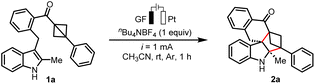Open Access Article
Open Access ArticleElectrochemically driven strain-release dearomative (3 + 2) cyclization for the synthesis of bicyclo[2.1.1]hexane-fused polycyclic spiroindolines
Yanren
Zhu
,
Enfan
Pu
,
Shaoxiong
Yang
,
Dongbo
Zhao
 ,
Qi
Xu
,
Piaopiao
Jiang
,
Xiong
Li
,
Hongbin
Zhang
,
Qi
Xu
,
Piaopiao
Jiang
,
Xiong
Li
,
Hongbin
Zhang
 * and
Jingbo
Chen
* and
Jingbo
Chen
 *
*
Key Laboratory of Medicinal Chemistry for Natural Resource, Ministry of Education, Yunnan Key Laboratory of Cross-Border Infectious Disease Control and Prevention and Novel Drug Development, School of Chemical Science and Technology, School of Pharmacy, Yunnan University, Kunming 650500, China. E-mail: zhanghb@ynu.edu.cn; chenjb@ynu.edu.cn
First published on 21st April 2026
Abstract
Spiroindolines are ubiquitous structural motifs in natural products and biologically active compounds, making the development of reliable synthetic methods for their construction highly important to the synthetic chemistry community. Moreover, the synthesis of polycyclic compounds featuring C(sp3)-rich and complex three-dimensional (3D) architectures and multiple contiguous quaternary carbon centers in a single step remains a formidable challenge. Herein, we report an efficient and environmentally friendly electrochemical strategy for a dearomative (3 + 2) cyclization reaction of C3-bicyclo[1.1.0]butane (BCB)-substituted indoles. This method enables the efficient assembly of structurally complex and novel bicyclo[2.1.1]hexane (BCH)-fused polycyclic spiroindolines in high yields, featuring four contiguous stereogenic centers, two of which are spirocyclic all-carbon quaternary carbon centers. This new methodology provides a series of valuable spiroindolines under mild reaction conditions in a practical and atom-economic manner without the need for external oxidants, and the products can be further transformed into more complex spiroindolines. Notably, these C(sp3)-rich products exhibit significant potential in the field of medicinal chemistry.
Introduction
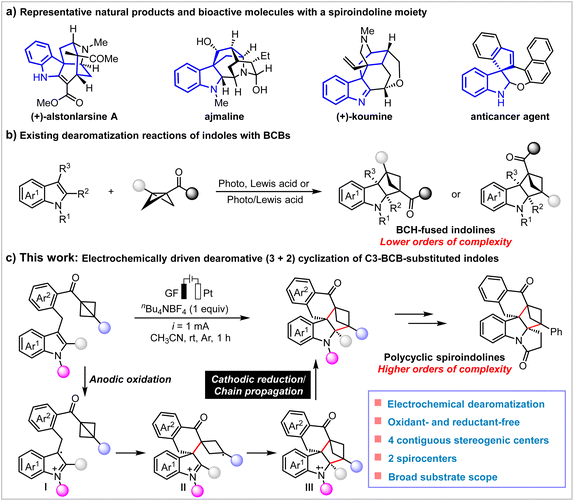
Dearomatization represents a powerful strategy for the efficient construction of sp3-rich, three-dimensional, and structurally complex polycyclic architectures from readily accessible aromatic feedstocks.1–4 Owing to the broad spectrum of biological activities associated with indole derivatives,5,6 the development of indole dearomatization reactions has emerged as an area of significant interest in synthetic chemistry.7 In particular, spiroindoline derivatives are privileged and valuable scaffolds that are prevalent in a great number of natural products and biologically active molecules, such as (+)-alstonlarsine A,8,9 ajmaline,10 (+)-koumine11,12 and anticancer agent (Scheme 1a).13 In drug structure optimization, the introduction of spirocycles and cyclobutane moieties significantly enhances the three-dimensional character of molecules and restricts bond stretching and rotation within molecular conformations, thereby stabilizing the presentation of a single pharmacophore conformation upon target binding, mediating desired pharmacological effects, and substantially reducing off-target effects and toxic side effects.14–19 Over the past decade, remarkable progress has been made in indole dearomative spiroannulation, driven primarily by transition-metal catalysis and visible-light catalysis,7,20,21 thereby enabling the efficient construction of spiroindolines and spiroindolenines. Notably, Tang and co-workers reported the preparation of a class of tetracyclic cyclopenta-fused spiroindoline skeletons through Cu(II)-catalyzed intramolecular (3 + 2) annulation reactions of donor–acceptor cyclopropanes with indoles.22 However, the single-step synthesis of polycyclic spiroindolines with C(sp3)-rich complex three-dimensional (3D) structures and multiple contiguous quaternary carbon centers represents the most rapid and efficient approach, yet it remains a formidable challenge.In recent years, bicyclo[1.1.0]butanes (BCBs) have gained widespread recognition as useful synthetic tools in (3 + n) cycloaddition reactions,23–38 enabling the formation of C(sp3)-rich bicyclic hydrocarbons that serve as bioisosteres of aromatic rings in medicinal chemistry, due to their improved pharmacokinetic and physicochemical properties.39–42 Benefiting from their distinctive ring strain and unique σ-bond framework, numerous BCB-based cycloaddition reactions have been developed, demonstrating exceptional reactivity and broad application potential in both polar and radical reaction systems. Among them, BCBs have been employed in the dearomatization cyclization reactions of indoles to access BCH-fused indolines. In 2022, Glorius et al. reported a representative photochemical [2π + 2σ] cycloaddition between indoles and monosubstituted BCBs, leading to the formation of BCHs with limited substrate scope.43 In 2023, Deng and co-workers developed a Yb(OTf)3 catalyzed intermolecular atom-economical cycloaddition of indoles with BCBs to provide a direct construction of polycyclic indoline skeletons.44 In the same year, Feng et al. developed a silver-catalyzed dearomative [2π + 2σ] cycloaddition of N-unprotected 2-substituted indoles and bicyclobutanes for the synthesis of indoline fused BCHs with opposite regioselectivity compared to the classical pathway.45 Very recently, Dong and coworkers investigated photocycloadditions of indole derivatives with monosubstituted BCBs by photoinduced chiral N,N′-dioxide/metal complex Lewis acid catalysis (Scheme 1b).46 Despite these achievements, the synthesis of polycyclic spiroindolines with more intricate 3D configurations remains an exceptionally intriguing and highly valuable frontier for researchers, and the development of environmentally friendly and sustainable methodologies to construct such structures holds significant importance for both synthetic and medicinal chemistry.
Electrochemistry, as a powerful and environmentally friendly platform for synthetic organic chemistry, is gaining increasing attention due to its inherent scalability, the ability to avoid harmful chemical oxidants and reductants, and the high tunability of reaction parameters.47–57 Furthermore, this strategy has also been successfully employed in dearomatization of indole to provide three-dimensional indolines of high-added value by Lei,58–61 Ling,62 Zhao,63,64 Guo,65et al.66–70 Nonetheless, reports on the application of BCBs in electrochemical systems remain very limited.37,71,72 To the best of our knowledge, the methods for obtaining polycyclic spiroindolines with two or more spirocenters through indole-based dearomatization reactions have not been reported. Moreover, to date, there have been no studies on the use of BCBs in electrocatalytic indole dearomatization reactions for the construction of polycyclic spiroindole derivatives. Based on previous literature and our research on indole dearomatization reactions,73 we speculate that the radical cation-mediated strain-release intramolecular (3 + 2) cycloaddition reaction may be achieved under mild and sustainable electrochemical conditions. Herein, we report a highly efficient and environmentally friendly electrochemical strategy for the intramolecular (3 + 2) dearomatization reaction of C3-BCB-substituted indoles, providing access to structurally complex and novel BCH-fused spiroindolines (Scheme 1c). This oxidant-free protocol enables the practical and atom-economic synthesis of a wide range of valuable spirocyclic indolines that bear four contiguous stereogenic centers, including two spiro quaternary carbon stereocenters in generally good yields with broad substrate scope and functional group compatibility.
Results and discussion
To validate the design strategy for constructing polycyclic spiroindolines, we selected the C3-BCB-substituted indole derivative 1a as the model substrate and explored a radical cation-mediated strain-release (3 + 2) cyclization reaction by the electrochemical method. Following systematic optimization, the polycyclic spiroindoline product 2a was successfully synthesized at a constant current of 1 mA in an undivided cell equipped with a graphite felt anode (GF) and a platinum plate cathode, using acetonitrile as the solvent. The reaction was conducted at room temperature, yielding 2a in 93% yield (see Table 1, entry 1). First, the screening of solvents indicated the importance of CH3CN in the reaction (see Table 1, entries 2 and 3). Additionally, replacing nBu4NBF4 with nBu4NBF6 or nBu4NI all resulted in diminished yield (see entries 4 and 5). The electrode screening results indicated that carbon cloth, carbon rod, and Pt plate anodes demonstrated inferior performance compared to the graphite felt anode (see entries 6–8). Considering the crucial role of current in the reaction, increasing the current did not lead to an improvement in yield (see entries 9 and 10). Control experiments confirmed the essential role of electricity, as no reaction was observed in the absence of current (see Table 1, entry 11). Notably, the transformation exhibits low sensitivity to ambient atmospheric conditions, delivering a comparable yield even in the presence of air (entry 12), which underscores the practical value of this protocol. Remarkably, control experiments conducted under conditions adapted from Lewis acid-catalyzed intermolecular reactions demonstrate that the transformation proceeds smoothly with 10 mol% In(OTf)3 as a Lewis acid catalyst, affording the desired product 2a in 71% yield (see Table S5 in the SI for more details).| Entry | Deviation from standard conditions | Yield%b |
|---|---|---|
| a Reaction condition: undivided cell, graphite felt anode (GF), Pt plate cathode, 1a (0.20 mmol), nBu4NBF4 (0.20 mmol), CH3CN (4 mL), room temperature, constant current = 1 mA, 1 h, Ar. b Isolated yields. | ||
| 1 | None | 93 |
| 2 | DCM instead of CH3CN | 86 |
| 3 | HFIP instead of CH3CN | N.D. |
| 4 | n Bu4NBF6 instead of nBu4NBF4 | 88 |
| 5 | n Bu4NI instead of nBu4NBF4 | 56 |
| 6 | Carbon cloth as an anode | 53 |
| 7 | Carbon rod as an anode | Trace |
| 8 | Pt as an anode | Trace |
| 9 | 3 mA instead of 1 mA | 92 |
| 10 | 5 mA instead of 1 mA | 90 |
| 11 | No electricity | N.R. |
| 12 | Under air | 85 |
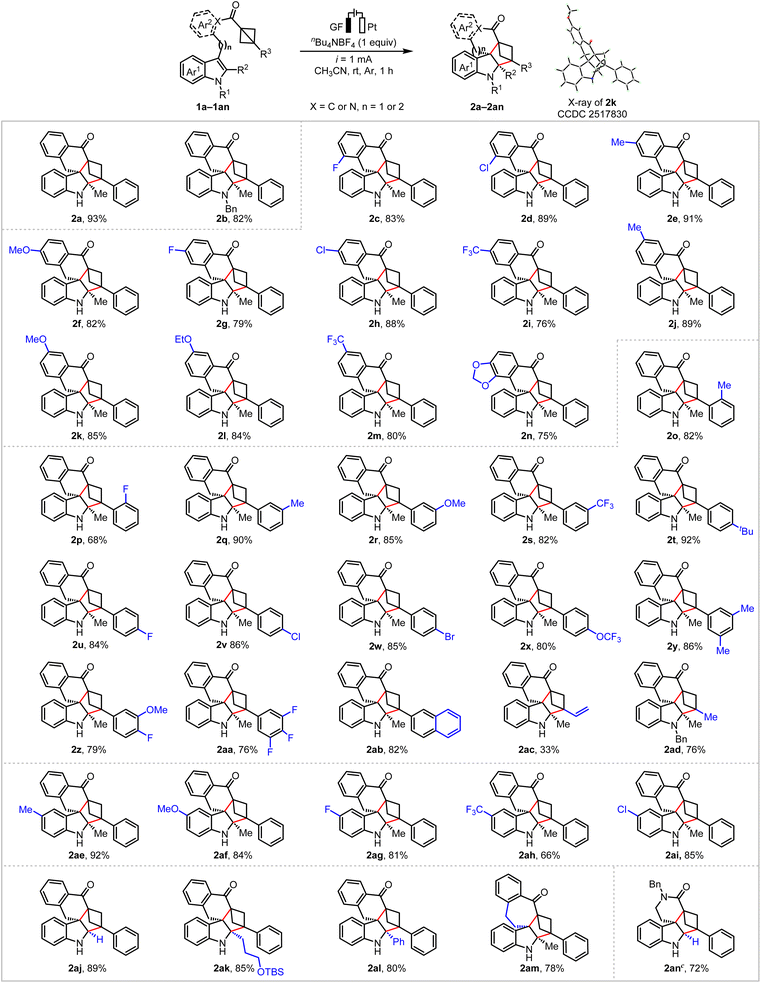
With the optimized reaction conditions established, the substrate scope of this electrocatalytic (3 + 2) cycloaddition reaction was subsequently explored. As illustrated in Scheme 2, initially, both unprotected and benzyl-protected BCB-substituted indole compounds 1a and 1b could be smoothly transformed into the corresponding spiroindolines 2a and 2b in 93% and 86% yields, respectively. Next, we investigated the influence of different substituents on the arene moiety of the aromatic ketone. Under optimal reaction conditions, a range of aromatic ketones bearing both electron-withdrawing (such as F, Cl, and CF3) and electron-donating (such as Me, OMe and OEt) substituents at different positions of the aromatic ring can efficiently undergo the reaction, producing the desired products 2c–2m in 76–91% yields. The structure of compound 2k was unambiguously verified by crystallographic analysis. In addition, a disubstituted aromatic ketone was applied to the reaction, successfully affording the corresponding polycyclic spiroindoline 2n in 75% yields. Subsequently, we turned our attention to the R3 moieties on the bridgehead bond in the BCBs. The cycloaddition reaction demonstrated remarkable tolerance toward both electron-deficient and electron-rich substituents at the ortho-, meta- and para-positions of the aromatic ring, including halogen (F, Cl, Br), methyl, tert-butyl, methoxy, trifluoromethoxy, and trifluoromethyl groups. These transformations delivered the desired products 2o–2aa in 68–92% yields. Replacing the R3 with a 2-naphthyl group was investigated and produced the desired product 2ab in 82% yield. However, when R3 was a vinyl group, the yield decreases significantly, with the desired 2ac obtained only in 33% yield. When R3 was a methyl group, the reaction proceeds smoothly, affording the target compound 2ad in 76% yield. Notably, the reaction of indole derivatives containing a C5-substituent (Me, OMe, F, CF3, Cl) was also tolerated, producing BCH-fused spiroindolines 2ae–2ai in 66–92% yields. Furthermore, a variety of functional groups on R2 moieties, including an H (as in 2aj), a silyl ether (as in 2ak) and a phenyl (as in 2al) were well tolerated, providing the desired cycloadducts 2aj–2al in good yields (80–89%). The experimental results indicate that the R2 on the indole ring has no significant effect on the reaction efficiency. More importantly, the homologous substrate with more than one carbon atom also exhibits good compatibility under the optimized reaction conditions, leading to the spirocycloheptane indoline 2am in decent yield (78%). Interestingly, an amide-linked BCB 1an was also well tolerated, affording the desired product 2an in 72% yield.
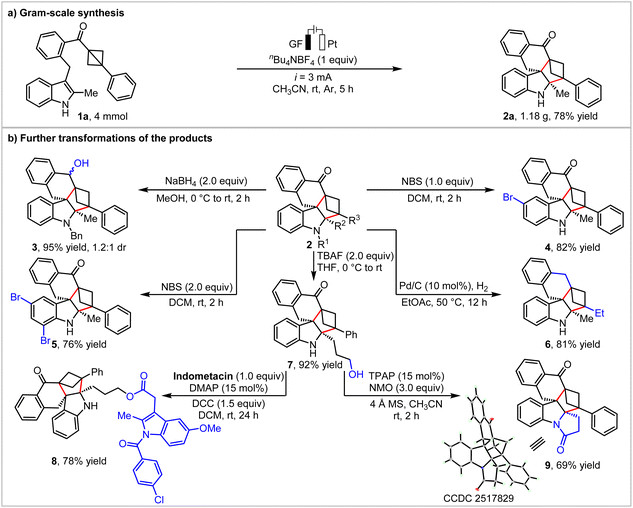
To showcase the potential application of this protocol, a scale-up synthesis of 2a was conducted. Under the standard reaction conditions, 4.0 mmol of BCB 1a reacted smoothly, resulting in the desired product 2a in 78% yield (Scheme 3a). Subsequently, we carried out additional late-stage transformations using BCH-fused polycyclic spiroindolines 2 to demonstrate the synthetic utility of these products. First, the carbonyl group of 2b was reduced to the hydroxyl group with NaBH4, affording alcohol 3 in high efficiency (95% yield, dr 1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). Furthermore, the spiroindoline 2a was subsequently elaborated into 4 and 5via electrophilic aromatic bromination. Interestingly, starting from 2ac, the carbonyl group and double bond were fully reduced to 6 by Pd/C-catalyzed hydrogenolysis in 81% yield. Next, the silyl group of 2ak could be removed with TBAF to give the primary alcohol 7. Alcohol 7 can undergo esterification with indomethacin to afford the complex derivative 8. Importantly, spiroindoline 7 can also undergo an intramolecular Ley oxidation reaction to access a complex polycyclic spiroindoline 9 containing three spiro carbon stereocenters. The structure of the 9 was unambiguously determined by X-ray crystallographic analysis (Scheme 3b).
1). Furthermore, the spiroindoline 2a was subsequently elaborated into 4 and 5via electrophilic aromatic bromination. Interestingly, starting from 2ac, the carbonyl group and double bond were fully reduced to 6 by Pd/C-catalyzed hydrogenolysis in 81% yield. Next, the silyl group of 2ak could be removed with TBAF to give the primary alcohol 7. Alcohol 7 can undergo esterification with indomethacin to afford the complex derivative 8. Importantly, spiroindoline 7 can also undergo an intramolecular Ley oxidation reaction to access a complex polycyclic spiroindoline 9 containing three spiro carbon stereocenters. The structure of the 9 was unambiguously determined by X-ray crystallographic analysis (Scheme 3b).
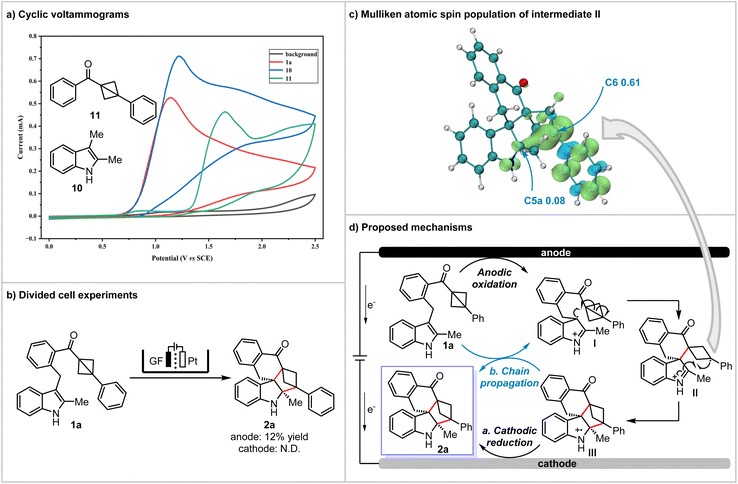
To elucidate the reaction mechanism, we conducted a series of mechanistic investigations (see the SI for details). Upon the introduction of TEMPO (2,2,6,6-tetramethylpiperidoxy) or BHT (butyl-hydroxytoluene) as a radical scavenger, the formation of the product was almost entirely suppressed. Furthermore, mass spectrometric analysis identified adducts between compound 1a and both TEMPO and BHT. These findings suggest that the reaction mechanism likely involves a radical pathway. Similarly, when C2-unsubstituted 1an was used as the substrate and P(OEt)3 was used as a trapping reagent, the desired product 2an was obtained in 38% yield. Meanwhile, an indole phosphorylation product was detected by HRMS, indicating the presence of a radical cation intermediate in the reaction (see the SI for details). Furthermore, according to cyclic voltammetry (CV) experiments, the oxidation peaks of 1a and its decomposition product, 2,3-dimethylindole 10, with BCB 11 were observed at +1.14 V, +1.22 V, and +1.66 V (vs. SCE), respectively (Scheme 4a). These results indicate that the oxidation potential of compound 1a is primarily attributed to the indole moiety, further suggesting that the reaction is likely initiated from the indole. Notably, the results from the divided-cell experiment revealed that the target product 2a, with a 12% yield, was detected in the anode compartment during substrate decomposition, whereas no reactions occurred in the cathode compartment (Scheme 4b). Importantly, the faradaic efficiency reveals that only 0.2 equiv. of charge is sufficient to achieve complete conversion (see the SI for details). This finding indicates that the reaction not only involves cathodic reduction but may also undergo chain propagation reactions.
According to the above studies and literature reports,58–61 a possible mechanism for the electrochemical dearomative (3 + 2) annulation of indole derivatives is proposed in Scheme 4d. To begin with, compound 1a undergoes anodic oxidation to generate the radical cation intermediate I. Subsequently, the intramolecular radical promotes the ring-opening of BCB, converting I into the benzyl radical species II. To validate the proposed intermediate, spin density analysis of radical cation II was performed. The results show that the unpaired electron is highly localized at the C6 position (Mulliken spin population of 0.61), while the spin density at C5a is negligible (0.08), indicating that intermediate II is a C6-localized radical, consistent with our hypothesis (Scheme 4c). Then, intermediate II undergoes an intramolecular radical addition cyclization to afford radical cation III. Ultimately, cathodic reduction or chain propagation of intermediate III delivers the target polycyclic spiroindolines.
Conclusions
In conclusion, we have developed a highly efficient and environmentally friendly electrochemical-driven radical cation-mediated strain-release dearomative (3 + 2) cyclization of C3-BCB-substituted indoles. Through this methodology, a series of structurally novel and densely functionalized BCH-fused spiroindolines were successfully synthesized as single diastereomers with good yields. This method involves mild conditions and produces BCH-fused spiroindolines with broad substrate scope, functional group compatibility and potential application, as demonstrated by late-stage synthetic transformations. Mechanistic studies provided strong evidence for a radical process. This practical and sustainable method is anticipated to see broader applications in organic synthesis and medicinal chemistry.Author contributions
Y. Zhu, E. Pu and S. Yang designed and performed the experimental work. D. Zhao performed calculations. Q. Xu, P. Jiang, and X. Li contributed to the analysis and interpretation of data. H. Zhang and J. Chen conceived the project. J. Chen and Y. Zhu wrote the manuscript. All authors contributed to or approved the final version of the paper.Conflicts of interest
There are no conflicts to declare.Data availability
CCDC 2517829 and 2517830 contain the supplementary crystallographic data for this paper.74a,bSupplementary information (SI): experimental procedures, crystallographic data, compound characterization data, and copies of NMR spectra of the products. See DOI: https://doi.org/10.1039/d6sc01271j.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (22361051 and U24A20802), the Yunnan Fundamental Research Projects (202301BF070001-022), and the Yunnan Key Laboratory of Crystalline Porous Organic Functional Materials (202449CE340024). D. Zhao was supported by the High-Level Talent Special Support Plan.Notes and references
- Y.-Z. Cheng, Z. Feng, X. Zhang and S.-L. You, Chem. Soc. Rev., 2022, 51, 2145–2170 RSC.
- P. Ji, K. Duan, M. Li, Z. Wang, X. Meng, Y. Zhang and W. Wang, Chem. Soc. Rev., 2024, 53, 6600–6624 RSC.
- C. J. Huck and D. Sarlah, Chem, 2020, 6, 1589–1603 CAS.
- S. P. Roche and J. A. Porco Jr., Angew. Chem., Int. Ed., 2011, 50, 4068–4093 CrossRef CAS PubMed.
- S. M. Umer, M. Solangi, K. M. Khan and R. S. Z. Saleem, Molecules, 2022, 27, 7586 CrossRef CAS PubMed.
- F. Omar, A. M. Tareq, A. M. Alqahtani, K. Dhama, M. A. Sayeed, T. B. Emran and J. Simal-Gandara, Molecules, 2021, 26, 2297 CrossRef CAS PubMed.
- Y.-Z. Liu, H. Song, C. Zheng and S.-L. You, Nat. Synth., 2022, 1, 203–216 CrossRef CAS.
- X.-X. Zhu, Y.-Y. Fan, L. Xu, Q.-F. Liu, J.-P. Wu, J.-Y. Li, J. Li, K. Gao and J.-M. Yue, Org. Lett., 2019, 21, 1471–1474 CrossRef CAS PubMed.
- J.-J. Yao, R. Ding, X. Chen and H. Zhai, J. Am. Chem. Soc., 2022, 144, 14396–14402 CrossRef CAS PubMed.
- M. F. Bartlett, R. Sklar, W. I. Taylor, E. Schlittler, R. L. S. Amai, P. Beak, N. V. Bringi and E. Wenkert, J. Am. Chem. Soc., 1962, 84, 622–630 CrossRef CAS.
- F. Khuong-Huu, A. Chiaroni and C. Riche, Tetrahedron Lett., 1981, 22, 733–734 CrossRef CAS.
- C.-T. Liu, Q.-W. Wang and C.-H. Wang, J. Am. Chem. Soc., 1981, 103, 4634–4635 CrossRef CAS.
- L. Peng, D. Xu, X. Yang, J. Tang, X. Feng, S.-L. Zhang and H. Yan, Angew. Chem., Int. Ed., 2019, 58, 216–220 CrossRef CAS PubMed.
- Y. Zheng, C. M. Tice and S. B. Singh, Bioorg. Med. Chem. Lett., 2014, 24, 3673–3682 CrossRef CAS PubMed.
- K. Hiesinger, D. Dar’in, E. Proschak and M. Krasavin, J. Med. Chem., 2021, 64, 150–183 CrossRef CAS PubMed.
- D. F. Veber, S. R. Johnson, H.-Y. Cheng, B. R. Smith, K. W. Ward and K. D. Kopple, J. Med. Chem., 2002, 45, 2615–2623 CrossRef CAS PubMed.
- M. V. S. Varma, R. S. Obach, C. Rotter, H. R. Miller, G. Chang, S. J. Steyn, A. El-Kattan and M. D. Troutman, J. Med. Chem., 2010, 53, 1098–1108 CrossRef CAS PubMed.
- M. R. van der Kolk, M. A. C. H. Janssen, F. P. J. T. Rutjes and D. Blanco-Ania, ChemMedChem, 2022, 17, e202200020 CrossRef CAS PubMed.
- V. M. Dembitsky, Phytomedicine, 2014, 21, 1559–1581 CrossRef CAS PubMed.
- J. Liang, Y. Wu, X. Ma and H. Li, Adv. Synth. Catal., 2025, 367, e202500003 CrossRef CAS.
- M. Zhu, X. Zhang, C. Zheng and S.-L. You, Acc. Chem. Res., 2022, 55, 2510–2525 CrossRef CAS PubMed.
- J. Zhu, Y. Liang, L. Wang, Z.-B. Zheng, K. N. Houk and Y. Tang, J. Am. Chem. Soc., 2014, 136, 6900–6903 CrossRef CAS PubMed.
- X. Liu, J. He, K. Lin, X. Wang and H. Cao, Org. Chem. Front., 2024, 11, 6942–6957 RSC.
- S. J. Sujansky and X. Ma, Asian J. Org. Chem., 2024, 13, e202400045 CrossRef CAS.
- R. I. Revie, J. Ragus and E. A. Anderson, Chem. Soc. Rev., 2026, 55, 941–954 RSC.
- P. Bellotti and F. Glorius, J. Am. Chem. Soc., 2023, 145, 20716–20732 CrossRef CAS PubMed.
- X.-C. Yang, J.-J. Wang, Y. Xiao and J.-J. Feng, Angew. Chem., Int. Ed., 2025, 64, e202505803 CrossRef CAS PubMed.
- Y. Xiao, L. Tang, X.-C. Yang, N.-Y. Wang, J. Zhang, W.-P. Deng and J.-J. Feng, CCS Chem., 2025, 7, 1903–1934 CrossRef CAS.
- H.-X. He, X.-C. Yang, Y. Xiao, L. Tang, W.-L. Xu and J.-J. Feng, Sci. China Chem., 2026, 69, 1120–1141 CrossRef CAS.
- M. Golfmann and J. C. L. Walker, Commun. Chem., 2023, 6, 9 CrossRef CAS PubMed.
- S. Nagasawa and Y. Iwabuchi, Synthesis, 2024, 57, 1153–1170 CrossRef.
- C. B. Kelly, J. A. Milligan, L. J. Tilley and T. M. Sodano, Chem. Sci., 2022, 13, 11721–11737 RSC.
- J. L. Tyler, F. Schäfer, H. Shao, C. Stein, A. Wong, C. G. Daniliuc, K. N. Houk and F. Glorius, J. Am. Chem. Soc., 2024, 146, 16237–16247 CrossRef CAS PubMed.
- H. Wang, H. Shao, A. Das, S. Dutta, H. T. Chan, C. Daniliuc, K. N. Houk and F. Glorius, Science, 2023, 381, 75–81 CrossRef CAS PubMed.
- S. Dutta, D. Lee, K. Ozols, C. G. Daniliuc, R. Shintani and F. Glorius, J. Am. Chem. Soc., 2024, 146, 2789–2797 CrossRef CAS PubMed.
- M. Golfmann, M. Reinhold, J. D. Steen, M. S. Deike, B. Rodemann, C. Golz, S. Crespi and J. C. L. Walker, ACS Catal., 2024, 14, 13987–13998 CrossRef CAS.
- J. T. Maddigan-Wyatt, D. A. Knyazev and D. B. Werz, Org. Lett., 2026, 28, 2573–2578 CrossRef CAS PubMed.
- P. Wipf and M. A. A. Walczak, Angew. Chem., Int. Ed., 2006, 45, 4172–4175 CrossRef CAS PubMed.
- P. K. Mykhailiuk, Org. Biomol. Chem., 2019, 17, 2839–2849 RSC.
- M. A. M. Subbaiah and N. A. Meanwell, J. Med. Chem., 2021, 64, 14046–14128 CrossRef CAS PubMed.
- J. Tsien, C. Hu, R. R. Merchant and T. Qin, Nat. Rev. Chem., 2024, 8, 605–627 CrossRef CAS PubMed.
- V. V. Levterov, Y. Panasyuk, V. O. Pivnytska and P. K. Mykhailiuk, Angew. Chem., Int. Ed., 2020, 59, 7161–7167 CrossRef CAS PubMed.
- R. Kleinmans, T. Pinkert, S. Dutta, T. O. Paulisch, H. Keum, C. G. Daniliuc and F. Glorius, Nature, 2022, 605, 477–482 CrossRef CAS PubMed.
- D. Ni, S. Hu, X. Tan, Y. Yu, Z. Li and L. Deng, Angew. Chem., Int. Ed., 2023, 62, e202308606 CrossRef CAS PubMed.
- L. Tang, Y. Xiao, F. Wu, J.-L. Zhou, T.-T. Xu and J.-J. Feng, Angew. Chem., Int. Ed., 2023, 62, e202310066 CrossRef CAS PubMed.
- J. Yang, L. Yang, Y. Zhao, L. Hou, Y. Qiu, G. Pan, X. Liu, X. Feng and S. Dong, J. Am. Chem. Soc., 2025, 147, 35755–35766 CrossRef CAS PubMed.
- M. Yan, Y. Kawamata and P. S. Baran, Chem. Rev., 2017, 117, 13230–13319 CrossRef CAS PubMed.
- R. Francke and R. D. Little, Chem. Soc. Rev., 2014, 43, 2492–2521 RSC.
- K. D. Moeller, Chem. Rev., 2018, 118, 4817–4833 CrossRef CAS PubMed.
- M. D. Kärkäs, Chem. Soc. Rev., 2018, 47, 5786–5865 RSC.
- L. F. T. Novaes, J. Liu, Y. Shen, L. Lu, J. M. Meinhardt and S. Lin, Chem. Soc. Rev., 2021, 50, 7941–8002 RSC.
- T. Fuchigami and S. Inagi, Acc. Chem. Res., 2020, 53, 322–334 CrossRef CAS PubMed.
- L. Zeng, J. Wang, D. Wang, H. Yi and A. Lei, Angew. Chem., Int. Ed., 2023, 62, e202309620 CrossRef CAS PubMed.
- X. Chang, Q. Zhang and C. Guo, Angew. Chem., Int. Ed., 2020, 59, 12612–12622 CrossRef CAS PubMed.
- X. Cheng, A. Lei, T.-S. Mei, H.-C. Xu, K. Xu and C. Zeng, CCS Chem., 2022, 4, 1120–1152 CrossRef CAS.
- M. C. Leech and K. Lam, Nat. Rev. Chem., 2022, 6, 275–286 CrossRef PubMed.
- P. Li, Y. Wang, H. Zhao and Y. Qiu, Acc. Chem. Res., 2025, 58, 113–129 CrossRef CAS PubMed.
- K. Liu, S. Tang, P. Huang and A. Lei, Nat. Commun., 2017, 8, 775 CrossRef PubMed.
- K. Liu, W. Song, Y. Deng, H. Yang, C. Song, T. Abdelilah, S. Wang, H. Cong, S. Tang and A. Lei, Nat. Commun., 2020, 11, 3 CrossRef PubMed.
- C. Song, K. Liu, X. Jiang, X. Dong, Y. Weng, C.-W. Chiang and A. Lei, Angew. Chem., Int. Ed., 2020, 59, 7193–7197 CrossRef CAS PubMed.
- X. Liu, D. Yang, Z. Liu, Y. Wang, Y. Liu, S. Wang, P. Wang, H. Cong, Y.-H. Chen, L. Lu, X. Qi, H. Yi and A. Lei, J. Am. Chem. Soc., 2023, 145, 3175–3186 CrossRef CAS PubMed.
- Z. Xu, C. Zheng, J. Lin, W. Huang, D. Song, W. Zhong and F. Ling, Angew. Chem., Int. Ed., 2025, 64, e202413601 CrossRef CAS PubMed.
- K. Mo, X. Zhou, J. Wu and Y. Zhao, Org. Lett., 2022, 24, 2788–2792 CrossRef CAS PubMed.
- K. Mo, X. Zhou, J. Wu and Y. Zhao, J. Org. Chem., 2022, 87, 16106–16110 CrossRef CAS PubMed.
- W. Zhao, Y. Lu, Y. Qiao, X. Yin, C. Liu, Z. Fang, J. Zhu and K. Guo, Org. Lett., 2023, 25, 7451–7456 CrossRef CAS PubMed.
- J. Zi, H. Tang, D. Wang, M. Li, Y. Zhou, S. Lv, D. Liang and L. Shi, Green Chem., 2024, 26, 10397–10403 RSC.
- Q.-H. Huang, S.-X. Li, J.-C. Kang, R.-X. Liu, Z.-H. Li, F. Xiong, T.-M. Ding and S.-Y. Zhang, Org. Lett., 2024, 26, 5657–5663 CrossRef CAS PubMed.
- X. Tan, Q. Wang and J. Sun, Nat. Commun., 2023, 14, 357 CrossRef CAS PubMed.
- X. Tan, Z. Zhou, M. Shao and J. Sun, Angew. Chem., Int. Ed., 2025, 64, e202510078 CrossRef CAS PubMed.
- H. Ren, R.-A. Wang, J. Shi, J.-R. Song, W. Wu, Q. Chi and N. Zhang, Org. Biomol. Chem., 2023, 21, 7290–7294 RSC.
- R. Maity, S. Paul, A. Sen, S. Garain and B. Maji, ChemSusChem, 2025, 18, e202401701 CrossRef CAS PubMed.
- J. Liu, Y. Qian, H. Zhao, Y. Liu, Z. Qin, Z. Zhang and L. Rong, J. Org. Chem., 2024, 89, 15914–15923 CrossRef CAS PubMed.
- Y. Zhu, S. Yang, E. Pu, L. Li, S. Ye, L. Wei, G. Ma, Y. Zhang, H. Zhang and J. Chen, Org. Lett., 2023, 25, 3533–3538 CrossRef CAS PubMed.
- (a) CCDC 2517829: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qj09l; (b) CCDC 2517830: Experimental Crystal Structure Determination, 2026, DOI:10.5517/ccdc.csd.cc2qj0bm.
| This journal is © The Royal Society of Chemistry 2026 |