Open Access Article
Open Access ArticleOrganic photochemistry for direct light-driven separations
Ariel Y.
Wang
 a,
Bayu I. Z.
Ahmad
a,
Bayu I. Z.
Ahmad
 b,
Carolyn
Ma
b,
Phillip J.
Milner
b,
Carolyn
Ma
b,
Phillip J.
Milner
 *b and
Richard Y.
Liu
*b and
Richard Y.
Liu
 *a
*a
aDepartment of Chemistry and Chemical Biology, Harvard University, 12 Oxford St., Cambridge, MA 02138, USA. E-mail: richardliu@chemistry.harvard.edu
bDepartment of Chemistry and Chemical Biology, Cornell University, Ithaca, NY, USA. E-mail: pjm347@cornell.edu
First published on 18th March 2026
Abstract
Compared to conventional methods, direct light-driven separations are promising strategies to achieve high selectivity while lowering energy cost. This perspective examines recent progress in photoswitch-enabled separations with a focus on concentration of CO2 and selective anion recovery from water. We highlight key design strategies for photo-pH-swing capture and identify current practical limitations toward translating solar-driven separations into technologies with meaningful impact.
Introduction
Efficient chemical separations are indispensable to the functioning of modern society. Beyond commodity chemical production, separation science underpins technologies that sustain and improve quality of life, from the purification of essential resources like air and water to the recycling of nutrients such as nitrogen and phosphorus. Despite their necessity, industrial scale separation operations remain wasteful and energy intensive. Separations account for more than half of the total industrial energy expenditure1 and up to ca. 15% of global energy consumption.2,3 In addition, up to 80% of these processes involve thermally driven steps (e.g., distillation, evaporation, drying, etc.), which depend heavily on heat from fossil fuel sources,3–5 further contributing to the current climate crisis. Recent efforts to integrate renewable energy sources into industrial systems, in the form of green electricity,6–9 have produced some environmental benefits. However, apart from technological challenges of integrating green energy with traditional separation methods, there are additional issues with the storage and distribution of clean energy.10 With rising energy demands, renewable electricity alone cannot meet long-term sustainability and emissions goals without also reducing energy requirements at the process level. Recognizing this challenge, several national and international agencies have identified the development of next-generation, low-energy separation technologies as a cornerstone of sustainable industrial practice and decarbonization strategy.4,5,11–13Unlike traditional thermal methods, non-thermal separations can operate without macroscopic phase transitions, instead exploiting molecular differences in size and/or chemical affinity to achieve selective transport or adsorption. Among these, membrane-based separations have emerged as a dominant platform,14,15 particularly in water desalination,16–20 gas purification,21,22 and bioproduct recovery.23–26 This expansion has been driven largely by progress in polymer synthesis and fabrication, which has led to improvements in membrane performance, lifetime, and applications.27,28 The impact of non-thermal separations is exemplified by reverse-osmosis desalination membranes, which have lowered energy demand from 16 kWh m−3 to ca. 2 kWh m−3.16 Apart from polymer-based porous membranes, the rise of porous crystalline materials, such as metal–organic frameworks (MOFs), further revolutionized sorbent design by offering unprecedented tunability in terms of pore size and chemistry.29–31
Although bulk purifications have advanced significantly, many modern challenges, such as carbon capture, uranium recovery from seawater, and rare earth metal purification from ores, require isolating targets from extremely dilute input streams. Concentrating from dilute sources not only requires high selectivity but also the ability to overcome a significant entropic penalty. One strategy to achieve this is the use of chemical sorbents that can form very strong bonds with the target species, thereby driving chemisorption. While this allows for selective capture from dilute sources, significant energy input is then required to drive the desorption process and regeneration of the sorbent. For amine-based systems, the prototypical chemisorbent, regeneration can account for up to 80% of the cost of operation.32,33 The trade-off between affinity, selectivity, and energetic cost continues to be challenging for both thermal and non-thermal separation systems.
Aiming to achieve a more favorable energetic profile, researchers have become interested in metastable systems that, upon activation of an external stimulus, can generate non-equilibrium states that participate in separations. Known approaches harness external changes in pressure,34 voltage,35 or electromagnetic fields,36 to generate reversible chemical changes that facilitate selective transport or desorption. This perspective highlights an emerging frontier: light-driven separation processes.37 Because sunlight is the most abundant renewable resource, directly coupling photon energy to separation steps bypasses the inefficiencies of generating, storing, and distributing electricity. Photoresponsive materials, especially those incorporating molecular photoswitches, offer a direct way to translate photon energy into chemical or structural transformations, generating out-of-equilibrium states that are capable of selective and efficient capture/release. In the following sections, we review recent advances in light-driven separations, with an emphasis on their applications in CO2 capture and water purification. We will discuss key design strategies, material considerations, and future directions for enabling scalable and low-energy photoresponsive separation technologies (Table 1).
| Ref | Method | System | pH range | Operating temperature | Capture capacity | Quantum yield | Reversibility |
|---|---|---|---|---|---|---|---|
| 52 | pH swing | Indazole mPAH and glycine in water | 8–10 | Room temperature | 15.6 µmol CO2 mL−1 h−1 (20 mM solution under DAC conditions) | — | 3 cycles with diminishing yields |
| 53 | pH and temperature swing | Pyranine and K2CO3 in water | 7.55–7.65 | ∼40 °C | 26.7 µmol CO2 mL−1 h−1 (0.1 M solution from 15% CO2 in N2) | 0.125 photon utilization | No degradation over 168 cycles (7 days) |
| 59 | pH swing | Merocyanine in 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 DMSO/water 1 DMSO/water |
6.9–9.9 | Room temperature | ∼6 µmol CO2 mL−1 h−1 (20 mM solution from 10% CO2 in N2) | 1.3% | No difference in activity over 3 capture and release cycles |
| 60 | pH swing | Fluorenol photobase in 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 ACN/water 1 ACN/water |
8.5–11.6 | Room temperature | 0.56 µmol CO2 mL−1 h−1 (10 mM solution under DAC conditions) | 20.1% | No difference in activity over 3 capture and release cycles |
| 61 | pH swing | PyDIG and GlyGly in water | 6.88–7.81 | 23–27 °C | 1.46–1.81 µmol CO2 mL−1 h−1 (1 mM solution under DAC conditions) | 4.01% | No difference in activity over 5 capture and release cycles |
| 72 | Photo-isomerization | Azobenzene-guanidine in ethanol | — | ∼40 °C | 2.4 µmol CO2 mL−1 h−1 (0.2 M solution under DAC conditions) | — | No degradation in activity over 3 capture and release cycles |
| 74 | Photo-tautomerization | 2MeBP and BzOK in DMSO | — | 60–65 °C | 26 µmol CO2 mL−1 h−1 (0.1 M solution from 10% CO2 + 3% O2 in N2) | — | No degradation over 5 capture and release cycles |
Light-driven CO2 concentration
Capturing CO2 from point sources or directly from the atmosphere is an important component of broader climate-mitigation strategies. CO2 also serves as a precursor to key commodities such as methanol and urea, giving it additional value as a chemical feedstock. Over the past 15 years, a multitude of new carbon-capture technologies have emerged.38–40 Most sorbents rely on highly exothermic interactions that overcome the entropy loss during concentration. Common functional groups such as amines and hydroxides form interactions on the order of tens of kJ mol−1 with CO2, which, while beneficial for capture, make regeneration a major hurdle.41,42 Thermal CO2 desorption is not only energy-intensive43 but also introduces secondary issues, such as water evaporation and sorbent decomposition.44,45 Thus, an important objective in CO2-capture system design is to enable low-energy, on-demand desorption. Light is an appealing trigger for this purpose because it is abundant, spatially addressable, and can be coupled selectively to specific molecular events. The following section outlines recent advances in light-driven strategies for separation of CO2, particularly highlighting methods involving pH swing and photoisomerization.Light-induced pH swing
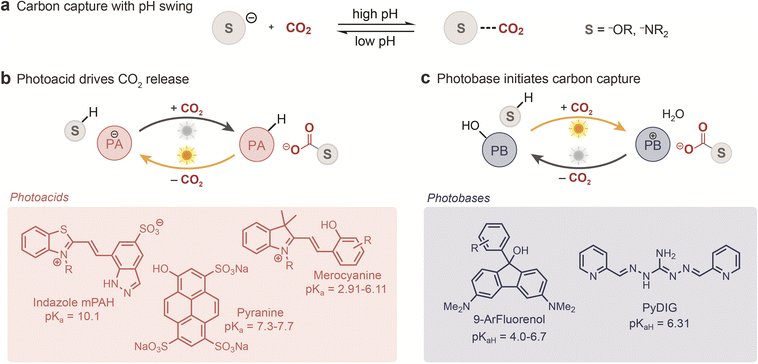
Capture-and-release using a photoinduced pH-swing has emerged as a leading strategy for light-driven CO2 separation.37,46 Because the solubility of CO2-derived inorganic species in water is strongly pH-dependent, photoresponsive chemistries capable of modulating pH upon irradiation offer a means to drive reversible CO2 absorption and release (Fig. 1a). Among these systems, metastable photoacids, molecules that induce a substantial, reversible decreases in pH upon excitation, have gained prominence. Photoacids have had a long history of applications in lithography,47 catalysis,48 and polymer synthesis,49 but more recently have been adapted as light-responsive agents for regenerating CO2 sorbents. Once excited, photoacids provide a transient reservoir of protons that can react with the CO2–sorbent adduct, thereby promoting CO2 release and sorbent regeneration (Fig. 1b).Although the formation of carbonic acid using an excited-state photoacid had been observed spectroscopically,50 the use of photoacids for triggering CO2 release in bulk solution was not explored until recently by Bennett et al.51 Using a merocyanine photoacid, the authors showed that an already acidic bicarbonate-containing solution could release CO2 in an accelerated fashion under irradiation. Although not integrated into a full capture-and-release cycle, this study provided a crucial proof of concept that photoacids can generate sufficiently large pH shifts to promote gaseous CO2 release. Building on this foundation, Ma and co-workers reported in 2023 that an indazole-based photoacid could drive CO2 release from glycine solutions saturated with ambient CO2, achieving up to 78% release after 1 h of irradiation.52 Although cyclic CO2 uptake and release was not performed, the results indicate that photoacids may offer a viable route for regenerating amine-based DAC sorbents.
Hatton and co-workers have shown that glycine-based aqueous capture systems can be photochemically regenerated using the water-soluble, excited-state photoacid pyranine.53 Pyranine's combination of photostability, high quantum yield and excellent water solubility makes it an ideal target for carbon capture applications. Mixtures of pyranine and up to 0.8 M glycine (a ca. 800-fold increase compared to previous studies)52 were able to sequester and release CO2 from an input stream of 15% v/v CO2 in N2 (simulating an industrial flue gas mixture). Control experiments indicated that the mechanism for bulk CO2 release was dominated by photothermal heating and water-vapor formation. In fact, photoacid-induced pH changes near the solution surface contributed significantly only during the first ∼30 seconds of simulated release, as calculations showed that the population and lifetime of excited states are too low to support significant bulk pH changes. To study photoacid-induced CO2 desorption in isolation, the authors constructed a continuous-flow photochemistry setup and demonstrated sustained CO2 release from solutions preloaded in the dark with a 15% v/v CO2 in N2 feed. Continuous operation of this flow system over 1 week yielded 60 mmol of CO2, with a photon utilization of 0.125 CO2 molecules liberated per photon. Under simulated DAC conditions (ca. 420 ppm CO2 in ambient air), continuous operation sequestered 3 µmol over 2 h. These results demonstrate that light-swing operation is feasible in practical flow architectures and highlight the strong dependence of photorelease efficiency on reactor design.
To bypass limitations such as water solubility and photoacid hydrolysis, Dawlaty and co-workers attempted to directly release CO2 from monoethanolamine (MEA) with a merocyanine photoacid.54 As in aqueous systems, CO2–MEA adducts can be reversibly driven toward CO2 release in the presence of added protons, and a total of ca. 30 mg (0.3%) mass loss from bulk solution was observed over 14 h. Relative to dark controls, irradiated MEA–photoacid solutions exhibited roughly twice the mass loss, attributed primarily to CO2 release. This study demonstrated the feasibility of photoacid-induced CO2 release in amines, but it also highlighted several practical constraints. Pure MEA is highly viscous at rt, over 20 times more viscous than water,55,56 requiring heating to 80 °C to permit efficient CO2 outgassing. Although this is below the temperature required for conventional thermal release (ca. 120 °C for amine sorbents),57 it still imposes a substantial energy burden for continuous operation. Furthermore, CO2 release is slower in MEA than in water-containing mixtures (e.g., a 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 H2O/MEA mixture), consistent with slower proton-transfer dynamics. These limitations pose significant barriers for scaling such systems, and alternative strategies to make aqueous-based capture systems more efficient and robust may be preferable.
1 H2O/MEA mixture), consistent with slower proton-transfer dynamics. These limitations pose significant barriers for scaling such systems, and alternative strategies to make aqueous-based capture systems more efficient and robust may be preferable.
Changes to the solvent system have been found to strongly impact both long term stability and release capacity of photoacids in water. To systematically probe the role of solvation, Lukatskaya and co-workers conducted an in-depth study of merocyanine photoacids in DMSO/water mixtures.58 Their results revealed that optimizing the solvent composition could profoundly enhance both photoacid performance and stability. At an optimal DMSO mole fraction of 0.15, the merocyanine photoacids exhibited a fivefold increase in CO2 release and more than doubled their lifetime relative to pure water. DMSO served as a stabilizing cosolvent, improving solubility and suppressing hydrolytic degradation, but at the expense of diminished pH swing and slower relaxation back to the ground state. To rationalize the observed trends, the authors used molecular dynamics simulations, which revealed a distinctive microsolvation pattern: DMSO primarily coordinated to the nonpolar regions of the photoacid, while water is localized near the polar phenolic and carbonyl sites. This selective solvation pattern explains the disproportionate impact of DMSO on stability. For instance, DMSO coordination at the alkene, the site most susceptible to hydrolytic attack, can impede decomposition even at relatively low concentrations.
The preceding examples have involved two separate components: a traditional sorbent to perform CO2 capture (e.g., amine and hydroxide), and a photoacid to regulate release. This raises a natural question: can a single compound mediate both capture and release steps? In principle, the merocyanine formed upon ring opening of spiropyran contains a phenoxide that may be sufficiently basic to form salts with carbonic acid. Leveraging this property, Liu and co-workers developed a one-component merocyanine photoacid system that enabled CO2 separation under simulated flue-gas conditions (10% v/v CO2 in N2).59 To achieve efficient and cyclic CO2 separation processes, they showed that the pKa (dark) of the photoacid must be carefully matched to the effective pKa of CO2 (ca. 6.25). If the photoacid is too basic, not enough protons are available for effective release; if it is too acidic, the dark pH becomes too low to enable effective capture. Although developed in the context of a single organic component, this principle can be extended naturally to multicomponent systems. During capture and release, protons are shuffled between the sorbent and the photoacid, so optimal performance requires matching the pKa values of the key components of the system.
Light can also be used not only to trigger CO2 release but also to drive CO2 capture itself. As systems achieving higher pH have better potential and greater capacity for fast CO2 uptake, photobases, which transiently raise solution pH upon irradiation, offer a more promising strategy for capturing CO2 from dilute streams (Fig. 1c). Recent work from the Liu group demonstrates that newly developed fluorenol photobases can indeed drive CO2 capture from dilute streams, including air.60 These Arrhenius photobases transiently release hydroxide ions upon photoexcitation, driving CO2 capture through bicarbonate formation. Once the light is removed, the photobase relaxes and the pH returns toward its initial value, promoting CO2 release. Liu and co-workers achieved efficient capture under simulated DAC conditions (400 ppm CO2) and enabled subsequent release at concentrations more than sevenfold higher than that of the input stream.
In a complementary approach, Custelcean and coworkers recently reported the use of a diiminoguanidine photobase (pyDIG) to initiate capture with peptide sorbents.61 Upon irradiation with UV light, the pH increase leads to deprotonation of the zwitterionic peptide, initiating carbon capture. Under this system, a 120 mL solution of 1 mM pyDIG/peptide captured up to 0.0467 mmol of CO2 within a 24 h period under irradiation, maintaining similar performance over four cycles. The thermal (“dark”) release process has a weaker driving force, taking up to 5 days to complete at rt, but the process can be sped up to 2 d by heating to 80 °C. Although photobase-driven capture remains in early stages of development, these results highlight a compelling complement to photoacid strategies – providing a light-capture, dark-release pathway particularly suited to low-temperature CO2 separations.
Guiding principles for pH-swing-driven carbon capture
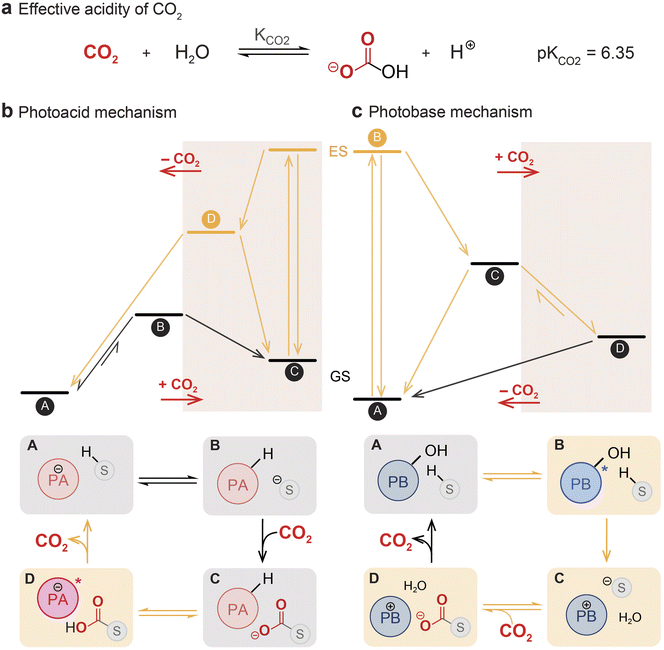
Given the number of carbon capture systems based on photoacids and photobases that have been reported to date, several fundamental principles governing their operation can be gleaned. Light-driven carbon capture systems that operate through a pH-swing mechanism involve proton transfer between dissolved CO2 and the photoswitch. In photoacid systems, the solutions typically begin at high pH, which decreases during CO2 uptake from the gas phase (Fig. 2b). When exposed to light, the photoacid transitions to a metastable state that favors releasing its proton to the surrounding medium. This proton transfer shifts the equilibrium of bicarbonate and/or carbamate species back toward molecular CO2, reversing the capture process. In comparison, photobases operate via the opposite pH trajectory. Upon photoexcitation, a photobase transiently increases the solution's pH by abstracting protons, thereby promoting the capture of CO2 by hydroxides. When the photobase relaxes to its ground state, its acidity is restored, resulting in proton donation and subsequent CO2 release (Fig. 2c). Thus, both photoacids and photobases rely on reversible, light-mediated pH modulation to toggle between CO2 capture and release.The efficiency of these proton relays is intimately tied to the acid–base equilibria governing CO2 speciation, from carbonic acid to its various basic forms (Fig. 2a). Although the true pKa of carbonic acid is closer to 3.4,50,62 it is a minor species under relevant conditions. Accounting for the hydration and solubility equilibria of CO2 results in the useful effective value of pKa = 6.35 for CO2 at 25 °C. Liu's work has shown that photoacids with ground-state pKa values near this threshold value are best suited for efficient CO2 release. Photoacids with lower pKa will have a decreased capacity for CO2 capture. In contrast, photoacids with higher pKa cannot efficiently release CO2, as the equilibrium always favors carbon capture over release. Similarly, in photobase systems, the optimal fluorenyl photobase used by Liu and co-workers has a conjugate acid pKa of 6.1, which allows for effective CO2 release in the dark. Some exceptions exist to this pH-matching principle: for example, pyranine, with a ground-state pKa of ca. 7.3, was found to release CO2, although this is largely through bulk photothermal heating rather than through a pH swing.
In general, a well-designed photoswitch should have a large pKa difference between ground and metastable states. In addition, the ground-state pKa of the photoacid (or conjugate pKa of the photobase) should be near the effective pKa of CO2 (6.35). Tuning the initial pH of the system with basic additives can improve the overall capture ability of the system, which was reflected in the increased capturing capacity observed by Ma and co-workers upon initial pH adjustment of their system with KOH.52 However, such pH modifications can also alter the speciation and photochemical behavior of the photoswitch, potentially reducing its capacity to donate or accept protons. Added sorbents that act as buffers (for example, amines) should be considered in combination with the photoactive agent, and the behavior of the composite system is not easily predicted from the pKa values of the photoswitch alone.
CO2 release using light-induced isomerization
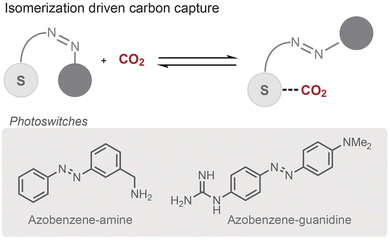
Another strategy for inducing CO2 release relies on light-driven structural changes within the sorbent compound. Light-induced geometric isomerization is a hallmark of photoswitches and has been leveraged in a number of materials applications, including biomacromolecules,63 polymers,64 and molecular machines.65 Many photoresponsive materials are based on azobenzene chemistry, which undergoes trans-to-cis isomerization upon irradiation with light. For CO2 sorbents, isomerization can introduce unfavorable steric clashes around adsorption sites that destabilize bound CO2 and shift the equilibrium towards release (Fig. 3). This approach has been applied largely in the solid state, including porous metal–organic and covalent organic frameworks.66–70 Although these studies establish the potential of isomerization-induced release for sorbent regeneration, they have not yet demonstrated separation from dilute streams such as ambient air.In principle, solid-state adsorption has several advantages over solution-state sorption, including bypassing limitations with solubility, offering improved long-term stability and gravimetric capacities, and reducing energy losses from solvent evaporation.71 However, such solid-state systems suffer from limitations of their own, including poor thermal transport, slow adsorption kinetics, and the need for additional mechanical processing. Recent work by Inagaki and co-workers illustrates several challenges with solid-state DAC.72 The authors developed an amine-based sorbent that incorporates an azobenzene switch to promote CO2 release. From elevated CO2 concentrations, the sorbent was able to achieve the theoretical capacity and release limit of the system (1 CO2 per 2 amine sites). However, the carbon-capture efficiency was highly dependent on the CO2 input concentration, and light-triggered release under DAC conditions yielded a lower loading ratio of 2 CO2 molecules per 5 amine sites. In addition, solid-state systems appear extremely sensitive to changes in the adsorption site: substituting the amine with guanidine eliminated CO2 capture entirely, even though the more basic guanidine was expected to enhance uptake (Fig. 3). By contrast, transferring the system from the solid state to ethanolic solution restored the activity of the guanidine-based sorbent, achieving up to a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 CO2 per amine capture ratio. The resulting sorbent–CO2 adduct could be isolated as a precipitate and the bound CO2 released over a 2 h period. The ability to sequester CO2 as a solid adduct can be valuable, improving the ease of handling and transportation, with release occurring at centralized hubs or points of use, such as greenhouses or chemical manufacturing facilities.73
1 CO2 per amine capture ratio. The resulting sorbent–CO2 adduct could be isolated as a precipitate and the bound CO2 released over a 2 h period. The ability to sequester CO2 as a solid adduct can be valuable, improving the ease of handling and transportation, with release occurring at centralized hubs or points of use, such as greenhouses or chemical manufacturing facilities.73
Photenol capture
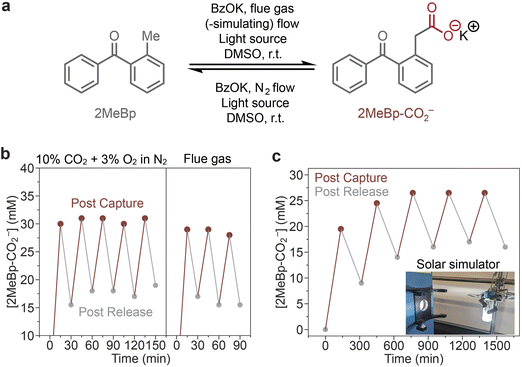
In all of the photoswing CO2 separations outlined above, one of the two steps—capture or release—was carried out in the dark. This invites the question: is a fully light-driven process for CO2 capture and release possible? Such a process would benefit from potential isothermal operation as well as the distribution of the energetic requirements across both steps. Milner and co-workers recently reported such a process using the inexpensive and photoresponsive organic molecule 2-methylbenzophenone (2MeBP, Fig. 4a).74 Previous work had shown that, upon UV light irradiation, 2-methylbenzophenone analogs can photo-tautomerize to the corresponding photoenols, which can then undergo cycloaddition with dienophiles such as CO2.75–78 Subsequent ring-opening (in the case of CO2) leads to the corresponding arylacetic acid derivative. While previous work had shown that these molecules are capable of CO2 release, UV light and long reaction times were required.79 Because the photodecarboxylation of 2-arylacetate derivatives is a common process in photoredox catalysis,80 the authors hypothesized that treatment of the obtained arylacetic acid product with base to generate the corresponding carboxylate (2MeBP–CO2−, Fig. 4a) would greatly accelerate CO2 photorelease. The successful combination of these two processes enabled the first light-driven process for CO2 capture and release.74Milner and co-workers commenced by demonstrating that CO2 capture with the photoenol derived from 2MeBP can be conducted under more realistic conditions than in previous studies, including with visible light irradiation (390 nm), dilute CO2 streams (10% v/v CO2 in N2), short reaction times (30 min), and in the presence of O2 as a contaminant. Next, the feasibility of CO2 release was evaluated. As expected, CO2 release from the carboxylic acid product was negligible upon light irradiation or heating to 60 °C. In contrast, irradiation of 2MeBP–CO2− with light (390 nm) led to rapid CO2 release and reformation of 2-MeBP upon back proton transfer. Remarkably, the presence of base during the capture step led to enhanced CO2 capture via the corresponding photoenolate. This is advantageous because it means that the base can remain in the reaction mixture during both the capture and release steps. Transient absorption (TA) spectroscopy measurements and density functional theory (DFT) calculations support that the decarboxylation step proceeds via a unique intramolecular photodecarboxylation pathway on the ps timescale.
To assess the ability of the combined CO2 capture and release system, the authors performed cycling experiments using more realistic mixtures. 5 cycles of capture and release under constant light irradiation were achieved, simply by switching between 10% v/v CO2 in N2 flow (capture) and pure N2 flow (release) to simulate a light-driven, pressure-swing process (Fig. 4b, left). CO2 removal was also achieved from a sample of flue gas from Cornell University's natural gas-fired power plant (Fig. 4b, right) and using a solar simulator instead of 390 nm light (Fig. 4c). Capture and release were also performed in the window of the authors' laboratory in Ithaca, NY, demonstrating the potential for this system to mediate CO2 removal using natural sunlight as the primary energy input. Future studies should focus on increasing the kinetics of the capture and release steps and improving the absorbance overlap of 2-methylbenzophenone variants with the solar spectrum.
Anion capture and water purification
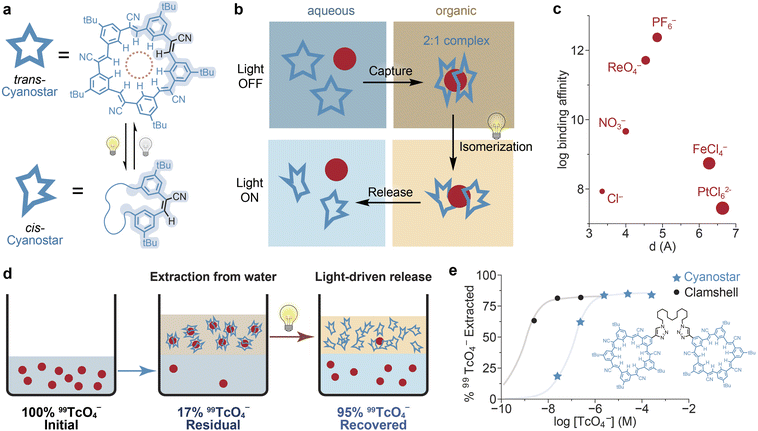
Contamination of drinking water with certain anions represents a significant challenge for water potability. These include inorganic anions such as sulfate, arsenate, nitrate, perchlorate, and selenate, as well as organic anions such as per- and poly-fluoroalkyl substances (PFAS). Previously overlooked anions such as chloronitramide have also recently been identified as hazardous contaminants of municipal water supplies.81 Numerous sorbents have been developed to remove these ions from water and make it fit for human consumption.82–85 However, the high binding affinities of these complexes hinder their subsequent regeneration. To address this issue, supramolecular complexes that change their anion-binding affinities upon light irradiation have been identified.86–95 However, most of these compounds have not been employed to achieve the effective removal of anions from water, but were merely shown to modulate the concentration of free anions in aqueous solution.Recently, Flood and co-workers reported a pioneering example of photoswitch-based anion removal from water.96 Specifically, they demonstrated that the cyanostilbene macrocycle cyanostar97 is capable of extracting the hexafluorophosphate (a byproduct of Li-ion battery manufacturing) and perrhenate (a surrogate for radioactive pertechnetate-99)98 anions from aqueous solution. Irradiation with light causes some of the trans-stilbene units to isomerize to their cis conformers, dramatically decreasing the anion-binding affinity of the macrocycle and triggering release (Fig. 5a and b). In its all-trans form, this macrocycle displays high affinities for larger anions (e.g., hexafluorophosphate) over common anion contaminants (e.g., chloride, nitrate, and sulfate), making it a suitable platform for selective ion extraction from water (Fig. 5c). The cyanostar binds large anions through the formation of a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex, with the anion trapped between two molecules of the macrocycle, as confirmed by single-crystal X-ray diffraction.97
1 complex, with the anion trapped between two molecules of the macrocycle, as confirmed by single-crystal X-ray diffraction.97
The ability of cyanostar to mediate selective anion removal from water was evaluated using a liquid–liquid extraction (LLE) setup. First, cyanostar was shown to facilitate the partitioning of tetraethylammonium hexafluorophosphate from the aqueous phase into the organic phase, even in the presence of competing chloride. Next, the release of free anions was evaluated by irradiating the anion-bound complex with 365 nm light. Release of the bound anion was supported by NMR spectroscopy, which confirmed both the trans → cis isomerization of the stilbene units in cyanostar as well as the recombination of released hexafluorophosphate anions with tetraethylammonium cations in solution. The authors then designed an LLE experiment by simply using anion-binding to extract hexafluorophosphate from the aqueous phase to the organic phase using cyanostar in the dark, and then releasing it back into fresh water upon light irradiation. Remarkably, the extraction of the anion was quantitative even in the presence of large excesses of competing chloride (up to 4 M), supporting the potential viability of this system for real-world applications.
The authors next turned their attention to extracting perrhenate from water. Both sodium and ammonium salts were employed, and the exchange of perrhenate was validated using UV-vis spectroscopy. Under similar LLE conditions as employed for hexafluorophosphate capture, quantitative perrhenate extraction from the aqueous phase to the organic phase was observed in the dark. Light irradiation allowed for 87% release of perrhenate back into the aqueous phase. A similar LLE for pertechnetate-99 was also demonstrated down to a concentration of 400 ppb, using the radioactivity of each phase to track anion extraction and release (Fig. 5d). Tethering two cyanostar macrocycles into a “clamshell” preorganizes the requisite 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 host–guest architecture and enabled extraction of pertechnetate-99 from solutions as dilute as 4 ppb, reflecting its exceptional binding affinity (Fig. 5e). Overall, Flood's work represents a significant step forward for the use of photoresponsive cages to mediate anion capture from aqueous solutions in an LLE setup. The reliance on a 2
1 host–guest architecture and enabled extraction of pertechnetate-99 from solutions as dilute as 4 ppb, reflecting its exceptional binding affinity (Fig. 5e). Overall, Flood's work represents a significant step forward for the use of photoresponsive cages to mediate anion capture from aqueous solutions in an LLE setup. The reliance on a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 binding mode (reducing the sorption capacity) and the need for UV-light irradiation are practical limitations that may be addressed in future work. The labor and resource intensity of LLE99 could also be overcome by adopting such a reactive motif within a porous material to enable solid-phase extraction.
1 binding mode (reducing the sorption capacity) and the need for UV-light irradiation are practical limitations that may be addressed in future work. The labor and resource intensity of LLE99 could also be overcome by adopting such a reactive motif within a porous material to enable solid-phase extraction.
Outlook
One of the central challenges in advancing light-driven separations is understanding and controlling how external factors, notably the solvation environment and additives, influence the behavior of photoresponsive molecules. For instance, subtle variations in solvent composition, initial pH, or irradiation wavelength have been shown to produce unexpectedly large shifts in photoacid performance, defying simple mechanistic interpretation.52,54,58 These sensitivities highlight that photoacid performance is not an intrinsic molecular property, but emerges from complex solvent–solute interactions that modulate proton transfer, stability, and switching dynamics. Beyond solvent composition, additives such as amino acids, bases, and electrolytes further complicate the chemical environment, and it is currently difficult to predict their effect on photoswitching from first principles. Progress in both experimental photophysics100,101 and computational modeling102 are crucial to improving our understanding of key intermediates and equilibria governing photoacid performance and long-term stability.For light-driven separations to advance beyond proof-of-concept demonstrations and mature into deployable technologies, laboratory discoveries must translate into systems that are scalable, robust, and process-relevant. To date, most experimental studies have emphasized fundamental aspects, such as photoacid stability, pH-swing dynamics, and mechanistic pathways for CO2 release, studied in small-scale, idealized environments. While such controlled investigations are essential for elucidating underlying mechanisms, the next stage of development demands integrated systems capable of operating under realistic process conditions and at meaningful throughput. This includes the use of realistic (sun)light fluxes and inclusion of realistic contaminants, including water, oxygen, SOx, and NOx, in input streams. In addition, long-term cycling stability (e.g., over thousands of cycles) has yet to be demonstrated for any of the systems discussed herein.
Process modeling can play a central role in identifying bottlenecks, guiding design trade-offs, and prioritizing the parameters critical for large-scale optimization. For example, a recent preprint by Lukatskaya and co-workers modeled a fully light-driven carbon-capture workflow based on spiropyran-mediated pH swings.103 This analysis explored the operational window required to achieve high CO2 recovery while minimizing energy input. The authors found that attaining >90% CO2 recovery with substantially reduced electricity demand would require photoacid concentrations exceeding 0.3 M. Reaching near-molar photoacid concentrations in aqueous media presents a formidable solubility challenge, and additional molecular or system-level modifications may be needed to maintain photochemical reversibility at such high loadings. Additional modeling of viable process and scrubber designs is needed to help push photoswing separations towards realistic operation.
Scaling present-day photoswing separations systems will require concurrent optimization across multiple dimensions, from the molecular design of the photoswitch to the architecture of realistic photoreactor platforms. Meeting these challenges will necessitate interdisciplinary collaboration, bridging synthetic chemistry, photophysics, and process engineering to establish a pathway toward practical, light-driven separations.
Author contributions
All authors contributed to writing of this manuscript.Conflicts of interest
P.J.M. and B.I.Z.A. are listed as inventors on a patent that includes a fully light-driven carbon capture process using photoenols (Cornell University, international patent application no. PCT/US24/42589, filed 15 August 2024). Harvard University is in the process of applying for patent(s) on the use of photobases for CO2 capture, on which R.Y.L. is listed as an inventor.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Acknowledgements
The development of photocycloaddition reactions for chemical separations was supported by the National Science Foundation (CBET-2047627) (B.I.Z.A., C.M., and P.J.M.). Any opinions, findings, and conclusions or recommendations expressed in this material are those of the author(s) and do not necessarily reflect the views of the National Science Foundation. Further support was provided by the Carbontech Development Initiative (B.I.Z.A. and P.J.M.), the Cornell Atkinson Center for Sustainability through a Graduate Research Grant to B.I.Z.A., and a Camille Dreyfus Teacher-Scholar Award to P.J.M. (TC-23-048). We also acknowledge the support of Arkema USA for a fellowship awarded to B.I.Z.A. R.Y.L. is grateful to the Salata Institute for Climate and Sustainability for partial support for the development of photoswitches for direct air capture. This work was also supported by the National Science Foundation through a CAREER award to R.Y.L. (CHE-2338206) and the Graduate Research Fellowship Program for A.Y.W.References
- J. A. C. Velasco, M. Tawarmalani and R. Agrawal, Systematic Analysis Reveals Thermal Separations Are Not Necessarily Most Energy Intensive, Joule, 2021, 5(2), 330–343, DOI:10.1016/j.joule.2020.12.002.
- E. C. D. Tan, E. P. Favvas and A. Brunetti, Editorial: Future Perspectives on Separation Technologies, Front. Sustainable, 2024, 5, 1411937, DOI:10.3389/frsus.2024.1411937.
- D. S. Sholl and R. P. Lively, Seven Chemical Separations to Change the World, Nature, 2016, 532(7600), 435–437, DOI:10.1038/532435a.
- National Academies of Sciences, Engineering, and Medicine. A Research Agenda for Transforming Separation Science, National Academies Press, Washington, D.C., 2019, p 25421 Search PubMed.
- IEA, Renewable Energy for Industry, IEA, Paris, 2017, https://www.iea.org/reports/renewable-energy-for-industry Search PubMed.
- M. Wei, C. A. McMillan and S. De La Rue Du Can, Electrification of Industry: Potential, Challenges and Outlook, Curr. Sustainable/Renewable Energy Rep., 2019, 6(4), 140–148, DOI:10.1007/s40518-019-00136-1.
- M. Martín, Challenges and Opportunities of Solar Thermal Energy towards a Sustainable Chemical Industry, Comput. Chem. Eng., 2022, 165, 107926, DOI:10.1016/j.compchemeng.2022.107926.
- IEA. Achieving Net Zero Heavy Industry Sectors in G7 Members, IEA: Paris, 2022 Search PubMed.
- D. S. Mallapragada, Y. Dvorkin, M. A. Modestino, D. V. Esposito, W. A. Smith, B.-M. Hodge, M. P. Harold, V. M. Donnelly, A. Nuz, C. Bloomquist, K. Baker, L. C. Grabow, Y. Yan, N. N. Rajput, R. L. Hartman, E. J. Biddinger, E. S. Aydil and A. D. Taylor, Decarbonization of the Chemical Industry through Electrification: Barriers and Opportunities, Joule, 2023, 7(1), 23–41, DOI:10.1016/j.joule.2022.12.008.
- I. Rattle, A. Gailani and P. G. Taylor, Decarbonisation Strategies in Industry: Going beyond Clusters, Sustainability Sci., 2024, 19(1), 105–123, DOI:10.1007/s11625-023-01313-4.
- ACS Green Chemistry Institute, Sustainable Separation Processes: A Roadmap to Accelerate Industrial Application of Less Energy-Intensive Alternative Separations, ACS, Washington, DC, 2019, https://www.acs.org/content/dam/acsorg/greenchemistry/redesign/roundtables/altsep-report-2019.pdf, accessed October 17, 2025 Search PubMed.
- A.SPIRE, Processes4Planet Strategic Research & Innovation Agenda (SRIA) Update, A.SPIRE/European Commission, Horizon Europe Partnership: Processes4Planet, Brussels, Belgium, 2024, https://www.aspire2050.eu/sites/default/files/pressoffice/publication/p4planet_sria_update_final_version_march_2024.pdf Search PubMed.
- U.S. Department of Energy, Industrial Efficiency & Decarbonization Office, Industrial Decarbonization Roadmap and Context, 2024, https://www.energy.gov/sites/default/files/2024-05/itiac-march2024-cresko-industrial-decarb-roadmap.pdf Search PubMed.
- M. Takht Ravanchi, T. Kaghazchi and A. Kargari, Application of Membrane Separation Processes in Petrochemical Industry: A Review, Desalination, 2009, 235(1), 199–244, DOI:10.1016/j.desal.2007.10.042.
- S. M. Dischinger, D. J. Miller, D. A. Vermaas and R. S. Kingsbury, Unifying the Conversation: Membrane Separation Performance in Energy, Water, and Industrial Applications, ACS ES&T Eng., 2024, 4(2), 277–289, DOI:10.1021/acsestengg.3c00475.
- M. Elimelech and W. A. Phillip, The Future of Seawater Desalination: Energy, Technology, and the Environment, Science, 2011, 333(6043), 712–717, DOI:10.1126/science.1200488.
- J. R. Werber, C. O. Osuji and M. Elimelech, Materials for Next-Generation Desalination and Water Purification Membranes, Nat. Rev. Mater., 2016, 1(5), 16018, DOI:10.1038/natrevmats.2016.18.
- M. Qasim, M. Badrelzaman, N. N. Darwish, N. A. Darwish and N. Hilal, Reverse Osmosis Desalination: A State-of-the-Art Review, Desalination, 2019, 459, 59–104, DOI:10.1016/j.desal.2019.02.008.
- O. E. Olufisayo and O. Olanrewaju, A Review of Renewable Energy Powered Seawater Desalination Treatment Process for Zero Waste, Water, 2024, 16(19), 2804, DOI:10.3390/w16192804.
- N. Ghaffour, T. M. Missimer and G. L. Amy, Technical Review and Evaluation of the Economics of Water Desalination: Current and Future Challenges for Better Water Supply Sustainability, Desalination, 2013, 309, 197–207, DOI:10.1016/j.desal.2012.10.015.
- D. Borah, G. Hazarika, A. Gogoi, S. Goswami, S. V. Sawake, D. Yadav, S. Karki, M. B. Gohain, L. R. Sahu and P. G. Ingole, Polymeric Membranes for Sustainable Gas Separation: A Comprehensive Review with Challenges, Innovations and Future Perspectives, Renewable Sustainable Energy Rev., 2025, 219, 115868, DOI:10.1016/j.rser.2025.115868.
- R. W. Baker and B. T. Low, Gas Separation Membrane Materials: A Perspective, Macromolecules, 2014, 47(20), 6999–7013, DOI:10.1021/ma501488s.
- A. L. Zydney, Continuous Downstream Processing for High Value Biological Products: A Review, Biotechnol. Bioeng., 2016, 113(3), 465–475, DOI:10.1002/bit.25695.
- A. C. Habert, C. P. Borges, F. d. A. Kronemberger, F. H. Conceição, R. NobregaMembrane Separation Processes, in Purification of Biotechnological Products, CRC Press, 2024 Search PubMed.
- L. R. Castilho, F. B. Anspach and W.-D. Deckwer, Comparison of Affinity Membranes for the Purification of Immunoglobulins, J. Membr. Sci., 2002, 207(2), 253–264, DOI:10.1016/S0376-7388(02)00257-0.
- R. Aguero, E. Bringas, M. F. S. Roman, I. Ortiz and R. Ibanez, Membrane Processes for Whey Proteins Separation and Purification. A Review, Curr. Org. Chem., 2017, 21(17), 1740–1752, DOI:10.2174/1385272820666160927122523.
- G. M. Geise, H. Lee, D. J. Miller, B. D. Freeman, J. E. McGrath and D. R. Paul, Water Purification by Membranes: The Role of Polymer Science, J. Polym. Sci., Part B:Polym. Phys., 2010, 48(15), 1685–1718, DOI:10.1002/polb.22037.
- L. R. Sahu, D. Yadav, D. Borah, A. Gogoi, S. Goswami, G. Hazarika, S. Karki, M. Borpatra Gohain, S. V. Sawake, S. V. Jadhav, S. Chatterjee and P. G. Ingole, Polymeric Membranes for Liquid Separation: Innovations in Materials, Fabrication, and Industrial Applications, Polymers, 2024, 16(23), 3240, DOI:10.3390/polym16233240.
- H. Zhou, J. R. Long and O. M. Yaghi, Introduction to Metal–Organic Frameworks, Chem. Rev., 2012, 112(2), 673–674, DOI:10.1021/cr300014x.
- D. Lowe, The 2025 Nobel Prize in Chemistry: Metal–Organic Frameworks, 2025, https://www.science.org/content/blog-post/2025-nobel-prize-chemistry-metal-organic-frameworks, accessed December 16, 2025 Search PubMed.
- F. Z. Zeggai, Z. Ait-Touchente, K. Bachari and A. Elaissari, Investigation of Metal-Organic Frameworks (MOFs): Synthesis, Properties, and Applications - An In-Depth Review, Chem. Phys. Impact, 2025, 10, 100864, DOI:10.1016/j.chphi.2025.100864.
- D. Aaron and C. Tsouris, Separation of CO2 from Flue Gas: A Review, Sep. Sci. Technol., 2005, 40(1–3), 321–348, DOI:10.1081/SS-200042244.
- F. Meng, Y. Meng, T. Ju, S. Han, L. Lin and J. Jiang, Research Progress of Aqueous Amine Solution for CO2 Capture: A Review, Renewable Sustainable Energy Rev., 2022, 168, 112902, DOI:10.1016/j.rser.2022.112902.
- A. D. Wiheeb, Z. Helwani, J. Kim and M. R. Othman, Pressure Swing Adsorption Technologies for Carbon Dioxide Capture, Separ. Purif. Rev., 2016, 45(2), 108–121, DOI:10.1080/15422119.2015.1047958.
- J. Z. Y. Tan, J. M. Uratani, S. Griffiths, J. M. Andresen and M. M. Maroto-Valer, Chemistry Advances Driving Industrial Carbon Capture Technologies, Nat. Rev. Chem., 2025, 9(10), 656–671, DOI:10.1038/s41570-025-00733-3.
- S. Zhong, X. Guo, A. Zhou, Z. Chen, D. Jin, M. Fan and T. Ma, Fundamentals and Recent Progress in Magnetic Field Assisted CO2 Capture and Conversion, Small, 2024, 20(5), 2305533, DOI:10.1002/smll.202305533.
- B. I. Z. Ahmad and P. J. Milner, Chemisorptive Carbon Capture Sees the Light, Trends Chem., 2025, 7(9), 529–540, DOI:10.1016/j.trechm.2025.07.002.
- T. Wilberforce, A. G. Olabi, E. T. Sayed, K. Elsaid and M. A. Abdelkareem, Progress in Carbon Capture Technologies, Sci. Total Environ., 2021, 761, 143203, DOI:10.1016/j.scitotenv.2020.143203.
- G. T. Rochelle, Amine Scrubbing for CO2 Capture, Science, 2009, 325(5948), 1652–1654, DOI:10.1126/science.1176731.
- E. S. Sanz-Pérez, C. R. Murdock, S. A. Didas and C. W. Jones, Direct Capture of CO2 from Ambient Air, Chem. Rev., 2016, 116(19), 11840–11876, DOI:10.1021/acs.chemrev.6b00173.
- A. C. Forse and P. J. Milner, New Chemistry for Enhanced Carbon Capture: Beyond Ammonium Carbamates, Chem. Sci., 2021, 12(2), 508–516, 10.1039/D0SC06059C.
- Y. Abdullatif, A. Sodiq, N. Mir, Y. Bicer, T. Al-Ansari, M. H. El-Naas and A. I. Amhamed, Emerging Trends in Direct Air Capture of CO2: A Review of Technology Options Targeting Net-Zero Emissions, RSC Adv., 2023, 13(9), 5687–5722, 10.1039/D2RA07940B.
- P. Panja, B. McPherson and M. Deo, Techno-Economic Analysis of Amine-Based CO2 Capture Technology: Hunter Plant Case Study, Carbon Capture Sci. Technol., 2022, 3, 100041, DOI:10.1016/j.ccst.2022.100041.
- T. Nguyen, M. Hilliard and G. T. Rochelle, Amine Volatility in CO2 Capture, Int. J. Greenh. Gas Control, 2010, 4(5), 707–715, DOI:10.1016/j.ijggc.2010.06.003.
- A. Krzemień, A. Więckol-Ryk, A. Smoliński, A. Koteras and L. Więcław-Solny, Assessing the Risk of Corrosion in Amine-Based CO2 Capture Process, J. Loss Prev. Process. Ind., 2016, 43, 189–197, DOI:10.1016/j.jlp.2016.05.020.
- U. I. Premadasa, B. Doughty, R. Custelcean and Y.-Z. Ma, Towards Energy-Efficient Direct Air Capture with Photochemically-Driven CO2 Release and Solvent Regeneration, ChemPlusChem, 2024, 89(10), e202300713, DOI:10.1002/cplu.202300713.
- C. J. Martin, G. Rapenne, T. Nakashima and T. Kawai, Recent Progress in Development of Photoacid Generators, J. Photochem. Photobiol., C, 2018, 34, 41–51, DOI:10.1016/j.jphotochemrev.2018.01.003.
- J. Saway, Z. M. Salem and J. J. Badillo, Recent Advances in Photoacid Catalysis for Organic Synthesis, Synthesis, 2021, 53(03), 489–497, DOI:10.1055/s-0040-1705952.
- N. Zivic, P. K. Kuroishi, F. Dumur, D. Gigmes, A. P. Dove and H. Sardon, Recent Advances and Challenges in the Design of Organic Photoacid and Photobase Generators for Polymerizations, Angew. Chem., Int. Ed., 2019, 58(31), 10410–10422, DOI:10.1002/anie.201810118.
- K. Adamczyk, M. Prémont-Schwarz, D. Pines, E. Pines and E. T. J. Nibbering, Real-Time Observation of Carbonic Acid Formation in Aqueous Solution, Science, 2009, 326(5960), 1690–1694, DOI:10.1126/science.1180060.
- R. Bennett, S. Clifford, K. Anderson and G. Puxty, Carbon Capture Powered by Solar Energy, Energy Procedia, 2017, 114, 1–6, DOI:10.1016/j.egypro.2017.03.1139.
- U. I. Premadasa, V. Bocharova, A. R. Miles, D. Stamberga, S. Belony, V. S. Bryantsev, A. Elgattar, Y. Liao, J. T. Damron, M. K. Kidder, B. Doughty, R. Custelcean and Y.-Z. Ma, Photochemically-Driven CO2 Release Using a Metastable-State Photoacid for Energy Efficient Direct Air Capture, Angew. Chem., Int. Ed., 2023, 62(29), e202304957, DOI:10.1002/anie.202304957.
- H. Seo, J. Schretter, M. Massen-Hane and T. A. Hatton, Visible Light-Driven CO2 Capture and Release Using Photoactive Pyranine in Water in Continuous Flow, J. Am. Chem. Soc., 2024, 146(39), 26777–26785, DOI:10.1021/jacs.4c07278.
- D. Cotton, T. Khuu, K. Takematsu, B. Delibas and J. M. Dawlaty, Photoinduced Carbon Dioxide Release via a Metastable Photoacid in a Nonaqueous Environment, J. Phys. Chem. Lett., 2024, 15(30), 7782–7787, DOI:10.1021/acs.jpclett.4c01577.
- T. G. Amundsen, L. E. Øi and D. A. Eimer, Density and Viscosity of Monoethanolamine + Water + Carbon Dioxide from (25 to 80) °C, J. Chem. Eng. Data, 2009, 54(11), 3096–3100, DOI:10.1021/je900188m.
- J. F. Swindells, J. R. Coe and T. B. Godfrey, Absolute Viscosity of Water at 20-Degrees-C, J. Res. Natl. Bur. Stand., 1952, 48(1), 1, DOI:10.6028/jres.048.001.
- F. de Meyer and C. Bignaud, The Use of Catalysis for Faster CO2 Absorption and Energy-Efficient Solvent Regeneration: An Industry-Focused Critical Review, Chem. Eng. J., 2022, 428, 131264, DOI:10.1016/j.cej.2021.131264.
- A. de Vries, K. Goloviznina, M. Reiter, M. Salanne and M. R. Lukatskaya, Solvation-Tuned Photoacid as a Stable Light-Driven pH Switch for CO2 Capture and Release, Chem. Mater., 2024, 36(3), 1308–1317, DOI:10.1021/acs.chemmater.3c02435.
- A. M. Alfaraidi, B. Kudisch, N. Ni, J. Thomas, T. Y. George, K. Rajabimoghadam, H. J. Jiang, D. G. Nocera, M. J. Aziz and R. Y. Liu, Reversible CO2 Capture and On-Demand Release by an Acidity-Matched Organic Photoswitch, J. Am. Chem. Soc., 2023, 145(49), 26720–26727, DOI:10.1021/jacs.3c08471.
- M. Purdy, A. Y. Wang, M. C. Drummer, D. G. Nocera and R. Y. Liu, Reversible Fluorenol Photobases That Perform CO2 Capture and Concentration from Ambient Air, Nat. Chem., 2025, 17, 1680–1687, DOI:10.1038/s41557-025-01901-0.
- J. D. Einkauf, D. Stamberga, N. Bhattacharjee, Y.-Z. Ma, V. S. Bryantsev and R. Custelcean, Photo-DAC: Light-Driven Ambient-Temperature Direct Air Capture by a Photobase, J. Am. Chem. Soc., 2026, 148(2), 2652–2659, DOI:10.1021/jacs.5c18831.
- D. Pines, J. Ditkovich, T. Mukra, Y. Miller, P. M. Kiefer, S. Daschakraborty, J. T. Hynes and E. Pines, How Acidic Is Carbonic Acid?, J. Phys. Chem. B, 2016, 120(9), 2440–2451, DOI:10.1021/acs.jpcb.5b12428.
- A. A. Beharry and G. A. Woolley, Azobenzene Photoswitches for Biomolecules, Chem. Soc. Rev., 2011, 40(8), 4422–4437, 10.1039/C1CS15023E.
- O. Bertrand and J.-F. Gohy, Photo-Responsive Polymers: Synthesis and Applications, Polym. Chem., 2017, 8(1), 52–73, 10.1039/C6PY01082B.
- I. Aprahamian, The Future of Molecular Machines, ACS Cent. Sci., 2020, 6(3), 347–358, DOI:10.1021/acscentsci.0c00064.
- D.-M. Xue, W.-J. Zhang, X.-Q. Liu, S.-C. Qi and L.-B. Sun, Fabrication of Azobenzene-Functionalized Porous Polymers for Selective CO2 Capture, Chin. J. Chem. Eng., 2022, 43, 24–30, DOI:10.1016/j.cjche.2021.10.029.
- Q. Huang, Z. Zhan, R. Sun, J. Mu, B. Tan and C. Wu, Light Triggered Pore Size Tuning in Photoswitching Covalent Triazine Frameworks for Low Energy CO2 Capture, Angew. Chem., 2023, 135(28), e202305500, DOI:10.1002/ange.202305500.
- Y. Zhu and W. Zhang, Reversible Tuning of Pore Size and CO2 Adsorption in Azobenzene Functionalized Porous Organic Polymers, Chem. Sci., 2014, 5(12), 4957–4961, 10.1039/C4SC02305F.
- R. Huang, M. R. Hill, R. Babarao and N. V. Medhekar, CO2 Adsorption in Azobenzene Functionalized Stimuli Responsive Metal–Organic Frameworks, J. Phys. Chem. C, 2016, 120(30), 16658–16667, DOI:10.1021/acs.jpcc.6b03541.
- Y. Jiang, X.-C. Shi, P. Tan, S.-C. Qi, C. Gu, T. Yang, S.-S. Peng, X.-Q. Liu and L.-B. Sun, Controllable CO2 Capture in Metal–Organic Frameworks: Making Targeted Active Sites Respond to Light, Ind. Eng. Chem. Res., 2020, 59(50), 21894–21900, DOI:10.1021/acs.iecr.0c04126.
- H. Zentou, B. Hoque, M. A. Abdalla, A. F. Saber, O. Y. Abdelaziz, M. Aliyu, A. M. Alkhedhair, A. J. Alabduly and M. M. Abdelnaby, Recent Advances and Challenges in Solid Sorbents for CO2 Capture, Carbon Capture Sci. Technol., 2025, 15, 100386, DOI:10.1016/j.ccst.2025.100386.
- R. Murakami, K. Shiota, A. Uchida and F. Inagaki, Light-Swing CO2 Capture: Photoirradiation-Based Chemical CO2 Release Based on Photoisomerization of Azobenzene-Amine/Guanidine Derivatives, Green Chem., 2024, 26(12), 7406–7413, 10.1039/D4GC00736K.
- J. Z. Y. Tan, J. M. Uratani, S. Griffiths, J. M. Andresen and M. M. Maroto-Valer, Chemistry Advances Driving Industrial Carbon Capture Technologies, Nat. Rev. Chem., 2025, 1–16, DOI:10.1038/s41570-025-00733-3.
- B. I. Z. Ahmad, K. Z. Colley, A. J. Musser and P. J. Milner, A Fully Light-Driven Approach to Separate Carbon Dioxide from Emission Streams, Chem, 2025, 102583, DOI:10.1016/j.chempr.2025.102583.
- Y. Masuda, N. Ishida and M. Murakami, Light-Driven Carboxylation of o-Alkylphenyl Ketones with CO2, J. Am. Chem. Soc., 2015, 137(44), 14063–14066, DOI:10.1021/jacs.5b10032.
- S. Paria, E. Carletti, M. Marcon, A. Cherubini-Celli, A. Mazzanti, M. Rancan, L. Dell'Amico, M. Bonchio and X. Companyó, Light-Triggered Catalytic Asymmetric Allylic Benzylation with Photogenerated C-Nucleophiles, J. Org. Chem., 2020, 85(6), 4463–4474, DOI:10.1021/acs.joc.0c00175.
- M. T. Blyth, B. B. Noble, I. C. Russell and M. L. Coote, Oriented Internal Electrostatic Fields Cooperatively Promote Ground- and Excited-State Reactivity: A Case Study in Photochemical CO2 Capture, J. Am. Chem. Soc., 2020, 142(1), 606–613, DOI:10.1021/jacs.9b12186.
- J. Y. J. Wang, M. T. Blyth, M. S. Sherburn and M. L. Coote, Tuning Photoenolization-Driven Cycloadditions Using Theory and Spectroscopy, J. Am. Chem. Soc., 2022, 144(2), 1023–1033, DOI:10.1021/jacs.1c12174.
- Y. Xu, X. Chen, W.-H. Fang and D. L. Phillips, pH- and Wavelength-Dependent Photodecarboxylation of Ketoprofen, Org. Lett., 2011, 13(20), 5472–5475, DOI:10.1021/ol202182k.
- L. Li, Y. Yao and N. Fu, Free Carboxylic Acids: The Trend of Radical Decarboxylative Functionalization, Eur. J. Org Chem., 2023, 26(21), e202300166, DOI:10.1002/ejoc.202300166.
- J. L. Fairey, J. R. Laszakovits, H. T. Pham, T. D. Do, S. D. Hodges, K. McNeill and D. G. Wahman, Chloronitramide Anion Is a Decomposition Product of Inorganic Chloramines, Science, 2024, 386(6724), 882–887, DOI:10.1126/science.adk6749.
- B. A. Moyer, R. Custelcean, B. P. Hay, J. L. Sessler, K. Bowman-James, V. W. Day and S.-O. Kang, A Case for Molecular Recognition in Nuclear Separations: Sulfate Separation from Nuclear Wastes, Inorg. Chem., 2013, 52(7), 3473–3490, DOI:10.1021/ic3016832.
- L. N. Pincus, H. E. Rudel, P. V. Petrović, S. Gupta, P. Westerhoff, C. L. Muhich and J. B. Zimmerman, Exploring the Mechanisms of Selectivity for Environmentally Significant Oxo-Anion Removal during Water Treatment: A Review of Common Competing Oxo-Anions and Tools for Quantifying Selective Adsorption, Environ. Sci. Technol., 2020, 54(16), 9769–9790, DOI:10.1021/acs.est.0c01666.
- Y. John, V. E. David Jr and D. Mmereki, A Comparative Study on Removal of Hazardous Anions from Water by Adsorption: A Review, Int. J. Chem. Eng., 2018, 2018(1), 3975948, DOI:10.1155/2018/3975948.
- I. Ravikumar and P. Ghosh, Recognition and Separation of Sulfate Anions, Chem. Soc. Rev., 2012, 41(8), 3077–3098, 10.1039/C2CS15293B.
- B. Shao, H. Fu and I. Aprahamian, A molecular anion pump, Science, 2024, 385, 544–549, DOI:10.1126/science.adp3506.
- Y. Li, M. Wang, F. Wang, S. Lu and X. Chen, Recent Progress in Studies of Photocages, Smart Mol., 2023, 1(1), e20220003, DOI:10.1002/smo.20220003.
- S. Xiong and Q. He, Photoresponsive Macrocycles for Selective Binding and Release of Sulfate, Chem. Commun., 2021, 57(99), 13514–13517, 10.1039/D1CC05506B.
- X. Chi, W. Cen, J. A. Queenan, L. Long, V. M. Lynch, N. M. Khashab and J. L. Sessler, Azobenzene-Bridged Expanded “Texas-Sized” Box: A Dual-Responsive Receptor for Aryl Dianion Encapsulation, J. Am. Chem. Soc., 2019, 141(16), 6468–6472, DOI:10.1021/jacs.9b01241.
- Z. Kokan and M. J. A. Chmielewski, Photoswitchable Heteroditopic Ion-Pair Receptor, J. Am. Chem. Soc., 2018, 140(47), 16010–16014, DOI:10.1021/jacs.8b08689.
- S. J. Wezenberg and B. L. Feringa, Photocontrol of Anion Binding Affinity to a Bis-Urea Receptor Derived from Stiff-Stilbene, Org. Lett., 2017, 19(2), 324–327, DOI:10.1021/acs.orglett.6b03423.
- S. J. Wezenberg, M. Vlatković, J. C. M. Kistemaker and B. L. Feringa, Multi-State Regulation of the Dihydrogen Phosphate Binding Affinity to a Light- and Heat-Responsive Bis-Urea Receptor, J. Am. Chem. Soc., 2014, 136(48), 16784–16787, DOI:10.1021/ja510700j.
- S. Lee and A. H. Flood, Photoresponsive Receptors for Binding and Releasing Anions, J. Phys. Org. Chem., 2013, 26(2), 79–86, DOI:10.1002/poc.2973.
- Y. Wang, F. Bie and H. Jiang, Controlling Binding Affinities for Anions by a Photoswitchable Foldamer, Org. Lett., 2010, 12(16), 3630–3633, DOI:10.1021/ol1014043.
- Y. Hua and A. H. Flood, Flipping the Switch on Chloride Concentrations with a Light-Active Foldamer, J. Am. Chem. Soc., 2010, 132(37), 12838–12840, DOI:10.1021/ja105793c.
- N. Bhattacharjee, A. Lutolli, J. D. Einkauf, Z. Zhang, A. R. Morgan, M. Pink, S. Jansone-Popova and A. H. Flood, Selective Binding and Light-Driven Release of Fluorous PF6– and Radioactive 99TcO4– Anions for All-to-Nothing Liquid–Liquid Extraction, J. Am. Chem. Soc., 2025, 147(18), 15707–15718, DOI:10.1021/jacs.5c03097.
- S. Lee, C.-H. Chen and A. H. Flood, A Pentagonal Cyanostar Macrocycle with Cyanostilbene CH Donors Binds Anions and Forms Dialkylphosphate [3]Rotaxanes, Nat. Chem., 2013, 5(8), 704–710, DOI:10.1038/nchem.1668.
- J. G. Darab and P. A. Smith, Chemistry of Technetium and Rhenium Species during Low-Level Radioactive Waste Vitrification, Chem. Mater., 1996, 8(5), 1004–1021, DOI:10.1021/cm950418+.
- J. A. Almohasin, J. Balag, V. G. Miral, R. V. Moreno, L. J. Tongco and E. C. R. Lopez, Green Solvents for Liquid–Liquid Extraction: Recent Advances and Future Trends, Eng. Proc., 2023, 56(1), 174, DOI:10.3390/ASEC2023-16278.
- Y.-Z. Ma, U. I. Premadasa, V. S. Bryantsev, A. R. Miles, I. N. Ivanov, A. Elgattar, Y. Liao and B. Doughty, Unravelling Photoisomerization Dynamics in a Metastable-State Photoacid, Phys. Chem. Chem. Phys., 2024, 26(5), 4062–4070, 10.1039/D3CP04454H.
- Y.-Z. Ma, U. I. Premadasa, M. Waters, N. Kumar, B. Doughty, M. Laud, Y. Liao and V. S. Bryantsev, Accessing Transient Isomers in the Photoreaction of Metastable-State Photoacid, ChemPhysChem, 2025, 26(13), e202500184, DOI:10.1002/cphc.202500184.
- J. Hernández, F. E. Curchod and B. F. Spectroscopic, Characterisation of Metastable Photoswitches for CO2 Capture and Release, Chem. Commun., 2025, 61, 17850–17853, 10.1039/D5CC04069H.
- A. De Vries, L. Rueben, P. Rehner, H. Schricker, L. Henz, H. Robin, A. Bardow and M. R. Lukatskaya, Light-Driven CO2 Capture with Photoacids from Concept to Scalable Technology, ChemRxiv, 2025 DOI:10.26434/chemrxiv-2025-q4h7c.
| This journal is © The Royal Society of Chemistry 2026 |