A spin polarization porous transport layer for anion exchange membrane water electrolyzers with a current density of 11.5 A cm−2
Tae Hyung
Kim†
a,
Chuan
Hu†‡
a,
Hyeon Keun
Cho
a,
Seung Hyun
Jae
a,
Sujin
Lee
b,
Bongjun
Yeom
 b,
Young Moo
Lee
b,
Young Moo
Lee
 *a and
Young-Hoon
Kim
*a and
Young-Hoon
Kim
 *a
*a
aDepartment of Energy Engineering, Hanyang University, 222 Wangsimni-ro, Seongdong-gu, Seoul 04763, Republic of Korea. E-mail: ymlee@hanyang.ac.kr; younghoonkim@hanyang.ac.kr
bDepartment of Chemical Engineering, Hanyang University, 222 Wangsimni-ro, Seongdong-gu, Seoul 04763, Republic of Korea
First published on 10th November 2025
Abstract
Alkaline anion exchange membrane water electrolyzers (AEMWEs) are a promising technology for hydrogen production from renewable energy sources. However, their performance is far lower than that of proton exchange membrane water electrolyzers and traditional alkaline water electrolyzers. Here, we demonstrate that chiral catalysts embedded in the porous transport layer (PTL) can enhance AEMWE performance. The chiral CoOx-based PTL achieves a current density of 8.21 A cm−2 at 2.0 V in AEMWEs, which is higher than that of the achiral meso-CoOx-PTL (5.42 A cm−2). The chiral CoOx-PTL provides additional active sites and facilitates interfacial charge transfer between the catalyst and electrolyte, thereby increasing the current density during electrocatalysis. Electrochemical analysis and measurement of H2O2 byproduct concentration confirmed that the chiral CoOx-PTL suppresses H2O2 formation even after surface reconstruction, supporting the persistence of the spin polarization. Extending this strategy to bimetallic systems, the chiral NiFe-based PTL achieves a remarkable current density of 11.5 A cm−2 at 2.0 V and exceptional operational stability, maintaining 1 A cm−2 for over 1000 hours in 1 M KOH. These results demonstrate the potential of spin-engineered catalysts for advancing AEMWEs toward industrial-scale hydrogen production.
Introduction
Anion-exchange membrane water electrolyzers (AEMWEs) have attracted interest as a promising technology for future hydrogen production due to the use of less expensive platinum group metal (PGM)-free catalysts, hydrocarbon-based membranes, and the possibility of using pure water or a low concentration of alkaline solution.1 In AEMWEs, the porous transport layer (PTL) is a crucial component, as it ensures intimate contact with the membrane and the electrolyte, serves as a conductive pathway for electrons between the catalyst layer and the current collector and facilitates the efficient transport of generated gases (such as O2 and H2) from the catalyst layer. Various efforts have been devoted to manipulate the PTL to increase the stability and efficiency for producing highly purified hydrogen through water electrolysis.2,3Controlling the spin degrees of freedom in injected charge carriers through the PTL is a fundamental strategy to increase the performance of AEMWEs.4 In AEMWEs, the oxygen evolution reaction (OER) is the rate-determining step because it requires four proton-coupled electron transfers, multiple reaction steps with complicated intermediates, formation of O–O bonds and a higher overpotential than the hydrogen evolution reaction (HER).5 The produced oxygen forms a triplet spin configuration in the ground state, where two electrons with parallel spin alignment occupy the frontier π* orbital. Therefore, spin injection creates spin-aligned intermediate hydroxyl radicals (˙OH↑), which favor the formation of triplet-state O2 rather than singlet-state hydrogen peroxide (H2O2), thereby increasing the OER activity and overall performance of the AEMWE.6,7
Chiral-induced spin selectivity (CISS) demonstrates that the spin states of electrons can be polarized to align in one specific direction when passing through chiral materials.8,9 CISS has been observed in self-assembled chiral molecular organic layers and metal halide layers, where spin-polarized charge carriers are injected into adjacent halide perovskite semiconductors10 and III–V semiconductors.11 A family of chiral metal oxides has been discovered to inject spin-polarized carriers and boost the formation of O2. For example, Ai et al. reported that chiral ZnO exhibited 1.6 times higher current density compared to achiral ZnO due to the suppressed byproduct formation and enhanced electron transfer.12 Similarly, Zhang et al. demonstrated that electrodeposited chiral Fe3O4 achieved 42% higher current density than achiral Fe3O4, which was ascribed to the elongated electron lifetime and reduced charge recombination through the CISS effect.13 Other chiral metal oxides such as CoOx have also been extensively studied owing to their superior catalytic activity and stability, exhibiting enhanced OER performance in half-cell configurations through the injection of spin-polarized electrons.14,15 Although these reports clearly explained that spin polarization improved OER activities, their practical demonstrations in single-cell AEMWE devices remain unexplored.
Recently, Garcés-Pineda et al. applied chiral Ni–Fe electrocatalysts to an AEMWE and demonstrated that spin polarization improved single-cell performance, achieving a current density of 200 mA cm−2 at 1.9 V.4 While this study successfully confirmed that spin polarization induced by chiral electrocatalysts increases single-cell AEMWE efficiency, the achieved current density (200 mA cm−2 at 1.9 V) remains far below that of proton exchange membrane water electrolyzers and traditional alkaline electrolyzers.
Here, we demonstrate that embedding chiral catalysts into the porous transport layer (PTL) can enhance the AEMWE performance. The chiral CoOx-based PTL achieves a current density of 8.21 A cm−2 at 2.0 V in the AEMWE, which is higher than that of the achiral meso-CoOx-PTL (5.42 A cm−2). Electrochemical analyses confirm that the improved AEMWE activity directly correlates with the intrinsically faster OER kinetics of the chiral electrocatalysts observed in half-cell tests. Our results show that the chiral electrocatalysts suppress the formation of H2O2 byproduct formation and thereby provide evidence for CISS-driven enhancement. Moreover, the chiral NiFe-based-PTL achieves a remarkable current density of 11.5 A cm−2 at 2.0 V and exceptional operational stability, maintaining 1 A cm−2 for over 1000 hours in 1 M KOH. These results demonstrate the potential of spin-engineered catalysts for advancing industrial-scale AEMWEs.
Results and discussion
Full cell anionic exchange membrane water electrolysis (AEMWE)
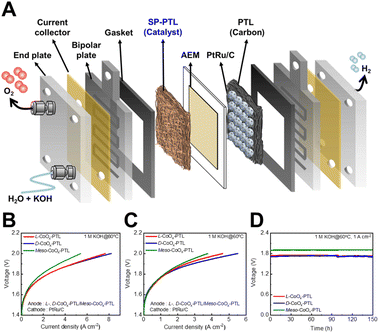
We fabricated the CoOx-based PTL by electrodepositing chiral CoOx amorphous films on a nickel fiber plate in a three-electrode electrochemical cell (Fig. S1). The scanning electron microscopy (SEM) images, elemental mapping images in transmission electron microscopy (TEM) energy dispersive spectroscopy (EDS), grazing incidence X-ray diffractometer (GIXRD), XRD and X-ray photoelectron spectroscopy (XPS) spectra (Fig. S2–S4) revealed that the CoOx amorphous thin film was successfully deposited on the nickel fiber plate. The CoOx-based PTL electrode maintains its original macrostructure after the electrodeposition of an amorphous CoOx film (Fig. S2), allowing the electrolyte and gases to penetrate the CoOx-based PTL, which are important for efficient water electrolysis performance and durability.16PtRu/C catalyst-coated carbon paper was prepared as a counter PTL cathode. A poly(aryl-co-aryl piperidinium) (PDTP-25) membrane with a thickness of ∼30 µm, pre-soaked in 1 M KOH at 60 °C for 3 hours, was prepared as an anion exchange membrane (AEM).17–19 The chiral CoOx/nickel fiber plate anode, AEM, and PtRu/C cathode with an effective area of 5 cm2 was assembled using polytetrafluoroethylene (PTFE) gaskets, gold-coated nickel plates (anode side), graphite plates (cathode side), and end plates (Fig. 1A, details in Methods).
The performance of the AEMWE was first evaluated at 80 °C in 1 M KOH. The spin-polarized AEMWE based on chiral L-CoOx-PTL and D-CoOx-PTL (0.3 mg cm−2) exhibited current densities of 7.68 A cm−2 and 8.21 A cm−2 at 2.0 V, respectively, both of which are higher than those of an AEMWE based on meso-CoOx-PTL (5.42 A cm−2) (Fig. 1B). At 60 °C (Fig. 1C), the spin-polarized AEMWE also demonstrated higher current densities (4.63 A cm−2 for L-CoOx-PTL, 5.52 A cm−2 for D-CoOx-PTL) compared to the meso-CoOx-PTL-based AEMWE (3.75 A cm−2). Additionally, temperature-dependent polarization curve analysis revealed that temperature significantly influences electrode kinetics, thereby improving current density as the temperature increases (Fig. S5). Note that L-CoOx-PTL consistently exhibited higher current densities than meso-CoOx-PTL at all tested temperatures. Furthermore, the spin-polarized AEMWE operated at a lower bias (1.71 V for L-CoOx-PTL, 1.7 V for D-CoOx-PTL) compared to 1.9 V for the spin-unpolarized AEMWE to achieve a current density of 1 A cm−2 and demonstrated highly stable operation for over 150 hours in 1 M KOH (Fig. 1D). These results indicate that a chiral electrocatalyst-PTL composed of chiral CoOx amorphous films on a nickel fiber plate effectively enhances both the efficiency and stability of the AEMWE.
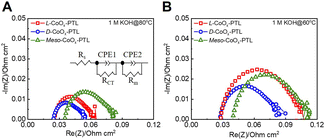
To investigate the effects of chirality on AEMWE performance, we conducted electrochemical impedance spectroscopy (EIS) measurements of the AEMWE in 1 M KOH at 80 °C (Fig. 2A). In the resulting semicircular arcs, the intercept observed at high frequency corresponds to the ohmic resistance (Rohm), while that from high to medium frequency reflects charge transfer resistance (RCT).20 The AEMWEs utilizing L-CoOx-PTL and D-CoOx-PTL exhibited Rohm values of 0.0252 Ω cm−2 and 0.0245 Ω cm−2, respectively, both of which were lower than the meso-CoOx-PTL (Rohm = 0.0354 Ω cm−2). Similarly, the RCT values for the AEMWE with the L-CoOx-PTL and D-CoOx-PTL were 0.0366 Ω cm−2 and 0.0298 Ω cm−2, respectively and were also lower than that of the meso-CoOx-PTL (RCT = 0.0457 Ω cm−2). At 60 °C, the AEMWEs with L-CoOx-PTL and D-CoOx-PTL constantly showed lower Rohm and RCT values (0.0281 Ω cm−2 and 0.0766 Ω cm−2 for L-CoOx-PTL, 0.0271 Ω cm−2 and 0.0541 Ω cm−2 for D-CoOx-PTL) compared to the meso-CoOx-PTL (0.0393 Ω cm−2 and 0.0714 Ω cm−2) (Fig. 2B). These results imply that the chiral CoOx-PTL facilitates more favorable interfacial charge transfer kinetics.
 | ||
| Fig. 2 EIS Nyquist plots of the AEMWE based on electrodeposited L-CoOx-PTL, D-CoOx-PTL and meso-CoOx-PTL measured in 1 M KOH at (A) 80 °C and (B) 60 °C. | ||
Origin of the enhanced mechanism: electrocatalytic oxygen evolution reaction
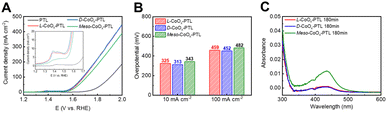
To further investigate the origin of the improved AEMWE performance induced by chiral CoOx-PTL, we performed circular dichroism (CD) measurements on the chiroptical properties of L-tartaric acid (TA) and D-TA precursor solutions, as well as the electrodeposited L-CoOx-spin polarization layer (SPL) and D-CoOx-SPL on ITO substrates under optimized deposition conditions (Fig. S6). The CD spectra confirmed that the chiral transfer from TA to SPL successfully occurred (Fig. S7). Subsequently, we analyzed the OER in a half-cell using a three-electrode system. All three electrodes exhibited typical polarization curves, displaying a redox reaction in the potential range of 1.30–1.45 V (vs. RHE), followed by an OER onset potential around 1.5 V (vs. RHE) (Fig. 3A). This redox reaction, typically exhibiting two peaks, has been previously attributed to the transition of CoOx into a hydroxide or oxyhydroxide phase during reconstruction.21 Notably, both L-CoOx-PTL and D-CoOx-PTL showed larger redox peaks than meso-CoOx-PTL, indicating greater formation of hydroxide/oxyhydroxide species in the chiral CoOx-PTL during reconstruction (Fig. 3A inset).21 We attribute this behavior to the fact that chiral CoOx-PTL possesses additional active sites which transform into the hydroxide or oxyhydroxide phase with higher catalytic reactivity due to the chiral geometry, therefore facilitating a more efficient OER and reducing resistance.The reconstruction of the chiral electrocatalysts was further confirmed by X-ray photoelectron spectroscopy (XPS). The XPS spectra of Co 2p (Fig. S8A) show that the Co3+/Co2+ ratio in the reconstructed L-CoOx-PTL increased from 1.12 to 1.29, indicating an increase in the oxidation state of Co, in agreement with an earlier report.22 Moreover, the O 1s spectra (Fig. S8B) reveal an increase in hydroxyl species (C–O) at 530.9 eV at the reconstructed L-CoOx-PTL, which increased from 48.9% in the fresh L-CoOx-PTL to 55.2% after the OER. This indicates that structural reconstruction occurs, with L-CoOx transitioning to a hydroxide or oxyhydroxide phase.23
Despite the surface reconstruction, L-CoOx-PTL and D-CoOx-PTL showed higher current densities and lower overpotentials at 10 and 100 mA cm−2 (325 and 459 mV for L-CoOx-PTL, and 313 and 452 mV for D-CoOx-PTL) compared to the meso-CoOx-PTL (343 and 482 mV) (Fig. 3B). Furthermore, L-CoOx-PTL and D-CoOx-PTL consistently exhibited lower overpotentials than meso-CoOx-PTL in KOH solutions of varying concentrations (419, 330, and 326 mV for L-CoOx-PTL, 397, 317, and 313 mV for D-CoOx-PTL and 443, 351, and 343 for meso-CoOx-PTL at 0.1, 0.5 and 1 M KOH, respectively) (Fig. S9).24
To determine whether chiral CoOx-PTLs, which maintain high current density even after reconstruction, also preserve the CISS effect in the electrolyte, we quantified hydrogen peroxide (H2O2) formation as indirect evidence of spin selectivity. H2O2 is generated through a competing two electron side reaction (2OH− → H2O2 + 2e− (1.76 V vs. RHE)) involving opposite or unpolarized spin directions (Fig. S10). The H2O2 concentration was measured by adding o-tolidine as a redox indicator after the OER at 2.2 V (vs. RHE) in 0.1 M Na2SO4 for 180 min. Both L-CoOx-PTL and D-CoOx-PTL showed lower absorption intensities corresponding to H2O2 concentration than meso-CoOx-PTL (Fig. 3C). Based on these results, we suggest that even after reconstruction, the chiral CoOx-PTLs maintain high current density and effectively suppress H2O2 formation possibly due to the CISS effect.
To understand the origin of this overall improved performance in chiral CoOx-PTLs after reconstruction, we considered various possible mechanisms. The Tafel slope was calculated to investigate the kinetics of the chiral electrocatalysts during the OER using the equation below:
E = A + B × log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) j j |
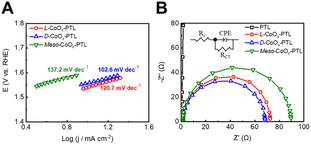 | ||
| Fig. 4 Half-cell oxygen evolution performance using L-CoOx-PTL, D-CoOx-PTL and meso-CoOx-PTL: (A) Tafel plots and (B) EIS Nyquist plots in 1 M KOH. | ||
Overall, chiral catalysts exhibited two major effects: (a) spin-induced effects and (b) increased active-site density. The spin-induced effects promote spin polarization, thereby suppressing the formation of spin-randomly oriented byproducts such as H2O2 while the increased active-site density reduces the overpotential and enhances the OER performance. Both effects originate from the intrinsic chiral geometry, which is absent in the achiral electrocatalysts. Thus, we propose that the chiral geometry fundamentally governs both the spin-induced effect and the increase in active-site density, resulting in the superior OER activity observed for the chiral systems.
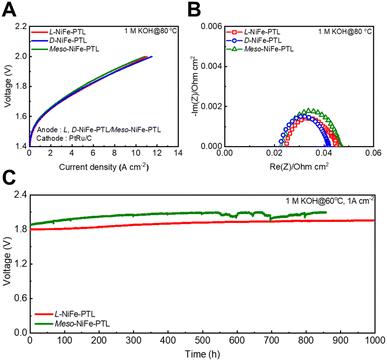
Next, to further extend spin selectivity into metal alloys and enhance the performance of spin-polarized AEMWEs, we synthesized chiral bimetallic Fe–Ni PTLs, which exhibit high electronic conductivity and are among the most promising OER catalysts. We then demonstrated spin-polarized AEMWEs based on these materials. For this purpose, we prepared chiral NiFe-PTL anodes using electrodeposition (see Methods). The performance of the AEMWE was measured at 80 °C in 1 M KOH. The spin-polarized AEMWE based on the chiral L-NiFe-PTL and D-NiFe-PTL (2.4 mg cm−2) exhibited current densities of 11.04 A cm−2 and 11.5 A cm−2 at 2.0 V, respectively, both of which are higher than those based on the meso-NiFe-PTL (10.83 A cm−2) (Fig. 5A). At 60 °C, the spin-polarized AEMWEs based on chiral L-NiFe-PTL and D-NiFe-PTL also showed higher current densities (7.06 A cm−2 for L-NiFe-PTL, 7.23 A cm−2 for D-NiFe-PTL) than the AEMWE based on meso-NiFe-PTL (6.85 A cm−2) (Fig. S11A).
The spin-polarized AEMWE with the L-NiFe-PTL and D-NiFe-PTL showed RCT values of 0.014 Ω cm−2 and 0.0136 Ω cm−2, respectively, both of which are lower than that of the spin-unpolarized AEMWE with meso-NiFe-PTL (RCT = 0.0177 Ω cm−2), indicating that the chiral NiFe-PTL induces more favorable interfacial charge transfer compared to the meso-NiFe-PTL (Fig. 5B). At 60 °C, AEMWEs with the L-NiFe-PTL and D-NiFe-PTL also showed lower RCT (0.0316 Ω cm−2 for L-NiFe-PTL, 0.0294 Ω cm−2 for D-NiFe-PTL) than the meso-NiFe-PTL (0.0336 Ω cm−2) (Fig. S11B). These results represent one of the highest performances reported so far among PGM-free anode AEMWEs to the best of our knowledge (Table S1). Therefore, we attribute the superior performance of the AEMWE based on chiral NiFe-PTL to the favorable interfacial charge transfer between the chiral NiFe electrocatalyst and electrolyte, as confirmed by lower RCT values than meso-NiFe-PTL observed in EIS measurements.
Furthermore, the spin-polarized AEMWE with L-NiFe-PTL showed lower operating bias (1.8 V) to achieve a current density of 1 A cm−2 compared to that with the meso-NiFe-PTL (1.88 V) due to the more favorable interfacial charge transfer in chiral NiFe-PTL (Fig. 5C). The spin-polarized AEMWE with L-NiFe-PTL exhibited more stable operation over 1000 hours with a lower voltage decay rate of 0.15 mV h−1 than did the spin-unpolarized AEMWE with meso-NiFe-PTL (stable operation up to 550 hours, voltage decay rate of 0.28 mV h−1) in 1 M KOH. Considering these results (spin-polarized AEMWE with chiral CoOx-PTL and chiral NiFe-PTL showed higher current density and durability than did spin-nonpolarized AEMWE ones) with the fact that chiral PTL exhibits reduced charge transfer resistance and suppresses H2O2 formation, we conclude that chiral electrocatalysts induce spin polarized current which enhances the overall performance of AEMWEs.
Conclusions
In summary, we have demonstrated an efficient spin-dependent alkaline anion exchange membrane water electrolyzer (AEMWE) by incorporating a spin polarization porous transport layer into the membrane electrode assembly (MEA). This chiral CoOx PTL-based spin-polarized AEMWE exhibited a current of 8.21 A cm−2 at 2.0 V, representing a remarkable 52% increase compared to the meso-CoOx PTL-based spin-unpolarized AEMWE. The chiral CoOx PTL provides additional active sites and enhances interfacial charge transfer between the catalyst and electrolyte, thereby increasing current density during electrocatalysis. Electrochemical analysis and measurement of H2O2 concentration further confirm that the chiral CoOx PTL suppresses H2O2 formation even after reconstruction, supporting the persistence of the CISS effect. Extending this strategy to bimetallic alloy electrocatalysts, the chiral NiFe-PTL showed a current density of 11.5 A cm−2 at 2.0 V, which is one of the highest performances so far reported among PGM-free anode AEMWEs to the best of our knowledge, and high durability (>1000 hours at 1 A cm−2). These results highlight a new approach for designing spin-polarized anodes, offering a promising pathway toward highly efficient and durable water electrolysis in AEMWE systems.Experimental
Materials
Cobalt chloride hexahydrate (CoCl2·6H2O, 98%), iron nitrate nonahydrate (Fe(NO3)3·6H2O, 98%), nickel nitrate hexahydrate (Ni(NO3)2·6H2O, 98%), L-tartaric acid (99.5%), D-tartaric acid (99.0%), meso-tartaric acid monohydrate (97.0%), and sodium carbonate (Na2CO3, 99.5%) were purchased from Sigma-Aldrich (USA). Potassium hydroxide (KOH, 85.0%) was purchased from Duksan Co., Ltd, South Korea. PtRu/C (20 wt% Ru, 40 wt% Pt) was obtained from Alfa Aesar, USA. Nickel foam (Ni-foam) (1.6 mm thickness, 110 ppi) was purchased from MTI Co., Ltd, USA. The nickel fiber plate porous transport layer (PTL) was purchased from Dioxide Materials, USA. Carbon paper PTL was purchased from SGL Carbon, Germany.Characterization
Absorption spectra were measured using a Jasco V-730 UV-Vis spectrophotometer. Circular dichroism (CD) measurements were carried out using a Jasco J-1500 spectrophotometer. The CD spectra were recorded between wavelengths of 300 and 500 nm at a scanning speed of 100 nm min−1.X-ray diffraction (XRD) and gazing incidence XRD (GIXRD) were measured using a Rigaku SmartLab (Japan). X-ray photoelectron spectroscopy (XPS) was conducted using a Thermo Fisher Scientific Theta probe VASE system (USA). Scanning electron microscopy (SEM) and elemental mapping images were obtained using a ZEISS GeminiSEM 560 with an SDD EDS system (Germany). Transmission electron microscopy (TEM), high-resolution transmission electron microscopy (HR-TEM), and elemental mapping images were acquired using a JEOL NEO ARM with 100 kV and a dual SDD EDS system (Japan).
Preparation of spin polarization cobalt oxide porous transport layer (CoOx-PTL) catalysts
For the synthesis of the chiral L-CoOx-PTL, D-CoOx-PTL and achiral meso-CoOx-PTL catalysts, 0.16 mmol of CoCl2·6H2O and 1.6 mmol of L-tartaric acid, D-tartaric acid, or meso-tartaric acid monohydrate were dissolved in 40 ml of DI (deionized) water.14 Subsequently, 4 mmol of Na2CO3 was added into the mixture to obtain a transparent solution. The solution was used as the electrolyte for electrochemical deposition on a working electrode. A three-electrode electrochemical cell was employed for the electrochemical deposition, with a silver/silver chloride (Ag/AgCl, 3.0 M) electrode as a reference and a platinum (Pt) electrode (25 mm2) as a counter electrode. For the deposition on the ITO substrate, a thin film was electrodeposited using galvanostatic chronopotentiometry (CP) mode at a current density of 0.1 mA cm−2 at room temperature with the potential reaching a minimum of 0.7 V over 30 min. For the deposition on the nickel fiber plate, a current density of 0.225 mA cm−2 was applied for 30 min (0.3 mg cm−2). The catalyst loading per geometric area was determined by measuring the weight difference of 1 cm × 1 cm nickel fiber plate before and after electrodeposition under completely dry conditions.Preparation of spin polarization nickel iron porous transport layer (NiFe-PTL) catalysts
In a typical synthesis of the chiral L-NiFe-PTL, D-NiFe-PTL and achiral meso-NiFe-PTL catalysts, 2 mmol Fe(NO3)3·9H2O, 1 mmol Ni(NO3)2·6H2O and 3 mmol of L-tartaric acid, D-tartaric acid or meso-tartaric acid monohydrate were dissolved in 50 ml of methanol and 2.5 ml DI water and stirred for 30 min.25 The solution was used as the electrolyte for electrochemical deposition on a working electrode. A three-electrode electrochemical cell was employed for the electrochemical deposition. For the deposition on the nickel fiber plate, a current density of −10 mA cm−2 was applied for 60 min (2.4 mg cm−2). After deposition, the samples were washed with DI water and ethanol and dried under vacuum conditions.Preparation of the membrane electrode assembly (MEA)
To measure the anion exchange membrane water electrolysis (AEMWE) performance, an electrodeposited anode with MEA was prepared. The nickel fiber plate was dipped into the prepared electrolyte and electrodeposited under the same conditions above. After the deposition, the sample was washed with DI water and dried under vacuum conditions. The cathode catalyst ink was prepared by mixing PtRu/C catalysts, ionomers, isopropanol, and DI water in a glass vial and ultrasonicated at 5 °C for an hour. The cathode was fabricated by hand-held spray coating with a catalyst loading amount of 0.7 mg cm−2 on the carbon paper PTL. A poly(aryl-co-aryl piperidinium) (PDTP-25) membrane with a thickness of approximately 30 µm was used as the AEM. Before testing, the AEM was soaked in 1 M KOH at 60 °C for 3 hours, refreshing the solution every hour. The assembly with an effective area of 5 cm2 was constructed with PTFE gaskets, PTL, bipolar plates, and end plates. Nickel fiber plate and carbon paper were used as the anode and cathode PTL, respectively. Gold-coated nickel plates and graphite plates were employed as anode and cathode bipolar plates, respectively.AEMWE measurements
AEMWE was evaluated using a single-cell test. Performance was measured using an electrochemical station (VSP, BioLogic SAS, France) with a current booster (VMP3 Booster, BioLogic SAS, France). The current range of the booster was 0 to 80 A. During the test, the anode was exposed to a circulating 1 M KOH at a flow rate of 36 mL min−1. The dry cathode was used to meet industrial standards. The I–V curves were measured by scanning the voltage from 1.3 to 2.0 V. Electrochemical impedance spectroscopy (EIS) was conducted at 1.6 V and 1.8 V in a frequency range of 1 Hz to 200 kHz at 60 °C and 80 °C with an amplitude of 50 mV. The stability of the AEMWE was evaluated using CP mode at a current density of 1 A cm−2.Electrochemical measurements
Electrochemical measurements were conducted using a potentiostat (BioLogic SP-200, France) with a three-electrode configuration. The catalysts loaded on Ni-foam were employed as the working electrode, Ag/AgCl was used as the reference electrode, and Pt was employed as the counter electrode. The applied voltage was converted into the reversible hydrogen electrode (RHE) scale using the equation:| ERHE = EAg/AgCl + 0.05916 × pH + 0.197 |
The scan rate of the linear sweep voltammetry (LSV) was 10 mV s−1. LSV was conducted in 1 M KOH. Electrochemical impedance spectroscopy (EIS) was analyzed in the frequency range between 100 mHz and 100 kHz at an AC amplitude of 10 mV. The Tafel slope was calculated by linear fitting based on the equation:
E = A + B![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) j j |
Hydrogen peroxide (H2O2) measurement
The generation of H2O2 during the OER was measured at 2.2 V (vs. RHE) using o-tolidine as the redox indicator. For the electrolyte, 0.1 M sodium sulfate (Na2SO4) solution with a pH of 6.55 was used. A 4 ml sample of reacted solution was taken every 30 min during the OER for 180 min and mixed with 0.8 ml of 0.1% (w/v) o-tolidine solution in 1 M HCl. If the mixture contains H2O2, it turns yellow, and the concentration can be measured using UV-vis spectroscopy. The solution was measured after being diluted 30-fold with D.I. water.Author contributions
T. H. Kim designed the study, performed experiments, analyzed data and prepared the manuscript. T. H. Kim, C. Hu and H. K. Cho evaluated the anion exchange membrane water electrolysis (AEMWE) performance. C. Hu was affiliated with the Department of Energy Engineering, Hanyang University at the time of the experiment and is currently affiliated with Southeast University, Nanjing, China. S. H. Jae and S. Lee assisted in the half-cell test. B. Yeom provided advice in the analysis of the chiroptical properties. Y. M. Lee and Y.-H. Kim designed and supervised the study, analyzed the data and prepared the manuscript. All authors discussed the results and commented on the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.Supplementary information: physical properties, chiroptical properties as well as additional OER performance data related to the AEMWE. See DOI: https://doi.org/10.1039/d5se01313e.
Acknowledgements
This work was supported by a National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (RS-2024-00408951, RS-2025-24534599 and RS-2025-02309471). This research was supported by the “Regional Innovation System & Education (RISE)” through the Seoul RISE Center, funded by the Ministry of Education (MOE) and the Seoul Metropolitan Government. (2025-RISE-01-027-04).References
- N. Du, C. Roy, R. Peach, M. Turnbull, S. Thiele and C. Bock, Chem. Rev., 2022, 122, 11830–11895 CrossRef CAS PubMed.
- F. Razmjooei, T. Morawietz, E. Taghizadeh, E. Hadjixenophontos, L. Mues, M. Gerle, B. D. Wood, C. Harms, A. S. Gago, S. A. Ansar and K. A. Friendrich, Joule, 2021, 5, 1776–1799 CrossRef CAS.
- D. Li, E. J. Park, W. Zhu, Q. Shi, Y. Zhou, H. Tian, Y. Lin, A. Serov, B. Zulevi, E. D. Baca, C. Fujimoto, H. T. Chung and Y. S. Kim, Nat. Energy, 2020, 5, 378–385 CrossRef CAS.
- F. A. Garcés-Pineda, J. Yu, C. A. Mesa, S. Plana-Ruiz, D. Ruano, Y. Liang, M. Lingenfelder, S. Gimenez and J. R. Galan-Mascaros, Chem. Sci., 2025, 13, 5475–5482 RSC.
- J. Zhang, J. Dong, Z. Li, J. Zhang and Z. Ma, Electron. Mater. Lett., 2025, 21, 228–234 CrossRef CAS.
- T. Wu, X. Ren, Y. Sun, S. Sun, G. Xian, G. G. Scherer, A. C. Fisher, D. Mandler, J. W. Ager, A. Grimaud, J. Wang, C. Shen, H. Yang, J. Gracia, H.-J. Gao and Z. J. Xu, Nat. Commun., 2021, 12, 3634 CrossRef CAS.
- X. Ren, T. Wu, Y. Sun, Y. Li, G. Xian, X. Liu, C. Shen, J. Gracia, H.-J. Gao, H. Yang and Z. J. Xu, Nat. Commun., 2021, 12, 2608 CrossRef CAS.
- B. P. Bloom, Y. Paltiel, R. Naaman and D. H. Waldeck, Chem. Rev., 2024, 124, 1950–1991 CrossRef CAS.
- R. Naaman, Y. Paltiel and D. H. Waldeck, Chem. Rev., 2020, 53, 2659–2667 CAS.
- Y.-H. Kim, Y. Zhai, H. Lu, X. Pan, C. Xiao, E. A. Gaulding, S. P. Harvey, J. J. Berry, Z. V. Vardeny, J. M. Luther and M. C. Beard, Science, 2021, 371, 1129–1133 CrossRef CAS.
- M. P. Hautzinger, X. Pan, S. C. Hayden, J. Y. Ye, Q. Jiang, M. J. Wilson, A. J. Philips, Y. Dong, E. K. Raulerson, I. A. Leahy, C.-S. Jiang, J. L. Blackburn, J. M. Luther, Y. Lu, K. Jungjohann, Z. V. Vardeny, J. J. Berry, K. Alberi and M. C. Beard, Nature, 2024, 631, 307–312 CrossRef CAS.
- M. Ai, L. Pan, C. Shi, Z.-F. Huang, X. Zhang, W. Mi and J.-J. Zou, Nat. Commun., 2023, 14, 4562 CrossRef CAS.
- W. Zhang, C. Jiang, H. Guan, Y. Wang, Y. Hu, W. Wang, W. Tian and L. Hao, Mater. Adv., 2024, 5, 1340–1347 RSC.
- S. Ghosh, B. P. Bloom, Y. Lu, D. Lamont and D. H. Waldeck, J. Phys. Chem. C, 2020, 124, 22610–22618 CrossRef CAS.
- J. Ran, M. Si and D. Gao, Chem. Eng. J., 2024, 493, 152545 CrossRef CAS.
- J. E. Park, S. Park, M.-J. Kim, H. Shin, S. Y. Kang, Y.-H. Cho and Y.-E. Sung, ACS Catal., 2022, 12, 135–145 CrossRef CAS.
- N. Chen, C. Hu, H. H. Wang, S. P. Kim, H. M. Kim, W. H. Lee, J. Y. Bae, J. H. Park and Y. M. Lee, Angew. Chem., Int. Ed., 2021, 60, 7710–7718 CrossRef CAS.
- N. Chen, S. Y. Paek, J. Y. Lee, J. H. Park, S. Y. Lee and Y. M. Lee, Energy Environ. Sci., 2021, 14, 6338–6348 RSC.
- N. Chen, H. H. Wang, S. P. Kim, H. M. Kim, W. H. Lee, C. Hu, J. Y. Bae, E. S. Sim, Y.-C. Chung, J.-H. Jang, S. H. Yoo, Y. Zhuang and Y. M. Lee, Nat. Commun., 2021, 12, 2367 CrossRef CAS.
- C. Hu, Y. J. Lee, Y. Ma, X. Zhang, S. W. Jung, H. Hwang, H. K. Cho, M.-G. Kim, S. J. Yoo, Q. Zhang and Y. M. Lee, ACS Energy Lett., 2024, 9, 1219–1227 CrossRef CAS.
- C. A. Mesa, F. A. Garces-Pineda, M. Garcia-Tecedor, J. Yu, B. Khezri, S. Plana-Ruiz, B. Lopez, R. Iturbe, N. Lopez, S. Gimenez and J. R. Galan-Mascaros, APL Energy, 2024, 2, 016106 CrossRef CAS.
- L. Reith, J. N. Hausmann, S. Mebs, I. Mondai, H. Dau, M. Driess and P. W. Menezes, Adv. Energy Mater., 2023, 13, 2203886 CrossRef CAS.
- Q. Bai, D. Liu, X. Yan, P. Guo, X. Ding, K. Xiang, X. Tu, Y. Guo and R. Wu, ACS Mater. Lett., 2024, 6, 2948–2956 CrossRef CAS.
- N. U. Hassan, Y. Zheng, P. A. Kohl and W. E. Mustain, J. Electrochem. Soc., 2022, 169, 044526 CrossRef CAS.
- T. Feng, W. Chen, J. Xue, F. Cao, Z. Chen, J. Ye, C. Xiao and H. Lu, Adv. Funct. Mater., 2023, 33, 2215051 CrossRef CAS.
Footnotes |
| † These authors contributed equally to this work. |
| ‡ Current address: Southeast University, Liangjiang W Rd, Jiangning District, Nanjing, Jiangsu 211102, China. |
| This journal is © The Royal Society of Chemistry 2026 |