Open Access Article
Open Access ArticleStepwise B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/h2_char_e002.gif) N bond cleavage by isocyanides: access to the 3-azaborole structural motif
N bond cleavage by isocyanides: access to the 3-azaborole structural motif
Libo Xiang†
a,
Ka Ho Kwan†b,
Yi Jingc,
Junyi Wangd,
Alexander Matlera,
Xuenian Chen c,
Zhenyang Lin
c,
Zhenyang Lin *b and
Qing Ye
*b and
Qing Ye *a
*a
aInstitute for Inorganic Chemistry and Institute for Sustainable Chemistry & Catalysis with Boron, Julius-Maximilians-Universität Würzburg, 97074 Würzburg, Germany. E-mail: qing.ye@uni-wuerzburg.de
bDepartment of Chemistry, The Hong Kong University of Science and Technology, Kowloon, Hong Kong. E-mail: chzlin@ust.hk
cCollege of Chemistry, Zhengzhou University, 450001, Zhengzhou, Henan, P. R. China
dDepartment of Chemistry, Southern University of Science and Technology, 518055, Shenzhen, Guangdong, P. R. China
First published on 7th April 2026
Abstract
The reaction of TMSoCb–B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N–SiMe3 (TMSoCb = 1-trimethylsilyl-ortho-carboranyl) with the sterically demanding (2,4,6-tri-tert-butylphenyl)isocyanide enables the isolation of the corresponding 1
N–SiMe3 (TMSoCb = 1-trimethylsilyl-ortho-carboranyl) with the sterically demanding (2,4,6-tri-tert-butylphenyl)isocyanide enables the isolation of the corresponding 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 iminoborane-isocyanide adduct, a species considered to be the key intermediate in isocyanide-catalyzed dimerization. In sharp contrast, the less hindered isocyanides, namely (2,4,6-trimethylphenyl)isocyanide and (2,6-di-iso-propylphenyl)isocyanide, react with the o-carboranyl iminoborane in a 3
1 iminoborane-isocyanide adduct, a species considered to be the key intermediate in isocyanide-catalyzed dimerization. In sharp contrast, the less hindered isocyanides, namely (2,4,6-trimethylphenyl)isocyanide and (2,6-di-iso-propylphenyl)isocyanide, react with the o-carboranyl iminoborane in a 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 fashion. Two equivalents of isocyanide undergo a [2 + 1 + 1] cycloaddition with the B
1 fashion. Two equivalents of isocyanide undergo a [2 + 1 + 1] cycloaddition with the B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N triple bond to furnish a 1,2-azaborete-3,4-diimine four-membered ring, while the third equivalent coordinates to the boron center of the ring. Subsequent reduction with Mg or Ca induces selective insertion of this third isocyanide into the BN bond, generating an unprecedented 3-azaborolediide species. Single crystal structure characterization and quantum chemical calculations reveal that the negative charge is extensively delocalized onto the o-carborane cage. Theoretical studies further establish a clear mechanistic pathway for the entire transformation.
N triple bond to furnish a 1,2-azaborete-3,4-diimine four-membered ring, while the third equivalent coordinates to the boron center of the ring. Subsequent reduction with Mg or Ca induces selective insertion of this third isocyanide into the BN bond, generating an unprecedented 3-azaborolediide species. Single crystal structure characterization and quantum chemical calculations reveal that the negative charge is extensively delocalized onto the o-carborane cage. Theoretical studies further establish a clear mechanistic pathway for the entire transformation.
Introduction
Iminoboranes (R–B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N–R′) feature a B
N–R′) feature a B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N triple bond that is isoelectronic with a C
N triple bond that is isoelectronic with a C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C triple bond, and can therefore be regarded as “inorganic alkynes”.1 Due to the pronounced polarity of the B
C triple bond, and can therefore be regarded as “inorganic alkynes”.1 Due to the pronounced polarity of the B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N bond, iminoboranes exhibit much higher reactivity than alkynes.2 For example, they tend to undergo (cyclo)oligomerization. In addition, the boron center is electrophilic and readily forms adducts with various Lewis bases.3 In addition, the B
N bond, iminoboranes exhibit much higher reactivity than alkynes.2 For example, they tend to undergo (cyclo)oligomerization. In addition, the boron center is electrophilic and readily forms adducts with various Lewis bases.3 In addition, the B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N triple bond of iminoboranes can react with the C
N triple bond of iminoboranes can react with the C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C triple bond of alkynes under the assistance of rhodium, proceeding via a 1,2-azaborete intermediate to ultimately afford 1,4-azaborines or 1,2-azaborines products, which are BN-isosteres of benzene derivatives.4 In these transformations, iminoboranes act as BN synthons.
C triple bond of alkynes under the assistance of rhodium, proceeding via a 1,2-azaborete intermediate to ultimately afford 1,4-azaborines or 1,2-azaborines products, which are BN-isosteres of benzene derivatives.4 In these transformations, iminoboranes act as BN synthons.
On the other hand, isocyanides, analogous to CO, constitute an important class of C1 synthons. They are stronger σ-donors than CO and can act as Lewis bases to stabilize low-valent boron compounds.5 In fact, isocyanides are sufficiently nucleophilic to attack the boron atom of the B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N unit, thereby triggering follow-up reactions. Paetzold and co-workers carried out seminal work, showing that iminoboranes can react with isocyanides to afford various cyclic adducts.6 In the presence of catalytic amounts of tBuNC, F5C6–B
N unit, thereby triggering follow-up reactions. Paetzold and co-workers carried out seminal work, showing that iminoboranes can react with isocyanides to afford various cyclic adducts.6 In the presence of catalytic amounts of tBuNC, F5C6–B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N–tBu readily dimerizes to form the diazadiboretidine product (I in Fig. 1). Increasing the loading of tBuNC to one equivalent affords a 1,3-diaza-2,4-diboracyclopentane-type product (II). Further reaction of II with a second equivalent of isocyanide has not been reported. However, a 1
N–tBu readily dimerizes to form the diazadiboretidine product (I in Fig. 1). Increasing the loading of tBuNC to one equivalent affords a 1,3-diaza-2,4-diboracyclopentane-type product (II). Further reaction of II with a second equivalent of isocyanide has not been reported. However, a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 reaction between an iminoborane and an isocyanide has been achieved for the B-amino iminoborane Me3Si(tBu)N–B
2 reaction between an iminoborane and an isocyanide has been achieved for the B-amino iminoborane Me3Si(tBu)N–B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N–tBu, which reacts with two equivalents of (2,6-dimethylphenyl)isocyanide to form the 1,2-azaborete-3,4-diimine product (III). In 2022, our laboratory successfully synthesized the first o-carboranyl iminoborane, TMSoCb–B
N–tBu, which reacts with two equivalents of (2,6-dimethylphenyl)isocyanide to form the 1,2-azaborete-3,4-diimine product (III). In 2022, our laboratory successfully synthesized the first o-carboranyl iminoborane, TMSoCb–B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N–R (TMSoCb = 1-trimethylsilyl-ortho-carboranyl, R = SiMe3, tBu), and demonstrated that it also undergoes isocyanide-catalyzed dimerization.7 DFT calculations indicate that the 1
N–R (TMSoCb = 1-trimethylsilyl-ortho-carboranyl, R = SiMe3, tBu), and demonstrated that it also undergoes isocyanide-catalyzed dimerization.7 DFT calculations indicate that the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 adduct formed between an iminoborane and an isocyanide is the key intermediate in this catalytic dimerization process.
1 adduct formed between an iminoborane and an isocyanide is the key intermediate in this catalytic dimerization process.
In this context, curiosity has turned toward: (1) whether the reaction could be halted at the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 adduct stage so that this key intermediate could be isolated; (2) whether the 1
1 adduct stage so that this key intermediate could be isolated; (2) whether the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 adduct is also a key intermediate for the formation of III; and (3) whether III could undergo a further ring-expansion reaction with a third equivalent of isocyanide to afford new BN heterocycles, such as the azaboroles shown in Scheme 1.
1 adduct is also a key intermediate for the formation of III; and (3) whether III could undergo a further ring-expansion reaction with a third equivalent of isocyanide to afford new BN heterocycles, such as the azaboroles shown in Scheme 1.
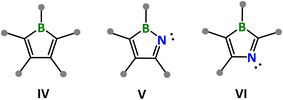
Boroles (IV in Scheme 1) are boracycle derivatives of cyclopentadiene in which the saturated carbon position is replaced by a sp2-hybridized boron with an empty p-orbital. are renowned for their 4π antiaromaticity, the electron deficiency of the boron center, and the resulting unique electronic properties and rich chemical reactivity (e.g. Lewis acid–base reaction, ring expansion, small-molecule activation, π-ligands in the formation of sandwich complexes).8 Very recently, our laboratory successfully synthesized the first 2-azaborole (V in Scheme 1) derivatives featuring a σ2π4 electronic configuration via ring-expansion reactions of benzoborirene C6H4{BN(SiMe3)2} with nitriles.9 In contrast, 3-azaboroles (VI in Scheme 1) have not yet been reported. Herein, we report our progress toward the construction of the 3-azaborole structural motif using an o-carboranyl-substituted iminoborane as a BN synthon and isocyanides as C1 synthons.
Results and discussion
To halt the reaction at the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 adduct stage, a highly sterically demanding isocyanide is expected to play a crucial role. Such steric congestion would kinetically block both (i) cycloaddition between the 1
1 adduct stage, a highly sterically demanding isocyanide is expected to play a crucial role. Such steric congestion would kinetically block both (i) cycloaddition between the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
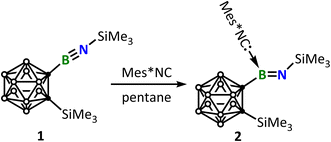
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 adduct and another free iminoborane molecule and (ii) further reaction of the adduct with a second equivalent of isocyanide to form III. Therefore, Mes*NC (Mes* = 2,4,6-tri-tert-butylphenyl) was chosen (Scheme 2). The reaction of iminoborane 1 with an equimolar amount of Mes*NC was monitored by the 11B-NMR spectrum, which revealed a slight upfield shift from δB 13.5 (1)7 to δB 13.4, accompanied by a color change from colorless to pale yellow. Orange single crystals suitable for X-ray diffraction analysis were obtained upon storing the light yellow pentane solution of the reaction mixture at −30 °C for 12 h. The solid-state structure unambiguously revealed a 1
1 adduct and another free iminoborane molecule and (ii) further reaction of the adduct with a second equivalent of isocyanide to form III. Therefore, Mes*NC (Mes* = 2,4,6-tri-tert-butylphenyl) was chosen (Scheme 2). The reaction of iminoborane 1 with an equimolar amount of Mes*NC was monitored by the 11B-NMR spectrum, which revealed a slight upfield shift from δB 13.5 (1)7 to δB 13.4, accompanied by a color change from colorless to pale yellow. Orange single crystals suitable for X-ray diffraction analysis were obtained upon storing the light yellow pentane solution of the reaction mixture at −30 °C for 12 h. The solid-state structure unambiguously revealed a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 adduct between 1 and Mes*NC (Fig. 2). The B1–N1 bond of (1.302(13) Å) is elongated by ca. 10 pm compared to that in 1 (1.220(2) Å), indicating a transformation from a B
1 adduct between 1 and Mes*NC (Fig. 2). The B1–N1 bond of (1.302(13) Å) is elongated by ca. 10 pm compared to that in 1 (1.220(2) Å), indicating a transformation from a B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N triple bond to a B
N triple bond to a B![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bond. In line with this observation, while the IR spectrum of 1 shows a characteristic B
N double bond. In line with this observation, while the IR spectrum of 1 shows a characteristic B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N stretching vibration at 1976 cm−1,7 the BN stretching vibration of 2 appears at 1605 cm−1, as supported by the corresponding 10B isotopic shift (Fig. S25).7 The C2–B1 bond distance of 1.639(13) Å is comparable to the Ccarbene–B bond distance of 1.626(3) Å observed in MeIiPr-1 adduct (MeIiPr = 1,3-di-iso-propyl-4,5-dimethyl-imidazole-2-ylidene).10
N stretching vibration at 1976 cm−1,7 the BN stretching vibration of 2 appears at 1605 cm−1, as supported by the corresponding 10B isotopic shift (Fig. S25).7 The C2–B1 bond distance of 1.639(13) Å is comparable to the Ccarbene–B bond distance of 1.626(3) Å observed in MeIiPr-1 adduct (MeIiPr = 1,3-di-iso-propyl-4,5-dimethyl-imidazole-2-ylidene).10
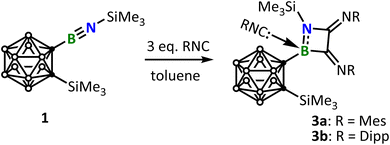
Next, we examined MesNC (Mes = 2,4,6-trimethylphenyl), which features reduced steric hindrance. In stark contrast, the treatment of 1 with one equivalent of MesNC resulted in a mixture consisting of considerable amount of unreacted 1 and a new species 3a, which according to the 1H-NMR spectrum, should be composed of one equivalent of 1 and three equivalents of MesNC. Addition of a second equivalent of MesNC led to an increased portion of 3a. Complete consumption of 1 was only achieved upon the addition of a third equivalent of MesNC, whereupon 1 was quantitatively converted into 3a. This further corroborates that 1 reacted with MesNC in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio (Scheme 3). Yellow single crystals of 3 suitable for X-ray diffraction analysis were obtained upon slow diffusion of pentane into the concentrated toluene solution of 3 at −30 °C. Likewise, the reaction employing DippNC (Dipp = 2,6-di-iso-propylphenyl), an isocyanide whose steric demand lies between that of Mes*NC and MesNC, furnished the same kind of product 3b, though in a lower yield.
3 ratio (Scheme 3). Yellow single crystals of 3 suitable for X-ray diffraction analysis were obtained upon slow diffusion of pentane into the concentrated toluene solution of 3 at −30 °C. Likewise, the reaction employing DippNC (Dipp = 2,6-di-iso-propylphenyl), an isocyanide whose steric demand lies between that of Mes*NC and MesNC, furnished the same kind of product 3b, though in a lower yield.
The single crystal structure is depicted in Fig. 3, suggesting that 3 should arise from the initial reaction of 1 with two equivalents of RNC (R = Mes, 3a; R = Dipp, 3b) to afford the 1,2-azaborete-3,4-diimine (III), followed by adduct formation with a third equivalent of RNC. The BNCC four-member ring is slighted twisted as indicated by the dihedral angles B1–C2–C3–N1 of 3a (−12.916°) and 3b (−12.162°), respectively. The exocyclic C2–N2 (3a, 1.2656(18) Å and 3b, 1.272(2) Å) and C3–N3 (3a, 1.2713(18) Å and 3b, 1.2680(19) Å) bond lengths fall in the range of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N double bonds. The major structural difference between 3a and 3b is that 3a exhibits pronounced Mes–Mes π–π stacking interactions with a centroid-to-centroid distance of 3.514 Å, whereas in 3b, the two Dipp groups adopt an orientation away from each other due to significant steric repulsion between their iso-propyl substituents (Fig. 3).
N double bonds. The major structural difference between 3a and 3b is that 3a exhibits pronounced Mes–Mes π–π stacking interactions with a centroid-to-centroid distance of 3.514 Å, whereas in 3b, the two Dipp groups adopt an orientation away from each other due to significant steric repulsion between their iso-propyl substituents (Fig. 3).
The calculated mechanistic pathway for the formation of compound 3′, a model compound for 3, is presented in Fig. 4. The process begins with the coordination of an isocyanide molecule to the compound 1 at the iminoborane boron center, forming the adduct Int1. Starting from Int1, an alternative route involving C–C coupling with a second PhNC molecule via nucleophilic attack on the coordinated carbon was examined, but found to be energetically unfavourable (see the blue energy profile in Fig. 4). Instead, Int1 undergoes structural rearrangement to form Int2, a three-membered ring species generated through N(iminoborane)–C(coordinated isocyanide) bond formation. Subsequent coordination of another PhNC molecule to the iminoborane boron center in Int2, accompanied by BN π-bond cleavage, yields Int3. From Int3, a 1,2-carbon shift produces the four-membered ring Int4, and coordination of a third PhNC molecule ultimately affords compound 3′. The overall barrier for this transformation is 20.5 kcal mol−1 in Gibbs free energy, with a reaction free energy of −12.8 kcal mol−1. The rate-determining transition state corresponds to the coordination of the second PhNC molecule.
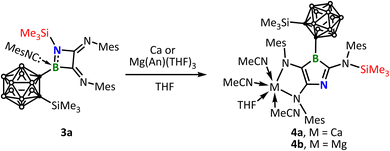
α-Diimines are known to undergo two-electron reduction to form α-diamides.11 In our earlier work, this approach enabled the synthesis of an antiaromatic benzo-fused borolediide.12 Thus, the diimine function in 3 immediately caught our attention. To this end, a THF solution of 3a was stirred with one equivalent of Mg(An)THF3 (An = anthracene) or with calcium metal at room temperature, resulting in a color change from yellow to deep purple. The reaction with Mg(An)THF3 proceeds much faster due to its homogeneous reaction nature, being completed within hours, whereas the reaction with calcium requires several days (Scheme 4). The 11B-NMR spectra displayed the formation of new boron-containing species 4a (δB 29.8) and 4b (δB 26.9), respectively. After removal of the solvent and thorough washing with hexane, the residue was extracted with acetonitrile and crystallized at −30 °C, yielding deep purple single crystals suitable for X-ray diffraction analysis (Fig. 5). The solid-state structure reveals the presence of coordinated acetonitrile ligand, which originates from the recrystallization solvent. The atom connectivity reveals that 3a has indeed undergone a two-electron reduction. More interestingly, the reduction triggers a selective insertion of the coordinated isocyanide into the BN bond, accompanied by migration of the silyl group (marked in red in Scheme 4) from the ring nitrogen atom to the nitrogen of the inserted isocyanide. Accordingly, at first glance, the crystal structure overall features an unprecedented 3-azaborole core (VI in Scheme 1). Further analysis of bond lengths and angles, such as the significantly elongated cage C–C distances (4a, 2.436 Å; 4b, 2.474 Å), indicates pronounced negative hyperconjugation between the o-carborane moiety and the 3-azaborolediide unit in 4a/4b. This is primarily manifested in the perpendicular orientation of the 3-azaborolediide fragment with respect to the C–C bond of the o-carborane cage (Fig. 6), which facilitates optimal overlap between the π orbitals of the 3-azaborole ring and the σ* antibonding orbital of the carborane C–C bond.13 In addition to the direct population of the antibonding orbital by chemical reduction,14d–f elongation of the cage C–C bonds arising from negative hyperconjugation has in fact also been observed in 1,2-diamino-o-carboranes,14a,b 1-imino-o-carboranes14c and o-carboranyl-substituted three-coordinate borane radical anion.13a In addition, the B1–Ccarborane bond is notably shortened (3a, 1.657(2) Å; 4a, 1.485(4) Å; 4b, 1.485(2) Å), further corroborating the presence of the negative hyperconjugation with the o-carborane moiety acting as an electron acceptor.
The calculated mechanistic pathway for the formation of compound 4a′ is presented in Fig. 7. The process begins with the reduction of 3′, resulting in the formation of Int5 which features N,N-coordination of the calcium ion. This formal two-electron reduction cleaves the two C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N π bonds in 3′ and generates a new C
N π bonds in 3′ and generates a new C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C π bond, accompanied by metal coordination. The reaction is associated with a free energy change of 68 kcal mol−1, indicating substantial gain in thermodynamic stability upon reduction. Starting from Int5, a 1,2-nitrogen shift expands the four-membered ring to yield Int6, a five-membered-ring intermediate with an envelope conformation. Subsequent planarization of the ring in Int6 leads to Int7 which features extended π-conjugation across the five-membered framework. This transformation provides significant stabilization, with a computed free energy gain of 36.6 kcal mol−1 (Int6 → Int7). Finally, a 1,3-silyl migration from Int7 yields 4a′, which can be described by two resonance structures, 4a′_closo and 4a′_nido, accounting for the observed C–C bond elongation in the product 4a. The participation of the resonance structure 4a′_nido is supported by the HOMO plot (Fig. S30), which clearly shows the C(cage)–B(exo) π-bonding character. The overall barrier for the transformation from Int5 to compound 4a′ is 12.2 kcal mol−1 in Gibbs free energy, with a reaction free energy of −30.1 kcal mol−1.
C π bond, accompanied by metal coordination. The reaction is associated with a free energy change of 68 kcal mol−1, indicating substantial gain in thermodynamic stability upon reduction. Starting from Int5, a 1,2-nitrogen shift expands the four-membered ring to yield Int6, a five-membered-ring intermediate with an envelope conformation. Subsequent planarization of the ring in Int6 leads to Int7 which features extended π-conjugation across the five-membered framework. This transformation provides significant stabilization, with a computed free energy gain of 36.6 kcal mol−1 (Int6 → Int7). Finally, a 1,3-silyl migration from Int7 yields 4a′, which can be described by two resonance structures, 4a′_closo and 4a′_nido, accounting for the observed C–C bond elongation in the product 4a. The participation of the resonance structure 4a′_nido is supported by the HOMO plot (Fig. S30), which clearly shows the C(cage)–B(exo) π-bonding character. The overall barrier for the transformation from Int5 to compound 4a′ is 12.2 kcal mol−1 in Gibbs free energy, with a reaction free energy of −30.1 kcal mol−1.
We have also examined the alternative pathway involving insertion of the third equivalent of isocyanide into the B–C bond of Int5 (Fig. 7). Our calculations show that this pathway has a higher activation barrier by 11.7 kcal mol−1 compared to insertion into the B–N bond. This insertion corresponds to a 1,2-carbon migration. The results indicate that the more covalent nature of the B–C bond relative to the B–N bond makes the 1,2-carbon migration significantly more energetically demanding and therefore kinetically less favorable.
Additionally, we performed a Nucleus Independent Chemical Shift (NICS) analysis on 4a′ to evaluate the aromatic character of the edge-fused five-membered ring system. The NICS values at the geometric centers of the two rings, as well as at positions 1.0 Å above and below the ring planes, fall within a range centered at about −1.5 ppm, indicating that the fused-ring structure is non-aromatic.
Conclusion
In conclusion, we have identified a stepwise reaction pattern in which the B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N triple bond of an o-carboranyl iminoborane engages sequentially with three equivalents of isocyanide to generate novel BN heterocycles. The first two equivalents undergo a [2 + 1 + 1] cycloaddition across the B
N triple bond of an o-carboranyl iminoborane engages sequentially with three equivalents of isocyanide to generate novel BN heterocycles. The first two equivalents undergo a [2 + 1 + 1] cycloaddition across the B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N unit to afford a 1,2-azaborete-3,4-diimine four-membered ring. Under reducing conditions, the third equivalent inserts selectively into the BN bond, expanding the ring to a 3-azaborolediide species. Structural and computational analyses show that the strong inductive electron-withdrawing o-carboranyl substituent15 participates in substantial electronic delocalization. Moreover, a sterically demanding isocyanide, i.e. Mes*NC, allowed the isolation of the corresponding 1
N unit to afford a 1,2-azaborete-3,4-diimine four-membered ring. Under reducing conditions, the third equivalent inserts selectively into the BN bond, expanding the ring to a 3-azaborolediide species. Structural and computational analyses show that the strong inductive electron-withdrawing o-carboranyl substituent15 participates in substantial electronic delocalization. Moreover, a sterically demanding isocyanide, i.e. Mes*NC, allowed the isolation of the corresponding 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 isocyanide-iminoborane adduct. This kind of 1
1 isocyanide-iminoborane adduct. This kind of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 adduct species, previously suggested as the key intermediate in isocyanide-catalyzed iminoborane dimerization,7 is further corroborated by quantum chemical calculations to be the intermediate leading to the formation of the 1,2-azaborete-3,4-diimine ring. The detailed computational study provides a clear mechanistic picture, illustrating how each elementary step transforms the B
1 adduct species, previously suggested as the key intermediate in isocyanide-catalyzed iminoborane dimerization,7 is further corroborated by quantum chemical calculations to be the intermediate leading to the formation of the 1,2-azaborete-3,4-diimine ring. The detailed computational study provides a clear mechanistic picture, illustrating how each elementary step transforms the B![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) N triple bond and ultimately generates the 3-azaborolediide-type structure.
N triple bond and ultimately generates the 3-azaborolediide-type structure.
Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2504504–2504508 contain the supplementary crystallographic data for this paper.16a–eThe data supporting this article have been included as part of the supplementary information (SI). Supplementary information: experimental details, synthesis, NMR and IR spectra, cyclic voltammetry, UV-vis spectra, computational details, crystallographic information, and additional characterization data. See DOI: https://doi.org/10.1039/d6sc01836j.
Acknowledgements
We thank Julius-Maximilians-Universität Würzburg (JMU) and the Research Grants Council of Hong Kong (HKUST 16301823) for financial support. We thank Christoph Mahler and Liselotte Michels for their help with testing high-resolution mass spectrometry (HRMS) and elemental analysis (EA), respectively.Notes and references
- L. Xiang, A. G. Albacar, J. Wang, A. Matler, T. Preitschopf, M. Finze and Q. Ye, Organometallics, 2025, 44, 1756–1759 CrossRef CAS.
- (a) Y. Fan, J. Cui and L. Kong, Eur. J. Org Chem., 2022, e202201086 CrossRef; (b) P. Paetzold, Adv. Inorg. Chem., 1987, 31, 123–170 CrossRef CAS.
- (a) H. Zhang, J. Wang, W. Yang, L. Xiang, W. Sun, W. Ming, Y. Li, Z. Lin and Q. Ye, J. Am. Chem. Soc., 2020, 142, 17243–17249 CrossRef CAS PubMed; (b) L. Xie, J. Zhang, H. Hu and C. Cui, Organometallics, 2013, 32, 6875–6878 CrossRef CAS; (c) H. Braunschweig, W. C. Ewing, K. Geetharani and M. Schäfer, Angew. Chem., Int. Ed., 2015, 54, 1662–1665 CrossRef CAS PubMed; (d) F. Dahcheh, D. W. Stephan and G. Bertrand, Chem.–Eur. J., 2015, 21, 199–204 CrossRef CAS PubMed; (e) F. Dahcheh, D. Martin, D. W. Stephan and G. Bertrand, Angew. Chem., Int. Ed., 2014, 53, 13159–13163 CrossRef CAS PubMed; (f) P. Cui, R. Guo, L. Kong and C. Cui, Inorg. Chem., 2020, 59, 5261–5265 CrossRef CAS PubMed; (g) R. Guo, X. Zhang, T. Li, Q. Li, D. A. Ruiz, L. L. Liu, C. H. Tung and L. Kong, Chem. Sci., 2022, 13, 2303–2309 RSC; (h) A. Koner, T. Sergeieva, B. Morgenstern and D. M. Andrada, Inorg. Chem., 2021, 60, 14202–14211 CrossRef CAS PubMed; (i) A. K. Swarnakar, C. Hering-Junghans, K. Nagata, M. J. Ferguson, R. McDonald, N. Tokitoh and E. Rivard, Angew. Chem., Int. Ed., 2015, 54, 10666–10669 CrossRef CAS PubMed; (j) A. K. Swarnakar, C. Hering-Junghans, M. J. Ferguson, R. McDonald and E. Rivard, Chem. Sci., 2017, 8, 2337–2343 RSC.
- (a) H. Braunschweig, A. Damme, J. O. C. Jimenez-Halla, B. Pfaffinger, K. Radacki and J. Wolf, Angew. Chem., Int. Ed., 2012, 51, 10034–10037 CrossRef CAS PubMed; (b) H. Braunschweig, K. Geetharani, J. O. C. Jimenez-Halla and M. Schäfer, Angew. Chem., Int. Ed., 2014, 53, 3500–3504 CrossRef CAS PubMed; (c) M. Schäfer, N. A. Beattie, K. Geetharani, J. Schäfer, W. C. Ewing, M. Krahfuß, C. Hörl, R. D. Dewhurst, S. A. Macgregor, C. Lambert and H. Braunschweig, J. Am. Chem. Soc., 2016, 138, 8212–8220 CrossRef PubMed; (d) M. Schäfer, J. Schäfer, R. D. Dewhurst, W. C. Ewing, M. Krahfuß, M. W. Kuntze-Fechner, M. Wehner, C. Lambert and H. Braunschweig, Chem.–Eur. J., 2016, 22, 8603–8609 CrossRef PubMed.
- H. Braunschweig, R. D. Dewhurst, F. Hupp, M. Nutz, K. Radacki, C. W. Tate, A. Vargas and Q. Ye, Nature, 2015, 522, 327 CrossRef CAS PubMed.
- C. Klöfkorn, M. Schmidt, T. Spaniol, T. Wagner, O. Costisor and P. Paetzold, Chem. Ber., 1995, 128, 1037–1043 CrossRef.
- J. Wang, P. Jia, W. Sun, Y. Wei, Z. Lin and Q. Ye, Inorg. Chem., 2022, 61, 8879–8886 CrossRef CAS PubMed.
- (a) H. Braunschweig, I. Krummenacher and J. Wahler, Adv. Organomet. Chem., 2013, 61, 1–53 CrossRef CAS; (b) J. He, F. Rauch, M. Finze and T. B. Marder, Chem. Sci., 2021, 12, 128–147 RSC; (c) A. Steffen, R. M. Ward, W. D. Jones and T. B. Marder, Coord. Chem. Rev., 2010, 254, 1950–1976 CrossRef CAS; (d) X. Su, T. A. Bartholome, J. R. Tidwell, A. Pujol, S. Yruegas, J. J. Martinez and C. D. Martin, Chem. Rev., 2021, 121, 4147–4192 CrossRef CAS PubMed.
- (a) L. Tan, J. Chen, X. Liu, A. Matler, N. Schopper, M. Finze, Z. Lin and Q. Ye, J. Am. Chem. Soc., 2024, 146, 31681–31690 CrossRef CAS PubMed; (b) S. Parua and Q. Ye, Coord. Chem. Rev., 2026, 558, 217742 CrossRef CAS; (c) J. Wang and Q. Ye, Chem.–Eur. J., 2023, 30, e202303695 CrossRef PubMed.
- L. Xiang, A. Matler, L. Tan and Q. Ye, Dalton Trans., 2024, 53, 11655–11658 RSC.
- (a) M. Ma, L. Shen, H. Wang, Y. Zhao, B. Wu and X.-J. Yang, Organometallics, 2020, 39, 1440–1447 CrossRef CAS; (b) R. Zhang, Y. Wang, Y. Zhao, C. Redshaw, I. L. Fedushkin, B. Wu and X.-J. Yang, Dalton Trans., 2021, 50, 13634–13650 RSC.
- X. Liu, M. Heinz, J. Wang, L. Tan, M. C. Holthhausen and Q. Ye, Angew. Chem., Int. Ed., 2023, 62, e202312608 CrossRef CAS PubMed.
- (a) J. Krebs, M. Haehnel, I. Krummenacher, A. Friedrich, H. Braunschweig, M. Finze, L. Ji and T. B. Marder, Chem.–Eur. J., 2021, 27, 8159–8167 CrossRef CAS PubMed; (b) L. Xiang, J. Wang, I. Krummenacher, K. Radacki, H. Braunschweig, Z. Lin and Q. Ye, Chem.–Eur. J., 2023, 29, e202301270 CrossRef CAS PubMed.
- (a) J. Li, R. Pang, Z. Li, G. Lai, X.-Q. Xiao and T. Müller, Angew. Chem., Int. Ed., 2019, 58, 1397–1401 CrossRef CAS PubMed; (b) Y. Wu, J. Zhang and Z. Xie, Chin. Chem. Lett., 2019, 30, 1530–1532 CrossRef CAS; (c) L. Xiang, J. Wang, L. Tan and Q. Ye, Inorg. Chem., 2026, 65, 1045–1050 CrossRef CAS PubMed; (d) S. Yao, T. Szilvási, Y. Xiong, C. Lorent, A. Ruzicka and M. Driess, Angew. Chem., Int. Ed., 2020, 59, 22043–22047 CrossRef CAS PubMed; (e) S. Yao, A. Kostenko, Y. Xiong, A. Ruzicka and M. Driess, J. Am. Chem. Soc., 2020, 142, 12608–12612 CrossRef CAS PubMed; (f) H. Wang, J. Zhang, H. K. Lee and Z. Xie, J. Am. Chem. Soc., 2018, 140, 3888–3891 CrossRef CAS PubMed.
- (a) R. G. Adler and M. F. Hawthorne, J. Am. Chem. Soc., 1965, 87, 4746–4750 CrossRef PubMed; (b) R. G. Adler and M. F. Hawthorne, J. Am. Chem. Soc., 1970, 92, 6174–6182 CrossRef; (c) C. Zhang, X. Liu, J. Wang and Q. Ye, Angew. Chem., Int. Ed., 2022, 61, e202205506 CrossRef CAS PubMed; (d) C. Zhang, J. Wang, Z. Lin and Q. Ye, Inorg. Chem., 2022, 61, 18275–18284 CrossRef CAS PubMed; (e) C. Zhang, J. Wang, W. Su, Z. Lin and Q. Ye, J. Am. Chem. Soc., 2021, 143, 8552–8558 CrossRef CAS PubMed; (f) M. O. Akram, J. R. Tidwell, J. L. Dutton and C. D. Martin, Angew. Chem., Int. Ed., 2023, 62, e202307040 CrossRef CAS PubMed; (g) L. Xiang, J. Wang, N. Knoblauch, A. Matler and Q. Ye, Angew. Chem., Int. Ed., 2025, e202501955 CAS; (h) L. Xiang, J. Wang, A. Matler and Q. Ye, Chem. Sci., 2024, 15, 17944–17949 RSC; (i) M. O. Akram, J. R. Tidwell, J. L. Dutton and C. D. Martin, Angew. Chem., Int. Ed., 2022, 134, e202212073 CrossRef; (j) L. Xiang, J. Wang, A. Matler, I. Krummenacher, H. Braunschweig and Q. Ye, Inorg. Chem., 2025, 64, 25428–25435 CrossRef CAS PubMed.
- (a) CCDC 2504504: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q24gf; (b) CCDC 2504505: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q24hg; (c) CCDC 2504506: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q24jh; (d) CCDC 2504507: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q24kj; (e) CCDC 2504508: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q24lk.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |