Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Correction: Enantioselective C(sp3)–H bond functionalization enabled by CpxM(III) catalysis (M = Co, Rh, Ir)
Shu-Bin
Mou†
ab,
Mu-Peng
Luo†
b,
Feifei
Fang†
b,
Shi
Cao
*b,
Dong
Wu
*a and
Shou-Guo
Wang
*b
aComputer Aided Drug Discovery Center, Zhuhai Institute of Advanced Technology, Chinese Academy of Sciences, Zhuhai 519003, P. R. China. E-mail: wudong@ziat.ac.cn
bCollege of Chemistry and Environmental Engineering, Shenzhen University, Shenzhen 518060, P. R. China. E-mail: shicaorganic@szu.edu.cn; shouguo.wang@szu.edu.cn
First published on 24th February 2026
Abstract
Correction for ‘Enantioselective C(sp3)–H bond functionalization enabled by CpxM(III) catalysis (M = Co, Rh, Ir)’ by Shu-Bin Mou et al., Chem. Sci., 2026, 17, 2990–3004, https://doi.org/10.1039/d5sc08394j.
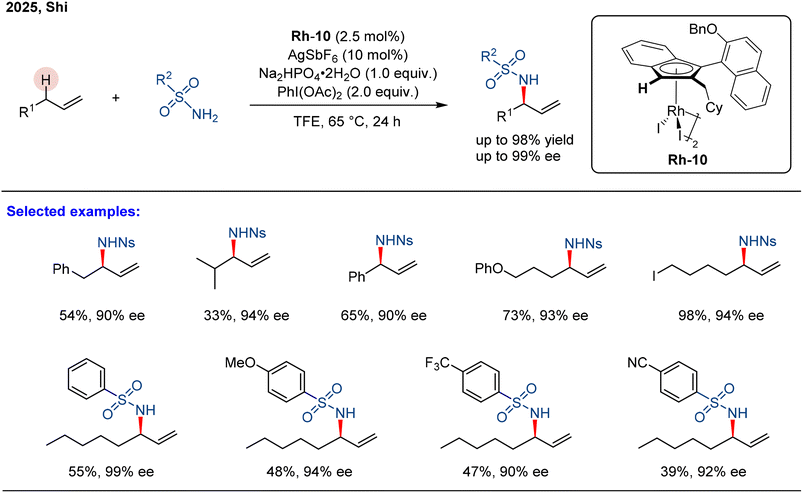
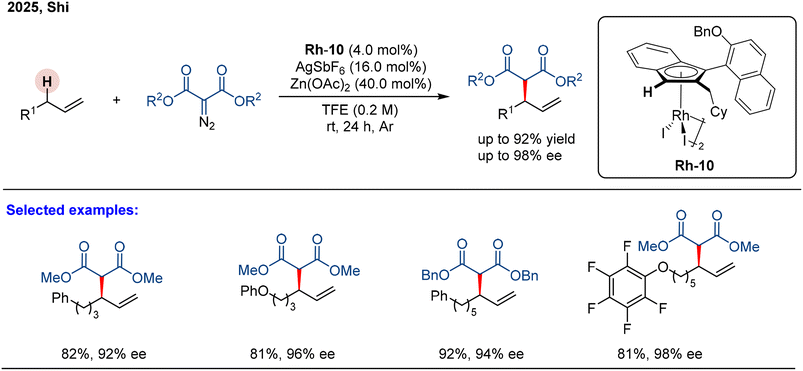
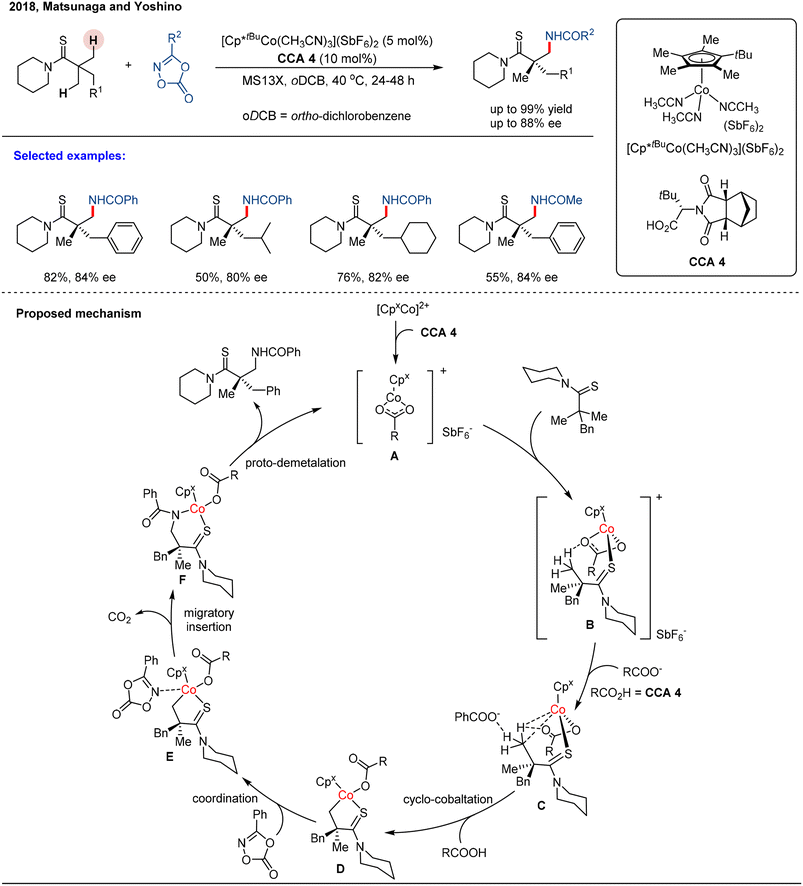
The authors regret that three of the schemes in the published manuscript (Schemes 16, 20 and 21) had errors present in them. The corrected schemes are given below.
The Royal Society of Chemistry apologises for these errors and any consequent inconvenience to authors and readers.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |