Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
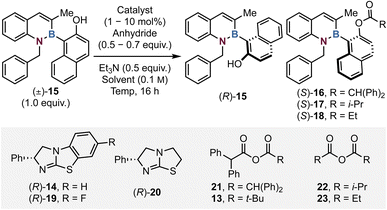
The acylative kinetic resolution of 1,2-azaborine naphthols
Martha I. Prindl,
Aidan P. McKay ,
David B. Cordes
,
David B. Cordes and
Andrew D. Smith
and
Andrew D. Smith *
*
EaStCHEM, School of Chemistry, University of St Andrews, St Andrews, KY16 9ST, Fife, UK. E-mail: ads10@st-andrews.ac.uk
First published on 9th April 2026
Abstract
The enantioselective synthesis of atropisomeric molecules containing stereogenic axes is becoming increasingly important due to their growing incorporation within medicinally relevant compounds. Organocatalytic routes to selectively prepare highly enantioenriched stereogenic axes containing a carbon(sp2)–boron(sp2) bond remain underdeveloped due to the inherent challenge of the longer C–B bond length (1.58 Å) compared to its C(sp2)–C(sp2) counterpart (1.49 Å). This manuscript showcases the development of an isothiourea catalysed acylative kinetic resolution of 1,2-azaborine frameworks to prepare configurationally stable carbon-boron stereogenic axes with good to excellent stereocontrol (24 examples, selectivity factors up to >200). The scope and limitations of this process have been investigated, with product derivatisation and racemisation studies providing insight into the configurational stability of these species and the association between boron hybridisation and atropisomeric stability. Building on insight gained from these studies preliminary proof of principle investigations concerning an acylative dynamic kinetic resolution in this system has been demonstrated.
Introduction
Atropisomerism is a fundamental chiral element defined as “stereoisomerism arising from restricted rotation around a single bond”.1–3 While configurationally stable chiral atropisomers were first described in 1922,4 Oki first defined a practical definition of atropisomers as having a half-life of greater than 1000 s (17 min) in 1983.5 A more sophisticated classification system was introduced by LaPlant et al.,6,7 organising atropisomers into three classes based on their racemisation half-lives and the resultant practical implications. For example, class 1 atropisomers (rotamers) have too short a lifetime to be isolated as distinct species (t1/2rac < 50 s, ΔG‡298 < 20 kcal mol−1) while class 2 atropisomers can be isolated but racemise quickly (50 s < t1/2rac < 430 d, ΔG‡298 = 20–28 kcal mol−1). Class 3 atropisomers (t1/2rac > 430 d, ΔG‡298 > 28 kcal mol−1) are considered configurationally stable and generally amenable for further applications and as such are important components in catalysts, pharmaceuticals, natural products and functional molecules such as photoswitches and chiral recognition reagents.2,8,9 For instance, there are currently four FDA approved drugs that are stable atropisomers with the anti-cancer KRAS inhibitor Sotorasib 1 which contains a highly stable C–N axis a representative example10–14 (Fig. 1A). Furthermore, it is estimated that ∼30% of small drug molecules approved since 2011 are class 1 atropisomers containing at least one unrestricted stereogenic axis.9,15 Commonly studied biaryl or heteroaryl atropisomeric systems incorporate C–C, C–N, C–S and C–O linkages, with methods for their selective preparation alongside their configurational stability having been well explored.16–20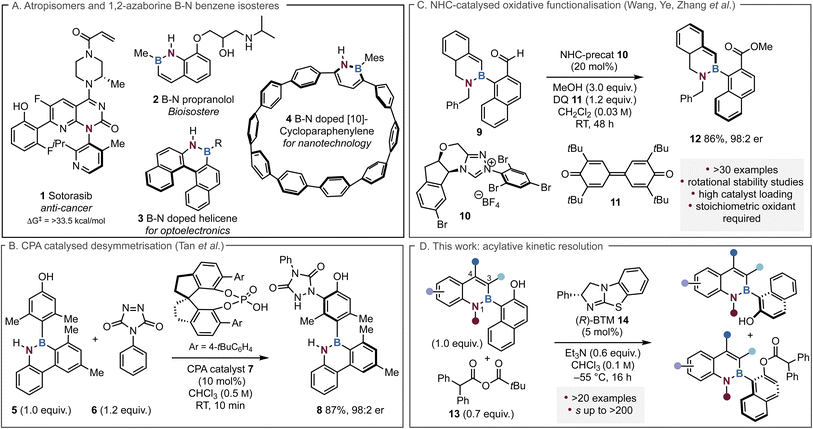 | ||
| Fig. 1 Applications, previous and current work on organocatalytic atropisomeric 1,2-azaborine scaffolds. | ||
To date limited synthetic routes to atropisomeric species that contain a C–B stereogenic axis have been developed, primarily due to the inherent challenge of a lower barrier to rotation that is enabled by a longer C(sp2)–B(sp2) bond (1.58 Å) compared to a C(sp2)–C(sp2) bond (1.49 Å).21,22 1,2-Azaborines are a distinctive class of boron-containing molecules that replace a C–C bond with a B–N bond, rendering them benzene isosteres.23–25 As such, applications have mainly focused on their potential as bioisosteres such as 2, along with their unique photophysical or electrochemical properties within molecules, including doped helicine motifs 3 and cycloparaphenylenes (CPPs) 4 (Fig. 1A).26–37 A handful of examples exemplified by Song and co-workers, have employed transition metal catalysts to develop highly enantioselective methods to generate atropisomeric C–B azaborine axes38–44 but notably only limited examples employ organocatalytic methods. The first state-of-the art organocatalytic approach was developed by Tan and co-workers in 2021, who used a chiral phosphoric acid (CPA) catalyst 7 to desymmetrise azaborine 5 to generate products such as 8 with excellent enantiocontrol (Fig. 1B).45 In an alternative dynamic kinetic resolution (DKR) approach, Zhang and co-workers employed an N-heterocyclic carbene (NHC) catalysed oxidative esterification process to resolve a wide range of azaborine scaffolds, giving the corresponding products in up to 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 er (Fig. 1C).46 Although effective, this approach requires high catalyst loadings of the NHC catalyst (20 mol%) as well as a stoichiometric amount of the oxidant 3,3′,5,5′-tetra-tert-butyldiphenoquinone (DQ).
1 er (Fig. 1C).46 Although effective, this approach requires high catalyst loadings of the NHC catalyst (20 mol%) as well as a stoichiometric amount of the oxidant 3,3′,5,5′-tetra-tert-butyldiphenoquinone (DQ).
The use of Lewis basic isothiourea catalysts within enantioselective acylation processes has been well established since Birman's founding work that demonstrated the effective kinetic resolution of secondary alcohols via selective benzotetramisole (BTM) 14 catalysed acyl transfer.47 Since this initial demonstration, multiple protocols for isothiourea catalysed kinetic resolutions for the preparation of point chiral molecules have been developed, including the resolution of secondary48,49 and more challenging tertiary alcohols,50 diols51 and fluorohydrins.52 The application of isothiourea catalysts for the generation of enantioenriched materials containing C–C, C–N or N–N stereogenic axes has previously been explored, including within a thia-Michael addition cyclisation,53 the N-acylation of sulfonamides54 and N-acylaminoindoles,55 and the resolution of biaryl diol species.56,57 At the outset of this project the organocatalytic development of a kinetic resolution process to introduce a C–B stereogenic axis via isothiourea catalysis had not been studied. In this context, this manuscript describes the effective kinetic resolution of a range of 1,2-azaborine naphthols to afford highly enantioenriched ester and alcohol products. The key role of substitution at C(3)-, C(4)- and N(1)- in determining the selectivity in these processes has been interrogated, with the configurational stability of a range of substrates probed (Fig. 1D). During the finalisation of this manuscript a related but orthogonal acylative kinetic resolution of 1,2-azaborines using an isothiourea catalyst (tetramisole) was developed by the Li group. This elegant work incorporated a C(3)-iodine substituent as a necessary constraint to achieve high enantioinduction using isobutyric anhydride as an acyl donor, and required a high 20 mol% catalyst loading over 48 hours reaction time.58 The barrier to rotation in the C(3)-iodoalcohol substrate was calculated to be 36.5 kcal mol−1, consistent with significant configurational stability, meaning that a DKR in this system would be unlikely. In comparison, this work employs lower catalyst loadings (5 mol% of (R)-14 over 16 hours) and focuses upon alternative less sterically hindered azaborine motifs bearing C(3)-Me or H-substituents. Furthermore, the sterically encumbered diphenylacetic pivalic anhydride was used in this process, delivering access to enantioenriched azaborine substrates not tolerated in the Li group publication. Significantly, mechanistic investigations concerning 1,2-azaborine racemisation and boron hybridisation, coupled with extending this approach to proof of concept acylative DKR processes distinguishes this from Li's work, and further expands the methods of developing enantioenriched C–B axes within azaborine motifs.
Results and discussion
Optimisation of the acylative kinetic resolution of a model 1,2-azaborine
To probe the viability of an acylative kinetic resolution process, initial optimisation investigated 1,2-azaborine substrate 15 bearing an N-benzyl and C(3)-Me substituents with diphenylacetic anhydride 21 as model reactants (Table 1). Initial conditions employed 10 mol% of isothiourea (R)-BTM 14 with 0.5 equivalents of anhydride 21 and 0.5 equivalents of Et3N in CHCl3 at room temperature for 16 hours, giving 53% conversion to ester 16 with promising selectivity (s = 9, entry 1), giving alcohol 15 (85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 er) and ester 16 (81
15 er) and ester 16 (81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19 er). Changing the Lewis base catalyst to tetramisole 20 gave reduced conversion and moderate stereoselectivity (entry 2). Further work using (R)-BTM sequentially decreased the catalyst loading to 5 and then 1 mol% of 14 (entries 3 and 4) with limited variation in conversion or selectivity. The introduction of isobutyric anhydride 22 with F-BTM 19 afforded poor selectivity (s = 3). Lowering the reaction temperature was found to have a significantly beneficial effect upon selectivity (entries 8–10), with optimal selectivity observed at −50 °C (c = 43, s = 50, entry 10). Further reducing the temperature to −75 °C afforded no conversion to ester 16 (entries 11–13). Altering the base and anhydride stoichiometry (to 0.6 and 0.7 equivalents respectively), changing to the mixed anhydride 13 and increasing the catalyst loading of 14 up to 5 mol% at −55 °C improved the conversion to 53%, providing optimal KR conditions with high selectivity (s = 43, entry 14), giving alcohol 15 (98
19 er). Changing the Lewis base catalyst to tetramisole 20 gave reduced conversion and moderate stereoselectivity (entry 2). Further work using (R)-BTM sequentially decreased the catalyst loading to 5 and then 1 mol% of 14 (entries 3 and 4) with limited variation in conversion or selectivity. The introduction of isobutyric anhydride 22 with F-BTM 19 afforded poor selectivity (s = 3). Lowering the reaction temperature was found to have a significantly beneficial effect upon selectivity (entries 8–10), with optimal selectivity observed at −50 °C (c = 43, s = 50, entry 10). Further reducing the temperature to −75 °C afforded no conversion to ester 16 (entries 11–13). Altering the base and anhydride stoichiometry (to 0.6 and 0.7 equivalents respectively), changing to the mixed anhydride 13 and increasing the catalyst loading of 14 up to 5 mol% at −55 °C improved the conversion to 53%, providing optimal KR conditions with high selectivity (s = 43, entry 14), giving alcohol 15 (98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 er) and ester 16 (93
2 er) and ester 16 (93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 er). Propionic anhydride 23 was also screened but gave reduced stereoselectivity (c = 45, s = 3), with the additional steric bulk provided by the diphenyl substitution on 13 being required for optimal selectivity. A 2.5 gram scale-up reaction on the model substrate 15 was performed and pleasingly no degradation in enantiocontrol or conversion was observed, with enantioenriched 15 and 16 obtained with a selectivity factor of 50 and a conversion of 49, corresponding to 50% isolated yield of the alcohol 15 (92
7 er). Propionic anhydride 23 was also screened but gave reduced stereoselectivity (c = 45, s = 3), with the additional steric bulk provided by the diphenyl substitution on 13 being required for optimal selectivity. A 2.5 gram scale-up reaction on the model substrate 15 was performed and pleasingly no degradation in enantiocontrol or conversion was observed, with enantioenriched 15 and 16 obtained with a selectivity factor of 50 and a conversion of 49, corresponding to 50% isolated yield of the alcohol 15 (92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 er), and 45% ester 16 (95
8 er), and 45% ester 16 (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 er, Fig. 2). The absolute (R)-configuration within resolved alcohol 15 was unambiguously determined by X-ray crystallographic analysis, with all other alcohols assigned by analogy and allowing the (S)-configuration within the ester to be assigned (See SI and Fig. 2).
5 er, Fig. 2). The absolute (R)-configuration within resolved alcohol 15 was unambiguously determined by X-ray crystallographic analysis, with all other alcohols assigned by analogy and allowing the (S)-configuration within the ester to be assigned (See SI and Fig. 2).
| Entry | Temp. (°C) | Catalyst (mol%) | Solvent (0.1 M) | Anhydride (equiv.) | Conversion (c) | Selectivity factor (s) | Erb |
|---|---|---|---|---|---|---|---|
| a All reactions were performed on a 0.1 mmol scale.b Ratio of alcohol/ester er, determined by HPLC analysis on a chiral stationary phase.c ∼7% ester observed.d Reacted for 24 hours.e Using 0.6 equiv. of Et3N. Following literature, s values under 50 are reported to nearest integer, above 50 to the nearest 10 and for very high s factor values > 200 is used.59 The following equations were used for calculations: c = ee(alcohol)/ee(alcohol)+ee(ester). s = ln[(1 − conv)(1 − ee(alcohol))]/(ln[(1 − conv)(1 + ee(alcohol))]. | |||||||
| 1 | RT | (R)-BTM 14 (10) | CHCl3 | 21 (0.5) | 53 | 9 | 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15/81 15/81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19 19 |
| 2 | RT | (R)-TM 20 (10) | CHCl3 | 21 (0.5) | 39 | 8 | 72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28/84 28/84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16 16 |
| 3 | RT | (R)-BTM 14 (5) | CHCl3 | 21 (0.5) | 44 | 10 | 78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22/85 22/85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
| 4 | RT | (R)-BTM 14 (1) | CHCl3 | 21 (0.5) | 50 | 10 | 83![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17/83 17/83![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 17 17 |
| 5 | RT | (R)-F-BTM 19 (1) | CHCl3 | 21 (0.5) | 43 | 12 | 78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 22/87 22/87![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 13 |
| 6 | RT | (R)-F-BTM 19 (1) | CHCl3 | 22 (0.5) | 52 | 3 | 71![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 29/69 29/69![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31 31 |
| 7 | RT | NONE | CHCl3 | 21 (0.5) | N.Ac | — | — |
| 8 | 0 | (R)-BTM 14 (1) | CHCl3 | 21 (0.5) | 47 | 14 | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16/88 16/88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 12 |
| 9 | −20 | (R)-BTM 14 (1) | CHCl3 | 21 (0.5) | 44 | 34 | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16/94 16/94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 6 |
| 10 | −50 | (R)-BTM 14 (1) | CHCl3 | 21 (0.5) | 43 | 50 | 85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15/96 15/96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 4 |
| 11d | −75 | (R)-BTM 14 (1) | Et2O | 21 (0.5) | N.R | — | — |
| 12d | −75 | (R)-BTM 14 (1) | PhMe | 21 (0.5) | N.R | — | — |
| 13d | −75 | (R)-BTM 14 (1) | CH2Cl2 | 21 (0.5) | N.R | — | — |
| 14e | −55 | (R)-BTM 14 (5) | CHCl3 | 13 (0.7) | 53 | 43 | 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2/93 2/93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 7 |
| 15e | −55 | (R)-BTM 14 (5) | CHCl3 | 23 (0.7) | 45 | 3 | 67![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 33/70 33/70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 30 |
Scope and limitations of the acylative kinetic resolution
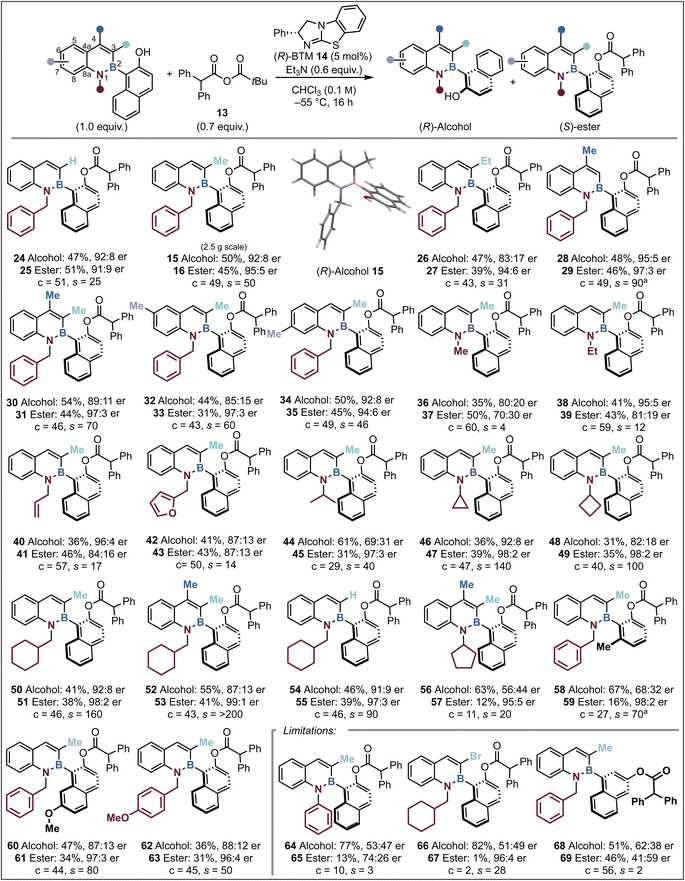
With the optimal conditions in hand the scope and limitations of the kinetic resolution process were probed. Variation at the C(3)-position was first considered as this was predicted to significantly change the steric environment around the C–B stereogenic axis which could directly alter both selectivity and conversion. Introduction of a C(3)-H or C(3)-Et substituent was readily tolerated, giving alcohol 24 and ester 25 (C(3)-H, c = 51, s = 25, erester = 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9) and alcohol 26 and ester 27 (C(3)-Et, c = 43, s = 31, erester = 94
9) and alcohol 26 and ester 27 (C(3)-Et, c = 43, s = 31, erester = 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6) with good but slightly diminished selectivity relative to the model substrate.
6) with good but slightly diminished selectivity relative to the model substrate.
Kinetic resolution of alcohols 24 (s = 25) and 26 (s = 31) proceeded with higher selectivity with anhydride 13, compared to Li's work (for 24 (s = 7) and 26 (s = 6) respectively) using anhydride 22, highlighting the substrate class orthogonality.58 Variation to a C(4)-Me substituent provided higher enantioselectivity than the model system, giving alcohol 28 and ester 29 (C(4)-Me, c = 49, s = 90, erester = 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), which is postulated to arise as a consequence of a “buttressing effect”, that harnesses the C(4)-Me interaction with the ortho-C(3)-H to hinder axis rotation.60,61 The incorporation of methyl substitution at both C(3) and C(4) was also tolerated, giving 30 and 31 with excellent levels of selectivity in the acylative kinetic resolution (c = 46, s = 70, erester = 97
3), which is postulated to arise as a consequence of a “buttressing effect”, that harnesses the C(4)-Me interaction with the ortho-C(3)-H to hinder axis rotation.60,61 The incorporation of methyl substitution at both C(3) and C(4) was also tolerated, giving 30 and 31 with excellent levels of selectivity in the acylative kinetic resolution (c = 46, s = 70, erester = 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). The addition of a methyl substituent to the C(6)- or C(7)-position gave similar results to that observed within the model substrate. These reactions afforded alcohol 32 and ester 33 (C(6)-Me, c = 43, s = 60, erester = 97
3). The addition of a methyl substituent to the C(6)- or C(7)-position gave similar results to that observed within the model substrate. These reactions afforded alcohol 32 and ester 33 (C(6)-Me, c = 43, s = 60, erester = 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) as well as alcohol 34 and ester 35 (C(7)-Me, c = 49, s = 46, erester = 94
3) as well as alcohol 34 and ester 35 (C(7)-Me, c = 49, s = 46, erester = 94![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6) respectively.
6) respectively.
Subsequent investigations focused upon the impact of changing the N(1)-substituent within the azaborine as this was also predicted to significantly impact the observed selectivity within the system. With a C(3)-Me substituent, sequential variation from an N(1)-Me (c = 60, s = 4, erester = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30), to N(1)-Et (c = 59, s = 12, erester = 81
30), to N(1)-Et (c = 59, s = 12, erester = 81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19) to N(1)-allyl substitution (c = 57, s = 17, erester = 84
19) to N(1)-allyl substitution (c = 57, s = 17, erester = 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 16) led to a sequential increase in stereoselectivity. In the latter two cases, allowing the reactions to proceed to >50% conversion allowed access to highly enantioenriched alcohols N(1)-Et 38 (41%, 95
16) led to a sequential increase in stereoselectivity. In the latter two cases, allowing the reactions to proceed to >50% conversion allowed access to highly enantioenriched alcohols N(1)-Et 38 (41%, 95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 er) and N(1)-allyl 40 (36%, 96
5 er) and N(1)-allyl 40 (36%, 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 er). Compared to the model substrate, incorporation of a N(1)-furanylmethyl substituent led to reduced selectivity (c = 50, s = 14, erester = 87
4 er). Compared to the model substrate, incorporation of a N(1)-furanylmethyl substituent led to reduced selectivity (c = 50, s = 14, erester = 87![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13). Incorporation of a branched N(1)-i-Pr substituent led to higher selectivity but reduced conversion (c = 29, s = 40) giving ester 45 with high enantiocontrol (31%, 97
13). Incorporation of a branched N(1)-i-Pr substituent led to higher selectivity but reduced conversion (c = 29, s = 40) giving ester 45 with high enantiocontrol (31%, 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 er). Building on this observation the introduction of branched but conformationally constrained N(1)-cycloalkyl groups was considered, with N(1)-cyclopropyl substitution affording significantly improved selectivity (c = 47, s = 140), giving good yields of highly enantioenriched ester 47 (39%, 98
3 er). Building on this observation the introduction of branched but conformationally constrained N(1)-cycloalkyl groups was considered, with N(1)-cyclopropyl substitution affording significantly improved selectivity (c = 47, s = 140), giving good yields of highly enantioenriched ester 47 (39%, 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 er) and alcohol 46 (36%, 92
2 er) and alcohol 46 (36%, 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 er). Likewise, variation to a N(1)-cyclobutyl group on 48 afforded excellent selectivity (s = 100, erester = 98
8 er). Likewise, variation to a N(1)-cyclobutyl group on 48 afforded excellent selectivity (s = 100, erester = 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). Extension of this approach to the β-branched N(1)-cyclohexylmethyl substituent led to good conversion and excellent stereoselectivity (c = 46, s = 160), giving good yields of highly enantioenriched ester 51 (38%, 98
2). Extension of this approach to the β-branched N(1)-cyclohexylmethyl substituent led to good conversion and excellent stereoselectivity (c = 46, s = 160), giving good yields of highly enantioenriched ester 51 (38%, 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 er) and alcohol 50 (41%, 92
2 er) and alcohol 50 (41%, 92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8 er). Further developments combined methyl substitution at both C(3)- and C(4)- with an N(1)-cyclohexylmethyl substituent leading to excellent selectivity (c = 43, s = >200), giving alcohol 52 (55%, 87
8 er). Further developments combined methyl substitution at both C(3)- and C(4)- with an N(1)-cyclohexylmethyl substituent leading to excellent selectivity (c = 43, s = >200), giving alcohol 52 (55%, 87![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 er) and ester 53 (41%, 99
13 er) and ester 53 (41%, 99![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 er). Further use of an N(1)-cyclohexylmethyl substituent with C(3)-H substitution also led to kinetic resolution with high selectivity (c = 46, s = 90), giving good yields of highly enantioenriched ester 55 (39%, 97
1 er). Further use of an N(1)-cyclohexylmethyl substituent with C(3)-H substitution also led to kinetic resolution with high selectivity (c = 46, s = 90), giving good yields of highly enantioenriched ester 55 (39%, 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 er) and alcohol 54 (46%, 91
3 er) and alcohol 54 (46%, 91![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 er). Disappointingly, the introduction of an N(1)-cyclopentyl substituent with methyl substitution at both C(3) and C(4) gave poor reactivity and reduced stereoselectivity (c = 11, s = 20). The effect of changing the naphthol scaffold to a 2,6-disubstituted phenol on 58 was tested, giving reduced conversion but high selectivity (c = 27, s = 70) affording ester 59 in low yield but with high enantiocontrol (16%, 98
9 er). Disappointingly, the introduction of an N(1)-cyclopentyl substituent with methyl substitution at both C(3) and C(4) gave poor reactivity and reduced stereoselectivity (c = 11, s = 20). The effect of changing the naphthol scaffold to a 2,6-disubstituted phenol on 58 was tested, giving reduced conversion but high selectivity (c = 27, s = 70) affording ester 59 in low yield but with high enantiocontrol (16%, 98![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 er). Further developing the napthol ring to an OMe-substituted variant was also well tolerated (c = 44, s = 80), giving enantioenriched ester 61 (34%, 97
2 er). Further developing the napthol ring to an OMe-substituted variant was also well tolerated (c = 44, s = 80), giving enantioenriched ester 61 (34%, 97![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 er) and alcohol 60 (47%, 87
3 er) and alcohol 60 (47%, 87![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 er). Comparative to the model substrate, the introduction of a N(1)-paramethoxybenzyl group on alcohol 62 was also readily tolerated, (c = 45, s = 50, erester = 96
13 er). Comparative to the model substrate, the introduction of a N(1)-paramethoxybenzyl group on alcohol 62 was also readily tolerated, (c = 45, s = 50, erester = 96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4). Several limitations within this system were also identified. For example, introduction of a bulky N(1)-phenyl substituent on 64 led to significantly reduced substrate reactivity, giving poor conversion (c = 10) and selectivity (s = 3). Incorporation of a C(3)-bromine substituent also led to very poor conversion (c = 2), presumably reflecting the significant steric effect of incorporating bromine substitution at this position, coupled with the use of a sterically encumbered acylating reagent. This limitation highlights the orthogonality in acylating reagent design with Li's work requiring a C(3)-halogen for high enantiodiscrimination while using isobutyric anhydride 22.58 Attempts to extend this process to selective acylation of a meta-substituted naphthol led to good reaction conversion (c = 56) but with very poor selectivity (s = 2), which is postulated to be due to the greater distance from the alcohol to the proposed recognition motif that dictates stereoselectivity.
4). Several limitations within this system were also identified. For example, introduction of a bulky N(1)-phenyl substituent on 64 led to significantly reduced substrate reactivity, giving poor conversion (c = 10) and selectivity (s = 3). Incorporation of a C(3)-bromine substituent also led to very poor conversion (c = 2), presumably reflecting the significant steric effect of incorporating bromine substitution at this position, coupled with the use of a sterically encumbered acylating reagent. This limitation highlights the orthogonality in acylating reagent design with Li's work requiring a C(3)-halogen for high enantiodiscrimination while using isobutyric anhydride 22.58 Attempts to extend this process to selective acylation of a meta-substituted naphthol led to good reaction conversion (c = 56) but with very poor selectivity (s = 2), which is postulated to be due to the greater distance from the alcohol to the proposed recognition motif that dictates stereoselectivity.
Quantifying the configurational stability of azaborines
To understand the configurational stability profile of a number of model azaborine substrates, the racemisation half-life (and barrier to rotation) of a series of enantioenriched alcohols and ester derivatives was investigated.62 Heating an enantioenriched substrate of known enantiopurity in xylene to a defined temperature and monitoring the degradation of enantiomeric excess over time was used for this purpose. Due to observed but inconsistent decomposition, a different temperature was used for each substrate. As a representative example, the barrier to rotation for model C(3)-Me substituted alcohol 15 was determined (t1/2rac = 0.27 h at 404 K, ΔG‡404 = 30.3 kcal mol−1). Assuming that ΔG‡ is invariant with temperature,62 this corresponds to a racemisation half-life at room temperature of ∼27 years. This level of configurational stability defines 15 as a class 3 atropisomer.9 The barrier to rotation along the C–B axis of the corresponding C(3)-H substituted alcohol 24 was calculated to be substantially lower than the C(3)-Me analogue (t1/2rac =12.1 h at 314 K, ΔG‡314 = 25.8 kcal mol−1), defining 24 as a class 2 atropisomer. This is consistent with the steric nature of the C(3)-substituent having a significant effect upon configurational lability. Extension of this approach to the corresponding C(3)-Me 16 and C(3)-H 25 substituted esters was also investigated. The same trend was observed, with the rotational barrier for C(3)-Me substituted ester 16 (t1/2rac = 280 h at 353 K, ΔG‡353 = 31.2 kcal mol−1) being significantly larger than the C(3)-H analogue 25 (t1/2rac = 1.3 h at 382 K, ΔG‡382 = 29.8 kcal mol−1). This also indicates that the ester products show significantly enhanced configurational stability compared to the respective alcohols, again consistent with the increased size of the ester in comparison to the alcohol leading to a higher barrier to rotation.Further studies noted that upon hydrolysis of enantioenriched ester 16 (95![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 er) using 3.0 equivalents of LiOH, while this afforded the alcohol 15 in good yield (73%), it was essentially racemic (52
5 er) using 3.0 equivalents of LiOH, while this afforded the alcohol 15 in good yield (73%), it was essentially racemic (52![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 48 er). It is postulated that racemisation of the alcohol occurs through reversible hydroxide anion addition to the boron atom, generating an sp3 boron centre which significantly lowers the barrier to rotation around the C–B bond, resulting in racemisation.38 Consistent with this proposal, racemisation of enantioenriched alcohol (R)-15 (93
48 er). It is postulated that racemisation of the alcohol occurs through reversible hydroxide anion addition to the boron atom, generating an sp3 boron centre which significantly lowers the barrier to rotation around the C–B bond, resulting in racemisation.38 Consistent with this proposal, racemisation of enantioenriched alcohol (R)-15 (93![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 er) was observed over 4 h upon treatment with KOtBu (1.2 equiv.) and Et3N (1.0 equiv.) in THF (Fig. 3). Intrigued by this effect, the introduction of a hydroxymethyl substituent within azaborine 70 was investigated. Interestingly, enantiomer interconversion through dynamic HPLC at 303 K was observed for 70 through a diagnostic “Batman” peak shape, indicative of enantiomerisation occurring at the HPLC timescale. Using the DCXplorer software developed by the Trapp group, the barrier to rotation within azaborine 70 was readily determined (t1/2rac = 1.23 min at 303 K, ΔG‡303 = 21.0 kcal mol−1), indicating a significantly lower energy barrier to rotation than the other substrates.63,64 While reversible intramolecular nucleophilic addition of the alcohol to the pendant boron atom to form an sp3-boron species (resulting in C–B bond elongation) is reasonable, the origin of the observed remarkable effect on configurational lability is unclear. It is speculated that the lability of the azaborine heterocycle is affected by this bond elongation, potentially allowing reversible ring-opening upon coordination, leading to enantiomerisation. The scope and limitations of this observation, as well as further studies concerning the mechanism of this process, is currently under investigation within this laboratory.
7 er) was observed over 4 h upon treatment with KOtBu (1.2 equiv.) and Et3N (1.0 equiv.) in THF (Fig. 3). Intrigued by this effect, the introduction of a hydroxymethyl substituent within azaborine 70 was investigated. Interestingly, enantiomer interconversion through dynamic HPLC at 303 K was observed for 70 through a diagnostic “Batman” peak shape, indicative of enantiomerisation occurring at the HPLC timescale. Using the DCXplorer software developed by the Trapp group, the barrier to rotation within azaborine 70 was readily determined (t1/2rac = 1.23 min at 303 K, ΔG‡303 = 21.0 kcal mol−1), indicating a significantly lower energy barrier to rotation than the other substrates.63,64 While reversible intramolecular nucleophilic addition of the alcohol to the pendant boron atom to form an sp3-boron species (resulting in C–B bond elongation) is reasonable, the origin of the observed remarkable effect on configurational lability is unclear. It is speculated that the lability of the azaborine heterocycle is affected by this bond elongation, potentially allowing reversible ring-opening upon coordination, leading to enantiomerisation. The scope and limitations of this observation, as well as further studies concerning the mechanism of this process, is currently under investigation within this laboratory.
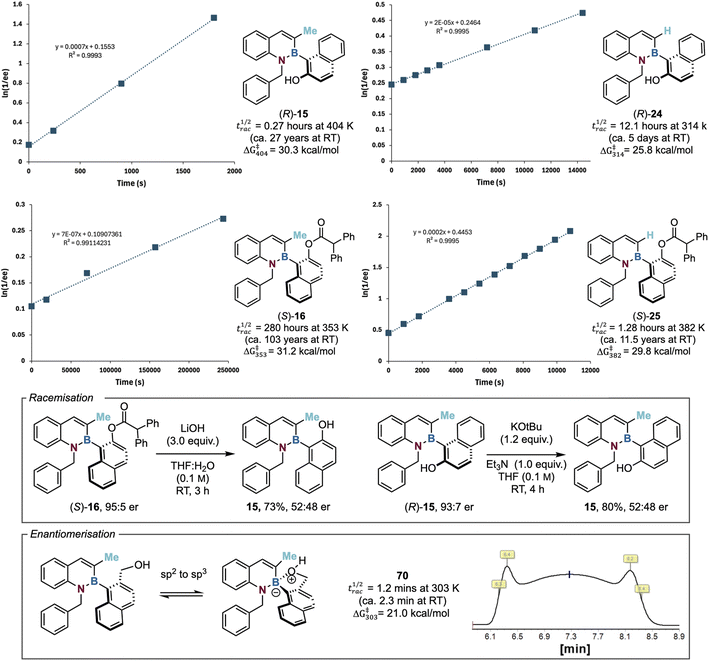 | ||
| Fig. 3 Quantifying the configurational stability of the 1,2-azaborines. With respect to half-life of racemisation calculations, RT = 298 K. N.B. The barrier to rotation for alcohol 24 was determined in good agreement with that from Li's group (26.1 kcal mol−1).58 | ||
Towards an acylative dynamic kinetic resolution process
Since the barrier to rotation of the C(3)-H substituted alcohol 24 (ΔG‡314 = 25.8 kcal mol−1) was calculated to be substantially lower than the corresponding C(3)-Me substituted analogue 15 (ΔG‡404 = 30.3 kcal mol−1), it was considered that 24 might undergo a DKR process. This was explored through heating 1.0 equivalents of azaborine 24 and 1.0 equivalents of anhydride 13 with a low catalyst loading of 14 (2.5 mol%) at 50 °C for 6.5 hours. Pleasingly this generated a 67% isolated yield of 25 with reasonable levels of enantioselectivity (81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19 er), with the remaining yield attributed to racemic alcohol 24, supporting the dynamic nature of this process (Fig. 4). To clarify that the moderate selectivity observed (81
19 er), with the remaining yield attributed to racemic alcohol 24, supporting the dynamic nature of this process (Fig. 4). To clarify that the moderate selectivity observed (81![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19 er) was not an artifact of ester racemisation, a control study was performed where enantioenriched ester (S)-25 was heated without catalyst to 50 °C in CHCl3. After 6.5 h, no degradation of enantiopurity was observed, indicating the lower selectivity is not due to in situ racemisation. An extensive optimisation process followed, including the screening of a variety of organic and inorganic bases, however no improvement in selectivity or acylation was observed (see SI for further details). The lower yield can be partly attributed to degradation of the azaborine starting material and product upon heating. Azaborines 28 and 54 that worked well within the KR process, were then introduced into the DKR. Disappointingly, the introduction of a C(4)-Me substituent did not improve the results, affording ester 29 in 61% yield and reduced enantioselectivity (72
19 er) was not an artifact of ester racemisation, a control study was performed where enantioenriched ester (S)-25 was heated without catalyst to 50 °C in CHCl3. After 6.5 h, no degradation of enantiopurity was observed, indicating the lower selectivity is not due to in situ racemisation. An extensive optimisation process followed, including the screening of a variety of organic and inorganic bases, however no improvement in selectivity or acylation was observed (see SI for further details). The lower yield can be partly attributed to degradation of the azaborine starting material and product upon heating. Azaborines 28 and 54 that worked well within the KR process, were then introduced into the DKR. Disappointingly, the introduction of a C(4)-Me substituent did not improve the results, affording ester 29 in 61% yield and reduced enantioselectivity (72![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 28 er), with the remaining alcohol showing some but poor enantioenrichment (56
28 er), with the remaining alcohol showing some but poor enantioenrichment (56![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 44 er) after 6.5 h. It is postulated the reduced enantioenrichment of 29 is related to the “buttressing effect” with C(3)-Me substitution that hinders rotation around the B–C axis and disfavours a dynamic process. Variation in the substitution from a N(1)-benzyl to a N(1)-cyclohexylmethyl group on 55 afforded a mediocre yield (55%), but with greatly improved enantioselectivity (88
44 er) after 6.5 h. It is postulated the reduced enantioenrichment of 29 is related to the “buttressing effect” with C(3)-Me substitution that hinders rotation around the B–C axis and disfavours a dynamic process. Variation in the substitution from a N(1)-benzyl to a N(1)-cyclohexylmethyl group on 55 afforded a mediocre yield (55%), but with greatly improved enantioselectivity (88![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12 er) while the remaining alcohol 54 was racemic. It is postulated that the moderate enantioselectivity observed within the dynamic process is due to the use of the higher reaction temperature required for alcohol racemisation. The azaborine kinetic resolution optimisation and previous work within the Smith group has observed improved selectivity upon reduced temperatures.57 Extension of this approach to the hydroxymethyl substituted analogue 70 that shows dynamic enantiomerization on the HPLC timescale was next attempted. Screening of a number of isothiourea catalysts showed that the Lewis base isoselenourea HyperSe 71 gave the best but still poor enantiocontrol in this transformation (Fig. 4). The enantiomeric ratio of product 72 did not deteriorate over 5 days of storage in solution and so it is assumed the poor enantioinduction is not a consequence of ester racemisation.
12 er) while the remaining alcohol 54 was racemic. It is postulated that the moderate enantioselectivity observed within the dynamic process is due to the use of the higher reaction temperature required for alcohol racemisation. The azaborine kinetic resolution optimisation and previous work within the Smith group has observed improved selectivity upon reduced temperatures.57 Extension of this approach to the hydroxymethyl substituted analogue 70 that shows dynamic enantiomerization on the HPLC timescale was next attempted. Screening of a number of isothiourea catalysts showed that the Lewis base isoselenourea HyperSe 71 gave the best but still poor enantiocontrol in this transformation (Fig. 4). The enantiomeric ratio of product 72 did not deteriorate over 5 days of storage in solution and so it is assumed the poor enantioinduction is not a consequence of ester racemisation.
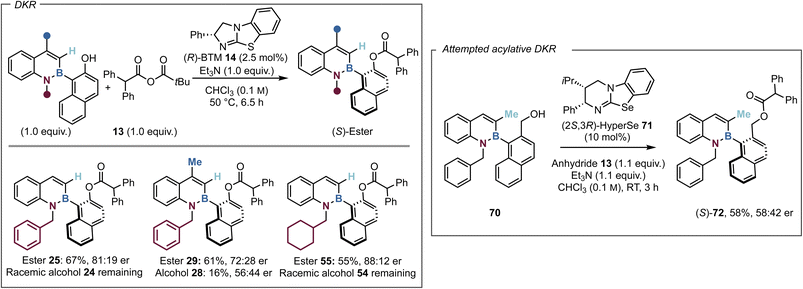 | ||
| Fig. 4 All reactions were performed on a 0.2 mmol scale. Isolated yields are reported. Product enantioselectivity was determined by HPLC analysis on a chiral stationary phase. | ||
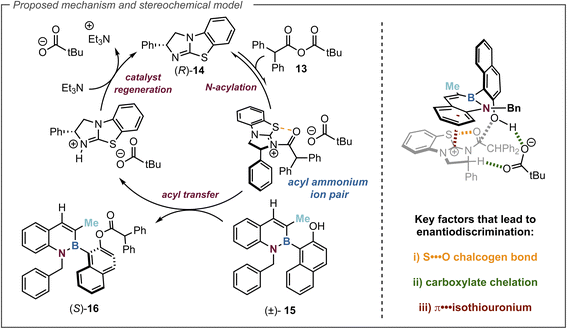
Proposed mechanism and stereochemical model
The proposed catalytic cycle for this transformation is set out in Fig. 5. Initially, (R)-BTM 14 is acylated with anhydride 13 leading to an acyl ammonium ion pair. The (S)-enantiomer of alcohol 15 preferentially reacts with this acyl ammonium ion pair in the stereodetermining step to afford the ester (S)-16, leading to enantioenrichment in the slower reacting (R)-15 azaborine alcohol. The resulting ammonium ion pair can be deprotonated by Et3N to regenerate the catalyst for turnover. By analogy to previous computational studies the observed selectivity in this acylation can be rationalised using the proposed transition state assembly in Fig. 5. Within this model it is assumed that the key factors that lead to enantiodiscrimination require a 1,5-chalcogen bonding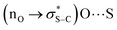 interaction between the acyl oxygen and the isothiourea catalyst sulfur that acts as a conformational lock.65–67 The carboxylate counterion is considered to activate the naphthol towards acylation by deprotonation, while simultaneously participating in non-classical H-bonding to the acylated isothiouronium ion benzylic C–H bond.68–74 To deliver high enantioselectivity, a donor substrate motif is needed to promote enantiorecognition through interaction with the positively charged acylated isothiouronium intermediate. A number of enantiorecognition motifs have been employed and recognised in isothiourea-catalysed acylations that include aryl,48,75–78 heteroaryl,79 alkenyl,78 alkynyl,75 heteroatom,80 C
interaction between the acyl oxygen and the isothiourea catalyst sulfur that acts as a conformational lock.65–67 The carboxylate counterion is considered to activate the naphthol towards acylation by deprotonation, while simultaneously participating in non-classical H-bonding to the acylated isothiouronium ion benzylic C–H bond.68–74 To deliver high enantioselectivity, a donor substrate motif is needed to promote enantiorecognition through interaction with the positively charged acylated isothiouronium intermediate. A number of enantiorecognition motifs have been employed and recognised in isothiourea-catalysed acylations that include aryl,48,75–78 heteroaryl,79 alkenyl,78 alkynyl,75 heteroatom,80 C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O,81,82 CF2,52 and P
O,81,82 CF2,52 and P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O substituents.83 In this case, utilising the benzofused aromatic substituent to participate in this capacity via a π-isothiouronium ion interaction (highlighted in red) leads to the observed selectivity for preferential acylation of the (S)-enantiomer.
O substituents.83 In this case, utilising the benzofused aromatic substituent to participate in this capacity via a π-isothiouronium ion interaction (highlighted in red) leads to the observed selectivity for preferential acylation of the (S)-enantiomer.
Conclusion
In conclusion, an isothiourea catalysed kinetic resolution of 1,2-azaborine substrates has been developed, with selectivity factors of up to >200 observed. Variation of the substitution within the azaborine at C(3)-, C(4)- or N(1)- that are in close proximity to the C–B axis directly impact the observed selectivity of acylation. The enantioenriched azaborines can be readily racemised through heating, or via the reversible formation of a tetracoordinate boron ate species. Using this knowledge, a moderately selective acylative dynamic kinetic resolution has been developed.Author contributions
M. I. P. and A. D. S. conceived the project; M. I. P. and A. D. S. designed the synthetic experiments; M. I. P. carried out all synthetic experimental studies and analysed the reactions. D. B. C. and A. P. M. carried out single crystal X-ray analysis. M. I. P. and A. D. S. wrote the manuscript. All other correspondence should be addressed to A. D. S.Conflicts of interest
The authors declare no competing interests.Data availability
CCDC 2536174 (compound (R)-15) contains the supplementary crystallographic data for this paper.108All data (experimental procedures and characterization data) that support the findings of this study are available within the article and its supplementary information (SI). The data underpinning this manuscript is available from the University of St Andrews Research Portal, Pure ID: 333325369 and can be accessed at https://doi.org/10.17630/e86c806f-4b19-4e4b-8842-1430c237adfa. References 84–107 were cited in the SI. Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc02046a.
Acknowledgements
The research leading to these results has received funding from the EaSI-CAT centre for Doctoral Training (M. I. P.). A. D. S. thanks the EPSRC Programme Grant “Boron: Beyond the Reagent” (EP/W007517) for support. We acknowledge support from Dr Tomas Lebl and Dr Siobhan Smith of the St Andrews solution state NMR facility (EP/X034747/1) plus Caroline Horsburgh and Dr Sally Shirran for help with the St Andrews mass spectrometry facility (EP/X034747/1). The authors also greatfully acknowledge Prof. Guy Lloyd-Jones (Edinburgh) for the suggestion of the homologated alcohol variant. D. B. C. and A. P. M. acknowledge support from EPSRC (UKRI381) and the University of St Andrews Strategic Equipment Fund for funding the single crystal XRD facilities at the University of St Andrews.Notes and references
- T. A. Schmidt, V. Hutskalova and C. Sparr, Nat. Rev. Chem., 2024, 8, 497–517 CrossRef PubMed.
- J. K. Cheng, S.-H. Xiang, S. Li, L. Ye and B. Tan, Chem. Rev., 2021, 121, 4805–4902 CrossRef CAS PubMed.
- J. K. Cheng, S.-H. Xiang and B. Tan, Acc. Chem. Res., 2022, 55, 2920–2937 CrossRef CAS PubMed.
- G. H. Christie and J. Kenner, J. Chem. Soc., Faraday Trans., 1922, 121, 614–620 RSC.
- M. Oki, in Topics in Stereochemistry, Wiley Interscience, 1983, vol. 1 Search PubMed.
- S. R. LaPlante, L. D. Fader, K. R. Fandrick, D. R. Fandrick, O. Hucke, R. Kemper, S. P. F. Miller and P. J. Edwards, J. Med. Chem., 2011, 54, 7005–7022 CrossRef CAS PubMed.
- S. R. LaPlante, P. J. Edwards, L. D. Fader, A. Jakalian and O. Hucke, ChemMedChem, 2011, 6, 505–513 CrossRef CAS PubMed.
- G.-J. Mei, W. L. Koay, C.-Y. Guan and Y. Lu, Chem, 2022, 8, 1855–1893 CAS.
- M. Basilaia, M. H. Chen, J. Secka and J. L. Gustafson, Acc. Chem. Res., 2022, 55, 2904–2919 CrossRef CAS PubMed.
- P. Eveleigh, E. C. Hulme, C. Schudt and N. J. Birdsall, Mol. Pharmacol., 1989, 35, 477–483 CrossRef CAS PubMed.
- J. Wang, W. Zeng, S. Li, L. Shen, Z. Gu, Y. Zhang, J. Li, S. Chen and X. Jia, ACS Med. Chem. Lett., 2017, 8, 299–303 CrossRef CAS PubMed.
- B. A. Lanman, J. R. Allen, J. G. Allen, A. K. Amegadzie, K. S. Ashton, S. K. Booker, J. J. Chen, N. Chen, M. J. Frohn, G. Goodman, D. J. Kopecky, L. Liu, P. Lopez, J. D. Low, V. Ma, A. E. Minatti, T. T. Nguyen, N. Nishimura, A. J. Pickrell, A. B. Reed, Y. Shin, A. C. Siegmund, N. A. Tamayo, C. M. Tegley, M. C. Walton, H.-L. Wang, R. P. Wurz, M. Xue, K. C. Yang, P. Achanta, M. D. Bartberger, J. Canon, L. S. Hollis, J. D. McCarter, C. Mohr, K. Rex, A. Y. Saiki, T. San Miguel, L. P. Volak, K. H. Wang, D. A. Whittington, S. G. Zech, J. R. Lipford and V. J. Cee, J. Med. Chem., 2020, 63, 52–65 CrossRef CAS PubMed.
- B. A. Lanman, A. T. Parsons and S. G. Zech, Acc. Chem. Res., 2022, 55, 2892–2903 CrossRef CAS PubMed.
- U. Berg, J. Deinum, P. Lincoln and J. Kvassman, Bioorg. Chem., 1991, 19, 53–65 CrossRef CAS.
- S. T. Toenjes and J. L. Gustafson, Future Med. Chem., 2018, 10, 409–422 CrossRef CAS PubMed.
- E. Kumarasamy, R. Raghunathan, M. P. Sibi and J. Sivaguru, Chem. Rev., 2015, 115, 11239–11300 CrossRef CAS PubMed.
- H. Sharif, L. A. McCall, M. G. Sanders and R. J. Phipps, Angew. Chem., Int. Ed., 2026, 65, e20698 CrossRef CAS PubMed.
- Y.-Y. Chen, J. Xue, Q. Shi, Y.-C. Zhang and F. Shi, Org. Lett., 2026, 28, 1481–1486 CrossRef CAS PubMed.
- J. Clayden, J. Senior and M. Helliwell, Angew. Chem., Int. Ed., 2009, 48, 6270–6273 CrossRef CAS PubMed.
- M. S. Betson, J. Clayden, C. P. Worrall and S. Peace, Angew. Chem., Int. Ed., 2006, 45, 5803–5807 CrossRef CAS PubMed.
- A. Mazzanti, E. Mercanti and M. Mancinelli, Org. Lett., 2016, 18, 2692–2695 CrossRef CAS PubMed.
- Y. Wang, Y. Nong, S. Li and Z. Jin, ChemistrySelect, 2026, 11, e04971 CrossRef CAS.
- M. J. S. Dewar, V. P. Kubba and R. Pettit, J. Chem. Soc., 1958, 3073–3076, 10.1039/JR9580003073.
- Z. Liu and T. B. Marder, Angew. Chem., Int. Ed., 2008, 47, 242–244 CrossRef CAS PubMed.
- P. G. Campbell, A. J. V. Marwitz and S.-Y. Liu, Angew. Chem., Int. Ed., 2012, 51, 6074–6092 CrossRef CAS PubMed.
- M. J. D. Bosdet and W. E. Piers, Can. J. Chem., 2009, 87, 8–29 CrossRef.
- K. Yuan, D. Volland, S. Kirschner, M. Uzelac, G. S. Nichol, A. Nowak-Król and M. J. Ingleson, Chem. Sci., 2022, 13, 1136–1145 RSC.
- F. J. R. Rombouts, F. Tovar, N. Austin, G. Tresadern and A. A. Trabanco, J. Med. Chem., 2015, 58, 9287–9295 CrossRef CAS PubMed.
- M. Chen, K. S. Unikela, R. Ramalakshmi, B. Li, C. Darrigan, A. Chrostowska and S.-Y. Liu, Angew. Chem., Int. Ed., 2021, 60, 1556–1560 CrossRef CAS PubMed.
- D. H. Knack, J. L. Marshall, G. P. Harlow, A. Dudzik, M. Szaleniec, S.-Y. Liu and J. Heider, Angew. Chem., Int. Ed., 2013, 52, 2599–2601 CrossRef CAS PubMed.
- P. Zhao, D. O. Nettleton, R. G. Karki, F. J. Zécri and S.-Y. Liu, ChemMedChem, 2017, 12, 358–361 CrossRef CAS PubMed.
- Y. Zhang, F. Cheng, X. Ye, X. Du, X. Li, Y. Deng and Q. Wu, Org. Lett., 2026, 28, 1054–1059 CrossRef CAS PubMed.
- Z. X. Giustra and S.-Y. Liu, J. Am. Chem. Soc., 2018, 140, 1184–1194 CrossRef CAS PubMed.
- Y. Zhang, C. Zhang, Y. Guo, J. Ye, B. Zhen, Y. Chen and X. Liu, J. Org. Chem., 2021, 86, 6322–6330 CrossRef CAS PubMed.
- J. S. A. Ishibashi, A. Dargelos, C. Darrigan, A. Chrostowska and S.-Y. Liu, Organometallics, 2017, 36, 2494–2497 CrossRef CAS.
- B. A. Haney, C. L. Schrank and W. M. Wuest, Tetrahedron Lett., 2021, 62, 152667 CrossRef CAS PubMed.
- L. Liu, A. J. V. Marwitz, B. W. Matthews and S.-Y. Liu, Angew. Chem., Int. Ed., 2009, 48, 6817–6819 CrossRef CAS PubMed.
- K. Yang, Y. Mao, Z. Zhang, J. Xu, H. Wang, Y. He, P. Yu and Q. Song, Nat. Commun., 2023, 14, 4438 CrossRef CAS PubMed.
- H. Wang, B. Qiao, J. Zhu, H. Guo, Z. Zhang, K. Yang, S.-J. Li, Y. Lan and Q. Song, Chem, 2024, 10, 317–329 CAS.
- Y. Ping, X. Shi, M. Lei and J. Wang, ACS Catal., 2024, 14, 5064–5076 CrossRef CAS.
- W. Qiu, R. Tao, Y. He, Y. Zhou, K. Yang and Q. Song, Nat. Commun., 2024, 15, 10432 CrossRef CAS PubMed.
- X.-L. Zhang, J. Gu, W.-H. Cui, Z. Ye, W. Yi, Q. Zhang and Y. He, Angew. Chem., 2022, 134, e202210456 CrossRef.
- J. Xu, W. Qiu, X. Zhang, Z. Wu, Z. Zhang, K. Yang and Q. Song, Angew. Chem., Int. Ed., 2023, 62, e202313388 CrossRef CAS PubMed.
- J.-P. Zhang, W.-J. Liu, Y. Hu, E. Hao and Z.-Y. Li, Org. Lett., 2026, 28, 394–399 CrossRef CAS PubMed.
- J. Yang, J.-W. Zhang, W. Bao, S.-Q. Qiu, S. Li, S.-H. Xiang, J. Song, J. Zhang and B. Tan, J. Am. Chem. Soc., 2021, 143, 12924–12929 CrossRef CAS PubMed.
- Y. Huang, X.-N. Li, X.-H. Wang, Z.-X. Wang, S. Ye and C.-L. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202501991 CrossRef CAS PubMed.
- V. B. Birman and X. Li, Org. Lett., 2006, 8, 1351–1354 CrossRef CAS PubMed.
- S. F. Musolino, O. S. Ojo, N. J. Westwood, J. E. Taylor and A. D. Smith, Chem. – Eur. J., 2016, 22, 18916–18922 CrossRef CAS PubMed.
- R. M. Neyyappadath, R. Chisholm, M. D. Greenhalgh, C. Rodríguez-Escrich, M. A. Pericàs, G. Hähner and A. D. Smith, ACS Catal., 2018, 8, 1067–1075 CrossRef CAS.
- M. T. Westwood, A. Omar Farah, H. B. Wise, M. Sinfield, C. Robichon, M. I. Prindl, D. B. Cordes, P. Ha-Yeong Cheong and A. D. Smith, Angew. Chem., 2024, 136, e202407983 CrossRef.
- S. Harrer, M. D. Greenhalgh, R. M. Neyyappadath and A. D. Smith, Synlett, 2019, 30, 1555–1560 CrossRef CAS.
- T. Desrues, J. Merad, D. Andrei, J.-M. Pons, J.-L. Parrain, M. Médebielle, A. Quintard and C. Bressy, Angew. Chem., Int. Ed., 2021, 60, 24924–24929 CrossRef CAS PubMed.
- A. J. Nimmo, A. S. Goodfellow, J. T. Guntley, A. P. McKay, D. B. Cordes, M. Bühl and A. D. Smith, Chem. Sci., 2025, 16, 10494–10502 RSC.
- J.-Y. Ong, X. Q. Ng, S. Lu and Y. Zhao, Org. Lett., 2020, 22, 6447–6451 CrossRef CAS PubMed.
- T.-Z. Li, S.-F. Wu, N.-Y. Wang, C.-S. Hong, Y.-C. Zhang and F. Shi, J. Org. Chem., 2024, 89, 12559–12575 CrossRef CAS PubMed.
- S. Qu, M. D. Greenhalgh and A. D. Smith, Chem. – Eur. J., 2019, 25, 2816–2823 CrossRef CAS PubMed.
- E. S. Munday, M. A. Grove, T. Feoktistova, A. C. Brueckner, D. M. Walden, C. M. Young, A. M. Z. Slawin, A. D. Campbell, P. H.-Y. Cheong and A. D. Smith, Angew. Chem., Int. Ed., 2020, 59, 7897–7905 CrossRef CAS PubMed.
- T.-T. Wang, Z.-P. Hu, J. Lin, H. Xu and X. Li, Chin. J. Chem., 2026, 44, 1539–1545 CrossRef CAS.
- M. D. Greenhalgh, J. E. Taylor and A. D. Smith, Tetrahedron, 2018, 74, 5554–5560 CrossRef CAS.
- M. Rieger and F. H. Westheimer, J. Am. Chem. Soc., 1950, 72, 19–28 CrossRef CAS.
- S. L. Chien and R. Adams, J. Am. Chem. Soc., 1934, 56, 1787–1792 CrossRef CAS.
- J.-P. Heeb, J. Clayden, M. D. Smith and R. J. Armstrong, Nat. Protoc., 2023, 18, 2745–2771 CrossRef CAS PubMed.
- DCXplorerMCXVII, https://www.cup.lmu.de/oc/trapp/tools.html, accessed 2026 Search PubMed.
- O. Trapp, in Differentiation of Enantiomers II, ed. V. Schurig, Springer International Publishing, Cham, 2013, pp. 231–269, DOI:10.1007/128_2013_453.
- S. Kolb, G. A. Oliver and D. B. Werz, Angew. Chem., Int. Ed., 2020, 59, 22306–22310 CrossRef CAS PubMed.
- C. Bleiholder, R. Gleiter, D. B. Werz and H. Köppel, Inorg. Chem., 2007, 46, 2249–2260 CrossRef CAS PubMed.
- R. Gleiter, G. Haberhauer, D. B. Werz, F. Rominger and C. Bleiholder, Chem. Rev., 2018, 118, 2010–2041 CrossRef CAS PubMed.
- E. Larionov, M. Mahesh, A. C. Spivey, Y. Wei and H. Zipse, J. Am. Chem. Soc., 2012, 134, 9390–9399 CrossRef CAS PubMed.
- P. Liu, X. Yang, V. B. Birman and K. N. Houk, Org. Lett., 2012, 14, 3288–3291 CrossRef CAS PubMed.
- X. Yang, P. Liu, K. N. Houk and V. B. Birman, Angew. Chem., Int. Ed., 2012, 51, 9638–9642 CrossRef CAS PubMed.
- R. C. Johnston and P. H.-Y. Cheong, Org. Biomol. Chem., 2013, 11, 5057–5064 RSC.
- C. E. Cannizzaro and K. N. Houk, J. Am. Chem. Soc., 2002, 124, 7163–7169 CrossRef CAS PubMed.
- V. Lutz, J. Glatthaar, C. Würtele, M. Serafin, H. Hausmann and P. R. Schreiner, Chem. Eur J., 2009, 15, 8548–8557 CrossRef CAS PubMed.
- S. Xu, I. Held, B. Kempf, H. Mayr, W. Steglich and H. Zipse, Chem. Eur J., 2005, 11, 4751–4757 CrossRef CAS.
- X. Li, H. Jiang, E. W. Uffman, L. Guo, Y. Zhang, X. Yang and V. B. Birman, J. Org. Chem., 2012, 77, 1722–1737 CrossRef CAS PubMed.
- Q. Xu, H. Zhou, X. Geng and P.-R. Chen, Tetrahedron, 2009, 65, 2232–2238 CrossRef CAS.
- P.-R. Chen, Y. Zhang, H. Zhou and Q. Xu, Acta Chim. Sin., 2010, 68, 1431–1436 CAS.
- D. Belmessieri, C. Joannesse, P. A. Woods, C. MacGregor, C. Jones, C. D. Campbell, C. P. Johnston, N. Duguet, C. Concellón, R. A. Bragg and A. D. Smith, Org. Biomol. Chem., 2011, 9, 559–570 RSC.
- I. Shiina, K. Ono and K. Nakata, Chem. Lett., 2011, 40, 147–149 CrossRef CAS.
- S. K. Agrawal, P. K. Majhi, A. S. Goodfellow, R. K. Tak, D. B. Cordes, A. P. McKay, K. Kasten, M. Bühl and A. D. Smith, Angew. Chem., Int. Ed., 2024, 63, e202402909 CrossRef CAS PubMed.
- I. Shiina, K. Nakata, K. Ono, M. Sugimoto and A. Sekiguchi, Chem. Eur J., 2010, 16, 167–172 CrossRef CAS PubMed.
- K. Nakata, K. Gotoh, K. Ono, K. Futami and I. Shiina, Org. Lett., 2013, 15, 1170–1173 CrossRef CAS PubMed.
- I. Shiina, K. Ono and T. Nakahara, Chem. Commun., 2013, 49, 10700–10702 RSC.
- W. R. Scaggs and T. N. Snaddon, Chem. – Eur. J., 2018, 24, 14378–14381 CrossRef CAS PubMed.
- H. B. Kagan and J. C. Fiaud, in Topics in Stereochemistry, John Wiley & Sons, Ltd, 1988, pp. 249–330, DOI:10.1002/9780470147276.ch4.
- S. R. Wisniewski, C. L. Guenther, O. A. Argintaru and G. A. Molander, J. Org. Chem., 2014, 79, 365–378 CrossRef CAS PubMed.
- H. Zhu, A. Manchado, A. Omar Farah, A. P. McKay, D. B. Cordes, P. H.-Y. Cheong, K. Kasten and A. D. Smith, Angew. Chem., 2024, 136, e202402908 CrossRef.
- R. Del Río-Rodríguez, M. T. Westwood, M. Sicignano, M. Juhl, J. A. Fernández-Salas, J. Alemán and A. D. Smith, Chem. Commun., 2022, 58, 7277–7280 RSC.
- X. Chang, P.-L. Ma, H.-C. Chen, C.-Y. Li and P. Wang, Angew. Chem., 2020, 132, 9022–9025 CrossRef.
- I. R. Hazelden, R. C. Carmona, T. Langer, P. G. Pringle and J. F. Bower, Angew. Chem., Int. Ed., 2018, 57, 5124–5128 CrossRef CAS.
- G. Chen, J. Gui, L. Li and J. Liao, Angew. Chem., Int. Ed., 2011, 50, 7681–7685 CrossRef CAS PubMed.
- M. Barbasiewicz, K. Błocki, M. Malińska and R. Pawłowski, Dalton Trans., 2013, 42, 355–358 RSC.
- H. Albright, H. L. Vonesh and C. S. Schindler, Org. Lett., 2020, 22, 3155–3160 CrossRef CAS PubMed.
- A.-M. L. Hogan and D. F. O'Shea, J. Am. Chem. Soc., 2006, 128, 10360–10361 CrossRef CAS PubMed.
- A.-M. L. Hogan and D. F. O'Shea, J. Org. Chem., 2008, 73, 2503–2509 CrossRef CAS PubMed.
- A. D. J. Calow, D. Dailler and J. F. Bower, J. Am. Chem. Soc., 2022, 144, 11069–11074 CrossRef CAS.
- M. Weimar, R. Correa Da Costa, F.-H. Lee and M. J. Fuchter, Org. Lett., 2013, 15, 1706–1709 CrossRef CAS PubMed.
- J. Caeiro, D. Peña, A. Cobas, D. Pérez and E. Guitián, Adv. Synth. Catal., 2006, 348, 2466–2474 CrossRef CAS.
- B. Satpathi, L. Dutta and S. S. V. Ramasastry, Org. Lett., 2019, 21, 170–174 CrossRef CAS.
- D. Moser, K. Jana and C. Sparr, Angew. Chem., 2023, 135, e202309053 CrossRef.
- G. A. Molander and S. R. Wisniewski, J. Org. Chem., 2014, 79, 6663–6678 CrossRef CAS PubMed.
- H. Xu, T. Du, J. Lin, F. You, Y. Shao, Q. Yang and X. Li, Angew. Chem., Int. Ed., 2026, 65, e21349 CrossRef CAS.
- T.-H. Doan, A. Chardon, N. Vanthuyne, T. N. Ramos, N. Tumanov, L. Fusaro, M. Albalat, L. Collard, J. Wouters, B. Champagne and G. Berionni, Angew. Chem., Int. Ed., 2025, 64, e202421931 CrossRef CAS PubMed.
- CrysAlisPro v1.171.44.121a, Rigaku Oxford Diffraction, Rigaku Corporation, Tokyo, Japan, 2025 Search PubMed.
- G. M. Sheldrick, Acta Crystallogr. A, 2015, 71, 3–8 CrossRef PubMed.
- G. M. Sheldrick, Acta Crystallogr. C, 2015, 71, 3–8 CrossRef PubMed.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. a. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- CCDC 2536174: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2r4323.
| This journal is © The Royal Society of Chemistry 2026 |