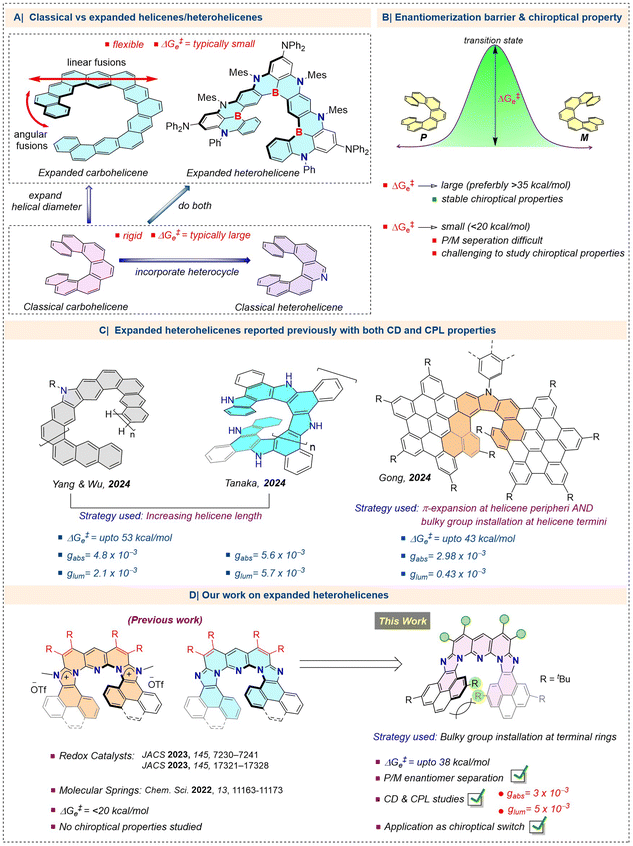
Open Access Article
Open Access ArticleInducing configurational stability in inherently flexible expanded heterohelicenes and unlocking stimuli-responsive chiroptical switching
Manisha
Pal†
 a,
Pirudhan
Karak†
a,
Pirudhan
Karak†
 a,
Debranjan
Hati
b,
Mrityunjay
Giri
a,
Debranjan
Hati
b,
Mrityunjay
Giri
 a,
Subi J.
George
a,
Subi J.
George
 *b and
Joyanta
Choudhury
*b and
Joyanta
Choudhury
 *a
*a
aOrganometallics & Smart Materials Laboratory, Department of Chemistry, Indian Institute of Science Education and Research (IISER) Bhopal, Bhopal 462 066, India. E-mail: joyanta@iiserb.ac.in
bNew Chemistry Unit, School of Advanced Materials, Jawaharlal Nehru Centre for Advanced Scientific Research (JNCASR), Bangalore 560064, India. E-mail: george@jncasr.ac.in
First published on 13th April 2026
Abstract
“π-Expanded” (hetero)helicenes have attracted special attention by virtue of their increased helical diameter, enlarged cavity, structural flexibility, and dynamics. However, their structural flexibility results in low enantiomerization barriers (ΔG‡e < 20 kcal mol−1), imposing a significant challenge to their enantiomeric separation, and hence, the study of their chiroptical properties. Therefore, there is a rising interest in increasing the ΔG‡e of “π-expanded” (hetero)helicenes to enable their chiral separation and chiroptical study. Previously, we disclosed a new type of expanded poly-aza[9]helicenes that possess low ΔG‡e (17–18 kcal mol−1) values at 25 °C, and thus, their P and M enantiomers are difficult to separate, suggesting the necessity of suitable structural modification. Herein, addressing this problem, we report the synthesis of configurationally-stable analogues of the expanded poly-aza[9]helicenes by introducing bulky tert-Bu groups at the terminal overlapping rings to restrict ring-flapping. The ΔG‡e values for these compounds are significantly higher (>35 kcal mol−1), allowing P/M enantiomer separation via chiral-HPLC. They show a gabs of ±3 × 10−3 and glum of ±5 × 10−3, and have stable chiroptical properties at elevated temperatures. Moreover, the basic imidazole units within these novel poly-aza[9]helicenes enable pH-responsive chiroptical switching. Overall, the stable chiroptical property and stimuli-responsive chiroptical function of this class of expanded poly-aza[9]helicenes could make them promising candidates for potential chirality-based applications.
Introduction
Helicenes are among the class of “molecules in distress”,1 as they “suffer” from significant steric strain between the terminal rings of their winding structure, leading to a conformational distortion of the π-conjugated polycyclic arene framework. However, this “suffering” is complemented by chirality, one of the most intriguing properties in chemical structures, stemming from the handedness of their helical shape. It is this chirality that endows the helicenes with fascinating chiroptical properties such as circular dichroism (CD) and circularly polarized luminescence (CPL). A variety of emerging (opto)electronic applications are associated with efficient CD/CPL properties, and consequently, there is an extensive research interest in developing helicene compounds that are suitable as materials for such applications.2 For practical applications, configurational stability with a high racemization barrier is the basic requirement.3 There have been several developments in the world of archetypal helicenes (possessing only angularly/ortho-fused rings along the helical backbone) toward isolating enantiopure compounds and studying their chiroptical properties (Fig. 1A), which have led to cutting-edge applications in materials science.1b,c,4 Very recently, a novel class of helicenes, called “expanded” (possessing both linearly- and angularly-fused rings along the helical rim) carbohelicenes (all-carbon helicenes)/heterohelicenes (heteroatom/s-containing helicenes) have been introduced in this field, with enriched structural diversity, modulated electronic and optoelectronic properties, dynamic chirality, and superior chiroptical properties (Fig. 1A).5 However, the inherent flexibility in these expanded versions, owing to their enlarged helical diameter, complicates the separation/isolation of the enantioenriched compounds and the study of their chiroptical properties because of their low ΔG‡e values (ΔG‡e < 20 kcal mol−1) (Fig. 1A and B). Therefore, commonly employed strategies to obtain enantiomerically pure expanded helicenes at ambient temperatures with high ΔG‡e values (ΔG‡e > 35 kcal mol−1), are: (a) increasing the length of the helical chain, (b) π-extension at the periphery of the helicenes, (c) multiplexing helicenic motifs on the same parent helicene backbone, and (d) bulky group substitution at the internal or terminal rings of the helical backbone.5e,6 However, the examples of enantiomerically pure expanded carbohelicenes are limited,7 and expanded heterohelicenes with a high racemization barrier have been very rare until now.5f,8,9 Just recently, in 2024, three independent studies by Yang and Wu,5f Tanaka,8a and Gong,9 reported expanded heterohelicenes with high ΔG‡e values (Fig. 1C). The previously mentioned strategies have been exploited in these works to achieve the required racemization barrier value and hence excellent absorption and luminescence dissymmetry factors. Notably, all of these expanded heterohelicenes possess carbazole as the heterocycle in their helical backbone. Systems incorporating electronically diverse heterocycles are highly desired in order to explore their modulated CD/CPL properties and new chiroptical functions such as stimuli-responsive chiroptical switches (vide infra).Recently, we developed a double ‘rollover π-expansion’ (RoPE) strategy-enabled synthesis of an intriguing class of neutral and cationic “expanded” poly-azahelicenes, embedded with an imidazole/imidazolium-fused pyridine-based mixed heterocyclic scaffold (Fig. 1D).10 While the cationic systems were exploited as a novel class of redox-active electrocatalysts for the CO2-reduction reaction (CO2RR) and hydrogen evolution reaction (HER),11 both the neutral and cationic compounds were used as stimuli (pH and light)-controlled soft molecular springs due to their flexible helical backbone.10b The same flexibility of these expanded heterohelicenes prevents configurational stability (calculated ΔG‡e value < 20 kcal mol−1 at 25 °C). We endeavoured to achieve the configurationally stable version of our expanded heterohelicenes by introducing bulky tert-butyl groups at the overlapping terminal rings of the helical rim (strategy (d) mentioned above) (Fig. 1D). Consequently, we successfully demonstrated the separation and isolation of the P and M enantiomers and evaluated the chiroptical properties, and finally, to showcase a potential application, uncovered a highly reversible on/off chiroptical switching function with the enantioenriched compounds by capitalizing on the embedded pH-responsive imidazole moieties (Fig. 1D).
Results and discussion
Synthesis
The precursor for the target helicenes, 2,6-bis(2,7-di-tert-butyl-9H-pyreno[4,5-d]imidazole-9-yl)pyridine (PYBIM) was synthesized by the reaction of 2,6-difluoropyridine with 2,7-di-tert-butyl-9H-pyreno[4,5-d]imidazole under basic conditions (Schemes S1–S4; see SI for details). The targeted expanded poly-aza[9]helicenes, HELI-1 and HELI-2, were obtained in good yields (62–73%) by applying our ‘rollover π-expansion’ protocol with the catalyst [RhCp*Cl2]2 on the precursor PYBIM with two different internal alkynes (Fig. 2A). The compounds HELI-1 and HELI-2 were both well characterized by NMR spectroscopy and mass spectrometry studies (see SI for details).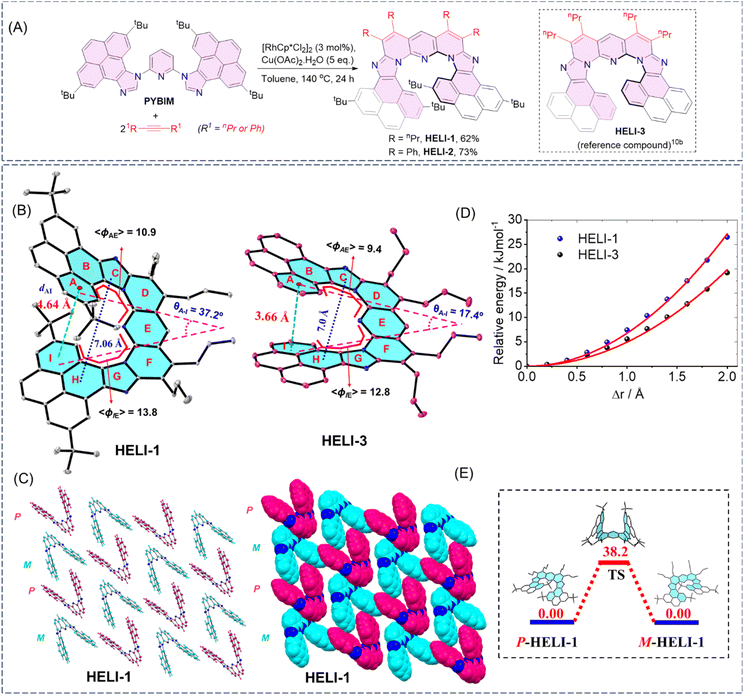 | ||
| Fig. 2 (A) Synthesis of π-expanded poly-aza[9]helicenes HELI-1 and HELI-2. The reference non-tBu analogue compound HELI-3 (reported previously by our group10b) is also shown. (B) Crystal structures and comparison of key metric parameters of HELI-1 (CCDC 2485128) and HELI-3. (C) Crystal packing and space-filling diagrams of HELI-1, showing the arrangement of the P/M isomers in the crystalline state. (D) Spring constant plots of HELI-1 and HELI-3.10b (E) Enantiomerization energy barrier (kcal mol−1) of HELI-1, computed via the DFT method (B3LYP/6-311g(2d,p) level of theory). TS = transition state. See SI for details. | ||
Crystal structure
The helical structure of HELI-1 (Fig. 2B) was unambiguously confirmed by single-crystal X-ray diffraction (SCXRD) analysis, using suitable crystals grown by diffusing hexane into a dichloromethane solution of the compound at room temperature. The crystal packing profile revealed alternating stacks of both the P and M enantiomers aligned along the vertical axis, resulting in a racemic columnar structure (Fig. 2C). At the molecular level, analysis revealed that the helical pitch (dAI), the vertical distance between the two terminal rings of HELI-1 was 4.64 Å, which was much higher than that of our previously reported non-tBu expanded heterohelicene analogue HELI-3 (dAI = 3.66 Å).10b The intersecting angle (θAI) between the two planes passing through the two terminal rings was 37.2°, which is markedly larger than that of HELI-3 (17.4°). The torsion angles of two helicinic wings were 10.9° and 13.8°, exceeding those observed for HELI-3 (9.4° and 12.8°). The higher values of the helical pitch, angle, and torsion angles in HELI-1 were attributed to the presence of the bulky tert-butyl groups on the overlapping terminal rings of the helicene rim, which imposed severe steric hindrance between these two rings. Although we could not obtain high-quality single crystals suitable for high-quality diffraction data for HELI-2 despite several attempts, the gross structure showed similar features to those of HELI-1 (see SI for details). The above structural characteristics suggested the incorporation of the desired rigidity in the inherently flexible helicenic backbone of the expanded heterohelicenes HELI-1 and HELI-2. Indeed, the molecular spring constant (k) value of HELI-1 calculated via the density functional theory (DFT) method (Fig. 2D and S5 in SI) was found to be 2.25 N m−1, which was significantly higher than that (k = 1.64 N m−1) of HELI-3, indicating that the tBu groups induced reduction of the flexibility of the helical rim in the former compound. Overall, it appeared that the presence of the bulky tBu groups at the two terminal overlapping rings would be able to restrict the easy flapping of the terminal rings to a great extent, and therefore, a high enantiomerization barrier was anticipated. Therefore, we investigated the enantiomerization barrier for HELI-1via DFT using dichloromethane solvent at 298.15 K (Fig. 2E). The ΔG‡e value was found to be 38.2 kcal mol−1, which was expectedly much higher compared to that of HELI-3 (ΔG‡e = 16.5 kcal mol−1 at 25 °C), as well as those of many other reported expanded carbo/heterohelicenes, such as expanded [13]-helicene5c (ΔG‡e theory = 13.0 kcal mol−1 at 25 °C), the aza-bora[9]-helicene7a (ΔG‡e theory = 14.3 kcal mol−1 at 25 °C) and [7]-heliphene12 (ΔG‡e experimental = 12.6 kcal mol−1 at −27 °C). Similarly, the DFT -calculated enantiomerization barrier of HELI-2 was found to be 39.1 kcal mol−1 (Fig. S4, SI), which is comparable to that of HELI-1.Chiroptical properties
The sufficiently high ΔG‡e value indicated the possibility of chiral resolution of the P and M enantiomers of these new expanded poly-[9]azahelicenes. We first chose HELI-2 for investigating chiral resolution and follow-up chiroptical properties. After a thorough screening of conditions, the P and M enantiomers of HELI-2 were successfully resolved via chiral HPLC using a Daicel CHIRALPAK IA-3 column and n-hexane/iPrOH (90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, v/v) as the eluent (Fig. S7, SI). The chiroptical properties and performances of the isolated P/M enantiomers were evaluated in both solution and solid state (thin film) using both CD and CPL studies.
10, v/v) as the eluent (Fig. S7, SI). The chiroptical properties and performances of the isolated P/M enantiomers were evaluated in both solution and solid state (thin film) using both CD and CPL studies.
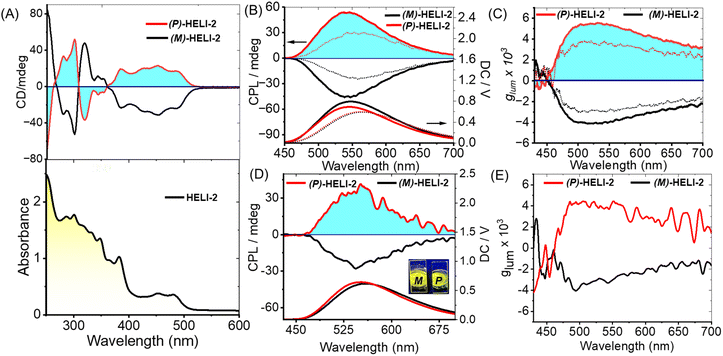
The UV-vis absorption and CD spectra of the isolated enantiomers (P/M) of HELI-2 are shown in Fig. 3A. The compound showed intense absorption bands at 350–375 nm and 430–500 nm (ε = 54![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 510 M−1 cm−1 at 355 nm; ε = 19
510 M−1 cm−1 at 355 nm; ε = 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 M−1 cm−1 at 445 nm in DCM; Fig. S9, SI). The absolute configuration of P/M-HELI-2 was determined by comparing the experimental and simulated CD spectra (calculated using the DFT method, Fig. S65, SI). The CD spectra of the two enantiomers as expected, showed mirror-imaged bisignate signals between 250 nm and 550 nm (the P and M isomers showed positive and negative ECD, respectively, at the absorption onset). The P/M-HELI-2 enantiomers displayed high absorption dissymmetry factors (gabs = Δε/ε) of ±3 × 10−3 in hexane at 425 nm (Fig. S15, SI), similar to those of many large π-extended helicenes.5e The linear dichroism (LD) experiments (Fig. S17 in the SI) ruled out any LD contribution to the observed chiroptical properties, implying that there is no macroscopic alignment of the chromophore under the experimental conditions in either hexane or dichloromethane. This is also supported by rotation-dependent CD spectra (Fig. S16 in SI), where similar spectral features were observed with rotation. HELI-2 showed strong fluorescence in the 490–680 nm range with λmax = 560 nm in DCM solution (Fig. S9b, SI) and λmax = 555 nm in hexane solution. The fluorescence quantum yield was calculated to be 0.38 and 0.30 in DCM and hexane, respectively (Table S4, SI). In the DC spectra, λmax = 560 nm matches the emission spectra (Fig. 3B). The P/M-HELI-2 showed mirror image CPL spectra with λmax = 550 nm in hexane and λmax = 560 nm in DCM, which matches both the emission and DC spectra (Fig. 3B). The CPL dissymmetry factor (glum) was measured to be ±5 × 10−3 in hexane, and ±3 × 10−3 in dichloromethane (Fig. 3C), which seems to be promisingly high and comparable to those of other relatively rigid carbo/heterohelicenes.5e,f,9,13 As HELI-2 was sparingly soluble in hexane, molecular aggregation occurred, which resulted in a higher glum value.13 The dynamic light scattering (DLS) data (Fig. S18 in SI) also suggested that the particle size is larger in hexane solution compared to that in DCM. Interestingly, the P and M isomers of HELI-2 also showed emission in the solid-state (Fig. 3D), and accordingly, they displayed mirror-image CPL profiles in the solid-state (drop-cast film), with a high glum value of ±4 × 10−3 (Fig. 3E). Thus, based on these encouraging chiroptical performance parameters, the P and MHELI-2 enantiomers exhibited potential for further practical solid-state applications.
800 M−1 cm−1 at 445 nm in DCM; Fig. S9, SI). The absolute configuration of P/M-HELI-2 was determined by comparing the experimental and simulated CD spectra (calculated using the DFT method, Fig. S65, SI). The CD spectra of the two enantiomers as expected, showed mirror-imaged bisignate signals between 250 nm and 550 nm (the P and M isomers showed positive and negative ECD, respectively, at the absorption onset). The P/M-HELI-2 enantiomers displayed high absorption dissymmetry factors (gabs = Δε/ε) of ±3 × 10−3 in hexane at 425 nm (Fig. S15, SI), similar to those of many large π-extended helicenes.5e The linear dichroism (LD) experiments (Fig. S17 in the SI) ruled out any LD contribution to the observed chiroptical properties, implying that there is no macroscopic alignment of the chromophore under the experimental conditions in either hexane or dichloromethane. This is also supported by rotation-dependent CD spectra (Fig. S16 in SI), where similar spectral features were observed with rotation. HELI-2 showed strong fluorescence in the 490–680 nm range with λmax = 560 nm in DCM solution (Fig. S9b, SI) and λmax = 555 nm in hexane solution. The fluorescence quantum yield was calculated to be 0.38 and 0.30 in DCM and hexane, respectively (Table S4, SI). In the DC spectra, λmax = 560 nm matches the emission spectra (Fig. 3B). The P/M-HELI-2 showed mirror image CPL spectra with λmax = 550 nm in hexane and λmax = 560 nm in DCM, which matches both the emission and DC spectra (Fig. 3B). The CPL dissymmetry factor (glum) was measured to be ±5 × 10−3 in hexane, and ±3 × 10−3 in dichloromethane (Fig. 3C), which seems to be promisingly high and comparable to those of other relatively rigid carbo/heterohelicenes.5e,f,9,13 As HELI-2 was sparingly soluble in hexane, molecular aggregation occurred, which resulted in a higher glum value.13 The dynamic light scattering (DLS) data (Fig. S18 in SI) also suggested that the particle size is larger in hexane solution compared to that in DCM. Interestingly, the P and M isomers of HELI-2 also showed emission in the solid-state (Fig. 3D), and accordingly, they displayed mirror-image CPL profiles in the solid-state (drop-cast film), with a high glum value of ±4 × 10−3 (Fig. 3E). Thus, based on these encouraging chiroptical performance parameters, the P and MHELI-2 enantiomers exhibited potential for further practical solid-state applications.
Similarly, HELI-1 exhibited absorption bands at 320–370 nm and 420–500 nm (ε = 59![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 M−1 L cm−1 at 345 nm; ε = 17
900 M−1 L cm−1 at 345 nm; ε = 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 M−1 L cm−1 at 435 nm in DCM; Fig. S8, SI). The chiral resolution of HELI-1 was also achieved, and the corresponding P and M enantiomers displayed mirror-image CD and CPL spectra (Fig. S12 and S13 SI). For HELI-1, the gabs and glum values are found to be ±4 × 10−3 and ±5 × 10−3, respectively, in DCM (Fig. S12–S14, SI).
600 M−1 L cm−1 at 435 nm in DCM; Fig. S8, SI). The chiral resolution of HELI-1 was also achieved, and the corresponding P and M enantiomers displayed mirror-image CD and CPL spectra (Fig. S12 and S13 SI). For HELI-1, the gabs and glum values are found to be ±4 × 10−3 and ±5 × 10−3, respectively, in DCM (Fig. S12–S14, SI).
Temperature-dependent chiroptical properties
The stable chiroptical properties of these compounds at high temperatures reinforced their suitability for further practical applications. As hexane or DCM are low-boiling solvents, toluene and ortho-dichlorobenzene were used for studying their temperature-dependent chiroptical properties. Before performing the experiment, we confirmed that HELI-2 was in monomeric form in toluene via a DLS study (Fig. S18 in SI), and ensured that similar chiroptical properties to those in DCM solvent were retained (Fig. S23 in SI). The variation of the CD signal at 450 nm as a function of temperature was measured in toluene with a temperature gradient of 10 °C/50 min (Fig. S24–S29 in SI). No change in the CD signal was observed up to 75 °C with increasing temperature (Fig. 4A), which supported the high ΔG‡e. In CP-OLEDs, chiral emitters often face heating-related issues that can affect their stability and performance during operation. Therefore, the retention of CPL properties at high temperatures is essential. Thus, we also measured variable-temperature CPL, and no change in λmax (550 nm) or CPL signal intensity was evident for either of the isomers (Fig. 4B, and S38–S43 in SI) in the 25–75 °C range. The glum value also remained constant up to 75 °C (Fig. S44 in SI). It is notable that we did not observe the decay of the CD or CPL signal in toluene solution up to 75 °C. Similarly, when ortho-dichlorobenzene was used as the solvent, the CD and CPL properties were retained up to 90 °C (Fig. S30–S37 and S45–S52 in SI).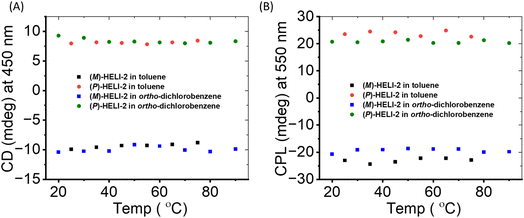 | ||
| Fig. 4 (A) Change in the CD signal (at 450 nm) and (B) CPL signal (at 550 nm) of (P/M)-HELI-2 with temperature in toluene and ortho-dichlorobenzene solution (concentration = 8 µM). | ||
In addition, we verified the stability of the representative chiral (M)-HELI-2 under constant external heating at 160 °C for up to 12 h, using ortho-dichlorobenzene as the solvent, and monitored the CD signal at 2-hour intervals. Notably, no detectable decrement in the CD signal was observed, which further confirmed the high racemization barrier value of these expanded helicenes (Fig. S54, SI). On that note, previously, Nowak-Król and coworkers reported azaborathia[9]helicene H1-Me4, with a racemization barrier of 39.1 kcal mol−1, but no racemization was observed at 170 °C in the solvent ortho-dichlorobenzene.2f
Furthermore, the half-life (t1/2) of the helical inversion process of HELI-2 was evaluated at different temperatures using the DFT method (Table S2, SI). At 25 °C, the t1/2 value was found to be very high (∼108 years); only at very high temperatures, such as at 300 °C, the t1/2 value is around 5.34 minutes. This clearly indicates that the racemization of HELI-2 can be achieved only at very high temperatures, which is experimentally very challenging.
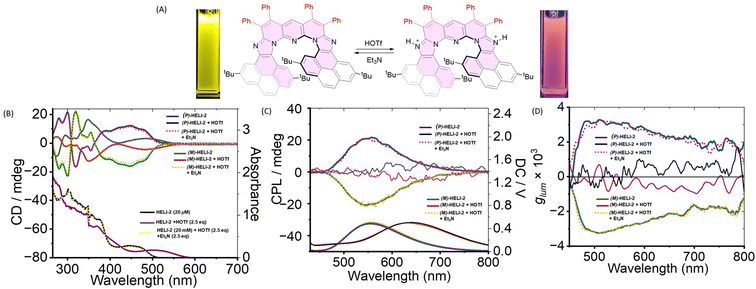
Stimuli-responsive chiroptical switching
As mentioned earlier, owing to the presence of the embedded pH-responsive imidazole moieties, this new class of expanded poly-aza[9]-helicenes opened up the opportunity to explore a stimuli (acid/base)-controlled on/off chiroptical switching function with the P/M enantiomers.14 Thus, consecutive treatment of an acid (triflic acid, CF3SO3H) and a base (triethylamine, Et3N) with a solution of HELI-2 (Fig. 5A) was first monitored via UV-vis absorption and fluorescence spectroscopy (Fig. S55, SI). Upon protonation, both the absorption and the emission bands of HELI-2 red-shifted from 446 nm to 514 nm and from 560 nm to 663 nm, respectively, as the LUMO was stabilized upon protonation (Fig. S62, SI). In the TD-DFT calculated UV-vis spectra, a similar red shift was present (Fig. S63, SI). Moreover, the protonation process was also monitored using 1H NMR spectroscopic studies (Fig S60, SI). The emission intensity of the protonated HELI-2 was reduced fivefold (Fig. S55 in SI). The original spectral profiles (both absorption and emission profiles) could be regenerated upon deprotonation by the addition of triethylamine (Fig. 5B and S55 in SI). Next, the pH-responsive reversible behavior was examined with the chiral P/M-HELI-2 isomers and monitored using CD and CPL spectroscopy. The CD spectra of the P/M-HELI-2 isomers also showed changes, with red-shifted (by 30–40 nm) peaks. Less-intense bands were observed at 350 nm and 488 nm for the protonated P/M-HELI-2 (Fig. 5B). Upon protonation, a red shift in DC spectra was observed (Fig. 5C). The CPL signal at λmax = 560 nm in DCM was found to be shifted to λmax ∼ 655 nm with much less CPL intensity when P/M-HELI-2 isomers were treated with triflic acid (Fig. 5C). Interestingly, upon the addition of triethylamine, the CPL characteristics were reversibly retained (Fig. 5C), very similar to previously reported N-doped octagon-containing hexabenzocoronene (HBC) system.14c The acid–base responsive reversible switching behavior was also confirmed by monitoring the glum parameter of both the enantiomers (Fig. 5D); upon protonation, the glum value was found to be reduced to ∼10−4. The above results demonstrated a significant stimulus-controlled chiroptical switching capability of chiroptically stable enantiomers of this new class of expanded poly-azahelicenes, which could be useful for further specialized applications in the field.In addition, for practical applications, the main requirement is solid-state functionality.15 Thus, we investigated the acid-base-responsive CPL switching of HELI-2 in the solid state (drop-cast film). Upon exposure to acid (triflic acid) vapour, the solid-state CPL intensity was found to be decreased significantly. The CPL intensity was recovered upon exposure to base (triethylamine) vapour (Fig. S61, SI), indicating pH control over the CPL output in the solid state. These results demonstrate the viability of stimuli-responsive CPL modulation in solid-state conditions relevant to device applications.
Conclusions
In conclusion, configurationally stable “π-expanded” 9-azahelicenes with a significantly enhanced enantiomerization energy barrier (>38 kcal mol−1) have been synthesized conveniently using a simple catalytic methodology. These unique helicenes can be easily separated into P and M isomers under ambient conditions. The optically pure chiral helicenes exhibited a high absorption dissymmetry factor gabs (±3 × 10−3 in solution) and luminescence dissymmetry factor glum (±5 × 10−3 in solution and ±4 × 10−3 in solid state), surpassing even those of classical [7]-helicenes. Additionally, the imidazole rings within the helicene core of these molecules endowed them with acid/base-responsive chiroptical switching properties. Crucially, the CPL properties exhibited thermal stability up to 160 °C, indicating their suitability for potential chiral applications.Author contributions
M. Pal: methodology, investigation, formal analysis, data curation, visualization, writing – original draft; P. Karak: methodology, investigation, formal analysis, data curation, visualization, writing – original draft; D. Hati: investigation, formal analysis, data curation; M. Giri: methodology, investigation; S. J. George: resources, supervision; J. Choudhury: conceptualization, supervision, resources, project administration, funding acquisition, writing. M. Pal, and P. Karak contributed equally to this work.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2485128 contains the supplementary crystallographic data for this paper.16All data of this research work are available in the manuscript and in the supplementary information (SI). Supplementary information: experimental procedures, computational details, and spectral details. See DOI: https://doi.org/10.1039/d6sc02610a.
Acknowledgements
J. C. thanks the Anusandhan National Research Foundation (ANRF; erstwhile Science and Engineering Research Board, SERB), Government of India (Grant No. CRG/2022/005562), and IISER Bhopal for generous financial support. S. J. G. thanks the ANRF JC Bose Grant (Grant no.: ANRF/2025/02766). M. P. thanks Ministry of Education, Government of India for the award of the Prime Minister's Research Fellowship (PMRF). P. K. acknowledges the Integrated PhD fellowship from IISER Bhopal. D. H. thanks JNCASR for fellowship. The authors acknowledge the extensive use of NMR, Mass, and SCXRD facilities of CIF, IISER Bhopal. The authors gratefully acknowledge the High-Performance Computation (HPC) facility of IISER Bhopal for DFT calculations. The authors would also like to thank the Department of Science and Technology, Government of India for awarding the DST-FIST grant (SR/FST/CS-II/2023/312) to support NMR facilities at IISER Bhopal.References
- (a) R. Hoffmann and H. Hopf, Angew. Chem., Int. Ed., 2008, 47, 4474–4481 CrossRef CAS PubMed; (b) M. Gingras, G. Félix and R. Peresutti, Chem. Soc. Rev., 2013, 42, 1007–1050 RSC; (c) M. Gingras, Chem. Soc. Rev., 2013, 42, 1051–1095 RSC.
- (a) K. Dhbaibi, L. Favereau and J. Crassous, Chem. Rev., 2019, 119, 8846–8953 CrossRef CAS PubMed; (b) L. Zhang, H.-X. Wang, S. Li and M. Liu, Chem. Soc. Rev., 2020, 49, 9095–9120 RSC; (c) V. Kumar, H. J. Bharathkumar, S. D. Dongre, R. Gonnade, K. Krishnamoorthy and S. S. Babu, Angew. Chem., Int. Ed., 2023, 62, e202311657 CrossRef CAS PubMed; (d) S. D. Dongre, G. Venugopal, V. Kumar, A. Badrinarayan Jadhav, J. Kumar and S. Santhosh Babu, Angew. Chem., Int. Ed., 2024, e202420767 Search PubMed; (e) F. Zhang, V. Brancaccio, F. Saal, U. Deori, K. Radacki, H. Braunschweig, P. Rajamalli and P. Ravat, J. Am. Chem. Soc., 2024, 146, 29782–29791 CrossRef CAS PubMed; (f) D. Volland, J. Niedens, P. T. Geppert, M. J. Wildervanck, F. Full and A. Nowak-Król, Angew. Chem., Int. Ed., 2023, 62, e202304291 CrossRef CAS PubMed.
- (a) P. Ravat, Chem.–Eur. J., 2021, 27, 3957–3967 CrossRef CAS PubMed; (b) J. M. Fernández-García, P. Izquierdo-García, M. Buendía, S. Filippone and N. Martín, Chem. Commun., 2022, 58, 2634–2645 RSC.
- T. Mori, Chem. Rev., 2021, 121, 2373–2412 CrossRef CAS PubMed.
- (a) G. R. Kiel, S. C. Patel, P. W. Smith, D. S. Levine and T. D. Tilley, J. Am. Chem. Soc., 2017, 139, 18456–18459 CrossRef CAS PubMed; (b) Y. Nakakuki, T. Hirose, H. Sotome, H. Miyasaka and K. Matsuda, J. Am. Chem. Soc., 2018, 140, 4317–4326 CrossRef CAS PubMed; (c) Y. Nakakuki, T. Hirose and K. Matsuda, J. Am. Chem. Soc., 2018, 140, 15461–15469 CrossRef CAS PubMed; (d) A. E. Samkian, G. R. Kiel, C. G. Jones, H. M. Bergman, J. Oktawiec, H. M. Nelson and T. D. Tilley, Angew. Chem., Int. Ed., 2021, 60, 2493–2499 CrossRef CAS PubMed; (e) G. R. Kiel, H. M. Bergman, A. E. Samkian, N. J. Schuster, R. C. Handford, A. J. Rothenberger, R. Gomez-Bombarelli, C. Nuckolls and T. D. Tilley, J. Am. Chem. Soc., 2022, 144, 23421–23427 CrossRef CAS; (f) G.-F. Huo, W.-T. Xu, Y. Han, J. Zhu, X. Hou, W. Fan, Y. Ni, S. Wu, H.-B. Yang and J. Wu, Angew. Chem., Int. Ed., 2024, 63, e202403149 CrossRef CAS PubMed.
- (a) P. Ravat, R. Hinkelmann, D. Steinebrunner, A. Prescimone, I. Bodoky and M. Juríček, Org. Lett., 2017, 19, 3707–3710 CrossRef CAS PubMed; (b) X. Guo, Z. Yuan, Y. Zhu, Z. Li, R. Huang, Z. Xia, W. Zhang, Y. Li and J. Wang, Angew. Chem., Int. Ed., 2019, 58, 16966–16972 CrossRef CAS PubMed; (c) K. Suzuki, H. Fukuda, H. Toda, Y. Imai, Y. Nojima, M. Hasegawa, E. Tsurumaki and S. Toyota, Tetrahedron, 2023, 132, 133243 CrossRef CAS; (d) K. Fujise, E. Tsurumaki, G. Fukuhara, N. Hara, Y. Imai and S. Toyota, Chem.–Asian J., 2020, 15, 2456–2461 CrossRef CAS PubMed; (e) Y. Matsuo, S. Seki and T. Tanaka, Chem. Lett., 2024, 53, upae159 CrossRef CAS.
- (a) J. Full, S. P. Panchal, J. Götz, A.-M. Krause and A. Nowak-Król, Angew. Chem., Int. Ed., 2021, 60, 4350–4357 CrossRef CAS; (b) K. Fujise, E. Tsurumaki, K. Wakamatsu and S. Toyota, Chem.–Eur. J., 2021, 27, 4548–4552 CrossRef CAS PubMed; (c) S. Oda, B. Kawakami, Y. Yamasaki, R. Matsumoto, M. Yoshioka, D. Fukushima, S. Nakatsuka and T. Hatakeyama, J. Am. Chem. Soc., 2022, 144, 106–112 CrossRef CAS.
- (a) Y. Matsuo, M. Gon, K. Tanaka, S. Seki and T. Tanaka, J. Am. Chem. Soc., 2024, 146, 17428–17437 CrossRef CAS PubMed; (b) J. Labella, W. R. Osterloh, K. Kuo, Y. Tsutsui, T. Tanaka and S. Seki, J. Am. Chem. Soc., 2026, 148, 9670–9679 CrossRef PubMed.
- X.-Y. Wang, J. Bai, Y.-J. Shen, Z.-A. Li and H.-Y. Gong, Angew. Chem., Int. Ed., 2025, 64, e202417745 CrossRef CAS.
- (a) P. Karak, C. Dutta, T. Dutta, A. L. Koner and J. Choudhury, Chem. Commun., 2019, 55, 6791–6794 RSC; (b) P. Karak and J. Choudhury, Chem. Sci., 2022, 13, 11163–11173 RSC.
- (a) P. Karak, S. K. Mandal and J. Choudhury, J. Am. Chem. Soc., 2023, 145, 7230–7241 CrossRef CAS PubMed; (b) P. Karak, S. K. Mandal and J. Choudhury, J. Am. Chem. Soc., 2023, 145, 17321–17328 CrossRef CAS.
- S. Han, A. D. Bond, R. L. Disch, D. Holmes, J. M. Schulman, S. J. Teat, K. P. C. Vollhardt and G. D. Whitener, Angew. Chem., Int. Ed., 2002, 41, 3223–3227 CrossRef CAS.
- Z. Qiu, C.-W. Ju, L. Frédéric, Y. Hu, D. Schollmeyer, G. Pieters, K. Müllen and A. Narita, J. Am. Chem. Soc., 2021, 143, 4661–4667 CrossRef CAS PubMed.
- (a) E. Yen-Pon, F. Buttard, L. Frédéric, P. Thuéry, F. Taran, G. Pieters, P. A. Champagne and D. Audisio, JACS Au, 2021, 1, 807–818 CrossRef CAS PubMed; (b) Y. Matsuo, C. Maeda, Y. Tsutsui, T. Tanaka and S. Seki, Angew. Chem., Int. Ed., 2023, 62, e202314968 CrossRef CAS PubMed; (c) M. A. Medel, L. Hortigüela, V. Lloveras, J. Catalán-Toledo, D. Miguel, A. J. Mota, N. Crivillers, A. G. Campaña and S. P. Morcillo, ChemistryEurope, 2023, 1, e202300021 CrossRef.
- (a) J. Han, S. Guo, H. Lu, S. Liu, Q. Zhao and W. Huang, Adv. Opt. Mater., 2018, 6, 1800538 CrossRef; (b) J. Ahn, S. H. Lee, I. Song, P. Chidchob, Y. Kwon and J. H. Oh, Device, 2023, 1, 100176 CrossRef.
- CCDC 2485128: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pdzfj.
Footnote |
| † M. P. and P. K. contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |