Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Recent progress in current and emerging techniques for the detection of PFAS – the forever chemicals
Vibhas
Chugh
a,
Paul
Gaskin
b and
Waye
Zhang
 *a
*a
aDepartment of Chemical Engineering, Swansea University, Swansea SA18EN, UK. E-mail: wei.zhang@swansea.ac.uk
bDwr Cymru Welsh Water, Newport, NP10 8FZ, UK
First published on 22nd January 2026
Abstract
Per- and polyfluoroalkyl substances (PFAS) are now regulated at ultra-trace levels in drinking water, with guideline values in the low ng L−1 (ppt) range in many jurisdictions, demanding highly sensitive, robust, and cost-effective monitoring tools. Regulatory drivers increasingly emphasize not only detection of legacy PFAS such as PFOS and PFOA, but also broader chemical coverage, routine compliance monitoring, and rapid screening at the point of use. This review critically surveys PFAS sensor technologies developed over the last decade, encompassing optical (colorimetric, fluorescence, and surface plasmon resonance), electrochemical (voltammetric, impedimetric, and potentiometric), and emerging biosensing and whole-cell reporter platforms. For each sensor class, typical limits of detection (from low ppb down to sub-ppt in optimized systems), dynamic ranges, regeneration, and compatibility with repeated measurements in real and complex water matrices are summarized. The underlying recognition and transduction principles—including molecularly imprinted polymers, host–guest interactions, ion-selective membranes, nanomaterial-enhanced interfaces, and biological recognition elements—are highlighted to connect materials design with analytical performance. Across these platforms, key advantages include miniaturization, rapid response, and potential integration into portable or on-line monitoring systems, whereas major limitations involve selectivity among structurally similar PFAS, matrix interferences, long-term stability, and limited multi-analyte capability. This review discusses how current research addresses these challenges through preconcentration strategies, sensor arrays, nanostructured materials, and integrated sample handling, and outlines future directions toward regulatory-grade, field-deployable PFAS sensors capable of continuous monitoring, multiplex detection, and scalable deployment in drinking water and environmental surveillance.
1. Introduction
Per- and polyfluoroalkyl substances are a diverse class of anthropogenic chemicals, characterized by aliphatic carbon chains (CnF2n+1), where hydrogen atoms are substituted by fluorine atoms.1 These compounds exhibit varying chain lengths and functional groups, including carboxylates, sulfonates, sulfonamides, alcohols, and phosphonates. PFAS are typically classified based on the extent of fluorine substitution, with “perfluorinated” compounds (where all hydrogen atoms are replaced by fluorine) and “polyfluorinated” compounds (with selective substitution) being the two most hazardous categories2 (Fig. 1). The defining feature of PFAS is their strong carbon–fluorine (C–F) bond, which imparts exceptional chemical stability, making these substances highly resistant to thermal, photolytic, and biological degradation.3,4 This unique characteristic is responsible for their widespread use in a broad spectrum of industrial and consumer products, including non-stick coatings, water- and oil-repellent fabrics, firefighting foams (particularly aqueous film-forming foams, AFFFs), electronics, automotive applications, and in the production of certain pesticides and lubricants as depicted in Fig. 1.5,6 Estimates suggest that approximately 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 different PFAS compounds have been synthesized for various applications, driven by their ability to perform under extreme conditions such as high temperatures, exposure to harsh chemicals, and in environments with high mechanical stress.7,8 While their use has been invaluable in diverse sectors—including the military, aviation, and consumer goods industries—the persistence of PFAS in environmental matrices has become a significant concern. Due to their hydrophobic and lipophobic nature, PFAS are easily mobilized in the environment, and their resistance to degradation leads to ubiquitous contamination of water, soil, wildlife, and food sources.9
000 different PFAS compounds have been synthesized for various applications, driven by their ability to perform under extreme conditions such as high temperatures, exposure to harsh chemicals, and in environments with high mechanical stress.7,8 While their use has been invaluable in diverse sectors—including the military, aviation, and consumer goods industries—the persistence of PFAS in environmental matrices has become a significant concern. Due to their hydrophobic and lipophobic nature, PFAS are easily mobilized in the environment, and their resistance to degradation leads to ubiquitous contamination of water, soil, wildlife, and food sources.9
Among the various types of PFAS, long-chain compounds, such as perfluorooctanesulfonic acid (PFOS) and perfluorooctanoic acid (PFOA), characterized by their long-fluorinated carbon chain, are often referred to as “legacy PFAS” and are considered persistent organic pollutants (POPs).8,10 The chemical structures of PFOA and PFOS are shown in Fig. 1. These compounds are particularly concerning due to their toxicity, bioaccumulation potential, and long-term environmental persistence.11,12 The toxicity of PFAS, especially in relation to their endocrine-disrupting effects and carcinogenicity, has driven regulatory agencies to phase out or restrict the use of these chemicals, particularly in industrial applications and consumer products. In contrast, short-chain PFAS typically refer to compounds such as sulfonates with five or fewer carbon atoms, and carboxylates with seven or fewer carbon atoms. Short-chain PFAS have emerged as alternatives to legacy compounds, becoming increasingly prevalent due to regulatory bans on their longer-chain counterparts.13,14 However, their environmental behaviour and toxicity are still under investigation, as they are often more mobile and can more readily accumulate in living organisms.15,16 Additionally, while short-chain PFAS may exhibit similar functional properties to long-chain compounds, higher concentrations are often required to achieve equivalent performance, further complicating toxicity assessments and regulatory guidelines.17,18 Among the different categories of PFAS, two major subgroups: perfluorocarboxylic acids (PFCAs) and perfluorosulfonic acids (PFSAs) represent some of the most encountered PFAS in environmental contamination studies.19,20 The increasing recognition of their environmental and health risks has prompted calls for comprehensive monitoring, remediation strategies, and the development of cost-effective and efficient analytical methods for PFAS detection.21,22 However, a significant knowledge gap remains regarding the full scope of PFAS toxicity, particularly concerning novel, short-chain variants and their long-term ecological and human health implications.
PFAS contamination is also widespread in soils globally, often entering surface and groundwater through waste disposal, industrial activities, agricultural runoff, and PFAS-containing products. These compounds are primarily ingested by humans through contaminated food, water, skin contact, and inhalation.23–26 Their bio-accumulative nature, combined with exceptional environmental persistence, makes PFAS particularly concerning, as they do not break down over time and can accumulate in the human body and ecosystems for decades.27–29 Studies have shown that PFAS concentrations in some water sources exceed established safety thresholds, contributing to growing public health concerns.30 Given their widespread presence and persistence, PFAS are implicated in a variety of health risks. These include thyroid dysfunction, immune system suppression, liver toxicity, kidney disease, developmental delays, and an increased risk of cancers.31,32 Epidemiological studies have shown links between PFAS exposure and adverse health outcomes such as reduced vaccine efficacy, changes in liver enzymes, and elevated cholesterol levels.25,33,34 Additionally, wildlife, especially aquatic species, is severely affected. PFAS accumulate in fish, shellfish, and marine mammals, leading to trophic transfer through the food chain.4 For example, polar bears, seals, and dolphins exhibit high concentrations of PFAS in their tissues, further exacerbating ecological concerns.35,36 As aquatic ecosystems are increasingly contaminated, bioaccumulation of PFAS poses a long-term threat to biodiversity and ecosystem function.
While the extensive environmental presence of PFAS is alarming, these compounds remain indispensable in many industrial applications, such as fire suppression, manufacturing, and consumer products like non-stick cookware and waterproof textiles. Despite growing concerns, their widespread use continues due to their unique properties. This creates a complex regulatory challenge. The U.S. Environmental Protection Agency (USEPA) has set a health advisory limit of 70 ng L−1 for PFOS and PFOA in drinking water, yet the broader spectrum of PFAS compounds complicates regulation.37,38 New PFAS compounds, some of which are not well studied, can break down into even more harmful substances, further complicating monitoring efforts. Moreover, some newer PFAS alternatives have not been extensively evaluated for their environmental and health impacts.3 With increasing regulatory pressure and public concern about PFAS contamination, the need for robust and accessible analytical methods has become ardent. Many of the existing detection techniques, while accurate, have significant limitations that hinder their widespread adoption and application in environmental monitoring. Standard laboratory methods, such as high-performance liquid chromatography coupled with tandem mass spectrometry (HPLC-MS/MS), are widely regarded as the gold standard for PFAS detection due to their sensitivity, specificity, and ability to quantify a wide range of PFAS compounds.39,40 However, these techniques are expensive and require highly specialized equipment and skilled personnel, making them impractical for routine, large-scale environmental testing or use under field conditions.13,41 The complexity of the analysis, combined with the time-consuming nature of the process (which can take hours to days), limits the ability to quickly assess contamination in real time. In addition, these laboratory methods can struggle to detect newer, less-studied PFAS compounds or their degradation products, especially given the vast number of PFAS variants and the continuous development of new chemical formulations.42,43 In contrast, sensor technologies such as electrochemical, optical, and fluorescence-based sensors are portable and cost-effective, and provide real-time, in situ monitoring, allowing for immediate data collection without the need to send samples to a laboratory. This makes sensors ideal for large-scale, on-site environmental assessments, enabling quicker response times and better management of contamination.44,45 Despite the potential of sensor-based technologies for PFAS detection, challenges persist in ensuring their accuracy across the diverse range of PFAS compounds. The chemical variability, persistence, and interactions of PFAS with environmental matrices require sensors to be highly specific and sensitive to detect these pollutants under varying conditions.46,47
This study provides an in-depth evaluation of current detection methods, challenges faced in widespread PFAS identification, and the potential of emerging sensor technologies. By highlighting the latest advancements in real-time, field-based monitoring, this research aims to contribute to the development of more effective, accessible solutions for PFAS contamination assessment and regulatory compliance. Addressing the growing issue of PFAS contamination requires not only improved detection technologies but also a more comprehensive regulatory framework that considers the entire lifecycle of these pollutants and their ever-expanding array of chemical variants.
2. Global prevalence of PFAS
Regulations throughout the world have been moving in the past two decades in response to mounting evidence of PFAS toxicity, human exposure, and extensive pollution of water supplies. Only lately have PFAS been classified as pollutants of rising concern, even though they were initially manufactured in the 1940s.48 In developed countries as well as economies, potable water, surface water, and groundwater have been shown to contain pervasive forms of persistent organic pollutants PFAS, which are the biggest class of chemicals commonly utilized in commercial goods. They have been found in large quantities in human serum, air, rainfall, ocean water, organisms, and food. Human health and the presence of perfluoroalkyl carboxylates and sulfonates in diverse biological matrices and pelagic levels have been the subject of recent concern.49 Government organizations worldwide are finding it challenging to create health-protective regulations to address PFAS pollution due to the lack of complete information on the vast majority of PFAS contaminants. Many PFAS have lately had concentration restrictions imposed in North American and European nations.50 Several states in the United States have set their own health-based standards for PFOS, PFOA, and other PFAS in drinking water (ASTSWMO, 2015; USEPA, 2017). UK, Germany, Canada, Norway, Sweden and Australia are among the nations that have released administrative standards for the amounts of PFAS in water.51The US Senate has approved further PFAS regulation legislation. The resolution mandates that the Defense Department cease deploying AFFFs carrying PFAS on October 1, 2023. The existing drinking water restriction of 70 ng L−1 for PFOA and PFOS lacks an appropriate enforcement mechanism, and manufacturers have already gradually discontinued using these substances.52 On April 10, 2024, the USEPA finalized the National Primary Drinking Water Regulation (NPDWR) for six PFAS, including PFOA and PFOS, establishing the first national, legally enforceable maximum contaminant levels (MCLs) for these chemicals in drinking water. Public water systems were given until 2029 to comply with these regulations, which require both monitoring and treatment to reduce PFAS concentrations to or below the specified limits. These standards are anticipated to significantly reduce PFAS exposure, preventing thousands of deaths and tens of thousands of serious PFAS-attributable illnesses.53,54
The Environmental Working Group in the US commissioned laboratory testing in 2019 that revealed PFAS to be present in the drinking water of dozens of American communities with varying population numbers and geographical locations. The examination encompassed 30 PFAS chemicals, with detection levels varying from 0.3 to 2.0 ng L−1. Combined PFAS concentrations constituted the range lower than 1 ng L−1 to 185 ng L−1.55 Having been considered the overall stats, these data suggest that Americans are exposed to PFAS from tainted tap water far more than the USEPA testing protocol indicates. In another research study, one sample had no PFAS at all, and two others had aggregate PFAS levels of less than 1 ng L−1 out of 44 potable water samples across 31 states and districts of Columbia. All PFAS detected in twenty sites were below the quantification limit but over the testing facility limit of detection. Samples from significant cities, such as New York, contained a few of the most elevated PFAS levels found. The USEPA and state government organizations have not made reports of PFAS contamination in 34 locations public, where the research identified it.56
The Organization for Economic Co-operation and Development (OECD) conducted a preliminary study to find out the number of PFAS available in the worldwide market to discover which ones are utilized in the EU and the UK. Between January 2017 and February 2018, this record was compiled, containing 4730 PFAS that were utilized in the worldwide market.57 77 of these PFAS have been registered for use in accordance with the REACH Regulation. REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) is the European Union regulation that governs the management and restriction of chemical substances. Any chemical produced or brought into the EU that weighs more than one tonne annually must register under the REACH law. As a result, it was decided that the PFAS listed in REACH dossiers had the greatest bearing on Europe and, consequently, Wales and England.58 Five of the 77 PFAS were duplicates as they belonged to various bands. As a result, the OECD (2018a) identified a final list of 72 PFAS that are utilized, manufactured, imported, or exported inside Europe. Overall, the information gathered from the literature suggests that it is improbable that any one of the PFAS will be present in drinking water at concentrations higher than 0.1 μg L (100 ng L−1). Based on simulated data, it can be concluded that most PFAS might not be expected to surpass a drinking water threshold of 0.1 μg L−1 if a norm were to be imposed for any one PFAS. The framework does, however, still need verification, and for those PFAS that are expected to be above the potable water threshold, it could be prudent to conduct targeted assessments of drinking water close to PFAS emission sites.57
The European Commission (EC) has designated PFOS and its derivatives as highly perilous compounds, and PFAS as emerging organic pollutants. To evaluate the quality of the environment, the EC set a threshold concentration for per- and polyfluoro caustic soda in drinking water and fish through the Water Framework Directive in 2012. Additionally, in 2013, the EC defined the Environmental Quality Standards to gauge the concentrations of PFOS in water bodies and biological communities. The average yearly concentration limit for water bodies is 0.65 ng L−1, while the highest permissible concentration is 36 μg kg−1 and 9.1 μg kg−1 for biological systems.59 According to Janousek et al. investigation of time-varying and general trends in the prevalence of PFAS, the highest levels and recognition frequency of PFOA decreased over the research period, but the rate of short-chained PFAS identification rose, particularly after 2014.21 Significant levels of PFAS have been documented in aquatic life, debris, and water of the Spanish waterways of the Guadalquivir and Ebro. The highest amount of PFAS detected in water samples from these locations ranged between 251.3 ng L−1 and 742.9 ng L−1.60 According to Llorca et al., PFAS were found in 54% of tap water samples, with values ranging from 2.4–27 ng L−1 in 32 German and Spanish towns. Eighty-eight percent of the 148 water samples that had their 21 PFAS content measured had a minimum of one detectable quantity of the chemical.61
Talking about the prevalence of PFAS in Asian countries, Tan et al. (2017) measured the amounts of 17 PFAS in water from large lakes and rivers that supply potable water to Eastern China.62 64 samples from cities, 51 water samples from rivers, and 45 samples from lakes were examined. The overall amount of PFAS in the tap water varied from 1.4 to 175 ng L−1, but the most concerning finding was that the drinking water quality threshold was surpassed by the PFOA levels (115–151 ng L−1) discovered in the potable water supply of 3 districts in Hangzhou. Pan et al. investigated the fate of 18 PFAS in the cycle of urban water in water treatment plants (WWTPs) and drinking water treatment plants (DWTPs) in Guangzhou.63 The total PFAS concentrations in the DWTPs ranged from 3.34 to 13.9 ng L−1 in effluents. As such, short-term interaction with PFAS discovered in potable water did not immediately endanger the well-being of humans. Other research studies have also been carried out to learn more about the prevalence, origins, and concentrations of PFAS from important drinking water sources in Tianjin,64 Fuxin,65 and Beijing.66
Takagi et al. (2011) assessed the concentrations of PFOA and PFOS in water samples taken from multiple Osaka-based water purification facilities that were employing cutting-edge water treatment technology at each stage of the drinking water treatment procedures. In all completed water tests, both PFAS were found at concentrations ranging from 1.3 to 3.7 ng L−1 for PFOS and from 6.5 to 48 ng L−1 for PFOA.67 Sharma et al. evaluated 21 PFAS forms of prevalence in drinking water reservoirs used by the local people in different Ganges River areas in India. All samples had detectable levels of perfluorohexanoic acid (PFHxA) and perfluoroheptanoic acid (PFHpA), which ranged from 0.8 to 4.9 ng L−1 and 0.5 to 3.5 ng L−1, respectively.68 Thompson et al. assessed the amount of PFAAs that are consumed daily in relation to potable water. At thirty-four regions, 62 samples were taken straight from the potable water taps. According to estimates, drinking water contributed an average of 2–3% of PFOS and PFOA, with highest amounts of 22% and 24%, respectively.69
Authorities worldwide should create more firm regulations, establish PFAS as a class rather than as distinct compounds, and limit the use of PFAS to essential applications while encouraging their phase-out, considering the lack of data about the adverse consequences of short-chain PFAS exposition.70 Furthermore, there is a great deal of ambiguity around PFAS monitoring because of several variables, such as source variability and sample location selection. Because there is currently a dearth of solid, trustworthy, and repeatable evidence, these substances are not controlled.
3. Overview of existing technologies
The detection and quantification of per- and polyfluoroalkyl substances (PFAS) present significant challenges due to their chemical diversity, persistence in environmental media, and the extremely low concentrations at which they typically occur, ranging from picograms to micrograms per litre in water, air, and soil. Effective identification of these compounds necessitates highly sensitive and reliable analytical techniques, capable of distinguishing PFAS from other environmental contaminants while also accounting for their varying chemical properties.71,72 A key characteristic of an effective detection system is its ability to provide accurate, reproducible results across a wide range of environmental matrices, while maintaining low detection limits and a broad dynamic range.Chromatographic methods, when coupled with mass spectrometry (MS), are currently the most used techniques for PFAS analysis due to their high sensitivity and selectivity. The liquid chromatography-tandem mass spectrometry (LC-MS/MS) method allows for the separation and identification of individual PFAS compounds within complex environmental samples, offering precise quantification even at trace levels. Solid-phase extraction (SPE) is often employed in conjunction with LC-MS/MS to concentrate PFAS from water samples and improve the sensitivity of detection.73,74 In addition, gas chromatography-mass spectrometry (GC-MS) is a frequently applied technique, particularly for volatile PFAS, although it is generally less effective for non-volatile, high-molecular-weight compounds.75 These chromatographic methods, while highly effective, require significant sample preparation, skilled personnel, and advanced instrumentation, which can limit their applicability for routine environmental monitoring, particularly in resource-limited settings.40,76
The prevailing gold standard for the detection of PFAS relies on mass spectrometry in conjunction with chromatographic techniques. In 2009, the US EPA released “Method 537,” an official analytical procedure that uses SPE to allow LC-MS/MS to assess potable water for 14 distinct PFAS, including PFOA and PFOS.77 The EPA released “Method 533,” an enhanced analytical technique in November 2019 that places greater emphasis on the identification of “short chain” PFAS. To increase the quality and specificity of the data collected, Method 533 makes use of isotope dilution anion exchange solid phase extraction and MS in multiple reaction monitoring (MRM) mode.78 These methods provide reliable data for regulatory compliance, but are time-consuming and costly, making them unsuitable for widespread, real-time monitoring.
In addition to the development of new detection methods, there has been growing interest in surrogate techniques that focus on the total PFAS contamination, rather than individual compounds. Methods such as liquid–liquid extraction (LLE),79 ion-pair extraction (IPE),80 and solid-phase microextraction (SPME)74 have been employed to capture a broader range of PFAS, including transformation products and other related compounds. These techniques, while effective in detecting total PFAS content, do not provide specific information about individual compounds, which can limit their utility for targeted risk assessments and regulatory compliance.81 While current analytical techniques such as LC-MS/MS remain the most accurate and reliable methods for PFAS detection, their complexity, high costs, and laboratory dependency hinder their widespread application. Sensor-based systems, offer promising solutions for real-time, field-based monitoring of PFAS. However, challenges related to sensitivity, specificity, and environmental interference of sensors are being addressed before these technologies can fully replace traditional methods for PFAS analysis. Continued research and development are ongoing to improve sensor performance, reduce costs, and enhance the ability of these systems to detect a broader range of PFAS compounds in diverse environmental matrices.
4. Sensors for PFAS detection
As the awareness surrounding the environmental and public health risks associated with PFAS continues to rise, there is an increasing imperative to develop efficient, rapid, and cost-effective detection methods. Traditional analytical techniques, such as mass spectrometry coupled with chromatographic separation, remain the reference standard for PFAS detection. However, these methods are hampered by their high operational costs, the need for highly trained personnel, time-consuming sample preparation, and reliance on laboratory infrastructure, limiting their applicability for large-scale, on-site, and real-time monitoring.82,83 Therefore, there is a pressing need for the advancement of sensor-based technologies that can provide rapid, portable, and sensitive detection of PFAS in a variety of environmental matrices, particularly water.The development of effective PFAS sensors faces several scientific and technical challenges. First, many of the current sensors exhibit detection limits that are insufficient to meet the ultra-low concentrations of PFAS mandated by regulatory agencies, especially in environmental samples that require high sensitivity (sub-ppt or femtomolar range).84 Furthermore, interference from various environmental factors—such as particulate matter, dissolved organic compounds, and salts—can degrade the performance of sensors, making it difficult to achieve reliable measurements in complex matrices. Addressing these challenges requires the development of new sensor materials and recognition elements that offer enhanced selectivity and sensitivity. Nanomaterials, such as carbon nanotubes, graphene, and metal–organic frameworks, have shown great promise for enhancing the performance of sensors by providing increased surface area for adsorption and improving the stability and reproducibility of sensor responses.85
PFAS compounds vary widely in terms of chain length, functional groups, and hydrophobicity, which complicates the development of a single detection method that can simultaneously recognize all relevant PFAS species.86 As a result, there is an increasing need for multi-functional or multi-modal sensor platforms that can detect various PFAS subclasses such as long-chain versus short-chain compounds, or those with different functional groups. In addition, for sensors to be deployed in real-world applications, they must be designed with robustness, durability, and ease of integration into existing monitoring frameworks.87 It is essential to ensure that sensor systems can be operated in a range of environmental conditions, such as varying pH, temperature, and ionic strength, while maintaining high levels of accuracy and reliability. Sensors also need to demonstrate long-term stability and reproducibility, ensuring consistent performance over time, especially when used for routine monitoring in diverse settings.82,88–90 The integration of sensors into environmental monitoring programs, water quality management systems, and public health assessments will not only provide more frequent and accurate data on PFAS contamination but will also facilitate the early identification of PFAS hotspots, enabling timely intervention and remediation strategies. The successful commercialization of PFAS sensors will have a transformative impact on the way we monitor and manage PFAS pollution, ultimately protecting human health and the environment from the pervasive and persistent threats posed by these compounds. Here, we overview the state-of-the-art PFAS detection assays and sensors, based on their detection mechanisms.
4.1 Optical sensors
Cutting-edge optical sensors, such as surface plasmon resonance (SPR), surface-enhanced Raman scattering (SERS) and fluorescence-based methods, are emerging as powerful tools for the detection of PFAS. These sensors detect changes in refractive indices or fluorescence intensities upon PFAS binding to functionalized sensor surfaces, providing label-free and non-invasive detection.91,92 While these techniques offer the advantage of direct, real-time monitoring, they typically struggle to achieve the required sensitivity for ultra-trace PFAS detection in environmental matrices.93 The major limitation of optical sensors lies in their susceptibility to environmental interference, such as turbidity, dissolved organic matter, and high ionic strength, which can significantly compromise the accuracy and reproducibility of the measurements.94 Furthermore, optical methods often lack the specificity needed to differentiate PFAS from structurally similar organic compounds, which limits their applicability for complex sample types.A recent study by Park et al. introduced a SERS-based sensor combining self-assembled polydiacetylene (SAp-PD) for the ultra-sensitive detection of PFOA.95 Detection relied on SERS intensity changes caused by SAp-PD conformational disruption upon binding to PFOA. The sensor achieved a low detection limit of 1.28 pM in distilled water and showed high selectivity against other fluorinated compounds. Practical applicability was demonstrated by detecting 1.69 nM PFOA in extracts from rice cooked on a damaged, PFOA-coated frying pan. This approach shows strong potential for early-stage detection of perfluorinated compounds in real-world samples. A SERS-based method using optimized Au@Ag nanorods enabling rapid, sensitive PFAS detection with a limit of 0.1 ppm was developed by Feng et al.96 The sandwich structure with AgNPs enhanced the SERS signal 3.6-fold over a monolayer, allowing dynamic capture of PFAS at hot spots. The assay, compatible with portable Raman devices, delivers results in under 30 minutes. While in situ detection is limited, the sensor shows strong potential for fast, field-based PFAS monitoring.
A fluorescent sensing array developed by Chen and colleagues enables rapid screening and differentiation of six distinct PFAS types in water.97 This array utilizes three highly stable zirconium porphyrinic metal–organic frameworks (MOFs), known as PCNs, each exhibiting different topological structures. The sensing principle is based on the static fluorescence quenching of PCNs by PFAS molecules upon adsorption. Because each PFAS interacts with the PCNs with varying affinities, their distinct fluorescence patterns serve as a basis for differentiation. Notably, the organized pore structure of the PCN sensors facilitates quick diffusion, resulting in a rapid reaction time—less than 10 seconds for PFAS detection. In a study by Dalapati et al., a fluorescent PDI-based MOF (U-1) was developed for PFOA detection via a fluorescence turn-on response.98 U-1 showed high chemical stability and selectivity for PFOA in both solution and solid phases. When deposited on filter paper, the MOF achieved a detection limit of 3.1 nM, aided by solid-phase extraction. The sensing mechanism involves complexation between the carboxylic group of PFOA and zirconium centres, along with hydrophobic interactions with the PDI ligand. This work highlights the promise of PDI-based MOFs for selective PFAS detection, with potential for further tuning via ligand and pore design. In another study, Niu et al. introduced a novel sensing method that employs poly(ethylene glycol)-terminated (PEG-thiols) and perfluoroalkyl-terminated (F-thiols) alkanethiol-modified gold nanoparticles (Au@PEG-F NPs) to detect perfluorinated chemicals (PFCs) in water samples.99 The PEG-thiols ensure that the gold nanoparticles remain well-dispersed and stable in solution, while the F-thiols promote strong fluorous–fluorous interactions, which cause PFCs to adsorb onto the nanoparticles. Combined with UV-vis spectrophotometry, the system demonstrates a broad linear detection range and is sensitive even at concentrations as low as 10 μg L−1, making it an effective and affordable technique for quantifying PFCs in environmental samples.
Amplifying fluorescent polymers (AFPs), which are highly fluorinated polymers based on poly(p-phenylene ethynylene) and polyfluorene backbones, incorporate pyridine selectors that undergo proton-transfer reactions with PFAS, resulting in red-shifted fluorescence due to exciton trapping. Concellon et al. developed AFPs for selective detection of aqueous PFOA and PFOS at low concentrations, reaching ng L−1 levels. AFP films detected PFAS at ∼1 ppb, while nanoparticle forms achieved ∼100 ppt sensitivity. Notably, the performance was consistent across various water types, including DI, MilliQ, and well water. Although further refinement is needed to meet regulatory limits, this platform shows strong potential for on-site PFAS detection in contaminated environments.100
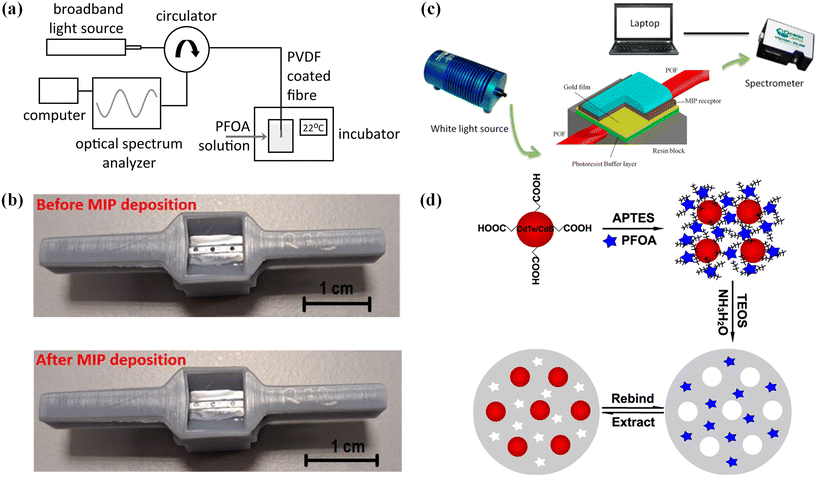
Faiz et al. explored a simple yet effective sensor fabrication technique using a liquid precipitation-based phase inversion method to deposit a thin polyvinylidene fluoride (PVDF) layer on freshly cut optical fibers101 (Fig. 2a). The β-phase of PVDF, which is electrically charged, enhances hydrophobic and dipole–dipole interactions with PFAS, particularly at binding sites. This sensor was able to detect PFOA with sensitivity measured through changes in the optical path difference, yielding a response range of 0.9–5 nm ppm−1 for PFOA concentrations. In a similar vein, Pitruzzella et al. designed an optical fibre sensor intended for ultra-low detection of PFOA in aqueous environments102 (Fig. 2b). This system integrates molecularly imprinted polymers (MIPs) and surface plasmon resonance (SPR) technologies, using a plastic optical fibre (POF) as the platform. The PFOA–MIP interaction within the fibre's micro-holes causes a shift in SPR phenomena, enabling the sensor to detect PFOA with high sensitivity. Cennamo et al. later built on this work, developing an SPR–POF–MIP sensor to enhance PFAS detection using MIPs as a receptor material103 (Fig. 2c). The MIP receptors, which can be easily deposited on a gold layer, provide a stable, repeatable, and cost-effective solution for PFAS detection.
Another promising method for PFAS detection involves carbon quantum dots (CQDs), as explored by multiple research teams. Jiao et al. and Walekar et al. utilized MIPs and nanoparticle-doped quantum dots (QDs) as fluorescence probes for PFAS detection.104,105 Zheng et al. advanced this approach by creating a photoluminescence sensor based on core–shell QDs coated with an MIP shell, specifically designed for PFOA detection.106 The QD–MIP composite showed both high specificity and stable photoluminescence, with PFOA molecules effectively quenching the fluorescence. This sensor demonstrated excellent linearity (0.25–15.00 μmol L−1) and a limit of detection (LOD) of 25 nmol L−1 (∼10 ppb), proving its reliability for detecting trace amounts of PFOA in environmental water samples. Chen et al. further investigated the interaction between carbon dots and PFOS, revealing that they form a ground-state complex that alters fluorescence, resonant light scattering (RLS), and UV-vis absorption signals107 (Fig. 2d). These signal changes correlate directly with PFOS concentration, providing a highly sensitive detection method with a fluorescence LOD of 18.27 nmol L−1. Building on this, the research group employed a dual-recognition technique using carbon dots and ethidium bromide for ratiometric sensing of PFOS.108 In a similar study, the researchers proposed a highly specific fluorescence-based detection method for PFOS using carbon dots and berberine chloride hydrate (BH).109 In this system, the addition of PFOS caused a noticeable fluorescence shift from blue to light yellow, allowing for clear visual detection of PFOS with high specificity. A summary of recent development in optical sensors for detection of different types of PFAS is presented in Table 1.
| Type of PFAS detected | Electrode material/matrix | Detection mechanism | LOD | Linear range | References |
|---|---|---|---|---|---|
| PFHxA – perfluorohexanoic acid; PFHpA – perfluoroheptanoic acid; PFNA – perfluorononanoic acid; PFDA – perfluorodecanoic acid; PFC – perfluorinated compounds; MOF – metal organic framework; PEG – polyethylene glycol; POF – plastic optical fiber; QD – quantum dot; NPs – nanoparticles. | |||||
| PFHxA (C-6), PFHpA (C-7), PFOA (C-8), PFNA (C-9), PFDA (C-10), PFOS (C-8) | Zirconium porphyrinic-based MOF | Static fluorescence quenching of MOFs by PFAS upon their adsorptive interactions | 10–8 mol L–1 (0.0041 ppb) | 0.1–2 μg mL–1 for PFHpA and PFNA; 0.1–1 μg mL–1 for PFDA and PFOS | 97 |
| PFCs | PEG-thiols + F-thiols + gold nanoparticles | Insolubility of Au@PEG-F NPs and precipitation due to the superhydrophobicity of perfluorocarbon monolayers, leading to color and absorbance response | 10 μg L–1 (10 ppb) | 0.011–13.8 μmol L–1 | 110 |
| PFOA (C-8) | Polyvinylidene fluoride coated optical fibres | Dipole–dipole and hydrophobic interaction | 178 ppb | — | 111 |
| PFOA (C-8) | D-shaped POF + MIP | Variation in the plasmonic response that occurs in a multimode POF | 0.81 ppt | — | 102 |
| PFOA, PFOS (C-8) | D-shaped POF + photoresist buffer layer + gold film | Molecular recognition | 0.13 ppb | — | 103 |
| PFOA (C-8) | D-shaped POF + MIP | Ionic interaction and van der Waals | 0.5 ppb | — | 112 |
| PFOA (C-8) | Graphitic carbon nitride nanosheets + MIP | Electrochemiluminescence | 0.01 ng m L−1 (0.01 ppb) | 0.02 to 40.0 ng mL−1 | 113 |
| PFOS (C-8) | MIP + chitosan + carbon QDs | The sulfonate group of PFOS can form a complex with the amino group through hydrogen bonding or electrostatic reaction, which can enhance the conjugation degree of H2N-passivated CQDs | 85 pg·L−1 (0.085 ppb) | 20–200 pg L−1 | 114 |
| PFOA (C-8) | Core–shell QDs + MIP | Photoluminescence | 25 nmol L–1 (10.4 ppb) | 0.25–15.00 μmol L–1 | 115 |
| PFOA (C-8) | Carbon QDs + selenium + nitrogen | Fluorescence quenching | 1.8 μM (745 ppb) | 10–70 μM | 116 |
| PFOS (C-8) | Carbon QDs | Triple channel fluorescence | 18.27 nmol L–1 (7.6 ppb) | 0.5–12 μmol L–1 | 107 |
| PFOS (C-8) | Nitrogen doped carbon QDs + ethidium bromide | Fluorescence and second-order scattering | 27.8 nM (11.5 ppb) | 0–2.0 μM | 108 |
| PFOA (C-8) | Cadmium sulphide QDs | Fluorescence quenching | 0.3 μmol L−1 (1 ppb) | 0.5 to 40 μmol L−1 | 117 |
| PFOS (C-8) | Carbon dots + berberine chloride hydrate | Fluorescence quenching | 21.7 nmol L–1 (9 ppb) | 0.5–40 μmol L–1 | 109 |
| PFOS (C-8) | SiO2 NPs | Acid–base pairing and hydrogen-bond interaction | 5.57 μg L−1 (5.57 ppb) | 5.57–48.54 μg L−1 | 118 |
| PFOA (C-8) | Polystyrene + gold NPs | Interparticle aggregation leading to colour change | 100 ppm | — | 119 |
| PFOA (C-8) | Perfluorosilane-functionalized nano-porous anodic alumina | Fluorous interaction induced Freundlich mechanism | 0.53 μg m L–1 (530 ppb) | — | 120 |
4.2 Biosensors
Biosensors offer a promising approach for PFAS detection by utilizing biological recognition elements, such as antibodies, enzymes, or aptamers, to selectively interact with PFAS molecules. These sensors convert the binding event into a measurable signal, providing high specificity even in complex matrices.121,122 Aptamer-based and antibody-based sensors are particularly noted for their ability to detect specific PFAS compounds with high affinity. One key advantage of biosensors is their potential for miniaturization and portability, allowing for on-site, real-time detection. These sensors typically offer quick response times and low LODs in ppb and ppt ranges, enabling field-deployable monitoring.123 However, challenges remain in terms of the stability and reproducibility of the biological components, as well as environmental factors that may impact sensor performance. Bioreceptors in biosensors demonstrate inadequate long-term stability owing to denaturation of recognition biomolecules, pronounced sensitivity to environmental variables including temperature excursions and pH alterations, and significant matrix effects from heterogeneous samples that induce measurement errors.124 Despite these limitations, biosensors are a rapidly evolving field with significant potential for advancing PFAS monitoring.To monitor the presence of PFOA/PFOS in seawater, Cennamo et al. developed, implemented, and evaluated an innovative biosensing system that utilized an optical fibre in conjunction with a proprietary antibody as a bio-receptor (Fig. 3c). One key advantage of this method is its ability to monitor perfluorinated chemicals in natural environments by leveraging the remote sensing capabilities of optical fibres. The system demonstrated both high selectivity and a low detection limit for PFOA/PFOS, achieving sensitivity levels below 0.21 ppb in seawater.125
In terms of molecular recognition, it is more likely that the perfluoroalkyl chain, rather than the terminal functional group, plays a primary role in the binding interaction between PFOA and the aptamer. Given the wide variety of fluorinated contaminants and persistent flame-retardant substances within the PFAS class—differing in alkyl chain lengths, functional groups, and degrees of fluorination—there remains a pressing need for further research to develop aptamers with high specificity for distinct PFAS structures. Addressing this, Park et al. demonstrated that a fluorescence-based aptasensor can serve as a promising tool for PFAS detection (Fig. 3d).126 They developed a sensor by isolating ssDNA aptamers with high affinity for PFAS compounds, enabling the fluorescence-based identification of PFOA. Their work highlights the potential of aptamer-based biosensors for the rapid, sensitive, and selective detection of PFAS and related fluorinated substances in environmental samples.
Peroxisome proliferator-activated receptor alpha (PPARα) is a nuclear receptor and transcription factor that plays a central role in the regulation of lipid metabolism, energy homeostasis, and inflammation. It is predominantly expressed in tissues with high fatty acid oxidation activity, such as the liver, heart, kidneys, and muscle. Many PFAS compounds can bind to and activate PPARα, leading to disruptions in lipid metabolism and other hepatic functions. Screening for PFAS that interact with PPARα is important because the binding affinity not only helps assess the potential metabolic toxicity of PFAS but also provides a mechanism-based biomarker for detection and risk assessment. Several types of PPARα ligands in the environment, including PFOS and PFOA, pose significant risks to human health. One promising method for screening PPARα ligands in ecological matrices involves a bioassay using modified nanogold probes that are sensitive to PPARα-responsive components (Fig. 3a).127 This bioassay is highly advantageous due to its exceptional sensitivity and broad detection range, making it particularly useful for identifying a variety of previously unidentified specimens. For cases where further analysis is required to determine the concentration of a specific compound, positive samples can be followed up with LC-MS (liquid chromatography-mass spectrometry) analysis.
When PFOS binds to PPARα, it can interact with PPAR response elements (PPREs) and form heterodimers with retinoid X receptors (RXRs). Zhang et al. developed a bioassay that utilizes immobilized PFOS on ELISA plates to compete with free PFOS in water samples for a fixed amount of PPARα–RXRα (Fig. 3b).128 The complex formed by PPARα–RXRα and PFOS can be indirectly quantified by binding it to a secondary plate coated with a PPARα antibody. The amount of PPARα–RXRα complex is then measured using biotin-modified PPARα–RXRα probes, quantum dots, and a streptavidin detection system. This method provides a sensitive and reliable means to assess PFOS binding and its potential to activate PPARα in environmental samples.
The immobilization of biomolecules onto screen-printed electrodes presents a persistent challenge in biosensing technologies. To address this issue, Moro et al. functionalized graphite-based screen-printed electrodes (G-SPEs) by electro-polymerizing pyrrole-2-carboxylic acid (Py-2-COOH), a pyrrole derivative containing multiple carboxyl functional groups, thus enhancing the electrode's bio-interfacing capabilities.129 In a proof-of-concept study, delipidated human serum albumin (hSA) was employed as a bio-receptor for the development of an impedimetric sensor targeting PFOA. Preliminary data from this work underscore the potential of this electrochemical biosensing platform for high-throughput analytical applications and toxicological screening, suggesting avenues for further optimization and integration into environmental monitoring assays. A summary of recent development in biosensors for detection of different types of PFAS is presented in Table 2.
| Type of PFAS detected | Electrode material/matrix | Detection mechanism | LOD | Linear range | References |
|---|---|---|---|---|---|
| PPARα – peroxisomal proliferator-activated receptor-α; MEHP – mono-(2-ethylhexyl) phthalate; ssDNA – single stranded deoxyribose nucleic acid. | |||||
| PFOS, PFOA (C-8) | Regulatory and response genes | Genetically engineered bacteria | 10–1000 ng L–1 (10–1000 ppt) | — | 130 |
| PFOA (C-8) | Graphite screen printed electrodes + pyrrole-2-carboxilic acid | Covalent immobilization of biomolecular recognition layers | — | — | 129 |
| PFOA (C-8) | Human liver fatty acid binding protein + circularly permuted green fluorescent protein | Electrostatic interactions and fluorescence | 236–330 ppb | — | 131 |
| PFOS, PFOA, (C-8) MEHP | PPARα + gold nanoparticles | Ligand–receptor interaction | 10 pM (1 ppb) | 100 pM to 1 μM | 127 |
| PFOS (C-8) | PPARα + quantum dots | Ligand–receptor interaction | 2.5 ng L−1 (0.0025 ppb) | 2.5–75 ng L−1 | 132 |
| PFOA (C-8) | ssDNA aptamers | Aptamer folding | 0.17 μM (70.4 ppb) | 0–75 μM | 133 |
| PFOS (C-8) | Multi-walled carbon nanohorns + glassy carbon electrodes | Inhibition influence | 1.6 nmol L–1 (0.8 ppb) | 5 to 500 nmol L–1 | 134 |
| PFOS, PFOA (C-8) | Ad hoc antibody + optical fibre | Bio-receptor | 0.2 ppb | — | 125 |
4.3 Electrochemical sensors
Electrochemical sensors, which rely on the measurement of changes in electrical properties (current, potential, or impedance) upon the interaction of PFAS with electrode surfaces, represent another promising avenue for on-site PFAS detection. These sensors are particularly advantageous due to their rapid response time, simplicity, and portability, making them ideal candidates for field deployment.135–137 The most employed electrochemical platforms for PFAS detection utilize MIPs as recognition elements, capitalizing on their affinity for specific PFAS molecules.138 However, the challenge remains in addressing the selectivity and sensitivity issues that arise from interference by other co-existing substances, such as metal ions, organic matter, and other environmental pollutants.139 To improve performance, electrochemical sensors may be coupled with additional pre-concentration steps, such as SPE, which enhances the sensor's sensitivity by concentrating PFAS from complex samples prior to detection.46,47,86,140 Nevertheless, despite the advantages of electrochemical sensors, they still fall short of meeting the stringent detection limits required for regulatory compliance but are the most promising for PFAS detection.To detect PFAS at sub-nanomolar concentrations, next-generation sensors are essential. The electrochemical properties of PFAS species offer a scalable platform for the detection of analytes at concentrations below 10 nM. A promising advancement in this field is the surface modification of electrodes using MIPs, which has garnered considerable interest. Clark et al. demonstrated the application of MIP-based carbon electrode sensors for the quantification of PFOS in river water141 (Fig. 4a). In their study, differential pulse voltammetry (DPV), the gold standard for electrochemical measurements, was initially employed for detection. However, their results also highlighted the advantages of electrochemical impedance spectroscopy (EIS) over traditional voltammetry. Notably, the authors demonstrated that EIS outperformed DPV in terms of sensitivity and reduced measurement uncertainty. EIS effectively mitigated the influence of liquid resistance, which is particularly relevant in complex matrices such as river water, thereby enhancing the reliability of PFOS detection.
Karimian et al. developed a highly sensitive electrochemical sensor for trace-level detection of PFOS by electrodepositing a MIP onto the surface of gold electrodes (Fig. 4d). The sensor leverages the principle of competitive binding, where PFOS, despite lacking a net electrical charge, competes with the electroactive probe ferrocenecarboxylic acid for the MIP's molecular recognition sites.142
Similarly, Glasscott et al. introduced a MIP-modified microelectrode sensor for the quantification of GenX, a widely distributed PFAS contaminant (Fig. 4c).143 To create specific molecular recognition sites, an o-phenylenediamine layer was electro-polymerized on the electrode surface, followed by solvent extraction to generate GenX-specific binding cavities. Their sensor exhibited a linear decrease in current with increasing concentrations of GenX, with ferrocene methanol oxidation serving as the electrochemical indicator. This system also incorporated oxygen reduction as a secondary sensing mechanism, demonstrating its capability to operate in moderately resistive ecological matrices. This makes the MIP-based microelectrode sensor a promising tool for the in situ detection of PFAS in environmental water. Most studies employ ortho-phenylenediamine (o-PD) for MIP polymerization networks due to its facile electropolymerization forming thin, uniform, stable films with precise imprinted cavities, excellent selectivity, mechanical strength, and pH/temperature tolerance—cost-effective for PFAS sensors. Other monomers include pyrrole, aniline, methacrylic acid (MAA), acrylamide (AAM), and EDOT derivatives, though o-PD dominates for electrochemical reproducibility.
Lu et al. introduced an ultra-sensitive electrochemical sensor for PFOS detection, which combines a glassy carbon electrode modified with a thin layer of gold nanostars and an electropolymerized MIP (Fig. 4e).144 The incorporation of gold nanostars enhanced the voltammetric response due to improved electrical conductivity and catalytic activity, particularly during the oxidation of ferrocenecarboxylic acid, a redox probe. The MIP layer was finely tuned to optimize sensor performance, allowing for enhanced sensitivity. However, interference from PFBA and PFBS was observed, leading to an approximate 10% underestimation of their concentrations. Despite this, the sensor showed robust performance for PFOS detection, with potential for the initial screening of unknown samples and the ability to offset interference effects in complex PFAS mixtures. Hafeez et al. developed a redox-active MIP platform based on electropolymerized EDOT–TEMPO on a glassy carbon electrode, exhibiting selective binding to PFOA via charge-assisted hydrogen bonding, which suppressed the redox activity of the TEMPO moieties. EDOT–TEMPO is a monomer consisting of a 3,4-ethylenedioxythiophene (EDOT) core modified with a stable 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) radical group that aids in electropolymerization. This suppression led to a concentration-dependent decrease in current density, enabling sensitive quantification with a detection limit of 0.28 ng L−1 and high selectivity against other PFAS.145 Notably, Amin et al. demonstrated AC electrokinetic (ACET) acceleration with a MIP on gold interdigitated electrodes, achieving ultra-sensitive PFOS detection (0.5 fg L−1 LOD in 10 s) via capacitance changes during selective binding, with 105![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 selectivity over PFOA. This work highlights ACET's role in enhancing enrichment and response speed, which is required for field-deployable PFAS monitoring.146
1 selectivity over PFOA. This work highlights ACET's role in enhancing enrichment and response speed, which is required for field-deployable PFAS monitoring.146
In another advancement, Solís et al. developed an electrochemical sensor utilizing a self-assembled monolayer of perfluorodecanethiol (PFDT) on gold nanoparticles deposited on glassy carbon electrodes for the selective detection of PFOA.147 The sensor demonstrated exceptional sensitivity, detecting PFOA at concentrations as low as parts per trillion (ppt). The electrochemical response was stable and reproducible, even in the presence of other perfluorinated compounds (PFCAs) and PFOS. Nanocomposites like MXene–AgNPs have shown strong potential as electrochemical sensors for PFAS detection using EIS. These materials selectively bind long-chain PFAS, inducing concentration-dependent changes in charge-transfer resistance, enabling quantification at parts-per-quadrillion (ppq) levels. Khan et al. demonstrated a high specificity sensor, with no interference from structurally similar compounds or common water constituents. Compared to conventional LC-MS/MS techniques, their platform offers a rapid (∼5 min), low-cost, and portable alternative for on-site PFAS screening.148 While MXene–AuNP sensors achieve ultra-low LODs, their linear ranges typically cover wastewater-relevant PFAS levels (ppt–ppb) but may saturate at highly contaminated sites (for e.g. >10 ppb), limiting their utility without dilution, highlighting the need for sensors with a low detection range.
Cheng et al. developed an integrated strategy for the tailored affinity-based capture of PFOS by incorporating permeable sorbent probes into a microfluidic device, significantly enhancing detection sensitivity.149 The system operates as an electrochemical sensor, providing direct measurement of PFOS concentration through a proportional change in current. The authors employed chromium-based MOFs to selectively capture PFOS, exploiting the chromium's strong affinity for both the fluorinated terminal groups and the sulfonate moiety (Fig. 4b). These MOF-based capture probes were strategically positioned between interdigitated microelectrodes within the microfluidic channel, addressing the need for an ultra-sensitive PFOS detection method suitable for real-time and in situ analysis. Another approach by Tian et al. utilizing a fluorine-functionalized Ce-UiO-66 MOF on a glassy carbon electrode (GCE) was developed for selective PFOA detection in water.150 The modified electrode leveraged electrostatic, anion–π, and fluorophilic interactions to enhance affinity toward PFOA, which blocked the interface and reduced the redox probe current. Quantification via Langmuir and Freundlich–Langmuir models yielded a wide detection range (0.4–450 nM) and a low detection limit (0.048 nM). While the method is promising for miniaturized sensors, further optimization of MOF structures and interface design is needed to enhance specificity, especially under complex environmental conditions. Additionally, deeper investigation into signal transduction mechanisms for electrochemically inert PFAS will be essential for advancing MOF-based sensor platforms. A summary of recent development in electrochemical sensors for detection of different types of PFAS is presented in Table 3.
| Type of PFAS detected | Electrode material/matrix | Polymerization method | Electrolyte used for polymerization | Washing solution | Detection technique | LOD | Linear range | References |
|---|---|---|---|---|---|---|---|---|
| PFOS (C-8) | Carbon based MIP | Electropolymerization | (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 0.1 M acetate buffer + methanol 1) 0.1 M acetate buffer + methanol |
50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 water/methanol 50 water/methanol |
DPV, EIS | 3.4 pM (0.0017 ppb) | 0.05–0.5 nM | 141 |
| PFOS (C-8) | o-Phenylenediamine based MIP | Electropolymerization | (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 0.1 M acetate buffer + methanol 1) 0.1 M acetate buffer + methanol |
50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 water/methanol 50 water/methanol |
CV, DPV | 0.5 nM (0.207 ppb) | 0.1–4.9 nM | 142 |
| GenX (C-8) | o-Phenylenediamine based MIP | Electropolymerization | Acetate buffer (pH 5.8) | 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 water/methanol + MilliQ water 50 water/methanol + MilliQ water |
CV, DPV | 250 fM (0.0825 ppt) | 1–5000 pM | 143 |
| PFOS (C-8) | Gold nanostar + MIP | Electropolymerization | (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 0.1 M acetate buffer + methanol 1) 0.1 M acetate buffer + methanol |
50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 water/methanol + MilliQ water 50 water/methanol + MilliQ water |
EIS, CV, DPV | 0.015 nM (0.0075 ppb) | — | 144 |
| PFOA and PFOS (C-8) | Methylene blue + MIP | Electropolymerization | — | — | CV | 100 nM (41.4 ppb) | 10 μM–10 mM | 151 |
| PFOS (C-8) | Gold based MIP | Electropolymerization | (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) 0.1 M acetate buffer + 10% methanol 10) 0.1 M acetate buffer + 10% methanol |
Methanol/MilliQ water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
CV, DPV, EIS | 0.1 nM (0.05 ppb) | — | 152 |
| PFOS (C-8) | Chromium based MOF | — | — | — | EIS | 0.5 ng L–1 (0.5 ppt) | — | 149 |
| PFOS (C-8), PFHxS (C-6), PFBS, and PFOA (C-8) | Micropipette-based interfaces between two immiscible electrolyte solutions | — | — | — | CV, DPV | 0.05 mM (50 ppt) | 1–9 μM | 153 |
| PFOA (C-8) | Perfluorodecanethiol + gold NPs | — | — | — | CV, LSV | 24 ppt | 100–5000 ppt | 147 |
| PFOA (C-8) | Polyaniline–chitosan | — | — | — | CV, DPV | 1.08 ppb | 5–150 ppb | 154 |
5. Current challenges and future perspectives
The global recognition of PFAS as persistent environmental contaminants has underscored the need for effective detection and remediation strategies. However, due to their complex chemical structure, these compounds present a unique set of challenges that hinder both their identification and removal. The amphiphilic nature of PFAS, combined with their exceptional resistance to degradation, renders them stable in diverse environmental matrices, where they accumulate and potentially cause long-term harm to ecosystems and human health. As these substances are not readily biodegradable, their management demands an integrated approach involving cutting-edge analytical methods, material innovations, and targeted remediation technologies. Although advanced analytical techniques, including mass spectrometry coupled with chromatography, have proven effective in detecting PFAS, they are constrained by the need for expensive instrumentation, skilled personnel, and complex sample preparation. These limitations hinder their suitability for real-time or widespread environmental monitoring. To address this gap, there is a critical demand for new methods that are not only cost-effective but also capable of rapid, on-site detection with minimal sample preprocessing.Sensor technologies offer clear advantages, such as their portability, ease of use, and potential for real-time, in situ measurements; however, several challenges remain. Many sensors are designed to detect only a subset of PFAS, limiting their applicability for comprehensive environmental monitoring. Furthermore, sensor performance can be influenced by environmental factors such as temperature, pH, and the presence of interfering substances, which may affect the reliability and accuracy of the measurements. Despite these challenges, the ongoing development of advanced sensor systems holds promise for enabling more widespread, real-time monitoring of PFAS contamination in diverse environmental contexts. Further advancements are required to enhance the performance of sensors, particularly in terms of improving their sensitivity to ultralow detection limits and ensuring robustness in complex, real-world samples. The primary difficulty arises from the inherently low optical or electrochemical activity of PFAS, which necessitates the use of specialized recognition elements that can selectively bind to these substances without interference from other chemicals. Research into the interactions between PFAS and various materials is essential for improving the efficiency and selectivity of detection methods. Although the role of hydrophobic and electrostatic interactions in PFAS adsorption is well-established, the effect of surface modifications and material composition on sensor performance remains an area of active exploration. Understanding these interactions in greater detail will allow for the design of materials and sensor platforms that can enhance PFAS capture without compromising selectivity. Handling high concentrations of PFAS is another concern for these sensors. Sensor linear ranges discussed in Tables 1–3 cover typical environmental levels, but their exceedance causes receptor saturation where they tend to signal plateau, yielding inaccurate high readings. The standard routine for wastewater includes diluting the sample 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10–1
10–1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 with clean water/methanol, re-measurement, and then multiplying the result by the dilution factor.
100 with clean water/methanol, re-measurement, and then multiplying the result by the dilution factor.
Another important area of focus is the development of pre-concentration techniques, which would allow for the isolation of PFAS from complex environmental matrices. Methods such as solid-phase extraction (SPE) and micro-SPE have been employed to improve the sensitivity of sensors by concentrating PFAS in samples prior to analysis. These techniques, when combined with portable sensor platforms, could lead to real-time detection capabilities, offering significant advantages in the field. Additionally, novel strategies, such as the integration of sensors with extraction technologies, could further reduce the potential for interference from other compounds, providing more accurate readings.
The commercialization of these sensor technologies will require further development of robust, field-deployable devices capable of providing accurate, real-time data. The integration of advanced sensor technologies into routine monitoring programs could significantly enhance our ability to manage PFAS contamination, enabling more efficient identification of hotspots, better-informed decision-making regarding clean-up efforts, and improved protection of both human and environmental health. A multi-disciplinary approach, involving collaboration between materials scientists, chemists, engineers, and environmental health experts, is essential for accelerating the development of next-generation sensors capable of addressing the complexity of PFAS contamination in diverse environmental settings. This transition from the laboratory to real-world applications will involve overcoming challenges related to sensor longevity, cost, and ease of use. The successful deployment of these technologies will not only aid in regulatory compliance but also contribute to broader public health and environmental safety by providing continuous monitoring capabilities in areas affected by PFAS contamination.
Conflicts of interest
There are no conflicts to declare.Data availability
Not applicable as no data were used in producing this review article.Acknowledgements
Authors would like to express their gratitude to for EPSRC International Doctoral Scholars – IDS grant NCZ1080-EP/W524694/1-2323187 and Dŵr Cymru Welsh Water providing an PhD scholarship to Mr. Vibhas Chugh and an EPSRC IAA grant (RIR1046-171).References
- L. G. T. Gaines, G. Sinclair and A. J. Williams, A Proposed Approach to Defining Per- and Polyfluoroalkyl Substances (PFAS) Based on Molecular Structure and Formula, Integr. Environ. Assess. Manage., 2023, 19(5), 1333–1347, DOI:10.1002/IEAM.4735.
- R. C. Buck, S. H. Korzeniowski, E. Laganis and F. Adamsky, Identification and Classification of Commercially Relevant Per- and Poly-Fluoroalkyl Substances (PFAS), Integr. Environ. Assess. Manage., 2021, 17(5), 1045–1055, DOI:10.1002/IEAM.4450.
- E. Gagliano, M. Sgroi, P. P. Falciglia, F. G. A. Vagliasindi and P. Roccaro, Removal of Poly- and Perfluoroalkyl Substances (PFAS) from Water by Adsorption: Role of PFAS Chain Length, Effect of Organic Matter and Challenges in Adsorbent Regeneration, Water Res., 2020, 171, 115381, DOI:10.1016/J.WATRES.2019.115381.
- J. Glüge, M. Scheringer, I. T. Cousins, J. C. Dewitt, G. Goldenman, D. Herzke, R. Lohmann, C. A. Ng, X. Trier and Z. Wang, An Overview of the Uses of Per- and Polyfluoroalkyl Substances (PFAS), Environ. Sci.: Processes Impacts, 2020, 22(12), 2345–2373, 10.1039/D0EM00291G.
- A. U. Rehman, M. Crimi and S. Andreescu, Current and Emerging Analytical Techniques for the Determination of PFAS in Environmental Samples, Trends Environ. Anal. Chem., 2023, 37, e00198, DOI:10.1016/J.TEAC.2023.E00198.
- L. G. T. Gaines, Historical and Current Usage of Per- and Polyfluoroalkyl Substances (PFAS): A Literature Review, Am. J. Ind. Med., 2023, 66(5), 353–378, DOI:10.1002/AJIM.23362.
- A. Manayil Parambil, E. Priyadarshini, S. Paul, A. Bakandritsos, V. K. Sharma and R. Zbořil, Emerging Nanomaterials for the Detection of Per- and Poly-Fluorinated Substances, J. Mater. Chem. A, 2025, 8246–8281, 10.1039/d4ta07045c.
- E. L. Schymanski, J. Zhang, P. A. Thiessen, P. Chirsir, T. Kondic and E. E. Bolton, Per- and Polyfluoroalkyl Substances (PFAS) in PubChem: 7 Million and Growing, Environ. Sci. Technol., 2023, 57(44), 16918–16928, DOI:10.1021/acs.est.3c04855.
- R. F. Menger, E. Funk, C. S. Henry and T. Borch, Sensors for Detecting Per- and Polyfluoroalkyl Substances (PFAS): A Critical Review of Development Challenges, Current Sensors, and Commercialization Obstacles, Chem. Eng. J., 2021, 417, 129133, DOI:10.1016/J.CEJ.2021.129133.
- Z. Jiao, S. Taniyasu, N. Yu, X. Wang, N. Yamashita and S. Wei, Two-Layer Homolog Network Approach for PFAS Nontarget Screening and Retrospective Data Mining, Nat. Commun., 2025, 16(1), 688, DOI:10.1038/s41467-025-56035-1.
- S. Y. Wee and A. Z. Aris, Revisiting the “Forever Chemicals”, PFOA and PFOS Exposure in Drinking Water, npj Clean Water, 2023, 6(1), 1–16, DOI:10.1038/s41545-023-00274-6.
- H. Brunn, G. Arnold, W. Körner, G. Rippen, K. G. Steinhäuser and I. Valentin, PFAS: Forever Chemicals—Persistent, Bioaccumulative and Mobile. Reviewing the Status and the Need for Their Phase out and Remediation of Contaminated Sites, Environ. Sci. Eur., 2023, 35(1), 1–50, DOI:10.1186/S12302-023-00721-8.
- D. Ackerman Grunfeld, D. Gilbert, J. Hou, A. M. Jones, M. J. Lee, T. C. G. Kibbey and D. M. O'Carroll, Underestimated Burden of Per- and Polyfluoroalkyl Substances in Global Surface Waters and Groundwaters, Nat. Geosci., 2024, 17(4), 340–346, DOI:10.1038/s41561-024-01402-8.
- D. T. Adamson, C. J. Newell, P. Kulkarni and H. Stroo, PFAS Monitored Retention: A Framework for Managing PFAS-Contaminated Groundwater Sites, Groundwater Monit. Rem., 2025, 45, 37–49, DOI:10.1111/gwmr.70001.
- J. Wang, Z. Dou, H. Zheng, C. Zhuang and M. L. Brusseau, Differential Sorption of Short-Chain versus Long-Chain Anionic Per- and Poly-Fluoroalkyl Substances by Soils, Environments, 2023, 10(10), 175, DOI:10.3390/ENVIRONMENTS10100175.
- D. Li, C. S. Lee, Y. Zhang, R. Das, F. Akter, A. K. Venkatesan and B. S. Hsiao, Efficient Removal of Short-Chain and Long-Chain PFAS by Cationic Nanocellulose, J. Mater. Chem. A, 2023, 11(18), 9868–9883, 10.1039/D3TA01851B.
- Z. Abunada, M. Y. D. Alazaiza and M. J. K. Bashir, An Overview of Per- and Polyfluoroalkyl Substances (PFAS) in the Environment: Source, Fate, Risk and Regulations, Water, 2020, 12(12), 3590, DOI:10.3390/W12123590.
- M. W. Sima and P. R. Jaffé, A Critical Review of Modeling Poly- and Perfluoroalkyl Substances (PFAS) in the Soil-Water Environment, Sci. Total Environ., 2021, 757, 143793, DOI:10.1016/J.SCITOTENV.2020.143793.
- U. Eriksson, J. F. Mueller, L. M. L. Toms, P. Hobson and A. Kärrman, Temporal Trends of PFSAs, PFCAs and Selected Precursors in Australian Serum from 2002 to 2013, Environ. Pollut., 2017, 220, 168–177, DOI:10.1016/J.ENVPOL.2016.09.036.
- L. Vierke, L. Ahrens, M. Shoeib, W. U. Palm, E. M. Webster, D. A. Ellis, R. Ebinghaus and T. Harner, In Situ Air–Water and Particle–Water Partitioning of Perfluorocarboxylic Acids, Perfluorosulfonic Acids and Perfluorooctyl Sulfonamide at a Wastewater Treatment Plant, Chemosphere, 2013, 92(8), 941–948, DOI:10.1016/J.CHEMOSPHERE.2013.02.067.
- R. M. Janousek, J. Mayer and T. P. Knepper, Is the Phase-out of Long-Chain PFASs Measurable as Fingerprint in a Defined Area? Comparison of Global PFAS Concentrations and a Monitoring Study Performed in Hesse, Germany from 2014 to 2018, TrAC, Trends Anal. Chem., 2019, 120, 115393, DOI:10.1016/J.TRAC.2019.01.017.
- R. A. Brase, E. J. Mullin and D. C. Spink, Legacy and Emerging Per- and Polyfluoroalkyl Substances: Analytical Techniques, Environmental Fate, and Health Effects, Int. J. Mol. Sci., 2021, 22(3), 995, DOI:10.3390/IJMS22030995.
- M. Menegatto and A. Zamperini, Health and Psychological Concerns of Communities Affected by Per- and Poly-Fluoroalkyl Substances: The Case of Residents Living in the Orange Area of the Veneto Region, Int. J. Environ. Res. Public Health, 2023, 20(22), 7056, DOI:10.3390/IJERPH20227056.
- W. Yao, J. Xu, W. Tang, C. Gao, L. Tao, J. Yu, J. Lv, H. Wang, Y. Fan, D. X. Xu and Y. Huang, Developmental Toxicity of Perfluorohexane Sulfonate at Human Relevant Dose during Pregnancy via Disruption in Placental Lipid Homeostasis, Environ. Int., 2023, 177, 108014, DOI:10.1016/J.ENVINT.2023.108014.
- D. M. Pizzurro, M. Seeley, L. E. Kerper and B. D. Beck, Interspecies Differences in Perfluoroalkyl Substances (PFAS) Toxicokinetics and Application to Health-Based Criteria, Regul. Toxicol. Pharmacol., 2019, 106, 239–250, DOI:10.1016/J.YRTPH.2019.05.008.
- K. Y. Christensen, M. Raymond, M. Blackowicz, Y. Liu, B. A. Thompson, H. A. Anderson and M. Turyk, Perfluoroalkyl Substances and Fish Consumption, Environ. Res., 2017, 154, 145–151, DOI:10.1016/J.ENVRES.2016.12.032.
- K. Steenland and A. Winquist, PFAS and Cancer, a Scoping Review of the Epidemiologic Evidence, Environ. Res., 2021, 194, 110690, DOI:10.1016/J.ENVRES.2020.110690.
- R. L. Birru, H. W. Liang, F. Farooq, M. Bedi, M. Feghali, C. L. Haggerty, D. D. Mendez, J. M. Catov, C. A. Ng and J. J. Adibi, A Pathway Level Analysis of PFAS Exposure and Risk of Gestational Diabetes Mellitus, Environ. Health, 2021, 20(1), 1–16, DOI:10.1186/S12940-021-00740-Z.
- B. Ruffle, U. Vedagiri, D. Bogdan, M. Maier, C. Schwach and C. Murphy-Hagan, Perfluoroalkyl Substances in U.S. Market Basket Fish and Shellfish, Environ. Res., 2020, 190, 109932, DOI:10.1016/J.ENVRES.2020.109932.
- H. Gardener, Q. Sun and P. Grandjean, PFAS Concentration during Pregnancy in Relation to Cardiometabolic Health and Birth Outcomes, Environ. Res., 2021, 192, 110287, DOI:10.1016/J.ENVRES.2020.110287.
- M. Kirk, K. Smurthwaite, J. Braunig, S. Trevenar, C. D'Este, R. Lucas, A. Lal, R. Korda, A. Clements, J. Mueller and B. Armstrong, The PFAS Health Study: Systematic Literature Review, 2018, DOI:10.25911/KW6T-7H44, https://rsph.anu.edu.au/files/PFAS%20Health%20Study%20Systematic%20Review_1.pdf.
- J. B. Brown, J. M. Conder, J. A. Arblaster and C. P. Higgins, Assessing Human Health Risks from Per- And Polyfluoroalkyl Substance (PFAS)-Impacted Vegetable Consumption: A Tiered Modeling Approach, Environ. Sci. Technol., 2020, 54(23), 15202–15214, DOI:10.1021/ACS.EST.0C03411/ASSET/IMAGES/LARGE/ES0C03411_0006.JPEG.
- F. C. Fischer, S. Ludtke, C. Thackray, H. M. Pickard, F. Haque, C. Dassuncao, S. Endo, L. Schaider and E. M. Sunderland, Binding of Per- and Polyfluoroalkyl Substances (PFAS) to Serum Proteins: Implications for Toxicokinetics in Humans, Environ. Sci. Technol., 2024, 58, 1055–1063, DOI:10.1021/ACS.EST.3C07415.
- J. Huang, Y. Liu, Q. Wang, J. Yi, H. Lai, L. Sun, J. A. Mennigen and W. Tu, Concentration-Dependent Toxicokinetics of Novel PFOS Alternatives and Their Chronic Combined Toxicity in Adult Zebrafish, Sci. Total Environ., 2022, 839, 156388, DOI:10.1016/J.SCITOTENV.2022.156388.
- L. Ahrens and M. Bundschuh, Fate and Effects of Poly- and Perfluoroalkyl Substances in the Aquatic Environment: A Review, Environ. Toxicol. Chem., 2014, 33(9), 1921–1929, DOI:10.1002/ETC.2663.
- F. Xiao, Emerging Poly- and Perfluoroalkyl Substances in the Aquatic Environment: A Review of Current Literature, Water Res., 2017, 124, 482–495, DOI:10.1016/J.WATRES.2017.07.024.
- A. Cordner, V. Y. De La Rosa, L. A. Schaider, R. A. Rudel, L. Richter and P. Brown, Guideline Levels for PFOA and PFOS in Drinking Water: The Role of Scientific Uncertainty, Risk Assessment Decisions, and Social Factors, J. Exposure Sci. Environ. Epidemiol., 2019, 29(2), 157–171, DOI:10.1038/s41370-018-0099-9.
- C. M. Teaf, M. M. Garber, D. J. Covert and B. J. Tuovila, Perfluorooctanoic Acid (PFOA): Environmental Sources, Chemistry, Toxicology, and Potential Risks, Soil Sediment Contam., 2019, 28(3), 258–273, DOI:10.1080/15320383.2018.1562420.
- R. B. Taylor and Y. Sapozhnikova, Comparison and Validation of the QuEChERSER Mega-Method for Determination of per- and Polyfluoroalkyl Substances in Foods by Liquid Chromatography with High-Resolution and Triple Quadrupole Mass Spectrometry, Anal. Chim. Acta, 2022, 1230, 340400, DOI:10.1016/J.ACA.2022.340400.
- O. A. Abafe, L. R. Macheka and J. O. Olowoyo, Confirmatory Analysis of Per and Polyfluoroalkyl Substances in Milk and Infant Formula Using UHPLC–MS/MS, Molecules, 2021, 26(12), 3664, DOI:10.3390/MOLECULES26123664.
- A. T. Aborode, R. O. Adesola, I. Idris, W. Sakariyau Adio, S. Olapade, G. Oluwafisayo, I. A. Onifade, S. Fakorede, T. Bakare-Abidola, J. Olaoye, A. D. Ogunyemi, O. A. Ogundijo, O. G. Banwo, A. A. Bakre, P. Oladoye, G. Adegoye and N. A. Jinadu, Challenges Associated With PFAS Detection Method in Africa, Environ. Health Insights, 2025, 19, 1–12, DOI:10.1177/11786302241310430.
- A. Manayil Parambil, E. Priyadarshini, S. Paul, A. Bakandritsos, V. K. Sharma and R. Zbořil, Emerging Nanomaterials for the Detection of Per- and Poly-Fluorinated Substances, J. Mater. Chem. A, 2025, 8246–8281, 10.1039/d4ta07045c.
- J. Kumar and M. Narayan, Advanced Nanoformulations for Detection and Removal of Poly- and Perfluoroalkyl Substances (PFAS), Pollutants, 2025, 5, 10, DOI:10.3390/pollutants5020010.
- S. C. Cullom and J. E. Dick, Time: The Potentially Powerful, Often-Overlooked Variable in Electrochemical PFAS Sensing, Curr. Opin. Electrochem., 2025, 101726, DOI:10.1016/j.coelec.2025.101726.
- M. A. A. Abdelhamid, M.-R. Ki, H. J. Yoon and S. P. Pack, Microfluidic Sensors for Micropollutant Detection in Environmental Matrices: Recent Advances and Prospects, Biosensors, 2025, 15(8), 474, DOI:10.3390/bios15080474.
- A. Dube, S. J. Malode, H. Akhdar, A. N. Alodhayb and N. P. Shetti, Electrochemical Detection of Per- and Polyfluoroalkyl Substances: A Review, Colloids Surf., B, 2025, 252, 114653, DOI:10.1016/j.colsurfb.2025.114653.
- N. Gondhiya, A. U. Rehman, D. Andreescu and S. Andreescu, Portable Electrochemical Sensors for Per- and Polyfluoroalkyl Substances: Design, Challenges, and Opportunities for Field Deployment, Curr. Opin. Electrochem., 2025, 53, 101725, DOI:10.1016/j.coelec.2025.101725.
- Y. Aminot, S. J. Sayfritz, K. V. Thomas, L. Godinho, E. Botteon, F. Ferrari, V. Boti, T. Albanis, M. Köck-Schulmeyer, S. Diaz-Cruz, M. Farré, D. Barceló, A. Marques and J. W. Readman, Environmental Risks Associated with Contaminants of Legacy and Emerging Concern at European Aquaculture Areas, Environ. Pollut., 2019, 809, 151003, DOI:10.1016/j.envpol.2019.05.133.
- S. Kurwadkar, J. Dane, S. R. Kanel, M. N. Nadagouda, R. W. Cawdrey, B. Ambade, G. C. Struckhoff and R. Wilkin, Per- and polyfluoroalkyl substances in water and wastewater: A critical review of their global occurrence and distribution, Sci. Total Environ., 2022, 809, 151003, DOI:10.1016/J.SCITOTENV.2021.151003.
- Per- and Polyfluoroalkyl Substances (PFAS) | US EPA, https://www.epa.gov/sdwa/and-polyfluoroalkyl-substances-pfas, Drinking water (national regulations in EU countries), DOI:10.1021/acs.est.0c03244.
- N. P. Cheremisinoff, Perfluorinated Chemicals (PFCs): Contaminants of Concern, Wiley, 2016 Search PubMed.
- M. S. Díaz-Cruz, M. J. García-Galán, P. Guerra, A. Jelic, C. Postigo, E. Eljarrat, M. Farré, M. J. Ló Pez De Alda, M. Petrovic and D. Barceló, Dean Analysis of Selected Emerging Contaminants in Sewage Sludge, TrAC, Trends Anal. Chem., 28, 1263–1275, DOI:10.1016/j.trac.2009.09.003.
- Per- and Polyfluoroalkyl Substances (PFAS) | US EPA, https://www.epa.gov/sdwa/and-polyfluoroalkyl-substances-pfas (accessed 2025-09-12).
- EPA Announces It Will Keep Maximum Contaminant Levels for PFOA, PFOS | US EPA, https://www.epa.gov/newsreleases/epa-announces-it-will-keep-maximum-contaminant-levels-pfoa-pfos (accessed 2025-09-12).
- F. Li, J. Duan, S. Tian, H. Ji, Y. Zhu, Z. Wei and D. Zhao, Short-Chain per- and Polyfluoroalkyl Substances in Aquatic Systems: Occurrence, Impacts and Treatment, Chem. Eng. J., 2020, 380, 122506, DOI:10.1016/j.cej.2019.122506.
- T. Stoiber, S. Evans, A. M. Temkin, D. Q. Andrews and O. V. Naidenko, WATER RESEARCH PFAS in Drinking Water: An Emergent Water Quality Threat, Water Solutions, 2020 Search PubMed.
- L. Rockett, Poly and Perfluorinated Alkyl Substances in Drinking Water, DWI 70/2/327, 2020 Search PubMed.
- Leading the Water Industry Research Agenda, https://ukwir.org/leading-the-water-industry-research-agenda (accessed 2024-02-29).
- E. Pignotti, G. Casas, M. Llorca, A. Tellbüscher, D. Almeida, E. Dinelli, M. Farré and D. Barceló, Seasonal Variations in the Occurrence of Perfluoroalkyl Substances in Water, Sediment and Fish Samples from Ebro Delta (Catalonia, Spain), Sci. Total Environ., 2017, 607–608, 933–943, DOI:10.1016/J.SCITOTENV.2017.07.025.
- M. Lorenzo, J. Campo, M. Farré, F. Pérez, Y. Picó and D. Barceló, Perfluoroalkyl Substances in the Ebro and Guadalquivir River Basins (Spain), Sci. Total Environ., 2016, 540, 191–199, DOI:10.1016/J.SCITOTENV.2015.07.045.
- M. Llorca, M. Farré, Y. Picó, J. Müller, T. P. Knepper and D. Barceló, Analysis of Perfluoroalkyl Substances in Waters from Germany and Spain, Sci. Total Environ., 2012, 431, 139–150, DOI:10.1016/J.SCITOTENV.2012.05.011.
- K. Y. Tan, G. H. Lu, H. T. Piao, S. Chen, X. C. Jiao, N. Gai, E. Yamazaki, N. Yamashita, J. Pan and Y. L. Yang, Current Contamination Status of Perfluoroalkyl Substances in Tapwater from 17 Cities in the Eastern China and Their Correlations with Surface Waters, Bull. Environ. Contam. Toxicol., 2017, 99(2), 224–231, DOI:10.1007/S00128-017-2109-3.
- C. G. Pan, Y. S. Liu and G. G. Ying, Perfluoroalkyl Substances (PFASs) in Wastewater Treatment Plants and Drinking Water Treatment Plants: Removal Efficiency and Exposure Risk, Water Res., 2016, 106, 562–570, DOI:10.1016/J.WATRES.2016.10.045.
- X. Cao, C. Wang, Y. Lu, M. Zhang, K. Khan, S. Song, P. Wang and C. Wang, Occurrence, Sources and Health Risk of Polyfluoroalkyl Substances (PFASs) in Soil, Water and Sediment from a Drinking Water Source Area, Ecotoxicol. Environ. Saf., 2019, 174, 208–217, DOI:10.1016/J.ECOENV.2019.02.058.
- J. Bao, W. J. Yu, Y. Liu, X. Wang, Y. H. Jin and G. H. Dong, Perfluoroalkyl Substances in Groundwater and Home-Produced Vegetables and Eggs around a Fluorochemical Industrial Park in China, Ecotoxicol. Environ. Saf., 2019, 171, 199–205, DOI:10.1016/J.ECOENV.2018.12.086.
- J. Meng, S. Liu, Y. Zhou and T. Wang, Are Perfluoroalkyl Substances in Water and Fish from Drinking Water Source the Major Pathways towards Human Health Risk?, Ecotoxicol. Environ. Saf., 2019, 181, 194–201, DOI:10.1016/J.ECOENV.2019.06.010.
- S. Takagi, F. Adachi, K. Miyano, Y. Koizumi, H. Tanaka, I. Watanabe, S. Tanabe and K. Kannan, Fate of Perfluorooctanesulfonate and Perfluorooctanoate in Drinking Water Treatment Processes, Water Res., 2011, 45(13), 3925–3932, DOI:10.1016/J.WATRES.2011.04.052.
- B. M. Sharma, G. K. Bharat, S. Tayal, T. Larssen, J. Bečanová, P. Karásková, P. G. Whitehead, M. N. Futter, D. Butterfield and L. Nizzetto, Perfluoroalkyl Substances (PFAS) in River and Ground/Drinking Water of the Ganges River Basin: Emissions and Implications for Human Exposure, Environ. Pollut., 2016, 208(Pt B), 704–713, DOI:10.1016/J.ENVPOL.2015.10.050.
- J. Thompson, G. Eaglesham and J. Mueller, Concentrations of PFOS, PFOA and Other Perfluorinated Alkyl Acids in Australian Drinking Water, Chemosphere, 2011, 83(10), 1320–1325, DOI:10.1016/J.CHEMOSPHERE.2011.04.017.
- J. K. Anderson, A. L. Luz, P. Goodrum and J. Durda, Perfluorohexanoic Acid Toxicity, Part II: Application of Human Health Toxicity Value for Risk Characterization, Regul. Toxicol. Pharmacol., 2019, 103, 10–20, DOI:10.1016/j.yrtph.2019.01.020.
- G. Jiménez-Skrzypek, J. González-Sálamo and J. Hernández-Borges, Analytical Methodologies and Occurrence of Per- and Polyfluorinated Alkyl Substances – A Review, J. Chromatogr. Open, 2023, 4, 100089, DOI:10.1016/J.JCOA.2023.100089.
- C. Barola, S. Moretti, D. Giusepponi, F. Paoletti, G. Saluti, G. Cruciani, G. Brambilla and R. Galarini, A Liquid Chromatography-High Resolution Mass Spectrometry Method for the Determination of Thirty-Three per- and Polyfluoroalkyl Substances in Animal Liver, J. Chromatogr. A, 2020, 1628, 461442, DOI:10.1016/J.CHROMA.2020.461442.
- B. A. Parker, D. R. U. Knappe, I. A. Titaley, T. A. Wanzek and J. A. Field, Tools for Understanding and Predicting the Affinity of Per- and Polyfluoroalkyl Substances for Anion-Exchange Sorbents, Environ. Sci. Technol., 2022, 56(22), 15470–15477, DOI:10.1021/ACS.EST.1C08345/ASSET/IMAGES/LARGE/ES1C08345_0006.JPEG.
- T. E. Lockwood, M. Talebi, A. Minett, S. Mills, P. A. Doble and D. P. Bishop, Micro Solid-Phase Extraction for the Analysis of per- and Polyfluoroalkyl Substances in Environmental Waters, J. Chromatogr. A, 2019, 1604, 460495, DOI:10.1016/J.CHROMA.2019.460495.
- Y. Liu, J. Bao, X. M. Hu, G. L. Lu, W. J. Yu and Z. H. Meng, Optimization of Extraction Methods for the Analysis of PFOA and PFOS in the Salty Matrices during the Wastewater Treatment, Microchem. J., 2020, 155, 104673, DOI:10.1016/J.MICROC.2020.104673.
- J. F. Ayala-Cabrera, E. Moyano and F. J. Santos, Gas Chromatography and Liquid Chromatography Coupled to Mass Spectrometry for the Determination of Fluorotelomer Olefins, Fluorotelomer Alcohols, Perfluoroalkyl Sulfonamides and Sulfonamido-Ethanols in Water, J. Chromatogr. A, 2020, 1609, 460463, DOI:10.1016/J.CHROMA.2019.460463.
- K. A. Oetjen, D. Tran, S. C. Roberts, C. Butt and C. Borton, Analysis of EPA Method 537 Per-and Polyfluoroalkyl Substances (PFASs) Using Microflow Liquid Chromatography, https://sciex.com/tech-notes/environmental-industrial/water-and-soil/optifl Search PubMed.
- J. S. Whitaker, R. B. Hrabak, M. Ramos, C. Neslund and Y. Li, An Interlaboratory Study on EPA Methods 537.1 and 533 for per- and Polyfluoroalkyl Substance Analyses, AWWA Water Sci., 2021, 3(4), e1234, DOI:10.1002/AWS2.1234.
- M. Zhong, T. Wang, C. Qi, G. Peng, M. Lu, J. Huang, L. Blaney and G. Yu, Automated Online Solid-Phase Extraction Liquid Chromatography Tandem Mass Spectrometry Investigation for Simultaneous Quantification of per- and Polyfluoroalkyl Substances, Pharmaceuticals and Personal Care Products, and Organophosphorus Flame Retardants in Environmental Waters, J. Chromatogr. A, 2019, 1602, 350–358, DOI:10.1016/J.CHROMA.2019.06.012.
- M. N. Goukeh, T. Abichou and Y. Tang, Measurement of Fluorotelomer Alcohols Based on Solid Phase Microextraction Followed by Gas Chromatography-Mass Spectrometry and Its Application in Solid Waste Study, Chemosphere, 2023, 345, 140460, DOI:10.1016/J.CHEMOSPHERE.2023.140460.
- T. L. Coggan, T. Anumol, J. Pyke, J. Shimeta and B. O. Clarke, A Single Analytical Method for the Determination of 53 Legacy and Emerging Per- and Polyfluoroalkyl Substances (PFAS) in Aqueous Matrices, Anal. Bioanal. Chem., 2019, 411(16), 3507–3520, DOI:10.1007/S00216-019-01829-8/TABLES/5.
- Y. Liang, Q. Jiang, M. Z. Hua, T. Zhang and X. Lu, Recent Advances in the Detection of Per- and Polyfluoroalkyl Substances (PFAS) in Food: A Review of Optical Sensors, Electrochemical Sensors, and Biosensors, Trends Food Sci. Technol., 2025, 164, 105239, DOI:10.1016/j.tifs.2025.105239.
- J. Park, J. H. Baik, S. Adjei-Nimoh and W. H. Lee, Advancements in Artificial Intelligence-Based Technologies for PFAS Detection, Monitoring, and Management, Sci. Total Environ., 2025, 980, 179536, DOI:10.1016/j.scitotenv.2025.179536.
- Y. D. Yang, Q. Zhang, X. Jin, C. V. Chau, J. Yang, G. Henkelman, X. Chi, H. Y. Gong, R. J. Hooley and J. L. Sessler, Readily Visualized Perfluorooctanoic Acid Detection Using a Small Molecule Chemosensor, Angew. Chem., Int. Ed., 2025, 64(19), e202501245, DOI:10.1002/anie.202501245.
- Y. P. Chung, J. T. Lin, Y. Y. Lee, P. T. Nguyen, T. T. T. Huynh, M. J. Lu, B. W. Huang, B. Sriram, S. F. Wang, S. Kogularasu and G. P. Chang-Chien, Comparative Review of Conventional and Emerging PFAS Detection Methods: A Focus on Detection Strategies, Microchem. J., 2025, 217, 115103, DOI:10.1016/j.microc.2025.115103.
- A. Khanam, A. Islam and W. Xu, Rapid Electrochemical Detection of Multi-PFAS via Novel Conductive Molecularly Imprinted Polymers (MIPs), https://ssrn.com/abstract=5304637.
- F. A. Tabar, J. W. Lowdon, M. Frigoli, R. D. Crapnell, T. J. Cleij, H. Diliën, C. E. Banks, K. Eersels, B. van Grinsven and P. Wagner, Tracking Perfluorooctanoic Acid in Tap and River Water Employing Screen-Printed Electrodes Modified with Molecularly Imprinted Polymers, ACS Omega, 2025, 10(15), 15018–15028, DOI:10.1021/acsomega.4c10473.
- Y. Xu, P. Zhou and T. Cui, Laser-Induced Graphene Electrochemical Sensors to Detect Perfluorooctanoic Acid (PFOA), https://ssrn.com/abstract=5287849.
- A. Lobitz, A. Steuber, S. Jia and L. Guo, Harnessing Fluorine Chemistry: Strategies for Per- and Polyfluoroalkyl Substances Removal and Enrichment, ChemPlusChem, 2025, 90, e202400784, DOI:10.1002/cplu.202400784.
- M. Khadka, Y. M. Pasternak, O. Sheinenzon, A. Snezhko, A. Ronen, M. Amiram and H. Ben-Yoav, Detection of Perfluorooctance Sulphonic Acid in Groundwater Using an Intelligent Array of Electrochemical Sensors, J. Hazard. Mater., 2025, 495, 138844, DOI:10.1016/j.jhazmat.2025.138844.
- R. Pitruzzella, PFAS Sensor Configurations Exploiting Plastic Optical Fibers and Molecularly Imprinted Polymers, in 2025 25th Anniversary International Conference on Transparent Optical Networks(ICTON), IEEE, 2025, pp. 1–6, DOI:10.1109/ICTON67126.2025.11125101.
- Z. Pu, X. Wang, W. Chen, Z. Liu and P. Wang, Denoising Algorithm for High-Resolution and Large-Range Phase-Sensitive SPR Imaging Based on PFA, Sensors, 2025, 25(15), 4641, DOI:10.3390/s25154641.
- R. Dalapati, S. Manickam, J. Shi, M. Hunter and L. Zang, Perylene Diimide Based Fluorescent Sensors for Aqueous Detection of Perfluorooctane Sulfonate(PFOS), Anal. Chim. Acta, 2025, 1341, DOI:10.1016/j.aca.2025.343670.
- A. Khitous, F. Arcadio, L. Zeni, N. Cennamo and O. Soppera, In Situ Synthesis of Molecularly Imprinted Polymers by Near-Field Photopolymerization for Ultrasensitive PFOA Plasmonic Plastic Fiber Optic Sensors, Sens. Actuators, B, 2025, 442, 137992, DOI:10.1016/j.snb.2025.137992.
- H. Park, J. Park, W. Kim, W. Kim and J. Park, Ultra-Sensitive SERS Detection of Perfluorooctanoic Acid Based on Self-Assembled p-Phenylenediamine Nanoparticle Complex, J. Hazard. Mater., 2023, 453, DOI:10.1016/j.jhazmat.2023.131384.
- Y. Feng, J. Dai, C. Wang, H. Zhou, J. Li, G. Ni, M. Zhang and Y. Huang, Ag Nanoparticle/Au@Ag Nanorod Sandwich Structures for SERS-Based Detection of Perfluoroalkyl Substances, ACS Appl. Nano Mater., 2023, 6(15), 13974–13983, DOI:10.1021/acsanm.3c01815.
- B. Chen, Z. Yang, X. Qu, S. Zheng, D. Yin and H. Fu, Screening and Discrimination of Perfluoroalkyl Substances in Aqueous Solution Using a Luminescent Metal-Organic Framework Sensor Array, ACS Appl. Mater. Interfaces, 2021, 13(40), 47706–47716, DOI:10.1021/ACSAMI.1C15528/SUPPL_FILE/AM1C15528_SI_001.PDF.
- R. Dalapati, M. Hunter, M. Sk, X. Yang and L. Zang, Fluorescence Turn-on Detection of Perfluorooctanoic Acid (PFOA) by Perylene Diimide-Based Metal-Organic Framework, ACS Appl. Mater. Interfaces, 2024, 16(25), 32344–32356, DOI:10.1021/acsami.4c03389.
- H. Niu, S. Wang, Z. Zhou, Y. Ma, X. Ma and Y. Cai, Sensitive Colorimetric Visualization of Perfluorinated Compounds Using Poly (Ethylene Glycol) and Perfluorinated Thiols Modified Gold Nanoparticles, Anal. Chem., 2014, 86(9), 4170–4177, DOI:10.1021/AC403406D/ASSET/IMAGES/MEDIUM/AC-2013-03406D_0009.GIF.
- A. Concellón, J. Castro-Esteban and T. M. Swager, Ultratrace PFAS Detection Using Amplifying Fluorescent Polymers, J. Am. Chem. Soc., 2023, 145(20), 11420–11430, DOI:10.1021/jacs.3c03125.
- F. Faiz, G. Baxter, S. Collins, F. Sidiroglou and M. Cran, Polyvinylidene Fluoride Coated Optical Fibre for Detecting Perfluorinated Chemicals, Sens. Actuators, B, 2020, 312, 128006, DOI:10.1016/J.SNB.2020.128006.
- R. Pitruzzella, F. Arcadio, C. Perri, D. Del Prete, G. Porto, L. Zeni and N. Cennamo, Ultra-Low Detection of Perfluorooctanoic Acid Using a Novel Plasmonic Sensing Approach Combined with Molecularly Imprinted Polymers, Chemosensors, 2023, 11(4), 211, DOI:10.3390/CHEMOSENSORS11040211.
- N. Cennamo, G. D'Agostino, G. Porto, A. Biasiolo, C. Perri, F. Arcadio and L. Zeni, A Molecularly Imprinted Polymer on a Plasmonic Plastic Optical Fiber to Detect Perfluorinated Compounds in Water, Sensors, 2018, 18(6), 1836, DOI:10.3390/S18061836.
- Z. Jiao, J. Li, L. Mo, J. Liang and H. Fan, A Molecularly Imprinted Chitosan Doped with Carbon Quantum Dots for Fluorometric Determination of Perfluorooctane Sulfonate, Microchim. Acta, 2018, 185(10), 1–9, DOI:10.1007/S00604-018-2996-Y/TABLES/2.
- L. S. Walekar, M. Zheng, L. Zheng and M. Long, Selenium and Nitrogen Co-Doped Carbon Quantum Dots as a Fluorescent Probe for Perfluorooctanoic Acid, Microchim. Acta, 2019, 186(5), 1–9, DOI:10.1007/S00604-019-3400-2/TABLES/1.
- L. Zheng, Y. Zheng, Y. Liu, S. Long, L. Du, J. Liang, C. Huang, M. T. Swihart and K. Tan, Core-Shell Quantum Dots Coated with Molecularly Imprinted Polymer for Selective Photoluminescence Sensing of Perfluorooctanoic Acid, Talanta, 2019, 194, 1–6, DOI:10.1016/J.TALANTA.2018.09.106.
- Q. Chen, P. Zhu, J. Xiong, L. Gao and K. Tan, A Sensitive and Selective Triple-Channel Optical Assay Based on Red-Emissive Carbon Dots for the Determination of PFOS, Microchem. J., 2019, 145, 388–396, DOI:10.1016/J.MICROC.2018.11.003.
- Q. Chen, P. Zhu, J. Xiong, L. Gao and K. Tan, A New Dual-Recognition Strategy for Hybrid Ratiometric and Ratiometric Sensing Perfluorooctane Sulfonic Acid Based on High Fluorescent Carbon Dots with Ethidium Bromide, Spectrochim. Acta, Part A, 2020, 224, 117362, DOI:10.1016/J.SAA.2019.117362.
- Z. Cheng, H. Dong, J. Liang, F. Zhang, X. Chen, L. Du and K. Tan, Highly Selective Fluorescent Visual Detection of Perfluorooctane Sulfonate via Blue Fluorescent Carbon Dots and Berberine Chloride Hydrate, Spectrochim. Acta, Part A, 2019, 207, 262–269, DOI:10.1016/J.SAA.2018.09.028.
- H. Niu, S. Wang, Z. Zhou, Y. Ma, X. Ma and Y. Cai, Sensitive Colorimetric Visualization of Perfluorinated Compounds Using Poly(Ethylene Glycol) and Perfluorinated Thiols Modified Gold Nanoparticles, Anal. Chem., 2014, 86(9), 4170–4177, DOI:10.1021/AC403406D/ASSET/IMAGES/MEDIUM/AC-2013-03406D_0009.GIF.
- F. Faiz, G. Baxter, S. Collins, F. Sidiroglou and M. Cran, Polyvinylidene Fluoride Coated Optical Fibre for Detecting Perfluorinated Chemicals, Sens. Actuators, B, 2020, 312, 128006, DOI:10.1016/J.SNB.2020.128006.
- N. Cennamo, G. D'agostino, F. Sequeira, F. Mattiello, G. Porto, A. Biasiolo, R. Nogueira, L. Bilro and L. Zeni, A Simple and Low-Cost Optical Fiber Intensity-Based Configuration for Perfluorinated Compounds in Water Solution, Sensors, 2018, 18(9), 3009, DOI:10.3390/S18093009.
- S. Chen, A. Li, L. Zhang and J. Gong, Molecularly Imprinted Ultrathin Graphitic Carbon Nitride Nanosheets–Based Electrochemiluminescence Sensing Probe for Sensitive Detection of Perfluorooctanoic Acid, Anal. Chim. Acta, 2015, 896, 68–77, DOI:10.1016/J.ACA.2015.09.022.
- Z. Jiao, J. Li, L. Mo, J. Liang and H. Fan, A Molecularly Imprinted Chitosan Doped with Carbon Quantum Dots for Fluorometric Determination of Perfluorooctane Sulfonate, Microchim. Acta, 2018, 185(10), 1–9, DOI:10.1007/S00604-018-2996-Y/TABLES/2.
- L. Zheng, Y. Zheng, Y. Liu, S. Long, L. Du, J. Liang, C. Huang, M. T. Swihart and K. Tan, Core-Shell Quantum Dots Coated with Molecularly Imprinted Polymer for Selective Photoluminescence Sensing of Perfluorooctanoic Acid, Talanta, 2019, 194, 1–6, DOI:10.1016/J.TALANTA.2018.09.106.
- L. S. Walekar, M. Zheng, L. Zheng and M. Long, Selenium and Nitrogen Co-Doped Carbon Quantum Dots as a Fluorescent Probe for Perfluorooctanoic Acid, Microchim. Acta, 2019, 186(5), 1–9, DOI:10.1007/S00604-019-3400-2/TABLES/1.
- Q. Liu, A. Huang, N. Wang, G. Zheng and L. Zhu, Rapid Fluorometric Determination of Perfluorooctanoic Acid by Its Quenching Effect on the Fluorescence of Quantum Dots, J. Lumin., 2015, 161, 374–381, DOI:10.1016/J.JLUMIN.2015.01.045.
- H. Feng, N. Wang, T. Trant, L. Yuan, J. Li and Q. Cai, Surface Molecular Imprinting on Dye–(NH2)–SiO2 NPs for Specific Recognition and Direct Fluorescent Quantification of Perfluorooctane Sulfonate, Sens. Actuators, B, 2014, 195, 266–273, DOI:10.1016/J.SNB.2014.01.036.
- M. Takayose, K. Akamatsu, H. Nawafune, T. Murashima and J. Matsui, Colorimetric Detection of Perfluorooctanoic Acid (PFOA) Utilizing Polystyrene-Modified Gold Nanoparticles, Anal. Lett., 2012, 45(18), 2856–2864, DOI:10.1080/00032719.2012.696225.
- C. S. Law, J. Wang, S. Gunenthiran, S. Y. Lim, A. D. Abell, L. Ahrens, T. Kumeria, A. Santos and N. H. Voelcker, Real-Time Detection of per-Fluoroalkyl Substance (PFAS) Self-Assembled Monolayers in Nanoporous Interferometers, Sens. Actuators, B, 2022, 355, 131340, DOI:10.1016/J.SNB.2021.131340.
- M. Nadal and J. L. Domingo, Non-Invasive Matrices for the Human Biomonitoring of PFAS: An Updated Review of the Scientific Literature, Toxics, 2025, 13, 134, DOI:10.3390/toxics13020134.
- X. Zhu, C. Cheng, X. Qin and Y. Wang, β-Cyclodextrin Imprinted Film Embedded with Methylene Blue: A Host-Guest Sensitive Electrochemical Strategy for PFAS Detection, J. Hazard. Mater., 2025, 485, 136870, DOI:10.1016/j.jhazmat.2024.136870.
- S. Bharti, PFAS in Water Systems: A Critical Review on Contamination Pathways, Analytical Methods, and Treatment Technologies, Water Conserv. Sci. Eng., 2025, 10, 89, DOI:10.1007/s41101-025-00415-7.
- Y. Xiao, Z. Du, Y. Li, L. Cao, B. Zhu, T. Kitaguchi and C. Huang, A Review on the Application of Biosensors for Monitoring Emerging Contaminants in the Water Environment, Sensors, 2025, 25, 4945, DOI:10.3390/s25164945.
- N. Cennamo, L. Zeni, P. Tortora, M. E. Regonesi, A. Giusti, M. Staiano, S. D'Auria and A. Varriale, A High Sensitivity Biosensor to Detect the Presence of Perfluorinated Compounds in Environment, Talanta, 2018, 178, 955–961, DOI:10.1016/J.TALANTA.2017.10.034.
- J. Park, K. A. Yang, Y. Choi and J. K. Choe, Novel SsDNA Aptamer-Based Fluorescence Sensor for Perfluorooctanoic Acid Detection in Water, Environ. Int., 2022, 158, 107000, DOI:10.1016/J.ENVINT.2021.107000.
- W. Xia, Y. J. Wan, X. Wang, Y. y. Li, W. J. Yang, C. X. Wang and S. q. Xu, Sensitive Bioassay for Detection of PPARα Potentially Hazardous Ligands with Gold Nanoparticle Probe, J. Hazard. Mater., 2011, 192(3), 1148–1154, DOI:10.1016/J.JHAZMAT.2011.06.023.
- J. Zhang, Y. Wan, Y. Li, Q. Zhang, S. Xu, H. Zhu and B. Shu, A Rapid and High-Throughput Quantum Dots Bioassay for Monitoring of Perfluorooctane Sulfonate in Environmental Water Samples, Environ. Pollut., 2011, 159(5), 1348–1353, DOI:10.1016/J.ENVPOL.2011.01.011.
- G. Moro, F. Bottari, S. Liberi, S. Covaceuszach, A. Cassetta, A. Angelini, K. De Wael and L. M. Moretto, Covalent Immobilization of Delipidated Human Serum Albumin on Poly(Pyrrole-2-Carboxylic) Acid Film for the Impedimetric Detection of Perfluorooctanoic Acid, Bioelectrochemistry, 2020, 134, 107540, DOI:10.1016/J.BIOELECHEM.2020.107540.
- G. Sunantha and N. Vasudevan, A Method for Detecting Perfluorooctanoic Acid and Perfluorooctane Sulfonate in Water Samples Using Genetically Engineered Bacterial Biosensor, Sci. Total Environ., 2021, 759, 143544, DOI:10.1016/J.SCITOTENV.2020.143544.
- M. M. Mann and B. W. Berger, A Genetically-Encoded Biosensor for Direct Detection of Perfluorooctanoic Acid, Sci. Rep., 2023, 13(1), 1–9, DOI:10.1038/s41598-023-41953-1.
- J. Zhang, Y. Wan, Y. Li, Q. Zhang, S. Xu, H. Zhu and B. Shu, A Rapid and High-Throughput Quantum Dots Bioassay for Monitoring of Perfluorooctane Sulfonate in Environmental Water Samples, Environ. Pollut., 2011, 159(5), 1348–1353, DOI:10.1016/J.ENVPOL.2011.01.011.
- J. Park, K. A. Yang, Y. Choi and J. K. Choe, Novel SsDNA Aptamer-Based Fluorescence Sensor for Perfluorooctanoic Acid Detection in Water, Environ. Int., 2022, 158, 107000, DOI:10.1016/J.ENVINT.2021.107000.
- Z. Tingting, Z. Huimin, L. Ayue and Q. Xie, Electrochemical Biosensor for Detection of Perfluorooctane Sulfonate Based on Inhibition Biocatalysis of Enzymatic Fuel Cell, Electrochemistry, 2014, 82(2), 94–99, DOI:10.5796/ELECTROCHEMISTRY.82.94.
- A. Dube, S. J. Malode, H. Akhdar, A. N. Alodhayb and N. P. Shetti, Electrochemical Detection of Per- and Polyfluoroalkyl Substances: A Review, Colloids Surf., B, 2025, 252, 114653, DOI:10.1016/j.colsurfb.2025.114653.
- S. C. Cullom and J. E. Dick, Time: The Potentially Powerful, Often-Overlooked Variable in Electrochemical PFAS Sensing, Curr. Opin. Electrochem., 2025, 101726, DOI:10.1016/j.coelec.2025.101726.
- N. Gondhiya, A. U. Rehman, D. Andreescu and S. Andreescu, Portable Electrochemical Sensors for Per- and Polyfluoroalkyl Substances: Design, Challenges, and Opportunities for Field Deployment, Curr. Opin. Electrochem., 2025, 53, 101725, DOI:10.1016/j.coelec.2025.101725.
- C. T. LeBarron, P. Shenoy and S. Hosseini, Recent Insights into Electrochemical Sensing of Per- and Polyfluoroalkyl Substances, ECS Sens. Plus, 2025, 4(3) DOI:10.1149/2754-2726/ade79d.
- E. O. Omotola, C. R. Ohoro, J. F. Amaku, J. Conradie, C. Olisah, K. G. Akpomie, A. Malloum, S. O. Akpotu, K. A. Adegoke and E. S. Okeke, Evidence of the Occurrence, Detection, and Ecotoxicity Studies of Perfluorooctanesulfonic Acid (PFOS) and Perfluorooctanoic Acid (PFOA) in Aqueous Environments, J. Environ. Health Sci. Eng., 2025, 23, 10, DOI:10.1007/s40201-025-00934-4.
- M. Zhou, D. Wei, J. Li, K. Fan and W. Wang, Low-Cost and Highly Sensitive Conductive Polymer-Based Lateral Flow Assay for per- and Polyfluoroalkyl Substances Detection, Innovation, 2025, 6, 100768, DOI:10.1016/j.xinn.2024.100768.
- R. B. Clark and J. E. Dick, Electrochemical Sensing of Perfluorooctanesulfonate (PFOS) Using Ambient Oxygen in River Water, ACS Sens., 2020, 5(11), 3591–3598, DOI:10.1021/ACSSENSORS.0C01894/ASSET/IMAGES/LARGE/SE0C01894_0006.JPEG.
- N. Karimian, A. M. Stortini, L. M. Moretto, C. Costantino, S. Bogialli and P. Ugo, Electrochemosensor for Trace Analysis of Perfluorooctanesulfonate in Water Based on a Molecularly Imprinted Poly(o-Phenylenediamine) Polymer, ACS Sens., 2018, 3(7), 1291–1298, DOI:10.1021/ACSSENSORS.8B00154/SUPPL_FILE/SE8B00154_SI_001.PDF.
- M. W. Glasscott, K. J. Vannoy, R. Kazemi, M. D. Verber and J. E. Dick, μ-MIP: Molecularly Imprinted Polymer-Modified Microelectrodes for the Ultrasensitive Quantification of GenX (HFPO-DA) in River Water, Environ. Sci. Technol. Lett., 2020, 7(7), 489–495, DOI:10.1021/ACS.ESTLETT.0C00341/ASSET/IMAGES/LARGE/EZ0C00341_0004.JPEG.
- D. Lu, D. Z. Zhu, H. Gan, Z. Yao, J. Luo, S. Yu and P. Kurup, An Ultra-Sensitive Molecularly Imprinted Polymer (MIP) and Gold Nanostars (AuNS) Modified Voltammetric Sensor for Facile Detection of Perfluorooctance Sulfonate (PFOS) in Drinking Water, Sens. Actuators, B, 2022, 352, 131055, DOI:10.1016/J.SNB.2021.131055.
- S. Hafeez, A. Khanam, H. Cao, B. P. Chaplin and W. Xu, Novel Conductive and Redox-Active Molecularly Imprinted Polymer for Direct Quantification of Perfluorooctanoic Acid, Environ. Sci. Technol. Lett., 2024, 11(8), 871–877, DOI:10.1021/acs.estlett.4c00557.
- N. Amin, J. Chen, Q. He, J. S. Schwartz and J. J. Wu, Ultra-Sensitive and Rapid Detection of Perfluorooctanesulfonic Acid by a Capacitive Molecularly-Imprinted-Polymer Sensor Integrated with AC Electrokinetic Acceleration, Sens. Actuators, B, 2024, 420, 136464, DOI:10.1016/J.SNB.2024.136464.
- J. J. C. Solís, S. Yin, M. Galicia, M. S. Ersan, P. Westerhoff and D. Villagrán, “Forever Chemicals” Detection: A Selective Nano-Enabled Electrochemical Sensing Approach for Perfluorooctanoic Acid (PFOA), Chem. Eng. J., 2024, 491, 151821 Search PubMed.
- R. Khan, Z. O. Uygun, D. Andreescu and S. Andreescu, Sensitive Detection of Perfluoroalkyl Substances Using MXene-AgNP-Based Electrochemical Sensors, ACS Sens., 2024, 9(6), 3403–3412, DOI:10.1021/acssensors.4c00776.
- Y. H. Cheng, D. Barpaga, J. A. Soltis, V. Shutthanandan, R. Kargupta, K. S. Han, B. P. McGrail, R. K. Motkuri, S. Basuray and S. Chatterjee, Metal-Organic Framework-Based Microfluidic Impedance Sensor Platform for Ultrasensitive Detection of Perfluorooctanesulfonate, ACS Appl. Mater. Interfaces, 2020, 12(9), 10503–10514, DOI:10.1021/ACSAMI.9B22445/SUPPL_FILE/AM9B22445_SI_001.PDF.
- Q. Tian, S. Chen, M. Shi, T. Gao, M. Zhang, C. Liao, X. Li, Q. Dong and C. Wang, Fluorine-Functionalized MOF Modified GCE for Highly Sensitive Electrochemical Detection of Persistent Pollutant Perfluorooctanoic Acid, Sens. Actuators, B, 2024, 404, DOI:10.1016/j.snb.2024.135309.
- C. Fang, Z. Chen, M. Megharaj and R. Naidu, E 9 Potentiometric Detection of AFFFs Based on MIP, Environ. Technol. Innovation, 2016, 5, 52–59, DOI:10.1016/J.ETI.2015.12.003.
- G. Moro, D. Cristofori, F. Bottari, E. Cattaruzza, K. De Wael and L. M. Moretto, Redesigning an Electrochemical MIP Sensor for PFOS: Practicalities and Pitfalls, Sensors, 2019, 19(20), 4433, DOI:10.3390/S19204433.
- G. J. Islam and D. W. M. Arrigan, Voltammetric Selectivity in Detection of Ionized Perfluoroalkyl Substances at Micro-Interfaces between Immiscible Electrolyte Solutions, ACS Sens., 2022, 7(10), 2960–2967, DOI:10.1021/ACSSENSORS.2C01100/SUPPL_FILE/SE2C01100_SI_001.PDF.
- N. F. Suhaimi, S. N. A. Baharin, N. A. Jamion, Z. Mohd Zain and K. P. Sambasevam, Polyaniline-Chitosan Modified on Screen-Printed Carbon Electrode for the Electrochemical Detection of Perfluorooctanoic Acid, Microchem. J., 2023, 188, 108502, DOI:10.1016/J.MICROC.2023.108502.
| This journal is © The Royal Society of Chemistry 2026 |