 Open Access Article
Open Access ArticleA DSE–ESIPT-active organic luminogen for “turn-on” enantioselective recognition of chiral amino alcohols and selective hydrazine sensing
Aakash Venkatesan a,
Baru Venkata Naga Sahithi
a,
Baru Venkata Naga Sahithi b,
Francis Joya,
Rupali Patel
b,
Francis Joya,
Rupali Patel c,
Aatika Nizam
c,
Aatika Nizam *a and
Vasantha Veerappa Lakshmaiah
*a and
Vasantha Veerappa Lakshmaiah *b
*b
aDepartment of Chemistry, Christ University, Bangalore-560029, India. E-mail: aatika.nizam@christuniversity.in
bDepartment of Life Sciences, Christ University, Bangalore-560029, India. E-mail: vasantha.vl@christuniversity.in
cDepartment of Physics and Electronics, Christ University, Bangalore-560029, India
First published on 23rd February 2026
Abstract
The development of dual-state emissive (DSE) organic luminogens has elevated the ease of recognition of various biological analytes, which demonstrates the multifaceted potential of dual-state emitters. Therefore, in this study, we synthesised a dual-state emissive excited-state intramolecular proton transfer (ESIPT)-based organic luminogen, (E)-4-(5-bromo-2-hydroxybenzylideneamino)-2,3-dimethyl-1-phenyl-1,2-dihydropyrazol-5-one (ANMB), exhibiting excitation-dependent phototunability with large Stokes shifts of 109 nm and 155 nm in both the solution and solid states, respectively, underscoring its potential as a biosensor. The metal-chelating ability of ANMB was investigated, revealing significant fluorescence quenching upon coordination with Cu2+ ions, leading to 96% reduction in emission intensity. The introduction of biological analytes, such as amino alcohols, enabled fluorescence recovery, where ANMB demonstrated enantioselective recognition: a single emission peak for the S-enantiomer and dual emission peaks for the R-enantiomer. Furthermore, ANMB demonstrated high selectivity for hydrazine detection in both the solution and solid states, with new emission bands observed at 411 nm and 432 nm, respectively, indicating a fluorescence shift from green to blue. Complementarily, ANMB was successfully applied for real-time imaging of hydrazine in food and plant samples, showcasing its practical adaptability. Additionally, in silico molecular docking studies were performed, revealing the potential therapeutic activity of ANMB against diarrheal targets. Overall, this work highlights the multifunctionality and tunability of DSE–ESIPT-based organic luminogens, positioning ANMB as a promising candidate for the selective recognition of biologically significant analytes in analytical and real-world contexts.
1. Introduction
Organic luminescent molecules have attracted the attention of researchers over the past few decades because of their ability to modify their optical signatures in response to external stimuli in the presence of specific substrates or variations in environmental physical parameters (pH, viscosity, temperature, pressure, polarity, etc.).1 This enables their diverse practical applications in numerous fields, such as biological sciences,2 pharmacology,3 environmental sciences4 and material sciences.5 There are two main application categories for organic fluorescent chromophores. Most organic molecules exhibit strong fluorescence emission when solvated in organic or aqueous media owing to their significant radiative deactivation channels.6 The chromophores demonstrating emission in solutions are used as fluorescent biomarkers for biomedical applications, including the imaging of cells,7 cellular organelles,8 tissues,9 macromolecules,10 and tumors.11 However, their high emission in the solution phase is often accompanied by detrimental aggregation in the solid state. This enables a strong non-radiative deactivation process, a phenomenon known as aggregation-caused quenching (ACQ), which limits their practical applications in films and devices.6 To avoid ACQ, Tang et al. proposed the concept of aggregation-induced emission (AIE), whereby molecular rotors in the chromophores facilitate non-radiative deactivation that causes fluorescence quenching in the solution state, whereas in the solid state, the restriction of the motion of the molecular rotors in the aggregated state activates radiative deactivation channels, resulting in strong fluorescence.12 The applications of solid-state emissive organic fluorophores include data encryption (logic gates and anti-counterfeiting security inks)13,14 and their use in devices such as organic photovoltaics (OPVs),15 organic light-emitting diodes (OLEDs)16,17 and organic field effect transistors (OFETs).18These materials have incentivized the research on molecules that exhibit fluorescence in both solution and solid states, which are popularized as dual-state emitters. Dual-state emissive (DSE) organic fluorophores were first reported by Tang et al. in 2015, and they have drawn a lot of interest as they allow for the use of a single fluorophore for a variety of applications, bridging the large functional gap between ACQ and AIE.19 To construct a molecule with DSE properties, a delicate balance of planarity, rigidity, and solubility is required, which is challenging, particularly in sensing applications.20 ESIPT-based molecules exhibit a range of capabilities, including reversible mechanochromism, aggregation-induced emission (AIE), ratiometric sensing, and anti-counterfeiting.21 They exhibit photophysical properties owing to the phototautomerism of the excited normal species (N*) and tautomeric species (T*), resulting in dual emission peaks, corresponding to their enol (E*) and keto (K*) forms, respectively.22 Thus, due to an easily apprehended proton transfer, this particular excited-state dynamics yields many advantageous characteristics, such as significant Stokes shifts, increased photostability, strong solid-state emission, and environment-sensitive optical properties.6
As a consequence, ESIPT-based DSE materials have emerged as attractive tools for the sensing of various biological analytes. Therefore, in this article, we present a DSE–ESIPT active organic luminogen, (E)-4-(5-bromo-2-hydroxybenzylideneamino)-2,3-dimethyl-1-phenyl-1,2-dihydropyrazol-5-one (ANMB), which exhibits chelation-induced quenching on complexation with Cu2+ ions, enabling ratiometric sensing of Cu2+ ions in the solution state. Furthermore, the retraction of the fluorescence was achieved by introducing phenylglycinol into the ANMB + Cu2+ system, attributed to the competitive affinity of phenylglycinol towards Cu2+ ions over ANMB, which triggers a “turn on” fluorescence emission. The selective detection of hydrazine in both solution and solid states was achieved through the formation of an imine bond between ANMB and hydrazine via the free carbonyl moiety in ANMB, leading to a hypsochromic shift in the emission spectrum from green to blue fluorescence. Henceforth, we put forth this ANMB organic luminogen as a multifaceted sensor for biological molecules. Additionally, in silico studies and bioimaging were performed to prove the potential adaptability of ESIPT hydrazone in multifaceted domains.
2. Experimental procedures
2.1 Materials and instrumentation
The experimental chemicals, such as 4-aminoantipyrine, 5-bromosalicylaldehyde, hydrazine hydrate, (S)-(+)-2-phenylglycinol and (R)-(−)-2-phenylglycinol, were purchased from Sigma Aldrich, and the solvents utilized were of spectroscopic grade and used without further purification. The absorption spectrum of the compounds was recorded using a Shimadzu UV1800 spectrophotometer in the range of 200–700 nm. Both solution and solid-state emission spectra of the compounds were recorded using a Shimadzu spectrofluorophotometer (RF-6000). The 1H NMR spectrum was recorded in DMSO-d6 using a Bruker Avance 400 MHz FT-NMR spectrometer.2.2 Synthesis and characterisation of ANMB
The antipyrine-based sensor (ANMB) was prepared by a Schiff's base condensation reaction involving 4-aminoantipyrine (0.203 g, 1.0 mmol) and 5-bromosalicylaldehyde (0.201 g, 1.0 mmol) in ethanol for 6 h at 70 °C following a previously reported procedure.23 A yellowish-orange color precipitate was procured on completion of the reaction and extracted using ethyl acetate. After equilibrium was reached, a rotary evaporator was employed to reduce the solvent volume, and a pure form of the product was obtained on slow evaporation. Techniques such as TLC and 1H NMR revealed the product formation.2.3 Preparation of fluorescent tags
Fluorescent tags were made from Whatman filter papers. The filter paper was dipped in a stock solution of ANMB (1.5 × 10−3 M) prepared in DMF-Tris buffer (9/1, v/v, pH 7.4) and dried in a hot air oven for 3 h to obtain fluorescent tags,24 which exhibited lime green fluorescence under UV light (365 nm). These fluorescent tags were used to investigate the solid-state response of ANMB towards various hydrazine derivatives.2.4 Molecular docking studies
The computational docking of ANMB was performed using AutoDock Vina as an extension in UCSF Chimera.6-7. The protein Mus musculus opioid receptors (ORs), including muOR (μOR) and deltaOR (δOR), and the M3 muscarinic acetylcholine receptor structures were retrieved from RCSB Protein DataBank (PDB) (http://www.rcsb.org/pdb), PDB-ID 5C1M, 4EJ4 and 4U14. The proteins and the ligand were prepared following the procedures in a published work.25 Discovery Studio Visualizer v.21.1.0.20298 was used to create the 2D diagrams.2.5 ADME prediction
In silico ADME analysis was conducted using SwissADME to investigate the physicochemical properties, lipophilicity, water solubility, pharmacokinetics, and drug likeness of the materials.2.6 Latent fingerprint analysis
A latent fingerprint study was performed using fingerprints voluntarily provided by an adult participant who provided informed consent. No personal or identifiable information was collected. The hands were washed thoroughly with soap and water, after which the fingers were directed to gently touch naturally oily regions of the face (nose and forehead) and then pressed onto glass substrates under ambient conditions for practical accessibility. To develop the latent fingerprints using the organic luminogen, a small amount of the powder was applied to the imprinted substrates using a soft brush, and excess material was removed by a gentle air flow. Optical images of the latent fingerprints before and after probe treatment were recorded using a mobile phone under UV illumination (365 nm). The experiment was repeated three times, yielding consistent visualization results.263. Results and discussions
3.1 Synthesis of ANMB
(E)-4-(5-Bromo-2-hydroxybenzylideneamino)-2,3-dimethyl-1-phenyl-1,2-dihydropyrazol-5-one (ANMB) was synthesized at 70 °C in ethanol and subsequently refluxed for 6 h to afford the product in 98% yield (Scheme S1, SI). 1H NMR analysis confirmed the product formation (Fig. S1, SI). 1H NMR (δ, ppm): 12.73 (OH, s); 9.65 (HC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, s); 7.74–7.73 (1H, d); 7.56–7.53 (2H, t); 7.44–7.37 (4H, m); 6.89–6.87 (1H, d); 3.22 (3H, s); 2.50–2.49 (3H, t).
N, s); 7.74–7.73 (1H, d); 7.56–7.53 (2H, t); 7.44–7.37 (4H, m); 6.89–6.87 (1H, d); 3.22 (3H, s); 2.50–2.49 (3H, t).
3.2 Investigation of optical properties of ANMB
The optical response of ESIPT-based molecules can be significantly influenced by various external stimuli, such as changes in solvent polarity, varying water fractions, and the coordination of various guest molecules and metal ions. In the present study, ANMB demonstrated an active excited state intramolecular proton transfer (ESIPT) due to the hydroxy group of the salicylaldehyde moiety and the imino nitrogen in both the solution and solid states.Thus, the heterogeneous properties of ANMB can be attributed to the photoisomerization process. Excitation at wavelengths greater than 400 nm resulted in a pronounced blue shift in the emission peak, suggesting the conversion of the enol tautomer to the keto form. The divergence of the keto and enol forms is due to the interaction between the hydroxy group of the salicylaldehyde moiety and the imine nitrogen in the probe. This evidence alludes to the attainable ESIPT mechanism for ANMB and illustrates the proposed emission process (Fig. 1c).
The compiled photophysical data of ANMB under various solvent-polarity conditions (Table S1 and Fig. S2, SI) are calculated, and the lowest quantum yield was observed in MeOH (φ = 0.0024). This can be attributed to the high polarity of the solvent that restricts the formation of intramolecular hydrogen bonds, which inhibits proton transfer in the ANMB system. The highest quantum yield was noticed in DMSO (φ = 0.0136) and DMF (φ = 0.0141). This can be ascribed to the polar aprotic and viscous nature of the solvents that restrict the rotation and promote the efficient transfer of intramolecular protons in the ANMB system, thereby stabilizing the bonds in the excited state. Therefore, based on the quantum yield value, DMF was selected as the optimal solvent for all of the studies conducted.
3.2.5.1. Absorption spectral studies. To examine the Cu2+-ion estimation by ANMB, the absorption spectra of ANMB were recorded upon adding the Cu2+ ions (Fig. 5a). Ratiometric sensing of Cu2+ ions was achieved, characterized by the initial absorption peak of ANMB centered at 360 nm. Upon increasing the concentration of Cu2+ ions, the absorption peak intensity at 360 nm decreased drastically and was accompanied by a bathochromic shift of 20 nm. Subsequently, a new absorption peak at 420 nm emerged, indicating the color change to yellow upon increasing the Cu2+-ion concentration. To estimate the limit of detection (LOD) and limit of quantification (LOQ), a linear plot between A420/A360 and the Cu2+-ion concentration was drawn (Fig. 5c). The LOD and LOQ were found to be 5.91 μM and 17.92 μM, respectively.
3.2.5.2. Photoluminescence spectral studies. To assess the photophysical response of ANMB towards Cu2+ ions, the Cu2+-ion solution was added to ANMB incrementally, which induced significant fluorescence quenching, as indicated by the emission peak at 509 nm (Fig. 5b). Therefore, an emission titration of ANMB was performed by introducing Cu2+ ions. This induced the sequential quenching of the fluorescence intensity, and complete quenching of fluorescence was noticed after the addition of 26 equiv of Cu2+ ions. A linear plot of the emission intensity vs. concentration of Cu2+ ions was utilized for the estimation of LOD and LOQ (Fig. 5d), which were found to be 3.11 μM and 9.41 μM, respectively.
3.2.5.3. Proposed mechanism for ANMB and Cu2+-ion chelation. Job's plot and Benesi–Hildebrand (BH) plot analyses were performed to gain insight into the binding interaction between ANMB and Cu2+ (Fig. 6a and b). The results suggest the possibility of a higher-order association, consistent with a 1
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (Cu2+
2 (Cu2+![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ANMB) binding model. The observed non-linearity in the BH plot further supports deviation from a simple 1
ANMB) binding model. The observed non-linearity in the BH plot further supports deviation from a simple 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding interaction, although definitive stoichiometry determination requires independent confirmation. As per the regulation of the U.S. Environmental Protection Agency, the allowed concentration of copper ions in drinking water is 20 μM.27 Therefore, in the presence of the ANMB probe, the naked eye can be adopted to assess whether the level of copper ions has exceeded the standard limit in actual samples. The observed fluorescence quenching can be attributed to electron or energy transfer from the excited fluorophore to Cu2+, which induces rapid non-radiative decay due to the inherent paramagnetic nature of Cu2+ ions. This interaction is likely to perturb the ESIPT and photo-induced electron transfer (PET) processes of the probe, leading to effective fluorescence quenching. Information from Job's plot suggested the formation of a 1
1 binding interaction, although definitive stoichiometry determination requires independent confirmation. As per the regulation of the U.S. Environmental Protection Agency, the allowed concentration of copper ions in drinking water is 20 μM.27 Therefore, in the presence of the ANMB probe, the naked eye can be adopted to assess whether the level of copper ions has exceeded the standard limit in actual samples. The observed fluorescence quenching can be attributed to electron or energy transfer from the excited fluorophore to Cu2+, which induces rapid non-radiative decay due to the inherent paramagnetic nature of Cu2+ ions. This interaction is likely to perturb the ESIPT and photo-induced electron transfer (PET) processes of the probe, leading to effective fluorescence quenching. Information from Job's plot suggested the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 stoichiometry complex; aided by literature reports, a plausible mechanism and complex structure were suggested (Fig. 6c and d).
2 stoichiometry complex; aided by literature reports, a plausible mechanism and complex structure were suggested (Fig. 6c and d).
 | ||
| Fig. 6 (a) Jobs plot, (b) BH plot, (c) structure of the ANMB–Cu2+ complex and (d) plausible mechanism of fluorescence quenching. | ||
3.2.5.4. Mathematical model studies. Mathematical modelling studies, such as Stern–Volmer plot analysis, were carried out to assess the fluorescence quenching efficiency of the Cu2+ ion upon addition to the ANMB solution (Fig. 7a). The results revealed that at higher concentrations of Cu2+, a discernible nonlinear behaviour with curvature is observed. This anomalous behaviour is well established in other stable copper complex systems based on molecular quenchers, which should thus provide a linear curve. In the present instance, however, the upward curve astonishingly changed to a linear rise at relatively high Cu2+ concentrations, subsequently producing a plateau that denotes equilibrium at higher concentrations. The product, τf kq, from the slope was found utilizing Stern–Volmer eqn (1) and (2) and the plot (Fig. 7b).
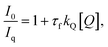 | (1) |
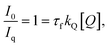 | (2) |
The binding interaction between Cu2+ and ANMB was analyzed (Fig. S5, SI) using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 binding model, yielding binding constants of K11 = 5.14 M−1 and K21 = 3.44 × 106 M−1. The error estimates for K11 and K21 were 13.89% and 1.03%, respectively. The significantly higher K21 value suggests a positive cooperativity effect, where the second ligand binds with much stronger affinity than the first. The model fitting was evaluated using residual analysis and sum of squared residuals (SSR = 1.44 × 107), indicating a reasonable fit to the experimental data.
2 binding model, yielding binding constants of K11 = 5.14 M−1 and K21 = 3.44 × 106 M−1. The error estimates for K11 and K21 were 13.89% and 1.03%, respectively. The significantly higher K21 value suggests a positive cooperativity effect, where the second ligand binds with much stronger affinity than the first. The model fitting was evaluated using residual analysis and sum of squared residuals (SSR = 1.44 × 107), indicating a reasonable fit to the experimental data.
3.2.5.5. Theoretical studies. The structures of ANMB in its enol (ANMB_enol) and keto (ANMB_keto) forms, along with the Cu2+ complex (ANMB_Cu2+), were optimized using DFT at the B3LYP functional level. For the free probe forms (ANMB_enol and ANMB_keto), the 6-311G(d,p) basis set was employed, while the LANL2DZ basis set was used for the Cu2+-containing complex. A comparative analysis of the optimized geometrical parameters and energy profiles of ANMB and its metal complex (Fig. 8a) provides insights into the response mechanism of the probe toward metal-ion binding. The orbital energies of the HOMO of ANMB_enol are primarily localized in the salicylaldehyde region, and the LUMO is localized over the pyrazolone ring of the antipyrine molecule. In contrast, both the HOMO and LUMO of the ANMB_keto are localized over the salicylaldehyde region, extending into the pyrazolone unit, suggesting a more symmetric electron distribution. Interestingly, the ANMB_Cu2+ complex exhibits similar orbital localization to that of the keto form, supporting the hypothesis that chelation is favoured in the keto tautomeric state. This electronic reorganization implies that ANMB_enol, due to the spatial separation of frontier orbitals, is less effective in coordinating Cu2+ ions, possibly leading to partial or weak binding. Conversely, in the keto form, the HOMO–LUMO density is localized in the region encompassing key donor atoms, which includes the phenolate oxygen (O−), imine nitrogen (N), and carbonyl oxygen of the pyrazolone ring (C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), enabling strong and complete chelation of Cu2+ ions. Therefore, the observed orbital behaviour aligns with the structural transition from enol to keto and correlates with the enhanced metal-binding capability of the probe in its keto form. Based on this, the Cu2+ ions coordinate with two molecules of ANMB through a six-coordination arrangement with two phenolate oxygen (O−), two imine nitrogen (N), and two carbonyl oxygen of the pyrazolone ring (C
O), enabling strong and complete chelation of Cu2+ ions. Therefore, the observed orbital behaviour aligns with the structural transition from enol to keto and correlates with the enhanced metal-binding capability of the probe in its keto form. Based on this, the Cu2+ ions coordinate with two molecules of ANMB through a six-coordination arrangement with two phenolate oxygen (O−), two imine nitrogen (N), and two carbonyl oxygen of the pyrazolone ring (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), adopting a distorted octahedral geometry. Additionally, the reduction of the HOMO–LUMO energy gap upon chelation was attributed to the increased conjugation, electron delocalization, and orbital interaction in the presence of the metal ion, which facilitates full chelation and significantly modifies the electronic structure of the probe.
O), adopting a distorted octahedral geometry. Additionally, the reduction of the HOMO–LUMO energy gap upon chelation was attributed to the increased conjugation, electron delocalization, and orbital interaction in the presence of the metal ion, which facilitates full chelation and significantly modifies the electronic structure of the probe.
The electrostatic potential (ESP) plot was generated to visualize the distribution of electrostatic charge over the molecular surfaces of ANMB in its different forms. In ANMB_enol, the ESP map revealed a localized region of negative potential concentrated around the hydroxyl (OH) group of the salicylaldehyde moiety, indicating limited electron delocalization. In contrast, the ANMB_keto exhibited a more delocalized negative electrostatic potential extending across the phenolate oxygen (O−), imine nitrogen (N), and carbonyl oxygen (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) of the pyrazolone ring. This delocalization suggests a more conjugated system in the keto form, which enhances its ability to coordinate with metal ions. Upon complexation with Cu2+ ions, the ESP plot of the resulting ANMB_Cu2+ complex showed a significant neutralization of the negative potential, and the electrostatic distribution became more uniform across the entire molecule. This transformation supports the formation of a stable, fully chelated complex, involving two ANMB ligands coordinating with a single Cu2+ ion. The loss of localized negative regions further confirms that the key donor atoms (phenolate O−, imine N, and pyrazolone C
O) of the pyrazolone ring. This delocalization suggests a more conjugated system in the keto form, which enhances its ability to coordinate with metal ions. Upon complexation with Cu2+ ions, the ESP plot of the resulting ANMB_Cu2+ complex showed a significant neutralization of the negative potential, and the electrostatic distribution became more uniform across the entire molecule. This transformation supports the formation of a stable, fully chelated complex, involving two ANMB ligands coordinating with a single Cu2+ ion. The loss of localized negative regions further confirms that the key donor atoms (phenolate O−, imine N, and pyrazolone C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) are involved in complete chelation, stabilizing the metal center and validating the binding mechanism suggested by the electronic structure analysis.
O) are involved in complete chelation, stabilizing the metal center and validating the binding mechanism suggested by the electronic structure analysis.
3.3 Sensing of biological analytes and biological activity
Upon investigating the multifaceted properties of ANMB, we tested its sensing performance towards biological analytes, such as (S)-(+)-2-phenylglycinol, (R)-(−)-2-phenylglycinol, and hydrazine, which exhibit selectively enhanced fluorescence responses.3.3.1.1. Photoluminescence studies. The ANMB-based amino alcohol chemosensor's potential was examined using the emission spectrum. The optimized condition, which included 50 μL of ANMB stock solution and 25 μM of Cu2+, was applied after the trials. For detection under the nearly neutral state for compatibility with biological systems, a pH of 7.4 was maintained. The fluorescence recovery was observed as the concentration of (S)-(+)-2-phenylglycinol increased from 0 to 64.71 μM, with an emission peak shouldered at 497 nm, along with a hypochromic shift of roughly 14 nm (Fig. 9a). Additionally, fluorescence recovery was observed as the concentration of (R)-(−)-2-phenylglycinol increased from 0 to 40.25 μM, with a dual emission peak shouldered at 464 and 502 nm (Fig. 9c), which enables ANMB to outperform as an enantioselective sensor of amino alcohols. No discernible change in the fluorescence emission was seen upon further addition, which indicates the equilibrium point of the chemosensor towards amino alcohols. The probe may exhibit this behaviour due to simultaneous association and dissociation with the metal ion. Consequently, the probe's free form is present at all times throughout the reaction. This anomalous fluorescence recovery behaviour of the ANMB–Cu2+ system is due to the stabilisation of both conformers or electronic states of ANMB by the R enantiomer, thereby exhibiting two distinct emission peaks, each associated with its corresponding electronic state. Contrary to that for the R enantiomer, a single emission peak was observed in the case of the S enantiomer, which was attributed to the conformation or steric hindrance that prevents ESIPT, allowing only normal emission. The LOD and LOQ were estimated using a linear plot of emission intensity vs. (S)-(+)-2-phenylglycinol and (R)-(−)-2-phenylglycinol concentration (Fig. 9b and d), and the results showed that they were 1.13 μM and 3.42 μM for (S)-(+)-2-phenylglycinol and 3.32 μM and 10.07 μM for (R)-(−)-2-phenylglycinol, respectively. The bindfit model studies (Fig. S6, SI) also suggested the strong interaction between the ANMB–Cu2+ system and (S)-(+)-2-phenylglycinol and (R)-(−)-2-phenylglycinol. Furthermore, fluorescence measurements performed with a mixture containing both R and S enantiomers (Fig. 9e) produced an emission spectrum distinct from those of the individual enantiomers, suggesting competitive binding and differential interaction within the chiral environment of the ANMB–Cu2+ ensemble. All experiments were conducted under identical conditions, including solvent composition, Cu2+ concentration, and total analyte concentration, minimizing the possibility of concentration- or aggregation-induced artifacts.
3.3.2.1. Photoluminescence studies. Emission spectra were measured to investigate the selective sensing of hydrazine in solution (Fig. 10a). On incremental addition of hydrazine to the probe solution, new dual peaks appeared at 411 and 432 nm, and the probe emission peak intensity at 520 nm increased, displaying a blueish green fluorescence that indicates the partial interaction of ANMB and hydrazine. This behaviour was observed until the hydrazine concentration reached 0.73 mM, and on further addition of hydrazine, the emission peak at 520 nm began to drop, and the peaks at 411 nm and 432 nm displayed a brilliant blue fluorescence. These results indicated the full interaction of the ANMB and hydrazine (Scheme S2, SI), which was monitored using NMR (Fig. S7, SI). The LOD and LOQ were estimated using a linear plot of the emission intensity (I432/I520) vs. hydrazine concentration (Fig. 10b), and the values are 0.122 mM and 0.371 mM, respectively.
The temporal response of the probe toward hydrazine was investigated by monitoring the emission intensities at 432 and 520 nm as a function of time after hydrazine addition (Fig. 10c). The emission at 432 nm increases progressively, while the emission at 520 nm exhibits an initial rise, followed by a gradual decrease. In contrast, the ratiometric signal (I432/I520) displays a clear and monotonic increase with time (Fig. 10d), reaching a steady value within approximately 8–9 min and remaining stable thereafter with negligible variation, indicating a reaction-mediated sensing mechanism and demonstrating that the ratiometric response provides a reliable, time-resolved analytical signal suitable for quantitative hydrazine detection. A selectivity bar chart comparing the reaction of ANMB to hydrazine versus interferences has been provided (Fig. S8, SI) to assess the selectivity of ANMB towards hydrazine, which demonstrated that the ANMB exhibits high specificity for hydrazine over other tested species.
3.3.2.2. Fluorescent tag studies. Fluorescent tags were made from filter paper to develop a simple and reliable detection method for analyzing hydrazine in the solid state. As demonstrated in the image, the tags emitted lime green fluorescence when exposed to UV light (365 nm) but remained colorless under visible light. Various concentrations (0 mM to 15 mM) of hydrazine were introduced to the tags, and images were captured under UV light (365 nm) and visible light. The image revealed that with increasing the concentration of hydrazine, a color shift from lime green to blue fluorescence under UV light (365 nm) and from colorless to pale yellow under visible light is observed (Fig. 11). This color shift with varied hydrazine concentrations reveals an interaction between hydrazine and ANMB, which changes the photophysical properties of ANMB.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P value of 3.49 and low log
P value of 3.49 and low log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) S demonstrate its moderate solubility and drug-likeness, as represented in Table S3.
S demonstrate its moderate solubility and drug-likeness, as represented in Table S3.3.4 Real-time applications
| Samples | Cu2+ spiked (μM) | Cu2+ recovered (μM) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| River water | 10 | 10.17 ± 0.10 | 101.7 | 1.02 |
| Tap water | 10 | 9.73 ± 0.13 | 97.3 | 1.29 |
| Drinking water | 10 | 9.83 ± 0.08 | 98.3 | 0.78 |
| Samples | NH2NH2 spiked (mM) | NH2NH2 recovered (mM) | Recovery (%) | RSD (%) |
|---|---|---|---|---|
| Drinking water | 0.5 | 0.484 ± 0.00513 | 96.94 | 1.06 |
| Neem leaf extract | 0.5 | 0.456 ± 0.00611 | 91.26 | 1.34 |
4. Conclusion
In this study, we developed and characterized a novel dual-state emissive (DSE) organic luminogen, ANMB, incorporating an excited-state intramolecular proton transfer (ESIPT) mechanism. ANMB displayed an exceptional Stokes shift—109 nm in solution and 155 nm in the solid state—providing strong evidence of its potential as a multifunctional sensor. Its structural design enabled both solution-phase and solid-state emission, which is a critical requirement for practical sensing platforms, especially for biological and environmental diagnostics. ANMB exhibited pronounced quenching upon chelation with Cu2+ ions, indicating high sensitivity and selectivity for metal-ion detection. This quenching effect served as a basis for fluorescence recovery experiments, where the probe demonstrated enantioselective recognition of amino alcohols. Notably, the S-enantiomer led to a single emission peak, while the R-enantiomer resulted in dual emission peaks, demonstrating ANMB's capacity to distinguish chiral molecules, which is of great importance in pharmaceutical and biochemical fields. Further, ANMB proved to be an effective and selective chemosensor for hydrazine. Its interaction with hydrazine induced a clear spectral shift with new emission peaks at 411 nm and 432 nm, corresponding to a visible change from green to blue fluorescence. In addition to its sensing capabilities, in silico docking studies suggested a potential therapeutic application of ANMB in targeting biological pathways relevant to diarrheal diseases. This highlights the compound's promise in not only diagnostic domains but also therapeutic domains, warranting further biological and pharmacological investigations. Taken together, this work underscores the power of integrating DSE and ESIPT mechanisms into organic luminogens to create highly sensitive, selective, and versatile materials for multifunctional applications. ANMB represents a valuable advancement in the field of luminescent materials, with prospective utility in chiral sensing, environmental monitoring, toxic-substance detection, and potential therapy development.Conflicts of interest
There are no conflicts to declare.Data availability
All the data supporting the findings of this study are included as text, figures, and tables. Other supporting data are available in the supplementary data. Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5sd00214a.Acknowledgements
The authors would like to thank Christ University for constant encouragement and support. The authors extend their appreciation to the Centre for Research at CHRIST University for funding this work through seed fund money under grant number CU-ORS-SM-24/12.References
- A. Mondal, E. Ahmmed, B. Ball and P. Chattopadhyay, ChemistrySelect, 2022, 7, e202103857, DOI:10.1002/slct.202103857.
- A. Venkatesan, A. Cherian and A. Nizam, Analyst, 2025, 150, 5124–5141, 10.1039/D5AN01012H.
- G. Vargas-Nadal, M. Köber, A. Nsamela, F. Terenziani, C. Sissa, S. Pescina, F. Sonvico, A. M. Gazzali, H. A. Wahab, L. Grisanti and M. E. Olivera, Pharmaceutics, 2022, 14, 2498, DOI:10.3390/pharmaceutics14112498.
- X. Tian, L. C. Murfin, L. Wu, S. E. Lewis and T. D. James, Chem. Sci., 2021, 12, 3406–3426, 10.1039/d0sc06928k.
- J. Kim, J. H. Oh and D. Kim, Org. Biomol. Chem., 2021, 19, 933–946, 10.1039/d0ob02387f.
- T. Stoerkler, T. Pariat, A. Laurent, D. Jacquemin, G. Ulrich and J. Massue, Molecules, 2022, 27, 2443, DOI:10.3390/molecules27082443.
- M. Jia, Y. Ren, S. Xiong, S. Li, P. Wang and C. Zhao, Spectrochim. Acta, Part A, 2025, 346, 126942, DOI:10.1016/j.saa.2025.126942.
- Y. Ren, M. Jia, S. Xiong, C. Pu, P. Wang and Y. An, Spectrochim. Acta, Part A, 2025, 216, 114732, DOI:10.1016/j.microc.2025.114732.
- J. Yin, Y. Kwon, D. Kim, D. Lee, G. Kim, Y. Hu, J. H. Ryu and J. Yoon, J. Am. Chem. Soc., 2014, 136, 5351–5358, DOI:10.1021/ja412628z.
- H. Tian Jr, A. C. Sedgwick, H. H. Han, S. Sen, G. R. Chen, Y. Zang, J. L. Sessler, T. D. James, J. Li and X. P. He, Coord. Chem. Rev., 2021, 427, 213577, DOI:10.1016/j.ccr.2020.213577.
- M. Gao, F. Yu, C. Lv, J. Choo and L. Chen, Chem. Soc. Rev., 2017, 46, 2237–2271, 10.1039/c6cs00908e.
- Y. Hong, W. Y. J. Lam and B. Z. Tang, Chem. Commun., 2009, 4332–4353, 10.1039/b904665h.
- D. C. Magri, Coord. Chem. Rev., 2021, 426, 213598, DOI:10.1016/j.ccr.2020.213598.
- X. F. Li, W. Zhou, Y. C. Liu, M. Hou, G. L. Feng, Y. M. Ji, Y. Zhang and G. W. Xing, Chem. Commun., 2022, 58, 11547–11550, 10.1039/D2CC04760H.
- A. Syed, H. Renuka, A. Mohitkar, R. Pallepogu, M. S. K. Challa, S. Goel and S. Jayanty, Mater. Adv., 2022, 3, 3151–3164, 10.1039/d1ma00778e.
- M. Shimizu and T. Hiyama, Chem. – Asian J., 2010, 5, 1516–1531, DOI:10.1002/asia.200900727.
- J. Gierschner, J. Shi, B. Milián-medina, D. Roca-sanjuán, S. Varghese and S. Y. Park, Adv. Opt. Mater., 2021, 9, 2002251, DOI:10.1002/adom.202002251.
- M. Gsänger, D. Bialas, L. Huang, M. Stolte and F. Würthner, Adv. Mater., 2016, 28, 3615–3645, DOI:10.1002/adma.201505440.
- G. Chen, W. Li, T. Zhou, Q. Peng, D. Zhai, H. Li, W. Z. Yuan, Y. Zhang and B. Z. Tang, Adv. Mater., 2015, 27, 4496–4501, DOI:10.1002/adma.201501981.
- X. Li, M. Li, Y. Chen, G. Qiao, Q. Liu, Z. Zhou, W. Liu and Q. Wang, Chem. Eng. J., 2021, 415, 128975, DOI:10.1016/j.cej.2021.128975.
- T. Pariat, M. Munch, M. Durko-Maciag, J. Mysliwiec, P. Retailleau, P. M. Verite, D. Jacquemin, J. Massue and G. Ulrich, Chem. – Eur. J., 2021, 27(10), 3483–3495, DOI:10.1002/chem.202004767.
- F. Joy, A. V. J. Devasia and A. Nizam, Appl. Spectrosc. Rev., 2024, 59, 959–988, DOI:10.1080/05704928.2023.2276925.
- F. Joy, A. V. J. Devasia and A. Nizam, Supramol. Chem., 2024, 1–13, DOI:10.1080/10610278.2024.2406269.
- F. Joy, J. Devasia, Y. Nair and A. Nizam, Food. Chem., 2023, 427, 136643, DOI:10.1016/j.foodchem.2023.136643.
- B. V. N. Sahithi and V. V. Lakshmaiah, RSC Adv., 2025, 15, 28721–28729, 10.1039/D5RA04728E.
- A. Venkatesan, A. Nizam, A. R. Cherian, R. Patel, J. Xavier, P. K R, H. R and N. G, RSC Adv., 2025, 15, 32638–32653, 10.1039/D5RA05696A.
- US-EPA and U.S.-E.P.A., National Primary Drinking Water Regulations, US-EPA, 2009 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |












