Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Correction: Polyterrylenes: synthesis and regioregularity effect on p-type charge transport and deep-red light photodetection in OFETs
Chitrak Ghosh†
a,
Minji Chung†b,
Hayeong Parkb,
Aniket Jitendra Talrejaa,
Ullrich Scherfc,
Joon Hak Oh*b and
Suman Kalyan Samanta*a
aDepartment of Chemistry, Indian Institute of Technology Kharagpur, Kharagpur 721302, India. E-mail: sksamanta@chem.iitkgp.ac.in
bSchool of Chemical and Biological Engineering, Institute of Chemical Processes, Seoul National University, Seoul 08826, Republic of Korea. E-mail: joonhoh@snu.ac.kr
cMacromolecular Chemistry Group (buwmakro), Wuppertal Center for Smart Materials and Systems (CM@S), D-42119 Wuppertal, Germany
First published on 24th March 2026
Abstract
Correction for ‘Polyterrylenes: synthesis and regioregularity effect on p-type charge transport and deep-red light photodetection in OFETs’ by Chitrak Ghosh et al., Chem. Sci., 2026, 17, 2703–2711, https://doi.org/10.1039/D5SC06452J.
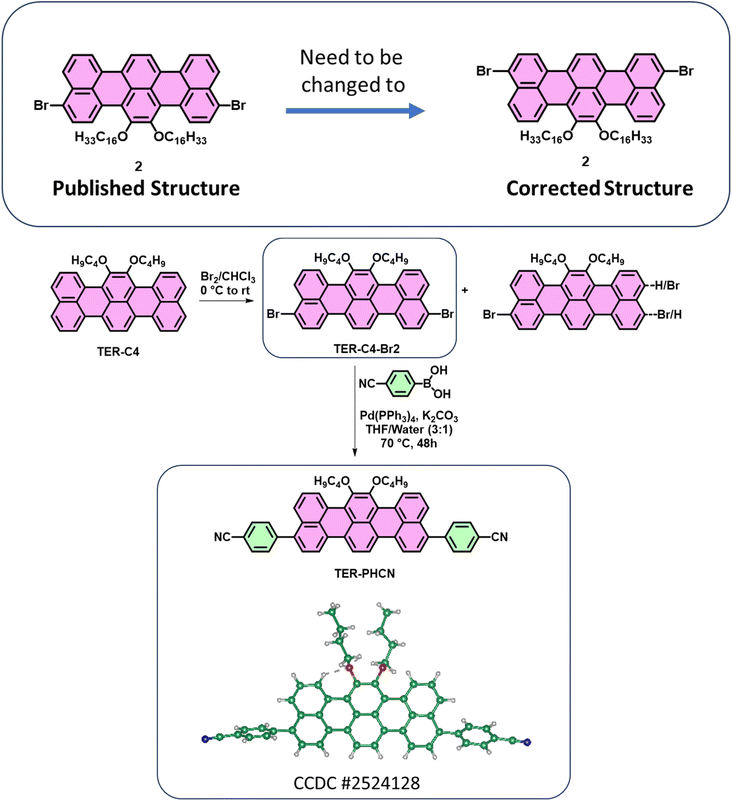
The authors regret that the chemical structure of compound 2 was incorrect in the original paper.
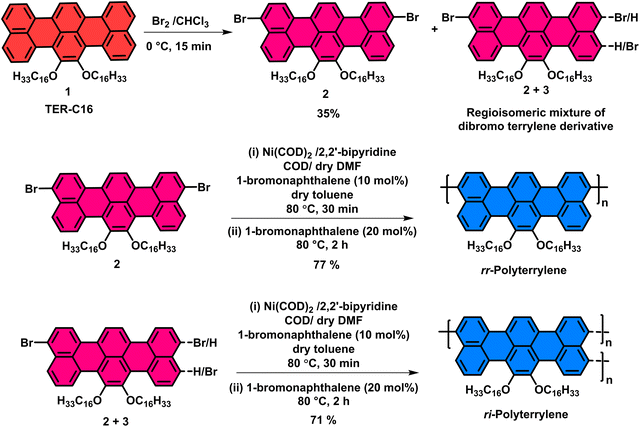
For the reported compound 2, the authors were not able to obtain a single crystal despite several attempts. However, during a follow-up work (shown below) using a shorter alkyl chain (n-butyl), and further reaction of the dibromo compound to generate the p-cyanobenzene derivative (TER-PHCN), a single crystal was obtained. The single crystal of TER-PHCN (CCDC # 2524128) showed that the bromines in TER-C4-Br2 are located at different peri-positions compared to the structure that was originally assigned in the published paper for compound 2. Based on this crystal structure, the authors realized that the published structure of compound 2 needs to be corrected.
Synthesis of TER-PHCN and its SCXRD structure:
Both the published and corrected structures of compound 2 are symmetric and would produce identical NMR spectra. The published structure of compound 2 was the most probable based on literature reports on perylene-based systems (Ref: Chem. Eur. J. 2017, 23, 9419–9424) and the electron-donating mesomeric effect of oxygen atoms. However, it is surprising and unexpected that the bromine atoms are located at the other peri-positions in TER-C4-Br2. Since the methods of bromination and isolation of the pure products were the same for both C4 and C16 chains, the authors believe that the published structure of compound 2 will adopt the location of bromine atoms similar to the TER-C4-Br2. Although the authors did not obtain the crystal structure for compound 2, based on the crystal structure of TER-PHCN (CCDC # 2524128), the published structure of compound 2 can be changed to the corrected structure.
Corrections to the main manuscript
Importantly, these changes in structure do not affect any spectral changes, observations, significance, direction, or the conclusions of the original article. All the compounds and their spectra, experimental data, and tables remain the same.The figures where the structures of compound 2 and its polymer appeared need to be replaced with the corrected structure, as given here (Graphical abstract image, Fig. 1, Scheme 1, Fig. 3).
Graphical abstract image:
 | ||
| Fig. 1 Schematic representation of functionalization of terrylene. Vial pictures showing the solutions of compounds 1, 2 and rr-Polyterrylene in chloroform under daylight. | ||
In the ‘Theoretical studies’ section, two sentences need to be modified slightly:
‘The dihedral angle between two terrylene subunits in the peri-position is about 76° as the other two remaining hydrogen atoms at the peri-position of terrylene prevent polyterrylene from achieving a fully planar polymer configuration (Fig. 3a).’
‘In context with DFT calculations, polyterrylene showed a slightly uplifted HOMO and low-lying LUMO energy levels compared to 7,8-bis(methyloxy)terrylene, delineating a modest reduction of the band gap of 0.14 eV, which correlates well with their experimental bandgap differences.’
Additional corrections and the new crystal structure data have been added to an updated version of the SI (Fig. S3 and S10, Table S2).The Royal Society of Chemistry apologises for these errors and any consequent inconvenience to authors and readers.
Footnote |
| † CG and MC contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |