Open Access Article
Open Access ArticleZrO2-induced d–d spatial coordination effects of Ru single-atom catalysts to boost the oxygen evolution reaction
Dandan Yua,
Bin Liua,
Dongming Lia,
Junkai Yua,
Xijie Lana,
Shuhong Liua,
Zhuxin Lib,
Pengyun Gaob,
Yong Zhang*a and
Hong Zhao *b
*b
aSchool of Materials Science and Engineering, Dalian Jiaotong University, Dalian, 116028, China. E-mail: zhangyong0411@126.com
bCollege of Material Science and Engineering, Sichuan University of Science and Engineering, Zigong, 643000, China. E-mail: zhaohong@suse.edu.cn
First published on 8th April 2026
Abstract
Proton exchange membrane water electrolysis (PEMWE) is a leading strategy for producing green hydrogen from intermittent renewable energy. However, the high anodic energy barrier of the oxygen evolution reaction (OER), in a strongly acidic and highly oxidative operating environment, poses severe challenges for catalyst design. Herein, based on a unique d–d spatial coordination design, tetragonal zirconia-supported Ru single-atom catalysts (Ru-RSZ) were synthesized, which exhibited an overpotential as low as 208 mV and a mass activity of 1452.61 A gRu−1 @ 1.51 V. Remarkably, these catalysts were stably operated for 500 h. X-ray absorption spectroscopy (XAS) and high-angle annular dark-field (HAADF) imaging demonstrated that Ru is atomically dispersed at Zr lattice sites in ZrO2, and the point-group symmetry of the Ru atom changes from D2h in RuO2 to D2d. Density functional theory (DFT) calculations elucidated that the d-band center of Ru atoms was lowered by d–d spatial coordination effects, which weakened the strong adsorption of oxygen at Ru sites, eventually reducing the activation barrier of the OER rate-determining step. This work paves a new avenue to design high-efficiency and stable catalysts for hydrogen production.
Introduction
Against the backdrop of advancing green, low-carbon development and the global energy transition, hydrogen production from water electrolysis has emerged as a cornerstone technology for building the future energy system.1–4 Proton exchange membrane water electrolysis (PEMWE) has garnered significant attention due to its higher current density, rapid kinetics and higher stack efficiency (70–77%) in contrast to alkaline water electrolysis.5,6 However, the oxygen evolution reaction (OER) of PEMWE systems involves complex multi-step proton-coupled electron transfer processes and demonstrates sluggish kinetics, which significantly limit the overall efficiency of water electrolysis.7,8 Furthermore, protonation and solvation effects in acidic media pose substantial challenges to the stability of anode catalysts.9 Currently, only iridium-based oxides are widely used as OER catalysts, but the disadvantages of high-cost and scarcity severely limit their large-scale applications.10,11 To address these challenges, extensive research has focused on finding alternative catalysts, with cost-effective (10 times cheaper than iridium)12 and Earth-abundant13 ruthenium (Ru) emerging as a promising iridium substitute owing to its comparable catalytic activity.14–16 Nevertheless, the durability of ruthenium oxides remains limited and insufficient to meet the long-term operational requirements of commercial-scale PEMWE systems.17 Moreover, as a noble metal, ruthenium still entails substantial cost.18 To satisfy the commercial demands of PEMWE for industrial hydrogen production, it is imperative to develop low-cost Ru electrocatalysts with high activity and long-term stability.Nowadays, the development of Ru-based electrocatalysts primarily encompasses doping and defect engineering, interface engineering, hybridized orbital engineering, etc.16,19,20 Among these, orbital coupling between RuO2 and heteroatom dopants has demonstrated pronounced enhancement and stabilization of active sites.21 For example, Pb doping produces Pb–O–Ru 6s–2p–4d orbital hybridization that increases the energy required to remove lattice oxygen, thereby suppressing excessive oxidation of Ru.22 Similarly, unique lanthanide dopants induce f–p–d gradient orbital coupling that can modify adsorption energies of reaction intermediates and inhibit involvement of lattice oxygen, thus preventing over-oxidation of Ru.23 However, Ru–M orbital coupling mediated by O bridges is weakened by the presence of O 2p orbitals. Moreover, studies of dopant-induced orbital coupling based on the rutile structure have frequently neglected the strong influence of local atomic coordination, although some studies have compared the OER performance of Ru located at sites of different symmetries in other structures such as pyrochlore24 and perovskites.25,26 Investigations specifically addressing how crystal-structure-driven differences alter orbital coupling remain scarce. More importantly, substantial amounts of Ru catalysts are still required in doped RuO2 systems, which does not satisfy the low-cost requirement of industrial hydrogen production.
In this work, an acid-resistant oxide, ZrO2, was adopted as the support to provide Ru atoms with a distinctive atomic coordination geometry (P42/nmc).27 A Ru single-atom catalyst supported on ZrO2 (Ru-RSZ) was synthesized via a hydrothermal method. X-ray diffraction (XRD) and Raman characterization indicate that introduction of Ru stabilizes the tetragonal phase of ZrO2. Further high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM) and X-ray absorption spectroscopy (XAS) analyses reveal that Ru occupies Zr lattice sites, with site symmetry transformed from D2h in RuO2 to D2d in ZrO2. Density functional theory (DFT) calculations reveal that the unique d–d interactions in Ru-RSZ furnish direct bonding, substantially lowering the Ru 4d-band center, suppressing strong adsorption of oxygen intermediates on Ru, reducing reaction barriers, and concurrently weakening Ru–O covalency to inhibit participation of lattice oxygen, thereby enhancing structural stability. The synthesized Ru-RSZ catalyst exhibits an overpotential reduced to 208 mV at a Ru loading of only 43.9 µgRu cm−2, and chronopotentiometry at a current density of 10 mA cm−2 shows that the catalyst remains stable for 500 h. Moreover, oxygen vacancies present in Ru-RSZ further reinforce the d–d interactions of Ru–Zr, contributing to the catalyst's activity and stability. This work provides an effective strategy for developing high-efficiency and low-cost electrocatalysts for industrial hydrogen production.
Results and discussion
Material design and theoretical calculation
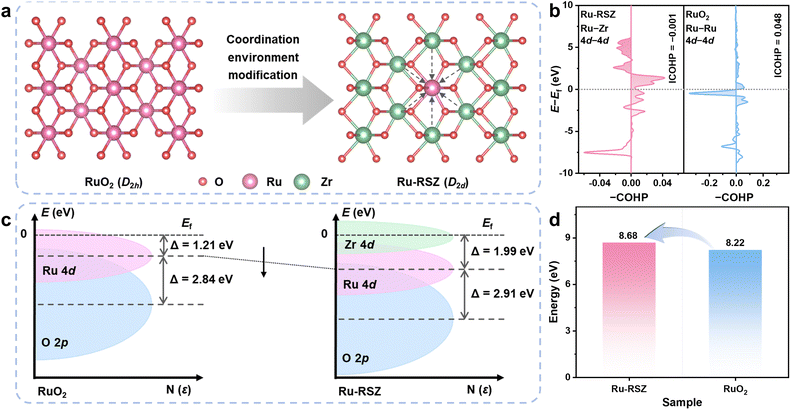
To validate the feasibility of the design strategy, DFT calculations were conducted to systematically compare the electronic structure and stability of the ZrO2-supported Ru catalyst (denoted as Ru-RSZ) and commercial RuO2 (Fig. 1a). Quantitative analysis of the intermetallic interactions provides direct evidence for the support stabilization effect: the integrated crystal orbital Hamilton population (ICOHP) for the Ru–Zr pair in Ru-RSZ is −0.001 eV, indicating a weak net bonding interaction.15 In contrast, the ICOHP for the Ru–Ru pair in RuO2 is +0.048 eV revealing a net antibonding interaction. Similarly, when Zr occupies the Ru sites in RuO2 with D2h symmetry, a net antibonding interaction (ICOHP = +0.021 eV) is also observed (Fig. S1). The direct d–d interaction between Zr and Ru not only reinforces the lattice framework but also markedly alters the 4d orbital distribution at Ru sites (Fig. 1b). Projected density of states (PDOS) and electronic band structure analyses (Fig. 1c and S2) reveal that the d-band center of the Ru 4d orbitals in Ru-RSZ shifts downward and farther from the Fermi level compared to that in com-RuO2.22 This modulation in the electronic structure suggests a weakened adsorption strength for oxygen evolution reaction intermediates, which is anticipated to mitigate the catalytic activity limitation inherent to conventional RuO2 caused by the over-binding of intermediates, thereby enhancing the intrinsic activity.28 Furthermore, the altered coordination environment induces a further separation between the Ru 4d and O 2p bands, characterized by a downward shift of both band centers, resulting in an enlarged characteristic band gap (εd–p) of 2.91 eV for Ru-RSZ compared to 2.84 eV for RuO2. This enlarged band gap indicates a reduced degree of covalent hybridization in the Ru–O bonds within Ru-RSZ, an electronic feature that contributes to suppressing the over-oxidation and dissolution of ruthenium under operational conditions, thereby enhancing the electrochemical stability of the catalyst.29 The obviously increased Ru vacancy formation energy (Fig. 1d) in Ru-RSZ further corroborates, from a thermodynamic perspective, the superior stabilization effect exerted by the ZrO2 support on the ruthenium active sites.20,30Material characterization and the coordination environment
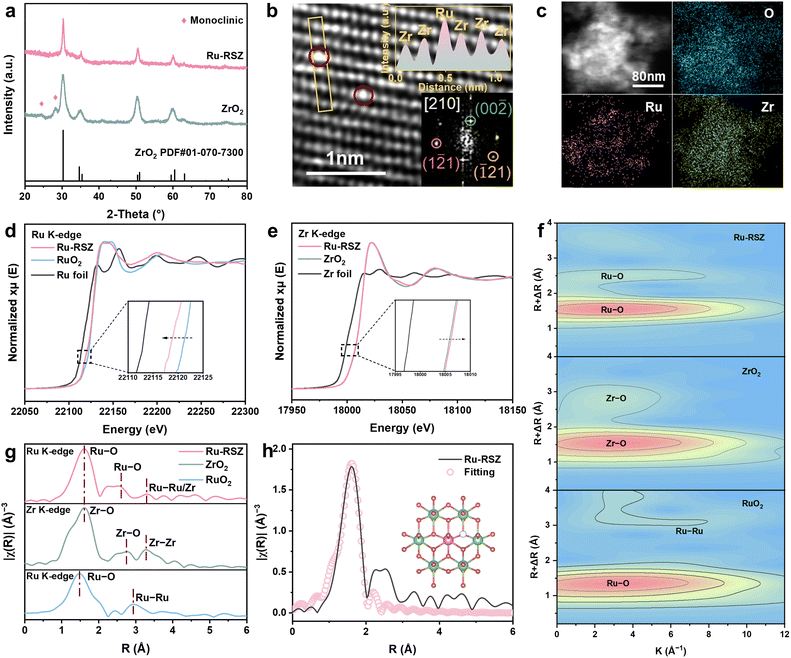
Ru-RSZ was synthesized via the co-precipitation method (Fig. S3). The resulting precipitate was then subjected to hydrothermal treatment at 200 °C for 12 hours, followed by centrifugation and drying to obtain Ru-RSZ powder. The XRD pattern confirms the presence of a tetragonal ZrO2 phase (JCPDS 01-070-7300) in Ru-RSZ (Fig. 2a). No detectable peaks corresponding to Ru were observed, indicating that the lattice structure of ZrO2 remains intact after Ru introduction (Fig. S4).31 Notably, the XRD pattern of Ru-RSZ shifts slightly to higher diffraction angles compared to that of ZrO2 (Fig. S5), indicating a decrease in the Ru-RSZ lattice constant owing to a smaller Ru atomic radius.32 After the introduction of Ru, the peaks associated with the monoclinic phase of ZrO2 were no longer detected, suggesting that Ru promotes the stabilization of the tetragonal phase. Raman spectroscopy (Fig. S6) confirmed the presence of the tetragonal ZrO2 phase (t-ZrO2) in the Ru-RSZ sample, with characteristic peaks at 284, 457 and 645 cm−1.33,34 No peaks from the monoclinic phase were observed. This result was in accordance with the above XRD results. Furthermore, two Raman bands corresponding to the tetragonal phase over the Ru-RSZ sample were blue-shifted, suggesting a strong metal–support interaction between Ru and ZrO2.35,36To investigate the morphology and distribution of Ru atoms in Ru-RSZ, scanning electron microscopy (SEM) and spherical aberration-corrected HAADF-STEM imaging (Fig. 2b) were performed.37 SEM and STEM images of Ru-RSZ reveal a morphology similar to that of ZrO2, with both materials consisting of nanoparticles approximately 8 nm in diameter (Fig. S7). The Z-contrast image acquired by HAADF-STEM (Fig. 2b), its Fast Fourier Transform (FFT) (inset) and the corresponding Z-contrast intensity profile show Ru (highlighted) randomly dispersed on Zr lattice sites in ZrO2 projected along the [210] crystallographic direction. STEM-energy-dispersive spectroscopy (STEM-EDS) mapping (Fig. 2c, S8 and Table S1) indicates uniform distribution of Ru, Zr, and O in Ru-RSZ with no obvious segregation (Ru atom percentage of metallic elements in Ru-RSZ is ≈10.8 at%).
The electronic structure of Ru-RSZ was further characterized by using X-ray photoelectron spectroscopy (XPS) and XAS. The full XPS spectra of Ru-RSZ, RuO2 and ZrO2 are presented in Fig. S9. X-ray absorption near-edge spectroscopy (XANES) of the Ru K-edge reveals a shift of the absorption edge to lower energy in Ru-RSZ compared to RuO2, suggesting a decrease in the Ru valence state (Fig. 2d). The Zr K-edge XANES spectra indicate a near-edge absorption energy slightly higher than that of ZrO2 (Fig. 2e). The Zr 3d XPS spectrum (Fig. S10) of Ru-RSZ exhibits a discernible binding energy upshift of approximately 0.14 eV and 0.2 eV for the Zr 3d5/2 and Zr 3d3/2 peaks, respectively, compared to ZrO2, suggesting a relatively higher oxidation state of Zr in Ru-RSZ, which agrees well with the XANES analysis results.38,39 Meanwhile, the deconvolution of the Ru 3p XPS spectrum (Fig. S11 and Table S2) indicates that the proportion of RuIII in Ru-RSZ is 74%, significantly higher than that in RuO2 (26%).40 This higher ratio suggests electron transfer from the ZrO2 support to Ru, leading to a reorganization of its local electronic structure, a conclusion further supported by the distinct negative shift in the Ru 3p binding energy peaks. The pre-reduction effect of Zr on Ru can effectively prevent the excessive oxidative dissolution of Ru during the OER process, thereby enhancing its stability.41
Wavelet Transform Extended X-ray Absorption Fine Structure (WT-EXAFS) indicates that the coordination environment of Ru in Ru-RSZ is the same as that of the Zr sites in ZrO2 (featuring two Ru/Zr–O coordination bonds),20 distinct from that in RuO2 (which exhibits Ru–O and Ru–Ru coordination bonds) (Fig. 2f). Concurrently, Ru K-edge EXAFS reveals Ru–O and Ru–Ru/Zr scattering peaks in Ru-RSZ at approximately 1.6 Å, 2.8 Å, and 3.2 Å. Zr K-edge EXAFS likewise shows Zr–O and Zr–Zr scattering peaks in Ru-RSZ at 1.6 Å, 2.7 Å, and 3.2 Å, demonstrating that Ru and Zr occupy the same metal sites in the Ru/Zr oxide phase, with a symmetry change from D2h to D2d (Fig. 2g, S12 and S13). In addition, EXAFS fitting results (Fig. 2h and Table S3) indicate a Ru–O coordination number of 7.4 in Ru-RSZ, evidencing a significant presence of oxygen vacancies.16 Furthermore, the O 1s peak (Fig. S14) of Ru-RSZ is deconvoluted into three peaks at 529.6, 530.7, and 532.7 eV, corresponding to the lattice oxygen (OL), surface oxygen vacancies (OV) and surface oxygen adsorbates (Oads), respectively.42,43 The peak fitting results of the O 1s XPS spectra (Table S4) indicate that the proportion of oxygen vacancies in Ru-RSZ is 43%, significantly higher than the 29% observed in ZrO2, suggesting a significant increase after Ru introduction. This finding is further corroborated by electron paramagnetic resonance (EPR) measurements (Fig. S15), which reveal a characteristic signal at g = 2.005 for Ru-RSZ (Fig. S15), corresponding to unpaired electrons trapped at oxygen vacancies, in good agreement with the XPS O 1s analysis.29
Electrochemical performance in acidic electrolytes
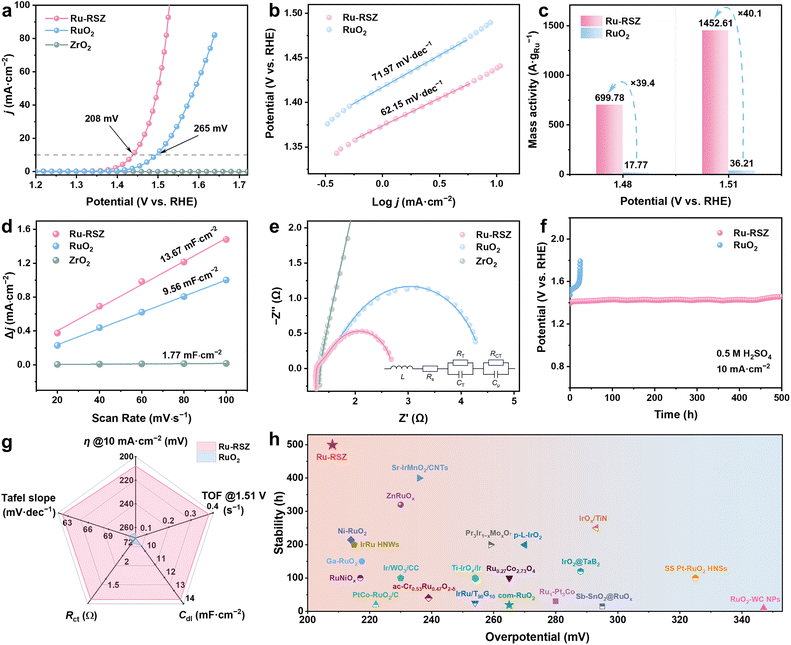
The OER performance of Ru-RSZ, ZrO2 and RuO2 catalysts was evaluated in a three-electrode system using 0.5 M H2SO4 as the electrolyte. Linear sweep voltammetry (LSV) and corresponding Tafel analysis (Fig. 3a and b) demonstrated that the Ru-RSZ catalyst exhibits outstanding electrocatalytic activity, requiring an overpotential (η) of only 208 mV to achieve a current density of 10 mA cm−2 along with a Tafel slope of 62.15 mV dec−1. These values are substantially lower than those of RuO2 (265 mV and 71.97 mV dec−1, respectively), while the ZrO2 support showed negligible OER activity. Notably, the Ru specific mass activity (Fig. 3cand S16) of the Ru-RSZ catalyst was calculated to be as high as 1452.61 A gRu−1 at η = 1.51 V, which is more than 40.1 times higher than that of RuO2 (36.21 A gRu−1 at 1.51 V). To further evaluate the intrinsic activity of the Ru species, the turnover frequency (TOF) was calculated and plotted against the overpotential (Fig. S17). The calculated TOF value for Ru-RSZ reached 0.38 s−1 at an overpotential of 1.51 V, representing a 38-fold enhancement over that of RuO2 (0.01 s−1). Double-layer capacitance (Cdl) was measured to determine the electrochemical active surface area (ECSA) for evaluating the OER performance (Fig. 3d, S18 and Table S5). Ru-RSZ demonstrates the highest Cdl of 13.67 mF cm−2 and ECSA (390.57 cm2), values approximately 1.43 times greater than those of RuO2 (9.56 mF cm−2 and 273.14 cm2, respectively). This indicates a significantly increased number of available active sites in Ru-RSZ, which effectively enhances its OER performance. Additionally, the OER activity normalized to the electrochemical surface area of Ru-RSZ and RuO2 was also calculated (Fig. S19). The ECSA-normalized mass activity of Ru-RSZ reached 0.16 mA cm−2 at 1.51 V (vs. RHE), which is 3.2 times higher than that of RuO2 (0.05 mA cm−2), further demonstrating its superior intrinsic OER catalytic activity.Electrochemical impedance spectroscopy (EIS) is a powerful technique to examine the electrical charge transfer behavior across interfaces and phases in electrochemical systems.44 The EIS measurements (Fig. 3e) revealed well-defined semicircular Nyquist plots for Ru-RSZ, RuO2, and ZrO2.45 Among these, Ru-RSZ exhibited a semicircle with a notably smaller radius compared to RuO2 and ZrO2. The fitting results (Table S6) indicated a significantly lower charge-transfer resistance (Rct) and more efficient electron transfer across the electrode/electrolyte interface. The stability of Ru-RSZ was evaluated by chronopotentiometry (CP) at a constant current density of 10 mA cm−2 (Fig. 3f). Compared to the commercial RuO2 catalyst, Ru-RSZ exhibited excellent stability, retaining most of its potential over a 500-hour period, with some cases even showing a slight performance enhancement as indicated by a decrease in the overpotential. Post-reaction characterization (Fig. S20) confirmed that the crystal structure and atomic dispersion of Ru remained intact, further supporting the structural robustness of the catalyst. With regard to the OER, Ru-RSZ outperformed RuO2 across all five considered dimensions (Fig. 3g and Table S7). The outstanding activity and stability of Ru-RSZ significantly exceed those of previously reported noble metal electrocatalysts in acidic electrolytes (Fig. 3h and Table S8).
Insights into the OER catalytic mechanism
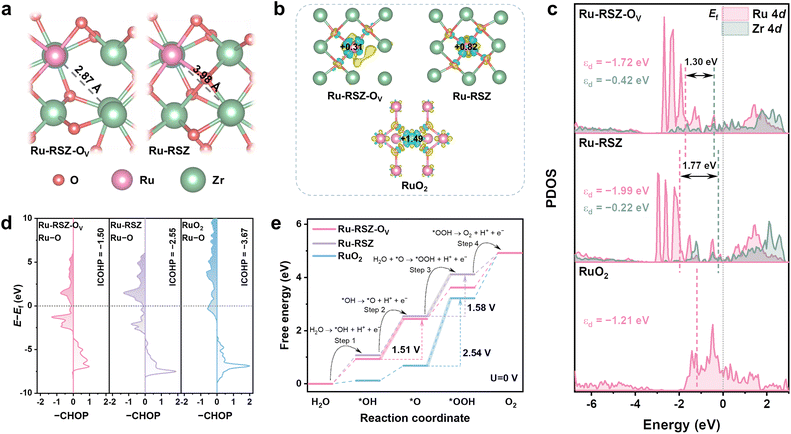
To elucidate the regulatory mechanism of oxygen vacancies on the electronic structure of catalytic sites, systematic DFT calculations were conducted on Ru-RSZ-OV, Ru-RSZ, and RuO2. Structural optimization (Fig. 4a) reveals that the formation of an oxygen vacancy induces notable local structural relaxation, reducing the distance between Ru and the adjacent Zr atom from 3.98 Å (Ru-RSZ) to 2.87 Å (Ru-RSZ-OV), thereby creating favorable geometric conditions for interatomic orbital interactions.22 Charge density difference and Bader charge analyses (Fig. 4b and Table S9) collectively unveil the charge redistribution triggered by the oxygen vacancies, with pronounced electron accumulation observed in the Ru–Zr bridging region, preliminarily indicating enhanced d–d orbital interaction.46 The Bader charge on the Ru atom in Ru-RSZ-OV decreases to +0.3099|e|, lower than its value of +0.8249|e| in Ru-RSZ, while the charge on the adjacent Zr atom also decreases from +1.6345|e| to +1.5131|e|, suggesting weakened electron localization and enhanced covalency.47 Notably, the Bader charge of Ru in Ru-RSZ-OV is lower than that of Ru in RuO2 (+1.4893|e|), while the charge of its Zr is higher than that of Zr in ZrO2 (+1.2822|e|), providing clear evidence for electron transfer from Zr to Ru, aligning with the valence state trend measured by XAS, jointly confirming the metal–support electronic synergy.In-depth analysis of the electronic structure shows that the PDOS (Fig. 4c) of the Ru-4d and Zr-4d orbitals in Ru-RSZ-OV exhibits significantly enhanced peak overlap and hybridization near the Fermi level, confirming the effective promotion of d–d orbital coupling between Ru and Zr by the oxygen vacancies.48 Furthermore, the separation between their d-band centers contracts from 1.77 eV in Ru-RSZ to 1.30 eV in Ru-RSZ-Ov, implying a reduced average energy difference and closer spatial distribution between the Ru-4d and Zr-4d orbitals, which further supports the conclusion of enhanced d–d orbital coupling. This coupling drives electron transfer from Zr to Ru, resulting in a moderate upshift of the Ru d-band center (from −1.99 eV to −1.72 eV) and a concomitant downshift of the Zr d-band center (from −0.42 eV to −0.22 eV) in Ru-RSZ-OV, a theoretically calculated charge redistribution that is consistent with the XAS analysis. The moderate upshift of the Ru d-band center facilitates the optimization of its adsorption free energy for OER intermediates, thereby enhancing the intrinsic activity.49 Concurrently, its energy level remains substantially lower than that of RuO2 (−1.21 eV), which is beneficial for suppressing the over-oxidation of Ru and consequently improving the electrochemical stability.
Direct evidence for chemical bond strengthening is provided by ICOHP analysis. The ICOHP value for the Ru–Zr atomic pair (Fig. S21) decreases significantly from −0.001 eV in Ru-RSZ to −0.089 eV in Ru-RSZ-OV, demonstrating a significant enhancement of the Ru–Zr bond induced by the oxygen vacancies. Simultaneously, analysis of the Ru site reveals that the ICOHP value (Fig. 5d) for the Ru–O bond in Ru-RSZ-Ov exhibits a less negative value (−1.50 eV) compared to those in Ru-RSZ (−2.55 eV) and RuO2 (−3.067 eV), indicating a weakened covalent character. This attenuation is attributed to the Ru–Zr d–d orbital coupling, which effectively modulates the adsorption strength of oxygenated intermediates on the Ru active sites, thereby optimizing the reaction kinetics of the OER. Furthermore, pH-dependent and tetramethylammonium cation (TMA+) probing measurements were further used to demonstrate the restricted lattice oxygen activity in Ru-RSZ (Fig. S22–24). According to the calculated proton reaction orders (ρ = ∂log(j)/∂pH), RuO2 shows significant pH-dependent (ρ = −0.165) OER activity, whereas Ru-RSZ reflects a pH-independent (ρ = −0.076) characteristic in which the current density nearly remains constant as the pH value changes. This result indicates that the degree of non-equilibrium proton–electron coupling transfer of Ru-RSZ has decreased.23 Moreover, the addition of TMA+ in 0.5 M H2SO4 does not affect the OER activity of Ru-RSZ, whereas a clear inhibition is observed for RuO2, indicating that the amount of *OO intermediates produced by Ru-RSZ is lower, demonstrating that the extent of lattice oxygen involvement in the OER process is reduced and the stability of the lattice structure has been optimized.50 Gibbs free energy calculations (Fig. 5e, S25 and S26) demonstrate that the adsorption energies of Ru-RSZ-OV for all OER intermediates lie between those of Ru-RSZ and RuO2, achieving a more balanced adsorption strength.51 Concurrently, the rate-determining step (RDS) energy barrier for Ru-RSZ-OV is the lowest (1.51 eV, *OH →*O + H+ + e−) among the studied catalysts, being lower than those for Ru-RSZ (1.58 eV, H2O + *O → *OOH + H+ + e−) and RuO2 (2.41 eV). The change in the RDS coupled with the significant reduction in the energy barrier collectively contributes to the enhanced OER activity.30 The vacancy formation energy calculations (Fig. S27) indicate that the Ru vacancy formation energy in Ru-RSZ-OV (8.94 eV) is higher than those in RuO2 (8.22 eV) and Ru-RSZ (8.68 eV), suggesting greater resistance of the Ru species to dissolution, which further corroborates, from a thermodynamic perspective, the crucial role of d–d orbital coupling in enhancing OER stability.
PEMWE performance evaluation
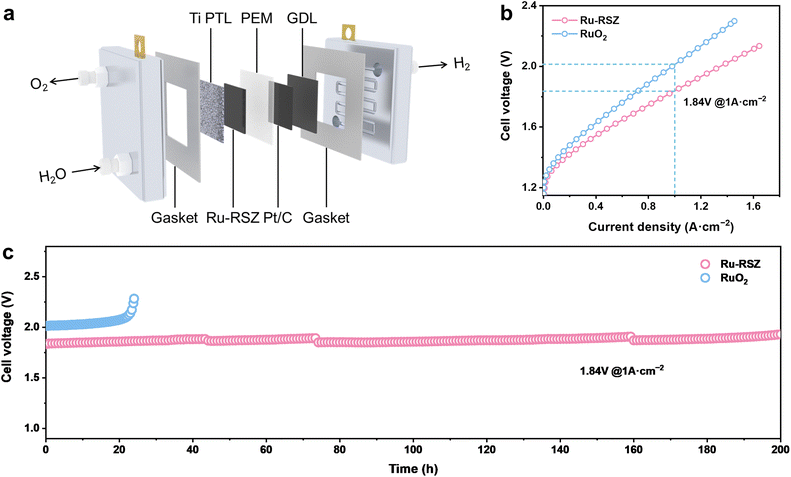
To further verify the performance of Ru-RSZ for practical applications in water electrolysis, a membrane electrode assembly was constructed utilizing a Nafion 211 membrane with Ru-RSZ as the anode catalyst and commercial Pt/C as the cathode catalyst, respectively (Fig. 5a). The mass loading of Ru in the anode of the PEMWE tests is 175.7 µgRu cm−2. The polarization curve (without iR compensation) confirms the superior catalytic activity of Ru-RSZ compared to commercial RuO2. Specifically, the Ru-RSZ-based PEMWE electrolyzer delivers a current density of 1 A cm−2 at a cell voltage of 1.84 V, which is 180 mV lower than the voltage required by commercial RuO2 (Fig. 5b).11,52 Furthermore, the long-term stability of the PEMWE with the Ru-RSZ anode was evaluated by the CP test at a constant current density of 1 A cm−2 (Fig. 5c). The result indicates that the cell voltage remained stable throughout the 200-hour test period, with no significant increase observed. These results strongly confirm that the electronic regulation of Ru by the ZrO2 support could enhance both the catalytic activity and stability of Ru-RSZ, demonstrating significant potential for practical application in PEMWE.Conclusions
In summary, this work reports the synthesis of a Ru electrocatalyst anchored on a zirconia support (Ru-RSZ), wherein the electronic structure of the Ru active sites is precisely modulated through the establishment of Ru–Zr 4d–4d coupling via oxygen vacancy generation, leading to substantially improved activity and stability for acidic OER. The successful stabilization of the tetragonal ZrO2 phase upon Ru incorporation was confirmed by XRD and Raman spectroscopy, while the formation of oxygen vacancies induced by Ru doping was evidenced by XAS and XPS analyses. DFT calculations provide a mechanistic foundation, showing that the coupling redistributes electron density and weakens the binding strength of oxygen intermediates, thereby optimizing the reaction kinetics. The optimized Ru-RSZ electrode delivers a low overpotential of 208 mV at 10 mA cm−2 and a mass activity (1452.61 A gRu−1 @ 1.51 V) 40.1 times greater than that of commercial RuO2. Its practical viability is further demonstrated in a PEMWE cell, achieving 1.84 V at 1 A cm−2 with stable operation for >200 h. Consequently, this work presents a rational and effective strategy for the development of efficient and durable Ru-based electrocatalysts for practical renewable energy conversion technologies.Author contributions
D. Yu conceived the project, performed all the electrochemical measurements, and wrote the initial draft of the manuscript. H. Zhao and Y. Zhang reviewed and edited the manuscript. B. Liu, D. Li, and J. Yu provided useful discussions and insights. X. Li and S. Liu contributed to the interpretation of the experimental results. Z. Li and P. Gao supervised the research. All authors contributed to the manuscript.Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting the findings of this article are available within the paper and the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc01575a.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (22478450), the Sichuan Province Science and Technology Education Joint Fund Project (2025NSFSC2057), the Liaoning Binhai Laboratory Project (LBLF-202306) and the Science and Technology Foundation of Dalian City (2022JJ11CG005).Notes and references
- H. Jun, E. Kang, J. Moon, H. Kim, S. Han, S. Choung, S. Kim, S. Y. Yi, E. Kang, C. H. Choi, J. W. Han and J. Lee, Chem, 2025, 11, 102367 CAS.
- Y. Jiao, Y. Zheng, M. Jaroniec and S. Z. Qiao, Chem. Soc. Rev., 2015, 44, 2060–2086 RSC.
- X. Wang, Z. Li, H. Jang, C. Chen, S. Liu, L. Wang, M. G. Kim, J. Cho, Q. Qin and X. Liu, Adv. Energy Mater., 2024, 15, 2403388 CrossRef.
- S. Yang, Q. Shu, B. Fu, S. Liu, Y. Zhang and H. Zhao, Chem. Eng. J., 2024, 488, 150905 CrossRef CAS.
- R.-T. Liu, Z.-L. Xu, F.-M. Li, F.-Y. Chen, J.-Y. Yu, Y. Yan, Y. Chen and B. Y. Xia, Chem. Soc. Rev., 2023, 52, 5652–5683 RSC.
- C.-Z. Yuan, S. Wang, K. San Hui, K. Wang, J. Li, H. Gao, C. Zha, X. Zhang, D. A. Dinh, X.-L. Wu, Z. Tang, J. Wan, Z. Shao and K. N. Hui, ACS Catal., 2023, 13, 2462–2471 CrossRef CAS.
- R. Ram, L. Xia, H. Benzidi, A. Guha, V. Golovanova, A. Garzon Manjon, D. Llorens Rauret, P. Sanz Berman, M. Dimitropoulos, B. Mundet, E. Pastor, V. Celorrio, C. A. Mesa, A. M. Das, A. Pinilla-Sanchez, S. Gimenez, J. Arbiol, N. Lopez and F. P. Garcia de Arquer, Science, 2024, 384, 1373–1380 CrossRef CAS PubMed.
- Z.-P. Wu, H. Zhang, S. Zuo, Y. Wang, S. L. Zhang, J. Zhang, S.-Q. Zang and X. W. Lou, Adv. Mater., 2021, 33, 2103004 CrossRef CAS PubMed.
- J. Chen, J. Ma, T. Huang, Q. Liu, X. Liu, R. Luo, J. Xu, X. Wang, T. Jiang, H. Liu, Z. Lv, T. Yao, G. Wang, X. Zheng, Z. Li and W. Chen, Angew. Chem., Int. Ed., 2025, 64, e202503330 CrossRef CAS PubMed.
- J. Park, D. W. Lee, J. Hyun, H. Lee, E. Oh, K. Seok, G. Doo and H.-T. Kim, Energy Environ. Sci., 2025, 18, 5577–5585 RSC.
- W. He, X. Tan, Y. Guo, Y. Xiao, H. Cui and C. Wang, Angew. Chem., Int. Ed. Engl., 2024, 63, e202405798 CrossRef CAS PubMed.
- M. Zhang, W. An, Q. Liu, Y. Jiang, X. Zhao, H. Chen, Y. Zou, X. Liang and X. Zou, Nat. Commun., 2025, 16, 7608 CrossRef CAS PubMed.
- J. Chen, Y. Ma, T. Huang, T. Jiang, S. Park, J. Xu, X. Wang, Q. Peng, S. Liu, G. Wang and W. Chen, Adv. Mater., 2024, 36, e2312369 CrossRef PubMed.
- G. Chen, R. Lu, C. Ma, X. Zhang, Z. Wang, Y. Xiong and Y. Han, Angew. Chem., Int. Ed. Engl., 2024, 63, e202411603 CrossRef CAS PubMed.
- L. Li, G. Zhang, C. Zhou, F. Lv, Y. Tan, Y. Han, H. Luo, D. Wang, Y. Liu, C. Shang, L. Zeng, Q. Huang, R. Zeng, N. Ye, M. Luo and S. Guo, Nat. Commun., 2024, 15, 4974 CrossRef CAS.
- Y. Shen, X.-L. Zhang, M.-R. Qu, J. Ma, S. Zhu, Y.-L. Min, M.-R. Gao and S.-H. Yu, Nat. Commun., 2024, 15, 7861 CrossRef CAS PubMed.
- J. Chen, Y. Ma, C. Cheng, T. Huang, R. Luo, J. Xu, X. Wang, T. Jiang, H. Liu, S. Liu, T. Huang, L. Zhang and W. Chen, J. Am. Chem. Soc., 2025, 147, 8720–8731 CrossRef CAS PubMed.
- J. Zhang, H. Qin, X. Cao, W. Jia, R. Ma, X. Chen, W. Xia, G. Lin and L. Jiao, ACS Mater. Lett., 2024, 6, 3016–3024 CrossRef CAS.
- Y. Qin, S. Deng, X.-Y. Zhou, Z. Yan, L. He, K. Li and T.-Y. Zhang, J. Am. Chem. Soc., 2025, 147, 30943–30955 CrossRef CAS PubMed.
- L. Deng, H. Chen, S.-F. Hung, Y. Zhang, H. Yu, H.-Y. Chen, L. Li and S. Peng, J. Am. Chem. Soc., 2024, 146, 35438–35448 CrossRef CAS PubMed.
- X. Li, H. Rong, J. Zhang, D. Wang and Y. Li, Nano Res., 2020, 13, 1842–1855 CrossRef CAS.
- C. Zhou, L. Li, Z. Dong, F. Lv, H. Guo, K. Wang, M. Li, Z. Qian, N. Ye, Z. Lin, M. Luo and S. Guo, Nat. Commun., 2024, 15, 9774 CrossRef CAS PubMed.
- X. Zhang, Y. Zhang, B. O. Protsenko, M. A. Soldatov, J. Zhang, C. Yang, S. Bo, H. Wang, X. Chen, C. Wang, W. Cheng and Q. Liu, Nat. Commun., 2025, 16, 6921 CrossRef CAS PubMed.
- Y. Li, J. Zhao, S. Zhang, Y. Fan, C. Y. Kuo, Y. C. Ku, T. S. Chan, C. W. Kao, Y. C. Huang, C. T. Chen, S. C. Haw, C. Jin, H. Zhao, D. Ye, C. Jing, Z. Hu and L. Zhang, J. Am. Chem. Soc., 2025, 147, 26854–26864 CrossRef CAS PubMed.
- B.-J. Kim, D. F. Abbott, X. Cheng, E. Fabbri, M. Nachtegaal, F. Bozza, I. E. Castelli, D. Lebedev, R. Schäublin, C. Copéret, T. Graule, N. Marzari and T. J. Schmidt, ACS Catal., 2017, 7, 3245–3256 CrossRef CAS.
- Z.-Q. Jiang, C.-Z. Fan, J.-Y. Pan, L. Shao, H. Chen, E. Pervaiz, Y. Dong, T.-Z. Wang, X.-R. Zheng, J.-H. Li and Y.-D. Deng, Rare Met., 2024, 43, 2891–2912 CrossRef CAS.
- H. Yu, C. Wang, X. Xin, Y. Wei, S. Li, Y. An, F. Sun, T. Lin and L. Zhong, Nat. Commun., 2024, 15, 5143 CrossRef CAS PubMed.
- Y. Lin, Z. Tian, L. Zhang, J. Ma, Z. Jiang, B. J. Deibert, R. Ge and L. Chen, Nat. Commun., 2019, 10, 162 CrossRef PubMed.
- F. Wang, L. Feng, M. Zhang and H. Cong, Nat. Commun., 2025, 16, 6624 CrossRef CAS PubMed.
- J. Zhang, L. Xu, X. Yang, S. Guo, Y. Zhang, Y. Zhao, G. Wu and G. Li, Angew. Chem., Int. Ed., 2024, 63, e202405641 CrossRef CAS PubMed.
- T. Wu, X. Zhu, Z. Xing, S. Mou, C. Li, Y. Qiao, Q. Liu, Y. Luo, X. Shi, Y. Zhang and X. Sun, Angew. Chem., Int. Ed. Engl., 2019, 58, 18449–18453 CrossRef CAS PubMed.
- W. Q. Zaman, W. Sun, M. Tariq, Z. Zhou, U. Farooq, Z. Abbas, L. Cao and J. Yang, Appl. Catal., B, 2019, 244, 295–302 CrossRef CAS.
- M. C. Uribe López, M. A. Alvarez Lemus, M. C. Hidalgo, R. López González, P. Quintana Owen, S. Oros-Ruiz, S. A. Uribe López and J. Acosta, J. Nanomater., 2019, 2019, 1–12 CrossRef.
- C. Li, Z. Han, Y. Hu, T. Liu and X. Pan, RSC Adv., 2022, 12, 27309–27320 RSC.
- C. Dong, R. Mu, R. Li, J. Wang, T. Song, Z. Qu, Q. Fu and X. Bao, J. Am. Chem. Soc., 2023, 145, 17056–17065 CrossRef CAS PubMed.
- Z. Zhang, S. Lu, M. Zhu, F. Wang, K. Yang, B. Dong, Q. Yao and W. Hu, J. Am. Chem. Soc., 2025, 147, 22806–22817 CrossRef CAS PubMed.
- N. Zhang, Y. Hu, L. An, Q. Li, J. Yin, J. Li, R. Yang, M. Lu, S. Zhang, P. Xi and C. H. Yan, Angew. Chem., Int. Ed. Engl., 2022, 61, e202207217 CrossRef CAS.
- X. Fang, S. Wu, Y. Wu, W. Yang, Y. Li, J. He, P. Hong, M. Nie, C. Xie, Z. Wu, K. Zhang, L. Kong and J. Liu, Appl. Surf. Sci., 2020, 518, 146226 CrossRef CAS.
- M. Yang, X. Guan, Z. Shi, H. Wu, Y. Cheng, Z. Wang, W. Liu, F. Xiao, M. Shao, M. Xiao, C. Liu and W. Xing, Small, 2025, 22, 2411117 CrossRef PubMed.
- Z. Li, H. Sheng, Y. Lin, H. Hu, H. Sun, Y. Dong, X. Chen, L. Wei, Z. Tian, Q. Chen, J. Su and L. Chen, Adv. Funct. Mater., 2024, 34, 2409714 CrossRef CAS.
- J. Zhao, Y. Guo, Z. Zhang, X. Zhang, Q. Ji, H. Zhang, Z. Song, D. Liu, J. Zeng, C. Chuang, E. Zhang, Y. Wang, G. Hu, M. A. Mushtaq, W. Raza, X. Cai and F. Ciucci, Nat. Nanotechnol., 2025, 20, 57–66 CrossRef CAS PubMed.
- R. Deng, F. Liu, S. Gao, Z. Xia, R. Wu, J. Kong, J. Yang, J. Wen, X. Zhang, C. Lv, Y. Wang, X. Li and Z. Wang, ACS Catal., 2025, 15, 1782–1794 CrossRef CAS.
- Q. Yao, J. Le, S. Yang, J. Cheng, Q. Shao and X. Huang, Chin. J. Catal., 2022, 43, 1493–1501 CrossRef CAS.
- J. M. P. Martirez and E. A. Carter, J. Am. Chem. Soc., 2019, 141, 693–705 CrossRef CAS PubMed.
- S. Wang, Q. Jiang, S. Ju, C. S. Hsu, H. M. Chen, D. Zhang and F. Song, Nat. Commun., 2022, 13, 6650 CrossRef CAS PubMed.
- Q. Ji, B. Tang, X. Zhang, C. Wang, H. Tan, J. Zhao, R. Liu, M. Sun, H. Liu, C. Jiang, J. Zeng, X. Cai and W. Yan, Nat. Commun., 2024, 15, 8089 CrossRef CAS PubMed.
- Q. Qin, Z. Li, X. Zhao, H. Zhao, L. Zhai, M. Gyu Kim, J. Cho, H. Jang, S. Liu and X. Liu, Angew. Chem., Int. Ed. Engl., 2025, 64, e202413657 CrossRef CAS PubMed.
- H. Wang, T. Zhai, Y. Wu, T. Zhou, B. Zhou, C. Shang and Z. Guo, Adv. Sci., 2023, 10, 2301706 CrossRef CAS PubMed.
- Y. Qin, T. Yu, S. Deng, X.-Y. Zhou, D. Lin, Q. Zhang, Z. Jin, D. Zhang, Y.-B. He, H.-J. Qiu, L. He, F. Kang, K. Li and T.-Y. Zhang, Nat. Commun., 2022, 13, 3784 CrossRef CAS PubMed.
- K. Zhou, Y. Wang, Z. Jiang, B. Dai and Z. J. Jiang, Adv. Mater., 2025, 37, 2420159 CrossRef CAS PubMed.
- Y. Xu, Z. Mao, J. Zhang, J. Ji, Y. Zou, M. Dong, B. Fu, M. Hu, K. Zhang, Z. Chen, S. Chen, H. Yin, P. Liu and H. Zhao, Angew. Chem., Int. Ed., 2024, 63, e202316029 CrossRef CAS PubMed.
- Z.-Y. Wu, F.-Y. Chen, B. Li, S.-W. Yu, Y. Z. Finfrock, D. M. Meira, Q.-Q. Yan, P. Zhu, M.-X. Chen, T.-W. Song, Z. Yin, H.-W. Liang, S. Zhang, G. Wang and H. Wang, Nat. Mater., 2023, 22, 100–108 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |