 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Mechanistic insights into azo compound back-isomerization from spin-flip time-dependent DFT combined with Marcus theory
Ari Serez ab,
Flavia Aleotti
ab,
Flavia Aleotti c,
Pascal Gerbaux
c,
Pascal Gerbaux b,
Luca Muccioli
b,
Luca Muccioli c and
Jérôme Cornil
c and
Jérôme Cornil *a
*a
aLaboratory for Chemistry of Novel Materials, University of Mons, Place du Parc 23, 7000 Mons, Belgium. E-mail: Jerome.Cornil@umons.ac.be
bOrganic Synthesis and Mass Spectrometry Laboratory, University of Mons, Place du Parc 23, 7000 Mons, Belgium
cDipartimento di Chimica Industriale “Toso Montanari”, University of Bologna, Via Gobetti 85, 40129 Bologna, Italy
First published on 27th April 2026
Abstract
Interest in photosensitive molecules has increased significantly over the past decade, with particular attention given to photoswitchable systems. Among these, azobenzene stands out as a reference compound due to its broad range of applications, in particular for solar energy storage. While the trans-to-cis photoisomerization has been relatively well characterized, the reverse cis-to-trans isomerization remains a complex process potentially involving multi-state physics. In this study, we compile recent theoretical advances aimed at modeling this process and introduce, through the spin-flip time-dependent density functional theory (SF-TDDFT) approach combined with the semi-classical Marcus equation, a fast and efficient method to investigate the mechanisms of thermal back-isomerization of azo derivatives. By comparing various exchange–correlation functionals with CASPT2 reference data, we demonstrate that the PBE0(D3BJ) functional provides an accurate description for the non-adiabatic rotational pathway. We successfully reproduce the experimental values (88.6 vs. 88.3 kJ mol−1 for the experimental enthalpy of activation, and −53.0 vs. −50.2 J mol−1 K−1 for the experimental entropy of activation) for azobenzene, thus motivating the extension of this methodology to other azo derivatives. This approach can be further generalized to a broader class of azo-based photo-switches in future studies.
1 Introduction
Modern society faces numerous energy-related challenges, especially in the transition from fossil fuels to renewable energy sources. To this end, many technologies have been developed, such as solar panels to produce electricity, and batteries to store it. To avoid inherent charge losses, an interesting new approach relies on the storage of energy in chemical structures. To do so, photo-switchable molecules that can isomerize by capturing a photon from the sun are used. A metastable isomer is then created, in which the solar energy is stored; this is referred to as a MOlecular Solar Thermal (MOST) system. This technology has been widely studied over the last decade.1–3 Among the various existing photochromic systems, azobenzene and derivatives appear very promising.1,4–6 Azobenzene can photo-isomerize from a trans (E) stable isomer to a cis (Z) metastable isomer, as shown in Fig. 1. These compounds offer many advantages, such as a low production cost, a high photo-isomerization quantum yield (44%), a large half-lifetime of the metastable form (about 4 days) and a decent storage energy density (0.20 MJ kg−1, or 36 kJ mol−1).7 Photo-isomerization, which can be considered the chemical analogue of the charging process in a battery, has been extensively studied.1,4,8 However, thermal back-isomerization, which can be considered the discharging process, has been largely overlooked. To the best of our knowledge, only a few studies have focused on this process.9–14 It is commonly accepted that the thermal back-isomerization can occur via two distinct mechanisms; the first involves the opening of the NNC angle, referred to as the inversion mechanism, while the second one proceeds via a rotation of a phenyl group around the double bond between the two nitrogen atoms and is referred to as the rotation mechanism (Fig. 1). | ||
| Fig. 1 Reaction pathways for the back isomerization mechanism. α (in blue) is the NNC bending angle involved in the inversion, and ω (in red) is the dihedral angle involved in the rotation. | ||
The minimum energy reaction path (MERP) was typically searched by DFT calculations, to describe the inversion mechanism.15 However, an inconsistency was pointed out recently: the activation entropy, Δ‡S, is calculated to be positive (about +10 J mol−1 K−1) at the DFT level against a negative experimental value for azobenzene (about −50.2 J mol−1 K−1).16,17 The experimental rate constant was well reproduced from a standard Eyring theory based on molecular parameters calculated at the DFT level, whereas Hartree-Fock and post-HF methods (such as MP2) were not able to provide the right order of magnitude.17 This discrepancy in the sign and amplitude of Δ‡S, known as the “entropy puzzle”, was studied recently in detail by Reimann and coworkers.10 Their highly correlated calculations suggest that the actual mechanism for the thermal back-isomerization of azobenzene is not an inversion but is rather based on a multi-state rotational process. Instead of a classical chemical reaction occurring on a single potential energy surface (PES), it was found that the lowest triplet state, T1, intersects with the ground state, S0 (Fig. 2). Estimating the rate constant thus requires accounting for a possible transition between these two states if the intersystem crossing is sufficiently fast (in other words, a non-adiabatic transition state description). The importance of the T1 state was supported experimentally by observing, in a few instances, an external heavy-atom effect on the kinetics of back-isomerization.10,13 Since the mixing between singlet and triplet states involves a spin–orbit coupling, adding heavy elements is expected to enhance this coupling, which is proportional to the fourth power of the nuclear charge.18
The electronic nature of this triplet state can be understood from a simple molecular orbital representation. During the rotation, the azo-π bond is broken since the 2pz orbitals of each nitrogen no longer overlap, which can promote a biradical form either in a singlet or triplet state. According to Hund's rule, the lowest triplet state will be lower in energy than the lowest singlet state at 90°, as presented in Fig. 2. A drawback of DFT is that it fails to locate a transition state for the rotation mechanism by systematically converging to the transition state associated with the inversion mechanism.10,13 A possibility to overcome this problem is to perform a constrained optimization by freezing the NNC angles.14 This discrepancy was explained by the fact that the ground state and the first singlet excited state S1 mix near 90°: this mixing can be quantitatively described only with multi-reference methods, or at least with a method that can capture the multi-configurational physics of the problem.10 A quasi-exact value for the activation entropy can thus be recovered by combining highly correlated methods with a formalism taking into account the amplitude of the spin orbit coupling at the crossing of the S0 and T1 states, such as the semi-classical Wentzel–Kramers–Brillouin (WBK) theory.10
In order to efficiently explore the vast chemical space of potential MOST candidates, there is a need to reduce the computational effort while maintaining a good level of accuracy. In the search for an alternative approach for the coupling of highly correlated methods to the WBK theory used in ref. 10, the present work aims to investigate the relevance of coupling the results of spin-flip(SF) TD-DFT calculations with the WKB theory. SF-TDDFT is currently emerging as a fast and accurate method to be used in the presence of a conical intersection or to deal with a ground state whose wave-function cannot be simply represented with a single determinant.19 A secondary aim is to assess whether the use of the semi-classical Marcus theory to describe the limiting kinetic step can provide rates similar to those obtained with WBK. It must be noted that Marcus theory has already been used to compute the rate of intersystem crossing processes by relying on three easily accessible molecular parameters (i.e., the reorganization energy, the spin–orbit coupling, and the free energy of the reaction). This approach will be first applied to the prototypical azobenzene molecule and will then be extended to other systems.
2 Theoretical background
We have first performed a benchmark of different DFT functionals to reproduce the potential energy curves of the S0 and T1 states taking CASPT2 results as the reference. We also employed the D3BJ dispersion correction, since DFT functionals usually do not correctly reproduce the inter/intramolecular van der Waals interactions.20 All the calculations were performed using Q-Chem 6.3 at the SF-TDDFT and DFT level and OpenMolcas 25.06 for the CASSCF/CASPT2 calculations. The spin-flip approach relies on the linear-response time-dependent DFT (LR-TDDFT) theory, which is well described by Casida's equation:
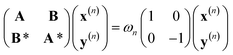 | (1) |
| Aiajb = δijδab(εa − εi) + (ia|fH|jb) + (ia|fxc|jb) | (2) |
| Biajb = (ia|fH|bj) + (ia|fxc|bj) | (3) |
Aiāj![[b with combining macron]](https://www.rsc.org/images/entities/i_char_0062_0304.gif) = δijδā = δijδā![[b with combining macron]](https://www.rsc.org/images/entities/i_char_0062_0304.gif) (εā − εi) − CHF(ij|fH|ā (εā − εi) − CHF(ij|fH|ā![[b with combining macron]](https://www.rsc.org/images/entities/i_char_0062_0304.gif) ) ) |
Biāj![[b with combining macron]](https://www.rsc.org/images/entities/i_char_0062_0304.gif) = −CHF(ib|fH|ā = −CHF(ib|fH|ā![[j with combining macron]](https://www.rsc.org/images/entities/i_char_006a_0304.gif) ) ) |
As mentioned above, it is necessary to go beyond the Eyring transition state theory (TST) and turn to a Non-Adiabatic TST (NA-TST); this formalism has already been applied to spin-forbidden reactions.21 If we consider two crossing potential energy surfaces associated with S0 and T1, as shown in Fig. 2, the hopping probability from S0 to T1 at the crossing point can be calculated from the Landau–Zener (LZ) theory as:21–24
| p = (1 − PLZ)(1 + PLZ) | (4) |
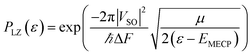 | (5) |
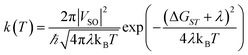 | (6) |
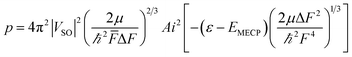 | (7) |
![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) is the average of the slopes. We can approximate this equation to:23
is the average of the slopes. We can approximate this equation to:23
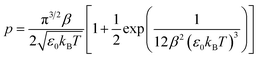 | (8) |
which is also known as the Wentzel–Kramers–Brillouin (WKB) approximation.10,23 All the parameters needed (µ,ΔF,
![[F with combining macron]](https://www.rsc.org/images/entities/i_char_0046_0304.gif) ) are calculated using the GLOWFreq suite.26 Assuming the S0 → T1 crossing as the rate-determining step, we can use p from eqn (8) as the transmission coefficient, γ(T) = p, in the Eyring equation:
) are calculated using the GLOWFreq suite.26 Assuming the S0 → T1 crossing as the rate-determining step, we can use p from eqn (8) as the transmission coefficient, γ(T) = p, in the Eyring equation:
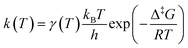 | (9) |
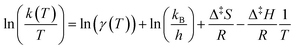 | (10) |
By plotting the left-hand side of eqn (10) with respect to 1/T, and neglecting the temperature dependence of γ(T), one obtains a straight line with slope −Δ‡H/R and intercept  . This is generally known as an Eyring plot, and it is often used in the analysis of experimental data, with the further assumption of γ = 1.
. This is generally known as an Eyring plot, and it is often used in the analysis of experimental data, with the further assumption of γ = 1.
However, as pointed out by Reimann et al.10 this latest assumption is no longer valid if spin-forbidden reactions are involved, for which γ ≪ 1. In this case, the apparent “measured” entropy Δ‡![[S with combining macron]](https://www.rsc.org/images/entities/i_char_0053_0304.gif) = Δ‡S + R
= Δ‡S + R![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) γ can acquire large negative values, even though the real entropy is probably small, as predicted by QM calculations.14,17 Concerning the non-adiabatic rotation mechanism, we computed Δ‡G, which corresponds to the free energy difference between the cis isomer and the MECP, and obtained Δ‡H and Δ‡S from an Eyring plot (eqn (10)). For the adiabatic rotation and inversion mechanisms, in the absence of intersystem crossing between the S0 and T1 states, Δ‡H and Δ‡S were directly obtained from frequency calculations at the cis and transition state geometries.
γ can acquire large negative values, even though the real entropy is probably small, as predicted by QM calculations.14,17 Concerning the non-adiabatic rotation mechanism, we computed Δ‡G, which corresponds to the free energy difference between the cis isomer and the MECP, and obtained Δ‡H and Δ‡S from an Eyring plot (eqn (10)). For the adiabatic rotation and inversion mechanisms, in the absence of intersystem crossing between the S0 and T1 states, Δ‡H and Δ‡S were directly obtained from frequency calculations at the cis and transition state geometries.
The spin–orbit coupling (SOC), VSO in eqn (6) and (8), is a relativistic interaction between the spin angular momentum and the orbital angular momentum of an electron. This interaction is described by the Breit–Pauli spin–orbit operator, as implemented in Q-Chem 6.3:18,27
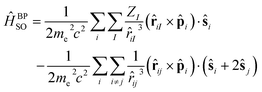 | (11) |
where i and j are the labels for electrons, ZI is the charge of the nucleus I, me is the electron mass, and c is the speed of light. The first term (eqn (11)) corresponds to the one-electron part, which is the interaction of the spin of the electron with its orbital angular momentum. The second term (eqn (11)) is the two-electron contribution which accounts for the electron–electron interaction. In Q-Chem 6.3, the two-electron part is treated via a mean-field approach.27,28
3 Results and discussion
The first step in the present work was to carefully validate a functional to be used with SF-TDDFT. It has been reported that BHHLYP is a good starting point within the spin-flip method, due to its high exchange contribution, which is important for this kind of calculation involving singlet and triplet excited states.29 Nevertheless, we decided to perform a comparison of different functionals in order to find the one that best reproduces the results of the complete active space perturbation theory (CASPT2) method, i.e., a highly-correlated multi-determinant approach taken here as the reference. We used the extended-multi state CASPT2 (XMS-CASPT2) formalism and the double zeta ANO-RCC-VDZP basis set with an active space (AS) of 10 electrons in 8 orbitals (Fig. 3).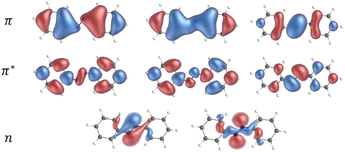 | ||
| Fig. 3 Orbitals involved in the active space for the CASPT2/ANO-RCC-VDZP calculations on trans azobenzene. | ||
The AS consists of two occupied lone-pair orbitals localized on the two nitrogen atoms and all the π orbitals except for the two most bonding and antibonding ones. The AS was selected following the procedure proposed by Aleotti et al.14 We performed a relaxed scan of the CNNC dihedral angle with steps of 10° for the ground state, and the triplet state energies were evaluated from single-point calculations at the ground state geometries. Our results are consistent with previously reported calculations.9,14 We repeated the same procedure for a set of 8 DFT functionals with the 6-311G(d,p) basis set, as shown in the SI (Fig. S1). Our approach, consisting of exploring different rungs of Jacob's ladder in DFT, (i.e., GGA, hybrid, range-separated, etc.) showed that PBE0(D3BJ)/6-311G(d,p) calculations are those that best reproduce the ground and triplet state curves provided by the CASPT2 calculations.
In Fig. 4 we show a comparison of the relative singlet and triplet state energies as a function of the dihedral angle, for two methods, with respect to the singlet state reference energies arbitrarily set as those predicted at 180°. The agreement for the triplet state is excellent at any angle, while for S0 there is a general overestimation of the energy along the pathway in the S0 state at the PBE0 level, compared to the CASPT2 results. We attribute this overestimation in part to the lack of dynamic correlation in DFT methods. Not surprisingly given the choice of our reference energies, for dihedral angles close to 180°, we observe that the energy difference between CASPT2 and SF-TDDFT curves is very small, with a mean value of c.a. 1.5 kJ mol−1 for angles ranging from 130° to 180°. Instead, the discrepancy is larger around the cis form: for angles between 10° and 60°, the deviation is about 10 kJ mol−1 while for angles between 70–110°, the deviation amounts to 18 kJ mol−1. This large deviation probably occurs because, at these angles, the two phenyl rings are very close to each other, a situation where electron correlation is expected to be more important. To further validate this hypothesis, we examined how the ground state energy curve is affected by the perturbation theory treatment by comparing CASSCF versus CASPT2 results; in CASPT2 calculations, the dynamic correlation is recovered by perturbation theory, exactly in the same way as in the Møller–Plesset approach at the second order (MP2 – perturbation theory applied to a HF single-reference wavefunction). Notably, the correlation energy accounts for only 0.33% of the total energy, which is consistent with the validity of a perturbative treatment, where the perturbation is expected to remain small. Fig. 5 clearly shows that the PBE0(D3BJ) potential energy curve lies between the CASSCF and CASPT2 curves, indicating that the recovery of dynamic electron correlation plays a significant role. In purple, we report the correlation energy with respect to the trans isomer, for which the correlation energy is minimal: the curve peaks at 90°, showing that dynamic correlation is most significant at this dihedral angle, reaching a value of 50 kJ mol−1, which is not negligible.
We have discussed so far the rotation mechanism, which is commonly assumed to be the minimum-energy reaction path. For completeness, we also analyzed the energy profile associated with the inversion mechanism. From Fig. 6, it is clear that standard DFT and the spin-flip approach give very similar results, which is in agreement with the hypothesis that the inversion path can be described with a single-reference method. Using the Møller–Plesset second-order perturbation theory (MP2) as the reference method, our DFT approach underestimates the activation barrier mostly near the transition state. However, CASPT2 results appear to be in better agreement with DFT (see Table 1).
 | ||
| Fig. 6 Pathway of the Z → E inversion mechanism following the bending angle NNC at different levels of theory. The basis set used for the MP2 calculations is cc-pVTZ. | ||
| Non-adiabatic rotation | Rotation | Inversion | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| WKB (eqn (8)) | Marcus (eqn (6)) | Adapted Marcus (eqn (13)) | Eyring (eqn (9)) | Eyring (eqn (9)) | ||||||
| Δ‡H | Δ‡![[S with combining macron]](https://www.rsc.org/images/entities/i_char_0053_0304.gif) |
Δ‡H | Δ‡![[S with combining macron]](https://www.rsc.org/images/entities/i_char_0053_0304.gif) |
Δ‡H | Δ‡![[S with combining macron]](https://www.rsc.org/images/entities/i_char_0053_0304.gif) |
Δ‡H | Δ‡S | Δ‡H | Δ‡S | |
| CASPT2 | 78.9 | −39 | 70 | −31 | 82.0 | −53 | 100.5 | −13.5 | 126.7 | 2.1 |
| PBE0(D3BJ) | 90.2 | −32 | 59.3 | −45 | 88.3 | −53 | 124.2 | −27 | 119.2 | 4.9 |
| MP2 | — | — | — | — | — | — | — | — | 137.1 | 16.9 |
| k (298 K) | 4.9 × 10−5 s−1 | 9.8 × 10−1 s−1 | 4.4 × 10−6 s−1 | 4.0 × 10−11 s−1 | 1.4 × 10−8 s−1 | |||||
| γ (298 K) | 4.98 × 10−2 | 1.5 × 10−2 | 8.35 × 10−3 | 1 | 1 | |||||
| Experimental16 | 88.6 | −50.2 | k = 8.5 × 10−6 s−1 | |||||||
| t.b.e10 | 95 ± 4 | −38 ± 2 | — | — | — | — | 118 ± 7 | −8 | 124 ± 3 | 11 |
The next step is to determine the activation parameters, i.e. Δ‡H and Δ‡S, of the back-isomerization process. There are thus three possible paths: the inversion mechanism, the purely ground state rotation mechanism, and the rotation mechanism combined with a spin-forbidden transition into the T1 state. The latter was referred to as non-adiabatic rotation in a recent article.30 Regarding the non-adiabatic rotation, using the WKB approximation (eqn (8)) fed by the results of highly correlated CCSD calculations, Reimann et al. found theoretical values in really good quantitative agreement with the experiment;10 however, CCSD is still a single-reference method, which implies that it will suffer from the same cusp problem as normal DFT (lack of strong correlation) when the dihedral angle is around 90°. We also employed eqn (8) and found a nice match between the values in ref. 10 and ours (see Table 1) when injecting parameters obtained at the spin-flip PBE0(D3BJ) level, and in particular a negative value for the entropy of activation (−32 J mol−1 K−1), strengthening the idea that its sign can be associated with a spin-forbidden process.21 The larger the spin–orbit coupling, the more pronounced these spin-forbidden effects become; azobenzene exhibits a spin–orbit coupling of approximately 15 cm−1, which is relatively large compared to typical organic conjugated systems.31 This coupling was calculated at the MECP geometry. Regarding the adiabatic rotation and inversion pathways, we evaluated the activation enthalpy and entropy based on frequency calculations by determining the partition function32 in the ground and transition state geometry. From the comparison of WKB enthalpies, entropies and rates with experimental ones (Table 1), it is clear that this approach performs well in describing the non-adiabatic rotational mechanism. Indeed, it yields the correct order of magnitude for the rate constant (order of 10−5 s−1) and provides activation parameters that are consistent with experimental data as well as with other theoretical methods. However, when the Marcus equation, as formulated in eqn (6), is applied (Table 1), although the activation entropy is well reproduced, it shows a significant discrepancy in the activation enthalpy (59.3 vs. 90.2 kJ mol−1). This inaccuracy originates from the ΔGST term: a more detailed analysis of the two key parameters in the Marcus equation – namely the reorganization energy λ and ΔGST, the free-energy difference between the triplet and singlet minima—reveals that the reorganization energy is in good agreement with the CASPT2 calculations. In contrast, the energy of the cis isomer is overestimated at the DFT level, leading to an incorrect estimation of ΔGST.
In the Marcus equation, ΔGST and λ are employed to estimate the transition state energy without actually measuring or calculating it directly with quantum chemistry methods:
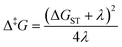 | (12) |
This formula further assumes that both potential energy surfaces are perfect parabolas with the same curvature, which is not necessarily the case. We have here adopted an alternative formulation of eqn (6):
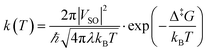 | (13) |
in which, while retaining the Marcus pre-exponential factor, featuring the spin–orbit coupling, VSO, we replaced the exponential term in eqn (6) with that used in the Eyring equation (eqn (9)). Δ‡G is estimated from energies almost equally overestimated at the DFT level. This modified expression is employed in Table 1 to compute the kinetic and activation parameters. It performs remarkably well, yielding activation parameters that are very close to the experimental values (88.3 vs. 88.6 kJ mol−1 for the enthalpy and −53 vs. −50.2 J mol−1 K−1 for the entropy) and to those predicted by WKB theory (90.2 kJ mol−1 and −32 J mol−1 K−1) at a much higher computational cost. Eqn (13) therefore offers a faster, more efficient, and simpler route to determine the rate constant (and the associated activation parameters) for the non-adiabatic rotational mechanism.
We have also assessed the influence of the S0/S1 mixing in the calculations. To do so, we performed single-point energy calculations with DFT and unrestricted (Broken-Symmetry, BS) DFT at geometries obtained from a SF-TDDFT scan of the singlet ground state. As depicted in Fig. 7, there is a perfect match of all three methods close to the cis and trans minima (green area) and a significant deviation (about 44 kJ mol−1 for DFT and 27 kJ mol−1 for UDFT) only in the TS region (red area). This demonstrates the importance of the SF-TDDFT to take into account the multi-configurational nature of the transition state, which in this case is important due to the strong mixing between the S0 and S1 states. Regardless, it is worth stressing that both MECPs between the S0 and T1 states are located in the green area of Fig. 7, which indicates that one could use single reference methods, such as classical DFT, to calculate the parameters needed for evaluating the rate constant via the WKB or Marcus equations. With the latter approach, we can derive (see the SI) an expression for the transmission coefficient used in the Eyring equation,
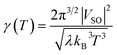 | (14) |
Finally, as a proof of concept, we have applied the proposed methodology to two other azo derivatives: para and meta methoxy-azothiophene (pATh and mATh, respectively), as presented in Fig. 8. These two heterocyclic azo compounds are of particular interest because they include a thiophene moiety that can be involved in a weak intramolecular interaction between the sulfur atom and the phenyl group on the opposite side of the azo bond, thereby stabilizing the cis isomer. These systems were recently described by Martins et al.30 who found, based on CASPT2 calculations, that these two systems will back-isomerize via a mix of non-adiabatic and adiabatic rotations since the inversion pathway is too high in energy. We therefore tracked the inversion and rotation pathways and reported the calculated activation parameters in Table 2; they are found to be in good agreement with the results in ref. 30. Indeed, based on Table 2, the spin-flip PBE0(D3BJ) reproduces well the CASPT2 results for the non-adiabatic and adiabatic rotations. However, there is a non-negligible difference for the inversion mechanism (about 10 kJ mol−1), which can compete as well in our case; note that our calculated activation entropies for the inversion mechanism are closer to the experimental ones. Regarding the inversion terms, we observed that this mechanism preferentially occurs on the side of the azo double bond bearing the thiophene substituent (see the SI); however we feel more cautious about the individuation of the actually prevailing mechanism, since all enthalpies terms are close, but still the non-adiabatic rotation appears to be the minimum-energy path. It should also be kept in mind that if the mechanisms exhibit similar Δ‡G values, the adiabatic pathways will always be the fastest. One should also consider that the experimental values were obtained in acetonitrile, which is a polar solvent that might thus strongly influence the kinetics of the back-isomerization process. A more expensive approach (e.g. QMMM) could provide further information on how solvents affect the mechanisms. We also suggest an experimental investigation of the heavy-atom effect to obtain information on whether a triplet state is involved or not in the actual mechanism.
 | ||
| Fig. 8 Chemical structures of meta-methoxy-azothiophene (mAth, left) and of para-methoxy-azothiophene (pAth, right). | ||
| Non-adiabatic rotation | Rotation | Inversion | ||||||
|---|---|---|---|---|---|---|---|---|
| WKB (eqn (8)) | Marcus (eqn (13)) | Eyring (eqn (9)) | Eyring (eqn (9)) | |||||
| Δ‡H | Δ‡![[S with combining macron]](https://www.rsc.org/images/entities/i_char_0053_0304.gif) |
Δ‡H | Δ‡![[S with combining macron]](https://www.rsc.org/images/entities/i_char_0053_0304.gif) |
Δ‡H | Δ‡S | Δ‡H | Δ‡S | |
| CASPT2 (pATh)30 | 92.1 | −39.7 | — | — | 105.3 | −0.6 | 135.1 | −0.2 |
| CASPT2 (mATh)30 | 97.2 | −43.2 | — | — | 111.8 | 0.2 | 138.1 | −0.1 |
| SF (pATh) | 90.8 | −32.0 | 88.4 | −56.2 | 115.5 | −4.9 | 100.0 | −7.5 |
| SF (mATh) | 95.0 | −31.0 | 92.4 | −55.5 | 123.5 | 17.32 | 102.2 | −2.9 |
| k(298 K, pATh) | 1.4 × 10−5 | 2.38 × 10−6 | 1.9 × 10−8 | 7.3 × 10−6 | ||||
| k(298 K, mATh) | 2.9 × 10−6 | 6.35 × 10−7 | 1.1 × 10−8 | 5.2 × 10−6 | ||||
| γ(298 K, pATh) | 3.5 × 10−2 | 5.8 × 10−3 | 1 | 1 | ||||
| γ(298 K, mATh) | 3.8 × 10−2 | 6.3 × 10−3 | 1 | 1 | ||||
| Exp.33 (pATh) | 86.6 | −12.5 | k = 3.9 × 10−6 | |||||
| Exp.33 (mATh) | 96.2 | −12.5 | k = 3.2 × 10−7 | |||||
4 Conclusions
The thermal back-isomerization of cis azobenzene is more complex than it first appears, since, although the inversion mechanism is well described using conventional DFT, the rotation mechanism exhibits pronounced multiconfigurational character at around 90°, where the S0 and S1 states strongly mix. Moreover, the emergence of a biradical character might lead to the involvement of a triplet state along the process. We found that the PBE0 functional with the D3BJ empirical dispersion and the 6-311G(d,p) basis set can reproduce highly correlated CASPT2 results by employing the SF-TDDFT formalism. This technique appears to be a powerful tool, as it reproduces highly correlated results in a shorter time. Subsequently, we showed that the Marcus equation can be used by considering the exponential term as described in the Eyring equation, which accounts for the activation energy (in our case, the energy required to reach the MECP), and retaining the actual pre-exponential factor of the Marcus equation. We obtain very accurate activation parameters (88.3 kJ mol−1 and −53 J mol−1 K−1), in excellent agreement with experimental data (88.6 kJ mol−1 and −50.2 J mol−1 K−1), which represents a major advance for the description of reaction mechanisms in azo compounds. Finally, we reproduced activation parameters for azothiophene derivatives as a proof of concept. This work is the first step towards exploring larger systems than azobenzene for the future, in order to understand how the molecular structure impacts the back isomerization kinetics.Author contributions
Ari Serez: writing – original draft, validation, methodology, investigation, conceptualization. Flavia Aleotti: methodology, writing – review & editing. Pascal Gerbaux: writing – review & editing, supervision, project administration, funding acquisition. Luca Muccioli: writing – review & editing, methodology. Jérôme Cornil: conceptualization, writing – review & editing, methodology, supervision, project administration, funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this study are available from the corresponding author upon request.Supplementary information (SI): Benchmark of SF-TDDFT functionals, transmission coefficients derived from Marcus theory, details on Marcus–Levich–Jortner and mechanistic details on azothiophene derivatives. See DOI: https://doi.org/10.1039/d6sc01578f.
Acknowledgements
A. S. was supported by the Action de Recherche Concertée (ARC) program funded by UMons on “MOlecular Solar Thermal systems: from molecules to energy capture/exchange prototypes”. The work in Mons has also been supported by the Fund for Scientific Research (FRS) of FNRS within the Consortium des Equipements de Calcul Intensif (CECI) under grant 2.5020.11 and by the Walloon Region (LUCIA Tier-1 supercomputer) under grant 1117545. J. C. is an FNRS research director. F. A. and L. M. acknowledge funding awarded by the International Foundation Big Data and Artificial Intelligence for Human Development (IFAB) to the project “Getting the MOST out of Sun” (GEM).Notes and references
- H. M. Bandara and S. C. Burdette, Chem. Soc. Rev., 2012, 41, 1809–1825 RSC.
- K. Börjesson, A. Lennartson and K. Moth-Poulsen, ACS Sustain. Chem. Eng., 2013, 1, 585–590 CrossRef.
- W. Sun, Z. Shangguan, X. Zhang, T. Dang, Z. Y. Zhang and T. Li, ChemSusChem, 2023, 16, 1–8 Search PubMed.
- Z. Wang, P. Erhart, T. Li, Z. Y. Zhang, D. Sampedro, Z. Hu, H. A. Wegner, O. Brummel, J. Libuda, M. B. Nielsen and K. Moth-Poulsen, Joule, 2021, 5, 3116–3136 CrossRef CAS.
- K. Moth-Poulsen, D. Ćoso, K. Börjesson, N. Vinokurov, S. K. Meier, A. Majumdar, K. P. C. Vollhardt and R. A. Segalman, Energy Environ. Sci., 2012, 5, 8534–8537 RSC.
- L. Dong, Y. Feng, L. Wang and W. Feng, Chem. Soc. Rev., 2018, 47, 7339–7368 RSC.
- Z. Wang, H. Hölzel and K. Moth-Poulsen, Chem. Soc. Rev., 2022, 51, 7313–7326 RSC.
- Z. Zhang, W. Wang, M. O'Hagan, J. Dai, J. Zhang and H. Tian, Angew. Chem., Int. Ed., 2022, 61, 1–24 Search PubMed.
- A. Cembran, F. Bernardi, M. Garavelli, L. Gagliardi and G. Orlandi, J. Am. Chem. Soc., 2004, 126, 3234–3243 CrossRef CAS PubMed.
- M. Reimann, E. Teichmann, S. Hecht and M. Kaupp, J. Phys. Chem. Lett., 2022, 13, 10882–10888 CrossRef CAS PubMed.
- S. Axelrod, E. Shakhnovich and R. Gómez-Bombarelli, ACS Cent. Sci., 2023, 9, 166–176 CrossRef CAS PubMed.
- J. Calbo, C. E. Weston, A. J. White, H. S. Rzepa, J. Contreras-García and M. J. Fuchter, J. Am. Chem. Soc., 2017, 139, 1261–1274 CrossRef CAS PubMed.
- T. Robert, G. Henrard, B. Tassignon, A. Serez, J. De Winter, P. Dugourd, J. Cornil, F. Chirot and P. Gerbaux, Anal. Chem., 2025, 97, 9405–9413 CrossRef CAS PubMed.
- F. Aleotti, L. Soprani, L. F. Rodríguez-Almeida, F. Calcagno, F. Loprete, I. Rivalta, S. Orlandi, E. Canè, M. Garavelli, I. Conti and L. Muccioli, Molecular Systems Design and Engineering, 2024 Search PubMed.
- C. R. Crecca and A. E. Roitberg, J. Phys. Chem. A, 2006, 110, 8188–8203 CrossRef CAS PubMed.
- T. Asano, T. Okada, S. Shinkai, K. Shigemats, K. Yumiko and O. Manabe, J. Am. Chem. Soc., 1981, 103, 5161–5165 CrossRef CAS.
- C. Rietze, E. Titov, S. Lindner and P. Saalfrank, J. Phys. Condens. Matter, 2017, 29, 314002–314024 CrossRef PubMed.
- C. M. Marian, Wiley Interdiscip. Rev.: Comput. Mol. Sci., 2012, 2, 187–203 CAS.
- D. Casanova and A. I. Krylov, Phys. Chem. Chem. Phys., 2020, 22, 4326–4342 RSC.
- S. Tsuzuki and T. Uchimaru, Phys. Chem. Chem. Phys., 2020, 22, 22508–22519 RSC.
- J. N. Harvey, Phys. Chem. Chem. Phys., 2007, 9, 331–343 RSC.
- E. J. Piechota and G. J. Meyer, J. Chem. Educ., 2019, 96, 2450–2466 Search PubMed.
- S. Liu, S. Srinivasan, J. Tao, M. C. Grady, M. Soroush and A. M. Rappe, J. Phys. Chem. A, 2014, 118, 9310–9318 CrossRef CAS PubMed.
- A. O. Lykhin, D. S. Kaliakin, G. E. Depolo, A. A. Kuzubov and S. A. Varganov, Int. J. Quantum Chem., 2016, 116, 750–761 CrossRef CAS.
- G. F. Bertsch, Derivations of Marcus's formula, https://archive.int.washington.edu/users/bertsch/marcus.1.pdf.
- K. L. Gannon, M. A. Blitz, C. H. Liang, M. J. Pilling, P. W. Seakins, D. R. Glowacki and J. N. Harvey, Faraday Discuss., 2010, 147, 173–188 RSC.
- S. Kotaru, P. Pokhilko and A. I. Krylov, J. Chem. Phys., 2022, 157, 224110 CrossRef CAS PubMed.
- Y. Shao, Z. Gan, E. Epifanovsky, A. T. Gilbert, M. Wormit, J. Kussmann, A. W. Lange, A. Behn, J. Deng, X. Feng, D. Ghosh, M. Goldey, P. R. Horn, L. D. Jacobson, I. Kaliman, R. Z. Khaliullin, T. Kus, A. Landau, J. Liu, E. I. Proynov, Y. M. Rhee, R. M. Richard, M. A. Rohrdanz, R. P. Steele, E. J. Sundstrom, H. L. Woodcock, P. M. Zimmerman, D. Zuev, B. Albrecht, E. Alguire, B. Austin, G. J. Beran, Y. A. Bernard, E. Berquist, K. Brandhorst, K. B. Bravaya, S. T. Brown, D. Casanova, C. M. Chang, Y. Chen, S. H. Chien, K. D. Closser, D. L. Crittenden, M. Diedenhofen, R. A. Distasio, H. Do, A. D. Dutoi, R. G. Edgar, S. Fatehi, L. Fusti-Molnar, A. Ghysels, A. Golubeva-Zadorozhnaya, J. Gomes, M. W. Hanson-Heine, P. H. Harbach, A. W. Hauser, E. G. Hohenstein, Z. C. Holden, T. C. Jagau, H. Ji, B. Kaduk, K. Khistyaev, J. Kim, J. Kim, R. A. King, P. Klunzinger, D. Kosenkov, T. Kowalczyk, C. M. Krauter, K. U. Lao, A. D. Laurent, K. V. Lawler, S. V. Levchenko, C. Y. Lin, F. Liu, E. Livshits, R. C. Lochan, A. Luenser, P. Manohar, S. F. Manzer, S. P. Mao, N. Mardirossian, A. V. Marenich, S. A. Maurer, N. J. Mayhall, E. Neuscamman, C. M. Oana, R. Olivares-Amaya, D. P. Oneill, J. A. Parkhill, T. M. Perrine, R. Peverati, A. Prociuk, D. R. Rehn, E. Rosta, N. J. Russ, S. M. Sharada, S. Sharma, D. W. Small, A. Sodt, T. Stein, D. Stück, Y. C. Su, A. J. Thom, T. Tsuchimochi, V. Vanovschi, L. Vogt, O. Vydrov, T. Wang, M. A. Watson, J. Wenzel, A. White, C. F. Williams, J. Yang, S. Yeganeh, S. R. Yost, Z. Q. You, I. Y. Zhang, X. Zhang, Y. Zhao, B. R. Brooks, G. K. Chan, D. M. Chipman, C. J. Cramer, W. A. Goddard, M. S. Gordon, W. J. Hehre, A. Klamt, H. F. Schaefer, M. W. Schmidt, C. D. Sherrill, D. G. Truhlar, A. Warshel, X. Xu, A. Aspuru-Guzik, R. Baer, A. T. Bell, N. A. Besley, J. D. Chai, A. Dreuw, B. D. Dunietz, T. R. Furlani, S. R. Gwaltney, C. P. Hsu, Y. Jung, J. Kong, D. S. Lambrecht, W. Liang, C. Ochsenfeld, V. A. Rassolov, L. V. Slipchenko, J. E. Subotnik, T. Van Voorhis, J. M. Herbert, A. I. Krylov, P. M. Gill and M. Head-Gordon, Mol. Phys., 2015, 113, 184–215 CrossRef CAS.
- M. Huix-Rotllant, B. Natarajan, A. Ipatov, C. Muhavini Wawire, T. Deutsch and M. E. Casida, Phys. Chem. Chem. Phys., 2010, 12, 12811–12825 RSC.
- F. A. Martins, R. V. Viesser, J. V. Schober, R. Herges and J. I. Wu, J. Am. Chem. Soc., 2025, 147(39), 35493–35500 CrossRef CAS PubMed.
- D. Beljonne, Z. Shuai, G. Pourtois and J. L. Bredas, J. Phys. Chem. A, 2001, 105, 3899–3907 CrossRef CAS.
- C. J. Cramer, Essentials of Computational Chemistry Theories and Models Second Edition, Wiley, 2nd edn, 2004, pp. 1–624 Search PubMed.
- A. H. Heindl and H. A. Wegner, Chem.–Eur. J., 2020, 26, 13730–13737 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |





