Open Access Article
Open Access ArticleHighly stereoselective synthesis of allylic β-lactams via enzymatic C(sp3)–H amidation
Nawal Zahra
Jafari†
 ,
Zheyuan
Wang†
,
Zheyuan
Wang†
 ,
Anwita
Chattopadhyay
,
Anwita
Chattopadhyay
 ,
Satyajit
Roy
,
Satyajit
Roy
 and
Rudi
Fasan
and
Rudi
Fasan
 *
*
Department of Chemistry and Biochemistry, The University of Texas at Dallas, Richardson, TX 75080, USA. E-mail: rudi.fasan@utdallas.edu
First published on 19th March 2026
Abstract
β-Lactams are versatile synthons for organic synthesis as well as valuable pharmacophores for drug development. Here, we describe a biocatalytic strategy for the enantioselective synthesis of allylic β-lactams via a hemoprotein-catalyzed intramolecular C(sp3)–H amidation reaction with dioxazolone substrates. Leveraging a stepwise radical mechanism and overriding the typical reactivity of metallonitrenes, this system provides access to a variety of β-lactam products with consistently high enantioselectivity (≥99% ee) by favoring the amination of an allylic C(sp3)-H bond over the more facile functionalization of the adjacent olefin group. This works expands the range of stereoselective strategies for C–N bond formation via C(sp3)–H functionalization and demonstrates the value of new-to-nature biocatalysis to promote chemical transformations not currently accessible through chemocatalysis.
Introduction
The selective functionalization of aliphatic C(sp3)–H bonds remains a keystone challenge in synthetic organic chemistry, especially when multiple potentially reactive functionalities coexist within a molecule. Among various C–H functionalization strategies, the direct amination of C(sp3)–H bonds has attracted significant interest,1–6 primarily due to the prevalence of nitrogen-containing moieties in pharmaceuticals and natural products,7,8 as well as the intrinsic difficulty associated with integrating nitrogen into complex molecular frameworks. Among nitrogen-containing heterocycles, β-lactams represent one of the most significant pharmacophores in medicinal chemistry.9 Since the discovery of penicillin, β-lactam scaffolds have been indeed central to the development of antimicrobial agents and enzyme inhibitors.9 The structural rigidity, ring strain, and unique reactivity make them invaluable in both drug development as well as versatile building blocks for the synthesis of organic molecules (Fig. 1A).10Because of their synthetic and pharmacological relevance, significant efforts have been devoted to the development of synthetic methods for the stereoselective synthesis of β-lactams.11,12 State-of-the-art catalytic methods include the use of palladium-catalyzed carbonylative cycloaddition reactions,13 copper-catalyzed Kinugasa/Michael domino reactions,14 and copper-catalyzed C(sp3)–C(sp2) cross coupling reaction,15 iron-catalyzed olefin oxyamidation,16 among others.11,12 The possibility to construct β-lactam rings via a direct, C(sp3)–H functionalization process is particularly attractive due to the ubiquitous presence of aliphatic C–H bonds in organic molecules. In this context, current methods include carbene C–H insertion with diazoamides17–19 and the palladium-catalyzed β-C(sp3)–H amidation of functionalized amides bearing a directing group such as quinolines or amide protecting groups. Despite this progress, chemocatalytic methods for the stereoselective synthesis of these β-lactam compounds via an undirected C(sp3)–H amination reaction are not available.
Over the past years, considerable progress has been made in the development of biocatalytic strategies for intra- and intermolecular C(sp3)–H amination reactions via abiological nitrene transfer chemistry using engineered hemoproteins and other metalloenzymes.20–33 Complementing and expanding beyond the scope of chemocatalytic nitrene transfer methods for C(sp3)–H amination,7,34–39 these methods have enabled the asymmetric synthesis of sultams, carbamates, sulfamide, lactams, as well as benzylic, allylic and propargylic amines with high catalyst-controlled selectivity. Relevant to the present work, we recently demonstrated the possibility to engage dioxazolone-based nitrene precursors in a hemoprotein-catalyzed intramolecular amination of benzylic C(sp3)–H bonds for the stereoselective synthesis of β-, γ-, and δ-lactams with high activity and enantioselectivity (Fig. 1C).32
Building on this progress, we were interested in targeting the enantioselective synthesis of allylic β-lactams via an intramolecular allylic C(sp3)–H amination process, as the resulting products combine the strained β-lactam pharmacophore with a versatile allylic functionality for further diversification. These compounds also constitute convenient precursors to allylic amines, which are valuable motifs in bioactive molecules in their own right (Fig. 1A).40 While intermolecular allylic C–H amination have been achieved with both transition metal catalysts5,8,34,36,41–46 and biocatalysts,25,29 there are no reported examples of allylic C–H amination to make β-lactams. This transformation indeed presents peculiar challenges in that functionalization of the more reactive olefin group tends to outcompete functionalization of the adjacent allylic C(sp3)–H bond in the presence of metallonitrene intermediates, in particular when the latter would lead to formation of a strained 4-membered ring.47–55 This reactivity bias has been indeed exploited to obtain γ- (or δ)-lactams via a variety of olefin difunctionalization strategies in the presence of both late and first-row transition metal catalysts (Fe, Os, Rh, Ir) (Fig. 1D).50–55
Herein, we demonstrate the possibility to overcome this inherent reactivity bias with the development of an enzyme-catalyzed β-lactam forming reaction via chemo- and stereoselective allylic C–H amination (Fig. 1E). This transformation, which is shown to proceed via a stepwise hydrogen atom transfer/radical rebound mechanism, enables the expeditious preparation of a variety of allylic β-lactams with high enantioselectivity from readily available starting materials.
Results and discussion
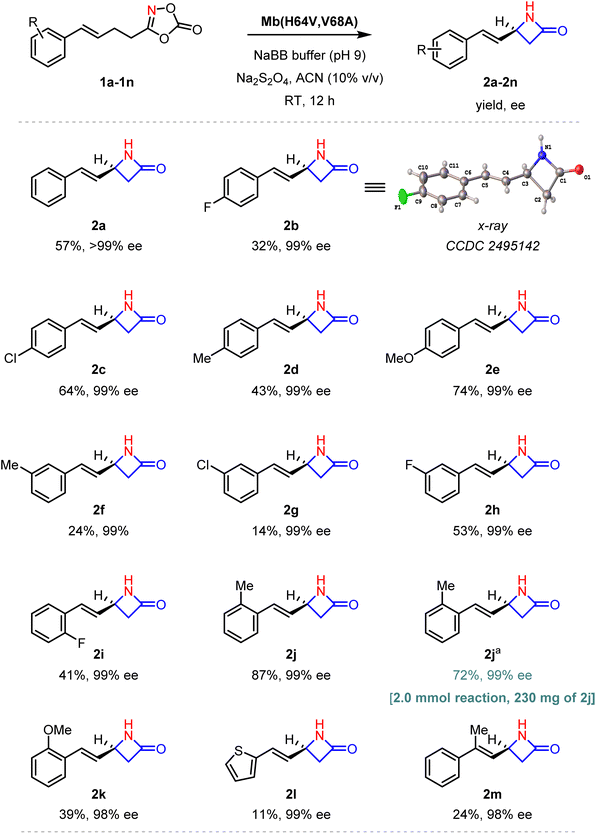
In initial studies, a diverse set of heme-containing enzymes and proteins, including myoglobin, various cytochromes P450 (e.g. P450BM3, CYP119, P411–CHF56 variants) cytochromes c, and others, were screened for their ability to promote the intramolecular C(sp3)–H amination of allylic dioxazolone (1a) to give the desired β-lactam 2a. However, none of the enzymes produced any detectable amount of the desired product (Table S1). Of note, the same reaction failed or gave minimal reactivity in the presence of several porphyrin-based catalysts commonly used for carbene transfer reactions such as FeIII(TPP)Cl and CoII(TPP) (Fig. 2). We then extended our screening to an in-house collection of purified myoglobin variants library containing a range of single to quadruple mutations at residues surrounding the heme cofactors. While the large majority of these variant showed no activity, Mb(H64V) displayed basal activity for formation of the desired product 2a (2% yield) with high enantioselectivity (99% ee; Fig. 2). Among these proteins, Mb(H64V,V68A), which was previously found to be an effective catalyst for many carbene transfer reactions57 as well as for intramolecular C–H amidation with dioxazolones,32 proved to be an optimal biocatalyst for the present reaction (Fig. 2). Interestingly, the activity and selectivity of Mb variants containing alanine and/or glycine mutations at the 64 or 68 positions significantly decreased when compared to Mb(H64V,V68A), and a similar effect was seen for Mb variants incorporating additional active site mutations in the Mb(H64V,V68A) background (e.g. L29A/F), indicating that the enzyme's reactivity is sensitive to subtle alterations in the configuration of the active site (Fig. 2B). Based on previous studies,32 we anticipated that the inclusion of an organic co-solvent would enhance the desired C–H amidation process by disfavoring formation of the amide byproduct 3a, which derives from reduction and protonation of the heme-nitrene intermediate.21 Indeed, screening showed that the use of acetonitrile (ACN) at 10% (v/v) was optimal to maximize the yield of the C–H amidation product 2a over the byproduct 3a (Tables S2 and S3). Additional tuning of the reaction showed that slightly alkaline (pH 9) conditions are optimal for the reaction (Table S4). Under these optimized conditions, Mb(H64V,V68A) catalyzes the formation of the allylic β-lactam product 1a in high enantiopurity (>99% ee) in 57% yield. The configuration of the β-lactam product was determined to be S based on crystallographic analysis of the fluorinated analog 2b (Table S5 and Fig. S1)With optimized reaction conditions in hand, the Mb(H64V,V68A)-catalyzed allylic C–H amination reaction was tested against various dioxolanone substrates (Fig. 3). Notably, a range of different electron withdrawing and electron donating groups at the ortho, meta or para position of the phenyl ring in the dioxazolone substrates were tolerated by the enzyme to give the desired allylic β-lactam products 2b–2k in good to satisfactory yields and with consistently high enantioselectivity (99% ee). Interesting structure–activity trends could be derived from this set of reactions. For the para-substituted substrates, the yields of C–H aminated product was found to depend, in part, on the electronic nature of the substituent, as indicated by the significantly higher yield for the methoxy-substituted product 2e (74% yield) compared to fluorine-containing counterpart 2b (32% yield) (Fig. 3). For the meta substituted dioxazolones, on the other hand, the yield of the C–H amination product appeared to be influenced in larger part by the size of the substituent, as suggested by the higher yield of the meta-fluorinated product 2h (53%) compared to the bulkier, methyl- and chloro-substituted counterparts 2f and 2g (24–14% yield). For the ortho-substituted dioxazolones, the ortho-methyl substituted substrate gave the highest yield of the β-lactam product 2j (87%) compared to methoxy (2k) and fluoro-substituted analogs (2i), which were produced in 39–41% yields. The thiophene-containing substrate 1l was also processed by the enzyme to give the desired β-lactam 2l with excellent enantioselectivity, albeit in low yield (11%). Importantly, the reactions with 1m further demonstrated that the substitution at the level of the olefin group is also compatible with the present system to afford the allylic β-lactam 2m in moderate yields (24%) but high enantiomeric excess (98% ee). To further demonstrate the synthetic utility of the present methodology, a preparative scale reaction (2 mmol) was carried out using 1j as the substrate, which afforded 230 mg of the β-lactam product 2j in 72% isolated yield and high enantiopurity (99% ee) (Fig. 3). Overall, the high enantioselectivity of this scaled-up reaction along with the preserved and consistently high enantioselectivity observed across all the products in Fig. 3 highlight the generality of the Mb(H64V,V64A) catalyst and methodology for the asymmetric synthesis of allylic β-lactams.
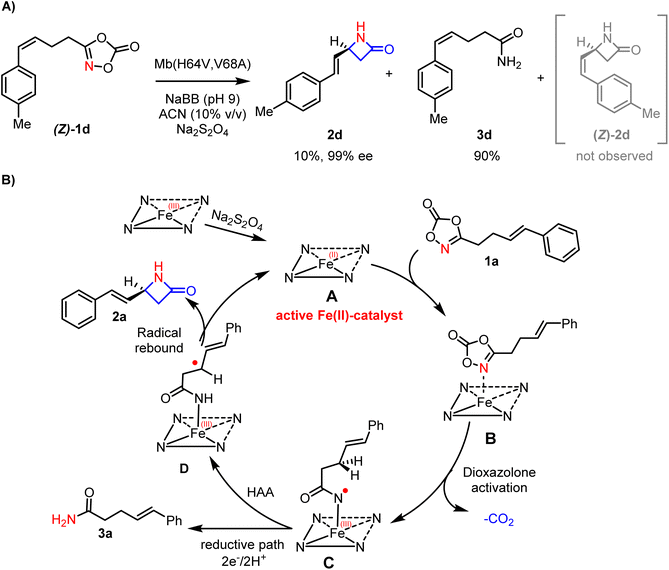
Experiments were then carried out to investigate the mechanism of the reaction, also for vis-a-vis comparison with previous mechanistic studies on biocatalytic formation of γ-lactams from dioxazolones.32 Consistent with the previous finding, transformation of the cis dioxazolone substrate (Z)-1d produced only the E-configured product 2d (Fig. 4A), indicating full isomerization of the double bond at the level of an allylic radical intermediate prior to the C–N bond formation step. Then intramolecular kinetic isotope effect (KIE) experiments were carried out using the monodeuterated substrate 1a–d, which showed the absence of a positive KIE at the level of the aminated C–H bond (KIE = 1.04 ± 0.1), indicating that C–H bond cleavage is not part of the rate determining steps of the reaction (see SI). Based on these results and previous studies,58 we propose a mechanism in which reaction of the catalytically active ferrous protein reacts with the dioxazolone substrate to form a heme–dioxazolone complex which undergoes decarboxylation to produce the acyl-nitrene intermediate C (Fig. 4B). The latter mediates an allylic C–H bond abstraction to the generate the allylic radical intermediate D, followed by C–N bond formation via a radical rebound process. As suggested by previous calculations,32 and consistent with the E/Z isomerization and KIE experiments of Fig. 4, the C–N bond-forming step is believed to be the rate-limiting and enantioselectivity-determining step of the reaction.
Conclusions
In conclusion, we have reported a first example of a biocatalytic strategy for the enantioselective synthesis of allylic β-lactams via an intramolecular C(sp3)–H amidation reaction. Using an engineered myoglobin variant, this approach provides access to medicinally and synthetically valuable β-lactam products with excellent enantioselectivity. Mechanistic studies support a stepwise radical pathway, where the formation of the C–N bond via a radical rebound mechanism plays a pivotal role in determining enantioselectivity. A most notable feature of this strategy is its exquisite biocatalyst-controlled chemoselectivity, which enables construction of a strained β-lactam ring by favoring functionalization of an allylic C(sp3)–H bond over the more facile functionalization of the adjacent olefin group, typically observed with synthetic transition metal catalysts (Fig. 1D).55 These results illustrate the value of new-to-nature biocatalysis not only for expanding the range of stereoselective strategies for C–N bond formation via direct C(sp3)–H functionalization but also for accessing chemical transformations not currently accessible through chemocatalysis.Author contributions
S. R. and R. F. conceptualized the study. N. Z. J. and Z. W. performed the bulk of the experiments with assistance by A. C. and S. R., under R. F. supervision. S. R., N. Z. J., Z. W. and R. F. wrote the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2495142 contains the supplementary crystallographic data for this paper.59All experimental procedures and spectroscopic data can be found in the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc01440b.
Acknowledgements
This work was supported by the U.S. National Institute of Health Grant R35GM158365 and grant RR230018 from the Cancer Prevention and Research Institute of Texas (CPRIT). R.F. acknowledges chair endowment support from the Robert A. Welch Foundation (Chair, AT-0051-20221212). The authors are grateful to the UTD Center for High-Throughput Reaction Discovery & Synthesis supported by grant RR230018 from the Cancer Prevention and Research Institute of Texas. The authors are grateful to Dr. William Brennessel (University of Rochester) for assistance with the crystallographic analyses.Notes and references
- K. W. Fiori and J. Du Bois, J. Am. Chem. Soc., 2007, 129, 562–568 CrossRef CAS PubMed.
- J. L. Roizen, D. N. Zalatan and J. Du Bois, Angew. Chem., Int. Ed., 2013, 52, 11343–11346 CrossRef CAS PubMed.
- C. Liang, F. Robert-Peillard, C. Fruit, P. Müller, R. H. Dodd and P. Dauban, Angew. Chem., Int. Ed., 2006, 45, 4641–4644 CrossRef CAS.
- H. Khatua, S. Das, S. Patra, S. K. Das, S. Roy and B. Chattopadhyay, J. Am. Chem. Soc., 2022, 144, 21858–21866 CrossRef CAS PubMed.
- S. M. Paradine and M. C. White, J. Am. Chem. Soc., 2012, 134, 2036–2039 CrossRef CAS.
- E. T. Hennessy and T. A. Betley, Science, 2013, 340, 591–595 CrossRef CAS PubMed.
- J. L. Roizen, M. E. Harvey and J. Du Bois, Acc. Chem. Res., 2012, 45, 911–922 CrossRef CAS PubMed.
- G. Dequirez, V. Pons and P. Dauban, Angew. Chem., Int. Ed., 2012, 51, 7384–7395 CrossRef CAS PubMed.
- L. M. Lima, B. N. M. d. Silva, G. Barbosa and E. J. Barreiro, Eur. J. Med. Chem., 2020, 208, 112829 CrossRef CAS PubMed.
- H. Öztürk, E. Ozkirimli and A. Özgür, PLoS One, 2015, 10, e0117874 CrossRef PubMed.
- A. Saura-Sanmartin and L. Andreu-Ardil, Org. Biomol. Chem., 2023, 21, 3296–3306 RSC.
- S. Hosseyni and A. Jarrahpour, Org. Biomol. Chem., 2018, 16, 6840–6852 RSC.
- T. Inagaki, T. Kodama and M. Tobisu, Nat. Catal., 2024, 7, 132–138 CrossRef CAS.
- M. M. C. Lo and G. C. Fu, J. Am. Chem. Soc., 2002, 124, 4572–4573 CrossRef CAS PubMed.
- F.-L. Wang, L. Liu, C.-J. Yang, C. Luan, J. Yang, J.-J. Chen, Q.-S. Gu, Z.-L. Li and X.-Y. Liu, Angew. Chem., Int. Ed., 2023, 62, e202214709 CrossRef CAS PubMed.
- J. Kweon, D. Kim, S. Kang and S. Chang, J. Am. Chem. Soc., 2022, 144, 1872–1880 CrossRef CAS PubMed.
- M. P. Doyle, W. Hu, A. G. H. Wee, Z. Wang and S. C. Duncan, Org. Lett., 2003, 5, 407–410 CrossRef CAS PubMed.
- C. H. Yoon, A. Nagle, C. Chen, D. Gandhi and K. W. Jung, Org. Lett., 2003, 5, 2259–2262 CrossRef CAS PubMed.
- M. K.-W. Choi, W.-Y. Yu and C.-M. Che, Org. Lett., 2005, 7, 1081–1084 CrossRef CAS PubMed.
- (a) Y. Yang, I. Cho, X. Qi, P. Liu and F. H. Arnold, Nat. Chem., 2019, 11, 987–993 CrossRef CAS PubMed; (b) J. A. McIntosh, P. S. Coelho, C. C. Farwell, Z. J. Wang, J. C. Lewis, T. R. Brown and F. H. Arnold, Angew. Chem., Int. Ed., 2013, 52, 9309–9312 CrossRef CAS.
- R. Singh, M. Bordeaux and R. Fasan, ACS Catal., 2014, 4, 546–552 CrossRef CAS PubMed.
- R. Singh, J. N. Kolev, P. A. Sutera and R. Fasan, ACS Catal., 2015, 5, 1685–1691 CrossRef CAS PubMed.
- P. Dydio, H. M. Key, H. Hayashi, D. S. Clark and J. F. Hartwig, J. Am. Chem. Soc., 2017, 139, 1750–1753 CrossRef CAS PubMed.
- V. Steck, J. N. Kolev, X. Ren and R. Fasan, J. Am. Chem. Soc., 2020, 142, 10343–10357 CrossRef CAS PubMed.
- Z.-J. Jia, S. Gao and F. H. Arnold, J. Am. Chem. Soc., 2020, 142, 10279–10283 CrossRef CAS PubMed.
- M. A. Vila, V. Steck, S. Rodriguez Giordano, I. Carrera and R. Fasan, ChemBioChem, 2020, 21, 1981–1987 CrossRef CAS PubMed.
- S. V. Athavale, S. Gao, Z. Liu, S. C. Mallojjala, J. S. Hirschi and F. H. Arnold, Angew. Chem., Int. Ed., 2021, 60, 24864–24869 CrossRef CAS PubMed.
- S. V. Athavale, S. Gao, A. Das, S. C. Mallojjala, E. Alfonzo, Y. Long, J. S. Hirschi and F. H. Arnold, J. Am. Chem. Soc., 2022, 144, 19097–19105 CrossRef CAS PubMed.
- Z. Liu, Z.-Y. Qin, L. Zhu, S. V. Athavale, A. Sengupta, Z.-J. Jia, M. Garcia-Borràs, K. N. Houk and F. H. Arnold, J. Am. Chem. Soc., 2022, 144, 80–85 CrossRef CAS PubMed.
- S. Gao, A. Das, E. Alfonzo, K. M. Sicinski, D. Rieger and F. H. Arnold, J. Am. Chem. Soc., 2023, 145, 20196–20201 CrossRef CAS PubMed.
- E. Alfonzo, D. Hanley, Z.-Q. Li, K. M. Sicinski, S. Gao and F. H. Arnold, J. Am. Chem. Soc., 2024, 146, 27267–27273 CrossRef CAS PubMed.
- S. Roy, D. A. Vargas, P. Ma, A. Sengupta, L. Zhu, K. N. Houk and R. Fasan, Nat. Catal., 2024, 7, 65–76 CrossRef CAS PubMed.
- R. Mao, S. Gao, Z.-Y. Qin, T. Rogge, S. J. Wu, Z.-Q. Li, A. Das, K. N. Houk and F. H. Arnold, Nat. Catal., 2024, 7, 585–592 CrossRef CAS PubMed.
- T. Ide, K. Feng, C. F. Dixon, D. Teng, J. R. Clark, W. Han, C. I. Wendell, V. Koch and M. C. White, J. Am. Chem. Soc., 2021, 143, 14969–14975 CrossRef CAS PubMed.
- A. Fanourakis and R. J. Phipps, Chem. Sci., 2023, 14, 12447–12476 RSC.
- F. Collet, C. Lescot and P. Dauban, Chem. Soc. Rev., 2011, 40, 1926–1936 RSC.
- M. Ju and J. M. Schomaker, Nat. Rev. Chem., 2021, 5, 580–594 CrossRef CAS PubMed.
- Y. Park, Y. Kim and S. Chang, Chem. Rev., 2017, 117, 9247–9301 CrossRef CAS PubMed.
- W.-C. C. Lee and X. P. Zhang, Angew. Chem., Int. Ed., 2024, 63, e202320243 CrossRef CAS PubMed.
- S. Nag and S. Batra, Tetrahedron, 2011, 67, 8959–9061 CrossRef CAS.
- D. N. Zalatan and J. Du Bois, J. Am. Chem. Soc., 2008, 130, 9220–9221 CrossRef CAS PubMed.
- C. Lescot, B. Darses, F. Collet, P. Retailleau and P. Dauban, J. Org. Chem., 2012, 77, 7232–7240 CrossRef CAS PubMed.
- F. Collet, C. Lescot, C. Liang and P. Dauban, Dalton Trans., 2010, 39, 10401–10413 RSC.
- H. Lebel, C. Spitz, O. Leogane, C. Trudel and M. Parmentier, Org. Lett., 2011, 13, 5460–5463 CrossRef CAS PubMed.
- Y. Nishioka, T. Uchida and T. Katsuki, Angew. Chem., Int. Ed., 2013, 52, 1739–1742 CrossRef CAS PubMed.
- V. Köhler, Y. M. Wilson, M. Dürrenberger, D. Ghislieri, E. Churakova, T. Quinto, L. Knörr, D. Häussinger, F. Hollmann, N. J. Turner and T. R. Ward, Nat. Chem., 2013, 5, 93–99 CrossRef PubMed.
- H. Lebel, K. Huard and S. Lectard, J. Am. Chem. Soc., 2005, 127, 14198–14199 CrossRef CAS PubMed.
- K. Guthikonda, P. M. Wehn, B. J. Caliando and J. Du Bois, Tetrahedron, 2006, 62, 11331–11342 CrossRef CAS.
- K. W. Fiori, C. G. Espino, B. H. Brodsky and J. Du Bois, Tetrahedron, 2009, 65, 3042–3051 CrossRef CAS.
- J. Kluegge, E. Herdtweck and T. Bach, Synlett, 2004, 2004, 1199–1202 Search PubMed.
- T. J. Donohoe, C. K. A. Callens and A. L. Thompson, Org. Lett., 2009, 11, 2305–2307 CrossRef CAS PubMed.
- J. H. Conway, Jr. and T. Rovis, J. Am. Chem. Soc., 2018, 140, 135–138 CrossRef PubMed.
- S. Y. Hong and S. Chang, J. Am. Chem. Soc., 2019, 141, 10399–10408 CrossRef CAS PubMed.
- H. Lei, J. H. Conway, Jr., C. C. Cook and T. Rovis, J. Am. Chem. Soc., 2019, 141, 11864–11869 CrossRef CAS PubMed.
- S. Kim, D. Kim, S. Y. Hong and S. Chang, J. Am. Chem. Soc., 2021, 143, 3993–4004 CrossRef CAS PubMed.
- R. K. Zhang, K. Chen, X. Huang, L. Wohlschlager, H. Renata and F. H. Arnold, Nature, 2019, 565, 67–72 CrossRef CAS PubMed.
- R. Fasan and M. G. Siriboe, Bull. Chem. Soc. Jpn., 2022, 80, 4–13 Search PubMed.
- D. A. Vargas, X. Ren, A. Sengupta, L. Zhu, S. Roy, M. Garcia-Borràs, K. N. Houk and R. Fasan, Nat. Chem., 2024, 16, 817–826 CrossRef CAS PubMed.
- CCDC2495142: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2prdgb.
Footnote |
| † Equal contribution. |
| This journal is © The Royal Society of Chemistry 2026 |


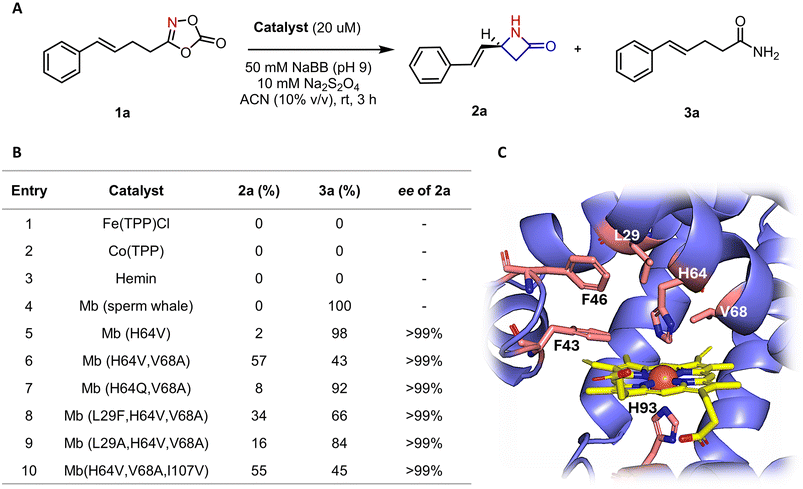
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) µM protein, 10 mM 1a, 10 mM Na2S2O4 in sodium borate buffer (50 mM, pH 7), 10% (v/v) acetonitrile, 3 h, room temperature, anaerobic conditions. The yields and product distribution were determined by GC using calibration curves of the isolated product.
µM protein, 10 mM 1a, 10 mM Na2S2O4 in sodium borate buffer (50 mM, pH 7), 10% (v/v) acetonitrile, 3 h, room temperature, anaerobic conditions. The yields and product distribution were determined by GC using calibration curves of the isolated product.