Open Access Article
Open Access ArticleRationally designed tetrahedral-configuration-matching methane trap in a metal–organic framework for efficient CH4/N2 separation
Yating Wanga,
Feifei Zhang*a,
Yanan Yanga,
Xiaoqing Wanga,
Jinping Li *ab and
Jiangfeng Yang
*ab and
Jiangfeng Yang *a
*a
aCollege of Chemistry and Chemical Engineering, Taiyuan University of Technology, Taiyuan 030024, Shanxi Province, China. E-mail: zhangfeifei0096@link.tyut.edu.cn; jpli211@hotmail.com; yangjiangfeng@tyut.edu.cn
bShanxi Research Institute of Huairou Laboratory, Taiyuan 030031, Shanxi Province, China
First published on 30th April 2026
Abstract
The enrichment and purification of CH4 from coalbed methane by adsorption are important but challenging. We propose a “tetrahedral-configuration-matching” metal–organic framework (MOF) methane trap, TUTJ-3Ni, with cyclopropyl groups precisely positioned to create a pre-configured pore structure that is geometrically complementary to tetrahedral CH4 molecules. TUTJ-3Ni exhibits a substantially higher CH4 adsorption heat (30.3 kJ mol−1) than its analogue TUTJ-2Ni (24.0 kJ mol−1), and this value is the highest among reported adsorbents. Moreover, TUTJ-3Ni exhibits a superior CH4/N2 selectivity of 11.1, the highest value reported for hydrophobic MOFs. In situ spectroscopy and theoretical modeling results elucidate that the matched tetrahedral binding pocket, constructed with two hydrogen atoms of the cyclopropyl group along with fluorine and oxygen atoms from the ligand, engages all four hydrogen atoms of CH4 via synergistic van der Waals interactions. Breakthrough experiment results verify that TUTJ-3Ni delivers high-purity CH4 (>99.9%) from coalbed methane and that its dynamic CH4 working capacity in humid environments is the highest among reported adsorbents. Furthermore, TUTJ-3Ni has good thermal and moisture stability and can easily be scaled up, making it promising for potential industrial applications.
1 Introduction
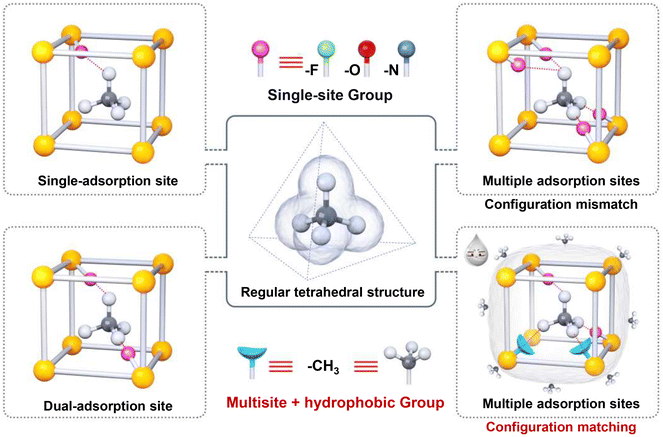
The global initiative to mitigate climate change and transition toward a low-carbon energy future has intensified the focus on unconventional natural gas resources. Among these, coalbed methane is a vast and promising reservoir.1–3 However, the efficient utilization of a significant portion of coalbed methane, often termed low-concentration coalbed methane (methane content less than 50%),4 remains a formidable challenge due to its inherently low calorific value. This gas stream is characterized by a high degree of dilution of methane (CH4) with inert nitrogen (N2).5,6 The core scientific challenge lies in achieving thermodynamically favorable and kinetically efficient separation of CH4 from N2. These two molecules exhibit remarkably similar physical properties, including nearly identical kinetic diameters of 3.8 Å for CH4 and 3.6 Å for N2, coupled with low polarizabilities (Fig. S1 and Table S1).7 This molecular similarity renders conventional separation technologies such as cryogenic distillation economically prohibitive.8,9 Consequently, billions of cubic meters of this valuable energy resource are wasted annually, exacerbating global energy insecurity while unnecessarily contributing to greenhouse gas emissions.10,11 The development of advanced separation materials capable of concurrently achieving high selectivity toward CH4 and exceptional stability under practical operating conditions has therefore emerged as a critical technological frontier with profound global implications.Metal–organic frameworks (MOFs), renowned for their ultrahigh surface areas and precisely tunable pore architectures, have emerged as promising candidates for gas separation.12–32 Their chemical versatility allows researchers to design pore environments that are tailored for specific molecule recognition.33,34 Researchers have made significant efforts to enhance the CH4 affinity of porous materials. One common strategy is to incorporate polar functional groups (F, N, and O) as single-site ligands, e.g., in fluorinated frameworks such as CuIn(3-Fina)4,35 nitrogen-containing structures such as Ni(ina)2,36 and oxygen-functionalized materials, including ATC-Cu.37 The limited efficacy of these materials arises from two fundamental issues. First, their isolated sites interact with the tetrahedral CH4 molecule via weak, non-specific van der Waals or electrostatic forces. Second, they fundamentally lack the complementary structural geometry required for specific molecular recognition. Consequently, they fail to provide a spatially matched and synergistic multi-point interaction that can enable the effective discrimination between CH4 and N2. This limitation is particularly relevant given the presence of a substantial amount of water vapor in practical coalbed methane extraction. Under such humid conditions, competitive water adsorption severely diminishes the CH4-adsorption performance. Water molecules readily form coordinate bonds with open metal sites or form extensive hydrogen-bonding networks, which in turn trigger irreversible structural collapse and pore blockage.38,39 While recent efforts have focused on developing hydrophobic MOFs, often through perfluoroalkyl functionalization, these strategies raise concerns about environmental persistence and cost.40,41 Overall, the establishment of a rational design principle that moves beyond mere hydrophobicity remains a formidable challenge. Such a principle must achieve specific, strong binding of the tetrahedral CH4 molecule instead of the linear N2, even in the presence of water.
Herein, we propose a “tetrahedral-configuration-matching” strategy inspired by the tetrahedral geometry of the methane molecule. As exhibited in Scheme 1, conventional adsorption sites—whether single, dual, or multiple—often suffer from insufficient binding strength or suboptimal spatial arrangement for effective CH4 capture. Our design focuses on engineering adsorption sites that mimic and synergistically interact with the CH4 tetrahedron. We focused on the methyl group (–CH3) as an ideal functional moiety. Its intrinsic tetrahedral symmetry offers natural geometric complementarity to the CH4 molecule, while the three outwardly directed hydrogen atoms increase the probability of optimal van der Waals contact. Furthermore, the well-established hydrophobicity of methyl groups ensures that performance is unaffected under moisture-rich conditions.42,43 Therefore, we conducted an accurate molecule-level experiment to rigorously verify the effectiveness of this strategy and clarify the relationship between the structure and performance. We selected two pharmaceutical ligands (norfloxacin and ciprofloxacin), which are structurally identical except for a single variation: norfloxacin bears an ethyl group (–CH2CH3), while ciprofloxacin features a cyclopropyl group (–CH2CH2–). By incorporating these ligands into MOFs constructed from 4-tert-butylsulfonylcalix[4]arene (H4SC4A-SO2) and Ni(II) nodes, we synthesized two isostructural MOFs, denoted as TUTJ-2Ni (ethyl) and TUTJ-3Ni (cyclopropyl).
Our investigation reveals that TUTJ-3Ni (TUT = Taiyuan University of Technology; J = methane) exhibits exceptional CH4 adsorption heat (30.3 kJ mol−1), CH4/N2 selectivity (11.1), and water resistance, and it substantially outperforms TUTJ-2Ni. Through a combination of experimental characterization and theoretical calculations, we demonstrate that the cyclopropyl group is not merely a hydrophobic unit but a key structural element that enforces a pre-organized, rigid pore geometry. This geometry facilitates “tetrahedral configuration matching”, enabling multi-point, synergistic dispersive interactions with the CH4 molecule. Thus, in this study, a new paradigm is established for designing hydrophobic porous materials through geometry-guided molecular engineering for challenging energy- and environment-related separations, offering a new pathway to mitigate the trade-off between selectivity and hydrothermal stability.
2 Results and discussion
2.1 Structure and characterization
The MOFs TUTJ-2Ni and TUTJ-3Ni were synthesized under solvothermal conditions by using Ni(II) salts, H4SC4A-SO2, and norfloxacin or ciprofloxacin ligands, respectively. Experimental details are provided in the SI. The powder X-ray diffraction (PXRD) patterns of both MOFs showed excellent agreement with that of TUTJ-3Co, confirming their isostructural relationship (Fig. S2–S4). Accordingly, the crystal structures of TUTJ-2Ni and TUTJ-3Ni (Fig. S5–S8 and Table S2) were determined via Rietveld refinement using TUTJ-3Co as the initial structural model.44As detailed in Fig. 1 and Table S2, structural analysis reveals that TUTJ-3Ni crystallizes in the monoclinic space group P21/n. Its fundamental secondary building unit consists of three Ni(II) ions, one H4SC4A-SO2 ligand, four ciprofloxacin molecules, and one nickel chloride hydrate unit. Two crystallographically equivalent Ni(II) centers exhibit identical coordination environments. Each Ni(II) ion forms coordinate bonds with sulfonyl and phenolic oxygen atoms from the H4SC4A-SO2 ligand, forming Ni–O bonds. In addition, they establish Ni–N and Ni–O bonds with nitrogen atoms and carboxylate oxygen atoms from two separate ciprofloxacin molecules. The third Ni(II) ion forms coordinate bonds with oxygen atoms of the calixarene skeleton and bridges two ciprofloxacin ligands via carboxylate oxygen atoms. The fourth Ni(II) center forms coordinate bonds with carboxylate oxygen atoms and carbonyl groups from two ciprofloxacin ligands, as well as with chloride ions and water molecules present in the starting materials.
These Secondary Building Units (SBUs) assemble into one-dimensional chains, wherein each calix[4]arene unit connects to four ciprofloxacin molecules. In the crystal lattice, these one-dimensional chains are not isolated. They undergo highly ordered parallel alignment along the other two crystallographic directions through interchain secondary π–π interactions, thereby assembling into a three-dimensional crystalline architecture.42 The resulting structure features well-defined micropores with channel dimensions of approximately 12 × 6.5 Å2 (Fig. S9 and S10). The structure of TUTJ-2Ni is similar to that of TUTJ-3Ni, differing only in the substitution of an ethyl moiety for the terminal cyclopropyl group in TUTJ-2Ni. This substitution results in a reduction of the channel dimensions from 12 × 7 Å2 in TUTJ-2Ni to 12 × 6.5 Å2 in TUTJ-3Ni. The narrowed pore size is expected to enhance host–guest compatibility. More importantly, the pre-organized cyclopropyl groups in TUTJ-3Ni create a geometrically optimized binding pocket that exhibits potential structural matching with the tetrahedral CH4 molecule, whereas the ethyl groups in TUTJ-2Ni lead to a less defined pore architecture. Consequently, such precise geometric matching is expected to contribute to superior gas separation performance in the cyclopropyl-functionalized framework.
The phase purity of the as-synthesized TUTJ-2Ni and TUTJ-3Ni was confirmed by performing PXRD and SEM analyses. As shown in Fig. S11–S13, the experimental diffraction patterns of both materials exhibit excellent agreement with their respective simulated patterns, confirming successful synthesis with high crystallinity and phase purity. Thermogravimetric analysis (TGA) results reveal outstanding thermal stability for both frameworks (Fig. S14). TUTJ-2Ni maintained its structural integrity up to 573 K, while TUTJ-3Ni exhibited even enhanced stability, remaining stable up to 593 K. The TGA curves of the activated samples further corroborated these findings, indicating excellent thermal stability suitable for practical applications. The permanent porosity of activated TUTJ-2Ni and TUTJ-3Ni was confirmed by N2 sorption measurements at 77 K (Fig. 2a and S15–S17). Both frameworks exhibit typical Type I isotherms, characteristic of microporous materials. The Brunauer–Emmett–Teller (BET) theory was used to determine the specific surface area that was found to be 614.5 m2 g−1 for TUTJ-2Ni and 605.5 m2 g−1 for TUTJ-3Ni, with corresponding total pore volumes of 0.41 cm3 g−1 and 0.40 cm3 g−1, respectively. Pore size distributions analyzed by the non-local density functional theory (NLDFT) revealed main peaks at 7.36 Å and 6.30 Å for TUTJ-2Ni and TUTJ-3Ni, respectively, demonstrating how the cyclopropyl substitution effectively reduces the pore dimensions while maintaining the structural integrity. Fourier-transform infrared (FT-IR) spectroscopy results enabled additional structural verification (Fig. 2b). Two sharp peaks observed at approximately 2965 cm−1 and 2867 cm−1 were attributed to C–H stretching vibrations of the –CH3 groups, confirming the presence of abundant methyl groups in the framework.45 Additional characteristic peaks at 1494 cm−1 were assigned to ν(C–H) vibrations, consistent with the expected chemical structure.
TUTJ-3Ni demonstrates exceptional stability under various extreme conditions, particularly maintaining complete structural integrity in highly humid environments. The framework remains unaffected after prolonged exposure to 100% relative humidity for a month, along with resistance to strong acids (pH = 1), bases (pH = 13), organic solvents, and high temperatures up to 573 K. The corresponding PXRD patterns and BET results (Fig. 2c, d and S18–S20) confirm the perfect preservation of the crystalline structure with no detectable degradation. This remarkable stability, especially the excellent water resistance, originates from the synergistic combination of several factors: (i) the rigid calix[4]arene scaffold that resists structural deformation; (ii) the fully coordinated Ni(II) centers with strong Ni–O and Ni–N bonds, which eliminate open metal sites prone to hydrolysis; (iii) the interdigitated chain architecture further stabilized by interchain π–π stacking interactions; and (iv) the hydrophobic pore surfaces decorated with abundant –CH3 and cyclopropyl groups that effectively repel water molecules.46 The outstanding moisture stability positions TUTJ-3Ni as an ideal candidate for practical separation applications under challenging humid conditions.
2.2 Gas adsorption performance
The gas adsorption performance of TUTJ-3Ni and TUTJ-2Ni was systematically investigated to evaluate their efficacy in performing CH4/N2 separation. Single-component adsorption isotherms measured at 298 K demonstrate that TUTJ-3Ni achieves a CH4 uptake of 18.6 cm3 g−1 at 1 bar, substantially exceeding both its N2 adsorption capacity (4.9 cm3 g−1) and the CH4 uptake of TUTJ-2Ni (10.5 cm3 g−1), as shown in Fig. 3a and S21–S23. The pronounced difference in gas affinities between the two frameworks highlights the significant impact of ligand functionalization on the adsorption behavior.The separation potential of these materials was further quantified through IAST selectivity calculations (Fig. 3b and S24). TUTJ-3Ni exhibits a CH4/N2 (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50) selectivity of 11.1, markedly superior to the value of 6.19 observed for TUTJ-2Ni. This value represents the highest CH4/N2 selectivity among all compared hydrophobic MOFs (Table S4), including NKMOF-8-Me (9.0),47 DMOF-A2 (7.2),48 TUT-100 (6.3),49 and MIL-120Al (6.0),50 positioning TUTJ-3Ni as the most effective material for CH4/N2 separation. Notably, IAST calculations for low-concentration CH4/N2 mixtures (20
50) selectivity of 11.1, markedly superior to the value of 6.19 observed for TUTJ-2Ni. This value represents the highest CH4/N2 selectivity among all compared hydrophobic MOFs (Table S4), including NKMOF-8-Me (9.0),47 DMOF-A2 (7.2),48 TUT-100 (6.3),49 and MIL-120Al (6.0),50 positioning TUTJ-3Ni as the most effective material for CH4/N2 separation. Notably, IAST calculations for low-concentration CH4/N2 mixtures (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80, 15
80, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85 and 5
85 and 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95) still give high selectivity, further supporting the practical applicability of TUTJ-3Ni for treating low-concentration coalbed methane.
95) still give high selectivity, further supporting the practical applicability of TUTJ-3Ni for treating low-concentration coalbed methane.
Moreover, the heat of adsorption (Qst), derived from adsorption isotherms collected at 273 and 298 K via the Clausius–Clapeyron equation, provides direct evidence of the enhanced CH4 affinity of TUTJ-3Ni (Fig. S25–S27 and Table S3). The CH4 adsorption heat of TUTJ-3Ni (30.3 kJ mol−1) is significantly higher than that of TUTJ-2Ni (24.0 kJ mol−1) (Fig. 3c). The CH4 adsorption heat of TUTJ-3Ni was compared with those of several representative benchmark adsorbents, as summarized in Fig. 3d and Table S5. TUTJ-3Ni demonstrates the highest Qst value among all the materials, such as Ni(ina)2 (28.0 kJ mol−1),36 NKMOF-8-Me (28.0 kJ mol−1),47 CoNi-TED (27.7 kJ mol−1),51 and ATC-Cu (26.8 kJ mol−1).37 This comparison directly confirms the effectiveness of the “tetrahedral-geometry-matching” strategy, where the pre-organized cyclopropyl groups create a pore structure that maximizes van der Waals interactions with the tetrahedral CH4 molecules.
A comprehensive three-dimensional performance assessment shows that TUTJ-3Ni is a promising CH4/N2 separation material (Fig. 3e), as it demonstrates an optimal balance among adsorption capacity, selectivity, and binding strength. The exceptional performance of TUTJ-3Ni is attributed to the introduction of cyclopropyl groups, which generate geometrically matched adsorption sites exhibiting optimal complementarity to the tetrahedral CH4 molecule—a feature absents in the ethyl-functionalized framework. The results of kinetic studies further demonstrate the separation capability of TUTJ-3Ni, which exhibits a kinetic selectivity of 1.59 for CH4 over N2, surpassing the value of 1.36 observed for TUTJ-2Ni (Fig. 3f and S28). Remarkably, complete desorption could be achieved within approximately 15 min under high vacuum conditions, indicating both favorable diffusion kinetics and excellent degree or regeneration of the material. From a practical perspective, TUTJ-3Ni demonstrates long-term stability, retaining over 97.5% of its initial CH4 adsorption capacity at 1 bar and 298 K after 120 consecutive adsorption–desorption cycles under vacuum-assisted regeneration (Fig. 3g). This remarkable retention of performance, achieved through a simple and energy-efficient regeneration process, highlights the structural robustness of the framework and its strong potential for industrial separation applications requiring durable adsorbent materials. Therefore, TUTJ-3Ni is an advanced adsorbent material that combines configuration matching for selective CH4 capture with rapid regeneration kinetics and long-term cycling stability, establishing a new standard for practical methane separation techniques.
2.3 Tetrahedral binding site identification
To elucidate the molecular-level mechanism underlying the superior CH4/N2 separation performance of TUTJ-3Ni, we employed a combination of in situ spectroscopic techniques and computational modeling. In situ infrared spectroscopy measurements were conducted to probe the host–guest interactions in both TUTJ-3Ni and TUTJ-2Ni frameworks (Fig. 4a–c, S29 and S30). As shown in Fig. 4a–c, upon CH4 loading, two distinct vibrational bands emerge at 1303 and 3014 cm−1, corresponding to δ(C–H) bending and ν(C–H) stretching modes, respectively.52,53 The progressive intensification of these bands with increasing CH4 exposure demonstrates the strong physical adsorption of methane within the framework, attributed to multiple van der Waals interactions between CH4 molecules and the pore walls. In contrast, parallel measurements with N2 revealed no discernible spectral features for either material (Fig. S30), confirming their weak affinity toward nitrogen. These spectroscopic results provide direct experimental evidence of the enhanced CH4 adsorption capacity and selectivity in TUTJ-3Ni.Theoretical modeling results provided molecular-level insight into the origin of the pronounced CH4/N2 separation performance of TUTJ-3Ni. Grand canonical Monte Carlo (GCMC) simulations at 298 K and 1 bar revealed that the adsorption density of CH4 is significantly higher than that of N2 within the framework, consistent with the experimental observations (Fig. S31). GCMC simulations initially identified three potential methane adsorption sites in TUTJ-3Ni. Analysis of site occupancy under reduced pressures showed that only two primary binding sites remain significantly populated at 0.1 bar. This finding allowed us to focus on the dominant adsorption site, which exhibits exceptional geometric complementarity to the tetrahedral CH4 molecule (Fig. S32). The results of density functional theory (DFT) calculations on the optimized geometry provided atomic-level details of the host–guest interactions (Fig. 4d, e and S33–S35). The results demonstrated that all four hydrogen atoms of a single CH4 molecule engage concurrently with various binding sites on the organic ligand. At this site, the CH4 molecule establishes multiple specific contacts: C–H⋯F (2.865 Å) and C–H⋯O (2.999 Å) interactions exist, and notably, two C–H⋯H–C contacts are formed with the cyclopropyl group at distances of 2.725 Å and 2.915 Å (Fig. 4d). This achievement is in sharp contrast to other reported materials. Al-CDC provides only two binding sites, while TUTJ-201Ni offers three, both insufficient for optimal CH4 engagement.20,54 Although Ni(ina)2 possesses four potential sites, their spatial arrangement permits interaction with merely three of the four hydrogen atoms of CH4 owing to geometric mismatch.36 In comparison, TUTJ-3Ni achieves complete tetrahedral coordination through its precisely aligned cyclopropyl functionalization. In stark contrast, the corresponding interactions in TUTJ-2Ni are substantially longer (C–H⋯F: 3.139 Å; C–H⋯O: 3.449 Å) and lack the defined geometric arrangement (Fig. S33). Furthermore, the calculated N2 binding distances (3.205–3.706 Å) are substantially longer than the CH4 binding distances (Fig. 4e), explaining the framework's pronounced selectivity toward methane capture through optimal geometric matching. As shown in Fig. S35, the binding energy calculated for the adsorption site I in TUTJ-3Ni reaches −32.07 kJ mol−1 for CH4, which is substantially higher than that for N2 (−18.29 kJ mol−1) and significantly exceeds the CH4 binding energy in the case of TUTJ-2Ni (−23.62 kJ mol−1). These results align remarkably well with the experimentally derived values of the heat of adsorption, confirming the reliability of our theoretical models.
We performed an independent gradient model (IGM) analysis based on the DFT-optimized structures. The results visually demonstrate multiple van der Waals interactions between the hydrogen atoms of the CH4 molecule and specific sites within the pore environment of TUTJ-3Ni. These interactions collectively establish a well-defined, tetrahedral-configuration-matching binding geometry that effectively encapsulates the CH4 molecule (Fig. 4f). In contrast, such synergistic multi-point interaction patterns are notably absent in the case of N2 adsorption on TUTJ-3Ni, and in both CH4 and N2 adsorption on TUTJ-2Ni (Fig. 4g and S36). TUTJ-2Ni cannot form a structurally complementary binding environment, and both frameworks exhibit inherently weak affinity toward the linear N2 molecule. Collectively, the results of these multiscale investigations establish a direct correlation between the exceptional separation performance of TUTJ-3Ni and its structurally complementary adsorption geometry. The cyclopropyl-functionalized framework creates pre-organized binding pockets that enable optimized van der Waals interactions with tetrahedral CH4 molecules, thus presenting a critical design principle for the development of advanced gas separation materials.
2.4 Dynamic breakthrough studies
We evaluated the industrial application potential of the developed materials for coalbed methane upgrading by performing dynamic breakthrough experiments using a custom-built apparatus (Fig. S37 and S38). When an equimolar CH4/N2 mixture at 5 mL min−1 was used, TUTJ-3Ni demonstrates exceptional separation efficacy, with N2 eluting at 2.2 min g−1, while CH4 breaks through significantly later at 5.8 min g−1, resulting in a substantial breakthrough time difference of 3.6 min g−1 (Fig. 5a). This pronounced retention contrast markedly surpasses the performance of TUTJ-2Ni, where both components break through at approximately 1.3 min g−1, indicating inadequate separation capability. The practical versatility of TUTJ-3Ni was further validated through systematic evaluation across varying operational parameters. TUTJ-3Ni retains a robust separation performance under different flow conditions and CH4/N2 compositions (50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50, 20
50, 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 80, 15
80, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85, and 5
85, and 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95), demonstrating its adaptability to fluctuating feed conditions commonly encountered in industrial settings (Fig. S39–S41). Notably, during He-purge desorption, high-purity CH4 (>99.9%) can be efficiently recovered with negligible N2 contamination (Fig. 5b), highlighting the material's renewability and potential for realizing pure-product collection. The framework exhibits outstanding cycling stability, maintaining identical breakthrough profiles over ten consecutive adsorption–desorption cycles (Fig. S42).
95), demonstrating its adaptability to fluctuating feed conditions commonly encountered in industrial settings (Fig. S39–S41). Notably, during He-purge desorption, high-purity CH4 (>99.9%) can be efficiently recovered with negligible N2 contamination (Fig. 5b), highlighting the material's renewability and potential for realizing pure-product collection. The framework exhibits outstanding cycling stability, maintaining identical breakthrough profiles over ten consecutive adsorption–desorption cycles (Fig. S42).
Because of the high-humidity environment that is characteristic of coalbed methane streams, we critically assessed the hydrophobicity and moisture stability of both materials. TUTJ-3Ni displays superior hydrophobicity with a water contact angle of 134° and minimal moisture uptake (Fig. S43). Most remarkably, the breakthrough performance of TUTJ-3Ni remains virtually unaffected under 100% relative humidity conditions (Fig. 5c), whereas TUTJ-2Ni suffers noticeable performance degradation (Fig. S44). This exceptional moisture tolerance is maintained over multiple cycles in saturated humidity environments, confirming the practical viability of TUTJ-3Ni for real-world methane purification applications (Fig. 5d). The comprehensive breakthrough studies establish TUTJ-3Ni as a technologically advanced adsorbent that combines CH4 affinity with exceptional hydrolytic stability, enabling efficient methane recovery from nitrogen-diluted streams under relevant practical-operation conditions. The material demonstrates robust performance with varying feed compositions, excellent regeneration, and sustained efficiency in humid environments. These qualities position it as a promising candidate for industrial coalbed methane upgrading.
2.5 Hydrolytic stability and practical performance validation
The practical applicability of TUTJ-3Ni under humid conditions was assessed through comparative studies with three benchmark CH4/N2 separation materials: ATC-Cu,37 Ni(ina)2,36 and CoNi-TED.51 Contact angle measurements revealed that TUTJ-3Ni exhibits exceptional hydrophobicity (134°), while water droplets were instantly absorbed by the benchmark materials (Fig. S45 and S46). After one week of aging at 100% relative humidity, TUTJ-3Ni retained its crystalline structure, whereas ATC-Cu and CoNi-TED showed significant peak attenuation, and Ni(ina)2 underwent complete structural collapse (Fig. S47). Correspondingly, TUTJ-3Ni retained over 93% of its original CH4 and N2 uptake capacity, while the reference materials suffered substantial capacity loss (Fig. S48–S52). Under dry feed conditions, all four materials demonstrated effective CH4/N2 separation (Fig. S53). However, upon exposure to 100% relative humidity, the benchmark materials exhibited progressive performance degradation with each cycle, while TUTJ-3Ni retained consistent separation efficiency over five consecutive cycles (Fig. S54–S58). Quantitative analysis showed that TUTJ-3Ni achieved the highest CH4 working capacity under dry conditions (18.27 cm3 g−1) and retained 98% of this capacity after five humid cycles, compared to only 5–42% retention for the reference materials (Fig. S59–S61). Radar chart analysis (Fig. S62) integrating breakthrough time, adsorption capacity, and dynamic performance metrics confirmed that TUTJ-3Ni retained >93% of all evaluated parameters under harsh humid conditions, significantly outperforming the benchmark materials (5–80%) and establishing it as a leading candidate for practical coalbed methane upgrading.The transition from laboratory-scale synthesis to industrial implementation represents a critical step in MOF development. To identify the scalability challenges, we successfully synthesized a TUTJ-3Ni sample with a 100-fold increase in scale while maintaining excellent phase purity (Fig. S63), as confirmed by PXRD analysis results (Fig. S64a). The scaled-up and laboratory-scale specimens exhibit nearly identical gas adsorption capacity and selectivity (Fig. S64b), demonstrating the viability of large-scale production without compromising performance. This successful scale-up, combined with the exceptional hydrolytic stability and separation performance, positions TUTJ-3Ni as a technologically advanced adsorbent ready for industrial applications in coalbed methane purification.
3 Conclusions
In summary, we have successfully demonstrated a geometry-guided molecular engineering strategy for overcoming the ubiquitous trade-off between selectivity and hydrothermal stability in CH4/N2 separation. By incorporating rigid cyclopropyl groups into the framework of TUTJ-3Ni, we constructed a pre-organized pore environment with exceptional geometric complementarity to the tetrahedral CH4 molecule, which enables synergistic multi-point van der Waals interactions for highly selective methane capture. This “tetrahedral-configuration-matching” strategy endows TUTJ-3Ni with remarkable CH4/N2 selectivity, and, most importantly, excellent hydrolytic stability, maintaining its structural integrity and separation performance even under 100% relative humidity. The material's practical viability is unequivocally confirmed by its excellent dynamic separation efficiency, robust cyclability, and successful scale-up. Therefore, this work establishes a new paradigm for designing hydrophobic adsorbents via geometric matching. It offers a promising path for advanced energy-related separations under challenging practical conditions.Author contributions
Y. W. and F. Z. conceived and designed the research. Y. W. and Y. Y. performed most of the experiments. X. W. and F. Z. analysed the data. Y. W. and F. Z. wrote the manuscript. J. L. and J. Y. supervised the project. All authors participated in and contributed to the preparation of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2492179 (for TUTJ-2Ni), 2492185 (for TUTJ-3Ni) and 2495218 (for TUTJ-3Co) contain the supplementary crystallographic data for this paper.44a–cThe data that support the findings of this study are openly available in the supplementary information (SI). Supplementary information: experimental procedures, gas adsorption isotherms, crystallographic data, and theoretical computational details. See DOI: https://doi.org/10.1039/d6sc02393b.
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (No. 22408258, 22478272, and 22378287), Joint Funds of the National Natural Science Foundation of China (U25A20624) and Natural Science Foundation of Shanxi Province (No. 202303021222012).Notes and references
- L. Grant, I. Vanderkelen, L. Gudmundsson, E. Fischer, S. I. Seneviratne and W. Thiery, Global emergence of unprecedented lifetime exposure to climate extremes, Nature, 2025, 641, 374–379 CrossRef CAS PubMed.
- J. C. Stroeve, D. Notz, J. Dawson, E. A. G. Schuur, D. Dahl-Jensen and C. Giesse, Disappearing landscapes: The Arctic at +2.7°C global warming, Science, 2025, 387, 616–621 CrossRef CAS PubMed.
- E. McFarland, Unconventional Chemistry for Unconventional Natural Gas, Science, 2012, 338, 340–342 CrossRef CAS PubMed.
- C. Liu, Y. Zhou, Y. Sun, W. Su and L. Zhou, Enrichment of coal-bed methane by PSA complemented with CO2 displacement, AIChE J., 2011, 57, 645–654 CrossRef CAS.
- H. Shang, F. Zhang, J. Liu, X. Zhang, J. Yang, L. Li and J. Li, Enriching Low-Concentration Coalbed Methane Using a Hydrophobic Adsorbent under Humid Conditions, Ind. Eng. Chem. Res., 2021, 60, 12689–12697 CrossRef CAS.
- T. A. Moore, Coalbed methane: A review, Int. J. Coal Geol., 2012, 101, 36–81 CrossRef CAS.
- L. Li, L. Yang, J. Wang, Z. Zhang, Q. Yang, Y. Yang, Q. Ren and Z. Bao, Highly efficient separation of methane from nitrogen on a squarate-based metal-organic framework, AIChE J., 2018, 64, 3681–3689 CrossRef CAS.
- D. M. Ruthven, Past Progress and Future Challenges in Adsorption Research, Ind. Eng. Chem. Res., 2000, 39, 2127–2131 CrossRef CAS.
- Y. Gensterblum, A. Busch and B. M. Krooss, Molecular concept and experimental evidence of competitive adsorption of H2O, CO2 and CH4 on organic material, Fuel, 2014, 115, 581–588 CrossRef CAS.
- D. S. Sholl and R. P. Lively, Seven chemical separations to change the world, Nature, 2016, 532, 435–437 CrossRef PubMed.
- S. J. Davis and C. Shearer, A crack in the natural-gas bridge, Nature, 2014, 514, 436–437 CrossRef CAS PubMed.
- X. Han, H. G. W. Godfrey, L. Briggs, A. J. Davies, Y. Cheng, L. L. Daemen, A. M. Sheveleva, F. Tuna, E. J. L. McInnes, J. Sun, C. Drathen, M. W. George, A. J. Ramirez-Cuesta, K. M. Thomas, S. Yang and M. Schröder, Reversible adsorption of nitrogen dioxide within a robust porous metal–organic framework, Nat. Mater., 2018, 17, 691–696 CrossRef CAS PubMed.
- S. Zhou, O. Shekhah, A. Ramírez, P. Lyu, E. Abou-Hamad, J. Jia, J. Li, P. M. Bhatt, Z. Huang, H. Jiang, T. Jin, G. Maurin, J. Gascon and M. Eddaoudi, Asymmetric pore windows in MOF membranes for natural gas valorization, Nature, 2022, 606, 706–712 CrossRef CAS PubMed.
- M. Eddaoudi, J. Kim, N. Rosi, D. Vodak, J. Wachter, M. O'Keeffe and O. M. Yaghi, Systematic Design of Pore Size and Functionality in Isoreticular MOFs and Their Application in Methane Storage, Science, 2002, 295, 469–472 CrossRef CAS PubMed.
- J.-R. Li, R. J. Kuppler and H.-C. Zhou, Selective gas adsorption and separation in metal–organic frameworks, Chem. Soc. Rev., 2009, 38, 1477–1504 RSC.
- H.-C. Zhou, J. R. Long and O. M. Yaghi, Introduction to Metal–Organic Frameworks, Chem. Rev., 2012, 112, 673–674 CrossRef CAS PubMed.
- H. Furukawa, K. E. Cordova, M. O'Keeffe and O. M. Yaghi, The Chemistry and Applications of Metal-Organic Frameworks, Science, 2013, 341, 1230444 CrossRef PubMed.
- F. Zhang, Z. Zhao, Y. Wang, X. Li, X. Bai, M. Lu, Y. Wang, X. Wang, L. Li, J. Li and J. Yang, Nitrogen Adsorption Sites with Low Polarizability for Benchmark N2/CH4 Separation, Angew. Chem., Int. Ed., 2025, 64, e202510242 CrossRef CAS PubMed.
- H. Zhang, J. Tang, C. Yu, M. Zhang, J. Wang and J. Duan, Oriented design and engineering of advanced metal–organic frameworks for light hydrocarbon separations, Chem. Sci., 2025, 16, 11768–11800 RSC.
- Z. Zhu, J. Xiao, M. Zhang, Y. Huang and S. Yuan, Precision pore engineering via fit-topology assembly in a Zn-porphyrin MOF for selective C2H2 capture, Chem. Sci., 2025, 16, 22638–22646 RSC.
- H. Li, M. Eddaoudi, M. O'Keeffe and O. M. Yaghi, Design and synthesis of an exceptionally stable and highly porous metal-organic framework, Nature, 1999, 402, 276–279 CrossRef CAS.
- S. Kitagawa, R. Kitaura and S.-i. Noro, Functional Porous Coordination Polymers, Angew. Chem., Int. Ed., 2004, 43, 2334–2375 CrossRef CAS PubMed.
- P. Li, N. A. Vermeulen, C. D. Malliakas, D. A. Gómez-Gualdrón, A. J. Howarth, B. L. Mehdi, A. Dohnalkova, N. D. Browning, M. O'Keeffe and O. K. Farha, Bottom-up construction of a superstructure in a porous uranium-organic crystal, Science, 2017, 356, 624–627 CrossRef CAS PubMed.
- R.-B. Lin, Z. Zhang and B. Chen, Achieving High Performance Metal–Organic Framework Materials through Pore Engineering, Acc. Chem. Res., 2021, 54, 3362–3376 CrossRef CAS PubMed.
- F. Zhang, H. Shang, L. Wang, Y. Wang, J. Yang, Y. Xia, H. Li, L. Li and J. Li, Construction of a Porous Metal–Organic Framework with a High Density of Open Cr Sites for Record N2/O2 Separation, Adv. Mater., 2021, 33, 2100866 CrossRef CAS PubMed.
- F. Zhang, H. Shang, B. Zhai, Z. Zhao, Y. Wang, L. Li, J. Li and J. Yang, Synergistic Nitrogen Binding Sites in a Metal-Organic Framework for Efficient N2/O2 Separation, Angew. Chem., Int. Ed., 2023, 62, e202316149 CrossRef CAS PubMed.
- Y.-L. Zhao, X. Bai, X. Zhang, Z.-Y. Han and J.-R. Li, Record high CH4/N2 adsorption separation selectivity in a scalable metal-organic framework, Sci. Bull., 2025, 70, 1215–1218 CrossRef CAS PubMed.
- R.-B. Lin, S. Xiang, W. Zhou and B. Chen, Microporous Metal-Organic Framework Materials for Gas Separation, Chem, 2020, 6, 337–363 CAS.
- K. M. Carsch, H. Z. H. Jiang, R. A. Klein, A. S. Rosen, P. S. Summerhill, J. L. Peltier, A. J. Huang, R. A. Murphy, M. N. Dods, H. A. Silva, Z. Hasanbasri, H. Kwon, S. L. Karstens, Y. Yabuuchi, J. Börgel, J. W. Taylor, K. R. Meihaus, K. C. Bustillo, A. M. Minor, K. A. Persson, C. M. Brown, R. D. Britt, N. P. Stadie and J. R. Long, Multigas adsorption with single-site cooperativity in a metal–organic framework, Science, 2025, 390, 808–812 CrossRef CAS PubMed.
- O. I.-F. Chen, C.-H. Liu, K. Wang, E. Borrego-Marin, H. Li, A. H. Alawadhi, J. A. R. Navarro and O. M. Yaghi, Water-Enhanced Direct Air Capture of Carbon Dioxide in Metal–Organic Frameworks, J. Am. Chem. Soc., 2024, 146, 2835–2844 CrossRef CAS PubMed.
- Z. Chen, K. O. Kirlikovali, P. Li and O. K. Farha, Reticular Chemistry for Highly Porous Metal–Organic Frameworks: The Chemistry and Applications, Acc. Chem. Res., 2022, 55, 579–591 CrossRef CAS PubMed.
- Z.-M. Ye, Y. Xie, K. O. Kirlikovali, S. Xiang, O. K. Farha and B. Chen, Architecting Metal–Organic Frameworks at Molecular Level toward Direct Air Capture, J. Am. Chem. Soc., 2025, 147, 5495–5514 CrossRef CAS PubMed.
- A. Schneemann, V. Bon, I. Schwedler, I. Senkovska, S. Kaskel and R. A. Fischer, Flexible metal–organic frameworks, Chem. Soc. Rev., 2014, 43, 6062–6096 RSC.
- G. Armstrong, Marvellous MOFs, Nat. Chem., 2008 DOI:10.1038/nchem.41.
- L.-P. Zhang, L. Xu, X.-T. Zhang, Y.-T. Li, H.-L. Lan, S.-C. Liu and Q.-Y. Yang, Pore Chemical Modification of Bimetallic Coordination Networks for Coal-Bed Methane Purification under Humid Conditions, Inorg. Chem., 2025, 64, 1596–1603 CrossRef CAS PubMed.
- S.-M. Wang, M. Shivanna and Q.-Y. Yang, Nickel-Based Metal–Organic Frameworks for Coal-Bed Methane Purification with Record CH4/N2 Selectivity, Angew. Chem., Int. Ed., 2022, 61, e202201017 CrossRef CAS PubMed.
- Z. Niu, X. Cui, T. Pham, P. C. Lan, H. Xing, K. A. Forrest, L. Wojtas, B. Space and S. Ma, A Metal–Organic Framework Based Methane Nano-trap for the Capture of Coal-Mine Methane, Angew. Chem., Int. Ed., 2019, 131, 10244–10247 CrossRef.
- J. J. Low, A. I. Benin, P. Jakubczak, J. F. Abrahamian, S. A. Faheem and R. R. Willis, Virtual High Throughput Screening Confirmed Experimentally: Porous Coordination Polymer Hydration, J. Am. Chem. Soc., 2009, 131, 15834–15842 CrossRef CAS PubMed.
- T. He, X.-J. Kong and J.-R. Li, Chemically Stable Metal–Organic Frameworks: Rational Construction and Application Expansion, Acc. Chem. Res., 2021, 54, 3083–3094 CrossRef CAS PubMed.
- S. Pal, S. Kulandaivel, Y.-C. Yeh and C.-H. Lin, Recent trends in superhydrophobic metal–organic frameworks and their diverse applications, Coord. Chem. Rev., 2024, 518, 216108 CrossRef CAS.
- M. Zheng, W. Xue, T. Yan, Z. Jiang, Z. Fang, H. Huang and C. Zhong, Fluorinated MOF-Based Hexafluoropropylene Nanotrap for Highly Efficient Purification of Octafluoropropane Electronic Specialty Gas, Angew. Chem., Int. Ed., 2024, 63, e202401770 CrossRef CAS PubMed.
- F. Zhang, Z. Zhao, Y. Wang, Y. Tang, X. Li, X. Bai, M. Lu, L. Li, J. Li and J. Yang, Installing Methyl Group Guards in a Metal–Organic Framework Featuring Multiple N/O Active Sites for Efficient C2H2/CO2 Separation under Moist Conditions, ACS Mater. Lett., 2024, 6, 4482–4490 CrossRef CAS.
- T. Li, L. Zhang, Y. Wang, X. Jia, H. Chen, Y. Li, Q. Shi, L.-B. Sun, J. Li, B. Chen and L. Li, Optimizing supramolecular interactions within metal–organic frameworks for ultra-high purity propylene purification, AIChE J., 2025, 71, e18646 CrossRef CAS.
- (a) CCDC 2492179: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pn9wk; (b) CCDC 2492185: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pnb2s; (c) CCDC 2495218: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2prgxv.
- Z. Zhao, Y. Li, Y. Wang, Y. Wang, Y. Tang, M. Lu, X. Yang, X. Wang, F. Zhang, J. Li and J. Yang, Hydrophobic metal-organic framework Featuring multiple O/S active sites for efficient CH4/N2 separation under humid conditions, Sep. Purif. Technol., 2025, 361, 131382 CrossRef CAS.
- I. V. Khariushin, V. Bulach, S. E. Solovieva, I. S. Antipin, A. S. Ovsyannikov and S. Ferlay, Thiacalix[4]arene macrocycles as versatile building blocks for the rational design of high-nuclearity metallic clusters, metallamacrocycles, porous coordination cages and containers, Coord. Chem. Rev., 2024, 513, 215846 CrossRef CAS.
- M. Chang, F. Wang, Y. Wei, Q. Yang, J.-X. Wang, D. Liu and J.-F. Chen, Separation of CH4/N2 by an ultra-stable metal–organic framework with the highest breakthrough selectivity, AIChE J., 2022, 68, e17794 CrossRef CAS.
- T. Li, X. Jia, H. Chen, Z. Chang, L. Li, Y. Wang and J. Li, Tuning the Pore Environment of MOFs toward Efficient CH4/N2 Separation under Humid Conditions, ACS Appl. Mater. Interfaces, 2022, 14, 15830–15839 CrossRef CAS PubMed.
- J. Liu, X. Tang, X. Liang, L. Wu, F. Zhang, Q. Shi, J. Yang, J. Dong and J. Li, Superhydrophobic zeolitic imidazolate framework with suitable SOD cage for effective CH4/N2 adsorptive separation in humid environments, AIChE J., 2022, 68, e17589 CrossRef CAS.
- F. Zhang, H. Shang, B. Zhai, X. Li, Y. Zhang, X. Wang, J. Li and J. Yang, Thermodynamic-kinetic synergistic separation of CH4/N2 on a robust aluminum-based metal-organic framework, AIChE J., 2023, 69, e18079 CrossRef CAS.
- P. Guo, J. Fu, B. Xue, T. Wang, L. Yang, Y. Ying, Q. Yang and D. Liu, One Flexible Metal–Organic Framework with Guest-Adaptive Rotational Valve for Gas Separation, Adv. Funct. Mater., 2025, 35, 2500210 CrossRef CAS.
- P. Liu, L. Fan, J. Li, Z. Wu, Y. Wang, Y. Chen, J. Gao, J. Yang, J. Li, B. Chen and L. Li, A Microporous Titanium Metal–Organic Framework with Double Nanotraps for Record CH4/N2 Separation, Chem. Mater., 2024, 36, 2925–2932 CrossRef CAS.
- M. Chang, Y. Li, M. Tong, J. Yang, T. Ma, Y. Wang and J. Zheng, Synergistic Binding Sites in a Robust and Scalable Metal–Organic Framework for Record CH4 Capture, Small, 2025, 21, 2412121 CrossRef CAS PubMed.
- M. Chang, Y. Zhao, D. Liu, J. Yang, J. Li and C. Zhong, Methane-trapping metal–organic frameworks with an aliphatic ligand for efficient CH4/N2 separation, Sustainable Energy Fuels, 2020, 4, 138–142 RSC.
| This journal is © The Royal Society of Chemistry 2026 |