Open Access Article
Open Access ArticleSynthesis, characterization, and imidogen photochemistry of a hydrazoic acid adduct of Rh2
Arpan Paikar ,
Phong Thai
,
Phong Thai ,
Matthew T. Figgins
,
Matthew T. Figgins ,
Bodhisattwa Mandal
,
Bodhisattwa Mandal ,
Matthew J. Lekas
,
Matthew J. Lekas ,
Joseph H. Reibenspies
,
Joseph H. Reibenspies ,
Gerard P. Van Trieste
,
Gerard P. Van Trieste and
David C. Powers
and
David C. Powers *
*
Department of Chemistry, Texas A&M University, College Station, Texas 77843, USA. E-mail: powers@chem.tamu.edu
First published on 28th April 2026
Abstract
Metal–imidogen (i.e., M–NH) intermediates are proposed in metal-catalyzed NH-transfer reactions. Transition metal complexes of hydrazoic acid (HN3) could serve as precursors to these transient intermediates; however, the coordination chemistry of HN3 is essentially unknown. Here, we report the synthesis and characterization of Rh2(esp)2(HN3)2, the first crystalline transition metal complex of HN3 (esp = α,α,α′,α′-tetramethyl-1,3-benzenedipropionate). Temperature-dependent multi-nuclear NMR, UV-vis, and IR spectroscopies demonstrate that HN3 is a weakly coordinating ligand (Keq = 1100 ± 100 M−2 at 243 K). Cryogenic photolysis (77 K) enabled observation of a non-steady state intermediate, which we assign to be a triplet Rh2–NH complex, that engages in olefin aziridination chemistry. The nitrene photochemistry of Rh2(esp)2(HN3)2 was corroborated by in crystallo synthesis and cryogenic spectroscopic characterization of a family of N-aryl nitrene complexes. Together, these results establish the coordination chemistry of HN3, confirm the triplet ground state of the Rh2–NH intermediate responsible for NH transfer, and demonstrate a photochemical platform to observe intermediates in NH-transfer catalysis.
Introduction
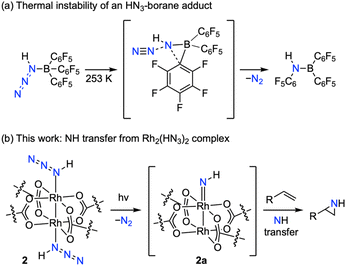
Imidogen (i.e., NH) is the simplest nitrene and cryogenic matrix isolation has established its triplet ground state (i.e., 3[NH]).1–4 Conceptually, imidogen transfer to C–H bonds and olefins would streamline access to primary amines and N–H aziridines, respectively.5–9 In the absence of a catalyst, NH transfer is rarely observed: even at low temperature (∼20 K), imidogen dimerizes to generate N2H2 (ref. 10 and 11) and reacts unselectively with olefinic substrates to yield mixtures of HCN, N2, and alkyl nitriles (Fig. 1a).12,13 Significant progress has been made towards the development of Rh2-,14,15 Co-,16 and Fe-catalyzed5,17,18 NH-transfer reactions, although at present, optimization of these reactions is empirically guided because the putative M–NH intermediates have eluded experimental observation.Hydrazoic acid (HN3)—the simplest molecular azide—is an explosive, toxic, and volatile liquid (b.p. = 310 K (37 °C)).19 HN3 is monomeric in the gas phase10,20–23 and displays a tetrameric structure in the solid state that persists up to its melting point (190(5) K).24,25 HN3 can serve as a precursor to imidogen via photochemical N2 extrusion.22,26–28 Application of HN3 photochemistry to the preparation of reactive M–NH complexes is currently stymied by the lack of HN3 coordination chemistry. In 1970, Schmidt suggested that reaction of HN3 with SbCl5—which ultimately affords NH4SbCl6, [SbCl4(N3)]2, and N2—proceeds through an unobserved HN3·SbCl5 adduct.29 Oxidative addition of HN3 with vanadium, chromium, and digermanium complexes have been observed and suggested to proceed via unobserved HN3 adducts.30–32 In 2019, Schulz reported the first—and thus far only—example of a crystalline HN3 coordination compound, HN3·B(C6F5)3 (Fig. 2a).33 Above 253 K (−20 °C), N2 loss is facile and results in aminoborane formation. The complete lack of transition metal complexes of HN3 prevents experimental evaluation of potential imidogen transfer chemistry from these platforms.
Rh2(esp)2 (1) catalyzes NH-transfer from hydroxylamine derivatives to olefinic substrates and Rh2(II,II)–NH intermediates have been proposed.14,15 Computational studies have suggested the proposed Rh2(II,II)–NH adopts a triplet ground state.34,35 Rh2(II,III) intermediates have been suggested as alternative intermediates,36–38 in part based on the facility of one-electron hydroxylamine redox chemistry.39 To investigate the putative Rh2–imidogen intermediates and establish photochemical access to M–NH species, here, we report the synthesis and characterization of the first crystalline transition metal complex of hydrazoic acid, Rh2(esp)2(HN3)2 (2) (Fig. 2b). Complex 2 is prepared by treatment of 1 with HN3 and is characterized by single-crystal X-ray diffraction (SCXRD), 1H, 13C, and 15N NMR, IR, and UV-vis spectroscopies. Photolysis of 2 in the presence of olefinic substrates promotes stereospecific NH transfer and cryogenic photolysis suggests the intermediacy of a transient triplet Rh2 imidogen adduct (i.e., 3[Rh2–NH]). The obtained spectroscopic data were further corroborated by in crystallo synthesis and spectroscopic characterization of a family of Rh2 N-aryl nitrene complexes. These findings indicate the chemical and kinetic competence of 3[Rh2–NH] intermediates in NH-transfer catalysis, detail the first spectroscopic characterization of Rh2 nitrenes, and introduce new opportunities in the synthesis of reactive M–NH intermediates.
Results and discussion
Synthesis and characterization
The solid-state IR spectrum of 2 (ATR) features a broad N–H peak at 3212 cm−1 and two peaks at 2143 cm−1 and 1176 cm−1 corresponding to the asymmetric and symmetric azide stretches, respectively (Fig. 3b). For comparison, we also prepared 2H- and 15N-labeled 2 (i.e., [2H]-2 and [15N]-2) by vacuum transfer of DN3 and H15NN2, respectively. [15N]-2 displays solid-state IR features that are slightly red-shifted compared to 2 at 3196 cm−1, 2119 cm−1, and 1166 cm−1 (Fig. 3b). [2H]-2 displays an N–D stretch at 2388 cm−1 (compared with an N–H stretch at 3112 cm−1 for 2) and no significant perturbation of the azide stretching modes.
The 1H NMR spectrum of 2 displays diamagnetically shifted peaks that integrate as expected for 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 coordination of HN3 to 1. The N–H chemical shift of 2 is temperature and concentration dependent (vide infra), but in general is sharper and shifted downfield compared to HN3 (Fig. 3c). For example, the N–H resonance of 2 is at 4.95 ppm (FWHM = 23.9 Hz) in CD2Cl2 at 298 K; the N–H resonance of HN3 is at 4.52 ppm (FWHM = 61.1 Hz). The N–H resonance is also solvent dependent: the N–H chemical shift of 2 is at 4.95 ppm in CD2Cl2, 3.6 ppm in toluene-d8 (Fig. S3), and 3.2 ppm in the solid state (measured via magic angle spinning (MAS) solid-state (SS) [2H]-NMR of [2H]-2 (Fig. S4)). Finally, the 1H spectrum of [15N]-2 displays a doublet for the N–H peak (JN–H = 69.0 Hz) (Fig. 3c). The 1H-coupled 15N NMR spectrum of [15N]-2 displays two peaks: a singlet at −171.2 ppm and a doublet at −325.2 ppm (JH–N = 69.7 Hz), which we assign as γ-N and α-N, respectively (Fig. 3d). The MAS-SS [15N]-NMR of [15N]-2 displays analogous features at −159.1 and −336.7 ppm (Fig. S5).
1 coordination of HN3 to 1. The N–H chemical shift of 2 is temperature and concentration dependent (vide infra), but in general is sharper and shifted downfield compared to HN3 (Fig. 3c). For example, the N–H resonance of 2 is at 4.95 ppm (FWHM = 23.9 Hz) in CD2Cl2 at 298 K; the N–H resonance of HN3 is at 4.52 ppm (FWHM = 61.1 Hz). The N–H resonance is also solvent dependent: the N–H chemical shift of 2 is at 4.95 ppm in CD2Cl2, 3.6 ppm in toluene-d8 (Fig. S3), and 3.2 ppm in the solid state (measured via magic angle spinning (MAS) solid-state (SS) [2H]-NMR of [2H]-2 (Fig. S4)). Finally, the 1H spectrum of [15N]-2 displays a doublet for the N–H peak (JN–H = 69.0 Hz) (Fig. 3c). The 1H-coupled 15N NMR spectrum of [15N]-2 displays two peaks: a singlet at −171.2 ppm and a doublet at −325.2 ppm (JH–N = 69.7 Hz), which we assign as γ-N and α-N, respectively (Fig. 3d). The MAS-SS [15N]-NMR of [15N]-2 displays analogous features at −159.1 and −336.7 ppm (Fig. S5).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 coordination of the aryl azide ligands to 1; during solvent removal from 3c, partial loss of the apical azides was evident by integration of the 1H NMR spectrum. The solid-state IR spectra display azide stretches at ∼2100 and ∼2130 cm−1 for 3a–3c (Fig. S6).43
1 coordination of the aryl azide ligands to 1; during solvent removal from 3c, partial loss of the apical azides was evident by integration of the 1H NMR spectrum. The solid-state IR spectra display azide stretches at ∼2100 and ∼2130 cm−1 for 3a–3c (Fig. S6).43
Ligand binding thermodynamics
The HN3 ligands of 2 are labile in solution. The N–H chemical shift in the 1H NMR of 2 is temperature (and concentration) dependent and shifts downfield upon cooling from 298 K to 203 K (Fig. 5a). Similar temperature dependence is reflected in VT-15N NMR experiments (Fig. S7). VT-IR spectroscopy similarly indicates facile equilibration of 2 and 1 + HN3 (Fig. 5b): a CH2Cl2 solution of 2 displays two azide stretching frequencies at 273 K—at 2138 and 2151 cm−1—which we assign to HN3 and 2, respectively (in the solid-state the azide stretches for 2 appears at 2143 cm−1).44,45 Upon cooling, the relative intensity of these spectral features changes, with the free HN3 stretch decreasing in intensity while the coordinated HN3 stretch increases in intensity. Similar temperature dependence is noted in analysis of the symmetric vibrational mode and in VT-UV-vis measurements (Fig. S8 and S9).To evaluate the thermodynamics of HN3 binding, we carried out an NMR titration of HN3 with 1 at 243 K (−30 °C). The N–H resonance of HN3 is systematically shifted downfield with increasing [1] (Fig. S10). Using equations derived analogously to those of Drago,46 these data enabled determination of temperature-dependent equilibrium constants (Keq): Keq = 1100 ± 100 M−2 at 243 K; Keq = 120 M−2 at 298 K. Van't Hoff analysis (Fig. S11), based on the obtained temperature-dependent equilibrium constants, provided ΔH0 = −5.5 ± 0.1 kcal mol−1 and ΔS0 = −9.0 ± 0.6 cal K−1 mol−1. The Keq for HN3 binding is similar to that determined by Albrecht for the coordination of AdN3 to Fe(HMDS)2 (62 M−1 at 298 K) (Ad = adamantyl).44
We similarly carried out an NMR titration of PhN3 with 1 (Fig. S12) to evaluate the thermodynamics of aryl azide binding. The obtained data provided Keq = 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ± 3000 M−2 at 243 K (Keq = 2700 M−2 at 298 K), which is slightly larger than that measured for HN3 (for VT-NMR of compound 3a, see Fig. S13). The relative binding affinities are consistent with enhanced Lewis basicity of Ph–N–N2 as compared with H–N–N2.47 Van't Hoff analysis, based on Keq measurements from 298 K to 203 K provided ΔH0 = −4.23 ± 0.06 kcal mol−1 and ΔS0 = 1.5 ± 0.2 cal K−1 mol−1 (Fig. S14). Temperature-dependent UV-vis spectroscopic data of 3a–c are collected in Fig. S15. In comparison to common nitrogen donors, such as pyridine and acetonitrile (Keq > 103 for binding to Rh2), HN3 and aryl azides are weakly binding ligands.48–51
000 ± 3000 M−2 at 243 K (Keq = 2700 M−2 at 298 K), which is slightly larger than that measured for HN3 (for VT-NMR of compound 3a, see Fig. S13). The relative binding affinities are consistent with enhanced Lewis basicity of Ph–N–N2 as compared with H–N–N2.47 Van't Hoff analysis, based on Keq measurements from 298 K to 203 K provided ΔH0 = −4.23 ± 0.06 kcal mol−1 and ΔS0 = 1.5 ± 0.2 cal K−1 mol−1 (Fig. S14). Temperature-dependent UV-vis spectroscopic data of 3a–c are collected in Fig. S15. In comparison to common nitrogen donors, such as pyridine and acetonitrile (Keq > 103 for binding to Rh2), HN3 and aryl azides are weakly binding ligands.48–51
NH-transfer photochemistry
To chemically evaluate the competence of photochemically generated intermediates to engage in N–H transfer, we pursued the study of the NH-transfer photochemistry of 2 with olefinic substrates. Photolysis (λ = 300 nm, 243 K, quartz reaction vessel) of a CD2Cl2 solution of 2 in the presence of 1-octene resulted in NH transfer to afford the corresponding N–H aziridine (4) in 39% yield (Fig. 6); compound 1 is observed as the Rh-containing byproduct by 1H NMR (for 1H and ESI-MS analysis see Fig. S16 and S17). Similarly, photolysis of [15N]-2 in the presence of 1-octene affords a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 4
1 mixture of 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) [15N]-4, which is expected from a mono-labeled [15N]-HN3 precursor (Fig. S18 and S19). In this experiment, [15N]-N2 was also observed by 15N-NMR (−70.3 ppm),52 which demonstrates the facility of N2 photoelimination under these reaction conditions (Fig. S20). NH transfer to 1-octene was also observed for photolysis of a toluene solution, albeit in lower yield (12%), which we attribute to limited solubility of 2 at low temperature in this solvent (Fig. S21). For comparison, photolysis of a CD2Cl2 solution of HN3 and 1-octene in the absence of 1 did not give rise to any observable NH-transfer products (Fig. S22).
[15N]-4, which is expected from a mono-labeled [15N]-HN3 precursor (Fig. S18 and S19). In this experiment, [15N]-N2 was also observed by 15N-NMR (−70.3 ppm),52 which demonstrates the facility of N2 photoelimination under these reaction conditions (Fig. S20). NH transfer to 1-octene was also observed for photolysis of a toluene solution, albeit in lower yield (12%), which we attribute to limited solubility of 2 at low temperature in this solvent (Fig. S21). For comparison, photolysis of a CD2Cl2 solution of HN3 and 1-octene in the absence of 1 did not give rise to any observable NH-transfer products (Fig. S22).
 | ||
| Fig. 6 N–H transfer photochemistry of 2 with olefins. Photochemical olefin aziridination by 2 is stereospecific. Yields obtained by 1H NMR spectroscopy. | ||
Photolysis of 2 in the presence of norbornene affords the corresponding N–H aziridine 5 in 25% yield (for 1H and ESI-MS analysis see Fig. S23 and S24).53 Photochemically promoted N–H transfer is stereospecific: photolysis of 2 in the presence of cis-4-octene afforded cis-6 exclusively (49% yield) (for 1H and ESI-MS analysis see Fig. S25 and S26). This observation is consistent with the stereospecific NH transfer that has been noted in Rh2-catalyzed imidogen-transfer reactions.14,15 The modest yields of aziridines in these reactions are likely due to competing reaction of free NH with olefins: N-chloromethylamine was also observed as reaction byproducts (Fig. S27–S29).13,54,55
In contrast to the observed NH-transfer photochemistry from 2, photolysis of 3a–3c in the presence of 1-octene did not result in nitrene transfer products (i.e., N-aryl aziridines or allylic amination products). This observation is consistent with facile intramolecular rearrangement of aryl nitrenes.56
Together, the results of these NH-transfer experiments indicate that the photogenerated intermediate from 2 is chemically and kinetically competent as an intermediate in the Rh2-catalyzed NH-transfer.
Observation of reactive nitrenes
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) tert-butylbenzene (TBB) (3
tert-butylbenzene (TBB) (3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) glass of 2 at 77 K in a quartz J-Young EPR tube. This solvent system was chosen to provide an optical glass while avoiding ligand exchange with more common glassy solvents such as 2-methyltetrahydrofuran.57 Photolysis of 2 at 77 K results in a small but reproducible blue shift of the absorbance centered at 570 nm to 560 nm and the concurrent growth of significant spectral intensity ∼500 nm (Fig. 7b). These spectral features arise from a non-steady state intermediate: thermal annealing of the sample to 298 K followed by re-freezing at 77 K results in the disappearance of the spectral intensity at 500 nm and the growth of a new absorbance centered at 543 nm, which overlays the spectrum of Rh2(esp)2 (1) in this solvent mixture (Fig. S30 and S31). We carried out analogous cryogenic photolysis of [15N]-2 in a 3
1) glass of 2 at 77 K in a quartz J-Young EPR tube. This solvent system was chosen to provide an optical glass while avoiding ligand exchange with more common glassy solvents such as 2-methyltetrahydrofuran.57 Photolysis of 2 at 77 K results in a small but reproducible blue shift of the absorbance centered at 570 nm to 560 nm and the concurrent growth of significant spectral intensity ∼500 nm (Fig. 7b). These spectral features arise from a non-steady state intermediate: thermal annealing of the sample to 298 K followed by re-freezing at 77 K results in the disappearance of the spectral intensity at 500 nm and the growth of a new absorbance centered at 543 nm, which overlays the spectrum of Rh2(esp)2 (1) in this solvent mixture (Fig. S30 and S31). We carried out analogous cryogenic photolysis of [15N]-2 in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 DMB
1 DMB![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TBB glass with the addition of 1-octene. Spectra obtained during this photolysis indicated the formation of the same non-steady state intermediate observed in the absence of 1-octene (Fig. S32). Thermal annealing of this sample led to disappearance of the non-steady state intermediate and the evolution of a 1
TBB glass with the addition of 1-octene. Spectra obtained during this photolysis indicated the formation of the same non-steady state intermediate observed in the absence of 1-octene (Fig. S32). Thermal annealing of this sample led to disappearance of the non-steady state intermediate and the evolution of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of 4 and [15N]-4, which demonstrates NH-transfer reactivity from the observed non-steady state intermediate (Fig. S33).
1 mixture of 4 and [15N]-4, which demonstrates NH-transfer reactivity from the observed non-steady state intermediate (Fig. S33).
We assign the non-steady state intermediate observed during 77 K photolysis to Rh2–NH complex 3[7]. The spectral features observed after cryogenic photolysis of 2 are consistent with TD-DFT calculations of 3[7] (Fig. 7c).58 DFT single points of the singlet and triplet imidogen complexes (i.e. 1[7] and 3[7] at the SMD-PBE0-D3/Def2-TZVP level of theory) were used to calculate the ΔEST (ΔEST = ESinglet − ETriplet) to be 11.9 kcal mol−1. To further confirm this, single points at the DLPNO-CCSD(T)/Def2-TZVP level of theory were run and provided a ΔEST of 15.2 kcal mol−1. The TD-DFT calculations of 2 and 3[7], using tert-butylbenzene as an implicit solvent model, suggest that conversion of 2 to 3[7] should be accompanied by minor changes in the absorbances at ∼570 and ∼425 nm but also the emergence of a new LMCT transition at 497 nm (Fig. 7d).59 This computed transition is well-matched to the growth of intensity observed experimentally at 500 nm (Fig. S34). In contrast, the observed spectral features are not well-matched to the computed spectrum of 1[7] (Fig. S35). Further, the observed spectral features are inconsistent with the formation of Rh2(II,III), which typically display low energy UV-vis transitions.35,36
Formulation as 3[7] is consistent with the metrical parameters that we previously reported for triplet adamantyl and biaryl nitrenes of Rh2 obtained by in crystallo photochemistry.40,60–62 Attempts to accomplish in crystallo synthesis of 7 were not productive due to loss of crystallinity during sample photolysis.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 DMB
1 DMB![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) TBB glass at 77 K gave rise to a new absorption features centered at 510 nm and 620 nm. Thermal annealing led to the disappearance of these spectroscopic features. Similar to 7, the experimental UV-vis spectra of 8a was qualitatively similar to the TD-DFT simulated spectrum computed for a triplet nitrene although the simulated spectrum is blue-shifted as compared to experiment (for full spectral evolution see Fig. S36). TD-DFT calculations of 3[8a] show transitions with moderate oscillator strength (>0.01) at 469 nm and 516 nm (Fig. S37a). Comparison of the simulated spectral shape and relative oscillator strengths suggests the transition at 469 nm corresponds to the sharper experimental feature at 510 nm whereas the transition at 516 nm corresponds to the feature at 620 nm. Natural Transition Orbital (NTO) analysis shows that the higher energy transition is MLCT from a Rh β d orbital into the SUMO that is delocalized between the nitrene N p orbital and the aryl group and the lower energy transition at 516 nm is mostly comprised of a Rh α d-to-d transition (Fig. 8c, for all the NTOs of 3[8a], see Fig. S37b).
TBB glass at 77 K gave rise to a new absorption features centered at 510 nm and 620 nm. Thermal annealing led to the disappearance of these spectroscopic features. Similar to 7, the experimental UV-vis spectra of 8a was qualitatively similar to the TD-DFT simulated spectrum computed for a triplet nitrene although the simulated spectrum is blue-shifted as compared to experiment (for full spectral evolution see Fig. S36). TD-DFT calculations of 3[8a] show transitions with moderate oscillator strength (>0.01) at 469 nm and 516 nm (Fig. S37a). Comparison of the simulated spectral shape and relative oscillator strengths suggests the transition at 469 nm corresponds to the sharper experimental feature at 510 nm whereas the transition at 516 nm corresponds to the feature at 620 nm. Natural Transition Orbital (NTO) analysis shows that the higher energy transition is MLCT from a Rh β d orbital into the SUMO that is delocalized between the nitrene N p orbital and the aryl group and the lower energy transition at 516 nm is mostly comprised of a Rh α d-to-d transition (Fig. 8c, for all the NTOs of 3[8a], see Fig. S37b).
Cryogenic photolysis of 3b also resulted in the evolution of a non-steady state intermediate (for full spectral evolution see Fig. S38). The spectral features of 3[8b] were observed at 507 nm and 571 nm and TD-DFT calculations result in a simulated spectrum (Fig. S39a) that qualitatively aligns with experiment much like in the case of 3[8a] and shows a blue shift for the higher energy peak relative to 3[8a] (463 nm for 3[8b] and 469 nm for 3[8a]). NTO analysis indicates the parentage of these transitions is similar to that of the features observed for 3[8a] (Fig. S39b). The observed blue-shift of the peaks is consistent with methylation raising the energy of the acceptor orbital for the observed transitions. Superficially, the results obtained from cryogenic photolysis of 3c also indicate the formation of a non-steady state intermediate (for full spectral evolution, see Fig. S40). In this case however, evacuation of the solvent during the preparation of the glassy solvent matrix resulted in partial loss of the azide ligand (vide supra) and thus quantitative comparisons of the resulting optical spectrum are not possible (for TD-DFT and NTO analysis of 3[8c], see Fig. S41).
In crystallo photolysis of 3 provided experimental metrical parameters for the corresponding Rh2 nitrenes (i.e., 3[8a], 3[8b], and 3[8c]) and confirmed the nitrene chemistry implied by the aforementioned spectroscopic and computational studies (Fig. 9, for 3[8a], and 3[8c], see Fig. S42 and S43, respectively). Photolysis of single crystals of 3 resulted in 30–50% photoconversion to the corresponding nitrenes before loss of crystallinity. In all cases, N2 loss was associated with shortening of both the Rh(1)–N(1) bonds (i.e., ∼0.3 Å for Rh(1)–N(1)). Though we cannot locate the N2 molecule in crystallo for 8a and 8c due to disorder, for 8b, in crystallo photoextrusion of N2 from 3b was observed. The metrical parameters of 8a, 8b, and 8c are consistent with formulation as triplet complexes. DFT single points at the SMD-PBE0-D3/Def2-TZVP level of theory were used to calculate the ΔEST to be 11.4 kcal mol−1, 9.1 kcal mol−1, and 12.4 kcal mol−1 for 3[8a], 3[8b], and 3[8c], respectively.
Conclusions
Here, we describe the first structurally characterized transition metal complex of hydrazoic acid, Rh2(esp)2(HN3)2 (2), by addition of gaseous HN3 to Rh2(esp)2 (1). Temperature-dependent NMR, UV-vis, and IR measurements indicate that the HN3 ligands are labile in solution; Keq = 1100 ± 100 M−2 at 243 K. Complex 2 engages in stereospecific NH-transfer photochemistry with olefins, which suggests that the same intermediate is accessed during photolysis of 2 as is generated during Rh2-catalyzed NH-transfer reactions.9 Cryogenic photolysis of 2 in a frozen solvent glass enabled observation of the transient Rh2–NH intermediate. Comparison of the spectroscopic data with results of DFT and TD-DFT calculations suggest this intermediate displays a triplet ground state (i.e., 3[7]).Analogous aryl azide complexes provided further support for the structural and spectroscopic assignments in the hydrazoic acid photochemistry. Cryogenic UV-vis spectroscopy provided evidence for the intermediacy of 3[Rh2–NAr] complexes and bolstered the computationally supported spectroscopic assignments for 3[Rh2–NH]. In addition, in crystallo synthesis provided experimental definition of the metrical parameters of the aryl nitrene adducts, which both confirms the nitrene photochemistry of this family of azide complexes as well as further supports the formulation as triplet adducts. Together, these results introduce HN3 as a ligand in transition metal coordination chemistry, enable photochemical generation of M–NH species, and provide new tools to study metal-catalyzed amination chemistry.
Author contributions
A. P., G. P. V. T., and D. C. P. conceptualized the project. A. P., P. T., M. T. F., B. M., M. J. L., J. H. R., and G. P. V. T. carried out experimental work. The manuscript was written with contributions from all authors.Conflicts of interest
There are no conflicts to declare.Data availability
Experimental details and spectral data are available in the supplementary information (SI). Supplementary information: experimental procedures, spectral data, Cartesian coordinates for the calculated structures, and X-ray diffraction details. See DOI: https://doi.org/10.1039/d6sc02258h.CCDC 2404479, 2514293, 2514304, 2516143, 2523419, 2523425 and 2524966 contain the supplementary crystallographic data for this paper.63a–g
Acknowledgements
The U.S. Department of Energy (DOE), Office of Science, Office of Basic Energy Sciences, Catalysis Program (DE-SC0024121) and the Welch Foundation (A-1907) are acknowledged for funding. Texas A&M University HPRC provided computational resources (https://hprc.tamu.edu).References
- R. F. Klima and A. D. Gudmundsdóttir, J. Photochem. Photobiol., A, 2004, 162, 239–247 CrossRef CAS.
- N. P. Gritsan and M. S. Platz, Chem. Rev., 2006, 106, 3844–3867 CrossRef CAS PubMed.
- D. Wang, W. Chen, H. Chen, Y. Chen, S. Ye and G. Tan, Nat. Chem., 2024, 17, 38–43 CrossRef PubMed.
- M. Janssen, T. Frederichs, M. Olaru, E. Lork, E. Hupf and J. Beckmann, Science, 2024, 385, 318–321 CrossRef CAS PubMed.
- Z.-J. Jia, S. Gao and F. H. Arnold, J. Am. Chem. Soc., 2020, 142, 10279–10283 CrossRef CAS PubMed.
- R. R. Anugu, S. Munnuri and J. R. Falck, J. Am. Chem. Soc., 2020, 142, 5266–5271 CrossRef CAS PubMed.
- A. Boullé, A. Doumbia, J.-P. Mahy and F. Avenier, Chem. Commun., 2023, 59, 79–81 RSC.
- J. L. Jat, D. Chandra, P. Kumar, V. Singh and B. Tiwari, Synthesis, 2022, 54, 4513–4520 CrossRef CAS.
- Y. Gelato, L. Marraffa, F. Pasca, P. Natho, G. Romanazzi, A. Tota, M. Colella and R. Luisi, J. Am. Chem. Soc., 2025, 147, 35567–35575 CrossRef CAS PubMed.
- E. Amble and B. P. Dailey, J. Chem. Phys., 1950, 18, 1422 CrossRef CAS.
- Dimerization of 3[NH] to generate 1[HN–NH] is a spin-allowed reaction according to the Carter–Goddard–Malrieu–Trinquier (CCMT) rules; see, M. Driess and H. Grützmacher, Angew. Chem., Int. Ed., 1996, 35, 828–856 CrossRef CAS; P. Power, Chem. Rev., 1999, 99, 3463–3504 CrossRef PubMed.
- E. Jacox and D. E. Milligan, J. Am. Chem. Soc., 1963, 85, 278–282 CrossRef.
- D. W. Cornell, R. S. Berry and W. Lwowski, J. Am. Chem. Soc., 1966, 88, 544–550 CrossRef CAS.
- Z. Ma, Z. Zhou and L. Kürti, Angew. Chem., Int. Ed., 2017, 56, 9886–9890 CrossRef CAS PubMed.
- J. L. Jat, M. P. Paudyal, H. Gao, Q.-L. Xu, M. Yousufuddin, D. Devarajan, D. H. Ess, L. Kürti and J. R. Falck, Science, 2014, 343, 61–65 CrossRef CAS PubMed.
- W. Xue, Z. Zhu, S. Chen, B. You and C. Tang, J. Am. Chem. Soc., 2023, 145, 4142–4149 CrossRef CAS PubMed.
- R. Mao, S. Gao, Z.-Y. Qin, T. Rogge, S. J. Wu, Z.-Q. Li, A. Das, K. N. Houk and F. H. Arnold, Nat. Catal., 2024, 7, 585–592 CrossRef CAS PubMed.
- F.-X. Fan, H. Xu, S.-X. Tang, Y. Dang and F. Wang, Nat. Commun., 2025, 16, 1471 CrossRef CAS PubMed.
- T. Curtius, Ber. Dtsch. Chem. Ges., 1890, 23, 3023–3033 CrossRef.
- E. H. Eyster, J. Chem. Phys., 1940, 8, 135–142 CrossRef CAS.
- V. Schomaker and R. Spurr, J. Am. Chem. Soc., 1942, 64, 1184–1187 CrossRef CAS.
- O. Kajimoto, T. Yamamoto and T. Fueno, J. Phys. Chem., 1979, 83, 429–435 CrossRef CAS.
- J. Lievin, J. Breulet and G. Verhaegen, Theor. Chim. Acta, 1979, 52, 75–88 CrossRef CAS.
- J. Evers, G. Oehlinger, F. X. Steemann and T. M. Klapötke, Inorg. Chem., 2020, 59, 17671–17677 CrossRef CAS PubMed.
- J. Evers, M. Göbel, B. Krumm, F. Martin, S. Medvedyev, G. Oehlinger, F. X. Steemann, I. Troyan, T. M. Klapötke and M. I. Eremets, J. Am. Chem. Soc., 2011, 133, 12100–12105 CrossRef CAS PubMed.
- H.-J. Himmel, M. Junker and H. Schnöckel, J. Chem. Phys., 2002, 117, 3321–3326 CrossRef CAS.
- S. L. Laursen, J. E. Grace, R. L. DeKock and S. A. Spronk, J. Am. Chem. Soc., 1998, 120, 12583–12594 CrossRef CAS.
- J. N. Russell, V. M. Bermudez and A. Leming, Langmuir, 1996, 12, 6492–6500 CrossRef CAS.
- A. Schmidt, Z. Anorg. Allg. Chem., 1971, 381, 31–39 CrossRef CAS.
- M. Morán and M. Gayoso, J. Organomet. Chem., 1983, 243, 423–426 CrossRef.
- H. Schäfer, W. Saak and M. Weidenbruch, J. Organomet. Chem., 2000, 604, 211–213 CrossRef.
- Z. D. Brown, J. D. Erickson, J. C. Fettinger and P. P. Power, Organometallics, 2013, 32, 617–622 CrossRef CAS.
- K. Bläsing, J. Bresien, R. Labbow, D. Michalik, A. Schulz, M. Thomas and A. Villinger, Angew. Chem., Int. Ed., 2019, 58, 6540–6544 CrossRef PubMed.
- R. Lorpitthaya, Z.-Z. Xie, K. B. Sophy, J.-L. Kuo and X.-W. Liu, Chem.–Eur. J., 2010, 16, 588–594 CrossRef CAS PubMed.
- X. Lin, C. Zhao, C.-M. Che, Z. Ke and D. L. Phillips, Chem.–Asian J., 2007, 2, 1101–1108 CrossRef CAS PubMed.
- K. P. Kornecki and J. F. Berry, Chem.–Eur. J., 2011, 17, 5827–5832 CrossRef CAS PubMed.
- D. N. Zalatan and J. Du Bois, J. Am. Chem. Soc., 2009, 131, 7558–7559 CrossRef CAS PubMed.
- R. H. Perry, T. J. Cahill, J. L. Roizen, J. Du Bois and R. N. Zare, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 18295–18299 CrossRef CAS PubMed.
- P. Bishnoi, A. Singh, Y. Maurya, V. Jhamb, A. Sharma, S. V. Sivapreetha and S. Chatterjee, ACS Org. Inorg. Au, 2025, 5, 385–399 CrossRef CAS PubMed.
- C. G. Espino, K. W. Fiori, M. Kim and J. Du Bois, J. Am. Chem. Soc., 2004, 126, 15378–15379 CrossRef CAS PubMed.
- A. Das, Y.-S. Chen, J. H. Reibenspies and D. C. Powers, J. Am. Chem. Soc., 2019, 141, 16232–16236 CrossRef CAS PubMed.
- A. Das, G. P. Van Trieste and D. C. Powers, Comments Inorg. Chem., 2020, 40, 116–158 CrossRef CAS.
- A. Das, C.-H. Wang, G. P. Van Trieste, C.-J. Sun, Y.-S. Chen, J. H. Reibenspies and D. C. Powers, J. Am. Chem. Soc., 2020, 142, 19862–19867 CrossRef CAS PubMed.
- W. Stroek, M. Keilwerth, L. A. Malaspina, S. Grabowsky, K. Meyer and M. Albrecht, Chem.–Eur. J., 2024, 30, e202303410 CrossRef CAS PubMed.
- Analogous VT-IR experiments were used to characterize the binding equilibrium of adamantyl azide (AdN3) with Fe(HMDS)2.
- R. S. Drago, Physical Methods for Chemists, Saunders College Publishing, 1992 Search PubMed.
- E. P. L. Hunter and S. G. Lias, J. Phys. Chem. Ref. Data, 1998, 27, 413–656 CrossRef CAS.
- R. S. Drago, S. P. Tanner, R. M. Richman and J. R. Long, J. Am. Chem. Soc., 1979, 101, 2897–2903 CrossRef CAS.
- R. S. Drago, J. R. Long and R. Cosmano, Inorg. Chem., 1981, 20, 2920–2927 CrossRef CAS.
- R. S. Drago, J. R. Long and R. Cosmano, Inorg. Chem., 1982, 21, 2196–2202 CrossRef CAS.
- A. F. Trindade, J. A. S. Coelho, C. A. M. Afonso, L. F. Veiros and P. M. P. Gois, ACS Catal., 2012, 2, 370–383 CrossRef CAS.
- K. Banert, N. Singh, B. Fiedler, J. Friedrich, M. Korb and H. Lang, Chem.–Eur. J., 2015, 21, 15092–15099 CrossRef CAS PubMed.
- Photopolymerization of styrenes upon UV irradiation prevented evaluation of potential N–H transfer to these substrates.
- S. Kodama, Bull. Chem. Soc. Jpn., 1983, 56, 2363–2370 CrossRef CAS.
- Attempts to achieve photocatalysis by carrying out photolysis of solutions of 1 with excess HN3 and olefinic substrates were unsuccessful, presumably due to preferential product binding to 1.
- H. Inui, K. Sawada, S. Oishi, K. Ushida and R. J. McMahon, J. Am. Chem. Soc., 2013, 135, 10246–10249 CrossRef CAS PubMed.
- K. S. Suslick, J. F. Bautista and R. A. Watson, J. Am. Chem. Soc., 1991, 113, 6111–6114 CrossRef CAS.
- 3[7] is presumably generated from 2 by initial excitation to a singlet excited state that undergoes intersystem crossing to a triplet excited state from which N2 dissociation proceeds.
- The low energy feature at around 1100 nm was not observed as the spectrometer collection range was 100–1060 nm.
- A. Paikar, G. P. Van Trieste, A. Das, C.-W. Wang, T. E. Sill, N. Bhuvanesh and D. C. Powers, Inorg. Chem., 2023, 62, 12557–12564 CrossRef CAS PubMed.
- Y. Baek, A. Das, S.-L. Zheng, J. H. Reibenspies, D. C. Powers and T. A. Betley, J. Am. Chem. Soc., 2020, 142, 11232–11243 CrossRef CAS PubMed.
- A. Sur and D. C. Powers, ACS Cent. Sci., 2025, 11, 8348–8352 Search PubMed.
- (a) CCDC 2404479: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2lq1v7; (b) CCDC 2514293: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qdb7q; (c) CCDC 2514304: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qdbl2; (d) CCDC 2516143: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qg7xc; (e) CCDC 2523419: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qptmw; (f) CCDC 2523425: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qptt2; (g) CCDC 2524966: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qrfjg.
| This journal is © The Royal Society of Chemistry 2026 |