron to nickel.13 This electronic redistribution lowers kinetic barriers, facilitating charge transfer and enhancing the binding of reaction intermediates. In its amorphous form, NixB benefits from a disordered structure that offers a high density of exposed active sites, particularly favourable for OH− adsorption, a key step in efficient water oxidation. The morphology of NixB nanoparticles further enhances these properties by maximising surface area and promoting rapid electron and ion transport, which are essential qualities for OER catalysis.14 In addition, NixB can transform into highly active NiOOH species through surface reconstruction, thereby exposing electron-rich Ni sites optimal for the conversion of OER intermediates.15 Despite these advantages, NixB particles tend to aggregate, reducing the accessibility of their active sites. Previous studies have explored strategies to improve NixB dispersion and performance by anchoring it to various supports.16–19 However, many of these supports are either catalytically inert, unstable under OER conditions or rely on platinum group metals (PGMs), which limit practicality for sustainable water-splitting systems.20
On the other hand, cobalt oxides such as Co3O4 and Co(OH)2, which similarly convert to CoOOH under anodic bias,20 offer complementary redox properties, high conductivity, and morphological tunability, making them ideal as supports for hybrid NixB/Co-based composites. Moreover, these cobalt oxides can be readily tailored into rod- and sheet-like morphologies, increasing the exposed surface area and improving contact between active sites and the electrolyte.21,22 Co3O4, a mixed-valence state and spinel-type oxide, offers high conductivity and multiple accessible oxidation states, while Co(OH)2 serves as a common in situ precursor to active oxyhydroxide species under OER conditions. These properties make them suitable as catalytically relevant supports for anchoring amorphous NixB, which are otherwise prone to aggregation. The combination of a cobalt-based support with NixB may therefore address the dual challenge of aggregation and catalytic stability and performance, whilst avoiding the limitations of unsuitable or PGM support systems.
In this work, we present a simple, scalable chemical reduction strategy to prepare a mixed-phase NixB/Co3O4/Co(OH)2 composite as a pre-catalyst for alkaline OER. In situ reduction of Ni2+ and partial transformation of Co3O4 rods into Co(OH)2 sheets yield a hybrid structure that supports active site exposure, while suppressing NixB agglomeration. Under anodic bias, the material undergoes surface reconstruction, forming the catalytically active nickel and cobalt (oxy)hydroxide species. Aiming to evaluate the true intrinsic behaviour of the NixB/Co3O4/Co(OH)2 catalyst without substrate interference, carbon cloth (CC) was used for the initial screening because it is largely inactive towards the OER. Therefore, it provides a cleaner baseline for comparing catalyst formulations without a strong substrate contribution, allowing the electrochemical response to be assigned primarily to the deposited catalyst rather than the substrate. This is particularly important for metrics such as electrochemical surface area (ECSA), Tafel slopes, CV redox features, and LSV-derived overpotentials, which can be distorted as an active current collector can also contribute to faradaic processes. However, carbon-based materials, including CC, are not ideal for extended anodic operation in alkaline media as carbon is prone to oxidative degradation under prolonged anodic conditions, which limits their suitability for durability studies.23 Therefore, following initial screening, the optimised composite was transferred onto a nickel foam (NF) substrate, which offers high electrical conductivity, a 3D porous architecture that improves electrolyte access and bubble release, and mechanical robustness under alkaline electrolysis conditions. The resulting material achieves an overpotential of 370 mV at 100 mA cm−2 with stability maintained over a 90-h period. Post-analysis XPS confirms complete surface reconstruction to catalytically active NiOOH and CoOOH, indicating that NixB/Co3O4/Co(OH)2 acts as a precursor for this active catalyst.
While NixB particles and Co3O4 have been used as electrocatalysts, this unique combination of NixB/Co3O4/Co(OH)2 has not, to the best of our knowledge, been previously utilised as a pre-catalyst for the OER. This makes it an interesting material, where the Co3O4 rods are partially converted to Co(OH)2 sheets, which can then readily form the active CoOOH surface phase, while the NixB serves as a source of NiOOH. This work contributes to the growing body of research on boride-based pre-catalysts, offering practical insights into hybrid support design, transformation-driven catalysis, and long-term OER stability in alkaline conditions.
2. Experimental
Detailed information about the materials, reagents, material characterisation, and electrochemical measurements on the prepared materials can be found in the SI.2.1 Synthesis of Co3O4 rods
In separate beakers, 1.19 g of CoCl2 (A1), 1.5 g of urea (B1), and 0.05 g of CTAB (cetyltrimethylammonium bromide) (C1) were each dissolved in 10 mL of deionised water (DI water) under stirring at 700 rpm, forming solutions A1, B1, and C1, respectively. After 10 min of mixing, solutions B1 and C1 were combined into solution A1 very slowly to ensure homogeneity. The combined mixture was then transferred to a Teflon-lined autoclave and heated at 120 °C for 12 h. After the reaction, the resulting product was washed thoroughly with deionised (DI) water and ethanol. The material was dried overnight at 50 °C. Finally, the dried product was calcined at 300 °C for 2 h. After calcination, the material was allowed to cool to room temperature and stored for further use. This methodology is illustrated in Scheme 1(a).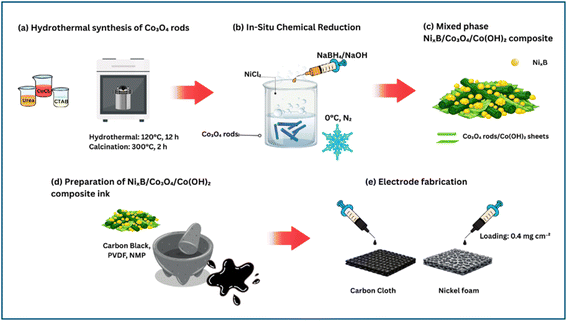 | ||
| Scheme 1 Schematic representation of the preparation and fabrication of the electrocatalysts on CC and NF. | ||
2.2 Synthesis of amorphous nickel boride (NixB)
0.742 g of NiCl2 was transferred to a beaker containing 25 mL of DI water and stirred at 700 rpm for 10 min, and labelled as solution A2. Separately, 0.189 g of NaBH4, a reducing agent and source of boron, was dissolved in 10 mL of DI water with 0.02 g of NaOH, and the mixture was labelled as solution B2. After 10 min, solution A2 was transferred to an ice-bath set up to maintain the reaction temperature at 0 °C. After 20 min of continuous stirring under a N2 atmosphere, solution B2 was slowly added dropwise to solution A2 using a syringe. Upon the addition of solution B2, an effervescence began, and a black precipitate formed immediately. The reaction was carried out for 30 min under an N2 atmosphere and stirring at 700 rpm at 0 °C. After the reaction, the precipitate was washed thoroughly with DI water and finally with ethanol. The powder was dried overnight at 50 °C and stored for further use.2.3 Preparation of NixB/Co3O4/Co(OH)2
The NixB/Co3O4/Co(OH)2 composites were synthesised via an in situ chemical reduction process, as shown in Scheme 1(b) and (c). Pre-synthesised Co3O4 (50, 100, or 150 mg) was dispersed in 25 mL of DI water and sonicated for 10 min to ensure uniform dispersion. These suspensions were transferred to a Buchner flask and stirred at 700 rpm, designated as solution A3. Separately, 0.742 g of NiCl2 was weighed out, and the nickel salt was slowly added to solution A3, which was then stirred for an additional 10 min to give solution B3. After 10 min, this solution was transferred to an ice-bath and maintained at 0 °C. Separately, 0.189 g of NaBH4 in 10 mL DI water with 0.02 g NaOH was prepared and labelled as solution C3. After 20 min of continuous stirring under a N2 atmosphere, solution B3 was slowly added dropwise to the Co3O4/Ni mixture (solution C3) using a syringe. The reaction proceeded under these conditions for an additional 30 min, with a gradual formation of Co(OH)2 sheets alongside the Co3O4 rods due to the basic and reducing atmosphere. The final composite materials, consisting of Co3O4 rods and Co(OH)2 sheets and amorphous NixB, were washed thoroughly with DI water and ethanol, dried overnight at 50 °C and stored for further analysis. Samples were designated as NixB/CoO-50, as NixB/CoO-100, and as NixB/CoO-150, corresponding to the initial Co3O4 rod mass used.2.4 Electrode preparation and coating
Carbon cloth (CC) electrodes were prepared by cutting a 1 × 2 cm2 piece of commercial CC. The CC was cleaned via 5-min sonication in 1.0 M HCl and washed with DI water and ethanol, followed by overnight drying at 60 °C. The catalyst slurry ink was then prepared by mixing 81% of the active material, 14% carbon black, and 5% polyvinylidene fluoride (PVDF) as a binder, with sufficient N-methyl-2-pyrrolidone (NMP) as a solvent. Finally, the slurry was evenly coated onto the CC in a 1 × 1 cm2 area of the CC substrate and dried at 50 °C between the layers, resulting in a final catalyst loading of 0.4 mg cm−2, as illustrated in Scheme 1(d) and (e). This was found to be the optimal loading. The influence of NixB/CoO-100 loadings is summarised in Table S1. While loadings between 0.1 and 0.6 mg cm−2 have little influence at lower overpotentials, the 0.4 mg cm−2 loading exhibits the lowest overpotential during the OER at a current density of 50 mA cm−2.Nickel foam (NF) electrodes were prepared by cutting a 1 × 1 cm2 piece of commercial NF. NF can be difficult to standardise due to its disordered porosity and variability between samples, leading to a wide range of baseline performances reported in the literature.24 To minimise variability and improve reproducibility, 1 × 1 cm2 NF pieces were pre-weighed and matched in mass across experiments. Each was connected to a nickel wire (all of equal weight), cleaned with acetone, and then electrochemically reduced by immersing the NF in 1.0 M KOH at −1.0 V (vs. Hg/HgO) for 10 min to remove surface oxides. It was then pressed to a uniform thickness of approximately 0.1 mm to provide a planar surface, facilitating the coating process. The slurry was prepared as above, but without the addition of conducting carbon black, as NF is intrinsically conducting. Like the CC, a final catalyst loading of 0.4 mg cm−2 was maintained.
3. Results and discussion
3.1 Characterisation of materials
The composite was prepared via an in situ chemical reduction process, in which amorphous NixB particles were deposited onto a cobalt-based support undergoing partial phase transformation. Specifically, the as-prepared Co3O4 rods were exposed to the highly alkaline and reducing environment generated by NaBH4 and NaOH. Under these conditions, Co3+ ions within the Co3O4 lattice are partially reduced to Co2+, and this, along with the existing Co2+ in the oxide phase, promotes the formation of Co(OH)2 sheets. Simultaneously, Ni2+ is reduced to form amorphous NixB, which nucleates and deposits across the Co3O4/Co(OH)2 surface. The result is a multidimensional composite comprising NixB particles interfacing with Co(OH)2 sheets as well as Co3O4 rods. The reaction pathway can be summarised in eqn (1) and (2).| Co3O4 + 3H2O → 3Co(OH)2 + ½O2 | (1) |
| Ni2+ + 2BH4− + 3H2O → NixB + B(OH)3 + 5½H2 | (2) |
The morphology and structural transformation within the NixB/Co3O4/Co(OH)2 composite were initially examined through field-emission scanning electron microscopy (FE-SEM), as shown in Fig. 1. FE-SEM images of pristine Co3O4 rods (Fig. 1(a)) display well-defined rod-like structures with varying lengths. These rod-like features are more readily seen in the inset in Fig. 1(a) and provide a favourable morphology for maximising surface area and enhancing electron transport, critical factors for electrochemical applications. In contrast, the amorphous NixB phase, prepared without the addition of the Co3O4 rods (Fig. 1(b) and the inset in Fig. 1(b)), is observed as spherical aggregated particles. FE-SEM analysis of the final composite (Fig. 1(c) and (d)) reveals a more complex architecture, where both Co3O4 rods and newly formed 2D sheet-like Co(OH)2 structures coexist, decorated with dispersed spherical NixB particles. This transformation from rods to sheets is likely promoted by the introduction of NaBH4 during the decoration process, resulting in a mixed-phase Co3O4/Co(OH)2 support that helps reduce NixB agglomeration and improve electrochemical accessibility. Energy-dispersive X-ray (EDX) mapping (Fig. 1(e–h)) further confirms the uniform distribution of Co, Ni, O, and B elements across the composite, indicating successful integration of NixB with Co3O4 and Co(OH)2.
X-ray diffraction (XRD) analysis was performed to confirm the incorporation of amorphous NixB and to investigate phase transformations within the Co3O4 rods during NixB deposition. As shown in Fig. 2(b), the XRD pattern of pristine Co3O4 exhibits sharp peaks at 19.0°, 31.3°, 36.8°, 38.5°, 44.8°, 59.3°, and 65.2°, corresponding to the (111), (220), (222), (311), (400), (511), and (440) planes of cubic Co3O4 (JCPDS 00-001-1152). In contrast, pristine NixB displays a broad, undefined peak between 40° and 50°, characteristic of its amorphous nature.25 In the composite samples, the XRD patterns display sharp reflections indicative of crystalline Co3O4, along with a broad feature associated with amorphous NixB. The broad amorphous NixB peak around 45° is more prominent in the sample with the highest NixB to Co3O4 ratio (CoONiB-50), but it is present in all composites, confirming successful integration of amorphous NixB into the Co3O4/Co(OH)2 support. Additionally, a broad peak appears in the 2θ range of 10–20°, consistent with the formation of a Co(OH)2 phase.26 This is attributed to the reducing and alkaline conditions generated during the chemical reduction process. Notably, the intensity of this broad peak increases with higher initial Co3O4 content, suggesting that the composites with a higher Co3O4 to NixB ratio exhibit a greater extent of Co(OH)2 sheet formation.
 | ||
| Fig. 2 (a) FTIR spectrum and (b) XRD pattern of NixB/CoO composite, and XPS spectra of (c) Co 2p, (d) B 1s, (e) Ni 2p, and (f) O 1s of the NixB/CoO-100 composite. | ||
Further evidence of cobalt phase transformation was obtained through Fourier-transform infrared (FTIR) spectroscopy (Fig. 2(a)). In pristine Co3O4, peaks corresponding to Co–O stretching vibrations are observed in the 560–660 cm−1 region, consistent with previous reports.27,28 In the composite, these peaks diminish and broaden, reflecting disruption in the lattice structure and supporting the formation of Co(OH)2.26 Additionally, a new peak observed at 972 cm−1 in the composites can be assigned to O–H deformation vibrations of Co(OH)2.29
Collectively, these analyses indicate that the composites undergo a significant partial phase transformation, resulting in a unique structure consisting of Co3O4 rods, partially converted into Co(OH)2 sheets, with amorphous NixB particles distributed throughout the cobalt-based support. The incorporation of amorphous NixB, known for its distinct electronic properties, contributes an additional catalytic component to the material, thereby enhancing its electrochemical performance. This mixed-phase structure increases the available surface area and active site density, enabling contributions from each component during electrochemical operation.
The elemental composition and electronic states of the NixB/CoO-100 composite material were investigated through X-ray photoelectron spectroscopy (XPS). The survey spectrum is shown in Fig. S1, confirming the presence of Co, Ni, O and B elements in the composite. As shown in Fig. 2, the high-resolution analysis of the Co 2p spectra (Fig. 2(c)) reveals two major peaks at binding energies of 780.2 and 782.1 eV, which are dominated by multiplet splitting and satellite features typical of mixed cobalt oxide/hydroxide systems, consistent with the presence of Co3O4 and Co(OH)2.30 Additionally, a peak appears at 778.5 eV, which may be attributed to the metallic cobalt.31 The B 1s region (Fig. 2(d)) exhibits two peaks at 187.9 and 191.6 eV, corresponding to Ni–B and B–O bonds, respectively. These binding energy values align well with those reported in the literature for nickel boride-based heterostructures.14 The Ni 2p spectrum (Fig. 2(e)) was deconvoluted into three peaks located at 851.9, 853.6 and 855.5 eV, which are assigned to Ni–B and Ni–O and Ni(OH)2 species, respectively.32 The O 1s (Fig. 2(f)) spectrum displays two distinct peaks at 529.1 and 531.0 eV corresponding with metal-oxide and metal hydroxide bonding, respectively.
3.2 Electrochemical performance of NixB/CoO on CC
The electrochemical performances of the NixB/CoO composites (NixB/CoO-50, NixB/CoO-100, NixB/CoO-150) were screened for the OER on the catalytically inert CC substrate in 1.0 M KOH. Prior to catalytic testing, each electrode was electrochemically activated by cyclic voltammetry (CV) between 0.0 V and 0.7 V (vs. Hg/HgO) at a scan rate of 50 mV s−1 in 1.0 M KOH until a stable state was achieved. This activation has previously been shown to be an effective strategy to improve the OER performance of transition metals.33 A stable state was reached after approximately 10 cycles, as illustrated in supplementary data, Fig. S2. Comparative CV curves for each composite are shown in Fig. 3(a) following 10 cycles. The composites exhibit higher anodic and cathodic peak currents relative to the control samples (NixB and Co3O4), suggesting improved electrochemical activity compared to the individual components. This enhancement may be due to increased surface area and more accessible redox sites provided by the composite structure. The CV recorded for Co3O4 reveals only a minor oxidation wave, associated with the oxidation of the metallic Co observed in the XPS, to Co2+, with no indication of the reverse reaction. Hence, the more prominent redox peaks in Fig. 3(a) signify the interconversion between Ni2+ and Ni3+ with quasi-reversible behaviour. Notably, the peak-to-peak separation varies with the amount of Co3O4 added, exhibiting the smallest separation and sharper peaks for NixB/CoO-100@CC. This indicates that this particular composite demonstrates superior electrical conductivity.Following activation, linear sweep voltammetry (LSV) was performed at 2 mV s−1 in 1.0 M KOH and cycled from 0.0 V to 1.5 V vs. Hg/HgO. These data were subjected to IR correction using an 85% correction. The impact of this correction is illustrated in Fig. S3, where the IR-corrected and non-corrected plots are compared. As expected, the IR correction has a greater influence at higher currents. In Fig. 3(b), the LSV plots are displayed for the different composites, while the LSV plots for CC and the benchmark RuO2 are provided in Fig. S4. Among the composites prepared with varying Co3O4 content, NixB/CoO-100@CC exhibited the highest OER activity with an overpotential of 410 mV at a current density of 50 mA cm−2 on CC compared to the values of 435, 478, 492 and 495 mV for NixB/CoO-150@CC, NixB/CoO-50@CC, Co3O4@CC and NixB@CC, respectively. In the LSV plots in Fig. 3(b), two redox events are also clearly evident. The peak centred at 1.38 to 1.40 V (vs. RHE) is consistent with the Ni2+/Ni3+ conversion. However, the conversion of Co3O4 to CoOOH, eqn (3), which has a standard reduction potential of 1.22 V vs. RHE,34 may also contribute to this peak. Indeed, this conversion is evident in the absence of nickel with the Co3O4@CC, where the oxidation wave is centred at 1.45 V. With the good conductivity of the NixB/CoO-100@CC composites, this reduction may be more thermodynamically favourable, shifting to a somewhat lower potential to coincide with the Ni2+/Ni3+ conversion. Interestingly, the shoulder peak observed for all the NixB/CoO composites may indicate the transformation of the Co(OH)2 sheets into CoOOH, eqn (4). This conversion has been observed at 1.58 V,35 which is in good agreement with the position of this shoulder peak in Fig. 3(b) at 1.52 V.
Co3O4 + OH− + H2O ![[left over right harpoons]](https://www.rsc.org/images/entities/char_21cb.gif) 3CoOOH + e− 3CoOOH + e− | (3) |
Co(OH)2 + OH− ![[left over right harpoons]](https://www.rsc.org/images/entities/char_21cb.gif) CoOOH + H2O + e− CoOOH + H2O + e− | (4) |
Clearly, the NixB/CoO-100@CC exhibits the highest OER activity, particularly at higher overpotentials, and was therefore selected for the OER studies and compared with its individual components, Co3O4@CC and NixB@CC.
To investigate OER kinetics, Tafel slope analysis was performed on NixB/CoO-100@CC. Notably, an oxidation peak linked to the formation of NiOOH, which obscures the Tafel region, was observed in the LSV curves (Fig. 3(b)), which complicates the determination of the true Tafel slope. Therefore, the Tafel plots were constructed from steady-state measurements rather than potentiodynamic data, which often suffer from non-steady-state effects, double-layer charging, and self-oxidation artefacts.36 The steady-state current densities (jss) were obtained by chronoamperometry at various overpotentials in the Tafel region (Fig. S5). The NixB/CoO-100@CC composite exhibited a Tafel slope of 69.0 mV dec−1, while NixB@CC and Co3O4@CC gave slopes of 46.3 and 80.4 mV dec−1 (Fig. 3(c)), respectively. It is well established that the apparent Tafel slope can shift with potential, surface coverage, and catalyst loading.37 These values were therefore used for comparison of the components of the NixB/CoO-100@CC composite, rather than a mechanistic analysis. Within this region, NixB@CC exhibits the lowest slope, meaning the current increases more rapidly per unit increase in overpotential, indicating simpler reaction dynamics with faster electron transfer and fewer intermediate transitions. However, as a single-phase material, NixB@CC does not benefit from the added active site diversity seen in the mixed-phase NixB/CoO-100@CC composite. The Co3O4@CC reference catalyst exhibited a Tafel slope of 98.4 mV dec−1, indicating moderate electron transfer efficiency without the additional catalytic effects provided by the composite structure. Overall, the NixB/CoO-100@CC composite demonstrated lower overpotentials in LSV measurements compared to both Co3O4@CC and NixB@CC, likely due to the higher density of accessible active sites and improved electron transport pathways created by its multi-phase architecture. The slightly higher Tafel slope in NixB/CoO-100@CC may indicate a shift in the rate-determining step or increased mechanistic complexity, consistent with recent work showing that Tafel slopes can reflect transitions in reaction pathways rather than intrinsic kinetics alone.38
ECSA estimation typically relies on double-layer capacitance (Cdl) measurements obtained from CVs in the non-faradaic region. However, Cdl measurements for transition metal oxides and hydroxides can be impacted by side reactions, including corrosion and specific adsorption, leading to potential overestimation of capacitance values.39 Additionally, it was observed that the NixB/CoO@CC composites exhibited limited capacitive regions within the required voltage range, complicating the determination of Cdl using CV measurements. To address these challenges, the ECSA was estimated through electrochemical impedance spectroscopy (EIS) in the non-faradaic region (Fig. 3(d)) by fitting to a simple Randles cell model, yielding capacitance values which were further used to calculate both the absolute and relative ECSA values, as illustrated in Table S2. To calculate the absolute ECSA, the specific capacitance of 40 µF cm−2 (a typical value for atomically flat transition metal oxides in KOH) was compared with the recorded capacitance values. This approach yielded estimated ECSA values of 97.84 cm2 mg−1 for NixB/CoO-100@CC, 30.44 cm2 mg−1 for NixB@CC, and 59.60 cm2 mg−1 for Co3O4@CC. Additionally, using the capacitance of the bare CC as a baseline reference, the relative ECSA ratios of 20.1, 6.25 and 12.24 for NixB/CoO-100@CC, Co3O4@CC and NixB@CC were obtained (Fig. 3(e)). This analysis indicates a substantial increase in electrochemically active sites in the mixed-phase composite NixB/CoO-100@CC.
Further analysis of the charge-transfer properties was conducted using EIS (Fig. S6) under catalytic turnover conditions (1.6 V vs. RHE), and the corresponding equivalent circuit, fitted data and circuit parameters are summarised in Table S3. A low charge-transfer resistance of 1.53 ± 0.04 Ω was observed for NixB/CoO-100@CC, with a similarly low value of 1.57 ± 0.06 Ω for NixB@CC, while a slightly higher resistance of 4.47 ± 0.05 Ω was seen with Co3O4@CC, indicating that the NixB enhances the conductivity of the composite. This low charge-transfer resistance and the structural characteristics of the NixB/CoO-100@CC likely contribute to its enhanced OER performance. The increased ECSA of the composite suggests an increase in catalytic sites and activity. Meanwhile, the hierarchical 0D/2D structure provides a conducting, high-surface-area structural support that stabilises the NixB particles, reducing agglomeration and thereby preserving site availability. Additionally, the presence of multiple redox-active species may enhance the adsorption of intermediates and catalytic turnover. The NixB/CoO-100@CC also exhibits a low charge-transfer resistance due to the presence of NixB, which enhances electron transport. Collectively, these factors contribute to the improved catalytic activity observed for the composite material.
Initial details regarding the stability of the NixB/CoO-100@CC were obtained at a constant potential of 1.6 V vs. RHE. These data are presented in Fig. 3(f). It is apparent that a slight current decay occurs over the 24-h period, which may be attributed to the oxidative degradation of the CC substrate under prolonged anodic polarisation at this relatively high potential.40 Subsequently, the LSV was recorded and compared to that of a freshly prepared electrocatalyst. These data are illustrated in the inset of Fig. 3(f). The LSV curves are comparable, showing only a slight reduction in OER activity after the 24-h stability test.
3.3 Electrochemical performance of NixB/CoO-100 on NF
To validate the applicability of the NixB/CoO catalyst on a practical electrode substrate, the optimised NixB/CoO-100 catalyst was applied to a 1 × 1 cm2, 0.1 mm thick piece of pressed NF and evaluated as an electrocatalyst for the OER. SEM analysis (Fig. 4(a)) reveals a uniform coating of the catalytic ink on the pressed NF. CV cycling (Fig. 4(b)) of the NixB/CoO-100 composite on the NF substrate (NixB/CoO-100@NF) shows a marked increase in current response compared to the bare NF electrode, indicating enhanced electrochemical activity and suggesting the deposited composite provides additional redox-active sites beyond the intrinsic contribution of the NF. Interestingly, on comparing these data to the corresponding study at CC, the high conductivity of the NF becomes apparent. The oxidation peak potential is centred at 0.52 V in Fig. 4(b), but at a higher potential of 0.57 V in Fig. 3(a). Likewise, there is a difference in the reduction peak potentials, which appear at 0.34 V for the NF and at 0.23 V for the CC substrate.The stability of the NixB/CoO-100@NF was studied over a 70-h period at a fixed potential of 1.6 V vs. RHE, and the corresponding current-time plot is shown in Fig. 4(c). After an initial slight decrease in the current over the first 10 h, it remains largely unchanged, confirming the retention of catalytic performance. Following the 70-h period, the electrode was polarised for a further 24 h at a higher current density of 50 mA cm−2. The corresponding plot is shown in the inset of Fig. 4(c). Again, very good stability is seen for this additional 24-h period. After this 94-h long-term stability testing period, the LSV was measured and compared to a newly prepared electrocatalyst. The corresponding LSVs are presented in Fig. 4(d), indicating excellent OER activity. The two plots show minimal differences, with nearly identical onset potentials. At a high current density of 200 mA cm−2, the overpotentials are 391 mV for the freshly prepared NixB/CoO-100@NF compared to 419 mV following a 94-h stability study. The minimal degradation over this extended period supports the suitability of NixB/CoO-100@NF catalyst for further development under more industrially relevant conditions. Indeed, the SEM observed after stabilisation, as shown in Fig. 4(e), clearly resembles the morphology presented in Fig. 4(a), with no evidence of any change in the surface morphology following stability.
It is evident from Fig. 4(d) that the NF exhibits OER activity, and this clearly contributes to the high OER activity of the NixB/CoO-100 electrocatalyst. To establish this contribution, the OER performances of NixB/CoO-100@NF, NixB/CoO-100@CC, CC and NF were assessed using LSV studies. The corresponding LSV data are presented in Fig. S7. Here, it is clear that the NixB/CoO-100 supported on the NF substrate has a higher OER activity, and this is associated with both the high conductivity of NF and its OER activity. However, the NixB/CoO-100@NF clearly enhances the OER performance of the NF, and more particularly the CC.
Furthermore, the NixB/CoO-100@NF compares very favourably with RuO2/NF, achieving higher OER activity. Indeed, the NixB/CoO-100@NF and RuO2/NF have similar onset potentials at 317 and 313 mV, respectively, while the overpotentials at a much higher current density of 200 mA cm−2 are 483 mV for RuO2/NF, but lower at 391 mV for NixB/CoO-100@NF. Furthermore, the NixB/CoO-100@NF performs well compared to other OER electrocatalysts, as highlighted in Table S4. Regarding the cobalt-based electrocatalysts, such as CoMoO4,41 with an overpotential of 366 mV at 10 mA cm−2 (η10), and stability over 80 h, the Co2CrO4/RuO2,42 with stability over 60 h and η10 = 209 mV, and Co3Fe7/Fe3O4,43 with η10 = 279 mV and stability for 12 h, the NixB/CoO-100@NF performs well, achieving an η10 of 316 with stability over 94 h. However, the benefit of the NixB/CoO-100@NF system lies in its high OER currents at relatively low overpotentials. Many of the electrocatalysts in Table S4 require overpotentials greater than 400 mV to achieve 100 mA cm−2. In contrast, the overpotential of NixB/CoO-100@NF at 100 mA cm−2 is only 391 mV. Furthermore, the NixB/CoO-100@NF compares well with the widely reported layered double hydroxides, including doped FeNi-LDH44 (Table S4).
3.4 Post-stability characterisation of NixB/CoO-100 on NF
To investigate surface reconstruction, high-resolution XPS was conducted on the NixB/CoO-100@NF composite after 70 h of continuous operation at 1.6 V vs. RHE in 1.0 M KOH (Fig. 5(a–c)). These spectra were compared with those in Fig. 2(c–f). In the Ni 2p region, a clear transformation from metallic Ni0/Ni–B (851.9 eV) to oxidised species is observed. Following prolonged exposure to OER conditions, the Ni0 disappears entirely, replaced by dominant features at 853.2 eV (Ni–O), 855.0 eV (Ni(OH)2), and a new feature at 856.3 eV attributed to NiOOH/Ni3+, consistent with progressive oxidation reported for nickel boride surfaces undergoing electrochemical activation and reconstruction.13 This progressive oxidation indicates the in situ formation of a catalytically active nickel (oxy)hydroxide phase during OER. Similarly, the Co 2p spectra support oxidation from the initial mixed Co3O4 to higher valent species. Following the long-term stability, prominent peaks at 782.1 and 784.0 eV are assigned to Co(OH)2 and CoOOH, respectively, consistent with the oxidation of the cobalt support under OER conditions.45 The O 1 s spectra support these findings. Initially, peaks at 531.0 and 529.1 eV correspond to hydroxide (Ni(OH)2/Co(OH)2) and oxide (Ni–O/Co–O) species, respectively. After extended testing, the hydroxide signal shifts to 531.6 eV, suggesting further oxidation and the formation of oxyhydroxide phases, consistent with the growth of NiOOH and CoOOH. Notably, B 1s signals present after activation, specifically B–O (191.9 eV) and B2O3 (193.0 eV) are no longer detectable following extended OER testing. This suggests substantial boron loss from the surface, likely as soluble borate species under alkaline conditions. This behaviour is consistent with reports for TM boride electrocatalysts, where surface boron is oxidised to soluble B(OH)4− at OER-relevant potentials, leaving behind a boron-deficient (oxy)hydroxide surface.46 While some studies have observed retained oxidised boron (e.g., BO2−) influencing OER-active sites, the complete disappearance of B species in our system supports a model where NixB serves as a pre-catalyst that transforms into a NiOOH/CoOOH-rich surface during OER. This aligns with the broader classification of TM borides as pre-catalysts rather than persistent active phases.7,47 In our system, the emergence of Ni3+ and Co3+ species alongside the disappearance of B signals supports a similar pre-catalytic mechanism, wherein the NixB/CoO interface reconstructs into a mixed (oxy)hydroxide-rich surface.48,49 This supports the role of the boride phase as enhancing catalytic activity through in situ transformation, rather than via direct involvement of B-containing species in the OER process. On the basis of this reconstructed oxyhydroxide-rich surface, the OER mechanism is assumed to proceed via the adsorbate evolution mechanism (AEM) described in eqn (5)–(8) and shown schematically in Scheme 2.| H2O + * → OH* + e− + H+ | (5) |
| OH* → O* + e− + H+ | (6) |
| O* + H2O → HOO* + e− + H+ | (7) |
| HOO* → * + O2 + e− + H+ | (8) |
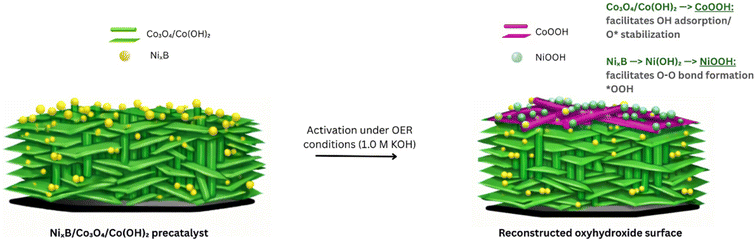 | ||
| Scheme 2 Schematic representation of the NixB/CoO-100 and its surface reconstruction to give oxyhydroxide CoOOH and NiOOH species. | ||
The first step involves OH* adsorption, followed by deprotonation of the hydroxyl species, O–O bond formation via OOH* and subsequent O2 release. In this system, surface CoOOH is expected to favour OH* adsorption and stabilisation of O* intermediates, while NiOOH sites facilitate O–O bond formation, providing a synergistic environment for the AEM which is consistent with the enhanced OER activity observed (Scheme 2).50
Consistent with previous studies, EDX (Fig. 5(e)) elemental mapping revealed the early-stage post-activation incorporation of Fe into the catalyst surface, and this appears to increase with prolonged electrochemical operation, as evidenced by post-stability EDX analysis (Fig. 5(f)). This incorporation, likely into the Ni(OH)2 layer, is well documented to enhance OER activity in alkaline media.51 The purification method developed by Marquez et al. was followed.52 As shown in Fig. 5(d), the performance was improved when using unpurified KOH (UnP-KOH) compared to the purified (P-KOH) electrolyte, further suggesting that trace Fe in the electrolyte contributes to the formation of a more active FeNi(OOH) layer at the surface of the NixB/CoO.
4. Conclusion
In this study, we demonstrate the development of a mixed-phase NixB/CoO-100@NF pre-electrocatalyst for OER in alkaline media. The catalyst, fabricated via a simple chemical reduction method, integrates amorphous nickel boride particles with cobalt oxide rods and hydroxide sheets to yield a highly active and stable composite. Among the series, the NixB/CoO-100 composite exhibited the best performance, achieving an overpotential of just 370 mV at 100 mA cm−2. Post-OER XPS analysis revealed a dynamic reconstruction mechanism wherein the initial boride and oxide phases transform into catalytically active Ni3+/Co3+ (oxy)hydroxide species. The disappearance of B 1s signals and the formation of NiOOH/CoOOH suggest a pre-catalyst behaviour, with the boride acting as a sacrificial precursor. Additional evidence of Fe incorporation during activation further highlights the dynamic nature of the catalytic surface and its role in boosting OER activity.Finally, the successful translation of the optimised catalyst coated onto an NF substrate underscores its potential for application in practical alkaline electrolysis systems. The stability of the material under prolonged electrolysis positions it as a promising candidate for scalable, cost-effective water splitting technologies.
Conflicts of interest
There are no conflicts to declare.Data availability
Data will be made available on request.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5se01506e.
Acknowledgements
This publication has emanated from research conducted with the financial support of Research Ireland under grant numbers IRC/GOIPG/2022/1605 and SFI/20/FFP-P/8793, the Sustainable Energy Authority of Ireland, grant number SEAI/22/RDD/841, and the EU Horizon 2020 program under the Marie Skłodowska-Curie grant no. 101106064.References
- M. Chen and J. Guan, Adv. Funct. Mater., 2025, 35(30), 2423552, DOI:10.1002/adfm.202423552.
- M. Yagi and M. Kaneko, Chem. Rev., 2001, 101(1), 21, DOI:10.1021/cr980108l.
- X. Long, J. Li, S. Xiao, K. Yan, Z. Wang, H. Chen and S. Yang, Angew. Chem., 2014, 126(29), 7584, DOI:10.1002/ange.201402822.
- M. R. Gao, Y. F. Xu, J. Jiang, Y. R. Zheng and S. H. Yu, J. Am. Chem. Soc., 2012, 134(6), 2930, DOI:10.1021/ja211526y.
- O. Mabayoje, A. Shoola, B. R. Wygant and C. B. Mullins, ACS Energy Lett., 2016, 1(1), 195, DOI:10.1021/acsenergylett.6b00084.
- Y. Zhu, H. C. Chen, C. S. Hsu, T. S. Lin, C. J. Chang, S. C. Chang, L. D. Tsai and H. M. Chen, ACS Energy Lett., 2019, 4(4), 987, DOI:10.1021/acsenergylett.9b00382.
- B. R. Wygant, K. Kawashima and C. B. Mullins, ACS Energy Lett., 2018, 3(12), 2956, DOI:10.1021/acsenergylett.8b01774.
- W. Chen, H. Wang, Y. Li, Y. Liu, J. Sun, S. Lee, J. S. Lee and Y. Cui, ACS Cent. Sci., 2015, 1(5), 244, DOI:10.1021/acscentsci.5b00227.
- W. Chen, Y. Liu, Y. Li, J. Sun, Y. Qiu, C. Liu, G. Zhou and Y. Cui, Nano Lett., 2016, 16(12), 7588, DOI:10.1021/acs.nanolett.6b03458.
- J. Liu and D. Xue, Adv. Mater., 2008, 20(13), 2622, DOI:10.1002/adma.200800208.
- F. Song, T. Zhang, Y. Qian, J. Shaw, S. Chen, G. Chen, Y. Sun and Y. Rao, Mater. Today Energy, 2021, 22, 100846, DOI:10.1016/j.mtener.2021.100846.
- X. Chen, Z. Yu, L. Wei, Z. Zhou, S. Zhai, J. Chen, Y. Wang, Q. Huang, H. E. Karahan, X. Liao and Y. Chen, J. Mater. Chem. A Mater., 2019, 7(2), 764, 10.1039/c8ta09130g.
- J. Masa, I. Sinev, H. Mistry, E. Ventosa, M. de la Mata, J. Arbiol, M. Muhler, B. Roldan Cuenya and W. Schuhmann, Adv. Energy Mater., 2017, 7(17), 1700381, DOI:10.1002/aenm.201700381.
- R. Sukanya, R. Karthik, M. Hasan, C. Breslin and J. J. Shim, Chem. Eng. J., 2023, 473, 145487, DOI:10.1016/j.cej.2023.145487.
- Y. Du, X. He, C. Yan, Q. Hu, J. Zhang and F. Yang, J. Mater. Chem. A Mater., 2025, 13(11), 7962, 10.1039/d4ta08295h.
- Y. Kang, B. Jiang, V. Malgras, Y. Guo, O. Cretu, K. Kimoto, A. Ashok, Z. Wan, H. Li, Y. Sugahara, Y. Yamauchi and T. Asahi, Small Methods, 2021, 5(10), 2100679, DOI:10.1002/smtd.202100679.
- X. Cao, X. Wang, L. Cui, D. Jiang, Y. Zheng and J. Liu, Chem. Eng. J., 2017, 327, 1085, DOI:10.1016/j.cej.2017.07.010.
- J. L. Hauser, G. Amberchan, M. Tso, R. Manley, K. Bustillo, J. Cooper, J. H. Golden, B. Singaram and S. R. J. Oliver, ACS Appl. Nano Mater., 2019, 2(3), 1472, DOI:10.1021/acsanm.8b02351.
- F. Yang, X. J. Ma, J. L. Yang, J. H. Liu, B. Chen, X. Y. Liu, K. Yang and W. Bin Zhang, Appl. Clay Sci., 2024, 251, 107305, DOI:10.1016/j.clay.2024.107305.
- F. Lyu, Q. Wang, S. M. Choi and Y. Yin, Small, 2019, 15, 1804201, DOI:10.1002/smll.201804201.
- Z. Liang, D. Shen, L. Wang and H. Fu, Nano Res., 2024, 17(4), 2234, DOI:10.1007/s12274-023-6219-4.
- Z. Liang, D. Shen, L. Wang and H. Fu, Small, 2021, 17, 2100129, DOI:10.1002/smll.202100129.
- S. Möller, S. Barwe, J. Masa, D. Wintrich, S. Seisel, H. Baltruschat and W. Schuhmann, Angew. Chem., Int. Ed., 2020, 59(4), 1585, DOI:10.1002/anie.201909475.
- W. Zheng, M. Liu and L. Y. S. Lee, ACS Energy Lett., 2020, 5(10), 3260, DOI:10.1021/acsenergylett.0c01958.
- W. Li, S. Wang, M. Wu, X. Wang, Y. Long and X. Lou, New J. Chem., 2017, 41(15), 7302, 10.1039/c7nj00222j.
- A. K. Dimble, N. D. Bagul, P. C. Walimbe, P. S. Kulkarni and S. D. Kulkarni, Langmuir, 2024, 40(35), 18631, DOI:10.1021/acs.langmuir.4c02149.
- G. Rathika, V. Suba, D. S. Lakshmi and R. Rani, J. Inorg. Organomet. Polym. Mater., 2022, 32(8), 3153, DOI:10.1007/s10904-022-02348-3.
- F. Zhang, C. Yuan, X. Lu, L. Zhang, Q. Che and X. Zhang, J. Power Sources, 2012, 203, 250, DOI:10.1016/j.jpowsour.2011.12.001.
- H. B. Li, M. H. Yu, X. H. Lu, P. Liu, Y. Liang, J. Xiao, Y. X. Tong and G. W. Yang, ACS Appl. Mater. Interfaces, 2014, 6(2), 745, DOI:10.1021/am404769z.
- T. J. Chuang, C. R. Brundle and D. W. Rice, Surf. Sci., 1976, 59(2), 413, DOI:10.1016/0039-6028(76)90026-1.
- S. Gupta, N. Patel, A. Miotello and D. C. Kothari, J. Power Sources, 2015, 279, 620, DOI:10.1016/j.jpowsour.2015.01.009.
- J. Wu, M. Hou, Z. Chen, W. Hao, X. Pan, H. Yang, W. Cen, Y. Liu, H. Huang, P. W. Menezes and Z. Kang, Adv. Mater., 2022, 34(32), 2202995, DOI:10.1002/adma.202202995.
- W. Wang, Y. Zhong, X. Zhang, S. Zhu, Y. Tao, Y. Zhang, H. Zhu, Y. Zhang, X. Wu and G. Hong, J. Colloid Interface Sci., 2021, 582, 803, DOI:10.1016/j.jcis.2020.08.095.
- C. Qiu, F. Maroun, M. Bouvier, I. Pacheco, P. Allongue, T. Wiegmann, C. Hendric Scharf, V. de Manuel-Gonzalez, F. Reikowski, J. Stettner, J. Stettner and O. M. Magnussen, ChemCatChem, 2024, 16(23), e202400988, DOI:10.1002/cctc.202400988.
- J. T. Mefford, A. R. Akbashev, M. Kang, C. L. Bentley, W. E. Gent, H. D. Deng, D. H. Alsem, Y.-S. Yu, N. J. Salmon, D. A. Shapiro, P. R. Unwin and W. C. Chueh, Nature, 2021, 593(7857), 67, DOI:10.1038/s41586-021-03454-x.
- S. Anantharaj, S. Noda, M. Driess and P. W. Menezes, ACS Energy Lett., 2021, 6, 4–1607, DOI:10.1021/acsenergylett.1c00608.
- A. Sengeni and S. Noda, Mater. Today Energy, 2022, 29, 101123, DOI:10.1016/j.mtener.2022.101123.
- O. van der Heijden, S. Park, R. E. Vos, J. J. J. Eggebeen and M. T. M. Koper, ACS Energy Lett., 2024, 9(4), 1871, DOI:10.1021/acsenergylett.4c00266.
- C. Wei, S. Sun, D. Mandler, X. Wang, S. Z. Qiao and Z. J. Xu, Chem. Soc. Reviews., 2019,(48), 2518, 10.1039/c8cs00848e.
- T. Wu, M. Han, X. Zhu, G. Wang, H. Zhang and H. Zhao, Chem. Commun., 2019, 55(16), 2344, 10.1039/c9cc00398c.
- X. Zhong, Y. Chen, T. Gan, Y. Huang, J. Li and S. Zhang, Nano Res., 2025, 18(3), 94907204, DOI:10.26599/NR.2025.94907204.
- M. Qi, X. Zheng, H. Tong, Y. Liu, D. Li, Z. Yan and D. Jiang, J. Colloid Interface Sci., 2025, 677, 548, DOI:10.1016/j.jcis.2024.07.260.
- R. Barath, S. Vadivel, S. Sarmila, P. Sujita, N. Murugan and Y. A. Kim, Mater. Lett., 2025, 399, 139056, DOI:10.1016/j.matlet.2025.139056.
- Y. Lv, X. Deng, J. Ding and Y. Zhou, Sci. Rep., 2024, 14, 902, DOI:10.1038/s41598-023-50361-4.
- T. Tran-Phu, R. Daiyan, J. Leverett, Z. Fusco, A. Tadich, I. Di Bernardo, A. Kiy, T. N. Truong, Q. Zhang, H. Chen, P. Kluth, R. Amal and A. Tricoli, Chem. Eng. J., 2022, 429, 132180, DOI:10.1016/j.cej.2021.132180.
- A. M. Zieschang, J. D. Bocarsly, J. Schuch, C. V. Reichel, B. Kaiser, W. Jaegermann, R. Seshadri and B. Albert, Inorg. Chem., 2019, 58(24), 16609, DOI:10.1021/acs.inorgchem.9b02617.
- K. Kawashima, R. A. Márquez, L. A. Smith, R. R. Vaidyula, O. A. Carrasco-Jaim, Z. Wang, Y. J. Son, C. L. Cao and C. B. Mullins, Chem. Rev., 2023, 123, 23–12795, DOI:10.1021/acs.chemrev.3c00005.
- S. Gul, J. W. D. Ng, R. Alonso-Mori, J. Kern, D. Sokaras, E. Anzenberg, B. Lassalle-Kaiser, Y. Gorlin, T.-C. Weng, P. H. Zwart, J. Z. Zhang, U. Bergmann, V. K. Yachandra, T. F. Jaramillo and J. Yano, Phys. Chem. Chem. Phys., 2015, 17(14), 8901, 10.1039/c5cp01023c.
- R.-Y. Fan, J.-Y. Xie, N. Yu, Y.-M. Chai and B. Dong, Int. J. Hydrogen Energy, 2022, 47(19), 10547, DOI:10.1016/j.ijhydene.2021.12.239.
- Y. Zhang, C. Han, J. Gao, L. Pan, J. Wu, X. Zhu and J.-J. Zou, ACS Catal., 2021, 11(20), 12485, DOI:10.1021/acscatal.1c03260.
- S. Anantharaj, S. Kundu and S. Noda, Nano Energy, 2021, 80, 105514, DOI:10.1016/j.nanoen.2020.105514.
- R. A. Márquez, K. Kawashima, Y. J. Son, G. Castelino, N. Miller, L. A. Smith, C. E. Chukwuneke and C. B. Mullins, ACS Energy Lett., 2023, 8(2), 1141, DOI:10.1021/acsenergylett.2c02847.
| This journal is © The Royal Society of Chemistry 2026 |




