Open Access Article
Open Access ArticleBreaking the limit – synthesis and properties of p-amino-triazatriangulenium, the most stable carbenium ion
Marko H. Nowack a,
Gennaro Pescitelli
a,
Gennaro Pescitelli b,
Arianna Lanza
b,
Arianna Lanza a and
Bo W. Laursen
a and
Bo W. Laursen *a
*a
aDepartment of Chemistry, University of Copenhagen, Universitetsparken 5, 2100, Copenhagen Ø, Denmark. E-mail: bwl@chem.ku.dk
bDipartimento di Chimica e Chimica Industriale, Università di Pisa, Via Giuseppe Moruzzi 13, 56124 Pisa, Italy
First published on 5th May 2026
Abstract
Highly stabilized tris(dialkylamino)-triazatriangulenium (A-TATA+) and tris(methoxy)-triazatriangulenium (MeO-TATA+) carbenium ions have been synthesized and characterized as fluorescent dyes. As a result of the extensive delocalization of positive charge in the planar triangulenium system and the presence of six strongly electron-donating groups, these new carbenium ions display extreme cation stability. A linear correlation between cation stability (pKR+) and reduction potential (Ered) has been established for the trianguleniums, leading to an estimated pKR+ value of 32 for A-TATA+, making A-TATA+ the most stable carbenium ion reported to date, surpassing the previous record by 7 pKR+ units. Furthermore A-TATA+ displays unique dye properties in the blue spectral region, with intense absorption at 447 nm (ε = 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1 M−1), and a high fluorescence quantum yield at 460 nm (ϕf = 45%).
000 cm−1 M−1), and a high fluorescence quantum yield at 460 nm (ϕf = 45%).
Introduction
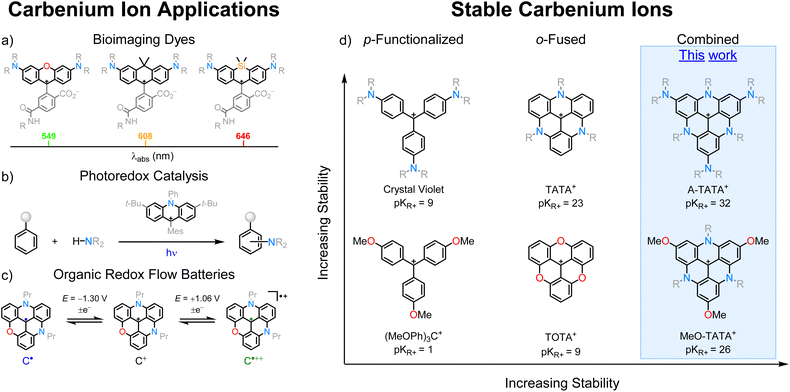
Stabilized carbenium ions such as triarylmethyliums, xantheniums, and acridiniums are organic compounds of great scientific and commercial importance.3 They are extensively used as textile and laser dyes, and are prevalent among the brightest fluorescent probes and cellular stains used for bioimaging and assays.4–6 The attractive spectral and photophysical properties of the carbenium ions originate from a conjugated π-system with an odd number of sp2 centers that results in red-shifted spectra and high absorption coefficients in moderate-sized conjugated chromophore structures.4,7–9 Spectral properties are readily tuned by position and electronegativity of substituents, such as the bridging group in rhodamine-type dyes.10 This is highlighted by the pioneering work from several researchers on the diversification of the rhodamine scaffold, toward red-shifted fluorescent probes (Fig. 1a).11–16 The cationic π-systems are in general electron deficient, which makes them strong one-electron acceptors in the excited state and thus useful as oxidative photocatalysts.17–21 In particular, acridinium systems have found use for a multitude of oxidative photoredox catalyzed reactions (Fig. 1b). For these applications, tuning of the redox potentials is a key feature that is achieved by adding groups either stabilizing or destabilizing the carbenium system.22–26 Stabilized carbenium ions have also seen applications in symmetrical organic redox flow batteries, where the excellent electrochemical stability makes them promising materials (Fig. 1c).27–30 For this and other redox applications, the number and nature of donor atoms/groups (mainly O and N) controls the electron affinity of the carbenium ion and thus the redox potential.31 It is not only the one-electron reduction/oxidation that is important for carbenium ions; nucleophilic addition of anions to the cationic dyes is also a key feature that leads to breaking of conjugation and formation of non-colored leuco adducts.32–35 This feature has been explored e.g. in fluorogenic rhodamine probes switched on at low pH or by interactions with various analytes. Also, in this case tuning of the cation stability is a vital design parameter. For redox related applications, the relevant measure of cation stability is the reduction potential (Ered) while the cation stability towards nucleophiles is measured by the pKR+ value, that quantifies the equilibrium between a carbenium ion and the corresponding alcohol (carbinol) formed by addition of hydroxide ions.36 The pKR+ values of carbenium ions have been extensively used to rank their stabilities, and are essential to evaluate reactivity towards nucleophiles, thus determining the operating conditions for dyes, probes, and catalysts.1,31,37–40Stabilized carbenium ions owe their stability to the addition of electron-donating groups, delocalizing the positive charge. Historically (Fig. 1d), this has been accomplished either by p-functionalization, exemplified by crystal violet, or by o-fusion of neighboring aryl groups via heteroatom bridges, exemplified by the acridinium and TriOxaTriangulenium (TOTA+) ions. The threefold bridging and extended conjugation of the triangulenium salts (Fig. 1d) make them among the most stable carbenium ions reported in the literature to date. The positive charge is delocalized onto the heterocyclic framework as a consequence of the electron-donating bridging heteroatoms, which planarize and rigidify the structure, leading to exceptional stabilities and bright fluorescence. The trianguleniums are named according to the identity of the bridging heteroatoms, as follows:
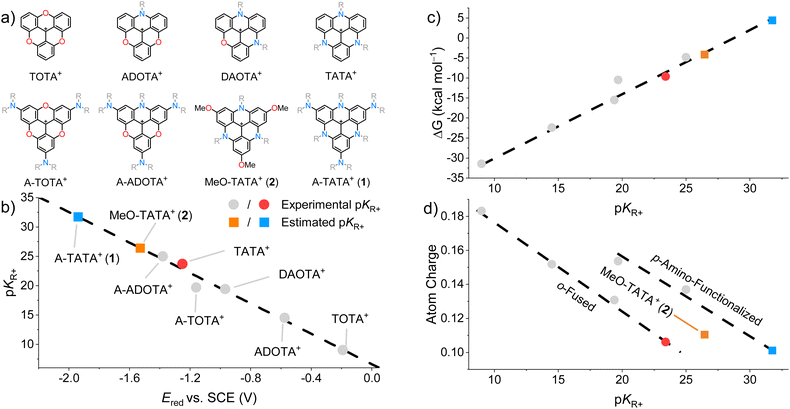
TriOxaTriangulenium (TOTA+), AzaDioxaTriangulenium (ADOTA+), DiAzaOxaTriangulenium (DAOTA+), and TriAzaTriangulenium (TATA+).1 High chemical stability and tunable optical and redox properties have paved the way for their applications as fluorescent dyes with a wide array of implementations, including pH probes and sensors,41–45 chiral emitters,46–49 fluorescence lifetime probes for proton transfer,50,51 and G-quadruplex DNA.52–56 In recent years, the applications have expanded into photoredox catalysis,57–64 CO2 reduction,65–67 hydrogen production,68,69 and electrolytes in organic redox flow batteries.27–30,70 The planar cationic nature of trianguleniums has been used to create ionic assemblies for applications as solid-state optical materials.71–75 We have previously combined p-functionalization with o-fusion to synthesize tris(dialkylAmino)-TOTA+ (A-TOTA+) and tris(dialkylAmino)-ADOTA+ (A-ADOTA+), leading to more highly stabilized carbenium ions.38,40 Among the triangulenium salts, TATA+ and A-ADOTA+ are the most stable carbenium ions reported to date, with pKR+ values of 23 and 25, respectively.1,40 Further stabilizing the triangulenium ions is critical for expanding their use as catalysts under highly basic and/or nucleophilic conditions, extending the redox window for energy storage applications, and lowering the reduction potential to increase the potency as photoreductants.
To create further stabilized triangulenium salts, we envisioned the extension of the TATA+ scaffold with electron-donating groups in the p-positions. However, it turns out that this cannot be realized through conventional nucleophilic aromatic substitution (SNAr) reactions on the tris(2,4,6-trimethoxyphenyl)carbenium precursor as realized for A-ADOTA+ and all other trianguleniums.40 The very successful SNAr synthesis strategy for trianguleniums is driven by the electron deficiency of the cationic substrates. However, as the cation stability increases the electrophilic reactivity decreases, with substitutions becoming fully suppressed for A-ADOTA+ (pKR+ = 25), setting a limit for this synthetic strategy. Therefore, we explored different avenues, inspired by recent literature on the Ir-catalyzed borylation of triangulenium species.76–78 This led to the successful synthesis of p-substituted TATA+ derivatives tris(dialkylAmino)-TriAzaTriangulenium (A-TATA+) and tris(Methoxy)-TriAzaTriangulenium (MeO-TATA+).
Results and discussion
Synthesis and solid-state structure analysis
Starting from N-octyl-TATA+, the p-borylated species (3) was obtained in excellent yield through Ir-catalyzed borylation with bis(1,5-cyclooctadiene)di-µ-methoxydiiridium(I) ([Ir(cod)OMe]2) and 3,4,7,8-tetramethyl-1,10-phenanthroline (Me4phen) as the catalyst system (Scheme 1a).76 From here, several attempts were made at Chan–Lam couplings with pyrrolidine to obtain the A-TATA+ framework directly from 3. However, they were not successful due to competing oxidation and protodeborylation reactions which led to complex mixtures of products. Therefore, other strategies were explored, starting with oxidation of the boronic esters and ion exchange with KPF6 to yield the poorly soluble tris(hydroxy)-TATA+ (4). From here, alkylation with methyl iodide and Cs2CO3 as the base yielded tris(methoxy)-TATA+ (MeO-TATA+, 2). Triflation of tris(hydroxy)-TATA+ 4 using trifluoromethanesulfonic anhydride (Tf2O) and pyridine as the base was achieved to afford the reactive tris(TfO)-TATA+ (5). A threefold SNAr reaction with pyrrolidine in anhydrous THF was possible with careful exclusion of water to form tris(pyrrolidine)-TATA+ (A-TATA+, 1). The compound was purified by column chromatography to remove the undesired S–O scission hydroxy-byproducts formed during the reaction. The crystal structure of MeO-TATA+ (2) was determined by electron diffraction (SI section 1.9), on microcrystals obtained by layering a CHCl3 solution with i-PrOH, followed by slow diffusion at −25 °C. The experimental solid-state structure of MeO-TATA·PF6 confirms the planarity of the triangulenium core (Fig. S56). In the crystal structure, MeO-TATA+ (2) packs in slipped stacks of dimers with two distinct interplanar distances of 3.48 Å and 3.58 Å (Scheme 1c and Fig. S56). Layers of parallel stacks are separated by the kinked octyl chains that form hydrophobic layers between the layers of stacked TATA+ cores. This crystal packing is in contrast to the parent TATA+, which persistently forms 2D layers of TATA+-dimers with inter planar distances of 3.38 Å separated by layers of straight interdigitated alkyl chains (Scheme 1b).1,79,80 We tentatively assign the difference in solid-state packing to the increased footprint of the planar TATA+ core due to the added methoxy-groups, which reduce the relative volume of the alkyl chains inducing their bending and the tilting of the TATA+ dimers in the ionic layers.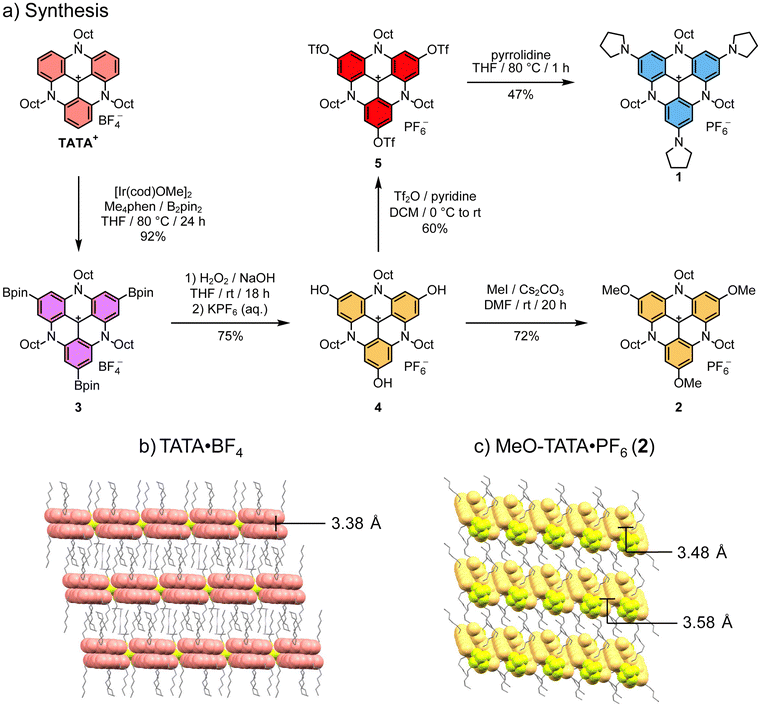 | ||
| Scheme 1 (a) Synthesis of the p-substituted triangulenium dyes A-TATA+ (1) and MeO-TATA+ (2). (b) Crystal packing of TATA·BF4 (redrawn from CIF file: CCDC 148692).1 (c) Crystal packing of MeO-TATA·PF6. | ||
Carbocation stability studies
With the p-substituted TATA+ dyes 1 and 2 in hand, we set out to characterize their cation stabilities. In previous work, a solvent system of DMSO and H2O with tetramethylammonium hydroxide as the hydroxide source was used to determine quantitative pKR+ values for stable carbenium ions.1,38,40,81–84 This system is, however, not applicable for pKR+ values above 25 due to competing irreversible reactions at the carbenium ions.40 Therefore, we first sought to probe the chemical stability of the p-substituted A-TATA+ using a qualitative method. The dyes TATA+ and A-TATA+ were dissolved in MeOH in a cuvette, where an excess of the strong hydride donor NaBH4 was subsequently added. Absorption spectra as a function of time after addition of NaBH4 are shown in Fig. 2 aiming to follow the addition of hydride to the cation in solution (C+ + H− → C–H). TATA+ is slowly reduced to the colorless TATA-H upon reaction with NaBH4, as indicated by the decrease in absorption at 525 nm, while A-TATA+ is not affected by the addition of NaBH4, clearly demonstrating the enhanced chemical stability of A-TATA+, suggesting a significantly higher pKR+ than TATA+ (pKR+ = 23.4). This enhanced cation stability is also expected to be reflected in a more negative reduction potential.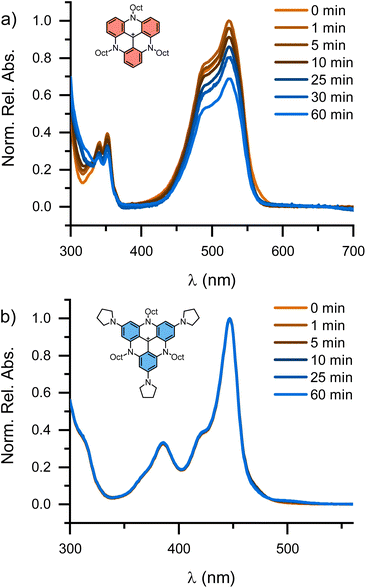 | ||
| Fig. 2 Absorption spectra of the triangulenium dyes TATA+ (a) and A-TATA+ (b) in MeOH after addition of solid NaBH4 at consecutive time points up to 60 min. Anions are omitted for clarity. | ||
We conducted cyclic voltammetry and differential pulse voltammetry experiments in MeCN with tetrabutylammonium hexafluorophosphate (TBAPF6, c = 0.1 M) as the electrolyte to determine the Ered of the trianguleniums presented herein, varying the scan rate from 0.1 to 1 V s−1 (SI section 1.3). The normalized voltammograms at scan-rates where reduction is quasi-reversible are shown in Fig. 3. The voltammograms show clear shifts in the reduction potentials due to the addition of electron-donating groups in the p-positions of TATA+, with Ered shifted by 300 mV for MeO-TATA+ (2) and by as much as 700 mV for A-TATA+ (1). This is a clear demonstration of the large cation-stabilizing effect of the p-donor groups and highlights potential applications of A-TATA+ as a stronger electron donor for photoredox catalysis.66–69
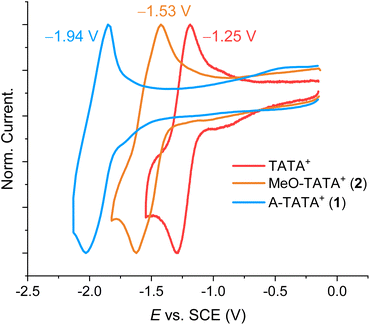 | ||
| Fig. 3 Normalized cyclic voltammograms of the triangulenium dyes TATA+ (red, ν = 0.1 V s−1), MeO-TATA+ (2) (orange, ν = 1 V s−1), and A-TATA+ (1) (blue, ν = 1 V s−1) at quasi-reversible scan-rates. Recorded in MeCN at rt with TBAPF6 (c = 0.1 M) as the supporting electrolyte. Analyte concentrations are 0.5 mM. WE, glassy carbon Ø = 2 mm; RE, Ag/Ag+; CE, Pt wire. All potentials are recorded vs. Fc/Fc+ and converted to SCE by addition of 380 mV.2 | ||
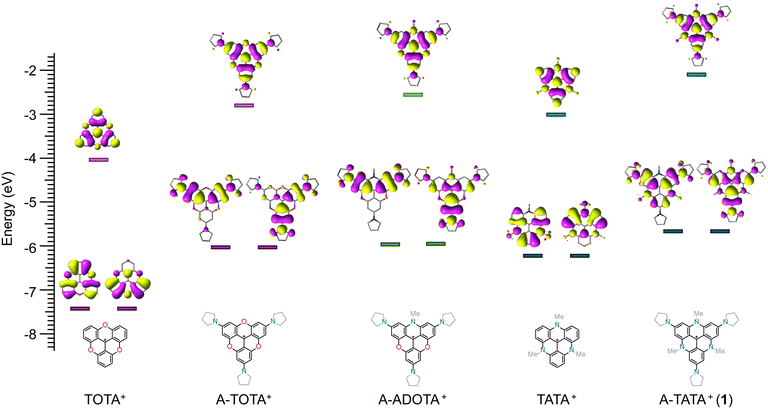
The one-electron reduction potential and the pKR+ value are both measures of carbenium ion stabilities, and work by Okamoto et al. suggests a correlation between the two parameters for structurally related tropyllium ions.85 The reduction potential can be considered to reflect mainly two terms: the electron affinity governed by the molecular structure, and the change in solvation energy between the cation and the neutral radical.86 The pKR+ value has an additional term, which depends on the strain energy difference between the cation and the carbinol product.85,86 Within a family of compounds, like the trianguleniums, the solvation and strain energies are expected to be similar due to the comparable structure of the compounds, so Ered and the pKR+ values should be proportional. To probe the proportionality, the pKR+ and Ered values for several families of carbenium ions were plotted (SI section 1.4). These relationships are linear, though each with different slopes (Fig. S21), suggesting Ered and pKR+ values are indeed proportional within families of carbenium ions. To explore the correlation between reduction potentials and pKR+ values for the trianguleniums, reported pKR+ values were plotted as a function of Ered determined by cyclic voltammetry in MeCN with TBAPF6 (0.1 M) as the supporting electrolyte (electrochemical data is reported in SI section 1.3).1,37,38,40 The proportional nature between pKR+ and Ered is clearly illustrated in Fig. 4b, showcasing a near-perfect linear relationship between the two parameters. The linear relationship for the trianguleniums allows us to estimate the pKR+ values for the p-substituted TATA+ dyes MeO-TATA+ (2) and A-TATA+ (1) to be 26 and 32, respectively. The obtained pKR+ values are far higher than any previously reported pKR+ values and clearly outside the range that can be measured experimentally with previously developed methods. To consolidate these findings and further investigate the cation stabilities of the trianguleniums, we approached the problem from a computational standpoint. The Gibbs free energy for the carbinol-forming reaction (C+ + OH− ⇌ COH) was determined at DFT level of theory (SI section 1.6, Table S7). The pKR+ values for the trianguleniums were plotted as a function of the computed ΔG values (Fig. 4c). These parameters are also linearly correlated, suggesting the estimated pKR+ values for A-TATA+ and MeO-TATA+ based on Ered are valid. These stability constants are the highest for any carbenium ion reported in the literature to our knowledge, making A-TATA+ the most stable carbenium ion reported to date. With this extension of the triangulenium family we can start to analyze and quantify the effects of p-substituents and bridging heteroatoms on the cation stability and reduction potentials (data collected in Table S4).1,37,38,40 Comparing TATA+ (pKR+ = 23) to TOTA+ (pKR+ = 9) the effect of N vs. O bridging is clear, increasing the stability by 14 units on the pKR+ scale. This effect is somewhat smaller for p-substitution as demonstrated by TOTA+ (pKR+ = 9) vs. A-TOTA+ (pKR+ = 19), where adding electron-donating amines increases the stability by 10 units on the pKR+ scale. Adding three p-amino groups to TATA+, giving A-TATA+ (pKR+ = 32), raises the pKR+ by another 9 pKR+ units indicating that the effects are additive and largely independent of the triangulenium core. To further explore the relationship between cation stability and electronic structure of the trianguleniums, the charge distribution on the central carbon atom was calculated using the DDEC6 charge-partitioning scheme (Table S8).87–89 DDEC6 is a modern and efficient net atomic charge (NAC) scheme that avoids inconsistencies between symmetry-related atoms and is tailored to provide an accurate description of ‘buried’ atoms. Plotting the atomic charge as a function of the pKR+ values for the trianguleniums reveals two linearly correlated populations (Fig. 4d), one with the strictly o-fused trianguleniums and one with the p-amino functionalized. The single case of p-methoxy triangulenium (2) falls in between. These results suggest that the impact of p-functionalization of the triangulenium system on Ered and pKR+ goes beyond simple charge delocalization. For instance, A-TATA+ and TATA+ have similar partial charges (+0.099 and +0.105, respectively) on the central carbon despite pKR+ values differing by 9 units. In fact, the main effect of p-amino substituents consists in raising both HOMO and LUMO energies, to different extents but in a correlated way, with respect to the unsubstituted analogs (Fig. 5). According to DFT calculations, HOMO and LUMO destabilization amounts to 0.57 eV and 0.91 eV, respectively, going from TATA+ to A-TATA+, consistent with the respective increase in pKR+ and |Ered| values.
Optical properties
The triangulenium family of carbenium ions is characterized by their exceptional cation stability combined with very favorable optical properties, enabling their applications as fluorescent dyes and probes, as well as photoredox catalysts.62 Trianguleniums without p-donor groups display unique photophysical properties with long fluorescence lifetimes (τ > 20 ns in the case of ADOTA+ and DAOTA+) which have been key to many fluorescence assays and imaging applications.52,54–56,90–96 The introduction of p-amino groups in A-TOTA+ and A-ADOTA+, on the other hand, leads to high molar absorption coefficients and short lifetimes (τ ≈ 4 ns) akin to rhodamines, and these dyes are used as chemically stable, bright fluorescent labels.97 Therefore, we investigated the photophysical properties of the new triangulenium dyes MeO-TATA+ (2) and A-TATA+ (1) in CH2Cl2, and compared them to the parent TATA+ fluorophore (SI section 1.5). The normalized absorption spectra and the emission spectra scaled to the fluorescence quantum yields are shown in Fig. 6. Detailed emission and excitation spectra are provided in SI section 1.5.1. The absorption coefficients, fluorescence lifetimes, fluorescence quantum yields, and associated rate constants are given in Table 1, with additional data provided in SI sections 1.5.2 and 1.5.3.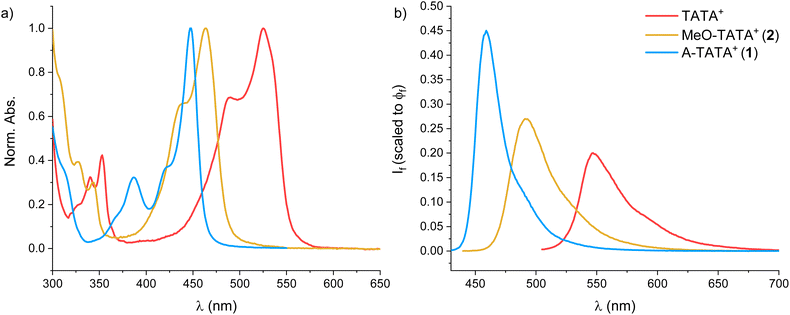 | ||
| Fig. 6 (a) Normalized absorption spectra of the TATA+ dyes in CH2Cl2, (b) Emission spectra scaled to the fluorescence quantum yield for the TATA+ dyes in CH2Cl2. | ||
| ε [M−1 cm−1] | λabs/λemi [nm] | τavg [ns] | ϕf [%]a | kf [107 s−1] | knr [107 s−1] | |
|---|---|---|---|---|---|---|
| a Quantum yields are determined relative to coumarin-153 in EtOH (ϕf = 54%).98 For TATA+ the quantum yield was determined relative to Rho-6G in EtOH (ϕf = 94%).99b Data from Laursen et al.100c Data from Laursen and Sørensen.40 | ||||||
| TATA+ | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
525/548 | 6.6 | 21 | 3.2 | 12 |
| MeO-TATA+ (2) | 26![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
463/485 | 4.6 | 28 | 6.0 | 16 |
| A-TATA+ (1) | 84![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
447/460 | 2.7 | 45 | 17 | 20 |
| A-TOTA+ | 132![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000b 000b |
472/495 | 3.0 | 93 | 31 | 2 |
| A-ADOTA+ | 120![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000c 000c |
458/479 | 2.7 | 73 | 27 | 10 |
From the absorption spectra, we observe a clear blue-shift in the absorption maxima as stronger electron donors are introduced in the p-positions, with A-TATA+ having an absorption maximum at 447 nm. The shift in absorption maxima is well reproduced by TD-DFT calculations (CAM-B3LYP/def2-TZVP/PCM level, Table S9 and Fig. S41). The same trend is observed in the emission spectra, drastically blue-shifting the fluorescence with increasing electron donor strength. Interestingly, the fluorescence quantum yield increases two-fold from TATA+ to A-TATA+ (from 20% to 45%). This is accompanied by a decrease in fluorescence lifetime from 6.6 to 2.7 ns for A-TATA+, reflecting a fivefold increase in the radiative rate constant (kf) from 3.0 × 107 to 16 × 107 s−1. The non-radiative rate constant (knr) increases marginally from 12 × 107 to 20 × 107 s−1, suggesting that p-functionalization also introduces additional non-radiative decay pathways.
All told, the introduction of electron donating groups in the p-positions makes the TATA+ scaffold a more efficient fluorophore with significantly blue-shifted optical properties. Excitation at 447 nm with a high molar absorption coefficient, good ϕf, and a small Stokes shift is rare for carbenium ions, and suggests that A-TATA+ derivatives could find applications in imaging and bioassays, especially when modified for water solubility and bioconjugation, or in other applications where cationic chromophores are needed.72,101,102 This spectral range is usually reserved for coumarins,5,103 substituted pyrenes,104–106 and multiple-resonant emitters for OLEDs.107 The changes in the optical properties can be rationalized using Dewar's perturbation molecular orbital theory approach, which has proven to be a good model for trianguleniums and related systems.108 The pair of HOMO orbitals of TATA+ has a node in the para positions (Fig. 5). Thus, introduction of electron-donating groups will predominantly destabilize the LUMO, and to a lesser extent the HOMO, leading to a blue-shift in the optical properties, namely S0–S1 and S1–S0 transitions, which are mainly associated with HOMO–LUMO single (de-)excitations. This explanation agrees with the redox properties, where A-TATA+'s Ered is significantly more negative (ΔE = −0.7 V, Fig. 3) compared to TATA+, while the oxidation potential is less affected (ΔE = −0.33 V, Table S1). These values agree with the shift in absorption maxima of 0.4 eV.
Conclusions
In summary, we have presented herein the synthesis of TATA+ derivatives with electron-donating groups (methoxy and amino) in the p-positions. These new trianguleniums MeO-TATA+ (2) and A-TATA+ (1) are among the most stable carbenium ions, with A-TATA+ having the highest cation stability ever reported with a pKR+ value of 32. The stability is accompanied by an exceptionally low Ered = −1.94 V vs. SCE, suggesting that A-TATA+ has potential applications as a photoreducant in photoredox catalysis. The new trianguleniums show promising dye properties with intense absorption in the blue region of the visible spectrum at 447 nm (ε = 84000 M−1 cm−1) and efficient fluorescence at 460 nm (ϕf = 45%) in CH2Cl2 solution.Author contributions
M. H. N contributed with writing – original draft & review & editing, conceptualization, investigation, formal analysis, data curation, and methodology. G. P. contributed with writing – review & editing, investigation, formal analysis, data curation, and methodology. A. L. contributed with writing – review & editing, investigation, data curation, and formal analysis. B. W. L. contributed with writing – review & editing, conceptualization, funding acquisition, methodology, supervision, and project administration.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2524713 MeO-TATA+ (2) contains the supplementary crystallographic data for this paper.123The supporting data has been provided as part of the supplementary information (SI). The authors have cited additional references within the SI.109–122 Supplementary information: experimental details and synthetic procedures, NMR spectra, HRMS spectra, detailed absorption and emission spectra of new compounds, additional computational details, electron difraction methods and crystal structure data. See DOI: https://doi.org/10.1039/d6sc01535b.
Acknowledgements
The Novo Nordisk Foundation is acknowledged for financially supporting this work (grant NNF20OC0062176). Electron diffraction experiments are supported by Novo Nordisk Foundation Research Infrastructure (grant NNF220C0074439). The Novo Nordisk Foundation is also acknowledged for NMR facility (grant NNF21OC0067315). G.P. acknowledges the support and computational resources provided by computing@unipi, a Computing Service provided by the University of Pisa.Notes and references
- B. W. Laursen and F. C. Krebs, Chem. – Eur. J., 2001, 7, 1773–1783 CrossRef CAS PubMed.
- V. V. Pavlishchuk and A. W. Addison, Inorg. Chim. Acta, 2000, 298, 97–102 CrossRef CAS.
- D. F. Duxbury, Chem. Rev., 1993, 93, 381–433 CrossRef CAS.
- K. H. Drexhage, in Dye Lasers, ed. F. P. Schäfer, Springer Berlin Heidelberg, Berlin, Heidelberg, 1973, pp. 144–193 Search PubMed.
- L. D. Lavis and R. T. Raines, ACS Chem. Biol., 2008, 3, 142–155 CrossRef CAS PubMed.
- L. D. Lavis, Annu. Rev. Biochem., 2017, 86, 825–843 CrossRef CAS PubMed.
- J. Griffiths, Colour and Constitution of Organic Molecules, Academic Press, 1976 Search PubMed.
- T. Terai and T. Nagano, Pflugers Arch., 2013, 465, 347–359 CrossRef CAS PubMed.
- H. Mustroph, Phys. Sci. Rev., 2020, 5, 20190145 Search PubMed.
- M. J. Schnermann and L. D. Lavis, Curr. Opin. Chem. Biol., 2023, 75, 102335 CrossRef CAS PubMed.
- J. Arden-Jacob, J. Frantzeskos, N. U. Kemnitzer, A. Zilles and K. H. Drexhage, Spectrochim. Acta, Part A, 2001, 57, 2271–2283 CrossRef CAS PubMed.
- K. Kolmakov, V. N. Belov, C. A. Wurm, B. Harke, M. Leutenegger, C. Eggeling and S. W. Hell, Eur. J. Org Chem., 2010, 2010, 3593–3610 CrossRef.
- T. Egawa, Y. Koide, K. Hanaoka, T. Komatsu, T. Terai and T. Nagano, Chem. Commun., 2011, 47, 4162–4164 RSC.
- Y. Koide, Y. Urano, K. Hanaoka, T. Terai and T. Nagano, ACS Chem. Biol., 2011, 6, 600–608 CrossRef CAS PubMed.
- J. B. Grimm, A. J. Sung, W. R. Legant, P. Hulamm, S. M. Matlosz, E. Betzig and L. D. Lavis, ACS Chem. Biol., 2013, 8, 1303–1310 CrossRef CAS PubMed.
- J. B. Grimm, T. A. Brown, A. N. Tkachuk and L. D. Lavis, ACS Cent. Sci., 2017, 3, 975–985 CrossRef CAS PubMed.
- H. Kotani, K. Ohkubo and S. Fukuzumi, J. Am. Chem. Soc., 2004, 126, 15999–16006 CrossRef CAS PubMed.
- C. Nicolas, C. Herse and J. Lacour, Tetrahedron Lett., 2005, 46, 4605–4608 CrossRef CAS.
- K. Ohkubo, K. Mizushima, R. Iwata and S. Fukuzumi, Chem. Sci., 2011, 2, 715 RSC.
- D. S. Hamilton and D. A. Nicewicz, J. Am. Chem. Soc., 2012, 134, 18577–18580 CrossRef CAS PubMed.
- N. A. Romero, K. A. Margrey, N. E. Tay and D. A. Nicewicz, Science, 2015, 349, 1326–1330 CrossRef CAS PubMed.
- A. Joshi-Pangu, F. Lévesque, H. G. Roth, S. F. Oliver, L.-C. Campeau, D. Nicewicz and D. A. Dirocco, J. Org. Chem., 2016, 81, 7244–7249 CrossRef CAS PubMed.
- C. Fischer, C. Kerzig, B. Zilate, O. S. Wenger and C. Sparr, ACS Catal., 2020, 10, 210–215 CrossRef CAS.
- V. Hutskalova and C. Sparr, Org. Lett., 2021, 23, 5143–5147 CrossRef CAS PubMed.
- J. D. Jensen, N. Bisballe, L. Kacenauskaite, M. S. Thomsen, J. Chen, O. Hammerich and B. W. Laursen, J. Org. Chem., 2021, 86, 17002–17010 CrossRef CAS PubMed.
- M. H. Nowack, M. B. Johansen, S. Sams, A. E. Hillers-Bendtsen, K. V. Mikkelsen and B. W. Laursen, Chem. – Eur. J., 2025, 31, e202403451 CrossRef CAS PubMed.
- J. Moutet, J. M. Veleta and T. L. Gianetti, ACS Appl. Energy Mater., 2021, 4, 9–14 CrossRef CAS.
- J. Moutet, D. Mills, M. M. Hossain and T. L. Gianetti, Mater. Adv., 2022, 3, 216–223 RSC.
- J. Moutet, M. H. Nowack, D. Mills, D. L. Lozier, B. W. Laursen and T. L. Gianetti, Mater. Adv., 2023, 4, 4598–4606 RSC.
- J. Moutet, T. H. El-Assaad, R. Kaur, D. D. Mills and T. L. Gianetti, Energy Mater., 2024, 4, 400024 CrossRef CAS.
- J. Bosson, J. Gouin and J. Lacour, Chem. Soc. Rev., 2014, 43, 2824–2840 RSC.
- V. Dujols, F. Ford and A. W. Czarnik, J. Am. Chem. Soc., 1997, 119, 7386–7387 CrossRef CAS.
- L. D. Lavis, T.-Y. Chao and R. T. Raines, ACS Chem. Biol., 2006, 1, 252–260 CrossRef CAS PubMed.
- X. Chen, T. Pradhan, F. Wang, J. S. Kim and J. Yoon, Chem. Rev., 2012, 112, 1910–1956 CrossRef CAS PubMed.
- A. N. Butkevich, G. Y. Mitronova, S. C. Sidenstein, J. L. Klocke, D. Kamin, D. N. H. Meineke, E. D'Este, P.-T. Kraemer, J. G. Danzl, V. N. Belov and S. W. Hell, Angew. Chem., Int. Ed., 2016, 55, 3290–3294 CrossRef CAS PubMed.
- V. Gold and B. W. V. Hawes, J. Chem. Soc., 1951, 2102–2111 RSC.
- J. C. Martin and R. G. Smith, J. Am. Chem. Soc., 1964, 86, 2252–2256 CrossRef CAS.
- B. W. Laursen, F. C. Krebs, M. F. Nielsen, K. Bechgaard, J. B. Christensen and N. Harrit, J. Am. Chem. Soc., 1998, 120, 12255–12263 CrossRef CAS.
- C. Nicolas and J. Lacour, Org. Lett., 2006, 8, 4343–4346 CrossRef CAS PubMed.
- B. W. Laursen and T. J. Sørensen, J. Org. Chem., 2009, 74, 3183–3185 CrossRef CAS PubMed.
- A. Wallabregue, D. Moreau, P. Sherin, P. Moneva Lorente, Z. Jarolímová, E. Bakker, E. Vauthey, J. Gruenberg and J. Lacour, J. Am. Chem. Soc., 2016, 138, 1752–1755 CrossRef CAS PubMed.
- T. J. Sørensen, M. Rosenberg, C. G. Frankær and B. W. Laursen, Adv. Mater. Technol., 2018, 4, 1800561 CrossRef.
- M. Rosenberg, A. K. R. Junker, T. J. Sørensen and B. W. Laursen, ChemPhotoChem, 2019, 3, 233–242 CrossRef CAS.
- C. G. Frankær, M. Rosenberg, M. Santella, K. J. Hussain, B. W. Laursen and T. J. Sørensen, ACS Sens., 2019, 4, 764–773 CrossRef PubMed.
- I. Dalfen, R. I. Dmitriev, G. Holst, I. Klimant and S. M. Borisov, Anal. Chem., 2019, 91, 808–816 CrossRef CAS PubMed.
- C. Herse, D. Bas, F. C. Krebs, T. Bürgi, J. Weber, T. Wesolowski, B. W. Laursen and J. Lacour, Angew. Chem., Int. Ed., 2003, 42, 3162–3166 CrossRef CAS PubMed.
- O. Kel, P. Sherin, N. Mehanna, B. Laleu, J. Lacour and E. Vauthey, Photochem. Photobiol. Sci., 2012, 11, 623–631 CrossRef CAS PubMed.
- S. Pascal, C. Besnard, F. Zinna, L. Di Bari, B. Le Guennic, D. Jacquemin and J. Lacour, Org. Biomol. Chem., 2016, 14, 4590–4594 RSC.
- B. Fabri, D. F. De Rosa, D. J. Black, R. Mucci, A. Krimovs, R. Pal and J. Lacour, Chem. – Eur. J., 2025, 31, e202501212 CrossRef CAS PubMed.
- R. K. Jakobsen, S. G. Stenspil, J. Chen and B. W. Laursen, Chem. Sci., 2025, 16, 7450–7458 RSC.
- M. H. Nowack and B. W. Laursen, Chemphotochem, 2025, 9, 2500183 CrossRef.
- A. Shivalingam, M. A. Izquierdo, A. L. Marois, A. Vyšniauskas, K. Suhling, M. K. Kuimova and R. Vilar, Nat. Commun., 2015, 6, 8178 CrossRef CAS PubMed.
- A. Kotar, B. Wang, A. Shivalingam, J. Gonzalez-Garcia, R. Vilar and J. Plavec, Angew. Chem., Int. Ed., 2016, 55, 12508–12511 CrossRef CAS PubMed.
- A. Shivalingam, A. Vyšniauskas, T. Albrecht, A. J. P. White, M. K. Kuimova and R. Vilar, Chem. – Eur. J., 2016, 22, 4129–4139 CrossRef CAS PubMed.
- B. W. Lewis, N. Bisballe, M. Santella, P. A. Summers, J. B. Vannier, M. K. Kuimova, B. W. Laursen and R. Vilar, Chem. – Eur. J., 2021, 27, 2523–2536 CrossRef CAS PubMed.
- P. A. Summers, B. W. Lewis, J. Gonzalez-Garcia, R. M. Porreca, A. H. M. Lim, P. Cadinu, N. Martin-Pintado, D. J. Mann, J. B. Edel, J. B. Vannier, M. K. Kuimova and R. Vilar, Nat. Commun., 2021, 12, 162 CrossRef CAS PubMed.
- L. Mei, J. M. Veleta and T. L. Gianetti, J. Am. Chem. Soc., 2020, 142, 12056–12061 CrossRef CAS PubMed.
- S. M. Stull, L. Mei and T. L. Gianetti, Synlett, 2021, 33, 1194–1198 Search PubMed.
- L. Mei, J. Moutet, S. M. Stull and T. L. Gianetti, J. Org. Chem., 2021, 86, 10640–10653 CrossRef CAS PubMed.
- M. M. Hossain, A. C. Shaikh, J. Moutet and T. L. Gianetti, Nat. Synth., 2022, 1, 147–157 CrossRef CAS.
- F. Calogero, G. Magagnano, S. Potenti, F. Pasca, A. Fermi, A. Gualandi, P. Ceroni, G. Bergamini and P. G. Cozzi, Chem. Sci., 2022, 13, 5973–5981 RSC.
- M. H. Nowack, J. Moutet, B. W. Laursen and T. L. Gianetti, Synlett, 2024, 35, 307–312 CrossRef CAS.
- M. M. Hossain, A. C. Shaikh, R. Kaur and T. L. Gianetti, J. Am. Chem. Soc., 2024, 146, 7922–7930 CrossRef CAS PubMed.
- K. A. Ryu, T. Reyes-Robles, T. P. Wyche, T. J. Bechtel, J. M. Bertoch, J. Zhuang, C. May, C. Scandore, N. Dephoure, S. Wilhelm, I. Quasem, A. Yau, S. Ingale, A. Szendrey, M. Duich, R. C. Oslund and O. O. Fadeyi, ACS Catal., 2024, 14, 3482–3491 CrossRef CAS.
- P.-Y. Ho, S.-C. Cheng, F. Yu, Y.-Y. Yeung, W.-X. Ni, C.-C. Ko, C.-F. Leung, T.-C. Lau and M. Robert, ACS Catal., 2023, 13, 5979–5985 CrossRef CAS.
- S. Hao, K.-K. Chen, P. Liang, Q. Huang, L. Zhou, Y. Wei and Z. Wei, Nat. Commun., 2025, 16, 6167 CrossRef CAS PubMed.
- S. Hao, Z. Wang, K. K. Chen, Y. Wei, L. Zhou and Z. Wei, ChemCatChem, 2025, 17, e00733 CrossRef CAS.
- R. Gueret, L. Poulard, M. Oshinowo, J. Chauvin, M. Dahmane, G. Dupeyre, P. P. Lainé, J. Fortage and M.-N. Collomb, ACS Catal., 2018, 8, 3792–3802 CrossRef CAS.
- S. Lyu, D. H. Cruz Neto, J. Aguirre-Araque, L. Termeau, F. Camara, S. Cherraben, D. Martin, E. Brémond, T. Pino, M.-H. Ha-Thi, P. P. Lainé, M.-N. Collomb and J. Fortage, Artif. Photosynth., 2025, 1, 251–266 CrossRef CAS.
- J. Moutet, D. D. Mills, D. L. Lozier and T. L. Gianetti, Batter. Supercaps, 2024, 7, e202300519 CrossRef CAS.
- Y. Haketa, S. Sasaki, N. Ohta, H. Masunaga, H. Ogawa, N. Mizuno, F. Araoka, H. Takezoe and H. Maeda, Angew. Chem., Int. Ed., 2010, 49, 10079–10083 CrossRef CAS PubMed.
- C. R. Benson, L. Kacenauskaite, K. L. VanDenburgh, W. Zhao, B. Qiao, T. Sadhukhan, M. Pink, J. Chen, S. Borgi, C.-H. Chen, B. J. Davis, Y. C. Simon, K. Raghavachari, B. W. Laursen and A. H. Flood, Chem, 2020, 6, 1978–1997 CAS.
- J. Chen, S. M. A. Fateminia, L. Kacenauskaite, N. Bærentsen, S. Grønfeldt Stenspil, J. Bredehoeft, K. L. Martinez, A. H. Flood and B. W. Laursen, Angew. Chem., Int. Ed., 2021, 60, 9450–9458 CrossRef CAS PubMed.
- Y. Haketa, K. Yamasumi and H. Maeda, Chem. Soc. Rev., 2023, 52, 7170–7196 RSC.
- P. Deshmukh, S. Satapathy, E. Michail, A. H. Olsson, R. Bushati, R. K. Yadav, M. Khatoniar, J. Chen, G. John, B. W. Laursen, A. H. Flood, M. Y. Sfeir and V. M. Menon, ACS Photonics, 2024, 11, 348–355 CrossRef CAS.
- H. Noguchi, T. Hirose, S. Yokoyama and K. Matsuda, CrystEngComm, 2016, 18, 7377–7383 RSC.
- L. Frédéric, B. Fabri, L. Guénée, F. Zinna, L. Di Bari and J. Lacour, Chem. – Eur. J., 2022, 28, e202201853 CrossRef PubMed.
- B. Fabri, T. Funaioli, L. Frédéric, C. Elsner, E. Bordignon, F. Zinna, L. Di Bari, G. Pescitelli and J. Lacour, J. Am. Chem. Soc., 2024, 146, 8308–8319 CrossRef CAS PubMed.
- T. J. Sørensen, C. B. Hildebrandt, J. Elm, J. W. Andreasen, A. Ø. Madsen, F. Westerlund and B. W. Laursen, J. Mater. Chem., 2012, 22, 4797–4805 RSC.
- T. J. Sørensen, C. B. Hildebrandt, M. Glyvradal and B. W. Laursen, Dyes Pigm., 2013, 98, 297–303 CrossRef.
- T. J. Sørensen, M. F. Nielsen and B. W. Laursen, ChemPlusChem, 2014, 79, 1030–1035 CrossRef.
- D. Dolman and R. Stewart, Can. J. Chem., 1967, 45, 911–924 CrossRef CAS.
- S. Ito, S. Kikuchi, N. Morita and T. Asao, J. Org. Chem., 1999, 64, 5815–5821 CrossRef CAS.
- S. Ito, S. Kikuchi, N. Morita and T. Asao, Bull. Chem. Soc. Jpn., 1999, 72, 839–849 CrossRef CAS.
- K. Okamoto, K. i. Takeuchi, K. Komatsu, Y. Kubota, R. Ohara, M. Arima, K. Takahashi, Y. Waki and S. Shirai, Tetrahedron, 1983, 39, 4011–4024 CrossRef CAS.
- M. R. Feldman and W. C. Flythe, J. Org. Chem., 1978, 43, 2596–2600 CrossRef CAS.
- N. G. Limas and T. A. Manz, RSC Adv., 2016, 6, 45727–45747 RSC.
- T. A. Manz and N. G. Limas, RSC Adv., 2016, 6, 47771–47801 RSC.
- N. G. Limas and T. A. Manz, RSC Adv., 2018, 8, 2678–2707 RSC.
- T. J. Sørensen, E. Thyrhaug, M. Szabelski, R. Luchowski, I. Gryczynski, Z. Gryczynski and B. W. Laursen, Methods Appl. Fluoresc., 2013, 1, 25001 CrossRef PubMed.
- B. P. Maliwal, R. Fudala, S. Raut, R. Kokate, T. J. Sørensen, B. W. Laursen, Z. Gryczynski and I. Gryczynski, PLoS One, 2013, 8, e63043 CrossRef CAS PubMed.
- R. M. Rich, D. L. Stankowska, B. P. Maliwal, T. J. Sørensen, B. W. Laursen, R. R. Krishnamoorthy, Z. Gryczynski, J. Borejdo, I. Gryczynski and R. Fudala, Anal. Bioanal. Chem., 2013, 405, 2065–2075 CrossRef CAS PubMed.
- R. Chib, M. Mummert, I. Bora, B. W. Laursen, S. Shah, R. Pendry, I. Gryczynski, J. Borejdo, Z. Gryczynski and R. Fudala, Anal. Bioanal. Chem., 2016, 408, 3811–3821 CrossRef CAS PubMed.
- A. Baibek, Z. Konieczna, M. Üçüncü, Z. S. Alghamdi, R. Sharma, M. H. Horrocks and M. Bradley, Chem. Biomed. Imaging, 2025, 3, 45–50 CrossRef CAS PubMed.
- K. Kumar, T. H. Braunstein, P. Hernandez-Varas, M. B. Liisberg and B. W. Laursen, J. Mater. Chem. B, 2025, 13, 13062–13074 RSC.
- C. Viedma-Barba, E. M. Ortega-Naranjo, F. Movilla, A. Reinoso, M. Andrades-Amate, P. Peñalver, J. C. Morales, J. A. González-Vera, M. Gutiérrez-Rodríguez and A. Orte, Sci. Rep., 2025, 16, 1925 CrossRef PubMed.
- Y. Zhang, J. Kæstel-Hansen, D. Teze, G. Huang, S. Schoffelen, M. Lisby, M. Zhang, N. S. Hatzakis and M. Meldal, Angew. Chem., Int. Ed., 2025, 64, e202504862 CrossRef CAS PubMed.
- K. Rurack and M. Spieles, Anal. Chem., 2011, 83, 1232–1242 CrossRef CAS PubMed.
- M. Fischer and J. Georges, Chem. Phys. Lett., 1996, 260, 115–118 CrossRef CAS.
- B. W. Laursen, J. Reynisson, K. V. Mikkelsen, K. Bechgaard and N. Harrit, Photochem. Photobiol. Sci., 2005, 4, 568–576 CrossRef CAS PubMed.
- L. Kacenauskaite, S. G. Stenspil, A. H. Olsson, A. H. Flood and B. W. Laursen, J. Am. Chem. Soc., 2022, 144, 19981–19989 CrossRef CAS PubMed.
- S. G. Stenspil, J. Chen, M. B. Liisberg, A. H. Flood and B. W. Laursen, Chem. Sci., 2024, 15, 5531–5538 RSC.
- W.-C. Sun, K. R. Gee and R. P. Haugland, Bioorg. Med. Chem. Lett., 1998, 8, 3107–3110 CrossRef CAS PubMed.
- D. Chercka, S.-J. Yoo, M. Baumgarten, J.-J. Kim and K. Müllen, J. Mater. Chem. C, 2014, 2, 9083–9086 RSC.
- T. Jiang, J. Qin, J. Lin, X. Lin, W. Liu, Z. Xie, C. Redshaw, Z. Zhao and X. Feng, ACS Appl. Mater. Interfaces, 2025, 17, 43339–43351 CrossRef CAS PubMed.
- Y. Liu, J. Zhang, X. Wang, Z. Xie, H. Zheng, S. Zhang, X. Cai, Y. Zhao, C. Redshaw, Y. Min and X. Feng, J. Org. Chem., 2024, 89, 3319–3330 CrossRef CAS PubMed.
- X. Wu, S. Ni, C.-H. Wang, W. Zhu and P.-T. Chou, Chem. Rev., 2025, 125, 6685–6752 CrossRef CAS PubMed.
- M. Rosenberg, K. R. Rostgaard, Z. Liao, A. Ø. Madsen, K. L. Martinez, T. Vosch and B. W. Laursen, Chem. Sci., 2018, 9, 3122–3130 RSC.
- H. E. Gottlieb, V. Kotlyar and A. Nudelman, J. Org. Chem., 1997, 62, 7512–7515 CrossRef CAS PubMed.
- Z. Gryczynski and I. Gryczynski, Practical Fluorescence Spectroscopy, Taylor & Francis Group, 2019 Search PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, G. A. Petersson, H. Nakatsuji, X. Li, M. Caricato, A. V. Marenich, J. Bloino, B. G. Janesko, R. Gomperts, B. Mennucci, H. P. Hratchian, J. V. Ortiz, A. F. Izmaylov, J. L. Sonnenberg, D. Williams-Young, F. Ding, F. Lipparini, F. Egidi, J. Goings, B. Peng, A. Petrone, T. Henderson, D. Ranasinghe, V. G. Zakrzewski, J. Gao, N. Rega, G. Zheng, W. Liang, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, K. Throssell, J. A. Jr, J. E. Peralta, F. Ogliaro, M. J. Bearpark, J. J. Heyd, E. N. Brothers, K. N. Kudin, V. N. Staroverov, T. A. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. P. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, J. M. Millam, M. Klene, C. Adamo, R. Cammi, J. W. Ochterski, R. L. Martin, K. Morokuma, O. Farkas, J. B. Foresman and D. J. Fox, 2016, Gaussian 16, Revision C.01, Wallingford, CT Search PubMed.
- T. A. Manz and N. G. Limas, ChargeMol program for performing DDEC analysis, Version 3.5, 2017, ddec.sourceforge.net Search PubMed.
- S. Ito, F. J. White, E. Okunishi, Y. Aoyama, A. Yamano, H. Sato, J. D. Ferrara, M. Jasnowski and M. Meyer, CrystEngComm, 2021, 23, 8622–8630 RSC.
- Rigaku Oxford Difraction Ltd, Rigaku Corporation, Wroclaw, Poland, 2025, CrysAlisPro.
- O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, J. A. K. Howard and H. Puschmann, J. Appl. Crystallogr., 2009, 42, 339–341 CrossRef CAS.
- G. Sheldrick, Acta Crystallogr. C, 2015, 71, 3–8 CrossRef PubMed.
- G. Sheldrick, Acta Crystallogr. A, 2015, 71, 3–8 CrossRef PubMed.
- L. M. Peng, Micron, 1999, 30, 625–648 CrossRef CAS.
- F. H. Allen and I. J. Bruno, Acta Crystallogr. B, 2010, 66, 380–386 CrossRef CAS PubMed.
- D. N. Rainer and J. Hester, CIVET – CIF Validation and Editing Tool, Zenodo, 2026, DOI:10.5281/zenodo.18178135.
- S. Ito, S. Kikuchi, N. Morita and T. Asao, Chem. Lett., 1996, 25, 175–176 CrossRef.
- E. M. Arnett, R. A. Flowers, R. T. Ludwig, A. E. Meekhof and S. A. Walek, J. Phys. Org. Chem., 1997, 10, 499–513 CrossRef CAS.
- CCDC 2524713: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qr5c1.
| This journal is © The Royal Society of Chemistry 2026 |