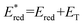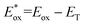Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Deep red photocatalysis via direct S0 → T1 excitation of an Ir(III) complex using 740 nm light
Robert J. Ortiz ,
Dion B. Nemez,
Mahtasim Bhuiyan,
Keighlynn A. Veilleux
,
Dion B. Nemez,
Mahtasim Bhuiyan,
Keighlynn A. Veilleux and
David E. Herbert
and
David E. Herbert *
*
Department of Chemistry and the Manitoba Institute for Materials, University of Manitoba, 144 Dysart Road, Winnipeg, MB R3T 2N2, Canada. E-mail: david.herbert@umanitoba.ca
First published on 17th April 2026
Abstract
We report the first examples of deep-red Ir(III) photochemistry using [Ir(ppy)2(p-biphe)]PF6 (ppy = 2-phenylpyridine; p-biphe = 6,6′,7,7′-biphenanthridine). Red light (740 nm) directly excites into the triplet manifold, populating a long-lived excited state (54 ± 3 ns) with mixed triplet metal-to-ligand charge-transfer/ligand-centered character capable of energy-transfer and electron-transfer photocatalysis.
While photocatalysis is by now an established pillar of modern synthetic chemistry,1 the majority of reactions utilize higher energy visible light (∼390–550 nm).2 Longer wavelengths, notably red (600–700 nm) and near infrared (NIR) light, can penetrate deeper into solutions compared with shorter wavelengths, decreasing the chance of side reactions involving direct absorption by substrates/products, and lowering the probability of catalyst degradation, alongside the safety benefits of utilizing lower energy radiation.3 In addition to metalloporphyrins and related molecules, and emerging work with abundant-element chromophores,4 coordination complexes of osmium(II) and ruthenium(II) have arguably received the most attention in this area (Fig. 1).5 In these latter cases, the photophysics exploits the ability of heavier elements to undergo direct, spin-forbidden S0 → T1 excitation thanks to large spin–orbit coupling (SOC).6 Despite this success with heavier group 8 elements7 and the general utility of Ir(III) photocatalysis,8 red-light mediated processes using group 9 chromophores has remained elusive.9,10 The lowest energy excitation reached with photochemically active Ir(III) has been orange light (590 nm).10 This presents an opportunity for ligand design to access deeper red excitation with Ir(III).
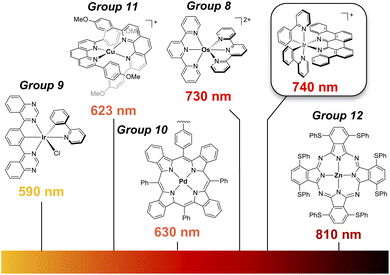 | ||
| Fig. 1 Low-energy photocatalysts in literature4,5 and the [Ir(ppy)2(p-biphe)]+ complex ([Ir]+) that is the focus of this work. | ||
We recently reported the synthesis of [Ir(ppy)2(p-biphe)]PF6 ([Ir]PF6), a heteroleptic Ir(III) complex supported by 2-phenylpyridine (ppy) and 6,6′,7,7′-biphenanthridine ligands (p-biphe).11 The planar, π-extended p-biphe framework12 allows [Ir]+ to absorb throughout the visible region of the electromagnetic spectrum past 800 nm and emit in the NIR. The broad absorption intrigued us with respect to the possibility of leveraging low-energy light in photocatalysis.
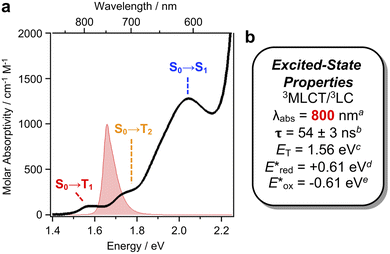
In particular, a weak band (ε ∼220 M−1 cm−1) is evident in the absorption spectrum of [Ir]+, assigned to direct S0 → Tn transitions. Exploiting this absorption using 740 nm red LEDs would represent a 150 nm (3400 cm−1) bathochromic shift compared to state-of-the-art low-energy Ir photocatalysts.9,10 The UV-Vis spectrum of a concentrated sample of [Ir]PF6 (0.8 mM) clearly resolves three peaks in the lowest energy region (Fig. 2a) which can be fit by Gaussian deconvolution (Fig. S1).13 Density functional theory (DFT) and time-dependent DFT simulations including SOC assigns these as spin-forbidden S0 → T1 (783 nm) and S0 → T2 (717 nm) transitions, overlapping with the spin-allowed S0 → S1 (605 nm; Fig. S2 and Table S1). Electron–hole density maps indicate the lowest energy S0 → T1 band has mixed metal-to-ligand charge-transfer/ligand centered (3MLCT/3LC) character, while the higher energy S0 → T2 and S0 → S1 bands present MLCT/inter-ligand charge-transfer (MLCT/ILCT) character. These bands should be directly accessible using red LEDs (red spectrum in Fig. 2a).
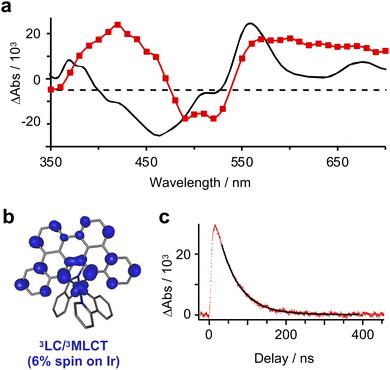
Phosphorescence from [Ir]PF6 (λem = 812 nm; Φlum = 0.26%) is sufficiently bright to record the lifetime of the emissive state (τ = 55 ns).11 Optical transient absorption (oTA) spectroscopy measurements of [Ir]PF6 in acetonitrile using a 540 nm pump confirm formation of a single, dominant excited state. The spectrum taken at a 13 ns delay shows excited-state absorptions (ESA) between 350–470 nm and 540–700 nm sandwiching a shallow bleach (480–530 nm; Fig. 3a). A linear combination of the absorption spectra of oxidized/reduced [Ir]PF6 measured in a spectroelectrochemical cell (Fig. S3 and S4) shows good but not perfect agreement with these features, supporting an interconfigurational 3LC/3MLCT assignment with characteristics of the MLCT state (Fig. 3b). Single-wavelength kinetic traces monitored at 600 nm can be fit to a single-exponential decay with a time constant of 54 ± 3 ns (Fig. 3c), in excellent agreement with the time constant extracted from luminescence decay measurements in dichloromethane.11 Overall, the 54 ns lifetime measured for the excited state of [Ir]+ should be more than sufficient for bimolecular quenching.
Cyclic voltammetry (CV) shows [Ir]+ can be reversibly reduced at −0.95 V and −1.53 V vs. FcH0/+ (FcH = ferrocene), with an irreversible oxidation at +0.95 V.11 The triplet state energy can be calculated at 1.56 eV using the highest energy emission band recorded at 77 K (Fig. S5).14 Using these data, we estimate excited-state redox potentials of 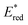 = +0.61 V, and
= +0.61 V, and 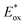 = −0.61 V vs. FcH0/+.15 [Ir]+ thus could conceivably serve both as a strong photo-oxidant and photo-reductant.16 Given the irreversible nature of the oxidation event recorded by CV, we chose to target reactions in which the substrate is oxidized, as well as energy-transfer photocatalysis. This choice was bolstered by the observation that known low-energy Ir(III) photocatalysts are generally reducing,10 differentiating the photochemical reactivity possible with [Ir]PF6. Important excited-state parameters are summarized in Fig. 2b.
= −0.61 V vs. FcH0/+.15 [Ir]+ thus could conceivably serve both as a strong photo-oxidant and photo-reductant.16 Given the irreversible nature of the oxidation event recorded by CV, we chose to target reactions in which the substrate is oxidized, as well as energy-transfer photocatalysis. This choice was bolstered by the observation that known low-energy Ir(III) photocatalysts are generally reducing,10 differentiating the photochemical reactivity possible with [Ir]PF6. Important excited-state parameters are summarized in Fig. 2b.
First, we attempted the aerobic hydroxylation of 4-methoxyphenylboronic acid (Fig. 4a).17 Irradiation of [Ir]+ in the presence of N,N-diisopropylethylamine (DIPEA; Eox(DIPEA/DIPEA+) = +0.28 V vs. FcH0/+)18,19 leads to photooxidation of DIPEA and reduction of [Ir]+ (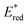 = +0.61 V). Electron-transfer from photoreduced [Ir] to O2 in oxygenated CD3CN then facilitates boronic acid hydroxylation via the superoxide radical.17 1H NMR spectroscopy shows 85 ± 11% conversion to the phenol in 20 h using 2 mol% [Ir]+ and 740 nm illumination (Fig. S6, S7 and Table S2). No conversion occurs without light, and only minor conversion is evident in the absence of [Ir]+ (Fig. S8, S9 and Table S2). Energy transfer mediated by 1O2 generation and reaction with furfural in alcoholic solution (Fig. 4b) is similarly effective. Using 1 mol% [Ir]+ excited at 740 nm in oxygenated CD3OD leads to 85 ± 9% conversion to 5-methoxyfuran-2-one over 23 h.20 When additional oxygen is bubbled in, the reaction progresses further, reaching 92 ± 4% conversion after another 3 h (Fig. S10, S11 and Table S3). A volatile side product (formic acid21) is evident in the 1H NMR spectrum and can be removed under reduced pressure (Fig. S12; see Fig. S45 for mechanism). Control reactions again confirmed the necessity of both [Ir]+ and light (Fig. S13, S14 and Table S3). These examples are the lowest energy photocatalysis using an Ir(III) photocatalyst reported to date.
= +0.61 V). Electron-transfer from photoreduced [Ir] to O2 in oxygenated CD3CN then facilitates boronic acid hydroxylation via the superoxide radical.17 1H NMR spectroscopy shows 85 ± 11% conversion to the phenol in 20 h using 2 mol% [Ir]+ and 740 nm illumination (Fig. S6, S7 and Table S2). No conversion occurs without light, and only minor conversion is evident in the absence of [Ir]+ (Fig. S8, S9 and Table S2). Energy transfer mediated by 1O2 generation and reaction with furfural in alcoholic solution (Fig. 4b) is similarly effective. Using 1 mol% [Ir]+ excited at 740 nm in oxygenated CD3OD leads to 85 ± 9% conversion to 5-methoxyfuran-2-one over 23 h.20 When additional oxygen is bubbled in, the reaction progresses further, reaching 92 ± 4% conversion after another 3 h (Fig. S10, S11 and Table S3). A volatile side product (formic acid21) is evident in the 1H NMR spectrum and can be removed under reduced pressure (Fig. S12; see Fig. S45 for mechanism). Control reactions again confirmed the necessity of both [Ir]+ and light (Fig. S13, S14 and Table S3). These examples are the lowest energy photocatalysis using an Ir(III) photocatalyst reported to date.
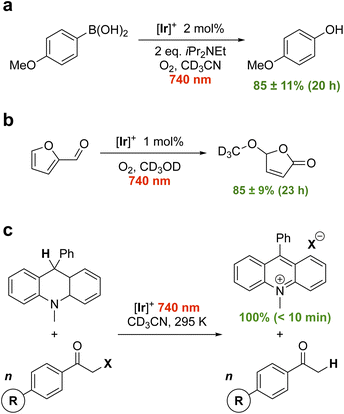 | ||
| Fig. 4 Proof-of-principle photocatalytic reactions (a–c) using [Ir]+ and red light (740 nm) illumination. | ||
We next turned our attention to the thermodynamically challenging hydrodehalogenation of phenacyl halides using 9,10-dihydro-10-methylacridine as a hydride source (DHA; Eox = +0.4 V vs. FcH0/+).19,22 DHA oxidation can been performed using [Ru(bpy)3]2+ (bpy = 2,2′-bipyridine) and 450 nm light.22,23 Here, we find photoexcited [Ir]+ can smoothly oxidize the even more difficult to oxidize 9-phenyl analogue 10-methyl-9-phenyl-9,10-dihydroacridine (MPA; Eox = +0.5 V vs. FcH0/+)19,24 using 740 nm red light, reducing phenacyl bromide to regenerate the starting catalyst (Fig. 4c). Given the debate in literature as to the reduction potential of phenacyl bromide,25,26 we measure it at Ered = −1.60 V vs. FcH0/+ (Fig. S15). In the presence of excess (3 equivalents) of phenacyl bromide, MPA is completely and cleanly consumed with only acetophenone and the acridinium bromide salt observed by 1H NMR within 30 min at a loading of 2 mol% [Ir]+ (Fig. S16–S18). Reducing the catalyst loading to 0.1 mol%, the reaction still cleanly goes to completion after 65 min (Fig. S19–S22; 1.6% min−1). With 2 mol% [Ir]+, the reaction progresses at a rate of 12.1% min−1 and is in fact complete after 10 min of illumination (Fig. S23 and S24). Control reactions again confirm the critical role of both [Ir]+ and light (Fig. S25 and S26). [Ir]+ proved quite durable and adding additional equivalents of MPA after the initial batch is consumed restarts catalysis with no loss in activity or selectivity (Fig. S27).
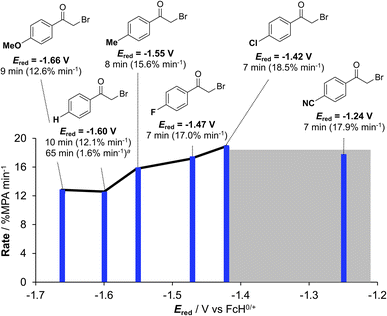
Using conditions of 2 mol% [Ir]+ at ambient temperature (295 K), we screened the compatibility of different functional groups using a variety of 4′-functionalized 2-bromoacetophenones (R = H, OMe, Me, F, Cl, CN). In each case, 100% conversion of MPA to the acridinium bromide is observed in the presence of excess (3 equivalents) phenacyl bromide. 1H NMR monitoring (Fig. S28–S33) allowed us to extract the initial rates (Fig. S34) which increase with more electron-withdrawing substituents. Comparing the rate versus Hammett parameters (σp),27 however, yielded a poor correlation (Fig. S35). A better correlation is evident using the substrate reduction potential (Fig. 5) measured via electrochemistry (Fig. S36–S40). The influence of the substrate reduction potential is noticeable up to a point: 2-bromo-4′-fluoro- (Ered = −1.47 V vs. FcH0/+) and 2-bromo-4′-cyanoacetophenone (Ered = −1.24 V vs. FcH0/+) are converted at similar rates (Fig. S41). We postulate that at a certain limit the reaction becomes controlled by diffusion, rather than thermodynamics (grey highlight in Fig. 5). Exchanging acylbromide for acylchloride in the form of 2-chloroacetophenone (Ered = −1.73 V vs. FcH0/+, Fig. S42), consumption of MPA is still observed, consistent with MPA photooxidation instigating catalysis, but without clean conversion to the hydrodehalogenated product (Fig. S43), in line with the thermodynamic challenge presented by this harder-to-reduce substrate.
Conclusions
In summary, we report that an Ir(III) complex of the planar, benzannulated π-extended ligand 6,6′,7,7′-biphenanthridine ([Ir]+)11 can effectively mediate photocatalysis using 740 nm deep-red light, the lowest energy illumination reported to enable such reactivity to date. This represents a 150 nm (3400 cm−1) bathochromic shift from the prior state-of-the-art in low-energy Ir(III) photocatalysis9,10 and showcases the ability of iridium photocatalysts to reach the deep red/near-infrared region of the electromagnetic spectrum. Direct excitation of [Ir]+ into its triplet Tn manifold provokes population of a long-lived (55 ns) excited state with strong photoredox properties. The promise of [Ir]+-mediated deep red photocatalysis is demonstrated through three proof-of-principle reactions, including the aerobic hydroxylation of 4-methoxyphenylboronic acid and 1O2-mediated synthesis of 5-methoxyfuran-2-one. These reactions had not yet been reported possible via red-light iridium photocatalysis.20 In addition, we also show [Ir]+ to be capable of hydrodehalogenation via the photooxidation of MPA, a reaction that to the best of our knowledge has been previously only accessible using blue light and the easier to oxidize DHA.23 Efforts to expand these findings to a broader scope of reaction classes are currently underway.Author contributions
Conceptualization: R. J. O., D. E. H. Formal analysis: R. J. O., D. E. H. Funding acquisition: D. E. H. Investigation: R. J. O., D. B. N., M. B., K. A. V. Supervision: D. E. H. Visualization: R. J. O., D. E. H. Writing – original draft: R. J. O., D. E. H. Writing – review & editing: R. J. O., D. E. H.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: supporting computational data, NMR spectra, electrochemical plots, time-resolved spectroscopy data, and further experimental details. See DOI: https://doi.org/10.1039/d6sc01462c.Acknowledgements
Support for this work came from the Natural Sciences and Engineering Research Council of Canada (RGPIN-2022-04501; CGS-D/Michael Smith Foreign Study Supplement) and Compute Canada. We are grateful to Prof. James K. McCusker for access to TA instrumentation, training, and insightful discussions, as well as to Bekah Bowers for assistance with TA measurements.References
- E. G. Moschetta, G. C. Cook, L. J. Edwards, M. A. Ischay, Z. Lei, F. Buono, F. Lévesque, J. A. O. Garber, M. MacTaggart, M. Sezen-Edmonds, K. P. Cole, M. G. Beaver, J. Doerfler, S. M. Opalka, W. Liang, P. D. Morse and N. Miyake, Org. Process Res. Dev., 2024, 28, 831–846 CrossRef CAS.
- N. A. Romero and D. A. Nicewicz, Chem. Rev., 2016, 116, 10075–10166 CrossRef CAS PubMed.
- D. C. Cabanero and T. Rovis, Nat. Rev. Chem., 2025, 9, 28–45 CrossRef CAS PubMed.
- N. Kumar, T. Sharma, N. Thakur, R. Jain and N. Sinha, Chem. - Eur. J., 2025, 31, e202500365 CrossRef CAS PubMed.
- L. Fortier, C. Lefebvre and N. Hoffmann, Beilstein J. Org. Chem., 2025, 21, 296–326 CrossRef CAS PubMed.
- M. Zhong and Y. Sun, Chem Catal., 2024, 4, 100973 CAS.
- G. Chacktas, B. Pfund, T. Kerackian, P. Yaltseva, M. Villeneuve, D. Durand, N. Fabre, C. Fiorini-Debuisschert, J.-C. Cintrat, O. S. Wenger and E. Romero, ACS Catal., 2025, 15, 13938–13947 CrossRef CAS.
- K. Teegardin, J. I. Day, J. Chan and J. Weaver, Org. Process Res. Dev., 2016, 20, 1156–1163 CrossRef CAS PubMed.
- K. A. Xie, E. Bednarova, C. L. Joe, C. Lin, T. C. Sherwood, E. M. Simmons, B. C. Lainhart and T. Rovis, J. Am. Chem. Soc., 2023, 145, 19925–19931 CrossRef CAS PubMed.
- E. Bednářová, R. Grotjahn, C. Lin, K. A. Xie, Y. Karube, J. S. Owen, C. L. Joe, B. C. Lainhart, T. C. Sherwood and T. Rovis, J. Am. Chem. Soc., 2025, 147, 12511–12522 CrossRef PubMed.
- D. B. Nemez, R. J. Ortiz, K. A. Veilleux, J. A. G. Williams and D. E. Herbert, Chem.–Eur. J., 2025, 31, e01802 CrossRef CAS PubMed.
- D. B. Nemez, I. B. Lozada, J. D. Braun, J. A. G. Williams and D. E. Herbert, Inorg. Chem., 2022, 61, 13386–13398 CrossRef CAS PubMed.
- J. T. Yarranton and J. K. McCusker, J. Am. Chem. Soc., 2022, 144, 12488–12500 CrossRef CAS PubMed.
- L. Schmid, F. Glaser, R. Schaer and O. S. Wenger, J. Am. Chem. Soc., 2022, 144, 963–976 CrossRef CAS PubMed.
- M. V. Wee-Léonard, B. Elias and L. Troian-Gautier, J. Am. Chem. Soc., 2024, 146, 11031–11035 Search PubMed.
- D. Kim and T. S. Teets, Chem. Phys. Rev., 2022, 3, 021302 CrossRef CAS.
- Y.-Q. Zou, J.-R. Chen, X.-P. Liu, L.-Q. Lu, R. L. Davis, K. A. Jørgensen and W.-J. Xiao, Angew. Chem., Int. Ed., 2012, 51, 784–788 CrossRef CAS PubMed.
- Y. Kwon, J. Lee, Y. Noh, D. Kim, Y. Lee, C. Yu, J. C. Roldao, S. Feng, J. Gierschner, R. Wannemacher and M. S. Kwon, Nat. Commun., 2023, 14, 92 CrossRef CAS PubMed.
- N. G. Connelly and W. E. Geiger, Chem. Rev., 1996, 96, 877–910 CrossRef CAS PubMed.
- B. D. Ravetz, N. E. S. Tay, C. L. Joe, M. Sezen-Edmonds, M. A. Schmidt, Y. Tan, J. M. Janey, M. D. Eastgate and T. Rovis, ACS Cent. Sci., 2020, 6, 2053–2059 CrossRef CAS PubMed.
- B. L. Feringa and R. Trav, Chim. Pays-Bas, 1987, 106, 469–488 CrossRef CAS.
- S. Fukuzumi, S. Mochizuki and T. Tanaka, J. Phys. Chem., 1990, 94, 722–726 CrossRef CAS.
- C. K. Prier, D. A. Rankic and D. W. C. MacMillan, Chem. Rev., 2013, 113, 5322–5363 CrossRef CAS PubMed.
- N. W. Koper, S. A. Jonker, J. W. Verhoeven, C. van Dijk and R. Trav, Chim. Pays-Bas, 1985, 104, 296–302 CrossRef CAS.
- D. D. Tanner and H. K. Singh, J. Org. Chem., 1986, 51, 5182–5186 CrossRef CAS.
- E. Speckmeier, P. J. W. Fuchs and K. Zeitler, Chem. Sci., 2018, 9, 7096–7103 RSC.
- C. Hansch, A. Leo and R. W. Taft, Chem. Rev., 1991, 91, 165–195 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |