Open Access Article
Open Access ArticleCascade charge-transport-chain engineering in alloy nanocluster–semiconductor artificial photosystems
Zi-Han Zhenga,
Qing Chena,
Peng Sua,
Lifeng Cai*b,
Jie Liangb,
Guangcan Xiaoc and
Fang-Xing Xiao *a
*a
aCollege of Materials Science and Engineering, Fuzhou University, New Campus, Fujian 350116, P. R. China. E-mail: fxxiao@fzu.edu.cn
bCollege of Environmental and Biological Engineering, Fujian Provincial Key Laboratory of Ecology-Toxicological Effects & Control for Emerging Contaminants, Putian University, Putian, Fujian 351100, P. R. China. E-mail: 89437499@qq.com
cInstrumental Measurement and Analysis Center, Fuzhou University, Fuzhou, 350108, P.R. China
First published on 23rd April 2026
Abstract
Atomically precise metal nanoclusters (NCs), featuring a discrete electronic structure and pronounced quantum confinement effects, are emerging as promising photosensitizers for artificial photosystems; however, their practical implementation remains fundamentally constrained by rapid charge recombination and poorly controlled charge transport. Here, we introduce a conceptual cascade charge-transport-chain engineering strategy that addresses this intrinsic bottleneck by constructing directional and continuous carrier transport pathways across NC–semiconductor interfaces. By integrating alloy NC photosensitization with atomic Ni doping of TiO2, robust interfacial electronic coupling is established, enabling directional and accelerated extraction of photogenerated carriers. The resulting BNC/Ni–TiO2 heterostructures exhibit markedly enhanced visible-light-driven hydrogen evolution, accompanied by effective suppression of charge recombination within alloy NCs. Combined experimental and theoretical investigations reveal that the performance enhancement originates from cascade charge-transport-chain engineering rather than simple binary synergy. This work provides a general design principle for constructing tunable charge-transport pathways with alloy NCs, advancing NC-based artificial photosystems toward solar-to-hydrogen energy conversion.
1. Introduction
Atomically precise nanoclusters (NCs) with discrete electronic structures and pronounced quantum confinement effects, have emerged as promising photosensitizers for artificial photosystems, owing to their superior light-harvesting properties.1,2 However, the widespread application of metal NCs is hindered by fundamental challenges, such as rapid charge recombination and poorly controlled charge transport, which prevent the construction of stable and efficient metal NC-based systems.3In recent years, heterometallic NCs particularly bimetallic NCs (BNCs) have garnered increasing attention as a new generation of light-harvesting antennae for solar energy conversion. BNCs are synthesized by incorporating two different metals into a single nanocluster unit, offer a promising strategy to overcome the inherent limitations of traditional monometallic NCs through bimetallic synergy. Through atomic-scale manipulation of metal composition and spatial arrangement, BNCs offer distinct advantages, including (i) precise control of their geometric and electronic structures; (ii) enhanced light-harvesting efficiency, and (iii) accelerated charge transport kinetics. These features position BNCs as ideal active centers for photon capture and catalytic reactions.4 Despite these compelling merits, the use of BNCs as photocatalysts remains limited by poor charge separation efficiency and low photocatalytic activity. To address these challenges, one promising strategy is the coupling of BNCs with metal oxide semiconductors, such as titanium dioxide (TiO2), to form heterostructures with optimized energy level alignment. This integration extends the photo-response range of the metal oxide substrate and enhances charge migration and separation over the BNCs, facilitating efficient photoredox catalysis. However, achieving efficient charge transport and separation remains a critical issue in the design of high-performance artificial photosystems.
A promising solution lies in the deposition of suitable co-catalysts (e.g., Ru, Pd, and Pt) on the TiO2 surface, which increases the number of active sites for accelerated photocatalytic reactions while mitigating carrier recombination.5 Among these co-catalysts, nickel (Ni) stands out as a low-cost and efficient option for photocatalytic hydrogen generation due to its ability to boost charge transfer kinetics and its strong affinity for active hydrogen species.6 Combining Ni with BNCs offers a dual-functional strategy for increasing the number of active sites on TiO2 and extending the visible light absorption capabilities of TiO2, which together maximize solar energy utilization and drive charge flow.6b,7 By strategically incorporating Ni co-catalysts and simultaneously anchoring alloy NCs (Au1−xAgx, Au1−xPtx, and Au1−xCux) onto TiO2, and fine-tuning the interface configuration, we hypothesize that a controllable charge transport pathway can be stimulated. Once the appropriate energy level alignment between the components is optimized, this will facilitate accelerated directional charge migration and suppress carrier recombination, ultimately leading to significantly improved photocatalytic hydrogen production performance under visible light. This integrated strategy paves the way for the development of novel alloy NC-based artificial photosystems.
Inspired by these insights, herein, we conceptually present a cascade charge-transport-chain engineering strategy via constructing TiO2–Ni/BNC (Au1−xAgx, Au1−xPtx, and Au1−xCux) heterostructure artificial photosystems for solar-to-hydrogen conversion. Specifically, Ni is doped onto the surface of TiO2 nanosheets via a molten-salt method (MSM), followed by electrostatic self-assembly of tailor-made alloy NCs onto the Ni-doped TiO2 (TiO2–Ni) to form the TiO2–Ni/BNC (Au1−xAgx, Au1−xPtx, and Au1−xCux) ternary photocatalysts. The results demonstrate that the photosensitization effect of BNCs broadens the light absorption range of pristine TiO2, while the accelerated interfacial charge transfer enabled by Ni doping significantly enhances the photocatalytic hydrogen evolution activities of the TiO2–Ni/BNC heterostructures under visible light. Through a combination of experimental and theoretical investigations, we elucidate the charge transfer mechanism within the TiO2–Ni/BNC photosystems, revealing a synergistic effect between atomic-scale BNCs and metal oxide supports. This work highlights the promising potential of BNCs as high-performance photosensitizers and presents a novel approach to engineering cascade charge-transport-chains that enhance the efficiency of metal NC–semiconductor hybrid artificial photosystems for efficient solar-to-hydrogen energy conversion.
2. Experimental section
2.1 Materials
All the materials were of analytical grade and used as received without further purification, and the detailed information is provided in the SI.2.2 Preparation of photocatalysts
2.3 Characterization
The crystal structure was determined by X-ray diffraction (XRD, Miniflex600). Morphologies were visualized by field emission scanning electron microscopy (FESEM, Supra55, Carl Zeiss) equipped with energy-dispersive spectroscopy (EDS) and transmission electron microscopy (TEM, Tecnai G2 F20). Fourier transform infrared (FTIR) spectra were recorded on an infrared spectrophotometer (TJ270-30A). Raman spectra were collected with a Raman spectrometer (Dxr-2xi, Thermo Scientific, America). UV-visible diffuse reflectance spectra (DRS) were measured on a Cary50 (Varian, America) with a reflectance background of 200 to 800 nm. X-ray photoelectron spectra (XPS) were recorded on a photoelectron spectrometer (ESCALAB 250, Thermo Scientific, America), and the binding energy (BE) of the elements was calibrated by the BE of C 1s (284.80 eV). Zeta potential (ξ) measurements were performed by dynamic light scattering analysis (Zeta sizer Nano ZS-90). Time-resolved photoluminescence (TRPL) spectra were measured on a FLS 920 fluorescence lifetime spectrophotometer (Edinburgh, Instruments, UK). The photoluminescence (PL) spectra were probed on a Varian Cary Eclipse spectrophotometer. Brunauer–Emmett–Teller (BET) specific surface area and N2 adsorption experiments were performed on an ASAP 2460. Concentrations of T-N0.25/10Au1−xAgx were quantified by inductively coupled plasma emission spectroscopy (ICP, XSERIES 2 ICP-MS). X-ray absorption fine structure (XAFS) spectroscopy was carried out using a Rapid XAFS 2M (Anhui Absorption Spectroscopy Analysis Instrument Co., Ltd) in transmission (or fluorescence) mode at 20 kV and 30 mA. Kelvin-Probe Force Microscopy (KPFM) was performed on a Dimension Icon (Bruker, America).2.4 Photocatalytic H2 production performances
Photocatalytic reactions were conducted on an online photocatalytic hydrogen production system using a Pyrex vessel. Specifically, 10 mg of the catalyst was dispersed in mixed solution (DI H2O, 2 mL), and then triethanolamine (TEOA, 0.5 mL) was added as the sacrificial agent. The photocatalytic system was thoroughly degassed and irradiated with a 300 W Xe lamp (PLS-SXE300D, Beijing Perfectlight Technology Co., Ltd) equipped with a 420 nm-cut-off optical filter (λ > 420 nm). Continuous magnetic stirring was applied at the bottom of the reactor to keep the catalyst in suspension during the whole experiment. The evolved H2 was monitored periodically using an online gas chromatograph (Shimadzu GC-8A with an MS-5A column using argon as carrier gas). Photoactivities were evaluated based on the H2 evolution amount in the first 2 h of the reaction. Cyclic photocatalytic H2 evolution reactions were carried out as follows. Specifically, the photocatalytic system was thoroughly degassed again after the first run without separating the catalysts and supplementing the TEOA. Subsequently, the thoroughly degassed system was irradiated again with a 300 W Xe lamp S10 with a UV-CUT filter to cut off light with a wavelength λ < 420 nm. Analogously, the following four runs of photocatalytic recycling tests were performed.2.5 Photoelectrochemical (PEC) measurements
PEC measurements were carried out on an electrochemical workstation (CHI660E, CHI Shanghai, Inc.) under visible light irradiation (λ > 420 nm) (PLS-SXE300D, Beijing Perfect Light Co. Ltd, China), with a conventional three-electrode system and 0.5 M Na2SO4 aqueous solution (pH = 6.69) was utilized as the electrolyte. The three-electrode system is composed of Pt foil (1 cm × 1 cm) as the counter electrode, an Ag/AgCl electrode as the reference electrode, and working electrodes. The working electrodes were prepared on fluorine-doped tin oxide (FTO) glass that was cleaned by sonication in DI H2O and ethanol for 30 min and dried at 60 °C in an oven. The specific operational information is provided in the SI.3. Results and discussion
3.1 Structural characterization
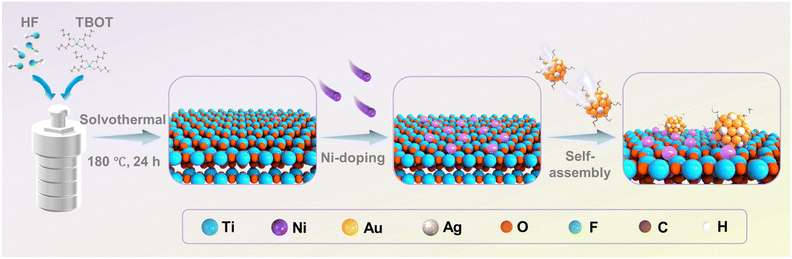
The synthesis procedures of the TiO2–Ni0.25/Au1−xAgx ternary heterostructure are depicted in Scheme 1. Initially, the Ni element was doped on the TiO2 nanosheets by MSM. Upon reaching the melting point of 625 K, the salts form a liquid phase, NiCl2 dissolves and is dispersed, generating a strong polarizing force (a pool of ionized anions and cations).11 This force destabilizes the Ti–O bonds on the surface of TiO2 nanosheets, facilitating the formation of Ni–O bonds between surface oxygen ions and mobile Ni2+ ions. Furthermore, inserting Ni2+ ions into the pre-formed, tightly bonded TiO2 lattice would require overcoming both lattice distortion energy and high diffusion energy barriers, which is thermodynamically unfavorable.12 Therefore, the MSM promotes the formation of stable Ni–O bonds on the TiO2 surface at relatively low temperature, thereby effectively preserving the structural integrity of the TiO2 structures. Subsequently, Au1−xAgx NCs were deposited onto the Ni-doped TiO2 (TiO2–Ni) through electrostatic self-assembly under ambient conditions, resulting in the TiO2–Ni/Au1−xAgx ternary heterostructures. More specifically, under acidic conditions, abundant hydroxyl (–OH) groups on the surface of TiO2 facilitate the proton (H+) migration and adsorption, rendering the surface positively charged.13 The surface of Au1−xAgx NCs (ca. 1.7 nm) is capped with glutathione (GSH) ligands bearing carboxyl (–COO−) functional groups, and thus they feature a negatively charged surface (Fig. S1). Thus, negatively charged Au1−xAgx NCs are attracted by the positively charged TiO2 substrate via electrostatic interaction, ultimately forming the TiO2/Au1−xAgx NCs heterostructure under ambient conditions. Similarly, Ni-doped TiO2 assembles with the Au1−xAgx NCs in the same way, giving rise to the TiO2–Ni/Au1−xAgx NCs heterostructure. After Ni doping and Au1−xAgx NC decoration, the color of the samples gradually changes from white to yellow (Fig. S2), indicating the successful modification of TiO2. This electrostatic self-assembly strategy is also utilized to fabricate the TiO2–Ni/alloy NC (Au1−xPtx and Au1−xCux) ternary heterostructures. The detailed characterization of Au1−xCux and Au1−xPtx NCs is specifically illustrated in Fig. S3 and S4, respectively.As shown in Fig. 1a, all the XRD results of TiO2, T-Ni0.25, T/Au1−xAgx, and T-Ni0.25/Au1−xAgx exhibit the characteristic diffraction peaks at 25.3°, 37.8°, and 48.0°, corresponding to the (101), (004), and (200) crystal planes of anatase TiO2 (JCPDS: 99-0008), respectively.8 This observation confirms that Ni doping and Au1−xAgx NC loading preserve the crystalline phase of TiO2. The absence of distinct diffraction peaks of Au1−xAgx NCs or metallic Ni species can be attributed to their low content and high dispersion in the nanocomposite. Raman results (Fig. S5) further corroborate the anatase phase of TiO2, with no observable signals for Au1−xAgx NCs or Ni dopant, aligning with the XRD results. It should be emphasized that the significant enhancement of the Eg peak intensity observed after Ni doping can be attributed to a surface-enhanced resonance Raman scattering (SERRS) effect.2a,14
Fourier transform infrared (FTIR) spectroscopy (Fig. 1b and Table S1) results of all the samples reveal an apparent peak at 1390 cm−1, which is assigned to the Ti–O vibrational modes.15 Particularly, T/Au1−xAgx and T-Ni0.25/Au1−xAgx exhibit additional peaks at 1640, 1530, 3241, and 2925 cm−1, corresponding to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, N–H, and C–H functional groups derived from the GSH ligands stabilizing the Au1−xAgx NCs.16 These features strongly confirm the successful deposition of Au1−xAgx NCs on the TiO2 substrate.
O, N–H, and C–H functional groups derived from the GSH ligands stabilizing the Au1−xAgx NCs.16 These features strongly confirm the successful deposition of Au1−xAgx NCs on the TiO2 substrate.
Diffuse reflectance spectroscopy (DRS) results (Fig. 1c and d) demonstrate that the incorporation of Au1−xAgx NCs and Ni doping significantly enhance the visible-light harvesting capacity of TiO2. The optical absorption edge of pristine TiO2 is located in the ultraviolet light region of approximately 400 nm, corresponding to its intrinsic bandgap excitation.17 According to the Kubelka–Munk function, the Eg value of TiO2 is determined to be approximately 3.00 eV. For the composite samples, Eg values of T-Ni0.25, T/Au1−xAgx, and T-Ni0.25/Au1−xAgx are determined to be 2.82 eV, 2.63 eV and 2.36 eV, respectively. Note that Eg decreases with Ni-doping and Au1−xAgx NC loading. Especially, T/Au1−xAgx and T-Ni0.25/Au1−xAgx exhibit substantially improved visible-light absorption intensity after Au1−xAgx NC loading, primarily due to the photosensitization effect of Au1−xAgx NCs. In contrast, pure Ni-doping samples only marginally enhance the light absorption of TiO2, implying that Ni acts as a co-catalyst rather than a light-harvesting component in the T-Ni0.25/Au1−xAgx composite.
N2 adsorption–desorption isotherms combined with Brunauer–Emmett–Teller (BET) and Barrett–Joyner–Halenda (BJH) analyses (Fig. S6 and Table S3) reveal the specific surface area and porosity of the samples. All the samples exhibit type IV isotherms and hysteresis loops at relative pressures (P/P0) of 0.8–1.0, indicative of a mesoporous structure.8 The BET specific surface areas of TiO2, T-Ni0.25, T/Au1−xAgx, and T-Ni0.25/Au1−xAgx are determined to be 86.63, 74.92, 42.95, and 40.51 m2 g−1, respectively. The decreased surface area of TiO2 with Ni and Au1−xAgx NC incorporation might stem from aggregation and pore filling, implying that the specific surface area does not contribute to the enhanced photoactivities of T/Au1−xAgx, and T-Ni0.25/Au1−xAgx heterostructures. Instead, atomic-scale dispersion of Ni in the oxide state and BNC deposition likely generate more surface reactive sites that are critical for charge transfer and catalytic activity.
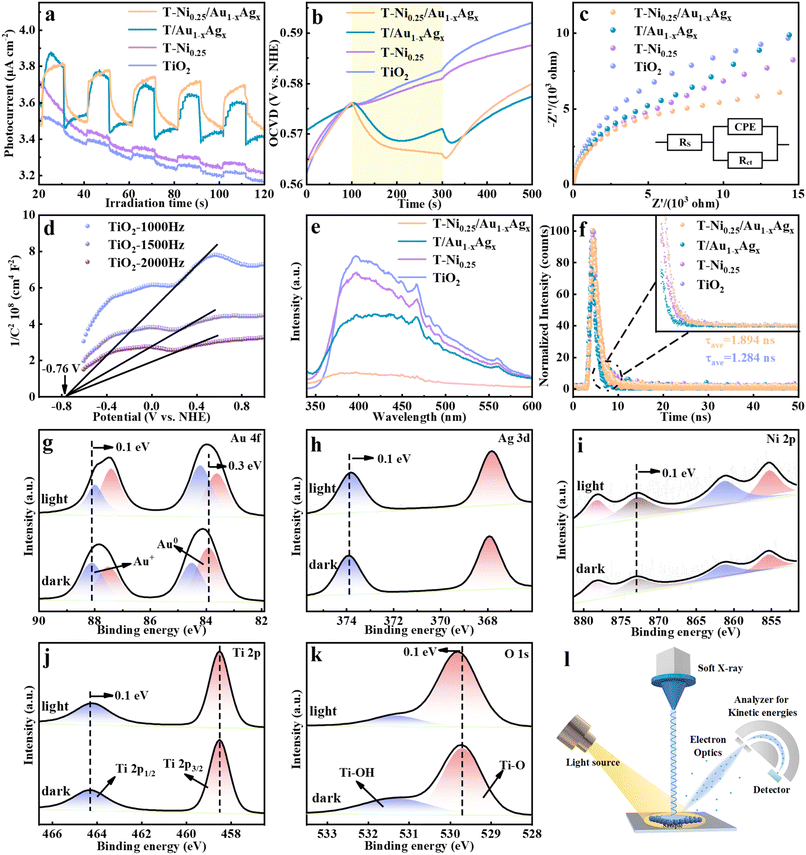
X-ray photoelectron spectroscopy (XPS) was used to analyze the surface composition and chemical bond states of pristine TiO2, T-Ni0.25, T/Au1−xAgx and T-Ni0.25/Au1−xAgx (Fig. S7 and Table S2). For pure TiO2, the peaks of Ti 2p (Fig. 1e) at 458.6 and 464.4 eV correspond to the Ti4+ species, while the O 1 s signals at 529.9 eV and 531.5 eV are assigned to the lattice oxygen and adsorbed oxygen, respectively.18 As for T-Ni0.25, the obvious shifts in the Ti 2p and O 1s binding energies (Fig. 1e and f) relative to pure TiO2 indicate the formation of strong Ni–O–Ti chemical bonds, confirming the covalent interaction between Ni species and the TiO2 substrate. In contrast, T/Au1−xAgx displays no discernible shifts in the Ti 2p or O 1s signals compared with those of pure TiO2 (Fig. S8), suggesting that Au1−xAgx NCs are physically adsorbed without disrupting the TiO2 lattice. For T-Ni0.25/Au1−xAgx, the peak shifts are dominated by the Ni–TiO2 interaction, while Au1−xAgx NCs are only adsorbed on the surface of the Ni-doped TiO2 and do not destroy the interaction between Ni species and the TiO2 substrate. The high-resolution Ni 2p spectrum of T-Ni0.25/Au1−xAgx (Fig. 1g) displays the characteristic peaks of Ni2+ at 855.3 eV, 861.3 eV, 872.9 eV, and 878.3 eV.19 The Au 4f signal of the T-Ni0.25/Au1−xAgx heterostructure (Fig. 1h) reveals a mixture of metallic Au (0) (84.1 eV and 87.6 eV) and oxidized Au(I) (84.5 eV and 88.2 eV) for Au1−xAgx NCs, while the Ag 3d peaks at 367.9 eV and 374.0 eV (Fig. 1i) verify the presence of Ag(I) species,2a aligning with the BNC composition. The XPS results of T-Ni0.25/Au1−xCux and T-Ni0.25/Au1−xPtx are specifically provided in Fig. S9 and S10, respectively. These XPS results confirm that Ni doping induces the atomic-scale interfacial bonding with TiO2, facilitating the interfacial charge transfer, while Au1−xAgx NCs act as efficient visible-light absorbers through physical adsorption. The XPS results strongly substantiate the successful Ni ion doping and BNC decoration in the ternary heterostructure. Moreover, the lack of structural disruption by BNC deposition ensures the integrity of the Ni–TiO2 interaction, thereby forming the synergistic heterostructure critical for enhanced photocatalytic performance.
As shown in Fig. 2a–f and S11, images at various magnifications reveal that all the samples maintain the two-dimensional (2D) nanosheet morphology of the TiO2 substrate, suggesting that Ni doping and Au1−xAgx NC loading do not alter the underlying architecture. As displayed in Fig. 2b and c and S12, TEM and HRTEM images of pristine TiO2 and T-Ni0.25 exhibit a regular 2D nanosheet morphology with lateral dimensions of 20–60 nm and a lattice spacing of 0.35 nm, corresponding to the (101) crystal plane of anatase TiO2.8 Critically, no distinct Ni nanoparticles or aggregates are observable in the TEM images of T-Ni0.25 (Fig. 2a–c), indicating that Ni is atomically dispersed or doped rather than forming bulk particles. In the TEM images of T-Ni0.25/Au1−xAgx (Fig. 2d–f), Au1−xAgx NCs appear as ultra-small size entities with a size of about 2 nm or less, uniformly distributed across the TiO2 nanosheets without disrupting the crystalline structure of the substrate.20 This confirms that the electrostatic self-assembly process effectively anchors the Au1−xAgx NCs onto the Ni-doped TiO2 nanosheet surface while preserving the nanosheet morphology. Energy-dispersive X-ray spectroscopy (EDS) mapping results (Fig. 2g–n) of the T-Ni0.25/Au1−xAgx composite material show homogeneous distributions of Ti, O, Ni, Au, and Ag elements, corroborating the aforementioned XPS results and once again providing direct evidence for the successful incorporation of both Ni and Au1−xAgx NCs ingredients. The uniform dispersion of Ni dopants and Au1−xAgx NCs across the TiO2 nanosheets underscores the structural integrity of the heterostructure. This morphological and compositional characterization collectively establishes that the synthetic strategy achieves atomic-level dispersion of Ni dopants while achieving ultrafine clustering of Au1−xAgx NCs on the TiO2 nanosheets, forming a well-defined heterostructure critical for efficient charge separation and photocatalytic activity.
 | ||
| Fig. 2 TEM and HRTEM images of (a–c) T-Ni0.25 and (d–f) T-Ni0.25/Au1−xAgx with corresponding (g–n) elemental mapping results for (h) Ti, (i) O, (j) N, (k) Ni, (l) Au, (m) Ag, and (n) C signals. | ||
3.2 Photocatalytic H2 generation activities
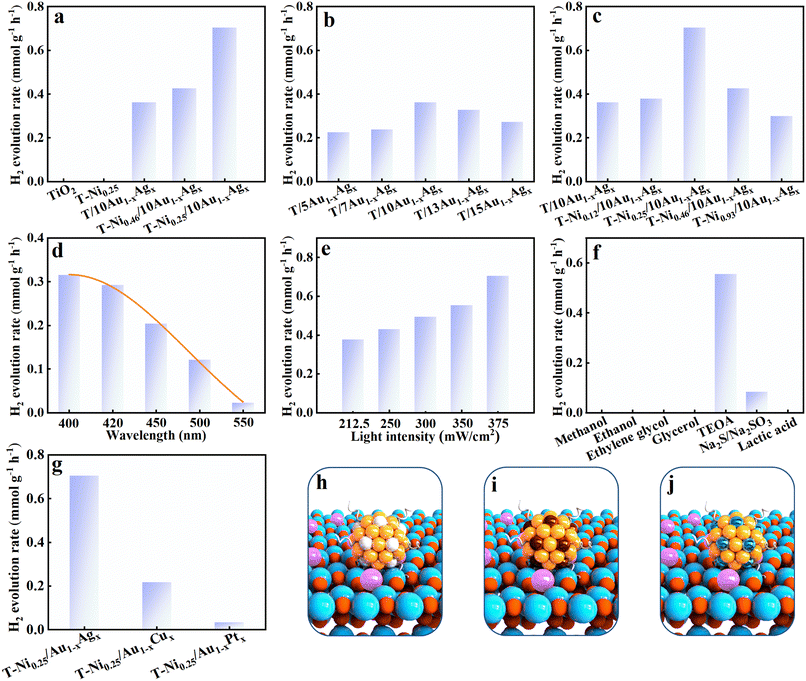
Using tri-ethanolamine (TEOA) as a hole-trapping agent, photocatalytic hydrogen evolution activities of the samples were systematically evaluated under visible light irradiation (λ > 420 nm). As shown in Fig. 3a, pristine TiO2 exhibited no hydrogen production activity under visible light, attributed to its wide bandgap which precludes the band-gap-photoexcitation.21 Upon Ni doping , the photocatalytic hydrogen evolution activity of T-Ni0.25 remains undetectable, indicating that Ni doping alone cannot confer TiO2 with visible-light responsiveness, which agrees with the DRS result. In contrast, T/10Au1−xAgx, T-Ni0.46/10Au1−xAgx, and T-Ni0.25/10Au1−xAgx display measurable hydrogen production, implying that Au1−xAgx NCs act as efficient photosensitizers, extending the photo-response range of TiO2 into the visible spectrum. The enhanced photocatalytic performance of T-Ni0.46/10Au1−xAgx compared with T/10Au1−xAgx strongly highlights the critical role of Ni dopants as a co-catalyst in accelerating the interfacial electron transfer, thereby improving the charge separation efficiency.9 Optimization of Au1−xAgx NC loading (Fig. 3b) revealed that T/10Au1−xAgx with 10 mL of BNCs exhibited the highest hydrogen evolution rate of 0.36 mmol g−1 h−1, reflecting the optimal balance between light absorption and charge transport.22 Further tuning of Ni doping levels (Fig. 3c) suggests that T-Ni0.25/10Au1−xAgx containing 0.25 wt% Ni and 10 mL of Au1−xAgx NCs achieved a significantly higher hydrogen evolution rate of 0.70 mmol g−1 h−1, establishing it as the optimal catalyst for subsequent systematic investigation.Inductively coupled plasma-mass spectrometry (ICP-MS) was employed to quantitatively determine the actual amount of Au1−xAgx NCs and Ni dopant in the T-Ni0.25/10Au1−xAgx heterostructure. As summarized in Table S4, the loading percentage of Au1−xAgx NCs and Ni dopant in the T-Ni0.25/10Au1−xAgx was approximately 3.83 and 0.09 wt%, respectively. This result signifies that even a small amount of Ni dopant surprisingly yielded a substantial enhancement in photoactivity, underscoring the critical role of Ni dopants as co-catalysts in facilitating the interfacial electron transfer. Monochromatic light irradiation tests (Fig. 3d) revealed that the photocatalytic hydrogen evolution performance of T-Ni0.25/10Au1−xAgx decreases with increasing wavelength in the visible range, with the highest activity observed under near-ultraviolet light (λ = 400–420 nm). This wavelength dependence aligns with the DRS results, confirming that while Au1−xAgx NCs broaden the light absorption of TiO2, the absorption efficiency remains primarily confined to the short-wavelength visible region. Light intensity measurements (Fig. 3e) show a linear increase in the photocatalytic hydrogen production rate with increasing light intensity, indicating that the photoreaction system operates under photon-limited conditions and possesses untapped potential for enhanced performance under higher irradiance, verifying it is indeed a photocatalytic reaction. Photocatalytic experiments using different sacrificial agents (Fig. 3f) demonstrate that TEOA outperformed other hole scavengers (methanol, ethanol, ethylene glycol, glycerol, lactic acid, and Na2S/Na2SO3), yielding the highest hydrogen evolution activity. This superiority is attributed to the suitable redox potential and strong hole-trapping ability of TEOA, facilitated by its tertiary amine group, which efficiently scavenges photogenerated holes and promotes sustained electron transfer to the catalyst surface, ultimately boosting the hydrogen generation.23 The chemical structure and redox properties of the sacrificial agent are critical determinants of catalytic performance, underscoring the importance of system-wide optimization in photocatalytic hydrogen production. As displayed in Fig. S13, cyclic stability tests indicate that photoactivity of the T-Ni0.25/10Au1−xAgx heterostructure exhibits apparent decay after the 2nd cycle. The relatively unfavorable cycling performance of the T-Ni0.25/10Au1−xAgx heterostructure is likely attributed to the vigorous stirring during continuous photocatalytic reactions, which causes the detachment of Au1−xAgx NCs from the TiO2 substrate, thereby weakening the visible-light absorption capacity of the composite and diminishing the number of active sites available for catalysis. More importantly, the intrinsically poor stability of metal NCs cannot be ignored, which leads to the gradual in situ transformation of metal NCs to conventional metal nanocrystals under continuous light irradiation, hence losing the advantageous photosensitization effect of metal NCs. Nevertheless, the significant enhancement in photocatalytic performance implies that the synergistic combination of Au1−xAgx NCs as photosensitizers and Ni dopants as a co-catalyst enables efficient visible-light-driven hydrogen evolution by expanding the spectral response of TiO2 and optimizing charge carrier dynamics. To further assess the generality of this strategy, a series of other alloy NC-based T-Ni0.25/[Au1−xCux, Au1−xPtx] heterostructures were prepared using analogous methodologies for visible-light-driven photocatalytic hydrogen production. The related characterization of T-Ni0.25/[Au1−xCux, Au1−xPtx] heterostructures is specifically provided in Fig. S9 and S10. As shown in Fig. 3g, the T-Ni0.25/Au1−xAgx heterostructure consistently exhibits the highest photoactivity among the tested systems under visible-light irradiation. Notably, all these alloy NC-modified samples (Fig. 3h–j) extend the light absorption spectrum of the TiO2 substrate into the visible region and demonstrate measurable photocatalytic activities, collectively highlighting the universal applicability of Au1−xAgx, Au1−xCux, and Au1−xPtx NCs as photosensitizers in this heterostructure design.
3.3 PEC and spectroscopic analysis
To elucidate the impact of BNC deposition and Ni doping on the interfacial charge separation efficiency of the T-Ni0.25/Au1−xAgx artificial photosystem, a comprehensive set of photoelectrochemical (PEC) analyses were performed on each photocatalyst. Transient photocurrent response is regarded as the most intuitive method for evaluating the carrier separation efficiency of photoelectrodes.24 Fig. 4a shows the periodic on/off transient photocurrent responses of pristine TiO2, T-Ni0.25, T/Au1−xAgx, and the T-Ni0.25/Au1−xAgx heterostructure under intermittent visible light irradiation (λ > 420 nm). Notably, the photocurrent density of T-Ni0.25/Au1−xAgx is significantly higher than that of pristine TiO2. Careful observation reveals that since TiO2 is a wide bandgap semiconductor, it cannot be photoexcited under visible light, and thus it generates almost no photocurrent. Moreover, after the introduction of the Ni co-catalyst, the increase in the photocurrent density of T-Ni0.25 is still relatively small since Ni doping fails to alter the light absorption of the TiO2 substrate, as unveiled by the DRS result. However, after loading the Au1−xAgx NCs, both T/Au1−xAgx and T-Ni0.25/Au1−xAgx generate apparent photocurrents under visible light, which strongly indicates that Au1−xAgx NCs serve as effective photosensitizers. The improved photocurrent of T-Ni0.25/Au1−xAgx compared with that of T/Au1−xAgx indicates that Ni doping can indeed accelerate the interfacial electron transport, thereby enhancing the carrier separation efficiency, which is consistent with the aforementioned photocatalytic performance analysis. To explore the charge recombination dynamics, open-circuit photovoltage decay (OCVD) results of the photoanodes were studied, which is regarded as an effective tool to monitor the open-circuit voltage (Voc) decay when the light is turned off in a stable state.25 As shown in Fig. 4b, TiO2 and T-Ni0.25 show almost no voltage attenuation owing to their large bandgaps and negligible visible light absorption, while T-Ni0.25/Au1−xAgx exhibits the maximum photovoltage, which once again confirms that the simultaneous introduction of Au1−xAgx and Ni doping leads to the most enhanced charge separation. Similarly, this result is fully consistent with the transient photocurrent response. Alternatively, Fig. 4c and Table S5 show the electrochemical impedance spectroscopy (EIS) results of the different samples under visible light irradiation. Among them, T-Ni0.25/Au1−xAgx shows the minimum semi-circular arc radius, indicating that Au1−xAgx NC loading and Ni doping effectively reduce the interfacial charge transfer resistance of the heterostructure. Therefore, the PEC results strongly validate that the significant improvement in the photoactivity of T-Ni0.25/Au1−xAgx is primarily attributed to the synergistic promotion of charge separation and migration afforded by Ni doping and Au1−xAgx NC photosensitization. According to the M–S results of TiO2, T-Ni0.25, T/Au1−xAgx and T-Ni0.25/Au1−xAgx (Fig. S14), T-Ni0.25/Au1−xAgx exhibits the largest charge carrier density (3.5 × 1019 cm−3), confirming the key role of Ni doping and Au1−xAgx decoration in accelerating the interfacial charge separation. Overall, the results of photocurrent, OCVD, EIS, M–S and ND analyses are consistent with the photocatalytic performances. In addition, according to the UV-Vis absorption spectra (Fig. S1e) and cyclic voltammetry (CV) results (Fig. S15), the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO) levels of Au1−xAgx NCs are determined to be approximately −2.235 V and 0.655 V vs. NHE (Fig. S18a), respectively. The HOMO and LUMO levels of Au1−xCux and Au1−xPtx NCs were also determined by analogous methods (Fig. S16–S18).To elucidate the photocatalytic mechanism, charge transport processes in TiO2, T-Ni0.25, T/Au1−xAgx and T-Ni0.25/Au1−xAgx were studied spectroscopically. Photoluminescence (PL) spectroscopy was employed to analyze the carrier recombination efficiency. As shown in Fig. 4e, steady-state PL spectra of the samples were recorded under 360 nm excitation. All the composite samples exhibit lower PL intensities compared with pure TiO2, indicating that Ni doping and Au1−xAgx NC photosensitization significantly suppress the electron–hole pair recombination. Among them, T-Ni0.25/Au1−xAgx shows the lowest PL intensity, signifying its maximally suppressed carrier recombination and optimal charge separation. Time-resolved photoluminescence (TRPL) decay curve spectroscopy was used to probe the carrier dynamics of the photosystems. The fitting results (Fig. 4f and Table S6) reveal that the average lifetime of the optimal sample T-Ni0.25/Au1−xAgx is 1.894 ns, compared with 1.598 ns for pristine TiO2. The extended average lifetime of T-Ni0.25/Au1−xAgx indicates a reduction in radiative recombination, suggesting an increased availability of carriers for the photocatalytic reaction. These results collectively demonstrate that concurrent Ni doping and Au1−xAgx NC loading synergistically inhibit the carrier recombination. Comparative analysis of the photoactivities, PEC performances and PL results (Fig. 4a–f) between T/Au1−xAgx and T-Ni0.25/Au1−xAgx further confirms that Ni doping enhances the overall performances of the samples. We speculate that the Ni dopant acts as an efficient electron transfer mediator, facilitating the unidirectional and smooth electron flow from Au1−xAgx NCs to the TiO2 substrate, thereby validating its role as a co-catalyst.
In situ XPS characterization, a powerful technique to analyze the surface chemistry and charge transport mechanisms of photocatalysts, was also performed.26 The changes in the binding energies of core elements directly reflect the variation in electron density. As illustrated in Fig. 4g–i, obvious shifts in the binding energies of Au 4f, Ag 3d, and Ni 2p for the T-Ni0.25/Au1−xAgx composite were observed under visible light irradiation compared with the dark-state conditions. These shifts verify the occurrence of photoelectron transfer upon light exposure, providing crucial evidence for determining the charge transfer pathways of the composite materials under visible light irradiation. Specifically, as TiO2 is inactive under visible light, photoelectrons generated in the Au1−xAgx NCs migrate to the TiO2 substrate, and the Ni dopant functioning as a co-catalyst accelerates the electron migration process. This is rationalized by the blue-shift in the binding energies of Ti 2p and O 1s for the T-Ni0.25/Au1−xAgx heterostructure (Fig. 4j–k). Noteworthily, the reduced binding energy of Ni 2p for the T-Ni0.25/Au1−xAgx heterostructure upon light irradiation also indicates the electron flow from Au1−xAgx NCs to the Ni dopant that acts as an interfacial electron transport mediator (Fig. 4i).
3.4 Photocatalytic mechanism
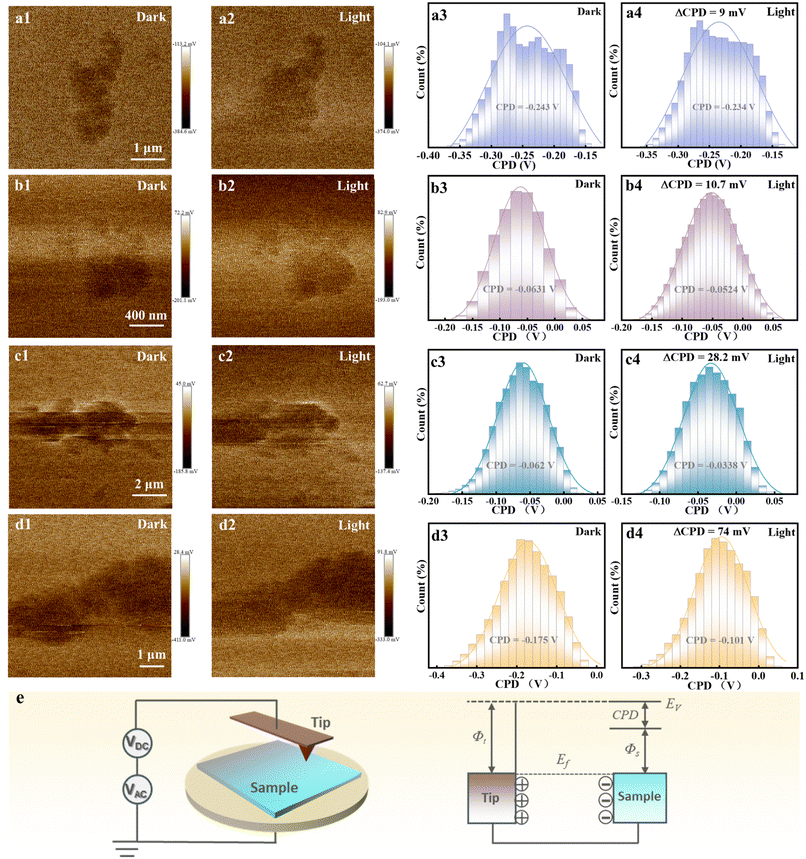
To deeply investigate the photogenerated charge separation behavior, Kelvin probe force microscopy (KPFM) was employed to obtain high-resolution nanoscale images of surface morphology and potential. KPFM operates based on the work function difference between the probe and the sample.27 Upon contact, electron flow equilibrates their Fermi levels, producing a contact potential difference (CPD) that depends on both the probe work function (Φt) and the sample work function (Φs), as shown in Fig. 5e.28 Thus, variations in CPD directly reveal the distribution and separation of charge carriers on the sample surface.29 Fig. S20 & Fig. 5 present the surface morphologies and potential images of all the samples. As shown in Fig. 5a1–d1, pristine TiO2, T-Ni0.25, T/Au1−xAgx and T-Ni0.25/Au1−xAgx exhibit non-uniform potential distribution across the scanned region, and differences in CPD before and after visible light irradiation (Fig. 5a2–d2) are clearly visible through color contrast in the potential maps. Under visible light illumination conditions, photogenerated electron–hole pairs separate, leading to an increase in CPD for all the samples. The corresponding ΔCPD values for pure TiO2, T-Ni0.25, T/Au1−xAgx and T-Ni0.25/Au1−xAgx are determined to be 9 mV, 10.7 mV, 28.2 mV, and 74 mV, respectively (Fig. 5a3d–3 and a4–d4). The positive ΔCPD indicates greater hole accumulation on the surface after light illumination, meaning the surface potential change is primarily governed by holes.30 Comparisons reveal that the ΔCPD changes of pure TiO2 and T-Ni0.25 are relatively small. In contrast, after depositing Au1−xAgx NCs, both T/Au1−xAgx and T-Ni0.25/Au1−xAgx exhibit significantly enhanced ΔCPD, with the latter showing the most pronounced increase. This suggests that T-Ni0.25/Au1−xAgx accumulates more photogenerated charges on the surface under visible light illumination.31 Additionally, the CPD line–scan profiles (Fig. S21a4–d4) further verify this trend, that is, T-Ni0.25/Au1−xAgx displays the largest CPD enhancement, strongly confirming its more efficient directional exciton transfer and improved spatial separation of electron–hole pairs. These findings align with the PEC results, jointly confirming the superior photogenerated charge separation and transfer capability of the T-Ni0.25/Au1−xAgx heterostructure.Elucidating the modulation of the electronic structure and atomic coordination environment of the doped Ni species in TiO2 is crucial for understanding the photocatalytic mechanism of the T-Ni0.25/Au1−xAgx heterostructure. Density functional theory (DFT) calculations were performed to determine the preferential location of Ni species in the TiO2 substrate. As shown in Fig. 6a, the configuration where Ni adsorbs on the TiO2 surface, forming direct Ni–O bonds, yields a binding energy of −4.20 eV. In contrast, the substitution of Ni at Ti sites in the TiO2 bulk results in a binding energy of only −0.05 eV. The significantly more negative binding energy for surface adsorption indicates that this configuration is energetically far more favorable. This computational conclusion is consistent with the EXAFS results below, which suggest the formation of Ni–O bonds on the TiO2 surface rather than replacement of Ti atoms with Ni.
The Ni doping of TiO2 by the MSM method, enabled by high-temperature conditions, facilitates Ni–O bond formation between freely mobile Ni2+ ions and surface oxygen ions of TiO2, as corroborated by extended X-ray absorption fine structure (EXAFS) and X-ray absorption near-edge structure (XANES) spectroscopy (Fig. 6a and b). As shown in Fig, 6b, the absorption edge of TiO2–Ni is positioned closer to that of NiO than to metallic Ni foil, indicating that the valence state of Ni in TiO2–Ni lies between +1 and +2, closely approaching the +2 state.32 Ni K-edge R-space EXAFS analysis (Fig, 6c) of TiO2–Ni reveals two characteristic peaks at 1.6 Å and 2.6 Å which are analogous to those of NiO, corresponding to the Ni–O and Ni–Ni coordinate ion bonds, respectively.33 As shown in Table S7, the fitting results of the EXAFS data are specifically summarized. Notably, the absence of the Ni–Ni bonding peak of metallic Ni foil within the TiO2–Ni spectrum indicates that Ni species are dispersed rather than forming metallic atoms, strongly suggesting their existence in an oxidized state.34 Additionally, wavelet transform (WT) analysis of the EXAFS spectra was performed to verify the atomic dispersion (Fig. 6e–g). In comparison with the WT profiles of Ni foil and NiO, TiO2–Ni exhibits two obvious coordination signals at approximately 5 Å−1 and 9 Å−1, corresponding to Ni–O and Ni–Ni coordination, respectively. This indicates that the Ni species on TiO2–Ni are in an oxidized state, which is consistent with the EXAFS results. Collectively, these results provide unambiguous evidence for the successful doping of Ni onto the TiO2 surface, with the formation of Ni–O surface linkages critical for modulating the electronic structure of the semiconductor.
Based on the M–S results (Fig. 4d), the conduction band (CB) potential (ECB) of TiO2 was determined to be −0.76 V relative to NHE (−0.34 V relative to RHE), and considering that the Efb of an n-type semiconductor is more positive by 0.1 V than the ECB, ECB of TiO2 is calibrated to be −0.86 V vs. NHE. Given that the bandgap of TiO2 determined from the DRS results is 3.00 eV (Fig. 1d), the valence band (VB) level of TiO2 is thus calculated to be 2.14 eV relative to NHE. Based on the experimental analyses, a photocatalytic mechanism is proposed, as illustrated in Fig. 6h. When the T-Ni0.25/Au1−xAgx heterostructure is irradiated with visible light, Au1−xAgx NCs undergo photoexcitation, inducing electron transitions from the HOMO to the LUMO level. Owing to the favorable energy level alignment between the LUMO level of Au1−xAgx NCs and the CB of TiO2, photoexcited electrons in the LUMO level of Au1−xAgx NCs rapidly inject into the CB of the TiO2 substrate, establishing a direct charge transfer pathway. The Ni species doped on the TiO2 surface function as a co-catalyst via strong Ni–O chemical bonding which plays a dual role by lowering the energy barrier for electron transfer from Au1−xAgx NCs to TiO2 through modulating the interfacial electronic structure and constructing a cascaded electron transport pathway, thereby accelerating the directional charge migration. This Ni-mediated synergy significantly suppresses the electron–hole recombination and enhances the separation efficiency of photogenerated charge carriers over Au1−xAgx NCs.9,35 Subsequently, the transferred electrons accumulate at the TiO2 surface and then react with protons (H+) adsorbed at the active sites on the TiO2 surface to drive the hydrogen evolution reaction. The discrete energy levels of BNCs combined with the uniform dispersion of Ni co-catalysts, create a robust “light absorption–charge separation–proton reduction” cascade charge transfer channel, enabling efficient conversion of solar energy into chemical energy. The synergy between molecular-like BNCs as light-harvesting units and Ni co-catalysts as charge-accelerating nodes represents a rational design strategy for constructing high-performance heterostructure photocatalysts, providing a foundational framework for advancing solar-to-hydrogen conversion technologies.
The TiO2–Ni/alloy NCs demonstrate a more superior photocatalytic mechanism compared to traditional semiconductor photocatalysts and metal nanoparticles. Conventional semiconductor photocatalysts generate electron–hole pairs via interband transitions but are often constrained by limited visible-light response and rapid carrier recombination.36 Metal nanoparticles can contribute via plasmonic effects or hot-electron injection, yet their function is typically confined to isolated steps.37 In contrast, metal NCs possess a discrete electronic structure with both molecule-like and nanomaterial-like characteristics, enabling more versatile light absorption and charge transfer pathways. Furthermore, BNCs allow compositional tuning to optimize energy-level alignment with the semiconductor. In this work, BNCs form a favorable cascade alignment with TiO2–Ni, promoting efficient electron transition, suppressing charge recombination, and prolonging excited-state carrier lifetimes.
4. Conclusions
To sum up, TiO2–Ni/alloy NC (Au1−xAgx, Au1−xPtx, and Au1−xCux) heterostructures were fabricated by a facile and universal molten salt method combined with an electrostatic self-assembly strategy. The results confirm that BNC (Au1−xAgx, Au1−xPtx, and Au1−xCux) decoration extends the light absorption of TiO2 into the visible region; meanwhile Ni doping accelerates the interfacial charge transport, thus synergistically boosting the electron separation from BNCs to TiO2. This synergy significantly enhances the visible-light-driven photocatalytic hydrogen evolution performances of TiO2–Ni/alloy NC (Au1−xAgx, Au1−xPtx, and Au1−xCux) heterostructures by virtue of their favorable energy level alignment. Furthermore, the charge transfer mechanism of TiO2–Ni/alloy NC artificial photosystems was unraveled, revealing a synergistic effect between atomic-scale metal NCs and metal oxide supports.Author contributions
Zi-Han Zheng performed the experiments, analyzed all the data, and drafted the manuscript. Qing Chen and Peng Su carried out and analyzed a portion of the experiments. Fang-Xing Xiao guided this work and corrected the manuscript. Lifeng Cai, Jie Liang, and Guangcan Xiao helped to check the manuscript. All the authors contributed to critical discussion of the data and the manuscript.Conflicts of interest
The authors declare no competing interests.Data availability
All data supporting the findings of this study are available within the paper and its supplementary information (SI) files. Source data are provided with this paper. Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc02262f.Acknowledgements
The support from the Award Program for Minjiang scholar professorship is greatly acknowledged. This work was financially supported by the National Natural Science Foundation of China (No. 21703038 and 22072025). The financial support from the State Key Laboratory of Structural Chemistry, Fujian Institute of Research on the Structure of Matter, Chinese Academy of Sciences (No. 20240018), and the Natural Science Foundation of Fujian Province (2024J01263) is acknowledged.References
- (a) R. C. Jin, C. J. Zeng, M. Zhou and Y. X. Chen, Chem. Rev., 2016, 116, 10346–13413 CrossRef CAS PubMed; (b) I. Chakraborty and T. Pradeep, Chem. Rev., 2017, 117, 8208–8271 CrossRef CAS PubMed; (c) X. Kang, Y. W. Li, M. Z. Zhu and R. C. Jin, Chem. Soc. Rev., 2020, 49, 6443–6514 RSC; (d) L. C. Liu and A. Corma, Chem. Rev., 2018, 118, 4981–5079 CrossRef CAS PubMed; (e) Y. X. Du, H. T. Sheng, D. Astruc and M. Z. Zhu, Chem. Rev., 2020, 120, 526–622 CrossRef CAS PubMed.
- (a) Q. L. Mo, B. J. Liu and F. X. Xiao, J. Phys. Chem. C, 2021, 125, 22421–22428 Search PubMed; (b) L. P. Zhang, J. R. Ran, S. Z. Qiao and M. Jaroniec, Chem. Soc. Rev., 2019, 48, 5184–5206 Search PubMed.
- (a) X. Y. Fu, Z. Q. Wei, S. Xu, X. Lin, S. Hou and F. X. Xiao, J. Phys. Chem. Lett., 2020, 11, 9138–9143 CrossRef CAS PubMed; (b) H. Liang, B. J. Liu, B. Tang, S. C. Zhu, S. Li, X. Z. Ge, J. L. Li, J. R. Zhu and F. X. Xiao, ACS Catal., 2022, 12, 4216–4226 CrossRef CAS; (c) H. Wang, F. Chen, W. Li and T. Tian, J. Power Sources, 2015, 287, 150–157 CrossRef CAS.
- (a) Y. Z. Bao, X. H. Wu, H. J. Gao, M. M. Zhou, S. Chen, S. Jin, H. Z. Yu and M. Z. Zhu, Dalton Trans., 2020, 49, 17164–17168 Search PubMed; (b) Y. Du, Z. J. Guan, Z. R. Wen, Y. M. Lin and Q. M. Wang, Chem. - Eur. J., 2018, 24, 16029–16035 Search PubMed; (c) A. G. Walsh and P. Zhang, J. Phys. Chem. Lett., 2021, 12, 257–275 Search PubMed; (d) X. Q. Ren, X. M. Fu, X. Z. Lin, J. Tang, H. Wang, C. Liu and J. H. Huang, Eur. J. Inorg. Chem., 2021, 2021, 2281–2283 Search PubMed; (e) X. Kang and M. Z. Zhu, Small, 2019, 15, e1902703 Search PubMed; (f) C. Zhu, Z. L. Chen, H. Li, L. Y. Lu, X. Kang, J. Xuan and M. Z. Zhu, J. Am. Chem. Soc., 2024, 146, 23212–23220 CrossRef CAS PubMed; (g) M. Kamachi, K. Yonesato, T. Okazaki, D. Yanai, S. Kikkawa, S. Yamazoe, R. Ishikawa, N. Shibata, Y. Ikuhara, K. Yamaguchi and K. Suzuki, Angew. Chem., Int. Ed., 2024, 63, e202408358 CrossRef CAS PubMed; (h) X. J. Zou, S. P. He, X. Kang, S. Chen, H. Z. Yu, S. Jin, D. Astruc and M. Z. Zhu, Chem. Sci., 2021, 12, 3660–3667 RSC; (i) E. H. Kim, M. H. Lee, J. Kim, E. C. Ra, J. H. Lee and J. S. Lee, Chin. J. Catal., 2023, 47, 214–221 Search PubMed.
- (a) L. Ji, D. Spanu, N. Denisov, S. Recchia, P. Schmuki and M. Altomare, Chem.–Asian J., 2020, 15, 301–309 Search PubMed; (b) Z. H. N. Al-Azri, W.-T. Chen, A. Chan, V. Jovic, T. Ina, H. Idriss and G. I. N. Waterhouse, J. Catal., 2015, 329, 355–367 Search PubMed; (c) Y. Liu, Y. Li, F. Peng, Y. Lin, S. Yang, S. Zhang, H. Wang, Y. Cao and H. Yu, Appl. Catal. B Environ., 2019, 241, 236–245 CrossRef CAS.
- (a) R. Shen, J. Xie, Q. Xiang, X. Chen, J. Jiang and X. Li, Chin. J. Catal., 2019, 40, 240–288 CrossRef CAS; (b) S. M. Thabet, H. N. Abdelhamid, S. A. Ibrahim and H. M. El-Bery, Sci. Rep., 2024, 14, 10115 CrossRef CAS PubMed; (c) J. S. Schubert, J. Popovic, G. M. Haselmann, S. P. Nandan, J. Wang, A. Giesriegl, A. S. Cherevan and D. Eder, J. Mater. Chem. A, 2019, 7, 18568–18579 RSC; (d) P. D. Tran, L. Xi, S. K. Batabyal, L. H. Wong, J. Barber and J. S. Chye Loo, Phys. Chem. Chem. Phys., 2012, 14, 11596–11599 Search PubMed; (e) X. Zhao, W. Xie, X. Shao, Z. Wang, B. Yang, C. Yang, J. Wang and X. Su, Mater. Sci. Semicond. Process., 2022, 148, 106775 CrossRef CAS.
- (a) M. A. Nazir, T. Najam, M. Altaf, K. Ahmad, I. Hossain, M. A. Assiri, M. S. Javed, A. u. Rehman and S. S. A. Shah, J. Alloys Compd., 2024, 990, 174378 Search PubMed; (b) Z. Fu, X. Ma, B. Xia, X. Hu, J. Fan and E. Liu, Int. J. Hydrogen Energy, 2021, 46, 19373–19384 CrossRef CAS; (c) M. Zhai, Z. Li, R. Li, Z. Feng, Q. Wang, C. Zhang, H. Chi, N. Ta and C. Li, J. Catal., 2025, 443, 115989 Search PubMed; (d) J. J. Zhao, P. F. Liu, Y. L. Wang, Y. H. Li, M. Y. Zu, C. W. Wang, X. L. Wang, L. J. Fang, H. D. Zeng and H. G. Yang, Chem. - Eur. J., 2017, 23, 16734–16737 CrossRef CAS PubMed; (e) J. Zhang, Z. Yu, Z. Gao, H. Ge, S. Zhao, C. Chen, S. Chen, X. Tong, M. Wang, Z. Zheng and Y. Qin, Angew. Chem., Int. Ed., 2016, 56, 816–820 Search PubMed.
- J. Shen, C. Luo, S. Qiao, Y. Chen, K. Fu, J. Xu, J. Pei, Y. Tang, X. Zhang, H. Tang, H. Zhang and C. Liu, Adv. Funct. Mater., 2023, 34, 2309056 Search PubMed.
- M. Xiao, L. Zhang, B. Luo, M. Lyu, Z. Wang, H. Huang, S. Wang, A. Du and L. Wang, Angew. Chem., Int. Ed., 2020, 59, 7230–7234 CrossRef CAS PubMed.
- D. Hikosou, S. Saita, S. Miyata, H. Miyaji, T. Furuike, H. Tamura and H. Kawasaki, J. Phys. Chem. C, 2018, 122, 12494–12501 CrossRef CAS.
- X. Liu, N. Fechler and M. Antonietti, Chem. Soc. Rev., 2013, 42, 8237–8265 RSC.
- (a) J. Ryu, J. H. Seo, G. Song, K. Choi, D. Hong, C. Wang, H. Lee, J. H. Lee and S. Park, Nat. Commun., 2019, 10, 2351 CrossRef PubMed; (b) Y. Yuan, W. Xiao, Z. Wang, D. J. Fray and X. Jin, Angew. Chem., Int. Ed., 2018, 57, 15743–15748 CrossRef CAS PubMed.
- (a) J. P. Holmberg, E. Ahlberg, J. Bergenholtz, M. Hassellöv and Z. Abbas, J. Colloid Interface Sci., 2013, 407, 168–176 CrossRef CAS PubMed; (b) A. M. Jastrzębska, P. Kurtycz, A. Olszyna, E. Karwowska, E. Miaśkiewicz-Pęska, M. Załęska-Radziwiłł, N. Doskocz and D. Basiak, Int. J. Appl. Ceram. Technol., 2014, 12, 1157–1173 CrossRef.
- (a) L. Yang, X. Jiang, W. Ruan, B. Zhao, W. Xu and J. R. Lombardi, J. Phys. Chem. C, 2008, 112, 20095–20098 CrossRef CAS; (b) X. Li, Y. Wu, Y. Shen, Y. Sun, Y. Yang and A. Xie, Appl. Surf. Sci., 2018, 427, 739–744 CrossRef CAS.
- Z. Y. Li, Y. H. Chen, J. R. Zhu, Q. Chen, S. J. Lu and F. X. Xiao, Inorg. Chem., 2023, 62, 16965–16973 CrossRef CAS PubMed.
- K. Kuroi, M. Yamada, I. Kawamura, M. Jung, C.-G. Pack and F. Fujii, Phys. Chem. Chem. Phys., 2022, 24, 13356–13364 RSC.
- A. Li, S. Chen, F. Yang, H. Gao, C. Dong and G. Wang, Inorg. Chem., 2021, 60, 18337–18346 CrossRef CAS PubMed.
- (a) H. Zhuang, W. Chen, W. Xu and X. Liu, Int. J. Energy Res., 2020, 44, 3224–3230 CrossRef CAS; (b) Y. Yang, X. Li, C. Lu and W. Huang, Catal. Lett., 2019, 149, 2930–2939 CrossRef CAS.
- (a) L. Ma, R. Guan, W. Kang, Z. Sun, H. Li, Q. Li, Q. Shen, C. Chen, X. Liu, H. Jia and J. Xue, J. Colloid Interface Sci., 2024, 660, 381–392 CrossRef CAS PubMed; (b) X. Shi, L. Mao, C. Dai, P. Yang, J. Zhang, F. Dong, L. Zheng, M. Fujitsuka and H. Zheng, J. Mater. Chem. A, 2020, 8, 13376–13384 RSC.
- G. Wu, Q. L. Mo, Y. Xiao, K. Wang, X. Z. Ge, S. R. Xu, J. L. Li, Y. Q. Shao and F. X. Xiao, Inorg. Chem., 2023, 62, 520–529 CrossRef CAS PubMed.
- Y. Wang, X. H. Liu, Q. Wang, M. Quick, S. A. Kovalenko, Q. Y. Chen, N. Koch and N. Pinna, Angew. Chem., Int. Ed., 2020, 59, 7748–7754 CrossRef CAS PubMed.
- K. Wang, X.-Z. Ge, Q.-L. Mo, X. Yan, Y. Xiao, G. Wu, S.-R. Xu, J.-L. Li, Z.-X. Chen and F.-X. Xiao, J. Catal., 2022, 416, 92–102 CrossRef CAS.
- (a) G. Malekshoar and A. K. Ray, Chem. Eng. Sci., 2016, 152, 35–44 CrossRef CAS; (b) W. Jones, D. J. Martin, A. Caravaca, A. M. Beale, M. Bowker, T. Maschmeyer, G. Hartley and A. Masters, Appl. Catal. B Environ., 2019, 240, 373–379 CrossRef CAS.
- J.-N. Yuan, X. Yan, B.-X. Zheng, J.-Q. Chen and F.-X. Xiao, Inorg. Chem. Front., 2025, 12, 1553–1567 RSC.
- P. Su, S. Li and F. X. Xiao, Small, 2024, 20, 2400958 CrossRef CAS PubMed.
- L. Wang, Y. Li, Y. Ai, E. Fan, F. Zhang, W. Zhang, G. Shao and P. Zhang, Adv. Funct. Mater., 2023, 33, 2306466 CrossRef CAS.
- W. Melitz, J. Shen, A. C. Kummel and S. Lee, Surf. Sci. Rep., 2011, 66, 1–27 CrossRef CAS.
- (a) J. Cheng, S. Wan and S. Cao, Angew. Chem., Int. Ed., 2023, 62, e202310476 CrossRef CAS PubMed; (b) J. Cheng, W. Wang, J. Zhang, S. Wan, B. Cheng, J. Yu and S. Cao, Angew. Chem., Int. Ed., 2024, 63, e202406310 CrossRef CAS PubMed.
- R. Li, H. Li, X. Zhang, B. Liu, B. Wu, B. Zhu, J. Yu, G. Liu, L. Zheng and Q. Zeng, Adv. Funct. Mater., 2024, 34, 2402797 CrossRef CAS.
- (a) C. Cheng, B. He, J. Fan, B. Cheng, S. Cao and J. Yu, Adv. Mater., 2021, 33, 2100317 CrossRef CAS PubMed; (b) X. Zhang, J. Xu, H. Long, J. Yu and H. Yu, ACS Catal., 2024, 14, 18669–18678 CrossRef CAS.
- (a) C. Yang, S. Wan, B. Zhu, J. Yu and S. Cao, Angew. Chem., Int. Ed., 2022, 61, e202208438 CrossRef CAS PubMed; (b) Y. Tang, Z.-F. Xu, Y. Sun, C. Wang, Y. Guo, W. Hao, X. Tan, J. Ye and T. Yu, Energy Environ. Sci., 2024, 17, 7882–7894 RSC.
- (a) W. Zhong, C. Yang, J. Wu, W. Xu, R. Zhao, H. Xiang, K. Shen, Q. Zhang and X. Li, Chem. Eng. J., 2022, 436, 134813 CrossRef CAS; (b) T. Chen, F. Wang, S. Cao, Y. Bai, S. Zheng, W. Li, S. Zhang, S. X. Hu and H. Pang, Adv. Mater., 2022, 34, 2201779 CrossRef CAS PubMed.
- M. Liu, Y. Ji, Y. Li, P. An, J. Zhang, J. Yan and S. Liu, Small, 2021, 17, 2102448 CrossRef CAS PubMed.
- B. He, P. Xiao, S. Wan, J. Zhang, T. Chen, L. Zhang and J. Yu, Angew. Chem., Int. Ed., 2023, 62, e202313172 CrossRef CAS PubMed.
- J. Yang, Z. Qiu, C. Zhao, W. Wei, W. Chen, Z. Li, Y. Qu, J. Dong, J. Luo, Z. Li and Y. Wu, Angew. Chem., Int. Ed., 2018, 57, 14095–14100 CrossRef CAS PubMed.
- (a) J. Deng, X. Xu, B. Su, M. Liu, X. Lin, W. Xing, X. F. Lu, Z. Lan, G. Zhang and S. Wang, Mater. Horiz., 2025, 12, 5702–5709 RSC; (b) Z. Wang, B. Su, J. Xu, Y. Hou and Z. Ding, Int. J. Hydrogen Energy, 2020, 45, 4113–4121 CrossRef CAS.
- (a) S. Sarina, H.-Y. Zhu, Q. Xiao, E. Jaatinen, J. Jia, Y. Huang, Z. Zheng and H. Wu, Angew. Chem., Int. Ed., 2014, 53, 2935–2940 CrossRef CAS PubMed; (b) B. Su, K. Tang, J. Cai, X. Lin, W. Xing, K. Liu, X. F. Lu, Y. Hou, W.-J. Ong and S. Wang, ACS Catal., 2026, 16, 5208–5217 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |