Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Using xenon difluoride and 2 Li[Al{OC(CF3)3}4] as an oxidant: from organoxenonium intermediates to (fluoro-)biphenyl radical cations
Konstantin Kloiber a,
Tim Heizmanna,
Benoît Lacombea,
Philipp Thielert
a,
Tim Heizmanna,
Benoît Lacombea,
Philipp Thielert bc,
Malte Sellin
bc,
Malte Sellin a,
Tim Schwandta,
Sabine Richertbc,
Stefan Weber
a,
Tim Schwandta,
Sabine Richertbc,
Stefan Weber b and
Ingo Krossing
b and
Ingo Krossing *a
*a
aInstitute of Inorganic and Analytical Chemistry, Freiburg Materials Research Center (FMF), University of Freiburg, Albertstr. 21, 79104 Freiburg, Germany. E-mail: krossing@uni-freiburg.de
bInstitute of Physical Chemistry, University of Freiburg, Albertstr. 21, 79104 Freiburg, Germany
cInstitute of Physical Chemistry II, University of Ulm, Lise-Meitner-Str. 16, 89081 Ulm, Germany
First published on 5th May 2026
Abstract
The feasibility of employing the XeF2/2Li[Al(ORF)4] (ORF = OC(CF3)3) system as a deelectronator, formally generating Xe(g) + 2LiF(s) + 2□+, was investigated. While this proved successful for substrates like ferrocene and 9,10-dichlorooctafluoroanthracene, the system was found to react with the solvents benzene, fluorobenzene (FB) and 1,2-difluorobenzene (2FB). The reaction led to the formation of biphenyl radical cations as arene coupling products. In contrast, the reaction with 1,2,3-trifluorobenzene (3FB) allowed the observation of a persistent organoxenonium ion. Low-temperature NMR control reactions and quantum chemical calculations suggest a xenonium-mediated coupling reaction with organoxenonium ions as intermediates. As a reference for the organoxenonium ions proposed, the perfluorinated tight ion-pair (C6F5)Xe-F-Al(ORF)3 and the salt [C6F5Xe][F{Al(ORF)3}2] were synthesized to assess their stability in combination with the alkoxyaluminate anions used in this work. Oxidation studies showed pronounced solvent effects, with strong oxidation observed in pentafluorobenzene, while in MeCN, ferrocene was found to be oxidized already by XeF2 alone. These findings not only expand the synthetic utility of XeF2-based oxidation systems but also provide new insight into the reactivity of organoxenonium ions, contributing to the growing understanding of the role of xenon chemistry in selective oxidative transformations.
Introduction
The development of high potential oxidants has experienced renewed interest over the past decade, particularly by combination of oxidant cations with weakly coordinating anions (WCAs).1–8 The availability of robust WCAs, e.g. [Al(ORF)4]−, [(RFO)3Al–F–Al(ORF)3]− (ORF = OC(CF3)3), [Al(OTeF5)4]− or [(C6F5)3B-CN-B(C6F5)3]−, enables the stabilization of highly reactive cations and allows redox potentials to be shifted to increasingly positive values.9–12 Very recently, this field has been expanded by the development of new aluminum-based Lewis superacids and related weakly coordinating anions derived from organotellurium ligands.13 Two general strategies are commonly employed to access reactive cations stabilized by WCAs: (1) metathesis reactions by using for example Li+, Ag+ or [CPh3]+ salts for halide abstraction,8,12,14,15 and (2) direct oxidation of the substrate with a high potential WCA-oxidant salt (![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Ox[WCA]).4,7,14,16,17 Here, inorganic reagents such as NO[Al(ORF)4], Cu[Al(ORF)4] or Ag[Al(ORF)4] are readily accessible through a simple metathesis reaction from the corresponding (even commercial)18 lithium salt and reach very high (solvent-dependent) oxidation potentials up to 1.52 V (NO+; 4FB = 1,2,3,4-F4C6H2) and 1.50 V (Cu+, Ag+, 5FB = C6F5H) vs. ferrocene/ferrocenium (Fc+/0).15,19–21 Notably, borate-WCAs such as [B(C6F5)4]− or [B(3,5-C6H3(CF3)2)4]− anodically oxidize already at moderate potentials to give fluorinated biphenyls electrosynthetically in good yields, showcasing their limited stability at higher potentials.22 In addition, the solvent-free Ag[B(C6F5)4] decomposes to AgC6F5 and B(C6F5)3.23 Furthermore, although classical WCAs such as [MF6]− (P, Sb, As) have high electrochemical stability, they tend to induce low solubilities and anion coordination in organic media; the latter is often followed by fluoride ion abstraction.24–26
Ox[WCA]).4,7,14,16,17 Here, inorganic reagents such as NO[Al(ORF)4], Cu[Al(ORF)4] or Ag[Al(ORF)4] are readily accessible through a simple metathesis reaction from the corresponding (even commercial)18 lithium salt and reach very high (solvent-dependent) oxidation potentials up to 1.52 V (NO+; 4FB = 1,2,3,4-F4C6H2) and 1.50 V (Cu+, Ag+, 5FB = C6F5H) vs. ferrocene/ferrocenium (Fc+/0).15,19–21 Notably, borate-WCAs such as [B(C6F5)4]− or [B(3,5-C6H3(CF3)2)4]− anodically oxidize already at moderate potentials to give fluorinated biphenyls electrosynthetically in good yields, showcasing their limited stability at higher potentials.22 In addition, the solvent-free Ag[B(C6F5)4] decomposes to AgC6F5 and B(C6F5)3.23 Furthermore, although classical WCAs such as [MF6]− (P, Sb, As) have high electrochemical stability, they tend to induce low solubilities and anion coordination in organic media; the latter is often followed by fluoride ion abstraction.24–26
Yet, despite the fact that inorganic oxidants paired with excellent WCAs have enabled the isolation and study of novel reactive species and unusual oxidation states throughout the periodic system,2,5,7,27–29 reagents with NO+, Cu+, Ag+ or related active cations are limited by two major challenges:30,31 first, their redox potential is strongly influenced by the solvent and environment, and second, the cations tend to undergo side reactions by forming complexes with the substrate (e.g. Ag+ and Fe(CO)5) or undergoing ligand substitution (e.g., NO+ and Fe(CO)5).13,17,28,29 Consequently, alternative oxidants have been developed in recent years to address these limitations. They act as selective deelectronators that do not react with the substrates through further bond-breaking and bond-making processes (Fig. 1a). With such reagents in hand, we and others were able to access new highly reactive species, e.g., the [Fe(CO)5]+ metalloradical or the [Ph3P-PPh3]2+ dication (Fig. 1b).4,17,32–34
 | ||
| Fig. 1 Selected examples for (a) selective deelectronators including their half wave potential given vs. Fc+/0 and (b) some selected applications to prepare unusual compounds in synthetic chemistry.4,17,32–34,42–46 The generated cation is shown in regular font with summary or structural formula and the deelectronator reagents in bold using the abbreviation defined in (a). Cp* = C5Me5; Dipp = 2,6-iPr2-C6H3. (c) Application of XeF2 as an oxidative system in combination with Lewis acids (LA = BF3, B(C6F5)3, Al(ORF)3) or silylated derivatives of strong Brønsted acids (A-SiMe3 = TfOSiMe3, Tf2NSiMe3, F5TeOSiMe3).3 (d) The oxidative system 2Ag[Al(ORF)4]/I2.47 (e) This work: investigation of the oxidative system XeF2/2Li[Al(ORF)4] with different (fluorinated) arenes and evaluation of its oxidative potential in acetonitrile and 5FB (anthraceneHal = 9,10-dichlorooctafluoroanthracene). | ||
In this realm, XeF2 is known as a versatile reagent with strong oxidizing and fluorinating properties and, unlike higher xenon fluorides, is kinetically stable and comparatively easy to handle, which has made it a widely used reagent in synthesis.35 In the presence of strong fluoride acceptors or Lewis acids, XeF2 can be converted into highly reactive and strongly oxidizing systems that are commonly rationalized in terms of fluoride abstraction and [XeF]+-type reactivity.36–38 In particular, combinations of XeF2 with strong Lewis acids such as BF3 and B(C6F5)3, or Lewis superacids such as Al(ORF)3, have been shown to display markedly enhanced reactivity, ranging from electrophilic fluorination/dearomatization of fluoroarenes to oxidation of suitable substrates, depending on the reaction partner and medium (Fig. 1c).3,37–41
Building on this chemistry, the present study explores the “activation” of XeF2 by a combination of two equivalents of Li[Al(ORF)4] as an oxidizing system. In analogy to the known XeF2/2LA and the closely related 2 Ag[WCA]/I2 systems (Fig. 1c and d), our approach aims to exploit the formally possible 2e− redox capabilities of the system XeF2 + 2Li+ → “Xe2+” + 2LiF → Xe(g) + 2LiF(s) + 2□+ (□+ = positive hole) by utilizing the thermodynamic driving forces of the formation of Xe gas and stable solid LiF (ΔLattH = 1030 kJ mol−1, Fig. 1e).48 Here, we found the XeF2/2Li[Al(ORF)4] system to act as a deelectronator for simple substrates like Fc0, but also delocalized high potential systems like anthraceneHal (1.42 V vs. Fc+/0) in MeCN or 5FB solution (Fig. 1a). This already underpins that the system reacts with the somewhat less oxidation stable solvents fluorobenzene (FB) and 1,2-difluorobenzene (2FB) to form biphenyl radical cations as arene coupling products. In contrast, 1,2,3-trifluorobenzene (3FB) does not yield radical cations but instead allows observation of a persistent organoxenonium ion. The mechanism of arene coupling and the stability of the involved organoxenonium ions were investigated.
Results and discussion
Initial observations
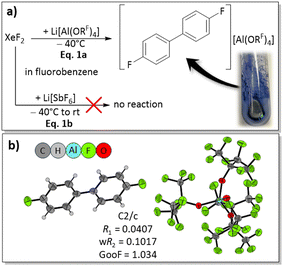
To investigate whether commercially available Li[Al(ORF)4] can serve to activate XeF2 as a potential two-electron oxidant, the reaction of XeF2 with two equivalents of Li[Al(ORF)4] in fluorobenzene (FB) was chosen as a starting point. Immediately after XeF2 was added to the Li[Al(ORF)4] solution, the reaction mixture turned intensely blue, potentially indicating the formation of radicals (Fig. 2a, eqn (1a)).49 The remarkable speed in color change suggested that the lithium salt of the alkoxyaluminate is a promising source for XeF2 activation. In contrast, the analogous reaction employing Li[SbF6] resulted in no observable changes, neither at −40 °C nor at room temperature (Fig. 2a). This is probably caused by the high lattice energy of Li[SbF6] and therefore its poor solubility in FB. When the deep blue reaction mixture resulting from reaction with Li[Al(ORF)4] was layered with n-pentane and stored at −35 °C, some blue crystals of [4,4′-difluorobiphenyl][Al(ORF)4] suitable for scXRD were isolated (scXRD = single crystal X-ray diffraction; Fig. 2b). Triggered by this observation, the reaction was investigated in more depth.Scope of the reaction and characterization
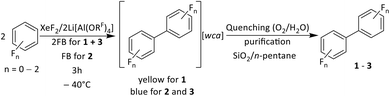
To investigate the generality of the reaction in Fig. 2a, the system XeF2/2Li[Al(ORF)4] was also used in combination with a mixture of benzene in 1,2-difluorobenzene (2FB) and in pure 2FB. The reaction of XeF2/2Li[Al(ORF)4] with benzene/2FB led to an intensely yellow colored solution, while the system in 2FB also had an intense blue color. After quenching these reactions by adding H2O and stirring under atmospheric conditions, followed by work up including purification by column chromatography (Silica 60/n-pentane), the neutral species biphenyl (1, bb), 4,4′-difluorobiphenyl (2, fbfb) and 3,3′,4,4′-tetrafluorobiphenyl (3, 2fb2fb) were isolated and characterized by NMR spectroscopy.EPR analyses were performed on the intensely colored solutions, leading after work up to 1–3. The simulations of the EPR spectra agree with their assignment as the radical cations [1]˙+, [2]˙+ and [3]˙+ (see Fig. 3) and prove the formation of the radical cations in Scheme 1.
In contrast to the three reactions of benzene, FB and 2FB, the reaction of 1,2,3-trifluorobenzene (3FB) in 5FB proceeds more slowly and is accompanied by a color change of the solution to brown. In the EPR spectrum of the reaction mixture, no paramagnetic species were detected. Low-temperature NMR analysis of the reaction mixture revealed several fluorinated arenes in the 19F NMR spectrum, including a species identified as the organoxenonium ion [2,3,4-C6H2F3Xe]+ (see Fig. 4). Remarkably, this species could also be observed at room temperature. Note that two factors likely contribute to the stability of xenonium ions in the present case: (1) stabilization by the electron-withdrawing effect of the fluorine substituents in the ortho- and para-position and (2) for ortho-fluorinated substrates, an additional fluorine-xenon interaction/chelate effect.49 Both likely account for the stability of this xenonium ion. Apparently, the enhanced stability of the observed xenonium ion and the low reactivity of the π-system of 3FB are the reasons why the reaction stops at this stage (see below, Scheme 5, proposed mechanism, step C). However, common side reactions of excess XeF2, such as the fluorination of arenes, predominate under these conditions.50
Stability of XeF2/2Li[Al(ORF)4] and its potential
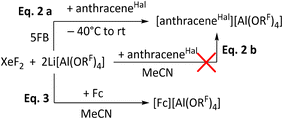
After establishing that XeF2/2Li[Al(ORF)4] induces coupling of lower fluorinated arenes, like FB and 2FB, the system was investigated using 5FB as solvent, which has the highest published positive electrochemical solvent limit of all fluorobenzenes of +2.67 V vs. Fc+/0.19 XeF2 alone was found to be stable at room temperature for up to two hours, after which fluorination of 5FB was observed (SI 4.1). Subsequently, the mixture of XeF2 and 2Li[Al(ORF)4] was analyzed by NMR spectroscopy at −35 °C. This analysis revealed that similar fluorination reactions occur rapidly in this mixture even at low temperatures. Therefore, the addition of XeF2 to a solution of Li[Al(ORF)4] in the presence of a redox active substrate appeared as a logical consequence to estimate its potential. For this purpose, 9,10-dichlorooctafluoroanthracene (anthraceneHal) was selected. Upon adding XeF2 and gradual warming to rt, a color change to deep green took place, characteristic of the radical cation [anthraceneHal]˙+ (Scheme 2). This indicates that XeF2/2Li[Al(ORF)4] in 5FB reaches a potential of at least 1.42 V vs. Fc+/0.32 In addition to 5FB, acetonitrile was also considered as a potential solvent. The observed stability of XeF2 in acetonitrile under the present conditions is consistent with previous literature reports, and XeF2 was found to remain stable for several days in this solvent (SI 4.2).51 While XeF2/2Li[Al(ORF)4] can oxidize Fc in acetonitrile, anthraceneHal could not be oxidized in this solvent (Scheme 2). Notably, control experiments demonstrated that Fc can already be oxidized by XeF2 alone in MeCN, suggesting that Li[Al(ORF)4] primarily acts as a metathesis reagent to afford Fc[Al(ORF)4]. These findings indicate that the enhanced reactivity of the Xe/2Li[Al(ORF)4] system is suppressed in MeCN, likely due to the increased solvation and consequently reduced reactivity of the Li+ ion in this system.Synthesis of [C6F5Xe]+-compounds and stability with aluminate WCAs
Various counterions have hitherto been used to synthesize and stabilize organoxenonium ions, including tetrafluoroborate and related fluoroborate-derived anions, and more recent work has further broadened the synthetic access to arylxenonium(II) salts.49,52–61 However, to date, no examples are reported utilizing perfluorinated alkoxyaluminate anions as WCAs. In combination with the above assignment of the [2,3,4-C6H2F3Xe]+ intermediate, this raised the question of whether organoxenonium species are compatible with aluminate anions, or if the observed decomposition of the anion is a result of such incompatibility. To address this, we aimed to synthesize salts with the well-established [C6F5Xe]+ cation and aluminate-WCAs as the counterion to directly assess their compatibility.A modified route to [C6F5Xe]+ salts
The established synthesis of the organoxenonium ion [C6F5Xe][AsF6] with the intermediate [C6F5Xe][BF2(C6F5)2] served as the starting point for our route (see Scheme 3).53 To obtain the xenonium compounds, our route uses the “ion-like” trimethylfluorosilane adduct (CH3)3SiF–Al(ORF)3 as a strong Lewis acid (FIA = 459 kJ mol−1)62,63 to abstract a fluoride ion from [BF2(C6F5)2]−. Depending on the number of equivalents of Me3SiF–Al(ORF)3 employed, we successfully synthesized [C6F5Xe][WCA] ([WCA]− = [FAl(ORF)3]− (4), [µ-F{Al(ORF)3}2]− (5), see Scheme 3). Both compounds 4 and 5 were characterized by NMR, IR and scXRD (SI 5.7, 5.8, 8.2 and 9).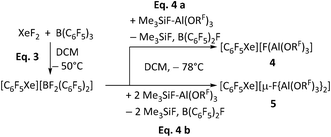 | ||
| Scheme 3 Synthesis of the compounds [C6F5Xe][WCA] ([WCA]− = [FAl(ORF)3]− (4), [µ-F{Al(ORF)3}2]− (5)) starting from the established synthesis of [C6F5Xe][BF2(C6F5)2] by Frohn et al.53 | ||
During these investigations, crystals of [C6F5Xe(OH2)][Al(ORF)4] were obtained accidentally when Li[Al(ORF)4] was used instead of Me3SiF–Al(ORF)3 (SI 8.3). While characterized only by scXRD, this species constitutes a rare structural example of a xenonium water complex (Fig. 5). The short Xe⋯O(H2O) contact of 2.624(3) Å supports coordination of water to the xenonium center. For comparison, recently characterized Xe(VI) hydrate complexes exhibit similarly short Xe⋯O(H2O) contacts, e.g. in [(18-crown-6)(H2O)XeO3]·H2O, the corresponding Xe⋯O(H2O) distance is 2.702(10) Å. However, the bonding situation in the present organoxenonium species is clearly different from that in XeO3.64
 | ||
| Fig. 5 Molecular structures of [C6F5Xe(H2O)][Al(ORF)4]. Selected bond length: Xe⋯O(H2O) = 2.624(3) Å. Thermal displacement ellipsoids are shown at the 50% probability level. | ||
Molecular structures
The molecular structure of the xenonium ion [C6F5Xe]+ shows distinct interactions depending on the counterion. With the anion [F-Al(ORF)3]−, the compound crystallizes in the orthorhombic space group Pbca, forming a contact ion pair. The shortest Xe⋯F(Al) contact is 2.595 Å, notably shorter than the 2.714 Å observed in [C6F5Xe][AsF6],53 indicating a stronger cation–anion interaction in the tetrahedral, monodentate anion [F-Al(ORF)3]−. By contrast, when the least coordinating WCA [µ-F(Al(ORF)3)2]− is employed,12 the salt [C6F5Xe][µ-F{Al(ORF)3}2] crystallizes in the monoclinic space group P21/n, and the closest Xe⋯F contact increases to 2.981 Å. This expanded distance reflects the much weaker interaction, yielding a nearly “naked” xenonium species that better represents the intrinsic properties of the isolated [C6F5Xe]+ cation. These structural differences demonstrate how the anion's coordination behavior and fluorine bridging capacity modulate cation–anion interactions.The Hirshfeld surface analysis of the scXRD structures of [C6F5Xe][F{Al(ORF)3}2] and C6F5Xe–F-Al(ORF)3 clearly demonstrates the stronger interaction of the [FAl(ORF)3]− anion with the cation (see Fig. 6c). On the Hirshfeld surface, the red regions correspond to contacts that are shorter than the sum of the van der Waals radii, indicating enhanced intermolecular interactions.
Stability of arene-xenonium ions
To revisit the literature statement that xenonium species derived from highly fluorinated arenes exhibit increased stability with rising degrees of fluorination65 and to support the stability of the observed 2,3,4-trifluorophenylxenonium ion, this trend was quantified by density functional theory (DFT) calculations (RI-r2scan-3c(D4)/def2-mTZVPP) by investigating the isodesmic transfer of a xenon atom from a phenyl xenonium ion onto a fluorinated phenyl cation: Scheme 4 shows that the reaction turns increasingly exergonic as the degree of fluorination increases.Proposed mechanism for biphenyl-formation and quantum chemical calculations
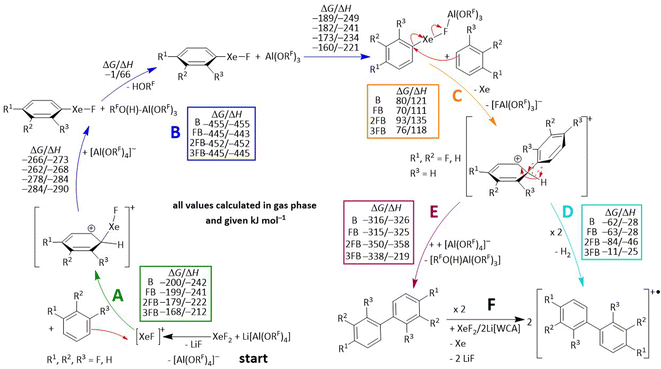
With this knowledge in mind, we now turn to the question of what mechanism leads to the observed radical cations of the coupling products [1]˙+, [2]˙+ and [3]˙+. Filler et al. proposed that the coupling of arenes induced by XeF2 in the presence of HF proceeds via a radical mechanism, involving the formation of the phenyl radical as a key intermediate, since no other intermediates were observed that could support an electrophilic mechanism.66 However, in the present XeF2/2Li[Al(ORF)4] system, we observe the concomitant formation of Li[FAl(ORF)3] and HORF, as well as in the low-temperature NMR studies the development of the 2,3,4-trifluorophenylxenonium ion. Therefore, we propose the electrophilic aromatic substitution mechanism shown in Scheme 5, based on experimental evidence that is further supported by DFT calculations in the subsequent section.Organoxenonium-ion formation
The hypothetical mechanism begins with the formation of LiF and [XeF][Al(ORF)4] in a medium that weakly solvates Li+. The xenonium species generated then acts as a strong electrophile and forms Wheland intermediates with the arenes benzene, FB, 2FB and 3FB (step A in Scheme 5). The +M-effect of the fluorine substituent accounts for the observed substitution patterns in the products. While formal elimination of HF from this Wheland intermediate could in principle yield the organoxenonium species directly, we presume instead that this intermediate reacts as a very strong Brønsted acid by protonation of one of the oxygen atoms of a second equivalent of the counterion [Al(ORF)4]− yielding the known, but weakly bound alcohol adduct RFO(H)–Al(ORF)3. The latter dissociates into the corresponding alcohol as well as the known Lewis super acid Al(ORF)3.67 The Lewis super acid then abstracts in step B a fluoride ion from the Xe-compound to probably generate a contact ion pair, related to the independently prepared [C6F5Xe–µ-F-Al(ORF)3]. Subsequently, the organoxenonium species acts as a [CH5−nFn]+ (n = 0–3) phenyl cation synthon and undergoes coupling with a second equivalent of the arene, accompanied by liberation of neutral xenon gas, to yield the coupling product in step C, again viewed as being formally a protonated (fluorinated) biphenyl [H(CH5−nFn)2]+ and thus a very strong Brønsted acid. For the final step, two mechanistic pathways are conceivable (D and E). Pathway D would directly lead to the observed radical cation coupling products via the formal elimination of a hydrogen atom (resulting in H2 evolution). Although this step is calculated to be exergonic, such a reaction is highly unusual. Notably, literature examples exist for protonated arenes, e.g. [H(mesitylene)]+,68 yet in those cases the formation of the corresponding radical cation was never reported. Instead, we assume that the deprotonation of this species, followed by rearomatization, takes place, which yields the neutral coupling products in step E. As Poleschner et al. have demonstrated, the combination of XeF2 with the Lewis acid Al(ORF)3 acts as a strongly oxidizing system.3 However, the identity of the final oxidant remains ambiguous, as both XeF2 in combination with the Lewis acid and Li[Al(ORF)4] could account for the formation of the observed radical cations of the coupling products in step G.DFT calculations
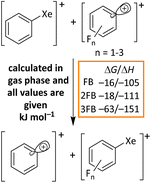
To support the proposed mechanism, we performed DFT calculations to assess the underlying thermodynamics in the gas phase, employing the cost-efficient but accurate D4 dispersion corrected r2scan-3c functional with the def2-mTZVPP basis set (Scheme 5).69 The first two reaction steps A and B are highly exergonic, which is in line with the experimental observation that the reactions even take place at −40 °C. Step C is moderately endergonic for all calculated arenes. Yet, this unfavorable thermodynamics is probably caused by the formation of two charged species from a neutral molecule. Bond heterolysis will be favored by polar solvents and should also be favorable in solution. By comparing steps D and E, the deprotonation and rearomatization are clearly thermodynamically favored, which underlines the assumption of forming the neutral coupling product followed by oxidation of another equivalent of XeF2/2Li[Al(ORF)4]. For benchmarking the protonation steps B and D, we also calculated the reaction [H(mesitylene)]+ + [Al(ORF)4]− → mesitylene + RFO(H)–Al(ORF)3, which exhibits gaseous ΔG/ΔH values of −216/–223 kJ mol−1 (RI-r2scan-3c(D4)/def2-mTZVPP). Note that [H(mesitylene)]+[Al(ORF)4]− is experimentally known to be stable in DCM solution at −20 °C, but decomposes slowly over hours at RT.67 This comparison is particularly relevant, since both reactants are charged and the products are neutral, like steps B and D in the proposed mechanism. However, both steps are considerably more exergonic than this reference reaction, further supporting the conclusion that anion protonation is also favorable in solution.Overall, experiments and calculations support the conclusion that the XeF2/2Li[Al(ORF)4] system mediates the coupling of arenes, followed by oxidation of the resulting biphenyl derivatives.
Conclusion
Through the combination of XeF2 and Li[Al(ORF)4] in polar but almost non-coordinating fluorobenzene solvents, activation of XeF2 was achieved at low temperature. With benzene (dissolved in 2FB), FB and 2FB, this led to coupling reactions forming the biphenyl derivatives 1–3 and their oxidation to the corresponding radical cations. A weakly solvated Li+ source is essential, as Li[SbF6] showed no reactivity under analogous conditions and acetonitrile blocked reactions in separate experiments. The observation of the 2,3,4-trifluorophenylxenonium ion using 3FB suggests an electrophilic mechanism, which was further explored through DFT studies. The successful synthesis of compounds 4 and 5 demonstrates the compatibility of organoxenonium species with alkoxy aluminates as anions. Stability studies of XeF2 at room temperature revealed that it remains stable for 2 h in 5FB before fluorination of 5FB occurs, whereas in acetonitrile XeF2 is stable for several days.By choosing suitable reagents, we showed that the XeF2/2Li+ system can react as an oxidant in suitable and compatible solvents. Upon reaction with anthraceneHal in 5FB, the characteristic intensely green colored solutions of [anthraceneHal]˙+ were formed, indicating a redox potential of the system of at least 1.42 V vs. Fc+/0 in 5FB. In contrast, no oxidation of anthraceneHal was observed in MeCN. Control experiments further showed that Fc can already be oxidized by XeF2 alone, whereas addition of Li[Al(ORF)4] affords [Fc][Al(ORF)4]. Thus, in acetonitrile, Li[Al(ORF)4] appears to mainly act as a metathesis reagent and yields a stable counterion for the ferrocenium cation. This led to the conclusion that the “price” for the higher stability of XeF2 in acetonitrile is the well solvated and hence less active Li+ in solution.
In summary, the XeF2/2Li[Al(ORF)4] system exhibits high reactivity even at low temperatures but is accompanied by challenges due to side reactions. In 5FB, it enables access to high potentials up to 1.42 V and potentially even higher, whereas acetonitrile offers better XeF2 stability but suppresses the enhanced reactivity of the combined XeF2/2Li+ system. Consequently, side reactions such as arene fluorination or coupling reactions and restricted solvent choice (favoring 5FB) represent key limitations. The isolated xenonium salts expand the field of xenon chemistry by demonstrating that readily available alkoxyaluminates can serve as anions. In addition, the observation of [C6F5Xe(OH2)][Al(ORF)4] provides a rare example of a xenonium water complex. The observed radical cations of fluorinated biphenyls exhibit significant potential as strong oxidants and are the subject of intense ongoing studies.
Author contributions
KK performed the majority of the syntheses and characterizations, carried out the DFT calculations and analyses, and co-wrote the manuscript together with IK. TH synthesized the [C6F5Xe][WCA] species together with MS. BL investigated the coupling products under the supervision of KK. TS synthesized the [C6F5Xe(OH2)]+ species under the supervision of KK. PT and SR performed the EPR measurements and simulations, and wrote and reviewed the corresponding section of the manuscript. IK conceived and supervised the project.Conflicts of interest
The authors declare no conflict of interest.Data availability
CCDC 2529884–2529886 and 2531335 contain the supplementary crystallographic data for this paper.70a–dExperimental details, procedures, weights, and 1D- and 2D-NMR spectra of the reactions are provided in the supplementary information (SI). Supplementary information: the general synthetic methods and characterization techniques used for this work together with the experimental procedures. Additional figures such as NMR, IR and EPR spectra are presented, as well as the crystallographic data of the isolated salts and details of the quantum chemical calculations. The authors have cited additional references within the SI. See DOI: https://doi.org/10.1039/d6sc01402j.
Acknowledgements
The authors acknowledge support by the state of Baden-Württemberg through bwHPC and the DFG through grant number INST 40/575-1 FUGG (JUSTUS 2 cluster). This work was supported by ERC Advanced Grant InnoChem. This work was supported by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – Project number 417643975 (S. Richert). S. Weber thanks the SIBW/DFG for financing magnetic resonance instrumentation that is operated within the MagRes Center of the University of Freiburg. K. Kloiber is grateful for a Manchot PhD fellowship from the “Jürgen Manchot Stiftung”.References
- M. Lehmann, A. Schulz and A. Villinger, Angew. Chem., Int. Ed., 2009, 48, 7444–7447 CrossRef CAS PubMed.
- Y. Schulte, C. Wölper, S. M. Rupf, M. Malischewski, D. J. SantaLucia, F. Neese, G. Haberhauer and S. Schulz, Nat. Chem., 2024, 16, 651–657 CrossRef CAS PubMed.
- H. Poleschner and K. Seppelt, Angew. Chem., Int. Ed., 2013, 52, 12838–12842 CrossRef CAS PubMed.
- R. Sievers, N. G. Kub, T. Streit, M. Reimann, G. Thiele, M. Kaupp and M. Malischewski, Angew. Chem., Int. Ed., 2025, 64, e202505783 CrossRef CAS.
- L. Fischer, M. H. Lee, I. Kim, A. Wiesner, K. F. Hoffmann and S. Riedel, Angew. Chem., Int. Ed., 2024, 63, e202407497 CrossRef CAS PubMed.
- R. Sievers, N. G. Kub, T. Streit, S. M. Rupf and M. Malischewski, Chem.–Eur. J., 2025, 31, e202500743 CrossRef CAS PubMed.
- A. N. Toraman, L. Fischer, A. Pérez-Bitrián, A. Wiesner, K. F. Hoffmann and S. Riedel, Chem. Commun., 2024, 60, 1711–1714 RSC.
- K. F. Hoffmann, D. Battke, P. Golz, S. M. Rupf, M. Malischewski and S. Riedel, Angew. Chem., Int. Ed., 2022, 61, e202203777 CrossRef CAS.
- T. He, M. A. Bruening, M. Espinosa and T. Agapie, Angew. Chem., Int. Ed., 2025, 64, e202417136 CrossRef CAS PubMed.
- J. Bader, L. Fischer, K. F. Hoffmann, N. Limberg, A. Millanvois, F. Oesten, A. Pérez-Bitrián, J. Schlögl, A. N. Toraman, D. Wegener, A. Wiesner and S. Riedel, Chem. Rev., 2025, 125, 9140–9186 CrossRef CAS PubMed.
- S. Kotsyuda, A. Wiesner, S. Steinhauer and S. Riedel, Z. Anorg. Allg. Chem., 2021, 647, 200–203 CrossRef CAS.
- A. Martens, P. Weis, M. C. Krummer, M. Kreuzer, A. Meierhöfer, S. C. Meier, J. Bohnenberger, H. Scherer, I. Riddlestone and I. Krossing, Chem. Sci., 2018, 9, 7058–7068 RSC.
- D. Wegener, N. Limberg, M. Bubenik, A. Pérez-Bitrián, A. Wiesner and S. Riedel, JACS Au, 2025, 5, 3565–3574 CrossRef PubMed.
- K. F. Hoffmann, A. Wiesner, N. Subat, S. Steinhauer and S. Riedel, Z. Anorg. Allg. Chem., 2018, 644, 1344–1348 CrossRef CAS.
- I. Krossing, Chem.–Eur. J., 2001, 7, 490–502 CrossRef CAS PubMed.
- J. J. Davidson, S. O. Gunther, D. W. Leong and O. V. Ozerov, Dalton Trans., 2023, 52, 16027–16031 RSC.
- F. Dankert, S. P. Muhm, C. Nandi, S. Danés, S. Mullassery, P. Herbeck-Engel, B. Morgenstern, R. Weiss, P. Salvador and D. Munz, J. Am. Chem. Soc., 2025, 147, 15369–15376 CrossRef CAS PubMed.
- AB155762|CAS 274933-96-9, https://abcr.com/de_de/ab155762, accessed December 4, 2025.
- C. Armbruster, M. Sellin, M. Seiler, T. Würz, F. Oesten, M. Schmucker, T. Sterbak, J. Fischer, V. Radtke, J. Hunger and I. Krossing, Nat. Commun., 2024, 15, 6721 CrossRef CAS PubMed.
- A. Decken, H. D. B. Jenkins, G. B. Nikiforov and J. Passmore, Dalton Trans., 2004, 2496–2504 RSC.
- J. Willrett, M. Sellin, M. Lapersonne, M. Seiler and I. Krossing, Chem.–Eur. J., 2025, 31, e202501134 CrossRef CAS PubMed.
- S. B. Beil, S. Möhle, P. Enders and S. R. Waldvogel, Chem. Commun., 2018, 54, 6128–6131 RSC.
- M. F. Ibad, A. Schulz and A. Villinger, Organometallics, 2019, 38, 1445–1458 CrossRef CAS.
- I.-C. Hwang and K. Seppelt, Inorg. Chem., 2003, 42, 7116–7122 CrossRef CAS PubMed.
- P. Gowik, T. Klapötke and U. Thewalt, J. Organomet. Chem., 1990, 385, 345–350 CrossRef CAS.
- Y. Nakajima, Y. Shiraishi, T. Tsuchimoto and F. Ozawa, Chem. Commun., 2011, 47, 6332 RSC.
- K. F. Hoffmann, A. Wiesner, C. Müller, S. Steinhauer, H. Beckers, M. Kazim, C. R. Pitts, T. Lectka and S. Riedel, Nat. Commun., 2021, 12, 5275 CrossRef CAS PubMed.
- F. S. T. Khan, A. L. Waldbusser, M. C. Carrasco, H. Pourhadi and S. Hematian, Dalton Trans., 2021, 50, 7433–7455 RSC.
- L. Fischer, M. H. Lee, A. Wiesner, C. Müller and S. Riedel, Chem. Sci., 2025, 16, 13229–13234 RSC.
- M. E. Moussa, M. Fleischmann, G. Balázs, A. V. Virovets, E. Peresypkina, P. A. Shelyganov, M. Seidl, S. Reichl and M. Scheer, Chem.–Eur. J., 2021, 27, 9742–9747 CrossRef CAS PubMed.
- J. Bohnenberger, W. Feuerstein, D. Himmel, M. Daub, F. Breher and I. Krossing, Nat. Commun., 2019, 10, 624 CrossRef CAS PubMed.
- M. Sellin, C. Friedmann, M. Mayländer, S. Richert and I. Krossing, Chem. Sci., 2022, 13, 9147–9158 RSC.
- M. Schorpp, T. Heizmann, M. Schmucker, S. Rein, S. Weber and I. Krossing, Angew. Chem., Int. Ed., 2020, 59, 9453–9459 CrossRef CAS PubMed.
- M. Sellin, J. Willrett, D. Röhner, T. Heizmann, J. Fischer, M. Seiler, C. Holzmann, T. A. Engesser, V. Radtke and I. Krossing, Angew. Chem., Int. Ed., 2024, 63, e202406742 CrossRef CAS PubMed.
- M. Tramsek and B. Zemva, Acta Chim. Slov., 2006, 53, 105–116 CAS.
- B. Zemva, A. Jesih, D. H. Templeton, A. Zalkin, A. K. Cheetham and N. Bartlett, J. Am. Chem. Soc., 1987, 109, 7420–7427 CrossRef CAS.
- H. Kim, D. Mandal, J. K. J. Ng, Z. Qu, S. Grimme and D. W. Stephan, Chem.–Eur. J., 2024, 30, e202401776 CrossRef CAS PubMed.
- S. Stavber and M. Zupan, J. Org. Chem., 1981, 46, 300–302 CrossRef CAS.
- A. Kraft, N. Trapp, D. Himmel, H. Böhrer, P. Schlüter, H. Scherer and I. Krossing, Chem.–Eur. J., 2012, 18, 9371–9380 CrossRef CAS PubMed.
- L. O. Müller, D. Himmel, J. Stauffer, G. Steinfeld, J. Slattery, G. Santiso-Quiñones, V. Brecht and I. Krossing, Angew. Chem., Int. Ed., 2008, 47, 7659–7663 CrossRef PubMed.
- V. V. Bardin, L. N. Shchegoleva and H. J. Frohn, J. Fluorine Chem., 1996, 77, 153–159 CrossRef CAS.
- L. Ai, X. Meng, S. Li, C. Zhang, D. Munz and J. Chu, Angew. Chem., Int. Ed., 2025, 64, e202515888 CrossRef CAS PubMed.
- M. Quiroz-Guzman and S. N. Brown, Acta Crystallogr., Sect. C:Cryst. Struct. Commun., 2010, 66, m171–m173 CrossRef CAS PubMed.
- N. G. Connelly and W. E. Geiger, Chem. Rev., 1996, 96, 877–910 CrossRef CAS PubMed.
- J. M. Rall, M. Schorpp, M. Keilwerth, M. Mayländer, C. Friedmann, M. Daub, S. Richert, K. Meyer and I. Krossing, Angew. Chem., Int. Ed., 2022, 61, e202204080 CrossRef CAS PubMed.
- M. Sellin, J. D. Watson, J. Fischer, G. E. Ball, L. D. Field and I. Krossing, Angew. Chem., Int. Ed., 2025, 64, e202507494 CrossRef CAS PubMed.
- P. J. Malinowski, D. Himmel and I. Krossing, Angew. Chem., Int. Ed., 2016, 55, 9262–9266 CrossRef CAS PubMed.
- CRC handbook of chemistry and physics: a ready-reference book of chemical and physical data, ed. D. R. Lide, CRC Press, CRC, Taylor & Francis, Boca Raton, Fla., 2006, p. 87, 2006–2007 Search PubMed.
- D. Naumann, H. Butler, R. Gnann and W. Tyrra, Inorg. Chem., 1993, 32, 861–863 CrossRef CAS.
- M. Zupan, J. Iskra and S. Stavber, J. Org. Chem., 1998, 63, 878–880 CrossRef CAS PubMed.
- M. A. Tius, Tetrahedron, 1995, 51, 6605–6634 CrossRef CAS.
- H.-J. Frohn, T. Schroer and G. Henkel, Angew. Chem., Int. Ed., 1999, 38, 2554–2556 CrossRef CAS PubMed.
- H.-J. Frohn, A. Klose, T. Schroer, G. Henkel, V. Buss, D. Opitz and R. Vahrenhorst, Inorg. Chem., 1998, 37, 4884–4890 CrossRef CAS PubMed.
- H. J. Frohn, S. Jakobs and G. Henkel, Angew. Chem., Int. Ed., 1989, 28, 1506–1507 CrossRef.
- K. Koppe, H.-J. Frohn, H. P. A. Mercier and G. J. Schrobilgen, Inorg. Chem., 2008, 47, 3205–3217 CrossRef CAS PubMed.
- D. Naumann, W. Tyrra, R. Gnann, D. Pfolk, T. Gilles and K. -F. Tebbe, Z. Anorg. Allg. Chem., 1997, 623, 1821–1834 CrossRef CAS.
- P. Cortés Soláns, M. L. Bubenik, M. H. Lee, P. S. Riemann and A. Pérez-Bitrián, Inorg. Chem., 2025, 64, 23184–23191 CrossRef PubMed.
- K. Koppe, V. Bilir, H.-J. Frohn, H. P. A. Mercier and G. J. Schrobilgen, Inorg. Chem., 2007, 46, 9425–9437 CrossRef CAS PubMed.
- D. Naumann, W. Tyrra, R. Gnann and D. Pfolk, J. Chem. Soc., Chem. Commun., 1994, 2651 RSC.
- D. Naumann and W. Tyrra, J. Chem. Soc., Chem. Commun., 1989, 47–50 RSC.
- H. J. Frohn and S. Jakobs, J. Chem. Soc., Chem. Commun., 1989, 625–627 RSC.
- H. Böhrer, N. Trapp, D. Himmel, M. Schleep and I. Krossing, Dalton Trans., 2015, 44, 7489–7499 RSC.
- M. Rohde, L. O. Müller, D. Himmel, H. Scherer and I. Krossing, Chem.–Eur. J., 2014, 20, 1218–1222 CrossRef CAS PubMed.
- K. M. Marczenko, H. P. A. Mercier, J. T. Goettel and G. J. Schrobilgen, Angew. Chem., Int. Ed., 2025, 64, e202425216 CrossRef CAS PubMed.
- H.-J. Frohn and V. V. Bardin, Organometallics, 2001, 20, 4750–4762 CrossRef CAS.
- R. Filler, Isr. J. Chem., 1978, 17, 71–79 CrossRef CAS.
- A. Kraft, J. Beck, G. Steinfeld, H. Scherer, D. Himmel and I. Krossing, Organometallics, 2012, 31, 7485–7491 CrossRef CAS.
- F. Scholz, D. Himmel, L. Eisele, W. Unkrig, A. Martens, P. Schlüter and I. Krossing, Chem.–Eur. J., 2015, 21, 7489–7502 CrossRef CAS PubMed.
- S. Grimme, A. Hansen, S. Ehlert and J.-M. Mewes, J. Chem. Phys., 2021, 154, 064103 CrossRef CAS PubMed.
- (a) CCDC 2529884: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxk5d; (b) CCDC 2529885: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxk6f; (c) CCDC 2529886: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qxk7g; (d) CCDC 2531335: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qz1zr.
| This journal is © The Royal Society of Chemistry 2026 |