Open Access Article
Open Access ArticleConfining ultrasmall Au nanoclusters in an ionic Ir(III)-based cage for selective photoreduction
Zhuolin Shia,
Fengyang Yu b,
Jinguo Wua,
Yongai Yu*a,
Hanshu Lia,
Xing Zhaoa,
Rong Zhanga,
Wenjing Jiangc,
Yiwei Liu
b,
Jinguo Wua,
Yongai Yu*a,
Hanshu Lia,
Xing Zhaoa,
Rong Zhanga,
Wenjing Jiangc,
Yiwei Liu a,
Jianwei Weid,
Xuezhao Li
a,
Jianwei Weid,
Xuezhao Li *a and
Cheng He
*a and
Cheng He a
a
aCentral Hospital of Dalian University of Technology, School of Chemistry, School of Chemical Engineering, Dalian University of Technology, Dalian 116024, China. E-mail: xuezhaoli@dlut.edu.cn; yuyongai@dlut.edu.cn
bCollege of Chemistry, Xinjiang University, Xinjiang 830046, China
cFujian Science & Technology Innovation Laboratory for Optoelectronic Information of China, Fuzhou, Fujian 350108, China
dHebei Technology Innovation Center for Energy Conversion Materials and Devices, College of Chemistry and Materials Science, Hebei Normal University, Shijiazhuang 050024, China
First published on 23rd April 2026
Abstract
Balancing activity and stability in metal nanoclusters (NCs) for efficient catalysis remains challenging, particularly in tuning their surrounding microenvironment to control selectivity. Here, we report ultrasmall Au nanoclusters (0.73 ± 0.14 nm) confined within a photoactive dinuclear Ir(III)-based ionic cage, synergistically coupling spatial confinement with electronic cooperativity for selective photoreduction. The ionic cage enables controlled synthesis of ultrasmall Au–NCs, ensures long-term stability (>6 months) and facilitates photoinduced electron transfer (PET) from Ir(III) photosensitizers to Au active sites. This multi-function design drives complete nitrobenzene-to-azobenzene conversion with >98% selectivity under visible light (450 nm) at room temperature, avoiding aniline byproducts. Operando spectroscopy, kinetic studies, and DFT calculations reveal that substrate-sieving at cage windows directs the stepwise reduction pathway via azoxybenzene intermediates. The demonstrated integration of photoinduced electronic and steric microenvironment control of cage-encapsulated NC-based composites establishes a promising strategy for developing nanocatalysts with exceptional selectivity steering capability.
Introduction
Metal nanoclusters (NCs)—typically less than 2 nm in size—have garnered significant attention due to their molecule-like electronic structures and high surface-area-to-volume ratios, which collectively endow them with outstanding catalytic activity.1,2 However, their inherently high surface energy renders them thermodynamically unstable, often leading to aggregation into larger particles.3 To address this, effective stabilization strategies employing organic ligands, polymers, or porous frameworks (e.g., MOFs, COFs) have been explored.4–8 Nevertheless, these approaches may hinder efficient substrate-catalyst interactions, introduce diffusion limitations, and importantly lack precise control over the microenvironment around the active sites, which limits achievable selectivity.9 Therefore, the challenge remains to design a confined yet accessible microenvironment that not only stabilizes ultrasmall NCs but also exerts precise control over substrate access to govern catalytic selectivity.10In natural systems, high catalytic activity and selectivity often arise from spatially confined microenvironments that regulate substrate access and reaction pathways, as exemplified by enzyme active-site pockets.11,12 Spatial confinement within well-defined nanocavities represents a powerful strategy for enhancing catalytic selectivity and steering reaction pathways.13–15 In this context, molecular cages (MCs) stand out as promising hosts for NCs. Their well-defined, processable cavities allow for precise spatial confinement of substrates and fine-tuning of the catalytic microenvironment.16–29 Compared to extended porous frameworks, MCs offer modular and atomically precise control over structure and functionality, which enables simultaneous protection of NCs and substrate accessibility to catalytic sites.30–34 Their capacity to organize multiple components within a confined volume mirrors the architecture of enzyme active pockets, facilitating cascade reactions and suppressing side pathways.35,36 Despite these advantages, most reported nanocatalysts based on purely organic molecular cages rely on flexible scaffolds that often lack sufficient structural rigidity and are prone to collapsing in solution.37–39 In addition, the integration of photoactive components into molecular cages to enable cooperative, light-driven catalysis with encapsulated metal NCs remains underexplored.40,41
In this study, we report an advanced catalytic platform comprising ultrasmall Au nanoclusters (Au–NCs, 0.73 ± 0.14 nm) confined within a photoactive dinuclear Ir(III)-based ionic cage. The Ir(III) cage not only directs the formation of ultrasmall Au–NCs but also serves as a built-in photosensitizer, enabling efficient PET to the surface of Au–NCs. This synergistic design enables complete photoreduction of nitrobenzene to azobenzene via azoxybenzene intermediates, achieving >98% selectivity under 450 nm light irradiation at room temperature, while effectively suppressing over-reduction to aniline. Mechanistic insights derived from kinetic analyses, operando spectroscopy, and DFT calculations comprehensively elucidate the origins of the system's high selectivity: size-exclusion effects at cage windows and the photoinduced formation of an electron-rich Au–NCs surface. Our results demonstrate the potential of integrating spatial confinement and photoactive functionality within a single cage skeleton to confine ultrasmall nanoclusters, creating advanced catalytic systems with specific selectivity.
Results and discussion
Design, synthesis, and characterization of the ionic dinuclear Ir(III)-based cage
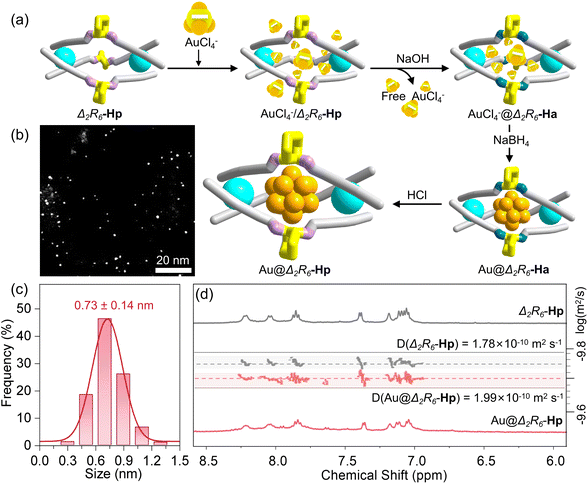
We previously reported a pair of homochiral dinuclear Ir(III)-based cages, Δ2R6-Hi and Λ2S6-Hi, constructed from three enantiopure trans-1,2-diaminocyclohexane (DACH, RR/SS-form) spacers and two fac-Ir(ppy)3 (ppy = 2-phenylpyridine) vertices in either Δ- or Λ-configurations, connected via imine linkages (Fig. S1–S3).42 Upon reduction of the imine bonds and subsequent acidification of the resulting neutral amine-linked cage, we envisioned that the resulting ionic Ir(III)-based cage could serve as an ideal host for ultrasmall NCs, owing to several distinct advantages:(1) The resulting cationic cage (+6) is highly water-soluble and stable. It can trap anionic metal precursors through electrostatic interactions while simultaneously preventing aggregation of the encapsulated NCs via coulombic repulsion—thus acting as a “cationic armor”.
(2) The rigid fac-Ir(ppy)3 vertices define a confined internal cavity suitable for size-controlled NC growth. Meanwhile, their photoactive nature enables potential synergy with encapsulated NCs in photocatalytic processes.43
(3) Crucially, the open cage windows are expected to allow for substrate diffusion and modulate interactions between the NC surface and incoming molecules, providing a spatially confined microenvironment conducive to selective transformations.
To this end, we selected the Δ2R6-Hp cage as a model system. As illustrated in Fig. 1a, its synthesis involved two steps: (i) reduction of Δ2R6-Hi with NaBH4 to obtain the amine-linked neutral cage Δ2R6-Ha, and (ii) subsequent protonation with dilute HCl to afford the final ionic cage Δ2R6-Hp. Both Δ2R6-Ha and Δ2R6-Hp were isolated in good yields and fully characterized by nuclear magnetic resonance (NMR) and high-resolution quadrupole time-of-flight mass spectrometry (HR-Q-TOF-MS) (Fig. S4–S33). In particular, the characteristic imine proton signals of Δ2R6-Hi disappeared, and new signals corresponding to NH (1.47 ppm) and methylene protons (3.18 and 3.50 ppm) appeared in the 1H NMR spectrum of Δ2R6-Ha (Fig. 1b). Moreover, diffusion-ordered NMR spectroscopy (1H DOSY) showed a single diffusion band (D = 7.94 × 10−11 m2 s−1), consistent with a single discrete species. Correspondingly, HR-Q-TOF-MS analysis of Δ2R6-Ha revealed two dominant peaks at m/z = 863.3407 ([M + 2H]2+, calc. 863.3415) and m/z = 575.8969 ([M + 3H]3+, calc. 575.8968), collectively confirming the complete conversion of imines to amines (Fig. 1d). Subsequent protonation of Δ2R6-Ha with dilute HCl (∼0.12 M, 12 h, r.t.) afforded the water-soluble ionic cage Δ2R6-Hp. In D2O, 1H NMR showed a single set of well-resolved signals, and 1H DOSY NMR displayed a consistent diffusion coefficient (D = 1.78 × 10−10 m2 s−1) (Fig. 1c), indicating the formation of a uniform species. HR-Q-TOF-MS confirmed the intact [M − 4H − 6Cl]2+ and [M − 3H − 6Cl]3+ species (Fig. S19). Furthermore, Δ2R6-Hp exhibits excellent aqueous solubility and thermal stability (Fig. S34), with no significant changes in the NMR spectra after heating at 90 °C for 48 h. Importantly, the protonation process was reversible: treatment with excess NaOH regenerated Δ2R6-Ha as a yellow precipitate, confirming that it is acid/base-switchable without compromising structural integrity.
Crystallization attempts for Δ2R6-Hp were unsuccessful, thus density functional theory (DFT) calculations were employed to model its structural properties. Notably, in contrast to many purely organic amine-linked cages that collapse after imine reduction due to backbone flexibility, the rigid Ir(III) modules in Δ2R6-Hp preserved the integrity of the cavity architecture.42 As illustrated in Fig. 1e, three RR-DACH spacers bridge two fac-Ir(ppy)3 vertices, both adopting the same Δ-configuration. The protonated –NH2+- groups introduce electrostatic repulsion between them, which expands the transverse dimension of the cavity, resulting in a more open and near-spherical geometry compared to the imine-linked Δ2R6-Hi (Fig. 1f).42 Correspondingly, the calculated average N⋯N distance increased to approximately 8.8 Å (from 8.1 Å), while the Ir⋯Ir distance decreased to 12.7 Å (from 13.3 Å). These robust and tunable structural features, combined with the enlarged internal cavity of Δ2R6-Hp, render it a promising platform for the encapsulation of ultrasmall Au–NCs.
The cationic skeleton of Δ2R6-Hp endows exceptional stability to the encapsulated Au–NCs in aqueous media via electrostatic repulsion, maintaining dispersion integrity without aggregation for over six months, as evidenced by consistent UV-vis spectra and HAADF-STEM imaging (Fig. S51). Importantly, employing Λ2S6-Hp as an alternative host also produced ultrasmall Au–NCs with comparable dimensions (Fig. S53), demonstrating the general applicability of these metallohelical cages as templates for nanocluster synthesis. In contrast, control experiments using Δ2R6-Ha, which lacks electrostatic trapping and stabilization abilities, yielded significantly larger nanoparticles (Au/Δ2R6-Ha, average size: 2.36 ± 0.61 nm, Fig. S54), indicating that the particles formed outside the cage cavity and were merely surrounded by cages. The PXRD pattern of Au/Δ2R6-Ha showed multiple diffraction peaks in the 30–70° range, consistent with the presence of crystalline Au nanoparticles (Fig. S55).33 These results highlight the critical role of electrostatic interactions in achieving both ultrasmall cluster size and long-term stability.
| Entry | Variation from the “standard conditions” | Conversion/% | Seletivity/% | ||
|---|---|---|---|---|---|
| AOB | AB | AN | |||
| a Reaction conditions: Au@Δ2R6-Hp (1 mol%), 0.1 mmol of NB, 2 mL of IPA, 0.3 M of NaOH, room temperature, Ar atmosphere, 450 nm LED, reaction time of 24 h. | |||||
| 1 | None | 100 | — | 98 | 2 |
| 2 | Au@Λ2S6-Hp | 100 | — | 98 | 2 |
| 3 | No catalyst | 5.5 | 5.5 | — | — |
| 4 | No light | 0 | — | — | — |
| 5 | Δ2R6-Hp | 33 | 33 | — | — |
| 6 | AuCl4−/Δ2R6-Hp | 20 | 88 | 12 | — |
| 7 | Au/Δ2R6-Ha | 100 | 57 | 29 | 14 |
| 8 | EtOH replaces IPA | 0 | — | — | — |
| 9 | None (48 h) | 100 | — | 98 | 2 |
| 10 | None (72 h) | 100 | — | 95 | 5 |
| 11 | None (after five cycles) | 100 | 17 | 83 | — |
The general applicability of Au@Δ2R6-Hp was evaluated in the selective photocatalytic coupling of various nitroarenes bearing diverse functional groups (Table 2). Halogenated nitroarenes, including F-, Cl-, and Br-substituted derivatives, were successfully reduced to their corresponding azobenzene products without any observable dehalogenation, affording high selectivity (3b–3f). Furthermore, substrates with para-substituents, whether electron-deficient (3g–3i) or electron-donating (3j and 3k), also underwent smooth conversion to azobenzenes with similarly high selectivity. These results underscore the robustness and functional group tolerance of the Au@Δ2R6-Hp system.
| a Reaction conditions: Au@Δ2R6-Hp (1 mol%), 0.1 mmol of substrate 1, 2 mL of IPA, 0.3 M of NaOH, room temperature, Ar atmosphere, 450 nm LED, reaction time of 12 h, 24 h or 36 h. C for conversion, and S for selectivity. |
|---|
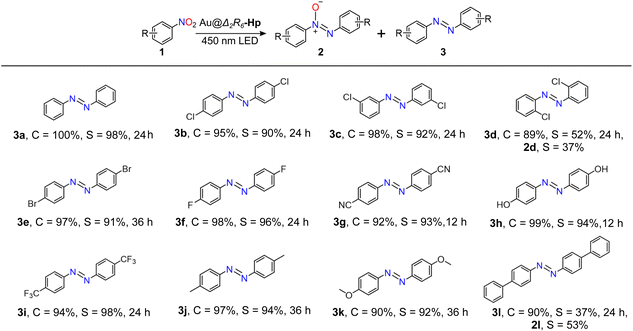 |
The catalytic performance was further examined with substrates featuring different degrees of steric hindrance. For Cl-substituted nitroarenes, a general selectivity trend of para ≈ meta > ortho was observed (3b–3d). In particular, the ortho-Cl substituted substrate 1d exhibited a moderate conversion of 89%, but afforded the desired azobenzene product 3d in only 52% selectivity, alongside 37% selectivity for the intermediate 2d. This reduced selectivity likely arises from steric interference near the active site, which impedes the further transformation of 2d into 3d. Notably, the catalytic efficiency decreased markedly when bulkier substrates such as biphenyl nitroarenes were employed. For example, substrate 1l gave significantly lower selectivity toward the final azobenzene product 3l (only 37%), while the intermediate 2l dominated with 53% selectivity. This diminished performance is attributed to the increased steric repulsion introduced by the biphenyl group, which likely restricts substrate accommodation within the narrow windows of the cage cavity. Such steric effects highlight the spatial sensitivity of the cage-confined catalytic environment.
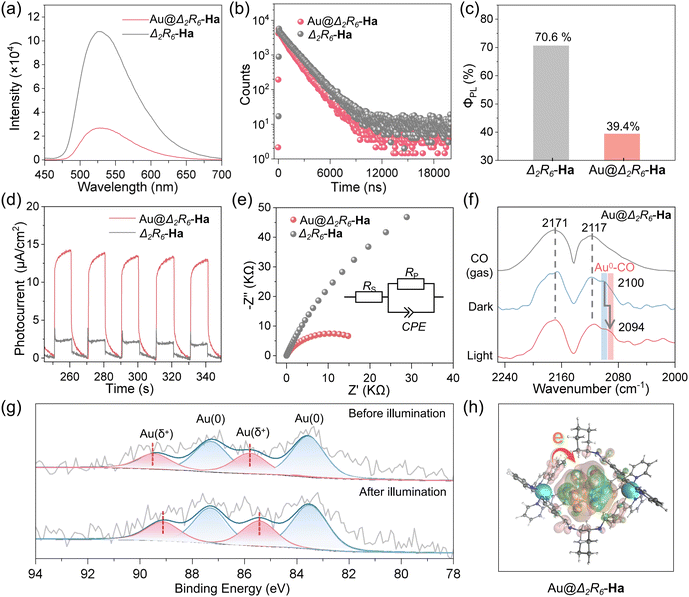
To further probe the surface electronic environment of Au–NCs, diffuse reflectance infrared Fourier transform spectroscopy (DRIFTS) was performed using CO as a probe molecule. As shown in Fig. 3f, two vibrational bands at 2171 cm−1 and 2117 cm−1 were observed after CO adsorption, characteristic of physisorbed CO on Au surfaces.53 After argon purging, a new band at 2100 cm−1 emerged, attributed to CO binding on neutral Au0 sites.54 Upon in situ 450 nm LED irradiation, this band red-shifted by 6 cm−1 to 2094 cm−1, indicating increased electron density on Au due to light-induced electron transfer. Given that CO adsorption frequencies inversely correlate with metal electron density, this shift suggests photoexcitation leads to electron accumulation on the Au surface.55 In agreement, X-ray photoelectron spectroscopy (XPS) analysis revealed a 0.5 eV shift in the binding energies of Auδ+ peaks after visible light irradiation (from 89.6/85.9 eV to 89.1/85.4 eV; Fig. 3g). This shift further corroborates light-driven electron enrichment at Au–NCs.1 As shown in Fig. 3h, electron density distribution (EDD) analysis showed that photoexcitation causes significant electron redistribution: electron density accumulates around the encapsulated Au–NCs (green regions) while depleting from the cage skeleton (pink regions).36 This spatial charge separation confirms the directionality of PET from the Ir-based cage to the Au–NCs. These experimental results collectively demonstrate that the cage's ability to donate electrons to the encapsulated Au–NCs under light irradiation enables the creation of electron-enriched catalytic centers for efficient substrate activation.
To probe the molecular basis of this selective reduction, we hypothesized that substrate binding affinities, modulated by the metallocage's sterically defined windows, determine access to the Au–NCs catalytic surface. Luminescence titration studies revealed a clear affinity hierarchy: NB exhibited the strongest binding with a high association constant (KNB = 3.24 × 106 M−1) and achieved saturation at 10 nM (Fig. 4c). In contrast, AOB and AB required 10-fold higher concentrations to reach saturation, correlating with their significantly weaker binding constants (KAOB = 3.36 × 105 M−1; KAB = 3.13 × 105 M−1) (Fig. 4d and e). Structural analysis of the cage suggests that its two bent DACH ligands and parallel ppy walls create a confined pocket with rhombic portal-like entrances, which can sieve substrates by steric exclusion. This structure explains why planar but bulky substrates like AB and AOB are likely to reside at the outer edge of the pocket due to steric hindrance, whereas compact NB can penetrate deeper for more efficient interactions. This steric exclusion mechanism was further corroborated by luminescence quenching assays using NB derivatives with increased bulk. For instance, ortho-chlorinated NB (NB-ortho-2Cl, 1d) and biphenyl-type NB (NB-2Phen, 1l) exhibited drastically reduced binding affinities (Ka ≈ 104 M−1), two orders of magnitude lower than NB (Fig. 4f and S68a), which is consistent with their lower reduction reactivity. Consequently, the Stern–Volmer plots (Fig. S68b and c) confirmed that the interaction with NB was one to two orders of magnitude stronger than that with the other compounds (AOB, AB, NB-ortho-2Cl and NB-2Phen), demonstrating unequivocally that increased steric bulk hinders effective binding.
To validate the spatial confinement mechanism, we employed DRIFTS to monitor substrate adsorption dynamics. NB showed strong N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O vibrational bands at ∼1348 and ∼1525 cm−1, with intensities increasing continuously over 15 minutes (Fig. 5a), confirming strong adsorption via its nitro group, while the benzene ring remained non-interacting.36 In contrast, AOB exhibited only weak peaks for C
O vibrational bands at ∼1348 and ∼1525 cm−1, with intensities increasing continuously over 15 minutes (Fig. 5a), confirming strong adsorption via its nitro group, while the benzene ring remained non-interacting.36 In contrast, AOB exhibited only weak peaks for C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, N–O, and N
C, N–O, and N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bonds (Fig. 5b), likely due to steric repulsion at the cage windows.56 AB showed even weaker adsorption, with minimal spectral changes (Fig. 5c), and faster equilibrium was reached for both AOB and AB within 3 minutes, compared to over 15 minutes for NB (Fig. 5d). Desorption studies further revealed stronger retention of NB (10 min to reach equilibrium) versus AOB/AB (5 min), reinforcing the idea of preferential NB binding and rapid AB release (Fig. 5e and S69). To match the cage cavity dimensions and the experimentally observed cluster size (∼0.73 nm), DFT calculations were performed using an icosahedral Au13 cluster (Fig. S71 and S72). As shown in Fig. 5g, NBs preferentially adsorb via their nitro groups in a vertical orientation within the pocket, achieving the strongest adsorption energy (−2.23 eV, Fig. 5f), while the bulkier AOB and AB adopt parallel orientations and exhibit significantly weaker adsorption energies (−1.85 eV, −1.76 eV). These findings confirm that the rhombic windows enable substrate sieving by favoring NB adsorption and AB desorption. Further DFT analysis revealed that NB reduction to AB proceeds with favorable stepwise energetics (Fig. S73), while the over-reduction of AB to 2AN faces a high activation barrier (3.62 eV), rendering it thermodynamically disfavored. Consequently, the spatially confined Au@Δ2R6-Ha system not only modulates adsorption and catalytic stages but also ensures product selectivity by blocking excessive hydrogenation.
N bonds (Fig. 5b), likely due to steric repulsion at the cage windows.56 AB showed even weaker adsorption, with minimal spectral changes (Fig. 5c), and faster equilibrium was reached for both AOB and AB within 3 minutes, compared to over 15 minutes for NB (Fig. 5d). Desorption studies further revealed stronger retention of NB (10 min to reach equilibrium) versus AOB/AB (5 min), reinforcing the idea of preferential NB binding and rapid AB release (Fig. 5e and S69). To match the cage cavity dimensions and the experimentally observed cluster size (∼0.73 nm), DFT calculations were performed using an icosahedral Au13 cluster (Fig. S71 and S72). As shown in Fig. 5g, NBs preferentially adsorb via their nitro groups in a vertical orientation within the pocket, achieving the strongest adsorption energy (−2.23 eV, Fig. 5f), while the bulkier AOB and AB adopt parallel orientations and exhibit significantly weaker adsorption energies (−1.85 eV, −1.76 eV). These findings confirm that the rhombic windows enable substrate sieving by favoring NB adsorption and AB desorption. Further DFT analysis revealed that NB reduction to AB proceeds with favorable stepwise energetics (Fig. S73), while the over-reduction of AB to 2AN faces a high activation barrier (3.62 eV), rendering it thermodynamically disfavored. Consequently, the spatially confined Au@Δ2R6-Ha system not only modulates adsorption and catalytic stages but also ensures product selectivity by blocking excessive hydrogenation.
Conclusions
In summary, by confining ultrasmall Au nanoclusters within a photoactive Ir(III)-based ionic cage, the complete nitrobenzene-to-azobenzene conversion with >98% selectivity was achieved under 450 nm LED irradiation at room temperature via an interesting stepwise pathway involving azoxybenzene intermediates. The system operates through a synergistic dual-regulation mechanism by integrating the light-harvesting ability of the Ir(III) metallocage with the catalytic function of the Au nanoclusters: (1) PET from the excited cage to the embedded Au nanoclusters generates electron-rich active sites, facilitating substrate activation; (2) the size-selective windows of the cage provide substrate-sieving functionality, which governs reactant adsorption and ensures high chemoselectivity. This work exemplifies how the fusion of photocatalytic activity and molecular recognition within a single supramolecular cage-nanocluster platform can yield catalysts with tailored activity and selectivity. Furthermore, extending this design concept to other redox-active metal clusters and functional cages presents a promising strategy for the simultaneous stabilization, microenvironment engineering, and catalytic selectivity tuning of nanoclusters.Author contributions
Zhuolin Shi performed molecular synthesis and experiments, analyzed the data, conducted the computational studies, and wrote the original draft. Fengyang Yu contributed to spherical aberration-corrected HAADF-STEM measurements and data analysis. Jinguo Wu assisted with molecular synthesis. Wenjing Jiang conducted XPS measurements. Rong Zhang performed HR-Q-TOF-MS measurements. Jianwei Wei assisted with kinetic analysis. Yongai Yu, Fengyang Yu, Hanshu Li, Xing Zhao, Yiwei Liu, and Cheng He contributed to scientific discussions and manuscript review and editing. Xuezhao Li conceived and designed the project, supervised the study, contributed to data analysis, acquired funding, contributed to the original draft, and revised the manuscript.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data generated in this study are provided in the article, supplementary information (SI) and from corresponding author(s) upon request. Supplementary information: synthesis, NMR and MS characterizations, photophysical measurements, photocatalytic experiments, and DFT. See DOI: https://doi.org/10.1039/d6sc01364c.Acknowledgements
This research work was financially supported by the National Natural Science Foundation of China (grant nos. 22477014, 22171033 and 22571032), the Fundamental Research Funds for the Central Universities [DUT25Z2504], and the Liaoning Provincial Science and Technology Plan Joint Program (Natural Science Foundation-General Program, 2024-MSLH-070). We thank Dr Cai Rui in Instrumental Analysis Center of Dalian University of Technology for assistance with photoluminescence spectroscopy.Notes and references
- J. Z. Zhang, Y. B. Zhang, H. L. Chai, H. L. Luo, C. X. Du, R. W. Huang and S. Q. Zang, Selectivity Modulation of Multistep Reduction Reactions by Gold Nanoclusters, Angew. Chem., Int. Ed., 2025, 64, e202413418 CrossRef CAS PubMed.
- X. Ma, Z. Zhang, D. Zhao, J. Peng, S. Xing, R. Huang, S. Li, N. Ma and Y. Liu, Spatially Confined Construction of Ultrasmall Pd Clusters Within Nitro-Bonded Covalent Organic Frameworks for Efficient Alkyne Semihydrogenation, Small, 2025, 21, 2410416 CrossRef CAS PubMed.
- H. Kang, J. T. Buchman, R. S. Rodriguez, H. L. Ring, J. He, K. C. Bantz and C. L. Haynes, Stabilization of Silver and Gold Nanoparticles: Preservation and Improvement of Plasmonic Functionalities, Chem. Rev., 2019, 119, 664–699 CrossRef CAS PubMed.
- E. L. Albright, T. I. Levchenko, V. K. Kulkarni, A. I. Sullivan, J. F. DeJesus, S. Malola, S. Takano, M. Nambo, K. Stamplecoskie, H. Häkkinen, T. Tsukuda and C. M. Crudden, N-Heterocyclic Carbene-Stabilized Atomically Precise Metal Nanoclusters, J. Am. Chem. Soc., 2024, 146, 5759–5780 CrossRef CAS PubMed.
- K. Ueda, R. Saito, K. Iseri, S. Sekiya, M. Nakamura and K. Isozaki, Accelerated Catalysis of Atomically Precise Thiolate-Protected Gold Nanoclusters by Supramolecular Ligand Engineering, ACS Catal., 2025, 15, 12260–12268 CrossRef CAS.
- Y. Zhong, P. Liao, P. Jiang, Y. Zhang, J. Kang, S. Xie, R. Feng, Y. Fan, Q. Liu and G. Li, Ionic-Fence Effect in Au Nanoparticle-Loaded UiO-66 Metal–Organic Frameworks for Highly Chemoselective Hydrogenation, Angew. Chem., Int. Ed., 2025, 64, e202501821 CrossRef CAS PubMed.
- C. He, Q. J. Wu, M. J. Mao, Y. H. Zou, B. T. Liu, Y. B. Huang and R. Cao, Multifunctional Gold Nanoparticles @Imidazolium-Based Cationic Covalent Triazine Frameworks for Efficient Tandem Reactions, CCS Chem., 2020, 3, 2368–2380 CrossRef.
- R. W. Y. Man, C. H. Li, M. W. A. MacLean, O. V. Zenkina, M. T. Zamora, L. N. Saunders, A. Rousina-Webb, M. Nambo and C. M. Crudden, Ultrastable Gold Nanoparticles Modified by Bidentate N-Heterocyclic Carbene Ligands, J. Am. Chem. Soc., 2018, 140, 1576–1579 CrossRef CAS PubMed.
- L. M. Cao, J. Zhang, X. F. Zhang and C. T. He, Confinement Synthesis in Porous Molecule-based Materials: A New Opportunity for Ultrafine Nanostructures, Chem. Sci., 2022, 13, 1569–1593 RSC.
- Q. Fu and X. Bao, Confined Microenvironment for Catalysis Control, Nat. Catal., 2019, 2, 834–836 CrossRef CAS.
- E. L. Bell, W. Finnigan, S. P. France, A. P. Green, M. A. Hayes, L. J. Hepworth, S. L. Lovelock, H. Niikura, S. Osuna, E. Romero, K. S. Ryan, N. J. Turner and S. L. Flitsch, Biocatalysis, Nat. Rev. Methods Primers, 2021, 1, 46 CrossRef CAS.
- L. Lancaster, W. Abdallah, S. Banta and I. Wheeldon, Engineering Enzyme Microenvironments for Enhanced Biocatalysis, Chem. Soc. Rev., 2018, 47, 5177–5186 RSC.
- H. Takezawa and M. Fujita, Molecular Confinement Effects by Self-Assembled Coordination Cages, Bull. Chem. Soc. Jpn., 2021, 94, 2351–2369 CrossRef CAS.
- J. Meeuwissen and J. N. H. Reek, Supramolecular catalysis beyond enzyme mimics, Nat. Chem., 2010, 2, 615–621 CrossRef CAS PubMed.
- Y. L. Lu, Y. P. Wang, K. Wu, M. Pan and C. Y. Su, Activating Metal–Organic Cages by Incorporating Functional M(ImPhen)3 Metalloligands: From Structural Design to Applications, Acc. Chem. Res., 2024, 57, 3277–3291 CrossRef CAS PubMed.
- R. McCaffrey, H. Long, Y. Jin, A. Sanders, W. Park and W. Zhang, Template Synthesis of Gold Nanoparticles with an Organic Molecular Cage, J. Am. Chem. Soc., 2014, 136, 1782–1785 CrossRef CAS PubMed.
- Y. Zhang, J. Zhou, K. Luo, W. Zhou, F. Wang, J. Li and Q. He, Ferritin-Inspired Encapsulation and Stabilization of Gold Nanoclusters for High-Performance Photothermal Conversion, Angew. Chem., Int. Ed., 2025, 64, e202500058 CrossRef CAS PubMed.
- X. X. Gou, T. Liu, Y. Y. Wang and Y. F. Han, Ultrastable and Highly Catalytically Active N-Heterocyclic-Carbene-Stabilized Gold Nanoparticles in Confined Spaces, Angew. Chem., Int. Ed., 2020, 59, 16683–16689 CrossRef CAS PubMed.
- M. Nihei, H. Ida, T. Nibe, A. M. P. Moeljadi, Q. T. Trinh, H. Hirao, M. Ishizaki, M. Kurihara, T. Shiga and H. Oshio, Ferrihydrite Particle Encapsulated within a Molecular Organic Cage, J. Am. Chem. Soc., 2018, 140, 17753–17759 CrossRef CAS PubMed.
- Y. Fang, J. Li, T. Togo, F. Jin, Z. Xiao, L. Liu, H. Drake, X. Lian and H. C. Zhou, Ultra-Small Face-Centered-Cubic Ru Nanoparticles Confined within a Porous Coordination Cage for Dehydrogenation, Chem, 2018, 4, 555–563 CAS.
- S. Y. Zhang, Z. Kochovski, H.-C. Lee, Y. Lu, H. Zhang, J. Zhang, J. K. Sun and J. Yuan, Ionic Organic Cage-encapsulating Phase-transferable Metal Clusters, Chem. Sci., 2019, 10, 1450–1456 RSC.
- C. Wang, F. Sun, G. He, H. Zhao, L. Tian, Y. Cheng and G. Li, Noble Metal Nanoparticles Meet Molecular Cages: A Tale of Integration and Synergy, Curr. Opin. Colloid Interface Sci., 2023, 63, 101660 CrossRef CAS.
- S. L. Hou, J. Dong, Z. H. Zhu, L. C. Geng, Y. Ma and B. Zhao, Size-Tunable Ultrafine Pt Nanoparticles in Soluble Metal–Organic Cages: Displaying Highly Stereoselective Hydrogenation of α-Pinene, Chem. Mater., 2020, 32, 7063–7069 CrossRef CAS.
- Y. Fang, J. Li, T. Togo, F. Jin, Z. Xiao, L. Liu, H. Drake, X. Lian and H. C. Zhou, Ultra-Small Face-Centered-Cubic Ru Nanoparticles Confined within a Porous Coordination Cage for Dehydrogenation, Chem, 2018, 4, 555–563 CAS.
- L. L. Metz, R. Ham, E. O. Bobylev, K. J. H. Brouwer, A. van Blaaderen, R. C. J. van de Poll, V. R. Drozhzhin, E. J. M. Hensen and J. N. H. Reek, M12L24 Nanospheres as Supramolecular Templates for the Controlled Synthesis of Ir-Nanoclusters and Their Use in the Chemo-selective Hydrogenation of Nitro Styrene, Chem. Sci., 2024, 15, 20022–20029 RSC.
- L. L. Metz, E. O. Bobylev, R. C. J. vandePoll, E. J. M. Hensen, I. Hoogsteder, W. Albrecht and J. N. H. Reek, Supramolecular Preorganization Rhodium and Iridium Metal Complexes Within M12L24 Self-Assembled Nanospheres for the Confined Synthesis Rh/Ir Alloyed Nanoparticles, ChemCatChem, 2025, 17, e202401161 CrossRef CAS.
- X. Hang, S. Wang, H. Pang and Q. Xu, A Coordination Cage Hosting Ultrafine and Highly Catalytically Active Gold Nanoparticles, Chem. Sci., 2022, 13, 461–468 RSC.
- Y. J. Du, J. H. Zhou, L. X. Tan, S. H. Liu, K. Zhao, Z. M. Gao and J. K. Sun, Porous Organic Cage Nanostructures for Construction of Complex Sequential Reaction Networks, ACS Appl. Nano Mater., 2022, 5, 7974–7982 CrossRef CAS.
- Y. Fang, Z. Xiao, J. Li, C. Lollar, L. Liu, X. Lian, S. Yuan, S. Banerjee, P. Zhang and H. C. Zhou, Formation of a Highly Reactive Cobalt Nanocluster Crystal within a Highly Negatively Charged Porous Coordination Cage, Angew. Chem., Int. Ed., 2018, 57, 5283–5287 CrossRef CAS PubMed.
- X. Yang, J. K. Sun, M. Kitta, H. Pang and Q. Xu, Encapsulating Highly Catalytically Active Metal Nanoclusters Inside Porous Organic Cages, Nat. Catal., 2018, 1, 214–220 CrossRef CAS.
- J. K. Sun, W. W. Zhan, T. Akita and Q. Xu, Toward Homogenization of Heterogeneous Metal Nanoparticle Catalysts with Enhanced Catalytic Performance: Soluble Porous Organic Cage as a Stabilizer and Homogenizer, J. Am. Chem. Soc., 2015, 137, 7063–7066 CrossRef CAS PubMed.
- M. Zhang, R. Liu, Z. He, L. Wang, X. Zhang and G. Li, Large Triazine Organic Cage Encapsulating Pd Nanoclusters for Selective Hydrogenation of Alkynes, ACS Catal., 2025, 15, 12247–12259 CrossRef CAS.
- B. Mondal and P. S. Mukherjee, Cage Encapsulated Gold Nanoparticles as Heterogeneous Photocatalyst for Facile and Selective Reduction of Nitroarenes to Azo Compounds, J. Am. Chem. Soc., 2018, 140, 12592–12601 CrossRef CAS PubMed.
- B. Mondal, K. Acharyya, P. Howlader and P. S. Mukherjee, Molecular Cage Impregnated Palladium Nanoparticles: Efficient, Additive-Free Heterogeneous Catalysts for Cyanation of Aryl Halides, J. Am. Chem. Soc., 2016, 138, 1709–1716 CrossRef CAS PubMed.
- M. Raynal, P. Ballester, A. Vidal-Ferran and P. W. N. M. van Leeuwen, Supramolecular Catalysis. Part 2: Artificial Enzyme Mimics, Chem. Soc. Rev., 2014, 43, 1734–1787 RSC.
- S. Ren, L. Tan, J. H. Zhou, J. Sun, P. Zhang, X. Cao, Y. H. Zhang and J. K. Sun, Eco-Friendly Encapsulation of Metal Clusters in Porous Organic Cages for Engineerable Microenvironment and Enhanced Catalysis, CCS Chem., 2024, 6, 2186–2197 CrossRef CAS.
- X. Zhou, Z. Wang, Z. N. Chen and Y. Yang, Phosphine-Built-In Porous Organic Cage Supported Ultrafine Pd Nanoclusters Enable Highly Efficient and Regioselective Hydrogenation of Epoxides, CCS Chem., 2024, 6, 2476–2488 CrossRef CAS.
- K. E. Jelfs, X. Wu, M. Schmidtmann, J. T. A. Jones, J. E. Warren, D. J. Adams and A. I. Cooper, Large Self-Assembled Chiral Organic Cages: Synthesis, Structure, and Shape Persistence, Angew. Chem., Int. Ed., 2011, 50, 10653–10656 CrossRef CAS PubMed.
- M. Liu, M. A. Little, K. E. Jelfs, J. T. A. Jones, M. Schmidtmann, S. Y. Chong, T. Hasell and A. I. Cooper, Acid- and Base-Stable Porous Organic Cages: Shape Persistence and pH Stability via Post-synthetic “Tying” of a Flexible Amine Cage, J. Am. Chem. Soc., 2014, 136, 7583–7586 CrossRef CAS PubMed.
- M. Wilms, L. V. Melendez, R. J. Hudson, C. R. Hall, S. P. Ratnayake, T. Smith, E. D. Gaspera, G. Bryant, T. U. Connell and D. E. Gómez, Photoinitiated Energy Transfer in Porous-Cage-Stabilised Silver Nanoparticles, Angew. Chem., Int. Ed., 2023, 62, e202303501 CrossRef CAS PubMed.
- N. Sun, C. Wang, H. Wang, L. Yang, P. Jin, W. Zhang and J. Jiang, Multifunctional Tubular Organic Cage-Supported Ultrafine Palladium Nanoparticles for Sequential Catalysis, Angew. Chem., Int. Ed., 2019, 58, 18011–18016 CrossRef CAS PubMed.
- J. Wu, X. Li, P. Wu, Z. Shi, L. Chen, R. Zhang, C. He and C. Duan, Engineering Homochiral Dinuclear Ir(III)-Metallohelix-Based Porous Molecular Crystals for Atropisomer Enantioseparation, Chem. Mater., 2022, 34, 4471–4478 CrossRef CAS.
- Z. Shi, Y. Wang, R. Zhang, H. Li, R. Cai, J. Wu, X. Wang, H. Li, X. Li and C. He, Confining Cu(I) ions within an Ir(III)-based Twin-cavity Cage for Photo-triggered Dioxygen Activation toward C(sp3)–H Oxidation, Inorg. Chem. Front., 2025, 12, 2515–2523 RSC.
- J. Chen, Y. L. Lu, Y. Huang, F. Zhang, H. Ye, Y.-H. Huang, X.-D. Zhang, Z. Jiao and C. Y. Su, Asymmetric Cascade Photocycloaddition-Acyloin Rearrangement Enabled by Cage-Confined Visible-Light Catalysis, J. Am. Chem. Soc., 2025, 147, 13008–13016 CrossRef CAS PubMed.
- S. Zhou, W. Cao, L. Shang, Y. Zhao, X. Xiong, J. Sun, T. Zhang and J. Yuan, Facilitating Alkaline Hydrogen Evolution Kinetics via Interfacial Modulation of Hydrogen-bond Networks by Porous Amine Cages, Nat. Commun., 2025, 16, 1849 CrossRef CAS PubMed.
- K. Esumi, A. Suzuki, N. Aihara, K. Usui and K. Torigoe, Preparation of Gold Colloids with UV Irradiation Using Dendrimers as Stabilizer, Langmuir, 1998, 14, 3157–3159 CrossRef CAS.
- E. Merino, Synthesis of Azobenzenes: the Coloured Pieces of Molecular Materials, Chem. Soc. Rev., 2011, 40, 3835–3853 RSC.
- K. Hüll, J. Morstein and D. Trauner, In Vivo Photopharmacology, Chem. Rev., 2018, 118, 10710–10747 CrossRef PubMed.
- A. Corma, P. Concepción and P. Serna, A Different Reaction Pathway for the Reduction of Aromatic Nitro Compounds on Gold Catalysts, Angew. Chem., Int. Ed., 2007, 46, 7266–7269 CrossRef CAS PubMed.
- H. Zhu, X. Ke, X. Yang, S. Sarina and H. Liu, Reduction of Nitroaromatic Compounds on Supported Gold Nanoparticles by Visible and Ultraviolet Light, Angew. Chem., Int. Ed., 2010, 49, 9657–9661 CrossRef CAS PubMed.
- Y. Dai, C. Li, Y. Shen, T. Lim, J. Xu, Y. Li, H. Niemantsverdriet, F. Besenbacher, N. Lock and R. Su, Light-tuned Selective Photosynthesis of Azo- and Azoxy-aromatics Using Graphitic C3N4, Nat. Commun., 2018, 9, 60–67 CrossRef PubMed.
- A. B. Tamayo, B. D. Alleyne, P. I. Djurovich, S. Lamansky, I. Tsyba, N. N. Ho, R. Bau and M. E. Thompson, Synthesis and Characterization of Facial and Meridional Tris-cyclometalated Iridium(III) Complexes, J. Am. Chem. Soc., 2003, 125, 7377–7387 CrossRef CAS PubMed.
- B. Wei, X. Liu, Y. Deng, K. Hua, J. Chen, H. Wang and Y. Sun, Efficient and Stable Co/β-Mo2C Catalyst for Hydroformylation, ACS Catal., 2021, 11, 14319–14327 CrossRef CAS.
- J. Luo, Y. Liu, Y. Niu, Q. Jiang, R. Huang, B. Zhang and D. Su, Insight into the Chemical Adsorption Properties of CO Molecules Supported on Au or Cu and Hybridized Au–CuO Nanoparticles, Nanoscale, 2017, 9, 15033–15043 RSC.
- H. Wang, X. Liu, W. Yang, G. Mao, Z. Meng, Z. Wu and H.-L. Jiang, Surface-Clean Au25 Nanoclusters in Modulated Microenvironment Enabled by Metal–Organic Frameworks for Enhanced Catalysis, J. Am. Chem. Soc., 2022, 144, 22008–22017 CrossRef CAS PubMed.
- G. Richner, J. A. van Bokhoven, Y. M. Neuhold, M. Makosch, K. Hungerbühler and K. In, Situ Infrared Monitoring of the Solid/liquid Catalyst Interface During the Three-phase Hydrogenation of Nitrobenzene Over Nanosized Au on TiO2, Phys. Chem. Chem. Phys., 2011, 13, 12463–12471 RSC.
| This journal is © The Royal Society of Chemistry 2026 |