Open Access Article
Open Access ArticlePromoting effect of nickel-chromium hydroxide on nickel phosphide nanosheets for efficient hydrogen production coupled with sulfion degradation
Xiaojun Wanga,
Jieting Lia,
Xinyu Zhua,
Zehua Xua,
Shuixiang Xiea,
Shutong Chena,
Yuxuan Wana,
Guangyu Zhang a,
Mingzheng Ge
a,
Mingzheng Ge a,
Wei Zhanga,
Chao Wang
a,
Wei Zhanga,
Chao Wang *b and
Rui-Qing Li
*b and
Rui-Qing Li *a
*a
aSchool of Textile and Clothing, Nantong University, Nantong 226019, PR China. E-mail: liruiqing@ntu.edu.cn
bSchool of Chemistry and Chemical Engineering, Yangzhou University, Yangzhou 225002, China. E-mail: wangchao@yzu.edu.cn
First published on 29th April 2026
Abstract
Electrochemical hydrogen production represents a sustainable approach for hydrogen generation. However, its large-scale practical application is largely limited by the sluggish oxygen evolution reaction (OER). In this study, the thermodynamically favorable sulfion oxidation reaction (SOR) is employed to replace the slow OER and nickel phosphide/nickel-chromium hydroxide (Ni2P/NiCr(OH)2) nanosheets are constructed to achieve low-energy hydrogen production. Benefiting from the interfacial water structure reorganization and electronic structure optimization caused by NiCr(OH)2 introduction, and uniform nanosheet morphology, Ni2P/NiCr(OH)2 can achieve a current density of 10 mA cm−2 at low overpotentials for both the hydrogen evolution reaction and SOR. The assembled hybrid water electrolyzer requires small voltages of 0.513 and 0.698 V to reach 10 and 100 mA cm−2, respectively, significantly lower than those of conventional water electrolysis. Moreover, Ni2P/NiCr(OH)2 exhibits excellent stability for 800 h at 200 mA cm−2 and realizes the conversion of sulfion into valuable elemental sulfur. This work presents a promising strategy for achieving energy-efficient hydrogen generation and resource utilization of sulfide-containing pollutants.
Introduction
In recent years, the continuous consumption of traditional fossil fuels has caused a series of serious environmental problems.1 These problems include greenhouse gas emissions and air pollution. They have pushed researchers to speed up the research, development, and industrial application of renewable clean energy and related technologies.2,3 Hydrogen (H2) has high energy density, yields pollution-free combustion products, and has abundant raw material sources.4,5 It is recognized as an ideal sustainable energy carrier to replace fossil fuels.6 Hydrogen plays a pivotal role in alleviating energy demand pressure and constructing a clean energy ecosystem.7,8 Currently, H2 production pathways include fossil fuel-based H2 production, industrial by-product H2 recovery and water electrolysis.9,10 Among them, water electrolysis technology with water as the raw material exhibits unique advantages and can produce high-purity green hydrogen with near-zero carbon emissions. It is a core hydrogen production technology integrating sustainability and environmental friendliness, and promoting the transformation of the energy structure.11–13Water electrolysis typically involves the hydrogen evolution reaction (HER) at the cathode to directly produce hydrogen, and the oxygen evolution reaction (OER) at the anode with a standard potential as high as 1.23 V vs. the reversible hydrogen electrode (RHE).14–16 Therefore, the OER with a high theoretical potential and sluggish kinetics results in substantial energy consumption is a major bottleneck of the overall water splitting (OWS) process. Specifically, actual anodic operating potentials are often significantly higher than 1.23 V due to limited mass transfer resistance, and ohmic polarization in practical electrolysis systems, further exacerbating the energy burden.17–19 To overcome these limitations, replacing the OER with the oxidation of thermodynamically favorable small molecules has emerged as an attractive strategy.20–23 This strategy not only effectively reduces the potentials of anodic reactions, but also enables the co-production of value-added chemicals and hydrogen.24–26 Thereinto, the sulfide oxidation reaction (SOR, S2− → S + 2e−) exhibits a lower standard potential of −0.48 V vs. RHE compared with the OER. Coupling the SOR with the HER in a hybrid water electrolyzer (HWE) can significantly reduce energy consumption of H2 production. Furthermore, this strategy can convert sulfur-containing wastewater into valuable elemental sulfur under mild conditions, realizing resource utilization of pollutants, which provides a promising pathway for developing efficient and low-energy-consumption H2 production.27–29
Although metal-based electrocatalysts exhibit high catalytic activity for the SOR, the active metal sites are susceptible to corrosion and poisoning by S2− when directly exposed to sulfur-containing electrolytes. This leads to the degradation of catalytic activity and stability. Therefore, developing catalysts with high activity and durability is imminent.30–32 Transition metal phosphides (TMPs) have emerged as a research hotspot due to their advantages of good electrical conductivity, chemical corrosion resistance and compositional tunability. Over the past few decades, researchers have developed various synthetic methods including the hydrothermal method, phosphorization and electrodeposition method, to adjust morphology, electronic structures and reaction energy barriers of TMPs, which display good performances for the SOR and HER. For example, Shi et al. prepared the Mo–Pt–S composite catalyst via electroreduction and pulse electrodeposition methods, which exhibited excellent hydrogen evolution reaction performance with an overpotential of only 30.12 mV at a current density of 10 mA cm−2.33 Yu et al. fabricated the FeCoNiCrMn high-entropy alloy (HEA) via a two-step process of hydrothermal sulfurization followed by phosphorization. Endowed with abundant lattice defects and lattice strains, the as-prepared HEA exhibits remarkably superior electrocatalytic performance toward the SOR.34 Liang et al. successfully synthesized a multiphase NiCoFe-based layered double hydroxide (NiCoFe-LDH) through a facile seed-assisted heterogeneous nucleation strategy. Featuring a unique microscale hydrangea-like morphology, the NiCoFe-LDH boasts an ultra-large specific surface area and synergistic active phases, which collectively provide abundant catalytic interfaces and reactive sites for the SOR.35 Li et al. synthesized a Ni3S2/CoNiP material, which shows excellent catalytic performances for the HER, SOR, and nitrate reduction reaction (NO3RR).36 Even though some progress has been made, most exploited catalysts show monofunctional catalytic performances, which can lead to the deterioration and incompatibility of catalysts when pairing them in an electrolyzer. Therefore, developing bifunctional catalysts for the HER and SOR to realize efficient hydrogen production and sulfion upgradation is of utmost urgency.
In this work, Ni2P/NiCr(OH)2 nanosheets grown on nickel foam (NF) were successfully prepared via hydrothermal and low-temperature calcination methods. The experimental results indicate that the NiCr(OH)2 introduction can reorganize the interfacial water structure and optimize the electronic structure of Ni2P, which lead to improved catalytic performances of Ni2P/NiCr(OH)2 for both the HER and SOR. The integrated Ni2P/NiCr(OH)2-based HWE requires a small cell voltage of 0.513 V at 10 mA cm−2 and long-term stability for 800 h, which can efficiently upgrade sulfion into valuable elemental sulfur, achieving efficient H2 production and resource utilization of sulfur pollution at low energy consumption.
Results and discussion
Synthesis and characterization
The synthesis route of Ni2P/NiCr(OH)2 is illustrated in Fig. 1a. A two-step hydrothermal reaction is employed to achieve uniform growth of the Ni(OH)2/NiCr(OH)2 precursor on NF, forming a nanosheet-like structure. The surface morphology of Ni(OH)2 is characterized using scanning electron microscopy (SEM). As displayed in Fig. 1b and S1a, the Ni(OH)2 product shows a smooth nanosheet structure. In contrast, Ni(OH)2/NiCr(OH)2 exhibits a hierarchical nanosheet structure after the second hydrothermal reaction (Fig. 1c and S1d), where small nanosheets grow on the original Ni(OH)2 nanosheets, implying the increased specific surface area.37 Subsequently, the target Ni2P/NiCr(OH)2 precursor is obtained through the phosphorization process. SEM images (Fig. 1d and S1c) show that Ni2P/NiCr(OH)2 preserves a similar hierarchical nanosheet structure to the precursor without obvious structural alteration. The X-ray diffraction (XRD) patterns of Ni2P and Ni2P/NiCr(OH)2 show that characteristic diffraction peaks can be well indexed to Ni2P (PDF#74-1385) and Ni(OH)2 (PDF#14-0117), confirming the formation of Ni2P and Ni2P/NiCr(OH)2 (Fig. 1e). The transmission electron microscopy (TEM) image in Fig. 1f further verifies the layered nanosheet structure. In the high-resolution TEM (HRTEM) image (Fig. 1g), distinct lattice fringes with lattice spacings of 0.174 and 0.198 nm correspond to the (102) plane of Ni(OH)2 and the (201) plane of Ni2P, respectively. In Fig. S2, the mass ratio of Ni![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) P
P![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cr
Cr![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) O in Ni2P/NiCr(OH)2 is calculated to be 38.8
O in Ni2P/NiCr(OH)2 is calculated to be 38.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 27.0
27.0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.8
3.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 26.3, which is close to the Ni
26.3, which is close to the Ni![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cr feeding ratio. Elemental mapping images of Ni2P/NiCr(OH)2 (Fig. 1h) confirm the coexistence and homogeneous distribution of Ni, P, Cr and O elements.
Cr feeding ratio. Elemental mapping images of Ni2P/NiCr(OH)2 (Fig. 1h) confirm the coexistence and homogeneous distribution of Ni, P, Cr and O elements.
X-ray photoelectron spectroscopy (XPS) was employed to further investigate the surface chemical composition and elemental valence states of Ni2P and Ni2P/NiCr(OH)2 catalysts. The survey XPS spectrum of Ni2P/NiCr (OH)2 (Fig. 2a) confirms the coexistence of Ni, P, Cr, and O elements, which is in good agreement with the elemental mapping results. While pristine Ni2P shows characteristic peaks of Ni 2p, P 2p and O 1s, the emerged Cr 2p signal in the Ni2P/NiCr(OH)2 confirms successful incorporation of Cr. In Fig. 2b, the Ni 2p spectrum of Ni2P displays two pairs of spin–orbit peaks at 852.4, 856.4 eV, and 872.7, 874.8 eV, belonging to Ni 2p3/2 and Ni 2p1/2, respectively. The additional peaks located at 861.6 and 880.2 eV correspond to satellite peaks.38–40 After coupling with NiCr(OH)2, the Ni 2p spectrum of Ni2P/NiCr(OH)2 reveals a negative shift of 0.7 eV compared with Ni2P, indicating that the electronic structure optimization of Ni2P and the presence of electronic interaction between Ni2P and NiCr(OH)2, which are beneficial to improve catalytic performances.41,42 The P 2p XPS spectrum (Fig. 2c) demonstrates that the characteristic peak at 129.5 eV is associated with the phosphorus–metal (P–M) bonding configuration, confirming the successful fabrication of Ni2P. The broad characteristic peak at 133.8 eV indicates the presence of P–O species on the sample surface, which originates from surface oxidation.43,44 In the high-resolution Cr 2p XPS spectrum (Fig. 2d), two characteristic peaks are observed at binding energies of 587.3 and 577.5 eV, which correspond to the Cr 2p1/2 and Cr 2p3/2 states, respectively. The characteristic peak of Cr 2p3/2 at 577.5 eV corresponds to Cr3+, which can modulate the electronic structure and surface active sites, playing a pivotal role in enhancing catalytic performance.45–47 The O 1s spectrum in Fig. S3 reveals the presence of lattice oxygen at 530.9 eV, deficient oxygen at 531.8 eV, and adsorbed oxygen species at 533.3 eV.48,49
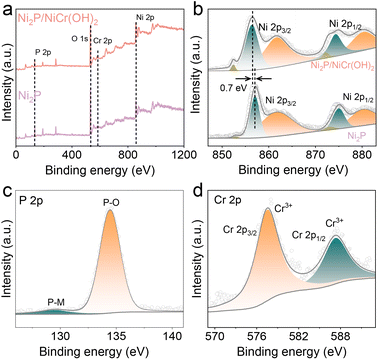 | ||
| Fig. 2 (a) XPS spectra and (b) Ni 2p XPS spectra of Ni2P and Ni2P/NiCr(OH)2. (c) P 2p XPS spectrum and (d) Cr 2p XPS spectrum of Ni2P/NiCr(OH)2. | ||
Electrochemical performances
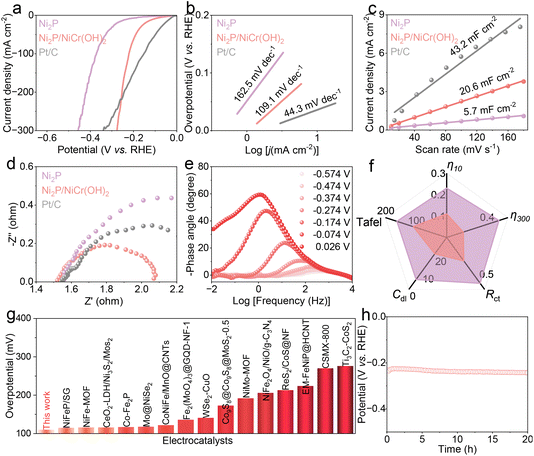
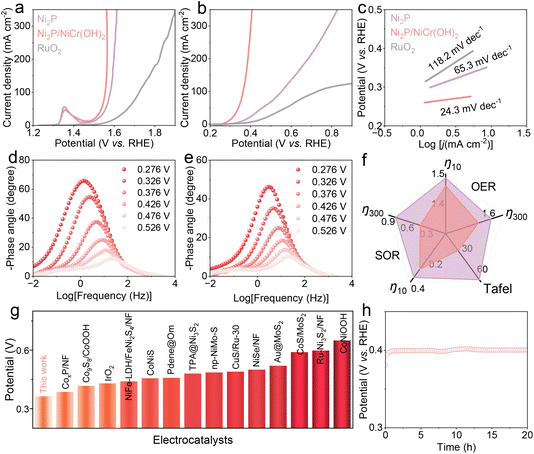
The electrocatalytic performances of Ni2P and Ni2P/NiCr(OH)2 catalysts toward the HER were evaluated in a three-electrode system. As shown in Fig. 3a and f, the catalytic activities are compared based on linear sweep voltammetry (LSV). The Ni2P/NiCr(OH)2 requires a low overpotential of 0.111 V to reach a current density of 10 mA cm−2, whereas Ni2P needs a high value of 0.230 V, indicating its relatively inferior catalytic activity. The HER performance of Ni2P/NiCr(OH)2 is outstanding and superior to that of most reported catalysts, as summarized in Fig. 3g and Table S1. When the current density increases to 300 mA cm−2, the required overpotential of Ni2P/NiCr(OH)2 is as low as 0.281 V, considerably lower than that of Ni2P (0.456 V) and Pt/C (0.340 V). To further investigate the reaction kinetics, Tafel plots derived from corresponding LSV curves were analyzed. As shown in Fig. 3b and f, the Tafel slope of Ni2P/NiCr(OH)2 is 109.1 mV dec−1, lower than that of Ni2P (162.5 mV dec−1), demonstrating that NiCr(OH)2 incorporation can remarkably accelerate catalytic kinetics and achieve high current densities at low overpotentials. The double-layer capacitance values (Cdl) were calculated from cyclic voltammetry (CV) curves in the non-faradaic region (Fig. S4) to obtain the electrochemical active surface area (ECSA). As shown in Fig. 3c and f, the Cdl value of Ni2P/NiCr(OH)2 is 20.6 mF cm−2, significantly larger than that of Ni2P (5.7 mF cm−2), suggesting abundant active sites and high intrinsic activity of Ni2P/NiCr(OH)2. To probe the electrode interfacial properties and charge transfer kinetics, electrochemical impedance spectroscopy (EIS) measurements were conducted. As presented in Fig. 3d and f, the charge transfer resistance (Rct) of Ni2P/NiCr(OH)2 is calculated to be 0.27 Ω, which is smaller than that of Ni2P (0.53 Ω), confirming that NiCr(OH)2 introduction is beneficial to reduce charge transfer resistance, accelerate charge transfer and deliver superior electrocatalytic activity. In addition, in situ Bode plots (Fig. 3e and S5) indicate that Ni2P/NiCr(OH)2 possesses strong capacitive behavior and efficient HER charge transfer kinetics in the high-frequency region. The above results indicate that NiCr(OH)2 introduction plays a crucial role in enhancing HER performances. The long-term stability of a catalyst is a key indicator for practical application. The stability test result in Fig. 3h shows that required potentials of Ni2P/NiCr(OH)2 remain nearly unchanged after continuous operation for 20 h. After the stability test, the SEM image of Ni2P/NiCr(OH)2 (Fig. S6) shows that there is no significant change in surface morphology, verifying its excellent structural stability.The OER performances of Ni2P/NiCr(OH)2 were further evaluated. As shown in Fig. 4a and f, Ni2P/NiCr(OH)2 exhibits excellent OER activity with small potentials of 1.542 and 1.564 V at 60 and 300 mA cm−2, respectively, lower than those of Ni2P (1.563 and 1.608 V, respectively) and commercial RuO2 (1.680 and 1.878 V, respectively). Moreover, the Cdl value of Ni2P/NiCr(OH)2 (Fig. S7) is higher than that of Ni2P, indicating that Ni2P/NiCr(OH)2 can expose more catalytically active sites. Considering that the high inherent potential of the OER leads to significant increased energy consumption, the thermodynamically favorable SOR was further explored in 1 M NaOH and 1 M Na2S. As shown in Fig. 4b and f, Ni2P/NiCr(OH)2 displays outstanding SOR activities with small voltages to achieve current densities of 10 and 300 mA cm−2 at 0.292 and 0.401 V, respectively, obviously lower than those of Ni2P (0.361 and 0.785 V, respectively), indicating that the introduction of NiCr(OH)2 can effectively regulate the surface electronic structure of Ni2P and significantly enhance SOR activity. The required potential for Ni2P/NiCr(OH)2 is lower than that of other previously developed catalysts (Fig. 4g and Table S2), fully demonstrating its performance advantage in electrocatalytic reactions. To analyze the SOR kinetic advantage of Ni2P/NiCr(OH)2, the Tafel curves were fitted and systematically analyzed. As shown in Fig. 4c and f, the Tafel slope of Ni2P/NiCr(OH)2 is 24.3 mV dec−1, which is much smaller than those of Ni2P (65.3 mV dec−1) and commercial RuO2 (118.2 mV dec−1), further implying that Ni2P/NiCr(OH)2 has fast SOR kinetics, which can guarantee the efficient progress of the SOR. To reveal the intrinsic mechanism of the OER and SOR, Bode plots were used to analyze reaction kinetic processes of Ni2P and Ni2P/NiCr(OH)2 at different potentials. As shown in Fig. 4d, e and S8, the phase angle of Ni2P/NiCr(OH)2 is significantly smaller than that of Ni2P in the low-frequency range, indicating that Ni2P/NiCr(OH)2 has fast electron transfer rate and reaction kinetics during the OER and SOR. Notably, when the potential increases to 1.501 V, the phase angles of both Ni2P and Ni2P/NiCr(OH)2 decrease sharply, indicating that a violent OER occurs, which is in agreement with previous results. As shown in Fig. 4f and S9, the potentials of Ni2P/NiCr(OH)2 remain nearly stable after continuous operation of 20 h under SOR and OER conditions, respectively. The post-test SEM images (Fig. S10 and S11) reveal that the nanosheet morphology of Ni2P/NiCr(OH)2 is well preserved, demonstrating its outstanding SOR and OER stabilities.
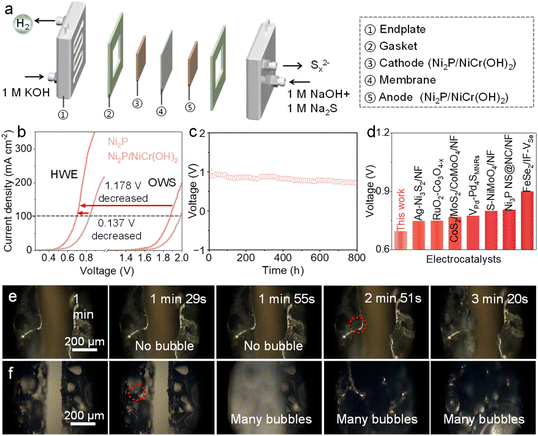
Given excellent bifunctional performances of Ni2P/NiCr(OH)2, the Ni2P/NiCr(OH)2-based flow electrolyzer (Fig. 5a) was integrated to verify its practical application, which employs a proton exchange membrane to separate the cathode and anode chambers, enabling efficient isolation and ion transport of cathodic and anodic reactions. As shown in Fig. 5b, the needed voltages of the HWE system are significantly lower than those of the OWS system at the same current density, confirming that replacing the OER with the SOR can effectively reduce energy input and achieve energy-saving H2 production. Specifically, the required voltages of HWE are only 0.513 and 0.698 V to attain 10 and 100 mA cm−2, respectively, much lower than those required in the OWS system (1.647 and 1.876 V, respectively) and many previously reported catalysts (Fig. 5d and Table S3). Meanwhile, Ni2P/NiCr(OH)2 shows much better electrocatalytic performance than Ni2P, where NiCr(OH)2 effectively boosts charge transfer and optimizes the surface structure. The long-term durability test of the Ni2P/NiCr(OH)2-based electrolyzer was performed. As shown in Fig. 5c, the HWE system driven by Ni2P/NiCr(OH)2 can ensure stable operation for up to 800 h with negligible voltage variation at 200 mA cm−2. Furthermore, the post-test SEM image (Fig. S12) reveals that its structure remains largely intact, fully confirming its excellent durability. After the durability test, post-treatment and characterization were performed by adding sulfuric acid into the anodic electrolyte to gain a yellow powder product, which is verified as elemental sulfur (S8, PDF#77-0145, Fig. S13). The above results indicate that the Ni2P/NiCr(OH)2-based HWE can achieve efficient conversion of sulfur-containing species into high-value-added elemental sulfur. To further confirm the impact of NiCr(OH)2 introduction on catalytic performances of Ni2P, in situ dark-field optical microscopy (DFLM) of Ni2P and Ni2P/NiCr(OH)2 was carried out.50–52 As shown in Fig. 5e and f, the H2 bubble begins to generate on the surface of Ni2P/NiCr(OH)2 at 1 min 29 s, while no gas bubble is observed on the surface of Ni2P. Obvious and intensive gas bubbles occur on Ni2P/NiCr(OH)2 at 1 min 55 s, whereas the surface of Ni2P still does not show any phenomenon of gas generation until 2 min 52 s and a large number of H2 bubbles begin to form in Ni2P at 3 min 20 s. These results directly demonstrate that Ni2P/NiCr(OH)2 possesses a higher catalytic performance and more intense electrochemical reaction process than Ni2P. Therefore, the Ni2P/NiCr(OH)2-based HWE by coupling the HER and SOR not only effectively reduces overall electrolysis voltages, but also simultaneously realizes the high-value conversion of sulfur-containing wastewater into valuable elemental sulfur.
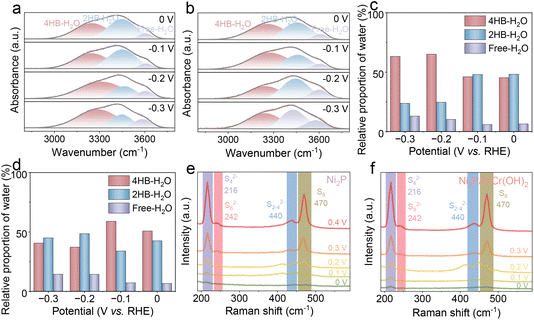
The in situ Raman spectroscopy of Ni2P and Ni2P/NiCr(OH)2 was performed to analyze the catalytic process of the HER and SOR in 1.0 M NaOH without and with 1.0 M Na2S electrolyte. For the HER, the interfacial water structures are analyzed (Fig. 6a and b), and the broad O–H stretching band (2800–3800 cm−1) is deconvolved into three Gaussian peaks located at 3250 cm−1, 3420 cm−1, and 3590 cm−1, which correspond to 4-coordinated H-bond water (4HB-H2O), 2-coordinated H-bond water (2HB-H2O), and the dangling O–H bonds of interfacial water (free-H2O), respectively.53 As we know, as the hydrogen-bond density among interfacial H2O molecules increases, the corresponding O–H stretching vibration energy reduces, indicating that interfacial H2O molecules with fewer hydrogen bonds are more easily dissociated. Consequently, the activation energy barrier for interfacial H2O dissociation follows the order: free-H2O < 2HB-H2O < 4HB-H2O. As shown n Fig. 6c and d, Ni2P/NiCr(OH)2 has a higher proportion of free-H2O compared with Ni2P, indicating that the enriched free H2O at the Ni2P/NiCr(OH)2 interface favors catalytic performance.54 These results show that the NiCr(OH)2 introduction can restructure the interfacial water structure and weaken the hydrogen-bond network, which are beneficial to H2O molecule mobility and reorientation, thus accelerating H2O dissociation to form hydrogen intermediates, thus remarkably enhancing the HER activity.55 For the SOR process, as the potentials gradually increase new characteristic peaks gradually appear at 216, 242, 440 and 470 cm−1 on the surface of Ni2P and Ni2P/NiCr(OH)2, which belong to S42−, S82−, S2–42− and S8, respectively (Fig. 6e and f). These results confirm that a strong SOR response occurs and the S2− gradually oxidizes to generate the short-chain polysulfur intermediates (Sx2−), which eventually form S8 molecules. However, the Raman characteristic peaks of Ni2P are weaker compared to those of Ni2P/NiCr(OH)2 (Fig. 6e and f) with increasing potentials, indicating that the introduction of NiCr(OH)2 effectively promotes the conversion of S2− to polysulfide and elemental S8.56,57 These results verify that Ni2P/NiCr(OH)2 has better adsorption and conversion capacities in terms of polysulfide ions than Ni2P, which effectively facilitates the SOR performance.
Conclusions
In summary, Ni2P/NiCr(OH)2 nanosheets were successfully fabricated via a two-step hydrothermal and phosphorization route. Ni2P/NiCr(OH)2 exhibits superior bifunctional activities for the HER and SOR, which deliver 10 mA cm−2 at 111 mV and 292 mV, respectively. In situ Raman spectroscopy and DFLM confirm that NiCr(OH)2 incorporation can reorganize the interfacial water structure and boost reaction kinetics. The assembled electrolyzer exhibits ultra-long stability for 800 h and enables high current densities at low voltages, obviously lower than those required for traditional water electrolysis. This study provides a strategy to modulate the interfacial water structure and achieve energy-saving hydrogen production and resource utilization of sulfion-rich wastewater.Author contributions
Xiaojun Wang: data curation and formal analysis. Jieting Li: investigation. Xinyu Zhu: formal analysis. Zehua Xu: investigation. Shuixiang Xie: data curation. Shutong Chen: formal analysis. Yuxuan Wan: data curation. Guangyu Zhang: investigation. Mingzheng Ge: investigation. Wei Zhang: investigation. Chao Wang: conceptualization & investigation. Rui-Qing Li: conceptualization, writing – review & editing.Conflicts of interest
There are no conflicts to declare.Data availability
The relevant experimental and characterization data are available in the article and the supplementary information (SI). Supplementary information: the relevant experimental and characterization data including SEM images, XPS spectrum, CV curves, Bode plots Cdl values. See DOI: https://doi.org/10.1039/d6sc02309f.Acknowledgements
This research was supported by the National Natural Science Foundation of China (No. 22302103), the Natural Science Foundation of Jiangsu Province (No. BK20230619), the Natural Science Foundation of the Jiangsu Higher Education Institutions of China (No. 23KJB540003), the Postgraduate Research & Practice Innovation Program of Jiangsu Province (No. KYCX25_3762), the National-Level Innovative Training Program Project for College Students of Nantong University (No. 202510304048), and Nantong University Analysis & Testing Center for SEM Characterization.Notes and references
- K. Modi, P. Pataniya, S. Siraj, P. Sahatiya, V. Patel and C. Sumesh, J. Energy Storage, 2023, 63, 107040 CrossRef.
- R. Li, X. Xu, J. Zeng, X. Zhang, X. Wan, S. Guo, X. Wang, S. Xie, Z. Cao, Y. Zhang, C. Wang, J. Deng, O. Fontaine, M. Ge, J. Dai, G. Zhang, W. Zhang, X. Wang and Y. Zhu, Nano Lett., 2025, 25, 1272 CrossRef CAS PubMed.
- J. Masnica, S. Sibt-e-Hassan, S. Potgieter-Vermaak, Y. Regmi, L. King and L. Tosheva, Green Carbon, 2023, 1, 160 CrossRef CAS.
- X. Zhang, Y. Chen, Z. Ye, H. Hu, L. Lei, F. You, J. Yao, H. Yang and X. Jiang, Chin. J. Struct. Chem., 2024, 43, 100200 CAS.
- S. Chauhan, K. Joshi, P. Pataniya and C. Sumesh, Sustainable Energy Fuels, 2025, 9, 3550 RSC.
- J. Liu, Y. Liu, B. Nan, D. Wang, C. Allen, Z. Gong, G. He, K. Fu, G. Ye and H. Fei, Angew. Chem., Int. Ed., 2025, 64, e202425196 CrossRef CAS PubMed.
- W. Song, J. Wu, J. Li, Z. Wang, T. He, W. Chen, D. Sun and J. Wu, Green Carbon, 2024, 2, 393 CrossRef CAS.
- S. Xie, X. Zhu, Y. Zhang, F. Zhang, W. Fan, Z. Yang, G. Zhang, M. Ge, W. Zhang and R. Li, Chem. Commun., 2026, 62, 1641 RSC.
- Y. Cheng, Y. wang, Z. Shi, H. Wu, J. Yang, J. Ni, M. Yang, Z. Wang, M. Xiao, C. Liu and W. Xing, EcoEnergy, 2025, 3, 131 CrossRef CAS.
- J. Liang, Z. Cai, X. He, Y. Luo, D. Zheng, S. Sun, Q. Liu, L. Li, W. Chu, S. Alfaifi, F. Luo, Y. Yao, B. Tang and X. Sun, Chem, 2024, 10, 3067 CAS.
- M. Song, X. Yang, C. Guo, S. Zhang, J. Ma and H. Gao, EcoEnergy, 2025, 3, 470 CrossRef CAS.
- S. Dristy, M. Habib, M. Joni, M. Najibullah, R. Mandavkar, S. Lin and J. Lee, Chin. J. Struct. Chem., 2025, 44, 100747 CAS.
- C. Wang, L. Zhu, X. Wang and M. Wu, Green Carbon, 2025, 3, 225 CrossRef.
- H. Tüysüz, Acc. Chem. Res., 2024, 57, 558 Search PubMed.
- Y. Chen, Y. Liu, L. Li, T. Sakthive, Z. Guo and Z. Dai, Adv. Funct. Mater., 2024, 34, 2406587 CrossRef CAS.
- S. Wang, Z. Geng, S. Bi, Y. Wang, Z. Gao, L. Jin and C. Zhang, Green Energy Environ., 2024, 9, 659 CrossRef CAS.
- Y. Wang, L. Chen, H. Zhang, M. Humayun, J. Duan, X. Xu, Y. Fu, M. Bououdina and C. Wang, Green Chem., 2023, 25, 8181 RSC.
- H. Sun, W. Zhang, J. Li, Z. Li, X. Ao, K. Xue, K. Ostrikov, J. Tang and C. Wang, Appl. Catal., B, 2021, 284, 119740 CrossRef CAS.
- R. L, X. Wang, S. Xie, S. Guo, Z. Cao, Z. Yan, W. Zhang and X. Wan, Chem. Sci., 2025, 16, 809 RSC.
- X. Zhang, J. Wang, K. Zong, Z. Chen, X. Yang, L. Yang, X. Wang and Z. Chen, Carbon Energy, 2025, 7, e679 CrossRef CAS.
- Y. Zhang, L. Liao, H. Zhou, Y. Qi, J. Sun, Y. Zhang, Q. Zhou, Y. Wang, D. Tang and F. Yu, Energy Environ. Sci., 2025, 18, 7695 RSC.
- Y. Ramli, V. Chaerusani and Z. Yang, Green Carbon, 2025, 3, 113 CrossRef.
- Y. Zhang, H. Wang, J. Lee and Q. Zhang, Chin. J. Catal., 2023, 55, 44 CrossRef.
- H. Thakkar, K. Modi, K. Joshi, G. Bhadu, S. Siraj, P. Sahatiya, P. Pataniya and C. Sumesh, ACS Sustainable Chem. Eng., 2024, 12, 8340 CrossRef CAS.
- R. Li, X. Wang, S. Xie, S. Guo, Z. Cao, Z. Yan, W. Zhang and X. Wan, Chem. Sci., 2025, 16, 809 RSC.
- Y. Nie, X. Liu, J. Li, R. Wang and B. Dong, Chem. Commun., 2026, 62, 594 RSC.
- L. Ai, Y. Tian, T. Xiao, J. Zhang, C. Zhang and J. Jiang, J. Colloid Interface Sci., 2024, 673, 607 CrossRef CAS.
- J. Huo, L. Jin, C. Chen, D. Chen, Z. Xu, C. Wilfred, Q. Xu and J. Lu, ACS Appl. Mater. Interfaces, 2023, 15, 43976 CrossRef CAS PubMed.
- S. Xie, X. Wang, Y. Li, S. Liu, J. Qian, Y. Zhang, L. Jiang, Z. Cao, Z. Yan, X. Wan, Z. Yang, L. Zou, W. Zhang and R. Li, Chem. Sci., 2025, 16, 12587 RSC.
- C. Duan, C. Tang, S. Yu, L. Li, J. Li and Y. Zhou, Appl. Catal., B, 2023, 324, 122255 CrossRef CAS.
- H. Yang, X. Long, F. Liu, J. Zhou, N. Chen, R. Feng, Y. Zhang, X. Fu, J. Luo and B. Zhao, Appl. Catal., B, 2025, 366, 125037 CrossRef CAS.
- X. Li, W. Yu, Y. Wang, R. Liu, Q. Yu, R. Hu, X. Jiang, Q. Gao, H. Liu, J. Yu and W. Zhou, Chin. Chem. Lett., 2024, 35, 109166 CrossRef CAS.
- S. Shi, M. Li, J. Zeng, J. Wang, G. Zeng and Q. Zhong, J. Electroanal. Chem., 2025, 986, 119076 CrossRef CAS.
- Z. Yu, D. Boukhvalov, H. Tan, D. Xiong, C. Feng, J. Wang, W. Wang, Y. Zhao, K. Xu, W. Su, X. Xiang, F. Lin, H. Huang, F. Zhang, L. Zhang, L. Meng and L. Liu, Chem. Eng. J., 2024, 494, 153094 CrossRef CAS.
- Z. Liang, Y. Nian, H. Du, P. Li, M. Wang and G. Ma, Materials, 2025, 18, 3377 CrossRef CAS PubMed.
- H. Li, J. Ding, Z. Wei, L. Zhuo, I. Shakir, J. Deng, G. Hu and X. Liu, Int. J. Hydrogen Energy, 2026, 199, 152878 CrossRef CAS.
- M. Koudahi and L. Naji, Electrochim. Acta, 2022, 434, 141314 CrossRef.
- M. Yang, J. Ding, Z. Wang, J. Zhang, Z. Peng and X. Liu, Chin. Chem. Lett., 2025, 36, 110861 CrossRef CAS.
- S. Liu, Y. Gu, J. Kang, J. Zhang, S. Liu, H. Yang, X. Zhu and M. Ming, Int. J. Hydrogen Energy, 2026, 198, 152670 CrossRef CAS.
- S. Zhang, X. Yang, L. Ma, J. Ma and H. Gao, Mater. Today Chem., 2025, 49, 103095 CrossRef CAS.
- L. Wu, L. Yu, F. Zhang, B. McElhenny, D. Luo, A. Karim, S. Chen and Z. Ren, Adv. Funct. Mater., 2021, 31, 2006484 CrossRef CAS.
- H. Huo, H. Hou, Y. Yu, J. Xu, W. Chen, S. Wang and D. Min, Electrochim. Acta, 2025, 544, 147697 CrossRef CAS.
- Z. Wang, G. Yang, P. Tian, X. Li, K. Deng, H. Yu, Y. Xu, H. Wang and L. Wang, Chem. Eng. J., 2023, 473, 145147 CrossRef CAS.
- J. Park, H. Yoon, D. Lee, S. Ji, W. Yang, S. Tilley, M. Sung, I. Park, J. Tan, H. Lee, J. Kim, D. Kim and J. Moon, Appl. Catal., B, 2022, 305, 121045 CrossRef CAS.
- Y. Pei, D. Li, C. Qiu, L. Yan, Z. Li, Z. Yu, W. Fang, Y. Lu and B. Zhang, Angew. Chem., Int. Ed., 2024, 136, e202411977 CrossRef.
- Y. Nie, H. Feng, H. Wang, Y. Hu, L. Cao and B. Dong, Chem. Eng. J., 2025, 523, 168370 CrossRef CAS.
- F. Sun, Y. Wang, H. Gao, X. Tian, R. Zhu, Y. Zheng, J. Zang and L. Dong, Appl. Catal. B Environ., 2025, 383, 126109 CrossRef.
- R. Li, S. Guo, X. Wang, X. Wan, S. Xie, Y. Liu, C. Wang, G. Zhang, J. Cao, J. Dai, M. Ge and W. Zhang, Chem. Sci., 2024, 15, 10084 RSC.
- Y. Dang, P. Düngen, J. Hao, X. Sun, Y. Ding and S. Heumann, Green Carbon, 2026 DOI:10.1016/j.greenca.2025.10.009.
- C. Wang, Q. Xie, G. Wang, Y. Lyu, Q. Wang, X. Ma, H. Wang, T. Guo, Y. Wu and J. Han, Nano Lett., 2024, 24, 13796 CrossRef CAS PubMed.
- C. Wang, H. Wang, X. Ma, T. Guo, A. Zheng, Y. Zhao, X. Li, Y. Wu, S. Li and J. Han, Nano Lett., 2025, 25, 6556 CrossRef CAS PubMed.
- N. Liu, G. Zhou, A. Yang, X. Yu, F. Shi, J. Sun, J. Zhang, B. Liu, C. Wu, X. Tao, Y. Sun, Y. Cui and S. Chu, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 765 CrossRef CAS.
- Z. Huang, Z. Chen, C. Xu, L. Wang, M. Gao, S. Zhang, T. Isimjan, X. Shi, Y. Ye, L. Chen, J. Wang, X. Yang, Y. Zhu and B. Wu, Adv. Funct. Mater., 2026, e14861 CrossRef CAS.
- Y. Wan, Y. Tang, Y. Zuo, K. Sun, Z. Zhuang, Y. Zheng, W. Yan, J. Zhang and R. Lv, Energy Environ. Sci., 2025, 18, 7460 RSC.
- J. Zhu, X. Sun, N. Feng, B. Zhao, M. Qiu, J. Xu and W. Luo, J. Am. Chem. Soc., 2025, 147, 47454 CrossRef CAS PubMed.
- Y. Nie, H. Feng, H. Wang, Y. Hu, L. Cao and B. Dong, Chem. Eng. J., 2025, 523, 168370 CrossRef CAS.
- C. Duan, C. Tang, S. Yu, L. Li, J. Li and Y. Zhou, Appl. Catal., B, 2023, 324, 122255 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |