Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Bidirectional skeletal remodelling of SF5-nitrobenzenes into azepine, bicyclic, and benzimidazole frameworks†
Muhamad Zulfaqar Bacho a,
Shiwei Wub,
Takuya Muramatsub,
Chavakula Nagababua,
Daiki Harano
a,
Shiwei Wub,
Takuya Muramatsub,
Chavakula Nagababua,
Daiki Harano b,
Seishu Ochiaib and
Norio Shibata
b,
Seishu Ochiaib and
Norio Shibata *ab
*ab
aDepartment of Nanopharmaceutical Sciences, Nagoya Institute of Technology, Gokiso, Showa-ku, Nagoya 466-8555, Japan. E-mail: nozshiba@nitech.ac.jp
bDepartment of Engineering, Nagoya Institute of Technology, Gokiso, Showa-ku, Nagoya 466-8555, Japan
First published on 20th March 2026
Abstract
The development of PFAS-free fluorinated scaffolds that preserve the desirable physicochemical attributes of perfluoroalkyl groups remains a central challenge in contemporary fluorine chemistry. Herein, we report a rapid and bidirectional skeletal-remodelling platform that enables controlled interconversion between aromatic, medium-sized, and bicyclic SF5-containing heterocycles from readily accessible SF5-nitrobenzenes. Phosphorus-catalyzed iterative deoxygenation of SF5-nitrobenzenes generates arylnitrene intermediates that undergo remarkably accelerated dearomative ring expansion, furnishing seven-membered SF5-azepines within dramatically shortened reaction times compared to non-SF5 analogues. These azepines function as versatile skeletal nodes, enabling divergent downstream transformations: photoinduced 4π-electrocyclization provides access to previously unexplored SF5-azabicyclo[3.2.0]hepta-2,6-diene frameworks, while selective fluoroacylative activation promotes reverse skeletal reconstruction to restore aromaticity and deliver SF5-substituted benzimidazoles. Collectively, this work demonstrates that strategic incorporation of the SF5 group not only expands accessible heterocyclic architectures but also fundamentally alters skeletal rearrangement kinetics, enabling rapid and controllable skeletal editing from a common, practical precursor. Given the OECD classification of SF5-containing molecules as non-PFAS, this unified skeletal-remodelling approach substantially broadens the design space of fluorinated scaffolds for applications in pharmaceuticals, agrochemicals, and functional materials, advancing the principles of sustainable fluorine chemistry.
Introduction
Fluorinated heterocycles are indispensable motifs in agrochemicals, pharmaceuticals, and functional materials, as fluorine substitution profoundly enhances lipophilicity, metabolic stability, and target engagement.1 In agrochemical design, CF3-substituted heterocycles are particularly dominant, accounting for more than 40% of all fluorinated agrochemicals.1b However, CF3 groups are classified as per- and polyfluoroalkyl substances (PFAS) under the OECD definition, and their extreme environmental persistence has become a serious global concern.2 The exceptional stability of C–F bonds leads to long-term accumulation and the formation of highly mobile degradation products such as trifluoroacetic acid (TFA, CF3CO2H).3 While CF3 groups remain manageable in pharmaceuticals,1a their intentional environmental release via agrochemicals1b,3 is increasingly unsustainable. Consequently, the development of PFAS-free fluorinated alternatives that retain the functional advantages of CF3 substituents has emerged as a critical challenge.The pentafluorosulfanyl (SF5) group represents a compelling solution.4,5 As a hypervalent sulfur–fluorine functionality, SF5 combines strong electron-withdrawing character, high lipophilicity, and substantial steric bulk, and is therefore widely regarded as a “super-CF3” substituent. Importantly, unlike CF3, SF5 does not rely on persistent C–F bonds and exhibits the potential for environmentally benign mineralization, positioning SF5-containing molecules as classified as PFAS-free.3d In molecular design, SF5 functions as a bioisostere for CF3, tert-butyl, and nitro groups, with comparable hydrophobicity and intrinsic volume (Fig. 1a). Despite these advantages, the structural diversity of SF5-containing molecules—particularly SF5-heterocycles—remains severely limited.5c
 | ||
| Fig. 1 Background of this work. (a) Properties of SF5 vs. CF3, NO2 and tBu-benzenes. (b) Previous work (c) This work. | ||
This limitation arises from a fundamental synthetic bottleneck. Traditional SF5 installation typically relies on radical reactions with alkenes or alkynes using SF5Cl or related reagents, which significantly limits substrate scope.5h In contrast, oxidative chloro-fluorination of aryl sulfides and related protocols4b require harsh reaction conditions, specialized equipment, and stringent moisture control, rendering their application to the synthesis of SF5-heterocycles highly limited.6 As a result, SF5 chemistry has remained largely limited to simple alkyl, alkenyl, and alkynyl motifs. Only recently have new classes of SF5 reagents or new protocols been developed,7 expanding the diversity of accessible substrates and reactivity profiles. Nevertheless, structurally complex heterocycles—particularly those incorporating SF5 or SF4 (ref. 8)—remain largely unexplored and synthetically inaccessible.
Concurrently, skeletal rearrangements have emerged as a powerful strategy for heterocycle synthesis, enabling rapid access to strained and unconventional architectures that are difficult to construct using classical cyclization methods.9 We recognized that this framework-reorganization approach is uniquely suited to SF5 chemistry, where conventional ring-construction strategies are intrinsically constrained. Consistent with this concept, we recently reported the photo-induced synthesis of SF5-azepine aryl ethers from SF5-azido benzene with phenols via skeletal rearrangement, thereby establishing this approach as a viable entry to medium-sized SF5-heterocycles (Fig. 1b).10 Notably, the strong electron-withdrawing nature of the SF5 substituent was found to facilitate the key ring-expansion step from benzene to azepine frameworks. Encouraged by these findings, we herein extend this concept to sequential skeletal remodelling, enabling access to a range of previously inaccessible SF5-containing heterocycles, including SF5-azepin-2-amines 3, SF5-azabicyclo[3.2.0]hepta-2,6-dienes 4, and SF5-benzimidazoles 5. In particular, the SF5-azabicyclo[3.2.0]hepta-2,6-diene derivatives represent the first examples of this rigid bicyclic scaffold bearing an SF5 substituent (Fig. 1c). Importantly, the synthesis originates from readily available SF5-nitrobenzenes 1, rather than inherently energetic SF5-azido benzenes, and proceeds through consecutive skeletal rearrangements. These transformations establish a unified skeletal-editing manifold that interconverts aromatic, medium-sized, and bicyclic SF5-containing architectures.
Results and discussion
Development of a rapid skeletal rearrangement to amino-SF5-azepines
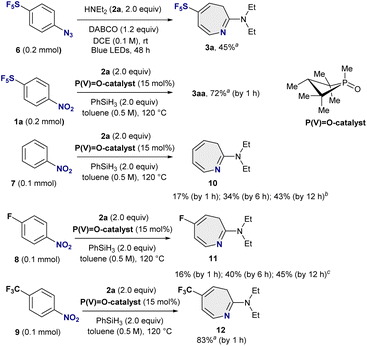
Seven-membered SF5-heterocycles are essentially unexplored, despite the prevalence of azepine frameworks in biologically active molecules and marketed drugs.11 Given our interest in 2-amino-SF5-azepines, we first examined the skeletal remodelling of SF5-azido benzene 6 using diethylamine 2a as the nucleophile in the presence of DABCO, following our reported LED conditions (Fig. 2).10 Under these conditions, the desired 2-diethylamino-SF5-azepine 3a was obtained in 45% yield after 48 h at room temperature, confirming the feasibility of azepine formation from SF5-azido benzene, albeit with limited efficiency and prolonged reaction times. We next turned to an alternative nitrene-generation strategy based on phosphorus-catalyzed deoxygenation of SF5-nitrobenzene, as developed by Radosevich and co-workers.12 Under these conditions, phenylnitrene are generated thermally via P(III)/P(V)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O redox cycling from P(V)
O redox cycling from P(V)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O-catalyst with phenylsilane. Pleasingly, application of this protocol to SF5-nitrobenzene 1a proved markedly more effective, delivering the corresponding SF5-azepine 3a in 72% yield within only 1 h at 120 °C. Notably, in non-SF5 systems, analogous nitrobenzene-to-azepine conversions under Radosevich conditions typically require approximately 12 h to reach completion.12 To further probe the origin of this pronounced rate enhancement, we evaluated nitrobenzene 7 and fluoro-substituted nitrobenzene 8 under identical conditions. In both cases, the corresponding azepin-2-amines (10 and 11) were obtained in only 16–17% yield after 1 h, with yields increasing gradually to 43–45% after 12 h. In sharp contrast, the SF5-substituted nitrobenzene underwent rapid and efficient skeletal rearrangement, suggesting that the strongly electron-withdrawing SF5 substituent significantly accelerates the ring-expansion process, likely by facilitating arylnitrene insertion and subsequent electrocyclic rearrangement.10 Given this substantial rate enhancement and the improved safety and practicality of using SF5-nitrobenzenes instead of SF5-azido benzenes, we undertook a systematic investigation of the phosphorus-catalyzed conversion of SF5-nitrobenzenes into SF5-azepines. A similar acceleration in the ring-expansion was observed for trifluoromethyl-substituted nitrobenzene 9 under standard conditions, affording azepine-2-amine 12 in 83% yield within 1 h, compared to 12 h in the reported method.12
O-catalyst with phenylsilane. Pleasingly, application of this protocol to SF5-nitrobenzene 1a proved markedly more effective, delivering the corresponding SF5-azepine 3a in 72% yield within only 1 h at 120 °C. Notably, in non-SF5 systems, analogous nitrobenzene-to-azepine conversions under Radosevich conditions typically require approximately 12 h to reach completion.12 To further probe the origin of this pronounced rate enhancement, we evaluated nitrobenzene 7 and fluoro-substituted nitrobenzene 8 under identical conditions. In both cases, the corresponding azepin-2-amines (10 and 11) were obtained in only 16–17% yield after 1 h, with yields increasing gradually to 43–45% after 12 h. In sharp contrast, the SF5-substituted nitrobenzene underwent rapid and efficient skeletal rearrangement, suggesting that the strongly electron-withdrawing SF5 substituent significantly accelerates the ring-expansion process, likely by facilitating arylnitrene insertion and subsequent electrocyclic rearrangement.10 Given this substantial rate enhancement and the improved safety and practicality of using SF5-nitrobenzenes instead of SF5-azido benzenes, we undertook a systematic investigation of the phosphorus-catalyzed conversion of SF5-nitrobenzenes into SF5-azepines. A similar acceleration in the ring-expansion was observed for trifluoromethyl-substituted nitrobenzene 9 under standard conditions, affording azepine-2-amine 12 in 83% yield within 1 h, compared to 12 h in the reported method.12
Substrate scope for skeletal rearrangement to amino-SF5-azepines
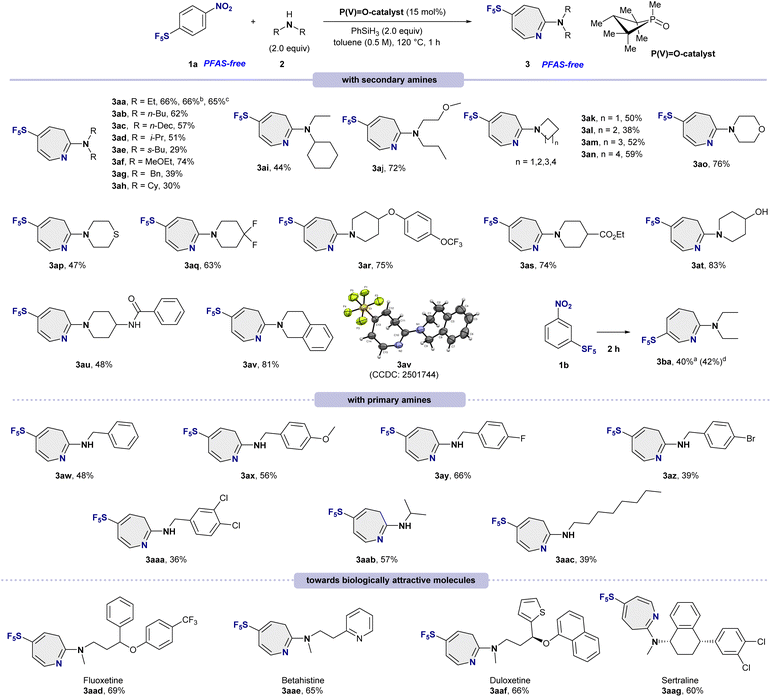
The generality of the phosphorus-catalyzed ring-expansion reaction was first evaluated with a broad range of acyclic secondary amines (Scheme 1, top). Straight-chain dialkylamines such as diethylamine (2a), dibutylamine (2b), and didecylamine (2c) were smoothly converted into the corresponding azepines 3aa–3ac in 57–66% yields. Branched secondary amines were also compatible, although increased steric demand led to somewhat diminished efficiencies, affording products 3ad and 3ae in 29–51% yields. An electron-rich ether-substituted amine (2f) was well tolerated, delivering 3af in 74% yield. Aromatic and alicyclic secondary amines, including dibenzylamine (2g), dicyclohexylamine (2h), and unsymmetrical amines (2i–2j), furnished azepines 3ag–3aj in 30–72% yields. The influence of cyclic amine ring size was next evaluated. Four-, five-, six-, and seven-membered cyclic amines (2k–2n) participated successfully, affording azepines 3ak–3an in moderate yields (38–59%). Notably, heteroatom-containing cyclic amines also proved to be competent nucleophiles: morpholine (2o), thiomorpholine (2p), and difluorinated piperidine (2q) delivered products 3ao–3aq in good to excellent yields (47–76%). In addition, a complex aryloxy-substituted piperidine (2r) underwent smooth coupling to afford 3ar in 75% yield, highlighting the functional-group tolerance of the process.To further assess the generality of the ketenimine intermediate derived from 1a, we investigated nucleophiles bearing diverse functional groups. Esters (2s), alcohols (2t), amides (2u), and bicyclic amines (2v) were all successfully incorporated, furnishing products 3as–3av in 48–83% yields. The structure of 3av was unambiguously confirmed by single-crystal X-ray diffraction (CCDC 2501744). Notably, this transformation is applicable not only to p-substituted SF5-nitrobenzene 1a but also to m-substituted analogue 1b, which afforded the regioselective product 3ba in 40% isolated yield.
Encouraged by the breadth of secondary amine compatibility, we extended the methodology to primary amines (Scheme 1, middle). Benzylamines bearing either electron-donating or electron-withdrawing substituents at the para position (2w–2aa) underwent efficient ring expansion to give products 3aw–3aaa in 36–66% yields. Primary aliphatic amines (2ab and 2ac) were likewise competent substrates, affording azepines 3aab and 3aac in 39–57% yields.
Finally, the synthetic utility of this transformation was demonstrated through late-stage functionalization of biologically relevant amines (Scheme 1, bottom). Complex drug-derived amines, including fluoxetine (2ad), betahistine (2ae), duloxetine (2af), and sertraline (2ag), were successfully converted into the corresponding SF5-azepine derivatives 3aad–3aag in good yields (60–69%). These results underscore the robustness of the method and its potential for late-stage diversification in medicinal and agrochemical discovery.
Anilines were not suitable as amine nucleophiles; instead of azepines 3, N–N bond coupling products were formed, consistent with previous reports.13
Second skeletal rearrangement to bicyclic SF5- azabicyclo[3.2.0]hepta-2,6-dienes
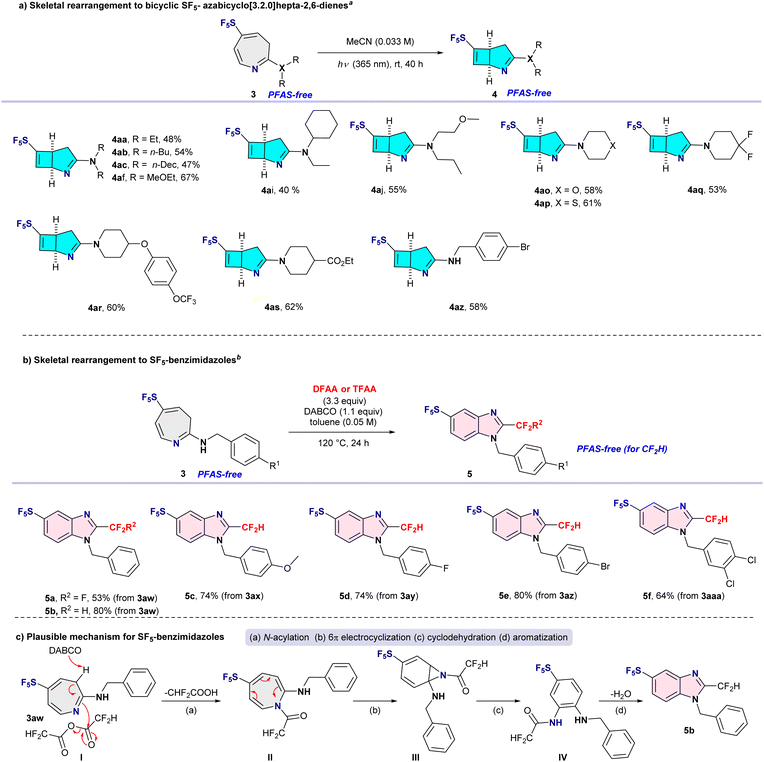
With gram-scale quantities of azepine 3aa available, we next investigated the photochemical 4π-electrocyclization developed by Leonori14 for the synthesis of bicyclic SF5-attached azabicyclo[3.2.0]hepta-2,6-dienes (Scheme 2a, top). Under simple irradiation of azepines 3 at 365 nm in acetonitrile at room temperature cleanly delivered the corresponding bicyclic products, SF5-attached azabicyclo[3.2.0]hepta-2,6-dienes 4 in moderate to good yields. SF5-azepines 3 bearing electron-donating alkyl substituents, including ethyl (3aa), n-butyl (3ab), n-decyl (3ac), and 2-methoxyethyl (3af) groups, were well tolerated, affording bicyclic derivatives 4aa, 4ab, 4ac, and 4af in yields of up to 67%. Azepines containing unsymmetrical secondary amines (3ai and 3aj) also underwent smooth skeletal rearrangement to furnish bicyclic products 4ai and 4aj in 40–55% yields. Notably, this photochemical transformation proved compatible with cyclic amine-substituted azepines: substrates 3ao–3as were efficiently converted into the corresponding bicyclic compounds 4ao–4as in consistently good yields (53–62%). Furthermore, SF5-azepines bearing primary amine substituents, exemplified by 4-bromobenzylamine-derived azepine 3az, participated smoothly to afford bicyclic product 4az in 58% yield. The relative stereochemistry of the bicyclic products 4 was established by detailed 2D NMR analysis of compound 4az (see SI) and was found to be fully consistent with previously reported azabicyclo[3.2.0] frameworks.14Third skeletal rearrangement to SF5-benzimidazoles
To further probe the structural plasticity of the SF5-azepine framework and to demonstrate bidirectional skeletal editing from a common intermediate, we investigated the re-aromatization of SF5-azepines to SF5-benzimidazoles. Following a protocol related to that reported by Radosevich and co-workers,12 a primary amine-substituted azepine (3aw, derived from benzylamine) was treated with trifluoroacetic anhydride (TFAA) in the presence of DABCO in toluene at 120 °C for 24 h. Under these conditions, re-aromatization of the azepine core was accompanied by intramolecular cyclodehydration, affording the corresponding SF5-benzimidazole 5a in 53% yield (Scheme 2b, middle). This cyclodehydration is plausibly driven by electrophilic activation of the azepine nitrogen by the acyl anhydride, followed by intramolecular nucleophilic attack of the pendant amine and subsequent loss of water to restore aromaticity (Scheme 2c, bottom)Because the CF3 group is classified as a PFAS motif, we next examined the transformation using difluoroacetic anhydride (DFAA) under otherwise identical conditions. As anticipated, the reaction proceeded efficiently to deliver the CF2H-substituted SF5-benzimidazole 5b in 80% yield. Notably, this skeletal reconstruction pathway proved tolerant of diverse substituents on the aromatic ring, including OMe, F, Br, and Cl, furnishing a series of CF2H-substituted SF5-benzimidazoles (5c–5f) in good to excellent yields.
Conclusions
This study demonstrates that skeletal rearrangement of SF5-nitrobenzenes provides a general and efficient strategy for constructing diverse SF5-heterocycles, including SF5-azepines, SF5-azabicyclo[3.2.0] frameworks, and SF5-benzimidazoles. Importantly, these motifs represent PFAS-free alternatives to CF3-based scaffolds, offering structurally rich and functionally attractive building blocks for pharmaceuticals, agrochemicals, and materials science. In light of the OECD classification of SF5-containing molecules as non-PFAS, this work contributes to the advancement of sustainable fluorine chemistry by demonstrating that strategic skeletal remodelling can unlock both reactivity and chemical space unique to SF5 substitution.Author contributions
MZB optimized the reaction conditions, surveyed the substrate scope, analyzed the data and discussed the results with NS. SW, TM, and CN prepared starting materials and attempted reactions. DH helped to prepare the starting materials. SO took the X-ray crystallography of 3av. MZB wrote the initial draft and NS wrote the manuscript. NS supervised the study. All authors contributed to the manuscript and approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2501744 contains the supplementary crystallographic data for this paper.15The data that support the findings of this study are available within the article and the supplementary information (SI). Supplementary information: materials and methods, experimental procedures, characterization data, and NMR spectra. See DOI: https://doi.org/10.1039/d6sc01441k.
Acknowledgements
This study was supported by the CREST program of the Japan Science and Technology Agency, entitled “Precise Material Science for Degradation and Stability” (grant number: JPMJCR21L1), by Dr Seiji Motojima (CMC Research Institute, Japan), and Kenji Koike (Tosoh Corporation).References
- (a) M. Inoue, Y. Sumii and N. Shibata, ACS Omega, 2020, 5, 10633–10640 CrossRef CAS PubMed; (b) Y. Ogawa, E. Tokunaga, O. Kobayashi, K. Hirai and N. Shibata, iScience, 2020, 23, 101467 CrossRef CAS PubMed; (c) R. Berger, G. Resnati, P. Metrangolo, E. Weber and J. Hulliger, Chem. Soc. Rev., 2011, 40, 3496–3508 RSC.
- (a) Z. Wang, A. M. Buser, I. T. Cousins, S. Demattio, W. Drost, O. Johansson, K. Ohno, G. Patlewicz, A. M. Richard, G. W. Walker, G. S. White and E. Leinala, Environ. Sci. Technol., 2021, 55, 15575–15578 CrossRef CAS PubMed; (b) E. L. Schymanski, J. Zhang, P. A. Thiessen, P. Chirsir, T. Kondic and E. E. Bolton, Environ. Sci. Technol., 2023, 57, 16918–16928 CrossRef CAS PubMed.
- (a) H. Joerss, F. Freeling, S. Van Leeuwen, J. Hollender, X. Liu, K. Nödler, Z. Wang, B. Yu, D. Zahn and G. Sigmund, Environ. Int., 2024, 193, 109061 CrossRef CAS PubMed; (b) A. Alinezhad, H. Shao, K. Litvanova, R. Sun, A. Kubatova, W. Zhang, Y. Li and F. Xiao, Environ. Sci. Technol., 2023, 57, 8796–8807 CrossRef CAS PubMed; (c) H. P. H. Arp, A. Gredelj, J. Glüge, M. Scheringer and I. T. Cousins, Environ. Sci. Technol., 2024, 58, 19925–19935 CrossRef CAS PubMed; (d) D. A. Jackson and S. A. Mabury, Environ. Toxicol. Chem., 2009, 28, 1866–1873 CrossRef CAS PubMed.
- (a) W. A. Sheppard, J. Am. Chem. Soc., 1960, 82, 4751–4752 CrossRef CAS; (b) T. Umemoto, L. M. Garrick and N. Saito, Beilstein J. Org. Chem., 2012, 8, 461–471 CrossRef CAS PubMed.
- (a) P. R. Savoie and J. T. Welch, Chem. Rev., 2015, 115, 1130–1190 CrossRef CAS PubMed; (b) G. Haufe, Tetrahedron, 2022, 109, 132656 CrossRef CAS; (c) P. Das, E. Tokunaga and N. Shibata, Tetrahedron Lett., 2017, 58, 4803–4815 CrossRef CAS; (d) M. F. Sowaileh, R. A. Hazlitt and D. A. Colby, Chem. Med. Chem., 2017, 12, 1481–1490 CrossRef CAS PubMed; (e) J. M. W. Chan, J. Mater. Chem. C, 2019, 7, 12822–12834 RSC; (f) M. Sani and M. Zanda, Synthesis, 2022, 54, 4184–4209 CrossRef CAS; (g) Y. Yang, L. Han, L. Brettnacher, L. Canavero and A. Tlili, Chem. Rev., 2025, 125, 8426–8476 CrossRef CAS PubMed; (h) J. Shou and F. Qing, Chin. J. Chem., 2025, 43, 3033–3042 CrossRef CAS.
- (a) O. S. Kanishchev and W. R. Dolbier, Angew. Chem., Int. Ed., 2015, 54, 280–284 CrossRef CAS PubMed; (b) M. Kosobokov, B. Cui, A. Balia, K. Matsuzaki, E. Tokunaga and N. Saito and Shibata, Angew. Chem., Int. Ed., 2016, 55, 10781–10785 CrossRef CAS PubMed; (c) B. Cui, M. Kosobokov, K. Matsuzaki, E. Tokunaga and N. Shibata, Chem. Commun., 2017, 53, 5997–6000 RSC; (d) B. Cui, S. Jia, E. Tokunaga, N. Saito and N. Shibata, Chem. Commun., 2017, 53, 12738–12741 RSC; (e) I. Saidalimu, Y. Liang, K. Niina, K. Tanagawa, N. Saito and N. Shibata, Org. Chem. Front., 2019, 6, 1157–1161 RSC; (f) K. Tanagawa, Z. Zhao, N. Saito and N. Shibata, Bull. Chem. Soc. Jpn., 2021, 94, 1682–1684 CrossRef CAS.
- Selected recent papers. (a) J. Shou, X. Xu and F. Qing, Angew. Chem., Int. Ed., 2021, 60, 15271–15275 CrossRef CAS PubMed; (b) Y. Jiang, X. Meng, J. Zhang, G. Wu, X. Lin and S. Guo, Nat. Commun., 2024, 15, 9705 CrossRef CAS PubMed; (c) M. Birepinte, P. A. Champagne and J. Paquin, Angew. Chem., Int. Ed., 2021, 61, e202112575 CrossRef PubMed; (d) L. Verret, K. Burchell-Reyes, J. F. Morin, J. F. Paquin and J. Fluor, Chem, 2025, 282, 110387 CAS; (e) J. Y. Shou and F. L. Qing, Angew. Chem., Int. Ed., 2022, 61, e202208860 CrossRef CAS PubMed; (f) Z. Fang, J. Y. Shou, M. N. Khan, X. B. He, X. Jia and F. L. Qing, J. Org. Chem., 2025, 90, 7841–7848 CrossRef CAS PubMed; (g) Y. Kraemer, J. A. Buldt, W. Kong, A. M. Stephens, A. N. Ragan, S. Park, Z. C. Haidar, A. H. Patel, R. Shey, R. Dagan, C. P. McLoughlin, J. C. Fettinger, D. J. Tantillo and C. R. Pitts, Angew. Chem., Int. Ed., 2024, 136, e202319930 CrossRef; (h) Y. Y. Jiang, J. S. Zhang, D. Sun, G. Wu, Z.-C. Kuang, N. Zhang, X. Y. Shen, X. L. Zhang, J. Jiang, H. Y. Yang, W. Zhang, X. S. Xue and S. Guo, J. Am. Chem. Soc., 2025, 147, 43785–43797 CrossRef CAS PubMed; (i) R. Li, C. Liu, C. Hu, J. Tsien, M. Hayes, S.-J. Chen, Y. Kanda, R. R. Merchant, B. S. Matsuura and T. Qin, Nat. Commun., 2025, 16, 10185 CrossRef CAS PubMed; (j) J. Y. Li, Y. Liu, D. Sun, G. Wu, S. S. Yue, H. Y. Yang, H. Zhang, W. Zhang, M. M. Zheng, X. S. Xue and S. Guo, J. Am. Chem. Soc., 2025, 147, 26124–26132 CrossRef CAS PubMed; (k) R. Li, C. Hu, C. Liu, T. Lyness, W. Li, C. Y. Cai, E. Crossley, Y. Kanda, R. R. Merchant, B. S. Matsuura, N. S. Williams and T. Qin, J. Am. Chem. Soc., 2025, 147, 34218–34224 CrossRef CAS PubMed; (l) Y. Kraemer, S. Park, W. Y. Kong, Y. Chen, A. J. Witt, J. A. Buldt, A. N. Ragan, L. M. Holder, D. J. Tantillo and C. R. Pitts, J. Am. Chem. Soc., 2025, 147, 27892–27904 CrossRef CAS PubMed; (m) Y. Yang, L. Han, L. Canavero, L. Brettnacher, L. Khrouz, C. Monnereau, M. Médebielle and A. Tlili, J. Am. Chem. Soc., 2025, 147, 27905–27911 CrossRef CAS PubMed; (n) H. Y. Yang, H. B. Du, Y. P. Bai, Y. H. Ding, Y.-F. Ma, G. Wu, N. Zhang, N. K. Szymczak and S. Guo, Sci. Adv., 2025, 11, eadw8408 CrossRef CAS PubMed.
- (a) P. Das, K. Niina, T. Hiromura, E. Tokunaga, N. Saito and N. Shibata, Chem. Sci., 2018, 9, 4931 RSC; (b) T. Aggarwal, K. Hada, Y. Murata, Y. Sumii, K. Tanagawa, K. Niina, S. Mori, J. Escorihuela and N. Shibata, Angew. Chem., Int. Ed., 2023, 62, e202307090 CrossRef CAS; (c) Y. Murata, K. Hada, T. Aggarwal, J. Escorihuela and N. Shibata, Angew. Chem., Int. Ed., 2024, 63, e202318086 CrossRef CAS PubMed; (d) S. R. Narra, M. Z. Bacho, M. Hattori and N. Shibata, Adv. Sci., 2024, 11, 2306554 CrossRef CAS PubMed.
- (a) J. Li, P. Tang, Y. Fan and H. Lu, Science, 2025, 389, 275–281 CrossRef CAS PubMed; (b) M. Puriņš, H. Nakahara and M. D. Levin, Science, 2025, 389, 295–298 CrossRef PubMed; (c) B. Ghosh, P. Kafle, R. Mukherjee, R. Welles, D. Herndon, K. M. Nicholas, Y. Shao and I. Sharma, Science, 2025, 387, 102–107 CrossRef CAS PubMed; (d) Z. Wang, P. Xu, S. M. Guo, C. G. Daniliuc and A. Studer, Nature, 2025, 642, 92–98 CrossRef CAS PubMed; (e) J. Qi, C. Wang, G. Wang, P. O'Neill, S. R. Dubbaka, H. T. Ang, X. Chen and J. Wu, Angew. Chem., Int. Ed., 2024, 64, e202413723 CrossRef PubMed; (f) R. T. Steele, M. Fujiu and R. Sarpong, Science, 2025, 388, 631–638 CrossRef CAS PubMed; (g) P. Rai, S. Naik, K. Gupta, K. Maji, G. Jindal and B. Maji, Nat. Commun., 2025, 16, 2991 CrossRef CAS PubMed.
- C. Nagababu, T. Muramatsu, M. Z. Bacho, S. Wu, S. Ochiai, J. Escorihuela and N. Shibata, Chem. Sci., 2025, 17, 1840–1849 RSC.
- (a) M. Kaur, S. Garg, D. S. Malhi and S. H. Sohal, Curr. Org. Chem., 2021, 25, 449–506 CAS; (b) A. Dorababu, Polycycl. Aromat. Compd., 2022, 43, 2250–2268 CrossRef; (c) D. McTavish and P. Benfield, Drugs, 1990, 39, 136–153 CrossRef CAS PubMed; (d) T. Miyazaki, H. Fujiki, Y. Yamamura, S. Nakamura and T. Mori, Cardiovasc. Drug Rev., 2007, 25, 1–13 CrossRef CAS PubMed; (e) J. A. Balfour and K. L. Goa, Drugs, 1991, 42, 511–539 CrossRef CAS; (f) M. Seto, Y. Aramaki, T. Okawa, N. Miyamoto, K. Aikawa, N. Kanzaki, S. Niwa, Y. Iizawa and M. Shiraishi, Chem. Pharm. Bull., 2004, 52, 577–590 CrossRef CAS PubMed.
- G. Li, M. N. Lavagnino, S. Z. Ali, S. Hu and A. T. Radosevich, J. Am. Chem. Soc., 2022, 145, 41–46 CrossRef PubMed.
- G. Li, S. P. Miller and A. T. Radosevich, J. Am. Chem. Soc., 2021, 143, 14464–14469 CrossRef CAS.
- E. Matador, M. J. Tilby, I. Saridakis, M. Pedrón, D. Tomczak, J. Llaveria, I. Atodiresei, P. Merino, A. Ruffoni and D. Leonori, J. Am. Chem. Soc., 2023, 145, 27810–27820 CrossRef CAS PubMed.
- CCDC2501744: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2pz8fd.
Footnote |
| † Dedicated to Professor David O'Hagan to mark his retirement from the University of St Andrews (UK) and his significant contributions to the field of organofluorine chemistry. |
| This journal is © The Royal Society of Chemistry 2026 |