Open Access Article
Open Access ArticlePhotoinduced intramolecular S-α-C(sp3)–H functionalization enabled by an electron donor–acceptor complex-Mediated radical relay
Tiantian
Xu
,
Ran
Hong
,
Tong
Lu
,
Denghua
Zhu
,
Jiong
Yu
,
Min
Wang
 * and
Pinhua
Li
* and
Pinhua
Li
 *
*
College of Chemical and Environmental Engineering, Anhui Polytechnic University, Wuhu, Anhui 241000, P. R. China. E-mail: wangmin204@chnu.edu.cn; pphuali@126.com
First published on 10th March 2026
Abstract
We describe a consecutive reaction of 2-alkynylthioanisoles and 4-cyanopyridine enabled by EDA complex photoactivation, which facilitates the sequential S-α-C(sp3)–H bond activation, cyclization and radical coupling under mild conditions. A variety of 3-(4-pyridylbenzyl)benzothiophenes and benzoselenophenes were synthesized via a single-step process. Mechanistic investigations indicate that EDA complexes between thioethers and 4-cyanopyridines are crucial to this process.
Introduction
Sulfur-containing molecules are widely present in biologically active natural products, pharmaceutical compounds, and functional materials.1 The direct α-C(sp3)–H functionalization of sulfides has emerged as an atom-economical and environmentally benign strategy for the efficient construction of diverse sulfur-containing compounds.2 Compared to the activation of α-C(sp3)–H bonds adjacent to oxygen or nitrogen atoms, the functionalization of α-sulfur C(sp3)-H bonds is considerably more challenging due to the susceptibility of thioethers toward oxidation and their strong coordinating ability.3 Notably, significant progress has been achieved over the past decade in the direct α-C(sp3)–H functionalization of thioethers, primarily through methods such as catalysis by rare-earth complexes4 or alkali metal amides,5 electrochemical oxidation-enabled S-α-C(sp3)–H functionalization,6 and photoinduced S-α-C(sp3)–H activation and modification (Scheme 1a).7–12 The generation of α-thioalkyl radicals from thioethers can be accomplished via a photoinduced single-electron transfer (SET) oxidation/deprotonation sequence or through direct hydrogen-atom transfer (HAT).7 MacMillan's group has developed a selective α-C(sp3)–H alkylation of sulfides with alkyl bromides via the combination of photoredox, nickel and hydrogen-atom transfer (HAT) catalysis.8 In addition, Alfonzo and Hande disclosed an α-C(sp3)–H activation of thioethers to generate nucleophilic α-thioalkyl radicals through dual photoredox/weak base catalysis, followed by coupling with pyridinium salts or addition with electron deficient olefins.9 Recently, Feng and Liu have reported a photoinduced chemo-, site- and stereoselective α-C(sp3)–H functionalization of sulfides using isatins as the photoredox reagent and coupling partner catalyzed by a chiral gallium(III)–N,N′-dioxide complex.10 The same group subsequently developed an enantioselective radical hydrofunctionalization of α-substituted vinylphosphine oxides or α,β-unsaturated pyrazoleamide through photoactivation of α-C(sp3)–H of sulfides using naphthacene-6,11-quinone (NQ) as photocatalyst.11 Very recently, Sawama reported a highly chemoselective α-C(sp3)–H deuteration of sulfides using a thioxanthone or anthraquinone as photocatalyst and D2O as an inexpensive deuterium source under 390 nm irradiation.12 Despite these advances, the photocatalyst-free and HAT reagent-free strategies for direct α-C(sp3)–H modification of sulfides remain highly desirable and still not realized.In the past few years, the EDA complex photochemistry,13 especially the catalytic EDA complex strategy14 has attracted the interest of a growing number of chemists, and which has been recognized as a powerful approach for expanding the potential of photo-driven radical synthetic chemistry. The most straightforward synthetic application of EDA complex photoactivation is based on light-driven intermolecular charge transfer, which generates a radical ion pair and leads to the coupling of stoichiometric donor and acceptor substrates. The transformation could conduct in the absence of external photocatalysts with high atomic economy because the moieties of the substrates will eventually end up in the core of the corresponding products. Among the reported strategies, amine has been extensively employed as donor of the EDA complex and successfully applied in organic synthesis.15 In 2020, Xu and coworkers realized the visible-light-promoted N-α-C(sp3)–H alkylation through intermolecular charge transfer within a glycine Katritzky salt EDA complex, providing a simple method for preparation of unnatural α-amino acids and late-stage modification of peptides.16 Subsequently, the group has also disclosed a photo-promoted C(sp3)-H bicyclopentylation enabled by an electron donor–acceptor complex-mediated chemoselective three component radical relay (Scheme 1b).17
However, these seminal strategies are frequently found in amine-based EDA complexes,18 and to the best of our knowledge, using sulfides as donors in EDA complexes for direct α-C(sp3)–H functionalization has rarely reported.19 Based on our ongoing interest in organic photochemistry20 and inspired by recent progress on visible-light excited EDA complex-enabled selective α-C(sp3)–H functionalization, we hypothesized that an electron-poor 4-cyanopyridine and an electron-rich sulfide could generate an EDA complex. Excitation of such an EDA complex under visible-light irradiation would promote single-electron transfer to generate a radical ion pair intermediate, and which will undergo further transformation for new molecular construction. To our pleasure, we have achieved this assumed process. Herein, we will present a novel photoinduced cascade radical annulation of 2-alkynylthioanisoles with 4-cyanopyridines through S-α-C(sp3)–H activation enabled by an electron donor–acceptor complex−mediated radical relay, providing an efficient approach to synthesize 3-(4-pyridylbenzyl)benzothiophenes and benzoselenophenes which are difficult to obtain via traditional methods (Scheme 1c). This protocol features photocatalyst-free and HAT reagent-free, simple operation, broad scope and good yield. Moreover, late-stage modification of pharmaceutical derivatives further highlights the utility of this strategy.
Results and discussion
We initially investigated the cascade radical annulation using methyl(2-(phenylethynyl)phenyl)sulfane (1) and 4-cyano-2,6-lutidine (2) as the model substrate (Table 1). Unfortunately, the reaction of 1 and 2 could not happen under irradiation of 390 nm (40 W) LEDs without any additives in acetonitrile and nitrogen atmosphere (entry 1). To our pleasure, the desired product 3 was obtained in 11–50% yield after a suitable acid, such as trifluoroacetic acid (TFA), p-toluenesulfonic acid (TsOH) and racemic 1,1′-binaphthyl-2,2′-diylhydrogenphosphate (BNDHP) was added to the reaction system (entries 2–4). Regrettably, the racemic products were still obtained when a chiral phosphoric acid, (S)-3,3′-bis(2-naphthalenyl)-1,1′-binaphthyl-2,2′-diylhydrogen phosphate was involved in this transformation (entry 5). Given that a weak acid proved beneficial for this reaction, we employed acetic acid as the solvent, achieving the target product 3 in 65% yield (entry 6). However, the yield of the desired product was dramatically decreased when the reaction was performed in the mixture of acetonitrile and acetic acid (entry 7). Interestingly as comparations, the amount of acetic acid also has a significant impact on the yield of the desired product (entry 8). Control experiments revealed that the acid, light and reaction temperature were vital for this transformation (entries 9–11). Further screening of the wavelength and power of the LEDs, the reaction temperature, the ratio of 1 and 2, as well as the reaction times were optimized, as also shown in Table 1 (entries 12–15).| Entry | Solvent | Additives | Yieldb (%) |
|---|---|---|---|
| a Reaction conditions: 1 (0.20 mmol) and 2 (4-cyano-2,6-lutidine, 0.24 mmol) in solvent were irradiated with 390 nm Kessil LED (40 W) in a reaction tube under N2 atmosphere at 55–60 °C for 24 h unless otherwise stated. b Isolated yield. c 20% mol of (S)-3,3′-bis(2-naphthalenyl)-1,1′-binaphthyl-2,2′-diylhydrogen phosphate was used, 0%ee. d 2 mL CH3COOH was used. e 6 mL CH3COOH was used. f With a fan, the reaction temperature is about 35–40 °C. g dark. h 80 °C in the dark. i 390 nm (10 W). j 390 nm (20 W). k 390 nm (30 W). l 370 nm (40 W). m 427 nm (40 W). n 0.24 mmol 1 was used. o 0.22 mmol 1 was used. p 12 h. q 36 h; BNDHP = 1,1′-binaphthyl-2,2′-diylhydrogenphosphate (racemic), n.d. = not detected. | |||
| 1 | CH3CN | — | n.d |
| 2 | CH3CN | 20% mol TFA | 11 |
| 3 | CH3CN | 20% mol TsOH | 17 |
| 4 | CH3CN | 20% mol BNDHP | 51 |
| 5 | CH3CN | — | 50c |
| 6 | CH3COOH | — | 65 |
| 7 | CH3CN/CH3COOH (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
— | 23 |
| 8 | CH3COOH (2 mL or 6 mL) | — | 32d, 69e |
| 9 | CH3COOH | — | 53f |
| 10 | CH3COOH | — | n.dg |
| 11 | CH3COOH | — | n.dh |
| 12 | CH3COOH | — | 12 i, 21j, 32k |
| 13 | CH3COOH | — | 38l, 51m |
| 14 | CH3COOH | — | 78n, 78o |
| 15 | CH3COOH | — | 48p, 76q |
With the optimized conditions in hand, we next sought to investigate the scope of this EDA complex-promoted cascade radical annulation. As shown in Scheme 2a, a broad range of 2- alkynylthioanisoles with different groups on the aryl rings (R1) readily underwent annulation with 4-cyano-2,6-lutidine or 4-cyanopyridine, affording the desired products (3–21) in moderate to good yields, and the structure of 18 was further confirmed by X-ray single crystal analysis. Notably, the electronic effect and position of the substituents had a significant influence on the cascade radical annulation, both strong electron-donating group (OCH3) and strong electron-withdrawing substituent (CN) decrease the reaction efficiencies, and the ortho-substituted substrates also afford the desired products in lower yields, probably due to the steric hindrance. Moreover, when R1 was replaced with naphthyl and thiophenyl group, the two substrates could also be tolerated well, undergoing the tandem reaction smoothly to give desired products 22–24 in 45–53% yields. Fortunately, when R1 was an alkyl substituent, such as tert-butyl and n-butyl group, the corresponding products 25–27 were also obtained in good yields. Moreover, 2-alkynylselenoanisoles were also suitable substrates to deliver the desired products 28 and 29 in 40% and 30% yields, respectively. Additionally, the reaction could be easily scalable, and when the reaction was carried out on 4.8 mmol (1.08 g) scale, 3 was isolated in 60% yield. Unfortunately, 1-methoxy-2-(phenylethynyl)benzene and N,N-dimethyl-2-(phenylethynyl)aniline failed in this reaction under present conditions, no targeted products were observed, and the starting materials were quantitatively recovered.
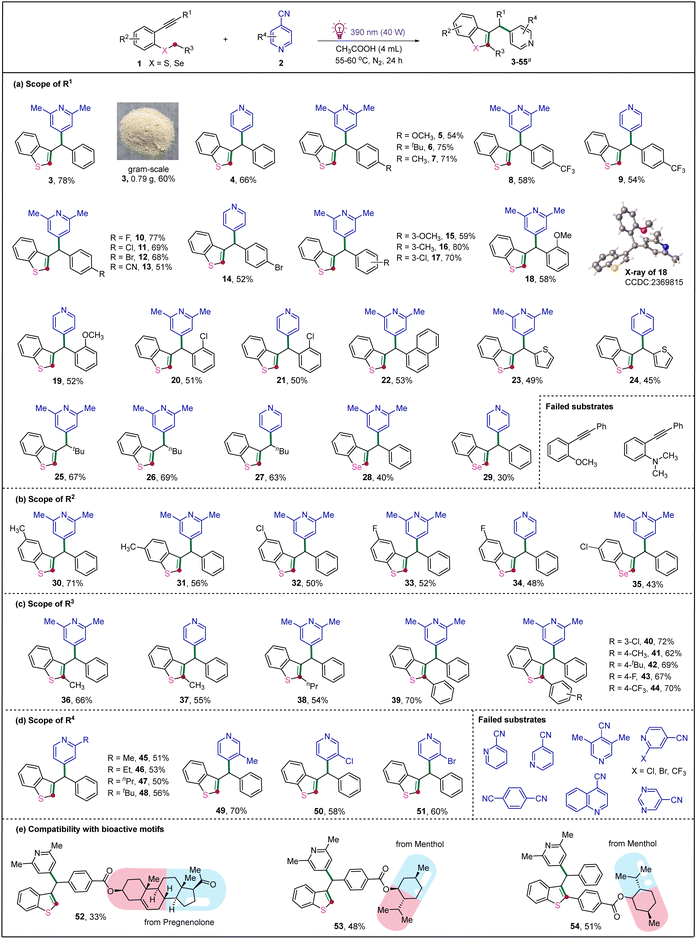 | ||
| Scheme 2 Substrate Scope. Reaction conditions: 1 (0.22 mmol), 2 (0.2 mmol), CH3COOH (4.0 mL), under N2 atmosphere, 55–60 °C, 390 nm Kessil lamp (40 W), 24 h, Isolated yield. | ||
Subsequently, different substituents (R2) on the aromatic ring of thioanisole were also investigated, both electronic and steric effects appear negligible in this transformation, affording the desired products 30–34 in 48–71% yields. Pleasingly, the substituted benzoselenophene 35 was also obtained in 43% yield under the optimized conditions (Scheme 2b).
Encouraged by our success with cascade radical annulation of 2-alkynylthioanisoles and 4-cyano-2,6-lutidine or 4-cyanopyridine, we further wondered whether this protocol could be applied to other 2-alkynylphenyl alkylsulfanes (Scheme 2c). We were delighted to find that a variety of 2-alkynylphenyl alkylsulfides, such as ethyl phenyl sulfide, butyl phenyl sulfide and benzyl phenyl sulfide, all underwent such an annulation to generate desired products 36–39 in moderate-to-good yield. Noteworthy, for benzyl phenyl sulfide, different kinds of substitutes on the phenyl ring of benzyl group, such as chloro, fluoro, trifluoromethyl, methyl and tert-butyl, did not significantly affect the reaction, providing the desired products 40–44 in 62–72% yields.
To further extend the reaction scope, we focused on evaluating substituted 4-cyanopyridines. As shown in Scheme 2d, 2-methyl, 2-ethyl, 2-npropyl and 2-tbutyl, 3-methyl, 3-chloro and 3-bromo substituted 4-cyanopyridine showed good compatibility with the reaction, delivering the corresponding products 45–51 in good yields (50−70%). To our surprise, 2,6-dimethylisonicotinonitrile, along with the 2-chloro, 2-bromo, and 2-CF3 substituted 4-cyanopyridines, did not undergo the desired transformation, and the starting materials were recovered. Unfortunately, 2-cyanopyridine and 3-cyanopyridine also failed to react under the current conditions, no desired products were detected, and the starting materials were recovered unchanged. Furthermore, when 1,4-dicyanobenzene, 4-cyanoquinoline, and 5-pyrimidinecarbonitrile were employed as substrates, no new products were observed and the starting materials were recovered in all cases. To demonstrate the utility of this EDA complex mediate S-α-C(sp3)–H activation strategy, the late-stage modification of segments of pharmaceutical molecule derivatives were also evaluated. The drug modified substrates, from pregnenolone and menthol, delivered the corresponding products 52–54 in 33–51% yields (Scheme 2e).
In order to gain a deeper mechanistic understanding of this annulation, a series of mechanistic studies were conducted. Firstly, we examined the ultraviolet-visible spectra of the reaction components and their mixture. As shown in Fig. 1a, both the appearance of a noticeable colour change and a bathochromic displacement absorption spectrum in the visible region were observed upon mixing 1 with 2 in the presence of acetic acid. Based on the UV-vis absorption of the EDA complex and the reaction conditions employed, we monitored the absorbance at 390 nm and plotted it as a function of the mole fraction of 2. The results confirmed the formation of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 complex between 1 and 2 (Fig. 1b). The association constant (Ka) was determined to be 1.74 M−1 by 1H NMR analysis in CD3COOD (Fig. 1c). On the other hand, the Stern–Volmer quenching experiment showed the exited state of 1 could be reductively quenched by 2, and the Stern–Volmer plot for 1 was fitted linearly with a quenching constant of Ksv = 5.22 × 10−3 M−1 (Fig. 1d and e). Notably, the fluorescence lifetime of 1 (τ1–0.8 ns) is not affected by the increasing concentration of 4-cyano-2,6-lutidine (2), which shows the static character of the quenching and further confirms the formation of an EDA complex (Fig. 1f). Moreover, NMR titration experiments revealed that upon complexation, the acceptor (2) exhibited upfield shifts in both its aliphatic and aromatic regions, while the donor (1) underwent corresponding downfield shifts (Fig. 1g). To elucidate the reaction mechanism, density functional theory (DFT) calculations were carried out to investigate the reaction pathway. Weak-interaction analysis of the most populated conformer revealed a pronounced interaction between the lone-pair electrons on the sulfur atom and the pyridinium ring (SI for details). All of these observations confirm an EDA complex of 1 and 2 was formed in this reaction system. In addition, the switching light experiment suggested that the continuous irradiation of visible light is needed (Fig. S5), and the quantum yield for the process was determined and calculated as Φ = 0.29, which suggested a photoinitiated chain transfer process could be excluded. It is worth noting that the isotope labeling experiments of the model reaction (Scheme 3a) revealed the 4-pyridylbenzyl hydrogen in the product 3 come from the reaction medium (acetic acid), ruling out the possibility of 1,3-HAT pathway to form the benzyl radical intermediate (IV′). Furthermore, to gain deeper insight into the rate-limiting step, we conducted a kinetic isotope effect (KIE) study of the reaction. The observed intermolecular competition KIE value of 1.8 suggests that deprotonation of the sulfur α-carbon is not the rate-determining step in this EDA-mediated transformation (Scheme 3b). Additionally, we found that radical scavengers, such as 2,2,6,6-tetramethyl-1-piperdinyloxy (TEMPO) and 2,6-ditert-butyl-4 methylphenol (BHT), inhibited the formation of the product (3), and the adducts of TEMPO or BHT with various radical species (A, A′, F, G) and the reaction intermediate D were all detected by high-resolution mass spectrometry (HRMS) analysis, demonstrating the radical intermediates in this cascade annulation process (Scheme 3c).
1 complex between 1 and 2 (Fig. 1b). The association constant (Ka) was determined to be 1.74 M−1 by 1H NMR analysis in CD3COOD (Fig. 1c). On the other hand, the Stern–Volmer quenching experiment showed the exited state of 1 could be reductively quenched by 2, and the Stern–Volmer plot for 1 was fitted linearly with a quenching constant of Ksv = 5.22 × 10−3 M−1 (Fig. 1d and e). Notably, the fluorescence lifetime of 1 (τ1–0.8 ns) is not affected by the increasing concentration of 4-cyano-2,6-lutidine (2), which shows the static character of the quenching and further confirms the formation of an EDA complex (Fig. 1f). Moreover, NMR titration experiments revealed that upon complexation, the acceptor (2) exhibited upfield shifts in both its aliphatic and aromatic regions, while the donor (1) underwent corresponding downfield shifts (Fig. 1g). To elucidate the reaction mechanism, density functional theory (DFT) calculations were carried out to investigate the reaction pathway. Weak-interaction analysis of the most populated conformer revealed a pronounced interaction between the lone-pair electrons on the sulfur atom and the pyridinium ring (SI for details). All of these observations confirm an EDA complex of 1 and 2 was formed in this reaction system. In addition, the switching light experiment suggested that the continuous irradiation of visible light is needed (Fig. S5), and the quantum yield for the process was determined and calculated as Φ = 0.29, which suggested a photoinitiated chain transfer process could be excluded. It is worth noting that the isotope labeling experiments of the model reaction (Scheme 3a) revealed the 4-pyridylbenzyl hydrogen in the product 3 come from the reaction medium (acetic acid), ruling out the possibility of 1,3-HAT pathway to form the benzyl radical intermediate (IV′). Furthermore, to gain deeper insight into the rate-limiting step, we conducted a kinetic isotope effect (KIE) study of the reaction. The observed intermolecular competition KIE value of 1.8 suggests that deprotonation of the sulfur α-carbon is not the rate-determining step in this EDA-mediated transformation (Scheme 3b). Additionally, we found that radical scavengers, such as 2,2,6,6-tetramethyl-1-piperdinyloxy (TEMPO) and 2,6-ditert-butyl-4 methylphenol (BHT), inhibited the formation of the product (3), and the adducts of TEMPO or BHT with various radical species (A, A′, F, G) and the reaction intermediate D were all detected by high-resolution mass spectrometry (HRMS) analysis, demonstrating the radical intermediates in this cascade annulation process (Scheme 3c).
 | ||
| Scheme 3 (a) Deuteration experiment; (b) KIE experiment; (c) radical capture experiment; (d) proposed reaction mechanism. | ||
Based on the results of experimental investigations, we considered that the formation and excitation of an EDA complex of 1 and 2 are involved in the reaction, and a reasonable mechanism of this transformation was proposed, as shown in Scheme 3d. Initially, the interaction between methyl(2-(phenylethynyl)phenyl)sulfane (1) and 4-cyano-2,6-lutidine (2) with the assistant of acetic acid, resulting the formation of an EDA complex. Next, excitation of such an EDA complex under visible-light irradiation accelerates the single-electron transfer, leading to the formation of radical ion pairs, a sulfide radical cation (I) and a pyridinium radical anion (II). Then, the intermediate I undergoes deprotonation to deliver a S-α-alkyl radical III, which then generate the vinyl radical intermediate VI through intramolecular radical addition. Under acidic conditions, II equilibrates with V. Subsequently, the radical−radical cross coupling of VI and V producing intermediate VI, which simultaneously eliminating a HCN to give the intermediates VII, which is then protonated to obtain a cation intermediate VIII, and ultimately yield the desired product 3 by deprotonation.
Conclusions
In summary, our study presents an efficient and mild strategy for the construction of various 3-(4-pyridylbenzyl)benzothiophenes and benzoselenophenes from 2-alkynylthioanisoles and their derivatives using substituted 4-cyanopyridines as pyridination reagents under mild conditions. This photochemical protocol features photocatalyst-free and HAT reagent-free, broad substrate scope, good yields, high step and atom economy, transition-metal-free reaction conditions. Notably, the late-stage modification of pharmaceutical derivatives highlights the potential application of this strategy. Mechanistically, visible-light induced the intermolecular charge transfer within the EDA complex of 2-alkynylthioanisole and 4-cyano-2,6-lutidine enabled the single electron transfer (SET), accomplishing the sequential reactions of selective S-α-C(sp3)–H bond activation, cyclization and radical coupling. Ongoing researches including further mechanism and the new applications of this photo-induced EDA complex mediated transformation are currently underway in our laboratory.Author contributions
P. L. and M. W. conceived and supervised the project, and edited the manuscript. T. X. designed and carried out the experiments, analyzed the data, and prepared the manuscript. R. H. performed the computational studies presented in this paper. T. L. and D. Z. provided valuable assistance with the synthesis of compounds. J. Y. contributed to experimental testing and analysis. All authors discussed the results and commented on the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6sc01281g.Acknowledgements
We gratefully acknowledge the Young Talent Key Project of Anhui Province (No. 170808J02), the University Synergy Innovation Program of Anhui Province (No. GXXT-2020-078) and the Research Fund of Anhui Polytechnic University (No. 2023YQQ0001, S022024011) for financial support of this work.Notes and references
- (a) C. Le, Y. Liang, R. W. Evans, X. Li and D. W. C. MacMillan, Construction of Sulfur-Containing Moieties in the Total Synthesis of Natural Products, Nat. Prod. Rep., 2020, 37, 246–275 RSC; (b) F, B. T, S. H. Liang and X. Jiang, Sulfur Containing Scaffolds in Drugs: Synthesis and Application in Medicinal Chemistry, Curr. Top. Med. Chem., 2016, 16, 1200–1216 CrossRef PubMed; (c) L. Bai and X. Jiang, Smelless/Stable/Sustainable Sulfur Chemistry, CCS Chem., 2025, 7, 1889–1902 CrossRef CAS; (d) Y. Zhou, Z. Zhu, K. Zhang and B. Yang, Molecular Structure and Properties of Sulfur-Containing High Refractive Index Polymer Optical Materials, Macromol. Rapid Commun., 2023, 44, 2300411 CrossRef CAS PubMed; (e) P. Sang, Q. Chen, D.-Y. Wang, W. Guo and Y. Fu, Organosulfur Materials for Rechargeable Batteries: Structure, Mechanism, and Application, Chem. Rev., 2023, 123, 1262–1326 CrossRef CAS PubMed.
- (a) A. M. F. Phillips and A. J. L. Pombeiro, Recent Developments in Transition Metal-Catalyzed Cross-Dehydrogenative Coupling Reactions of Ethers and Thioethers, ChemCatChem, 2018, 10, 3354–3383 CrossRef; (b) L. Tang, Q. Hu, K. Yang, M. Elsaid, C. Liu and H. Ge, Recent Advances in Direct α-C(sp3)-H Bond Functionalization of Thioethers, Green Synth. Catal., 2022, 3, 203–211 Search PubMed; (c) N. S. Shlapakov, A. D. Kobelev, J. V. Burykina, Y.-Z. Cheng, S.-L. You and V. P. Ananikov, Sulfur in Waste-Free Sustainable Synthesis: Advancing Carbon-Carbon Coupling Techniques, Angew. Chem., Int. Ed., 2024, 63, e202402109 CrossRef CAS PubMed; (d) S. R. Atriardi, Y. Anita and S. K. Woo, Harnessing Photoredox and Weak Brønsted Base Dual Catalysis for Selective C(sp3)-H Bond Activation, Synthesis, 2025, 57, 953–964 CrossRef CAS; (e) N. Seling, M. Atobe, K. Kasten, J. D. Firth, P. B. Karadakov, F. W. Goldberg and P. O'Brien, α-Functionalisation of Cyclic Sulfides Enabled by Lithiation Trapping, Angew. Chem., Int. Ed., 2024, 63, e202314423 CrossRef CAS PubMed.
- (a) T. G. Saint-Denis, N. Y. S. Lam, Ni. Chekshin, P. F. Richardson, J. S. Chen, J. Elleraas, K. D. Hesp, D. C. Schmitt, Y. Lian, C. W. Huh and J.-Q. Yu, Mechanistic Study of Enantioselective Pd-Catalyzed C(sp3)-H Activation of Thioethers Involving Two Distinct Stereomodels, ACS Catal., 2021, 11, 9738–9753 CrossRef CAS PubMed; (b) L. Jin, P. Zhang, Y. Li, X. Yu and B.-F. Shi, Atroposelective Synthesis of Conjugated Diene-Based Axially Chiral Styrenes via Pd(II)-Catalyzed Thioether-Directed Alkenyl C−H Olefination, J. Am. Chem. Soc., 2021, 143, 12335–12344 CrossRef CAS PubMed; (c) B. Zhao, G. B. Hammond and B. Xu, Modulation of photochemical oxidation of thioethers to sulfoxides or sulfones using an aromatic ketone as the photocatalyst, Tetrahedron Lett., 2021, 82, 153376 CrossRef CAS.
- (a) Y. Luo, H.-L. Teng, C. Xue, M. Nishiura and Z. Hou, Yttrium-Catalyzed Regioselective α-C-H Silylation of Methyl Sulfides with Hydrosilanes, ACS Catal., 2018, 8, 8027–8032 CrossRef CAS; (b) Y. Luo, Y. Ma and Z. Hou, α-C-H Alkylation of Methyl Sulfides with Alkenes by a Scandium Catalyst, J. Am. Chem. Soc., 2018, 140, 114–117 CrossRef CAS PubMed.
- (a) Y.-F. Liu, L. Zheng, D.-D. Zhai, X.-Y. Zhang and B.-T. Guan, Dimeric Potassium Amide-Catalyzed α-Alkylation of Benzyl Sulfides and 1,3-Dithianes, Org. Lett., 2019, 21, 5351–5356 CrossRef CAS PubMed; (b) H.-Z. Du and B.-T. Guan, Alkali Metal Amide-Catalyzed α-Deuteration of Sulfides, Org. Lett., 2024, 26, 10458–10463 CrossRef CAS PubMed.
- (a) H. Wang, M. He, Y. Li, H. Zhang, D. Yang, M. Nagasaka, Z. Lv, Z. Guan, Y. Cao, F. Gong, Z. Zhou, J. Zhu, S. Samanta, A. D. Chowdhury and A. Lei, Electrochemical Oxidation Enables Regioselective and Scalable α-C(sp3)-H Acyloxylation of Sulfides, J. Am. Chem. Soc., 2021, 143, 3628–3637 CrossRef CAS PubMed; (b) H. Wang, M. Yua, P. Zhang, H. Wan, H. Cong and A. Lei, Electrochemical dual-oxidation strategy enables access to a-chlorosulfoxides from sulfides, Sci. Bull., 2022, 67, 79–84 CrossRef CAS PubMed.
- A. S. Anderton, O. J. Knowles, J. A. Rossi-Ashton and D. J. Procter, Flavin-Mediated Photocatalysis Providesa General Platform for Sulfide C−H Functionalization, ACS Catal., 2024, 14, 2395–2401 CrossRef CAS PubMed.
- (a) C. Le, Y. Liang, R. W. Evans, X. Li and D. W. C. MacMillan, Selective sp3 C-H Alkylation via Polarity-Match Based Cross-Coupling, Nature, 2017, 547, 79–83 CrossRef CAS PubMed; (b) J. Kim, B. X. Li, R. Y.-C. Huang, J. X. Qiao, W. R. Ewing and D. W. C. MacMillan, Site-Selective Functionalization of Methionine Residues via Photoredox Catalysis, J. Am. Chem. Soc., 2020, 142, 21260–21266 CrossRef CAS PubMed.
- (a) E. Alfonzo and S. M. Hande, Photoredox and Weak Brønsted Base Dual Catalysis: Alkylation of α-Thio Alkyl Radicals, ACS Catal., 2020, 10, 12590–12595 CrossRef CAS; (b) E. Alfonzo and S. M. Hande, α-Heteroarylation of Thioethers via Photoredox and Weak Brønsted Base Catalysis, Org. Lett., 2021, 23, 6115–6120 CrossRef CAS PubMed.
- Z. Tan, S. Zhu, Y. Liu and X. Feng, Photoinduced Chemo-, Site- and Stereoselective α-C(sp3)-H Functionalization of Sulfides, Angew. Chem., Int. Ed., 2022, 61, e202203374 CrossRef CAS PubMed.
- (a) Z. Tan, Y. Liu and X. Feng, Photoredox-Catalyzed C(sp3)-H Radical Functionalization to Enable Asymmetric Synthesis of α- Chiral Alkyl Phosphine, Sci. Adv., 2024, 10, eadn9738 CrossRef CAS PubMed; (b) Y. Luo, Y. Zhou, F. Xiao, X. He, Z. Zhong, Q.-L. Zhou, W. Cao, X. Liu and X. Feng, Quinone-Initiated Photocatalytic Enantioselective Giese Radical Addition with Ethers, Thioethers, Amines, and Alkanes, ACS Catal., 2024, 14, 12031–12041 CrossRef CAS.
- R. Ogasahara, M. Mae, Y. Itabashi, K. Ohkubo, K. Matsuura, H. Shimizu, K. Ban, M. Togami, T. Udagawa, H. Fujioka, M. Kamiya, S. Akai and Y. Sawama, Photocatalytic and Chemoselective H/D Exchange at α-Thio C(sp3)-H Bonds, J. Am. Chem. Soc., 2025, 147, 15499–15509 CrossRef CAS PubMed.
- (a) G. E. M. Crisenza, D. Mazzarella and P. Melchiorre, Synthetic Methods Driven by the Photoactivity of Electron Donor-Acceptor Complexes, J. Am. Chem. Soc., 2020, 142, 5461–5476 CrossRef CAS PubMed; (b) H. Li, Y. Liu and S. Chiba, Leveraging of Sulfur Anions in Photoinduced Molecular Transformations, JACS Au, 2021, 1, 2121–2129 CrossRef CAS PubMed; (c) Z. Yang, Y. Liu, K. Cao, X. Zhang, H. Jiang and J. Li, Synthetic Reactions Driven by Electron-Donor–Acceptor (EDA) Complexes, Beilstein J. Org. Chem., 2021, 17, 771–799 CrossRef CAS PubMed; (d) Y. Sumida and H. Ohmiya, Direct Excitation Strategy for Radical Generation in Organic Synthesis, Chem. Soc. Rev., 2021, 50, 6320–6332 RSC; (e) S. Wang, H. Wang and B. König, Light-Induced Single-Electron Transfer Processes involving Sulfur Anions as Catalysts, J. Am. Chem. Soc., 2021, 143, 15530–15537 CrossRef CAS PubMed; (f) T. Tasnim, M. J. Ayodele and S. P. Pitre, Recent Advances in Employing Catalytic Donors and Acceptors in Electron Donor-Acceptor Complex Photochemistry, J. Org. Chem., 2022, 87, 10555–10563 CrossRef CAS PubMed; (g) B. Saxena, R. Patel and A. Sharma, Recent Advances in Electron Donor-Acceptor (EDA)-Complex Reactions involving Quaternary Pyridinium Derivatives, Adv. Synth. Catal., 2023, 365, 1538–1564 CrossRef CAS.
- (a) Z.-Y. Cao, T. Ghosh and P. Melchiorre, Enantioselective Radical Conjugate Additions Driven by a Photoactive Intramolecular Iminium-Ion-Based EDA Complex, Nat. Commun., 2018, 9, 3274 CrossRef PubMed; (b) E. de Pedro Beato, D. Spinnato, W. Zhou and P. Melchiorre, A General Organocatalytic System for Electron Donor-Acceptor Complex Photoactivation and Its Use in Radical Processes, J. Am. Chem. Soc., 2021, 143, 12304–12314 CrossRef CAS PubMed; (c) E. Le Saux, M. Zanini and P. Melchiorre, Photochemical Organocatalytic Benzylation of Allylic C-H Bonds, J. Am. Chem. Soc., 2022, 144, 1113–1118 CrossRef CAS PubMed; (d) D. J. Castillo-Pazos, J. D. Lasso, E. Hamzehpoor, J. Ramos-Sánchez, J. M. Salgado, G. Cosa, D. F. Perepichka and C.-J. Li, Triarylamines as Catalytic Donors in Light-Mediated Electron Donor-Acceptor Complexes, Chem. Sci., 2023, 14, 3470–3481 RSC; (e) K. Devi, A. Shehzad and M. P. Wiesenfeldt, Organophotocatalytic Reduction of Benzenes to Cyclohexenes, J. Am. Chem. Soc., 2024, 146, 34304–34310 CrossRef CAS PubMed; (f) Y.-X. Jiang, L.-L. Liao, T.-Y. Gao, W.-H. Xu, W. Zhang, L. Song, G.-Q. Sun, J.-H. Ye, Y. Lan and D.-G. Yu, Visible-Light-Driven Synthesis of N-Heteroaromatic Carboxylic Acids by Thiolate-Catalysed Carboxylation of C(sp2)–H Bonds Using CO2, Nat. Synth., 2024, 3, 394–405 CrossRef CAS; (g) K. Almagambetova, K. Murugesan and M. Rueping, Transition-Metaland Photocatalyst-Free, Redox-Neutral Heteroarylation of C(sp3)-H Bonds, ACS Catal., 2024, 14, 12664–12670 CrossRef CAS.
- (a) D. Xie, W. Zeng, J. Yang and X. Ma, Visible-Light-Promoted Direct Desulfurization of Glycosyl Thiols to Access C-Glycosides, Nat. Commun., 2024, 15, 9187 CrossRef CAS PubMed; (b) J. Wei, J. Meng, C. Zhang, Y. Liu and N. Jiao, Dioxygen Compatible Electron Donor Acceptor Catalytic System and Its Enabled Aerobic Oxygenation, Nat. Commun., 2024, 15, 1886 CrossRef CAS PubMed; (c) K. Sachidanandan, C. Stenftenagel, A. M. Cluff, G. A. McAlary, A. Joshy, B. Niu and S. Laulhé, Sequential EDA/HAT Enables C(sp3)-C(sp3) Cross-Dehydrogenative Coupling: Accessing Non-Canonical Amino Acids with Quaternary Carbon Centers from Activated and Non-Activated Alkanes, Org. Lett., 2025, 27, 5619–5624 CrossRef CAS PubMed; (d) Y. Huo, B. Chen, Z. Xu, X. Ren, R. Qia and C. Wang, Visible-Light-Driven C(sp3)-H Alkylation of Heterobenzylic Amines via Electron Donor-Acceptor Complexes, Org. Chem. Front., 2024, 11, 205–210 RSC; (e) X. Xie, K. Qiao, B.-R. Shao, W. Jiang and L. Shi, Visible-Light-Promoted Radical Cross-Dehydrogenative Coupling of C(sp3)-H/C(sp3)-H and C(sp3)-H/C(sp2)-H via Intermolecular HAT, Org. Lett., 2023, 25, 4264–4269 CrossRef CAS PubMed.
- C. Wang, R. Qi, H. Xue, Y. Shen, M. Chang, Y. Chen, R. Wang and Z. Xu, Visible-Light-Promoted C(sp3)-H Alkylation by Intermolecular Charge Transfer: Preparation of Unnatural a-Amino Acids and Late-Stage Modification of Peptides, Angew. Chem., Int. Ed., 2020, 59, 7461–7466 CrossRef CAS PubMed.
- X. Dang, Z. Li, J. Shang, C. Zhang, C. Wang and Z. Xu, Photoinduced C(sp3)-H Bicyclopentylation Enabled by an Electron Donor-Acceptor Complex-Mediated Chemoselective Three-Component Radical Relay, Angew. Chem., Int. Ed., 2024, 63, e202400494 CrossRef CAS PubMed.
- (a) A. Dewanji, L. van Dalsen, J. A. Rossi-Ashton, E. Gasson, G. E. M. Crisenza and D. J. Procter, A General Arene C-H Functionalization Strategy via Electron Donor-Acceptor Complex Photoactivation, Nat. Chem., 2023, 15, 43–52 CrossRef CAS PubMed; (b) J. D. Lasso, D. J. Castillo-Pazos, J. M. Salgado, C. Ruchlin, L. Lefebvre, D. Farajat, D. F. Perepichka and C.-J. Li, A General Platform for Visible Light Sulfonylation Reactions Enabled by Catalytic Triarylamine EDA Complexes, J. Am. Chem. Soc., 2024, 146, 2583–2592 CrossRef CAS PubMed; (c) Y. Wang, Y. Xu, X. Qi, L.-C. Wang, C. Xu, G. Huang and X.-F. Wu, Amines Tuned Controllable Carbonylation for the Synthesis of γ-Lactones and 1,4-Diones, Nat. Commun., 2025, 16, 6305 CrossRef CAS PubMed.
- P. K. Roy and Y. Hitomi, A Simple Method for the Formation of α-Thioalkyl Radicals and the Promotion of Carbon-Carbon Bond Formation by Photoirradiation of Electron Donor-Acceptor Complexes, Asian J. Org. Chem., 2023, 12, e202300378 CrossRef CAS.
- (a) T. Xu, F.-D. Wang, W.-C. Yang, T. Lu, M. Wang and P. Li, Photo-Induced Intramolecular Alkyl/Aryl Group Transfer and SO2 Insertion: A New Strategy For the Synthesis of 3-(Alkyl/Arylsulfonyl)Benzothiophenes, Green Chem., 2025, 27, 2386–2391 RSC; (b) H. Yan, F.-D. Wang, M. Wang, L. Ye and P. Li, Photoinduced Radical Cyclization of 2-Alkynylthioanisoles with Disulfides without an External Photocatalyst, J. Org. Chem., 2023, 88, 15288–15297 CrossRef CAS PubMed; (c) K. Yan, X. Yang, J. Gao, Z. Zhang and P. Li, Visible-Light-Induced Cyclization of 2-Alkenyl-1,1’Biphenyls, Org. Chem. Front., 2023, 10, 2453–2458 RSC; (d) F.-D. Wang, J. Jiang, T. Xu, W.-C. Yang, S.-P. Zhang, M. Wang and P. Li, Synthesis of Acylbenzo[b]thiophenes, Benzofurans and Indoles via Intramolecular Oxidative Cyclization Enabled by Photocatalytic Hydrogen Atom Transfer (HAT), Tetrahedron Chem., 2025, 13, 100119 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |