Open Access Article
Open Access Article18F-labelling of (hetero)aryl halides via sequential Miyaura borylation/copper-mediated radiofluorination
Abdias N. Noel a,
Samuel G. Grecoa,
Mami Horikawa
a,
Samuel G. Grecoa,
Mami Horikawa a,
Taylor E. Spillera,
Diana L. Nicholsa,
Jason A. Witek
a,
Taylor E. Spillera,
Diana L. Nicholsa,
Jason A. Witek b,
Allen F. Brooks
b,
Allen F. Brooks b,
Peter J. H. Scott
b,
Peter J. H. Scott *b and
Melanie S. Sanford
*b and
Melanie S. Sanford *a
*a
aDepartment of Chemistry, University of Michigan, 930 North University Avenue, Ann Arbor, Michigan 48109, USA. E-mail: mssanfor@umich.edu
bDepartment of Radiology, University of Michigan, 1301 Catherine, Ann Arbor, Michigan 48109, USA. E-mail: pjhscott@umich.edu
First published on 8th May 2026
Abstract
This article describes the development of a mild, general, and highly reproducible method for the radiofluorination of (hetero)aryl iodide, bromide, and chloride substrates. This transformation proceeds via sequential Pd-catalysed Miyaura borylation with B2Pin2 followed by in situ Cu-mediated radiofluorination of the (hetero)aryl-BPin intermediate with K18F. Successful implementation of this method required identifying and replacing/removing components of the borylation reaction that impede the radiofluorination step. This method is applied to electronically and sterically diverse (hetero)aryl halides and enables the synthesis of 18F-labelled analogues of bioactive molecules such as indomethacin, dimethomorph, and metaxalone. In addition, this sequence is effective for the radiofluorination of a variety of amino acids and peptides.
Introduction
Fluorine-18 is the most common radionuclide used in positron emission tomography (PET) imaging, based on its attractive half-life (t1/2 ∼ 110 min) and favorable imaging properties.1 More than 60% of FDA-approved radiotracers for PET imaging contain a C–18F bond, and among these 25% are (hetero)aryl fluorides (Ar18F).2 As the applications of nuclear medicine in drug discovery, diagnostic imaging, and disease therapy3–5 expand, the demand for new and increasingly complex 18F-labelled (hetero)aromatic molecules continues to accelerate.6Over the past decade, the copper-mediated radiofluorination (CMRF) of (hetero)aryl boron compounds [ArB(OR)2] has emerged as a versatile method for late-stage construction of Ar–18F bonds.7 These transformations are particularly powerful in the context of electron-neutral and electron-rich (hetero)arene scaffolds, where traditional nucleophilic aromatic substitution (SNAr)8 reactions fail. CMRF was developed independently by our team9,10 and by Gouverneur and coworkers.11 It has since been expanded by other groups12,13 and applied to the synthesis of numerous radiotracers, including [18F]F-DOPA,14 [18F]flumazenil,15 and [18F]TRACK.16 Despite this progress, there are challenges that limit even wider application and clinical translation of CMRF. First, preparing the requisite ArB(OR)2 precursors in high yield and purity remains a bottleneck. While a variety of synthetic methods are available for C(sp2)–B bond formation,17 many of these afford poor yields with densely functionalized pharmaceutical candidates and/or peptides. Second, even when borylation is successful, the isolation and purification of structurally complex ArB(OR)2 is often challenging. Finally, once isolated, many of the ArB(OR)2 products have limited shelf-stability due to degradation via hydrolysis and/or protodeboronation.14,18,19 This can detrimentally impact the radiolabeling yield, tracer purification, and reproducibility, all of which are critical for clinical applications.14,18
We sought to address these challenges by developing sequences that generate ArB(OR)2 in situ from stable, readily available, and inexpensive starting materials. These ArB(OR)2 intermediates could then be telescoped directly to CMRF without the need to isolate, purify, or store sensitive organoboron species. We recently demonstrated proof-of-concept for this approach in a tandem Ir-catalysed C–H borylation/CMRF reaction.20 This method leverages abundant and stable arene precursors and enables the late-stage C–H radiofluorination of a range of substrates. However, the sequence is limited by the often-poor site selectivity of C–H borylation with complex bioactive scaffolds. It also exhibits moderate functional group compatibility and suffers from reproducibility issues, with standard deviations in radiochemical yields of up to 20%. Overall, a selective, general, mild, and reproducible alternative would be highly enabling for the radiofluorination of bioactive scaffolds, including amino acids and peptides.
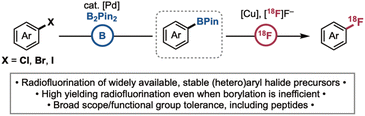
This article describes a borylation/radiofluorination sequence that starts from abundant, readily available, and stable (hetero)aryl halides (Scheme 1). Currently, the direct radiofluorination of aryl halides (particularly electron neutral or -rich derivatives that are not reactive in traditional SNAr pathways) remains challenging. The few known examples exhibit modest scope and functional group compatibility and/or afford low molar activity products. For instance, a recent report by Nicewicz and Li demonstrated photochemical cation radical-accelerated (CRA) substitution21 as a novel mechanistic manifold for the radiofluorination of electron rich (hetero)aryl halides. However, the transformation is limited to arenes with a relatively narrow range of redox potentials; furthermore, it works best with aryl fluoride precursors, which then afford low molar activity products. Our team recently reported several complementary Cu-mediated methods for the direct radiofluorination of aryl halides; however, these are limited by the requirement for high energy UV irradiation22 and/or the installation of directing groups proximal to the C(sp2)–X bond.23
 | ||
| Scheme 1 This work: 18F-fluorination of (hetero)aryl halides via sequential Miyaura borylation/copper-mediated radiofluorination. | ||
We demonstrate herein the development of a versatile radiofluorination of (hetero)aryl halides (halide = Cl, Br, I) involving sequential Pd-catalysed Miyaura borylation/Cu-mediated radiofluorination (Scheme 1). This method is highly reproducible and exhibits broad substrate scope and functional group compatibility. Furthermore, it proceeds in high radiochemical yield even when the initial borylation yield is modest. This sequence is showcased in the radiofluorination of bioactive molecules including a series of amino acids and peptides. Note: since radioactivity is hazardous, work was conducted by trained personnel in a dedicated radiochemistry facility, according to all institutional, state and federal rules, and the As Low As Reasonably Achievable (ALARA) principles.
Results and discussion
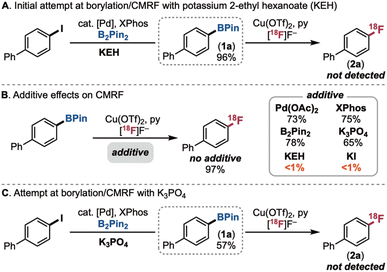
The first step of the proposed sequence involves the selective borylation of a C(sp2)–halogen bond. We targeted Pd-catalysed Miyaura borylation, since it is well-documented to proceed with high functional group tolerance under mild conditions and low catalyst loading.24 Using 4-iodobiphenyl as a test substrate, Miyaura borylation was performed under literature conditions (1 mol% of Pd(OAc)2, 3 mol% of XPhos, 1.2 equiv. of B2Pin2, 2.2 equiv. of potassium 2-ethyl hexanoate (KEH) in isopropyl acetate).25,26 The crude mixture was then telescoped directly to Cu-mediated radiofluorination (using Cu(OTf)2, pyridine, and 51.4 MBq (1.39 mCi) K18F in DMA/n-BuOH). However, this sequence failed to afford any of the radiofluorinated product 2a (Scheme 2A).Analysis following the first step revealed that the borylated intermediate 1a was formed in 96% yield (Scheme 2A). Furthermore, an isolated sample of 1a underwent radiofluorination in 97% radiochemical yield (RCY, calculated using a combination of radio-TLC (for yield of organic and inorganic 18F) and radio-HPLC (for purity of organic 18F products) as detailed on p. S32 of the SI). These results indicate that some component(s) carried over from the borylation reaction impedes the radiofluorination step. To diagnose the problem, the CMRF of 1a was conducted in the presence of each of the borylation reagents (added in the maximum concentration that could be carried forward from the first step). As shown in Scheme 2B, Pd(OAc)2, XPhos, and B2Pin2 led to modest decreases in the yield of 2a. In contrast, KEH completely inhibited CMRF, suggesting that this salt is the problematic component. We hypothesize that KEH reacts with Cu(OTf)2 to form the carboxylate complex Cu(KEH)2. Control studies reveal that analogous Cu(II) carboxylates (e.g., Cu(OAc)2) do not mediate CMRF (see SI, p. S28).
While carboxylates are the most common bases used in Miyaura borylation, several reports have shown that phosphate salts are also effective.25 Encouragingly, additive studies revealed that K3PO4 is compatible with the CMRF step (Scheme 2B). Furthermore, Pd(OAc)2/XPhos-catalysed borylation with K3PO4 gives 57% yield of the borylated product 1a. However, the sequential borylation/CMRF with K3PO4 still failed to afford any of the 18F-labelled product 2a (Scheme 2C).
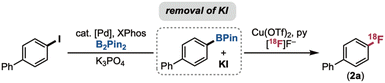
We next reasoned that KI, a by-product of the borylation step, was likely impeding radiofluorination. The negative impact of KI was confirmed by additive studies, with <1% RCY of 2a obtained for CMRF in the presence of 1 equiv. of KI (Scheme 2B). To remove this byproduct, we first explored filtration of the crude borylation mixture using sorbents that are expected to retain the salt. Using either a quaternary methyl ammonium (QMA) ion exchange resin (Table 1, entry 2) or a silica gel filter (entry 3) led to complete recovery of radiofluorination reactivity (>90% RCY) and high reproducibility (standard deviations of ±1–2%). Further experiments revealed that sorbent is not required in this filtration: simple mechanical filtration with a nylon syringe filter led to 86 ± 6% RCY of 2a (entry 4) due to the low solubility of KI in the isopropyl acetate solvent. Allowing the insoluble salts to settle for ∼30 min and then carefully transferring the solution phase also led to good radiochemical yield, albeit with higher variability (85 ± 9 RCY; entry 5). We ultimately moved forward with the nylon syringe filter procedure, since this proved inexpensive, simple, fast, and reproducible.
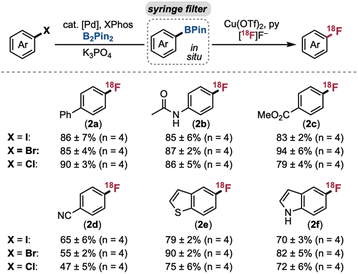
A test set of six electronically diverse (hetero)aryl iodide, -bromide, and -chloride substrates was next evaluated using the optimised procedure: (i) Pd-catalysed Miyaura borylation with K3PO4, (ii) filtration of the crude mixture through a nylon syringe filter, and (iii) Cu-mediated radiofluorination. All of the (hetero)aryl iodides and bromides afforded good to excellent radiochemical yields using commercial reagents stored and dispensed on the benchtop (Scheme 3). Minimal difference in yield was observed upon changing from –I to –Br, and the reactions were all highly reproducible (standard deviations of ≤±7). The (hetero)aryl chlorides gave comparable yields after three modifications to the borylation conditions: (i) the Pd pre-catalyst was changed from Pd(OAc)2 to Pd2(dba)3, (ii) the XPhos ligand was stored and dispensed under N2 atmosphere, and (iii) the temperature was increased to 110 °C. These changes enhanced both reproducibility and yield with these less activated substrates.33
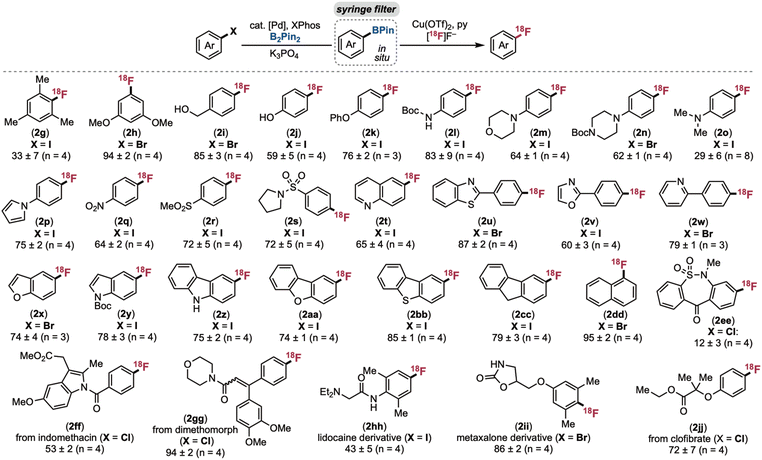
A broader set of (hetero)aryl halide derivatives was next evaluated to establish the scope and limitations of the method. As shown in Scheme 4, borylation/radiofluorination proved effective for accessing electronically diverse 18F-labelled (hetero)arenes. Products bearing electron-donating alkyl, phenol, ether, and aniline substituents (2g–2o) are particularly noteworthy, since these are not accessible via traditional SNAr radiofluorination of the aryl halide starting materials. Notably, even the 4-dimethylamino substrate, which was reported as unreactive starting from the isolated aryl boronate ester,19 afforded 2jj in 29% RCY. Various heterocycles proved compatible, including quinoline (2t), benzothiazole (2u), oxazole (2v), pyridine (2w), and benzofuran (2x). Across all these substrates, the borylation/radiofluorination sequence afforded moderate to high RCC and standard deviations of <±9%.27
Bioactive molecules containing aryl halides, including indomethacin, dimethomorph, and clofibrate were converted to the corresponding 18F derivatives (2ff, 2gg, and 2jj) in good to excellent radiochemical yields. Products 2ff and 2gg are the first 18F-analogues of these scaffolds. For 2jj, the radiochemical yield (72 ± 7%) was significantly higher than the 40 ± 3% RCY obtained using Nicewicz and Li's photochemical CRA approach.21 Notably, with dimethomorph, the borylation step proceeded in just 35% yield. Despite this, the radiofluorinated product 2gg was obtained in excellent radiochemical yield (94 ± 2%). This result highlights a key advantage of this method – high borylation yield is not required to obtain high radiochemical yield due to the change of limiting reagent between the two steps (from (hetero)aryl halide in step 1 to K18F in step 2). An 18F-labelled analogue of metaxalone (2ii) was also prepared in 86 ± 2% RCY using this method, demonstrating compatibility with a sterically hindered aryl bromide substrate.
Many of these 18F-labelled bioactive molecules are challenging to access from aryl halides using literature methods. For instance, nucleophilic aromatic substitution28 is not viable for the radiofluorination of electron-rich/neutral aryl halides to form 2gg, 2hh, 2ii, and 2jj (or the 18F-amino acids and peptides in Scheme 5; vide infra). Nicewicz and Li's CRA-SNAr radiofluorination would likely struggle to generate 2ff, 2gg, or many of the amino acids/peptides in Scheme 5 due to the redox potentials of the halide-containing rings.21 Our (NHC)Cu (NHC = N-heterocyclic carbene)-mediated radiofluorination23 only works with pyridine or imine directing groups proximal to the C(sp2)–halogen bond, a feature that is not present in any of these substrates. Finally, our UV light-promoted Cu-mediated radiofluorination affords modest yield and functional group compatibility even with simple substrates.22
The bioactive substrates in Schemes 4 and 5 also illustrate the advantages of this new approach compared to our prior tandem C–H borylation/CMRF method.20 For instance, 18F-lidocaine derivative 2hh was accessed in 43 ± 5% RCY yield using the current method versus just 15 ± 8% RCY via C–H borylation/CMRF. 18F-Phenylalanine derivative 3a (Scheme 5) was obtained in 88 ± 4% RCY yield as a single isomer from the corresponding aryl iodide. In contrast, C–H borylation/CMRF afforded a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.7 mixture regioisomers in 51 ± 17% RCY yield. Finally, products like 2ii are not accessible via the sterically-controlled C–H borylation approach due the high degree of congestion at the labelling site.
1.7 mixture regioisomers in 51 ± 17% RCY yield. Finally, products like 2ii are not accessible via the sterically-controlled C–H borylation approach due the high degree of congestion at the labelling site.
A final set of studies focused in applying this sequence to the 18F-labelling of amino acid and peptide substrates. As peptide-based therapeutics emerge at an accelerating pace,29 there is high demand for radiolabelled analogues to study biodistribution, quantify target engagement, and conduct in vivo disease diagnosis and treatment.30 Most current approaches to peptide radiofluorination utilize large 18F-containing prosthetic groups.31 While these enable late-stage radiolabelling and generate readily separable products, prosthetics can alter target affinity and physiochemical properties. As such, strategies for the direct 18F-radiolabelling of peptides with minimal structural perturbation (e.g. converting phenylalanine to [18F]fluorophenylalanine) are in high demand.32
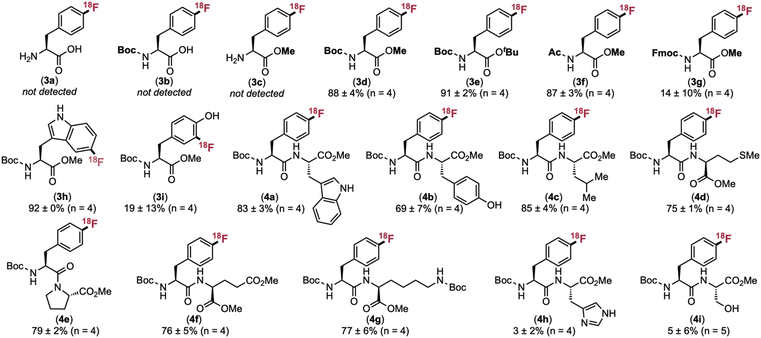
We first sought to identify protecting group schemes compatible with the borylation/CMRF of 4-iodophenylalanine derivatives. Unprotected, N-Boc protected, and C-methyl protected 4-iodo-L-phenylalanine afforded none of the corresponding radiofluorinated products 3a–c. Control studies revealed that both the borylation and CMRF steps proceed in <10% yield with these substrates. However, protection of the C- and N-termini with Me and Boc, respectively, led to excellent (88 ± 4%) radiochemical yield of 3d. N-Acyl and C-tert-butyl protecting groups also proved compatible (3e and 3f). In contrast, N-Fmoc led to large drop in both yield and reproducibility of the borylation/radiofluorination sequence (14 ± 10% RCY of 3g). Here, the borylation step leads to competing deprotection of the Fmoc group, likely due to the basic conditions. Boc-5-Br-L-Trp-OMe was also a viable substrate, yielding 83 ± 3% RCY of 3h. This is another example where a high RCY is obtained despite a low (25%) yield in the borylation step.
We next explored a series of dipeptides containing an N-terminal Boc-4-iodo-L-phenylalanine to assess compatibility with common side chain functional groups. Various C-terminal amino acids with unprotected side chains, including tryptophan, tyrosine, and methionine, were well tolerated (see products 4a, 4b, and 4d). However, modest radiochemical conversion (≤5%) was obtained for the C-terminal histidine and serine products 4h and 4i, indicating that protection of some side chain functional groups will be required in application of this method to more complex peptides.
Finally, we pursued the automation of this method on a TracerLab FXN-pro synthesis module. Translation of new radiochemistry methods to automated synthesis is crucial for accessing clinical doses of radiotracers, which typically require at least 20 mCi of activity at end-of-synthesis to allow for a 10 mCi injection after completion of QC testing and transport to the PET imaging suite. The manual reactions in Schemes 4 and 5 were performed using ∼51.4 MBq (1.39 mCi) of starting 18F per vial, while a typical automated run uses 66.6 GBq (∼1.8 Ci) of activity. Protected bromophenylalanine was used as a test case for automation. The borylation step and filtration were conducted manually, and then the crude mixture was loaded into the TracerLab FXN-pro synthesis module for Cu-mediated radiofluorination. Following semi-preparative HPLC purification, product 3d was obtained in a 9.2% decay-corrected radiochemical yield (RCY), 99% radiochemical purity (RCP), and a molar activity of 578 TBq mmol−1 (15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 605 Ci mmol−1 n = 2). Importantly the bromo precursor, borylated intermediate, and radiofluorinated product exhibit baseline separation by HPLC (pp. S100–S102), enabling facile purification of the target product 3d.
605 Ci mmol−1 n = 2). Importantly the bromo precursor, borylated intermediate, and radiofluorinated product exhibit baseline separation by HPLC (pp. S100–S102), enabling facile purification of the target product 3d.
The automated radiochemical yield (9.2%) is significantly lower than the manual yield (88%) for this substrate. However, automated synthesis starting from the isolated BPin precursor afforded 3d in comparable (13%) radiochemical yield, demonstrating that this drop in yield between manual and automated radiosynthesis is a general feature of CMRF rather than the telescoped sequence.34 In general, automated yields are well-precedented to be significantly lower than those for manual reactions for several reasons. First automation involves multiple steps and transfers of material, while the manual reactions are performed in a single step in a single vessel. Second, manual reactions start from soluble fluoride, with yields calculated relative to the fluoride added to the reaction mixture. In contrast, automated yields are calculated based on the total isolated product (non-decay-corrected) and starting amount of fluoride delivered from the cyclotron. Despite a lower automated yield compared to the manual test reactions, this automated sequence nevertheless delivers >5.5 GBq (>150 mCi) total activity of 3d. This is sufficient for both preclinical evaluation and several human doses of this material from a single production batch, demonstrating the feasibility of applying the sequential borylation/radiofluorination sequence to PET radiotracer synthesis in a (pre)clinical setting (Scheme 6).
Conclusions
In conclusion, this report showcases a tandem borylation–radiofluorination reaction of (hetero)aryl halides that provides access to a wide variety of 18F-labelled molecules. The (hetero)aryl halide (halide = Cl, Br or I) are readily available, inexpensive, and stable, and numerous functionalized derivatives (including bioactive molecules, amino acids, and dipeptides) are viable substrates. Key advantages of this approach include: (1) a wide substrate scope that is insensitive to the electronic and steric properties of the (hetero)aryl halide, (2) the ability to obtain high radiochemical yield even if the borylation step is inefficient, (3) the effectiveness of this transformation for (hetero)aryl halide substrates that are challenging to radiofluorinate using existing methods, and (4) the feasibility of high yielding radiofluorination of peptides, substrates of high interest for emerging diagnostic and therapeutic radiopharmaceuticals. These benefits widen the chemical space compatible with CMRF, enabling straightforward access to more diverse radiopharmaceuticals for PET imaging.Author contributions
A. N. N. developed and optimized the reaction sequence, designed and evaluated the substrate scope, and synthesized reference standards and labelling precursors of all non-peptide substrates. S. G. G. optimized the peptide radiolabelling reactions and synthesized labelling precursors and reference standards of all peptide substrates. M. H. and T. E. S. provided critical guidance on experimental design and planning, particularly at the outset of the project. A. N. N., S. G. G., J. A. W., and A. F. B. performed radiolabelling studies. D. N. and S. G. G. performed preliminary studies on the compatibility of different protecting groups for peptides in CMRF. M. S. S. and P. J. H. S. directed all aspects of the project and secured funding. All the authors contributed to writing/editing the manuscript.Conflicts of interest
The authors declare no competing financial interests.Data availability
Further details of experimental methods, additional analysis, and analytical data, as well as supplemental figures including NMR spectra, analytical radioTLC and radioHPLC traces, and additional screening and control experiments are included in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc01625a.Acknowledgements
The National Institutes of Health (R01EB021155) is gratefully acknowledged for support of this work. We thank Dr Karsten Donebauer and Dr Eric Webb for helpful discussions on the sequential reaction sequence, and Dr Monica Rivas for helpful discussions on peptide synthesis and purification.Notes and references
- (a) W. A. Weber, J. Czernin, C. J. Anderson, R. D. Badawi, H. Barthel, F. Bengel, L. Bodei, I. Buvat, M. DiCarli, M. M. Graham, J. Grimm, K. Herrmann, L. Kostakoglu, J. S. Lewis, D. A. Mankoff, T. E. Peterson, H. Schelbert, H. Schöder, B. A. Siegel and H. W. Strauss, J. Nucl. Med., 2020, 61, 263S–272S CrossRef PubMed; (b) S. Dhoundiyal, S. Srivastava, S. Kumar, G. Singh, S. Ashique, R. Pal, N. Mishra and F. Taghizadeh-Hesary, Eur. J. Med. Res., 2024, 29, 26 CrossRef PubMed.
- R. Halder and T. Ritter, J. Org. Chem., 2021, 86, 13873–13884 CrossRef CAS PubMed.
- D. J. Donnelly, PET Imaging in Drug Discovery & Development, in Handbook of Radiopharmaceuticals: Methodology and Applications, ed. Scott P. J. H., and Kilbourn M., John Wiley & Sons, Chichester, 2020, pp. 703–725 Search PubMed.
- M. Djekidel, Front. Nucl. Med., 2023, 3, 1213714 CrossRef PubMed.
- W. A. Weber, J. Czernin, C. J. Anderson, R. D. Badawi, H. Barthel, F. Bengel, L. Bodei, I. Buvat, M. DiCarli, M. M. Graham, J. Grimm, K. Herrmann, L. Kostakoglu, J. S. Lewis, D. A. Mankoff, T. E. Peterson, H. Schelbert, H. Schöder, B. A. Siegel and H. W. Strauss, J. Nucl. Med., 2020, 61, 263S–272S CrossRef PubMed.
- (a) A. L. Vāvere and P. J. H. Scott, Semin. Nucl. Med., 2017, 47, 429–453 CrossRef PubMed; (b) C. Mason, G. R. Gimblet, S. E. Lapi and J. S. Lewis, Radiol. Clin. North Am., 2021, 59, 887–918 CrossRef PubMed.
- (a) J. S. Wright, T. Kaur, S. Preshlock, S. S. Tanzey, W. P. Winton, L. S. Sharninghausen, N. Wiesner, A. F. Brooks, M. S. Sanford and P. J. H. Scott, Clin. Transl. Imaging, 2020, 8, 167–206 CrossRef; (b) J. S. Wright, L. S. Sharninghausen, A. Lapsys, M. S. Sanford and P. J. H. Scott, ACS Cent. Sci., 2024, 10, 1674–1688 CrossRef CAS PubMed; (c) G. D. Bowden, M. M. Muller, M. M. Herth, M. S. Sanford and P. J. H. Scott, npj Imaging, 2025, 3, 25 CrossRef CAS PubMed.
- (a) H. Sun and S. G. DiMagno, Angew. Chem., Int. Ed., 2006, 45, 2720–2725 CrossRef CAS PubMed; (b) O. Jacobson, D. O. Kiesewetter and X. Chen, Bioconjugate Chem., 2015, 26, 1–18 CrossRef CAS PubMed.
- N. Ichiishi, A. F. Brooks, J. J. Topczewski, M. Rodnick, M. S. Sanford and P. J. H. Scott, Org. Lett., 2014, 16, 3224–3227 CrossRef CAS PubMed.
- (a) Y. Ye, S. D. Schimler, P. S. Hanley and M. S. Sanford, J. Am. Chem. Soc., 2013, 135, 16292–16295 CrossRef CAS PubMed; (b) A. Mossine, A. F. Brooks, K. J. Makaravage, J. M. Miller, N. Ichiishi, M. S. Sanford and P. J. H. Scott, Org. Lett., 2015, 17, 5780–5783 CrossRef CAS PubMed.
- M. Tredwell, S. M. Preshlock, N. J. Taylor, S. Gruber, M. Huiban, J. Passchier, J. Mercier, C. Génicot and V. Gouverneur, Angew. Chem., Int. Ed., 2014, 53, 7751–7755 CrossRef CAS PubMed.
- (a) R. Richarz, P. Krapf, F. Zarrad, E. A. Urusova, B. Neumaier and B. D. Zlatopolskiy, Org. Biomol. Chem., 2014, 12, 8094–8099 RSC; (b) J. Zischler, N. Kolks, D. Modemann, B. Neumaier and B. D. Zlatopolskiy, Chem. – Eur. J., 2017, 23, 3251–3256 CrossRef CAS PubMed.
- (a) D. Antuganov, M. Zykov, V. Timofeev, K. Timofeeva, Y. Antuganova, V. Orlovskaya, O. Fedorova and R. Krasikova, Eur. J. Org Chem., 2019, 2019, 918–922 CrossRef CAS; (b) D. Zhou, W. Chu and J. A. Katzenellenbogen, J. Comp. Radiopharm., 2022, 65, 13–20 CrossRef CAS; (c) D. Zhou, W. Chu, H. Chen and J. Xu, ACS Med. Chem. Lett., 2023, 15, 116–122 CrossRef PubMed; (d) C. Hoffmann, N. Kolks, D. Smets, A. Haseloer, B. Gröner, E. A. Urusova, H. Endepols, F. Neumaier, U. Ruschewitz, A. Klein, B. Neumaier and B. D. Zlatopolskiy, Chem. – Eur. J., 2023, 29, e202202965 CrossRef CAS PubMed; (e) J. Sun, C. Jaworski, R. Schirrmacher and D. G. Hall, Chem. – Eur. J., 2024, 30, e202400906 CrossRef CAS PubMed.
- (a) A. V. Mossine, S. S. Tanzey, A. F. Brooks, K. J. Makaravage, N. Ichiishi, J. M. Miller, B. D. Henderson, M. B. Skaddan, M. S. Sanford and P. J. H. Scott, Org. Biomol. Chem., 2019, 17, 8701–8705 RSC; (b) A. V. Mossine, S. S. Tanzey, A. F. Brooks, K. J. Makaravage, N. Ichiishi, J. M. Miller, B. D. Henderson, T. Erhard, C. Bruetting, M. B. Skaddan, M. S. Sanford and P. J. H. Scott, Nat. Protoc., 2020, 15, 1742–1759 CrossRef CAS PubMed.
- T. Gendron, G. Destro, N. J. W. Straathof, J. B. I. Sap, F. Guibbal, C. Vriamont, C. Caygill, J. R. Atack, A. J. Watkins, C. Marshall, R. Hueting, C. Warnier, V. Gouverneur and M. Tredwell, EJNMMI Radiopharm. Chem., 2022, 7, 5 CrossRef PubMed.
- R. Schirrmacher, J. J. Bailey, A. V. Mossine, P. J. H. Scott, L. Kaiser, P. Bartenstein, S. Lidner, D. R. Kaplan, A. Kostikov, G. Fricker, A. Mahringer, P. Rosa-Neto, E. Schirrmacher, C. Wängler, B. Wängler, A. Thiel, J. P. Soucy and V. Bernard-Gauthier, et al., J. Med. Chem., 2018, 61, 1737–1743 CrossRef PubMed.
- (a) F. W. Friese and A. Studer, Chem. Sci., 2019, 10, 8503–8518 RSC; (b) M. Wang and Z. Shi, Chem. Rev., 2020, 120, 7348–7398 CrossRef CAS PubMed; (c) Z.-H. Shang, J. Pan, Z. Wang, Z.-X. Zhang and J. Wu, Eur. J. Org Chem., 2023, 26, e202201379 CrossRef CAS; (d) J. S Wright, P. J. H. Scott and P. G. Steel, Angew. Chem., Int. Ed., 2020, 60, 2796–2821 CrossRef PubMed.
- T. Kaur, X. Shao, M. Horikawa, L. S. Sharninghausen, S. Preshlock, A. F. Brooks, B. D. Henderson, R. A. Koeppe, A. F. DaSilva, M. S. Sanford and P. J. H. Scott, Org. Process Res. Dev., 2023, 27, 373–381 CrossRef CAS PubMed.
- N. Hadjipaschalis, S. Ortalli, Z. Chen, R. S. Paton, J. Ford, M. Tredwell and V. Gouverneur, Org. Lett., 2025, 27, 6545–6550 CrossRef CAS PubMed.
- J. S. Wright, L. S. Sharninghausen, S. Preshlock, A. F. Brooks, M. S. Sanford and P. J. H. Scott, J. Am. Chem. Soc., 2021, 143, 6915–6921 CrossRef CAS.
- W. Chen, H. Wang, N. E. S. Tay, V. Pistritto, K.-P. Li, T. Zhang, Z. Wu, D. A. Nicewicz and Z. Li, Nat. Chem., 2022, 14, 216–223 CrossRef CAS PubMed.
- T. E. Spiller, K. Donabauer, A. F. Brooks, J. A. Witek, G. D. Bowden, P. J. H. Scott and M. S. Sanford, Org. Lett., 2024, 36, 6433–6437 CrossRef PubMed.
- L. S. Sharninghausen, A. F. Brooks, W. P. Winton, K. J. Makaravage, P. J. H. Scott and M. S. Sanford, J. Am. Chem. Soc., 2020, 142, 7362–7367 CrossRef CAS.
- W. K. Chow, O. Y. Yuen, C. M. So, C. P. Lau, W. T. Wong and F. Y. Kwong, RSC Adv., 2012, 3, 12581–12539 Search PubMed.
- S. Barroso, M. Joksch, P. Puylaert, S. Tin, S. J. Bell, L. Donnellan, S. Duguid, C. Muir, P. Zhao, V. Farina, D. N. Tran and J. G. de Vries, J. Org. Chem., 2021, 86, 103–109 CrossRef CAS PubMed.
- T. Ishiyama, M. Murata and N. Miyaura, J. Org. Chem., 1995, 60, 7508–7510 CrossRef CAS.
- Unsuccessful substrates (giving <1% radiochemical yield) are summarized in SI page 25. In most cases, these are also unsuccessful starting from the pure Ar-BPin, indicating that the CMRF step is the problem..
- S. J. Lee, M. T. Morales-Colón, A. F. Brooks, J. S. Wright, K. J. Makaravage, P. J. H. Scott and M. S. Sanford, J. Org. Chem., 2021, 86, 14121–14130 CrossRef CAS PubMed.
- (a) L. Wang, N. Wang, W. Zhang, X. Cheng, Z. Yan, G. Shao, X. Wang, R. Wang and C. Fu, Signal Transduct. Targeted Ther., 2022, 7, 48 CrossRef CAS PubMed; (b) L. Costa, E. Sousa and C. Fernandes, Pharmaceuticals, 2023, 16, 996 CrossRef CAS PubMed; (c) X. Ji, A. L. Nielsen and C. Heinis, Angew Chem. Int. Ed. Engl., 2024, 63, e202308251 CrossRef CAS PubMed.
- (a) S. Richter and F. Wuest, Molecules, 2014, 19, 20536–20556 CrossRef PubMed; (b) I. M. Jackson, P. J. H. Scott and S. Thompson, Semin. Nucl. Med., 2017, 47, 493–523 CrossRef PubMed; (c) G. P. Nicolas, A. Morgenstern, M. Schottelius and M. Fani, J. Nucl. Med., 2019, 60, 167–171 CrossRef CAS PubMed; (d) A. J. Hall and M. B. Haskali, Aust. J. Chem., 2022, 75, 34–54 CrossRef CAS.
- (a) D. Van der Born, A. Pees, A. J. Poot, R. V. A. Orru, A. D. Windhorst and D. Vugts, Chem. Soc. Rev., 2017, 46, 4709–4773 RSC; (b) O. Morris, O. M. Fairclough, J. Grigg, C. Prenant and A. McMahonm, J. Labelled Compd. Radiopharm., 2019, 62, 4–23 CrossRef CAS PubMed; (c) C. Rangger and R. Haubner, Pharmaceuticals, 2020, 13, 22 CrossRef CAS PubMed.
- (a) J. Rickmeier and T. Ritter, Angew. Chem., Int. Ed., 2018, 57, 14207–14211 CrossRef CAS PubMed; (b) R. Halder, G. Ma, J. Rickmeier, J. W. McDaniel, R. Petzold, C. N. Neumann, J. M. Murphy and T. Ritter, Nat. Protoc., 2023, 18, 3614–3651 CrossRef CAS PubMed.
- K. Billingsley, T. E. Barder and S. L. Buchwald, Angew. Chem., Int. Ed., 2007, 46, 5359–5363 CrossRef CAS PubMed.
- The radiochemical yield for the automation is within the expected range for current standards for CMRF. (a) F. Guibbal, P. G. Isenegger, T. C. Wilson, A. Pacelli, D. Mahaut, J. B. I. Sap, N. J. Taylor, S. Verhoog, S. Preshlock, R. Hueting, B. Cornelissen and V. Gouverneur, Nat. Protoc., 2020, 15, 1525–1541 CrossRef CAS PubMed; (b) A. V. Mossine, A. F. Brooks, V. Bernard-Gauthier, J. J. Bailey, N. Ichiishi, R. Schirrmacher, M. S. Sanford and P. J. H. Scott, J. Labelled Compd. Radiopharm., 2018, 61, 228–236 CrossRef CAS PubMed; (c) J. S. Wright, L. S. Sharninghausen, S. Preshlock, A. F. Brooks, M. S. Sanford and P. J. H. Scott, J. Am. Chem. Soc., 2021, 143, 6915–6921 CrossRef CAS PubMed; (d) M. A. Nadporojskii, V. V. Orlovskaya, O. S. Fedorova, D. S. Sysoev and R. N Krasikova, Molecules, 2024, 29, 3342 CrossRef CAS PubMed; (e) K. J. Makaravage, A. F. Brooks, A. V. Mossine, M. S. Sanford and P. J. H. Scott, Org. Lett., 2016, 18, 5440–5443 CrossRef CAS PubMed; (f) S. J. Lee, M. T. Morales-Colon, A. F. Brooks, J. S. Wright, K. J. Makaravage, P. J. H. Scott and M. S. Sanford, J. Org. Chem., 2021, 86, 14121–14130 CrossRef CAS PubMed; (g) A. V. Mossine, S. S. Tanzey, A. F. Brooks, K. J. Makaravage, N. Ichiishi, J. M. Miller, B. D. Henderson, M. B. Skaddan, M. S. Sanford and P. J. H. Scott, Org. Biomol. Chem., 2019, 17, 8701–8705 RSC.
| This journal is © The Royal Society of Chemistry 2026 |