Open Access Article
Open Access ArticleSynergistic Cu nanoparticles and Cu single atoms leveraging hydrogen spillover for selective CO electroreduction to acetate
Qinglong Wangab,
Tianfu Liu*ad,
Xinhui Guoad,
Pengfei Weiac,
Dunfeng Gao *ad,
Guoxiong Wang
*ad,
Guoxiong Wang ac and
Xinhe Bao
ac and
Xinhe Bao acd
acd
aState Key Laboratory of Catalysis, Dalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian 116023, China. E-mail: ltianfu@dicp.ac.cn; dfgao@dicp.ac.cn
bKey Laboratory of Green Chemical Process of Ministry of Education, Hubei Key Laboratory of Novel Reactor and Green Chemical Technology, School of Chemical Engineering and Pharmacy, Wuhan Institute of Technology, Wuhan 430205, China
cDepartment of Chemistry, Shanghai Key Laboratory of Molecular Catalysis and Innovative Materials, iChEM (Collaborative Innovation Center of Chemistry for Energy Materials), Fudan University, Shanghai 200438, China
dUniversity of Chinese Academy of Sciences, Beijing 100049, China
First published on 13th April 2026
Abstract
The electrochemical reduction of CO to acetate on Cu-based catalysts is constrained by a kinetic trade-off between CO activation and the availability of active hydrogen (*H) species. Herein, we overcome this limitation by constructing a synergistic catalyst comprising Cu nanoparticles and Cu single atoms (CuNP–Cu1), which leverages a hydrogen spillover mechanism. The Cu1 sites facilitate water dissociation to generate *H, which subsequently spills over to adjacent Cu NPs, enabling efficient hydrogenation of the *OCCO intermediate, with subsequent formation of *CCO selectively. This synergy delivers an acetate faradaic efficiency of 60.6% with an acetate partial current density of 111.1 mA cm−2 and stable operation for 132 h. Operando spectroscopy measurements and theoretical calculations collectively reveal that the Cu1-mediated hydrogen spillover shifts the reaction pathway toward acetate formation. This work establishes hydrogen spillover as a design principle for coordinating multistep reactions in CO electroreduction.
Introduction
The electrochemical reduction of CO to multicarbon (C2+) products represents a promising strategy for the sustainable production of high-value chemicals. Acetate, a key intermediate, finds extensive applications in fine chemicals, pesticides, fuels, and pharmaceuticals.1,2 Although the formation of acetate is thermodynamically favorable, with a theoretical CO/acetate redox potential of 0.09 V versus the reversible hydrogen electrode (vs. RHE),3 the catalytic process remains kinetically sluggish. The multi-proton–electron coupled transfer reaction poses a significant kinetic barrier, limiting the efficient conversion of CO to acetate.4 To improve acetate selectivity, significant efforts have been focused on enhancing C–C coupling and increasing current density.5–7 Strategies including optimizing crystal facets,8,9 morphology,10 interfaces,11 and alloying12–14 have been reported in recent years.Cu stands out among metal catalysts owing to its unique ability to facilitate C–C coupling during CO electrochemical reduction, enabling the synthesis of valuable C2+ compounds.15–17 Cu nanoparticles (NPs) exhibit favorable *CO adsorption thermodynamics and low C–C coupling barriers, making them efficient for generating C2+ products via *CO electroreduction.18,19 As a result, Cu NPs typically enable appreciable formation of ethylene and ethanol.20,21 However, the formation of the *OCCOH intermediate is widely recognized as a pivotal step in C2+ product generation, with its subsequent hydrogenation and dehydration via the ketene intermediate (*C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) selectively driving the reaction pathway toward acetate formation, which is kinetically constrained by the limited availability of active hydrogen (*H) species on Cu surfaces.22–25
O) selectively driving the reaction pathway toward acetate formation, which is kinetically constrained by the limited availability of active hydrogen (*H) species on Cu surfaces.22–25
Hydrogen spillover has emerged as a key mechanism to regulate interfacial *H abundance, lowering the energy barrier for C–C coupling.26,27 Recently, single-atom Cu catalysts (Cu1) have been shown to facilitate water dissociation for efficient *H generation, promoting the conversion of *CO to *CHO.28 It is widely reported that the synergistic increase in the *H and *CO surface coverage kinetically steers the reaction pathway toward asymmetric C–C coupling via preferential *CO hydrogenation.29–31 In such a scenario, synergistic catalysis is expected to be a feasible strategy to balance the *CO adsorption and the hydrogenation kinetics of carbon intermediates.
Herein, we construct a synergistic catalyst by introducing Cu single atoms onto Cu NPs (CuNP–Cu1) for electrochemical CO reduction to acetate, with a faradaic efficiency of 60.6%, a partial current density of 111.1 mA cm−2 and durability for 132 hours. Electrochemical CO stripping experiments, operando Raman spectroscopy and H/D kinetic isotope effect (KIE) studies reveal that Cu NPs facilitate C–C coupling by effectively adsorbing CO molecules and Cu single atoms enhance the dissociation of H2O to generate active *H species, promoting hydrogen spillover from Cu1 sites to CuNP. Density functional theory (DFT) calculations incorporating ab initio molecular dynamics (AIMD) simulations indicate that the modulated *H coverage thermodynamically drives the formation of key intermediates, ultimately enhancing acetate production. This work establishes hydrogen spillover engineering as a general strategy to coordinate multistep reactions in CO electroreduction systems.
Results and discussion
Structural and morphological characterization
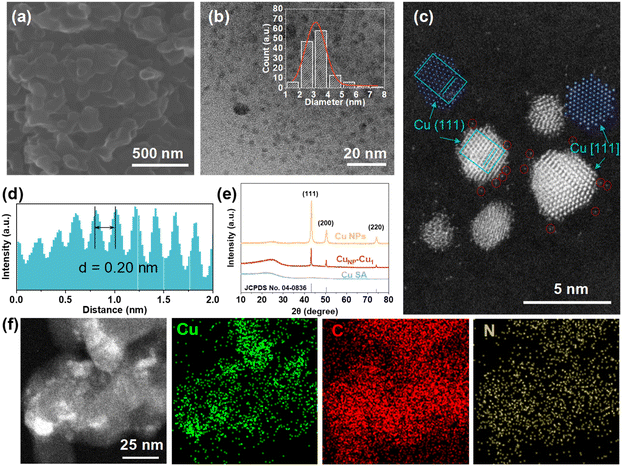
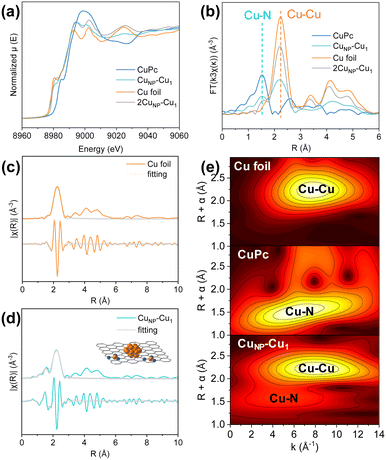
The CuNP–Cu1 catalyst was synthesized via the pyrolysis of a Cu coordination polymer under Ar flow (Fig. S1). The scanning electron microscopy (SEM) image, as illustrated in Fig. 1a, reveals a smooth and granular texture. The transmission electron microscopy (TEM) image of CuNP–Cu1 (Fig. 1b) reveals the presence of Cu NPs (CuNP) with an average size of 3.1 nm within the CuNP–Cu1 catalyst (see SEM and TEM images in Fig. S2). Aberration-corrected high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) images (Fig. 1c) reveal that Cu single atoms (highlighted by red circles) are densely dispersed around Cu nanoparticles with an average inter-site distance of approximately 0.22 nm (Fig. S3). Such atomic-level spatial proximity is essential for efficient hydrogen spillover, as it minimizes the migration distance of *H species between Cu1 and CuNP surfaces.32 A lattice spacing of 0.20 nm for the CuNP is consistent with the (111) crystal plane of metallic Cu, confirmed by the intensity profile derived from the STEM image (Fig. 1d). For comparison purposes, we prepared 2CuNP–Cu1 (doubled copper salt, Fig. S4), Cu NPs (Cu nanoparticles loaded on XC72R, Fig. S5a) and CuSA (Cu single atom, CuNP–Cu1 etched with H2SO4 to remove CuNP, Fig. S5b and c). X-ray diffraction (XRD) patterns of CuNP–Cu1, and Cu NPs (Fig. 1e) show characteristic peaks attributed to metallic copper and carbon, with a prominent peak at 43.2° corresponding to the Cu (111) plane. In contrast, the XRD pattern of CuSA exhibited typical diffraction patterns of amorphous carbon, with no discernible peaks attributable to copper. Energy-dispersive X-ray spectroscopy (EDS) elemental mapping (Fig. 1f) indicates a uniform distribution of C, N, and Cu on the carbon matrix, suggesting that these elements are evenly dispersed.To further elucidate the precise coordination structure and chemical state of Cu within the catalyst, X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) characterization studies were employed. Fig. 2a shows the Cu K-edge XANES spectra of CuNP–Cu1, 2CuNP–Cu1, Cu foil, and CuPc. The absorption edge position for CuNP–Cu1 lies between those of Cu foil and CuPc, indicating that the average oxidation state of Cu in the catalyst is between 0 and +2. This suggests the coexistence of Cu0 and Cu2+ species within the CuNP–Cu1 catalyst. The Fourier transform (FT) EXAFS spectra in R-space, shown in Fig. 2b, reveal distinct features. Cu foil exhibits a characteristic first shell Cu–Cu bond at 2.2 Å, while CuPc shows a Cu–N bond at 1.5 Å. In contrast, CuNP–Cu1 and 2CuNP–Cu1 display two bond lengths at 1.5 Å and 2.2 Å, corresponding to Cu–N and Cu–Cu bonds, respectively.33,34 The Fourier-transformed EXAFS fitting plots and k3-weighted Cu K-edge EXAFS spectra of Cu foil and CuNP–Cu1 catalysts demonstrate a good fitting quality of the EXAFS data (Fig. 2c, d and S6). Additionally, wavelet transform (WT)-EXAFS analysis was conducted on the Cu K-edge of CuNP–Cu1, Cu foil, and CuPc (Fig. 2e). Cu foil exhibits a maximum WT intensity at k = 7.0 Å−1, indicative of Cu–Cu coordination, whereas CuPc shows a maximum WT intensity at k = 6.0 Å−1, corresponding to Cu–N coordination. Notably, CuNP–Cu1 presents maxima at both k = 6.0 Å−1 and k = 7.0 Å−1, confirming the presence of both Cu–Cu and Cu–N bonds. The EXAFS fitting yields an average Cu–N coordination number of 2.7, consistent with a distribution of coordination environments where Cu–N3 is the dominant motif (Table S1). DFT calculations based on a representative Cu–N3 model were employed to elucidate the mechanistic role of the Cu single-atom sites. This evidence further corroborates the coexistence of Cu single atoms and Cu NPs in CuNP–Cu1, aligning with HAADF-STEM observations.
Electrochemical CO reduction performance
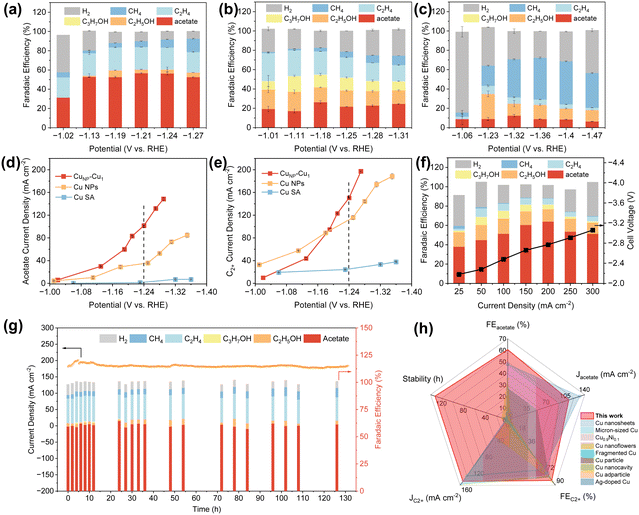
The electrochemical performance of CuNP–Cu1 was evaluated in a flow cell (Fig. S7). Fig. S8 shows a notable increase in cathodic current density when the feed gas was switched from Ar to CO. The mass loading of the CuNP–Cu1 catalyst on carbon paper was optimized to assess CO reduction reaction (CORR) performance. As shown in Fig. 3a and S9, the catalyst with a mass loading of 0.5 mg cm−2 led to a prominent acetate partial current density. The catalytic performance of CuNP–Cu1 was benchmarked against Cu NPs and CuSA under identical conditions (Fig. 3a–e). Notably, the faradaic efficiency (FE) for acetate over the CuNP–Cu1 catalyst reaches 57.6% at −1.24 V vs. RHE with a partial current density of 103.9 mA cm−2. In contrast, the sum of acetate FE from Cu NPs (FE ∼20%) and CuSA (FE ∼10%) individually is substantially lower than that of CuNP–Cu1, providing a clear indication of synergistic enhancement rather than simple additive effects (Fig. 3a–d). The partial current densities of acetate and C2+ products on CuNP–Cu1, Cu NPs, and CuSA show a sustained increase as the applied potential shifts negatively (Fig. 3d and e). The faradaic efficiency for C2+ products on CuNP–Cu1 reaches 83.4% with a partial current density of 150.5 mA cm−2 (Fig. 3e and S10). The carbon selectivity distribution among CO electrolysis products exhibits pronounced disparities in carbonaceous product formation. CuNP–Cu1 demonstrates an exceptional acetate selectivity of approximately 80%, whereas Cu NPs and CuSA catalysts show substantial reductions in acetate production efficiency (Fig. S11). Moreover, commercial Cu NPs (25 nm) also exhibit a product distribution similar to Cu NPs (Fig. S12).To further probe the origin of this synergy, we systematically varied the CuNP/Cu1 ratio by quantifying CuSA and Cu NPs in the 2CuSA, CuSA, 0.5CuSA, CuNP–Cu1, 2CuNP–Cu1 and 0.5CuNP–Cu1 catalysts by inductively coupled plasma-optical emission spectrometry (ICP-OES) measurements (Table S2) and evaluated their CORR performance (Fig. S13). A volcano-shaped relationship emerges between the CuNP/Cu1 ratio and acetate FE, with the optimal performance centered at CuNP–Cu1. Catalysts with insufficient Cu1 sites suffer from limited *H supply, while those with excessive Cu1 sites exhibit diminished C–C coupling due to insufficient Cu NP surfaces. This volcano trend underscores the necessity of balancing the *CO activation and the *H generation for selective acetate formation. These results confirm that the selective acetate formation is a result of the synergistic effect between Cu nanoparticles and Cu single atoms. Additionally, electrochemical impedance spectroscopy (EIS) and double-layer capacitance (Cdl) measurements indicate that the coexistence of Cu nanoparticles and single atoms accelerates electron transfer within CuNP–Cu1 and provides more active sites (Fig. S14 and S15).
To further investigate the role of *CO coverage in steering product selectivity, we evaluated the CO electroreduction performance of CuNP–Cu1 in an alkaline membrane electrode assembly (MEA) electrolyzer under both 0.1 and 1.0 MPa CO. As illustrated in Fig. 3f and S16, at 0.1 MPa CO, the acetate FE reaches 48.5%, with partial current densities of 184.7 mA cm−2 for C2+ products and 94.8 mA cm−2 for acetate. Upon increasing the CO pressure to 1.0 MPa, the acetate FE markedly increases to 64.0%, and the corresponding partial current density increases to 127.9 mA cm−2. This pronounced shift in product distribution provides strong experimental evidence for the proposed synergy between *CO coverage and hydrogen spillover. Elevated CO pressure significantly increases *CO surface coverage on Cu nanoparticles, which not only promotes C–C coupling kinetics but also synergizes with the high *H coverage sustained by hydrogen spillover from adjacent Cu single atoms. These pressure-dependent results thus independently validate the proposed hydrogen spillover mechanism, confirming that the synergy between *CO coverage and *H supply dictates product selectivity.35–37
Furthermore, the durability test conducted at −1.24 V vs. RHE reveals that the CuNP–Cu1 catalyst remains stable for 132 hours without obvious current decay, maintaining an average acetate faradaic efficiency of 60.6% and an average current density of 111.1 mA cm−2 (Fig. 3g). Compared with literature benchmarks, this work outperforms state-of-the-art CORR electrodes in terms of acetate partial current density and operation stability (Fig. 3h and Table S3).
Mechanistic investigation of hydrogen spillover
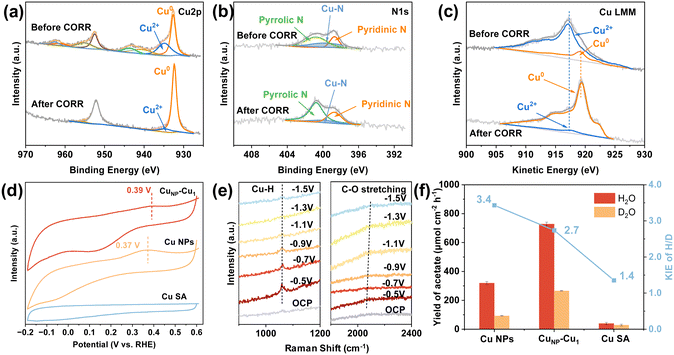
We characterized the structural changes in the catalyst using quasi in situ X-ray photoelectron spectroscopy (XPS) under oxygen-free conditions to accurately determine the oxidation state of Cu during CO electroreduction. The Cu 2p spectrum (Fig. 4a) reveals peaks corresponding to metallic Cu (Cu0) at 932.8 eV and single-atom Cu (Cu2+) at 934.6 eV for Cu 2p3/2.38,39 After performing CO electroreduction at 150 mA cm−2 in a 1.0 M KOH solution, the single-atom Cu in the catalyst is partially reduced to metallic copper. The N 1s spectrum (Fig. 4b) indicates the presence of pyridinic N at 398.7 eV and pyrrolic N at 400.8 eV. Additionally, a Cu–N peak observed near 399.8 eV suggests coordination between copper sites and nitrogen atoms.40,41 The decrease in the proportion of pyridinic N after the reaction may be attributed to the breaking of Cu–N bonds during the reaction, leading to the release of single-atom copper. As shown in Fig. 4c, nonlinear least squares fitting (NLLSF) of the Cu LMM Auger spectrum further confirms the coexistence of Cu2+ and Cu0, with kinetic energies of 917.2 eV and 919.4 eV, respectively.42–44 The catalyst surface contains a higher abundance of single-atom Cu before the CORR than that after the reaction. Although minor coarsening of the CuNP is observed in Fig. S17, this structural modification has no detrimental effects on the CORR catalytic performance based on long-term stability results and the unchanged contact angle (Fig. S18). Taken together, these findings demonstrate that the CuNP–Cu1 architecture maintains structural integrity through adaptive restructuring during prolonged electrochemical operation.Electrochemical CO stripping voltammetry tests were conducted to evaluate the CO adsorption performance of CuNP–Cu1, Cu NPs, and CuSA in a flow cell. The CO desorption peaks for CuNP–Cu1 and Cu NPs are located at 0.39 and 0.37 V, respectively, indicating strong CO adsorption on the CuNP–Cu1 catalyst (Fig. 4d). No CO desorption peaks were observed for catalysts without adsorbing CO (Fig. S19). A higher CO desorption potential indicates stronger CO adsorption ability.45,46 Cu SA shows no detectable CO desorption peak, confirming that Cu NPs are the primary sites for *CO activation. This functional separation is the prerequisite for the proposed synergistic mechanism. To probe surface species evolution on CuNP–Cu1 during the CORR, operando Raman spectroscopy was employed. As depicted in Fig. 4e, spectra acquired from −0.5 to −1.5 V vs. RHE exhibit two characteristic bands, the peak at 1060 cm−1 corresponding to Cu–H bending vibrations,47 and the band at 2080 cm−1 assigned to atop-adsorbed *CO (C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) O stretching).35,48 The inverse intensity correlation, that is, a decreasing Cu–H signal with increasing *CO adsorption, reveals consumption of adsorbed hydrogen species during CO adsorption at cathodic potentials.49 In contrast, control experiments with the Cu NP catalyst show no detectable *CO or Cu–H signals until more negative potentials (Fig. S20), demonstrating that Cu1 sites critically enhance both *CO adsorption and hydrogen spillover kinetics on adjacent CuNP surfaces.
O stretching).35,48 The inverse intensity correlation, that is, a decreasing Cu–H signal with increasing *CO adsorption, reveals consumption of adsorbed hydrogen species during CO adsorption at cathodic potentials.49 In contrast, control experiments with the Cu NP catalyst show no detectable *CO or Cu–H signals until more negative potentials (Fig. S20), demonstrating that Cu1 sites critically enhance both *CO adsorption and hydrogen spillover kinetics on adjacent CuNP surfaces.
To investigate the role of Cu1 in water dissociation, the H/D KIE experiments were conducted. When D2O replaces H2O in the electrolyte, the production rates of products decrease for CuNP–Cu1, Cu NPs, and CuSA catalysts. The magnitude of this decrease was closely correlated with the ratio of CuNP to Cu1 content. The KIE of H/D is defined as the ratio of production rates in H2O and D2O electrolytes. A KIE value close to 1 indicates minimal impact of water dissociation on product formation, whereas a larger KIE suggests a significant influence of hydrogen spillover on the production rate.28,29 A calibration curve between the concentration of deuterated acetate and deuterated dimethyl sulfoxide (DMSO) was plotted to quantify acetate-d (Fig. S21 and S22). As shown in Fig. 4f, the KIE value for acetate production on Cu SA is 1.4, close to unity, indicating that water dissociation is not rate-limiting. In contrast, Cu NPs exhibit a KIE of 3.4, suggesting that *H supply substantially limits acetate formation on pure Cu NPs. CuNP–Cu1 displays a moderate KIE of 2.7, consistent with Cu1 sites accelerating water dissociation and supplying *H to Cu NPs. Besides, KIE values for C2H4 and H2 yield present similar trends to that of acetate (Fig. S23). An increase in Cu1 content within the catalysts accelerates the water dissociation rate, leading to a decrease in the KIE value. The integration of Cu1 into CuNP amplifies the KIE value for C2+ formation, confirming that Cu1-mediated hydrogen spillover occurs between Cu1 and CuNP.
DFT calculations
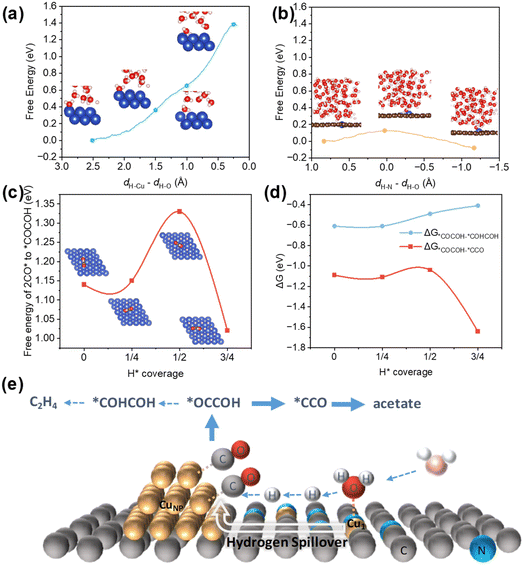
DFT calculations incorporating AIMD simulations were conducted to mechanistically decode the dual-site synergy in the CO electroreduction pathway. The investigation focuses on the H2O dissociation kinetics for the hydrogen source and C–C coupling. The computational models Cu(100) and Cu–N3 moiety were constructed to model CuNP and Cu1 sites.50,51 To identify the primary source of *H supply, we calculated the free energy profiles for H2O dissociation on both sites using AIMD simulations. The reaction coordinate Δd was defined to track the O–H bond scission, which was expressed as the difference between the forming and breaking bond lengths: Δd = dH–Cu − dH–O on CuNP, and Δd = dH–N − dH–O on Cu1 sites52 (Fig. 5a, b, S24 and S25). The results revealed a clear distinction between the two sites. The energy barrier on Cu(100) increased inversely with Δd, reaching 1.38 eV at the final state of H2O dissociation. The Cu–N3 site exhibited a dramatically lower barrier of 0.13 eV, indicating kinetically favorable water dissociation. This direct comparison demonstrates that Cu1 sites reduce the H2O dissociation barrier by 1.25 eV relative to CuNP, confirming that the single-atom sites are the kinetically favorable centers for generating *H species. This theoretical insight aligns with experimental operando Raman spectroscopy and KIE values, corroborating the synergistic mechanism wherein Cu1 sites efficiently generate *H, which then spills over to adjacent CuNP surfaces to facilitate the hydrogenation of key intermediates within the overall CORR pathway.Given the established significance of the *OCCOH to ketene pathway as selectivity-determining for acetate production, the energetics of *OCCOH formation can be considered a key descriptor for a catalyst's C–C coupling performance and overall C2+ selectivity. We therefore systematically investigated the impact of *H coverage on this crucial step.53 DFT calculations uncovered a coverage-dependent energetic switching behavior. *OCCOH formation becomes increasingly endergonic (ΔG up to 1.33 eV) at low *H coverage (θH ≤ 1/2 ML). However, at high *H coverage (θH = 3/4 ML), the reaction barrier is dramatically reduced to 1.02 eV (Fig. 5c, S26 and Table S4), enabling the pathway kinetically accessible under operational potentials. These computational insights demonstrate that high *H coverage (>1/2 ML) is favorable for *OCCOH formation, indicating that a high *H concentration is crucial for promoting the overall reaction pathway towards C2+ products. As shown in Fig. 5d and Table S4, the energy of the acetate intermediate *CCO at 3/4 ML coverage is −1.64 eV, which is lower than −1.09 eV at 0 ML coverage. In contrast, the energy of the ethylene intermediate *COHCOH at 3/4 ML coverage is −0.41 eV, higher than −0.61 eV at 0 ML coverage. Since a more negative energy indicates a more favorable process, acetate formation becomes more favorable at high *H coverage. This coverage-dependent energetics explains that Cu1-mediated hydrogen spillover directs the selectivity toward acetate over ethylene.
Collectively, our experimental and theoretical results converge on a unified mechanistic picture. Cu1 sites in the CuNP–Cu1 catalyst function as *H flux amplifiers to accelerate water dissociation and enable hydrogen spillover onto adjacent CuNP, where *CO is activated (Fig. 5e). The resulting high *H coverage on Cu NPs selectively promotes *OCCO hydrogenation toward acetate, circumventing the kinetic trade-off inherent in conventional Cu catalysts. This mechanism dynamically enhances acetate selectivity by 2.7-fold and 6.5-fold versus CuNP-only and Cu1-only catalysts, respectively.
Conclusions
In summary, we have demonstrated that the spatial integration of Cu nanoparticles and Cu single atoms enables a hydrogen spillover mechanism that decouples the kinetic trade-off between CO activation and *H supply in CO electroreduction. The Cu1 sites accelerate water dissociation to form *H, while the proximal Cu NPs enable the *H-assisted C–C coupling and acetate pathway. This synergy results in impressive acetate selectivity and operation stability, and more importantly, establishes hydrogen spillover as a general strategy for coordinating multistep reactions in complex electrocatalytic systems.Author contributions
Conceptualization: D. G. and T. L.; methodology: Q. W., T. L., X. G., P. W., and D. G.; investigation: Q. W. and T. L.; visualization: Q. W. and T. L.; funding acquisition: G. W., D. G., T. L., and Q. W.; supervision: D. G., T. L., G. W., and X. B.; writing—original draft: Q. W. and T. L.; writing—review and editing: D. G., T. L., G. W., and X. B.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data underlying this article are available in the article and in its online supplementary information (SI). Supplementary information: experimental and theoretical details as well as additional characterization and performance data. See DOI: https://doi.org/10.1039/d6sc02439d.Acknowledgements
This work was supported by the National Key Research and Development Program of China (2021YFA1501500), the National Natural Science Foundation of China (22372171, 22472176, 22494711, 22125205, and 22321002), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0600200), the Liaoning Revitalization Talents Program (XLYC2203178), the Liaoning Binhai Laboratory (LBLF-2023-02, LBLD-2024-02), the Natural Science Foundation of Liaoning Province (2026JH6/101100020), the Dalian Outstanding Young Scientist Foundation (2024RJ003), the Dalian Institute of Chemical Physics (DICP I202504), the China Postdoctoral Science Foundation (2023M743427), the Postdoctoral Fellowship Program of CPSF (GZC20232586), and the Photon Science Center for Carbon Neutrality (JZHKYPT-2021-07). We thank the Shanghai Synchrotron Radiation Facility of BL14W1 (https://cstr.cn/31124.02.SSRF.BL14W1) for assistance in XAS measurements.Notes and references
- J. Y. T. Kim, C. Sellers, S. Hao, T. P. Senftle and H. Wang, Nat. Catal., 2023, 6, 1115–1124 CrossRef CAS.
- W. Ma, X. He, W. Wang, S. Xie, Q. Zhang and Y. Wang, Chem. Soc. Rev., 2021, 50, 12897–12914 RSC.
- H. Du, J. Fu, L.-X. Liu, S. Ding, Z. Lyu, Y.-C. Chang, X. Jin, F. O. Kengara, B. Song, Q. Min, J.-J. Zhu, D. Du, C. Gu, Y. Lin, J.-S. Hu and W. Zhu, Mater. Today, 2022, 59, 182–199 CrossRef CAS.
- Y. Zhao, Y. Li, J. Chen, B. Sun, L. Fan, J. Chen, Y. Xiao, H. Yang, D. Wang, J. Chen, X. Han, S. Xi, J. Zhang and L. Wang, ACS Catal., 2024, 14, 8366–8375 CrossRef CAS.
- J. Wu, T. Sharifi, Y. Gao, T. Zhang and P. M. Ajayan, Adv. Mater., 2019, 31, 1804257 CrossRef PubMed.
- T. Wang, Q. Zhao, Y. Fu, C. Lei, B. Yang, Z. Li, L. Lei, G. Wu and Y. Hou, Small Methods, 2019, 3, 1900210 CrossRef CAS.
- N. Meng, Z. Wu, Y. Huang, J. Zhang, M. Chen, H. Ma, H. Li, S. Xi, M. Lin, W. Wu, S. Han, Y. Yu, Q.-H. Yang, B. Zhang and K. P. Loh, Nat. Commun., 2024, 15, 3892 CrossRef CAS PubMed.
- J. Liu, Y. Wen, W. Yan, Z. Huang, X. Liu, X. Huang, C. Zhan, Y. Zhang, W.-H. Huang, C.-W. Pao, Z. Hu, D. Su, S. Xie, Y. Wang, J. Han, H. Xiong, X. Huang and N. Chen, Energy Environ. Sci., 2025, 18, 4396–4404 RSC.
- Z.-Z. Wu, X.-L. Zhang, Z.-Z. Niu, F.-Y. Gao, P.-P. Yang, L.-P. Chi, L. Shi, W.-S. Wei, R. Liu, Z. Chen, S. Hu, X. Zheng and M.-R. Gao, J. Am. Chem. Soc., 2022, 144, 259–269 CrossRef CAS PubMed.
- P. Zhu, C. Xia, C.-Y. Liu, K. Jiang, G. Gao, X. Zhang, Y. Xia, Y. Lei, H. N. Alshareef and T. P. Senftle, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2010868118 CrossRef CAS PubMed.
- W. Wei, Z. Li, J. Shang, C. Xia, X. Hu, Y. Hu, J. Xu, Y. Zhao and M. Ding, Appl. Catal., B, 2025, 371, 125220 CrossRef CAS.
- Y. Ji, Z. Chen, R. Wei, C. Yang, Y. Wang, J. Xu, H. Zhang, A. Guan, J. Chen, T.-K. Sham, J. Luo, Y. Yang, X. Xu and G. Zheng, Nat. Catal., 2022, 5, 251–258 CrossRef CAS.
- J. Jin, J. Wicks, Q. Min, J. Li, Y. Hu, J. Ma, Y. Wang, Z. Jiang, Y. Xu, R. Lu, G. Si, P. Papangelakis, M. Shakouri, Q. Xiao, P. Ou, X. Wang, Z. Chen, W. Zhang, K. Yu, J. Song, X. Jiang, P. Qiu, Y. Lou, D. Wu, Y. Mao, A. Ozden, C. Wang, B. Y. Xia, X. Hu, V. P. Dravid, Y.-M. Yiu, T.-K. Sham, Z. Wang, D. Sinton, L. Mai, E. H. Sargent and Y. Pang, Nature, 2023, 617, 724–729 CrossRef CAS PubMed.
- Q. Sun, Y. Zhao, X. Tan, C. Jia, Z. Su, Q. Meyer, M. I. Ahmed and C. Zhao, ACS Catal., 2023, 13, 5689–5696 CrossRef CAS.
- H. Li, T. Liu, P. Wei, L. Lin, D. Gao, G. Wang and X. Bao, Angew. Chem., Int. Ed., 2021, 60, 14329–14333 CrossRef CAS PubMed.
- M. Li, Y. Ma, J. Chen, R. Lawrence, W. Luo, M. Sacchi, W. Jiang and J. Yang, Angew. Chem., Int. Ed., 2021, 60, 11487–11493 CrossRef CAS PubMed.
- R. Chen, H.-Y. Su, D. Liu, R. Huang, X. Meng, X. Cui, Z.-Q. Tian, D. H. Zhang and D. Deng, Angew. Chem., Int. Ed., 2020, 59, 154–160 CrossRef CAS PubMed.
- Y. Wu, C. Chen, S. Liu, Q. Qian, Q. Zhu, R. Feng, L. Jing, X. Kang, X. Sun and B. Han, Angew. Chem., Int. Ed., 2024, 63, e202410659 CrossRef CAS PubMed.
- W. Wu, L. Luo, Z. Li, J. Luo, J. Zhao, M. Wang, X. Ma, S. Hu, Y. Chen and W. Chen, Angew. Chem., Int. Ed., 2024, 136, e202404983 CrossRef.
- S. Li, G. Zhang, X. Ma, H. Gao, D. Fu, T. Wang, J. Zeng, Z.-J. Zhao, P. Zhang and J. Gong, J. Am. Chem. Soc., 2024, 146, 31927–31934 CrossRef CAS PubMed.
- Z. Wang, J. Guo, D. Wang, J. Ma, X. Feng, Z. Cui, D. Ruan, X. Luo, J. Fan, J. Yang, B.-Q. Xiong and X. Ren, Angew. Chem., Int. Ed., 2026, 65, e23475 CrossRef CAS PubMed.
- R. Dorakhan, I. Grigioni, B.-H. Lee, P. Ou, J. Abed, C. O'Brien, A. Sedighian Rasouli, M. Plodinec, R. K. Miao, E. Shirzadi, J. Wicks, S. Park, G. Lee, J. Zhang, D. Sinton and E. H. Sargent, Nat. Synth., 2023, 2, 448–457 CrossRef CAS.
- P. Zhu, C. Xia, C.-Y. Liu, K. Jiang, G. Gao, X. Zhang, Y. Xia, Y. Lei, H. N. Alshareef, T. P. Senftle and H. Wang, Proc. Natl. Acad. Sci. U. S. A., 2021, 118, e2010868118 CrossRef CAS PubMed.
- L. Zhang, J. Feng, R. Wang, L. Wu, X. Song, X. Jin, X. Tan, S. Jia, X. Ma, L. Jing, Q. Zhu, X. Kang, J. Zhang, X. Sun and B. Han, J. Am. Chem. Soc., 2025, 147, 713–724 CrossRef CAS PubMed.
- F. Ni, H. Yang, Y. Wen, H. Bai, L. Zhang, C. Cui, S. Li, S. He, T. Cheng, B. Zhang and H. Peng, Sci. China Mater., 2020, 63, 2606–2612 CrossRef.
- H. Kang, L. Zhu, S. Li, S. Yu, Y. Niu, B. Zhang, W. Chu, X. Liu, S. Perathoner, G. Centi and Y. Liu, Nat. Catal., 2023, 6, 1062–1072 CrossRef CAS.
- K. Jenkinson, M. C. Spadaro, V. Golovanova, T. Andreu, J. R. Morante, J. Arbiol and S. Bals, Adv. Mater., 2023, 35, 2306447 CrossRef CAS PubMed.
- J. Feng, L. Zhang, S. Liu, L. Xu, X. Ma, X. Tan, L. Wu, Q. Qian, T. Wu, J. Zhang, X. Sun and B. Han, Nat. Commun., 2023, 14, 4615 CrossRef CAS PubMed.
- S. Li, G. Zhang, X. Ma, H. Gao, D. Fu, T. Wang, J. Zeng, Z.-J. Zhao, P. Zhang and J. Gong, J. Am. Chem. Soc., 2024, 146, 31927–31934 CrossRef CAS PubMed.
- C. Du, J. P. Mills, A. G. Yohannes, W. Wei, L. Wang, S. Lu, J.-X. Lian, M. Wang, T. Guo, X. Wang, H. Zhou, C.-J. Sun, J. Z. Wen, B. Kendall, M. Couillard, H. Guo, Z. Tan, S. Siahrostami and Y. A. Wu, Nat. Commun., 2023, 14, 6142 CrossRef CAS PubMed.
- D. Cheng, Z. Wei and P. Sautet, J. Am. Chem. Soc., 2025, 147, 10954–10965 CrossRef CAS PubMed.
- Q. Wu, Y. Li, Y. Cao, J. Li, J. Mao, X. Liu, W. Qu, P. Yan, Z. Cai, H. Lv, Y. Huang and Q. Li, Angew. Chem., Int. Ed., 2026, 138, e17120 CrossRef.
- G. Wang, Y. Liu, X. Zhang, X. Zong, X. Zhang, K. Zheng, D. Qu, L. An, X. Qi and Z. Sun, J. Am. Chem. Soc., 2024, 146, 8668–8676 CrossRef CAS PubMed.
- Y. Dai, H. Li, C. Wang, W. Xue, M. Zhang, D. Zhao, J. Xue, J. Li, L. Luo, C. Liu, X. Li, P. Cui, Q. Jiang, T. Zheng, S. Gu, Y. Zhang, J. Xiao, C. Xia and J. Zeng, Nat. Commun., 2023, 14, 3382 CrossRef CAS PubMed.
- P. Wei, D. Gao, T. Liu, H. Li, J. Sang, C. Wang, R. Cai, G. Wang and X. Bao, Nat. Nanotechnol., 2023, 18, 299–306 CrossRef CAS PubMed.
- Y. Rong, T. Liu, J. Sang, R. Li, P. Wei, H. Li, A. Dong, L. Che, Q. Fu, D. Gao and G. Wang, Angew. Chem., Int. Ed., 2023, 62, e202309893 CrossRef CAS PubMed.
- Y. Rong, X. Guo, G. Zhang, J. Sang, H. Li, D. Gao and G. Wang, ACS Energy Lett., 2024, 9, 3204–3211 CrossRef CAS.
- S. Roy, Z. Li, Z. Chen, A. C. Mata, P. Kumar, S. C. Sarma, I. F. Teixeira, I. F. Silva, G. Gao, N. V. Tarakina, M. G. Kibria, C. V. Singh, J. Wu and P. M. Ajayan, Adv. Mater., 2024, 36, 2300713 CrossRef CAS PubMed.
- L. Liu, J. Hu, Y. Sheng, H. Akhoundzadeh, W. Tu, W. J. S. Siow, J. H. Ong, H. Huang and R. Xu, ACS Nano, 2024, 18, 26271–26280 CAS.
- H. Cheng, X. Wu, X. Li, X. Nie, S. Fan, M. Feng, Z. Fan, M. Tan, Y. Chen and G. He, Chem. Eng. J., 2021, 407, 126842 CrossRef CAS.
- K. Lee, E. J. Kim, J. Kim, K. H. Kim, Y. J. Lee, M. J. Lee, K. Ryu, S. Shin, J. Choi, S. H. Kwon, H. Lee, J. K. Kim, B.-H. Kim, B. J. Kim and S. W. Lee, Adv. Energy Mater., 2024, 14, 2303803 CrossRef CAS.
- C. Zhan, F. Dattila, C. Rettenmaier, A. Herzog, M. Herran, T. Wagner, F. Scholten, A. Bergmann, N. López and B. Roldan Cuenya, Nat. Energy, 2024, 9, 1485–1496 CrossRef CAS PubMed.
- R. M. Arán-Ais, F. Scholten, S. Kunze, R. Rizo and B. Roldan Cuenya, Nat. Energy, 2020, 5, 317–325 CrossRef.
- Y. Liu, X. Wang, Z. Wang, C. Chen, J. Song, L. Zhang, W. Bao, B. Sun, L. Wang and D. Liu, ACS Catal., 2024, 14, 12610–12622 CrossRef CAS.
- Y. Yang, J. Zhang, Z. Tan, J. Yang, S. Wang, M. Li and Z. Su, Angew. Chem., Int. Ed., 2024, 136, e202408873 CrossRef.
- T. Hou, J. Zhu, H. Gu, X. Li, Y. Sun, Z. Hua, R. Shao, C. Chen, B. Hu, L. Mai, S. Chen, D. Wang and J. Zhang, Angew. Chem., Int. Ed., 2025, 137, e202424749 CrossRef.
- B. M. Tackett, D. Raciti, A. R. Hight Walker and T. P. Moffat, J. Phys. Chem. Lett., 2021, 12, 10936–10941 CrossRef CAS PubMed.
- R. Chen, Q. Wu, J. Zhu, S. Wang, Z. Hu, J. Hu, J. Zhu, H. Zhang, B. Ye, Y. Sun and Y. Xie, J. Am. Chem. Soc., 2025, 147, 7921–7931 CrossRef CAS PubMed.
- X. Guo, T. Liu, Y. Song, R. Li, P. Wei, Z. Liao, Z. Wu, D. Gao, Q. Fu, G. Wang and X. Bao, Angew. Chem., Int. Ed., 2025, 64, e202507062 CrossRef CAS PubMed.
- H. Matsushima, C. Haak, A. Taranovskyy, Y. Gründer and O. M. Magnussen, Phys. Chem. Chem. Phys., 2010, 12, 13992–13998 RSC.
- X. Huang, H. Yao, Y. Cui, W. Hao, J. Zhu, W. Xu and D. Zhu, ACS Appl. Mater. Interfaces, 2017, 9, 40752–40759 CrossRef CAS PubMed.
- Y. Qin, C. Xia, T. Wu, J. Zhang, G. Gao, B. Y. Xia, M. L. Coote, S. Ding and Y. Su, J. Am. Chem. Soc., 2024, 146, 32539–32549 CrossRef CAS PubMed.
- J. Sang, T. Liu, P. Wei, H. Li, C. Liu, Y. Wang, Y. Rong, Q. Wang, G. Wang and X. Bao, Energy Environ. Sci., 2024, 17, 3594–3603 RSC.
| This journal is © The Royal Society of Chemistry 2026 |