Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Highly Cα-regio-, enantio- and diastereoselective Mukaiyama-type annulation of siloxyfurans: stereodivergent synthesis of multi-stereogenic tricyclic γ-lactones
Lifei Gan†
a,
Zi-Qing Li†b,
Tao Chen†a,
Xuanchen Wanad,
Junyang Zhanga,
Jiangtao Rena,
Ming Jianga,
Penglong Caoa,
Jinhai Huanga,
Yu-Hua Deng *a,
Fangzhi Penga,
Run Tianc,
Yingcheng Wang*a,
Zhihan Zhang*b and
Zhihui Shao
*a,
Fangzhi Penga,
Run Tianc,
Yingcheng Wang*a,
Zhihan Zhang*b and
Zhihui Shao *ad
*ad
aKey Laboratory of Medicinal Chemistry for Natural Resource, Ministry of Education, School of Chemical Science and Technology, School of Pharmacy, State Key Laboratory for Conservation and Utilization of Bio-Resources in Yunnan, Yunnan University, Kunming 650091, China. E-mail: dengyuhua@ynu.edu.cn; ycwang@ynu.edu.cn; zhihui_shao@hotmail.com
bCollege of Chemistry, Central China Normal University, Wuhan, 430079, China. E-mail: zhihanzhang@ccnu.edu.cn
cYunnan University Affiliated Hospital, Yunnan University, Kunming, 650000, China
dSouthwest United Graduate School, Kunming 650092, China
First published on 29th April 2026
Abstract
The first Cα-selective asymmetric reaction of 2-siloxyfurans, a class of versatile nucleophiles, has been developed with both high enantioselectivity and diastereoselectivity. Moreover, by changing the achiral co-catalyst to a newly developed combined co-catalyst, a rare diastereoselective reversal was achieved, selectively yielding the thermodynamically less stable diastereomer. DFT and experimental studies reveal that the observed Cα-selectivity results from dispersion and electrostatic interactions between 2-siloxyfurans and the electrophile/catalyst, while the diastereodivergent synthesis arises from a divergent C–O bond formation via dynamic kinetic lactonization-driven epimerization. This work not only provides a method to overcome the challenges of Cα-selective asymmetric reactions of 2-siloxyfurans, but also offers a stereodivergent synthesis of chiral tricyclic γ-lactones. Importantly, the resulting chiral tricyclic γ-lactones are not only the core structures in natural products and bioactive molecules but also serve as an appealing platform for diversity-oriented synthesis (DOS), streamlining the construction of other valuable enantioenriched compounds.
Introduction
Stereodivergent synthesis of multi-stereogenic chiral compounds is of significant importance, as different absolute or relative configurations often dictate distinct physiological and pharmacological profiles.1–3 Multi-stereogenic γ-lactones are widely found in natural products, pharmaceuticals, flavors, and fragrances.4–8 Thus, the development of catalytic asymmetric methods for constructing these structures, especially in a stereodivergent manner, is of great interest. Although many elegant methods have been developed for the synthesis of enantioenriched γ-butyrolactones with one or two stereocenters on the γ-butyrolactone ring,9–18 there are few catalytic asymmetric protocols for the diastereo- and enantioselective synthesis of γ-butyrolactones with three stereocenters. The tricyclic γ-lactone core [A], shown in Fig. 1a, represents a privileged subclass of γ-lactone architectures.19–24 Nevertheless, there is only one catalytic enantioselective approach available—based on asymmetric transfer hydrogenation followed by syn-selective lactonization—to deliver a single diastereomer of such tricyclic γ-lactones.16–18 To date, there has been no approach involving catalytic asymmetric carbon–carbon (C–C) bond formation25 that would provide a more direct and efficient route to these chiral tricyclic γ-lactones. Moreover, despite remarkable progress in the area of stereodivergent synthesis,26–30 a diastereodivergent synthesis of these architectures has not been achieved. Thus, it is highly desirable to establish a modular and stereodivergent catalytic strategy to access structural diversified chiral tricyclic γ-lactone scaffolds based on C–C bond formation from simple building blocks.2-Siloxyfurans, as unstabilized dienolate nucleophiles, are a class of versatile platforms in organic synthesis.31,32 In asymmetric catalysis, the Cγ-selective vinylogous Mukaiyama-type reactions of 2-siloxyfurans with various electrophiles have been widely exploited to afford chiral γ,γ-disubstituted butenolides, due to the existence of a thermodynamically more stable conjugated π-system in the resulting products (Fig. 1b).33–40 In contrast, catalytic asymmetric Cα-selective Mukaiyama-type reactions have lagged behind and remain a significant challenge.41–43 In 2012, Feringa and Hartwig independently reported the asymmetric Cα-selective allylation reactions of 2-siloxyfurans using Pd and Ir catalysis, respectively.41,42 Both studies indicate that 2-siloxyfurans are activated by the carboxylate leaving group of the allylic electrophiles to generate dienolate anions I. In Feringa's Pd catalyst system, hydrogen-bonding interaction between the oxygen anion and the chiral ligand was proposed to play a key role in controlling regioselectivity.41 Despite this insight, both reactions still failed to control the stereochemistry at the 2-siloxyfuran nucleophiles, resulting in either a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 diastereomeric ratio (dr) or producing conjugated γ-butenolides via double bond isomerization. Meanwhile, Mlynarski and co-workers described a chiral Zn(II)-catalyzed asymmetric Mukaiyama aldol reaction to produce conjugated γ-butenolides, in which water-containing solvents played a crucial role in directing Cα-regioselectivity.43 However, the enantioselectivity remained modest, with only up to 70% ee. Currently, there is still no general catalytic protocol that can achieve both high enantio- and diastereo-selectivity in the Cα-selective reaction of 2-siloxyfurans, and a diastereodivergent version also remains an elusive goal, due to multiple challenges in controlling regioselectivity and enantioselectivity as well as tuning diastereoselectivity. These challenges have severely limited the potential applications of the Cα-selective reaction of 2-siloxyfurans.
1 diastereomeric ratio (dr) or producing conjugated γ-butenolides via double bond isomerization. Meanwhile, Mlynarski and co-workers described a chiral Zn(II)-catalyzed asymmetric Mukaiyama aldol reaction to produce conjugated γ-butenolides, in which water-containing solvents played a crucial role in directing Cα-regioselectivity.43 However, the enantioselectivity remained modest, with only up to 70% ee. Currently, there is still no general catalytic protocol that can achieve both high enantio- and diastereo-selectivity in the Cα-selective reaction of 2-siloxyfurans, and a diastereodivergent version also remains an elusive goal, due to multiple challenges in controlling regioselectivity and enantioselectivity as well as tuning diastereoselectivity. These challenges have severely limited the potential applications of the Cα-selective reaction of 2-siloxyfurans.
Herein we describe the first successful Cα-regioselective, enantioselective and diastereodivergent reaction of 2-siloxyfurans through the development of new catalytic systems (Fig. 1c). The reaction of unstabilized 2-siloxyfuran nucleophiles with oxabicyclic alkenes44–46 as electrophilic partners proceeded with high regio-, enantio- and diastereoselectivity via a cascade annulation, providing a diastereodivergent synthesis of chiral tricyclic lactone frameworks with three stereocenters in a single step. Different from the traditional diastereodivergent C–C bond formation strategy,47–51 this stereodivergent approach involves common stereoselective C–C bond formation, followed by divergent lactonization processes (C–O bond formation) directed by a co-catalyst. By tuning the co-catalyst, a rare diastereoselective reversal occurs, selectively yielding the thermodynamically less stable diastereomer. In contrast with traditional stoichiometric thermodynamic-driven epimerization,13,52 this stereoinversion involves a catalytic dynamic kinetic lactonization-driven epimerization process. Furthermore, the observed Cα-regioselectivity is attributed to the distinctive dispersion and electrostatic interactions between 2-siloxyfurans and the electrophile/catalyst. The resulting chiral tricyclic γ-lactones as new versatile platforms for diversity-oriented synthesis53,54 have been demonstrated by the diastereodivergent synthesis of multi-stereogenic monocyclic γ-lactones and diverse asymmetric syntheses of chiral quaternary carbon-embedded tricyclic γ-lactones as well as other important chiral frameworks, such as dihydropyridazin-3(2H)-ones and dioxabicyclo[3.2.1]octanes,55a thus significantly expanding the chemical space of stereochemical diversity, skeletal diversity, and functional-group diversity.
Results and discussion
Reaction development
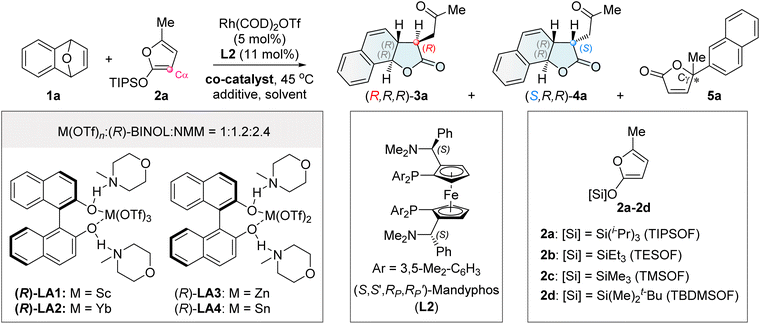
We initiated our studies by performing the reaction of oxabenzonorbornadiene 1a with 2-siloxyfuran 2a (TIPSOF) at 45 °C in the presence of Rh(COD)2OTf and Mandyphos L2 as the chiral catalyst (Table 1). The reaction led to the formation of phenol via the decomposition of oxabenzonorbornadiene 1a (entry 1). Interestingly, the reaction did not proceed when ZnF2 was used to activate 2-siloxyfuran 2a to generate the corresponding dienolate anion I (entry 2).41,42 After extensive efforts,55b the desired chiral tricyclic γ-lactone product 3a with an R,R,R-configuration56a–e was finally obtained in 71% yield with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Cα/Cγ selectivity, >20
1 Cα/Cγ selectivity, >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr and 92% ee (entry 3), when Zn(OTf)2 was used as a co-catalyst.57 Reducing the loading of Zn(OTf)2 to 20 mol% led to a sharp decrease in diastereoselectivity, indicating that Zn(OTf)2, acting as a Lewis acid, was involved in the key stereodetermining transition state, either by activating the bridgehead oxygen to facilitate the Rh oxidative insertion46,57 or by activating the nucleophile (entry 4). When Zn(OTf)2 was combined with ZnF2—an additive that promotes the desilylation of 2-siloxyfuran to dienolate anion I—the yield and diastereoselectivity significantly decreased (entry 5). This indicates that dienolate anion I is not an effective intermediate in this reaction. The silyl groups of 2-siloxyfurans affected both reactivity and selectivity. Smaller silyl groups, such as triethylsilyl (TES, 2b) and trimethylsilyl (TMS, 2c), resulted in decomposition of both substrates (entry 6), whereas the bulkier TBDMS group (2d) led to a poor diastereomeric ratio of 2
1 dr and 92% ee (entry 3), when Zn(OTf)2 was used as a co-catalyst.57 Reducing the loading of Zn(OTf)2 to 20 mol% led to a sharp decrease in diastereoselectivity, indicating that Zn(OTf)2, acting as a Lewis acid, was involved in the key stereodetermining transition state, either by activating the bridgehead oxygen to facilitate the Rh oxidative insertion46,57 or by activating the nucleophile (entry 4). When Zn(OTf)2 was combined with ZnF2—an additive that promotes the desilylation of 2-siloxyfuran to dienolate anion I—the yield and diastereoselectivity significantly decreased (entry 5). This indicates that dienolate anion I is not an effective intermediate in this reaction. The silyl groups of 2-siloxyfurans affected both reactivity and selectivity. Smaller silyl groups, such as triethylsilyl (TES, 2b) and trimethylsilyl (TMS, 2c), resulted in decomposition of both substrates (entry 6), whereas the bulkier TBDMS group (2d) led to a poor diastereomeric ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, along with a small amount of 5a (entry 7).56f Both the anion and cation of the metal Lewis acid salts influenced reactivity and selectivity. For example, Zn(OAc)2 was unreactive (entry 8). Intriguingly, KOTf as a co-catalyst afforded the major diastereomeric tricyclic γ-lactone 4a with an S,R,R-configuration, albeit with only moderate yield and enantioselectivity (entry 9).
1, along with a small amount of 5a (entry 7).56f Both the anion and cation of the metal Lewis acid salts influenced reactivity and selectivity. For example, Zn(OAc)2 was unreactive (entry 8). Intriguingly, KOTf as a co-catalyst afforded the major diastereomeric tricyclic γ-lactone 4a with an S,R,R-configuration, albeit with only moderate yield and enantioselectivity (entry 9).
| Entry | Co-catalyst | Solvent, additive | Yield (%) | dr (3a/4a) | ee (%) (3a/4a) | rr (Cα/Cγ) |
|---|---|---|---|---|---|---|
a Unless otherwise noted, the reaction was conducted with 1a (0.1 mmol), 2a (0.5 mmol), Rh(COD)2OTf (5 mol%), L2 (11 mol%), co-catalyst (50 mol%), and additive (2.0 equiv. ZnF2 or 3.5 equiv. TFE), in indicated solvent (2 mL, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 mixed solvents) under an argon atmosphere at 45 °C. The yield refers to the combined yield of 3a and 4a. The dr value was determined by 1H-NMR analysis for the ratio of 3a and 4a. The ee value of 3a/4a was determined by chiral HPLC analysis. The regioselective ratio (rr) was determined by 1H-NMR spectroscopy for the ratio of (3a + 4a) and 5a.b Co-catalyst (20 mol%) was used.c Using 2b or 2c (0.5 mmol) in place of 2a.d Using 2d (0.5 mmol) in place of 2a. NR = no reaction. ND = not detected. w/o = without. 1 mixed solvents) under an argon atmosphere at 45 °C. The yield refers to the combined yield of 3a and 4a. The dr value was determined by 1H-NMR analysis for the ratio of 3a and 4a. The ee value of 3a/4a was determined by chiral HPLC analysis. The regioselective ratio (rr) was determined by 1H-NMR spectroscopy for the ratio of (3a + 4a) and 5a.b Co-catalyst (20 mol%) was used.c Using 2b or 2c (0.5 mmol) in place of 2a.d Using 2d (0.5 mmol) in place of 2a. NR = no reaction. ND = not detected. w/o = without. |
||||||
| 1 | None | DCE/CHCl3, w/o | ND | — | — | — |
| 2 | ZnF2 | DCE/CHCl3, w/o | NR | — | — | — |
| 3 | Zn(OTf)2 | DCE/CHCl3, w/o | 71 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1 1 |
92/— | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1 1 |
| 4b | Zn(OTf)2 | DCE/CHCl3, w/o | 52 | 3.7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
92/91 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 5 | Zn(OTf)2 | DCE/CHCl3, ZnF2 | 39 | 4.6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
87/98 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 6c | Zn(OTf)2 | DCE/CHCl3, w/o | ND | — | — | — |
| 7d | Zn(OTf)2 | DCE/CHCl3, w/o | 65 | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
80/91 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 8 | Zn(OAc)2 | DCE/CHCl3, w/o | ND | — | — | — |
| 9 | KOTf | DCE/CHCl3, w/o | 37 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 13 |
76/59 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 10b | (R)-LA1 | DCE/CHCl3, w/o | 77 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 11 11 |
79/97 | 9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 11b | (R)-LA2 | DCE/CHCl3, w/o | 34 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4.6 4.6 |
92/93 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 12b | (R)-LA3 | DCE/CHCl3, w/o | 50 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
96/97 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 13b | (R)-LA4 | DCE/CHCl3, w/o | 45 | <1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
—/93 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 14b | (R)-LA4 | CHCl3, TFE | 76 | <1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 20 20 |
—/95 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/b_char_2009.gif) 1 1 |
| 15b | (±)-LA4 | CHCl3, TFE | 71 | <1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
—/95 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 16b | w/o (R)-BINOL in (R)-LA4 | CHCl3, TFE | 73 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9 9 |
—/94 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 17b | w/o NMM in (R)-LA4 | CHCl3, TFE | 61 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.4 1.4 |
97/93 | >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
| 18b | w/o Sn(OTf)2 in (R)-LA4 | CHCl3, TFE | NR | — | — | — |
Inspired by the potential capability of Kobayashi's combined chiral Lewis acid catalysts based on rare-earth metals, such as (R)-LA1 and (R)-LA2, in asymmetric reactions,58,59 we evaluated several related systems to improve the inversion of diastereoselectivity toward (S,R,R)-4a. The combined Lewis acids (R)-LA1 (Sc(OTf)3-(R)-BINOL-NMM) and (R)-LA2 (Yb(OTf)3-(R)-BINOL-NMM) as co-catalysts successfully switched the diastereoselectivity toward (S,R,R)-4a as the major diastereomer (entries 10 and 11). Encouraged by this result, other combined chiral Lewis acid co-catalysts were examined (entries 12 and 13 and Table S6 in the SI). Among them, (R)-LA4 (Sn(OTf)2-(R)-BINOL-NMM)—a previously unreported catalyst based on main-group metals—provided (S,R,R)-4a with excellent regio-, diastereo-, and enantio-selectivity, albeit in 45% yield (entry 13). The yield was significantly improved to 76% by adding the proton source CF3CH2OH (TFE) in CHCl3, affording the desired product (S,R,R)-4a with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Cα/Cγ selectivity, >20
1 Cα/Cγ selectivity, >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, and 95% ee (entry 14: conditions B).56a–e In contrast to Kobayashi's work where the chirality of BINOL in combined Lewis acids controlled enantioselectivity, the chirality of BINOL in LA4 had no effect on stereoselectivity. Even racemic LA4 (Sn(OTf)2-(±)-BINOL-NMM) afforded excellent results (entry 15 and Table S8 in the SI). The absence of BINOL resulted in the major diastereoisomer 4a with diminished diastereoselectivity (entry 16). No NMM led to a low dr, delivering the mixed products 3a and 4a (entry 17). Without Sn(OTf)2, the reaction did not work (entry 18). This study not only provides an unprecedented method for diastereodivergent asymmetric synthesis but also expands the chemistry of combined Lewis acid catalysis by demonstrating its unique ability to switch diastereocontrol, opening a new avenue in asymmetric synthesis.60
1 dr, and 95% ee (entry 14: conditions B).56a–e In contrast to Kobayashi's work where the chirality of BINOL in combined Lewis acids controlled enantioselectivity, the chirality of BINOL in LA4 had no effect on stereoselectivity. Even racemic LA4 (Sn(OTf)2-(±)-BINOL-NMM) afforded excellent results (entry 15 and Table S8 in the SI). The absence of BINOL resulted in the major diastereoisomer 4a with diminished diastereoselectivity (entry 16). No NMM led to a low dr, delivering the mixed products 3a and 4a (entry 17). Without Sn(OTf)2, the reaction did not work (entry 18). This study not only provides an unprecedented method for diastereodivergent asymmetric synthesis but also expands the chemistry of combined Lewis acid catalysis by demonstrating its unique ability to switch diastereocontrol, opening a new avenue in asymmetric synthesis.60
Mechanistic study
To gain insight into the reaction mechanism underlying the diastereodivergent formation of tricyclic γ-lactones, a series of mechanistic experiments were conducted. First, no direct Cα-epimerization between (R,R,R)-3a and (S,R,R)-4a was observed under standard conditions B and A, respectively (Scheme 1a(i and ii)). Interestingly, upon treatment with lithium hexamethyldisilazide (LiHMDS, 4.0 equiv.) at an elevated temperature (90 °C), (S,R,R)-4a was converted into (R,R,R)-3a in 43% yield with 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, accompanied by a significant decomposition of 4a (Scheme 1b(i)). In contrast, (R,R,R)-3a could not be converted into (S,R,R)-4a (Scheme 1b(ii)).55b These results suggest that the chiral tricyclic γ-lactone (S,R,R)-4a is a thermodynamically less stable isomer, while the thermodynamically stable diastereomer is (R,R,R)-3a.
1 dr, accompanied by a significant decomposition of 4a (Scheme 1b(i)). In contrast, (R,R,R)-3a could not be converted into (S,R,R)-4a (Scheme 1b(ii)).55b These results suggest that the chiral tricyclic γ-lactone (S,R,R)-4a is a thermodynamically less stable isomer, while the thermodynamically stable diastereomer is (R,R,R)-3a.
Next, the reaction evolution for the formation of product (S,R,R)-4a was monitored in deuterated solvent under standard conditions B at various time points. As shown in Fig. 2a, intermediate 6 initially appeared—then its diastereomeric counterpart 7—and subsequently the product (S,R,R)-4a. Notably, intermediate 6 as the major diastereoisomer can also be obtained under modified conditions A in air at room temperature with the catalysis of [Rh]-L2/Zn(OTf)2 (Fig. 2b: 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, 89% yield by 1H-NMR analysis). However, silica-gel chromatographic separation afforded a mixture of intermediates 6 and 7 with a 2
1 dr, 89% yield by 1H-NMR analysis). However, silica-gel chromatographic separation afforded a mixture of intermediates 6 and 7 with a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 3
1 to 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr in 89% yield, indicating that intermediate 6 is readily prone to Cα-racemization. Under conditions B, the isolated mixture of 6 and 7 (2
1 dr in 89% yield, indicating that intermediate 6 is readily prone to Cα-racemization. Under conditions B, the isolated mixture of 6 and 7 (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 3
1 to 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr) led to the product (S,R,R)-4a in 93% yield with >20
1 dr) led to the product (S,R,R)-4a in 93% yield with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. These results indicate that, under conditions B, intermediate 6 with an (S,R,R)-configuration is unlikely to undergo the direct lactonization to form the product (R,R,R)-3a. A significant loss of deuterium content was observed in both 4a and the recovered intermediate mixture 6/7, indicating that during Cα-epimerization from intermediate 6 to 7 and the subsequent lactonization to product (S,R,R)-4a (Fig. 2c), H/D exchange occurs via a deprotonation–reprotonation step.
1 dr. These results indicate that, under conditions B, intermediate 6 with an (S,R,R)-configuration is unlikely to undergo the direct lactonization to form the product (R,R,R)-3a. A significant loss of deuterium content was observed in both 4a and the recovered intermediate mixture 6/7, indicating that during Cα-epimerization from intermediate 6 to 7 and the subsequent lactonization to product (S,R,R)-4a (Fig. 2c), H/D exchange occurs via a deprotonation–reprotonation step.
Under conditions A, a high deuterium retention ratio of 96% was observed in (R,R,R)-d-3a, based on d-2a (93% D) (Fig. 2d).55b High-resolution mass spectrometry (HRMS) further identified the reactive ARO adduct II—not intermediate 6—as the key species leading to (R,R,R)-3a.
Based on the above experimental findings, it was proposed that both dual catalytic systems proceed through a common stereocontrolled transition state for C–C bond formation, followed by divergent lactonization pathways (Fig. 2e). Crucially, the diastereoselective inversion is governed by the synergistic action of both the combined multifunctional co-catalyst LA4 and the proton additive TFE.55b This system promotes the protonation of the ARO-adduct II (to (S,R,R)-6)—thereby suppressing the formation of (R,R,R)-3a—while concurrently enabling a dynamic kinetic lactonization-driven epimerization pathway of (S,R,R)-6 to (R,R,R)-7 that selectively yields the thermodynamically less stable isomer (S,R,R)-4a. This proposed mechanism was supported by the density functional theory (DFT) calculations elucidating the observed diastereoselective reversal (Fig. S6–S8 in SI). DFT results confirm that the combined catalyst (R)-LA4 acts as a multifunctional catalytic system, integrating the capabilities of both a Lewis acid and a proton-transfer catalyst across the process. Computational data indicate that these two diastereomers 6 and 7 are thermodynamically similar, which is consistent with the experimental observation that the epimerization from 6 to 7 occurs relatively easily while maintaining a consistently low dr. Conversely, the pathway from 6 to (R,R,R)-3a was calculated to be energetically disfavored.
According to the results above, DFT calculations were performed to elucidate both regioselectivity and stereoselectivity in the C–C bond formation step (Fig. 3, S4 and S5 in the SI). The computational results were consistent with the experimental observations regarding the regioselectivity and stereoselectivity. Distortion/interaction analysis and the independent gradient model based on Hirshfeld partition (IGMH)61,62 revealed that non-covalent interactions between π-allylic [Rh]* species (IM1) and TIPSOF 2a dominate the energy trends among different transition states. In the most favored transition state TS0-Si-Cα, significant dispersion forces and electrostatic attraction were observed between the furan ring of 2a and the π-allylic [Rh]* species (IM1), contributing to the stabilization of the transition state. In contrast, the three disfavored transition states lacked electrostatic attraction and exhibited diminished dispersion due to steric hindrance from silyl groups, as evidenced by elongated C–C bond lengths in these transition states.
Based on the above results and related studies,44,45,50,57 a rational mechanism is proposed in Fig. 4. The chiral rhodium catalyst ([Rh]*) initially coordinates with oxabenzonorbornadiene 1a, followed by desymmetric oxidative insertion to generate the π-allylic [Rh]* species C (or IM1). Subsequently, a Cα-selective nucleophilic attack of 2a on C proceeds via the favored transition state D (or TS0-Si-Cα), yielding the ARO adduct E. This adduct undergoes direct lactonization to furnish the tricyclic γ-lactone (R,R,R)-3a while regenerating the Rh catalyst. In the presence of a proton source ((R)-LA4 or TFE), adduct E is rapidly protonated to form intermediate 6, which undergoes Cα-epimerization toward intermediate 7, followed by lactonization of 7 to deliver the tricyclic γ-lactone (S,R,R)-4a.
Substrate scope
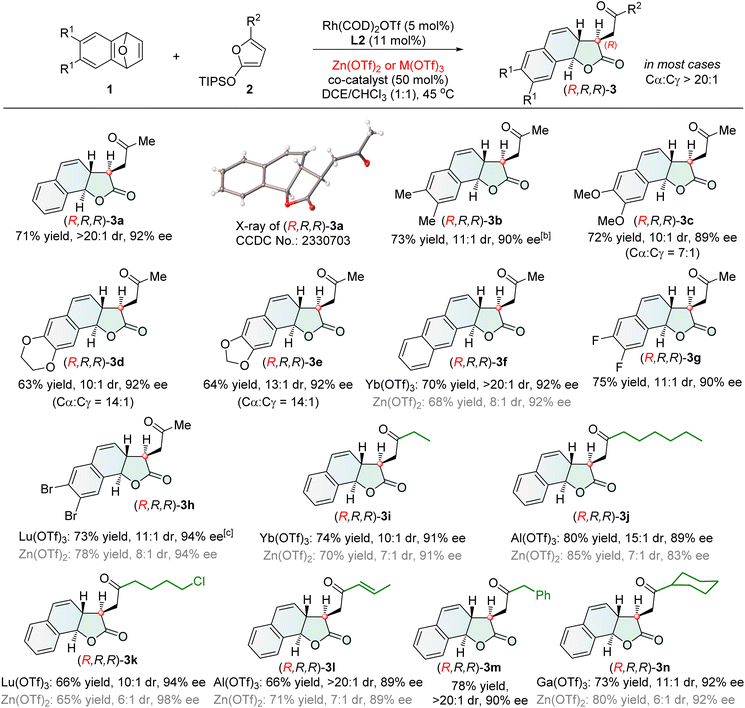
With the optimized chiral catalytic systems and reaction conditions in hand, we investigated the generality of Cα-ARO/lactonization reactions. In the protocol catalyzed by Rh(I)/L2 and Zn(OTf)2, the scope of oxabenzonorbornadienes 1 was firstly examined (Scheme 2). A range of oxabenzonorbornadienes 1 bearing electron-rich, electron-neutral and electron-deficient groups on the benzene ring performed well in this reaction, affording tricyclic γ-lactones 3a–h. The naphthalene-containing substrate 1f smoothly participated in the reaction, and Yb(OTf)3 as a co-catalyst provided the desired tricyclic γ-lactone 3f in 70% yield with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, 92% ee, and >20
1 dr, 92% ee, and >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Cα/Cγ selectivity. Replacing Zn(OTf)2 with Lu(OTf)3 as the co-catalyst improved the diastereoselectivity for 3h from 8
1 Cα/Cγ selectivity. Replacing Zn(OTf)2 with Lu(OTf)3 as the co-catalyst improved the diastereoselectivity for 3h from 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to 11
1 to 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr while maintaining good enantioselectivity. Subsequently, the scope of 2-siloxyfurans 2 was investigated. Various 2-siloxyfurans bearing different substituents performed well in this process, producing the corresponding tricyclic γ-lactones (3a and 3i–n). Functional groups such as halides and olefins were well tolerated (3k and 3l). Notably, the 2-siloxyfuran with R = allyl led to tricyclic γ-lactone 3l featuring a conjugated enone moiety suitable for further transformation, resulting from olefin isomerization of the Cα-ring opening/lactonization product.
1 dr while maintaining good enantioselectivity. Subsequently, the scope of 2-siloxyfurans 2 was investigated. Various 2-siloxyfurans bearing different substituents performed well in this process, producing the corresponding tricyclic γ-lactones (3a and 3i–n). Functional groups such as halides and olefins were well tolerated (3k and 3l). Notably, the 2-siloxyfuran with R = allyl led to tricyclic γ-lactone 3l featuring a conjugated enone moiety suitable for further transformation, resulting from olefin isomerization of the Cα-ring opening/lactonization product.
 | ||
| Scheme 2 Asymmetric synthesis of tricyclic γ-lactones (R,R,R)-3[a]. [a] Unless otherwise noted, the reaction was conducted under the conditions described in entry 3 of Table 1, and the dr value refers to the ratio of 3 and 4. [b] L2 (15 mol%) was used. [c] At 50 °C. | ||
The diastereodivergent Cα-ARO/lactonization reaction, catalyzed by Rh(I)/L2 in combination with the multifunctional co-catalyst (R)-LA4, demonstrated a broad substrate scope. A variety of tricyclic γ-lactones (S,R,R)-4 were obtained in generally good yields, exhibiting excellent diastereoselectivity (>20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr), exclusive Cα-selectivity, and high enantioselectivity (90–98% ee) (Scheme 3).
1 dr), exclusive Cα-selectivity, and high enantioselectivity (90–98% ee) (Scheme 3).
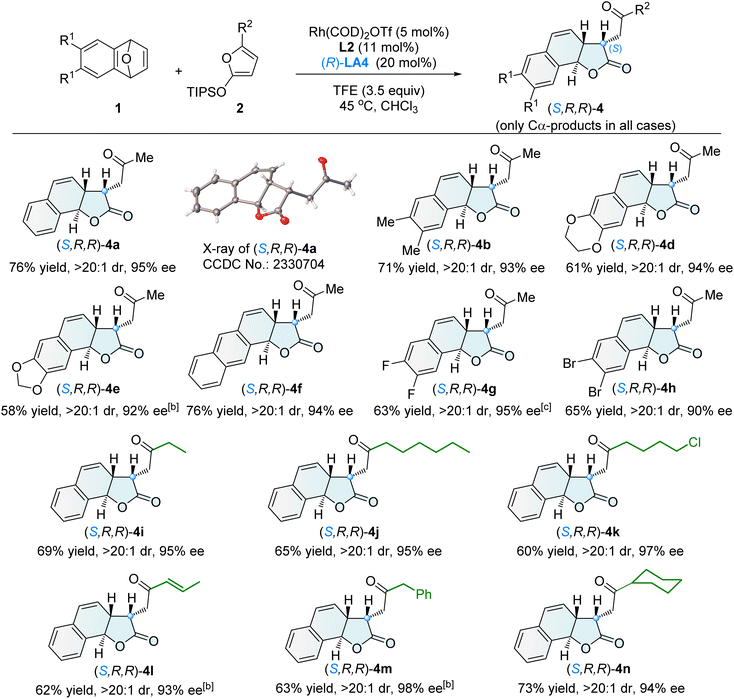 | ||
| Scheme 3 Asymmetric synthesis of tricyclic γ-lactones (S,R,R)-4[a]. [a] Unless otherwise noted, the reaction was conducted under the conditions described in entry 14 of Table 1, and the dr value refers to the ratio of 4 and 3. [b] 2 (3.0 equiv.) was used, at 40 °C. [c] At 40 °C. | ||
Stereodivergent synthesis and synthetic applications
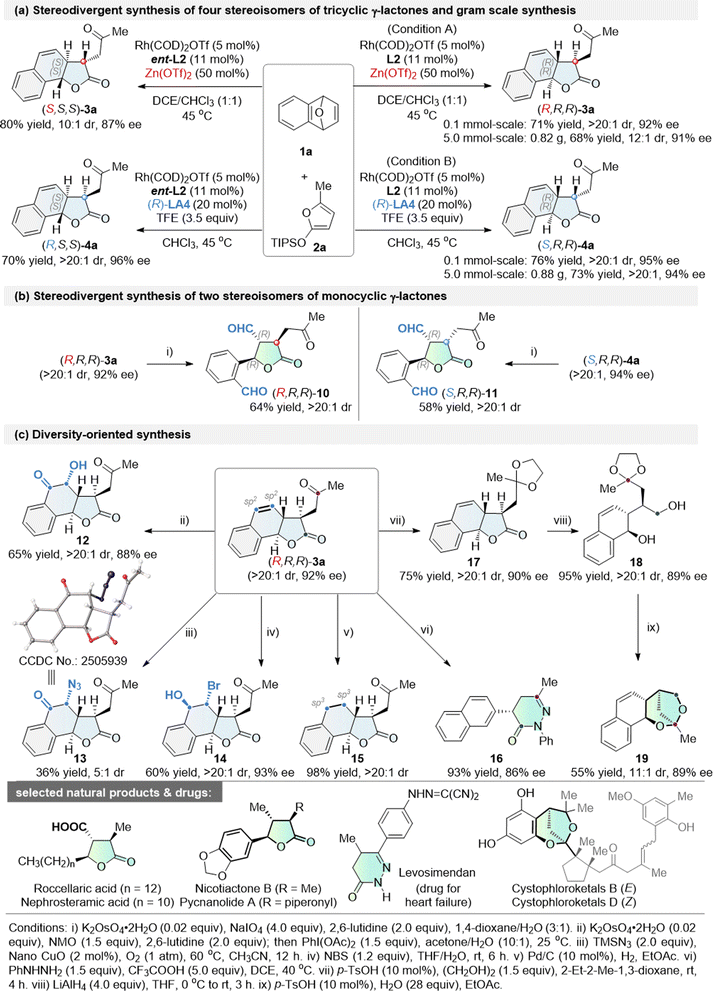
As indicated in Scheme 4a, changing the chiral ligand L2 to ent-L2 in the Cα-ARO/cyclization cascade reaction between 1a and 2a yielded (S,S,S)-3a in 80% yield with a 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr and 87% ee. Similarly, (R,S,S)-4a was obtained in 70% yield, with a >20
1 dr and 87% ee. Similarly, (R,S,S)-4a was obtained in 70% yield, with a >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr and 96% ee.56a–e Thus, four stereoisomers were synthesized in good yields, exhibiting both excellent enantioselectivity and diastereoselectivity. Moreover, the tricyclic γ-lactones (R,R,R)-3a and (S,R,R)-4a were prepared on a gram scale with comparable reactivity and selectivity (Scheme 4a). Although a slight decrease in diastereoselectivity was observed for (R,R,R)-3a, it still maintained a high dr of 12
1 dr and 96% ee.56a–e Thus, four stereoisomers were synthesized in good yields, exhibiting both excellent enantioselectivity and diastereoselectivity. Moreover, the tricyclic γ-lactones (R,R,R)-3a and (S,R,R)-4a were prepared on a gram scale with comparable reactivity and selectivity (Scheme 4a). Although a slight decrease in diastereoselectivity was observed for (R,R,R)-3a, it still maintained a high dr of 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, demonstrating the robustness of this diastereodivergent protocol.
1, demonstrating the robustness of this diastereodivergent protocol.
Diversity-oriented synthesis (DOS), a powerful tool for drug discovery, aims to efficiently construct compound libraries with structural complexity, stereochemical, and functional diversity.53,54 By leveraging the multiple functional groups embedded in the resulting tricyclic γ-lactone products, we successfully executed a series of intriguing DOS protocols starting from (R,R,R)-3a (Schemes 4b, c and 5).
The alkene moiety in 3a serves as a versatile linchpin for downstream diversification, enabling rapid expansion of three-dimensional chemical space and modular elaboration of skeletal and functional complexity. Oxidative cleavage of the olefin under catalytic K2OsO4/NaIO4 conditions—avoiding the use of volatile, toxic OsO4—yields γ-aryl-substituted monocyclic γ-lactone (R,R,R)-10 in 64% yield with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr (Scheme 4b). The same cleavage protocol applied to (S,R,R)-4a furnishes the complementary diastereomer (S,R,R)-11 in 58% yield with >20
1 dr (Scheme 4b). The same cleavage protocol applied to (S,R,R)-4a furnishes the complementary diastereomer (S,R,R)-11 in 58% yield with >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr. Thus, these transformations provide a diastereodivergent gateway to valuable monocyclic γ-lactones bearing three contiguous stereocenters.
1 dr. Thus, these transformations provide a diastereodivergent gateway to valuable monocyclic γ-lactones bearing three contiguous stereocenters.
Interestingly, switching the co-oxidant manifold to NMO/PhI(OAc)2 while retaining catalytic K2OsO4 redirected the reaction toward ketohydroxylation, delivering α-hydroxy ketone 12 (Scheme 4c: 65% yield, >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, 88% ee). Notably, a heterogeneous copper-based catalyst diversified the alkene functionalization manifold, facilitating oxyazidation to furnish α-azido ketones 13 in 36% yield (5
1 dr, 88% ee). Notably, a heterogeneous copper-based catalyst diversified the alkene functionalization manifold, facilitating oxyazidation to furnish α-azido ketones 13 in 36% yield (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr), instead of the previously reported C
1 dr), instead of the previously reported C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double-bond cleavage.63 The structure and absolute configuration of 13 were confirmed by single-crystal X-ray analysis. Exposure to NBS in THF/H2O promoted hydroxyl-bromination, ultimately yielding the densely functionalized product 14 (60% yield, >20
C double-bond cleavage.63 The structure and absolute configuration of 13 were confirmed by single-crystal X-ray analysis. Exposure to NBS in THF/H2O promoted hydroxyl-bromination, ultimately yielding the densely functionalized product 14 (60% yield, >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, 93% ee). Pd/C-mediated hydrogenation of (R,R,R)-3a cleanly saturated the double bond to provide 15 in 98% yield, enhancing the sp3 character of this scaffold.
1 dr, 93% ee). Pd/C-mediated hydrogenation of (R,R,R)-3a cleanly saturated the double bond to provide 15 in 98% yield, enhancing the sp3 character of this scaffold.
Treatment with PhNHNH2 led to a cascade amidation/condensation cyclization of 3a, affording the privileged dihydropyridazin-3(2H)-one 16 with 86% ee and 93% yield. Due to the poor diastereoselectivity (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr) in the direct LiAlH4-mediated reduction of 3a, an alternative cascade reduction protocol was developed. This protocol commenced with the protected ketal 17, followed by the reduction of lactone to give the diol 18. Finally, hydrolysis-initiated cyclization of 18 delivered the complex fused 2,7-dioxabicyclo[3.2.1]octane 19, thereby enabling skeletal diversity synthesis. Ring-distortion reactions represent a hallmark strategy in diversity-oriented synthesis, involving structural reshaping of existing ring systems through predictable chemical operations—such as ring cleavage, rearrangement, aromatization, and fusion—to rapidly access diverse structural and skeletal molecular platforms. With this strategy in mind, we have successfully transformed the tricyclic γ-lactone 3a into privileged chiral scaffolds including monocyclic γ-lactones,4,5,64 dihydropyridazin-3(2H)-ones,65 and fused dioxabicyclo[3.2.1]-octanes66—structural motifs that are widely found in natural products and pharmaceutical agents.55a
1 dr) in the direct LiAlH4-mediated reduction of 3a, an alternative cascade reduction protocol was developed. This protocol commenced with the protected ketal 17, followed by the reduction of lactone to give the diol 18. Finally, hydrolysis-initiated cyclization of 18 delivered the complex fused 2,7-dioxabicyclo[3.2.1]octane 19, thereby enabling skeletal diversity synthesis. Ring-distortion reactions represent a hallmark strategy in diversity-oriented synthesis, involving structural reshaping of existing ring systems through predictable chemical operations—such as ring cleavage, rearrangement, aromatization, and fusion—to rapidly access diverse structural and skeletal molecular platforms. With this strategy in mind, we have successfully transformed the tricyclic γ-lactone 3a into privileged chiral scaffolds including monocyclic γ-lactones,4,5,64 dihydropyridazin-3(2H)-ones,65 and fused dioxabicyclo[3.2.1]-octanes66—structural motifs that are widely found in natural products and pharmaceutical agents.55a
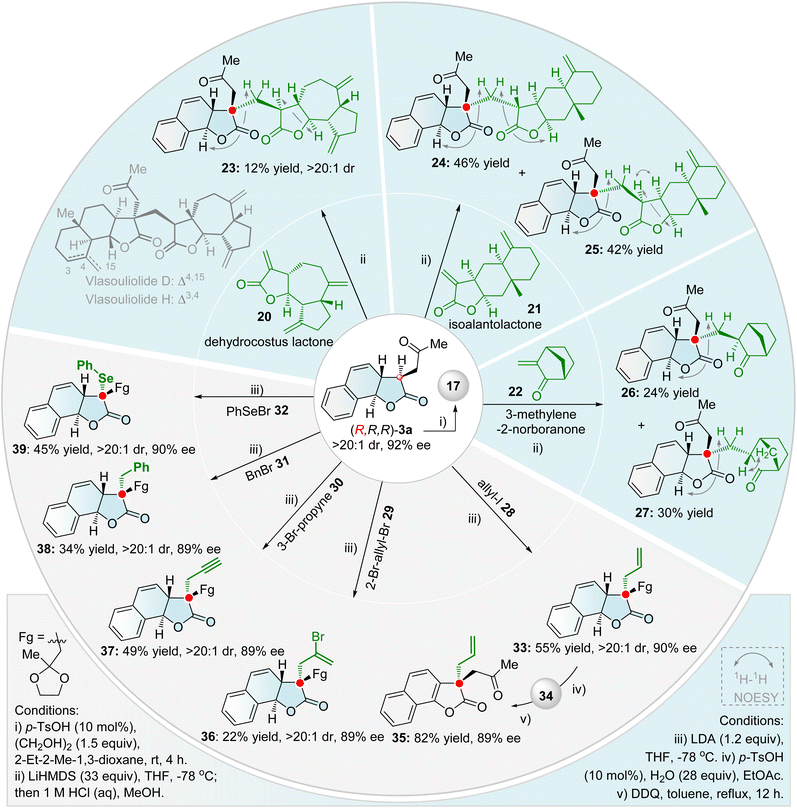
The efficient construction of chiral quaternary stereocenters represents a fundamental yet challenging objective in modern organic synthesis, due to their three-dimensional structural rigidity and unique biological activities.67,68 By employing two kinds of efficient enantioselective sequential processes, diverse chiral quaternary carbon stereocenter-embedded tricyclic γ-lactones were synthesized (Scheme 5). These transformations circumvent the reliance on natural chiral sources typical of traditional synthetic methods and achieve precise control over complex three-dimensional architectures and functional group diversity starting from simple feedstocks. To avoid competitive regioselectivity issues, the ketal-protected tricyclic γ-lactone 17 was used as the starting substrate. First, the LiHMDS-mediated Michael protocol efficiently introduces the natural scaffolds (20–22) onto tricyclic γ-lactones, delivering variants bearing chiral quaternary stereocenters (23–27). Coupling with dehydrocostus lactone 20, followed by acid-promoted deprotection, rapidly furnished the single diastereoisomer 23—an analogue of the natural bis-sesquiterpene lactones Vlasouliolide D and H69—with excellent diastereoselectivity (>20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr). As we know, this is the first enantioselective synthesis of the bis-sesquiterpene lactone core. When isoalantolactone 21 was employed as a Michael acceptor in a similar sequential procedure, column chromatographic isolation afforded two stereoisomeric products 24 (46% yield) and 25 (42% yield). Reaction with 3-methylene-2-norbornanone 22 similarly afforded the stereoisomers 26 (24% yield) and 27 (30% yield). 1H–1H NOESY correlations confirmed the absolute configuration of all these stereoisomeric products, revealing uniform stereochemistry at the newly formed quaternary carbon center. Next, the direct LDA-mediated SN-type Cα-alkylation of γ-lactone with various alkyl halides 28–32 provided an alternative strategy to access chiral γ-lactones bearing quaternary stereocenters. Several synthetically useful functional groups—including allyl (33, 36), propargyl (37), and benzyl group (38)—were successfully incorporated into the lactone core, demonstrating the method's versatility. Employing PhSeBr under these conditions smoothly afforded the Cα-selenylated product 39 (45% yield, >20
1 dr). As we know, this is the first enantioselective synthesis of the bis-sesquiterpene lactone core. When isoalantolactone 21 was employed as a Michael acceptor in a similar sequential procedure, column chromatographic isolation afforded two stereoisomeric products 24 (46% yield) and 25 (42% yield). Reaction with 3-methylene-2-norbornanone 22 similarly afforded the stereoisomers 26 (24% yield) and 27 (30% yield). 1H–1H NOESY correlations confirmed the absolute configuration of all these stereoisomeric products, revealing uniform stereochemistry at the newly formed quaternary carbon center. Next, the direct LDA-mediated SN-type Cα-alkylation of γ-lactone with various alkyl halides 28–32 provided an alternative strategy to access chiral γ-lactones bearing quaternary stereocenters. Several synthetically useful functional groups—including allyl (33, 36), propargyl (37), and benzyl group (38)—were successfully incorporated into the lactone core, demonstrating the method's versatility. Employing PhSeBr under these conditions smoothly afforded the Cα-selenylated product 39 (45% yield, >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr, 90% ee). Additionally, DDQ-mediated oxidative aromatization of the dihydronaphthyl unit in deprotected 34 readily afforded chiral quaternary carbon-embedded naphthofuranones 35—a π-expanded homologue of the privileged benzofuranone scaffold.55a
1 dr, 90% ee). Additionally, DDQ-mediated oxidative aromatization of the dihydronaphthyl unit in deprotected 34 readily afforded chiral quaternary carbon-embedded naphthofuranones 35—a π-expanded homologue of the privileged benzofuranone scaffold.55a
Conclusions
In summary, we have established the first Cα-selective, diastereodivergent asymmetric Mukaiyama-type ring-opening/lactonization of 2-siloxyfurans with oxabicyclic alkenes. A series of chiral tricyclic γ-lactones with three contiguous stereocenters were synthesized in excellent enantioselectivity (up to 98% ee), diastereoselectivity (up to >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 dr) and regioselectivity (up to >20
1 dr) and regioselectivity (up to >20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 Cα
1 Cα![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cγ). By simply changing the co-catalyst (Zn(OTf)2 or Yb(OTf)3) to a novel multifunctional co-catalyst (Sn(OTf)2-BINOL-NMM), the diastereoselectivity was efficiently switched. Four stereoisomers with (S,S,S), (R,R,R), (R,S,S), and (S,R,R)-configurations were readily obtained. This protocol features several advances: (a) the first highly enantioselective and diastereoselective Cα-selective reaction of 2-siloxyfurans, as well as the first diastereodivergent asymmetric Cα-selective reaction of 2-siloxyfurans; (b) developing an alternative catalytic asymmetric route to privileged chiral tricyclic γ-lactones based on a stereoselective C–C bond formation strategy; (c) successfully accomplishing a transition-metal-catalyzed ARO reaction of oxabenzonorbornadienes with unstabilized enolates, exhibiting high stereocontrol of nucleophiles; (d) discovering a distinctive and rare diastereoselective reversal strategy—a catalytic dynamic kinetic lactonization-driven epimerization—that selectively delivers the thermodynamically less stable diastereomer. Moreover, DFT and experimental studies revealed that the origin of Cα-regioselectivity arises from the dispersion and electrostatic interactions between 2-siloxyfurans and the electrophilic partner/chiral catalyst, opening a new platform for reaction design. Additionally, these tricyclic γ-lactones serve as versatile platforms for diversity-oriented synthesis. This utility was demonstrated by the diastereodivergent synthesis of monocyclic γ-lactones with three contiguous stereocenters and the construction of several complex scaffolds, including chiral quaternary carbon-embedded tricyclic γ-lactones, dihydropyridazin-3(2H)-ones, and fused dioxabicyclo[3.2.1]octanes.
Cγ). By simply changing the co-catalyst (Zn(OTf)2 or Yb(OTf)3) to a novel multifunctional co-catalyst (Sn(OTf)2-BINOL-NMM), the diastereoselectivity was efficiently switched. Four stereoisomers with (S,S,S), (R,R,R), (R,S,S), and (S,R,R)-configurations were readily obtained. This protocol features several advances: (a) the first highly enantioselective and diastereoselective Cα-selective reaction of 2-siloxyfurans, as well as the first diastereodivergent asymmetric Cα-selective reaction of 2-siloxyfurans; (b) developing an alternative catalytic asymmetric route to privileged chiral tricyclic γ-lactones based on a stereoselective C–C bond formation strategy; (c) successfully accomplishing a transition-metal-catalyzed ARO reaction of oxabenzonorbornadienes with unstabilized enolates, exhibiting high stereocontrol of nucleophiles; (d) discovering a distinctive and rare diastereoselective reversal strategy—a catalytic dynamic kinetic lactonization-driven epimerization—that selectively delivers the thermodynamically less stable diastereomer. Moreover, DFT and experimental studies revealed that the origin of Cα-regioselectivity arises from the dispersion and electrostatic interactions between 2-siloxyfurans and the electrophilic partner/chiral catalyst, opening a new platform for reaction design. Additionally, these tricyclic γ-lactones serve as versatile platforms for diversity-oriented synthesis. This utility was demonstrated by the diastereodivergent synthesis of monocyclic γ-lactones with three contiguous stereocenters and the construction of several complex scaffolds, including chiral quaternary carbon-embedded tricyclic γ-lactones, dihydropyridazin-3(2H)-ones, and fused dioxabicyclo[3.2.1]octanes.
Author contributions
Z. H. S. conceived and directed the project. Y. H. D. and Y. C. W. directed the project. Z. H. Z. and Z. Q. L. performed the DFT calculations. L. F. G. performed most of the chemistry experiments. X. C. W., J. T. R., M. J., P. L. C. and J. H. H. assisted in the separation and purification of some target products. J. Y. Z. assisted in the synthesis of some substrates. T. C., F. Z. P. and R. T. provided guidance for the project. All authors participated in the discussion. Z. H. S. and Y. H. D. prepared this manuscript.Conflicts of interest
All authors declare no competing interests.Data availability
CCDC 2330703 ((R,R,R)-3a), 2330704 ((S,R,R)-4a), 2333474 ((R,S,S)-4a), 2341397 (9) and 2505939 (13) contain the supplementary crystallographic data for this paper.56a–eThe authors declare that the data supporting the findings of this study are available within the article and the supplementary information (SI) as well as from the authors upon request. The coordinates of the optimized structure are available from the source data. Supplementary information: experimental procedures, experimental equipment, synthetic applications, characterization data, computational results, X-ray data, HRMS, HPLC and NMR spectra for all new compounds. See DOI: https://doi.org/10.1039/d6sc01491g.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22371248, 22261053, 22571272, and 22361047), Yunnan Fundamental Research Projects (202301AS070021, 202501AT070206, 202301AU070002, and 202401AW070001), Project of Innovative Research Team of Yunnan Province (202405AS350010), Yunnan Provincial Science and Technology Project at Southwest United Graduate School (202302AP370004), and Medical Research Fund of Yunnan University (Key project: YDYXJJ2024-0010). We thank the Advanced Analysis and Measurement Center of Yunnan University for the sample testing service.Notes and references
- K. Jozwiak, W. J. Lough and I. W. Wainer, Drug Stereochemistry: Analytical Methods and Pharmacology, Informa, 3rd edn, 2012, p. 332 Search PubMed.
- W. H. Brooks, W. C. Guida and K. G. Daniel, Curr. Trends Med. Chem., 2011, 11, 760–770 CrossRef CAS PubMed.
- I. P. Silvestri and P. J. J. Colbon, ACS Med. Chem. Lett., 2021, 12, 1220–1229 CrossRef CAS PubMed.
- T. D. Tran, N. B. Pham, R. Booth, P. I. Forster and R. J. Quinn, J. Nat. Prod., 2016, 79, 1514–1523 CrossRef CAS PubMed.
- P. A. Onocha and M. S. Ali, Res. J. Phytochem., 2011, 5, 136–145 CrossRef CAS.
- T. Janecki, Natural Lactones and Lactams: Synthesis, Occurrence and Biological Activity, John Wiley & Sons, Weinheim, Germany, 2013, p. 392 Search PubMed.
- J. Hur, J. Jang and J. Sim, Int. J. Mol. Sci., 2021, 22, 2769 CrossRef CAS PubMed.
- M. Seitz and O. Reiser, Curr. Opin. Chem. Biol., 2005, 9, 285–292 CrossRef CAS PubMed.
- B. Mao, M. Fananas-Mastral and B. L. Feringa, Chem. Rev., 2017, 117, 10502–10566 CrossRef CAS PubMed.
- J. Fournier, O. Lozano, C. Menozzi, S. Arseniyadis and J. Cossy, Angew. Chem., Int. Ed., 2013, 52, 1257–1261 CrossRef CAS PubMed.
- Z.-J. He, M.-H. Hong, L.-H. Zhang, W.-H. Xiong and F.-M. Zhang, J. Am. Chem. Soc., 2025, 147, 31639–31649 CrossRef CAS PubMed.
- S. Singha, E. Serrano, S. Mondal, C. G. Daniliuc and F. Glorius, Nat. Catal., 2020, 3, 48–54 CrossRef CAS.
- M. Xue, J. Cui, X. Zhu, F. Wang, D. Lv, Z. Nie, Y. Li and H. Bao, Angew. Chem., Int. Ed., 2023, 62, e202304275 CrossRef CAS PubMed.
- Q. Xiong, K. Tian, X.-L. Liu, L. He, Y.-W. Song, Y. Liu, Z.-F. Wang, X.-Q. Dong and C.-J. Wang, Angew. Chem., Int. Ed., 2026, 65, e19537 CrossRef CAS PubMed.
- K. M. Steward, E. C. Gentry and J. S. Johnson, J. Am. Chem. Soc., 2012, 134, 7329–7332 CrossRef CAS PubMed.
- T. Touge, K. Sakaguchi, N. Tamaki, H. Nara, T. Yokozawa, K. Matsumura and Y. Kayaki, J. Am. Chem. Soc., 2019, 141, 16354–16361 CrossRef CAS PubMed.
- Z. Xiong, J. Tian, P. Xue and H. Lv Zhang, Org. Chem. Front., 2020, 7, 104–108 RSC.
- M. Shi, Y. Yao, X. Fan, K. Li, X. Yu, Y. Liu, Z. Wu and N. Wang, ACS Catal., 2024, 14, 17480–17488 CrossRef CAS.
- J. Wang, S. Su, S. Zhang, S. Zhai, R. Sheng, W. Wu and R. Guo, Eur. J. Med. Chem., 2019, 175, 215–233 CrossRef CAS PubMed.
- A. T. Kulyyasov, T. S. Seitembetov and S. M. Adekenov, Chem. Nat. Compd., 1997, 33, 185–186 CrossRef CAS.
- A. Coricello, A. El-Magboubb, M. Luna, A. Ferrari, I. S. Haworth, C. J. Gomer, F. Aiello and J. D. Adams, Bioorg. Med. Chem. Lett., 2018, 28, 993–996 CrossRef CAS PubMed.
- R. Yang, M. Han, C. Wu, J. Yang and Y. Guo, J. Agric. Food Chem., 2025, 73, 14882–14890 CrossRef CAS PubMed.
- X. Shi, S. Dai, J. Song, S. Zhang, W. Zhang, Y. Guo, S. Zhang, Y. Wang, W. Ye, J. Zheng, X. Ma and W. Zhao, Bioorg. Chem., 2025, 154, 108048 CrossRef CAS PubMed.
- B. Sandargo, B. Thongbai, D. Praditya, E. Steinmann, M. Stadler and F. Surup, Molecules, 2018, 23, 2697–2708 CrossRef PubMed.
- A. Rahman and Z. Shah, in Stereoselective Synthesis in Organic Chemistry, ed. H. H. Wasserman, S. F. Martin and Y. Yamamoto, Springer-Verlag, New York, 1999, pp. 185–396 Search PubMed.
- L. Lin and X. Feng, Chem.–Eur. J., 2017, 23, 6464–6482 CrossRef CAS PubMed.
- M. Bihani and J. C.-G. Zhao, Adv. Synth. Catal., 2017, 359, 534–575 CrossRef CAS.
- I. P. Beletskaya, C. Nájera and M. Yus, Chem. Rev., 2018, 118, 5080–5200 CrossRef CAS PubMed.
- D. Moser, T. A. Schmidt and C. Sparr, JACS Au, 2023, 3, 2612–2630 CrossRef CAS PubMed.
- L. Wei, C. Fu, Z.-F. Wang, H.-Y. Tao and C.-J. Wang, ACS Catal., 2024, 14, 3812–3844 CrossRef CAS.
- G. Rassu, F. Zanardi, L. Battistini and G. Casiraghi, Chem. Soc. Rev., 2000, 29, 109–118 RSC.
- L. Hoppmann and O. G. Mancheño, Molecules, 2021, 26, 6902 CrossRef CAS PubMed.
- Q. Zhang, X. Liu and X. Feng, Curr. Org. Synth., 2013, 10, 764–785 CrossRef CAS.
- R. P. Singh, B. M. Foxman and L. Deng, J. Am. Chem. Soc., 2010, 132, 9558–9560 CrossRef CAS PubMed.
- S. P. Brown, N. C. Goodwin and D. W. C. MacMillan, J. Am. Chem. Soc., 2003, 125, 1192–1194 CrossRef CAS PubMed.
- Y.-Q. Jiang, Y.-L. Shi and M. Shi, J. Am. Chem. Soc., 2008, 130, 7202–7203 CrossRef CAS PubMed.
- L. C. Wieland, E. M. Vieira, M. L. Snapper and A. H. Hoveyda, J. Am. Chem. Soc., 2009, 131, 570–576 CrossRef CAS PubMed.
- J. Li, R. Huang, Y.-K. Xing, G. Qiu, H.-Y. Tao and C.-J. Wang, J. Am. Chem. Soc., 2015, 137, 10124–10127 CrossRef CAS PubMed.
- F. Richard, S. Aubert, T. Katsina, L. Reinalda, D. Palomas, R. Crespo-Otero, J. Huang, D. C. Leitch, C. Mateos and S. Arseniyadis, Nat. Synth., 2022, 1, 641–648 CrossRef CAS.
- J. Cui, R. Oriez, H. Noda, T. Watanabe and M. Shibasaki, Angew. Chem., Int. Ed., 2022, 61, e202203128 CrossRef CAS PubMed.
- B. Mao, Y. Ji, M. F. Mastral, G. Caroli, A. Meetsma and B. L. Feringa, Angew. Chem., Int. Ed., 2012, 51, 3168–3173 CrossRef CAS PubMed.
- W. Chen and J. F. Hartwig, J. Am. Chem. Soc., 2012, 134, 15249–15252 CrossRef CAS PubMed.
- M. Woyciechowska, G. Forcher, S. Buda and J. Mlynarski, Chem. Commun., 2012, 48, 11029–11031 RSC.
- M. Lautens, K. Fagnou and S. Hiebert, Acc. Chem. Res., 2003, 36, 48–58 CrossRef CAS PubMed.
- S. V. Kumar, A. Yen, M. Lautens and P. J. Guiry, Chem. Soc. Rev., 2021, 50, 3013–3093 RSC.
- L. Zhang, C. M. Le and M. Lautens, Angew. Chem., Int. Ed., 2014, 53, 5951–5954 CrossRef CAS PubMed.
- S. Krautwald, D. Sarlah, M. A. Schafroth and E. M. Carreira, Science, 2013, 340, 1065–1067 CrossRef CAS PubMed.
- X. Huo, R. He, X. Zhang and W. Zhang, J. Am. Chem. Soc., 2016, 138, 11093–11096 CrossRef CAS PubMed.
- Y.-H. Wen, F. Yang, S. Li, X. Yao, J. Song and L.-Z. Gong, J. Am. Chem. Soc., 2023, 145, 4199–4207 CrossRef CAS PubMed.
- J. Huang, H. Xu, Y. Pang, L. Liu, X. Wan, W. Wang, L. Gan, J. Ren, F. Peng, Y. Dang, Y. Ou, Y.-H. Deng and Z. Shao, Angew. Chem., Int. Ed., 2025, 64, e202507941 CrossRef CAS PubMed.
- L. Xiao, L. Wei and C.-J. Wang, Angew. Chem., Int. Ed., 2021, 60, 24930–24940 CrossRef CAS PubMed.
- C. Che, Y.-N. Lu and C.-J. Wang, J. Am. Chem. Soc., 2023, 145, 2779–2786 CrossRef CAS PubMed.
- M. D. Burke and S. L. Schreiber, Angew. Chem., Int. Ed., 2004, 43, 46–58 CrossRef PubMed.
- W. R. J. D. Galloway, A. Isidro-Llobet and D. R. Spring, Nat. Commun., 2010, 1, 80 CrossRef PubMed.
- (a) More selected natural products and pharmaceutical agents containing these chiral privileged scaffolds, including monocyclic γ-lactones, dihydropyridazin-3(2H)-ones, dioxabicyclo[3.2.1]-octanes, benzofuranones, and chiral quaternary-bearing tricyclic γ-lactones, are shown in Section 6 in SI.; (b) For more details, please see the SI..
- (a) CCDC 2330703: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2j78z1; (b) CCDC 2330704: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2j7903; (c) CCDC 2333474: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2jb5cf; (d) CCDC 2341397: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2jldyh; (e) CCDC 2505939: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2q3mr7; (f) S. Li, Q. Chen, J. Yang and J. Zhang, Angew. Chem., Int. Ed., 2022, 61, e202202046 CrossRef CAS PubMed . According to Zhang's report, the absolute configuration and structure of 5a was assigned to be R..
- T. Chen, L. Gan, R. Wang, Y. Deng, F. Peng, M. Lautens and Z. Shao, Angew. Chem., Int. Ed., 2019, 58, 15819–15823 CrossRef CAS PubMed.
- S. Kobayashi, M. Araki and I. Hachiya, J. Org. Chem., 1994, 59, 3758–3759 CrossRef CAS.
- S. Kobayashi, H. Ishitani, I. Hachiya and M. Araki, Tetrahedron, 1994, 50, 11623–11636 CrossRef CAS.
- For a review on combined acid catalysis, see: H. Yamamoto and K. Futatsugi, Angew. Chem., Int. Ed., 2005, 44, 1924–1942 CrossRef CAS PubMed.
- T. Lu and Q. Chen, J. Comput. Chem., 2022, 43, 539–555 CrossRef CAS PubMed.
- T. Lu, Angew. Chem., Int. Ed., 2025, 64, e202504895 CrossRef CAS PubMed.
- Z. Cheng, K. Huang, C. Wang, L. Chen, X. Li, Z. Hu, X. Shan, P.-F. Cao, H. Sun, W. Chen, C. Li, Z. Zhang, H. Tan, X. Jiang, G. Zhang, Z. Zhang, M. Lin, L. Wang, A. Zheng, C. Xia, T. Wang, S. Song, X. Shu and N. Jiao, Science, 2025, 387, 1083–1090 CrossRef CAS PubMed.
- R. Bandichhor, B. Nosse and O. Reiser, in Natural Product Synthesis I: Targets, Methods, Concepts, ed. J. Mulzer, Springer Berlin Heidelberg, Berlin, Heidelberg, 2005, vol. 243, pp. 43–72 Search PubMed.
- A. F. E. Rump, D. A. R. Rosen and W. Klaus, Pharmacol. Toxicol., 1994, 74, 244–248 CrossRef CAS PubMed.
- M. E. Hattab, G. Genta-Jouve, N. Bouzidi, A. Ortalo-Magne, C. Hellio, J.-P. Marechal, L. Piovetti, O. P. Thomas and G. Culioli, J. Nat. Prod., 2015, 78, 1663–1670 CrossRef PubMed.
- K. W. Quasdorf and L. E. Overman, Nature, 2014, 516, 181–191 CrossRef CAS PubMed.
- T. T. Talele, J. Med. Chem., 2020, 63, 13291–13315 CrossRef CAS PubMed.
- D. M. Cárdenas, F. J. R. Mejías, J. M. G. Molinillo and F. A. Macías, J. Org. Chem., 2020, 85, 7322–7332 CrossRef PubMed.
Footnote |
| † L. Gan, Z.-Q. Li and T. Chen contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |