Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Fabrication methods and applications of printed wearable sensors for real-time sweat analysis: a comprehensive review
Pegah Bavafa ac,
Yahya Sorkhe
ac,
Yahya Sorkhe a,
Lia A. Stanciub and
Aytekin Uzunoglu
a,
Lia A. Stanciub and
Aytekin Uzunoglu *a
*a
aDepartment of Materials and Metallurgical Engineering, Faculty of Chemistry-Metallurgical Engineering, Istanbul Technical University, Istanbul, Turkiye. E-mail: auzunoglu@itu.edu.tr
bDepartment of Materials Engineering, Collage of Engineering, Purdue University, West Lafayette, IN, USA
cGraduate School of Science and Engineering, Koç University, Istanbul, Türkiye
First published on 28th April 2026
Abstract
Sweat analysis has emerged as a new approach for non-invasive and continuous health monitoring. One of the significant advances in non-invasive sweat analysis has been leveraged by the implementation of printing technologies in electrochemical (bio)sensors. This in-depth review analyzes the fast-changing landscape of these cutting-edge sensors, emphasizing their strong potential to deliver continuous, real-time health information conveniently and affordably. We thoroughly examined various printing methods, including screen printing, inkjet printing, 3D printing, laser printing, roll-to-roll printing, and electrohydrodynamic jet printing, for the fabrication of the sweat sensors. In addition, an extensive section examines electrode and substrate materials, ranging from conventional carbon-based and metallic materials to advanced nanomaterials and conductive polymers. We also evaluated the shift from traditional carbon and metallic electrodes to advanced nanomaterials. A detailed assessment is provided of recent progress in the detection of critical analytes such as glucose, lactate, electrolytes, cortisol, and other ions across fitness and sports tracking, disease management, workplace health and safety, and personalized medicine. The last portion of the paper is devoted to unraveling the challenges against practical and commercial adaptation: long-term stability, sensitivity, and selectivity in complex bio-environments, and integration with communication technologies.
1. Introduction
The development of high-performance (bio)sensors has pushed the boundaries of detection for a wide range of analytes, especially in healthcare, biology, and chemistry, thanks to their fast, sensitive, reliable, cost-effective, and easy-to-use nature. The main working principle of a biosensor relies on the interaction of analytes with the sensor surface, which produces measurable signals from a physicochemical transducer. Continuous biomarker detection is of great interest in some cases; a well-known example is the continuous monitoring of glucose level. The advent of (bio)sensor technology has ushered in a new era, especially in healthcare applications, for detecting biomarkers associated with diseases, driven by the discovery of innovative electrode materials and the development of scalable, low-cost, and reproducible electrode fabrication methods. The development of flexible and wearable analytical devices has leveraged the point of care (POC) sensors for the skin-interface related diagnosis method, eliminating the requirement of expensive laboratory equipment.1 In addition, skin-interfaced wearable sensors enable biomarker detection via a noninvasive approach with a small or limited sample volume. The integration of wearable analytical devices with microfluidics has emerged as a next-generation sensor design enabling continuous, real-time data collection.2 Another significant breakthrough was achieved when those next-generation sensors were integrated with wireless or Bluetooth devices, which transmit the collected data to mobile applications.Sweat analysis is a promising non-invasive technique for real-time monitoring of various physiological measures and biomarkers.3,4 Compared with traditional biofluids such as urine and blood, sweat sampling is minimally painful, easy to perform, and can be continuously monitored without requiring highly trained personnel.5 The complex composition of sweat, including electrolytes, metabolites, proteins, and other biomolecules, can be used to assess hydration status, electrolyte homeostasis, metabolic disease, and some biomarkers associated with diseases or physiological conditions. For example, elevated sweat chloride can be used to diagnose cystic fibrosis, sweat glucose can support non-invasive diabetes monitoring, and sweat sodium and potassium reflect dehydration or renal dysfunction.6–8 In addition, some proteins and peptides in sweat composition may serve as biomarkers for detrimental conditions, including atopic dermatitis or psoriasis.9 Sweat analysis also enables drug abuse testing, therapeutic drug monitoring, early cancer biomarker discovery, stress assessment via cortisol, and VOC-based screening for diseases, including tuberculosis and lung cancer.10–13
In recent years, a great effort has been devoted to developing innovative wearable sensors capable of measuring analytes in sweat, such as lactate,13 Ca2+, glucose,14 cortisol,15 sodium,15,16 potassium,16 chloride,17,18 uric acid,19 and heavy metals.20 Electrochemical (bio)sensors have shown exceptional promise due to their high sensitivity, selectivity, miniaturization, and compatibility with wearable platforms.8–10 While screen-printing, photolithography, and micromachining techniques have been widely exploited in sensor fabrication, the advent of printing technologies has revolutionized the field, enabling rapid prototyping, cost-effective manufacturing, and scalable production of highly customized, flexible sensor designs.21,22
Printing techniques, such as inkjet printing, screen printing, and 3D printing, offer unique advantages, including precise material deposition, compatibility with a wide range of functional inks (e.g., conductive, semiconductive, and insulating), and the ability to create complex patterns and architectures on substrates.15,16,23 Over the past decade, extensive research efforts have been dedicated to developing printed electrochemical (bio)sensors for sweat analysis, leveraging various printing methodologies and material combinations.16,17 Printed electrochemical (bio)sensors have been successfully applied to measure electrolytes, metabolites, and disease-related biomarkers in sweat.20,24,25
Despite the progress in sweat analysis using printed electrodes, there is no dedicated review focusing specifically on printed sweat sensors. The published reviews are dedicated to wearable biosensors, electrochemical sensors, or sweat sampling with a broad scope.14,21,22,24,26 The literature on printed sensors for sweat analysis is fragmented, with research scattered across various journals, making it difficult for researchers and practitioners to gain a holistic understanding of the field and identify emerging trends.5,15,22 This review addresses that gap by systematically summarizing printed electrochemical (bio)sensors for sweat analysis, critically evaluating the state-of-the-art, and outlining key challenges, limitations, and future research directions.
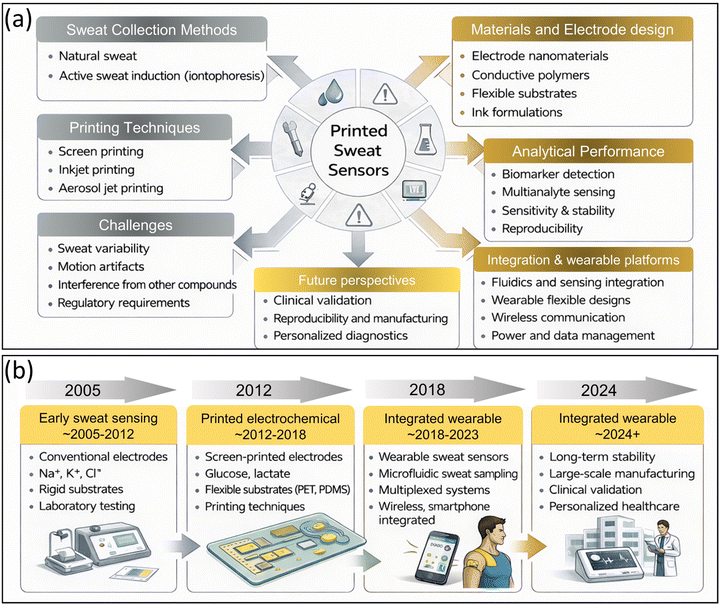
Fig. 1(a) summarizes the fundamental components and research directions involved in the development of printed sweat sensing platforms. Sweat has emerged as a highly attractive biofluid for non-invasive health monitoring because it contains numerous physiologically relevant biomarkers, including electrolytes (Na+, K+, Cl−), metabolites (glucose, lactate), hormones, and trace elements that reflect metabolic status and hydration levels.27–29 Continuous monitoring of these analytes using wearable sweat sensors provides valuable insights into human physiology and personalized healthcare applications30,31 flexible electrochemical sensing technologies are particularly well suited for such applications due to their high sensitivity, rapid response, miniaturization capability, and compatibility with wearable electronics.32,33 Early developments in sweat sensing focused primarily on electrochemical detection of individual biomarkers; however, recent advances have enabled multiplexed sensing platforms capable of simultaneously detecting multiple analytes in real time. These systems typically combine flexible substrates, microelectronic signal processing units, and wireless communication modules to create fully integrated wearable health monitoring devices.34–37 As illustrated in the schematic, the design of printed sweat sensors therefore involves a multidisciplinary integration of materials science, electrochemistry, microfluidics, and biomedical engineering.27,29,38,39
Another key aspect highlighted in Fig. 1(a) is the combination of sweat collection methods, printing technologies, and electrode materials that determine the overall performance of wearable sweat sensors. Sweat sampling can occur naturally during physical activity or can be actively induced using iontophoresis to enable controlled sweat extraction without exercise33,40 efficient sampling strategies are frequently combined with microfluidic systems that guide sweat toward sensing electrodes and allow continuous monitoring of analytes with minimal sample volumes.41,42 From a fabrication perspective, printing techniques such as screen printing, inkjet printing, and aerosol jet printing have become dominant approaches because they enable scalable, low-cost manufacturing of flexible electrochemical sensors on substrates such as PET, PDMS, or textiles.33,41 Screen-printed electrodes are particularly attractive since their composition can be easily tuned using conductive inks containing carbon, metals, enzymes, nanomaterials, or conductive polymers, enabling highly customizable sensing interfaces. Despite rapid progress, several challenges remain for practical deployment of sweat sensing devices, including variability in sweat composition, motion artifacts during on-body measurements, interference from coexisting biomolecules, and the need for standardized calibration and regulatory validation.2,29,40 Consequently, current research efforts are increasingly focused on improving reproducibility, integrating microfluidics and electronics into wearable patches, and advancing large-scale manufacturing strategies to enable clinical translation of printed sweat sensing technologies for personalized diagnostics and health monitoring.43–46
Over the past two decades, the field of sweat sensing has progressed from laboratory-based electrochemical detection to fully integrated wearable sensing platforms.30,46 Early studies primarily focused on conventional electrodes and in vitro measurements of electrolyte biomarkers.47,48 With the emergence of printing technologies such as screen printing, inkjet printing, and aerosol jet printing, flexible and low-cost sensors capable of detecting a broader range of analytes have been developed.47–49 More recently, advances in microfluidics, nanomaterials, and wireless electronics have enabled the development of wearable systems capable of real-time monitoring of biomarkers directly on the human body.5,30,49,50 Fig. 1 summarizes the evolution of printed wearable sweat sensors and highlights the future roadmap toward clinically validated wearable diagnostic platforms.
2. Sweat as a biofluid
Sweat is a promising biofluid for continuous, real-time health monitoring outside the skin due to its rich biomarker content of electrolytes, metabolites, proteins, and hormones.51,52 Unlike blood or interstitial fluid, sweat can be collected without invasive procedures, enabling continuous, real-time wearable-based health monitoring.3 Moreover, its non-invasive nature offers excellent potential for various applications, ranging from sports physiology, where it is beneficial for balancing electrolyte status, to clinical diagnosis, such as measuring chloride content for cystic fibrosis screening in neonates. Advances in wearable and microfluidic sensor technology have propelled the potential for correct determination of sweat composition and reporting on physiological as well as pathological status.53,542.1. Sweat collection method
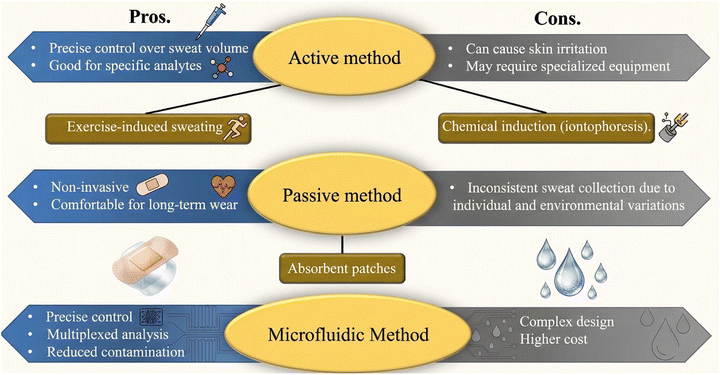
The integrity and composition of the collected sweat sample are among the main concerns, as they are influenced by the sweat collection method; deviations in the results may occur. Moreover, any contaminant introduced during sample collection may alter the actual analyte concentrations.55 The technique of sweat stimulation, whether through exercise, iontophoresis, or passive collection, can affect sweat rate and composition.7 Exercise-induced sweat, for instance, may have different electrolyte concentrations than in the resting state due to physiological changes during physical activity. Furthermore, the material used for sweat collection can also affect results; some materials may adsorb specific analytes or leach substances into the sample, leading to false measurements.10 Temporal factors also play a role, as sweat composition can change over time due to evaporation, leading to the concentration of some analytes and the potential loss of volatile components.56 The skin's microbiome can metabolize specific sweat components, altering sample composition if not promptly analyzed or properly preserved.57Additionally, the anatomical site of sweat collection can influence results due to variations in sweat gland density and local skin chemistry across different body regions.58 Sample processing techniques, such as centrifugation or filtration, may inadvertently remove or alter specific analytes, particularly larger molecules like proteins or cellular components.59 Storage conditions and the time between collection and analysis can lead to the degradation of unstable compounds or the growth of microorganisms, further altering the sample composition.60 Lastly, the analytical method itself, including sample preparation steps such as dilution or extraction, can introduce variability and potentially change the original composition of the sweat sample.56 Understanding and addressing these factors is crucial to developing standardized protocols that ensure accurate and reliable sweat analysis. A detailed examination of the sample collection methods is described below.
Furthermore, researchers have explored combining different sweat collection methods to enhance the performance and reliability of printed biosensors. For instance, Munje et al.68 proposed a hybrid approach that combines passive and active sweat-collection techniques for continuous monitoring of glucose and pH levels in sweat using a printed biosensor system. Sweat collection methods and their pros and cons are summarized in Fig. 2. The active, passive, and microfluidic sweat-collection methods have been extensively employed in the printed biosensors, each offering unique advantages and applications. The choice of sweat collection method depends on factors such as the target analyte, desired monitoring duration, and the specific requirements of the biosensor system. Ongoing research aims to further optimize and integrate these sweat-collection techniques with printed biosensors, enabling more accurate, reliable, and continuous monitoring of biomarkers for personalized healthcare and sports performance.
3. Printed electrochemical biosensors
3.1. Printing methods for electrochemical sensor fabrication
Printing methods have been widely used in the production of electrochemical sensors and biosensors owing to their ability to fabricate miniaturized, inexpensive, and repeatable devices.69 It involves the controlled deposition of functional materials (e.g., conductive inks, enzymes, or other biomaterials) onto a substrate. Table 1 lists the significant ink properties of five typical printing processes: screen, gravure, inkjet, flexography, and offset. Gravure and inkjet printing processes use low-viscosity inks with tiny particles to facilitate precise jetting, whereas screen and offset printing processes use much thicker, high-viscosity inks.14,70,71 While surface tension is less significant in screen printing, it is crucial in inkjet printing to ensure perfect droplet formation. While all those processes use inks containing small particles, they differ in their solids content. Inkjet and gravure printing methods use lower solid content, whereas screen and offset printing methods use higher solid content, which affects drying time and final print properties.72–74| Method | Viscosity | Surface tension | Particle size | Other traits | Ref. |
|---|---|---|---|---|---|
| Inkjet | Low | Medium | Very small | Fast-drying, low solids | 47–49 |
| Screen printing | Very high | Not critical | Small | High solids, thick | 75 |
| Gravure | Medium | Low | Very small | Fast-drying, low solids | 76 |
| Flexography | Medium | Medium | Small | Fast-drying, medium solids | 77 |
| Offset | Very high | Not critical | Small | High solids, sticky | 49, 50 |
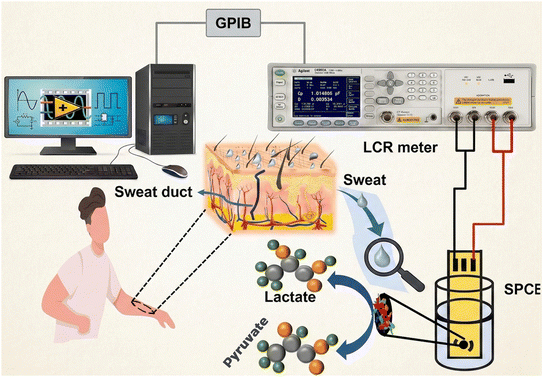 | ||
| Fig. 3 The process of lactate detection using a benchtop E4980A precision LCR meter and a screen-printed carbon electrode. Reprinted with permission from ref. 80. Copyright (2023) Elsevier. | ||
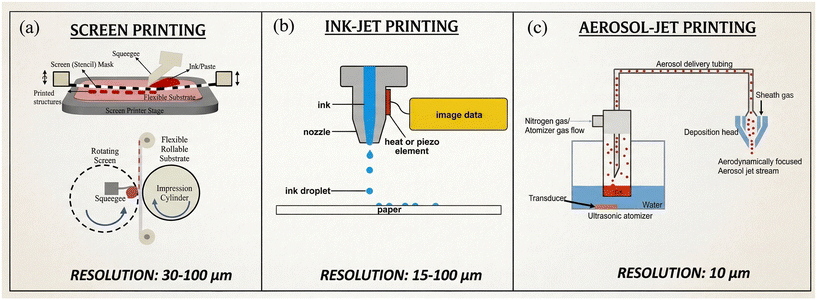 | ||
| Fig. 4 The schematic of (a) screen-printing, (b) inkjet printing, and (c) aerosol jet printing method. Reprinted with permission from ref. 14. | ||
Among all 3D printing processes, the FDM process, schematically shown in Fig. 5(a), is widely recognized as the most popular, attributed to its simplicity, ease of operation, and low cost.16 While in FDM, a thermoplastic is melted and squeezed out of a nozzle, a photosensitive resin is selectively cured with UV light or a laser in SLA both techniques support the creation of complex geometries as well as the integration of various functional materials, such as conductive inks, enzymes, and polymers, into the printed architecture.16,89–92 Ability to construct complex 3D structures and geometries,93 shape flexibility and rapid prototyping, integration of multiple materials and functionalities in a single device, minimal material loss, and cost-effectiveness for small-scale production are among the advantages of the 3D printing method. However, a limited range of compatible materials, especially conductive inks and biological components, and relatively low printing rates for mass production are disadvantages of the 3D printing method.
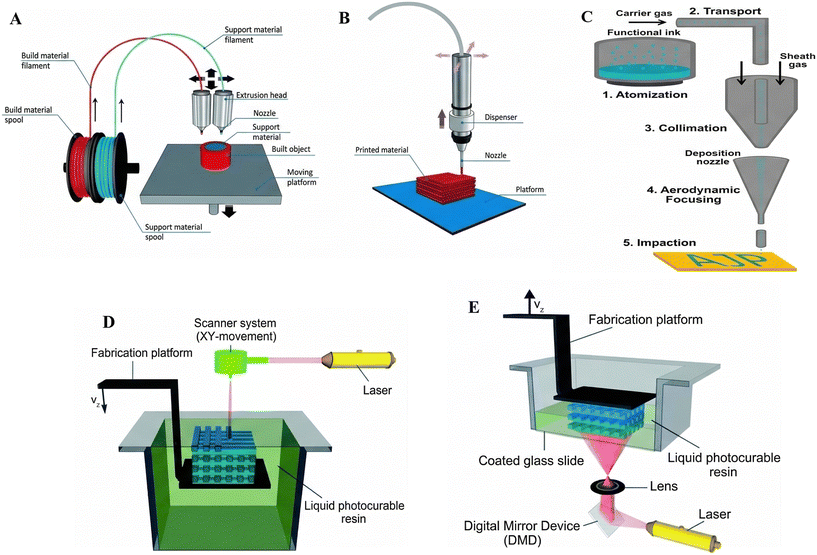 | ||
| Fig. 5 Schematic of the 3D printing method showing a heated nozzle precisely depositing semi-molten thermoplastic material layer by layer to build a 3D structure (A). The DIW technique uses a controlled dispenser to lay down viscous ink material in a similar layer-by-layer fashion (B). With permission from The Royal Society of Chemistry.94 AJP additive manufacturing method for printing with functional ink (C). Reproduced from ref. 95, with permission from IOP science. Laser-scanning SL with a free surface/bath configuration (D), and DLP SLA employing a constrained-surface “bat” configuration (E), with permission from Elsevier.97 | ||
Apart from FDM and SLA, the direct ink writing (DIW) method, schematically depicted in Fig. 5(b), is a new 3D printing technology that revolutionizes additive manufacturing by enabling one-step printing of functional inks and minimizing post-processing. DIW significantly expands 3D printing capabilities, enabling rapid prototyping and low-cost fabrication of complex, functional structures across industries.94 The aerosol jet printing (AJP) additive manufacturing pathway used for functional ink printing is schematically illustrated in Fig. 5(c).95 Stereolithography (SLA) is a 3D printing technology that builds objects layer by layer using UV light to cure liquid resin. The two settings are bath and bat. Under the bath setting, a stream of UV traces a 2D cross-section on a submerged substrate submerged in a pool of liquid resin. The substrate is lowered after each layer, and a blade cuts off the top surface of the resin. The bat structure features a suspended substrate and a light source beneath the tank. The two configurations outlined schematically in Fig. 5(d) and (e) are either laser- or digital light processing (DLP)-powered for controlled photopolymerization.96,97
A novel 3D-printed wearable patch was fabricated in a study by Kim et al.98 (Fig. 6). This patch incorporated a microfluidic channel and versatile ion-selective sensors, enabling in situ monitoring of sweat electrolytes, specifically Na+, K+, and Ca2+ ions. To achieve selective detection of these target ions, ionophores made of polyvinyl chloride (PVC) were incorporated into the 3D-printed electrode. Fig. 6(a–e) shows the AIIW (all-in-one wearable) patch, which consists of two main elements. First, a flexible, 3D-printed sensing unit responsible for detecting and analyzing sweat biomarkers. Second, a flexible WMFSH (wearable microfluidic fresh sweat harvesting) unit is designed to establish seamless skin contact and efficiently collect fresh sweat for analysis. This WMFSH unit is securely attached to the skin using a skin-friendly double-sided adhesive layer, ensuring continuous sweat sampling and accurate readings by the sensing unit.98 Fig. 6(f–h) shows two main parts of the sweatainer system: the sweatainer device itself and the epidermal port interface that connects it to the skin. The sweatainer device comprises a 3D-printed microfluidic network with enclosed channels and open reservoirs, a 200 μm-thick PDMS layer that seals the reservoirs, and a 60 μm-thick gasket made from medical-grade adhesive. It shows how it's put together and how it attaches to the skin. The sweatainer itself is a 3D printed structure with tiny channels and open areas for sweat to collect. It's sealed with a thin PDMS layer and attached to the skin using a special medical adhesive.99 Real-time sweat monitoring with the AIIW patch is demonstrated in Fig. 6(i) and (j). The patch, secured to the subject's forearm with a double-sided adhesive band, continuously tracked sodium, potassium, and calcium levels in sweat during and after exercise.98
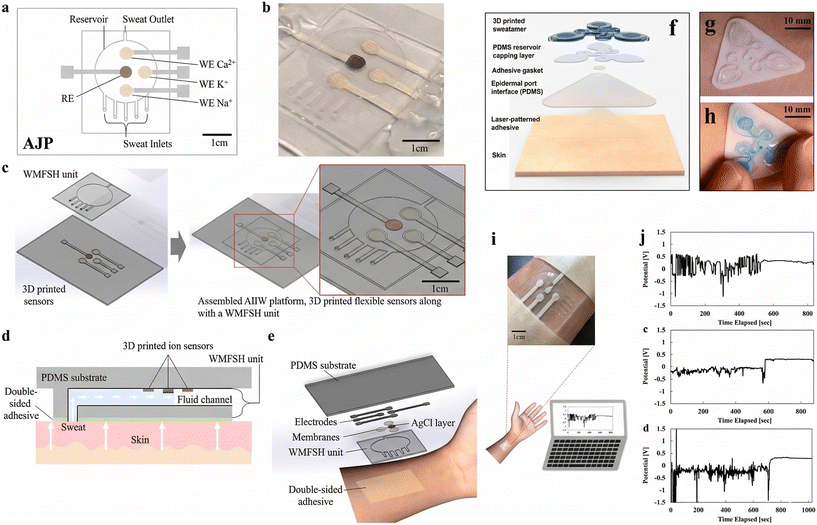 | ||
| Fig. 6 Schematic representation (a), an optical image of the actual device (b), its individual units (c), a cross-section illustrating its attachment to the skin (d), and a breakdown of its various components.98 (e) An illustration of 3D printed microfluidic devices designed for epidermal sweat detection (f–h) reprinted from ref. 99. Real-time sweat analysis for measuring sodium, potassium, and calcium levels (i) and their relative results (j). Reprinted with permission from ref. 98. | ||
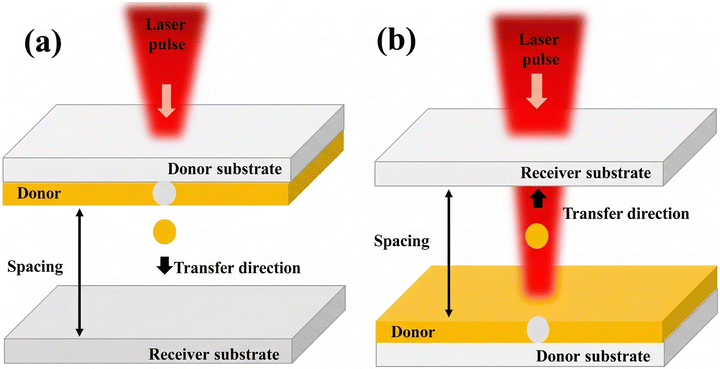 | ||
| Fig. 7 Schematics of setups for (a) LIFT and (b) LIBT. Reprinted with permission from ref. 101. | ||
A schematic of the R2R printing method is shown in Fig. 8(a). A flexible PET substrate is fed through a series of processing units, including a screen-print unit, an aligner, a UV curing unit, and an oven. This process is repeated multiple times to create the biosensor's layered structure. A sample of a biosensor produced by this method is shown in Fig. 8(b). A printed patch made by the R2R method, attached to a body part, is shown in Fig. 8(c). This type of printed biosensor can be used to evaluate the composition and dynamics of exercise- and iontophoretic-sweat samples. In research conducted by Nyein et al., R2R-printed biosensors were used to measure analytes in sweat and sweat rate simultaneously. These studies aim to explore the connections between sweat parameters and an individual's health or fitness level.104
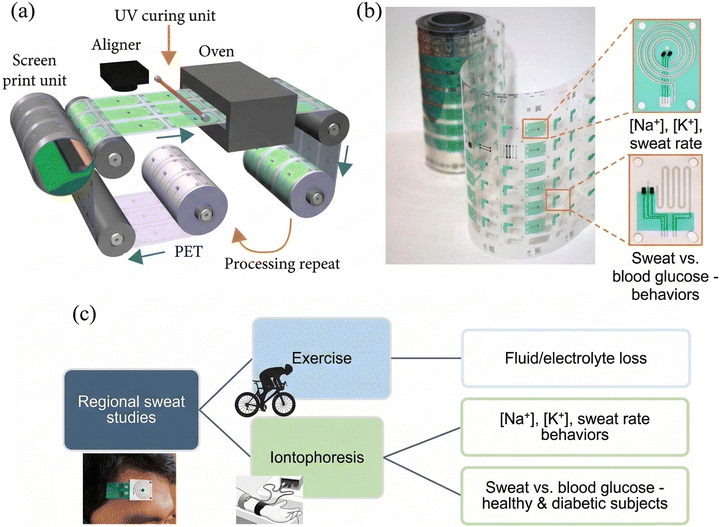 | ||
| Fig. 8 Schematic of roll-to-roll screen printing (a), biosensor fabricated by roll-to-roll printing method (b), and application of a printed patch attached to part of a body, which can be used to study the composition and dynamics of exercise and iontophoretic sweat (c).104 | ||
3.2. Electrode materials
Various electrode materials are applied in printed electrochemical (bio)sensors, and the choice is based on factors such as conductivity, electrochemical activity, and compatibility with the printing method. Carbon-based materials, such as carbon pastes, carbon nanotubes, and graphene, are traditionally used for their high electrical conductivity, low cost, and ease of printing. In addition, metal nanoparticles, such as gold, platinum, and silver, exhibit high electrocatalytic activity and are commonly used in printed electrodes to improve sensitivity and selectivity.72,74 Flexible polymers such as poly(3,4-ethylenedioxythiophene) polystyrene sulfonate (PEDOT:PSS) and polyaniline have also been studied for their flexibility, biocompatibility, and potential for printing using a broad range of methods.16,90,92 The selection of electrode materials is critical for the enhanced performance of printed electrochemical sensors for various applications.96,100,103 Table 2 provides information on the functional materials and electrolytes used in printed electrochemical sweat analysis sensors, along with the printing techniques employed. While potassium chloride (KCl) and sodium chloride (NaCl) are ubiquitous electrolytes in most methods, others, such as phosphate-buffered saline (PBS) and artificial sweat, are also used to mimic physiological conditions.27,33,55,103 Notably, functional materials such as silver/silver chloride ink are commonly used in inkjet, laser, roll-to-roll, aerosol jet, and electrohydrodynamic jet printing, indicating their ease of use in these processes. This inconsistency in the selection of electrode and electrolyte materials is a testament to the ongoing interest in enhancing sensor performance and tailoring them for specific analytes and printing techniques.23,99,113,114| Printing method | Electrode materials | Electrolyte materials |
|---|---|---|
| Screen printing | Carbon4,26,115,116 gold72,117 silver118–120 platinum92,121 | Potassium chloride (KCl)4,26,115,116 sodium chloride (NaCl)122 phosphate buffer saline (PBS)92 artificial sweat120 sodium sulfate (Na2SO4)116 |
| Inkjet printing | Silver123–126 gold125,126 carbon127 PEDOT:PSS128–130 | Silver/silver chloride (Ag/AgCl) ink124,131,132 potassium chloride (KCl)125,126 sodium chloride (NaCl)132 phosphate buffered saline (PBS)123,133,134 potassium nitrate (KNO3)21,135 |
| 3D printing | Carbon136–138 gold139 silver139,140 platinum93,141 | Aqueous electrolytes with potassium chloride (KCl) or sodium chloride (NaCl)94,142 |
| Laser printing | Silver,142 gold,142 carbon,33 graphene102,103 | Silver/silver chloride (Ag/AgCl) ink143 potassium chloride (KCl),144 sodium chloride (NaCl)144 |
| Roll-to-roll printing | Silver,145 gold,145 carbon146 | Silver/silver chloride (Ag/AgCl) ink147 potassium chloride (KCl),124 sodium chloride (NaCl)146 |
| Aerosol jet printing | Silver,148 gold105 | Silver/silver chloride (Ag/AgCl) ink73,149 potassium chloride (KCl)150 sodium chloride (NaCl)151 |
| Electrohydrodynamic jet printing | Silver,152 gold153 | Silver/silver chloride (Ag/AgCl) ink153,154 sodium chloride (NaCl)155 |
3.3. Substrate materials
The success of the printing process depends heavily on the compatibility of the inks with the substrate surface. In addition, the choice of substrate will have a profound impact on the mechanical and thermal properties, as well as the performance, durability, and wearability of the printed electrodes. Therefore, the choice of substrate is a critical factor in the development of printed electrochemical sensors for sweat analysis.26 Traditionally, rigid substrates such as glass, silicon, and ceramics have been used due to their excellent chemical stability and well-defined surface properties.156 However, the growing demand for flexible and wearable sensors has shifted focus towards more pliable materials. Polymer-based substrates such as polyethylene terephthalate (PET), polyimide (PI), and polyethylene naphthalate (PEN) have gained popularity due to their flexibility, low cost, and compatibility with various printing techniques.157 These materials enable the fabrication of conformable sensors that can adapt to the curvature of the human body, enhancing comfort and maintaining consistent skin contact.4In recent years, there has been increasing interest in stretchable substrates further to improve the wearability and durability of sweat sensors. Materials such as polydimethylsiloxane (PDMS) and thermoplastic polyurethane (TPU) offer excellent elasticity and biocompatibility, allowing sensors to withstand repeated deformation without compromising their performance.158 These stretchable substrates enable the development of sensors that can accommodate the dynamic nature of human skin during movement, maintaining sensor functionality and skin contact even during physical activity.159 The motivation for using different substrates is to tailor sensor properties to meet the requirements of specific applications. Paper-based substrates, for instance, have gained attention for their low cost, biodegradability, and potential for disposable sensors.160 Conversely, cloth-based substrates enable the direct embedding of sensors into clothing in an unobtrusive manner, providing a convenient platform for continuous sweat monitoring.161 In addition, the substrate properties can affect printing and the final properties of the resultant sensors. For example, some substrates require surface pretreatments and the use of primers to enhance adhesion and the uniformity of printed layers.162 Thermal and chemical stability of the substrate should also be considered, particularly when post-processing at high temperatures is used, or the sensor is exposed to various chemical environments in sweat.8
Briefly, the selection of substrate material plays a vital role in the flexibility, stretchability, biocompatibility, chemical resistance, and printability of the printed sensors. With advancements in the field, new substrate materials and composites continue to be developed to meet the evolving demands of wearable sweat-sensing technology, pushing the boundaries of sensor performance and wearer comfort.163
4. Printed electrochemical (bio)sensors for sweat analysis
Table 3 summarizes various studies on printed electrochemical sensors for sweat analysis. It encompasses a range of printing methods, including laser scribing, screen printing, inkjet printing, and 3D printing. The analytes detected by these sensors vary widely, from ions such as sodium, potassium, and chloride to metabolites such as glucose and lactate, and even complex biomolecules such as cortisol. The substrate materials used for printing also vary, including polyethylene terephthalate (PET), polydimethylsiloxane (PDMS), PVC paper, nonwoven cloth, and even stretchable substrates. The table highlights the diverse applications of those printed sensors, ranging from hydration status assessment and disease diagnosis for athlete performance monitoring to drug detection. It also showcases the versatility of printing technologies in fabricating wearable sensors with integrated features like microfluidics, wireless communication, and energy harvesting. However, the table also reveals some of the challenges in this field, such as the need for improved sensitivity and stability of printed sensors, especially under mechanical stress and in complex biological fluids. Additionally, integrating multiple functionalities into a single device while maintaining miniaturization and wearability remains an ongoing area of research.| Printing method | Analyte type | Substrate materials | Ink material | Main focus of the paper | Ref. |
|---|---|---|---|---|---|
| Laser scribing | H2O2 | Polyimide | — | Wearable sweat-capture device with patterned graphene arrays | 164 |
| Screen printing | K+, Na+ | Polyethylene terephthalate (PET) | Ag/AgCl, carbon, insulator inks | Sweat sensor for assessing hydration status | 165 |
| Screen printing | Creatinine, urea, pH | Polydimethylsiloxane (PDMS) | — | Passive sweat collection and colorimetric analysis | 3 |
| Screen printing | Glucose | PVC paper | Carbon ink, MXene/NiSm-LDH | Enzyme-free electrochemical glucose sensor | 166 |
| Screen printing | 167 | ||||
| Screen printing/heat-transfer | Cl−, Na+, K+, NH4+ | PET, nonwoven cloth | Carbon, silver, resist inks | Wearable ion sensor for sweat monitoring | 168 |
| Screen printing | Na+, NH4+, lactate | Polyethylene terephthalate (PET) | Gold nanoparticle, Ag/AgCl inks | Flexible printed electrochemical sensing platform | 169 |
| Screen printing | Glucose, lactate | Paper | Carbon, silver inks, Ti3C2Tx/MB | Low-cost integrated sensing paper for sweat analysis | 170 |
| Screen printing | Na+, K+, glucose, lactate, pH | Stretchable substrate | Elastomeric or silver inks | Stretchable sweat-sensing platform with integrated battery and display | 171 |
| Screen printing | Uric acid, tyrosine, ascorbic acid | Polyimide (PI) | Cu, Au, Ag/AgCl ink | Wearable sweat sensor with laser-induced graphene electrodes | 172 |
| Screen printing | Glucose, lactate, Na+, K+, Cl−, pH | PET | Carbon, Ag/AgCl | Highly integrated sensing paper (HIS) for real-time sweat analysis | 170 |
| Inkjet printing | Glucose | Tattoo paper | Prussian blue, glucose oxidase, chitosan | Wearable tattoo-based biosensor for glucose monitoring | 173 |
| Screen printing | Cd2+, Pb2+, uric acid, estradiol | Microbial nanocellulose (MNC) | Carbon paste | Microbial nanocellulose as a substrate for wearable electrochemical sensors to detect various analytes in sweat | 174 |
| 3D printing | Cortisol | Nanoporous flexible polyamide membrane | Palladium, MoS2 nanosheets | Non-faradaic label-free cortisol biosensor using MoS2 nanosheets integrated into a nanoporous flexible electrode system | 175 |
| Screen printing | Cortisol | Flexible nanoporous polyamide substrate | Gold | Four-channel electrical impedance spectroscopy (EIS) module for cortisol biosensing in sweat | 176 |
| Screen printing | Cortisol | Paper (Whatman no. 1 filter paper) | Polydimethylsiloxane (PDMS) | Portable 3D microfluidic origami biosensor for cortisol detection in human sweat | 177 |
| Inkjet printing, screen printing | Sweat rate, conductivity, copper ions | Polyethylene terephthalate (PET) | Silver nanoparticles (AgNPs), carbon, Ag/AgCl inks | Fully printed wearable microfluidic nanosensor for sweat rate, conductivity, and copper detection | 178 |
| Screen printing | Lactate | MgO-templated carbon (MgOC) | Poly(ethylene glycol)diglycidyl ether (PEGDGE), 1,2-naphthoquinone (1,2-NQ), lactate oxidase (LOx) | Continuous sweat lactate monitoring system with integrated screen-printed MgO-templated carbon-lactate oxidase biosensor and microfluidic sweat collector | 179 |
| Screen printing | Urea | Polyethylene terephthalate (PET) | Polyaniline (PANI) ink, urease bioink, polyvinylchloride (PVC) membrane | Wearable potentiometric biosensor for on-body and on-site monitoring of urea in sweat | 180 |
| Screen printing | Urea, ammonium | Polyethylene terephthalate (PET) | Carbon, silver/silver chloride (Ag/AgCl) inks | Touch-based potentiometric sensors for simultaneous detection of urea and ammonium from fingertip sweat | 181 |
| — | Cortisol | — | — | Wearable and implantable cortisol-sensing electronics for stress monitoring | 182 |
| Screen printing inkjet printing, 3D printing, aerosol jet, printing | Urea, glucose, lactate, electrolytes | — | — | Wearable organic electrochemical transistor patch for multiplexed sensing of calcium and ammonium ions from human perspiration | 183 |
| Screen printing, inkjet printing, 3D printing | Zn, Cd, Pb, Cu, Hg | — | — | Wearable sensors: modalities, challenges, and prospects | 1 |
| — | Na+, K+ | — | — | A thread/fabric-based band as a flexible and wearable microfluidic device for sweat sensing and monitoring | 184 |
| — | Glucose, lactate | — | — | Flexible textile-based sweat sensors for wearable applications | 185 |
| Screen printing | Sweat rate and electrolyte concentration | — | — | An unconventional vertical fluidic-controlled wearable platform for synchronously detecting sweat rate and electrolyte concentration | 186 |
| — | Glucose | Flexible materials | — | Graphene-interfaced flexible and stretchable micro-nano electrodes: from fabrication to sweat glucose detection | 187 |
| Screen printing | Cortisol | Paper | — | A portable 3D microfluidic origami biosensor for cortisol detection in human sweat | 177 |
The implementation of the R2R printing method for microfluidic chip fabrication and its use in sweat analysis was reported by Javey et al.104 The authors modified the PET substrate surface by forming silver and carbon electrodes via R2R printing. It should be noted that before printing, the PET surface was heated to 140 °C to enhance substrate compatibility. After printing, the electrodes were cured in an oven. The fabricated microfluidic wearable paths were used for sweat analysis, specifically to determine [Na+], [K+], glucose concentrations, and sweat rate in both exercise and chemically induced sweat conditions. Whilst the R2R printing method has been widely adopted for stretchable and flexible wearable electronic devices, its use in sweat analysis is rare. A recent study demonstrated the applicability of the R2R screen-printing method for sweat-sensing devices.188 The authors developed R2R screen-printed single-use sweat-sensing devices with high reusability and a robust interface, and the performance of the sensing systems was elucidated under both environmental heating and exercise conditions. To minimize sample (sweat) loss during transport from the sweat collector to the detection area, a microfluidic adhesive tape was implemented. The developed device designs are shown in Fig. 10. While the sensors showed excellent promise for real-time monitoring of sweat rate, the small sensor capacity hinders the use of those microfluidic devices for more than 42 active minutes. Design limitations, such as motion artifacts, limit the design's sample capacity.
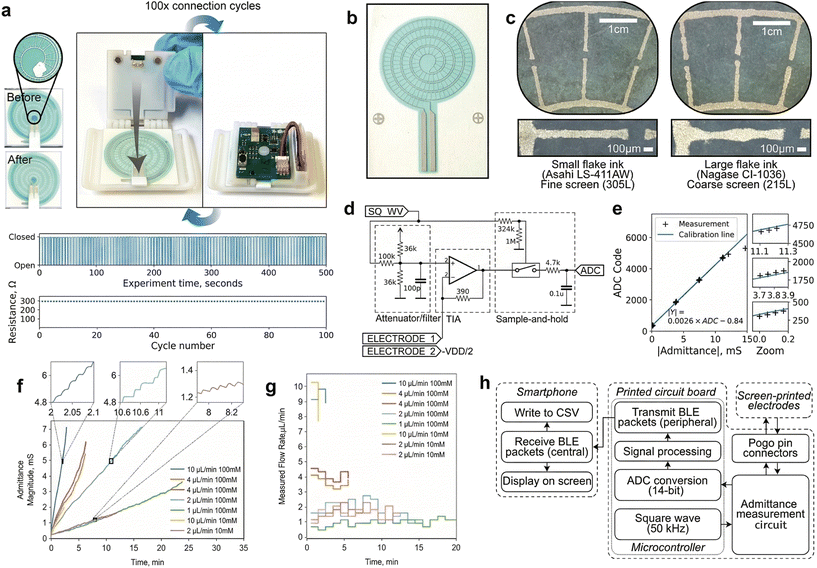 | ||
| Fig. 10 The design and testing of a new integrated circuit for measurements. (a) The first step involved testing the durability of the connection between the PCB and electrodes, which was achieved with spring-loaded pins and magnets; over 100 cycles, there was no significant increase in either contact or electrode resistance. (b) The electrode was fabricated by silk-screen printing silver ink onto a TPU 2D substrate (c), with LS-411 AW ink producing sharper patterns due to a finer mesh screen. (d) The core of the system is an admittance measurement circuit that includes a configurable excitation signal, a transimpedance amplifier, and a sample-and-hold detector, with its output being a voltage read by a microcontroller's ADC. (e) This channel was calibrated using discrete resistors. (f) and (g) Raw admittance signal obtained from benchtop calibrations. (h) A block diagram illustrates the complete measurement system used for all admittance measurements, with permission from ref. 188. | ||
One of the most widely used printing technologies for developing wearable sweat sensors is screen printing. As shown in Table 3, various functional materials, including carbon, silver NPs, Ag/AgCl, Cu, Au, PDMS, PANI, and TMDs, were printed onto different substrates (PDMS, PET, PI, paper, cloth, PVC paper, carbon template, etc.). Yang et al.,165 for instance, prepared screen-printed, flexible, and wearable sweat sensors to measure Na+ and K+ ions in human sweat. The sensors consisted of six layers: a PET substrate, Ag/AgCl, carbon, PEDOT:PSS, and ISM. While the PET, Ag/AgCl, and carbon layers were implemented to facilitate signal conduction, the PEDOT:PSS layer served as the transducer in the sensor design. The sensor surface was then covered with an insulating layer formed by screen-printing insulating material and UV curing. The performance of independently prepared sensors was evaluated, yielding RSD% values of 9.73% and 8.86%, indicating acceptable deviation. The sensors displayed 55.731 and 54.480 mV per decade slopes between 1–32 mM K+ and 5–160 mM Na+ concentrations, respectively. In another study, Vinoth et al.189 designed a microfluidic sweat-sensing device with low-dimensional sensing components via screen printing. The sampling efficiency was also enhanced by enhancing the hydrophobicity of the microfluidic channels. The microfluidic channels were prepared by the screen-printing of polystyrene-block-polybutadiene-block-polystyrene (SBS, styrene 30 wt% %) binder-based screen-printable carbon inks. The silver reference and carbon-based working electrode patterns were printed on flexible PI films (50 μm). It is essential to note that the authors devoted significant effort to developing printable silver and carbon inks with the desired viscosity and physical properties to meet the requirements of the screen-printing method. The performance of the wearable sensors was assessed against lactate, Na+, K+, and pH levels. The analytical responses of the ion-selective sensors were evaluated in vitro, and sensitivities of 9.5 and 60.1 mV per decade were obtained in the linear ranges of 0.02–200 mM and 0.01–100 mM for Na-ISE and K-ISE, respectively. The developed wearable sensors were also employed in pH sensing, and the pH values could be measured within the 3–8 pH window with a sensitivity of 71.4 mV pH−1. Besides carbon-based materials, metal nanoparticles (Au, Cu, Ag, Pd, etc.) have also been used in the screen-printing method to develop wearable sensors, as reported by Chen et al.,172 wearable sweat sensors with high analytical performance and mechanical stability were developed by screen-printing Cu and Au layers to form the conductive pathway and prevent electronic resistance, respectively. Apart from the above-mentioned wearable sensors, the authors included laser-induced graphene formation technology to enhance sensor performance by increasing the physical surface area. The sensor's performance was tested for uric acid (UA), tyrosine (Tyr), and ascorbic acid (AA), demonstrating high sensitivity. Bimetallic compositions were also implemented in screen printing technology. Two-dimensional (2D) materials offer significant advantages for printing technology due to their tailorable surface properties, excellent electron transport, and large physical surface area. On the other hand, the restacking issue in layered 2D materials, driven by van der Waals (vdW) forces between layers and by hydrophobic surfaces, should be carefully addressed to achieve inks with high homogeneity, stability, and printability. The discovery of graphene revolutionized 2D material research, and it has been one of the most widely used 2D materials in printing technology. Graphene-based materials have been used in screen printing in various forms: graphene oxide, reduced graphene oxide, and pristine graphene.
While a vast range of conductive or non-conductive materials can be used as functional material in the ink composition, the printing parameters play a crucial role in achieving high-quality printed patterns. Therefore, the key printing parameters, including deposition height, contact angle, pseudoplasticity, filament size, pressure applied during the printing process, mesh geometry, interaction of the ink with the substrate, ink's cohesive and adhesive forces, material loading, viscosity of the ink, viscosity recovery time, and yield stress are essential parameters to achieve high print resolution and quality.190 Therefore, in general, inks contain organic binders and rheological property modifiers, in addition to the functional material and the solvent—for example, as reported by Sen et al.191 prepared flexible sensors by screen printing of MWCNT on acetate paper. The inks were prepared by mixing MWCNT, acrylic varnish, hardener, and liquid paraffin. Li et al.170 investigated the electroanalytical performance of a printed sensing paper for direct analysis of sweat. The authors emphasized the importance of sweat collection, the use of wearable sensors for long-term study, and skin comfort.
Within this purpose, the researchers developed a cost-effective, disposable sensing paper (HIS) for sweat analysis. In the given sensor design, MXene/methylene blue (Ti3C2Tx/MB) hybrids were incorporated as active material. On the other hand, it should be noted that while the electrodes were fabricated by screen-printing of silver and carbon pastes, the Ti3C2Tx/MB functional material was drop-cast onto the working electrode surface. Thereafter, the working electrodes were modified with biological components to detect glucose and lactate levels. Bandodkar et al.173 developed a fully printed tattoo-based iontophoretic biosensor with a different electrode pattern by screen printing. The in vitro performance of the tattoo sensor showed a linear response over 0–100 μM glucose, with a sensitivity of 23 nA μM−1 and a limit of detection of 3 μM.
Li et al.164 documented a sweat collector based on graphene-patterned-array wearable devices. This work discussed the device's capability to collect and quantify sweat droplets. The electrochemical function of the device was demonstrated by H2O2 detection, with a limit of detection of 6 μM. Shitanda et al.179 developed an online continuous lactate sensor system with an in-built screen-printed MgO-templated carbon-lactate oxidase biosensor. The system was determined to have a lactate detection limit of 0.3 mM and a dynamic range of 50 mM. The most common printing method is screen printing, which is used in most of the listed studies in Table 3. It is so popular because it has the ability to print functional inks of different types on various substrates, such as flexible and stretchable materials. This ability makes it a strong contender for wearables—for instance, Yang et al.165 employed screen printing to fabricate a sweat sensor on polyethylene terephthalate (PET) for hydration status monitoring by measuring K+ and Na+ ions. Similarly, Li et al.170 employed screen printing to create a highly integrated sensing paper (HIS) on PET for real-time multi-sweat analyte analysis (glucose, lactate, and electrolytes).
The choice of substrate materials also varies significantly, with PET, PDMS, and paper being commonly used. For example, Bandodkar et al.173 developed a tattoo-based glucose sensor on tattoo paper via inkjet printing, demonstrating the potential for discreet, non-invasive monitoring. On the other hand, Kinnamon et al.175 used a 3D-printed, nonporous, flexible polyamide membrane for cortisol detection, highlighting the use of advanced manufacturing techniques to enhance sensor performance.
The performance of sweat sensors is evaluated using metrics such as sensitivity, linear range, and limit of detection (LOD). These metrics provide insights into the sensor's ability to accurately and precisely measure the target analyte. For instance, the glucose sensor developed by Li et al.170 exhibited a linear range of 0.08–1.25 mM with a LOD of 17.05 μM, demonstrating its capability to detect glucose at physiologically relevant concentrations. Similarly, the lactate sensor by Shitanda et al.179 achieved a LOD of 0.3 mM and a dynamic range of up to 50 mM, making it suitable for monitoring lactate levels during exercise.
This table shows emerging trends in sweat-sensing technologies, such as the use of laser scribing to create patterned graphene arrays for sweat capture and the integration of microfluidics for continuous sweat monitoring. Furthermore, the development of wearable and implantable cortisol-sensing electronics and organic electrochemical transistor patches for multiplexed sensing, which point towards the future of personalized health monitoring, is well established in the literature.
5. Analytical performance metrics in sweat sensing
The analytical performance of wearable sweat sensors is typically evaluated on sensitivity, selectivity, stability, and response time. Among these, device sensitivity is the most important given the often-low concentrations of sweat biomarkers. Many printed electrochemical sensors now achieve sensitivities comparable to those of benchtop sensors. For example, a recently reported textile-based printed enzymatic sensor for metabolites showed high glucose sensitivity of 18.4 μA mM−1 cm−2 with a linear range of 20–1000 μM,192 which is sufficient to detect low (tens to hundreds of μM) glucose levels in sweat. Leveraging novel advanced nanomaterials and ink formulations (e.g., MXene or NiO-modified inks) further improved sensor sensitivity. For example, a recently reported lactate sensor demonstrated sensitivities up to hundreds of μA mM−1 cm−2 in vitro, which is an advancement for intense exercise monitoring.193The following analytical metric to be monitored in such sensors is selectivity. The target analytes must be measured without encountering interference from the sweat's complex matrix. Printed sensors employ ion-selective membranes, enzyme specificity, or molecularly imprinted polymers (MIPs) to achieve high selectivity. For example, a printed chloride sensor was reported to show no significant response to other sweat ions.168 Similarly, the screen-printed textile glucose sensor noted above used a Nafion diffusion barrier to block interfering substances.192 For cortisol, which is present at nM levels in sweat, published wearable sensors used MIP-based recognition. In one such flexible printed cortisol, the authors showed high specificity for cortisol, with negligible response to structurally similar hormones.194 Thus, it is indeed clear that printed sensors can be engineered for excellent specificity, even ones comparable to lab assays.
Stability and reproducibility are also parameters that need to be monitored for practical wearables. Printed sweat sensors have shown encouraging stability both in storage and during operation. The printed textile glucose sensor reference above retained ∼100% of its response after 30 days of storage, which demonstrates excellent shelf-stability1. Mechanical durability is also important, as such wearables undergo bending and exposure to sweat. Recent reports indicate that printed electrodes on flexible substrates indeed withstand repeated flexing without performance loss.195
When the sensing element is functionalized with biological recognition elements (e.g., enzymes), many designs include protective coatings (e.g., epoxies or hydrogels) to prevent enzyme leaching or ink degradation and extend sensor operational stability. In terms of signal stability, printed ion-selective electrodes (ISEs) can maintain stable potentials for hours of continuous use if properly calibrated and a stable reference electrode is integrated.168 Reproducibility between devices is generally high in these sensors due to the uniformity of the printing processes. For example, the printed ion sensors for Ca2+ and Cl− were reported to exhibit minimal sensor-to-sensor variance and repeatable calibration curves across multiple devices.168
Printed-on-skin sweat sensors can now be fabricated to display fast response times as well. The typical response time is on the order of seconds to several minutes, limited mainly by the transport of sweat samples rather than by intrinsic sensor kinetics. Electrochemical transducers (amperometric or potentiometric) respond almost instantaneously once sweat reaches the sensing interface. In practice, designs that minimize the required sample volume and improve sweat access yield the fastest responses. Notably, a recent breakthrough “meta-garment” with printed microfluidic fibers achieved a response time of only ∼1.4 s and required just 0.1 μL of sweat – the fastest and smallest volume reported to date.196 This is one example of a highly fast-response sensor; however, more commonly, printed sweat patches have response times on the order of tens of seconds as sweat wets the sensor. For example, a flexible pH sensor printed on a stretchable substrate reached steady-state readings in ∼8 s.197 Overall, leveraging thin and film-printed electrodes and efficient sweat collection enables modern devices to provide near-real-time monitoring of changing sweat chemistry.
6. Sensor effectiveness for common sweat biomarkers
Printed wearable sensors have been developed for a wide array of sweat biomarkers, with electrolytes and metabolites being the most commonly targeted. Analytes of interest include electrolyte ions (such as sodium (Na+), potassium (K+), chloride (Cl−), and calcium (Ca2+)), metabolites (such as lactate, glucose, urea, and amino acids), and hormones or other markers (such as cortisol for stress or ethanol for alcohol monitoring). The effectiveness of printed sensors in detecting these targets has significantly improved in recent years, with many devices now capable of real-time, on-body measurements that correlate with physiological levels.Electrolytes in sweat provide information on hydration status, electrolyte balance, and even disease diagnosis (for example, high sweat Cl− is a marker for cystic fibrosis).198 Printed potentiometric sensors (ISEs) are the leading technology for detecting electrolytes. These typically consist of a screen-printed reference electrode (Ag/AgCl or solid-state) and a working electrode coated with an ion-selective membrane specific to the ion of interest. The printed ISE approach is highly effective; for example, sensors for Na+ and K+ often exhibit near-Nernstian sensitivity (∼50–60 mV per decade concentration) and cover the whole physiological range of sweat (e.g., 10–100 mM Na+, 5–50 mM K+).168
A recent fully printed Na+/K+ patch demonstrated real-time tracking of electrolyte loss during exercise, with sensor readings closely matching expected changes in sweat composition.199 For chloride, as noted earlier, a printed sensor yielded an ideal −59.5 mV per decade response and showed no cross-sensitivity to other anions.168 As sweat contains a mix of ions (Na+, K+, Cl−, typically in the tens of millimolar range, and others like Ca2+ or Mg2+ in the lower millimolar range), such devices must meet these sensitivity levels. By incorporating ion-specific membrane cocktails (e.g., valinomycin for K+, selective ionophores for Na+ and Cl−), printed sensors effectively discriminate between ions. Many designs combine multiple ion sensors on one platform, such as when multiplexed printed patches can simultaneously measure Na+, K+, and Cl−, or pH, providing comprehensive sweat electrolyte analysis.34,200,201 The accuracy of these ion sensors in real sweat has been validated against standard analytical techniques. In one of the cited studies, for instance, a wearable Na+/Cl− sensor patch was compared with ion chromatography of collected sweat. It showed deviations of only a few percent, confirming the sensors' quantitative reliability.168
Printed wearable electrolyte sensors are highly effective, as they can provide continuous readings of ionic concentrations, support hydration monitoring, and even issue electrolyte imbalance warnings (as might be needed for athletes or workers in hot environments). Metabolic biomarkers in sweat, on the other hand, are indicators of physiological stress, exercise intensity, and metabolic health. Lactate in sweat, for example, correlates with muscle exertion and oxygenation, while glucose in sweat has been explored as a noninvasive proxy for blood glucose.164,165,170,173,189–191 Printed enzymatic biosensors are commonly used for these targets. In such sensors, an enzyme (e.g., lactate oxidase or glucose oxidase) is immobilized on a printed electrode and selectively reacts with the target metabolite. The resulting product (often hydrogen peroxide) is then detected electrochemically. The effectiveness of printed lactate sensors has advanced tremendously, and they can now measure both resting and exercise-induced levels. Typical sweat lactate might range from ∼1 mM at rest to >15 mM during intense exercise. A recent wearable lactate patch reported a linear range of 1–20 mM with sensitivity around 0.6 μA mM−1 (in a three-electrode configuration).202 This allows the user to track lactate build-up and clearance during workouts.
Glucose is a more challenging analyte due to its lower sweat concentration (often 100–200 μM, though highly variable). However, recently reported sensors, such as the textile-based sensor described earlier, had a detection range starting at 20 μM,192 which covers the upper end of typical sweat glucose levels. In on-skin tests, this sensor could clearly detect a rise in sweat glucose after the subject ingested a high-sugar meal. Although sweat glucose does not track blood glucose with as much precision as other targets, as it typically lags and is much lower in concentration, these sensors are effective for observing relative changes or screening for abnormal levels. They also tend to have high selectivity, as, for example, in the case of using a Prussian blue mediator and a Nafion coating in the printed glucose sensor, which minimized interference from co-existing lactate or ascorbate.192 Thus, it can be said that sweat glucose readings can be obtained reliably in real time, which represents a significant step toward noninvasive glucose monitoring.
Sweat sensors have successfully targeted other metabolites and small molecules. Cortisol, a stress hormone, is present in sweat in low nanomolar concentrations and has traditionally been measured via lab assays. Recent wearable designs have used printed electrodes functionalized with aptamers or MIPs for cortisol, demonstrating the ability to detect physiologically relevant fluctuations in cortisol levels. For example, a flexible cortisol patch with a printed CNT-MIP composite electrode detected a cortisol increase (∼10–20 nM) in sweat within 15 minutes of a stress event (ice-water hand dip), followed by its return to baseline.194 Such devices indicate that even hormones can be monitored by printed sweat sensors with reasonable sensitivity and time resolution.
Other biomarkers measured include sweat pH. Printable pH sensors are often made by screen-printing pH-sensitive polymer or oxide inks, which easily cover the normal sweat pH range of ∼4.5–7.5.183,202 Urea and ammonia are indicators of metabolic waste and kidney function and can now be measured using printed colorimetric patches or amperometric enzymatic sensors.192–195,203 Moreover, sweat sensor patches can also detect heavy metals or drugs. For example, sweat sensors on flexible substrates have been used to detect lead and cadmium in sweat for environmental exposure monitoring.196–198 While these latter sensors are less common, their development demonstrates the broad applicability of printed sweat sensing. Virtually, at the time of this review, any analyte for which a selective chemistry can be devised (enzyme, ionophore, molecular receptor, etc.) can find a printable ink that can be deployed in a sweat sensor for said analyte. The effectiveness of detecting each biomarker ultimately depends on how well the pairing between the appropriate sensing mechanism and the most performant printed platform is done.
7. Application
The development of wearable electrochemical sweat sensors has opened a wide range of applications across diverse fields, from sport and fitness monitoring and disease control to industrial safety and health, to individualized healthcare. Such applications leverage sweat sensors' ability to trace, in real time and non-invasively, a variety of biomarkers, providing valuable insights into an individual's physiological status and enabling targeted interventions.7.1. Sports and fitness monitoring
Wearable sweat sensors offer a powerful tool for athletes and fitness enthusiasts to monitor their hydration status, electrolyte balance, and metabolic markers during physical activity. By continuously tracking sweat composition, these sensors can provide real-time insights into an individual's physiological state, enabling informed decision-making and personalized training strategies.13,90,203Fig. 11 shows a collection of innovative wearable biosensors designed for sweat analysis, offering a glimpse into the future of healthcare and disease monitoring. Examples of sensors integrated into patches, smart bands, and even ear-worn devices that aim to capture and analyze various biomarkers in sweat are shown in this figure. These examples highlight the diversity of sensing mechanisms, from enzymatic reactions for lactate detection to electrochemical sensing for electrolytes and metabolites. Additionally, it emphasizes the trend towards non-invasive, real-time health monitoring, with devices capable of wirelessly transmitting data to smartphones for convenient tracking and analysis. These advancements in wearable biosensor technology hold immense promise for personalized healthcare, fitness tracking, and early disease detection.205–209
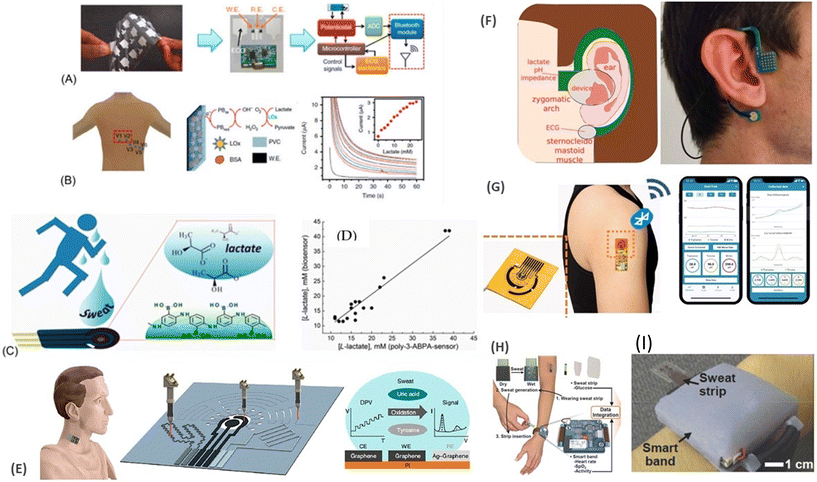 | ||
| Fig. 11 Device photographs of a sweat-based sensor on the body and its sensor features (A and B) with permission from Elsevier,204 schematic representations of screen-print, block diagram of the circuit, enzymatic sensing mechanism, and amperometric response of wearable sweat-based lactate biosensor (C) with permission from Elsevier,204 example of non-enzymatic lactate sensor in human sweat, and correlation diagram for lactate (D) with permission from Elsevier204 a wearable laser-engraved sensor for the sensitive detection of uric acid and tyrosine in sweat (E) with permission from ref. 205. A wireless ear-worn device capable of pH, lactate, and ECG sensing during physical exercise (F) with permission from ref. 206. A wearable electrochemical biosensor for monitoring of multiple metabolites and nutrients (G) with permission from ref. 207. A wearable smart band for detecting sweat-based glucose and vital signs (H), a real image of smart band on a human hand (I) with permission from ref. 208. | ||
Sodium, potassium, and chloride are electrolytes that serve essential functions in hydration and muscle function.209–211 Sweat sensors can quantify levels of these electrolytes, allowing athletes to optimize their hydration practices and electrolyte replacement.212 For example, Pirovano et al.16 developed a wearable sensor system that quantifies sodium and potassium ions in sweat during exercise. The researchers found that real-time feedback enabled players to maintain an ideal electrolyte balance, which is key to performance and recovery.
Lactate is a byproduct of anaerobic metabolism and a significant indicator of exercise and muscle fatigue.213–216 Real-time lactate monitoring using sweat sensor-based technology can help athletes track their intensity levels, prevent overtraining, and adjust their training regimens accordingly. Lee et al.217 developed a flexible, wearable glucose biosensor that accurately measured glucose concentration in sweat during exercise. Scientists demonstrated that this sensor can provide rivals with valuable information on exercise intensity and the capability to adjust their training programs. Dehydration also significantly impacts an athlete's performance and recovery. Sweat sensors can monitor biomarkers of hydration, including sodium, potassium, and chloride concentrations in sweat, as well as sweat flow rate and fluid intake. By providing instant feedback regarding hydration levels, such sensors will help athletes ensure they are optimally hydrated and prevent complications associated with dehydration.217,218
7.2. Disease management
Wearable electrochemical sweat sensors have the potential to revolutionize disease management by enabling continuous and non-invasive monitoring of biomarkers associated with various conditions. Sweat sensors have been developed to detect glucose levels, offering a promising alternative to traditional blood glucose monitoring for individuals with diabetes.219,220 Continuous glucose monitoring via sweat can provide better insights into glucose fluctuations, enabling more effective disease management. For instance, Xuan et al.221 developed a flexible, wearable electrochemical sensor that accurately monitors glucose levels in sweat. This technology could potentially eliminate the need for frequent finger-prick blood tests, improving patient compliance and quality of life. Fig. 11b demonstrates the schematic of an enzymatic biosensor to detect glucose in sweat.203Cystic fibrosis is a genetic disorder characterized by abnormally high chloride ion levels in sweat. Sweat sensors can be designed to detect chloride ion concentrations, enabling better management and treatment of this condition.222,223 Coyle et al.224 developed a wearable sweat sensor capable of measuring chloride levels in real time, which could be used to monitor cystic fibrosis patients and adjust their treatment accordingly. Specific biomarkers in sweat, such as creatinine and urea, can provide insights into kidney function.225 Wearable sweat sensors can continuously monitor these biomarkers, enabling early detection of kidney disorders and facilitating timely interventions. Alizadeh et al.41 developed a flexible, wearable sensor for monitoring creatinine levels in sweat, demonstrating its potential for noninvasive kidney function monitoring.
Sweat sensors can also be used to monitor biomarkers associated with metabolic diseases, such as diabetes and obesity.226,227 For example, Karpova et al.228 developed a wearable sensor to monitor metabolites, such as glucose and lactate, in sweat, providing valuable insights into metabolic processes and aiding in the management of metabolic disorders. Specific biomarkers in sweat, such as cytokines and other inflammatory markers, can indicate the presence of inflammatory conditions. Wearable sweat sensors can monitor these biomarkers, enabling early detection and personalized treatment strategies for conditions like rheumatoid arthritis, inflammatory bowel disease, and other autoimmune disorders.33,228
7.3. Occupational health and safety
In industrial and occupational settings, wearable sweat sensors can play a crucial role in monitoring worker health and safety. Sweat sensors can detect the presence of toxic substances or environmental pollutants, alerting workers to potential exposure and enabling timely intervention.178,229 For example, Gao et al.230 developed a wearable sensor that could detect heavy metal ions, such as lead and cadmium, in sweat. This technology could be valuable in industries where workers are at risk of exposure to toxic substances, such as mining, manufacturing, and construction. Certain chemicals and volatile organic compounds (VOCs) can be excreted through sweat, and their presence can indicate exposure to potentially harmful substances. Wearable sweat sensors can detect these chemicals, enabling monitoring and early intervention for workers in industries where chemical exposure is a risk, such as manufacturing, agriculture, and chemical processing.24,231 Sweat sensors can monitor physiological markers of heat stress, dehydration, and fatigue, which are critical considerations in physically demanding or extreme environments.232,233 By providing real-time feedback, these sensors can help prevent heat-related illnesses, improve worker productivity, and enhance overall safety conditions. Sonner et al. developed a wearable sweat sensor that could monitor electrolyte levels and sweat rate, enabling early detection of heat stress and dehydration in workers exposed to high temperatures or physically demanding tasks.7.4. Personalized healthcare
The integration of wearable electrochemical sweat sensors into personalized healthcare systems holds immense promise. By continuously monitoring an individual's sweat composition, these sensors can provide a comprehensive picture of their physiological state, enabling proactive and personalized healthcare interventions. Sweat sensors can be used to monitor medication adherence and therapeutic drug levels, ensuring optimal dosing and treatment efficacy.234–236 For example, Emaminejad et al.67 developed a wearable sensor that could detect specific drugs in sweat, enabling monitoring of medication adherence and adjusting dosages accordingly.8. Challenges and outlook
The field of printed wearable sweat sensors has seen notable breakthroughs in the past few years (2022–2025),237–241 even as specific challenges remain focal points of research. Improved materials design, innovative device architectures, and system-level integration are all contributing to these advances and to the high analytical performance of wearable sweat sensors. One significant advance in recent years is in multi-analyte sensing and system integration. Early sweat sensors typically measured a single biomarker, but recent devices can detect a panel of biomarkers simultaneously. For example, researchers demonstrated a fully integrated “smart wristband” that incorporated multiple printed sensors (for Na+, K+, and lactate) along with a microcontroller and a Bluetooth module on a flexible printed circuit, essentially representing a lab-on-a-watch for sweat analysis.189 In 2025, a “meta-garment” for firefighters was reported, requiring only 0.1 μL of sweat and 1.4 s to obtain readings.196Nanostructured materials such as MXene, conductive polymers, and nanoparticle composites have been formulated into printable inks to improve sensitivity and stability.115,217,222–225,227 For example, incorporating carbon nanotubes or graphene into the ink can increase electrode surface area and electron transfer, yielding higher currents for a given analyte concentration.194 Enzyme immobilization techniques were also improved by using polymer matrices or crosslinkers printed alongside the enzyme to prolong its activity. There have also been enzyme-free sensor developments, such as an MXene/Ni-LDH-based glucose sensor that achieves low detection limits via catalytic electrooxidation of glucose.167 These improvements often lead to better sensor stability and broader operating conditions. From a fabrication standpoint, 3D printing is emerging as a complementary approach for creating structural components of wearable sensors. One such example is 3D-printed microfluidic networks or scaffoldings that hold the printed electrodes in contact with the skin.
Despite these advances, several challenges remain to be addressed to achieve ideal printed sweat sensor designs. One challenge is the long-term stability during continuous wear. While sensors can last days, requirements for truly continuous health monitoring might demand weeks or months of operation without maintenance. This raises issues such as biofouling (skin debris accumulating on sensors) and long-term drift. Researchers are investigating antifouling coatings and on-the-fly calibration schemes to counter this.33,227 Another challenge is calibrating and standardizing sweat readings. Sweat composition can be influenced by sweat rate, and therefore, the same sensor reading might mean different things under different conditions. To address this, some wearable devices now include sweat rate sensors, such as printed microfluidic channels that measure sweat volume alongside chemical sensors, enabling normalization for dilution effects.242,243 Additionally, more work is needed to correlate sweat analyte levels with blood levels for specific biomarkers, such as glucose or cortisol. Extensive clinical studies are underway to correlate those relationships, which will inform the calibration models used in devices. Interference and cross-reactions pose additional challenges, especially in multiplexed devices. These challenges show there is still a need for improvements in system design when combining functionalities. Although printed sensors themselves require little power, another challenge is that wireless transmission and data logging do. Here, an exciting development is self-powered sweat sensors that harvest energy from the body or the environment. For example, biofuel cells that generate power from lactate in sweat, or flexible solar cells, can be used in such sensors.244,245
Another critical challenge arises when scaling up the fabrication process, as it is associated with maintaining the performance and reproducibility of both the fabrication process and the analytical response. It should be emphasized that analytical response repeatability is a key requirement for clinical applications. While the printing methods discussed in the given review are ready for scale-up operations, additional parameters should be considered when (bio)sensing technology is the intended application.
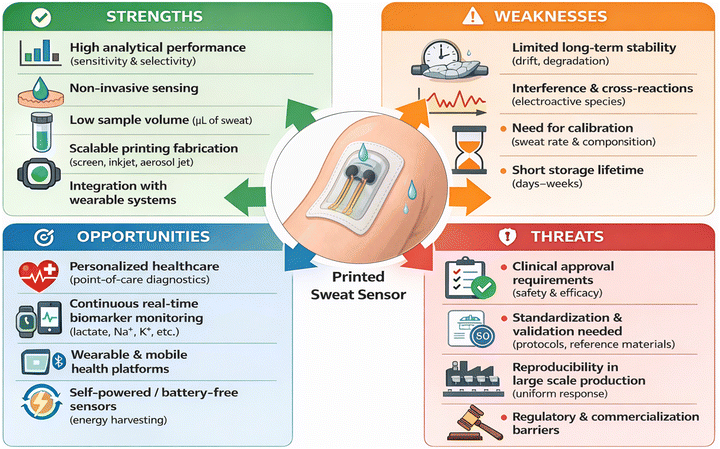
A SWOT (strengths, weaknesses, opportunities, and threats) analysis was performed to clearly demonstrate the strengths and weaknesses of the printed sensors (Fig. 12). High analytical performance, non-invasive and low-volume sensing capability, and a scalable fabrication process are among the strengths of printed sweat sensors. In addition, those sensors can be integrated into different systems, enabling the development of fully integrated sensing platforms. Therefore, the printed sensors demonstrate the strong potential in electrochemical sweat analysis in health, safety, and sports applications. On the other hand, some challenges, including limited long-term stability, interference, and cross-reaction problems, should be alleviated for real-world applications. In addition, to minimize false readings, the sensor response should be calibrated against fluctuations that happen in samples (sweat rate, changes in sweat composition depending on body condition) and across experiments. The current literature indicates insufficient storage time (e.g., days or weeks) and should be extended to months. Addressing the mentioned weaknesses in printed sweat-sensing systems will create significant opportunities in personalized healthcare, continuous and real-time biomarker monitoring, and the use of a non-invasive method. In addition, the design of self-powered analysis platforms will enable the development of battery-free wearable sensors. It should be noted that to have commercialized printed sweat sensors, several treats should be addressed, such as the approval of those systems for clinical applications, and validated and standardized results are crucial. Moreover, scalable, reproducible sensing platforms with uniform responses should be developed.
9. Conclusion
The development of printing methods and their application in fabricating low-cost, flexible electrodes has opened a new path in electrochemical sensors, especially for sweat analysis. Printed wearable sensors offer a transformative approach, especially in healthcare, by enabling noninvasive, continuous monitoring of biomarkers in sweat. As discussed, the implementation of versatile printing methods, including screen printing, inkjet printing, 3D printing, and aerosol jet printing, has revolutionized the fabrication of customized, flexible, and low-cost sweat sensors. Besides the fabrication methods, the development of new materials, including MXenes, carbon nanotubes, conductive polymers, metal nanoparticles, and graphene, with a wide range of electrochemical properties, has enabled the development of high-performance sweat sensors with low limits of detection and selectivity. The integration of sweat sensors with microfluidics, microcontrollers, and wireless or Bluetooth communication modules accelerated the development of sweat-sensing technology. While printed sweat sensors are important for future sensing technology, several challenges should be addressed, including insufficient sensor stability, issues with sweat collection and sampling, and nonuniform analytical response. The stability of the sweat sensors should be extended to weeks and even months for continuous monitoring. The variation in sensor response associated with fabrication and the analytical response reproducibility should be less than 10% RSD. Moreover, drifts in sensor performance and sensor voltage noise should be minimized. Non-enzymatic detection routes are considered a promising approach to enhance storage stability. Mitigating those technical challenges will make printed sensors indispensable, especially in sports physiology and the diagnosis of chronic diseases. For practical deployment, printed sweat sensors should be evaluated against several quantitative benchmarks. Recent reports indicate that excellent storage stability can reach ∼100% signal retention after 30 days, while future wearable platforms should target stability over weeks to months.165,246–248 For rapid and low-volume operation, state-of-the-art microfluidic systems have demonstrated response times of ∼1.4 s with only 0.1 μL of sweat. In terms of reproducibility, device-to-device variation should ideally remain below 10% relative standard deviation (RSD), as independently prepared screen-printed sensors have shown RSD values of 9.73% and 8.86%.165,249,250Conflicts of interest
The author declares they have no conflict of interest.Abbreviations
| 2D | Two-dimensional |
| 3D | Three-dimensional |
| Ag NP | Silver nanoparticle |
| AJP | Aerosol jet printing |
| CNT | Carbon nanotube |
| DIW | Direct ink writing |
| DLP | Digital light processing |
| EHD | Electrohydrodynamic |
| FDM | Fused deposition modeling |
| ISEs | Ion-selective electrodes |
| LCR | Inductance, capacitance, resistance |
| LDH | Layered double hydroxide |
| LIBT | Llaser-induced backward transfer |
| LIFT | Laser-induced forward transfer |
| LOD | Limit of detection |
| MgOC | MgO-templated carbon |
| MIP | Molecularly imprinted sensor |
| MNC | Microbial nanocellulose |
| MXene | Two-dimensional transition metal carbide/nitride |
| PANI | Polianilin |
| PBS | Phosphate-buffered saline |
| PDMS | Polydimethylsiloxane |
| PEDOT:PSS | Poly(3,4-ethylenedioxythiophene) polystyrene sulfonate |
| PEN | Polyethylene naphthalate |
| PET | Polyethylene terephthalate |
| PI | Polyimide |
| PVC | Polyvinyl chloride |
| R2R | Roll to roll |
| SLA | Stereolithography |
| SPCE | Screen-printed carbon electrode |
| SWOT | Strengths, weaknesses, opportunities, and threats |
| TPU | Thermoplastic polyurethane |
| UV | Uultraviolet |
| VOC | Volatile organic compounds |
| WMFSH | Wearable microfluidic fresh sweat harvesting |
Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
A. U. acknowledges the financial support of TUBITAK for the project: 122 N920 and TÜBA-GEBİP Award (Outstanding Young Scientist Awards-2025) provided by Turkish Academy of Sciences.References
- J. Heikenfeld, A. Jajack, J. Rogers, P. Gutruf, L. Tian, T. Pan, R. Li, M. Khine, J. Kim, J. Wang and J. Kim, Wearable sensors: modalities, challenges, and prospects, Lab Chip, 2018, 18, 217–248 RSC.
- R. Ghaffari, J. A. Rogers and T. R. Ray, Recent progress, challenges, and opportunities for wearable biochemical sensors for sweat analysis, Sens. Actuators, B, 2021, 332, 129447 CrossRef CAS.
- Y. Zhang, H. Guo, S. B. Kim, Y. Wu, D. Ostojich, S. H. Park, X. Wang, Z. Weng, R. Li, A. J. Bandodkar, Y. Sekine, J. Choi, S. Xu, S. Quaggin, R. Ghaffari and J. A. Rogers, Passive sweat collection and colorimetric analysis of biomarkers relevant to kidney disorders using a soft microfluidic system, Lab Chip, 2019, 19, 1545–1555 RSC.
- W. Gao, S. Emaminejad, Y. Y. N. Nyein, S. Challa, K. Chen, A. Peck and H. M. Fahad, Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis | Nature, Nature, 2016, 529, 509–514 CrossRef CAS PubMed.
- A. Koh, D. Kang, Y. Xue, S. Lee, R. M. Pielak, J. Kim, T. Hwang, S. Min, A. Banks, P. Bastien, M. C. Manco, L. Wang, K. R. Ammann, K.-I. Jang, P. Won, S. Han, R. Ghaffari, U. Paik, M. J. Slepian, G. Balooch, Y. Huang and J. A. Rogers, A soft, wearable microfluidic device for the capture, storage, and colorimetric sensing of sweat, Sci. Transl. Med., 2016, 8, 366ra165 Search PubMed.
- A. Mishra, R. Greaves and J. Massie, The Relevance of Sweat Testing for the Diagnosis of Cystic Fibrosis in the Genomic Era, Clin. Biochem. Rev., 2005, 26, 135–153 Search PubMed.
- L. B. Baker, Sweating Rate and Sweat Sodium Concentration in Athletes: A Review of Methodology and Intra/Interindividual Variability, Sports Med., 2017, 47, 111–128 CrossRef.
- M. Bariya, H. Y. Y. Nyein and A. Javey, Wearable sweat sensors, Nat. Electron., 2018, 1, 160–171 CrossRef.
- S. M. Cristescu, H. A. Gietema, L. Blanchet, C. L. J. J. Kruitwagen, P. Munnik, R. J. van Klaveren, J. W. J. Lammers, L. Buydens, F. J. M. Harren and P. Zanen, Screening for emphysema via exhaled volatile organic compounds, J. Breath Res., 2011, 5, 046009 CrossRef CAS.
- S. Jadoon, S. Karim, M. R. Akram, A. Kalsoom Khan, M. A. Zia, A. R. Siddiqi and G. Murtaza, Recent Developments in Sweat Analysis and Its Applications, Int. J. Anal. Chem., 2015, 2015, 164974 Search PubMed.
- M. M. Raiszadeh, M. M. Ross, P. S. Russo, M. A. Schaepper, W. Zhou, J. Deng, D. Ng, A. Dickson, C. Dickson, M. Strom, C. Osorio, T. Soeprono, J. D. Wulfkuhle, E. F. Petricoin, L. A. Liotta and W. M. Kirsch, Proteomic analysis of eccrine sweat: Implications for the discovery of schizophrenia biomarker proteins, J. Proteome Res., 2012, 11, 2127–2139 CrossRef CAS PubMed.
- M. Jia, W. M. Chew, Y. Feinstein, P. Skeath and E. M. Sternberg, Quantification of cortisol in human eccrine sweat by liquid chromatography – tandem mass spectrometry, Analyst, 2016, 141, 2053–2060 RSC.
- D. R. Seshadri, R. T. Li, J. E. Voos, J. R. Rowbottom, C. M. Alfes, C. A. Zorman and C. K. Drummond, Wearable sensors for monitoring the physiological and biochemical profile of the athlete, NPJ Digit. Med., 2019, 2, 72 CrossRef PubMed.
- E. Sardini, M. Serpelloni and S. Tonello, Printed Electrochemical Biosensors: Opportunities and Metrological Challenges, Biosensors, 2020, 10, 166 CrossRef CAS.
- B. Schazmann, D. Morris, C. Slater, S. Beirne, C. Fay, R. Reuveny, N. Moyna and D. Diamond, A wearable electrochemical sensor for the real-time measurement of sweat sodium concentration, Anal. Methods, 2010, 2, 342–348 RSC.
- P. Pirovano, M. Dorrian, A. Shinde, A. Donohoe, A. J. Brady, N. M. Moyna, G. Wallace, D. Diamond and M. McCaul, A wearable sensor for the detection of sodium and potassium in human sweat during exercise, Talanta, 2020, 219, 121145 CrossRef CAS.
- A. Hauke, S. Oertel, L. Knoke, V. Fein, C. Maier, F. Brinkmann and M. P. M. Jank, Screen-Printed Sensor for Low-Cost Chloride Analysis in Sweat for Rapid Diagnosis and Monitoring of Cystic Fibrosis, Biosensors, 2020, 10, 123 CrossRef CAS PubMed.
- D.-H. Choi, J. S. Kim, G. R. Cutting and P. C. Searson, Wearable Potentiometric Chloride Sweat Sensor: The Critical Role of the Salt Bridge, Anal. Chem., 2016, 88, 12241–12247 CrossRef CAS PubMed.
- X. Wei, M. Zhu, J. Li, L. Liu, J. Yu, Z. Li and B. Ding, Wearable biosensor for sensitive detection of uric acid in artificial sweat enabled by a fiber structured sensing interface, Nano Energy, 2021, 85, 106031 CrossRef CAS.
- B. Ramachandran and Y.-C. Liao, Microfluidic wearable electrochemical sweat sensors for health monitoring, Biomicrofluidics, 2022, 16, 051501 CrossRef CAS PubMed.
- N. Komuro, S. Takaki, K. Suzuki and D. Citterio, Inkjet printed (bio)chemical sensing devices, Anal. Bioanal. Chem., 2013, 405, 5785–5805 CrossRef CAS PubMed.
- S. Khan, L. Lorenzelli and R. S. Dahiya, Technologies for Printing Sensors and Electronics Over Large Flexible Substrates: A Review, IEEE Sens. J., 2015, 15, 3164–3185 Search PubMed.
- J. Wei, X. Zhang, Q. Chang, S. M. Mugo and Q. Zhang, An Integrated Sweat Sensor for Synchronous Detection of Multiple Atherosclerosis Biomarkers, Anal. Chem., 2023, 95, 15786–15794 CrossRef CAS.
- J. Wang, Electrochemical glucose biosensors, Chem. Rev., 2008, 108, 814–825 CrossRef CAS PubMed.
- Q.-F. Li, X. Chen, H. Wang, M. Liu and H.-L. Peng, Pt/MXene-Based Flexible Wearable Non-Enzymatic Electrochemical Sensor for Continuous Glucose Detection in Sweat, ACS Appl. Mater. Interfaces, 2023, 15, 13290–13298 CrossRef CAS.
- A. J. Bandodkar and J. Wang, Non-invasive wearable electrochemical sensors: a review, Trends Biotechnol., 2014, 32, 363–371 CrossRef CAS PubMed.
- E. J. M. Moonen, J. R. Haakma, E. Peri, E. Pelssers, M. Mischi and J. M. J. den Toonder, Wearable sweat sensing for prolonged, semicontinuous, and nonobtrusive health monitoring, VIEW, 2020, 1, 20200077 CrossRef.
- S. K. Nagarkoti, R. Yadav and P. Bhati, in 2025 International Conference on Emerging Trends in Networks and Computer Communications (ETNCC), 2025, pp. 898–905 Search PubMed.
- Z. Lihong and L. Qiuping, Advancements and Obstacles in Sweat-Based Biosensors for Health Monitoring, Crit. Rev. Anal. Chem., 2025, 1–32 CrossRef.
- A. Childs, B. Mayol, J. A. Lasalde-Ramírez, Y. Song, J. R. Sempionatto and W. Gao, Diving into Sweat: Advances, Challenges, and Future Directions in Wearable Sweat Sensing, ACS Nano, 2024, 18, 24605–24616 CrossRef CAS.
- K. M. Clark and T. R. Ray, Recent Advances in Skin-Interfaced Wearable Sweat Sensors: Opportunities for Equitable Personalized Medicine and Global Health Diagnostics, ACS Sens., 2023, 8(10), 3606–3622 CrossRef CAS PubMed.
- Y. Yang and W. Gao, Wearable and flexible electronics for continuous molecular monitoring, Chem. Soc. Rev., 2019, 48, 1465–1491 RSC.
- F. Gao, C. Liu, L. Zhang, T. Liu, Z. Wang, Z. Song, H. Cai, Z. Fang, J. Chen, J. Wang, M. Han, J. Wang, K. Lin, R. Wang, M. Li, Q. Mei, X. Ma, S. Liang, G. Gou and N. Xue, Wearable and flexible electrochemical sensors for sweat analysis: a review, Microsyst. Nanoeng., 2023, 9, 1–21 CrossRef.
- T. Terse-Thakoor, M. Punjiya, Z. Matharu, B. Lyu, M. Ahmad, G. E. Giles, R. Owyeung, F. Alaimo, M. Shojaei Baghini, T. T. Brunyé and S. Sonkusale, Thread-based multiplexed sensor patch for real-time sweat monitoring, npj Flexible Electron., 2020, 4, 18 CrossRef.
- G. Xu, X. Huang, R. Shi, Y. Yang, P. Wu, J. Zhou, X. He, J. Li, Y. Zen, Y. Jiao, B. Zhang, J. Li, G. Zhao, Y. Liu, Y. Huang, M. Wu, Q. Zhang, Z. Yang and X. Yu, Triboelectric Nanogenerator Enabled Sweat Extraction and Power Activation for Sweat Monitoring, Adv. Funct. Mater., 2024, 34, 2310777 CrossRef CAS.
- Y. Song, J. Min, Y. Yu, H. Wang, Y. Yang, H. Zhang and W. Gao, Wireless battery-free wearable sweat sensor powered by human motion, Sci. Adv., 2020, 6, eaay9842 CrossRef CAS.
- A. M. V. Mohan, V. Rajendran, R. K. Mishra and M. Jayaraman, Recent advances and perspectives in sweat based wearable electrochemical sensors, TrAC, Trends Anal. Chem., 2020, 131, 116024 CrossRef CAS.
- J. Choi, R. Ghaffari, L. B. Baker and J. A. Rogers, Skin-interfaced systems for sweat collection and analytics, Sci. Adv., 2018, 4, eaar3921 CrossRef PubMed.
- R. Vinoth, T. Nakagawa, J. Mathiyarasu and A. M. V. Mohan, Fully Printed Wearable Microfluidic Devices for High-Throughput Sweat Sampling and Multiplexed Electrochemical Analysis, ACS Sens., 2021, 6, 1174–1186 CrossRef CAS PubMed.
- J. Min, J. Tu, C. Xu, H. Lukas, S. Shin, Y. Yang, S. A. Solomon, D. Mukasa and W. Gao, Skin-Interfaced Wearable Sweat Sensors for Precision Medicine, Chem. Rev., 2023, 123, 5049–5138 CrossRef CAS PubMed.
- A. Alizadeh, A. Burns, R. Lenigk, R. Gettings, J. Ashe, A. Porter, M. McCaul, R. Barrett, D. Diamond, P. White, P. Skeath and M. Tomczak, A wearable patch for continuous monitoring of sweat electrolytes during exertion, Lab Chip, 2018, 18, 2632–2641 RSC.
- T. Zhang, G. K. M. Kabandana, J. A. Terrell, H. Chen and C. Chen, Recent Advances in Wearable Sweat Sensor Development, Wiley Interdiscip. Rev.:Nanomed. Nanobiotechnol., 2025, 17, e70006 Search PubMed.
- Y. Zhang, S. Qiu, K. Du, S. Wu, T. Xiang, K. Zheng, Z. Liu, H. Chen, N. Ji, F. Wang, W. Wu and Y.-T. Zhang, Artificial Intelligence-Enhanced Wearable Blood Pressure Monitoring in Resource-Limited Settings: A Co-Design of Sensors, Model, and Deployment, Nano-Micro Lett., 2026, 18, 164 CrossRef PubMed.
- M. Gong, Z. Wu, Y. He, J. Xiao, X. Men and H. Chen, Noninvasive Point-of-Care Testing: From Sensing Technologies and Wearable Platforms to Intelligent Diagnostics, J. Innovative Opt. Health Sci., 2026 DOI:10.1142/S1793545826300053.
- B. Xie, J. Lin, L. Cui, X. Shi and S. Wu, Microfluidic Biomaterials for Next-Generation Biomedical Platforms: Advances in Fabrication, Functionality, and Applications, J. Biomed. Mater. Res., Part A, 2026, 114, e70032 CrossRef CAS.
- W. Nie, J. Jiang, X. Wei, L. Zhuo, D. Li, M. Jia, T. Zheng and H. Cui, An All-Soft Wearable Electrochemiluminescence Chip for Sweat Metabolite Detection, Adv. Sci., 2026, 13(10), e19435 CrossRef CAS.
- W. Gao, H. Ota, D. Kiriya, K. Takei and A. Javey, Flexible Electronics toward Wearable Sensing, Acc. Chem. Res., 2019, 52, 523–533 CrossRef CAS.
- H. Zhao, X. Zhang, W. U. Khan, M. Karbarz and Q. Zhang, Research Progress in Electrochemical Sweat Sensors Driven by Advanced Materials and Structural Design for Health Monitoring, Adv. Mater. Technol., 2026, 11(7), e02468 CrossRef CAS.
- E. Gulgosteren, S. Agrali Ermis, A. Algin Toros, T. Toros, E. Serin, M. O. Sekeroglu and N. Bahadır Kayisoglu, Sweat, tears, and beyond: advanced wearable sensors for personalized health and athletic performance, Front. Bioeng. Biotechnol., 2025, 13, 1684674 CrossRef PubMed.
- Ö. Erdem, E. Derin, S. Zeibi Shirejini, K. Sagdic, E. G. Yilmaz, S. Yildiz, G. A. Akceoglu and F. Inci, Carbon-Based Nanomaterials and Sensing Tools for Wearable Health Monitoring Devices, Adv. Mater. Technol., 2022, 7, 2100572 CrossRef.
- P. Calvert, Inkjet Printing for Materials and Devices, Chem. Mater., 2001, 13(10) DOI:10.1021/cm0101632.
- J. P. Hart, A. Crew, E. Crouch, K. C. Honeychurch and R. Pemberton, Chapter 23 Screen-printed electrochemical (bio)sensors in biomedical, environmental and industrial applications, Compr. Anal. Chem., 2007, 49, 497–557 CAS.
- H. Yu and J. Sun, Sweat detection theory and fluid driven methods: A review, Nanotechnol. Precis. Eng., 2020, 3, 126–140 CrossRef CAS.
- A. J. Bandodkar, W. J. Jeang, R. Ghaffari and J. A. Rogers, Wearable Sensors for Biochemical Sweat Analysis, Annu. Rev. Anal. Chem., 2019, 12, 1–22 CrossRef PubMed.
- Z. Sonner, E. Wilder, J. Heikenfeld, G. Kasting, F. Beyette, D. Swaile, F. Sherman, J. Joyce, J. Hagen, N. Kelley-Loughnane and R. Naik, The microfluidics of the eccrine sweat gland, including biomarker partitioning, transport, and biosensing implications, Biomicrofluidics, 2015, 9, 031301 CrossRef CAS.
- A. Mena-Bravo and M. D. L. de Castro, Sweat: a sample with limited present applications and promising future in metabolomics, J. Pharm. Biomed. Anal., 2013 DOI:10.1016/j.jpba.2013.10.048.
- C. Callewaert, B. Buysschaert, E. Vossen, V. Fievez, T. Van de Wiele and N. Boon, Artificial sweat composition to grow and sustain a mixed human axillary microbiome, J. Microbiol. Methods, 2014, 103, 6–8 CrossRef CAS PubMed.
- N. A. Taylor and C. A. Machado-Moreira, Regional variations in transepidermal water loss, eccrine sweat gland density, sweat secretion rates and electrolyte composition in resting and exercising humans, Extrem. Physiol. Med., 2013, 2, 4 CrossRef PubMed.
- É. Csősz, G. Emri, G. Kalló, G. Tsaprailis and J. Tőzsér, Highly abundant defense proteins in human sweat as revealed by targeted proteomics and label-free quantification mass spectrometry, J. Eur. Acad. Dermatol. Venereol., 2015, 29, 2024–2031 CrossRef.
- M. M. Delgado-Povedano, M. Calderón-Santiago, F. Priego-Capote and M. D. Luque de Castro, Development of a method for enhancing metabolomics coverage of human sweat by gas chromatography-mass spectrometry in high resolution mode, Anal. Chim. Acta, 2016, 905, 115–125 CrossRef CAS.
- Y. Chen, B. Ma, Y. Zuo, G. Chen, Q. Hao, C. Zhao and H. Liu, Versatile sweat bioanalysis on demand with hydrogel-programmed wearables, Biosens. Bioelectron., 2023, 235, 115412 CrossRef CAS.
- J.-L. Lafuente, S. González, C. Aibar, D. Rivera, E. Avilés and J.-J. Beunza, Continuous and Non-Invasive Lactate Monitoring Techniques in Critical Care Patients, Biosensors, 2024, 14, 148 CrossRef CAS PubMed.
- J. B. Eckenhoff and F. Theeuwes, US Pat., US4756314A, 1988 Search PubMed.
- R. Peng, Z. Sonner, A. Hauke, E. Wilder, J. Kasting, T. Gaillard, D. Swaille, F. Sherman, X. Mao, J. Hagen, R. Murdock and J. Heikenfeld, A new oil/membrane approach for integrated sweat sampling and sensing: sample volumes reduced from μL’s to nL’s and reduction of analyte contamination from skin, Lab chip, 2016, 16, 4415–4423 RSC.
- W. Gao, H. Y. Y. Nyein, Z. Shahpar, L.-C. Tai, E. Wu, M. Bariya, H. Ota, H. M. Fahad, K. Chen and A. Javey, in 2016 IEEE International Electron Devices Meeting (IEDM), 2016, pp. 6.6.1–6.6.4 Search PubMed.
- Y. Zhang, J. Li, S. Jiao, Y. Li, Y. Zhou, X. Zhang, B. Maryam and X. Liu, Microfluidic sensors for the detection of emerging contaminants in water: A review, Sci. Total Environ., 2024, 929, 172734 CrossRef CAS PubMed.
- S. Emaminejad, W. Gao, E. Wu, Z. A. Davies, H. Yin Yin Nyein, S. Challa, S. P. Ryan, H. M. Fahad, K. Chen, Z. Shahpar, S. Talebi, C. Milla, A. Javey and R. W. Davis, Autonomous sweat extraction and analysis applied to cystic fibrosis and glucose monitoring using a fully integrated wearable platform, Proc. Natl. Acad. Sci. U. S. A., 2017, 114, 4625–4630 CrossRef CAS.
- R. D. Munje, S. Muthukumar, A. Panneer Selvam and S. Prasad, Flexible nanoporous tunable electrical double layer biosensors for sweat diagnostics, Sci. Rep., 2015, 5, 14586 CrossRef CAS PubMed.
- J. S. Stefano, L. O. Orzari, H. A. Silva-Neto, V. N. de Ataíde, L. F. Mendes, W. K. T. Coltro, T. R. Longo Cesar Paixão and B. C. Janegitz, Different approaches for fabrication of low-cost electrochemical sensors, Curr. Opin. Electrochem., 2022, 32, 100893 CrossRef CAS.
- D. Zhang, Y. Bai, H. Niu, L. Chen, J. Xiao, Q. Guo and P. Jia, Enzyme Immobilization by Inkjet Printing on Reagentless Biosensors for Electrochemical Phosphate Detection, Biosensors, 2024, 14, 168 CrossRef CAS PubMed.
- C. Cooper and B. Hughes, in 2020 Pan Pacific Microelectronics Symposium (Pan Pacific), IEEE, HI, USA, 2020, pp. 1–11 Search PubMed.
- M. Chung, G. Fortunato and N. Radacsi, Wearable flexible sweat sensors for healthcare monitoring: a review, J. R. Soc., Interface, 2019, 16, 20190217 CrossRef CAS PubMed.
- E. B. Secor, Principles of aerosol jet printing, Flexible Printed Electron., 2018, 3, 035002 CrossRef.
- E. J. Carrasco-Correa, E. F. Simó-Alfonso, J. M. Herrero-Martínez and M. Miró, The emerging role of 3D printing in the fabrication of detection systems, TrAC, Trends Anal. Chem., 2021, 136, 116177 CrossRef CAS.
- H.-W. Lin, C.-P. Chang, W.-H. Hwu and M.-D. Ger, The rheological behaviors of screen-printing pastes, J. Mater. Process. Technol., 2008, 197, 284–291 CrossRef CAS.
- R. Szentgyörgyvölgyi, in Printing on Polymers, ed. J. Izdebska and S. Thomas, William Andrew Publishing, 2016, pp. 199–215 Search PubMed.
- J. Izdebska, in Printing on Polymers, ed. J. Izdebska and S. Thomas, William Andrew Publishing, 2016, pp. 179–197 Search PubMed.
- A. A. Paul, A. D. Aladese and R. S. Marks, Additive Manufacturing Applications in Biosensors Technologies, Biosensors, 2024, 14, 60 CrossRef CAS PubMed.
- A. Kalkal, S. Kumar, P. Kumar, R. Pradhan, M. Willander, G. Packirisamy, S. Kumar and B. D. Malhotra, Recent advances in 3D printing technologies for wearable (bio)sensors, Addit. Manuf., 2021, 46, 102088 CAS.
- N. Kumar, Y.-J. Lin, Y.-C. Huang, Y.-T. Liao and S.-P. Lin, Detection of lactate in human sweat via surface-modified, screen-printed carbon electrodes, Talanta, 2023, 265, 124888 CrossRef CAS.
- D. R. Thévenot, K. Toth, R. A. Durst and G. S. Wilson, Electrochemical biosensors: recommended definitions and classification, Biosens. Bioelectron., 2001, 16, 121–131 CrossRef PubMed.
- A. J. Bard and L. R. Faulkner, Electrochemical Methods: Fundamentals and Applications, Wiley, New York, 2001 Search PubMed.
- N. J. Ronkainen, H. B. Halsall and W. R. Heineman, Electrochemical biosensors, Chem. Soc. Rev., 2010, 39, 1747–1763 RSC.
- I.-H. Cho, D. H. Kim and S. Park, Electrochemical biosensors: perspective on functional nanomaterials for on-site analysis, Biomater. Res., 2020, 24, 6 CrossRef CAS PubMed.
- B. D. Malhotra and A. Chaubey, Biosensors for clinical diagnostics industry, Sens. Actuators, B, 2003, 91, 117–127 CrossRef CAS.
- H. H. Ipekci, Z. Gozutok, N. Celik, M. Serdar Onses and A. Uzunoglu, Ink-jet printing of particle-free silver inks on fabrics with a superhydrophobic protection layer for fabrication of robust electrochemical sensors, Microchem. J., 2021, 164, 106038 CrossRef CAS.
- S. Sarıkaya, H. H. Ipekci, H. Kotan and A. Uzunoglu, Inkjet printing of highly dispersed, shortened, and defect-rich MWCNTs to construct flexible electrochemical sensors for the detection of bisphenol A in milk samples, Carbon, 2023, 214, 118362 CrossRef.
- M. Dilsen, H. H. Ipekci and A. Uzunoglu, Inkjet printing of Pd/SO3H-modified graphene on different polymeric substrates to construct flexible electrochemical sensors, J. Mater. Res., 2023, 38, 3572–3584 CrossRef CAS.
- Y. Qin, H.-J. Kwon, M. M. R. Howlader and M. J. Deen, Microfabricated electrochemical pH and free chlorine sensors for water quality monitoring: recent advances and research challenges, RSC Adv., 2015, 5, 69086–69109 RSC.
- G. Paimard, E. Ghasali and M. Baeza, Screen-Printed Electrodes: Fabrication, Modification, and Biosensing Applications, Chem, 2023, 11, 113 CAS.
- A. Crew, D. Lonsdale, N. Byrd, R. Pittson and J. P. Hart, A screen-printed, amperometric biosensor array incorporated into a novel automated system for the simultaneous determination of organophosphate pesticides, Biosens. Bioelectron., 2011, 26, 2847–2851 CrossRef CAS.
- O. D. Renedo, M. A. Alonso-Lomillo and M. J. A. Martínez, Recent developments in the field of screen-printed electrodes and their related applications, Talanta, 2007, 73, 202–219 CrossRef CAS.
- R. M. Cardoso, C. Kalinke, R. G. Rocha, P. L. dos Santos, D. P. Rocha, P. R. Oliveira, B. C. Janegitz, J. A. Bonacin, E. M. Richter and R. A. A. Munoz, Additive-manufactured (3D-printed) electrochemical sensors: A critical review, Anal. Chim. Acta, 2020, 1118, 73–91 CrossRef CAS.
- A. Ambrosi and M. Pumera, 3D-printing technologies for electrochemical applications, Chem. Soc. Rev., 2016, 45, 2740–2755 RSC.
- L. De Waele, M. Di Pietro, S. Perilli, E. Mantini, G. Trevisan, M. Simoncini, M. Panella, V. Betti, M. Laffranchi and D. Mantini, Aerosol Jet Printing for Neuroprosthetic Device Development, Bioengineering, 2025, 12, 707 CrossRef CAS PubMed.
- S. Waheed, J. M. Cabot, N. P. Macdonald, T. Lewis, R. M. Guijt, B. Paull and M. C. Breadmore, 3D printed microfluidic devices: enablers and barriers, Lab Chip, 2016, 16, 1993–2013 RSC.
- T. S. Tran, R. Balu, S. Mettu, N. Roy Choudhury and N. K. Dutta, 4D Printing of Hydrogels: Innovation in Material Design and Emerging Smart Systems for Drug Delivery, Pharmaceuticals, 2022, 15, 1282 CrossRef CAS PubMed.
- T. Kim, Q. Yi, E. Hoang and R. Esfandyarpour, A 3D Printed Wearable Bioelectronic Patch for Multi-Sensing and In Situ Sweat Electrolyte Monitoring, Adv. Mater. Technol., 2021, 6, 2001021 CrossRef CAS.
- C.-H. Wu, H. J. H. Ma, P. Baessler, R. K. Balanay and T. R. Ray, Skin-interfaced microfluidic systems with spatially engineered 3D fluidics for sweat capture and analysis, Sci. Adv., 2023, 9, eadg4272 CrossRef CAS PubMed.
- P. Serra and A. Piqué, Laser-Induced Forward Transfer: Fundamentals and Applications, Adv. Mater. Technol., 2019, 4, 1800099 CrossRef.
- Z. Ur Rehman, F. Yang, M. Wang and T. Zhu, Fundamentals and Advances in Laser-Induced Transfer, Opt. Laser Technol., 2023, 160, 109065 CrossRef.
- M. Bahri, M. Amin Elaguech, S. Nasraoui, K. Djebbi, O. Kanoun, P. Qin, C. Tlili and D. Wang, Laser-Induced graphene electrodes for highly sensitive detection of DNA hybridization via consecutive cytosines (polyC)-DNA-based electrochemical biosensors, Microchem. J., 2023, 185, 108208 CrossRef CAS.
- S. K. Dhinesh and K. L. Senthil Kumar, A Review on 3D Printed Sensors, IOP Conf. Ser.: Mater. Sci. Eng., 2020, 764, 012055 Search PubMed.
- H. Y. Y. Nyein, M. Bariya, L. Kivimäki, S. Uusitalo, T. S. Liaw, E. Jansson, C. H. Ahn, J. A. Hangasky, J. Zhao, Y. Lin, T. Happonen, M. Chao, C. Liedert, Y. Zhao, L.-C. Tai, J. Hiltunen and A. Javey, Regional and correlative sweat analysis using high-throughput microfluidic sensing patches toward decoding sweat, Sci. Adv., 2019, 5, eaaw9906 CrossRef CAS PubMed.
- J.-U. Park, M. Hardy, S. J. Kang, K. Barton, K. Adair, D. kishore Mukhopadhyay, C. Y. Lee, M. S. Strano, A. G. Alleyne, J. G. Georgiadis, P. M. Ferreira and J. A. Rogers, High-resolution electrohydrodynamic jet printing, Nat. Mater., 2007, 6, 782–789 CrossRef CAS.
- D. P. Rocha, R. B. A. Albuquerque, G. P. Oliveira, R. M. Cardoso, F. S. Semaan, R. M. Dornellas, E. M. Richter and R. A. Abarza Muñoz, in Encyclopedia of Sensors and Biosensors, Elsevier, 2023, vol. 2, pp. 73–88 Search PubMed.
- A. Erdem, E. Yildiz, H. Senturk and M. Maral, Implementation of 3D printing technologies to electrochemical and optical biosensors developed for biomedical and pharmaceutical analysis, J. Pharm. Biomed. Anal., 2023, 230, 115385 CrossRef CAS PubMed.
- J. S. Stefano, C. Kalinke, R. G. da Rocha, D. P. Rocha, V. A. O. P. da Silva, J. A. Bonacin, L. Angnes, E. M. Richter, B. C. Janegitz and R. A. A. Muñoz, Electrochemical (Bio)Sensors Enabled by Fused Deposition Modeling-Based 3D Printing: A Guide to Selecting Designs, Printing Parameters, and Post-Treatment Protocols, Anal. Chem., 2022, 94, 6417–6429 CrossRef CAS PubMed.
- T. T. T. Can, T. C. Nguyen and W.-S. Choi, Patterning of High-Viscosity Silver Paste by an Electrohydrodynamic-Jet Printer for Use in TFT Applications, Sci. Rep., 2019, 9, 9180 CrossRef PubMed.
- C. Wei, H. Qin, N. A. Ramírez-Iglesias, C.-P. Chiu, Y. Lee and J. Dong, High-resolution ac-pulse modulated electrohydrodynamic jet printing on highly insulating substrates, J. Micromech. Microeng., 2014, 24, 045010 CrossRef CAS.
- Y. Han and J. Dong, Electrohydrodynamic (EHD) Printing of Molten Metal Ink for Flexible and Stretchable Conductor with Self-Healing Capability, Adv. Mater. Technol., 2018, 3, 1700268 CrossRef.
- Y. Han and J. Dong, High-resolution direct printing of molten-metal using electrohydrodynamic jet plotting, Manuf. Lett., 2017, 12, 6–9 Search PubMed.
- G. Xu, C. Cheng, Z. Liu, W. Yuan, X. Wu, Y. Lu, S. S. Low, J. Liu, L. Zhu, D. Ji, S. Li, Z. Chen, L. Wang, Q. Yang, Z. Cui and Q. Liu, Battery-Free and Wireless Epidermal Electrochemical System with All-Printed Stretchable Electrode Array for Multiplexed In Situ Sweat Analysis, Adv. Mater. Technol., 2019, 4, 1800658 CrossRef CAS.
- V. Marinov, O. Swenson, R. Miller, F. Sarwar, Y. Atanasov, M. Semler and S. Datta, Laser-Enabled Advanced Packaging of Ultrathin Bare Dice in Flexible Substrates, IEEE Trans. Compon., Packag., Manuf. Technol., 2012, 2, 569–577 Search PubMed.
- J. R. Sempionatto, J. A. Lasalde-Ramírez, K. Mahato, J. Wang and W. Gao, Wearable chemical sensors for biomarker discovery in the omics era, Nat. Rev. Chem., 2022, 6, 899–915 CrossRef PubMed.
- A. J. Bandodkar, D. Molinnus, O. Mirza, T. Guinovart, J. R. Windmiller, G. Valdés-Ramírez, F. J. Andrade, M. J. Schöning and J. Wang, Epidermal tattoo potentiometric sodium sensors with wireless signal transduction for continuous non-invasive sweat monitoring, Biosens. Bioelectron., 2014, 54, 603–609 CrossRef CAS.
- A. Nag, S. C. Mukhopadhyay and J. Kosel, Wearable Flexible Sensors: A Review, IEEE Sens. J., 2017, 17, 3949–3960 CAS.
- F. F. Franco, L. Manjakkal and R. Dahiya, in 2020 IEEE International Conference on Flexible and Printable Sensors and Systems (FLEPS), 2020, pp. 1–4 Search PubMed.
- V. A. T. Dam, M. A. G. Zevenbergen and R. van Schaijk, Toward wearable patch for sweat analysis, Sens. Actuators, B, 2016, 236, 834–838 CrossRef CAS.
- A. Zoerner, S. Oertel, M. P. M. Jank, L. Frey, B. Langenstein and T. Bertsch, Human Sweat Analysis Using a Portable Device Based on a Screen-printed Electrolyte Sensor, Electroanalysis, 2018, 30, 665–671 CrossRef CAS.
- Z. Taleat, A. Khoshroo and M. Mazloum-Ardakani, Screen-printed electrodes for biosensing: a review (2008–2013), Microchim. Acta, 2014, 181, 865–891 CrossRef CAS.
- V. Mazzaracchio, A. Serani, L. Fiore, D. Moscone and F. Arduini, All-solid state ion-selective carbon black-modified printed electrode for sodium detection in sweat, Electrochim. Acta, 2021, 394, 139050 Search PubMed.
- P. Sundriyal and S. Bhattacharya, Inkjet-Printed Sensors on Flexible Substrates, in Energy, Environment, and Sustainability, ed. S. Bhattacharya, A. K. Agarwal, N. Chanda, A. Pandey and A. K. Sen, Springer, Singapore, 2018, pp. 89–113 Search PubMed.
- P. Calvert, Inkjet Printing for Materials and Devices, Chem. Mater., 2001, 13, 3299–3305 Search PubMed.
- G. Cummins and M. P. Y. Desmulliez, Inkjet printing of conductive materials: a review, Circuit World, 2012, 38, 193–213 Search PubMed.
- M. Singh, H. M. Haverinen, P. Dhagat and G. E. Jabbour, Inkjet Printing—Process and Its Applications, Adv. Mater., 2010, 22, 673–685 CrossRef CAS PubMed.
- T. H. da Costa, E. Song, R. P. Tortorich and J.-W. Choi, A Paper-Based Electrochemical Sensor Using Inkjet-Printed Carbon Nanotube Electrodes, ECS J. Solid State Sci. Technol., 2015, 4, S3044 CrossRef CAS.
- K. Nagamine, A. Nomura, Y. Ichimura, R. Izawa, S. Sasaki, H. Furusawa, H. Matsui and S. Tokito, Printed Organic Transistor-based Biosensors for Non-invasive Sweat Analysis, Anal. Sci., 2020, 36, 291–302 CrossRef CAS PubMed.
- A. M. Khalaf, H. H. Issa, J. L. RamíRez and S. A. Mohamed, All Inkjet-Printed Temperature Sensors Based on PEDOT:PSS, IEEE Access, 2022, 10, 61094–61100 Search PubMed.
- C. Bali, A. Brandlmaier, A. Ganster, O. Raab, J. Zapf and A. Hübler, Fully Inkjet-Printed Flexible Temperature Sensors Based on Carbon and PEDOT:PSS1, Mater. Today: Proc., 2016, 3, 739–745 Search PubMed.
- A. M. Zamarayeva, N. A. D. Yamamoto, A. Toor, M. E. Payne, C. Woods, V. I. Pister, Y. Khan, J. W. Evans and A. C. Arias, Optimization of printed sensors to monitor sodium, ammonium, and lactate in sweat, APL Mater., 2020, 8(10), 100905 CrossRef CAS.
- N. Ruecha, O. Chailapakul, K. Suzuki and D. Citterio, Fully Inkjet-Printed Paper-Based Potentiometric Ion-Sensing Devices, Anal. Chem., 2017, 89, 10608–10616 CrossRef CAS PubMed.
- K.-L. Tsou, K.-Y. Chen, Y.-D. Chou, Y.-T. Cheng, H.-E. Tsai and C.-K. Lee, Inkjet-printed flexible non-enzymatic lactate sensor with high sensitivity and low interference using a stacked NiOx/NiOx-Nafion nanocomposite electrode with clinical blood test verification, Talanta, 2022, 249, 123598 CrossRef CAS PubMed.
- E. Bihar, S. Wustoni, A. M. Pappa, K. N. Salama, D. Baran and S. Inal, A fully inkjet-printed disposable glucose sensor on paper, npj Flexible Electron., 2018, 2, 30 CrossRef.
- F. Xie, J. Wang, S. Ganguly, S. Singh, W. Wang, J. Baum and R. R. Jha, in 2024 IEEE Energy Conversion Congress and Exposition (ECCE), 2024, pp. 804–811 Search PubMed.
- A. Ambrosi and M. Pumera, 3D-printing technologies for electrochemical applications, Chem. Soc. Rev., 2016, 45, 2740–2755 RSC.
- A. Tuantranont, A. Wisitsoraat, C. Sriprachuabwong, C. Karuwan, P. Pasakon and D. Phokharatkul, in Graphene Science Handbook: Mechanical and Chemical Properties, 2016, pp. 147–161 Search PubMed.
- Y. Chang, Q. Cao and B. J. Venton, 3D printing for customized carbon electrodes, Curr. Opin. Electrochem., 2023, 38, 101228 CrossRef CAS PubMed.
- M. R. Khosravani and T. Reinicke, 3D-printed sensors: Current progress and future challenges, Sens. Actuators, A, 2020, 305, 111916 CrossRef CAS.
- Y. Xu, X. Wu, X. Guo, B. Kong, M. Zhang, X. Qian, S. Mi and W. Sun, The Boom in 3D-Printed Sensor Technology, Sensors, 2017, 17, 1166 CrossRef PubMed.
- E. M. Richter, D. P. Rocha, R. M. Cardoso, E. M. Keefe, C. W. Foster, R. A. A. Munoz and C. E. Banks, Complete Additively Manufactured (3D-Printed) Electrochemical Sensing Platform, Anal. Chem., 2019, 91, 12844–12851 CrossRef CAS PubMed.
- T. Han, A. Nag, N. Afsarimanesh, S. C. Mukhopadhyay, S. Kundu and Y. Xu, Laser-Assisted Printed Flexible Sensors: A Review, Sensors, 2019, 19, 1462 CrossRef PubMed.
- J. Liao, X. Zhang, Z. Sun, H. Chen, J. Fu, H. Si, C. Ge and S. Lin, Laser-Induced Graphene-Based Wearable Epidermal Ion-Selective Sensors for Noninvasive Multiplexed Sweat Analysis, Biosensors, 2022, 12, 397 CrossRef CAS PubMed.
- C.-W. Lee, S.-Y. Jeong, Y.-W. Kwon, J.-U. Lee, S.-C. Cho and B.-S. Shin, Fabrication of laser-induced graphene-based multifunctional sensing platform for sweat ion and human motion monitoring, Sens. Actuators, A, 2022, 334, 113320 CrossRef CAS.
- M. Riza and X. Du, Roll-To-Roll Printing of Wearable Sensors and Devices for Personalized Health Monitoring, Am. J. Biomed. Sci. Res., 2020, 7, 495 CrossRef.
- M. Bariya, Z. Shahpar, H. Park, J. Sun, Y. Jung, W. Gao, H. Y. Y. Nyein, T. S. Liaw, L.-C. Tai, Q. P. Ngo, M. Chao, Y. Zhao, M. Hettick, G. Cho and A. Javey, Roll-to-Roll Gravure Printed Electrochemical Sensors for Wearable and Medical Devices, ACS Nano, 2018, 12, 6978–6987 Search PubMed.
- S. Khan, S. Ali and A. Bermak, Recent Developments in Printing Flexible and Wearable Sensing Electronics for Healthcare Applications, Sensors, 2019, 19, 1230 CrossRef CAS PubMed.
- M. N. Tousignant, V. Tischler, K. Wagner, Z. S. Lin, J. Brusso, R. Izquierdo and B. H. Lessard, Aerosol jet printed temperature sensors using an environmentally friendly bilayer dielectric, Flexible Printed Electron., 2024, 9, 015012 CrossRef CAS.
- A. Agrawal and C. M. Hussain, in Flexible and Wearable Sensors, CRC Press, 2023 Search PubMed.
- J. Dominiczak, J. Krzemiński, J. Wojcieszek, D. Baraniecki, F. Budny, I. Wojciechowska, P. Walter, A. Pepłowski, Ł. Górski and M. Jakubowska, Aerosol-jet-printed potentiometric pH sensor for sweat measurements in smart patches, Sens. Bio-Sens. Res., 2024, 43, 100636 CrossRef.
- C. Fisher, L. N. Skolrood, K. Li, P. C. Joshi and T. Aytug, Aerosol-Jet Printed Sensors for Environmental, Safety, and Health Monitoring: A Review, Adv. Mater. Technol., 2023, 8, 2300030 Search PubMed.
- H. Zhou and Y. Song, Fabrication of Electronics by Electrohydrodynamic Jet Printing, Adv. Electron. Mater., 2022, 8, 2200728 CrossRef CAS.
- T. Chang, Z. Wang, X. Tian, D. Ma, S. Hussain, L. Xia and J. Han, Electrohydrodynamic Jet Printing Mechanisms and Applications to Flexible Electronic Devices, Proceedings of the ASME 2024 19th International Manufacturing Science and Engineering Conference, Volume 1: Additive Manufacturing; Advanced Materials Manufacturing; Biomanufacturing; Life Cycle Engineering, ASME, Knoxville, Tennessee, USA, 2024, V001T03A014, DOI:10.1115/MSEC2024-130799.
- J. Lu, T. Wang, X. Li, F. Wu, H. Yang and W. Hu, Flexible Sensors Based on Electrohydrodynamic Jet Printing, Prog. Chem., 2022, 34, 1982–1995 CAS.
- M. S. Onses, E. Sutanto, P. M. Ferreira, A. G. Alleyne and J. A. Rogers, Mechanisms, Capabilities, and Applications of High-Resolution Electrohydrodynamic Jet Printing, Small, 2015, 11, 4237–4266 CrossRef CAS PubMed.
- J. R. Windmiller and J. Wang, Wearable Electrochemical Sensors and Biosensors: A Review, Electroanalysis, 2013, 25, 29–46 CrossRef CAS.
- Y. Khan, A. E. Ostfeld, C. M. Lochner, A. Pierre and A. C. Arias, Monitoring of Vital Signs with Flexible and Wearable Medical Devices, Adv. Mater., 2016, 28, 4373–4395 CrossRef CAS PubMed.
- S. Xu, Y. Zhang, L. Jia, K. E. Mathewson, K.-I. Jang, J. Kim, H. Fu, X. Huang, P. Chava, R. Wang, S. Bhole, L. Wang, Y. J. Na, Y. Guan, M. Flavin, Z. Han, Y. Huang and J. A. Rogers, Soft microfluidic assemblies of sensors, circuits, and radios for the skin, Science, 2014, 344, 70–74 CrossRef CAS PubMed.
- W.-H. Yeo, Y.-S. Kim, J. Lee, A. Ameen, L. Shi, M. Li, S. Wang, R. Ma, S. H. Jin, Z. Kang, Y. Huang and J. A. Rogers, Multifunctional epidermal electronics printed directly onto the skin, Adv. Mater., 2013, 25, 2773–2778 CrossRef CAS PubMed.
- W. Dungchai, O. Chailapakul and C. S. Henry, Electrochemical detection for paper-based microfluidics, Anal. Chem., 2009, 81, 5821–5826 CrossRef CAS PubMed.
- W. Gong, C. Hou, J. Zhou, Y. Guo, W. Zhang, Y. Li, Q. Zhang and H. Wang, Continuous and scalable manufacture of amphibious energy yarns and textiles, Nat. Commun., 2019, 10, 868 CrossRef PubMed.
- S. Lim, M. Joyce, P. D. Fleming, A. T. Aijazi, M. Atashbar, M. Joyce, P. D. Fleming, A. T. Aijazi and M. Atashbar, Inkjet Printing and Sintering of Nano-Copper Ink, J. Imaging Sci. Technol., 2013, 57, 1–7 Search PubMed.
- Y. Liu, M. Pharr and G. A. Salvatore, Lab-on-Skin: A Review of Flexible and Stretchable Electronics for Wearable Health Monitoring, ACS Nano, 2017, 11(10) DOI:10.1021/acsnano.7b04898.
- G. Li, X. Mo, W. C. Law and K. C. Chan, Wearable Fluid Capture Devices for Electrochemical Sensing of Sweat, ACS Appl. Mater. Interfaces, 2019, 11, 238–243 CrossRef CAS PubMed.
- M. Yang, N. Sun, X. Lai, Y. Li, X. Zhao, J. Wu and W. Zhou, Screen-Printed Wearable Sweat Sensor for Cost-Effective Assessment of Human Hydration Status through Potassium and Sodium Ion Detection, Micromachines, 2023, 14, 1497 Search PubMed.
- Z. Damirchi, A. Firoozbakhtian, M. Hosseini and M. R. Ganjali, Ti3C2/Ni/Sm-based electrochemical glucose sensor for sweat analysis using bipolar electrochemistry, Microchim. Acta, 2024, 191, 137 Search PubMed.
- J. Gilnezhad, A. Firoozbakhtian, M. Hosseini, S. Adel, G. Xu and M. R. Ganjali, An enzyme-free Ti3C2/Ni/Sm-LDH-based screen-printed-electrode for real-time sweat detection of glucose, Anal. Chim. Acta, 2023, 1250, 340981 CrossRef CAS PubMed.
- I. Shitanda, N. Muramatsu, R. Kimura, N. Takahashi, K. Watanabe, H. Matsui, N. Loew, M. Motosuke, T. Mukaimoto, M. Kobayashi, T. Mitsuhara, Y. Sugita, K. Matsuo, S. Yanagita, T. Suzuki, H. Watanabe and M. Itagaki, Wearable Ion Sensors for the Detection of Sweat Ions Fabricated by Heat-Transfer Printing, ACS Sens., 2023, 8, 2889–2895 CrossRef CAS PubMed.
- A. M. Zamarayeva, N. A. D. Yamamoto, A. Toor, M. E. Payne, C. Woods, V. I. Pister, Y. Khan, J. W. Evans and A. C. Arias, Optimization of printed sensors to monitor sodium, ammonium, and lactate in sweat, APL Mater., 2020, 8, 100905 CrossRef CAS.
- M. Li, L. Wang, R. Liu, J. Li, Q. Zhang, G. Shi, Y. Li, C. Hou and H. Wang, A highly integrated sensing paper for wearable electrochemical sweat analysis, Biosens. Bioelectron., 2021, 174, 112828 CrossRef CAS PubMed.
- L. Yin, M. Cao, K. N. Kim, M. Lin, J.-M. Moon, J. R. Sempionatto, J. Yu, R. Liu, C. Wicker, A. Trifonov, F. Zhang, H. Hu, J. R. Moreto, J. Go, S. Xu and J. Wang, A stretchable epidermal sweat sensing platform with an integrated printed battery and electrochromic display, Nat. Electron., 2022, 5, 694–705 CrossRef.
- S. Chen, Z. Cao, K. Zhou, S. Li, H. Li, K. Xu, H. Tang, H. Deng, Q. Zhou, J. Pan and F. Xia, Screen printing and laser-induced flexible sensors for the simultaneous sensitive detection of uric acid, tyrosine, and ascorbic acid in sweat, Analyst, 2023, 148, 2965–2974 RSC.
- A. J. Bandodkar, W. Jia, C. Yardımcı, X. Wang, J. Ramirez and J. Wang, Tattoo-Based Noninvasive Glucose Monitoring: A Proof-of-Concept Study, Anal. Chem., 2015, 87, 394–398 CrossRef CAS PubMed.
- R. R. Silva, P. A. Raymundo-Pereira, A. M. Campos, D. Wilson, C. G. Otoni, H. S. Barud, C. A. R. Costa, R. R. Domeneguetti, D. T. Balogh, S. J. L. Ribeiro and O. N. Oliveira, Microbial nanocellulose adherent to human skin used in electrochemical sensors to detect metal ions and biomarkers in sweat, Talanta, 2020, 218, 121153 CrossRef CAS PubMed.
- D. Kinnamon, R. Ghanta, K.-C. Lin, S. Muthukumar and S. Prasad, Portable biosensor for monitoring cortisol in low-volume perspired human sweat, Sci. Rep., 2017, 7, 13312 CrossRef PubMed.
- D. Sankhala, S. Muthukumar and S. Prasad, A Four-Channel Electrical Impedance Spectroscopy Module for Cortisol Biosensing in Sweat-Based Wearable Applications, SLAS Technol., 2018, 23, 529–539 CrossRef PubMed.
- X. Weng, Z. Fu, C. Zhang, W. Jiang and H. Jiang, A Portable 3D Microfluidic Origami Biosensor for Cortisol Detection in Human Sweat, Anal. Chem., 2022, 94, 3526–3534 CrossRef CAS PubMed.
- Q. Yang, G. Rosati, V. Abarintos, M. A. Aroca, J. F. Osma and A. Merkoçi, Wearable and fully printed microfluidic nanosensor for sweat rate, conductivity, and copper detection with healthcare applications, Biosens. Bioelectron., 2022, 202, 114005 CrossRef CAS PubMed.
- I. Shitanda, M. Mitsumoto, N. Loew, Y. Yoshihara, H. Watanabe, T. Mikawa, S. Tsujimura, M. Itagaki and M. Motosuke, Continuous sweat lactate monitoring system with integrated screen-printed MgO-templated carbon-lactate oxidase biosensor and microfluidic sweat collector, Electrochim. Acta, 2021, 368, 137620 CrossRef CAS.
- G. Ibáñez-Redín, G. Rosso Cagnani, N. O. Gomes, P. A. Raymundo-Pereira, S. A. S. Machado, M. A. Gutierrez, J. E. Krieger and O. N. Oliveira, Wearable potentiometric biosensor for analysis of urea in sweat, Biosens. Bioelectron., 2023, 223, 114994 CrossRef PubMed.
- T. Laochai, C. Moonla, J. Moon, K. Sakdaphetsiri, L. Yin, L. F. Mendes, A. Abbas, O. Djassemi, S. Seker, K. Mahato, O. Chailapakul, J. Wang and N. Rodthongkum, Touch–based potentiometric sensors for simultaneous detection of urea and ammonium from fingertip sweat, Sens. Actuators, B, 2024, 413, 135898 CrossRef CAS.
- J. Ok, S. Park, Y. H. Jung and T. Kim, Wearable and Implantable Cortisol-Sensing Electronics for Stress Monitoring, Adv. Mater., 2024, 36, 2211595 CrossRef CAS PubMed.
- S. T. Keene, D. Fogarty, R. Cooke, C. D. Casadevall, A. Salleo and O. Parlak, Wearable Organic Electrochemical Transistor Patch for Multiplexed Sensing of Calcium and Ammonium Ions from Human Perspiration, Adv. Healthcare Mater., 2019, 8, 1901321 CrossRef CAS PubMed.
- Z. Zhao, Q. Li, L. Chen, Y. Zhao, J. Gong, Z. Li and J. Zhang, A thread/fabric-based band as a flexible and wearable microfluidic device for sweat sensing and monitoring, Lab Chip, 2021, 21, 916–932 RSC.
- E. Asadian, F. Hekmat, M. Hafezi Kahnamouei, R. Mohammadpour, S. Shahrokhian and P. Sasanpour, Supercapacitor-powered wearable biosensor for continuous lactate monitoring from sweat, Biosens. Bioelectron., 2025, 275, 117226 CrossRef CAS PubMed.
- S. Wang, M. Liu, X. Yang, Q. Lu, Z. Xiong, L. Li, H. Zheng, S. Feng and T. Zhang, An unconventional vertical fluidic-controlled wearable platform for synchronously detecting sweat rate and electrolyte concentration, Biosens. Bioelectron., 2022, 210, 114351 CrossRef CAS PubMed.
- A. Qureshi and J. H. Niazi, Graphene-interfaced flexible and stretchable micro–nano electrodes: from fabrication to sweat glucose detection, Mater. Horiz., 2023, 10, 1580–1607 RSC.
- N. Davis, A. Kang, E. Hakola, L. Gillan, Y. Zhan, J. Hiltunen and A. Javey, Reusable, Fully Integrated Sweat Monitor Band with Peel-and-Stick-Replacement Printed Microfluidic Sensor, Adv. Mater. Technol., 2025, 10, 2500477 CrossRef CAS.
- R. Vinoth, T. Nakagawa, J. Mathiyarasu and A. M. V. Mohan, Fully Printed Wearable Microfluidic Devices for High-Throughput Sweat Sampling and Multiplexed Electrochemical Analysis, ACS Sens., 2021, 6, 1174–1186 CrossRef CAS PubMed.
- N. Zavanelli and W.-H. Yeo, Advances in Screen Printing of Conductive Nanomaterials for Stretchable Electronics, ACS Omega, 2021, 6, 9344–9351 CrossRef CAS PubMed.
- M. Şen, M. Oğuz and İ. Avcı, Non-toxic flexible screen-printed MWCNT-based electrodes for non-invasive biomedical applications, Talanta, 2024, 268, 125341 CrossRef PubMed.
- S. Khosravi, S. Soltanian, A. Servati, A. Khademhosseini, Y. Zhu and P. Servati, Screen-Printed Textile-Based Electrochemical Biosensor for Noninvasive Monitoring of Glucose in Sweat, Biosensors, 2023, 13, 684 CrossRef CAS PubMed.
- P. Li, P. K. Kalambate, K. D. Harris, A. B. Jemere and X. (Shirley) Tang, Robust and flexible electrochemical lactate sensors for sweat analysis based on nanozyme-enhanced electrode, Biosens. Bioelectron.: X, 2024, 17, 100455 CAS.
- C. Wang, Z. Wang, W. Wei, Z. Zhang, A. A. Li, G. Huang, X. Li, S. S. Ge, L. Zhou and H. Kong, High-precision flexible sweat self-collection sensor for mental stress evaluation, npj Flexible Electron., 2024, 8, 47 CrossRef.
- Z. Xing, J. Hui, B. Lin, Z. Wu and H. Mao, Recent Advances in Wearable Sensors for the Monitoring of Sweat: A Comprehensive Tendency Summary, Chem, 2023, 11, 470 CAS.
- M. Li, G. Li, Z. Li, Y. Tang, R. Xu, T. Xu, Y. Wang, Y. Liu, L. Qu, B. Wang, Y. Yang and M. Tian, Ultrasensitive biosensing meta-garment via wetting gradient effect for heat-exhaustion warning, npj Flexible Electron., 2025, 9, 17 CrossRef.
- R. F. R. Ursem, A. Steijlen, M. Parrilla, J. Bastemeijer, A. Bossche and K. D. Wael, Worth your sweat: wearable microfluidic flow rate sensors for meaningful sweat analytics, Lab Chip, 2025, 25, 1296–1315 RSC.
- A. Stojković, N. Radlović, B. Vuletić, B. Nestorović, Z. Leković, S. Obradović and A. Vujić, Presentation of an infant with nutritional deficiency dermatitis as the initial manifestation of cystic fibrosis, Srp. Arh. Celok. Lek., 2013, 141, 810–813 CrossRef PubMed.
- W. Ji, H. Liu, Y. Liu, W. Zhang, T. Zhou, X. Liu, C. Tao, J. Dai, B. Zha, R. Xie, J. Wu, Q. Wu, W. Zhang, L. Li and F. Huo, Large-scale fully printed “LEGO® Bricks” type wearable sweat sensor for physical activity monitoring, npj Flexible Electron., 2023, 7, 53 CrossRef.
- A. R. Sharifi, S. Ardalan, R. S. Tabatabaee, S. Soleimani Gorgani, H. Yousefi, K. Omidfar, M. A. Kiani, C. Dincer, T. Naghdi and H. Golmohammadi, Smart Wearable Nanopaper Patch for Continuous Multiplexed Optical Monitoring of Sweat Parameters, Anal. Chem., 2023, 95, 16098–16106 CrossRef CAS PubMed.
- W. He, C. Wang, H. Wang, M. Jian, W. Lu, X. Liang, X. Zhang, F. Yang and Y. Zhang, Integrated textile sensor patch for real-time and multiplex sweat analysis, Sci. Adv., 2019, 5, eaax0649 CrossRef CAS PubMed.
- L. J. Currano, F. C. Sage, M. Hagedon, L. Hamilton, J. Patrone and K. Gerasopoulos, Wearable Sensor System for Detection of Lactate in Sweat, Sci. Rep., 2018, 8, 15890 CrossRef PubMed.
- A. Sharma, M. Badea, S. Tiwari and J. L. Marty, Wearable Biosensors: An Alternative and Practical Approach in Healthcare and Disease Monitoring, Molecules, 2021, 26, 748 CrossRef CAS PubMed.
- O. Parlak, V. F. Curto, E. Ojeda, L. Basabe-Desmonts, F. Benito-Lopez and A. Salleo, in Wearable Bioelectronics, Elsevier, 2020, pp. 65–88 Search PubMed.
- Y. Yang, Y. Song, X. Bo, J. Min, O. S. Pak, L. Zhu, M. Wang, J. Tu, A. Kogan, H. Zhang, T. K. Hsiai, Z. Li and W. Gao, A laser-engraved wearable sensor for sensitive detection of uric acid and tyrosine in sweat, Nat. Biotechnol., 2020, 38, 217–224 CrossRef CAS PubMed.
- B. Gil, S. Anastasova and G. Z. Yang, A Smart Wireless Ear-Worn Device for Cardiovascular and Sweat Parameter Monitoring During Physical Exercise: Design and Performance Results, Sensors, 2019, 19, 1616 CrossRef CAS PubMed.
- M. Wang, Y. Yang, J. Min, Y. Song, J. Tu, D. Mukasa, C. Ye, C. Xu, N. Heflin, J. S. McCune, T. K. Hsiai, Z. Li and W. Gao, A wearable electrochemical biosensor for the monitoring of metabolites and nutrients, Nat. Biomed. Eng., 2022, 6, 1225–1235 CrossRef CAS PubMed.
- Y. J. Hong, H. Lee, J. Kim, M. Lee, H. J. Choi, T. Hyeon and D.-H. Kim, Multifunctional Wearable System that Integrates Sweat-Based Sensing and Vital-Sign Monitoring to Estimate Pre-/Post-Exercise Glucose Levels, Adv. Funct. Mater., 2018, 28, 1805754 CrossRef.
- M. N. Sawka and S. J. Montain, Fluid and electrolyte supplementation for exercise heat stress, Am. J. Clin. Nutr., 2000 DOI:10.1093/ajcn/72.2.564S.
- American College of Sports Medicine, M. N. Sawka, L. M. Burke, E. R. Eichner, R. J. Maughan, S. J. Montain and N. S. Stachenfeld, American College of Sports Medicine position stand. Exercise and fluid replacement, Med. Sci. Sports Exercise, 2007, 39, 377–390 CrossRef PubMed.
- S. M. Shirreffs and M. N. Sawka, Fluid and electrolyte needs for training, competition, and recovery, J. Sports Sci., 2011, 29(1), S39–S46 CrossRef PubMed.
- R. J. Maughan and S. M. Shirreffs, Development of individual hydration strategies for athletes, Int. J. Sport Nutr. Exercise Metab., 2008, 18, 457–472 Search PubMed.
- A. Uzunoglu and L. A. Stanciu, Novel CeO2–CuO-decorated enzymatic lactate biosensors operating in low oxygen environments, Anal. Chim. Acta, 2016, 909, 121–128 CrossRef CAS PubMed.
- M. L. Goodwin, J. E. Harris, A. Hernández and L. B. Gladden, Blood Lactate Measurements and Analysis during Exercise: A Guide for Clinicians, J. Diabetes Sci. Technol., 2007, 1, 558–569 CrossRef PubMed.
- J. Anzai, H. Takeshita, Y. Kobayashi, T. Osa and T. Hoshi, Layer-by-Layer Construction of Enzyme Multilayers on an Electrode for the Preparation of Glucose and Lactate Sensors: Elimination of Ascorbate Interference by Means of an Ascorbate Oxidase Multilayer, Anal. Chem., 1998, 70, 811–817 CrossRef CAS PubMed.
- A. Uzunoglu, CeO2-ZrO2 Nanoparticle-Modified Enzymatic Lactate Biosensors with Reduced Oxygen Susceptibility, J. Electrochem. Soc., 2018, 165, B436 CrossRef CAS.
- H. Lee, C. Song, Y. S. Hong, M. Kim, H. R. Cho, T. Kang, K. Shin, S. H. Choi, T. Hyeon and D.-H. Kim, Wearable/disposable sweat-based glucose monitoring device with multistage transdermal drug delivery module, Sci. Adv., 2017, 3, e1601314 CrossRef PubMed.
- L. B. Baker, J. R. Stofan, A. A. Hamilton and C. A. Horswill, Comparison of regional patch collection vs. whole body washdown for measuring sweat sodium and potassium loss during exercise, J. Appl. Physiol., 2009, 107, 887–895 CrossRef CAS PubMed.
- J. Moyer, D. Wilson, I. Finkelshtein, B. Wong and R. Potts, Correlation between sweat glucose and blood glucose in subjects with diabetes, Diabetes Technol. Ther., 2012, 14(5) DOI:10.1089/dia.2011.0262.
- S. Imani, A. J. Bandodkar, A. M. V. Mohan, R. Kumar, S. Yu, J. Wang and P. P. Mercier, A wearable chemical-electrophysiological hybrid biosensing system for real-time health and fitness monitoring, Nat. Commun., 2016, 7, 11650 CrossRef CAS PubMed.
- X. Xuan, H. S. Yoon and J. Y. Park, A wearable electrochemical glucose sensor based on simple and low-cost fabrication supported micro-patterned reduced graphene oxide nanocomposite electrode on flexible substrate, Biosens. Bioelectron., 2018, 109, 75–82 CrossRef CAS PubMed.
- E. V. Karpova, E. V. Shcherbacheva, A. A. Galushin, D. V. Vokhmyanina, E. E. Karyakina and A. A. Karyakin, Noninvasive Diabetes Monitoring through Continuous Analysis of Sweat Using Flow-Through Glucose Biosensor, Anal. Chem., 2019, 91, 3778–3783 CrossRef CAS PubMed.
- V. A. LeGrys, Sweat analysis proficiency testing for cystic fibrosis, Pediatr. Pulmonol., 2000, 30, 476–480 CrossRef CAS PubMed.
- S. Coyle, D. Morris, K.-T. Lau, D. Diamond and N. Moyna, in 2009 Sixth International Workshop on Wearable and Implantable Body Sensor Networks, 2009, pp. 307–311 Search PubMed.
- Y. Jiang, Md. N. Islam, R. He, X. Huang, P.-F. Cao, R. C. Advincula, N. Dahotre, P. Dong, H. F. Wu and W. Choi, Recent Advances in 3D Printed Sensors: Materials, Design, and Manufacturing, Adv. Mater. Technol., 2023, 8, 2200492 CrossRef.
- P. J. Derbyshire, H. Barr, F. Davis and S. P. J. Higson, Lactate in human sweat: a critical review of research to the present day, J. Physiol. Sci., 2012, 62, 429–440 CrossRef CAS PubMed.
- S. Imani, A. J. Bandodkar, A. M. V. Mohan, R. Kumar, S. Yu, J. Wang and P. P. Mercier, A wearable chemical–electrophysiological hybrid biosensing system for real-time health and fitness monitoring, Nat. Commun., 2016, 7, 11650 CrossRef CAS PubMed.
- E. V. Karpova, E. E. Karyakina and A. A. Karyakin, Wearable non-invasive monitors of diabetes and hypoxia through continuous analysis of sweat, Talanta, 2020, 215, 120922 CrossRef CAS PubMed.
- J. Kim, W. R. de Araujo, I. A. Samek, A. J. Bandodkar, W. Jia, B. Brunetti, T. R. L. C. Paixão and J. Wang, Wearable temporary tattoo sensor for real-time trace metal monitoring in human sweat, Electrochem. Commun., 2015, 51, 41–45 CrossRef CAS.
- W. Gao, H. Y. Y. Nyein, Z. Shahpar, H. M. Fahad, K. Chen, S. Emaminejad, Y. Gao, L.-C. Tai, H. Ota, E. Wu, J. Bullock, Y. Zeng, D.-H. Lien and A. Javey, Wearable Microsensor Array for Multiplexed Heavy Metal Monitoring of Body Fluids, ACS Sens., 2016, 1, 866–874 CrossRef CAS.
- K. C. Honeychurch and J. P. Hart, Screen-printed electrochemical sensors for monitoring metal pollutants, TrAC, Trends Anal. Chem., 2003, 22, 456–469 CrossRef CAS.
- G. Matzeu, L. Florea and D. Diamond, Advances in wearable chemical sensor design for monitoring biological fluids, Sens. Actuators, B, 2015, 211, 403–418 CrossRef CAS.
- V. F. Curto, C. Fay, S. Coyle, R. Byrne, C. O'Toole, C. Barry, S. Hughes, N. Moyna, D. Diamond and F. Benito-Lopez, Real-time sweat pH monitoring based on a wearable chemical barcode micro-fluidic platform incorporating ionic liquids, Sens. Actuators, B, 2012, 171–172, 1327–1334 CrossRef CAS.
- W. Ji, J. Zhu, W. Wu, N. Wang, J. Wang, J. Wu, Q. Wu, X. Wang, C. Yu, G. Wei, L. Li and F. Huo, Wearable Sweat Biosensors Refresh Personalized Health/Medical Diagnostics, Research, 2021, 2021, 9757126 CrossRef CAS PubMed.
- Y.-J. Guo, C.-C. Li, J.-A. Huang, K. Singh, H. Mellini, Y.-H. Lin, C.-C. Yu, T.-C. Fang, C.-C. Pi, P.-C. Chen, C.-Y. Li, S.-Y. Fan, W.-C. Huang, T.-H. Tsai, Y.-T. Liao and L.-C. Tai, Wearable sensors for monitoring drug pharmacokinetics in patients with Parkinson’s disease, Sci. Rep., 2026, 16 DOI:10.1038/s41598-026-43825-w.
- Y. Liu, J. Li, S. Xiao, Y. Liu, M. Bai, L. Gong, J. Zhao and D. Chen, Revolutionizing Precision Medicine: Exploring Wearable Sensors for Therapeutic Drug Monitoring and Personalized Therapy, Biosensors, 2023, 13, 726 CrossRef CAS PubMed.
- C.-B. Ma, X. Shang, M. Sun, X. Bo, J. Bai, Y. Du and M. Zhou, Emerging Multifunctional Wearable Sensors: Integrating Multimodal Sweat Analysis and Advanced Material Technologies for Next-Generation Health Monitoring, ACS Sens., 2025, 10, 2388–2408 CrossRef CAS PubMed.
- Z. Chen, Y. Liu, W. Yu, S. Liu and Y. Huang, Machine Learning-Driven Wearable Sweat Sensors with AgNW/MXene for Non-Invasive SERS-Based Cardiovascular Disease Detection, ACS Appl. Nano Mater., 2025, 8, 5602–5610 CrossRef CAS.
- J. Wang, S. Wang and J. Zhang, Recent advances in wearable sweat sensors using functional nucleic acids: progress and future directions, Flexible Printed Electron., 2025, 10, 013502 CrossRef CAS.
- J. Sun, W. Dai, Q. Guo, Y. Gao, J. Chen, J. L. Chen, G. Mao, H. Sun and Y.-K. Peng, Self-powered wearable electrochemical sensor based on composite conductive hydrogel medium for detection of lactate in human sweat, Biosens. Bioelectron., 2025, 277, 117303 CrossRef CAS PubMed.
- H. Zafar, A. Channa, V. Jeoti and G. M. Stojanović, Comprehensive Review on Wearable Sweat-Glucose Sensors for Continuous Glucose Monitoring, Sensors, 2022, 22, 638 CrossRef CAS PubMed.
- S. Anastasova, B. Crewther, P. Bembnowicz, V. Curto, H. M. Ip, B. Rosa and G.-Z. Yang, A wearable multisensing patch for continuous sweat monitoring, Biosens. Bioelectron., 2017, 93, 139–145 CrossRef CAS PubMed.
- H. Y. Y. Nyein, L.-C. Tai, Q. P. Ngo, M. Chao, G. B. Zhang, W. Gao, M. Bariya, J. Bullock, H. Kim, H. M. Fahad and A. Javey, A Wearable Microfluidic Sensing Patch for Dynamic Sweat Secretion Analysis, ACS Sens., 2018, 3, 944–952 CrossRef CAS PubMed.
- J. R. Sempionatto, M. Lin, L. Yin, E. De la Paz, K. Pei, T. Sonsa-Ard, A. N. de Loyola Silva, A. A. Khorshed, F. Zhang, N. Tostado, S. Xu and J. Wang, An epidermal patch for the simultaneous monitoring of haemodynamic and metabolic biomarkers, Nat. Biomed. Eng., 2021, 5, 737–748 CrossRef CAS PubMed.
- N. Gao, G. Xu, G. Chang and Y. Wu, From Lab to Life: Self-Powered Sweat Sensors and Their Future in Personal Health Monitoring, Adv. Sci., 2025, 12, e2409178 CrossRef PubMed.
- T. Laochai, J. Yukird, N. Promphet, J. Qin, O. Chailapakul and N. Rodthongkum, Non-invasive electrochemical immunosensor for sweat cortisol based on L-cys/AuNPs/MXene modified thread electrode, Biosens. Bioelectron., 2022, 203, 114039 CrossRef CAS PubMed.
- L. Xu, X. Hu, F. Zhang, S. Zhou, Z. Zhang, J. Zhang, H. Liang, H. Liu, G. Chen, Q. Zhang, S. Das, J. Tao and J. Wan, Simple, Low-Cost, Hydrophilic, and Permeable Sensors for Sweat Ascorbic Acid Detection with Screen-Printed Carbon Electrodes and Flexible Wood Substrates, Adv. Mater. Technol., 2026, 11, e01602 CrossRef CAS.
- Y. Hua, J. Mai, R. Su, C. Ma, J. Liu, C. Zhao, Q. Zhang, C. Liao and Y. Wang, A Highly Stable Electrochemical Sensor Based on a Metal–Organic Framework/Reduced Graphene Oxide Composite for Monitoring the Ammonium in Sweat, Biosensors, 2024, 14, 617 CrossRef CAS PubMed.
- B. Hosseinzadeh, S. Tonello, N. F. Lopomo and E. Sardini, Fully Printed Flexible Sensor With a Cobalt-Based Metal–Organic Framework Sensing Layer for Humidity Monitoring in Wearable Systems, IEEE Sens. J., 2026, 26, 3644–3657 Search PubMed.
- Y. Yin, Z. Tan, W. Zhu, Z. Pu, H. Yu, R. Wang and D. Li, A wearable microfluidic system for efficient sweat collection and real-time detection, Talanta, 2024, 274, 125967 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |