Open Access Article
Open Access ArticleBeyond static paradigms: defect dynamic evolution and advanced applications in water electrolysis
Xiaojun
Wang
a,
Huilin
Zhao
a,
Lei
Li
 *b,
Weiping
Xiao
*b,
Weiping
Xiao
 c,
Guangrui
Xu
c,
Guangrui
Xu
 d,
Guangying
Fu
*e,
Lei
Wang
d,
Guangying
Fu
*e,
Lei
Wang
 *a and
Zexing
Wu
*a and
Zexing
Wu
 *a
*a
aState Key Laboratory of Advanced Optical Polymer and Manufacturing Technology, Key Laboratory of Eco-chemical Engineering, Ministry of Education, International Science and Technology Cooperation Base of Eco-chemical Engineering and Green Manufacturing, College of Chemistry and Molecular Engineering, Qingdao University of Science & Technology, 53 Zhengzhou Road, Qingdao, 266042, P. R. China. E-mail: splswzx@qust.edu.cn; inorchemwl@126.com
bCollege of Biological, Chemical Sciences and Engineering, Jiaxing University, Jiaxing, Zhejiang 314001, China. E-mail: leili@mail.zjxu.edu.cn
cCollege of Science, Nanjing Forestry University, Nanjing, 210037, P. R. China
dCollege of Materials Science and Engineering, Qingdao University of Science & Technology, 53 Zhengzhou Road, Qingdao, 266042, P. R. China
eKey Laboratory of Photoelectric Conversion and Utilization of Solar Energy, Qingdao Institute of Bioenergy and Bioprocess Technology, Chinese Academy of Sciences, CN-266101 Qingdao, China. E-mail: fugy@qibebt.ac.cn
First published on 6th May 2026
Abstract
Electrocatalytic reactions play crucial roles in energy conversion and green chemical synthesis; however, their practical applications are often constrained by sluggish reaction kinetics, high overpotentials, and insufficient operational stability. In recent years, defect engineering has emerged as an effective structural regulation strategy, providing new opportunities to enhance electrocatalytic performance through the deliberate introduction of non-ideal structural features, such as vacancies, heteroatom dopants and interfacial architectures. Accumulating studies have demonstrated that defects not only modulate the electronic structure of catalysts and reaction pathways, but that their dynamic evolution under specific reaction conditions also exerts profound influence on the real working-state structure and long-term catalytic performance. Owing to central importance in green hydrogen production, water-splitting reactions serve as representative model systems for elucidating defect–structure–activity relationships. In this review, we systematically summarize recent advances in defect engineering for electrocatalysis, with particular focus on water electrolysis. Emphasis is placed on defect types and structural characteristics, construction strategies, dynamic evolution behaviors, and their typical applications, while current challenges and future research directions are also discussed, aiming to provide valuable guidance for the rational design and practical implementation of high-performance defect-engineered electrocatalysts.
1 Introduction
Electrocatalytic reactions play central roles in energy conversion and green chemical synthesis, with broad applications in hydrogen production, carbon dioxide conversion, nitrogen-cycle reactions, and electrochemical energy storage systems.1–3 Compared with conventional thermocatalytic processes, electrocatalysis relies on externally applied electric fields to drive electron-transfer reactions, offering advantages such as mild operating conditions, high energy utilization efficiency, and strong process tunability.4 However, most key electrocatalytic reactions intrinsically involve complex multi-electron transfer processes and coupled reaction intermediates, leading to sluggish reaction kinetics, high overpotentials, and insufficient operational stability. These issues continue to severely limit the further development of electrocatalytic technologies.5,6 Consequently, achieving fundamental understanding and rational regulation of structure–activity relationships from the materials-structure perspective has emerged as one of the central scientific challenges in contemporary electrocatalysis.The electronic structure of catalysts, their local coordination environments, and interfacial reaction properties are widely recognized as decisive factors governing electrocatalytic activity and selectivity.7 Conventional catalysts based on ideal crystalline structures often struggle to simultaneously optimize active-site exposure, charge-transfer efficiency, and structural stability, which has prompted increasing research interest in non-ideal structural systems. As ubiquitous non-periodic features in crystalline materials, defects can effectively break local symmetry and electronic neutrality, generating low-coordination, highly reactive catalytic sites on surfaces or interfaces, while markedly modulating the electronic states and interfacial reaction behavior of catalysts.8
In recent years, defect engineering has emerged as a key strategy for electrocatalytic material design. By introducing vacancy defects, heteroatom dopants, grain boundaries (GBs), and heterointerfaces, the electronic structure and catalytic performance can be precisely regulated across different spatial scales.9 For example, Arandiyan et al. systematically investigated the effect of oxygen vacancies (VO) on the oxygen evolution reaction (OER) performance using LaNiO3 with abundant VO and vacancy-free LaNiO3 as model systems, revealing the crucial role of defects in enhancing electrical conductivity, modulating the electronic structure, and improving catalytic activity.10 In addition, CeO2−x/MoO3−x/RuO2 heterointerface defect systems were employed to further elucidate the influence of different defect configurations on the hydrogen evolution reaction (HER) and catalytic ozonation performance.11 Moreover, defect engineering strategies have also been extended to the nitrate reduction reaction, highlighting their broad applicability across diverse electrocatalytic processes.12 Overall, defect-engineered catalysts often exhibit superior performance compared with their ideal crystalline counterparts across wide ranges of electrocatalytic reactions.13
Notably, defects are no longer regarded as static structural features with the continued advancement of research. Increasing evidence from in situ and operando characterization studies indicates that defects undergo continuous generation, migration, aggregation, and reconstruction during material synthesis and practical electrocatalytic operation. Such dynamic evolution processes largely determine the true working-state structure of catalysts and their long-term performance.14 Consequently, structure–activity relationships established solely on the basis of static characterization exhibit inherent limitations, and reassessing the role of defects in electrocatalysis from dynamic perspectives has emerged as an important research direction.
Among various electrocatalytic reaction systems, water electrolysis occupies a particularly prominent status due to critical roles in green hydrogen production and renewable energy conversion.15 Along with its well-defined reaction pathways and significant industrial relevance, it serves as an ideal model system for elucidating the dynamic evolution mechanisms of defects and their structure–activity relationships.16,17 In the context of the HER and OER, defect engineering has demonstrated pronounced advantages in enhancing catalytic activity, regulating reaction pathways, and improving structural stability, thereby providing representative model systems for elucidating the intrinsic relationships among defects, structure, and catalytic performance.18
Based on this background, this review systematically surveys and critically analyzes recent progress in defect engineering for electrocatalytic systems, with particular focus on roles in structural regulation and structure–activity relationships. Emphasis is placed on defect types and structural characteristics, construction strategies, dynamic evolution behavior, and representative applications in water electrolysis (Scheme 1), with the aim of serving as comprehensive guidance for rational design and practical implementation of defect-engineered electrocatalysts.
2 Classification of defects and structure–activity relationships
Defects generally refer to localized disordered states in crystalline materials that deviate from ideal periodic lattice arrangements, including the absence, displacement, and substitution of atoms or ions, as well as the incorporation of heteroatoms. The introduction of defects disrupts local charge neutrality and lattice symmetry, inducing charge redistribution and generating non-ideal coordination sites with distinct chemical reactivity. From a structure–property perspective, deliberately engineered defects with tailored types and spatial distributions can effectively modulate the electronic density of states, surface energy, and adsorption behavior of reaction intermediates.19,20 These modifications in turn govern the catalytic reaction energetics and kinetics, enabling the synergistic optimization of catalytic activity and stability.21,222.1 Types of defects
From the perspective of structural scale and spatial extension, crystal defects can generally be classified into point defects, line defects, planar defects, and bulk defects (Fig. 1 illustrates various defects and their corresponding features in multi-dimensional crystals). This classification framework provides a fundamental basis for understanding defect characteristics and their structure–activity relationships. Point defects refer to localized lattice imperfections at the atomic scale (approximately 0.1–1 nm), involving the absence, displacement, or additional introduction of single atoms or a small number of atoms. They represent the most common type of defects. Typical point defects include vacancies and interstitial defects. Vacancy defects arise when atoms or ions that should occupy lattice sites in an ideal crystal are missing. Among them, VO are particularly representative, as they can significantly alter local charge distribution and coordination environments, exposing more low-coordinated and highly reactive sites on catalyst surfaces. This effect facilitates the adsorption and transformation of key reaction intermediates, reducing reaction energy barriers and overpotentials.23,24 In contrast, interstitial defects are formed when relatively small atoms or ions occupy interstitial sites within the lattice. Such defects can induce local lattice expansion or distortion, regulate interlayer spacing, and improve electronic transport properties, while simultaneously increasing the accessibility of active sites and enhancing overall electrocatalytic performance.25 Line defects are typically represented by dislocations. The presence of dislocations leads to continuous lattice distortion, causing atoms in the vicinity of the dislocation core to adopt high-energy and asymmetrically coordinated configurations, which can act as potential highly active reaction sites. Moreover, dislocations are often accompanied by pronounced local strain fields. These strain effects can effectively modulate the electronic structure of metal centers and optimize the adsorption energies of reaction intermediates (such as *OH and *O), thereby accelerating electrocatalytic reaction kinetics.26,27 | ||
| Fig. 1 Schematic illustrating various defects and their respective features in crystals with multiple dimensions.33 Copyright 2020 Elsevier B.V. | ||
Surface defects mainly include GBs and stacking faults. GBs are interfacial regions between crystallites with different orientations, characterized by highly disordered atomic arrangements, abundant dangling bonds, and elevated interfacial energies, making them intrinsically active regions for electrocatalytic reactions. In electrocatalytic systems, the introduction of GBs is often associated with an increased concentration of vacancy defects (e.g., VO), which enhances electron transport and markedly improves catalytic activity.28 In addition, stacking faults refer to deviations from the ideal periodic stacking sequence of atomic layers in crystals. Such faults disrupt the inherent stacking symmetry and can stabilize associated dislocations, leading to localized lattice distortion regions. These regions are typically enriched with twin boundaries, steps, and corner atomic configurations, which introduce strain effects and strongly modulate the electronic structure of the material, ultimately influencing its electrocatalytic performance.29,30 Bulk defects are three-dimensionally distributed imperfections that deviate from ideal crystal periodicity, with characteristic sizes ranging from the atomic scale to micrometers or even larger. They are predominantly located within the interior of materials rather than being confined to surfaces or interfaces. Cavities represent representative forms of bulk defects. The internal space and edge regions of cavities can provide large numbers of exposed low-coordination active sites, while also facilitating adsorption, mass transport, and rapid release of reaction intermediates and gaseous products. These features promote sustained catalytic reactions and contribute to improved long-term stability of the electrocatalytic system.31,32 In the following sections, we will systematically review three representative types of defects and discuss in detail their structure–activity relationships as well as the underlying mechanisms in electrocatalytic processes.
In recent years, anion vacancies-such as oxygen, sulfur, selenium, and phosphorus vacancies-have been extensively investigated in electrocatalysis and proven to be effective for tuning catalytic performance.34,35 Among them, VO are the most representative anion vacancies in metal oxides. Their formation directly modifies the local coordination environment of metal centers and is often accompanied by localized electron trapping and redistribution. Consequently, defect states are introduced near the band edges, leading to bandgap narrowing or enhanced effective electrical conductivity, facilitating electron transport and interfacial charge-transfer kinetics.36 Extensive experimental and theoretical studies demonstrated that the introduction of VO in TiO2 induced bandgap narrowing and localized electronic states (Fig. 2a). These electronic structure modulations facilitate charge transport and optimize reaction-related electronic processes, thereby establishing a clear structure–property relationship between defect configuration and catalytic performance.37 For metal sulfides, intrinsically low electrical conductivity and sluggish interfacial charge-transfer kinetics severely limit their practical electrocatalytic applications. To address these challenges, the introduction of sulfur vacancies (VS) has been recognized as an effective regulation strategy.38 Researchers39 proposed VS-induced local strain strategies to enhance d–p orbital hybridization. The concentrated tensile strain introduced by VS significantly lowered the Co d-orbital energy levels and shifted the d-band center upward, strengthening Co-3dx2−y2/O-2pz orbital hybridization and facilitating charge transfer and accelerated redox kinetics, underscoring the strong coupling between defect-induced structural distortion and catalytic performance (Fig. 2b). However, it should be noted that VS often impart high lattice distortion energies and complex atomic rearrangements to metal sulfides. Moreover, their elevated surface energies may lead to structural instability during long-term cycling, ultimately resulting in performance degradation.40 To overcome the insufficient stability associated with VS, Hui et al.41 introduced In atoms into VS-MoS2 to construct an In-VS-MoS2 system. It was found that the formation energy of VS near In sites (1.61 eV) was substantially lower than that near Mo sites (2.99 eV, Fig. 2c). Meanwhile, the surface energy of pristine MoS2 was 2.94 eV, which increased to 8.23 eV after VS formation, whereas In doping reduced it to 4.39 eV (Fig. 2d). These results further reveal the role of dopants in regulating defect energetics and stabilizing the catalyst framework.
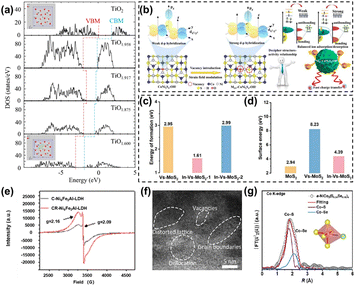 | ||
| Fig. 2 (a) Total density of states (TDOS) of TiO2 and TiO2−x near the Fermi level.36 Copyright 2023 Springer Nature. (b) Theoretical models of CoNi2S4–OH− and MSV-CoNi2S4–OH− and schematic illustration for their various d–p orbital hybridization states induced by the VS-modulated strain field and the corresponding material feature.39 Copyright 2025 Wiley-VCH. (c) Energy of formation of VS-MoS2, In-VS-MoS2-1, and In-VS-MoS2-2.41 Copyright 2025 Wiley-VCH. (d) Surface energy of MoS2, VS-MoS2, and In-VS-MoS2.41 Copyright 2025 Wiley-VCH. (e) Electron paramagnetic resonance (EPR) spectra of C-Ni6Fe2Al-LDH and CR-Ni6Fe2Al-LDH.45 Copyright 2024 Royal Society of Chemistry. (f) High-resolution transmission electron microscopy (HRTEM) image of Px-CoMoO4−x/NF.50 Copyright 2023 Wiley-VCH. (g) X-ray absorption spectroscopy (XAS) fitting in R space at the Co K-edge of a-NiCo2(S0.84Se0.16)4.52 Copyright 2025 Tsinghua University Press. | ||
Similar to anion vacancies, the construction of cation vacancies is also an important strategy for enhancing intrinsic catalytic activity, with comparable mechanisms for regulating electronic structures and reaction kinetics. Notably, cation vacancies generally exhibit higher structural stability under electrocatalytic conditions than anion vacancies.42 However, owing to the relatively large atomic mass of metal cations, high migration barriers, and strong metal–anion bonding, the removal of metal cations from lattices is thermodynamically and kinetically unfavorable, making the construction of high-purity single cation vacancies under mild conditions highly challenging. To address this limitation, sacrificial dopants have been introduced as universal strategies for generating cation vacancies.43 Wang et al.44 selectively leached Zn via electrochemical reconstruction to successfully fabricate Zn-vacancy-modified NiFeOOH. The introduction of Zn vacancies stabilized Fe sites as “hydroxyl (OH) pumps”, enhancing local OH adsorption and accelerating VO refilling through atomically unhindered spillover pathways. In addition, Zn vacancies optimized the O 2p band center and strengthened metal–oxygen (M–O) covalency, activating lattice oxygen (OL) participation in the reaction. This work clearly linked vacancy-induced electronic and geometric modifications with enhanced OER kinetics and structural robustness under lattice oxygen mechanism (LOM) operation. Beyond single vacancies, cation–anion composite vacancies can integrate multiple vacancy effects to synergistically optimize reaction pathways. Researchers45 selectively removed Al from NiFeAl-LDH via alkaline etching to generate cation vacancies, followed by electrochemical redox treatment to introduce VO, successfully constructing composite vacancies. EPR measurements confirmed the presence of such composite defects (Fig. 2e). Electrochemical evaluations demonstrated that these composite vacancies not only enhanced electrical conductivity, charge-transfer capability, and ion transport rates but also reduced the activation energy barrier for the OER, providing valuable insights for constructing uniformly distributed cation–anion composite vacancy defects.
Non-metal elements, such as C, N, P, and S, are commonly used dopants in electrocatalytic materials. Their incorporation can effectively tune electronic structures,48,49 create additional active sites, and lower reaction kinetic barriers. Qi et al.50 synthesized a series of P-doped CoMoO4 nanowires. HRTEM and X-ray photoelectron spectroscopy (XPS) analyses identified high densities of lattice defects induced by P doping, including vacancies, dislocations and lattice distortions (Fig. 2f), which increased the density of unsaturated active sites and significantly modulated the electronic structure of the active metal centers. The formation of these defects was mainly attributed to charge imbalance caused by partial substitution of MoO42− groups with PO43−, as well as structural distortions induced by differences in the atomic radius. These results indicate that P-doping-driven defect engineering plays a decisive role in tuning the structure–electronic property relationship of CoMoO4. Compared with S and P doping, Se exhibits a broader oxidation-state range and more stable intermediate valence states, endowing it with unique advantages in electronic structure regulation.51 Researchers52 reported that Se-doped NiCo2S4 samples with different Se contents all exhibited amorphous characteristics, highlighting that defect-rich amorphous phases could be effectively engineered via heteroatom doping. Owing to the larger atomic radius of Se, lattice strain and mismatch were introduced, promoting strain relaxation in NiCo2S4 hollow nanorods and inducing localized structural dissociation and pore formation. XAS further confirmed that Se incorporation significantly increased the electronic density around Co centers and induced severe local lattice distortion (Fig. 2g), indicating enhanced metal–ligand bond flexibility in the amorphous framework and ultimately reducing the OER rate-determining step (RDS) energy barrier from 4.11 eV to 2.81 eV and markedly accelerating reaction kinetics. In addition to non-metal dopants, metal doping has also been widely employed to construct efficient electrocatalysts. Zhang et al.53 successfully fabricated an interstitial Mn-doped NiFeSe2 (Ni0.8Fe0.2Mn0.1Se2) self-supported anode. Mn doping enhanced the electronic structure of NiFeSe2 through orbital hybridization and interstitial [MnSe4] tetrahedral formation, which elevated the Fermi level and narrowed the bandgap. These electronic structure modulations governed charge transport properties and were responsible for the accelerated charge-transfer kinetics.
Overall, vacancy defects can significantly enhance the intrinsic catalytic activity by introducing unsaturated coordination sites and modulating the electronic structure. However, owing to their high surface energy, they often suffer from limited structural stability during long-term operation. In contrast, doping-induced defects offer a more controllable strategy for tuning the electronic structure and defect energetics, which not only improves electrical conductivity but also stabilizes the catalyst framework; nevertheless, their effectiveness in enhancing catalytic activity strongly depends on the nature and physicochemical properties of the dopants. Heterointerface defects, arising from lattice mismatch and electronic coupling between different phases, enable synergistic regulation of charge distribution and adsorption energetics, thereby allowing simultaneous improvement in catalytic activity and structural stability to a certain extent. Clarifying the functional roles of different defect types provides important guidance for the rational design of defect-engineered catalysts.
3 Synthesis methods
The rational selection and optimized combination of synthetic strategies are the technical foundation for the precise construction and controllable regulation of defects, and the characteristics of different methods in defect control are systematically summarized as follows.3.1 Chemical approaches
Chemical treatments represent one of the most commonly employed and effective strategies for constructing defect-rich electrocatalysts. Compared with physical methods, chemical approaches are generally conducted under relatively mild conditions. The generation of defects is jointly governed by thermodynamic equilibrium and reaction kinetics, which affords greater tunability but with relatively limited defect concentration.56 Various defect structures favorable for electrocatalytic performance, including vacancies, heteroatom substitutions and localized structural reconstructions, are achievable through the rational design of reaction environments and tailored chemical treatment pathways. | ||
| Fig. 3 (a) Illustration of the synthetic procedure of GA/NCO-2 and the effect of reaction duration on the structure.60 Copyright 2024 Wiley-VCH. (b) Aberration-corrected transmission electron microscopy (AC-TEM) image of Ru1/D-NiFe LDH (isolated Ru atoms are marked with yellow circles).66 Copyright 2021 Springer Nature. (c) E-CoFe LDHs after HNO3 etching.67 Copyright 2017 Royal Society of Chemistry. (d) Evolution of the different structures of MoS2.68 Copyright 2019 American Chemical Society. | ||
Beyond the construction of point defects, conventional pyrolysis can also be employed to fabricate heterojunction electrocatalysts through in situ oxidation or nitridation of the host matrix. Zhang et al.72 synthesized Mo2N/MoO3@NC-30 heterojunction porous nanosheets via two-step pyrolysis routes (Fig. 4a). In this process, urea first decomposed at 500 °C under an inert atmosphere to generate carbon nitride and ammonia. The generated ammonia subsequently serves as a nitrogen source to react with the molybdenum precursor at 800 °C, forming Mo2N. Meanwhile, carbon nitride further decomposed at elevated temperatures to yield nitrogen-doped carbon (NC), resulting in Mo2N nanoparticles supported on an NC matrix (Mo2N@NC). On this basis, controlled partial oxidation of Mo2N at 300 °C produced the Mo2N/MoO3@NC-30 heterostructure enriched with interfacial vacancy defects arising from lattice mismatch. XPS analysis demonstrated enhanced interfacial polarization arising from interface defects, which promoted intermediate adsorption and charge transfer. In addition, conventional pyrolysis is widely used to introduce heteroatom dopants such as O, S, F, B, and P into defect-rich electrocatalysts.73–77 The incorporation of these heteroatoms not only induces defect structures within the lattice but also provides anchoring sites for stabilizing metal active centers. By regulating local electronic distributions, such dopants further optimize reaction kinetics and contribute to enhanced electrocatalytic performance.
 | ||
| Fig. 4 (a) Schematic diagram of the preparation process for Mo2N/MoO3@NC-30 heterojunction catalysts.72 Copyright 2026 IOP Publishing Ltd. (b) Schematic diagram of the synthesis process of VO-Co3O4@NF and VS-Co3S4@NF.80 Copyright 2022 Elsevier B.V. (c) Schematic illustration of Pt single-atom-supported α-NixFeγ(OH)2.81 Copyright 2022 Wiley-VCH. | ||
3.2 Physical approaches
Physical methods constitute an important class of strategies for defect engineering in electrocatalysts. The fundamental concept lies in breaking the intrinsic chemical bonds and long-range atomic ordered structure of materials through externally applied high-energy fields, thereby inducing lattice defects and modulating the electronic structure and surface reactivity of catalysts. Unlike synthesis routes that rely on chemical reaction pathways, physical methods typically achieve nonequilibrium structural regulation via transient energy input, featuring short reaction times, rapid kinetics and the ability to generate diverse defect types.82–84 Compared with conventional thermal treatments or pyrolysis processes that often require hours or even days, physical irradiation processes can usually be completed within seconds to minutes, making them particularly advantageous for the efficient construction of defect-rich and tunable nano-electrocatalysts.3.2.1.1 Plasma treatment. Plasma is a partially ionized gas composed of electrons, ions, excited or ground-state atoms and molecules, free radicals, and photons. It is typically generated by gas discharge at high temperature, or under strong electric fields or high-energy conditions and is regarded as the “fourth state of matter”, distinct from solids, liquids, and gases.85–87 During plasma treatment, energy is transferred to the material surface in the form of ion bombardment or photon irradiation, inducing surface structural reconstruction as the energy dissipates. When high-energy ions interact with surface atoms of the catalyst, energy transfer can cause localized covalent bond breakage and surface atom ejection, thereby generating various vacancies and coordinatively unsaturated sites on the material surface.88 This makes plasma treatment an effective approach for constructing defect-rich catalysts. Liu et al.89 employed Ar plasma etching to fabricate NiSe2 nanosheet arrays enriched with Se defects. The introduced Se vacancies not only significantly increased the number of HER active sites but also accelerated charge-transfer kinetics by modulating the local electronic structure. Notably, plasma treatment can also enable the simultaneous construction of multiple types of vacancies. Wang et al.88 exfoliated bulk CoFe-LDHs into ultrathin nanosheets using Ar plasma and concurrently introduced O, Co, and Fe vacancies during the exfoliation process (Fig. 5a). These vacancies markedly promoted the adsorption of water molecules and reaction intermediates. Benefiting from the high specific surface area and abundant exposed active sites enabled by the ultrathin architecture, the obtained material demonstrated excellent OER activity. It should be emphasized that plasma treatment parameters, such as temperature and treatment duration, have a pronounced impact on the structural integrity of materials. At elevated temperatures or extended treatment durations, surface cracking and even structural disintegration became pronounced, as demonstrated by the evolution of basal-plane cracks in monolayer MoS2 with plasma treatment time (Fig. 5b-d).90 In order to alleviate the structural degradation induced by high temperatures, Jiang et al.91 employed low-temperature plasma (<500 °C) to simultaneously achieve nitridation, defect construction and interfacial coupling, effectively avoiding phase separation induced by high temperatures. Scanning electron microscope (SEM) and EPR analyses showed severe particle aggregation in untreated NiSe2/Ni/NCNT@CC, whereas plasma-treated catalysts displayed uniformly dispersed nanoparticles and porous architecture, leading to an increased specific surface area of 46.1 m2 g−1 and providing abundant active sites for electrochemical reactions. Overall, plasma technology features rapid reaction kinetics and a relatively low environmental footprint, which has led to its widespread adoption in defect engineering.92 However, it should be noted that as a typical surface-modification technique, plasma treatment still faces challenges related to the uniformity of defect distribution during large-scale production.
 | ||
| Fig. 5 (a) CoFe LDH nanosheets produced by Ar plasma exfoliation.86 Copyright 2017 Wiley-VCH. SEM images (b–d) show morphology of monolayer MoS2 with 10, 20, and 30 s oxygen plasma exposure, where the cracks appear in the basal plane.90 Copyright 2016 American Chemical Society. (e) Schematic diagram depicting the synthesis of Pt NPs, clusters and SACs under laser planting with low, moderate and high fluences and frequencies, respectively.101 Copyright 2023 American Chemical Society. | ||
3.2.1.2 Microwave-assisted methods. Microwave-assisted methods are rapid thermal-treatment techniques based on the interaction between electromagnetic waves and matter, and they have been widely employed in recent years for the construction of defect-engineered electrocatalysts.93,94 During microwave irradiation, materials with high dielectric loss can undergo rapid and uniform temperature elevation within an extremely short time, followed by instantaneous cooling once the irradiation is terminated. This process enables nonequilibrium structural evolution to occur on the timescale of seconds or even milliseconds.95 Such “rapid heating-instant quenching” thermal shock readily induces inhomogeneous lattice strain, localized coordination defects, and VO or VM within the crystal lattice. Simultaneously, it enables the cooperative promotion of precursor decomposition, elemental doping, and phase-structure reorganization, rendering microwave methods highly efficient for constructing multidimensional defect architectures. According to the physical state of the reaction system, microwave-assisted synthesis can be broadly classified into three main categories: solid-state, liquid-phase, and quasi-solid-state methods. Solid-state microwave-assisted synthesis employs solvent-free solid precursors as the reaction system, typically requiring mechanical grinding or similar processes to ensure homogeneous mixing.96 Energy absorption relies on the intrinsic dielectric properties of the solids or the presence of added susceptors. Fan et al.97 used urea as a nitrogen source to synthesize O/N co-doped commercial conductive carbon (ONCB) in a household microwave oven via a 40 s rapid irradiation process. This strategy not only introduced abundant oxygen- and nitrogen-containing functional groups but also generated numerous defects and porous structures within the carbon framework, thereby promoting the uniform dispersion of Ru nanoparticles on the support. However, due to limitations in solid-state diffusion, localized overheating may occur, making precise temperature control challenging. In contrast, liquid-phase microwave-assisted synthesis employs polar solvents such as water, alcohols or ionic liquids as reaction media. Upon dissolution of the precursors, homogeneous or multiphase reaction systems containing mobile dipoles and ions are formed, which enables efficient microwave energy transfer through dipolar rotation and ionic conduction mechanisms. Compared with solid-state approaches, liquid-phase microwave synthesis generally offers more rapid volumetric heating and a faster mass transfer rate. Alam Khan et al. achieved the rapid synthesis of TiO2 gel spheres by microwave treatment of a biphasic system consisting of polymeric titanium ethoxide dispersed in kerosene, pioneering liquid-phase microwave synthesis of inorganic materials.98 Owing to its versatility and controllability, this strategy has become the most widely applied microwave-assisted synthesis route. In practice, the scalability of this method is limited, thereby hindering its large-scale application.
Situated between solid-state and liquid-phase approaches, quasi-solid-state microwave synthesis has recently attracted increasing attention as a transitional strategy. In this approach, small quantities of solvent are incorporated to transform the precursors into a semi-solid or paste-like state. This configuration improves thermal uniformity in solid-state systems and is conducive to large-scale application. Using quasi-solid-state microwave methods, Wu et al. achieved the effective coupling of low loadings of noble metals with transition-metal oxide, carbide and hydroxide matrices, successfully constructing a series of electrocatalysts enriched with VO,55,93,94 which exhibited favorable catalytic activity and stability. Despite the pronounced advantages of microwave-induced thermal shock in defect construction, significant challenges remain in terms of process controllability and scalability, which continue to limit its large-scale implementation.
3.2.1.3 Laser-assisted methods. Laser-assisted synthesis is an advanced physical approach that relies on ultrashort interaction time and ultrahigh power density. In this process, high-energy laser pulses are delivered to materials within an exceptionally short duration, inducing localized chemical bond cleavage and thereby generating various defect structures.99,100 The type and concentration of defects generated during laser-assisted synthesis are strongly dependent on laser parameters, including fluence, pulse width and scanning mode. Zou et al.101 proposed a laser implantation strategy for the universal preparation of high-loading SACs (Fig. 5e). Specifically, laser pulses concurrently generated substrate defects and isolated metal atoms, with the defect sites serving as effective anchors through enhanced electronic interactions.
Their study demonstrated that increasing laser fluence markedly enhanced bond rupture and local energy fluctuations, thereby facilitating the migration of metal atoms into lattice interstitials or inducing the formation of abundant surface and bulk vacancies. Thus, the metal loading of SACs reached a record-high level of 41.8 wt%. In addition, laser techniques enable effective regulation of the catalyst morphology and microstructure. Gary J. Cheng and co-workers102 constructed a laser shock-induced nanoreactor through precise regulation of the experimental setup and laser parameters, thereby enabling directional control over the reaction process and successfully fabricating coral-like LDH substructures featuring an ultrahigh specific surface area, abundant defects, and uniformly distributed dopants (Fig. 6a and b). Using this approach, layered double hydroxide nanosheets with a thickness of approximately 3 nm were obtained, representing ∼75% reduction compared with those synthesized via conventional solvothermal methods. This strategy effectively overcomes the limitations associated with insufficient morphological control in laser-based fabrication of layered materials.103 The above synthetic strategies are predominantly focused on the preparation of defect-rich catalysts via laser ablation in liquid environments. Notably, this approach is not limited to liquid-phase systems and can also be extended to solid-state conditions for defect engineering. Using a mixture of indium chloride and carbon nanotubes as the precursor, Ye et al.104 employed the transient laser-assisted strategy to rapidly generate In-based catalysts (L-In) featuring abundant stacking faults, vacancies, and interstitial defects (Fig. 6c). The synergistic coexistence of these defects significantly enhanced the activity and selectivity of the catalysts toward the CO2 reduction reaction.
 | ||
| Fig. 6 (a) The illustration of the experimental set-up for the synthesis of a LDH nano-coral.102 Copyright 2022 Wiley-VCH. (b) The illustration of the laser-induced nanoreactor.102 Copyright 2022 Wiley-VCH. (c) Spherical aberration corrected HRTEM images of L-In showing the defects such as vacancies, interstitial atoms and stacking faults.104 Copyright 2022 Wiley-VCH. (d and e) TEM and HRTEM images for GB-(Fe0.66Co0.34)2B.108 Copyright 2022 Elsevier B.V. (f) EPR spectra of S–CuFe2O4/BC and VQ S–CuFe2O4/BC.109 Copyright 2024 American Chemical Society. | ||
4 Dynamic evolution of defects
In existing studies, the characterization and analysis of defects have predominantly relied on ex situ characterization techniques, which focus on elucidating how defects in relatively stable structural states regulate catalytic performance, thereby laying the foundation for establishing structure–performance relationships between defects and catalytic behavior. However, accumulating experimental evidence indicates that defects undergo dynamic evolution processes under synthesis and realistic reaction conditions prior to establishing definitive structure–property relationships.111,112 This approach facilitates more comprehensive understanding of defect formation processes and their underlying mechanisms. Investigating the dynamic evolution of defects during the synthesis stage benefits the elucidation of their formation pathways and stabilization mechanisms, thereby providing a solid foundation for the controllable construction of defects. In contrast, probing defect dynamics under electrocatalytic operating conditions helps to elucidate the mechanisms underlying the generation, transformation and deactivation of active sites under realistic reaction conditions, deepening the understanding of intrinsic electrocatalytic processes. Overall, defect dynamic evolution is an intrinsic property of defect-rich electrocatalysts, and understanding its driving forces and evolution pathways is fundamental to elucidating the true working-state structure of catalysts. Furthermore, elucidating the dynamic evolution behaviours of defects is a prerequisite for achieving their precise and controllable regulation. In the following sections, we systematically analysed the driving forces and typical pathways of defect dynamic evolution under electrocatalytic conditions.4.1 Driving forces for defect dynamic evolution
The dynamic evolution of defects in catalysts is typically governed by the synergistic effects of thermodynamic, electrochemical, mechanical, and chemical environments. Given that defects inherently reside in high-energy and metastable states, their structures are highly susceptible to reconstruction in these complex environments, enabling dynamic optimization of the electronic structure and electrocatalytic performance.Among the various driving forces, thermodynamic effects play a fundamental and decisive role. Elevated temperature enhances atomic mobility, thereby accelerating defect formation, diffusion, aggregation and order-disorder transitions. Under relatively low-temperature conditions (e.g., ∼300 °C), limited atomic mobility favored energetically stable ordered configurations of VO, such as the Cu4O3 (corresponds to the CuO phase containing 25% ordered VO) superstructure formed at the subsurface of CuO.113 In contrast, entropy-driven processes governed defect behavior at elevated temperatures on the order of 900 °C, leading to defect disordering and vacancy diffusion into the bulk, ultimately leading to the formation of subsurface one-dimensional VO channels in CeO2.114 Beyond thermal energy input, mechanical actions, such as localized high temperatures (>1000 °C) generated during ball milling, could induce substantial oxygen displacement in catalysts such as ZnO1−x/Cu, resulting in a high concentration of VO (Fig. 7a). Correspondingly, Zn 2p peaks at 1021.8 and 1044.8 eV corresponded to Zn 2p3/2 and Zn 2p1/2, and Zn LMM auger electron spectroscopy spectra (LMM AES spectra) features at 988.8 and 992 eV were assigned to Zn2+ and Znδ+ (δ < 2) (Fig. 7b), confirming that there existed some reduced Zn species, indicating the presence of VO.115 Moreover, severe lattice distortions116 represent localized thermodynamic instabilities that tend to promote defect aggregation and are often accompanied by the formation of bulk defects. At the macroscopic level, thermodynamic parameters including GBs curvature, interfacial energy and reaction barriers collectively drive GBs migration to minimize the total free energy of the system.117 These observations collectively indicate that thermodynamic driving forces govern the dynamic evolution of defects in electrocatalysts throughout the entire process and operate across multiple length scales.
 | ||
| Fig. 7 (a) Schematic illustration of the synthesis of ZnO1−x/Cu.115 Copyright 2024 American Chemical Society. (b) Zn 2p XPS and Zn LMM AES spectra of the reduced ZnO1−x/Cu catalysts.115 Copyright 2024 American Chemical Society. (c) The lowest-energy configuration of stable OH groups formed by H adsorption on the CuO surface.124 Copyright 2020 Springer Nature. (d) The adsorption of the 6th H atom triggers the spontaneous desorption of H2O and the formation of VO on the CuO surface.124 Copyright 2020 Springer Nature. (e) Comparing the free adsorption energy of different nitrate reduction (NO3−RR) intermediates on the structural models of Cu (111) and Cu (100).125 Copyright 2021 Royal Society of Chemistry. | ||
As one of the most critical external stimuli in electrocatalytic systems, electric fields or applied bias can also regulate the direction and rate of defect migration through directional energy input, thereby profoundly influencing catalytic performance. In TiO2 systems, the application of an external bias induced the “dragging effect” that drove subsurface VO toward the surface, where they aggregated into clusters consisting of two to three vacancies.118 Furthermore, progressive oxidation of metal ions to high-valence states under anodic polarization generates local charge imbalance, which weakens M–O bonds and accumulates lattice stress, thereby facilitating the formation of VO and VM. These vacancy defects modulate the band structure and local electronic structure, promoting reactant adsorption and activation.38,119 Nevertheless, with further increases in current density and potential, enhanced electron-transfer rates and elevated surface atomic kinetic energy facilitate defect migration and aggregation, which may induce bulk phase transformations or even structural collapse of the catalyst.120 For instance, Wang et al.121 introduced Cr and V into FeCoNi/CC to modulate the electronic structure. During the OER, Cr and V acted as sacrificial dopants and underwent controlled dissolution, generating abundant in situ VO and significantly increasing the density of active sites. This process represented a typical defect-engineering strategy in high-entropy systems, where selective element leaching could dynamically regulate defect concentration and surface reconfiguration. Despite partial Cr/V dissolution, the remaining high-entropy alloy retained high configurational entropy, effectively suppressing phase separation and maintaining structural integrity. Such entropy-stabilized frameworks are widely recognized as a key advantage of high-entropy materials for accommodating defect formation while preserving phase stability under specific reaction conditions.
Beyond external fields, reaction intermediates generated during catalysis can be regarded as “molecular switches” governing defect dynamic evolution. Strong adsorption of intermediates often introduces substantial local stress or induces charge redistribution at the atomic scale, thereby triggering atomic rearrangements and leading to the transient formation or healing of defects.122,123 Such dynamic adaptation is widely considered as an intrinsic origin of high catalytic activity. Specifically, VO gradients introduced by surface reactions, such as OH/H2O adsorption–desorption, can drive defect diffusion into the subsurface region and induce dynamic order-disorder structural oscillations. For example, Zhou et al.124 investigated the accumulation adsorption of H on CuO surfaces and the subsequent formation of H2O using DFT (Fig. 7c and d). Sustained adsorption of H atoms led to the formation of surface OH groups, which remained stable on the surface up to an OH coverage of 0.625 monolayers. Upon further H adsorption, H atoms bound to the surface OH groups and formed H2O molecules, which desorbed from the surface in a barrier-less manner. This H2O desorption process resulted in OL depletion and concomitant generation of VO on the oxide surface. The vacancies subsequently diffused into the subsurface, thereby disrupting the ordering of the Cu4O3 superstructure. The subsequent re-accumulation of surface OH species promoted vacancy reordering, ultimately giving rise to a periodic structural transformation process. Guided by this understanding, He et al.125 exploited *N intermediates generated during NO3−RR as capping agents to control the growth of Cu-based nanocrystals with dominant Cu (111) facets, successfully obtaining highly active catalysts enriched with Cu (100) facets and surface defects (Cu (100) exhibited higher activity toward the NO3−RR among the two facets).
DFT calculations revealed that the synergistic exposure of Cu (100) facets and surface defects enhanced the adsorption of both *NO3 and *H, lowering the reaction barrier for the NO3−RR, while effectively suppressing the competing HER (Fig. 7e). In summary, driven by the cooperative effects of thermodynamic, electrochemical, mechanical and reaction chemical environments, catalyst defects undergo continuous self-adaptive dynamic evolution, achieving a delicate balance between activity enhancement and structural stability. This understanding not only deepens fundamental insights into defect-mediated catalytic mechanisms but also provides a solid theoretical basis for the rational design of high-performance electrocatalysts through the emerging paradigm of “defect regulation under specific reaction conditions”.
4.2 Typical pathways of defect dynamic evolution
Under the continuous influence of external stimuli, the dynamic evolution of defects in materials typically proceeds along several key pathways, including defect generation and healing, defect migration and aggregation and defect-induced structural reconstruction. These dynamic pathways define the continuous evolution of defects from their initial formation to their actual working states, providing a comprehensive framework for elucidating the mechanisms governing defect regulation.Dynamic defect generation constitutes the initial stage of this evolutionary process. Esch et al.114 systematically investigated the formation and time-dependent evolution of VO on CeO2 surfaces, using high-temperature vacuum annealing to induce vacancy formation and employing scanning tunneling microscopy (STM) in combination with DFT calculations for structural and mechanistic analysis. In the early stages of annealing, CeO2 (111) surfaces were primarily populated by isolated VO, which could stably occupy either surface or subsurface positions with comparable coverages. According to theoretical calculations, VO introduced localized electrostatic perturbations that promoted lattice relaxation, charge redistribution and the reduction of neighboring Ce ions to Ce3+, ultimately leading to localized structural distortions. With prolonged annealing, initially isolated VO gradually migrated and interacted with one another, exhibiting pronounced tendency toward spontaneous aggregation. STM observations showed that short-chain vacancy clusters formed first and subsequently evolved into extended chain-like defect structures composed of multiple vacancy units, including double-chain and triple-chain configurations. These results indicate that individual defects possess an intrinsic propensity to aggregate into vacancy clusters under thermodynamic driving forces and charge reorganization, while the continuous aggregation process is often accompanied by further reconstruction of local atomic structures and electronic states.
Furthermore, the continuous accumulation of defects not only alters the local electronic structure of materials but can also triggers pronounced lattice reconstruction, thereby inducing the dynamic annihilation and redistribution of defects. With CuO as a model system,113in situ TEM studies showed that lattice collapse was induced when the VO concentration exceeded the accommodation limit of local superstructures, enabling efficient vacancy annihilation. Specifically, excessive VO formation led to the localized collapse of subsurface Cu–O layers, giving rise to stacking-fault structures within the crystal. HRTEM snapshots (Fig. 8a-c) clearly captured the lateral propagation of stacking faults generated by the collapse of subsurface Cu–O layers (marked by the orange, cyan and red arrows). From a crystallographic perspective, the CuO lattice consisted of alternately stacked Cu–O layers and Cu atomic layers. When the specific Cu–O layer collapsed owing to excessive VO accumulation, structural defects such as edge dislocations were introduced into the lattice. DFT calculations further confirm that the sustained accumulation of VO significantly destabilized the Cu–O coordination environment, rendering Cu atoms severely undercoordinated and thereby driving their migration toward intrinsic Cu vacancy sites in adjacent layers (Fig. 8d). This process directly illustrated the coupled dynamic evolution of defect migration, aggregation and annihilation. Such structural transformations ultimately exerted a direct influence on the catalytic performance of defect-engineered catalysts.
 | ||
| Fig. 8 (a–c) Time-resolved HRTEM images of CuO at p(H2) ≈ 0.5 Pa and T ≈ 300 °C showing that a stacking fault in the bulk propagates laterally toward the left.113 Copyright 2021 American Chemical Society. (d) Atomic model of the stacking fault and migration of a Cu atom from site 1 to site 2 and then to site 3, leading to propagation of the stacking fault upon the aggregation of the VO in the Cu–O plane.113 Copyright 2021 American Chemical Society. | ||
At a higher level, phase transitions can be regarded as an advanced manifestation of defect evolution. By disrupting the ideal periodicity of the crystal lattice, phase transitions introduce localized strain accumulation and energy fluctuations, providing favorable nucleation sites and lower-energy pathways for the emergence of new phases.126,127 Conversely, phase transitions are inherently coupled with defect generation and annihilation, owing to the proliferation of interfaces, dislocations, and other defect structures during new-phase formation.128 This dynamic interplay between defects and phase transitions is widely recognized as a key factor governing the material structure and performance. For example, Mn doping induced Jahn–Teller (J–T) distortions that caused pronounced deformation of MnO6 octahedra centered on Mn, generating VO, coordination unsaturation and other types of defects. These defects substantially lowered the phase-transition energy barriers from Co4N to Co3O4, CoOOH and ultimately CoO2, accelerating the generation of LOM-relevant high-valence metal active species. Nitrogen desorption and the increase in Co oxidation states further promoted the generation of new defects during the phase-transition process. In turn, these defects regulate local electronic configurations and lattice stability, effectively suppressing the aggregation of phase-transition products while maintaining continuous exposure of active sites.129
5 Capturing defects
To verify the mechanisms of defect dynamic evolution and overcome the limitations of conventional ex situ characterization, advanced in situ/operando techniques are indispensable. Conventional defect characterization primarily relies on structural, compositional and electronic-structure analysis techniques to elucidate the types and functional roles of defects in materials across multiple length scales. At the crystallographic level, XRD is widely employed to identify lattice distortion and variations in long-range order. Features such as peak shifts, broadening and asymmetry are commonly regarded as indirect indicators of strain accumulation and dislocation introduction.130 Raman and Fourier-transform infrared (FTIR) spectroscopies are sensitive to short-range order and local bonding environments, enabling the identification of defect-induced local structural perturbations via variations in vibrational peak positions and intensities.131 At microstructural and atomic scales, electron microscopy techniques allow the direct observation of diverse defect structures, ranging from dislocations and grain boundaries to stacking faults, amorphous regions, and heterogeneous interfaces.132 Of these, STEM imaging allows for the direct observation of most 0D and 1D defects. This technique is also applicable to semi-quantitative analysis of defect concentrations, where statistical counting of defects within localized regions, combined with appropriate extrapolation, enables estimation of the overall defect density in the material.133 The chemical composition and electronic structure are commonly probed using XPS for surface valence, vacancy, and doping analysis, while EPR provides complementary insight into defect-related species and low-coordination metal sites owing to its high sensitivity to unpaired electrons.134 Among them, XPS can be employed for the indirect identification and semi-quantitative analysis of 0D and 2D defects. For instance, detailed deconvolution of the O 1s spectra across different samples allows for the relative quantification of VO concentrations.135 Moreover, energy-dispersive X-ray spectroscopy (EDS) enables compositional analysis by analyzing the cation/anion stoichiometric ratios and can be used to estimate defect concentrations; however, the results are generally semi-quantitative and lack absolute quantitative accuracy.133 Overall, although these techniques provide critical insights into defect configurations and structural characteristics, they are largely limited to “static” analyses before and after the reaction, and thus insufficient for capturing the real-time evolution of defects during formation and under working conditions. Consequently, in situ and quasi-in situ characterization techniques have emerged as major development directions in defect research. Accurate and real-time characterization of defects is the key support for the precise construction and controllable regulation of defects, and conventional and in situ/operando characterization techniques for defect detection and dynamic evolution monitoring are summarized in this section.Conventional electron microscopy generally provides only static snapshots of defects before and after the reaction, whereas key processes such as defect generation, evolution, healing and annihilation under realistic operating conditions remain largely inaccessible. In situ electron microscopy overcomes this limitation by precisely reproducing catalytic reaction environments within the microscope chamber, enabling real-time visualization of catalyst evolution from the atomic to nanometer scale and revealing dynamic correlations between defect evolution and catalytic activity enhancement.136–138 For example, Patrick R. Unwin et al.139 employed scanning electrochemical cell microscopy (SECCM) to generate potential-dependent electrochemical “videos” under H2 and CO2 atmospheres, simultaneously mapping the electrocatalytic activity distribution and surface topography of gold electrodes. By combining these measurements with high-resolution electron backscatter diffraction (HR-EBSD), grain orientation, lattice strain and dislocation density maps were constructed (Fig. 9a). The results showed that dislocations were enriched near GBs, which likely accounted for the enhanced localized catalytic activity previously observed at these sites. Moreover, dislocations also tend to aggregate at other structural features, such as slip bands (SBs), thereby giving rise to multiple potential localized regions of high catalytic activity within the material. Guo et al.140 further integrated in situ electrochemical atomic force microscopy with scanning electrochemical microscopy (EC-AFM-SECM) to synchronously track nanoscale morphological evolution and local catalytic activity (Fig. 9b). Under operando conditions, they clarified the spatial distribution and dynamic formation of defect sites, demonstrating that surface reconstruction of amorphous SnO2/GO catalysts during the NO3−RR induced the proliferation of edge- and wrinkle-like defects, which were identified as catalytic “hot spots”. This technique establishes dynamic and causal linkage between microstructural evolution and macroscopic performance, substantially advancing materials research from empirical exploration toward rational design. Benefiting from the highly disordered intrinsic structure, amorphous materials underwent dynamic bond rearrangement under specific reaction conditions, thereby enabling continuous regeneration of active defects.
 | ||
| Fig. 9 (a) The dislocation density schematic on the surface of the gold electrode.139 Copyright 2021 Springer Nature. (b) Schematic diagram of the EC-AFM-SECM setup for evaluating the NO3−RR responsiveness of the catalyst.140 Copyright 2025 American Chemical Society. (c) In situ EPR spectra of the 0.75Pd–5In2O3–ZrO2 catalyst prepared.145 Copyright 2022 Springer Nature. Operando XAS of the Co K-edge of (d) pure Co3O4 and (e) VO-Co3O4. The insets show magnified views of the regions marked by dotted boxes.149 Copyright 2020 American Chemical Society. (f) In situ ATR-FTIR spectra of V-Bi NS at different potentials.155 Copyright 2024 Wiley-VCH. | ||
In situ EPR/electron spin resonance (ESR) is among the most powerful techniques for directly probing the dynamic evolution of defects in electrocatalysts.141,142 This method selectively detects signals originating from defects containing unpaired electrons, such as VO/M and dangling bonds. By monitoring signal intensity changes at characteristic g-factor positions as potential or time varies, the evolution of specific defect concentrations can be directly and quantitatively evaluated,143 establishing a direct link between electrochemical stimuli and defect generation or annihilation kinetics. Gunnar Jeschke and co-workers144,145 systematically investigated the evolution of VO in ZrO2-supported In2O3/ZnO-based catalytic systems using the in situ EPR technique, including both pristine materials and those doped with Cu, Pd, and Ni. Under a flowing H2 atmosphere, vacancy-related signals were observed to broaden progressively with increasing time, which was attributed to the continuous generation of additional VO. Upon subsequently switching the gas atmosphere to CO2 and maintaining it for 2 h, the vacancy signals gradually narrowed, indicating that part of the newly formed defects was partially healed under oxidizing conditions (Fig. 9c). These observations provide compelling evidence that VO undergo pronounced dynamic evolution.
During electrocatalytic reactions, in situ XAS enables real-time monitoring of metal oxidation states and local coordination environments, thereby directly elucidating how defects respond to applied potentials, interact with reaction intermediates and ultimately govern catalytic performance.146–148 For instance, Wang et al.,149 by tracking the evolution of Co coordination environments, demonstrated that VO do not act as direct OER active sites but instead follow the dynamic pathway of “OH capture → induced Co pre-oxidation → Co–OOH formation”, effectively lowering the reaction energy barrier (Fig. 9d and e). Zhao et al.150 revealed via in situ XAS that the absorption edge shifted to the low-energy region at 1.25 V, indicating a decrease in the valence state of Ni sites. Upon increasing the potential to 1.35 V, the Ni sites were oxidized to higher valence states, resulting in a gradual shift of the absorption edge toward the high-energy region. The variation of operando XAS spectra was attributed to the structural distortion of lower symmetry unsaturated Ni sites by adsorption of OH− and removal of the terephthalic acid (BDC) ligand at low potential, and evolution to a highly symmetric NiO6-liganded octahedron (β-NiOOH) at high potential. And β-NiOOH would further generate some VO (VO–NiOOH) to promote the reaction under potential application, intuitively capturing the dynamic evolution behavior of defects.
Raman and infrared spectroscopies are highly sensitive to molecular vibrational modes, and variations in defect states often induce pronounced changes in surface bond vibrational frequencies. Consequently, these techniques not only allow quantitative evaluation of defect density and type through enhancement of defect-related vibrational features,151 but also enable sensitive detection of key reaction intermediates at defect sites, thereby elucidating the role of defects in stabilizing intermediates and optimizing reaction pathways.152,153 For example, Li et al.154 identified the presence of initial VM through characteristic peak assignments in in situ Raman spectra, and further revealed the dynamic evolution of VM (VM → VMOH → VMOH-H). Zhang et al.155 employed in situ attenuated total reflection-Fourier transform infrared spectroscopy (ATR-FTIR) to capture signals of key intermediates (*CO2 and *OCHO). The vacancy-containing catalyst enabled the detection of relevant signals at lower potential compared with the vacancy-free control catalyst, demonstrating that vacancy defects enhanced adsorption and activation of inert CO2 molecules via their electron-rich nature (Fig. 9f). Vacancy defects could also stabilize the adsorption of the key intermediate *OCHO, thus facilitating the reaction kinetics of formate production.
In addition, several other in situ techniques have been widely applied to monitor defect evolution. The synergistic application of these techniques provides a comprehensive experimental framework for elucidating the formation and dynamic evolution mechanisms of defects in electrocatalysts.
6 Applications of defect engineering in water electrolysis
Defect engineering has become a core strategy for regulating the electronic structure and surface reaction environment of electrocatalysts, and has been widely applied across various electrocatalytic systems. Among these, water splitting is considered as a representative model system for elucidating the role of defect engineering, owing to its well-defined reaction pathways, well-established mechanistic understanding, and significant industrial relevance.156 Accordingly, defect engineering has been particularly extensively explored in the HER and OER, and has emerged as a key strategy for enhancing electrocatalytic performance. Unlike conventional strategies such as morphology regulation or compositional optimization, defect engineering typically avoids extensive modification of the bulk crystalline framework. Instead, it introduces localized structural imperfections to precisely tailor electronic states, coordination environments, intermediate adsorption energetics and reaction pathways. In this context, this section systematically reviews the dynamic modulatory roles and underlying mechanisms of several representative defect types, namely vacancy defects, doping-induced defects, and heterointerface defects, in conventional and complex water electrolysis systems (Table 1).| Catalyst | Synthesis methods | Defect types | Target reactions | Performance metrics | Relevant mechanisms | Ref. |
|---|---|---|---|---|---|---|
| VCr,Co–NiFeOOH | Corrosion and electrochemical self-activation | Cation vacancy | OER in 1 M KOH | 198 mV @ 10 mA cm−2 | Local electronic structure optimization and surface adsorption energy modulation | 162 |
| NiFeCe-LDH@CP | Hydrothermal method | Doping defects | OER in 1 M KOH | 267 mV @ 100 mA cm−2 | Local electronic structure optimization and surface adsorption energy modulation | 32 |
| Ni,B–RuOx | Solid-liquid reaction and annealing | Doping defects | OER in 0.5 M H2SO4 | 206.8 mV @ 10 mA cm−2 | AEM-LOM coupling mechanism and grain boundary-bulk synergy | 180 |
| W–NiCo2O4 | Electrodeposition and annealing | Doping defects | OER in 0.5 M H2SO4 | 206.8 mV and stable operation for 100 h (10 mA cm−2) | Dynamic oxygen replenishment mechanism, local electronic structure modulation, and VO healing | 181 |
| Mo2N/MoO3@NC-30 | Annealing | Heterointerface defects | HER in 1 M KOH | 94 mV @ 10 mA cm−2, stable operation for 1000 h at 500 mA cm−2 | Interfacial polarization effect and interfacial electron delocalization and accumulation | 72 |
| IrNi/F-MWCNTs | Thermal shock | Doping defects | HER in 1 M KOH | 31 mV @ 10 mA cm−2 | Interfacial water restructuring and d-band center theory | 188 |
| MoO3−x/Mo2AlB2 | Hydrothermal method and intercalation | Mixed defects | HER in 0.5 M H2SO4 | 38 mV @ 10 mA cm−2 | Active microdomains theory andelectronic structure modulation | 199 |
| V–NiII–Fe | Impregnation | Vacancy | OER in 1 M KOH + seawater | 333 mV @ 1 A cm−2 | VO-induced Ni4+ formation mechanism and the selective adsorption effect of hard acids | 218 |
6.1 Conventional electrolysis system
Vacancy defects engineering (e.g. VO and VM) are widely regarded as among the most direct and effective strategies for tuning OER activity. By modifying the local atomic coordination environment, vacancy defects can reconstruct intermediate adsorption behavior and thereby optimize overall reaction kinetics. Guo et al.162 constructed Cr-vacancy-rich catalysts (VCr,Co–NiFeOOH) via two-step strategies involving room-temperature corrosion and subsequent electrochemical self-activation. DFT calculations based on the extended microenvironment model comprising approximately 40 metal atoms demonstrated that the introduction of VCr markedly weakened the adsorption of reaction intermediates on adjacent Fe sites (Fig. 10a), thereby alleviating excessive adsorption. In synergy with Co sites, this vacancy-induced modulation enabled fine-tuning and spatial redistribution of surface adsorption energetics. As a result, the energy barrier of the OER RDS decreased from 2.34 eV to 2.20 eV, markedly accelerating the kinetics of the critical reaction step. Beyond the surface regulation of adsorption energy, vacancies formed dynamically could activate OL by enhancing M–O covalency, facilitating mechanistic transitions and inducing in situ reconstruction of the active phase. Based on this strategy, He et al.163 systematically compared the electrochemical reconstruction behaviors of crystalline NiMoO (hc-NiMoO) and amorphous NiMoO (a-NiMoO, usually with various defects164) during the OER, elucidating the intrinsic correlation between “covalency-reconstruction-activity”. In situ Raman spectroscopy demonstrated the disappearance of M–O vibrational modes and the emergence of characteristic Ni–OOH peaks in a-NiMoO at ∼1.40 V, while the same structural evolution in hc-NiMoO required a higher potential of ∼1.60 V (Fig. 10b and c). Further analysis of the δ(Ni3+–O)/ν(Ni3+–O) ratio indicated that a-NiMoO preferentially transformed into disordered d-NiOOH, while hc-NiMoO evolved into relatively ordered o-NiOOH, suggesting an enhanced propensity of amorphous structures for reaction-front reconstruction and the concomitant formation of disordered phases. Post-OER XPS analysis revealed an exceptionally high Mo leaching extent of up to 97.5% in a-NiMoO, resulting in the dynamic formation of VM. Positron annihilation spectroscopy (PAS) was used to quantitatively probe defect characteristics, where the shortest lifetime component (τ1) corresponded to small vacancies, while longer lifetimes signified clustered or extended vacancy defects.162 The τ1 value of d-NiOOH (187 ps) was significantly higher than that of o-NiOOH (156 ps), confirming higher concentration of Mo vacancies in the former (Fig. 10d). XAS analysis identified lower Debye–Waller factors (σ2) for the Ni–O scattering path in NiOOH–C (0.0066 Å2) reconstructed from amorphous NiO, compared with d-NiOOH (0.0078 Å2), reflecting enhanced structural disorder in d-NiOOH arising from dynamic VM generation. Benefiting from vacancy-induced disorder and electronic structure modulation, d-NiOOH achieved a current density of 100 mA cm−2 at an overpotential of only 201 mV, significantly outperforming NiOOH–C (297 mV). Mechanistically, vacancy-induced structural distortion strengthened Ni–O covalency and effectively activated OL, driving the OER pathway from the conventional AEM toward LOM and thereby substantially improving overall kinetics. However, the intrinsic mechanism by which catalysts maintain long-term structural stability after the leaching of metal species and OL has not been thoroughly investigated in this study.
 | ||
| Fig. 10 (a) Reallocation of adsorption energy with a uniform mixture of local relatively high and low states.162 Copyright 2025 American Chemical Society. In situ Raman spectra of (b) a-NiMoO and (c) hc-NiMoO.163 Copyright 2025 Wiley-VCH. (d) Measured lifetime spectrum of d-NiOOH by the PAS test.163 Copyright 2025 Wiley-VCH. (e) HRTEM image of NiFeCe-LDH@CP.32 Copyright 2023 Elsevier B.V. (f) Gibbs free energy diagram for the four steps of the OER on NiFe-LDH@CP and NiFeCe-LDH@CP.32 Copyright 2023 Elsevier B.V. (g and h) O 1s XPS spectra of HEO-Al and HEO catalysts, along with their corresponding relative oxygen contents.166 Copyright 2025 American Chemical Society. (i) In situ XAS spectra of NiSe2/FeSe2-NC.169 Copyright 2023 Elsevier B.V. (j) Formation energy of Se vacancies in NiSe2, FeSe2 and NiSe2/FeSe2.169 Copyright 2023 Elsevier B.V. (k) Proposed nature of ΔΦ during the OER in CeO2/Fe–Co(OH)2 catalysts.170 Copyright 2025 Royal Society of Chemistry. | ||
Doping is generally regarded as an effective approach for optimizing intrinsic electronic structures by modulating lattice electron density and valence states.165 The defect-inducing effect of doping arises from disparities in the valence state, ionic radius, electronegativity and coordination environment between dopant atoms and the host lattice. These mismatches require local lattice compensation, which in turn leads to structural distortion, vacancy formation, or the development of disordered regions. Liao et al.32 introduced trace amounts of Ce into NiFe-LDH nanosheets to construct a NiFeCe-LDH@CP catalyst for the OER. Combined HRTEM, Raman and EPR analyses demonstrated pronounced Ce-doping-induced lattice distortion and disorder, accompanied by disruption of the layered framework and generation of dislocation defects (Fig. 10e). These structural perturbations not only increased the accessible surface area but also markedly enhanced surface VO concentrations. DFT calculations further showed that the distortion-defect synergy optimized the local electronic structure of Ni sites and modulated intermediate adsorption energies, lowering the energy barrier of the rate-determining *OH → *O step from 2.3 eV to 1.66 eV (Fig. 10f). Beyond improving catalytic activity, achieving an optimal balance between activity and stability remained central to OER catalyst design. To this end, Shen et al.166 introduced Al into high-entropy oxide (HEO) via molten-salt pyrolysis methods, named HEO-Al. VO concentration in HEO-Al reached 32.75%, markedly higher than the 18.15% observed in undoped HEO, as determined by XPS (Fig. 10g and h). DFT results highlighted the critical role of moderate VO concentrations in promoting OER deprotonation and strengthening O 2p-metal d orbital coupling, which effectively lowered the LOM energy barrier. Meanwhile, Al incorporation not only modulated defect formation but also increased the configurational entropy of the system, improving structural stability. This synergistic effect between defect engineering and high-entropy design enabled a dynamic balance between catalytic activity and durability by effectively suppressing structural degradation during electrolysis.
Heterointerface defects originate from interfacial structural and electronic mismatches, giving rise to local coordination distortion, electronic reconstruction, and defect accumulation, with pronounced implications for catalytic regulation.167,168 Song et al.169 constructed the NiSe2/FeSe2-NC nano-heterojunction enriched with interfacial structures and high-density vacancies via an in situ growth strategy, offering an ideal model system to elucidate interfacial regulation in the OER. Above 1.4 V, in situ XAS captured the development of pronounced Fe–M shells associated with in situ-formed Ni3+–O–Fe3+ coordination motifs, identified as the genuine OER active centers (Fig. 10i). Notably, Se exhibited partial dissolution during operation, inducing transient Se-vacancy intermediates. Combined DFT and operando XAS analyses demonstrated pronounced weakening of local Fe–Se bonds by reaction-induced vacancy intermediates, which facilitated Fe oxidation to highly active (oxy)hydroxide species and lowered the energy barrier for active-phase formation. A comparison of Se vacancy formation energies indicated facile Se activation and dissolution in NiSe2 (−1.69 eV), in contrast to the significantly higher vacancy formation energy in FeSe2 (−0.38 eV) (Fig. 10j). Consequently, NiSe2/FeSe2 with moderate vacancy formation energy enabled dual regulation: controlled Se dissolution induced electronically favorable active vacancies at the interface, while excessive dissolution was suppressed to maintain overall structural integrity. In NiSe2/FeSe2-NC, dissolution-induced vacancies were stabilized by interfacial confinement, simultaneously improving conductivity, lowering the formation energy of the active phase, and preserving long-term structural integrity. Furthermore, the synergistic interplay among multiple types of defects enables multifaceted and precise modulation of electrocatalytic performance. Wang et al.170 constructed the vacancy-rich CeO2/Fe–Co(OH)2 catalytic system.
DFT calculations demonstrated the thermodynamic spontaneity of Fe incorporation into Co(OH)2 and further showed that Fe doping markedly lowered the VO formation energy of Co(OH)2. EPR results revealed substantially higher VO concentrations in Fe–Co(OH)2 than in pristine Co(OH)2, with these concentrations being further amplified upon heterostructure formation with CeO2. The introduction of additional VO effectively activated OL and promoted active phase (Co4+–Ox) formation during the OER, thereby enhancing catalytic activity. However, to address the inherent activity–stability trade-off associated with excessive OL participation, a dynamic interfacial electron backflow mechanism (CeO2 → Co4+–Ox) was established during the steady-state reaction. Owing to the higher work function of Co4+ (3.99 eV) relative to CeO2 (2.92 eV), electrons were transferred from CeO2 to the Co centers, partially reducing Co4+ to the more stable Co3+ state. This electron redistribution stabilized Co(IV)–O bonds and suppressed excessive OL involvement, thereby mitigating over-oxidation-induced structural degradation (Fig. 10k). Meanwhile, interfacial bonding (e.g., Ce–O–Co) effectively anchored VO, suppressing their migration and aggregation at high potentials. Benefiting from these synergistic mechanisms, the catalyst maintained nearly unchanged VO concentrations after continuous operation at 1000 mA cm−2 for 800 h, enabling long-term, high-current-density OER dominated by the LOM pathway.
While LOM can markedly boost OER kinetics, it also triggers persistent OL loss and metal dissolution, thereby undermining structural stability and accelerating catalyst deactivation.176,177 Building on this concept, Zhang et al.178 introduced rare-earth Nd during combined pyrolysis-acid leaching. Nd ion-coupled oxygen dissolution during acid treatment, followed by the subsequent leaching of Nd ions at the early reaction stage, triggered the generation of high-density and spatially controllable OL vacancies. Meanwhile, the short-range quasi-crystalline rutile order was retained, ultimately leading to the formation of the IrOx-3Nd catalyst. This structure provided abundant oxygen-unsaturated sites that facilitated intermediate adsorption and deprotonation, while simultaneously maintaining the integrity of the Ir–O–Ir network. Consequently, large-scale Ir–O bond cleavage and Ir ion dissolution under high potentials were effectively suppressed. DFT calculations demonstrated a defect-induced shift of the Fermi level in IrOx-3Nd toward the O 2p band center (−3.43 eV), accompanied by a reduced energy gap between the Ir 5d and O 2p band centers (Fig. 11a), indicative of enhanced Ir–O covalency. This electronic modulation facilitated OL activation by lowering the energy barrier for OL oxidation, thereby promoting the LOM pathway and improving catalytic activity. To address the inherent activity–stability trade-off associated with LOM, the introduced VO could be rapidly replenished by water molecules during catalysis, with a low energy barrier of only 0.42 eV. This dynamic vacancy refilling process enabled continuous OL self-repair, preventing vacancy accumulation and suppressing structural degradation. In situ Raman spectroscopy indicated preferential adsorption of surface *OH species at vacancy sites, enabling reversible oxygen exchange processes that maintained the structural integrity of Ir–O bonds even under high anodic potentials (Fig. 11b and c). These findings highlighted a synergistic strategy in which defect-induced electronic structure modulation activated the LOM pathway, while dynamic oxygen replenishment and reversible intermediate adsorption collectively stabilized the catalyst structure, thereby achieving a balance between high catalytic activity and long-term stability.
 | ||
| Fig. 11 (a) Density of states (DOS) plots of Nd leaching-induced defect IrO2 and rutile IrO2.178 Copyright 2025 Wiley-VCH. (b and c) In situ Raman spectra of IrOx-3Nd and C–IrOx after 0–30 min OER tests.178 Copyright 2025 Wiley-VCH. Differential electrochemical mass spectrometry (DEMS) signals of 16O18O and 18O18O from the OER products for 18O-marked (d) Ru1−xO2 and (e) RuO2−x.179 Copyright 2024 American Chemical Society. (f) Free energy diagrams for AEM and LOM on the surfaces of Ru1−xO2, RuO2−x, and RuO2 under an electrode potential of 0 and 1.23 V.179 Copyright 2024 American Chemical Society. (g) Reaction free energy and subsurface O loss enthalpy change volcano against the symmetry-breaking.177 Copyright 2025 Springer Nature. | ||
Notably, redirecting the OER pathway serves as more straightforward strategy. As the classic electrocatalyst for acidic OER, the activity decay of RuO2 mainly originates from structural instability induced by the LOM.173 In this context, Pan et al.179 systematically investigated the roles of cation- and anion-vacancy-modified RuO2 catalysts (Ru1−xO2 and RuO2−x) in acidic OER. As determined by operando DEMS, oxygen evolution on all catalysts was dominated by the formation of 32O2, supporting an AEM-dominated OER pathway. Additionally, RuO2−x produced only 0.56% 34O2, compared with 3.4% for Ru1−xO2 and 7.2% for pristine RuO2 (Fig. 11d and e). This behavior confirmed the stability of the AEM pathway and highlighted the role of defect engineering in suppressing the intrinsically unstable LOM. Meanwhile, the synergistic modulation of Ru and VO optimized Ru–O bond strength and the electronic structure, reducing the energy barrier of the RDS from 2.04 eV to 1.61 eV and 1.74 eV, respectively (Fig. 11f). Accordingly, this approach achieved a synergistic balance between improved OER kinetic behavior and maintained structural stability. Moreover, Huang et al.177 proposed a symmetry-breaking design strategy induced by anion doping for M–RuO2 (M = F, S, Se, P, N, Cl, Br) on the basis of DFT calculations. Among these candidates, F-doped RuO2 exhibited the lowest free energy barrier for the RDS (1.80 eV), highlighting its superior potential for achieving both high activity and enhanced stability in acidic OER (Fig. 11g). A higher maximum electron localization function (ELF) value was further observed for surface *OOH species on F–RuO2 compared with pristine RuO2, suggesting enhanced interaction strength between the *OOH intermediate and the catalyst surface. DOS analysis revealed effective modulation of the Ru d and O 2p electronic states upon F incorporation, accompanied by a downward shift of the O 2p band center from −2.96 eV to −3.23 eV. This downward shift increased the VO formation energy, suppressing Ru dissolution and stabilizing OL (Fig. 12a).
 | ||
| Fig. 12 (a) DOS of Ru 3d and O 2p for F–RuO2 and RuO2.177 Copyright 2025 Springer Nature. (b) HRTEM image of Ni,B–RuOx, with typical GBs regions highlighted by white dashed lines.180 Copyright 2025 American Chemical Society. (c) Ru K-edge XAS spectra.180 Copyright 2025 American Chemical Society. (d) Calculated energies for Ru structural degradation and surface oxygen loss in RuO2 and Ni,B–RuOx.180 Copyright 2025 American Chemical Society. (e) Ab initio molecular dynamics (AIMD) snapshots of H2O dissociation and *O migration into a lattice vacancy.181 Copyright 2025 American Chemical Society. (f) Full-cell operation at 1 A cm−2 in pure water.181 Copyright 2025 American Chemical Society. (g) High-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) image of the core/shell RuCo/RuCoOx nanospheres.184 Copyright 2024 American Chemical Society. | ||
In addition, both the enthalpy change associated with subsurface oxygen loss and the energy consumption required for Ru demetalation in F–RuO2 were markedly higher than those in RuO2, further confirming that symmetry breaking played critical roles in reinforcing lattice stability. Overall, the doping strategy simultaneously enhanced O-species adsorption and suppressed Ru demetalation, achieving synergistic improvement in catalytic activity and durability for acidic OER, while effectively shifting the reaction pathway from the LOM to the AEM. Considering that excessive suppression of the LOM pathway may lead to activity loss, Dai et al.180 achieved cooperative regulation of LOM and AEM by co-doping Ni and B into RuO2via the solid–liquid reaction route. In this system, B doping induced the formation of GBs (Fig. 12b), significantly increasing the concentration of VO and exposing more Ru active sites, while Ni preferentially accumulated near GBs under strain-driven effects, enabling synergistic structural modulation. XAS characterization revealed that Ni incorporation broke the symmetry of Ru–O octahedra and induced Ru–O bond contraction, thereby enhancing structural robustness (Fig. 12c).
DFT calculations further showed that GB regions exhibited a relatively low RDS barrier for the LOM pathway, whereas non-GB regions preferentially followed the AEM pathway with a barrier of 1.88 eV. This spatially resolved distribution of reaction pathways establishes a cooperative AEM-LOM mechanism between GBs and bulk regions. Importantly, to address the activity–stability trade-off associated with LOM-dominated systems, Ni and B co-doping significantly increased the Ru demetalation energy (Fig. 12d), effectively suppressing metal dissolution under acidic conditions. As a result, this system achieved the synergistic balance in which LOM-driven high activity at GBs regions was coupled with AEM-mediated stability in bulk regions.
The majority of existing studies focus on regulating the intrinsic activity of noble-metal-based catalysts, whereas the dynamic evolution of defects during catalysis has received far less attention, and noble-metal dependence remains persistently limiting. To address these limitations, Peng et al.181 extended the investigation to non-precious metal systems (doping W into NiFe2O4) and proposed the dynamic oxygen replenishment mechanism (DORM) for the first time based on AEM and LOM. This strategy directly addressed the intrinsic instability of LOM, where OL consumption often led to irreversible oxygen depletion and structural degradation. In this mechanism, OL participating in O–O coupling could be continuously replenished by water-derived species (Fig. 12e). DFT calculations identified W–Co sites with lower water adsorption energy (−0.74 eV) and dissociation barriers (−0.63 eV), markedly promoting interfacial water activation and facilitating oxygen supply for OL regeneration. As evidenced by structural characterization, the lattice distortion formation induced by W doping effectively lowered the surface protonation and vacancy formation energy, establishing a favorable structural foundation for OL activation and dynamic replenishment. ELF analysis further revealed pronounced enhancement in the electronic polarization of Co–O bonds upon W incorporation, which promoted VO healing and stabilized the OL network. Meanwhile, AIMD simulations revealed that VO induced by W doping could be rapidly filled by water dissociation products within approximately 360 fs, avoiding irreversible structural collapse caused by OL depletion in conventional LOM pathways. Collectively, these results established a dynamic repair paradigm in which OL activation and real-time oxygen replenishment are coupled, enabling sustained LOM activity while suppressing structural degradation. Benefiting from these advantages, W–NiCo2O4 achieved stable operation for 280 h at 1 A cm−2 in a PEM electrolyzer (Fig. 12f), providing a new design paradigm for non-precious-metal catalysts toward acidic OER. Regarding the activity–stability trade-off in the LOM-dominated OER, this work addresses the issue from multiple perspectives. First, appropriate defect engineering can modulate the d-band center to enhance M–O covalency and lower the energy barrier for OL oxidation, thereby effectively activating the LOM pathway. The stability challenges of this mechanism have been addressed in previous work, where OL replenishes depleted VO through the DORM. In addition, electron back-donation can suppress over-oxidation of metal centers. Together, these effects mitigate structural degradation caused by OL depletion, achieving a balance between efficient LOM activation and long-term structural stability. Furthermore, synergistic defect engineering strategies can construct spatially coupled AEM-LOM pathways while maintaining moderate LOM participation, thereby further reconciling catalytic activity and stability. These insights provide useful guidance for the rational design of high-performance OER catalysts.
On the basis of conventional regulation of vacancy and doping defects, heterointerfaces can endow materials with multilevel structural stability through interfacial bonding and synergistic effects of components.182,183 Liu et al.184 precisely controlled the oxidation conditions of RuCo alloy nanospheres to construct RuCo/RuCoOx core-shell heterostructures with tunable oxide-shell thicknesses. This structure was confirmed by HAADF-STEM characterization (Fig. 12g). GPA revealed a periodic distribution of maximum εxx strain at the interface, corresponding to dislocation-enriched regions that effectively exposed depletion-layer active sites and optimized the electronic structure. Computational simulations identified elongated Ru–O and Co–O bonds together with weakened covalency with increasing RuCoOx shell thickness, leading to energetically unfavorable OL oxidation and improved structural stability.
Layered MoS2, the prototypical HER catalyst, serves as the representative example in which VS are widely recognized as key active motifs that activate otherwise inert basal planes and expose edge-like active sites, enhancing HER activity.186 Li et al.68 introduced VS into MoS2 nanosheets via H2 annealing at temperatures ranging from 400 to 800 °C. As evidenced by EPR measurements, the VS concentration exhibited nonmonotonic dependence on temperature, increasing initially to the maximum at 600 °C and decreasing upon further heating. This behavior originated from the formation of isolated point defects at relatively low temperatures, which generated Mo–S dangling bonds, whereas progressive sulfur stripping leaded to the formation of extended defect regions at higher temperatures, reducing the number of Mo–S dangling bonds and thus weakening the EPR signal (Fig. 13a). Electrochemical measurements demonstrated that such vacancy defects significantly enhanced HER activity. To further elucidate the influence of the local vacancy environment on catalytic performance, Zhou et al.187 employed the confined growth strategy, in which excessive adsorption of reactant molecules on nascent nanocrystal surfaces during hydrothermal synthesis inhibited continuous oriented growth, yielding MoS2 samples enriched with vacancy defects. Modulating the Mo/S ratio allowed the formation of vacancies with distinct atomic environments (S–MoO2 and M–MoO2, Fig. 13b), enabling the construction of seven Mo/S ratios and sixteen vacancy models with different local coordination structures. Consistent experimental observations and DFT calculations identified M–MoO2 vacancies, characterized by higher density of terminal Mo atoms, as exhibiting more favorable hydrogen adsorption and delivering optimal HER activity. Further analysis highlighted the critical role of the local atomic configuration around vacancies in tuning the surface electronic structure and orbital distribution, enabling near-thermodynamically optimal Mo–H interactions and balanced *H adsorption–desorption behavior.
 | ||
| Fig. 13 (a) EPR spectra of the different defected MoS2.68 Copyright 2019 American Chemical Society. (b) Schematic diagram of the synthesis of different vacancy states MoS2 by controlling the ratio of sulfur to molybdenum in the reactants.187 Copyright 2024 American Chemical Society. Atomic configurations of an equilibrated (c) H2O–IrNi/F-MWCNTs interface and (d) H2O–IrNi/MWCNTs interface in alkaline media.188 Copyright 2025 Wiley-VCH. (e) Atomic configuration of an equilibrated H2O–IrNi/F-MWCNTs interface at ≈10.1 ps in alkaline media.188 Copyright 2025 Wiley-VCH. (f) The calculated d-band center for spin-up and spin-down.191 Copyright 2025 Wiley-VCH. | ||
Besides vacancy defects, doping defects have also been widely employed to optimize the catalytic performance of alkaline HER systems. Zong et al.188 synthesized F-doped multiwalled carbon nanotubes loaded with IrNi nanoclusters using a high-temperature ultrafast shock process (1200 °C for 10 s) under vacuum. AIMD simulations identified hydrogen-bond interactions between F dopants and interfacial water molecules, which guided water molecules toward the catalyst surface and promoted an ordered interfacial water structure. This reorganization shortened the average water-surface distance from 5.35 to 5.05 Å, strengthening interfacial adsorption (Fig. 13c and d). Operando attenuated total reflection-surface-enhanced infrared absorption spectroscopy (ATR-SEIRAS) measurements revealed a pronounced red shift and intensity enhancement of the ν-OH vibration, further confirming the optimized alignment of interfacial water molecules. On the IrNi/F-MWCNTs surface, metadynamics simulations captured H–O bond dissociation of water molecules within 10.1 ps, in sharp contrast to the undoped counterpart where no dissociation was observed, thereby evidencing a markedly lowered water dissociation barrier for the Volmer step upon F doping (Fig. 13e). When assembled into an anion exchange membrane water electrolyzer (AEMWE), this catalyst delivered an industrially relevant current density of 1 A cm−2 at 1.82 V, highlighting its promising application potential. In addition, cobalt phosphide (Co2P), owing to its near-optimal hydrogen binding energy (HBE) under alkaline conditions, had been extensively investigated as a HER catalyst.189,190 Yuan et al.191 systematically introduced transition-metal dopants into Co2P to construct a series of metal-doped Co2P nanocomposite frameworks (M-CPNCF), aiming to elucidate the role of secondary transition metals in Co2P. Metal centers with distinct coordination environments enabled precise modulation of the d-orbitals due to their differences in d-band filling and spin polarization, thereby effectively tuning the adsorption energies of reaction intermediates (Fig. 13f). Specifically, active sites with lower d-band filling possessed higher d-orbital energy levels and a greater number of unoccupied orbitals, which facilitated electron transfer from these unoccupied states to bonding orbitals, thereby strengthening adsorption interactions.192 From a spin-polarization perspective, the elevation of antibonding states in spin-down d orbitals enhanced bonding interactions, whereas the lowering of antibonding states in spin-up d orbitals weakened bonding strength, and the competition between these effects ultimately determined the adsorption energy.193 Combined theoretical and experimental results identified Fe doping as the most effective strategy to synergistically regulate water dissociation kinetics, OH binding energy, and *H adsorption–desorption behavior at Co active sites, leading to thermodynamically favorable conditions and enhanced HER activity.
Heterointerface defects represent another effective strategy for enhancing catalytic performance toward the alkaline HER. Structural mismatch at heterointerfaces frequently induces vacancies and strain, which act as key electronic modulation sites. These defect-rich interfaces generate localized polarized electric fields, facilitate charge redistribution, and cooperatively optimize the adsorption/desorption energetics of water and reaction intermediates, thereby overcoming the limitations of single-phase catalysts.194,195 Zhang et al.72 prepared IrNi nanoclusters at 300 °C and constructed a Mo2N/MoO3@NC-30 heterojunction by exploiting lattice mismatch to induce interfacial vacancies. Experimental and theoretical results collectively identified interfacial vacancies as key contributors to enhanced interfacial polarization and stabilization of high-valence Mo species in the vicinity of N sites. Such electronic regulation suppressed *OH adsorption while favoring *H desorption to H2, ultimately boosting catalytic activity. Moreover, the “spillover effect” induced by heterointerfaces optimized intermediate coverage, maintained an optimal adsorption balance, and suppressed active-site poisoning, providing an intrinsic driving force for HER enhancement.196,197 Zhang et al.197 anchored Ru clusters onto Mn3O4via Ru–O–Mn linkages to construct an Ru@Mn3O4 heterostructure with compressive strain. Compared with Mn3O4 obtained by simple physical mixing without compressive strain, this structure exhibited superior HER catalytic performance. In Ru and Mn3O4 single-component systems, in situ Raman spectra showed progressively intensified M–O (M = Ru, Mn) vibrations with increasing potential, reflecting sustained water dissociation and the accumulation of surface OH species. In contrast, the OH adsorption signal weakened with increasing potential for Ru@Mn3O4 with compressive strain, suggesting that the heterointerface provided an efficient OH desorption pathway (Fig. 14a). According to DFT analysis, Mn3O4 favored water dissociation owing to its high oxophilicity, while Ru–O–Mn interfacial sites with weaker OH binding acted as desorption channels for accumulated OH species, enabling efficient OH spillover (Fig. 14b). This offered a new insight into optimizing the HER. More intriguingly, beyond the deliberate construction of heterojunctions, catalysts may also undergo in situ structural reconstruction under electrochemical operating conditions, spontaneously generating new heterointerfaces. Wang et al.198 tracked the dynamic surface evolution of BaRuO3 (BRO) by TEM, showing a transition from an initially smooth surface to a stabilized Ru/BaRuO3 heterostructure during operation. Nanoscale clusters appeared within 127 s, evolved into metallic Ru domains with clear lattice fringes by 453 s and eventually stabilized as in situ exsolved Ru nanoparticles at longer times (918–1458 s), accompanied by abundant VO (Fig. 14c), which resulted in significantly enhanced HER performance. This study demonstrated that the synergistic interplay between in situ reconstruction and defect generation could substantially improve the intrinsic catalytic properties, offering an important design paradigm for alkaline HER catalysts.
 | ||
| Fig. 14 (a) In situ Raman spectra of Ru@Mn3O4.197 Copyright 2025 Wiley-VCH. (b) *OH adsorption energies on Ru NPs, Ru–O–Mn sites, Ru-side, Ru-top, Ru NCs, and Mn3O4.197 Copyright 2025 Wiley-VCH. (c) HRTEM images of initial BRO showing in situ Ru exsolution over time under TEM observation (time indicated in seconds).198 Copyright 2025 American Chemical Society. (d) Annular correlative HAADF-STEM (AC HAADF-STEM, the jigsaw in the figure is the active microregion).199 Copyright 2025 Wiley-VCH. (e) Front and side views of differential charge density for the optimal O sites in different microregions.199 Copyright 2025 Wiley-VCH. | ||
Defects can effectively break local coordination symmetry and regulate metal–hydrogen interactions, driving the ΔGH* toward the thermodynamically optimal region. Zhao et al. and Tsai et al.204,205 introduced controllable VS into the basal plane of MoS2. According to DFT analysis, the approach of ΔGH* toward thermoneutrality at vacancy-adjacent sites enabled the catalytic activation of the intrinsically inert basal plane. Building on this concept, Han et al.206 employed NiPS3 as the model system to systematically investigate the effects of neighboring Ni vacancies (VNi) and VS on water adsorption/dissociation and the electrochemical adsorption behavior of generated protons on the catalyst surface. With further removal of Ni atoms adjacent to VS, water molecules underwent self-orientation, enabling H atoms to form hydrogen bonds with neighboring S atoms and thereby stabilizing molecular physisorption. Meanwhile, the synergistic introduction of dual vacancies dramatically reduced the water dissociation energy from 0.88 eV to −0.91 eV. In this process, water molecules adsorbed at VS sites in a “pseudo-substitutional” manner, followed by dissociation, with the resulting OH and H species occupying VS and VNi, respectively. The resulting Ni–S dual-vacancy structure exhibited ΔGH* (−0.09 eV) close to the ideal value, leading to an overall enhancement in HER activity. It is worth noting that these studies are primarily based on static structural and energetic analyses. Direct experimental evidence for capturing the real-time occupation, release and dynamic evolution of vacancies during catalysis remains scarce and represents a critical challenge for future investigations.
In addition, Zheng et al.199 constructed a porous layered MoO3−x/Mo2AlB2 structure via an alkaline hydrothermal etching-intercalant intercalation synergistic strategy. The resulting material was subsequently mixed with ruthenium chloride (RuCl3) and treated via ultrafast Joule heating to obtain Ru–MoO3−x/Mo2AlB2 composite catalysts. As evidenced by EPR and XAS analyses, the introduction of abundant VO and lattice distortions within molybdenum oxide layers could be attributed to the synergistic action of etching and doping. These defect-rich regions promoted the formation of active microdomains, in which Ru atoms and VO were preferentially enriched and acted as the dominant catalytically active centers (verified by AC HAADF-STEM characterization, Fig. 14d). AIMD analysis of the active microdomains showed preferential stabilization of Ru atoms at sites adjacent to VO, corresponding to the most thermodynamically favorable configurations. Further analysis revealed that within the microdomains, the ΔGH* values of Mo and Ru sites were significantly higher than those of O atoms adjacent to Ru–Ov–Mo motifs, whereas the latter exhibited ΔGH* values close to zero, indicating that electron-rich oxygen sites served as the primary HER active centers (Fig. 14e). This finding challenged the conventional paradigm of metal-centered activity, demonstrating that the cooperative formation of VO and Ru doping induced electron accumulation on oxygen sites, rendering them highly efficient hydrogen evolution centers. Moreover, coupling the layered conductive support with low-crystallinity active phases endowed the system with both high structural stability and electron-unsaturated metal dangling bonds characteristic of amorphous materials, thereby markedly enhancing HER activity. Benefitting from these structural and electronic modulations, the catalyst delivered a current density of 10 mA cm−2 at an overpotential of only 38 mV in acidic media and exhibited excellent long-term stability.
6.2 Complex water electrolysis systems
With the continuous advancement of research, water electrolysis systems have gradually evolved from conventional acidic and alkaline electrolytes with simple compositions to more complex aqueous environments.207 Among these, seawater electrolysis, as the representative complex system, has emerged as a prominent research focus in the field of renewable energy. Currently, seawater electrolysis research has primarily emphasized alkaline and neutral-media electrolysis systems. While the preceding sections mainly discussed the role of defect engineering in regulating electronic configurations, intermediate adsorption, and reaction pathways for the HER/OER in freshwater systems, the complex ionic environment inherent to seawater electrolysis exerts a much more complicated influence on catalytic activity and structural stability (Fig. 15). On the anodic side of seawater electrolysis, high concentrations of Cl− exhibit pronounced thermodynamic and kinetic competition with the OER.208,209 On the one hand, chloride oxidation can proceed in parallel with the OER, significantly compromising reaction selectivity. On the other hand, Cl− and its oxidative derivatives are highly corrosive toward anode materials, readily inducing metal dissolution, lattice degradation and active-site passivation,210,211 thereby accelerating catalyst deactivation. Simultaneously, on the cathodic side, water reduction (H2O → H2 + OH−) leads to a sharp local increase in pH, providing direct thermodynamic driving force for the precipitation of Ca2+ and Mg2+ as hydroxides. The resulting Mg(OH)2/Ca(OH)2 deposits severely impede charge transfer and mass transport, representing another dominant degradation pathway in seawater electrolysis. The following section systematically summarizes the regulation strategies and underlying mechanisms of defect engineering in addressing the typical challenges of seawater electrolysis (Fig. 15). | ||
| Fig. 15 Schematic illustration of the main challenges and corresponding mitigation strategies in seawater electrolysis. | ||
To mitigate anodic Cl− corrosion, current strategies primarily rely on the construction of dense oxide protective layers or the incorporation of polyanionic species with inherent Cl−-repelling capability.212,213 While these approaches can partially suppress chloride-induced degradation, dense protective layers often impede access to OER active sites. Meanwhile, polyanionic species tend to suffer from gradual depletion at industrially relevant current densities owing to intense gas evolution and electrolyte convection, ultimately compromising catalytic activity and long-term stability.214,215 To overcome these limitations, Li et al.216 proposed an alternative interface design strategy distinct from the conventional “coating-isolation” paradigm. By epitaxially depositing catalytic nanoparticles onto heterophase nanorods, the NiTe/Ni2FeP heterojunction catalyst was constructed, which intrinsically avoided physical blockage of active sites and enabled both high activity and durability for direct seawater electrolysis. The abundant interfacial regions in this architecture induced a built-in electric field (BEF). Molecular dynamics simulations revealed that the BEF, in synergy with the applied electric field, enhanced interfacial enrichment and adsorption of OH− while effectively suppressing competitive Cl− dsorption, thereby markedly improving OER selectivity. As a result, this catalyst exhibited stable operation for over 6800 h at industrial current densities, demonstrating outstanding activity and structural robustness (Fig. 16a and b). Moreover, hard acidic metal sites preferentially adsorbed OH− during anion competition processes according to the hard–soft acid–base (HSAB) theory.217 Building on this principle, Wang et al.218 introduced VO into Fe-doped nickel hydroxide (NiII–Fe) with an area of approximately 2000 cm−2via two-step impregnation synthesis. Experimental and theoretical analyses revealed the “maturation” mechanism of VO that significantly promoted the formation of high-valence Ni4+ species, thereby enhancing the hard-acid character of Ni sites. Specifically, VO regulated local charge redistribution, reduced proton desorption energy and tuned the antibonding orbitals of Ni–O bonds, narrowing the bandgap and substantially lowering the energy barriers for the Ni2+ → Ni3+ → Ni4+ oxidation transitions (Fig. 16c). The enrichment of OH− species near vacancy-rich catalysts was supported by AIMD simulations, with a concentration of 4.4 M at 6 Å above the electrode surface compared to 1.3 M for vacancy-free samples. In contrast, the Cl− concentration at 14 Å is reduced to ∼45% of that in the control. These results collectively highlighted the exceptional Cl− corrosion resistance and high OER selectivity of VO-engineered NiFe catalysts. When applied in AEMWE, the catalyst delivered an operating voltage of only 1.68 V at 0.5 A cm−2, achieving hydrogen production cost lower than 50% of the U.S. DOE's 2026 target, thereby demonstrating promising scalability and practical application potential.
 | ||
| Fig. 16 (a) Long-term durability test of NiTe/Ni2FeP/NF in 1 M KOH + seawater measured at 1.6, 1.8, and 2.2 V vs. RHE, corresponding to current densities of 100, 300, and 1000 mA cm−2, respectively.216 Copyright 2026 Wiley-VCH. (b) OH− and Cl− concentrations at positions (distance) perpendicular to Ni2FeP and NiTe/Ni2FeP.216 Copyright 2026 Wiley-VCH. (c) Schematic illustration of orbital hybridization between Ni and reaction intermediates under Ni3+ and Ni4+.218 Copyright 2025 Wiley-VCH. (d) Linear sweep voltammetry (LSV) curves and overpotentials of Ru/V–NiO in simulated seawater with varying NaCl concentrations.219 Copyright 2025 Elsevier B.V. (e) In situ Raman spectra in natural seawater of B–Os at differing potentials.220 Copyright 2025 Wiley-VCH. (f) The DCD of B–Os.220 Copyright 2025 Wiley-VCH. | ||
In addition to anodic chloride corrosion, Ca2+/Mg2+ precipitation at the cathode represents another critical challenge in seawater electrolysis, originating from cathode-induced local alkalization that thermodynamically drives hydroxide formation. Liu et al.219 synthesized an Ru/V–NiO heterostructure composed of Ru nanoparticles supported on vanadium-doped nickel oxide via hydrothermal treatment followed by N2 annealing. The reduction in the water dissociation energy barrier, together with weakened Cl− adsorption at active sites, as supported by in situ Raman and DFT analyses, could be attributed to the synergistic effects of Ru/V–NiO interfacial construction and VO introduction. Consequently, the catalytic performance remained nearly unchanged even at elevated chloride concentrations, endowing the material with excellent resistance to seawater corrosion (Fig. 16d). Furthermore, Fu et al.220 rapidly synthesized interstitial boron-doped osmium (B–Os) catalysts using the microwave-assisted quasi-solid-state method, enabling precise regulation of the interfacial microenvironment. TEM- and GPA-based strain analysis demonstrated the coexistence of compressive and shear strains in the lattice, leading to lattice distortion and local atomic disorder that facilitate the exposure of catalytically active sites. In situ Raman results highlighted the role of B–Os in creating a locally acidic interfacial environment that mitigated OH− co-precipitation with Ca2+/Mg2+ during the HER (Fig. 16e). Differential charge density (DCD) analysis further indicated that interstitial boron triggers electron accumulation and the formation of negatively charged centers at Os sites (Fig. 16f). The resulting electronic structure not only electrostatically suppressed Cl− corrosion but also promoted preferential H3O+ adsorption, thereby achieving simultaneous resistance to corrosion and precipitation. When paired with a RuO2 anode in an AEM electrolyzer, the system operated in natural seawater at 1 A cm−2 with a cell voltage of only 1.73 V and maintained stable performance for over 400 h. In summary, defect engineering not only substantially enhances the intrinsic HER/OER activity in seawater systems but also strengthens electrodes against precipitation, chloride corrosion and interfacial mass-transport limitations through multidimensional regulation of interfacial electronic structures and reaction microenvironments. As such, it has emerged as the core design paradigm for the development of efficient and durable seawater electrolysis systems.
7 Conclusions and perspectives
This review focuses on the structural regulation effects and structure–activity relationships of defect engineering in electrocatalytic systems, and provides a systematic survey and critical analysis of recent advances in water electrolysis-related electrocatalysis. The main contributions of this review can be summarized as follows. First, from the perspectives of structural hierarchy and chemical nature, various types of defects in electrocatalytic materials are systematically classified. The common characteristics and distinct roles of different defects in regulating local coordination environments, electronic structures, and interfacial reaction properties are comparatively analyzed, thereby establishing a unified structural framework for understanding defect-performance relationships. Second, the characteristics of chemical and physical synthesis strategies in controlling defect type, concentration, and structural stability are comprehensively summarized, highlighting the critical influence of synthesis pathways on the controllability of defect construction. Furthermore, particular emphasis is placed on the dynamic evolution behavior of defects. By integrating recent advances in in situ and operando characterization techniques, this review elucidates the intrinsic nature of defect reconstruction during material synthesis, activation, and electrocatalytic operation, underscoring the decisive importance of working-state structures over static characterization in establishing reliable structure–activity correlations. The significance of this dynamic investigation lies in two aspects: (1) it reveals the pronounced “non-static” nature of defects in electrocatalytic systems, challenging the conventional view of defects as fixed structural entities and demonstrating that they undergo continuous dynamic evolution, including-formation or self-healing, migration or aggregation, and reconstruction, under multiple external stimuli. This advances the research paradigm from static structural characterization toward dynamic mechanistic understanding. (2) It establishes a structure–activity relationship framework based on “operando defect dynamics”, overcoming the limitations of ex situ characterization in capturing real active sites and reaction pathways. By integrating in situ/operando techniques with the dynamic evolution of defects, it elucidates that catalytic performance originates from dynamic equilibrium of defect structures during the reaction, thereby providing a more reliable theoretical foundation for elucidating electrocatalytic mechanisms. Finally, the regulatory roles of defects in water electrolysis reactions are systematically summarized. The critical functions of defects in providing low-coordination active sites, modulating intermediate adsorption behavior, and facilitating key reaction pathways are clarified, further highlighting the essential role of defect engineering in overcoming the intrinsic performance limitations of conventional water-splitting electrocatalysts.Despite the significant progress achieved, defect engineering in water-electrolysis electrocatalysis still faces several fundamental scientific challenges and practical bottlenecks that require further breakthroughs. First, regarding precise defect construction and controllable regulation, existing strategies still lack sufficient capability for independent control of defect type, spatial distribution, and concentration at the atomic scale. To address this limitation, future research should adopt a thermodynamics-kinetics co-regulation framework, in which defect formation energy landscapes are tuned via precursor chemistry, reaction temperature, electrochemical potential, and strain engineering. In parallel, predictive theoretical approaches should be employed to pre-evaluate defect stability and guide application-oriented defect design, enabling truly “on-demand” construction of defect structures. Moreover strengthening multimodal characterization integration and data fusion to establish an integrated framework linking defect evolution, electronic structure, and reaction kinetics. These approaches enable precise tuning of defect density, distribution, and local coordination environments in a controllable manner. Second, the causal relationship between defect dynamic evolution and catalytic performance is yet to be fully elucidated. Although in situ techniques can capture certain aspects of defect evolution, the true evolution pathways of defects under multiscale and multiphysical-field coupling conditions remain poorly understood. Strengthening multimodal characterization and data integration is therefore essential to establish a unified framework linking defect evolution, electronic structure, and reaction kinetics. Third, achieving a balance between high catalytic activity and long-term stability remains a central challenge for defect-rich catalysts in practical applications. Excessive defect density may significantly enhance initial activity but can also induce structural collapse, uncontrolled phase transitions, or metal dissolution. Designing defect architectures that are “evolvable yet structurally stable” through heterointerface engineering, synergistic doping, or dynamic self-repair mechanisms, represents a key future direction. Finally, from an application perspective, the adaptability of defect-engineered catalysts under seawater electrolysis, wide-pH conditions, and at industrial-level high current densities still requires systematic evaluation. Integrating defect-engineering concepts with device-level design, reaction engineering, and scalable manufacturing processes is essential for translating laboratory-scale advances into practical energy-conversion technologies. Overall, with the continued advancement of in situ characterization techniques, theoretical modeling, and materials synthesis, defect engineering is expected to evolve from empirical regulation toward rational and predictive design, playing an increasingly central role in the development of efficient, stable, and sustainable electrocatalytic systems for water electrolysis (Fig. 17).
Author contributions
Xaiojun Wang and Zexing Wu wrote and edited the original draft. All authors contributed to the figures, review, and editing of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results or new data were generated or analysed as part of this review.Acknowledgements
The authors are thankful for funding support from the National Natural Science Foundation of China (52371227; 22278175), Taishan Scholar Young Talent Program (tsqn202408200), Shandong Province “Double-Hundred Talent Plan” (WST2020003), Zhejiang Provincial Natural Science Foundation of China (Grant No. LZ24B060001), and State Key Laboratory of New Textile Materials and Advanced Processing, Wuhan Textile University (FZ 2025022).References
- Y. Zhang, D. Gong, X. Yang, H. Qu, Z. Tong, X. Meng and K. Huang, Environ. Chem. Saf., 2025, 1(2), 9600033–9603940 CrossRef.
- Q.-L. Hong, X. Xiao, X. Ai, H. Liu, G.-R. Xu, Q. Xue, X. Wang, B. Y. Xia and Y. Chen, Chem. Soc. Rev., 2025, 54, 9849–9875 RSC.
- W. Song, J. Wu, J. Li, Z. Wang, T. He, W. Chen, D. Sun and J. Wu, Green Carbon, 2024, 2, 393–400 CrossRef CAS.
- J. Gao, S. C. S. Shiong and Y. Liu, Chem. Eng. J., 2023, 472, 145033 CrossRef CAS.
- Q. Zhang, Y. Li, F. Luo and Z. Yang, Chem. Commun., 2025, 61, 10747–10763 RSC.
- R. Deng, M. Guo, C. Wang and Q. Zhang, Nano Mater. Sci., 2024, 6, 139–173 CrossRef CAS.
- Y. Lu, L. Zhou, S. Wang and Y. Zou, Nano Res., 2023, 16, 1890–1912 CrossRef CAS.
- Y. Zhang, J. Liu, Y. Xu, C. Xie, S. Wang and X. Yao, Chem. Soc. Rev., 2024, 53, 10620–10659 RSC.
- Y. Wang, P. Han, X. Lv, L. Zhang and G. Zheng, Joule, 2018, 2, 2551–2582 CrossRef CAS.
- H. Arandiyan, S. S. Mofarah, Y. Wang, C. Cazorla, D. Jampaiah, M. Garbrecht, K. Wilson, A. F. Lee, C. Zhao and T. Maschmeyer, Chem.–Eur. J., 2021, 27, 14418–14426 CrossRef CAS PubMed.
- X. Zheng, S. S. Mofarah, C. Cazorla, R. Daiyan, A. A. Esmailpour, J. Scott, Y. Yao, S. Lim, V. Wong, E. Y. Chen, H. Arandiyan, P. Koshy and C. C. Sorrell, Adv. Funct. Mater., 2021, 31, 2103171 CrossRef CAS.
- D. Hao, J. Ren, Y. Wang, H. Arandiyan, M. Garbrecht, X. Bai, H. K. Shon, W. Wei and B.-J. Ni, Adv. Energy Mater., 2021, 2021, 12 Search PubMed.
- X. Geng, M. Vega-Paredes, Z. Wang, C. Ophus, P. Lu, Y. Ma, S. Zhang, C. Scheu, C. H. Liebscher and B. Gault, Nat. Commun., 2024, 15, 8534 CrossRef PubMed.
- Y. Zhang, H. Liu, S. Zhao, C. Xie, Z. Huang and S. Wang, Adv. Mater., 2023, 35, 2209680 CrossRef CAS PubMed.
- M. Zhang, B. Zhou, L. Guo, H. Li, W. Xiao, G. Xu, D. Chen, C. Li, Y. Du, Z. Wu and L. Wang, Inorg. Chem. Front., 2024, 11, 5884–5893 RSC.
- T. Wu, C. Dong, D. Sun and F. Huang, Nanoscale, 2021, 13, 1581–1595 RSC.
- X. Zhou, T. Yang, T. Li, Y. Zi, S. Zhang, L. Yang, Y. Liu, J. Yang and J. Tang, Nano Res. Energy, 2023, 2, e9120086 CrossRef.
- J. Li, R. Guo, X. Zang, Y. Wang, H. Li, W. Xiao, L. Xin, Y. Zong, G. Fu, Z. Wu and L. Wang, Inorg. Chem. Front., 2025, 12, 821–832 RSC.
- B. Sun, W. Zhong, H. Liu, X. Ai, S. Han and Y. Chen, EnergyChem, 2025, 7, 100148 CrossRef CAS.
- Z.-H. Yuan, T.-J. Wang, B. Sun, Q.-L. Hong, X. Ai, S.-N. Li, J. Bai and Y. Chen, Appl. Catal., B, 2025, 366, 125041 CrossRef CAS.
- J. X. Zheng, D. P. Meng, J. X. Guo, X. B. Liu, L. Zhou and Z. Wang, Adv. Mater., 2024, 36, 2405129 CrossRef CAS PubMed.
- Y. Q. Chen, X. Y. Li and P. F. Ou, Front. Energy, 2025, 19, 59–68 CrossRef CAS.
- H. Y. Zhang, L. L. Wu, R. H. Feng, S. H. Wang, C. S. Hsu, Y. M. Ni, A. Ahmad, C. R. Zhang, H. F. Wu, H. M. Chen, W. Zhang, Y. Li, P. Liu and F. Song, ACS Catal., 2023, 13, 6000–6012 CrossRef CAS.
- Z. Z. Liu, T. Liu, Q. N. Lin, C. Y. Bao and L. Y. Zhu, Angew. Chem., Int. Ed., 2015, 54, 174–178 CrossRef CAS PubMed.
- V. H. Hoa, S. Sidra, H. T. Dao, M. Mai, V. Rajendiran, S. W. Han and D. W. Kim, Appl. Catal., B, 2026, 380, 125800 CrossRef CAS.
- M. Zhou, Y. Zhao, Z. W. Liu, X. R. Zhao, E. Z. Liu, L. Y. Xiao, P. F. Yin, C. K. Dong, H. Liu, X. W. Du and J. Yang, Nano Res., 2024, 17, 4711–4719 CrossRef CAS.
- Y. J. Liu, Y. B. Xu, Y. Tian, S. Guo, H. F. Li, X. Y. Yuan, Z. J. Zhao, B. C. Xu, X. J. Wang, P. Wang and Z. M. Liu, ACS Catal., 2025, 15, 3378–3390 CrossRef CAS.
- X. Zhong, E. X. Yuan, F. Yang, Y. Liu, H. Lu, J. Yang, F. Gao, Y. Zhou, J. M. Pan, J. W. Zhu, C. Yu, C. Z. Zhu, A. H. Yuan and E. H. Ang, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2306673120 CrossRef CAS.
- R. Jiang, Y. M. Da, J. F. Zhang, H. Wu, B. B. Fan, J. H. Li, J. J. Wang, Y. D. Deng, X. P. Han and W. B. Hu, Appl. Catal., B, 2022, 316, 121682 CrossRef CAS.
- Y. Wang, H. Arandiyan, S. S. Mofarah, X. J. Shen, S. A. Bartlett, P. Koshy, C. C. Sorrell, H. Y. Sun, C. Pozo-Gonzalo, K. Dastafkan, S. Britto, S. K. Bhargava and C. Zhao, Adv. Mater., 2024, 36, 2402156 CrossRef CAS.
- J. H. Yang, L. L. An, S. Wang, C. H. Zhang, G. Y. Luo, Y. Q. Chen, H. Y. Yang and D. L. Wang, Chin. J. Catal., 2023, 55, 116–136 CrossRef CAS.
- Y. Y. Liao, R. C. He, W. H. Pan, Y. Li, Y. Y. Wang, J. Li and Y. X. Li, Chem. Eng. J., 2023, 464, 142669 CrossRef CAS.
- Z. Xiao, C. Xie, Y. Wang, R. Chen and S. Wang, J. Energy Chem., 2021, 53, 208–225 CrossRef CAS.
- S. N. Fan, G. C. Yang, Y. Q. Jiao, Y. Liu, J. Q. Wang, H. J. Yan and H. G. Fu, Adv. Mater., 2025, 37, 2502523 CrossRef CAS PubMed.
- K. W. Bo, C. Z. Feng, S. S. Tang, Y. T. Wu, Q. J. Tao, Q. Lan, H. J. Zhang and Y. Wang, Appl. Catal., B, 2026, 385, 126272 CrossRef CAS.
- S. J. Guan, Y. L. Cheng, L. Hao, H. Yoshida, C. Tarashima, T. Z. Zhan, T. Itoi, T. B. Qiu and Y. Lu, Sci. Rep., 2023, 13, 14105 CrossRef CAS PubMed.
- X. S. Kang, G. H. Dong and T. T. Dong, ACS Appl. Energy Mater., 2023, 6, 1025–1036 CrossRef CAS.
- J. W. Guo, H. B. Zhao, Z. W. Yang, L. W. Wang, A. Z. Wang, J. Zhang, L. H. Ding, L. F. Wang, H. Liu and X. Yu, Adv. Funct. Mater., 2024, 34, 2315714 CrossRef CAS.
- G. Gao, Y. T. Deng, X. K. Wang, J. Z. Wang, T. Li, C. Chen, Y. Q. Wang, Y. M. Li, M. M. Hu, G. C. Li, L. Wang, Z. J. Li, G. M. Zhou, J. Zhao and D. S. Wang, Adv. Funct. Mater., 2025, 35, 2508406 CrossRef CAS.
- Y. Zhao, B. Zhang, B. Xia, Z. Liu, C. Tan, C. Song, M. Fujii, L. Ma and M. Song, ACS ES&T Eng., 2024, 4, 2025–2035 Search PubMed.
- Q. B. Jiang, H. F. Xu, K. S. Hui, Y. J. Wei, L. W. Liu, Z. Q. Ye, C. Y. Zha, M. T. Zheng, J. Lu and K. N. Hui, Adv. Mater., 2025, 37, 2415986 CrossRef CAS PubMed.
- J. J. Yang, Z. Y. Cao, Y. J. Wan, S. H. Guan, B. Jiang, Y. Yamauchi and H. X. Li, Adv. Energy Mater., 2025, 15, 2404936 CrossRef CAS.
- W. J. He, R. Zhang, J. Y. Zhang, F. Q. Wang, Y. Li, J. L. Zhao, C. Chen, H. Liu and H. L. Xin, J. Catal., 2022, 410, 112–120 CrossRef CAS.
- Z. H. Yin, Y. M. Fan, K. P. Song, L. Chen, H. Tan, C. Q. Li, Y. P. Zhang, H. G. Zhang and J. J. Wang, Adv. Funct. Mater., 2025, 36, e12013 CrossRef.
- Z. Y. Li, D. Wang, H. G. Kang, Z. N. Shi, X. W. Hu, H. B. Sun and J. L. Xu, J. Mater. Chem. A, 2024, 13, 587–594 RSC.
- Y. S. Lv, S. X. Bi, Z. M. Ma, Y. Shu, P. F. Zhang and Z. H. Luo, ACS Catal., 2025, 15, 19135–19147 CrossRef CAS.
- A. K. Rai, A. A. Shah, A. B. Dar, J. Kumar and M. Shrivastava, Small Methods, 2025, 9, 2401001 CrossRef CAS.
- Z. Chen, N. Han, W. Wei, D. Chu and B.-J. Ni, EcoEnergy, 2024, 2, 114–140 CrossRef CAS.
- Y.-N. Zhou, W.-L. Yu, H.-J. Liu, R.-Y. Fan, G.-Q. Han, B. Dong and Y.-M. Chai, EcoEnergy, 2023, 1, 425–436 CrossRef.
- Y. Qi, B. W. Liu, X. Q. Qiu, X. Z. Zeng, Z. C. Luo, W. D. Wu, Y. C. Liu, L. H. Chen, X. H. Zu, H. F. Dong, X. L. Lin and Y. L. Qin, Adv. Mater., 2023, 35, 2208284 CrossRef CAS PubMed.
- S. Dyjak, B. J. Jankiewicz, S. Kaniecki and W. Kicinski, Green Chem., 2024, 26, 2985–3020 RSC.
- G. Wang, Y. Sun, Y. Zhao, C. Deng, Y. Zhu and Y. Li, Nano Res., 2025, 18, 94907165 CrossRef CAS.
- S. H. Wang, M. Li, H. L. Tang and H. N. Zhang, Small, 2025, 21, 2411397 CrossRef CAS PubMed.
- Q. M. Peng, J. Q. Zhu, F. L. Wei, T. T. Isimjan, T. X. Sun, X. L. Yang, Y. F. Zhu and B. Wu, Appl. Catal., B, 2026, 384, 126158 CrossRef CAS.
- X. W. Fu, X. C. Zang, J. X. Gao, H. D. Li, W. P. Xiao, Y. X. Zong, G. Y. Fu, J. S. Wang, T. Y. Ma, W. Jin, Z. X. Wu and L. Wang, Adv. Energy Mater., 2025, 15, 2501054 CrossRef CAS.
- F. Liu and Z. X. Fan, Chem. Soc. Rev., 2023, 52, 1723–1772 RSC.
- K. Zhou, Y. S. Zhang, M. Y. Zhang, A. Rezayan, Z. Quan, D. D. Han, J. S. Wang, D. Wu and C. B. Xu, Chem. Eng. J., 2024, 492, 152347 CrossRef CAS.
- B. B. Liu, H. L. Liu, S. Z. Sun, Z. X. Ma, Y. X. Zhang, R. F. Nie and J. Fu, Adv. Funct. Mater., 2025, 35, 2504172 CrossRef CAS.
- Y. T. Li, C. R. Zhao, W. Wang, X. Wu and Y. D. Huang, Mater. Chem. Front., 2025, 9, 243–252 RSC.
- S. D. Yang, X. Guo, X. N. Li, T. Z. Wu, L. H. Zou, Z. Y. He, Q. Xu, J. J. Zheng, L. Chen, Q. Y. Wang and Z. J. Xu, Angew. Chem., Int. Ed., 2024, 63, e202317957 CrossRef CAS PubMed.
- Z. Sofer, Y. Zuo, N. Antonatos, L. Dekanovsky, J. Luxa, J. D. Elliott, D. Gianolio, J. Saturala, F. Guzzetta, S. Mourdikoudis, J. Regner and R. Melek, ACS Catal., 2023, 13, 2601–2609 CrossRef.
- J. L. Zhu, J. M. Qian, X. B. Peng, B. R. Xia and D. Q. Gao, Nano-Micro Lett., 2023, 15, 30 CrossRef CAS.
- L. S. Peng, N. Yang, Y. Q. Yang, Q. Wang, X. Y. Xie, D. Sun-Waterhouse, L. Shang, T. R. Zhang and G. I. N. Waterhouse, Angew. Chem., Int. Ed., 2021, 60, 24612–24619 CrossRef CAS PubMed.
- Y. Zhou, D. F. Yan, Q. F. Gu, S. H. Zhu, L. Wang, H. G. Peng and Y. Zhao, Appl. Catal., B, 2021, 285, 119792 CrossRef CAS.
- J. Jin, X. Han, Y. Y. Fang, Z. D. Zhang, Y. P. Li, T. Y. Zhang, A. J. Han and J. F. Liu, Adv. Funct. Mater., 2022, 32, 2109218 CrossRef CAS.
- P. L. Zhai, M. Y. Xia, Y. Z. Wu, G. H. Zhang, J. F. Gao, B. Zhang, S. Y. Cao, Y. T. Zhang, Z. W. Li, Z. Z. Fan, C. Wang, X. M. Zhang, J. T. Miller, L. C. Sun and J. G. Hou, Nat. Commun., 2021, 12, 4587 CrossRef CAS PubMed.
- P. Zhou, Y. Y. Wang, C. Xie, C. Chen, H. W. Liu, R. Chen, J. Huo and S. Y. Wang, Chem. Commun., 2017, 53, 11778–11781 RSC.
- L. Li, Z. D. Qin, L. Ries, S. Hong, T. Michel, J. Yang, C. Salameh, M. Bechelany, P. Miele, D. Kaplan, M. Chhowalla and D. Voiry, ACS Nano, 2019, 13, 6824–6834 CrossRef CAS PubMed.
- M. M. Yan, Z. X. Wei, Z. C. Gong, B. Johannessen, G. L. Ye, G. C. He, J. J. Liu, S. L. Zhao, C. Y. Cui and H. L. Fei, Nat. Commun., 2023, 14, 368 CrossRef CAS.
- K. Huang, Z. X. Wei, J. B. Liu, Z. C. Gong, J. J. Liu, M. N. Yan, G. C. He, H. S. Gong, Y. F. Hu, Y. M. He, S. L. Zhao, G. L. Ye and H. L. Fei, Small, 2022, 18, 2201139 CrossRef CAS PubMed.
- Q. C. Wang, Q. G. Feng, Y. P. Lei, S. H. Tang, L. Xu, Y. Xiong, G. Z. Fang, Y. C. Wang, P. Y. Yang, J. J. Liu, W. Liu and X. Xiong, Nat. Commun., 2022, 13, 3689 CrossRef CAS PubMed.
- X. Zhang, Y. Jiang, T. Wang, C. Wang, D. Wang, Y. Qiu and Y. Zhang, Mater. Futures, 2025, 5, 015103 CrossRef.
- J. J. Liu, Z. X. Wei, Z. C. Gong, M. M. Yan, Y. F. Hu, S. L. Zhao, G. L. Ye and H. L. Fei, Appl. Catal., B, 2023, 324, 122267 CrossRef CAS.
- H. Ma, W. S. Yan, Y. L. Yu, L. H. Deng, Z. Hong, L. Song and L. Li, Nanoscale, 2023, 15, 1357–1364 RSC.
- X. Zhou, M. K. Ke, G. X. Huang, C. Chen, W. X. Chen, K. Liang, Y. T. Qu, J. Yang, Y. Wang, F. T. Li, H. Q. Yu and Y. E. Wu, Proc. Natl. Acad. Sci. U. S. A., 2022, 119, e2119492119 CrossRef CAS.
- Z. Liu, X. X. Wang, R. Guo, J. J. Richardson, T. Z. Wang, W. J. Xu, F. Caruso and S. J. Pan, Adv. Funct. Mater., 2023, 33, 2211563 CrossRef CAS.
- P. Shen, X. C. Li, Y. J. Luo, Y. L. Guo, X. L. Zhao and K. Chu, ACS Nano, 2022, 16, 7915–7925 CrossRef CAS PubMed.
- H. L. Fei, J. C. Dong, Y. X. Feng, C. S. Allen, C. Z. Wan, B. Volosskiy, M. F. Li, Z. P. Zhao, Y. L. Wang, H. T. Sun, P. F. An, W. X. Chen, Z. Y. Guo, C. Lee, D. L. Chen, I. Shakir, M. J. Liu, T. D. Hu, Y. D. Li, A. I. Kirkland, X. F. Duan and Y. Huang, Nat. Catal., 2018, 1, 63–72 CrossRef CAS.
- Y. Q. Zhang, L. Tao, C. Xie, D. D. Wang, Y. Q. Zou, R. Chen, Y. Y. Wang, C. K. Jia and S. Y. Wang, Adv. Mater., 2020, 32, 1905923 CrossRef CAS PubMed.
- Q. Wang, H. Xu, X. Y. Qian, G. Y. He and H. Q. Chen, Appl. Catal., B, 2023, 322, 122104 CrossRef CAS.
- L. M. Wang, L. L. Zhang, W. Ma, H. Wan, X. J. Zhang, X. Zhang, S. Y. Jiang, J. Y. Zheng and Z. Zhou, Adv. Funct. Mater., 2022, 32, 2203342 CrossRef CAS.
- H. W. Zhao, S. L. Zhu, Z. D. Cui, Z. Y. Li, S. L. Wu, W. C. Xu, Z. H. Gao, Y. Q. Liang, L. L. Ma and H. Jiang, Adv. Funct. Mater., 2025, 35, 2423549 CrossRef CAS.
- Y. Q. Zhang, J. J. Liu, Y. F. Xu, C. Xie, S. Y. Wang and X. D. Yao, Chem. Soc. Rev., 2024, 53, 10620–10659 RSC.
- H. Z. Yu, M. Y. Hu, C. Chen, C. J. Hu, Q. H. Li, F. Hu, S. J. Peng and J. Ma, Angew. Chem., Int. Ed., 2023, 62, e202314569 CrossRef CAS PubMed.
- M. Boutonnet Kizling and S. G. Järås, Appl. Catal., A, 1996, 147, 1–21 CrossRef CAS.
- W. Chu, L. N. Wang, P. A. Chernavskii and A. Y. Khodakov, Angew. Chem., Int. Ed., 2008, 47, 5052–5055 CrossRef CAS PubMed.
- Z. Wang, Y. Zhang, E. C. Neyts, X. X. Cao, X. S. Zhang, B. W. L. Jang and C. J. Liu, ACS Catal., 2018, 8, 2093–2110 CrossRef CAS.
- Y. Y. Wang, Y. Q. Zhang, Z. J. Liu, C. Xie, S. Feng, D. D. Liu, M. F. Shao and S. Y. Wang, Angew. Chem., Int. Ed., 2017, 56, 5867–5871 CrossRef CAS PubMed.
- F. Q. Wang, Y. Y. Zhang, W. H. Yuan, J. Mao, K. Wang, Y. Li, C. Chen, L. M. Liang and C. C. Liu, ACS Appl. Nano Mater., 2023, 6, 3848–3855 CrossRef CAS.
- G. L. Ye, Y. J. Gong, J. H. Lin, B. Li, Y. M. He, S. T. Pantelides, W. Zhou, R. Vajtai and P. M. Ajayan, Nano Lett., 2016, 16, 1097–1103 CrossRef CAS PubMed.
- Z. J. Xu, J. L. Wu, W. H. Chen, Z. Q. Jiang, J. Cao, G. L. Chen and Z. J. Jiang, Adv. Funct. Mater., 2025, 35, e11117 CrossRef CAS.
- C.-J. Liu, J. Zou, K. Yu, D. Cheng, Y. Han, J. Zhan, C. Ratanatawanate and B. W. L. Jang, Pure Appl. Chem., 2006, 78, 1227–1238 CrossRef CAS.
- X. R. Zhuge, C. F. Wang, Y. H. Li, Y. X. Zong, W. P. Xiao, D. H. Chen, J. S. Wang, T. Y. Ma, M. Song, Z. X. Wu and L. Wang, Sci. China Mater., 2025, 68, 4471–4480 CrossRef.
- E. Z. Li, Y. Z. Shen, Y. H. Li, Y. X. Zong, W. P. Xiao, G. R. Xu, J. S. Wang, H. Li, T. Y. Ma, Z. X. Wu and L. Wang, Appl. Catal., B, 2026, 382, 126022 CrossRef CAS.
- C. Gabriel, S. Gabriel, E. H. Grant, E. H. Grant, B. S. J. Halstead and D. M. P. Mingos, Chem. Soc. Rev., 1998, 27, 213–224 RSC.
- J. Huang, X. Xu, Y. Yan, Y. Zheng, Y. Yao, Z. Li, Y. Yan, K. N. Hui, J. Zou and M. Liu, Adv. Energy Mater., 2025, 15, 2500360 CrossRef CAS.
- Y. H. Zhang, S. Wu, T. Sun, Q. G. Li and G. Y. Fan, J. Colloid Interface Sci., 2025, 681, 71–81 CrossRef CAS PubMed.
- M. A. Khan, M. S. Akhtar and O.-B. Yang, Sol. Energy, 2010, 84, 2195–2201 CrossRef CAS.
- D. S. Zhang, B. Goekce and S. Barcikowski, Chem. Rev., 2017, 117, 3990–4103 CrossRef CAS PubMed.
- R. Q. Ye, D. K. James and J. M. Tour, Acc. Chem. Res., 2018, 51, 1609–1620 CrossRef CAS PubMed.
- B. Wang, X. Zhu, X. D. Pei, W. G. Liu, Y. C. Leng, X. W. Yu, C. Wang, L. H. Hu, Q. M. Su, C. P. Wu, Y. F. Yao, Z. Q. Lin and Z. G. Zou, J. Am. Chem. Soc., 2023, 145, 13788–13795 CrossRef CAS.
- W. D. Yi, H. Q. Jiang and G. J. Cheng, Small, 2022, 18, 2202403 CrossRef CAS PubMed.
- S. Y. Liu and C. R. Liu, Cryst. Growth Des., 2019, 19, 2912–2918 CrossRef CAS.
- W. H. Guo, Y. F. Zhang, J. J. Su, Y. Song, L. B. Huang, L. Cheng, X. H. Cao, Y. B. Dou, Y. B. Ma, C. Y. Ma, H. Zhu, T. T. Zheng, Z. Y. Wang, H. Li, Z. X. Fan, Q. Liu, Z. Y. Zeng, J. C. Dong, C. A. Xia, B. Z. Tang and R. Q. Ye, Small, 2022, 18, 2201311 CrossRef CAS.
- Z. H. Xiao, X. B. Huang, L. Xu, D. F. Yan, J. Huo and S. Y. Wang, Chem. Commun., 2016, 52, 13008–13011 RSC.
- X. L. Fan, D. W. Chang, X. L. Chen, J. B. Baek and L. M. Dai, Curr. Opin. Chem. Eng., 2016, 11, 52–58 CrossRef.
- I. Y. Jeon, S. Y. Bae, J. M. Seo and J. B. Baek, Adv. Funct. Mater., 2015, 25, 6961–6975 CrossRef CAS.
- X. S. Qiao, H. J. Kang, Y. Li, K. Cui, X. Jia, X. H. Wu and W. Qin, Appl. Catal., B, 2022, 305, 121034 CrossRef CAS.
- Y. Zhao, B. Zhang, B. Xia, Z. Liu, C. Tan, C. Song, M. Fujii, L. Ma and M. Song, ACS ES&T Eng., 2024, 4, 2025–2035 Search PubMed.
- A. P. Amrute, J. De Bellis, M. Felderhoff and F. Schüth, Chem.–Eur. J., 2021, 27, 6819–6847 CrossRef CAS PubMed.
- E. L. Lawrence, B. D. A. Levin, T. Boland, S. L. Y. Chang and P. A. Crozier, ACS Nano, 2021, 15, 2624–2634 CrossRef CAS PubMed.
- C. Zhang, B. H. Chen, D. H. Mei and X. Liang, J. Mater. Chem. A, 2019, 7, 5475–5481 RSC.
- X. H. Sun, D. X. Wu, W. H. Zhu, X. B. Chen, R. Sharma, J. C. Yang and G. W. Zhou, J. Phys. Chem. Lett., 2021, 12, 9547–9556 CrossRef CAS PubMed.
- F. Esch, S. Fabris, L. Zhou, T. Montini, C. Africh, P. Fornasiero, G. Comelli and R. Rosei, Science, 2005, 309, 752–755 CrossRef CAS.
- F. X. Zhang, B. Y. Li, X. Quan, K. Wang, J. Y. Xu, T. T. Wu, Z. L. Li, M. Yan, S. J. Liu, Y. He, Y. Shi, Y. Q. Su and P. F. Xie, ACS Catal., 2024, 14, 7136–7148 CrossRef CAS.
- Z. X. Li, H. C. Yang, C. W. Zhong, S. W. Deng and E. Z. Li, Acta Mater., 2026, 303, 121740 CrossRef CAS.
- J. G. Lin, F. Liu, W. Q. Cheng, Y. Ma, G. Xue, S. L. Liang, Y. C. Wang, P. Y. Cao, Y. Zhang and X. L. Wu, Acta Mater., 2026, 303, 121730 CrossRef CAS.
- M. Setvin, M. Schmid and U. Diebold, Phys. Rev. B, 2015, 91, 195403 CrossRef.
- L. P. Bao, S. Ali, C. H. Dai, Q. Zeng, C. Zeng, Y. S. Jia, X. Liu, P. Wang, X. H. Ren, T. Yang, M. Bououdina, Z. H. Lu, Y. C. Wei, X. Yu and Y. T. Zhou, ACS Nano, 2024, 18, 5878–5889 CAS.
- H. J. Li, K. Xi, W. Wang, S. Liu, G. R. Li and X. P. Gao, Energy Storage Mater., 2022, 45, 1229–1237 CrossRef.
- D. Q. Wang, Y. Zhang, C. Zhao, Z. Q. Li, R. L. Qiu, Q. L. Shuai, H. Q. Wang and Q. S. Hua, ACS Appl. Mater. Interfaces, 2025, 17, 45687–45695 CrossRef CAS.
- M. Harrison, D. Woodruff, J. Robinson, D. Sander, W. Pan and J. Kirschner, Phys. Rev. B: Condens. Matter Mater. Phys., 2006, 74, 165402 CrossRef.
- Y. Grunder, C. A. Lucas, P. B. Thompson, Y. Joly and Y. Soldo-Olivier, J. Phys. Chem. C, 2022, 126, 4612–4619 CrossRef CAS.
- X. H. Sun, W. H. Zhu, D. X. Wu, C. R. Li, J. Y. Wang, Y. G. Zhu, X. B. Chen, J. A. Boscoboinik, R. Sharma and G. W. Zhou, Nat. Commun., 2020, 11, 305 CrossRef CAS PubMed.
- Q. Hu, Y. J. Qin, X. D. Wang, Z. Y. Wang, X. W. Huang, H. J. Zheng, K. R. Gao, H. P. Yang, P. X. Zhang, M. H. Shao and C. X. He, Energy Environ. Sci., 2021, 14, 4989–4997 RSC.
- C. Yang, C. S. Chen, T. Y. Bian, C. Xu, X. L. Che, D. Y. Li, K. Liang, X. Z. Dong, J. Yin, G. Li and Y. Zhu, ACS Nano, 2025, 19, 9225–9231 CrossRef CAS PubMed.
- J. L. Qi, T. X. Xu, J. Cao, S. Guo, Z. X. Zhong and J. C. Feng, Nanoscale, 2020, 12, 6204–6210 RSC.
- C. L. Zhang, G. J. Liu, P. Wu, G. F. Zeng and Y. H. Sun, Chemcatchem, 2020, 12, 3512–3522 CrossRef CAS.
- J. R. Han, H. B. Wang, Y. T. Wang, H. Zhang, J. Li, Y. J. Xia, J. S. Zhou, Z. Y. Wang, M. C. Luo, Y. H. Wang, N. Wang, E. Cortes, Z. M. Wang, A. Vomiero, Z. F. Huang, H. X. Ren, X. M. Yuan, S. H. Chen, D. H. Feng, X. H. Sun, Y. C. Liu and H. Y. Liang, Angew. Chem., Int. Ed., 2024, 63, e202405839 CrossRef CAS.
- S. Li, H. Yang, H. Tong, W. Xu, Z. Wang, W. Lu, X. Deng and L. Li, Adv. Mater., 2025, 37, e11910 CrossRef CAS PubMed.
- Y. Zou, X. Li, Y. Zhao, X. Liu, S. Xie, F. Liu and T. Zhu, ACS Catal., 2025, 15, 6346–6360 CrossRef CAS.
- Y. Li, Z. Zhang, R. Wei, X. Zhang, R. Geng, F. Zheng, L. Wang, Y. Wang and S. Yin, Chem. Eng. J., 2025, 527, 171700 CrossRef.
- H. Arandiyan, S. S. Mofarah, C. C. Sorrell, E. Doustkhah, B. Sajjadi, D. Hao, Y. Wang, H. Sun, B.-J. Ni, M. Rezaei, Z. Shao and T. Maschmeyer, Chem. Soc. Rev., 2021, 50, 10116–10211 RSC.
- Z. J. Chen, Q. Shao, J. Wu, J. Zheng, S. Bao, L. Zhang, T. Zhang, X. Lan, Y. Sun and D. Wang, Angew. Chem., Int. Ed., 2025, 64, e202500678 CrossRef CAS PubMed.
- Y. Wang, T. Wang, H. Arandiyan, G. Song, H. Sun, Y. Sabri, C. Zhao, Z. Shao and S. Kawi, Adv. Mater., 2024, 36, 2313378 CrossRef CAS.
- K. Y. Zhu, X. F. Zhu and W. S. Yang, Angew. Chem., Int. Ed., 2019, 58, 1252–1265 CrossRef CAS.
- Y. Jia, L. Z. Zhang, L. Z. Zhuang, H. L. Liu, X. C. Yan, X. Wang, J. D. Liu, J. C. Wang, Y. R. Zheng, Z. H. Xiao, E. Taran, J. Chen, D. J. Yang, Z. H. Zhu, S. Y. Wang, L. M. Dai and X. D. Yao, Nat. Catal., 2019, 2, 688–695 CrossRef CAS.
- T. C. Liu, L. P. Lin, X. X. Bi, L. L. Tian, K. Yang, J. J. Liu, M. F. Li, Z. H. Chen, J. Lu, K. Amine, K. Xu and F. Pan, Nat. Nanotechnol., 2019, 14, 50–56 CrossRef CAS PubMed.
- R. G. Mariano, M. Kang, O. J. Wahab, I. J. McPherson, J. A. Rabinowitz, P. R. Unwin and M. W. Kanan, Nat. Mater., 2021, 20, 1000–1006 CrossRef CAS PubMed.
- Y. Zhao, S. Gao, X. Y. Chen, Y. Liu, M. J. Zhang, Y. F. Liu, J. X. Kang, T. Q. Guo, L. M. Liu and L. Guo, J. Am. Chem. Soc., 2025, 147, 31722–31730 CrossRef CAS PubMed.
- S. A. Bonke, T. Risse, A. Schnegg and A. Brückner, Nat. Rev. Methods Primers, 2021, 1, 33 CrossRef CAS.
- A. Brückner, Chem. Soc. Rev., 2010, 39, 4673–4684 RSC.
- J. Fischer, M. Agrachev, J. Forrer, R. Tschaggelar, O. Oberhänsli and G. Jeschke, Chimia, 2024, 78, 326–332 CrossRef CAS PubMed.
- T. P. Araújo, J. Morales-Vidal, T. S. Zou, M. Agrachev, S. Verstraeten, P. O. Willi, R. N. Grass, G. Jeschke, S. Mitchell, N. López and J. Pérez-Ramírez, Adv. Energy Mater., 2023, 13, 2204122 CrossRef.
- T. Pinheiro Araújo, C. Mondelli, M. Agrachev, T. Zou, P. O. Willi, K. M. Engel, R. N. Grass, W. J. Stark, O. V. Safonova and G. Jeschke, Nat. Commun., 2022, 13, 5610 CrossRef PubMed.
- F. Z. Sun, Z. Li, H. Xu, Y. Fu, H. Li, Y. Y. Yao, L. Ren, X. Q. He, Y. H. Li, R. Yang, N. Zhang, Z. G. Hu, T. Y. Ma and J. X. Zou, Adv. Energy Mater., 2024, 14, 2400875 CrossRef CAS.
- H. Z. Yang, N. Guo, S. B. Xi, H. Y. Zou, J. Y. Chen, L. Fan, Y. K. Xiao, Q. He, L. L. Duan, P. F. Wei, G. X. Wang, C. Zhang and L. Wang, Adv. Mater., 2025, 37, 2510192 CrossRef CAS PubMed.
- P. P. Zhang, Y. R. Yu, K. Z. Li, R. L. Yang, R. H. Hou, Y. K. Li, Y. X. Wei, M. Cai, G. S. Shao and P. Zhang, Adv. Energy Mater., 2025, 16, 2501940 CrossRef.
- Z. H. Xiao, Y. C. Huang, C. L. Dong, C. Xie, Z. J. Liu, S. Q. Du, W. Chen, D. F. Yan, L. Tao, Z. W. Shu, G. H. Zhang, H. G. Duan, Y. Y. Wang, Y. Q. Zou, R. Chen and S. Y. Wang, J. Am. Chem. Soc., 2020, 142, 12087–12095 CrossRef CAS PubMed.
- L. Z. Sun, X. Pan, Y. N. Xie, J. G. Zheng, S. H. Xu, L. Li and G. H. Zhao, Angew. Chem., Int. Ed., 2024, 63, e202402176 CrossRef CAS.
- A. C. Ferrari and D. M. Basko, Nat. Nanotechnol., 2013, 8, 235–246 CrossRef CAS PubMed.
- F. Y. Hu, R. P. Ye, C. K. Jin, D. Liu, X. H. Chen, C. Li, K. H. Lim, G. Q. Song, T. C. Wang, G. Feng, R. B. Zhang and S. Kawi, Appl. Catal., B, 2022, 317, 121715 CrossRef CAS.
- T. L. Hou, J. X. Zhu, H. F. Gu, X. Y. Li, Y. Q. Sun, Z. Hua, R. W. Shao, C. Chen, B. T. Hu, L. Q. Mai, S. H. Chen, D. S. Wang and J. T. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202424749 CrossRef CAS PubMed.
- Y. J. Wu, J. Yang, T. X. Tu, W. Q. Li, P. F. Zhang, Y. Zhou, J. F. Li, J. T. Li and S. G. Sun, Angew. Chem., Int. Ed., 2021, 60, 26829–26836 CrossRef CAS PubMed.
- X. W. Wang, Y. Y. Zhang, S. Wang, Y. F. Li, Y. F. Feng, Z. C. Dai, Y. X. Chen, X. M. Meng, J. Xia and G. Q. Zhang, Angew. Chem., Int. Ed., 2024, 63, e202407665 CrossRef CAS PubMed.
- M. S. A. Sher Shah, G. Y. Jang, K. Zhang and J. H. Park, EcoEnergy, 2023, 1, 344–374 CrossRef.
- X. Wang, H. Zhao, H. Li, W. Xiao, G. Xu, G. Fu, L. Wang and Z. Wu, Inorg. Chem. Front., 2025, 12, 8118–8127 RSC.
- X. Liu, R. Y. Liu, J. X. Qiu, X. Cheng and G. G. Li, Angew. Chem., Int. Ed., 2020, 132, 14066–14071 CrossRef.
- Y. J. Pan, Z. C. Wang, K. X. Wang, Q. Ye, B. S. Shen, F. S. Yang and Y. L. Cheng, Adv. Funct. Mater., 2024, 34, 2402264 CrossRef CAS.
- J. X. Zhao, K. J. Wang, X. Y. Li, X. M. Li, X. Y. Cui and X. Zhao, Angew. Chem., Int. Ed., 2025, 64, e202513592 CrossRef CAS PubMed.
- S. W. Ding, Z. H. Li, G. X. Lin, Y. X. Ding, L. Q. Wang and L. C. Sun, Angew. Chem., Int. Ed., 2025, 64, e202517132 CrossRef CAS PubMed.
- H. J. Niu, N. Ran, W. Zhou, W. X. An, C. X. Huang, W. X. Chen, M. Zhou, W. F. Lin, J. J. Liu and L. Guo, J. Am. Chem. Soc., 2025, 147, 2607–2615 CrossRef CAS PubMed.
- S. Qi, J. You, X. Y. Liufu, Y. Zhang, R. B. Chen, J. P. Zhuang, T. Y. Liang, L. L. Li, Q. H. Huo, C. Y. Shang, X. Zhang, H. P. Yang, Q. Hu and C. X. He, Adv. Mater., 2025, 38, e12188 CrossRef PubMed.
- T. Guo, P. Hu, L. Li, Z. Wang and L. Guo, Chem, 2023, 9, 1080–1093 CAS.
- B. Zhou, J. Zhou, L. Wang, J. H. Kang, A. Zhang, J. Zhou, D. Zhang, D. Xu, B. Hu and S. Deng, Nat. Synth., 2024, 3, 67–75 CrossRef CAS.
- B. X. Shen, S. Li, M. T. Yang, K. Ge, H. J. Xia, Q. Y. Li, F. Ge and Y. D. Hu, ACS Appl. Mater. Interfaces, 2025, 17, 29628–29638 CrossRef PubMed.
- X. M. Li, B. Han, R. X. Zhu, R. C. Shi, M. Wu, Y. W. Sun, Y. H. Li, B. Y. Liu, L. F. Wang, J. M. Zhang, C. B. Tan, P. Gao and X. D. Bai, Proc. Natl. Acad. Sci. U. S. A., 2023, 120, e2213650120 CrossRef CAS PubMed.
- J. Wu, X. H. Gao and Z. W. Chen, Chem. Eng. J., 2024, 492, 152241 CrossRef CAS.
- S. Tao, G. K. Zhang, B. Qian, J. Yang, S. Q. Chu, C. C. Sun, D. J. Wu, W. S. Chu and L. Song, Appl. Catal., B, 2023, 330, 122600 CrossRef CAS.
- Z. M. Li, J. H. Yi, Y. Tang, Z. F. Zhang, C. P. Li, R. Bao and J. S. Wang, Energy Environ. Sci., 2025, 18, 7188–7202 RSC.
- J. J. Zhang, L. L. Xu, X. X. Yang, S. Guo, Y. F. Zhang, Y. Zhao, G. Wu and G. Li, Angew. Chem., Int. Ed., 2024, 63, e202405641 CrossRef CAS PubMed.
- Y. C. Lin, Z. Q. Tian, L. J. Zhang, J. Y. Ma, Z. Jiang, B. J. Deibert, R. X. Ge and L. Chen, Nat. Commun., 2019, 10, 162 CrossRef PubMed.
- R. X. Ge, L. Li, J. W. Su, Y. C. Lin, Z. Q. Tian and L. Chen, Adv. Energy Mater., 2019, 9, 1901313 CrossRef.
- J. W. Tao, R. Q. Gao, G. Y. Lin, C. Y. Chu, Y. Sun, C. Y. Yu, Y. H. Ma and H. B. Qiu, Nat. Commun., 2025, 16, 4996 CrossRef CAS PubMed.
- S.-Y. Yuan, T.-T. Li, J.-Y. Cui, J.-K. Sun, Y.-S. Gong, A. Braun, H. Liu and J.-J. Wang, EcoEnergy, 2024, 2, 322–335 CrossRef CAS.
- H. F. Wang, C. Lin, L. Tan, J. Shen, X. T. Wu, X. X. Pan, Y. H. Zhao, H. J. Zhang, Y. Sun, B. B. Mei, H. D. Um, Q. Xiao, W. Jiang, X. P. Li and W. Luo, Nat. Commun., 2025, 16, 3976 CrossRef CAS PubMed.
- M. Jin, J. Chen, X. Han, Y. Guo, Y. Liu, J. Wang, J. Li, Y. Yuan, H. Jin, S. Wang and X. Huang, Nat. Commun., 2025, 16, 11615 CrossRef CAS PubMed.
- N. Zhang, X. Y. Liu, H. X. Zhong, W. Liu, D. Bao, J. R. Zeng, D. P. Wang, C. N. Ma and X. B. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202503246 CrossRef CAS PubMed.
- J. Yang, K. Y. An, Z. C. Yu, L. L. Qiao, Y. P. Cao, Y. J. Zhuang, C. F. Liu, L. Li, L. S. Peng and H. Pan, ACS Catal., 2024, 14, 17739–17747 CrossRef CAS.
- H. Liu, Y. Zhang, Z. Y. Liu, S. C. Wang, X. Q. Mu, Q. H. Zhang, W. Cao, S. L. Liu, D. S. Wang and Z. H. Dai, J. Am. Chem. Soc., 2025, 147, 44141–44151 CrossRef CAS PubMed.
- S. X. Li, S. Zhao, S. F. Hung, L. M. Deng, L. Q. Wang, F. K. Shi, A. Dong, Y. Zhang, T. Y. Chen, F. Hu, L. L. Li, S. Ramakrishna, Y. P. Wu and S. J. Peng, J. Am. Chem. Soc., 2025, 147, 33770–33779 CrossRef CAS PubMed.
- J. Han, M. K. Nam, S. Shin, I. Cho, H. Jeon, J. Heo, J. Lee, M. K. Cho, D. J. Preston, M. C. Hersam, I. S. Kim, B. Shong and W. K. Lee, J. Am. Chem. Soc., 2025, 147, 41096–41108 CrossRef CAS PubMed.
- Z. X. Wu, Y. L. Wang, D. Z. Liu, B. W. Zhou, P. F. Yang, R. Z. Liu, W. P. Xiao, T. Y. Ma, J. S. Wang and L. Wang, Adv. Funct. Mater., 2023, 33, 2307010 CrossRef CAS.
- Y. Liu, Y. Huang, D. J. Wu, H. Jang, J. H. Wu, H. R. Li, W. X. Li, F. Zhu, M. G. Kim, D. L. Zhou, X. K. Xi, Z. W. Lei, Y. C. Zhang, Y. Deng, W. S. Yan, M. D. Gu, J. Jiang, S. H. Jiao and R. G. Cao, J. Am. Chem. Soc., 2024, 146, 26897–26908 CrossRef CAS PubMed.
- Y. Ou, L. Liu, X. Peng, L. Zhang, Z. Ou, W. Zhang and Y. Zhang, Nano Mater. Sci., 2024, 6, 565–575 CrossRef CAS.
- X. W. Guo, E. H. Song, W. Zhao, S. M. Xu, W. L. Zhao, Y. J. Lei, Y. Q. Fang, J. J. Liu and F. Q. Huang, Nat. Commun., 2022, 13, 5954 CrossRef PubMed.
- W. Q. Zhan, X. W. Zhai, Y. H. Li, M. Wang, H. Wang, L. Wu, X. F. Tang, H. J. Zhang, B. J. Ye, K. B. Tang, G. M. Wang and M. Zhou, ACS Nano, 2024, 18, 10312–10323 CrossRef CAS PubMed.
- G. H. Zhang, S. L. Qi, H. D. Wang, X. F. Lei, Z. X. Zhang, W. Liu, X. Y. Liu, Y. Zhao, L. Yang, W. G. Ma, W. J. Fan and X. Zong, Adv. Funct. Mater., 2025, e17113 Search PubMed.
- H. Zhang, A. R. Chen, Z. H. Bi, X. Z. Wang, X. J. Liu, Q. Q. Kong, W. Zhang, L. Q. Mai and G. Z. Hu, ACS Nano, 2023, 17, 24070–24079 CrossRef CAS PubMed.
- H. Y. Wang, S. X. Zhai, H. Wang, F. X. Yan, J. T. Ren, L. Wang, M. L. Sun and Z. Y. Yuan, ACS Nano, 2024, 18, 19682–19693 CAS.
- L. Wang, X. W. Lv, H. Y. Wang, J. T. Ren, Y. Feng, M. L. Sun and Z. Y. Yuan, Adv. Energy Mater., 2025, 15, e04036 CrossRef.
- Z. Zhao, J. P. Sun, X. Li, S. Y. Qin, C. H. Li, Z. S. Zhang, Z. Z. Li and X. C. Meng, Nat. Commun., 2024, 15, 7475 CrossRef CAS PubMed.
- Y. M. Zhang, L. L. Zhao, J. Wang, Y. Liu, Z. D. Zhang, W. W. Cai, J. Z. Ma and J. T. Zhang, J. Am. Chem. Soc., 2025, 147, 27367–27376 CrossRef CAS PubMed.
- Y. Yao, E. L. Hu, Z. Y. Wang, Y. J. Cui and G. D. Qian, ACS Omega, 2022, 7, 2244–2251 CrossRef CAS PubMed.
- C. A. Zhou, K. Ma, Z. C. Zhuang, M. L. Ran, G. Q. Shu, C. Wang, L. Song, L. R. Zheng, H. R. Yue and D. S. Wang, J. Am. Chem. Soc., 2024, 146, 21453–21465 CrossRef PubMed.
- Q. Wang, S.-F. Hung, K. Lao, X. Huang, F. Li, H. B. Tao, H. B. Yang, W. Liu, W. Wang and Y. Cheng, Nat. Catal., 2025, 8, 1–11 CAS.
- C. Xu, H. Yu, H. Huang, S. Li, Y. Cao, W. Peng, Y. Li, H. Ke, S. Xu, H. Mo, C. Wu, H. Wang, Y. Zhang and X. Li, Angew. Chem., Int. Ed., 2025, 64, e202504667 CrossRef CAS PubMed.
- L. Wang, B. Li, M. Li, Y. Zhang, T. Yuan, Y. Ding, T. Li, C. Peng, S. Jiang and X. Xiong, J. Am. Chem. Soc., 2025, 148, 3128–3138 CrossRef PubMed.
- Y. Q. Yang, D. W. Pang, C. J. Wang, Z. H. Fu, N. Y. Liu, J. J. Liu, H. J. Wu, B. B. Jia, Z. L. Guo, X. Y. Fan and J. L. Zheng, Angew. Chem., Int. Ed., 2025, 64, e202504084 CrossRef CAS PubMed.
- J. Li, Q. Wang, R. Guo, Y. Zong, J. Xing, W. Xiao, G. Xu, D. Chen, L. Wang and Z. Wu, Inorg. Chem. Front., 2025, 12, 5063–5072 RSC.
- Y. H. He, X. P. Chen, Y. C. Lei, Y. Q. Liu and L. L. Wang, Nanomaterials, 2023, 13, 2522 CrossRef CAS PubMed.
- N. N. Zhang, S. Y. Sun, C. Q. Cheng, F. F. Zhang, S. W. Yan, Z. L. Zhang, L. Q. Liu, Y. X. Li, L. Shang, R. Zhang, P. F. Yin, J. Yang, C. K. Dong, X. W. Du and H. Liu, ACS Energy Lett., 2025, 10, 5596–5604 CrossRef CAS.
- J. Ni, Z. P. Shi, Y. B. Wang, J. H. Yang, H. X. Wu, P. B. Wang, M. L. Xiao, C. P. Liu and W. Xing, eScience, 2025, 5, 100295 CrossRef.
- C. Tsai, H. Li, S. Park, J. Park, H. S. Han, J. K. Norskov, X. L. Zheng and F. Abild-Pedersen, Nat. Commun., 2017, 8, 15113 CrossRef PubMed.
- Y. X. Zhao, M. T. Tang, S. D. Wu, J. Geng, Z. J. Han, K. Chan, P. Q. Gao and H. Li, J. Catal., 2020, 382, 320–328 CrossRef CAS.
- H. G. Han, J. W. Choi, M. Son and K. C. Kim, eScience, 2024, 4, 100204 CrossRef.
- J. Liu, J. Sun, X. Wang, Y. Wang, M. Li, M. Li, X. Zhang, H. Xia, J. Sun, D. Li and L. Zhang, EcoEnergy, 2025, 3, e70021 CrossRef CAS.
- Q. Shi, P. Wang, M. Niu, Y. Zhang, H. Wang, J. Zhang, Y. Yamauchi, Y.-Z. Long and J. Zheng, EcoEnergy, 2026, 4, e70029 CrossRef CAS.
- H. Meskher, A. R. Woldu, P. K. Chu, F. Lu and L. Hu, EcoEnergy, 2024, 2, 630–651 CrossRef CAS.
- Y. Gu, Y. Zhang, X. Song, Y. Wang, A. Bao, D. Zhang, J. Lv, Z. Zheng, X. Zhang, Z. Liu and X. Qi, Nano Res. Energy, 2025, 4, e9120189 CrossRef.
- M. Song, X. Yang, C. Guo, S. Zhang, J. Ma and H. Gao, EcoEnergy, 2025, 3, 470–481 CrossRef CAS.
- Z. Li, Y. Yao, S. Sun, J. Liang, S. Hong, H. Zhang, C. Yang, X. Zhang, Z. Cai and J. Li, Angew. Chem., Int. Ed., 2024, 63, e202316522 CrossRef CAS PubMed.
- H. Chen, Y.-Q. Wang, R. Ding, Z.-W. Zeng, B.-W. Liu, F.-R. Zeng, Y.-Z. Wang and H.-B. Zhao, Matter, 2024, 7, 3189–3204 CrossRef CAS.
- P. Wang, P. Wang, T. Wu, X. Sun and Y. Zhang, Adv. Sci., 2024, 11, 2407892 CrossRef CAS PubMed.
- J. Hu, X. Tang, Q. Dai, Z. Liu, H. Zhang, A. Zheng, Z. Yuan and X. Li, Nat. Commun., 2021, 12, 3409 CrossRef CAS PubMed.
- H. Tang, J. Ding, Z. Feng, K. Shen, L. Chen and Y. Li, Adv. Funct. Mater., 2025, e28467 Search PubMed.
- X. Wang, H. Hu, J. Song, J. Ma, H. Du, J. J. Wang, M. Wang, W. Chen, Y. Zhou and J. Wang, Adv. Energy Mater., 2025, 15, 2402883 CrossRef CAS.
- H. Mao, X. Liu, T. Cui, J. Tang, Z. Su, J. Chi, Y. Chai, Z. Wu and L. Wang, Angew. Chem., Int. Ed., 2025, 64, e202511867 CrossRef CAS PubMed.
- J. Duan, X. Zhao, Z. Luo, H. Sun, M. Chen, Q. Lu, B. Wang, B. Xiao, T. Zhou, D. Li, H. Cui, T. He and Q. Liu, Appl. Catal., B, 2025, 385, 126271 CrossRef.
- X. Fu, H. Li, Y. Zong, W. Xiao, J. Wang, H. Li, T. Ma, Z. Wu and L. Wang, Angew. Chem., Int. Ed., 2025, 64, e202512710 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |