Study on the influence of hydrogen addition on ethane/air laminar burning velocity, key species production, and implied physicochemical effects
Yanfei
Zhang
ab,
Zewen
Yu
ab,
Yujie
Xu
c,
Wenlong
Wang
d,
Mingming
Huang
 *ab,
Lingbo
Kong
e,
Zhiyong
Zhao
e and
Xiao
Zhang
f
*ab,
Lingbo
Kong
e,
Zhiyong
Zhao
e and
Xiao
Zhang
f
aSchool of Mechanical Engineering, Shandong Key Laboratory of CNC Machine Tool Functional Components, Qilu University of Technology (Shandong Academy of Sciences), Jinan 250353, China. E-mail: dahuangby@sina.com
bShandong Institute of Mechanical Design and Research, Jinan 250031, China
cShandong Engineering Consulting Institute, Jinan 251401, China
dCollege of Mechanical and Electrical Engineering, Huangshan University, Huangshan, Anhui 245041, China
eShandong Chuangyu Energy Technology Co., Ltd, Jinan 251401, China
fShandong Torch Creation Energy Science and Technology, Co., Ltd, Jinan 251401, China
First published on 27th November 2025
Abstract
In the context of carbon emission reduction, this study investigates the mixed combustion characteristics of zero-carbon hydrogen and ethane—the second major component of natural gas—under gas turbine operating conditions. Ethane not only has fuel properties but also serves as a chemical feedstock (for ethylene production). The quantitative relationship between laminar flame speed and key free radicals (O, H, and OH) was analyzed, and the underlying mechanism of the influence of free radicals on combustion speed was decoupled, considering the effects of heat, mass transfer, and chemical interactions. The study shows that under heated and pressurized conditions in the gas turbine, the hydrogen addition can significantly increase the laminar burning velocity (LBV) of ethane, as hydrogen enhances the concentrations of H, O, and OH free radicals. More importantly, a clear linear positive correlation exists between the LBV and the peak mole fractions of H, O, and OH species. The addition of hydrogen alters the linear correlation coefficient of the above linear relationship, but the linear relationship between LBV and the peak molar fractions of H, O and OH is not affected by pressure and equivalent ratio. The change in the linear correlation coefficient caused by hydrogenation is mainly influenced by chemical effects, followed by thermal effects, with the least impact from transport effects. These numerical results can provide a valuable reference for the design and operational condition selection of gas turbine combustors.
1 Introduction
Ethane (C2H6) serves as both a fuel and a chemical feedstock, attracting significant attention in the energy and chemical engineering fields.1,2 Ethane is the second most important component in natural gas, after methane (CH4), and can be used both as a fuel for combustion and as a feedstock for ethylene (C2H4) production.3,4 The volumetric composition of C2H6 in natural gas ranges from 0.5% to 13.3%, and can even reach as high as 16% in some regions of the Middle East. Compared to methane, ethane has a higher combustion speed and a more stable flame.5,6 In comparison to liquefied natural gas (LNG), ethane offers stronger low-cost advantages and better commercial viability.7 With the continued extraction of shale gas in recent years,8 global ethane production has been steadily increasing, and scientists are increasingly focusing on the application of ethane fuel in energy power equipment, such as internal combustion engines and gas turbines.Scholars have studied the combustion characteristics of ethane–air mixtures under high temperature and high pressure conditions in gas turbines. Qi et al.9 investigated the effects of inert gases, temperature, and pressure on the flammability limits of ethane under high-temperature and high-pressure conditions. Their study showed that the upper flammability limit of ethane exhibits a quadratic linear relationship with temperature. The addition of nitrogen reduces the flammability of ethane, but this effect decreases with increasing pressure and increases with increasing temperature. Mitu et al.10 studied the pressure and temperature dependence of ethane flame propagation velocity, calculating the normal burning velocity (NBV) using a cubic law of pressure increase, and determining the flame propagation velocity using NBV and the expansion coefficient of unburned gases. The study by Hwang et al.11 indicates that the laminar lift-off flame of ethane can stabilize within a triangular steady state defined by the Reynolds number parameter and the normalized lift-off height, and theoretically predicts how the effective Schmidt number changes with fuel type and lift-off height. Ghosh et al.12 studied the laminar burning velocity of ethane/air mixtures at atmospheric pressure, down to temperatures as low as 160 K. The experimental results obtained align well with the LBV calculated using the power law based on the ratio of unburned gas temperature to ambient temperature. Hashemi et al.6 conducted experimental studies on the oxidation of ethane under high-pressure conditions, reporting that the onset temperature of ethane oxidation is influenced by equivalence ratio, pressure, and residence time, and is generally in the range of 700 to 825 K.
Although ethane is a well-established chemical feedstock and fuel, its combustion efficiency and pollutant emissions are less satisfactory compared to hydrogen (H2).13 Hydrogen is considered a high-quality clean energy source, with a high calorific value and energy conversion efficiency. Its only combustion product is H2O, making it a true zero-carbon fuel.14 In addition, the diversification of hydrogen production methods and its technological breakthroughs in storage and transportation have also promoted the application of hydrogen,15 with the potential to gradually replace hydrocarbon fuels in the context of carbon peak and carbon neutrality in the future.16 Before hydrogen fully replaces hydrocarbon fuels, it is necessary to conduct research on hydrogen-blended gas based on application scenarios. Moreover, in gas turbine conditions, blending hydrocarbon fuels with hydrogen can mitigate issues such as backfire and thermoacoustic oscillations caused by the rapid flame speed of hydrogen.12,17 Ethane is an important hydrocarbon fuel, and its combustion when mixed with hydrogen has broad application prospects. Fig. 1 shows the process flow diagram of the industrial application of ethane–hydrogen blended fuel, with its infrastructure being relatively mature.
In recent years, the combustion of hydrogen-blended hydrocarbon fuels has been extensively studied as a strategy to reduce carbon footprints. Numerous studies have demonstrated that the addition of hydrogen to hydrocarbon fuels such as methane, propane, dimethyl ether, methanol, and ethanol not only enhances combustion efficiency and engine performance but also mitigates the emissions of major pollutants to varying degrees. Sun et al.18 investigated hydrogen–methane mixtures at ambient temperature and pressure using GRI-Mech 3.0 and compared the simulated laminar flame speeds with experimentally measured values. Their study found that the addition of hydrogen increased the mole fractions of H, O, and OH as well as the flame temperature, but it also promoted the formation of thermal NO emissions. As the hydrogen content increased from 0 to 100%, NO emissions rose while CO emissions decreased. Wang et al.19 investigated the combustion and emission characteristics of propane when blended with hydrogen. Their results indicated that, in propane–hydrogen mixtures, increasing the hydrogen fraction led to higher flow velocities and temperatures within the combustion chamber, while emissions of carbon oxides and nitrogen oxides decreased. Eckart et al.20 studied the laminar flame speed and emission characteristics of dimethyl ether with hydrogen addition through both experimental and numerical approaches. The study found that the hydrogen-enriched mixtures exhibited a relatively low sensitivity to initial temperature, and the addition of hydrogen increased the heat release rate of dimethyl ether. Prasad et al.21 investigated the combustion characteristics of methanol–hydrogen mixtures in internal combustion engines. Their study indicated that, at higher compression ratios, the addition of hydrogen to methanol significantly improved overall engine performance and markedly reduced CO, CO2, and HC emissions. Zhen et al.22 reported that, in high-compression-ratio engines burning ethanol–hydrogen blends, hydrogen addition led to an increase in both combustion duration and ignition delay. When hydrogen was added at the same fraction, engines running on methanol–hydrogen mixtures exhibited superior performance compared with ethanol–hydrogen mixtures, whereas ethanol–hydrogen blends produced lower HC and NOx emissions than methanol–hydrogen blends.
In summary, blending hydrogen into hydrocarbon fuels has emerged as a significant approach for reducing carbon emissions and improving combustion characteristics. Against this research background, some scholars have extended their investigations to ethane, focusing on its combustion mechanisms and emission characteristics under hydrogen blending, with the aim of providing theoretical insights and technical support for broader applications of hydrogen–hydrocarbon blended fuels. Li et al.23 compared the effects of hydrogen addition on the LBV of methane, ethane, and propane, indicating that hydrogen addition has a stronger promoting effect on the LBV of methane than on that of ethane and propane. This is because the reaction H + O2 = O + OH has the strongest promoting effect on the laminar burning velocity of methane/hydrogen mixtures, resulting in a more significant increase in the concentrations of radicals such as O, H, and OH in the flame. Xiang et al.24 introduced three hypothetical species, FH2, FO2, and FN2, into the detailed chemical reaction mechanism GRI 3.0 Mech to analyze the effects of physical and chemical effects on the ethane/hydrogen mixture. The numerical analysis found that the chemical effect of H2 promotes the LBV, AFT, and net heat release rate (NHRR) of ethane, while the physical effect reduces these three parameters. This indicates that the chemical effect accelerates the combustion reaction process, while the physical effect suppresses the combustion chemical reactions. Li et al.25 experimentally measured key combustion characteristics of the ethane/hydrogen mixture, including laminar burning velocity, Markstein length, flame thickness, and effective Lewis number. They proposed a general correlation between the Peclet number and Markstein number, covering a wide range of equivalence ratios and temperatures, aimed at defining the stable and unstable states of flame propagation. Nilsson et al.26 were the first to study the effect of hydrogen addition on heavy hydrocarbons, such as methane–ethane–propane mixtures. Their study indicates that the H radical is crucial for combustion enhancement, particularly under lean burn conditions.
In recent years, some scholars have also conducted research on the quantitative relationship between laminar flame speed and key radicals. Cai et al.27,28 used numerical analysis methods to investigate the quantitative relationships between ammonia/syngas laminar flame speed and radical concentration, LBV and IDT, indicating that these relationships vary with changes in initial temperature and pressure, generally following either a linear or power-law function. Furthermore, to explore the theoretical basis of these quantitative relationships, they also examined four global flame parameters: AFT, activation energy, Zeldovich number, and Lewis number. Liu et al.29 conducted a fitting study on the linear relationship between the combustion characteristics of ammonia–methane mixtures. Their study indicated that LBV exhibits a linear relationship with the maximum concentration of O + H + OH, and the slope of this linear relationship changes with the ammonia content. Chen et al.30 investigated the effect of hydrogen/carbon monoxide/syngas additions (0–40%) on the relationship between ammonia flame LBV and H + NH2 radicals, and derived fitting expressions for the relationship between LBV and H and H + NH2 radicals, respectively.
Compared with pure ethane combustion, blending zero-carbon hydrogen with ethane can significantly reduce carbon emissions throughout the production process. Laminar flame speed and key radicals are critical factors for studying the combustion process and effectively reducing carbon emissions. The literature review indicates that, although some researchers have explored hydrogen–ethane as an alternative fuel, the studies remain very limited. Moreover, most studies have focused on qualitative analyses of combustion characteristics. Currently, quantitative analyses of combustion characteristics are scarce. Investigating the quantitative relationships between radicals and laminar flame speed is crucial for gas turbine combustion efficiency, emission reduction, and safe operation. Such research can provide critical guidance for the design of gas turbine combustion chambers.
This study comprehensively examines the impact of hydrogen addition, pressure, and equivalence ratio on ethane laminar flame speed, key radical concentrations, and chemical heat transport effects through flame propagation analysis, key component generation rate sensitivity analysis, parametric studies, quantitative fitting, reaction path analysis, and effect analysis.
2 Models and validation
2.1 Numerical model
Numerical simulations were conducted on the laminar premixed combustion process of ethane–air mixtures with varying hydrogen content using the premixed flame propagation model in ANSYS CHEMKIN. The combustion zone is divided into reaction zone and preheating zone.Building on this, a premixing method based on the velocity of the mixed gas was proposed, with an initial value of 0.04 g (cm−2 s−1). The computational distance was set between 0 and 1 cm. The GRAD and CURV methods were applied, with values set to 0.06 for both, generating approximately 500 grids in total, with 50 adaptive grids. In the iteration process, the relative error was set to 10−4, and the absolute error was set to 10−9. Unless otherwise specified, the initial temperature for the simulation was set to 300 K, with a fixed temperature of 400 K applied. The mixed-mean transport model is used under all operating conditions, and the Soret effect is taken into account. The upwind difference method was used to discretize the convection terms, while the average method was employed to solve for the diffusion coefficient of the mixed gas. Considering the actual operating conditions of gas turbines, the initial pressures were set to 1, 3, 5, 6, 7, 8, 9, 10, 12.6, 15, and 16.5 atm, respectively. The equivalence ratios ranged from 0.7 to 1.3 in increments of 0.1, as shown in Table 1. The calculation formula for the hydrogen mixing ratio is as follows:
| XH2 = [VH2/(VH2 + VC2H6)] × 100% | (1) |
| Equivalence ratio (φ) | Fuel component (XH2) | Temperature (K) | Pressure (atm) |
|---|---|---|---|
| 0.7–1.3 | 10–90% | 300 | 1–16.5 |
The formula for the flame propagation velocity of the ethane/hydrogen mixed gas flame can be expressed as:
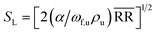 | (2) |
 represents the average reaction rate of the mixed gas in the reaction zone, α is the thermal diffusivity, ωf,u is the mass fraction of the unburned gas, and ρu is the density of the unburned gas.
represents the average reaction rate of the mixed gas in the reaction zone, α is the thermal diffusivity, ωf,u is the mass fraction of the unburned gas, and ρu is the density of the unburned gas.
Using the reaction rate constant, the reaction speed and the production rates of various chemical reaction components can be determined. The forward chemical reaction rate constant k can be calculated using the Arrhenius equation:
| k = ATbexp(−Ea/RT) | (3) |
2.2 Verification of reaction mechanism
Building on previous research, four reaction mechanisms applicable to the combustion of ethane–hydrogen mixtures were selected, namely GRI 3.0,31 San Diego,32 Aramco 1.3,33 and USC II,34 to analyze the laminar combustion characteristics of ethane–hydrogen mixtures. The basic details of these four mechanisms are shown in Table 2.Fig. 2 compares the simulated results of the laminar burning velocity (LBV) for ethane/hydrogen mixed fuel at an initial temperature of 300 K, a hydrogen doping ratio of 50%, and pressures of 1 atm and 5 atm, with the experimental results of Li et al.25 From Fig. 2(a), it can be observed that at a pressure of 1 atm, the San Diego and USC II mechanisms match the experimental data well under lean fuel conditions, while Aramco 1.3 and USC II align better with the experimental data under rich fuel conditions. All four mechanisms significantly underestimate the experimental value at φ = 1.1. But in general, the simulated data of these four mechanisms are basically consistent with the measured values at atmospheric pressure. When the pressure increased to 5 atm, the simulation values from the GRI 3.0 mechanism were lower than the experimental data, with significant deviation as the equivalence ratio varied. The simulation values of the San Diego mechanism were consistent with experimental data when φ < 0.9, but predicted lower values when φ ≥ 0.9. Overall, both the Aramco 1.3 and USC II mechanisms showed good agreement with experimental data. However, it is evident that the Aramco 1.3 mechanism exhibited smaller deviations from experimental values when φ ≥ 1, except at φ = 1.1, where the difference was larger. This could be due to the limitations of the mechanism or experimental errors.
 | ||
| Fig. 2 Under conditions of (a) P = 1 atm and (b) P = 5 atm, T = 300 K, XH2 = 50%, LBV changes with the equivalent ratio; point: experimental data; line: analog data (φ = 0.7–1.3, the experimental data are from Li et al.25). | ||
The Aramco 1.3 mechanism includes the most species, making it more widely applicable. Compared to the USC II mechanism, it reduces some unnecessary elementary reactions, thereby maintaining accuracy while reducing computational effort. Furthermore, the thermodynamic data of the Aramco 1.3 mechanism have been carefully determined and validated through experiments, providing accurate thermodynamic parameters for simulation calculations. Using the Aramco 1.3 mechanism for calculation effectively solved the problem of insufficient specificity of GRI 3.0 and San Diego. Therefore, the combustion of C2H6/air mixtures under different hydrogen doping ratios should be calculated using the Aramco 1.3 mechanism.
3 Results and discussion
3.1 The effect of hydrogen addition on laminar burning velocity
Combustion in gas turbines occurs under high temperature and high pressure. Investigating the effects of varying hydrogen content on ethane laminar premixed flame under high-pressure conditions can provide valuable data and experimental guidance for gas turbine combustion.Fig. 3 shows the relationship between laminar burning velocity (LBV) and equivalence ratio under different hydrogen contents at P = 5 atm and T = 300 K. It is evident from Fig. 3 that, for different equivalence ratios, the LBV increases with the rise in hydrogen content. Under varying hydrogen content, the trend of LBV with equivalence ratio first rises and then falls. Additionally, as the hydrogen blending ratio increases, the equivalence ratio corresponding to the maximum LBV also increases. The equivalent ratio corresponding to the maximum LBV gradually increases under different hydrogen doping ratios. For example, when the hydrogen doping ratio is between 10% and 70%, the mixture achieves the maximum LBV at φ = 1.1. When the hydrogen doping ratio is between 80% and 90%, the maximum LBV of the mixture occurs at φ = 1.2. Within the investigated equivalence ratio range (0.7–1.3), the LBV increases with hydrogen content. As the hydrogen blending ratio rises from 10% to 90%, the maximum LBV increases from 30.43 cm s−1 to 82.69 cm s−1, approximately tripling. According to eqn (3), hydrogen, as the lightest combustible gas, reduces the density of the unburned mixture (ρu), thereby increasing the laminar burning velocity. In addition, the higher the hydrogen content, the greater the increase in LBV, which is also found in the CH4/H2 mixture.24
 | ||
| Fig. 3 The variation relationship of LBV with the equivalent ratio (φ = 0.7–1.3) and hydrogen blending ratio (XH2 = 10–90%) at P = 5 atm and T = 300 K. | ||
3.2 The effect of hydrogen addition on the relationship between Max (H + O + OH) and laminar burning velocity
In the combustion process of ethane/hydrogen fuel, studying the impact of H, O, and OH radicals on the LBV is crucial. According to the sensitivity analysis of various reactions of ethane/hydrogen to temperature in ref. 24, the reaction H + O2 = O + OH, which characterizes the influence on LBV, is also the most significant reaction in promoting temperature rise. An increase in XH2 can accelerate the reaction H2 + OH = H + H2O, producing more H radicals, which in turn promote the reaction H + O2 = O + OH, continuously generating active radicals.18 Studies by many scholars have shown that LBV is closely related to the concentration of key free radicals such as H, O and OH during the combustion of hydrocarbons.35–39Fig. 4 shows the effect of hydrogen addition on the mole fractions of O, H, and OH under different equivalence ratios, with an initial pressure of 5 atm and a temperature of 300 K. Under varying hydrogen content conditions, the mole fractions of the three radicals increase first and then decrease with changes in the equivalence ratio. For the H radical, when XH2 is 10–80%, the peak occurs at φ = 1.2; when XH2 is 90%, the maximum H radical concentration is observed at φ = 1.3. When XH2 is 10–90%, the O radical reaches its peak at φ = 0.9, and the OH radical peaks at φ = 1.0. The trend of the single radical's variation with hydrogen addition is shown in SI Fig. S1. The concentration of H, O and OH radicals increased with the increase of hydrogen content. Moreover, the higher the hydrogen content, the greater the increase in these radical concentrations, which is similar to the trend observed for LBV with changes in hydrogen blending ratio. Therefore, there may be some quantitative relationship between Max (H + O + OH) and LBV.
 | ||
| Fig. 4 The relationship between hydrogen addition and the concentration of H + O + OH radicals at different equivalence ratios. | ||
To quantitatively verify this relationship, the data from Table 1 under the same initial temperature and hydrogen content were fitted using Origin software and MATLAB fitting tools. Three equivalence ratios were selected: 0.8, 1.1, and 1.3. The relationship between LBV and Max (H + O + OH) is shown in Fig. 5(a). Different symbols represent different equivalence ratios, and within a cluster of symbols, the pressure decreases from left to right. As shown in Fig. 5(a), under different equivalence ratios, the correlation between LBV and Max (H + O + OH) mole fractions is strong. An increase in pressure reduces both the LBV and Max (H + O + OH), but pressure changes do not affect the linear relationship between LBV and Max (H + O + OH). Changes in equivalence ratio result in variations in the slope and intercept of this linear relationship. When the pressure P = 5 atm, XH2 = 50% and T = 300 K, LBV and Max (H + O + OH) under different equivalent ratios are shown in Fig. 5(b). It can be observed that within the range φ = 0.7–1.3, both Max (H + O + OH) and LBV exhibit consistent trends with equivalence ratio changes, first increasing and then decreasing, peaking at φ = 1.1. By drawing a tangent at φ = 0.8, it can be seen that the slope remains relatively constant within the range φ = 0.7–1.0. The LBV increases with the concentration of radicals, exhibiting a linear correlation. At this stage, changes in equivalence ratio do not affect the linear relationship between LBV and Max (H + O + OH). The curve shows an inflection point at φ = 1.1, indicating that changes in equivalence ratio begin to affect the linear fitting relationship between LBV and Max (H + O + OH). To eliminate the influence of equivalence ratio, subsequent research will focus on the range φ = 0.7–1.0. Fig. 6 illustrates the effect of pressure and equivalence ratio range on the relationship between LBV and Max (H + O + OH), showing that this relationship is not affected by pressure or equivalence ratio. At XH2 = 50% and T = 300 K, the quantitative relationship between LBV and Max (H + O + OH) can be expressed as:
| SL = 1907 × Max (H + O + OH) + 5.667 | (4) |
 | ||
| Fig. 5 (a) Effect of pressure on the relationship between LBV and Max (H + O + OH). (b) Effect of equivalence ratio on the relationship between LBV and Max (H + O + OH). | ||
Further analysis indicates that variations in hydrogen content affect the slope and intercept of this linear relationship. Table 3 shows the slope, intercept and R2 obtained by fitting under different hydrogen content conditions. The slope (p1) and intercept (p2) obtained from Table 3 were fitted against hydrogen content using MATLAB R2020a, as shown in Fig. 7. The results show that the p1 and p2 of the curve exhibit a power-law relationship with hydrogen content, which can be used in the linear formula relating LBV to Max (H + O + OH). At T = 300 K, XH2 = 10–90%, P = 1–16.5 atm, and φ = 0.7–1.0, The laminar burning velocity (SL) is expressed as a function of Max (H + O + OH), hydrogen content, and radical concentrations as follows:
| SL = (1171XH23.976 + 1807) × Max (H + O + OH) + (4.273XH25.501 + 5.452) | (5) |
| Case | X H2 | Slope (p1) | Intercept (p2) | R 2 |
|---|---|---|---|---|
| 1 | 10% | 1772 | 5.343 | 0.9929 |
| 2 | 20% | 1799 | 5.329 | 0.9936 |
| 3 | 30% | 1825 | 5.451 | 0.9926 |
| 4 | 40% | 1858 | 5.552 | 0.9925 |
| 5 | 50% | 1907 | 5.667 | 0.9919 |
| 6 | 60% | 1976 | 5.832 | 0.9915 |
| 7 | 70% | 2079 | 6.094 | 0.9908 |
| 8 | 80% | 2256 | 6.52 | 0.9894 |
| 9 | 90% | 2595 | 7.913 | 0.9842 |
In summary, increasing hydrogen content changes LBV by changing the concentration of O, H, and OH radicals, because it changes the linear relationship between the free radicals (O, H, and OH) and LBV.
3.3 The behavior of H, O, and OH radicals
Considering that gas turbines mostly operate under lean combustion conditions with varying pressures, this study examines the relationship between LBV and the behavior of H, O, and OH radicals at an equivalence ratio of φ = 0.8 under medium and high pressures of 5 atm and 16.5 atm. Fig. 8 analyzes the variations in LBV and the concentrations of O, H, and OH radicals with hydrogen content under medium and high-pressure conditions. Under different pressure conditions, both the LBV and radical concentrations increase with rising hydrogen content, particularly at high hydrogen fractions (XH2 ≥ 70%), where the LBV rises sharply. This is mainly due to the increase of molar concentrations of O, H and OH caused by the addition of hydrogen, which expands the branched chain reaction and accelerates the laminar combustion rate, which has been proved by a large number of studies.18,24,40 | ||
| Fig. 8 (a) Variation of LBV with hydrogen content at pressures of 5 atm and 16.5 atm. (b) Variation of OH, H, and O radicals with hydrogen content (XH2 = 10–90%, φ = 0.8, T = 300 K). | ||
When the pressure increases from 5 atm to 16.5 atm, both the LBV and radical concentrations decrease significantly. The higher the hydrogen content, the greater the reduction, indicating that the promoting effect of hydrogen on LBV weakens under high pressure. The radicals and LBV exhibit similar trends under different pressure conditions, confirming the fitting results in Fig. 6, which show that the linear relationship between LBV and Max (H + O + OH) is unaffected by pressure. Under lean combustion conditions, the OH concentration is significantly higher than those of H and O, and it is also the radical most affected by variations in hydrogen content. As shown in Fig. 9, the OH radical consistently has the highest mole fraction during flame propagation, making it the primary radical in the structure of lean flames. Increases in pressure and hydrogen content do not change the significance of OH in lean combustion flames.
3.4 Yield and sensitivity analysis
As mentioned in Section 3.2, the LBV of ethane/hydrogen mixtures maintains a linear relationship with Max (H + O + OH), and the slope of this linear relationship varies with changes in hydrogen content. To investigate the reason for maintaining this linear relationship, a sensitivity analysis was conducted on the free radicals O, H, and OH, as well as LBV, selecting the 8 reactions that have the greatest impact.From the analysis of Fig. 10, it can be seen that for these three free radicals, the R1 reaction H + O2 = O + OH is the most important branching reaction, promoting an increase in their concentrations and having the highest sensitivity coefficient. Under medium pressure conditions (P = 5 atm), the sensitivity coefficient of the R1 reaction gradually decreases with the addition of hydrogen. This is because hydrogen addition enhances the sensitivity coefficients of the R2 O + H2 = H + OH and R3 OH + H2 = H + H2O reactions, leading to competition with R1. Under high-pressure conditions (P = 16.5 atm), the sensitivity coefficient of the R1 reaction for H radicals exhibits the same trend with hydrogen content as observed under medium pressure conditions, for the same reasons. For O and OH, the sensitivity coefficient of the R1 reaction increases with rising hydrogen content, but the effect of the increase is not significant.
R9 H + O2 (+M) = HO2 (+M), as a chain-terminating reaction, is the most important reaction in inhibiting the increase in the concentrations of O, H, and OH, because under fuel-lean conditions, the oxygen concentration is sufficiently high. The forward reaction of R9 not only consumes H radicals but also competes with R1 for H radicals, thereby significantly suppressing the generation of O and OH radicals. Under two pressure conditions, the absolute value of the negative sensitivity coefficient of R9 for the three free radicals gradually decreases with the increase in hydrogen content. Hydrogen addition reduces the sensitivity coefficients of branching reactions and the absolute value of the negative sensitivity coefficient of chain-termination reactions. However, within the overall reaction system, the promoting effect outweighs the inhibitory effect, resulting in an increase in radical concentrations (as shown in Fig. 8b).
When the pressure increases from 5 atm to 16.5 atm, the absolute values of the sensitivity coefficients of key reactions increase, while radical concentrations decrease (as shown in Fig. 8b and 9), which is opposite to the effect of hydrogen addition. In the reaction system, it is likely that the inhibitory effect of increased pressure outweighs the promoting effect.16
A sensitivity analysis was conducted on the reactions affecting the LBV of ethane/hydrogen, and the results are shown in Fig. 11. Notably, compared to methane/hydrogen combustion,23 reaction R201 C2H4 + H (+M) = C2H5 (+M) shows significantly increased importance in promoting the laminar burning velocity (LBV), making it a critical reaction influencing the production rate of H radicals. The addition of hydrogen supplies more H radicals, driving the forward rate of R1 while suppressing the forward rate of R201, a phenomenon that has been confirmed in numerous studies.23,24,26 The reason R9 inhibits the increase in LBV is the same as its inhibitory effect on O, H, and OH radicals. Increasing pressure amplifies the absolute sensitivity coefficients of reactions R1 and R9.
 | ||
| Fig. 11 Influence of hydrogen content on the reaction and its sensitivity coefficient related to laminar combustion velocity, with pressure of 5 atm and 16.5 atm, φ = 0.8, T = 300 K, respectively. | ||
Overall, the key reactions that promote and inhibit the increase in flame speed (R1 and R9) are the same as those that promote and inhibit the increase in radicals (O, H, and OH), and their sensitivity coefficients exhibit the same pressure-dependent behavior. This is the main reason why the LBV maintains a linear relationship with Max (H + O + OH) and remains unaffected by pressure changes. The effect of hydrogen addition on LBV differs from its impact on the sensitivity of key radicals, particularly R1 and R9. This discrepancy may account for the variation in the slope of eqn (5) as hydrogen content changes.
3.5 Reaction pathway analysis
To understand the effect of varying hydrogen content on the ethane oxidation reaction pathways under medium-to-high pressure conditions, a reaction pathway analysis was conducted using the Aramco 1.3 mechanism, with the percentages in parentheses corresponding to results at P = 16.5 atm, as shown in Fig. 12. The “%” next to each radical (H, O, and OH) indicates the percentage of conversion through the corresponding elementary reaction. For example, at XH2 = 20% and P = 5 atm, 44.6% of the conversion during ethane dehydrogenation to form ethyl is caused by OH attack. The thick arrows in the reaction pathway indicate the primary reaction routes for ethane, representing pathways through which the majority of intermediate products are converted.H, O, and OH are critical radicals driving ethane oxidation, almost directly or indirectly participating in all combustion reactions. As a result, Max (H + O + OH) is closely correlated with the laminar burning velocity. The first step in ethane oxidation involves extracting H from the fuel molecule and oxidizing it to ethyl.41 As the hydrogen content increases from 20% to 80%, the percentages of H, O, and OH involved in the C2H6 → C2H5 process decrease, but the percentage of H radicals involved in the conversion of ethyl to ethane increases. Furthermore, the conversion pathways C2H5 → CH3 → CH4 and C2H5 → C3H8 are also enhanced with the increase in hydrogen content. In the oxidation pathway of C2H3, the addition of hydrogen causes C2H3 to preferentially react with oxygen to form formaldehyde (CH2O) rather than CH2CHO. Since CH2CHO contains an unsaturated aldehyde group, its chemical bond stability is poor and less stable than formaldehyde.42
Under varying hydrogen content conditions, when the pressure increases from 5 atm to 16.5 atm, the importance of OH and O in the C2H6 → C2H5 reaction increases, while the percentage of H radicals decreases, thereby reducing the reaction rate of C2H6 + H = C2H5 + H2. Increased pressure enhances the reaction pathway C2H5 → C2H4 while reducing the conversion rates of the pathways C2H5 → CH3 → CH4 and C2H5 → C3H8, which is opposite to the effects of hydrogen addition. For the reaction pathways C2H3 → CH2O and CH2CHO → CH2O, increased pressure enhances the conversion rates, with this effect being most pronounced at XH2 = 60% (see SI Fig. S4). However, when the hydrogen content increases to 80%, increased pressure reduces the conversion rates. As the pressure increases, the collision frequency between molecules rises, making the decomposition reaction more likely to occur. Additionally, as pressure increases, the reaction equilibrium shifts toward a decrease in the number of gas molecules.43
In summary, the effects of hydrogen addition and pressure increase on reaction pathways are opposite. An increase in hydrogen content causes ethane to favor the formation of CH4, C3H8, and CH2O, while weakening the reaction pathway leading to CO2 formation. In contrast, increased pressure drives ethane to follow the primary reaction pathway leading to CO2 formation, with this effect being particularly pronounced at 80% hydrogen content. This explains why the higher the hydrogen content, the greater the reduction in laminar burning velocity caused by increased pressure (as shown in Fig. 8). This change in reaction pathways results in hydrogen addition enhancing the laminar burning velocity, while pressure increase has a suppressive effect.
3.6 Chemical effects, thermal effects, and transport effects
Hydrogen as an additive affects the laminar premixed combustion characteristics of fuels in three ways: (1) chemical effects caused by hydrogen's participation in combustion reactions; (2) thermal effects due to changes in flame temperature; and (3) transport effects resulting from the reduction in reactant concentration and collision frequency. This study employs the method proposed by Liu et al.37 to distinguish the contributions of these three effects in numerical analysis. This method introduces a virtual substance, FH2, which shares the same thermal properties, transport characteristics, and three-body collision rates as H2, but does not participate in chemical reactions or molecular dissociation. To isolate the thermal and transport effects from the physical effects, another hypothetical substance, IH2, is introduced. IH2 matches FH2 in chemical effects and thermal properties but has transport properties identical to N2.Fig. 13 presents the calculation methods for the chemical effects, thermal effects, and transport effects of H2 at different equivalence ratios and hydrogen contents, with the results shown in Fig. 14. It can be seen that the chemical effect has the greatest influence, followed by the thermal effect, and the transport effect has the least influence. Tang et al.44 also found this phenomenon in the study of n-butane hydrogenation mixture. Hydrogen addition and pressurization do not alter the relative importance of these three effects. The influence of transport effects increases gradually with rising equivalence ratio, while the effects of chemical and thermal processes first increase and then decrease, reaching a maximum around φ = 0.9. This is because the relative proportions of fuel and oxygen change with the equivalence ratio. When the equivalence ratio is low, oxygen is relatively abundant, promoting the generation of H, O, and OH radicals, which triggers chain reactions and enhances the chemical effect. However, when the equivalence ratio exceeds a certain value, insufficient oxygen limits the generation of radicals, making it difficult for the chain reactions to persist, thus weakening the chemical effect.45,46
 | ||
| Fig. 14 Chemical, thermal, and transport effects vary with (a) equivalent ratio and (b) hydrogen content under different pressure conditions. | ||
At a fixed equivalence ratio (φ = 0.8), hydrogen addition has the greatest impact on the chemical and thermal effects, which increase with higher hydrogen content. Chemical effects are the primary reason for the increase in LBV with higher hydrogen content, as hydrogen and pressure have almost no impact on transport effects. As pressure rises from 5 atm to 16.5 atm, the thermal effect decreases, but the chemical effect is more significantly affected. Therefore, in the process where pressurization reduces LBV, the chemical effect is the dominant factor. As pressure increases, the number of effective collisions per unit time increases, thereby accelerating the chemical reaction rate and enhancing the chemical effect.47
4 Conclusion
A chemical kinetic simulation was conducted using the Chemkin premixed free propagation flame model to systematically study the effects of different hydrogen additions (10–90%) on the laminar premixed combustion characteristics of ethane/air under medium pressure (P = 5 atm) and high pressure (P = 16.5 atm) conditions. The main conclusions are summarized as follows:(1) Hydrogenation can significantly increase LBV, especially at XH2 ≥ 70%. The reason why hydrogenation leads to an increase in the laminar burning velocity is the rise in the concentrations of H, O and OH radicals.
(2) LBV exhibits a linear positive correlation with Max (H + O + OH). The slope (p1) and intercept (p2) of this linear relationship both follow a power-law relationship with hydrogen content, while being independent of pressure and equivalence ratio.
(3) The sensitivity coefficients of reactions R1 (H + O2 = O + OH) and R9 (H + O2 (+M) = HO2 (+M)) vary with pressure and hydrogen content, and are a significant factor influencing the linear relationship between laminar burning velocity and Max (H + O + OH).
(4) Hydrogen addition promotes the formation of CH4, C3H8, and CH2O from ethane while reducing the reaction pathway that converts ethane into CO2. In contrast, pressure has the opposite effect. The change in the linear correlation coefficient caused by hydrogenation is mainly influenced by chemical effects, followed by thermal effects, with the least impact from transport effects.
In summary, this study investigates the laminar flame speed and key free radicals of hydrogen–ethane blended combustion. The research findings provide theoretical foundations and practical references for the sustainable development of similar industrial processes. They are also expected to offer critical technological support for carbon-neutral pathways and contribute to the transition of industrial systems towards a net-zero emission future.
Nomenclature
Greek letters
| φ | Equivalence ratio |
| α | Thermal diffusivity |
| ω f,u | Mass fraction of the unburned gas |
| ρ u | Density of the unburned gas |
| k | Reaction rate constant |
Abbreviation
| LBV | Laminar burning velocity |
| AFT | Adiabatic flame temperature |
| IDT | Ignition delay time |
| NHRR | Net heat release rate |
Conflicts of interest
The authors declare no conflicts of interest.Data availability
Data is provided within the manuscript or supplementary information (SI) files. Supplementary information is available. See DOI: https://doi.org/10.1039/d5se01256b.Acknowledgements
This work was supported by “Mount Taishan Industrial Leading Talents Program of Shandong Province” (No. tscx202606082), “Qilu University of Technology (Shandong Academy of Sciences) – Weihai City Industry-University-Research Collaborative Innovation Fund” (2022CXY-03), “Shandong Science and Technology SMEs Innovation Capacity Improvement Project” (Grant No. 2023TSGC0270), “Shandong Enterprise Technology Innovation Project” (Grant No. 2024537010000271 and 2025537010000492), “Jinan City science and technology plan project social livelihood special”(Grant No. 202317003) and “Jinan City science and technology SMEs innovation ability improvement project” (Grant No. 201212201631).References
- J. Qin, P. Zhao, J. Meng, S. Zuo, X. Wang and W. Zhu, et al., Surface exposure engineering on LaMnO3@Co2MnO4 for high-efficiency ethane catalytic combustion, Appl. Surf. Sci., 2024, 653, 159393 CrossRef.
- L. Liu, L. Wang and F.-S. Xiao, Zeolite catalysts for non-oxidative ethane dehydrogenation to ethylene, EES Catal., 2024, 2(4), 923–931 RSC.
- M. Li, S. Mao, X. Zhou and H. Wang, One-step purification of ethylene from ethane/ethylene mixtures by a stable vanadium-based metal-organic framework, Sep. Purif. Technol., 2025, 360, 131049 CrossRef CAS.
- D. Maporti, F. Galli, P. Mocellin and G. Pauletto, Flexible ethylene production: Electrified ethane cracking coupled with oxidative dehydrogenation, Energy Convers. Manage., 2023, 298, 117761 CrossRef CAS.
- Z. Jiang, S. Dong, W. Gao, B. Wang, H. Ding and J. Xie, et al., Probing the effect of fuel components on the auto-ignition behavior of ammonia/natural gas blends: A case study of ethane addition, Combust. Flame, 2024, 259, 113186 CrossRef CAS.
- H. Hashemi, J. G. Jacobsen, C. T. Rasmussen, J. M. Christensen, P. Glarborg and S. Gersen, et al., High-pressure oxidation of ethane, Combust. Flame, 2017, 182, 150–166 CrossRef CAS.
- Y. Hu, S. I. Badrawi, J. Kumar, H. N. Sabeh, T. R. Alsenani and F. Riaz, et al., Gas turbine-based system taking advantage of LNG regasification process for multigeneration purposes; Techno-economic-environmental analysis and machine learning optimization, Process Saf. Environ. Prot., 2023, 179, 10–26 CrossRef CAS.
- X. Li, C. Pei and J. Gong, Shale gas revolution: Catalytic conversion of C1–C3 light alkanes to value-added chemicals, Chem, 2021, 7(7), 1755–1801 CAS.
- C. Qi, X. Yu, Y. Wang, L. Chen, X. Yan and X. Lv, et al., Investigating the effect of temperature, pressure, and inert gas on the flammability range of ethane/oxygen mixtures, Fuel, 2023, 354, 129296 CrossRef CAS.
- M. Mitu, D. Razus, V. Giurcan and D. Oancea, Normal burning velocity and propagation speed of ethane–air: Pressure and temperature dependence, Fuel, 2015, 147, 27–34 CrossRef CAS.
- G. J. Hwang, D. S. Jeon and N. I. Kim, Characteristics of laminar lifted flames of pure ethane in a non-premixed jet under various pressures, Combust. Flame, 2024, 264, 113422 CrossRef CAS.
- A. Ghosh, N. M. Munoz-Munoz, K. P. Chatelain and D. A. Lacoste, Laminar burning velocity of hydrogen, methane, ethane, ethylene, and propane flames at near-cryogenic temperatures, Appl. Energy Combust. Sci., 2022, 12, 100094 Search PubMed.
- E. A. Shchepakina, I. A. Zubrilin, A. Y. Kuznetsov, K. D. Tsapenkov, D. V. Antonov and P. A. Strizhak, et al., Physical and Chemical Features of Hydrogen Combustion and Their Influence on the Characteristics of Gas Turbine Combustion Chambers, Appl. Sci., 2023, 13(6), 3754 CrossRef CAS.
- N. Hajialigol and B. F. Ahmadi, Pollution formation and the response of premixed hydrogen blending flame to the external excitation using large eddy simulation: The application of an artificial neural network and genetic algorithm, J. Taiwan Inst. Chem. Eng., 2023, 148, 104650 CrossRef CAS.
- E. Giacomazzi, G. Troiani, A. Di Nardo, G. Calchetti, D. Cecere and G. Messina, et al., Hydrogen Combustion: Features and Barriers to Its Exploitation in the Energy Transition, Energies, 2023, 16(20), 7174 CrossRef CAS.
- Y. Zhang, J. Fu, J. Shu, M. Xie and J. Liu, A chemical kinetic investigation of laminar premixed burning characteristics for methane-hydrogen-air mixtures at elevated pressures, J. Taiwan Inst. Chem. Eng., 2020, 111, 141–154 CrossRef CAS.
- S. J. M. Algayyim, K. Saleh, A. P. Wandel, I. M. R. Fattah, T. Yusaf and H. A. Alrazen, Influence of natural gas and hydrogen properties on internal combustion engine performance, combustion, and emissions: A review, Fuel, 2024, 362, 130844 CrossRef CAS.
- Y. Sun, Y. Zhang, M. Huang, Q. Li, W. Wang and D. Zhao, et al., Effect of hydrogen addition on the combustion and emission characteristics of methane under gas turbine relevant operating condition, Fuel, 2022, 324, 124707 CrossRef CAS.
- Y. Wang, C. H. Sohn and J.-Y. Kim, Effects of hydrogen blending on combustion and pollutant emission of propane/air in a model furnace with a rotary kiln burner, Therm. Sci. Eng. Prog., 2024, 47, 102330 CrossRef CAS.
- S. Eckart, S. Benaissa, R. A. Alsulami, K. A. Juhany, H. Krause and A. Mohammad, Laminar burning velocity, emissions, and flame structure of dimethyl ether-hydrogen air mixtures, Int. J. Hydrogen Energy, 2023, 48(91), 35771–35785 CrossRef CAS.
- B. S. Nuthan Prasad, J. K. Pandey and G. N. Kumar, Effect of hydrogen enrichment on performance, combustion, and emission of a methanol fueled SI engine, Int. J. Hydrogen Energy, 2021, 46(49), 25294–25307 CrossRef CAS.
- X. Zhen, X. Li, Y. Wang, D. Liu and Z. Tian, Comparative study on combustion and emission characteristics of methanol/hydrogen, ethanol/hydrogen and methane/hydrogen blends in high compression ratio SI engine, Fuel, 2020, 267, 117193 CrossRef CAS.
- Z. Li, X. Cheng, W. Wei, L. Qiu and H. Wu, Effects of hydrogen addition on laminar flame speeds of methane, ethane and propane: Experimental and numerical analysis, Int. J. Hydrogen Energy, 2017, 42(38), 24055–24066 CrossRef CAS.
- L. Xiang, H. Jiang, F. Ren, H. Chu and P. Wang, Numerical study of the physical and chemical effects of hydrogen addition on laminar premixed combustion characteristics of methane and ethane, Int. J. Hydrogen Energy, 2020, 45(39), 20501–20514 CrossRef CAS.
- J. Li, Y. Xie, M. Elsayed Morsy and J. Yang, Laminar burning Velocities, Markstein numbers and cellular instability of spherically propagation Ethane/Hydrogen/Air premixed flames at elevated pressures, Fuel, 2024, 364, 131078 CrossRef CAS.
- E. J. K. Nilsson, A. van Sprang, J. Larfeldt and A. A. Konnov, The comparative and combined effects of hydrogen addition on the laminar burning velocities of methane and its blends with ethane and propane, Fuel, 2017, 189, 369–376 CrossRef CAS.
- Z. Cai, M. Huang, G. Wei, Z. Liu, H. Fang and Y. Song, et al., Numerical study of the effect of pressure on the combustion characteristics of ammonia/coal-derived syngas mixture under gas turbine operating conditions, Fuel, 2023, 347, 128463 CrossRef CAS.
- Z. Cai, M. Huang, G. Wei, Z. Liu, H. Zhang and Q. Hao, et al., Theoretical study on the inherent relationship between laminar burning velocity, ignition delay time, and NO emission of ammonia-syngas-air mixtures under gas turbine conditions, Int. J. Hydrogen Energy, 2024, 50, 690–705 CrossRef CAS.
- D. Liu, Y. Zhang, Q. Li, M. Huang, Z. Liu and H. Zhang, Numerical study of the effect of NH3 addition on CH4/air combustion characteristics under gas turbine operating conditions, Sustainable Energy Fuels, 2024, 8(3), 573–587 RSC.
- Z. Chen and Y. Jiang, Numerical investigation of the effects of H2/CO/syngas additions on laminar premixed combustion characteristics of NH3/air flame, Int. J. Hydrogen Energy, 2021, 46(21), 12016–12030 CrossRef CAS.
- S. GPJhwmbegm. GRI-Mech 3.0. 1999 Search PubMed.
- A. Valera-Medina, H. Xiao, M. Owen-Jones, W. I. F. David and P. J. Bowen, Ammonia for power, Prog. Energy Combust. Sci., 2018, 69, 63–102 CrossRef.
- W. K. Metcalfe, S. M. Burke, S. S. Ahmed and H. J. Curran, A Hierarchical and Comparative Kinetic Modeling Study of C1–C2 Hydrocarbon and Oxygenated Fuels, Int. J. Chem. Kinet., 2013, 45(10), 638–675 CrossRef CAS.
- C. Zamfirescu and I. Dincer, Using ammonia as a sustainable fuel, J. Power Sources, 2008, 185(1), 459–465 CrossRef CAS.
- F. Liu, Z. Liu, Z. Sang, X. He, F. Liu and C. Liu, et al., A numerical and comparative study of the laminar combustion characteristics and kinetics of dimethyl ether and ethanol flames at elevated temperature, Fuel, 2021, 284, 118934 CrossRef CAS.
- F. Liu, Z. Liu, Z. Sang, X. He, M. Sjöberg and D. Vuilleumier, et al., Numerical study and cellular instability analysis of E30-air mixtures at elevated temperatures and pressures, Fuel, 2020, 271, 117458 CrossRef CAS.
- G. Liu, J. Zhou, Z. Wang, W. Yang, J. Liu and K. Cen, Adiabatic laminar burning velocities of C3H8-O2-CO2 and C3H8-O2-N2 mixtures at ambient conditions-PART II: Mechanistic interpretation, Fuel, 2020, 276, 117946 CrossRef CAS.
- Z. Wang, S. Wang, R. Whiddon, X. Han, Y. He and K. Cen, Effect of hydrogen addition on laminar burning velocity of CH4/DME mixtures by heat flux method and kinetic modeling, Fuel, 2018, 232, 729–742 CrossRef CAS.
- Q. Zhou, C. S. Cheung, C. W. Leung, X. Li, X. Li and Z. Huang, Effects of fuel composition and initial pressure on laminar flame speed of H2/CO/CH4 bio-syngas, Fuel, 2019, 238, 149–158 CrossRef CAS.
- Q. Li, G. Hu, S. Liao, Q. Cheng, C. Zhang and C. Yuan, Kinetic Effects of Hydrogen Addition on the Thermal Characteristics of Methane–Air Premixed Flames, Energy Fuels, 2014, 28(6), 4118–4129 CrossRef CAS.
- X. Chen, D. Dang, H. An, B. Chu and Y. Cheng, MnOx promoted phase-pure M1 MoVNbTe oxide for ethane oxidative dehydrogenation, J. Taiwan Inst. Chem. Eng., 2019, 95, 103–111 CrossRef CAS.
- Y.-l. Han, Z. Zheng, C.-h. Yin, P.-f. Li, H. Zhang and Y.-m. Hu, Catalytic oxidation of formaldehyde on iron ore tailing, J. Taiwan Inst. Chem. Eng., 2016, 66, 217–221 CrossRef CAS.
- G. Subbiah, R. P. T, J. R. P, M. M. Al-Ansari and C. B. B. Arun, Assessment of compression ignition engines performance using Spirulina microalgae biodiesel and gaseous fuel blends: A comparative study on efficiency, emissions, and combustion dynamics, J. Taiwan Inst. Chem. Eng., 2024, 105916 Search PubMed.
- C. L. Tang, Z. H. Huang and C. K. Law, Determination, correlation, and mechanistic interpretation of effects of hydrogen addition on laminar flame speeds of hydrocarbon–air mixtures, Proc. Combust. Inst., 2011, 33(1), 921–928 CrossRef CAS.
- S. Jin, Z. Zi, P. Yang, J. Zhang and B. Wu, The influence of exhaust gas recirculation on combustion and emission characteristics of ammonia-diesel dual-fuel engines: Heat capacity, dilution and chemical effects, J. Energy Inst., 2024, 117, 101778 CrossRef CAS.
- Y. Zhang, J. Fu, J. Shu, M. Xie, F. Zhou and J. Liu, et al., A numerical investigation of the effect of natural gas substitution ratio (NGSR) on the in-cylinder chemical reaction and emissions formation process in natural gas (NG)-diesel dual fuel engine, J. Taiwan Inst. Chem. Eng., 2019, 105, 85–95 CrossRef CAS.
- H. Zhang, B. Yao, H. Zhang, F. Yang, M. Yuan and Z. Li, et al., Synergistic effects of dilution gases and hydrogen on methane/air laminar combustion characteristics and NOX-emission concentrations, Int. J. Hydrogen Energy, 2024, 88, 779–791 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |