Open Access Article
Open Access ArticleCapturing elusive alkenyl radicals in the Wharton reaction and its interrupted cyclization
Kornkamon Akkarasereenon,
Yichen Liu,
Yin Tung Lai and
Rongbiao Tong *
*
Department of Chemistry, The Hong Kong University of Science and Technology, Clearwater Bay, Kowloon, Hong Kong, China. E-mail: rtong@ust.hk; Fax: +86 23581594; Tel: +86 23587357
First published on 20th April 2026
Abstract
The Wharton reaction is a widely used transformation in organic synthesis, enabling the conversion of α,β-epoxy ketones into versatile allylic alcohols. Despite its discovery more than 60 years ago, the detailed mechanism has remained elusive and controversial. While the prevailing mechanism involves the intermediate of an alkenyl anion, several sporadic reports have challenged this pathway, instead suggesting the involvement of an alkenyl radical. Yet no direct evidence has been provided for either species. Herein, we report the first direct capture of the elusive alkenyl radical intermediate in the Wharton reaction using well-established radical traps, including TEMPO (a persistent radical scavenger), PhSSPh, Me3SnSnMe3, and a vinyl cyclopropane radical clock. These experiments furnish the first unambiguous chemical evidence for the generation of alkenyl radicals and effectively rule out the alkenyl anion pathway. Furthermore, we discovered an interrupted Wharton reaction in which the alkenyl radicals are efficiently intercepted by tethered arenes under the standard Wharton reaction conditions, leading to novel cyclized products. These cycloadducts offer valuable intermediates for the total synthesis of polycyclic diarylheptanoid musellarins.
Introduction
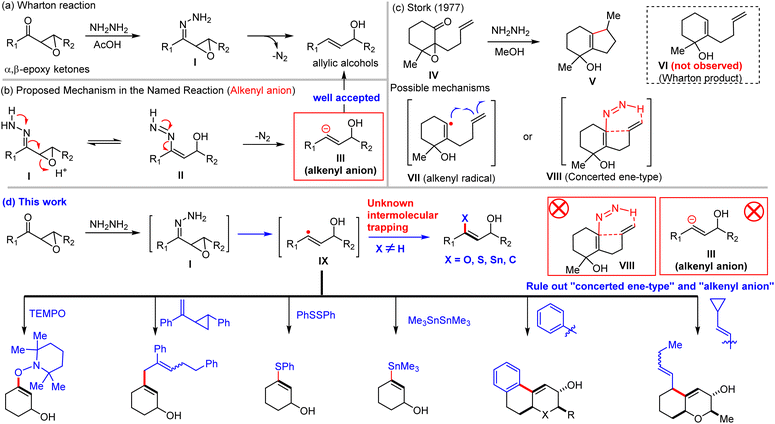
The Wharton reaction (WR), also known as Wharton rearrangement and Wharton olefin synthesis, was discovered in 1961 as a hydrazine-mediated reduction of α,β-epoxy ketones to afford allylic alcohols (Scheme 1a).1 The transformation has found widespread application in the total synthesis of natural products,2–10 as exemplified by O'Doherty's de novo synthesis of various (deoxy-) sugars via Achmatowicz rearrangement and the Wharton reaction,8,9 Lee's synthesis of platensimycin and platencin,2 Tu's construction of morphine/codeine precursors,7 and the use of the Wharton reaction on steroid substrates by Zhang5 and Gademann4 to access chlionastatins A/B and withanolide A, respectively. These examples highlight the reaction's broad synthetic utility and remarkable functional group tolerance under mild conditions.Despite its long-standing utility, the detailed mechanism—particularly the nitrogen extrusion step and the formation of the C(sp2)–H bond—has remained controversial for over six decades. The prevailing mechanism involves generation of an alkenyl anion intermediate (III), followed by protonation,11–13 analogous to the Wolff–Kishner reduction of ketones under strong basic conditions.14 However, this proposal is questionable, as the highly basic alkenyl anion (pKa of vinyl C-H ≈ 43)15 would be rapidly protonated in the protic reaction medium (acetic acid, methanol, and hydrazine hydrate). This anion pathway was first challenged in the 1970s by Ohloff16 and Stork's17,18 observation of cyclized product V (Scheme 1c), which could not arise from alkenyl anion addition to an unactivated alkene. To rationalize the formation of the cyclized product, Stork proposed either alkenyl radical (VII) cyclization or a concerted ene-type process (VIII). These two pathways could not be distinguished at the time, and Ohloff/Stork's findings were largely regarded as exceptional. Subsequent literature, including major reference studies (e.g., Kürti/Czakó, 2005; Jørgensen, 2010),11–13 has continued to favor the alkenyl anion mechanisms, while internet sources such as Wikipedia and Google Search (AI Mode) suggest the involvement of either a vinyl anion or radical intermediates. The lack of direct evidence for either species has sustained this long-standing mechanistic controversy. Herein, we report the first direct capture of the elusive alkenyl radical19,20 in the Wharton reaction using well-established radical traps: TEMPO (persistent radical scavenger), PhSSPh, Me3SnSnMe3, and vinyl cyclopropane (radical clock). These experiments provide unambiguous chemical evidence for the generation of short-lived alkenyl radicals, thereby ruling out the alkenyl anion and concerted ene-type pathways (Scheme 1d). Furthermore, we disclose an interrupted Wharton reaction, in which the transient alkenyl radicals are efficiently intercepted by tethered arenes under classical conditions, yielding novel cyclized products. This new transformation holds promise for the total synthesis of musellarin A21,22 and would be useful for the total synthesis of ergoline indole alkaloids.23–25
Results and discussion
Capturing alkenyl radicals in the Wharton reaction
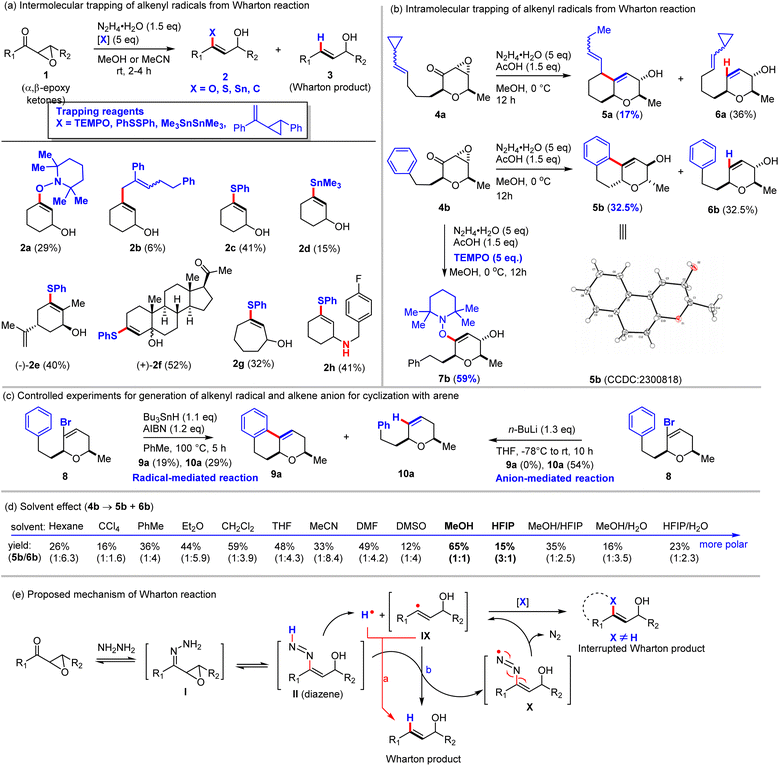
Ohloff and Stork's observation of cyclized products clearly refuted the alkenyl anion pathway for the specific substrates examined, raising the key question whether the Wharton reaction proceeds via an anionic, radical, or concerted mechanism—and whether the mechanism is substrate-dependent. To address this ambiguity, we designed and prepared a series of α,β-epoxy ketones (1a–1g) and investigated radical trapping under standard Wharton reaction conditions (Scheme 2a). To our delight, treatment of α,β-epoxycyclohexanone (1a) with hydrazine hydrate in the presence of TEMPO [(2,2,6,6-tetramethylpiperidin-1-yl)oxyl] afforded the TEMPO-trapped adduct 2a in 29% yield, along with the expected volatile Wharton product 3a (>30% yield). This experiment provided the first direct evidence for the involvement of an alkenyl radical intermediate26–30 in the Wharton reaction. To further corroborate this finding, we employed a vinylcyclopropane derivative as a radical clock.31,32 The reaction of α,β-epoxycyclohexanone (1a) under these conditions delivered the ring-opening adduct 2b in 6% yield, confirming that the transient alkenyl radical can be captured with vinylcyclopropane. We next examined additional established radical traps. Diphenyl disulfide (PhSSPh)33,34 and hexamethylditin (Me3SnSnMe3)35–37 proved effective, with optimized conditions [N2H4·H2O (1.5 equiv.), radical trap (5 equiv.), no acetic acid], to yield alkenyl sulfide 2c (41% yield) and alkenyl stannane 2d (15% yield) from 1a. Notably, diphenyl disulfide outperformed TEMPO, the vinylcyclopropane radical clock, and hexamethylditin, and thus it was used as the preferred trap for broader substrate scope. Pleasingly, α,β-epoxy ketones derived from carvone, progesterone and cycloheptenone furnished the corresponding alkenyl sulfides 2e (40%), 2f (52% yield), and 2g (32% yield) in good yields. This approach represents a potentially valuable new method for preparing synthetically useful alkenyl sulfides. Furthermore, we extended radical trapping to the aza-Wharton reaction12,38,39 of an appropriate acylaziridine, successfully isolating β-amino alkenyl sulfide 2h in 41% yield when using PhSSPh as the radical trap. This product holds promise as a versatile building block for natural product total synthesis and medicinal chemistry applications. Collectively, these trapping experiments furnish consistent evidence that the Wharton reaction proceeds via a short-lived alkenyl radical intermediate, thereby resolving long-standing mechanistic uncertainty and excluding the alkenyl anion and concerted ene-type pathways. Moreover, this work unveils a mild, in situ method for generating elusive alkenyl radicals—typically accessed only from haloalkenes or alkynes40–44—with broad potential applications in organic synthesis.Intramolecular trapping of alkenyl radicals: interrupted Wharton reaction
Building on the successful intermolecular capture of alkenyl radicals, we next explored intramolecular trapping, as originally observed by Ohloff and Stork, which presents a more stringent test due to the challenge of distinguishing radicals from concerted pathways. To probe this, we first designed substrate 4a, featuring a vinylcyclopropane radical clock tethered in a 1,6-relationship to the α,β-epoxy ketone (Scheme 2b). Under standard Wharton conditions, 4a afforded the cyclopropane ring-opened cyclized product 5a (17% yield) along with the normal Wharton product 6a (36% yield). The absence of the cyclopropane moiety in 5a unambiguously indicates a radical cyclization mechanism involving cyclopropane ring opening, thereby excluding a concerted process and providing strong support for Stork's proposed alkenyl radical intermediate VII (Scheme 1c). We then examined substrate 4b bearing a tethered arene, which was expected to preclude a concerted ene-type cyclization (as proposed by Stork for VIII, Scheme 1c). Treatment of 4b hydrazine hydrate under standard conditions yielded a ∼1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixture of the uncyclized Wharton product 6b (32.5% yield) and the cyclized product 5b (32.5% yield, structure confirmed by X-ray crystallography), which were consistent with an alkenyl radical pathway. Notably, we were able to trap this alkenyl radical with TEMPO (5 eq.) and obtained TEMPO-trapped adduct 7b in 59% yield. To further validate that cyclization proceeds via the alkenyl radical rather than an anion, we designed and synthesized vinyl bromide 8 as a model system and compared its behavior under radical and anionic conditions (Scheme 2c). Under classical radical conditions (AIBN, Bu3SnH, PhMe, 100 °C), vinyl bromide 8 produced a mixture of cyclized product 9a (19% yield) and uncyclized product 10a (29% yield),45,46 mirroring the Wharton reactivity and confirming alkenyl radical involvement. In contrast, generation of the alkenyl anion from 8 using n-BuLi followed by aqueous workup afforded only the uncyclized alkene 10a (54% yield), without detectable cyclized product 9a. These complementary experiments consistently demonstrate that the interrupted cyclization in 4a and 4b arises from an alkenyl radical intermediate, not an alkenyl anion. Finally, solvent effects on the reaction of 4b were investigated (Scheme 2d). Both non-polar and polar (including protic) solvents supported the Wharton reaction, affording a mixture of cyclized and uncyclized products in moderate yields with a varying ratio. The minimal dependence on solvent polarity strongly supports a radical mechanism, as polar solvents would be expected to stabilize charged intermediates and significantly alter product distributions in an ionic pathway.
1 mixture of the uncyclized Wharton product 6b (32.5% yield) and the cyclized product 5b (32.5% yield, structure confirmed by X-ray crystallography), which were consistent with an alkenyl radical pathway. Notably, we were able to trap this alkenyl radical with TEMPO (5 eq.) and obtained TEMPO-trapped adduct 7b in 59% yield. To further validate that cyclization proceeds via the alkenyl radical rather than an anion, we designed and synthesized vinyl bromide 8 as a model system and compared its behavior under radical and anionic conditions (Scheme 2c). Under classical radical conditions (AIBN, Bu3SnH, PhMe, 100 °C), vinyl bromide 8 produced a mixture of cyclized product 9a (19% yield) and uncyclized product 10a (29% yield),45,46 mirroring the Wharton reactivity and confirming alkenyl radical involvement. In contrast, generation of the alkenyl anion from 8 using n-BuLi followed by aqueous workup afforded only the uncyclized alkene 10a (54% yield), without detectable cyclized product 9a. These complementary experiments consistently demonstrate that the interrupted cyclization in 4a and 4b arises from an alkenyl radical intermediate, not an alkenyl anion. Finally, solvent effects on the reaction of 4b were investigated (Scheme 2d). Both non-polar and polar (including protic) solvents supported the Wharton reaction, affording a mixture of cyclized and uncyclized products in moderate yields with a varying ratio. The minimal dependence on solvent polarity strongly supports a radical mechanism, as polar solvents would be expected to stabilize charged intermediates and significantly alter product distributions in an ionic pathway.
Proposed mechanism for the Wharton reaction
Based on the direct experimental evidence for alkenyl radical intermediates presented above and the established literature on diazene decomposition,47–57 we propose a revised, unambiguous, and more detailed mechanism for the Wharton reaction (Scheme 2e). The process begins with dehydrative condensation of α,β-epoxy ketone with hydrazine to form hydrazone I, which tautomerizes to the alkenyl diazene II. Homolytic cleavage of II with release of nitrogen gas initiates the radical pathway, generating the alkenyl radical IX47–57 and hydrogen radical (H˙) in the initiation step. In the absence of external traps, two competing pathways account for product formation. Path a involves direct radical rebound58–61 between alkenyl radical IX and H˙ to deliver the conventional Wharton allylic alcohol. More favorably, however, the reaction of alkenyl radical IX with another molecule of diazene II affords the observed Wharton product along with diazene radical X. Subsequent fragmentation of X releases nitrogen gas and re-generates the alkenyl radical IX sustaining the radical propagation. When an appropriate radical trap (TEMPO, PhSSPh, Me3SnSnMe3, and C(sp2)-based traps such as vinylcyclopropane derivatives) is present either intermolecularly or intramolecularly, the transient alkenyl radical IX is efficiently intercepted, leading to the corresponding interrupted Wharton products, including the cyclized products observed in this work and in Stork's work. This radical mechanism fully accommodates our trapping experiments, the solvent independence of cyclization ratios, the failure of alkenyl anions to cyclize in model studies, and the known behavior of diazene intermediates, thereby resolving the long-standing mechanistic controversy surrounding the Wharton reaction.Interrupted Wharton reaction: development as a general cyclization method
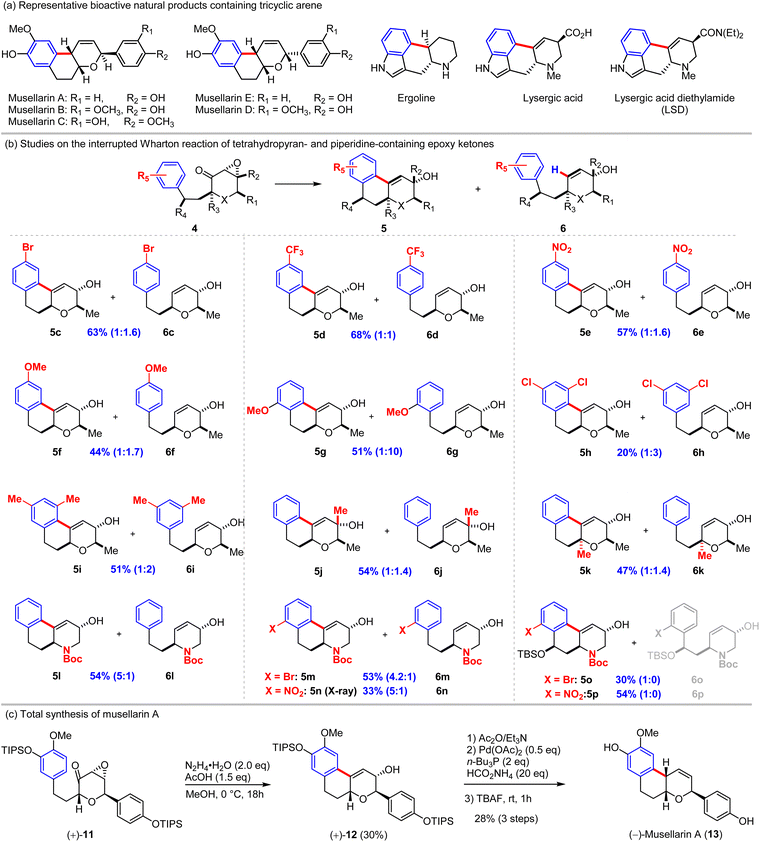
Our studies provided consistent evidence of an alkenyl radical intermediate from the Wharton reaction and demonstrated the feasibility of intercepting the transient species with tethered arenes to afford cyclized products (e.g., 5b). Encouraged by these findings, we sought to develop this interrupted Wharton reaction into a general cyclization strategy with potential applications in the total synthesis of bioactive polycyclic natural products such as musellarins (diarylheptanoid-derived phenols with cytotoxic activity) and ergoline alkaloids (tetracyclic indole derivatives featuring the characteristic ergoline core) (Scheme 3a). To this end, we designed and synthesized a focused series of arene-tethered α,β-epoxy ketones incorporating a pyran or piperidine motif (Scheme 3b). We found that substituents on the aromatic ring—whether electron-withdrawing or electron-donating—exerted negligible influence on the reaction efficiency or the cyclized-to-normal Wharton product ratio (5c–5f and 6c–6f). However, an ortho-methoxy group on the phenyl ring strongly disfavored cyclization, leading predominantly to the normal Wharton product 6g (6g/5g = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, >50% combined yield). We further examined meta-disubstituted benzene substrates. The 2,4-dichlorophenyl derivative 4h afforded the cyclized product 5h in poor overall yields (∼20%) with the normal Wharton product 6h predominating (6h/5h = 3
1, >50% combined yield). We further examined meta-disubstituted benzene substrates. The 2,4-dichlorophenyl derivative 4h afforded the cyclized product 5h in poor overall yields (∼20%) with the normal Wharton product 6h predominating (6h/5h = 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), possibly due to inhibition of alkenyl radical generation by the aryl chloride. In contrast, the 2,4-dimethylphenyl analog 4i delivered a balanced mixture of 5i and 6i (51% combined yield), indicating that steric hindrance from the ortho-methyl group does not significantly impede radical cyclization. Introduction of a methyl substituent on the epoxide (5j/6j) or on the tether linkage (5k/6k) had no discernible effect, further consistent with a radical mechanism.
1), possibly due to inhibition of alkenyl radical generation by the aryl chloride. In contrast, the 2,4-dimethylphenyl analog 4i delivered a balanced mixture of 5i and 6i (51% combined yield), indicating that steric hindrance from the ortho-methyl group does not significantly impede radical cyclization. Introduction of a methyl substituent on the epoxide (5j/6j) or on the tether linkage (5k/6k) had no discernible effect, further consistent with a radical mechanism.
Piperidine-containing α,β-epoxyketones were next evaluated. These substrates underwent smooth interrupted Wharton reactions, furnishing the desired cyclized products 5l, 5m, and 5n in moderate yields (33–54%), accompanied by minor amounts of the normal Wharton products 6l–6n. Notably, substrates 4o and 4p exhibited a strong preference for cyclization, exclusively delivering the cyclized products 5o (30%) and 5p (54%) on milligram and gram scales, respectively. These fully functionalized tetracyclic adducts, with all key stereocenters and functional groups appropriately positioned, represent promising advanced intermediates for the total synthesis of ergoline alkaloids.
This work establishes the interrupted Wharton reaction as a mild, radical-based cyclization method with good substrate tolerance and practical scalability, offering a novel entry into complex polycyclic frameworks relevant to natural product synthesis. To showcase the synthetic application of this newly interrupted Wharton reaction, we accomplished the total synthesis of musellarin A.21,22 As depicted in Scheme 3c, the α,β-epoxy ketone 11 was prepared according to our previously reported procedures (10 steps, see the SI for details)21 and subjected to the Wharton reaction conditions to provide the expected cyclized product 12 in 30% yield (20% yield of the normal Wharton product). Deoxygenation with double-bond migration was found to be challenging when the Myers method62,63 or a hydrogenation/elimination was used. Finally, we found that the Pd-catalyzed deoxygenation of acetylated-allylic alcohol 12 can realize the deoxygenation with alkene migration in 29% yield (2 steps).64–67 Subsequent desilylation with TBAF furnished (−)-musellarin A in 96% yield. The spectroscopic data of musellarin A matched well with those from the previous report.21,22
Conclusions
In summary, this study provides the first direct chemical evidence for the involvement of alkenyl radicals in the Wharton reaction through successful trapping experiments with radical scavengers such as TEMPO, PhSSPh, Me3SnSnMe3, and vinyl cyclopropane as a radical clock. These findings consistently refute the long-standing hypothesis of an alkenyl anion intermediate, which is incompatible with the protic reaction conditions and instead support a radical-mediated mechanism for nitrogen extrusion and C(sp2)–H formation. Furthermore, we have uncovered a novel interrupted Wharton reaction, wherein the transient alkenyl radicals are captured intramolecularly by tethered arenes, leading to cyclized products with high synthetic utility. This methodology has been demonstrated in the concise total synthesis of (–)-musellarin A, highlighting its potential for accessing complex polycyclic structures in natural product synthesis. The elucidation of the radical mechanism in the Wharton reaction opens new avenues for exploiting in situ-generated alkenyl radicals in organic synthesis, particularly for the preparation of functionalized alkenes such as alkenyl sulfides and stannanes, which could serve as versatile building blocks in cross-coupling reactions. Applications of this methodology toward the total synthesis of ergoline alkaloids will be reported in due course.Author contributions
K. A., Y. L., and Y. T. L. performed the experiments and analysed the experimental results and data. K. A. and R. T. prepared the manuscript. R. T. conceptualized, supervised, and funded this work.Conflicts of interest
There are no conflicts to declare.Data availability
Experimental procedures and characterization data are available within this article and its supplementry information (SI). Data are also available from the corresponding author on request.CCDC 2300818, 2300819, and 2520928 contain the supplementary crystallographic data for this paper.68a–c
Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc01519k.
Acknowledgements
This research was financially supported by the Research Grants Council of Hong Kong (C6022-22W, 16308922, 16304023, and 16303524).Notes and references
- P. Wharton and D. Bohlen, J. Org. Chem., 1961, 26, 3615–3616 CrossRef CAS.
- L. Zhu, C. Zhou, W. Yang, S. He, G.-J. Cheng, X. Zhang and C.-S. Lee, J. Org. Chem., 2013, 78, 7912–7929 CrossRef CAS PubMed.
- J. Wang, M. A. Márquez-Cadena and R. Tong, Org. Lett., 2020, 22, 5074–5078 CrossRef CAS.
- R. Liffert, J. Hoecker, C. K. Jana, T. M. Woods, P. Burch, H. J. Jessen, M. Neuburger and K. Gademann, Chem. Sci., 2013, 4, 2851–2857 RSC.
- H. Cui, Y. Shen, Y. Chen, R. Wang, H. Wei, P. Fu, X. Lei, H. Wang, R. Bi and Y. Zhang, J. Am. Chem. Soc., 2022, 144, 8938–8944 CrossRef CAS PubMed.
- J. Liu, R. P. Hsung and S. D. Peters, Org. Lett., 2004, 6, 3989–3992 CrossRef CAS PubMed.
- Q. Zhang, F.-M. Zhang, C.-S. Zhang, S.-Z. Liu, J.-M. Tian, S.-H. Wang, X.-M. Zhang and Y.-Q. Tu, Nat. Commun., 2019, 10, 2507 CrossRef PubMed.
- H.-Y. L. Wang and G. A. O'Doherty, Chem. Commun., 2011, 47, 10251–10253 RSC.
- M. F. Cuccarese, H.-Y. L. Wang and G. A. O'Doherty, Eur. J. Org Chem., 2013, 2013, 3067–3075 CrossRef CAS.
- W. Yu and Z. Jin, J. Am. Chem. Soc., 2002, 124, 6576–6583 CrossRef CAS PubMed.
- C. Dupuy and J. Luche, Tetrahedron, 1989, 45, 3437–3444 CrossRef CAS.
- H. Jiang, N. Holub and K. Anker Jørgensen, Proc. Natl. Acad. Sci. U. S. A., 2010, 107, 20630–20635 CrossRef CAS PubMed.
- L. Kürti and B. Czakó, Strategic Applications of Named Reactions in Organic Synthesis, Elsevier Academic Press, 2005 Search PubMed.
- H. H. Szmant, Angew. Chem., Int. Ed., 1968, 7, 120–128 CrossRef CAS.
- F. G. B. H. Riech, R. Williams, D. A. Evans and D. H. Ripin, pKa Tables, https://organicchemistrydata.org/hansreich/resources/pka/.
- G. Ohloff and G. Uhde, Helv. Chim. Acta, 1970, 53, 531–541 CrossRef CAS.
- G. Stork and P. G. Williard, J. Am. Chem. Soc., 1977, 99, 7067–7068 CrossRef CAS.
- G. Stork, in Current Trends in Organic Synthesis, Elsevier, 1983, pp. 359–370 Search PubMed.
- C. Galli, A. Guarnieri, H. Koch, P. Mencarelli and Z. Rappoport, J. Org. Chem., 1997, 62, 4072–4077 CrossRef CAS.
- G. D. Sargent and M. W. Browne, J. Am. Chem. Soc., 1967, 89, 2788–2790 CrossRef CAS.
- Z. Li, F. C. F. Ip, N. Y. Ip and R. Tong, Chem. - Eur. J., 2015, 21, 11152–11157 CrossRef CAS PubMed.
- Z. Li, T.-F. Leung and R. Tong, Chem. Commun., 2014, 50, 10990–10993 RSC.
- D. Jakubczyk, J. Z. Cheng and S. E. O'Connor, Nat. Prod. Rep., 2014, 31, 1328–1338 RSC.
- P. L. Schiff, Am. J. Pharm. Educ., 2006, 70, 98 CrossRef.
- M. K. Jastrzębski, A. A. Kaczor and T. M. Wróbel, Molecules, 2022, 27, 7322 CrossRef PubMed.
- L. Li, C. Hao, R. Zhai, W. He and C. Deng, Fuel, 2023, 331, 125853 CrossRef CAS.
- D. H. R. Barton, V. N. Le Gloahec and J. Smith, Tetrahedron Lett., 1998, 39, 7483–7486 CrossRef CAS.
- E. G. Bagryanskaya and S. R. A. Marque, Chem. Rev., 2014, 114, 5011–5056 CrossRef CAS PubMed.
- H. Yan, G. Rong, D. Liu, Y. Zheng, J. Chen and J. Mao, Org. Lett., 2014, 16, 6306–6309 CrossRef CAS PubMed.
- X.-F. Xia, S.-L. Zhu, Y.-N. Niu, D. Zhang, X. Liu and H. Wang, Tetrahedron, 2016, 72, 3068–3072 CrossRef CAS.
- D. Griller and K. U. Ingold, Acc. Chem. Res., 1980, 13, 317–323 CrossRef CAS.
- D. C. Nonhebel, Chem. Soc. Rev., 1993, 22, 347–359 RSC.
- S. Nevejans, N. Ballard, J. I. Miranda, B. Reck and J. M. Asua, Phys. Chem. Chem. Phys., 2016, 18, 27577–27583 RSC.
- X. Li, Z. Xu, L. Wang, F. Wang, J. Yang and P. Li, ChemPhotoChem, 2021, 5, 142–148 CrossRef CAS.
- W. R. Bowman, C. F. Bridge, M. O. Cloonan and D. C. Leach, Synlett, 2001, 2001, 0765–0768 CrossRef.
- W. R. Bowman, C. F. Bridge, P. Brookes, M. O. Cloonan and D. C. Leach, J. Chem. Soc., Perkin Trans. 1, 2002, 58–68 CAS.
- W. R. Bowman, M. O. Cloonan, A. J. Fletcher and T. Stein, Org. Biomol. Chem., 2005, 3, 1460–1467 RSC.
- M. Sasaki and A. K. Yudin, J. Am. Chem. Soc., 2003, 125, 14242–14243 CrossRef CAS PubMed.
- S. Silva, P. Rodrigues, I. Bento and C. D. Maycock, J. Org. Chem., 2015, 80, 3067–3074 CrossRef CAS PubMed.
- T. P. M. Goumans, K. van Alem and G. Lodder, Eur. J. Org Chem., 2008, 2008, 435–443 CrossRef.
- F. Le Vaillant and J. Waser, Chem. Sci., 2019, 10, 8909–8923 RSC.
- S. Kumar and B. Ali Shah, Chem.–Asian J., 2023, 18, e202300693 CrossRef CAS PubMed.
- H. F. Piedra and M. Plaza, Photochem. Photobiol. Sci., 2024, 23, 1217–1228 CrossRef CAS PubMed.
- M.-H. Li, X.-R. Shu, Y.-J. Lu, W.-C. Tian, Y. He, Q. Li, Z.-J. Wang, G.-P. Ge and W.-T. Wei, Org. Chem. Front., 2024, 11, 5202–5231 RSC.
- M. T. Erben and S. Bywater, J. Am. Chem. Soc., 1955, 77, 3710–3711 CrossRef CAS.
- J. P. Van Hook and A. V. Tobolsky, J. Am. Chem. Soc., 1958, 80, 779–782 CrossRef CAS.
- D. J. Cram and J. S. Bradshaw, J. Am. Chem. Soc., 1963, 85, 1108–1118 CrossRef CAS.
- T. Tsuji and E. M. Kosower, J. Am. Chem. Soc., 1969, 91, 3375 CrossRef CAS.
- E. M. Kosower and T. Tsuji, J. Am. Chem. Soc., 1971, 93, 1992–1999 CrossRef CAS.
- T. Tsuji and E. M. Kosower, J. Am. Chem. Soc., 1970, 92, 1429–1430 CrossRef CAS.
- P.-K. C. Huang and E. M. Kosower, J. Am. Chem. Soc., 1968, 90, 2367–2376 CrossRef CAS.
- E. M. Kosower, P.-K. C. Huang and T. Tsuji, J. Am. Chem. Soc., 1969, 91, 2325–2329 CrossRef CAS.
- R. O. Hutchins, M. Kacher and L. Rua, J. Org. Chem., 1975, 40, 923–926 CrossRef CAS.
- G. W. Kabalka, J. D. Baker Jr and G. W. Neal, J. Org. Chem., 1977, 42, 512–517 CrossRef CAS.
- G. W. Kabalka and S. T. Summers, J. Org. Chem., 1981, 46, 1217–1218 CrossRef CAS.
- A. Jabbari, E. J. Sorensen and K. Houk, Org. Lett., 2006, 8, 3105–3107 CrossRef CAS PubMed.
- M. L. Shrestha, W. Qi and M. C. McIntosh, J. Org. Chem., 2017, 82, 8359–8370 CrossRef CAS PubMed.
- X. Huang and J. T. Groves, J. Biol. Inorg. Chem., 2017, 22, 185–207 CrossRef CAS PubMed.
- X. He and P. R. O. de Montellano, J. Biol. Chem., 2004, 279, 39479–39484 CrossRef CAS PubMed.
- T. Krämer, M. R. Gyton, I. Bustos, M. J. G. Sinclair, S.-y. Tan, C. J. Wedge, S. A. Macgregor and A. B. Chaplin, J. Am. Chem. Soc., 2023, 145, 14087–14100 CrossRef PubMed.
- K.-B. Cho, H. Hirao, S. Shaik and W. Nam, Chem. Soc. Rev., 2016, 45, 1197–1210 RSC.
- A. G. Myers and B. Zheng, Tetrahedron Lett., 1996, 37, 4841–4844 CrossRef CAS.
- A. G. Myers, B. Zheng and M. Movassaghi, J. Org. Chem., 1997, 62, 7507 CrossRef CAS PubMed.
- J. Tsuji and T. Yamakawa, Tetrahedron Lett., 1979, 20, 613–616 CrossRef.
- G. Majetich, Y. Zhang, X. Tian, J. E. Britton, Y. Li and R. Phillips, Tetrahedron, 2011, 67, 10129–10146 CrossRef CAS.
- Y. Aoyama, Y. Araki and T. Konoike, Synlett, 2001, 2001, 1452–1454 CrossRef.
- M. Welker, S. Woodward and A. Alexakis, Org. Lett., 2010, 12, 576–579 CrossRef CAS PubMed.
- (a) CCDC 2300818: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2h75yw; (b) CCDC 2300819: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2h75zx; (c) CCDC 2520928: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qm78w.
| This journal is © The Royal Society of Chemistry 2026 |