Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Synergistic decatungstate and Brønsted acid catalysis enables direct C–H indolation of alkanes and aldehydes
Gangqi Peng†
a,
Yangling Deng†ab,
Mengxuan Zhanga,
Yuxuan Hea,
Hao Chenga,
Xinbin Wanga,
Yuanyuan An *a,
Guanyinsheng Qiu
*a,
Guanyinsheng Qiu *b and
Danqing Zheng
*b and
Danqing Zheng *a
*a
aState Key Laboratory of Materials-Oriented Chemical Engineering, College of Chemical Engineering, Nanjing Tech University, Nanjing, 211816, China. E-mail: zhengd@njtech.edu.cn
bCollege of Biological and Chemical Engineering, Jiaxing University, Jiaxing, 314001, China. E-mail: qiuguanyinsheng@mail.zjxu.edu.cn
First published on 27th April 2026
Abstract
Indole skeletons are ubiquitous in natural products, pharmaceuticals, and agricultural agents, making direct C–H indolation strategies highly significant. Direct radical alkylation of indoles, however, is often hampered by the inherent polarity mismatch between nucleophilic alkyl radicals and the electron-rich indole ring. Herein, we report that synergistic decatungstate and Brønsted acid catalysis enables the direct radical C–H indolation of alkanes and aldehydes with 2-indolylmethanols. The acid-mediated ionization of 2-indolylmethanols generates delocalized carbocations, which efficiently capture alkyl or acyl radicals derived from the decatungstate-catalyzed hydrogen atom transfer (HAT) process. This cascade delivers C3-functionalized indoles with high regioselectivity under mild conditions. Mechanistic studies suggest that the Brønsted acid plays a dual role by facilitating the formation of key carbocation intermediates and accelerating the HAT step. The late-stage C–H indolation of complex natural products and pharmaceutical agents further demonstrates the synthetic versatility of this protocol.
Introduction
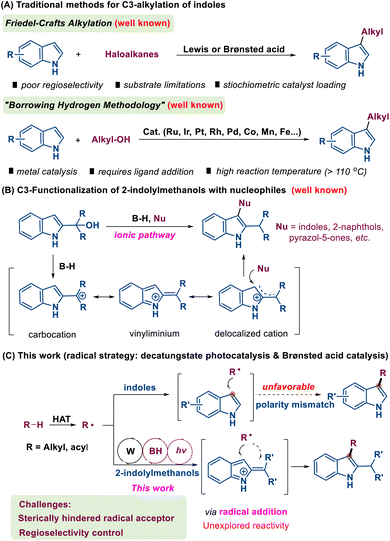
The indole scaffold represents a highly adaptable and valuable heterocyclic system that is extensively distributed across various natural products, medicinal compounds, and agricultural chemicals (Fig. 1).1 This prevalence stems from its remarkable structural versatility and diverse biological functionalities. In organic synthesis, significant research efforts have been consistently directed toward the exploration and advancement of innovative strategies for both the assembly and modification of indole-based structures.2Among the various established methodologies, C3-alkylation of indoles has emerged as a particularly promising approach, attracting significant research attention due to its effectiveness in generating C3-substituted indole derivatives (Scheme 1A).3 Traditional C3-alkylation methods mainly rely on Friedel–Crafts reactions using Lewis or Brønsted acids. However, these approaches face several limitations, including poor compatibility with green chemistry principles, the toxicity of alkyl halides and issues such as over-alkylation.4 In addition, the borrowing hydrogen strategy provides an alternative approach for the C3-alkylation of indoles. This methodology is advantageous due to its utilization of non-toxic and readily accessible alcohols as starting materials, with water being the only byproduct, thus aligning with green chemistry principles. Nevertheless, this method presents certain limitations, such as the use of metal catalysts (e.g., Ru, Ir, Rh, Pd, Co, Mn, Fe, and Cu complexes) and the requirement for additional ligands. Moreover, the process typically requires a higher reaction temperature, which may limit its practical applications.5,6
Over the last decade, Shi and coworkers have disclosed a remarkable polarity reversal phenomenon that the C3-position of 2-indolemethanol exhibits electrophilic characteristics rather than its conventional nucleophilic behavior when exposed to Brønsted or Lewis acids.7 This intriguing transformation can be rationalized by the in situ formation of a delocalized cation intermediate derived from 2-indolemethanol (Scheme 1B). On this foundation, numerous reactions involving 2-indolylmethanol have subsequently been reported, encompassing diverse nucleophilic substitutions8 and various intermolecular cycloadditions, including [3 + 2],9 [3 + 3],10 and [4 + 3]11 variants. These groundbreaking findings have not only challenged the traditional paradigm of C3-nucleophilicity but have also significantly expanded the scope of substrate compatibility. Despite these advancements, it is noteworthy that both conventional and newly established methods predominantly proceed through ionic mechanisms. Crucially, such ionic pathways are inherently biased towards ‘soft’ nucleophiles, rendering them ineffective for the introduction of ‘hard’ alkyl groups derived from unactivated alkanes. Consequently, the development of a complementary radical-based strategy to overcome this limitation and achieve direct C3-alkylation remains a significant, yet largely unexplored frontier.
However, due to the intrinsic chemical properties of the C3 position of indoles, it is difficult for them to undergo attack by alkyl radicals due to polarity mismatch, making direct radical C3-alkylation of indoles unfavorable. To address this limitation, herein we report a synergistic catalytic strategy that combines hydrogen atom transfer (HAT) photocatalysis with Brønsted acid catalysis, achieving C3-alkylation of indoles through a radical process (Scheme 1C). This methodology provides a versatile platform for the synthesis of diverse C3-substituted indole derivatives, offering significant improvements in both efficiency and substrate scope.
Results and discussion
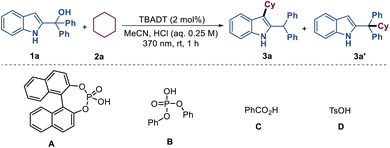
In recent years, decatungstate-catalyzed hydrogen atom transfer (HAT) of alkanes has represented a powerful strategy for direct functionalization of inert C(sp3)-H bonds.12 Drawing on this established reactivity, we employed tetrabutylammonium decatungstate (TBADT) as the photocatalyst to generate alkyl radicals from alkanes for this investigation. To optimize the reaction conditions, 2-indolemethanol 1a (0.1 mmol) and cyclohexane 2a (0.1 mmol) were used as model substrates (Table 1). When the reaction was carried out in the presence of 2 mol% TBADT in a mixture of MeCN/0.25 M HCl (0.85![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.15, 0.1 M) under nitrogen and irradiated with a Kessil 40 W 370 nm lamp for 1 hour, the desired product 3a was isolated in 81% yield (entry 1). Replacement of aqueous HCl with organic Brønsted acids such as phosphonic acid A–B, benzoic acid C and p-toluenesulfonic acid (TsOH) D resulted in lower yields of 3a and the generation of regioisomer 3a′ (entries 2–5). Varying the concentration of HCl led to a slight decrease in yield (Table 1, entries 6–7). When an aqueous solution of TsOH, H2SO4 and H3PO4 was used instead of aqueous HCl, the target product 3a was obtained in 61–69% yield (Table 1, entries 8–10). Comparing the results of entries 5 and 8, it can be concluded that H2O played a significant role in this reaction system. The use of other solvents such as DCE, EA, and DMSO all led to negative outcomes (Table 1, entries 11–13). Replacement of TBADT with other HAT photocatalysts such as Eosin Y, FeCl3, and benzophenone resulted in trace yields of 3a (entries 14–16). Reducing the amount of TBADT from 2 mol% to 1 mol% resulted in a slight decrease of yield (Table 1, entry 17). Altering the amount of 2a to 0.1 mmol and 1a to 0.15 mmol afforded product 3a in 50% yield (Table 1, entry 18). Additionally, the results from controlled experiments demonstrated the necessity of TBADT, HCl and light for this transformation (Table 1, entries 19–21).
0.15, 0.1 M) under nitrogen and irradiated with a Kessil 40 W 370 nm lamp for 1 hour, the desired product 3a was isolated in 81% yield (entry 1). Replacement of aqueous HCl with organic Brønsted acids such as phosphonic acid A–B, benzoic acid C and p-toluenesulfonic acid (TsOH) D resulted in lower yields of 3a and the generation of regioisomer 3a′ (entries 2–5). Varying the concentration of HCl led to a slight decrease in yield (Table 1, entries 6–7). When an aqueous solution of TsOH, H2SO4 and H3PO4 was used instead of aqueous HCl, the target product 3a was obtained in 61–69% yield (Table 1, entries 8–10). Comparing the results of entries 5 and 8, it can be concluded that H2O played a significant role in this reaction system. The use of other solvents such as DCE, EA, and DMSO all led to negative outcomes (Table 1, entries 11–13). Replacement of TBADT with other HAT photocatalysts such as Eosin Y, FeCl3, and benzophenone resulted in trace yields of 3a (entries 14–16). Reducing the amount of TBADT from 2 mol% to 1 mol% resulted in a slight decrease of yield (Table 1, entry 17). Altering the amount of 2a to 0.1 mmol and 1a to 0.15 mmol afforded product 3a in 50% yield (Table 1, entry 18). Additionally, the results from controlled experiments demonstrated the necessity of TBADT, HCl and light for this transformation (Table 1, entries 19–21).
| Entry | Variations from standard conditions | 3a [%]b | 3a′ [%]b |
|---|---|---|---|
| a Reaction conditions: 1a (0.1 mmol), 2a (1.0 mmol), TBADT (2 mol%), 0.25 M HCl (0.15 mL), MeCN (0.85 mL), Kessil 370 nm, 1 h, rt. M = mol L−1. n.d. = not detected. n.r. = no reaction.b Yields were determined by GC.c Isolated yields based on 1a. | |||
| 1 | None | 87(81)c | n.d. |
| 2 | A (10 mol%) instead of HCl | 44 | 12 |
| 3 | B (10 mol%) instead of HCl | 19 | n.d. |
| 4 | C (10 mol%) instead of HCl | n.d. | n.d. |
| 5 | D (10 mol%) instead of HCl | 13 | n.d. |
| 6 | HCl (0.1 M) | 72 | 10 |
| 7 | HCl (0.5 M) | 81 | n.d. |
| 8 | 0.25 M TsOH instead of HCl | 63 | 11 |
| 9 | 0.125 M H2SO4 instead of HCl | 69 | 10 |
| 10 | 0.08 M H3PO4 instead of HCl | 61 | 6 |
| 11 | DCE as solvent | Trace | n.d. |
| 12 | EA as solvent | n.d. | n.d. |
| 13 | DMSO as solvent | n.d. | n.d. |
| 14 | Eosin Y instead of TBADT | Trace | n.d. |
| 15 | FeCl3 instead of TBADT | Trace | n.d. |
| 16c | Benzophenone instead of TBADT | n.d. | n.d. |
| 17 | TBADT (1 mol%) | 76 | n.d. |
| 18 | 1.0 eq. of 2a, 1.5 eq. of 1a | 50 | n.d. |
| 19c | No TBADT | n.d. | n.d. |
| 20 | No HCl | n.r. | n.r. |
| 21 | In the dark | n.r. | n.r. |
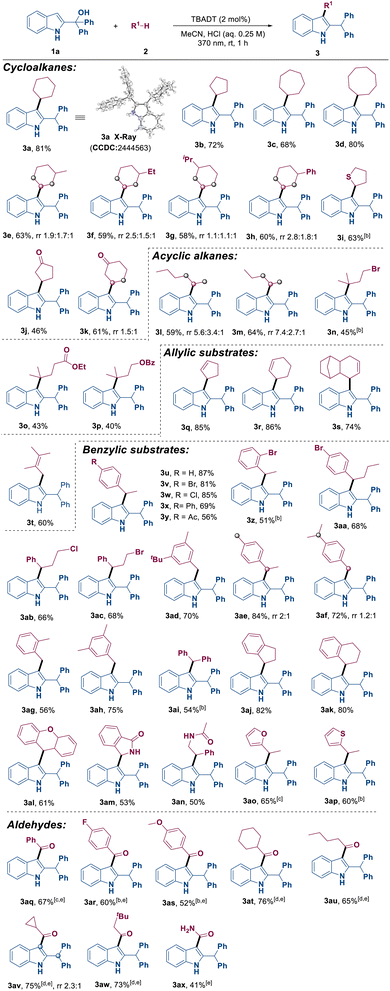
With the optimized conditions identified, we then explored the substrate scope and site selectivity of this transformation. As illustrated in Table 2, a broad range of hydrocarbon compounds R1–H 2 including cycloalkanes, chain alkanes, olefins, benzylic substrates and aldehydes were demonstrated to be suitable as radical precursors in this reaction. It was observed that cycloalkanes with varying ring sizes and substituents all yielded the corresponding products 3a–3h in 58–81% yields. Different alkyl- or aryl-substituted cyclohexanes were also compatible, delivering 3e–3h in good yields. Thioethers and cyclic ketones were also viable substrates, providing the desired products 3i–3k with yields of 46–63%. Linear and branched alkanes could be converted into products 3l–3p in moderate yields. Notably, the reaction with linear alkanes produced a mixture of positional isomers, indicating that indolylation of the secondary C(sp3)−H position was more favorable than other positions (3l and 3m). Additionally, exclusive site-selectivity on the tertiary C–H bond was observed in the reaction with branched alkanes (3n–3p). The divergent regioselectivity observed between cyclic and acyclic alkanes is characteristic of decatungstate-mediated HAT. For acyclic precursors (3n–3p), the hydrogen abstraction is driven by bond dissociation energy (BDE), favoring the weaker tertiary C–H bonds. However, the excited state [W10O32]4− is highly sensitive to steric hindrance. In rigid, substituted cyclic alkanes (3e–3h), the tertiary C–H bonds are sterically shielded, leading the bulky photocatalyst to preferentially abstract hydrogen from the more accessible secondary C–H positions. Moreover, olefins furnished indolated products 3q–3t at the allylic position with yields ranging from 60% to 86%. The reactivity of a variety of substrates containing benzylic C(sp3)−H bonds was investigated. It was found that electron-neutral or electron-poor alkyl benzenes, diphenylmethane, indane, tetralin, 9H-xanthene, isoindolin-1-one and even heterocyclic substrates all reacted smoothly with 2-indolemethanol 1a to yield the corresponding products (3u–3ap) in moderate to good yields. It should be noted that the reaction resulted in two regioisomers when there were two benzylic reactive sites in the substrate (3ae–3af).
Besides that, to our delight, both aromatic and aliphatic aldehydes (3aq–3ax) were found to be compatible with this transformation. This compatibility was achieved by extending the reaction time and varying the type and concentration of Brønsted acid.
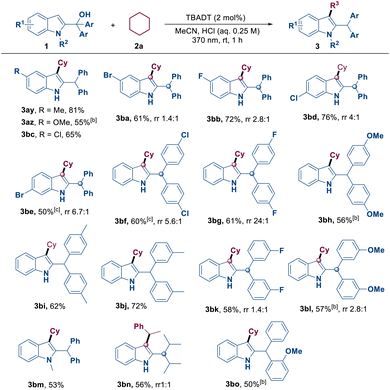
Subsequently, we assessed the reactivity of 2-indolemethanol derivatives 1 with cyclohexane 2a, and a series of indolated products 3ay–3bo were obtained in moderate to excellent yields (Table 3). 2-Indolemethanols with different electronic properties, either due to different Ar groups or due to electron-donating or electron-withdrawing substituents on the indole ring, were all compatible with this reaction. The reaction produced the corresponding products 3ay–3bl with yields ranging from moderate to good. One should note that when the substrates contain F, Cl, Br, and OMe groups, this reaction may often lead to the formation of alkylated by-products at the aryl benzylic position (3ba–3bg and 3bk–3bl). A 2-indolemethanol derivative with an N–CH3 substituent was found to be a suitable substrate, yielding the expected product 3bm in 53% yield. Notably, when the reaction was performed using ethylbenzene and a 2-indolylmethanol derivative with isopropyl groups replacing the aryl groups, product 3bn was obtained with a yield of 56% and a rr (regioisomeric ratio) value of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Most importantly, 2-indolylmethanol bearing two distinct aryl groups efficiently afforded the desired product 3bo in 50% yield.
1. Most importantly, 2-indolylmethanol bearing two distinct aryl groups efficiently afforded the desired product 3bo in 50% yield.
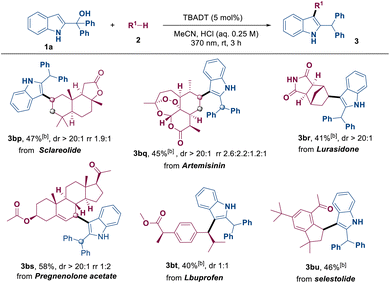
Furthermore, the reaction proceeded smoothly with natural products and drug molecules, including sclareolide, artemisinin and lurasidone, yielding the corresponding products 3bp–3bu in moderate yields (Table 4). For late-stage functionalizations, the C–H substrate was reduced to 3.0 equivalents; thus, a higher TBADT loading (5 mol%) was employed to offset the slower HAT kinetics and maintain efficient turnover. The successful indolylation of these substrates demonstrates the high robustness and reliability of the reaction and its broad applicability to a diverse array of chemical structures, particularly those found in biologically active compounds. The observed regioselectivity is highly consistent with established empirical trends for decatungstate-mediated HAT processes, where hydrogen abstraction preferentially occurs at the most sterically accessible and thermodynamically favored C–H bonds within the complex molecular frameworks.12
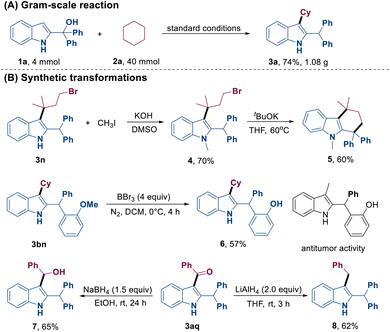
A gram-scale reaction involving 4.0 mmol of 2-indolemethanol 1a with cyclohexane 2a was successfully conducted, which resulted in the formation of product 3a in 74% yield. This result demonstrated the feasibility of this protocol (Scheme 2A). Subsequently, straightforward transformations of the obtained products were studied (Scheme 2B). For instance, compound 5 could be synthesized from 3n through successive N-methylation and intramolecular cyclization. The demethylation of 3bn could lead to the formation of 6, which is presumably a bioactive compound. Moreover, treatment of 3aq with NaBH4 or LiAlH4 as the reducing agent afforded compounds 7 and 8, respectively.
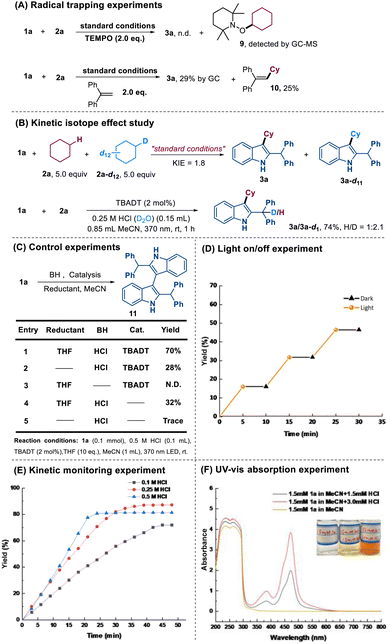
To gain insights into the reaction mechanism, a series of mechanistic experiments were carried out (Scheme 3). Adding 2 equivalents of 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO) as a radical scavenger completely inhibited the reaction. While product 3a was undetectable, GC-MS analysis revealed the formation of TEMPO-Cy 9. Using 1,1-diphenylethylene as a radical trapper afforded product 3a in only 29% yield, along with the trapped cyclohexyl radical adduct 10 (25% yield) (Scheme 3A). Based on these results, a cyclohexyl radical-involved pathway was confirmed. Next, we examined the kinetic isotope effect (KIE) via an intermolecular competition experiment. The observed KIE value of 1.8 indicated that C–H bond homolysis plays a non-negligible role in the rate-determining step. Further mechanistic insights were gained by employing an aqueous solution of HCl (0.25 M in D2O); product 3a/3a–d1 was isolated in 74% yield with an H/D ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.1 (Scheme 3B). This prominent solvent isotope effect suggested water as the predominant proton donor for the benzylic position of product 3a. As shown in Scheme 3C, a range of control experiments were conducted. Notably, the formation of a dimer product 11 was significantly enhanced when the reaction was conducted in the presence of THF (acting as a hydrogen atom donor/reductant), HCl, and photocatalyst TBADT. This observation suggests that the reduced photocatalyst species [W10O32]5−H+, generated via HAT from THF, engages in a single electron transfer (SET) with the acid-generated cationic intermediate. This reduction yields a persistent benzylic radical, which subsequently undergoes homocoupling to furnish the dimer 11. A light on–off experiment revealed that the reaction could only occur under illumination conditions (Scheme 3D). Kinetic studies revealed a noticeable acceleration of the reaction rate with increasing HCl concentration (Scheme 3E). These measurements were completed within 0.5–1.0 hours, demonstrating significantly faster kinetics than most reported HAT-mediated transformations, which generally require more than 10 hours for completion. According to the reported literature, this phenomenon can be explained by the dual catalytic role of HCl in this system, both facilitating the formation of carbocation intermediates and synergistically promoting the HAT process with TBADT.13 Furthermore, UV-vis spectroscopic analysis was performed to investigate the electronic properties of 1a under acidic conditions (Scheme 3F). While compound 1a itself exhibited no detectable absorption in the visible light region, the addition of HCl induced obvious absorption bands in this spectral region. Notably, the absorption intensity showed a clear concentration-dependent enhancement with increasing HCl concentration.
2.1 (Scheme 3B). This prominent solvent isotope effect suggested water as the predominant proton donor for the benzylic position of product 3a. As shown in Scheme 3C, a range of control experiments were conducted. Notably, the formation of a dimer product 11 was significantly enhanced when the reaction was conducted in the presence of THF (acting as a hydrogen atom donor/reductant), HCl, and photocatalyst TBADT. This observation suggests that the reduced photocatalyst species [W10O32]5−H+, generated via HAT from THF, engages in a single electron transfer (SET) with the acid-generated cationic intermediate. This reduction yields a persistent benzylic radical, which subsequently undergoes homocoupling to furnish the dimer 11. A light on–off experiment revealed that the reaction could only occur under illumination conditions (Scheme 3D). Kinetic studies revealed a noticeable acceleration of the reaction rate with increasing HCl concentration (Scheme 3E). These measurements were completed within 0.5–1.0 hours, demonstrating significantly faster kinetics than most reported HAT-mediated transformations, which generally require more than 10 hours for completion. According to the reported literature, this phenomenon can be explained by the dual catalytic role of HCl in this system, both facilitating the formation of carbocation intermediates and synergistically promoting the HAT process with TBADT.13 Furthermore, UV-vis spectroscopic analysis was performed to investigate the electronic properties of 1a under acidic conditions (Scheme 3F). While compound 1a itself exhibited no detectable absorption in the visible light region, the addition of HCl induced obvious absorption bands in this spectral region. Notably, the absorption intensity showed a clear concentration-dependent enhancement with increasing HCl concentration.
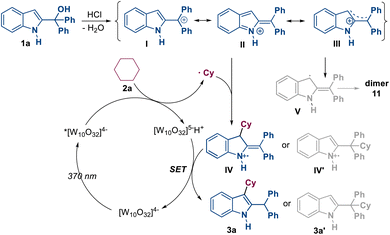
Based on the mechanistic verification presented above and previous reports,13,14 a plausible mechanism for the photoredox and Brønsted acid dual-catalyzed C3-alkylation of indoles is proposed in Scheme 4. On the one hand, in the presence of HCl, 2-indolemethanol 1 transforms into resonant intermediates, including carbocation I, vinyliminium II and delocalized cation III. On the other hand, photoexcitation of [W10O32]4− generates its excited state *[W10O32]4−, which can directly abstract a hydrogen atom from R5–H 2, leading to the formation of alkyl radical R5˙ and [W10O32]5−H+. The alkyl radical R5˙ can be trapped by the vinyliminium intermediate II, yielding intermediates IV or IV′. Following the SET process between [W10O32]5−H+ and intermediates IV or IV′, two distinct products are generated. 3a′ is produced directly from the SET process, whereas 3a is obtained after an intramolecular rearrangement of the intermediate formed in the SET process. Additionally, the delocalized cation III can be transformed into a radical intermediate V via an SET process with [W10O32]5−H+, and a dimer product 11 is further produced from intermediate V.
Conclusions
In summary, we report a synergistic catalytic protocol that merges decatungstate photocatalysis and Brønsted acid catalysis to achieve the direct C–H indolation of alkanes and aldehydes with 2-indolylmethanols. This method effectively overcomes the inherent polarity mismatch through a radical-polar crossover strategy, enabling the direct coupling of 2-indolylmethanols with a broad range of radical precursors with high regioselectivity. This protocol was applied to 73 examples, notably involving the late-stage functionalization of complex natural products and pharmaceuticals. Mechanistic investigations reveal the critical dual role of the Brønsted acid: it not only facilitates the dehydration of 2-indolylmethanols to generate the electrophilic cationic acceptor but also synergistically accelerates the decatungstate-mediated HAT process. Given the ubiquity of indole scaffolds in pharmaceutically important compounds, we anticipate that this methodology will find widespread application in medicinal chemistry and synthesis.Author contributions
D. Z. conceptualized the project. G. P. and Y. D. performed the experiments and analyzed the results. M. Z., Y. H., H. C. and X. W. assisted in performing the experiments. G. Q., Y. A., and D. Z. supervised and directed the project. Y. A. and D. Z. wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: further details of the experimental procedures, 1H and 13C NMR spectra, HRMS data, and X-ray crystallographic data for 3a. See DOI: https://doi.org/10.1039/d6sc01827k.CCDC 2444563 contains the supplementary crystallographic data for this paper.15
Acknowledgements
We gratefully acknowledge the financial support of the “Jiangsu Specially-Appointed Professors Program”, the National Natural Science Foundation of China (22201130, 22401148, and 22208158) and the State Key Laboratory of Materials-Oriented Chemical Engineering, Nanjing Tech University.Notes and references
- (a) A. J. Kochanowska-Karamyan and M. T. Hamann, Marine Indole Alkaloids: Potential New Drug Leads for the Control of Depression and Anxiety, Chem. Rev., 2010, 110, 4489–4497 CrossRef CAS PubMed; (b) W. Gul and M. T. Hamann, Indole Alkaloid Marine Natural Products: An Established Source of Cancer Drug Leads with Considerable Promise for the Control of Parasitic, Neurological and Other Diseases, Life Sci., 2005, 78, 442–453 CrossRef CAS PubMed; (c) E. J. Thomas, in Science of Synthesis: Houben-Weyl Methods of Molecular Transformation, Thieme: Stuttgart, Germany, 2000 Search PubMed; (d) Y. Wang, X. Tang, Z. Shao and J. Ren, J, Indole-Based Alkaloids from Deep-Sea Bacterium Shewanella piezotolerans with Antitumor Activities, J. Antibiot., 2014, 67, 395–398 CrossRef CAS PubMed.
- (a) S. Cacchi and G. Fabrizi, Update 1 of: Synthesis and Functionalization of Indoles Through Palladium-Catalyzed Reactions, Chem. Rev., 2011, 111, PR215–PR283 CrossRef PubMed; (b) R. Dalpozzo, Catalytic Strategies for the Asymmetric Functionalization of Indoles: An Update, Chem. Soc. Rev., 2015, 44, 742–778 RSC; (c) M. Bandini and A. Eichholzer, Catalytic Functionalization of Indoles in a New Dimension, Angew. Chem., Int. Ed., 2009, 48, 9608–9644 CrossRef CAS PubMed; (d) T.-Z. Li, S.-J. Liu, W. Tan and F. Shi, Catalytic Asymmetric Construction of Axially Chiral Indole-Based Frameworks: An Emerging Area, Chem. – Eur. J., 2020, 26, 15779–15792 CrossRef CAS PubMed; (e) W. Wang, X. Bai, S. Jin, J. Guo, Y. Zhao, H. Miao, Y. Zhu, Q. Wang and Z. Bu, An Unexpected FeCl3-Catalyzed Cascade Reaction of Indoles and o-Hydroxychalcones for the Assembly of Chromane-Bridged Polycyclic Indoles, Org. Lett., 2018, 20, 3451–3454 Search PubMed; (f) J.-B. Chen and Y.-X. Jia, Recent Progress in Transition-Metal-Catalyzed Enantioselective Indole Functionalizations, Org. Biomol. Chem., 2017, 15, 3550–3567 Search PubMed; (g) P. Wu, W.-T. Zhang, J.-X. Yang, X.-Y. Yu, S.-F. Ni, W. Tan and F. Shi, Synthesis of Alkene Atropisomers with Multiple Stereogenic Elements via Catalytic Asymmetric Rearrangement of 3-Indolylmethanols, Angew. Chem., Int. Ed., 2024, 63, e202410581 CAS; (h) Y. Kang, F. Wang and X. Li, Rhodium-Catalyzed Asymmetric Hydroselenation of 1-Alkynylindoles for Atroposelective Synthesis of Vinyl Selenoethers, ACS Catal., 2024, 14, 13055–13064 CrossRef CAS; (i) Z. Deng, L. Meng, X. Bing, S. Niu, X. Zhang, J. Peng, Y. Luan, L. Chen and P. Tang, Silver-Enabled Dearomative Trifluoromethoxylation of Indoles, J. Am. Chem. Soc., 2024, 146, 2325–2332 CrossRef CAS PubMed; (j) F. Jiang, K.-W. Chen, P. Wu, Y.-C. Zhang, Y. Jiao and F. Shi, A Strategy for Synthesizing Axially Chiral Naphthyl-Indoles: Catalytic Asymmetric Addition Reactions of Racemic Substrates, Angew. Chem., Int. Ed., 2019, 58, 15104–15110 CrossRef CAS PubMed; (k) K.-W. Chen, Z.-H. Chen, S. Yang, S.-F. Wu, Y.-C. Zhang and F. Shi, Organocatalytic Atroposelective Synthesis of N−N Axially Chiral Indoles and Pyrroles by De Novo Ring Formation, Angew. Chem., Int. Ed., 2022, 61, e202116829 CrossRef CAS PubMed; (l) J.-Y. Zhang, J.-Y. Chen, C.-H. Gao, L. Yu, S.-F. Ni, W. Tan and F. Shi, Asymmetric (4+n) Cycloadditions of Indolyldimethanols for the Synthesis of Enantioenriched Indole-Fused Rings, Angew. Chem., Int. Ed., 2023, 62, e202305450 CrossRef CAS PubMed; (m) Z.-H. Chen, T.-Z. Li, N.-Y. Wang, X.-F. Ma, S.-F. Ni, Y.-C. Zhang and F. Shi, Organocatalytic Enantioselective Synthesis of Axially Chiral N,N’-Bisindoles, Angew. Chem., Int. Ed., 2023, 62, e202300419 CrossRef CAS PubMed.
- (a) S. Lancianesi, A. Palmieri and M. Petrini, Synthetic Approaches to 3-(2-Nitroalkyl) Indoles and Their Use to Access Tryptamines and Related Bioactive Compounds, Chem. Rev., 2014, 114, 7108–7149 Search PubMed; (b) C. Pradhan and B. Punji, Advancement in the C-H Bond Alkylation of (Hetero)arenes Catalyzed by the Most Abundant Transition Metal-Iron, Org. Chem. Front., 2024, 11, 2397–2417 RSC; (c) L. Wang, Y. Chen and J. Xiao, Alkylideneindoleninium Ions and Alkylideneindolenines: Key Intermediates for the Asymmetric Synthesis of 3-Indolyl Derivatives, Asian J. Org. Chem., 2014, 3, 1036–1052 CrossRef CAS.
- (a) X. Zhang, E. Jones-Mensah, J. Deobald and J. Magolan, Alkylation of Indoles with α,β-Unsaturated Ketones using Alumina in Hexanes, Adv. Synth. Catal., 2019, 361, 5548–5551 CrossRef CAS; (b) J. Li, W. Lu, Y. Lu, Z. Zha and Z. Wang, Construction of Chiral All-Carbon Quaternary Stereocenters by Asymmetric Friedel–Crafts Reaction of Isatin Derivatives, Chin. J. Chem., 2022, 40, 195–200 CrossRef CAS; (c) T. Suzuka, Y. Ooshiro and K. Ogihara, Friedel–Crafts-Type Alkylation of Indoles in Water Using Amphiphilic Resin-Supported 1,10-Phenanthroline–Palladium Complex under Aerobic Conditions, Catalyst, 2020, 10, 193 Search PubMed; (d) J. Wei, B. Cao, C.-W. Tse, X.-Y. Chang, C.-Y. Zhou and C.-M. Che, Chiral cis-Iron(II) Complexes with Metal- and Ligand-Centered Chirality for Highly Regio- and Enantioselective Alkylation of N-Heteroaromatics, Chem. Sci., 2020, 11, 684–693 RSC; (e) C.-C. Xie, R. Tan and Y.-K. Liu, Asymmetric Construction of Polycyclic Indole Derivatives with Different Ring Connectivities by an Organocatalysis Triggered Two-Step Sequence, Org. Chem. Front., 2019, 6, 919–924 RSC; (f) Y. Zhang, N. Yang, X. Liu, J. Guo, X. Zhang, L. Lin, C. Hu and X. Feng, Reversal of Enantioselective Friedel-Crafts C3-Alkylation of Pyrrole by Slightly Tuning the Amide Units of N,N’-Dioxide Ligands, Chem. Commun., 2015, 51, 8432–8435 RSC.
- (a) S. Lerch, L.-N. Unkel and M. Brasholz, Tandem Organocatalysis and Photocatalysis: An Anthraquinone-Catalyzed Indole-C3-Alkylation/Photooxidation/1,2-Shift Sequence, Angew. Chem., Int. Ed., 2014, 53, 6558–6562 CrossRef CAS PubMed; (b) A. K. Bains, A. Biswas and D. Adhikari, Nickel-Catalyzed Chemoselective C-3 Alkylation of Indoles with Alcohols through a Borrowing Hydrogen Method, Chem. Commun., 2020, 56, 15442–15445 RSC; (c) M. Hu, Y. Jiang, N. Sun, B. Hu, Z. Shen, X. Hu and L. Jin, Nickel-Catalyzed C3-Alkylation of Indoles with Alcohols via a Borrowing Hydrogen Strategy, New J. Chem., 2021, 45, 10057–10062 RSC; (d) B. Zhou, Z. Ma, A. M. Alenad, C. Kreyenschulte, S. Bartling, M. Beller and R. V. Jagadeesh, Cobalt-Catalyzed C–H Alkylation of Indoles with Alcohols by Borrowing Hydrogen Methodology, Green Chem., 2022, 24, 4566–4572 RSC; (e) E. J. Pazur, N. R. Tasker and P. Wipf, C3-Functionalization of Indoles with α-Heteroaryl-Substituted Methyl Alcohols, Org. Biomol. Chem., 2023, 21, 8651–8657 RSC; (f) G. D. Gregorio, M. Mari, F. Bartoccini and G. Piersanti, Iron-Catalyzed Direct C3-Benzylation of Indoles with Benzyl Alcohols through Borrowing Hydrogen, J. Org. Chem., 2017, 82, 8769–8775 CrossRef PubMed; (g) X. Jiang, W. Tang, D. Xue, J. Xiao and C. Wang, Divergent Dehydrogenative Coupling of Indolines with Alcohols, ACS Catal., 2017, 7, 1831–1835 CrossRef CAS; (h) M. Kamal, S. Khatua, B. Rani, S. Goswami and S. Samanta, Alcohol Dehydrogenation-Triggered Selective C3-Alkylation of Indoles by Homogeneous Azo-aromatic Cobalt Catalysts, J. Org. Chem., 2023, 88, 5827–5843 CrossRef CAS PubMed; (i) A. K. Guin, S. Pal, S. Chakraborty, S. Chakraborty and N. D. Paul, Oxygen Dependent Switchable Selectivity during Ruthenium Catalyzed Selective Synthesis of C3-Alkylated Indoles and Bis(indolyl)methanes, J. Org. Chem., 2023, 88, 16755–16772 CrossRef CAS PubMed.
- For selected examples of other strategies for the C3-alkylation of indoles, see: (a) T. Zha, J. Rui, Z. Zhang, D. Zhang, Z. Yang, P. Yu, Y. Wang, F. Peng and Z. Shao, Direct Catalytic Asymmetric and Regiodivergent N1- and C3-Allenylic Alkylation of Indoles, Angew. Chem., Int. Ed., 2023, 62, e202300844 CrossRef CAS PubMed; (b) Z. Wang, G. Xu, S. Tang, Y. Shao and J. Sun, Catalyst-Controlled Selective Alkylation/Cyclopropanation of Indoles with Vinyl Diazoesters, Org. Lett., 2019, 21, 8488–8491 CrossRef CAS PubMed; (c) Y. Peng, Y. Wang, K. Wang, Q. Sun and X. Bao, Visible-Light Photocatalyzed C3–H Alkylation of 2H-Indazoles/Indoles with Sulfoxonium Ylides via Diversified Mechanistic Pathways, ACS Catal., 2024, 14, 1193–1204 CrossRef CAS; (d) J. Pan, R. Zhao, J. Guo, D. Ma, Y. Xia, Y. Gao, P. Xu and Y. Zhao, Three-Component 3-(Phosphoryl)methylindole Synthesis from Indoles, H-Phosphine Oxides and Carbonyl Compounds under Metal-Free Conditions, Green Chem., 2019, 21, 792–797 RSC; (e) S. Basak, A. Alvarez-Montoya, L. Winfrey, R. L. Melen, L. C. Morrill and A. P. Pulis, B(C6F5)3-Catalyzed Direct C3 Alkylation of Indoles and Oxindoles, ACS Catal., 2020, 10, 4835–4840 CrossRef CAS PubMed.
- (a) S.-J. Liu, X. Wang, J.-X. Yang, X.-S. Ao, S.-F. Ni, Y.-C. Zhang and F. Shi, Atroposelective Construction of Axially Chiral Alkenylindole-Fused Nine-Membered Rings via Catalytic Asymmetric Formal (4+5) Cycloaddition, Nat. Commun., 2025, 16, 6605 CrossRef CAS PubMed; (b) H.-H. Zhang and F. Shi, Organocatalytic Atroposelective Synthesis of Indole Derivatives Bearing Axial Chirality: Strategies and Applications, Acc. Chem. Res., 2022, 55, 2562–2580 CrossRef CAS PubMed; (c) J.-Y. Wang, C.-H. Gao, C. Ma, X.-Y. Wu, S.-F. Ni, W. Tan and F. Shi, Design and Catalytic Asymmetric Synthesis of Furan-Indole Compounds Bearing both Axial and Central Chirality, Angew. Chem., Int. Ed., 2024, 63, e202316454 CrossRef CAS PubMed; (d) C. Li, H.-H. Zhang, T. Fan, Y. Shen, Q. Wu and F. Shi, Brønsted Acid-Catalyzed Regioselective Reactions of 2-Indolylmethanols with Cyclic Enaminone and Anhydride Leading to C3-Functionalized Indole Derivatives, Org. Biomol. Chem., 2016, 14, 6932–6936 RSC; (e) M.-M. Xu, H.-Q. Wang, Y.-J. Mao, G.-J. Mei, S.-L. Wang and F. Shi, Cooperative Catalysis-Enabled Asymmetric α-Arylation of Aldehydes Using 2-Indolylmethanols as Arylation Reagents, J. Org. Chem., 2018, 83, 5027–5034 CrossRef CAS PubMed; (f) C. Ma, F. Jiang, F.-T. Sheng, Y. Jiao, G.-J. Mei and F. Shi, Design and Catalytic Asymmetric Construction of Axially Chiral 3,3’-Bisindole Skeletons, Angew. Chem., Int. Ed., 2019, 58, 3014–3020 CrossRef CAS PubMed.
- (a) H.-H. Zhang, C.-S. Wang, C. Li, G.-J. Mei, Y. Li and F. Shi, Design and Enantioselective Construction of Axially Chiral Naphthyl-Indole Skeletons, Angew. Chem., Int. Ed., 2017, 56, 116–121 CrossRef CAS PubMed; (b) Z.-Q. Zhu, Y. Shen, J.-X. Liu, J.-Y. Tao and F. Shi, Enantioselective Direct α-Arylation of Pyrazol-5-ones with 2-Indolylmethanols via Organo-Metal Cooperative Catalysis, Org. Lett., 2017, 19, 1542–1545 CrossRef CAS PubMed; (c) Y.-Y. He, X.-X. Sun, G.-H. Li, G.-J. Mei and F. Shi, Substrate-Controlled Regioselective Arylations of 2-Indolylmethanols with Indoles: Synthesis of Bis(indolyl)methane and 3,3’-Bisindole Derivatives, J. Org. Chem., 2017, 82, 2462–2471 CrossRef CAS PubMed; (d) C. Hu, G. Hong, Y. He, C. Zhou, M. C. Kozlowski and L. Wang, Lewis Acid-Controlled Regioselective Phosphorylation of 2-Indolylmethanols with Diarylphosphine Oxides: Synthesis of Highly Substituted Indoles, J. Org. Chem., 2018, 83, 4739–4753 CrossRef CAS PubMed; (e) Y. Zhou, W.-B. Cao, L.-L. Zhang, X.-P. Xu and S.-J. Ji, Ag(I)-Promoted Dehydroxylation and Site-Selective 1,7-Disulfonylation of Diaryl(1H-indol-2-yl)methanols, J. Org. Chem., 2018, 83, 6056–6065 CrossRef CAS PubMed; (f) S. Zhu, Y. Zhang, J. Luo, F. Wang, X. Cao and S. Huang, Temperature-Controlled Regioselective Thiolation of 2-Indolylmethanols under Aqueous Micellar Conditions, Green Chem., 2020, 22, 657–661 RSC; (g) C. Ma, J.-Y. Zhou, Y.-Z. Zhang, Y. Jiao, G.-J. Mei and F. Shi, Synergistic-Catalysis-Enabled Reaction of 2-Indolymethanols with Oxonium Ylides for the Construction of 3-Indolyl-3-Alkoxy Oxindole Frameworks, Chem.–Asian J., 2018, 13, 2549–2558 CrossRef CAS PubMed; (h) L. Chen, Y.-X. Zou, X.-Y. Fang, J. Wu and X.-H. Sun, Brønsted Acid-Catalyzed Regiodivergent Phosphorylation of 2-Indolylmethanols to Synthesize Benzylic Site or C3-Phosphorylated Indole Derivatives, Org. Biomol. Chem., 2018, 16, 7417–7424 RSC.
- (a) S.-C. Zhan, J. Sun, Q. Sun, Y. Han and C.-G. Yan, Acid-Modulated Construction of Cyclopenta[b]indole and Cyclohepta[b]indole via Unprecedented C3/C2 Carbocation Rearrangement, J. Org. Chem., 2023, 88, 5440–5456 CrossRef CAS PubMed; (b) J. Mao, H. Zhang, X.-F. Ding, X. Luo and W.-P. Deng, Synergistic Catalysis for Asymmetric [3 + 2] Cycloadditions of 2-Indolylmethanols with α,β-Unsaturated Aldehydes, J. Org. Chem., 2019, 84, 11186–11194 CrossRef CAS PubMed; (c) Z.-Q. Zhu, Y. Shen, X.-X. Sun, J.-Y. Tao, J.-X. Liu and F. Shi, Catalytic Asymmetric [3+2] Cycloadditions of C-3 Unsubstituted 2-Indolylmethanols: Regio-, Diastereo- and Enantioselective Construction of the Cyclopenta[b]indole Framework, Adv. Synth. Catal., 2016, 358, 3797–3808 CrossRef CAS; (d) M.-M. Xu, H.-Q. Wang, Y. Wan, S.-L. Wang and F. Shi, Enantioselective Construction of Cyclopenta[b]indole Scaffolds via the Catalytic Asymmetric [3 + 2] Cycloaddition of 2-Indolylmethanols with p-Hydroxystyrenes, J. Org. Chem., 2017, 82, 10226–10233 Search PubMed.
- (a) T.-Z. Li, S.-J. Liu, Y.-W. Sun, S. Deng, W. Tan, Y. Jiao, Y.-C. Zhang and F. Shi, Regio- and Enantioselective (3+3) Cycloaddition of Nitrones with 2-Indolylmethanols Enabled by Cooperative Organocatalysis, Angew. Chem., Int. Ed., 2021, 60, 2355–2363 CrossRef CAS PubMed; (b) Z.-Q. Zhu, L. Yu, M. Sun, G.-J. Mei and F. Shi, Regioselective [3+3] Cyclization of 2-Indolymethanols with Vinylcyclopropanes via Metal Catalysis, Adv. Synth. Catal., 2018, 360, 3109–3116 CrossRef CAS; (c) Y.-C. Shi, X.-Y. Yan, P. Xu, S. Jiang, R. Xu, W. Tan and F. Shi, Design and Application of m-Hydroxybenzyl Alcohols in Regioselective (3+3) Cycloadditions of 2-Indolymethanols, Chin. J. Chem., 2023, 41, 27–36 CrossRef CAS; (d) S. Nawaz, Y. Huang, X. Bao, S. Wei, X. Wei, J. Qu and B. Wang, Construction of a Spiro[pyrazolone-4,2’-pyridoindole] Scaffold via a [3+3] Cycloaddition of 2-Indolylmethanol with a 4-Aminopyrazolone-Derived Azomethine Ylide, Org. Biomol. Chem., 2021, 19, 8530–8538 RSC; (e) L.-Z. Qin, Y.-L. Cheng, X. Wen, Q.-L. Xu and L. Zhen, Synthesis of Indole-Fused Scaffolds via [3+3] Cyclization Reaction of 2-Indolylmethanols with Quinone Imines, Tetrahedron, 2021, 77, 131742 CrossRef CAS.
- (a) M. Sun, C. Ma, S.-J. Zhou, S.-F. Lou, J. Xiao, Y. Jiao and F. Shi, Catalytic Asymmetric (4+3) Cyclizations of In Situ Generated ortho-Quinone Methides with 2-Indolylmethanols, Angew. Chem., Int. Ed., 2019, 58, 8703–8708 Search PubMed; (b) S. Deng, C. Qu, Y. Jiao, W. Liu and F. Shi, Insights into 2-Indolylmethanol-Involved Cycloadditions: Origins of Regioselectivity and Enantioselectivity, J. Org. Chem., 2020, 85, 11641–11653 CrossRef CAS PubMed; (c) Z.-W. Qiu, B. Q. Li, H.-F. Liu, Z.-Q. Zhu, H.-P. Pan, N. Feng, A.-J. Ma, J.-B. Peng and X.-Z. Zhang, Formal (3 + 4)-Annulation of Propargylic p-Quinone Methides with 2-Indolylmethanols: Synthesis of Polysubstituted Indole-Fused Oxepines, J. Org. Chem., 2021, 86, 7490–7499 CrossRef CAS PubMed; (d) S.-J. Zhou, M. Sun, J.-Y. Wang, X.-Y. Yu, H. Lu, Y.-C. Zhang and F. Shi, Metal-Catalyzed Regiospecific (4+3) Cyclization of 2-Indolylmethanols with ortho-Quinone Methides, Eur. J. Org Chem., 2020, 2020, 4301–4308 CrossRef CAS.
- (a) J. Zhang and A. Studer, Decatungstate-Catalyzed Radical Disulfuration through Direct C–H Functionalization for the Preparation of Unsymmetrical Disulfides, Nat. Commun., 2022, 13, 3886 CrossRef CAS PubMed; (b) A. Businski, D. Hugenbusch, T. C. Ta, R. Tayaran, L. Unterriker, J. Glasenapp, C. Näther and R. Herges, Triphenylene Diazocines: Butterfly-Type Rigid Photoswitches with Annulated Aromatic Ring Systems and Increased Switching Amplitude, Org. Lett., 2026, 28, 1–6 CrossRef CAS PubMed; (c) M. B. Nodwell, A. Bagai, S. D. Halperin, R. E. Martin, H. Knust and R. Britton, Chem. Commun., 2015, 51, 11783–11786 RSC; (d) S. Yang, H. Li, J. Lv, L. Wang, Y. Lu, G. Sun, X. Wang, Q. Yin, Y. Bi and X. Fang, Modifications of Terpenoids via Inert Aliphatic C–H Bond Heteroarylation with Heteroarenes, Chem. Commun., 2025, 61, 8564–8567 RSC.
- (a) L. Capaldo, D. Ravelli and M. Fagnoni, Direct Photocatalyzed Hydrogen Atom Transfer (HAT) for Aliphatic C–H Bonds Elaboration, Chem. Rev., 2022, 122, 1875–1924 CrossRef CAS PubMed; (b) H. Cao, X. Tang, H. Tang, Y. Yuan and J. Wu, Photoinduced Intermolecular Hydrogen Atom Transfer Reactions in Organic Synthesis, Chem Catal., 2021, 1, 523–598 CAS; (c) H. Tan, C. Zhang, Y. Deng, M. Zhang, X. Cheng, J. Wu and D. Zheng, Photoinduced Radical Sulfinylation of C(sp3)–H Bonds with Sulfinyl Sulfones, Org. Lett., 2023, 25, 2883–2888 CrossRef CAS PubMed; (d) S. Xu, Y. Ping, Y. Su, H. Guo, A. Luo and W. Kong, A Modular Approach to Catalytic Stereoselective Synthesis of Chiral 1,2-Diols and 1,3-Diols, Nat. Commun., 2025, 16, 364 CrossRef CAS PubMed; (e) A. M. Nair, P. Martínez-Balart, S. Barbeira-Arán and M. Fañanás-Mastral, Cross-Coupling of Gaseous Alkanes with (Hetero)Aryl Bromides via Dual Nickel/Photoredox Catalysis, Angew. Chem., Int. Ed., 2025, 64, e202416957 CrossRef CAS PubMed; (f) I. B. Perry, T. F. Brewer, P. J. Sarver, D. M. Schultz, D. A. DiRocco and D. W. C. MacMillan, Direct Arylation of Strong Aliphatic C–H Bonds, Nature, 2018, 560, 70–75 CrossRef CAS PubMed; (g) W. Ding, Z. He, M. Sayed, Y. Zhou, Z. Han and L. Gong, Enantioselective Synthesis of β- and α-Amino Ketones through Reversible Alkane Carbonylation, Nat. Synth., 2024, 3, 507–516 CrossRef CAS; (h) J. Chen, C. Tan, J. Rodrigalvarez, S. Zhang and R. Martin, Site-Selective Distal C(sp3)–H Bromination of Aliphatic Amines as a Gateway for Forging Nitrogen-Containing sp3 Architectures, Angew. Chem., Int. Ed., 2024, 63, e202406485 CrossRef CAS; (i) H. Zeng, R. Yin, Y. Zhao, J. Ma and J. Wu, Modular Alkene Synthesis from Carboxylic Acids, Alcohols and Alkanes via Integrated Photocatalysis, Nat. Chem., 2024, 16, 1822–1830 CrossRef CAS PubMed; (j) X. Hu, I. Cheng-Sánchez, W. Kong, G. A. Molander and C. Nevado, Nickel-Catalyzed Enantioselective Alkene Dicarbofunctionalization Enabled by Photochemical Aliphatic C–H Bond Activation, Nat. Catal., 2024, 7, 655–665 CrossRef CAS PubMed; (k) N. Marie, J.-A. Ma, V. Tognetti and D. Cahard, Photocatalyzed Cascade Hydrogen Atom Transfers for Assembly of Multi-Substituted α-SCF3 and α-SCF2H Cyclopentanones, Angew. Chem., Int. Ed., 2024, 63, e202407689 CrossRef CAS PubMed; (l) V. Palani and A. E. Wendlandt, Strain-Inducing Positional Alkene Isomerization, J. Am. Chem. Soc., 2023, 145, 20053–20061 CrossRef CAS PubMed; (m) Y.-A. Zhang, V. Palani, A. E. Seim, Y. Wang, K. J. Wang and A. E. Wendlandt, Stereochemical Editing Logic Powered by the Epimerization of Unactivated Tertiary Stereocenters, Science, 2022, 378, 383–390 CrossRef CAS PubMed; (n) G. Laudadio, Y. Deng, K. Ravelli, M. Nuño, M. Fagnoni, D. Guthrie, Y. Sun and T. Noël, C(sp3)–H Functionalizations of Light Hydrocarbons Using Decatungstate Photocatalysis in Flow, Science, 2020, 369, 92–96 CrossRef CAS PubMed; (o) Q. Wang, S. Ni, L. Yu, Y. Pan and Y. Wang, Photoexcited Direct Amination/Amidation of Inert Csp3–H Bonds via Tungsten–Nickel Catalytic Relay, ACS Catal., 2022, 12, 11071–11077 CrossRef CAS; (p) K. Zheng, C. Chen, H. Xu, J. Mao and C. Shen, Unlocking the Accessibility of Alkyl/Aryl Radicals from Boronic Acids through EDA Complex Photoactivation, J. Org. Chem., 2025, 90, 15567–15577 CrossRef CAS PubMed; (q) J. Xu, J. Zhang, Z. Wang, J. Li, J. Shen and P. Zhang, Photocatalyzed Site-Selective Formal β-C(sp3)–H Sulfonylation of Ketones with Sulfinates, Org. Lett., 2026, 28, 2777–2782 CrossRef CAS PubMed; (r) Y.-Y. Zeng, J. Jiang, Y.-C. Wen, C.-L. Zhuang, L.-J. Ou, Z. Yang, H.-T. Zhu, Z.-L. Wang and W.-M. He, Decatungstate-Photocatalyzed Heterogeneous Direct Benzylation of N-heterocycles with Benzaldehydes, Chin. Chem. Lett., 2026, 37, 111776 CrossRef CAS.
- (a) H. Cao, D. Kong, L.-C. Yang, S. Chanmungkalakul, T. Liu, J. L. Piper, Z. Peng, L. Gao, X. Liu, X. Hong and J. Wu, Brønsted Acid-Enhanced Direct Hydrogen Atom Transfer Photocatalysis for Selective Functionalization of Unactivated C(sp3)–H Bonds, Nat. Synth., 2022, 1, 794–803 CrossRef CAS; (b) R. F. Renneke, M. Kadkhodayan, M. Pasquali and C. L. Hill, Roles of Surface Protonation on the Photodynamic, Catalytic, and Other Properties of Polyoxometalates Probed by the Photochemical Functionalization of Alkanes. Implications for Irradiated Semiconductor Metal Oxides, J. Am. Chem. Soc., 1991, 113, 8357–8367 CrossRef CAS.
- CCDC 2444563: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2n1rwc.
Footnote |
| † G. P. and Y. D. contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |