Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Enantioselective synthesis of configurationally stable [5]helicenes containing 1,2-azaborine units
Catherine Olguina,
Christian Tabacarua,
Lennart Bessea,
Martin Simona,
Christopher Golza,
Marcos Humanesb,
Manuel A. Fernández-Rodríguez b,
Patricia García-García
b,
Patricia García-García *b,
Maike Mückec,
Ricardo A. Mata*c and
Manuel Alcarazo
*b,
Maike Mückec,
Ricardo A. Mata*c and
Manuel Alcarazo *a
*a
aInstitut für Organische und Biomolekulare Chemie, Georg-August-Universität Göttingen, Tammannstr 2, Göttingen 37077, Germany. E-mail: manuel.alcarazo@chemie.uni-goettingen.de
bUniversidad de Alcalá (IRYCIS). Departamento de Química Orgánica y Química Inorgánica, Instituto de Investigación Química “Andrés M. del Río” (IQAR), 28805 Alcalá de Henares, Madrid, Spain
cInstitut für Physikalische Chemie, Georg-August-Universität Göttingen, Tammannstr 6, Göttingen 37077, Germany
First published on 23rd April 2026
Abstract
Two different families of BN-doped [5]helicenes have been efficiently synthesized through a highly enantioselective, intramolecular, Au-catalyzed alkyne hydroarylation reaction. Key for the success of the method is the use of BINOL-derived cationic phosphonites as ancillary ligands (BINOL: 1,1-bi-2-naphthol). The inversion barriers of the structures obtained have been determined both experimentally and theoretically, and are essentially identical to those reported for non-dopped carbo[5]helicenes of otherwise identical structure. Contrarily, the newly prepared BN-doped helicenes exhibit intensified absorption spectra at long wavelength (λ ≈ 400 nm) and fluorescence when compared with their only-carbon counterparts. These effects are particularly pronounced for the naphtho[2,1-c]phenanthro[1,2-e][1,2]azaborinine series, in which the BN-unit is located at the rim of the helix. Preliminary studies on the post-synthetic functionalization of these structures are also described; specifically, the naphtho[2,1-c]phenanthro[1,2-e][1,2]azaborinine structure can be site-selective brominated at position 4. In addition, the unprecedented deborilation of these helices to afford axially chiral anilines has been observed by treatment with DDQ.
Introduction
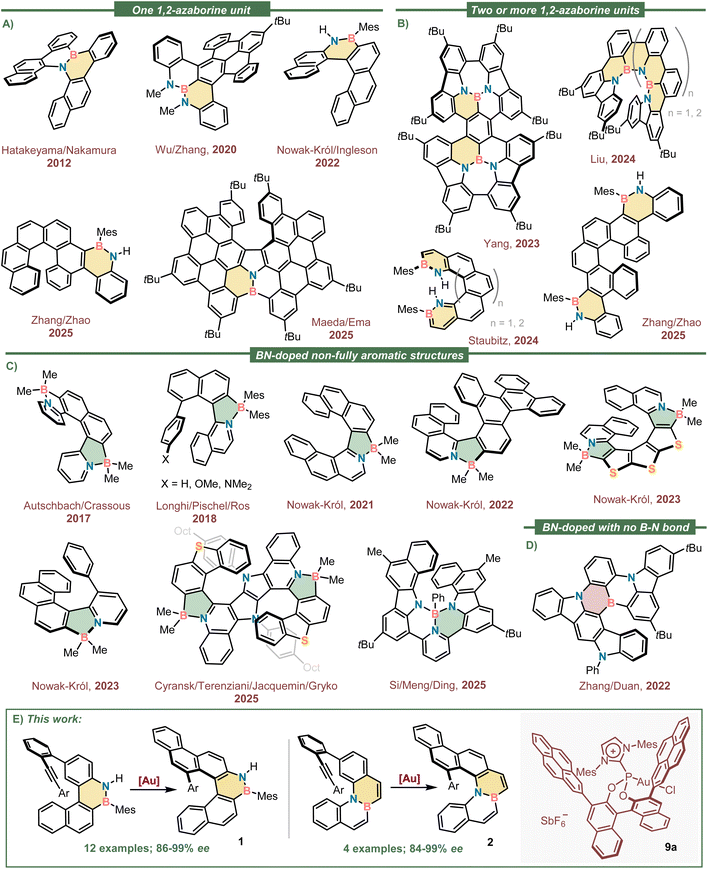
In polycyclic aromatic scaffolds, the formal replacement of two consecutive carbon atoms by an isoelectronic BN-unit barely modifies the geometric parameters of the original structure because the atomic radii of the three elements is quite similar, and the aromatized framework is retained.1–5 Yet, that exchange generates a perturbation on the electronic distribution that completely redefines the reactivity and photophysical properties of the resulting BN-doped materials.6–8 For example, it is well-documented that electrophilic aromatic substitution more readily occurs after formal BN-doping than in their carbon-only precursors;9,10 moreover, the substitution is highly regioselective and takes place at the position(s) adjacent to boron if these are unsubstituted and sterically accessible.11–13 Hydrogenation,14 photoisomerization reactions,15 and [4 + 2] cycloadditions with dienophiles of diverse nature are reactions that also get facilitated in 1,2-azaborinine derivatives due to the reduced aromaticity of this ring.16,17 An enhanced dipolar moment that affects solubility and molecular stacking is also expected by BN-doping; however, the most significant impact is often observed in the photophysical properties of the resulting materials.18 The asymmetry generated by the formal CC/BN-replacement deeply modifies the shapes and energetic distribution of the original frontier orbitals in a magnitude that depends upon the number of BN-units introduced,19,20 and their specific location within the polyaromatic scaffold.21 It is for this reason that BN-doping has emerged as an appealing strategy to optimize the luminescent properties of polycyclic aromatic hydrocarbon materials.22,23 In fact, BN-doped arenes often depict higher photoluminescence quantum yields (φPL) than their all-carbon counterparts, making them superior components in electronic devices such as organic light emitting diodes (OLEDs),24 organic field-effect transistors (OFETs)25 or solar cells.26 Their use for selective fluoride anion detection has been reported as well.27In addition, helical shape structures have been recognized as promising circularly polarized luminescence (CPL) dyes28,29 with potential application in diverse areas such as bioimaging,30 chiral switches,31–33 and data storage34 among others;35 however, unmodified carbohelicenes are typically characterized by low performance in terms of φPL and luminescence dissymmetry factors (glum).36,37 It is for that reason that the installment of BN-moieties in helicenes has not gone unnoticed as a tool to fine tune their chiroptical properties without altering their shape.38 Fig. 1A and B shows a selection of conformationally stable helicenes containing one,39–43 two,42,44,45 or more 1,2-azaborinine rings;46 while Fig. 1C depicts non-completely aromatic helical scaffolds in which the BN-doping unit is a 1,2-azaborole,47–53 and Fig. 1D shows a 1,4-diazaborinine containing helicene.
Looking at the structures shown in Fig. 1, it is surprising that with no exception they all were obtained through racemic syntheses that invariably require a final chromatographic separation of the enantiomeric constituents using a chiral stationary phase. In an attempt to address this synthetic limitation, we speculated that the enantioselective π-extension of 1,2-azaborine containing polyarenes through an intramolecular alkyne hydroarylation reaction might be the appropriate entry to enantioenriched BN-doped helicenes of different sizes and substitution patterns.54,55 In addition, our previous experience in the topic made us believe that Au(I)-catalysts bearing chiral α-cationic ancillary ligands56 are predestined to successfully achieve that transformation.57–61 Herein, we describe the materialization of that idea with the highly enantioselective syntheses of two different families of boraaza[5]helicenes sharing a common naphtho[2,1-c]chrysene scaffold (Fig. 1E). In compounds of general structure 1 the BN-unit is incorporated at the rim of the skeleton (7-,8-positions), while in 2 it is placed internally, in the bridging 8a, 14b-positions. The racemization barriers of both heterohelicenes are investigated through theoretical and experimental methods, and their photochemical characterization reported (UV-vis absorption, φPL and glum spectra). As expected, the introduction of the polar BN-moiety within the hydrocarbon framework drastically improved the φPL in both cases when compared with the parent naphtho[2,1-c]chrysene (φPL = 2%), albeit more significantly in 1-type structures (φPL(1b) = 20%).
Results and discussion
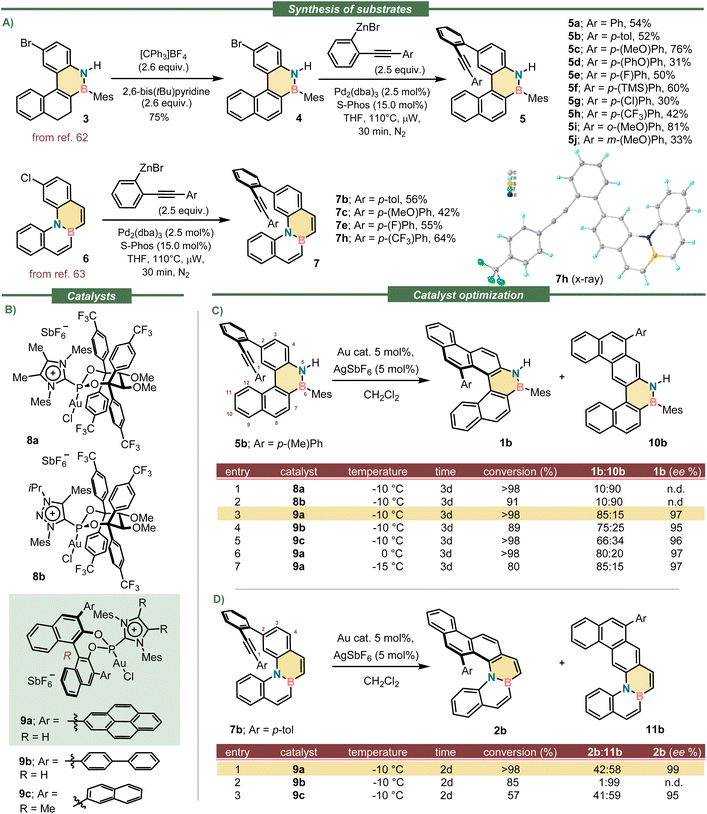
Synthesis of precursors, catalyst optimization and scope
We initiated our studies with the synthesis of appropriate substrates for the planned enantioselective hydroarylation. Thus, alkynes 5a–j were prepared from reported bromide 3 through an initial oxidation to obtain the fully aromatized benzo[e]naphtho[2,1-c][1,2]azaborine 4,62 followed by the installation of the necessary alkyne moiety via Negishi coupling. Substrates of general formula 7 were prepared following a similar route but starting from the already aromatic precursor 6 (Scheme 1A).63 The experimental details regarding the syntheses and the spectroscopic characterization of all new substrates are described in the SI.Compound 5b was used as model and initially submitted to the effect of the five Au-catalysts that, considering our previous experience, were the most promising for the key cyclization (Scheme 1B).57–61 Unfortunately, catalysts 8a and 8b, both sharing an acyclic version of TADDOL (tetraaryl-2,2-disubstituted-1,3-dioxolane-4,5-dimethanol), and decorated with p-(CF3)phenyl substituents, performed badly; they provided the undesired planar regioisomer 10b as the main product of the hydroarylation reaction (Scheme 1C, entries 1–2).
On the other hand, catalysts 9a–c derived from the BINOL (1,1′-bi-2-naphthol) platform demonstrated to be suitable for this reaction. They three delivered helicene 1b with excellent enantioselectivity (Scheme 1C, entries 3–5), and importantly, 9a was even able to override the natural tendency of the substrate to direct the intramolecular hydroarylation to 10b. Specifically, the use of 9a raised the regioselectivity to a remarkable 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 ratio for the azabora[5]helicene 1b while providing excellent optical purity as well (97% ee). Catalyst 9a also promoted the cyclisation of 7b into the corresponding azabora[5]helicene 2b with excellent optical purity (99% ee; Scheme 1D, entry 1); yet, the main product was the BN-doped dibenzo[a,m]tetraphene 11b. The use of catalysts 9b–c did not improve the regioselectivity of this hydroarylation step, which is highly substrate dependent.
15 ratio for the azabora[5]helicene 1b while providing excellent optical purity as well (97% ee). Catalyst 9a also promoted the cyclisation of 7b into the corresponding azabora[5]helicene 2b with excellent optical purity (99% ee; Scheme 1D, entry 1); yet, the main product was the BN-doped dibenzo[a,m]tetraphene 11b. The use of catalysts 9b–c did not improve the regioselectivity of this hydroarylation step, which is highly substrate dependent.
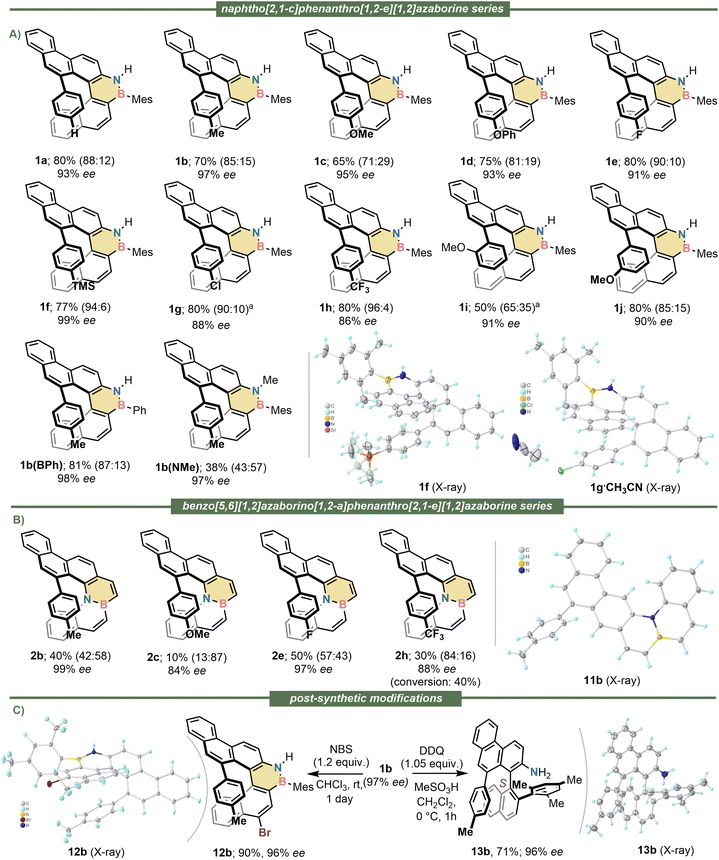
Having identified catalyst 9a as the most suitable, the scope of the cyclisation was evaluated using alkynes 5a–j, which contain substituents of diverse electron nature at their aromatic termini. We were pleased to observe that all reactions proceeded until complete conversion of the substrates; very high levels of enantioinduction were imparted in all cases as well (90–99% ee). The regioselectivity of the cyclisation ranged from mediocre (1i; 65![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35) to excellent (1f; 94
35) to excellent (1f; 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6), but despite that inconvenience, the helicene products were always the major component of the reaction mixtures and were gained in analytically pure form and good yields through preparative HPLC separation. It was also observed that the mesityl substituent at boron does not play a prominent role in this transformation; its formal exchange by a phenyl group neither alters the remarkable level of enantioselectivity, nor the regioselectivity in which 1b(BPh) is obtained. Nonetheless, the N-atom from substrates 5 must be kept unsubstituted; just methylation at that position inverts the regioselectivity of the cyclisation making planar S10b(NMe) the main product of the reaction. Even in that unfavourable case the azabora [5]helicene 1b(NMe) could be isolated in 38% yield with excellent optical purity (97% ee). The isolation and characterization of all side products of general formula 10 can be found in the SI. Crystals suitable for X-ray diffraction analysis of enantiopure 1b, 1d, 1e, 1f, 1g and 1h were obtained; from these measurements the absolute configuration of the six helicenes was assigned as P (Fig. 2A and S39–S44). By extension, we assumed the configuration of the whole 1a–j series to be the same.
6), but despite that inconvenience, the helicene products were always the major component of the reaction mixtures and were gained in analytically pure form and good yields through preparative HPLC separation. It was also observed that the mesityl substituent at boron does not play a prominent role in this transformation; its formal exchange by a phenyl group neither alters the remarkable level of enantioselectivity, nor the regioselectivity in which 1b(BPh) is obtained. Nonetheless, the N-atom from substrates 5 must be kept unsubstituted; just methylation at that position inverts the regioselectivity of the cyclisation making planar S10b(NMe) the main product of the reaction. Even in that unfavourable case the azabora [5]helicene 1b(NMe) could be isolated in 38% yield with excellent optical purity (97% ee). The isolation and characterization of all side products of general formula 10 can be found in the SI. Crystals suitable for X-ray diffraction analysis of enantiopure 1b, 1d, 1e, 1f, 1g and 1h were obtained; from these measurements the absolute configuration of the six helicenes was assigned as P (Fig. 2A and S39–S44). By extension, we assumed the configuration of the whole 1a–j series to be the same.
Subsequently, the extension of the synthetic protocol to the internally BN-doped helicenes 2 was attempted. By submitting alkynes 7 to the reaction conditions already optimized the expected 6-endo-dig cyclisation took place in all cases, but the regioselectivity of the process was strongly substrate dependent. Electron donating substituents at the hanging alkyne facilitate the transformation in terms of conversion, but they preferentially direct the cyclisation to the undesired 3-position of the benzo[e]benzo[5,6][1,2]azaborino[1,2-a][1,2]azaborine core. Thus, for substrates 7b and 7c the main products isolated are 11b and S11c, respectively. Helicenes 2b and 2c are still obtained from these reactions with high ee's but diminished yield. In contrast, electron withdrawing groups at the hanging alkyne preferentially guide the hydroarylation towards the formation of the helicenes 2e and 2h. Hence, azabora[5]helicenes 2b, 2c, 2e and 2h were isolated with high optical purity (84–99% ee) (See Fig. 2B). No X-ray quality crystals of these compounds were obtained, but comparison of the shape and sign of their electronic circular dichroism spectra with those of the 1 series allows us to confidently assign their absolute configuration also as P.
We also evaluated the possibility of functionalizing the helicenes just prepared. Thus, treatment of 1b with N-bromosuccinimide (NBS) cleanly afforded 12b, in which the electrophilic bromination occurred exclusively at the 4-position of the naphtho[2,1-c]phenanthro[1,2-e][1,2]azaborine skeleton (For the X-ray structure of 12b see Fig. 2C and S53). This exquisite regioselectivity is remarkable considering that apart of the BN-doping no directing groups were employed.
More unexpected was the clean DDQ-promoted oxidative deborylation of 1b with concomitant C–C coupling of the two aryl moieties originally at boron to deliver aniline 13b (For the X-ray structure of 13b see Fig. 2C and S55). The formation of 13b, which occurs with complete helical to axial chirality transfer, suggests that the oxidation-induced deborylation/C–C coupling manifold known to be operative in the ring contraction of chlorodibenzoborepines into phenanthrenes64 might be more general than anticipated, and also functional for structurally related dibenzo[c,e][1,2]azaborines.
Racemization dynamics
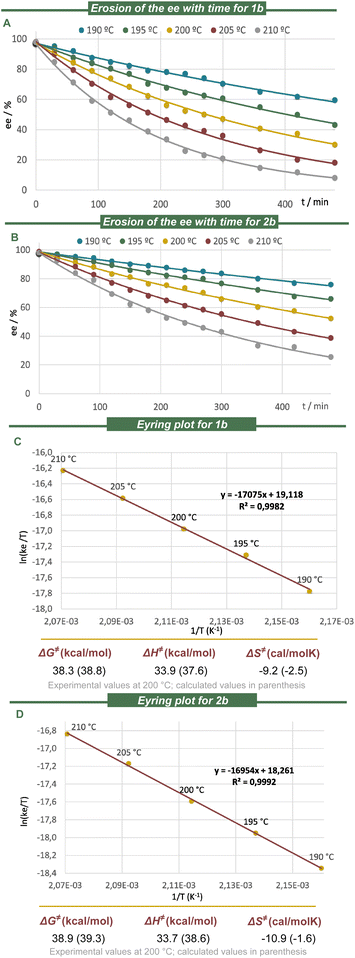
The racemization dynamics of the newly prepared azabora[5]helicenes were experimentally determined by heating enantiopure samples of model substrates 1b and 2b in 1,2,4-trichlorobenzene. The racemization rate constants were measured between 190 and 210 °C, and the activation-free energies (ΔG‡) for the inversion were calculated through an Eyring plot (Fig. 3). As expected from the minimal geometric distortion introduced by BN-doping, the racemization of both compounds, 1b (ΔG‡ = 38.3 kcal mol−1 at 200 °C) and 2b (ΔG‡ = 38.9 kcal mol−1 at 200 °C) is very similar to that of the parent carbo[5]helicene 14 (ΔG‡ = 37.6 kcal mol−1 at 200 °C).57 We also conclude that the location of the BN-unit, either in the rim or internally, has very minor effect on the enantiomerization barriers because the wedge angles (φ) of benzene and [1,2]-azaborine are basically identical, and B–N bond cleavage is not involved in the process.65 The calculated free energies of racemization at the B3LYP-D3/def2-TZVP level closely match the experimentally determined values (Fig. 3C and D);66–69 the geometric data of the corresponding transition states are collected in the SI. All structure optimisations were carried out with the Gaussian 16 (Rev. A.03) program package.70Photophysical and chiroptical characterization
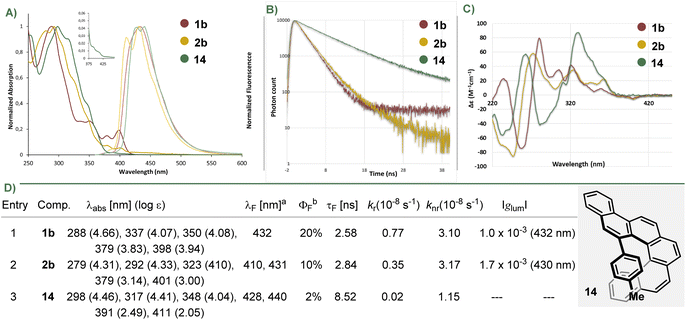
The UV-vis absorption and fluorescence spectra, as well as fluorescence life times of 1b, 2b were recorded in CH2Cl2 and compared with those of their parent hydrocarbon 14; they are shown in Fig. 4 (also see Fig. S1–S5). The three model compounds show comparable absorption maxima (λabs = 288 nm, 1b; 279 nm, 2b; 298 nm, 14) with similar molar extinction coefficients (log ε = 4.66, 1b; 4.31, 2b; 4.46, 14), but helicenes 1b and 2b are characterized by a significant intensification of the least energetic band (1b, λabs = 398 nm, log ε = 3.94; 2b, λabs = 401 nm, log ε = 3.00), an effect related with BN-doping.In the fluorescence spectrum of 1b, only one band centered at λF = 432 nm is observed, while in that of 2b, at least two vibronic bands (λF = 410, 431 nm) are recognized. In terms of fluorescence quantum yields, the BN-doped helicenes were substantially brighter (φPL = 0.20, 1b; 0.10, 2b) than the barely emissive parent carbohelicene (φPL = 0.02, 14). The fluorescence lifetime are very similar for both BN-doped structures (τF = 2.6 ns, 1b; 2.8 ns, 2b) and shorter than that of 14 (τF = 8.5 ns). The small Stokes shifts, and the observable vibrational structures of the emission spectra are indicative of high rigid structures.
The electronic circular dichroism (ECD) spectra of 1b, 2b and 14 were recorded (Fig. 4c; for the complete study see Fig. S10–30). Enantiomer P-1b exhibits an intense negative CD band at 258 nm (Δε = −74.6 M−1 cm−1) and positive cotton effects in the region from 271 to ca. 377 nm (Δε = 78.3 M−1 cm−1 at 281 nm; Δε = 35.1 M−1 cm−1 at 306 nm, and Δε = 41.1 M−1 cm−1 at 322 nm). Similarly, P-2b depicts an intense negative CD band in the high energy region centred at 247 nm (Δε = −85.6 M−1 cm−1) and positive cotton effects in the region from 259 to ca. 380 nm (Δε = 58.0 M−1 cm−1 at 274 nm; Δε = 33.7 M−1 cm−1 at 326 nm, and Δε = 22.4 M−1 cm−1 at 364 nm). CPL spectra for 1b and 2b were recorded (Fig. 4D and S31–34). Both compounds depict comparable luminescence dissymmetry factors (|glum| = 1–2 × 10−3), which are in the same range as these of [5]helicenes.
To obtain deeper understanding of the impact of BN-doping in these helicenes, we compared the measured UV/vis absorption spectra of the model compounds with simulated results. In line with a previous study carried out by some of us,71 we employed simplified time-dependent density functional theory (sTD-DFT) as a fast method to calculate a large portion of the spectra.72 The basis set employed was def2-TZVP, with all sTD-DFT calculations carried out with the Orca program package (version 6.0.1).73 A uniform shift of −0.4 eV was applied to the transition energies in order to better align the theoretical and experimental spectra. In our calculations, we found necessary to make use of a long-range corrected functional, as some of the bands exhibited a significant charge-transfer character. The study was focused on the first absorption bands of 1b, 2b and 14 around 400 nm (Fig. 5).
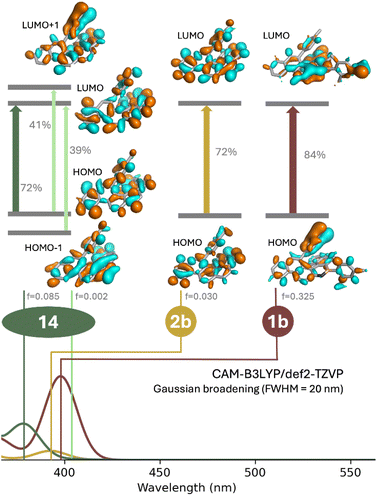 | ||
| Fig. 5 Plots of the frontier molecular orbitals with the transition oscillator strength f for the major compositions of S0 → S1 and the TD-DFT simulated spectra. | ||
The TD-DFT calculations for 1b and 2b revealed the lowest energy transitions to be dominated in both cases by the HOMO–LUMO transition, with weights of 84% and 72% respectively. In the case of 14, CAM-B3LYP/def2-TZVP registers this transition as the second absorption band, with the first transition exhibiting a very low oscillator strength. In analogy to previous reports with carbo[5]helicenes, the S0 → S1 transition can be primarily described as a combination of two major contributions: HOMO → LUMO +1 and HOMO −1 → LUMO with a low calculated oscillator strength (0.002) if compared to those of 1b (0.325) and 2b (0.030). The isodensity surfaces for the frontier orbitals of all three compounds is provided in Fig. 5. It is noticeable that 2b and 14 show a similar picture, with the transition largely delocalised across the aromatic rings. The main difference is found in 1b, which depicts the most intense band. This comes about through a charge transfer from the non-fused aromatic ring to the BN-containing helicene scaffold. In the case of 2b such an effect is not observed because a charge accumulation in a structure containing the BN bond at a junction between two rings is more penalising to the aromaticity of the helical moiety.
Conclusions
In summary, we have prepared two families of BN-doped [5]helicenes through a synthetic route that uses a highly enantioselective Au-catalysed hydroarylation as key step. Comparison of the racemization dynamics and photophysical properties of these structures with those of their all-carbon analogue allows an accurate evaluation of the impact of BN-doping. The influence on the inversion barriers is negligible, and also seem to be rather small on λabs at long wavelength (Δλ ≈ 10–15 nm); however, both the intensities of the absorptions and the φF values were significantly enhanced for BN-doped helicenes; particularly, in the case of 1b. TD-DFT calculations attribute this observation to a charge transfer from a non-fused aromatic ring to the BN-containing helicene scaffold, which is specially favoured for 1b.Author contributions
C. O., C. T. and L. B. performed the syntheses, routine analytics, racemization kinetics, and chiroptical properties measurements. M. S. performed the HPLC analyses. C. G. performed the X-ray diffraction analyses. M. H, M. A. F. R and P. G. G. developed the synthetic route to compound 6. M. M. and R. M. carried out the DFT calculations. M. A. conceived the project and wrote the manuscript. All authors reviewed the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2506342–2506358 contain the supplementary crystallographic data for this paper.74a–qAll data regarding this article can be found in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc02344d.
Acknowledgements
Funding: Financial support from the Deutsche Forschungsgemeinschaft (INST 186/1237-1, INST 186/1318-1, INST 186/1324-1, INST 186/1622-1 and 389479699/GRK2455) and the Ministerio de Ciencia, Innovación y Universidades (grant PID2023-146343NB-I00 financed by MCIU/AEI/10.13039/501100011033/FEDER, UE) is gratefully acknowledged. We thank the NMR and MS services at the Faculty of Chemistry (University of Göttingen, DE) for technical assistance and Profs. J. M. Cuerva and D. Miguel (University of Granada, ES) for fruitful discussions. C.O. thanks the DAAD for support (Ph.D. fellowship) and M. H. thanks Comunidad de Madrid for a predoctoral contract.Notes and references
- C. A. Jaska, D. J. H. Emslie, M. J. D. Bosdet, W. E. Piers, T. S. Sorensen and M. Parvez, J. Am. Chem. Soc., 2006, 128, 10885–10896 CrossRef CAS PubMed.
- M. J. D. Bosdet, C. A. Jaska, W. E. Piers, T. S. Sorensen and M. Parvez, Org. Lett., 2007, 9, 1395–1398 CrossRef CAS PubMed.
- J. S. A. Ishibashi, J. L. Marshall, A. Mazière, G. J. Lovinger, B. Li, L. N. Zakharov, A. Dargelos, A. Graciaa, A. Chrostowska and S.-Y. Liu, J. Am. Chem. Soc., 2014, 136, 15414–15421 CrossRef CAS PubMed.
- X. Liu, P. Wu, J. Li and C. Cui, J. Org. Chem., 2015, 80, 3737–3744 CrossRef CAS PubMed.
- C. Zhang, L. Zhang, C. Sun, W. Sun and X. Liu, Org. Lett., 2019, 21, 3476–3480 CrossRef CAS PubMed.
- Q. Feng, Y. Zhou, H. Xu, J. Liu, Z. Wan, Y. Wang, P. Yang, S. Ye, Y. Zhang, X. Cao, D. Cao and H. Huang, Chem. Soc. Rev., 2025, 54, 5995–6061 RSC.
- L. Jia, Q. Wu, T. Yang, B. Xie, J. Sheng, W. Xie and J. Shi, Molecules, 2025, 30, 4252 CrossRef CAS PubMed.
- C. R. McConnell and S.-Y. Liu, Chem. Soc. Rev., 2019, 48, 3436–3453 RSC.
- Y. Zhang, W. Dan and X. Feng, Organometallics, 2017, 36, 1677–1680 CrossRef CAS.
- Y. Zhang, F. Sun, W. Dan and X. Fang, J. Org. Chem., 2017, 82, 12877–12887 CrossRef CAS PubMed.
- J. Pan, J. W. Kampf and A. J. Ashe, Org. Lett., 2007, 9, 679–681 CrossRef CAS PubMed.
- G. A. Molander and S. R. Wisniewski, J. Org. Chem., 2014, 79, 6663–6678 CrossRef CAS PubMed.
- A. N. Brown, B. Li and S.-Y. Liu, J. Am. Chem. Soc., 2015, 137, 8932–8935 CrossRef CAS PubMed.
- J. Liu, Y. Dai, D. Robinson, B. Li, K. Miqueu and S.-Y. Liu, Angew. Chem., Int. Ed., 2025, 64, e202504419 CrossRef CAS PubMed.
- T. Ozaki, S. K. Bentley, N. Rybansky, B. Li and S.-Y. Liu, J. Am. Chem. Soc., 2024, 146, 24748–24753 CrossRef CAS PubMed.
- R. J. Burford, B. Li, M. Vasiliu, D. A. Dixon and S.-Y. Liu, Angew. Chem., Int. Ed., 2015, 54, 7823–7827 CrossRef CAS PubMed.
- Y. García-Rodeja and I. Fernández, J. Org. Chem., 2016, 81, 6554–6562 CrossRef PubMed.
- Z. Liu and T. B. Marder, Angew. Chem., Int. Ed., 2008, 47, 242–244 CrossRef CAS PubMed.
- X. Chen, D. Tan and D.-T. Yang, J. Mater. Chem. C, 2022, 10, 13499–13532 RSC.
- X.-Y. Wang, J.-Y. Wang and J. Pei, Chem.–Eur. J., 2015, 21, 3528–3539 CrossRef CAS PubMed.
- T. Kaehler, M. Bolte, H.-W. Lerner and M. Wagner, Angew. Chem., Int. Ed., 2019, 58, 11379–11384 CrossRef CAS PubMed.
- M. R. Rapp, W. Leis, F. Zinna, L. Di Bari, T. Arnold, B. Speiser, M. Seitz and H. F. Bettinger, Chem.–Eur. J., 2022, 28, e202104161 CrossRef CAS PubMed.
- J. Ruhl, N. Oberhof, A. Dreuw and H. A. Wegner, Angew. Chem., Int. Ed., 2023, 62, e202300785 CrossRef CAS PubMed.
- X. Wang, F. Zhang, J. Liu, R. Tang, Y. Fu, D. Wu, Q. Xu, X. Zhuang, G. He and X. Feng, Org. Lett., 2013, 15, 5714–5717 CrossRef CAS PubMed.
- X.-Y. Wang, H.-R. Lin, T. Lei, D.-C. Yang, F.-D. Zhuang, J.-Y. Wang, S.-C. Yuan and J. Pei, Angew. Chem., Int. Ed., 2013, 52, 3117–3120 CrossRef CAS PubMed.
- X. Liu, S. Pang, L. Zeng, W. Deng, M. Yang, X. Yuan, J. Li, C. Duan, F. Huang and Y. Cao, Chem. Commun., 2022, 58, 8686–8689 RSC.
- J. Zhang, F. Liu, Z. Sun, C. Li, Q. Zhang, C. Zhang, Z. Liu and X. Liu, Chem. Commun., 2018, 54, 8178–8181 RSC.
- L. Arrico, L. Di Bari and F. Zinna, Chem.–Eur. J., 2021, 27, 2920–2934 CrossRef CAS PubMed.
- W.-L. Zhao, M. Li, H.-Y. Lu and C.-F. Chen, Chem. Commun., 2019, 55, 13793–13803 RSC.
- P. Stachelek, L. MacKenzie, D. Parker and R. Pal, Nat. Commun., 2022, 13, 553 CrossRef CAS PubMed.
- N. Saleh, B. Moore II, M. Srebro, N. Vanthuyne, L. Toupet, J. A. G. Williams, C. Roussel, K. K. Deol, G. Muller, J. Autschbach and J. Crassous, Chem.–Eur. J., 2015, 21, 1673–1681 CrossRef CAS PubMed.
- H. Isla, M. Srebro-Hooper, M. Jean, N. Vanthuyne, T. Roisnel, J. L. Lunkley, G. Muller, J. A. G. Williams, J. Autschbach and J. Crassous, Chem. Commun., 2016, 52, 5932–5935 RSC.
- E. Yen-Pon, F. Buttard, L. Frédéric, P. Thuéry, F. Taran, G. Pieters, P. A. Champagne and D. Audisio, JACS Au, 2021, 1, 807–818 CrossRef CAS PubMed.
- N. P. M. Huck, W. F. Jager, B. de Lange and B. L. Feringa, Science, 1996, 273, 1686–1688 CrossRef CAS.
- Y. Zhang, S. Yu, B. Han, Y. Zhou, X. Zhang, X. Gao and Z. Tang, Matter, 2022, 5, 837–875 CrossRef CAS.
- M. Sapir and E. Vander Donckt, Chem. Phys. Lett., 1975, 36, 108–110 CrossRef CAS.
- N. I. Nijegorodov and W. S. Downey, J. Phys. Chem., 1994, 98, 5639–5643 CrossRef CAS.
- A. Nowak-Król, P. T. Geppert and K. R. Naveen, Chem. Sci., 2024, 15, 7408–7440 RSC.
- T. Hatakeyama, S. Hashimoto, T. Oba and M. Nakamura, J. Am. Chem. Soc., 2012, 134, 19600–19603 CrossRef CAS PubMed.
- Z. Sun, C. Yi, Q. Liang, C. Bingi, W. Zhu, P. Qiang, D. Wu and F. Zhang, Org. Lett., 2020, 22, 209–213 CrossRef CAS PubMed.
- K. Yuan, D. Volland, S. Kirschner, M. Uzelac, G. S. Nichol, A. Nowak-Król and M. J. Ingleson, Chem. Sci., 2022, 13, 1136–1145 RSC.
- M. Wang, M.-Y. Zhang and C.-H. Zhao, Org. Lett., 2025, 27, 1823–1828 CrossRef CAS PubMed.
- C. Maeda, S. Michishita, I. Yasutomo and T. Ema, Angew. Chem., Int. Ed., 2025, 64, e202418546 CrossRef CAS PubMed.
- D. Tan, J. Dong, T. Ma, Q. Feng, S. Wang and D.-T. Yang, Angew. Chem., Int. Ed., 2023, 62, e202304711 CrossRef CAS PubMed.
- Y. Appiarius, S. Míguez-Lago, P. Puylaert, N. Wolf, S. Kumar, M. Molkenthin, D. Miguel, T. Neudecker, M. Juríček, A. G. Campaña and A. Staubitz, Chem. Sci., 2024, 15, 466–476 RSC.
- Y. Yu, C. Wang, F.-F. Hung, C. Chen, D. Pan, C.-M. Che and J. Liu, J. Am. Chem. Soc., 2024, 146, 22600–22611 CrossRef CAS PubMed.
- C. Shen, M. Srebro-Hooper, M. Jean, N. Vanthuyne, L. Toupet, J. A. G. Williams, A. R. Torres, A. J. Riives, G. Muller, J. Autschbach and J. Crassous, Chem.–Eur. J., 2017, 23, 407–418 CrossRef CAS PubMed.
- Z. Domínguez, R. López-Rodríguez, E. Álvarez, S. Abbate, G. Longhi, U. Pischel and A. Ros, Chem.–Eur. J., 2018, 24, 12660–12668 CrossRef PubMed.
- J. Full, S. P. Panchal, J. Götz, A.-M. Krause and A. Nowak-Król, Angew. Chem., Int. Ed., 2021, 60, 4350–4357 CrossRef CAS PubMed.
- D. Volland, J. Niedens, P. T. Geppert, M. J. Wildervanck, F. Full and A. Nowak-Król, Angew. Chem., Int. Ed., 2023, 62, e202304291 CrossRef CAS PubMed.
- J. Full, M. J. Wildervanck, C. Dillmann, S. P. Panchal, D. Volland, F. Full, K. Meerholz and A. Nowak-Król, Chem.–Eur. J., 2023, 29, e202302808 CrossRef CAS PubMed.
- W. D. Petrykowski, N. Vanthuyne, C. Naim, F. Bertocchi, Y. M. Poronik, A. Ciesielski, M. K. Cyrański, F. Terenziani, D. Jacquemin and D. T. Gryko, Chem. Sci., 2025, 16, 8338–8345 RSC.
- Y. He, H. Yang, Y. Yue, X. Gan, S. Xiao, X. Chen, S. Zhu, D. Wan, R. He, H. Si, G. Meng, P. Chen and J. Ding, J. Mater. Chem. C, 2025, 13, 18092–18100 RSC.
- Y. Wang, Z.-G. Wu and F. Shi, Chem Catal., 2022, 2, 3077–3111 CAS.
- W. Liu, T. Qin, W. Xie and X. Yang, Chem.–Eur. J., 2022, 28, e20220236 Search PubMed.
- L. D. M. Nicholls and M. Alcarazo, Chem. Lett., 2019, 48, 1–13 CrossRef CAS.
- W. Fu, V. Pelliccioli, R. Casares-López, J. M. Cuerva, M. Simon, C. Golz and M. Alcarazo, CCS Chem., 2024, 6, 2439–2451 CrossRef CAS.
- W. Fu, V. Pelliccioli, M. von Geyso, P. Redero, C. Böhmer, M. Simon, C. Golz and M. Alcarazo, Adv. Mater., 2023, 2211279 CrossRef CAS PubMed.
- V. Pelliccioli, T. Hartung, M. Simon, C. Golz, E. Licandro, S. Cauteruccio and M. Alcarazo, Angew. Chem., Int. Ed., 2022, 61, e202114577 CrossRef CAS PubMed.
- P. Redero, T. Hartung, J. Zhang, L. D. M. Nicholls, G. Zichen, M. Simon, C. Golz and M. Alcarazo, Angew. Chem., Int. Ed., 2020, 59, 23527–23531 CrossRef CAS PubMed.
- T. Hartung, R. Machleid, M. Simon, C. Golz and M. Alcarazo, Angew. Chem., Int. Ed., 2020, 59, 5660–5664 CrossRef CAS PubMed.
- E. Sans-Panadés, J. J. Vaquero, M. A. Fernández-Rodríguez and P. García-García, Org. Lett., 2022, 24, 5860–5865 CrossRef PubMed.
- A. Abengózar, P. García-García, D. Sucunza, A. Pérez-Redondo and J. J. Vaquero, Chem. Commun., 2018, 54, 2467–2470 RSC.
- Y. Shoji, N. Tanaka, S. Muranaka, N. Shigeno, H. Sugiyama, K. Takenouchi, F. Hajjaj and T. Fukushima, Nat. Commun., 2016, 7, 12704 CrossRef PubMed.
- T. Hensel, N. N. Andersen, M. Plesner and M. Pittelkow, Synlett, 2016, 27, 498–525 CAS.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648–5652 CrossRef CAS.
- S. Grimme, J. Antony, S. Ehrlich and H. Krieg, J. Chem. Phys., 2010, 132, 154104 CrossRef PubMed.
- S. Grimme, S. Ehrlich and L. Goerigk, J. Comb. Chem., 2011, 32, 1456–1465 CAS.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman, D. J. Fox, Gaussian 16, Revision A.03, Gaussian, Inc, Wallingford CT, 2016 Search PubMed.
- S. Suárez-Pantiga, P. Redero, X. Aniban, M. Simon, C. Golz, R. A. Mata and M. Alcarazo, Chem.–Eur. J., 2021, 27, 13358–13366 CrossRef PubMed.
- C. Bannwarth and S. Grimme, Comput. Theor. Chem., 2014, 1040–1041, 45–53 CrossRef CAS.
- F. Neese, WIREs Comput. Mol. Sci., 2025, 15, e70019 CrossRef.
- (a) CCDC 2506342: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q41rp; (b) CCDC 2506343: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q41sq; (c) CCDC 2506344: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q41tr; (d) CCDC 2506345: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q41vs; (e) CCDC 2506346: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q41wt; (f) CCDC 2506347: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q41xv; (g) CCDC 2506348: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q41yw; (h) CCDC 2506349: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q41zx; (i) CCDC 2506350: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q420z; (j) CCDC 2506351: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4210; (k) CCDC 2506352: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4221; (l) CCDC 2506353: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4232; (m) CCDC 2506354: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4243; (n) CCDC 2506355: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4254; (o) CCDC 2506356: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4265; (p) CCDC 2506357: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4276; (q) CCDC 2506358: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q4287.
| This journal is © The Royal Society of Chemistry 2026 |