Hollow Sb nanocrystals confined in N, S Co-doped carbon nanofibers boosting sodium-ion transport for high-performance sodium-ion batteries
Qiushi
Huang
a,
Yejian
Yu
a,
Rujia
Zou
 *a,
Jinqi
Zhu
*a and
Huifang
Chen
*b
*a,
Jinqi
Zhu
*a and
Huifang
Chen
*b
aState Key Laboratory of Advanced Fiber Materials, College of Materials Science and Engineering, Donghua University, Shanghai 201620, China. E-mail: rjzou@dhu.edu.cn; zhujinqi.dhu@foxmail.com
bKey Laboratory of High Performance Fibers & Products, Ministry of Education, Donghua University, Shanghai, 201620, P. R. China. E-mail: hfchen@dhu.edu.cn
First published on 20th November 2025
Abstract
Herein, N, S co-doped carbon fibers encapsulating hollow Sb nanocrystals (h-Sb@NS-CNFs) were synthesized by a simple ion exchange and electrospinning process. The hollow Sb nanocrystals, in conjunction with the confinement effect of the carbon fibers, can offer faster Na+ pathways, decrease the Na+ diffusion barriers, and effectively mitigate the structural degradation of the electrode caused by the volume changes of Sb, thereby extending the cycle life of batteries. Additionally, the dual-element co-doping strategy employing nitrogen and sulfur provides more active sites for the Na+ reaction and increases the electronic conductivity while simultaneously enhancing the ionic diffusion kinetics, as indicated by density functional theory (DFT) and kinetic analysis. Therefore, h-Sb@NS-CNF exhibits excellent cyclic stability (305.3 mAh g−1 at 2 A g−1 for 900 cycles) and a high-rate capacity (209.3 mAh g−1 at 10 A g−1) as an anode material for sodium-ion batteries.
1. Introduction
Sodium-ion batteries (SIBs) are a competitive substitute for lithium-ion batteries (LIBs) due to their lower cost and similar working principle to LIBs.1 Since the graphite carbon anodes used in commercial LIBs are not suitable for SIBs, many anode materials for SIBs have been developed, such as carbon-based materials, conversion-type materials and alloy-type materials.2–10 Among these anode materials, Sb has attracted much attention because of its high theoretical capacity (660 mAh g−1), suitable voltage platform (0.5 to 0.7 V vs. Na+/Na) and good electrical conductivity (2.56 × 106 S m−1).11–15 However, Sb undergoes a volume change of 390% during charging and discharging that will cause fracture of the electrode materials, resulting in reduced capacity and poor stability.16 Additionally, compared with Li+, the larger size of Na+ leads to slower diffusion during the charge and discharge processes, resulting in even slower reaction kinetics for the Sb anode in SIBs.17In order to overcome the disadvantages of volume change and the low reaction kinetics of Sb electrodes, many strategies have been designed, including the design of Sb–M alloys, composites with conductive carbon materials, and the design of hollow nanostructures.18,19 Carbon materials, such as carbon nanofibers (CNFs), graphene, carbon nanotubes, and porous carbon, are widely used for enhancing the electrical conductivity of SIB electrodes while also serving as a cushion for the electrode materials against volume changes.20–23 A hollow nanostructure design of anode materials has been widely employed for enhancing the reaction kinetics of Na+ and accommodating volume changes, thereby effectively improving the cycling stability and rate performance of SIBs.24–26
In recent years, CNF materials fabricated via electrospinning have exhibited superior mechanical characteristics, with inherent self-supporting properties and flexibility, indicating their potential applicability in the realm of flexible battery technology.27,28 Liu et al. designed porous Sb/CNF by electrospinning and a self-activation method, the structure of the porous carbon fibers confining ultrasmall Sb nanocrystals, which demonstrate good stability, also enhances the battery's rate capability.29 However, the huge volume change of Sb particles during the charge–discharge process can disrupt their integration with carbon materials, leading to poor cycling stability of SIBs. Therefore, stably encapsulating hollow Sb nanoparticles in CNFs by a simple method while ensuring excellent ionic diffusion kinetics remains challenging.
Herein, we used ZIF-8 as a precursor and synthesized N, S co-doped carbon fibers for encapsulating hollow Sb nanocrystals (h-Sb@NS-CNFs). The combination of the hollow nanostructured design and carbon fiber composite is an effective strategy for providing sufficient space for the volume expansion of Sb during sodiation and desodiation, improving the cycle stability of batteries. At the same time, the small particle size of h-Sb@NS-CNFs shorten the diffusion path of Na+, decrease the Na+ diffusion barriers, and increase the electronic conductivity. In addition, the N, S co-doped carbon fibers provide more adsorption sites for Na+, while also enhancing the ionic diffusion properties. As a result, h-Sb@NS-CNFs exhibits excellent cyclic stability of 373.8 mAh g−1 after 200 cycles at 0.5 A g−1 and 305.3 mAh g−1 after 900 cycles at 2 A g−1. Furthermore, h-Sb@NS-CNFs demonstrates outstanding rate performance of 209.3 mAh g−1 at 10 A g−1 in SIBs.
2. Experimental section
2.1 Synthesis of Sb2S3 nanoboxes
A mixture of 4.0 g 2-methylimidazole and 1.68 g Zn(NO3)2·6H2O in 80 mL methanol (total volume) was vigorously stirred for 1 h, then aged at room temperature for 12 h. The resulting suspension was centrifuged at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 5 min, washed with methanol, and vacuum-dried at 60 °C for 12 h to obtain ZIF-8 nanopolyhedrals. Forty milligrams of ZIF-8 was ultrasonically dispersed in 30 mL ethanol for 10 min, heated to 120 °C in an oil bath, and then treated with a mixture of 2.0 g thioacetamide in 20 mL ethanol for 3 h under reflux. After cooling, the ZnS nanobox suspension was centrifuged (12
000 rpm for 5 min, washed with methanol, and vacuum-dried at 60 °C for 12 h to obtain ZIF-8 nanopolyhedrals. Forty milligrams of ZIF-8 was ultrasonically dispersed in 30 mL ethanol for 10 min, heated to 120 °C in an oil bath, and then treated with a mixture of 2.0 g thioacetamide in 20 mL ethanol for 3 h under reflux. After cooling, the ZnS nanobox suspension was centrifuged (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 5 min), washed with ethanol, and redispersed in 20 mL ethanol. This suspension was combined with a solution of 40 mg SbCl3 in 20 mL ethanol under magnetic stirring for 2 h, then centrifuged (12
000 rpm, 5 min), washed with ethanol, and redispersed in 20 mL ethanol. This suspension was combined with a solution of 40 mg SbCl3 in 20 mL ethanol under magnetic stirring for 2 h, then centrifuged (12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 5 min) and washed with ethanol to yield yellow Sb2S3 nanoboxes.
000 rpm, 5 min) and washed with ethanol to yield yellow Sb2S3 nanoboxes.
2.2 Preparation of Sb2S3@PAN
The obtained Sb2S3 nanoboxes were added to 3 mL DMF in a beaker, followed by ultrasonic dispersion for 30 minutes, resulting in the formation of a yellow-colored dispersed liquid. Following this step, 0.4 g PAN was added into the liquid and stirred for 12 h at room temperature, achieving a homogeneous spinning dope. The spinning dope was loaded into a 5-mL plastic syringe with a 21-gauge tip needle for electrospinning. The voltage, feeding rate, and the distance between the collector and stainless-steel needle were fixed at 12 kV, 0.15 mL h−1 and 20 cm, respectively.2.3 Preparation of h-Sb@NS-CNFs
The Sb2S3@PAN composite fibers were subjected to an annealing process under an Ar atmosphere. The fibers were heated at a uniform rate of 5 °C per minute from room temperature to a final temperature of 530 °C, which was maintained for 2 hours. The resulting h-Sb@NS-CNFs composite fibers, which were then collected for further use or analysis, exhibited self-supporting properties and maintained good flexibility.2.4 Preparation of Sb@NS-CNFs and Sb@CNP
The synthesis of Sb@NS-CNFs was similar to that of h-Sb@NS-CNFs, except that Sb2S3 was replaced with SbCl3 during the electrospinning process, and 100 mg of sulfur powder was added during the carbonization process to achieve N and S co-doping. For the preparation of Sb@CNP, 194 mg of ammonium persulfate was dissolved in 50 mL of deionized water, followed by the addition of Sb2S3 and ultrasonic dispersion. Subsequently, 0.2 mL of pyrrole was added, and the mixture was stirred for 12 h. The resulting product was centrifuged, dried, and annealed under the same conditions as h-Sb@NS-CNFs.2.5 Material characterization
Field-emission scanning electron microscopy (FESEM, Hitachi S-4800) and high-resolution transmission electron microscopy (TEM, JEOL JEM-2100 F equipped with an energy dispersive X-ray spectrometer) were performed to investigate the morphologies and structures of the samples. Powder XRD patterns were acquired using an X-ray diffractometer D/MAX-2550VB+ to confirm the components and the phases of the composites. XPS data for exploring the chemical valence states were obtained using an EALALab MIKII spectrometer with a Mg-Kα excitation source. The carbon content of the samples was determined by thermogravimetric analysis (TGA, Libra/209F1).2.6 Electrochemical measurements
The electrochemical performance was tested in half 2032 coin-type cells with sodium metal foil as the counter and reference electrodes. A glass fiber membrane (Guangdong Canrd New Energy Technology Co., Ltd) acted as the separator. The organic electrolyte for the half-cells was composed of 1 M NaPF6/DME. The fabrication of the coin cells was completed in an argon-filled glovebox (with both the moisture and the oxygen concentration below 0.1 ppm). Galvanostatic discharge–charge measurements and GITT were performed with a LANHE battery testing system (LAND2001A) within 0.01 to 3.0 V. Cyclic voltammetry (CV, voltage range from 0.01 to 3.0 V) and electrochemical impedance spectroscopy (EIS, frequency range from 100 kHz to 0.01 Hz) were performed on an Autolab (PGSTA302N) electrochemical workstation.2.7 Computational details
The density functional theory (DFT) calculations were performed using the CP2K package based on the hybrid Gaussian and plane-wave scheme,30 with the generalized gradient approximation (GGA) and Perdew–Burke–Ernzerhof (PBE) functional, which were used to describe electron exchange and correlation.31 A plane-wave density cutoff of 800 Ry was adopted, and the vdWs interactions were included by using an empirical DFT-D3(BJ) method.32 During structure optimization, the molecular orbitals of the valence electrons were expanded into the DZVP-MOLOPT-SR-GTH basis sets,33 whereas the atomic core electrons were described using Goedecker–Teter–Hutter (GTH) pseudopotentials.34 In all the calculations, a k-point sampling of 2 × 2 × 1 was employed. The illustrations of the structures and visualizations of the charge densities were generated using VESTA.35 The adsorption energy Eads was defined as:| Eads = Esub+Na − Esub − ENa |
3. Results and discussion
The simple fabrication of h-Sb@NS-CNFs is shown in detail in Fig. 1a. Firstly, hollow Sb2S3 nanoboxes were obtained by an ion exchange method using ZIF-8 nanopolyhedrals as the templates. The h-Sb2S3 nanoboxes were then mixed with a PAN solution to synthesize the h-Sb2S3@PAN nanofibers via electrospinning. Finally, the h-Sb2S3@PAN nanofibers were calcined under Ar atmosphere. During carbonization of the PAN fibers, the h-Sb2S3 nanoboxes were carbothermally reduced to hollow Sb nanocrystals and then encased in the carbon nanofibers, forming the h-Sb@NS-CNFs. During the carbonization process, a portion of the nitrogen atoms from the cyano groups in PAN was retained within the CNFs. Meanwhile, during the reduction process, sulfur atoms from Sb2S3 diffused into the CNFs, ultimately forming a N, S co-doped structure. | ||
| Fig. 1 (a) Schematic of the formation of the h-Sb@NS-CNFs. (b) SEM images of the h-Sb@NS-CNFs. (c) TEM image and (d) HRTEM image of the h-Sb@NS-CNFs. | ||
The morphologies of the ZIF-8 nanopolyhedrals and h-Sb2S3 nanoboxes were characterized by scanning electron microscopy (SEM) and transmission electron microscopy (TEM), as shown in Fig. S1. The ZIF-8 nanopolyhedrals have a uniform morphology with an average particle size of ∼150 nm (Fig. S1a). After the in situ sulfurization process, the h-Sb2S3 nanoboxes with a void inner space and size of approximately 200 nm exhibit morphologies akin to that of the ZIF-8 nanopolyhedrals (Fig. S1b–d). The SEM images of h-Sb@NS-CNFs (Fig. 1b and S2) demonstrate that the h-Sb@NS-CNFs possess a smooth surface with an average diameter of approximately 400 nm. There were no obvious fractures or damage on the fiber, indicating that carbonization did not cause the collapse of the fiber structure or the precipitation of active Sb. The TEM images in Fig. 1c and d clearly demonstrate that the h-Sb nanocrystals were successfully prepared and confined within the void space in the nanofibers provided by the hollow Sb2S3 nanoboxes. The high-resolution TEM (HRTEM) image (Fig. 1d) displays a 0.31-nm lattice spacing, clearly corresponding to the (0 1 2) plane distance of Sb. Energy dispersive X-ray spectroscopy (EDX) elemental mapping (Fig. S3) reveals that the h-Sb nanocrystals were encapsulated within the box-like spaces inside the fibers. The Sb@NS-CNFs were synthesized by replacing the hollow Sb2S3 with SbCl3. The SEM and TEM images reveal that the Sb nanoparticles were encapsulated within the solid CNFs, which exhibited good structural uniformity. The ZIF8-derived Sb2S3 nanoboxes were coated with polypyrrole (PPy) by a self-assembly method. Sb2S3@PPy was calcined to obtain a carbon layer, and the Sb2S3 nanoboxes were reduced to Sb nanocrystals in this process. The SEM and TEM images (Fig. S5) show that the Sb nanocrystals were clad by a carbon layer, forming Sb@CNP with an average diameter of ∼200 nm.
The XRD results of h-Sb@NS-CNFs and Sb@CNP are displayed in Fig. 2a. These peaks are perfectly consistent with the standard patterns of Sb (JCPDS:35-0732), indicating that Sb2S3 in CNFs was reduced to Sb during carbonization. The h-Sb@NS-CNFs were further characterized by Raman spectroscopy (Fig. 2b). The peak at 147.0 cm−1 was assigned to the A1g vibration mode of the characteristic Sb peak, thus indicating the existence of Sb in the CNFs. The two distinct peaks at 1358.8 cm−1 and 1594.0 cm−1 can be assigned to the D and G bands of the CNFs. The D band represents the stretching and vibration of the disordered carbon layer with sp3 hybrid orbitals, corresponding to the degree of amorphous carbon in the fibers; the G band is the vibration peak of the ordered carbon layer with sp2 hybrid orbitals, corresponding to the degree of graphitization of carbon in the fibers. The ID/IG ratio of 1.40 is primarily attributed to the doping effects caused by nitrogen and sulfur from PAN and Sb2S3, respectively. The electronic conductivity of h-Sb@NS-CNFs annealed at different temperatures was evaluated by chronoamperometric polarization measurements (Fig. S7). The carbonized fibers obtained at 530 °C exhibited an electronic conductivity of 1.27 S cm−1, indicating that this temperature was sufficient for endowing the fibers with excellent electrical conductivity.
 | ||
| Fig. 2 (a) XRD patterns of the h-Sb@NS-CNFs and Sb@CNP. (b) Raman spectra of the h-Sb@NS-CNFs. (c–f) XPS spectra of the h-Sb@NS-CNFs. | ||
The chemical compositions and bonding structure were further analyzed via XPS analysis. Fig. S8 shows the full spectrum of h-Sb@NS-CNFs; the peaks of C 1s, N 1s, S 2p and Sb 3d illustrate that the elements C, N, S and Sb were present in h-Sb@NS-CNFs. The two peaks in the high-resolution C 1s spectrum (Fig. 2c) can be assigned to the C–C bonds with sp3 (283.3 eV) and sp2 (284.8 eV) hybridizations.37 Moreover, the peak at 286.2 eV was assigned to the C–N bond. The peaks at 396.7 eV and 398.3 eV for N 1s (Fig. 2d) correspond to the pyridinic nitrogen and pyrrolic nitrogen, revealing nitrogen doping in the carbon layer.38 In the high-resolution S 2p spectrum (Fig. 2e), the peaks at 168.5 eV and 167.3 eV correspond to sulfur oxides, such as SO3 and SO4, while the peaks at 164.8 eV and 163.9 eV correspond to C–S covalent bonds, and the peaks at 162.8 eV and 161.7 eV correspond to metal sulfides.39 The doping with nitrogen and sulfur introduced a multitude of defects into the carbon fibers, which were conducive for enhancing the transport of electrons and ions.40 Additionally, doping provided a greater number of active sites for the material. There are three peaks in the high-resolution Sb 3d spectrum (Fig. 2f), which can be assigned to Sb 3d5/2 (528.8 eV), Sb 3d3/2 (538.3 eV) and O 1s (530.1 eV).41 The peak of O 1s can be attributed to the existence of an oxide film on the surface of the carbon fibers.
To investigate the influence of N and S doping on the electrochemical properties of h-Sb@NS-CNFs for SIBs, density functional theory (DFT) was used for analyzing the adsorption of Na on the different doping models and their diffusion behavior (Fig. S16). Fig. 3a shows the top views of the optimized geometric configurations of Na adsorbed on the graphitic nitrogen (NG)–, pyridinic nitrogen (N6)–, pyrrolic nitrogen (N5)–, sulfur (S)– and nitrogen–sulfur (N6–S and N5–S)-doped graphene structures. The adsorption energies of Na on the doping models of NG, N6, N5 and S were −0.50 eV, −1.49 eV, −2.51 eV, and −1.32 eV, respectively (Fig. 3c). In contrast, the nitrogen-sulfur-co-doped models, (N6–S) and (N5–S) exhibited higher adsorption energies of −1.91 eV and −2.57 eV, indicating that the introduction of sulfur enhanced the adsorption of Na, which could have improved their charge–discharge capacity. The differential charge density maps of N5–S for Na adsorption confirmed the transfer of electrons from the sodium atom to the sulfur and nitrogen atoms in N5–S (Fig. 3b). The Bader charge analysis confirmed that the co-doping of N and S jointly promoted the adsorption of Na (Fig. S17). The diffusion behavior of Na migrating from the stable state to the metastable state on the material surface was further investigated, and the energy profiles of Na along the diffusion path were determined (Fig. 3d and e). The diffusion energy barriers of N5–S and N6–S were observed to be lower than those of N5 and N6 (0.83 vs. 0.90 eV and 0.42 vs. 0.65 eV respectively), indicating that the N, S co-doping structures were more favorable for Na diffusion than pure N doping, and that they decreased the Na+ diffusion barriers between Na+ and the active materials, thereby allowing for a better rate performance.
The electrochemical properties of the h-Sb@NS-CNFs composite as an anodic material for SIBs were evaluated. Fig. 4a displays a representative cyclic voltammogram (CV) for the first, second and third cycles at a scan rate of 0.1 mV s−1 in the voltage window of 0.01–3 V vs. Na/Na+.42 The first CV curve exhibits two obvious cathodic peaks located at 0.57 V and 0.41 V, which did not appear in subsequent cycles. These irreversible peaks can be ascribed to electrolytic decomposition on the electrode surface of h-Sb@NS-CNFs and the formation of the SEI film.43 In addition, the peak at 0.42 V in the first cathodic scan was attributed to the alloying process: Sb + xNa+ + xe− ⇔ NaxSb (x < 3). There are two reversible peaks at 0.46 V and 1.44 V in the subsequent cathodic scans, which can be ascribed to the multi-step alloying process: NaxSb + (3 − x)Na+ + (3 − x)e− ⇔ Na3Sb (x < 3). The sharp peak observed at 0.01 V following cathode scanning and the peak at 0.05 V following anode scanning correspond to the intercalation and extraction of Na+ in the carbon layer. The oxidation peaks at 0.79 V and 0.89 V can be ascribed to the dealloying process: Na3Sb → NaxSb → Sb. The high coincidence of oxidation peaks during the three anodic scans indicates that the h-Sb@NS-CNFs electrode exhibits good reversibility and little polarization. With the exception of the first circle, the subsequent voltage platforms basically overlapped, which confirms that the CV results, indicating that the material has excellent reversibility and electrical conductivity.44 The CD curve (Fig. S9) displays a distinct charge–discharge plateau, which corresponds to the peak positions in the CV curve.
The rate performance of h-Sb@NS-CNFs, Sb@NS-CNFs and Sb@CNP in SIBs was tested at different current densities in the voltage range from 0.01 V to 3.0 V. As shown in Fig. 4b, h-Sb@NS-CNFs delivered specific capacities of 415.7, 382.5, 357.1, 341.5, 331.4, 320.9 and 320.7 mAh g−1 at current densities of 0.1, 0.2, 0.5, 1, 2, 5 and 10 A g−1 respectively. When the current density was returned to 0.1 A g−1, the reversible specific capacity increased to 379.5 mAh g−1. h-Sb@NS-CNFs exhibited outstanding performance among the various Sb-based, anode materials (Table S1). In contrast, Sb@NS-CNFs and Sb@CNP demonstrated poor stability, with decreasing specific capacities during the cycles at 0.1 A g−1. Moreover, the cycling performance (0.5 A g−1, 0.01 V to 3.0 V) of h-Sb@NS-CNFs, Sb@NS-CNFs and Sb@CNP are shown in Fig. 4c. The batteries were activated at 0.1 A g−1, with an initial coulombic efficiency of 64.1%. The initial capacity loss and low coulombic efficiency can be ascribed to the formation of the SEI layer on the surface of the battery's electrode. h-Sb@NS-CNFs delivered specific capacities of 370.4 mAh g−1 and 373.8 mAh g−1 after 5 and 200 cycles, respectively, with a capacity retention ratio of 100.7%, revealing the good cycle stability of h-Sb@NS-CNFs. The dQ/dV curves of h-Sb@NS-CNFs from the 50th to 200th cycle nearly overlapped, further confirming the excellent cycling stability of the electrode (Fig. S10). In comparison, Sb@NS-CNFs and Sb@CNP exhibited low-capacity retention rates of 72.5% and 78.1%, respectively, at a current density of 0.5 A g−1. The long cycling test of h-Sb@NS-CNFs, Sb@NS-CNFs and Sb@CNP at 2 A g−1 is shown in Fig. 4d. The initial charge and discharge capacities of h-Sb@NS-CNFs were 596.1 mAh g−1 and 782 mAh g−1 with an initial coulombic efficiency of 76.2%. h-Sb@NS-CNFs delivered a specific capacity of 305.3 mAh g−1 after 900 cycles. Compared with the initial capacity of 305.3 mAh g−1, the capacity retention ratio reached 78.8%. In contrast, Sb@NS-CNFs exhibited a significantly lower capacity than h-Sb@NS-CNFs, while Sb@CNP showed a rapid capacity decay during cycling.
To further understand the improvement in the electrochemical performance of the as-designed h-Sb@NS-CNFs electrode, especially its cycling stability at high current density, a series of analyses for evaluating the electrochemical kinetics were performed. Firstly, to understand the diffusion of Na+ and electrons in the h-Sb@NS-CNFs electrode, CV measurements at different scan rates were performed. As the scanning rate increased, the response of current to voltage became more pronounced (Fig. 5a and b). The oxidation and cathodic peaks are clearly visible with a slight shift at high scanning rate. The contributions of diffusion (k1ν) and capacitance (k2ν1/2) can be calculated from i = aνb or log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i = blog
i = blog![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ν + log
ν + log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a.45 In these equations, i and ν represent the peak current and the corresponding scanning rate, respectively, while a and b are constants. The b constant can be calculated by a linear fitting of log(i) and log(ν). A b value close to 0.5 corresponds to a diffusion-controlled process, while a value approaching 1.0 indicates capacitive behavior. The b values of the anode and cathode were 0.549 and 0.728, proving that both diffusion and capacitance are present in the electrochemical reaction for the h-Sb@NS-CNFs electrode. As shown in Fig. S11, the CV curve at 0.6 mV s−1 corresponds to a green area, which the capacitive contribution corresponds to a blue area. The area ratio obtained by integrating the capacitance curve with the CV curve is the capacitance contribution value.46 The capacitive contribution ratio of the material at different scanning rates was accurately calculated using the following formula: i = k1ν + k2ν1/2. Fig. 5c shows that the values of the capacitance contribution were 76%, 81%, 83%, 86% and 87% at 0.2 mV s−1, 0.4 mV s−1, 0.6 mV s−1, 0.8 mV s−1 and 1.0 mV s−1, respectively, suggesting that a capacitive process dominated the charge and discharge processes of h-Sb@NS-CNFs. The relatively high capacitive contribution of the h-Sb@NS-CNFs electrode is attributable to its hollow nanostructure and highly conductive N, S co-doped carbon framework.
a.45 In these equations, i and ν represent the peak current and the corresponding scanning rate, respectively, while a and b are constants. The b constant can be calculated by a linear fitting of log(i) and log(ν). A b value close to 0.5 corresponds to a diffusion-controlled process, while a value approaching 1.0 indicates capacitive behavior. The b values of the anode and cathode were 0.549 and 0.728, proving that both diffusion and capacitance are present in the electrochemical reaction for the h-Sb@NS-CNFs electrode. As shown in Fig. S11, the CV curve at 0.6 mV s−1 corresponds to a green area, which the capacitive contribution corresponds to a blue area. The area ratio obtained by integrating the capacitance curve with the CV curve is the capacitance contribution value.46 The capacitive contribution ratio of the material at different scanning rates was accurately calculated using the following formula: i = k1ν + k2ν1/2. Fig. 5c shows that the values of the capacitance contribution were 76%, 81%, 83%, 86% and 87% at 0.2 mV s−1, 0.4 mV s−1, 0.6 mV s−1, 0.8 mV s−1 and 1.0 mV s−1, respectively, suggesting that a capacitive process dominated the charge and discharge processes of h-Sb@NS-CNFs. The relatively high capacitive contribution of the h-Sb@NS-CNFs electrode is attributable to its hollow nanostructure and highly conductive N, S co-doped carbon framework.
The galvanostatic intermittent titration technique (GITT) was adopted to further reveal the electrochemical kinetics of Na+ storage behavior of the h-Sb@NS-CNFs and Sb@CNP electrodes (Fig. 5d and e), which can be confirmed by the equation:.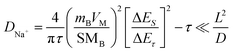 47,48 In the above equation D+Na, mB, VM, S, ΔES and ΔEτ stand for the diffusion coefficient of Na+, the active mass, the molar volume, the electrochemically active area of electrode, the open-circuit voltage difference after two adjacent relaxations and the voltage difference between the start and end of a single pulse step, respectively. As illustrated in Fig. 5f and S12, the Na+ diffusion coefficient of h-Sb@NS-CNFs was superior to that of Sb@CNP, primarily ranging from 10−15 to 10−8 cm2 s−1. This can be attributed to the unique structure of h-Sb@NS-CNFs, which offered an abundance of reactive sites, thereby shortening the Na+ transport pathway and effectively enhancing the reaction kinetics.
47,48 In the above equation D+Na, mB, VM, S, ΔES and ΔEτ stand for the diffusion coefficient of Na+, the active mass, the molar volume, the electrochemically active area of electrode, the open-circuit voltage difference after two adjacent relaxations and the voltage difference between the start and end of a single pulse step, respectively. As illustrated in Fig. 5f and S12, the Na+ diffusion coefficient of h-Sb@NS-CNFs was superior to that of Sb@CNP, primarily ranging from 10−15 to 10−8 cm2 s−1. This can be attributed to the unique structure of h-Sb@NS-CNFs, which offered an abundance of reactive sites, thereby shortening the Na+ transport pathway and effectively enhancing the reaction kinetics.
Electrochemical impedance spectroscopy (EIS) was performed to study the interfacial impedance and electrochemical kinetics of the h-Sb@NS-CNFs and Sb@CNP electrodes. The Nyquist plots of h-Sb@NS-CNFs and Sb@CNP after 1 cycle, 100 cycles and 500 cycles are shown in Fig. S13, 5g and h, respectively. The EIS profiles are characterized by a semicircle in the high-frequency domain and a linear segment in the low-frequency domain. These features are indicative of the electron-transfer resistance (Rct) and Warburg impedance associated with Na+ diffusion (Zw), respectively. Upon completion of 100 and 500 cycles, a smaller semicircle of h-Sb@NS-CNFs was observed, in comparison to that of Sb@CNP, suggesting accelerated charge transfer and a more stable electrochemical interface for the h-Sb@NS-CNFs electrode. Furthermore, the slope of the linear portion of the Nyquist plot at low frequencies for h-Sb@NS-CNFs was close to 1, substantiating the lower Warburg impedance, indicating enhanced Na+ diffusion within the h-Sb@NS-CNFs. The hollow structure of the Sb nanoboxes and the protection provided by the N, S co-doped carbon fibers ensured that h-Sb@NS-CNFs maintained excellent electrical conductivity and the Na+ diffusion rates even after 500 cycles. To further characterize the interfacial electrochemical behavior, DRT analysis was performed on the impedance spectra of h-Sb@NS-CNFs and Sb@CNP after 100 and 500 cycles (Fig S12). The relaxation peak area of h-Sb@NS-CNFs in the mid-frequency region was smaller than that of Sb@CNP, indicating a lower charge-transfer resistance. Furthermore, the EIS spectra of h-Sb@NS-CNFs after 500 cycles confirm the material's robust stability throughout the cyclic charge–discharge process. Finally, to further understand the morphology of h-Sb@NS-CNFs after 900 cycles and Sb@CNP after 500 cycles, SEM images are shown in Fig. S15. h-Sb@NS-CNFs maintains a complete fiber structure and diameter after 900 cycles without fracture and Sb leakage. The stability of the hollow fiber encased structural confirmed that the carbon fibers inhibited the volume expansion of Sb. In contrast, Sb@CNP was unable to maintain structural integrity, forming a large amount of crushed Sb nanocrystals, which indicated that the carbon layer was broken up during the alloying processes of Sb. Based on this comprehensive investigation, the h-Sb@NS-CNFs can maintain electrical interconnectivity throughout the entire measured area and it effectively relieved volume expansion, showing superior ion and electron transfer kinetics, which contribute to its superior cycle stability.
4. Conclusion
In summary, we developed N, S co-doped carbon fibers encapsulating hollow Sb nanocrystals through hydrothermal and electrostatic spinning methods. Characterizations revealed that sufficient space in the hollow nanostructure of h-Sb@NS-CNFs can alleviate size expansion and prevent cracking and fracture of the electrode during sodiation and desodiation. Furthermore, the large specific surface area conferred by the hollow structure of the composite not only provides a large number of active sites, but also shortens the Na+ diffusion route. Particularly, the three-dimension interconnection and conductive network of the N, S co-doped carbon nanofibers provide more adsorption sites for Na+, while also accelerating ion transfer at the interface. As a result, the h-Sb@NS-CNFs electrode exhibits extremely good cycling stability and rate performance in SIBs. The nanostructured design and dual-element co-doping strategy can be extended to other high-performance electrode materials.Author contributions
Qiushi Huang: conceptualization, methodology, validation, formal analysis, data curation, visualization, writing – original draft, writing – review & editing. Yejian Yu: conceptualization, methodology, formal analysis, investigation. Jinqi Zhu: conceptualization, validation. Rujia Zou: funding acquisition, writing-review & editing. Huifang Chen: writing-review & editing.Conflicts of interest
The authors declare no conflict of interest.Data availability
All relevant data are available within the manuscript and its additional files.Supplementary information (SI): additional characterization data and electrochemical testing results. See DOI: https://doi.org/10.1039/d5se01268f.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (grant no. 52372281), the Fundamental Research Funds for the Central Universities (2232025G-02), and the Program for Professor of Special Appointment (Eastern Scholar) at Shanghai Institutions of Higher Learning. Q. Huang and Y. Yu contributed equally to this work.References
- J. Y. Hwang, S. T. Myung and Y. K. Sun, Chem. Soc. Rev., 2017, 46, 3529–3614 RSC.
- L. Q. Zhu, K. L. Xu, Y. Y. Fang, X. J. Wu, L. Song, Y. C. Zhu and Y. T. Qian, Adv. Funct. Mater., 2023, 33, 202302444 Search PubMed.
- G. Y. Ren, T. T. Tang, S. S. Song, Y. X. Li, J. Y. Gao, Y. T. Wang, Z. J. Yao, S. H. Shen, L. Q. Zhang, Y. N. Guo and Y. F. Yang, ACS Appl. Mater. Interfaces, 2024, 16, 18991–19002 CrossRef CAS PubMed.
- Y. Chu, J. Zhang, Y. B. Zhang, Q. Li, Y. R. Jia, X. M. Dong, J. Xiao, Y. Tao and Q. H. Yang, Adv. Mater., 2023, 35, 202212186 Search PubMed.
- J. Zhang, M. Xiao, T. Liu, Y. Meng, F. Zhu and Z. Fan, Green Chem., 2024, 26, 5546–5555 RSC.
- M. Xiao, D. Qi, H. Sun, Y. Meng and F. Zhu, Inorg. Chem. Front., 2024, 11, 4107–4140 RSC.
- R. Lin, M. Xiao, Y. Xu, L. Zeng, F. Zhu, Y. Zhang and Y. Meng, J. Energy Storage, 2024, 92, 112291 CrossRef.
- M. Xiao, B. Ma, H. Zhang, X. Li, Q. Wang, Y. Peng and H. Zhang, J. Mater. Chem. A, 2022, 10, 21492–21502 RSC.
- M. Xiao, H. Zhang, B. Ma, Z. Zhang, X. Li, Q. Xiao, Q. Wang, Y. Peng and H. Zhang, J. Mater. Chem. A, 2021, 9, 24548–24559 RSC.
- M. Xiao, B. Ma, Z. Zhang, Q. Xiao, X. Li, Z. Zhang, Q. Wang, Y. Peng and H. Zhang, J. Mater. Chem. A, 2021, 9, 7227–7237 RSC.
- M. He, K. Kraychyk, M. Walter and M. V. Kovalenko, Nano Lett., 2014, 14, 1255–1262 CrossRef CAS PubMed.
- Y. T. Yuan, S. Jan, Z. Y. Wang and X. B. Jin, J. Mater. Chem. A, 2018, 6, 5555–5559 RSC.
- X. Xu, L. Si, X. S. Zhou, F. Z. Tu, X. S. Zhu and J. C. Bao, J. Power Sources, 2017, 349, 37–44 CrossRef CAS.
- A. G. Nguyen, H. T. T. Le, R. Verma, D. L. Vu and C. J. Park, Chem. Eng. J., 2022, 429, 132359 CrossRef CAS.
- P. X. Li, X. Guo, S. J. Wang, R. Zang, X. M. Li, Z. M. Man, P. Li, S. S. Liu, Y. H. Wu and G. X. Wang, J. Mater. Chem. A, 2019, 7, 2553–2559 RSC.
- M. M. Lao, Y. Zhang, W. B. Luo, Q. Y. Yan, W. P. Sun and S. X. Dou, Adv. Mater., 2017, 29, 1700622 CrossRef PubMed.
- S. Y. Qiao, Q. W. Zhou, M. Ma, H. K. Liu, S. X. Dou and S. K. Chong, ACS Nano, 2023, 17, 11220–11252 CrossRef CAS.
- J. Liu, Y. R. Wen, P. A. van Aken, J. Maier and Y. Yu, Nano Lett., 2014, 14, 6387–6392 CrossRef CAS PubMed.
- J. Liu, Z. Z. Yang, J. Q. Wang, L. Gu, J. Maier and Y. Yu, Nano Energy, 2015, 16, 389–398 CrossRef CAS.
- Y. J. Zhu, X. G. Han, Y. H. Xu, Y. H. Liu, S. Y. Zheng, K. Xu, L. B. Hu and C. S. Wang, ACS Nano, 2013, 7, 6378–6386 CrossRef CAS PubMed.
- Y. E. Xiang, X. Y. Hu, X. Zhong, W. T. Deng, G. Q. Zou, H. S. Hou and X. B. Ji, Adv. Funct. Mater., 2024, 34, 2311478 CrossRef CAS.
- W. X. Wang, Y. J. Wang, Z. Q. Jiang, B. L. Deng and Z. J. Jiang, J. Mater. Chem. A, 2023, 11, 21561–21568 RSC.
- L. Yu, G. Q. Zhang, C. Z. Yuan and X. W. Lou, Chem. Commun., 2013, 49, 137–139 RSC.
- N. N. Wang, Z. C. Bai, Y. T. Qian and J. Yang, Adv. Mater., 2016, 28, 4126–4133 CrossRef CAS.
- Z. M. Liu, X. Y. Yu, X. W. Lou and U. Paik, Energy Environ. Sci., 2016, 9, 2314–2318 RSC.
- B. Selvaraj, C. C. Wang, Y. F. Song, H. S. Sheu, Y. F. Liao and N. L. Wu, J. Mater. Chem. A, 2020, 8, 22620–22625 RSC.
- C. L. Zhao, Y. X. Lu, L. Q. Chen and Y. S. Hu, Infomat, 2020, 2, 126–138 CrossRef CAS.
- J. Wang, Z. Z. Wang, J. F. Ni and L. Li, Energy Storage Mater., 2022, 45, 704–719 CrossRef.
- Z. M. Liu, H. R. Sun, X. J. Wang, Z. Y. Gu, C. M. Xu, H. F. Li, G. X. Zhang, Y. He and X. L. Wu, Energy Storage Mater., 2022, 48, 90–100 CrossRef.
- J. Hutter, M. Iannuzzi, F. Schiffmann and J. VandeVondele, Wiley Interdiscip. Rev.:Comput. Mol. Sci., 2014, 4, 15–25 CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1997, 78, 3865–3868 CrossRef.
- S. Grimme, S. Ehrlich and L. Goerigk, J. Comput. Chem., 2011, 32, 1456–1465 CrossRef CAS PubMed.
- J. VandeVondele and J. Hutter, J. Chem. Phys., 2007, 127, 114105 CrossRef PubMed.
- S. Goedecker, M. Teter and J. Hutter, Phys. Rev. B:Condens. Matter Mater. Phys., 1996, 54, 1703–1710 CrossRef CAS PubMed.
- K. Momma and F. Izumi, J. Appl. Crystallogr., 2008, 41, 653–658 CrossRef CAS.
- G. Henkelman, B. P. Uberuaga and H. Jónsson, J. Chem. Phys., 2000, 113, 9901–9904 CrossRef CAS.
- Y. Liu, Y. Qing, B. Zhou, L. D. Wang, B. Pu, X. F. Zhou, Y. B. Wang, M. Z. Zhang, J. Bai, Q. Tang and W. Q. Yang, ACS Nano, 2023, 17, 2431–2439 CrossRef CAS PubMed.
- J. T. Xu, M. Wang, N. P. Wickramaratne, M. Jaroniec, S. X. Dou and L. M. Dai, Adv. Mater., 2015, 27, 2042–2048 CrossRef CAS PubMed.
- Y. Yan, Y. X. Yin, S. Xin, Y. G. Guo and L. J. Wan, Chem. Commun., 2012, 48, 10663–10665 RSC.
- X. T. Yuan, A. A. Razzaq, Y. J. Chen, Y. B. Lian, X. H. Zhao, Y. Peng and Z. Deng, Chin. Chem. Lett., 2021, 32, 890–894 CrossRef CAS.
- H. L. Zhai, H. F. Jiang, Y. Qian, X. Y. Cai, H. M. Liu, Y. T. Qiu, M. M. Jin, F. Xiu, X. Liu and L. F. Lai, Mater. Chem. Phys., 2020, 240, 122139 CrossRef CAS.
- L. Y. Hu, X. S. Zhu, Y. C. Du, Y. F. Li, X. S. Zhou and J. C. Bao, Chem. Mater., 2015, 27, 8138–8145 CrossRef CAS.
- P. K. Allan, J. M. Griffin, A. Darwiche, O. J. Borkiewicz, K. M. Wiaderek, K. W. Chapman, A. J. Morris, P. J. Chupas, L. Monconduit and C. P. Grey, J. Am. Chem. Soc., 2016, 138, 2352–2365 CrossRef CAS PubMed.
- C. Liu, X. Fu, S. Z. Liao, G. Q. Zou and H. Yang, Nanomaterials, 2023, 13, 13020254 Search PubMed.
- P. L. Lou, Z. H. Cui, Z. Q. Jia, J. Y. Sun, Y. B. Tan and X. X. Guo, ACS Nano, 2017, 11, 3705–3715 CrossRef CAS PubMed.
- X. J. Pu, D. Zhao, C. L. Fu, Z. X. Chen, S. N. Cao, C. S. Wang and Y. L. Cao, Angew. Chem., Int. Ed., 2021, 60, 21310–21318 CrossRef CAS PubMed.
- X. Zhong, J. M. Duan, Y. E. Xiang, X. Y. Hu, Y. J. Huang, Y. J. Li, W. T. Deng, G. Q. Zou, H. S. Hou and X. B. Ji, Adv. Funct. Mater., 2023, 33, 2306574 CrossRef CAS.
- S. P. Zhang, G. Wang, B. B. Wang, J. M. Wang, J. T. Bai and H. Wang, Adv. Funct. Mater., 2020, 30, 2001592 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |



