Open Access Article
Open Access ArticleA unified strategy for remote C–H fluorination of phenylacetic acids and their homologues at the meta-position
Kun Zhou ,
Xin Chen,
Yue Wang,
Yang-Jie Mao
,
Xin Chen,
Yue Wang,
Yang-Jie Mao *,
Xin Yang,
Kai Liu,
Qi Pan,
Dong-Liang Pan,
Dan-Qian Xu
*,
Xin Yang,
Kai Liu,
Qi Pan,
Dong-Liang Pan,
Dan-Qian Xu * and
Shao-Jie Lou
* and
Shao-Jie Lou *
*
Catalytic Hydrogenation Research Center, State Key Laboratory of Green Chemical Synthesis and Conversion, Key Laboratory of Green Pesticides and Cleaner Production Technology of Zhejiang Province, Zhejiang University of Technology, Hangzhou 310014, P. R. China. E-mail: chrc@zjut.edu.cn; loushaojie@zjut.edu.cn; maoyangjie@zjut.edu.cn
First published on 24th April 2026
Abstract
The site-selective C–H bond fluorination of substituted aromatics at the electronically and geometrically disfavored meta-position is of great interest and importance in medical chemistry and organic chemistry, yet has remained unexplored to date. By employing a newly developed U-shaped template, we herein report for the first time the Pd-catalysed meta-selective C–H bond fluorination of phenylacetic acids and their homologues. The reaction features high site-selectivity and is compatible with diverse homologues of phenylacetic acids, constituting a unified strategy for the remote aromatic C–H bond fluorination of carboxylic acids.
Introduction
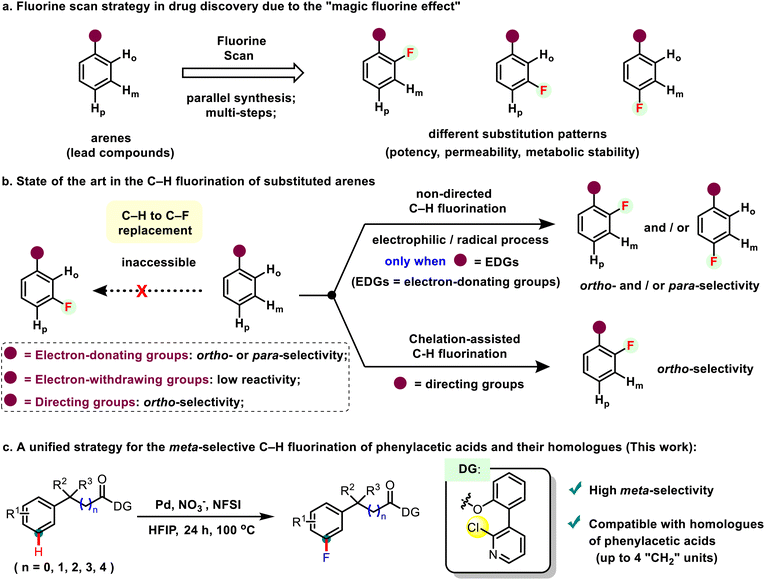
Aryl fluoride compounds are among the fastest-growing classes of organic molecules, playing a vital role in pharmaceuticals, agrochemicals, and functional materials due to the magic fluorine effect.1,2 Notably, the substitution patterns of the fluorine atom on aromatics could significantly influence the physical properties (e.g. permeability, metabolic stability, and potency) of these compounds. To this end, the fluorine scan of a lead compound, which involves the systematic replacement of hydrogen with fluorine, has emerged as a powerful and common strategy for drug discovery (Scheme 1a).3 Despite the success, given the challenging C–F bond formation, parallel de novo synthetic routes were often required to install a fluorine atom onto a specific position of the non-fluorinated lead molecules, which would significantly attenuate the efficiency of fluorine scan in drug discovery.4In this context, the direct and selective replacement of the aromatic C–H bond with the C–F bond, known as C–H fluorination, has emerged as an attractive alternative pathway to assemble fluorine-containing molecules.5 Many groups,6 including our group,7 have developed a variety of strategies for the selective and late-stage C–H fluorination of aromatics. Generally, the non-directed electrophilic or radical C–H fluorination of aromatics is only suitable with those possessing electron-donating groups, and therefore the dominant ortho- and/or para-selective fluorination are often observed due to the intrinsic electronic effect.8 To better control the ortho-/para-selectivity, chelation assisted C–H fluorination has been developed and the geometrically favoured ortho-selectivity could be achieved (Scheme 1b). Despite the recent advances, the selective C–H fluorination at the meta-position of substituted aromatics has remained unexplored due to significant electronic and geometric challenges.
Phenylacetic acids are ubiquitous in many bioactive molecules. More interestingly, changing the chain length of a phenylacetic acid lead compound towards its homologues might significantly modulate the bioactivity.9 Given the importance of meta-fluorinated phenylacetic acid derivatives in pharmaceuticals, a unified meta-selective C–H fluorination of phenylacetic acids and their homologues is of great interest and importance both in organic chemistry and medical chemistry, yet has remained unknown to date. It has been previously reported by the groups of Yu, Maiti, Li, and many others that well-designed U-shaped templates could facilitate the remote C–H functionalization (e.g. arylation, alkenylation, halogenation, and hydroxylation) of arenes.10 However, the analogous meta-selective remote C–H bond fluorination of aromatics has yet not been reported, probably due to the challenging C–F bond formation. Herein, in light of these literature studies and our previous findings on the selective C–H fluorination, we report for the first time the Pd-catalysed C–H fluorination of phenylacetic acid derivatives at the remote meta-position by using a pyridine-containing directing group. In addition, diverse homologues of phenylacetic acid (up to 4 “CH2” units) could also be compatible in this protocol, constituting a unified strategy for the remote aromatic C–H fluorination of carboxylic acids.
Results and discussion
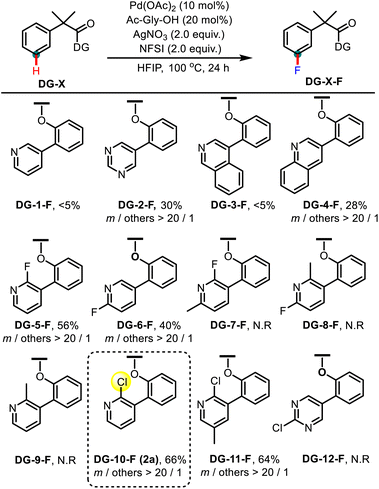
Inspired by the previously reported U-shaped templates for the remote C–H bond functionalization,11 we prepared diverse N-heterocycle-based biaryl directing groups (DGs) to test the remote C–H fluorination of phenylacetic acids (Table 1). On the basis of our previous nitrate promoted C–H fluorination and the general reaction conditions for remote C–H functionalization,7b phenylacetic acid derivatives with different DGs were treated with Pd(OAc)2, AgNO3, amino acid ligand, and N-fluorobenzenesulfonimide (NFSI) using hexafluoro-2-propanol (HFIP) as the solvent. In our preliminary attempts, the influence of coordinating N-heterocycles was investigated. It was found that the non-substituted 3-pyridyl directing group was inefficient in this reaction (DG-1-F). Changing the pyridyl moiety to the 4-pyrimidyl group could provide the desired fluorinated product in a promising 30% yield with excellent meta-selectivity (DG-2-F). 4-Isoquinolinyl (DG-3-F) and 3-quinolinyl (DG-4-F) groups were also tested and a comparable result was obtained in the case of DG-3-F. These results indicated that the substitution pattern of N-heterocycles could significantly influence the coordinating ability of the nitrogen centre and therefore manipulate the reactivity. With this consideration in mind, more directing groups possessing different substituted N-heterocycles were prepared. Indeed, the directing group bearing a 2-fluoro-3-pyridyl moiety could dramatically improve the yield of the fluorination product to 56% with high meta-selectivity (DG-5-F). Switching the fluorine atom to the C6-position diminished the yield (DG-6-F). Introducing a methyl group at the C2-position of the pyridine moiety would shut down the reaction, probably due to increased steric repulsion (DG-7-F, DG-8-F, and DG-9-F). Interestingly, replacing the C2-fluoro group with the C2-chloro group could further improve the reaction yield (DG-10-F, 66%).12 Installing an extra methyl group at the para-position of the chloro substituent did not improve the yield (DG-11-F) and the chloro-substituted 4-pyrimidyl group was inefficient in this reaction (DG-12-F).| a Reaction conditions: DG-X (0.2 mmol), Pd(OAc)2 (10 mol%), Ac-gly-OH (20 mol%), NFSI (2.0 equiv.), AgNO3 (2.0 equiv.), HFIP (2.0 mL), 24 h, and 100 °C; yield was determined by GC using dodecane as an internal standard. |
|---|
 |
Having established the optimised directing group, we proceeded to further screen the reaction conditions using DG-10 (1a) as the substrate. Firstly, Pd(OAc)2 still outperformed other Pd catalysts, such as PdCl2 and Pd(PPh3)4 (Table 2, entries 1–4). The absence of an amino acid ligand did not significantly influence the reaction outcome even with a slightly increased yield (entry 5). However, the removal of silver nitrate resulted in a sharp decrease in the yield of 2a, which is consistent with our previous finding in the selective C–H fluorination (entry 6). Replacing AgNO3 with AgSbF6 gave a slightly lower yield, while other nitrates, e.g. KNO3, were much less effective in this reaction (entries 7 and 8). It was speculated that the SbF6− ion might also play a similar role to nitrate anions to generate the cationic Pd species, which would promote the C–H activation and C–F bond reductive elimination. Adding more AgNO3 would further enhance the production of 2a (entry 10, 77%). Either lowering or elevating the reaction temperature diminished the yield (entries 11 and 12). Among the different solvents tested in this work (see Table S2 in the SI for details), HFIP still outcompeted others, e.g. trifluoroacetic acid, CH3NO2, and 1,2-dichloroethane. This is in agreement with the previously reported unique solvent effect of HFIP in the palladium-catalysed C–H functionalization reactions, particularly those for remote aromatic C–H functionalization.11n,13 Finally, a better yield of 86% was obtained when the reaction was run for 48 h (entry 13, 72% isolated yield). The re-evaluation of different U-shaped directing groups under the optimised ligand-free conditions was also conducted and DG-10-F still outperformed others (see Fig. S3 in the SI for details). The highly meta-selective C–H fluorination of the phenylacetic acid derivative is remarkable, constituting the first successful example of remote C–H fluorination of aromatics.
| Entry | Catalyst (10 mol%) | Additive (2.0 or 3.0 equiv.) | Ligand (0 or 20%) | Yield of 2a (%) |
|---|---|---|---|---|
| a Reaction conditions unless otherwise noted: 1a (0.2 mmol), Pd(OAc)2 (10 mol%), NFSI (2.0 equiv.), AgNO3 (2.0 or 3.0 equiv.), HFIP (2.0 mL), 24 h, and 100 °C. Yield was determined by GC using dodecane as an internal standard, n.r. = no reaction.b 80 °C.c 120 °C.d 48 h.e Isolated yield. | ||||
| 1 | Pd(OAc)2 | AgNO3 (2.0 equiv.) | Ac-gly-OH | 66 |
| 2 | PdCl2 | AgNO3 (2.0 equiv.) | Ac-gly-OH | 45 |
| 3 | Pd(PPh3)4 | AgNO3 (2.0 equiv.) | Ac-gly-OH | 65 |
| 4 | — | AgNO3 (2.0 equiv.) | Ac-gly-OH | n.r. |
| 5 | Pd(OAc)2 | AgNO3 (2.0 equiv.) | — | 70 |
| 6 | Pd(OAc)2 | — | — | 29 |
| 7 | Pd(OAc)2 | AgSbF6 (2.0 equiv.) | — | 65 |
| 8 | Pd(OAc)2 | KNO3 (2.0 equiv.) | — | 31 |
| 9 | Pd(OAc)2 | AgNO3 (1.0 equiv.) | — | 55 |
| 10 | Pd(OAc)2 | AgNO3 (3.0 equiv.) | — | 77 |
| 11b | Pd(OAc)2 | AgNO3 (3.0 equiv.) | — | 55 |
| 12c | Pd(OAc)2 | AgNO3 (3.0 equiv.) | — | 65 |
| 13d | Pd(OAc)2 | AgNO3(3.0 equiv.) | — | 86 (72)e |
Having optimized the reaction conditions, we proceeded to evaluate the substrate scope of this protocol. As summarized in Table 3, various phenylacetic acids bearing different α-substituents were examined. The substrates featuring a gem-dimethyl group at the α-position (2a–2d) showed enhanced reactivity compared to those without this substitution, probably due to the Thorpe–Ingold effect.14 Besides, α,α-disubstituted cyclic alkyl moieties, e.g. strained cyclopropyl (2e, 2f, and 2g), unstrained cyclopentyl (2h), and cyclohexyl (2i) groups, could also survive the reaction conditions. Moreover, the solid state structure of the meta-fluorinated product 2h was unambiguously determined by single crystal X-ray diffraction analysis. We further explored other α-substituted phenylacetic acid derivatives. As expected, the reaction of compounds that possessed a single methyl (2j) or ethyl group (2k) at the α-position also afforded the desired products in good yields. In addition, the more flexible α-free phenylacetic acid derivatives were also tested and comparable yields were obtained (2l–2s).
| a Reaction conditions: 1a (0.2 mmol), Pd(OAc)2 (10 mol%), NFSI (3.0 equiv.), AgNO3 (3.0 equiv.), HFIP (2.0 mL), 48 h, and 100 °C; isolated yield. |
|---|
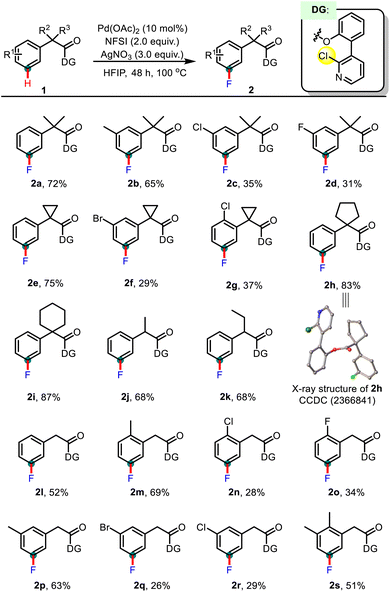 |
Notably, the type and position of substituents on the aromatic ring could significantly influence the reactivity. In general, the substrates bearing electron-donating groups were more reactive than those with electron-withdrawing groups, suggesting that the electrophilic C–H palladation might be involved in the C–H cleavage step. Substituents tethered at the ortho or meta positions of the directing group were well tolerated, while the sterically demanding para-substituted substrates failed to give the desired meta-fluorinated products (see page S37 in the SI for details).
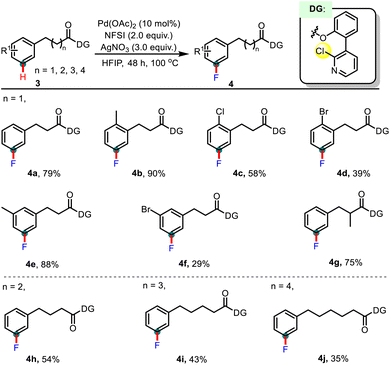
Given the importance of phenylacetic acid homologues in pharmaceuticals, a general and unified strategy that enables the remote C–H fluorination of these compounds is of great interest in the fields of both organic chemistry and drug discovery. With this consideration in mind, we next attempted to extend the study to other phenyl substituted aliphatic acid derivatives with longer carbon chain linkers. As summarized in Table 4, the reaction tolerated substrates with various chain lengths (n up to 4). Phenylpropionic acids reacted successfully to afford the desired meta-fluorinated products (4a–4g). For instance, electron-donating groups such as alkyl substituents (4b and 4e) proceeded smoothly. Halogen groups (4c, 4d, and 4f) and an α-methyl substituent (4g) were also compatible with this transformation. Additionally, carboxylic acids with even longer-chains, e.g. phenylbutyric acid (4h), phenylvaleric acid (4i), and phenylhexanoic acid (4j), proved to be viable substrates as well. The results aligned with our expectations, consistently demonstrating good meta-selectivity and acceptable yields. Notably, a gradual decrease in yield was observed with further prolonged chain length. We presume that the enhanced flexibility of the molecular skeleton in longer-chain acids reduces the effective interaction probability between the directing group and the aromatic ring, thereby compromising reaction efficiency.15
| a Reaction conditions: 3 (0.2 mmol), Pd(OAc)2 (10 mol%), NFSI (3.0 equiv.), AgNO3 (3.0 equiv.), HFIP (2.0 mL), 48 h, and 100 °C; isolated yield. |
|---|
 |
To gain mechanistic insight into this remote C–H fluorination process, kinetic isotope effect (KIE) experiments were conducted. The intramolecular competition experiment between the deuterated substrate 1l-d1 and 1l afforded a primary KIE value of 4.0. Moreover, the KIE measured from two parallel reactions that used 1l-d5 and 1l respectively was 2.0. These results indicate that C–H bond cleavage is probably involved in the rate-determining steps (Scheme 2a).16 To further demonstrate the synthetic practicality of this meta-C–H fluorination protocol, a gram-scale reaction of phenylacetate was performed, delivering the desired product 2h without a significant loss in yield (81%, Scheme 2b(I)). Under mild conditions, the pyridine-based directing group could readily be removed, furnishing both fluorinated phenylacetic acid 5a and the biaryl directing auxiliary (1a–10) in good yields. Owing to the transformation ability of the carboxylic acid moiety, the obtained 5a could easily be transformed into other compounds, such as the reductive product alcohol 5b and cyclization products 5c and 5d. Next, a gram-scale fluorination of phenylpropionic acid also proceeded smoothly to give 4a in 78% yield (81%, Scheme 2b(II)). Subsequent removal of the directing group provided phenylpropionic acid 5e along with the corresponding biphenyl auxiliary. The utility of this fluorinated product was further demonstrated by the following transformations: the hydrogenation of 5e yielded 3-fluorophenylpropanol (5f), a versatile synthon that was efficiently converted to other derivatives, e.g. halide (5g and 5h), amine (5i), and ether (5j) products.
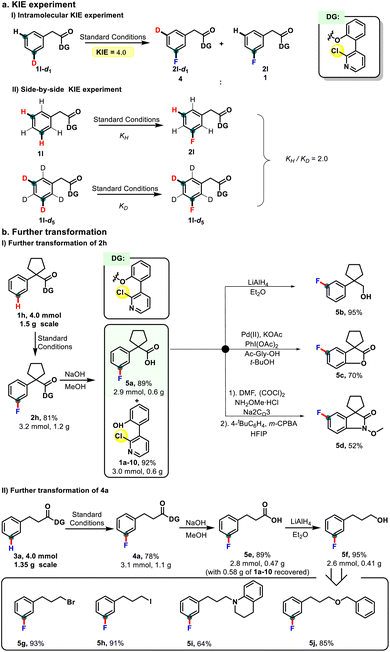 | ||
| Scheme 2 (a) KIE experiment and (b) further transformation: (I) further transformation of 2h; (II) further transformation of 3a. | ||
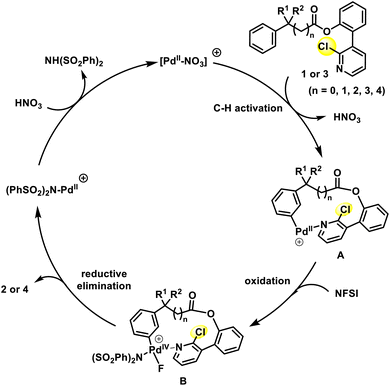
Based on our previous reports and other literature studies, a possible mechanism for the meta-selective aromatic C–H fluorination is proposed and depicted in Scheme 3.7a–e,17 The catalytic cycle is initiated by the in situ generation of a highly electrophilic cationic [Pd(NO3)]+ species, which interacts and coordinates with substrate 1 or 3. Owing to the unique steric hindrance and positioning effect of the U-shaped long-chain directing group, the coordinated [PdII] species selectively activates the meta-C–H bond on the benzene ring through electrophilic palladation, affording a macro-palladacycle intermediate A. Subsequently, the divalent palladium species A is oxidized by NFSI to form the tetravalent palladium species B. Reductive elimination from B affords the fluorinated product 2 or 4, concurrently reducing the palladium center to form the [(PhSO2)2N-PdII]+ species. Finally, anion exchange of this resulting [PdII] intermediate with HNO3 regenerates the reactive palladium nitrate species and closes the catalytic cycle. However, an alternative mechanism involving the possible oxidation of substrate-binding PdII species to PdIV prior to the C–H bond palladation could not be excluded. In this case, the C–H activation at the PdIV center would take place.17a
Conclusions
In summary, through the design of a novel directing group, we have achieved for the first time the meta-selective aromatic C–H fluorination of phenylacetic acid derivatives. Notably, this protocol was also feasible for the analogous C–H fluorination of other homologues of phenylacetic acid, constituting a unified strategy for the precision H/F exchange at the remote position of the phenyl substituted carboxylic acids. This work not only opens a new avenue for aromatic C–H fluorination but also enriches the reaction types for remote aromatic C–H functionalization.Author contributions
S.-J. Lou and D.-Q. Xu conceived and directed the project. K. Zhou, X. Chen, Y. Wang, X. Yang, K. Liu, Q. Pan and D.-L. Pan performed the experiments and analysed the data. S.-J. Lou, Y.-J. Mao and K. Zhou wrote the manuscript with input from all the authors. All authors have read and approved the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting the findings of this study are available as follows.CCDC 2366841 (2h) contains the supplementary crystallographic data for this paper.18
Supplementary information: additional experimental data, including characterisation data and NMR spectra for new compounds. See DOI: https://doi.org/10.1039/d6sc01331g.
Acknowledgements
This work was supported in part by the National Natural Science Foundation of China (No. 22301275), the Central Government Guided Local Science and Technology Development Fund Project (No. 2025ZY01113), the “Jianbing” and “Lingyan” Research and Development Program of Zhejiang province (2023C02059), the Beidou Scientific Research Project of Zhejiang University of Technology, Zhejiang Key Laboratory of Green Pesticides and Cleaner Production Technology, Zhejiang Provincial Natural Science Foundation of China under Grant No. LMS25B020004, and the Research Foundation for Advanced Talents from ZJUT (No. 2021101016929).Notes and references
- (a) G. Balz and G. Schiemann, Ber. Dtsch. Chem. Ges. (A and B), 1927, 60, 1186–1190 CrossRef; (b) D. J. Adams and J. H. Clark, Chem. Soc. Rev., 1999, 28, 225–231 RSC; (c) M. E. Phelps, Proc. Natl. Acad. Sci. U. S. A., 2000, 97, 9226–9233 CrossRef CAS PubMed; (d) K. Müller, C. Faeh and F. Diederich, Science, 2007, 317, 1881–1886 CrossRef PubMed; (e) D. O'Hagan, Chem. Soc. Rev., 2008, 37, 308–319 RSC; (f) M. Döbele, S. Vanderheiden, N. Jung and S. Bräse, Angew. Chem., Int. Ed., 2010, 49, 5986–5988 CrossRef PubMed; (g) T. Furuya, A. S. Kamlet and T. Ritter, Nature, 2011, 473, 470–477 CrossRef CAS PubMed; (h) C. Hollingworth and V. Gouverneur, Chem. Commun., 2012, 48, 2929–2942 RSC; (i) T. Liang, C. N. Neumann and T. Ritter, Angew. Chem., Int. Ed., 2013, 52, 8214–8264 CrossRef CAS PubMed; (j) J. Wang, M. Sánchez-Roselló, J. L. Aceña, C. del Pozo, A. E. Sorochinsky, S. Fustero, V. A. Soloshonok and H. Liu, Chem. Rev., 2014, 114, 2432–2506 CrossRef CAS PubMed; (k) P. A. Champagne, J. Desroches, J.-D. Hamel, M. Vandamme and J.-F. Paquin, Chem. Rev., 2015, 115, 9073–9174 CrossRef CAS PubMed; (l) D. E. Yerien, S. Bonesi and A. Postigo, Org. Biomol. Chem., 2016, 14, 8398–8427 RSC; (m) C. Thiehoff, Y. P. Rey and R. Gilmour, Isr. J. Chem., 2017, 57, 92–100 CrossRef CAS; (n) S. Caron, Org. Process Res. Dev., 2020, 24, 470–480 CrossRef CAS; (o) F.-G. Zhang, X.-Q. Wang, Y. Zhou, H.-S. Shi, Z. Feng, J.-A. Ma and I. Marek, Chem. – Eur. J., 2020, 26, 15378–15396 CrossRef CAS PubMed; (p) J.-C. Agopian, O. Téraube, K. Charlet and M. Dubois, J. Fluorine Chem., 2021, 251, 109887 CrossRef CAS; (q) S. K. Padamata, A. Yasinskiy, S. Stopic and B. Friedrich, J. Fluorine Chem., 2022, 255–256, 109964 CrossRef CAS; (r) Y. Du, Y. Bian, D. Baecker, G. Dhawan, A. Semghouli, L. Kiss, W. Zhang, A. E. Sorochinsky, V. A. Soloshonok and J. Han, Chem. – Eur. J., 2025, 31, e202500662 CrossRef CAS PubMed; (s) V. Kumar, Meenu, M. Shaquiquzzaman, M. M. Alam, M. Akhter, D. Parveen, M. Kaleem, S. Charan, M. K. Saifullah and S. Tasneem, J. Fluorine Chem., 2026, 290, 110527 CrossRef CAS.
- (a) E. Berrino, B. Michelet, A. Martin-Mingot, F. Carta, C. T. Supuran and S. Thibaudeau, Angew. Chem., Int. Ed., 2021, 60, 23068–23082 CrossRef CAS PubMed; (b) C. Guo, M. P. Fritz, J. Struppe, S. Wegner, J. Stringer, I. V. Sergeyev, C. M. Quinn, A. M. Gronenborn and T. Polenova, Anal. Chem., 2021, 93, 8210–8218 CrossRef CAS PubMed; (c) G. Chandra, D. V. Singh, G. K. Mahato and S. Patel, Chem. Pap., 2023, 77, 4085–4106 CrossRef CAS PubMed; (d) P. Kumar, N. Goel and S. Bhagat, ChemistrySelect, 2024, 9, e202401371 CrossRef CAS.
- (a) I. Hyohdoh, N. Furuichi, T. Aoki, Y. Itezono, H. Shirai, S. Ozawa, F. Watanabe, M. Matsushita, M. Sakaitani, P.-S. Ho, K. Takanashi, N. Harada, Y. Tomii, K. Yoshinari, K. Ori, M. Tabo, Y. Aoki, N. Shimma and H. Iikura, ACS Med. Chem. Lett., 2013, 4, 1059–1063 CrossRef CAS PubMed; (b) Z. Feng, Q. Li, S. Li, Y. Zhang, X. Liu, C. Zhang, A. Dong, D. Kong, W. Wang and P. Huang, Adv. Healthcare Mater., 2023, 12, 2201894 CrossRef CAS PubMed; (c) Y. Wang, X. Yang, Y. Meng, Z. Wen, R. Han, X. Hu, B. Sun, F. Kang, B. Li, D. Zhou, C. Wang and G. Wang, Chem. Rev., 2024, 124, 3494–3589 CrossRef CAS PubMed; (d) D. Datta, B. Selvakumar, A. D. Goel, S. Chhibber and T. Gupta, Eur. J. Nucl. Med. Mol. Imaging, 2025, 52, 3846–3858 CrossRef PubMed; (e) Z. Luo, H. Qiu, X. Peng, Q. Tan, B. Chen, Q. Gu, H. Liu and H. Zhou, Eur. J. Med. Chem., 2025, 291, 117647 CrossRef CAS PubMed.
- (a) T. Furuya and T. Ritter, J. Am. Chem. Soc., 2008, 130, 10060–10061 CrossRef CAS PubMed; (b) N. D. Ball and M. S. Sanford, J. Am. Chem. Soc., 2009, 131, 3796–3797 CrossRef CAS PubMed; (c) T. Furuya, D. Benitez, E. Tkatchouk, A. E. Strom, P. Tang, W. A. Goddard, III and T. Ritter, J. Am. Chem. Soc., 2010, 132, 3793–3807 CrossRef CAS PubMed; (d) M. G. Campbell and T. Ritter, Org. Process Res. Dev., 2014, 18, 474–480 CrossRef CAS PubMed; (e) M. G. Campbell and T. Ritter, Chem. Rev., 2015, 115, 612–633 CrossRef CAS PubMed; (f) M. Khandelwal, G. Pemawat and R. Kanwar Khangarot, Asian J. Org. Chem., 2022, 11, e202200325 CrossRef CAS; (g) S. Liu, J. Zhou, L. Yu, Y. Liu, Y. Huang, Y. Ouyang, G.-K. Liu, X.-H. Xu and N. Shibata, Chem. Rev., 2025, 125, 4603–4764 CrossRef CAS PubMed; (h) M. Wang, J. Ruskin, J. Marques, N. Garrison and T. Lectka, Chem. Rev., 2025, 125, 9382–9428 CrossRef CAS PubMed; (i) B. Zhao, J. Liu and J.-P. Wan, Chem. Commun., 2025, 61, 13852–13863 RSC.
- (a) Y. Li, Y. Wu, G.-S. Li and X.-S. Wang, Adv. Synth. Catal., 2014, 356, 1412–1418 CrossRef CAS; (b) A. Lin, C. B. Huehls and J. Yang, Org. Chem. Front., 2014, 1, 434–438 RSC; (c) D. A. Petrone, J. Ye and M. Lautens, Chem. Rev., 2016, 116, 8003–8104 CrossRef CAS PubMed; (d) Q. Cheng and T. Ritter, Trends Chem., 2019, 1, 461–470 CrossRef CAS; (e) R. Szpera, D. F. J. Moseley, L. B. Smith, A. J. Sterling and V. Gouverneur, Angew. Chem., Int. Ed., 2019, 58, 14824–14848 CrossRef CAS PubMed.
- (a) K. L. Hull, W. Q. Anani and M. S. Sanford, J. Am. Chem. Soc., 2006, 128, 7134–7135 CrossRef CAS PubMed; (b) X. Wang, T.-S. Mei and J.-Q. Yu, J. Am. Chem. Soc., 2009, 131, 7520–7521 CrossRef CAS PubMed; (c) K. S. L. Chan, M. Wasa, X. Wang and J.-Q. Yu, Angew. Chem., Int. Ed., 2011, 50, 9081–9084 CrossRef CAS PubMed; (d) X.-G. Zhang, H.-X. Dai, M. Wasa and J.-Q. Yu, J. Am. Chem. Soc., 2012, 134, 11948–11951 CrossRef CAS PubMed; (e) T. Truong, K. Klimovica and O. Daugulis, J. Am. Chem. Soc., 2013, 135, 9342–9345 CrossRef CAS PubMed; (f) C. Testa, J. Roger, S. Scheib, P. Fleurat-Lessard and J.-C. Hierso, Adv. Synth. Catal., 2015, 357, 2913–2923 CrossRef CAS; (g) D. A. Gutierrez, W.-C. C. Lee, Y. Shen and J. J. Li, Tetrahedron Lett., 2016, 57, 5372–5376 CrossRef CAS; (h) C. Testa, É. Gigot, S. Genc, R. Decréau, J. Roger and J.-C. Hierso, Angew. Chem., Int. Ed., 2016, 55, 5555–5559 CrossRef CAS PubMed; (i) X.-Y. Chen and E. J. Sorensen, J. Am. Chem. Soc., 2018, 140, 2789–2792 CrossRef CAS PubMed; (j) K. Yamamoto, J. Li, J. A. O. Garber, J. D. Rolfes, G. B. Boursalian, J. C. Borghs, C. Genicot, J. Jacq, M. van Gastel, F. Neese and T. Ritter, Nature, 2018, 554, 511–514 CrossRef CAS PubMed; (k) So Jeong Lee, K. J. Makaravage, A. F. Brooks, P. J. H. Scott and M. S. Sanford, Angew. Chem., Int. Ed., 2019, 131, 3151–3154 CrossRef; (l) J. Tang, F. Lu, Y. Sun, G. Zhang, E. Zhang and Y.-Y. Jiang, J. Org. Chem., 2023, 88, 14165–14171 CrossRef CAS PubMed.
- (a) S.-J. Lou, D.-Q. Xu, A.-B. Xia, Y.-F. Wang, Y.-K. Liu, X.-H. Du and Z.-Y. Xu, Chem. Commun., 2013, 49, 6218–6220 RSC; (b) S.-J. Lou, D.-Q. Xu and Z.-Y. Xu, Angew. Chem., Int. Ed., 2014, 53, 10330–10335 CrossRef CAS PubMed; (c) S.-J. Lou, Q. Chen, Y.-F. Wang, D.-Q. Xu, X.-H. Du, J.-Q. He, Y.-J. Mao and Z.-Y. Xu, ACS Catal., 2015, 5, 2846–2849 CrossRef CAS; (d) X.-Q. Ning, S.-J. Lou, Y.-J. Mao, Z.-Y. Xu and D.-Q. Xu, Org. Lett., 2018, 20, 2445–2448 CrossRef CAS PubMed; (e) M. Yang-Jie, S.-J. Lou, H. Hong-Yan and D.-Q. Xu, Angew. Chem., Int. Ed., 2018, 130, 14281–14285 CrossRef; (f) Y.-J. Mao, G. Luo, H.-Y. Hao, Z.-Y. Xu, S.-J. Lou and D.-Q. Xu, Chem. Commun., 2019, 55, 14458–14461 RSC; (g) Q. Wu, Y.-J. Mao, K. Zhou, S. Wang, L. Chen, Z.-Y. Xu, S.-J. Lou and D.-Q. Xu, Chem. Commun., 2021, 57, 4544–4547 RSC; (h) Q.-Z. Wu, Y.-J. Mao, K. Zhou, H.-Y. Hao, L. Chen, S. Wang, Z.-Y. Xu, S.-J. Lou and D.-Q. Xu, Chem. Commun., 2021, 57, 765–768 RSC.
- (a) T. Tian, W.-H. Zhong, S. Meng, X.-B. Meng and Z.-J. Li, J. Org. Chem., 2013, 78, 728–732 CrossRef CAS PubMed; (b) D. Liang, Y. Li, S. Gao, R. Li, X. Li, B. Wang and H. Yang, Green Chem., 2017, 19, 3344–3349 RSC; (c) M. Betourney, H. Ashton, G. Costello, J. Hunter, S. H. Park, B. Shepler, J. C. Sloop, M. Stancea and K. Wilmott, J. Fluorine Chem., 2025, 285–286, 110455 CrossRef CAS.
- (a) S. Sakurai, N. Ogawa, T. Suzuki, K.-i. Kato, T. Ohashi, S. Yasuda, H. Kato and Y. Ito, Chem. Pharm. Bull., 1996, 44, 765–777 CrossRef CAS PubMed; (b) M. L. Lídia, A. A. Marina and N. d. A. Daniel, Curr. Top. Med. Chem., 2019, 19, 1734–1750 CrossRef PubMed; (c) J. Jurczyk, J. Woo, S. F. Kim, B. D. Dherange, R. Sarpong and M. D. Levin, Nat. Synth., 2022, 1, 352–364 CrossRef CAS PubMed; (d) R. Zhang, T. Yu and G. Dong, Science, 2023, 382, 951–957 CrossRef CAS PubMed.
- (a) M. Zhang, Y. Zhang, X. Jie, H. Zhao, G. Li and W. Su, Org. Chem. Front., 2014, 1, 843–895 RSC; (b) J. Yang, Org. Biomol. Chem., 2015, 13, 1930–1941 RSC; (c) A. Dey, S. Agasti and D. Maiti, Org. Biomol. Chem., 2016, 14, 5440–5453 RSC; (d) Y.-F. Yang, X. Hong, J.-Q. Yu and K. N. Houk, Acc. Chem. Res., 2017, 50, 2853–2860 CrossRef CAS PubMed; (e) U. Dutta and D. Maiti, Acc. Chem. Res., 2022, 55, 354–372 CrossRef CAS PubMed; (f) N. Y. S. Lam, Z. Fan, K. Wu, H. S. Park, S. Y. Shim, D. A. Strassfeld and J.-Q. Yu, J. Am. Chem. Soc., 2022, 144, 2793–2803 CrossRef CAS PubMed; (g) S. K. Sinha, S. Guin, S. Maiti, J. P. Biswas, S. Porey and D. Maiti, Chem. Rev., 2022, 122, 5682–5841 CrossRef CAS PubMed; (h) A. T. Sebastian, S. Maji, M. Rajashekhar, S. Maiti, R. Kowalczyk and D. Maiti, Angew. Chem., Int. Ed., 2024, 63, e202410806 CrossRef CAS PubMed; (i) R. S. Chauhan, Y. Bairagi, O. Desai, R. Kowalczyk and D. Maiti, Chem. Commun., 2025, 61, 4293–4315 RSC; (j) W.-B. Li, X.-C. Huang, P. Liu, J. Kong and G.-P. Yang, Chin. Chem. Lett., 2025, 36, 110543 CrossRef CAS.
- (a) D. Leow, G. Li, T.-S. Mei and J.-Q. Yu, Nature, 2012, 486, 518–522 CrossRef CAS PubMed; (b) S. Lee, H. Lee and K. L. Tan, J. Am. Chem. Soc., 2013, 135, 18778–18781 CrossRef CAS PubMed; (c) L. Wan, N. Dastbaravardeh, G. Li and J.-Q. Yu, J. Am. Chem. Soc., 2013, 135, 18056–18059 CrossRef CAS PubMed; (d) M. Bera, A. Modak, T. Patra, A. Maji and D. Maiti, Org. Lett., 2014, 16, 5760–5763 CrossRef CAS PubMed; (e) G.-J. Cheng, Y.-F. Yang, P. Liu, P. Chen, T.-Y. Sun, G. Li, X. Zhang, K. N. Houk, J.-Q. Yu and Y.-D. Wu, J. Am. Chem. Soc., 2014, 136, 894–897 CrossRef CAS PubMed; (f) R.-Y. Tang, G. Li and J.-Q. Yu, Nature, 2014, 507, 215–220 CrossRef CAS; (g) G. Yang, P. Lindovska, D. Zhu, J. Kim, P. Wang, R.-Y. Tang, M. Movassaghi and J.-Q. Yu, J. Am. Chem. Soc., 2014, 136, 10807–10813 CrossRef CAS PubMed; (h) Y.-F. Yang, G.-J. Cheng, P. Liu, D. Leow, T.-Y. Sun, P. Chen, X. Zhang, J.-Q. Yu, Y.-D. Wu and K. N. Houk, J. Am. Chem. Soc., 2014, 136, 344–355 CrossRef CAS PubMed; (i) M. Bera, A. Maji, S. K. Sahoo and D. Maiti, Angew. Chem., Int. Ed., 2015, 54, 8515–8519 CrossRef CAS; (j) L. Chu, M. Shang, K. Tanaka, Q. Chen, N. Pissarnitski, E. Streckfuss and J.-Q. Yu, ACS Cent. Sci., 2015, 1, 394–399 CrossRef CAS PubMed; (k) Y. Deng and J.-Q. Yu, Angew. Chem., Int. Ed., 2015, 54, 888–891 CrossRef CAS PubMed; (l) S. Li, H. Ji, L. Cai and G. Li, Chem. Sci., 2015, 6, 5595–5600 RSC; (m) S. Li, L. Cai, H. Ji, L. Yang and G. Li, Nat. Commun., 2016, 7, 10443 CrossRef; (n) A. Maji, B. Bhaskararao, S. Singha, R. B. Sunoj and D. Maiti, Chem. Sci., 2016, 7, 3147–3153 RSC; (o) T. Patra, R. Watile, S. Agasti, T. Naveen and D. Maiti, Chem. Commun., 2016, 52, 2027–2030 RSC; (p) B. E. Haines, J.-Q. Yu and D. G. Musaev, ACS Catal., 2017, 7, 4344–4354 CrossRef CAS; (q) P. Wang, P. Verma, G. Xia, J. Shi, J. X. Qiao, S. Tao, P. T. W. Cheng, M. A. Poss, M. E. Farmer, K.-S. Yeung and J.-Q. Yu, Nature, 2017, 551, 489–493 CrossRef CAS PubMed; (r) H.-J. Xu, Y. Lu, M. E. Farmer, H.-W. Wang, D. Zhao, Y.-S. Kang, W.-Y. Sun and J.-Q. Yu, J. Am. Chem. Soc., 2017, 139, 2200–2203 CrossRef CAS PubMed; (s) L. Yang, S. Li, L. Cai, Y. Ding, L. Fu, Z. Cai, H. Ji and G. Li, Org. Lett., 2017, 19, 2746–2749 CrossRef CAS; (t) C. Deng, J.-X. Zhang and Z. Lin, ACS Catal., 2018, 8, 2498–2507 CrossRef CAS; (u) H. Shi, A. N. Herron, Y. Shao, Q. Shao and J.-Q. Yu, Nature, 2018, 558, 581–585 CrossRef CAS; (v) M. Li, M. Shang, H. Xu, X. Wang, H.-X. Dai and J.-Q. Yu, Org. Lett., 2019, 21, 540–544 CrossRef CAS PubMed; (w) H.-J. Xu, Y.-S. Kang, H. Shi, P. Zhang, Y.-K. Chen, B. Zhang, Z.-Q. Liu, J. Zhao, W.-Y. Sun, J.-Q. Yu and Y. Lu, J. Am. Chem. Soc., 2019, 141, 76–79 CrossRef CAS PubMed; (x) J. Xu, J. Chen, F. Gao, S. Xie, X. Xu, Z. Jin and J.-Q. Yu, J. Am. Chem. Soc., 2019, 141, 1903–1907 CrossRef CAS PubMed; (y) G. Meng, N. Y. S. Lam, E. L. Lucas, T. G. Saint-Denis, P. Verma, N. Chekshin and J.-Q. Yu, J. Am. Chem. Soc., 2020, 142, 10571–10591 CrossRef CAS; (z) G. Li, Y. Yan, P. Zhang, X. Xu and Z. Jin, ACS Catal., 2021, 11, 10460–10466 CrossRef CAS; (a a) P. Muthuraja, R. Usman, R. Sajeev and P. Gopinath, Org. Lett., 2021, 23, 6014–6018 CrossRef CAS PubMed; (a b) P. Ramesh, C. Sreenivasulu, D. R. Kishore, D. Srinivas, K. R. Gorantla, B. S. Mallik and G. Satyanarayana, J. Org. Chem., 2022, 87, 2204–2221 CrossRef CAS PubMed; (a c) W. Ali, A. Saha, H. Ge and D. Maiti, JACS Au, 2023, 3, 1790–1799 CrossRef CAS PubMed; (a d) N. Goswami, N. Kumar, S. Bag, P. Gupta and D. Maiti, ACS Catal., 2023, 13, 11091–11103 CrossRef CAS; (a e) Y.-L. Zhu, Q.-X. Sun, J.-S. Feng, Y.-D. Shao and J. Chen, Org. Lett., 2023, 25, 4416–4421 CrossRef CAS PubMed; (a f) K. Mounika and G. Satyanarayana, Org. Lett., 2024, 26, 8899–8903 CrossRef CAS PubMed; (a g) D. Srinivas and G. Satyanarayana, J. Org. Chem., 2024, 89, 433–451 CrossRef CAS; (a h) S. Ghosh, N. Jain and R. B. Sunoj, ACS Catal., 2025, 15, 20251–20269 CrossRef CAS; (a i) X. Yang, H. Liu, Y.-D. Li, J. Wang, H.-J. Xu and Y. Hu, J. Org. Chem., 2025, 90, 7279–7289 CrossRef CAS PubMed.
- X. Chen, S. Fan, M. Zhang, Y. Gao, S. Li and G. Li, Chem. Sci., 2021, 12, 4126–4131 RSC.
- H. F. Motiwala, A. M. Armaly, J. G. Cacioppo, T. C. Coombs, K. R. K. Koehn, V. M. I. V. Norwood and J. Aubé, Chem. Rev., 2022, 122, 12544–12747 CrossRef CAS PubMed.
- (a) N. T. Patil, V. S. Raut, R. D. Kavthe, V. V. N. Reddy and P. V. K. Raju, Tetrahedron Lett., 2009, 50, 6576–6579 CrossRef CAS; (b) H.-X. Dai, G. Li, X.-G. Zhang, A. F. Stepan and J.-Q. Yu, J. Am. Chem. Soc., 2013, 135, 7567–7571 CrossRef CAS PubMed; (c) M. J. O'Neill, T. Riesebeck and J. Cornella, Angew. Chem., Int. Ed., 2018, 57, 9103–9107 CrossRef; (d) R. Zhao and W. Lu, Organometallics, 2018, 37, 2188–2192 Search PubMed; (e) H. Xu, M. Liu, L.-J. Li, Y.-F. Cao, J.-Q. Yu and H.-X. Dai, Org. Lett., 2019, 21, 4887–4891 CrossRef CAS PubMed; (f) H.-Y. Hao, S.-J. Lou, S. Wang, K. Zhou, Q.-Z. Wu, Y.-J. Mao, Z.-Y. Xu and D.-Q. Xu, Chem. Commun., 2021, 57, 8055–8058 RSC; (g) D. Srinivas and G. Satyanarayana, Org. Lett., 2021, 23, 7353–7358 Search PubMed; (h) K. Mounika, D. Srinivas and G. Satyanarayana, J. Org. Chem., 2025, 90, 7888–7901 CrossRef CAS PubMed.
- (a) A. Modak, T. Patra, R. Chowdhury, S. Raul and D. Maiti, Organometallics, 2017, 36, 2418–2423 CrossRef CAS; (b) R. Jayarajan, J. Das, S. Bag, R. Chowdhury and D. Maiti, Angew. Chem., Int. Ed., 2018, 57, 7659–7663 CrossRef CAS PubMed; (c) T. Bhattacharya, C. Teja, N. Kumar, K. K. Bhagat, G. K. Lahiri, P. Gupta, S. Tyagi and D. Maiti, ACS Catal., 2024, 14, 2216–2228 CrossRef CAS; (d) Y. Bairagi, S. Porey, I. Mahato and D. Maiti, ACS Catal., 2025, 15, 7536–7542 CrossRef CAS.
- E. M. Simmons and J. F. Hartwig, Angew. Chem., Int. Ed., 2012, 51, 3066–3072 Search PubMed.
- (a) J. J. Topczewski and M. S. Sanford, Chem. Sci., 2015, 6, 70–76 Search PubMed; (b) I. J. S. Fairlamb, Angew. Chem., Int. Ed., 2015, 54, 10415–10427 CrossRef CAS PubMed; (c) C. Testa, J. Roger, P. Fleurat-Lessard and J.-C. Hierso, Eur. J. Org Chem., 2019, 2019, 233–253 CrossRef CAS; (d) X. Xiong, Y.-J. Mao, H.-Y. Hao, Y.-T. He, Z.-Y. Xu, G. Luo, S.-J. Lou and D.-Q. Xu, Org. Biomol. Chem., 2020, 18, 6732–6737 RSC.
- CCDC 2366841: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2kfwqm.
| This journal is © The Royal Society of Chemistry 2026 |